Physiological Responses of the Submerged Macrophyte Stuckenia pectinata to High Salinity and Irradiance Stress to Assess Eutrophication Management and Climatic Effects: An Integrative Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Experimental Design and Treatments
2.3. Chlorophyll-a Fluorescence Measurements
2.4. Chlorophyll (a, b) Content
2.5. Relative Growth Rate
3. Statistical Analysis
4. Results
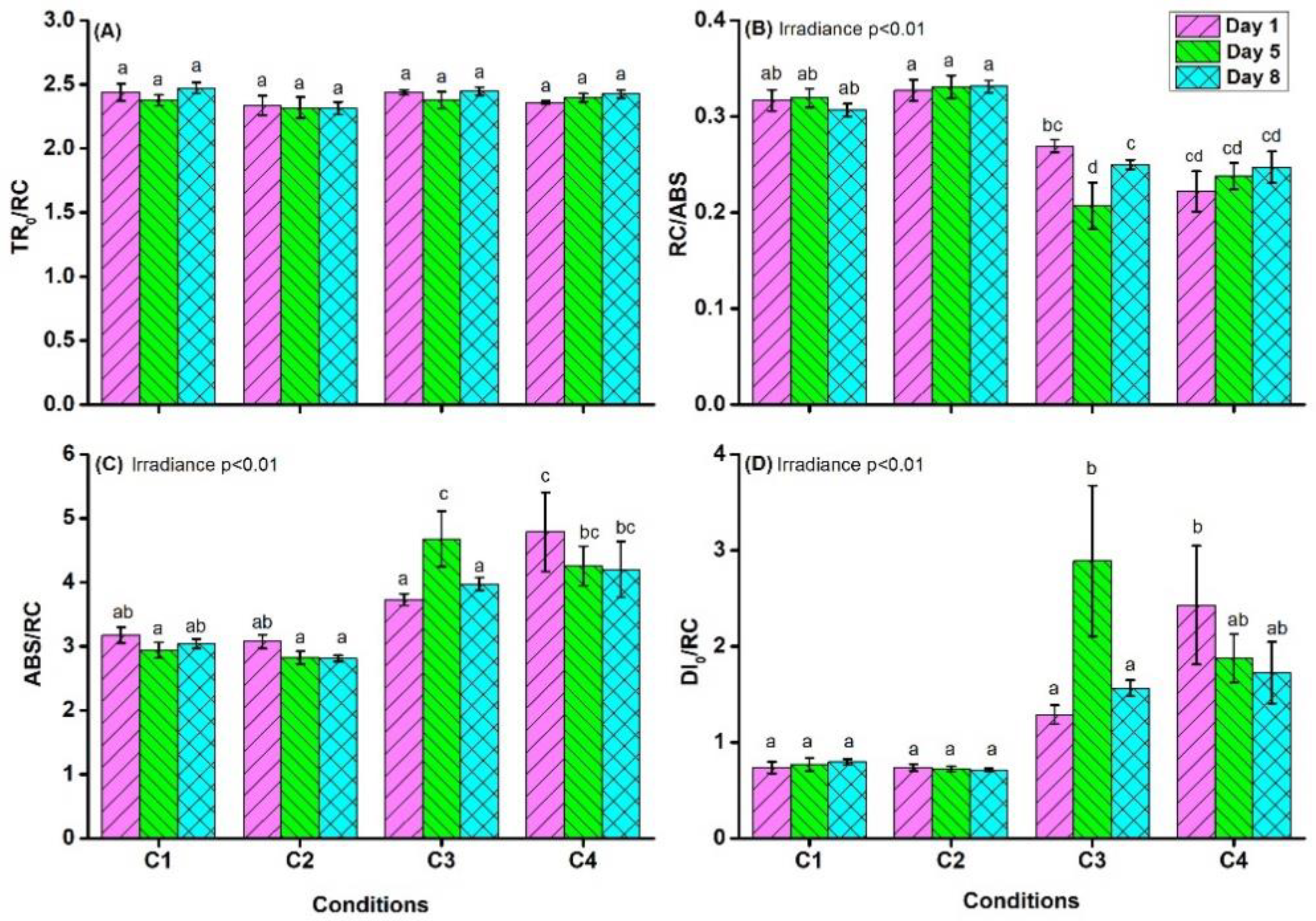
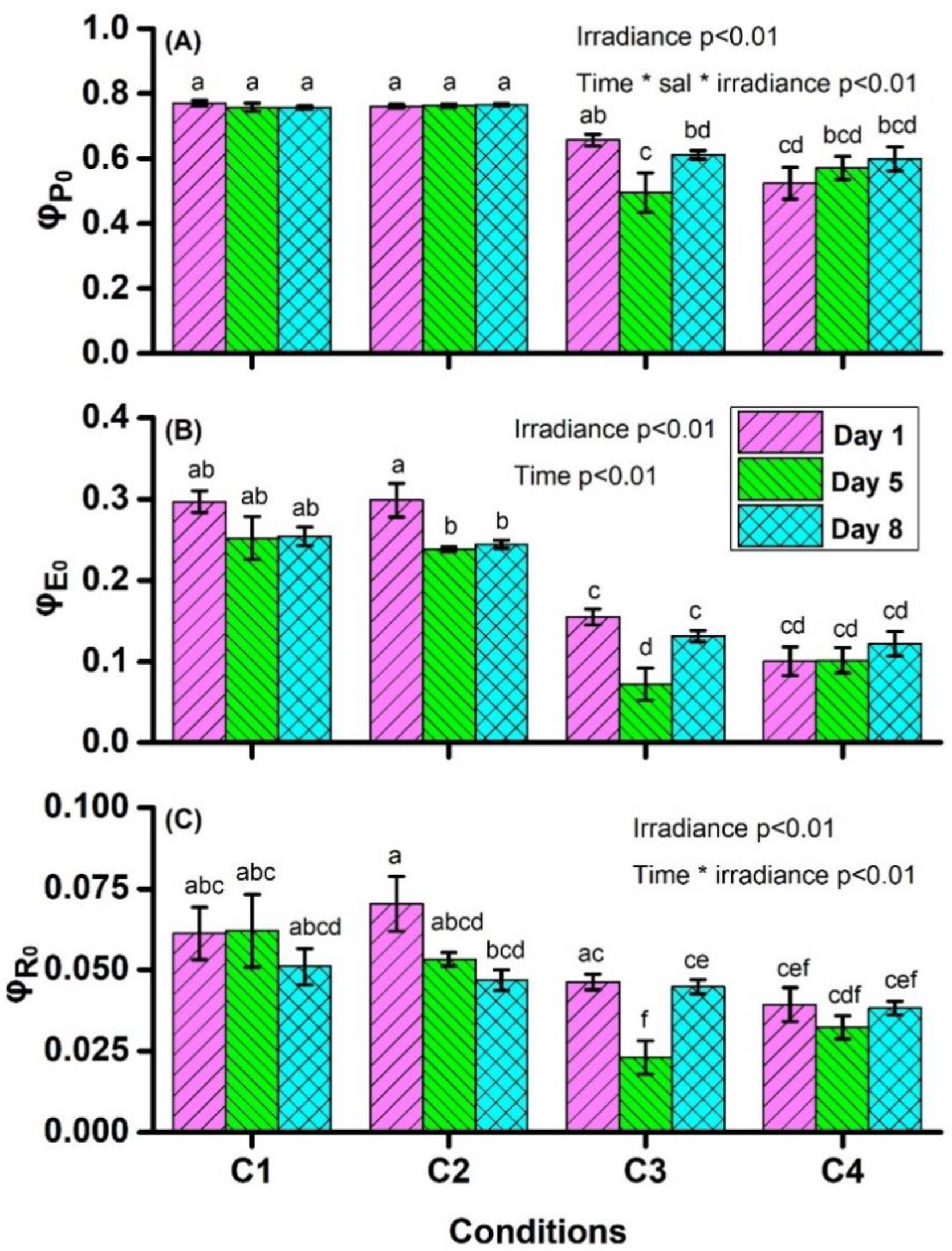
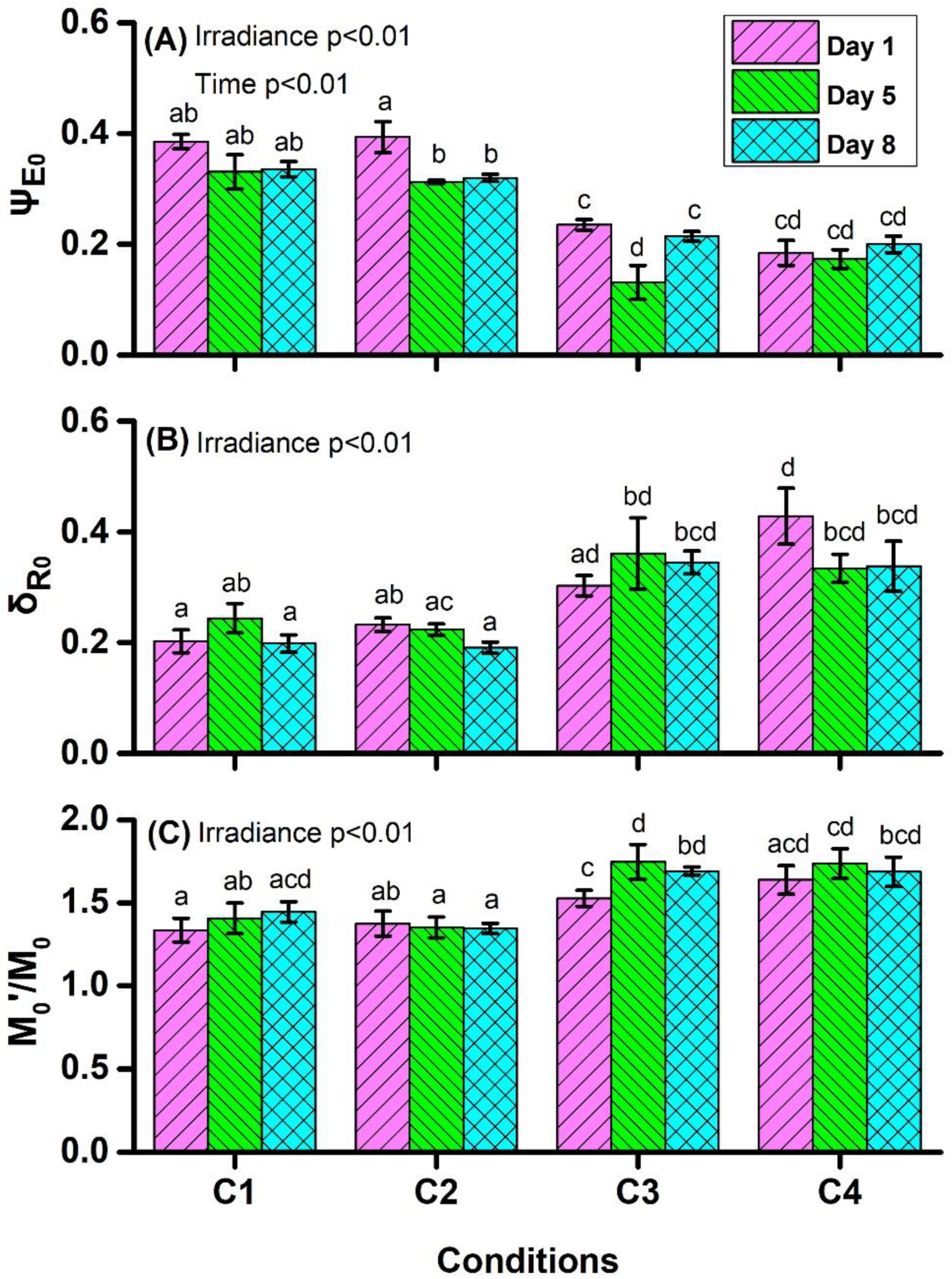
4.1. Chlorophyll a Fluorescence Measurements
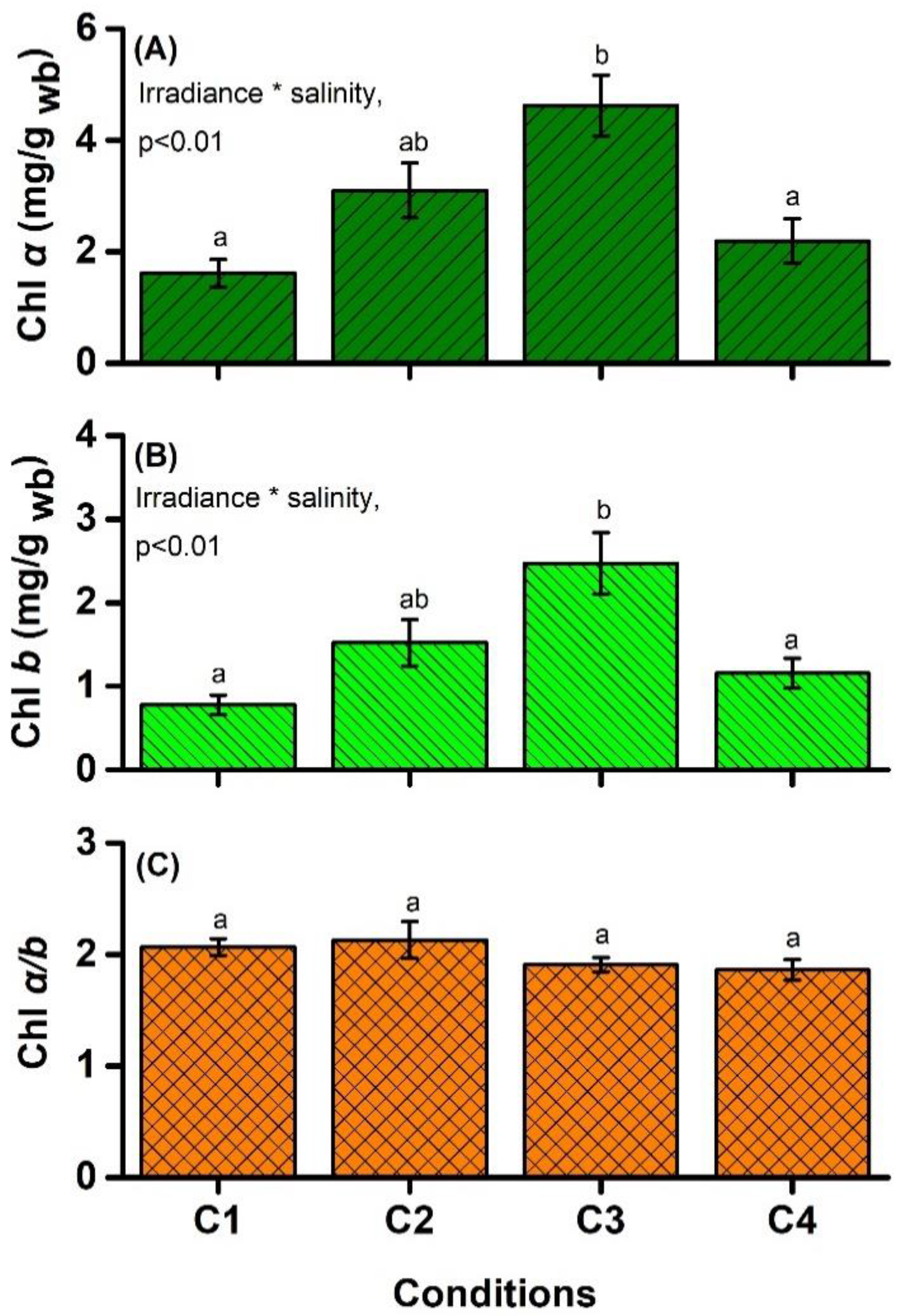
4.2. Chlorophyll Content
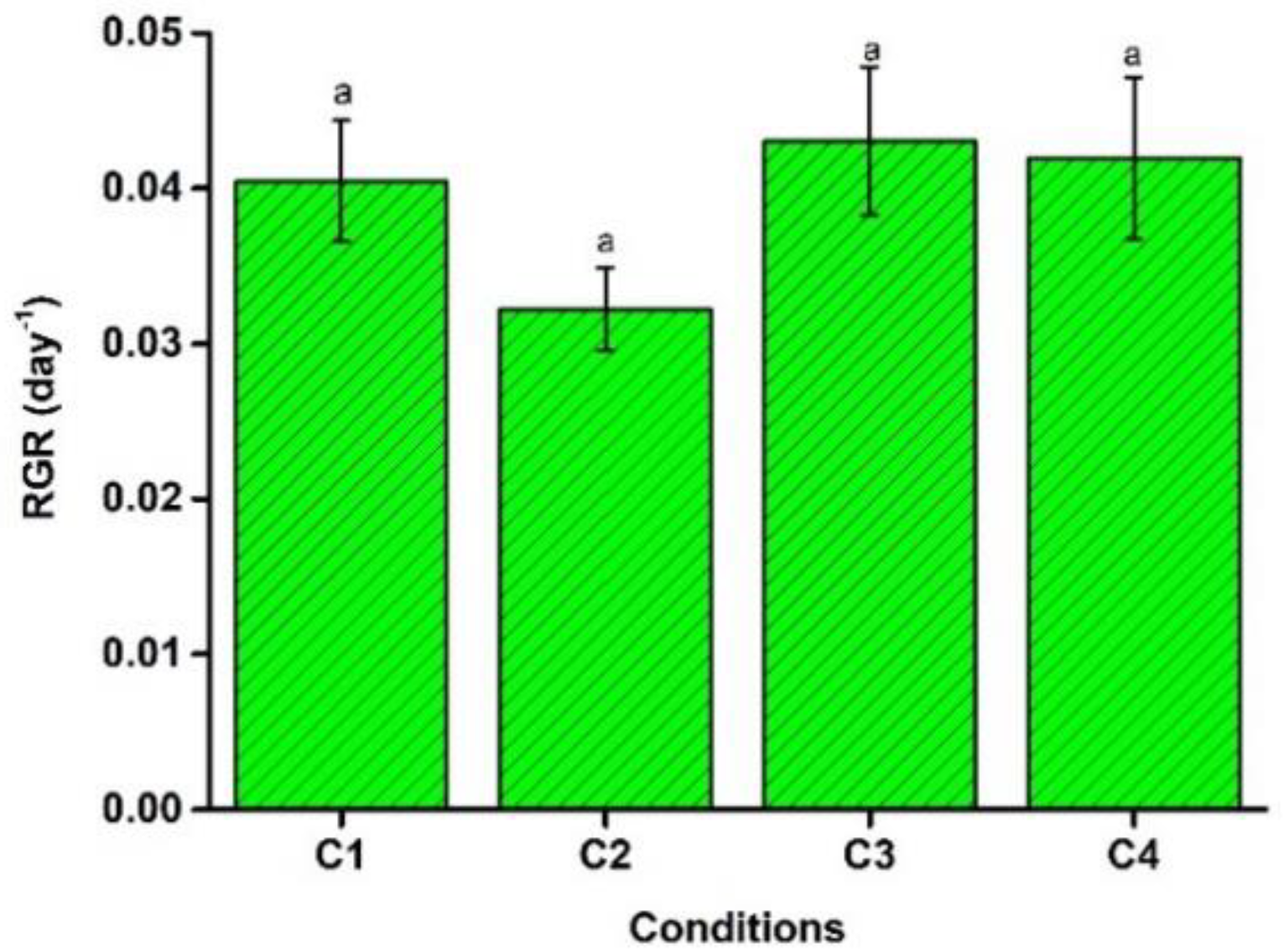
4.3. Relative Growth Rate
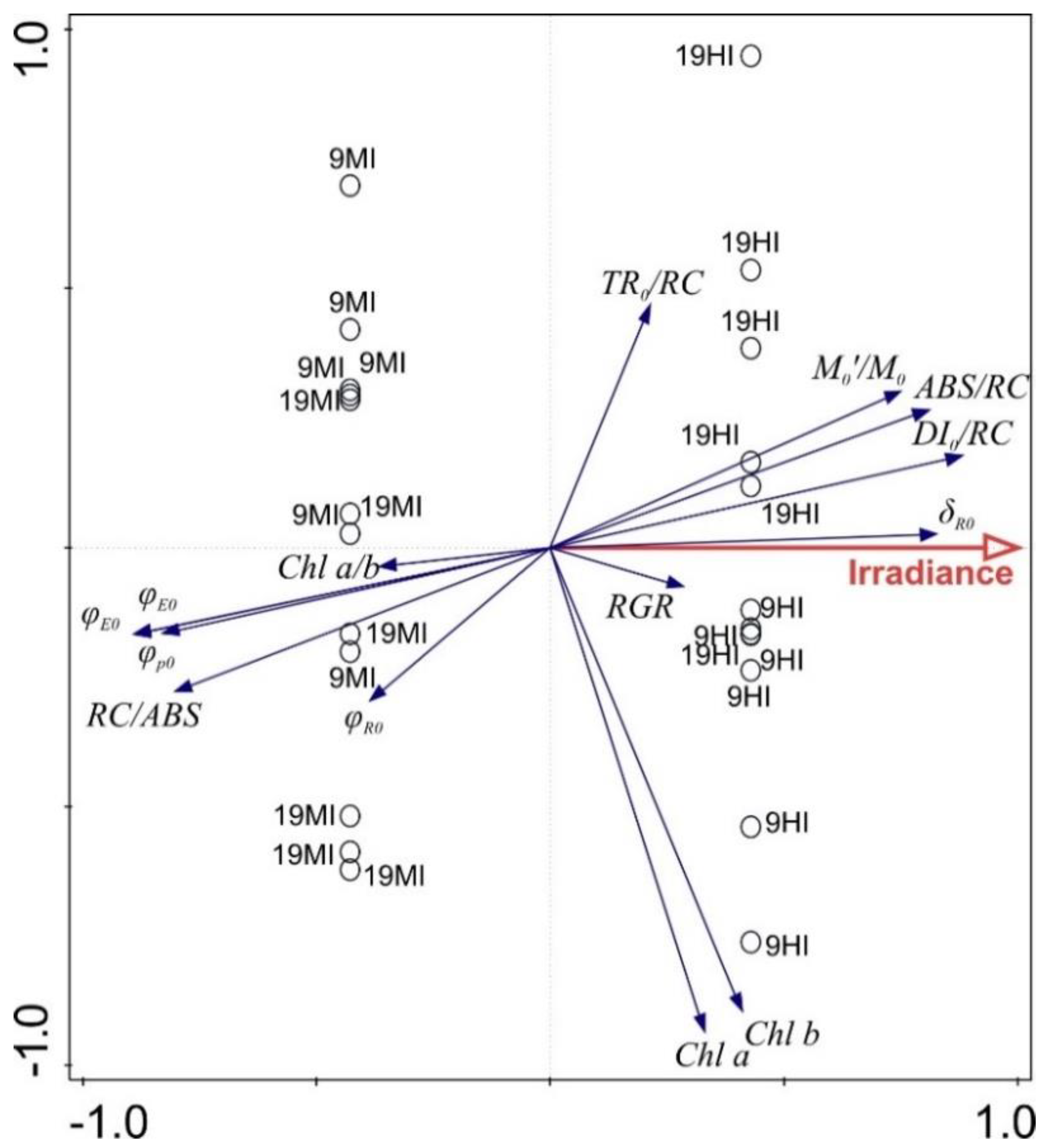
4.4. Redundancy Analysis (RDA)
5. Discussion
5.1. Irradiance Effects
5.2. Interactive Effects between Irradiance and Salinity
5.3. Interactive Effects with Time
5.4. Stuckenia pectinata in Vistonis Lake
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Pilon, J.; Santamaría, L. Clonal variation in morphological and physiological responses to irradiance and photoperiod for the aquatic angiosperm Potamogeton pectinatus. J. Ecol. 2002, 90, 859–870. [Google Scholar] [CrossRef]
- Scheffer, M.; de Redelijkheid, M.R.; Noppert, F. Distribution and dynamics of submerged vegetation in a chain of shallow eutrophic lakes. Aquat. Bot. 1992, 42, 199–216. [Google Scholar] [CrossRef]
- Borgnis, E.; Boyer, K.E. Salinity tolerance and competition drive distributions of native and invasive submerged aquatic vegetation in the upper San Francisco Estuary. Estuaries Coasts 2016, 39, 707–717. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Søndergaard, M.; Kirsten, C. Alternative Stable states. In The Structering Role of Submerged Macrophytes in Lakes; Jeppesen, E., Søndergaard, M., Christoffersen, K., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 427. [Google Scholar]
- Gao, Y.N.; Dong, J.; Fu, Q.Q.; Wang, Y.P.; Chen, C.; Li, J.H.; Li, R.; Zhou, C.J. Allelopathic effects of submerged macrophytes on phytoplankton. Allelopath. J. 2017, 40, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Zhang, H.; Han, Z.; Ye, J.; Liu, Z. The influence of aquatic macrophytes on Microcystis aeruginosa growth. Ecol. Eng. 2012, 42, 130–133. [Google Scholar] [CrossRef]
- Engelhardt, K.A.M.; Ritchie, M.E. Effects of macrophyte species richness on wetland ecosystem functioning and services. Nature 2001, 411, 687–689. [Google Scholar] [CrossRef]
- Hilt, S.; Brothers, S.; Jeppesen, E.; Veraart, A.J.; Kosten, S. Translating regime shifts in shallow lakes into changes in ecosystem functions and services. Bioscience 2017, 67, 928–936. [Google Scholar] [CrossRef]
- Albertoni, E.F.; Palma-Silva, C.; Trindade, C.R.T.; Furlanetto, L.M. Field evidence of the influence of aquatic macrophytes on water quality in a shallow eutrophic lake over a 13-year period. Acta Limnol. Bras. 2014, 26, 176–185. [Google Scholar] [CrossRef] [Green Version]
- Bakker, E.S.; Van Donk, E.; Declerck, S.A.J.; Helmsing, N.R.; Hidding, B.; Nolet, B.A. Effect of macrophyte community composition and nutrient enrichment on plant biomass and algal blooms. Basic Appl. Ecol. 2010, 11, 432–439. [Google Scholar] [CrossRef]
- Hidding, B.; Brederveld, R.J.; Nolet, B.A. How a bottom-dweller beats the canopy: Inhibition of an aquatic weed (Potamogeton pectinatus) by macroalgae (Chara spp.). Freshw. Biol. 2010, 55, 1758–1768. [Google Scholar] [CrossRef]
- Triest, L.; Tran Thi, V.; Le Thi, D.; Sierens, T.; van Geert, A. Genetic differentiation of submerged plant populations and taxa between habitats. Hydrobiologia 2010, 656, 15–27. [Google Scholar] [CrossRef]
- Abbasi, S.; Afsharzadeh, S.; Saeidi, H.; Triest, L. Strong genetic differentiation of submerged plant populations across mountain ranges: Evidence from Potamogeton pectinatus in Iran. PLoS ONE 2016, 11, 1–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ganie, A.H.; Reshi, Z.A.; Wafai, B.A. Reproductive ecology of Potamogeton pectinatus L. (= Stuckenia pectinata (L.) Börner) in relation to its spread and abundance in freshwater ecosystems of the Kashmir Valley, India. Trop. Ecol. 2016, 57, 787–803. [Google Scholar]
- Pilon, J.; Santamaría, J. Seasonal acclimation in the photosynthetic and respiratory temperature responses of three submerged freshwater macrophyte species. New Phytol. 2001, 151, 659–670. [Google Scholar] [CrossRef]
- Sandsten, H.; Beklioglu, M.; Ince, O. Effects of waterfowl, large fish and periphyton on the spring growth of Potamogeton pectinatus L. in Lake Mogan, Turkey. Hydrobiologia 2005, 537, 239–248. [Google Scholar] [CrossRef]
- Abbasi, S.; Afsharzadeh, S.; Saeidi, H. Genetic diversity of Potamogeton pectinatus L. in Iran as revealed by ISSR markers. Acta Bot. Croat. 2017, 76, 177–182. [Google Scholar] [CrossRef] [Green Version]
- Hootsmans, M.J.M.; Drovandi, A.A.; Soto Perez, N.; Wiegman, F. Photosynthetic plasticity in Potamogeton pectinatus L. from Argentina: Strategies to survive adverse light conditions. Hydrobiologia 1996, 340, 1–5. [Google Scholar] [CrossRef]
- Shabnam, N.; Sharmila, P.; Sharma, A.; Strasser, R.J.; Govindjee; Pardha-Saradhi, P. Mitochondrial electron transport protects floating leaves of long leaf pondweed (Potamogeton nodosus Poir) against photoinhibition: Comparison with submerged leaves. Photosynth. Res. 2015, 125, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Poikane, S.; Portielje, R.; Denys, L.; Elferts, D.; Kelly, M.; Kolada, A.; Mäemets, H.; Phillips, G.; Søndergaard, M.; Willby, N.; et al. Macrophyte assessment in European lakes: Diverse approaches but convergent views of ‘good’ ecological status. Ecol. Indic. 2018, 94, 185–197. [Google Scholar] [CrossRef]
- Kondo, K.; Kawabata, H.; Ueda, S.; Hasegawa, H.; Inaba, J.; Mitamura, O.; Seike, Y.; Ohmomo, Y. Distribution of aquatic plants and absorption of radionuclides by plants through the leaf surface in brackish Lake Obuchi, Japan, bordered by nuclear fuel cycle facilities. J. Radioanal. Nucl. Chem. 2003, 257, 305–312. [Google Scholar] [CrossRef]
- Kantrud, H.A. Sago pondweed (Potamogeton pectinatus L.): A literature review. Resour. Publ. US Fish Wildl. Serv. 1990, 176, 23. [Google Scholar]
- Rodríguez-Gallego, L.; Sabaj, V.; Masciadri, S.; Kruk, C.; Arocena, R.; Conde, D. Salinity as a major driver for submerged aquatic vegetation in coastal lagoons: A Multi-Year Analysis in the Subtropical Laguna de Rocha. Estuaries Coasts 2014, 38, 451–465. [Google Scholar] [CrossRef]
- Dhir, B. Status of aquatic macrophytes in changing climate: A perspective. J. Environ. Sci. Technol. 2015, 8, 139–148. [Google Scholar] [CrossRef]
- van Wijck, C.; Grillas, P.; de Groot, C.J.; Ham, L.T. A comparison between the biomass production of Potamogeton pectinatus L. and Myriophyllum spicatum L. in the Camargue (southern France) in relation to salinity and sediment characteristics. Vegetatio 1994, 113, 171–180. [Google Scholar] [CrossRef]
- Hu, Q.; Turnbull, M.; Hawes, I. Salinity restricts light conversion efficiency during photo-acclimation to high irradiance in Stuckenia pectinata. Environ. Exp. Bot. 2019, 165, 83–91. [Google Scholar] [CrossRef]
- Moustaka-Gouni, M.; Hiskia, A.; Genitsaris, S.; Katsiapi, M.; Manolidi, K.; Zervou, S.K.; Christophoridis, C.; Triantis, T.M.; Kaloudis, T.; Orfanidis, S. First report of Aphanizomenon favaloroi occurrence in Europe associated with saxitoxins and a massive fish kill in Lake Vistonis, Greece. Mar. Freshw. Res. 2017, 68, 793–800. [Google Scholar] [CrossRef]
- Vardaka, E.; Moustaka-Gouni, M.; Cook, C.M.; Lanaras, T. Cyanobacterial blooms and water quality in Greek waterbodies. J. Appl. Phycol. 2005, 17, 391–401. [Google Scholar] [CrossRef]
- Lüning, K. Seaweeds: Their Environment, Biogeography, and Ecophysiology; Wiley: Hoboken, NJ, USA, 1990. [Google Scholar]
- Markou, D.A.; Sylaios, G.K.; Tsihrintzis, V.A.; Gikas, G.D.; Haralambidou, K. Water quality of Vistonis Lagoon, Northern Greece: Seasonal variation and impact of bottom sediments. Desalination 2007, 210, 83–97. [Google Scholar] [CrossRef]
- Dolbeth, M.; Crespo, D.; Leston, S.; Solan, M. Realistic scenarios of environmental disturbance lead to functionally important changes in benthic species-environment interactions. Mar. Environ. Res. 2019, 150, 104770. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide Kate. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli-Michael, M.; Dangre, D.; Rai, M. Biophysical phenomics reveals functional building blocks of plants systems biology: A case study for the evaluation of the impact of mycorrhization with Piriformospora indica. Adv. Tech. Soil Microbiol. 2007, 11, 319–341. [Google Scholar] [CrossRef]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The fluorescence transient as a tool to characterize and screen photosynthetic samples. In Probing Photosynthesis: Mechanism, Regulation & Adaptation; CRC: Boca Raton, FL, USA, 2000; pp. 443–480. [Google Scholar]
- Tsimilli-Michael, M. Revisiting JIP-test: An educative review on concepts, assumptions, approximations, definitions and terminology. Photosynthetica 2020, 58, 275–292. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Chen, L.; Hao, Z.; Li, X.; Liu, Y. Effects of salinity stress on the photosynthesis of Wolffia arrhiza as probed by the OJIP test. Fresenius Environ. Bull. 2011, 20, 432–438. [Google Scholar]
- Xu, C.; Wang, M.T.; Yang, Z.Q.; Zheng, Q.T. Low temperature and low irradiation induced irreversible damage of strawberry seedlings. Photosynthetica 2020, 58, 156–164. [Google Scholar] [CrossRef]
- Lepeduš, H.; Vidaković-Cifrek, Ž.; Šebalj, I.; Antunović Dunić, J.; Cesar, V. Effects of low and high irradiation levels on growth and PSII efficiency in Lemna minor L. Acta Bot. Croat. 2020, 79, 185–192. [Google Scholar] [CrossRef]
- Parihar, P.; Singh, S.; Singh, R.; Singh, V.P.; Prasad, S.M. Effect of salinity stress on plants and its tolerance strategies: A review. Environ. Sci. Pollut. Res. 2015, 22, 4056–4075. [Google Scholar] [CrossRef] [PubMed]
- Tuller, J.; Marquis, R.J.; Andrade, S.M.M.; Monteiro, A.B.; Faria, L.D.B. Trade-offs between growth, reproduction and defense in response to resource availability manipulations. PLoS ONE 2018, 13, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Gleeson, S.K.; Tilman, D. Plant allocation and the multiple limitation hypothesis. Am. Nat. 1992, 139, 1322–1343. [Google Scholar] [CrossRef]
- Schubert, N.; Freitas, C.; Silva, A.; Costa, M.M.; Barrote, I.; Horta, P.A.; Rodrigues, A.C.; Santos, R.; Silva, J. Photoacclimation strategies in northeastern Atlantic seagrasses: Integrating responses across plant organizational levels. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Tsimilli-Michael, M.; Strasser, R.J. In vivo assessment of stress impact on plant’s vitality: Applications in detecting and evaluating the beneficial role of mycorrhization on host plants. Mycorrhiza 2008, 679–703. [Google Scholar] [CrossRef]
- Granger, S.; Lizumi, H. Water quality measurement methods for seagrass habitat. Glob. Seagrass Res. Methods 2001, 393–406. [Google Scholar] [CrossRef]
- Underwood, A.J. Experiments in Ecology; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Owens, T.G.; Falkowski, P.G.; Whitledge, T.E. Diel periodicity in cellular chlorophyll content in marine diatoms. Mar. Biol. 1980, 59, 71–77. [Google Scholar] [CrossRef]
- Ruban, A.V.; Berera, R.; Ilioaia, C.; Van Stokkum, I.H.M.; Kennis, J.T.M.; Pascal, A.A.; Van Amerongen, H.; Robert, B.; Horton, P.; Van Grondelle, R. Identification of a mechanism of photoprotective energy dissipation in higher plants. Nature 2007, 450, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.J. Efficient high light acclimation involves rapid processes at multiple mechanistic levels. J. Exp. Bot. 2015, 66, 2401–2414. [Google Scholar] [CrossRef] [Green Version]
- Stirbet, A. Govindjee On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and Photosystem II: Basics and applications of the OJIP fluorescence transient. J. Photochem. Photobiol. B Biol. 2011, 104, 236–257. [Google Scholar] [CrossRef] [PubMed]
- Wientjes, E.; Van Amerongen, H.; Croce, R. LHCII is an antenna of both photosystems after long-term acclimation. Biochim. Biophys. Acta Bioenerg. 2013, 1827, 420–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raven, J.A. The cost of photoinhibition. Physiol. Plant. 2011, 142, 87–104. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.X.; Chen, L.S.; Zheng, J.G.; Han, S.; Tang, N.; Smith, B.R. Aluminum-induced effects on Photosystem II photochemistry in Citrus leaves assessed by the chlorophyll a fluorescence transient. Tree Physiol. 2008, 28, 1863–1871. [Google Scholar] [CrossRef]
- Broetto, F.; Monteiro Duarte, H.; Lüttge, U. Responses of chlorophyll fluorescence parameters of the facultative halophyte and C3-CAM intermediate species Mesembryanthemum crystallinum to salinity and high irradiance stress. J. Plant Physiol. 2007, 164, 904–912. [Google Scholar] [CrossRef]
- Hazrati, S.; Tahmasebi-Sarvestani, Z.; Modarres-Sanavy, S.A.M.; Mokhtassi-Bidgoli, A.; Nicola, S. Effects of water stress and light intensity on chlorophyll fluorescence parameters and pigments of Aloe vera L. Plant Physiol. Biochem. 2016, 106, 141–148. [Google Scholar] [CrossRef]
- Stirbet, A.D.; Strasser, R.J. Numerical simulation of the in vivo fluorescence in plants. Math. Comput. Simul. 1996, 42, 245–253. [Google Scholar] [CrossRef]
- Yan, K.; Chen, P.; Shao, H.; Shao, C.; Zhao, S.; Brestic, M. Dissection of photosynthetic electron transport process in Sweet Sorghum under Heat Stress. PLoS ONE 2013, 8, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Goltsev, V.; Zaharieva, I.; Chernev, P.; Kouzmanova, M.; Kalaji, H.M.; Yordanov, I.; Krasteva, V.; Alexandrov, V.; Stefanov, D.; Allakhverdiev, S.I.; et al. Drought-induced modifications of photosynthetic electron transport in intact leaves: Analysis and use of neural networks as a tool for a rapid non-invasive estimation. Biochim. Biophys. Acta Bioenerg. 2012, 1817, 1490–1498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Zhou, Z.; Liang, J.; Lv, R. In vivo evaluation of the high-irradiance effects on PSII activity in photosynthetic stems of Hexinia polydichotoma. Photosynthetica 2015, 53, 621–624. [Google Scholar] [CrossRef]
- Ruban, A.V. Plants in light. Commun. Integr. Biol. 2009, 2, 50–55. [Google Scholar] [CrossRef]
- Bertelli, C.M.; Unsworth, R.K.F. Light stress responses by the eelgrass, Zostera marina (L). Front. Environ. Sci. 2018, 6, 1–13. [Google Scholar] [CrossRef]
- Esteban, R.; Barrutia, O.; Artetxe, U.; Fernández-Marín, B.; Hernández, A.; García-Plazaola, J.I. Internal and external factors affecting photosynthetic pigment composition in plants: A meta-analytical approach. New Phytol. 2015, 206, 268–280. [Google Scholar] [CrossRef]
- Collier, C.J.; Waycott, M.; Ospina, A.G. Responses of four Indo-West Pacific seagrass species to shading. Mar. Pollut. Bull. 2012, 65, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Paolacci, S.; Harrison, S.; Jansen, M.A.K. The invasive duckweed Lemna minuta Kunth displays a different light utilisation strategy than native Lemna minor Linnaeus. Aquat. Bot. 2018, 146, 8–14. [Google Scholar] [CrossRef]
- Wu, G.; Ma, L.; Sayre, R.T.; Lee, C.H. Identification of the optimal light harvesting antenna size for high-light stress mitigation in plants. Front. Plant Sci. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Zhu, S.; Qin, L.; Feng, P.; Shang, C.; Wang, Z.; Yuan, Z. Treatment of low C/N ratio wastewater and biomass production using co-culture of Chlorella vulgaris and activated sludge in a batch photobioreactor. Bioresour. Technol. 2019, 274, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Casagranda, C.; Boudouresque, C.F. Biomass of Ruppia cirrhosa and Potamogeton pectinatus in a Mediterranean brackish lagoon, Lake Ichkeul, Tunisia. Fundam. Appl. Limnol. 2007, 168, 243–255. [Google Scholar] [CrossRef] [Green Version]
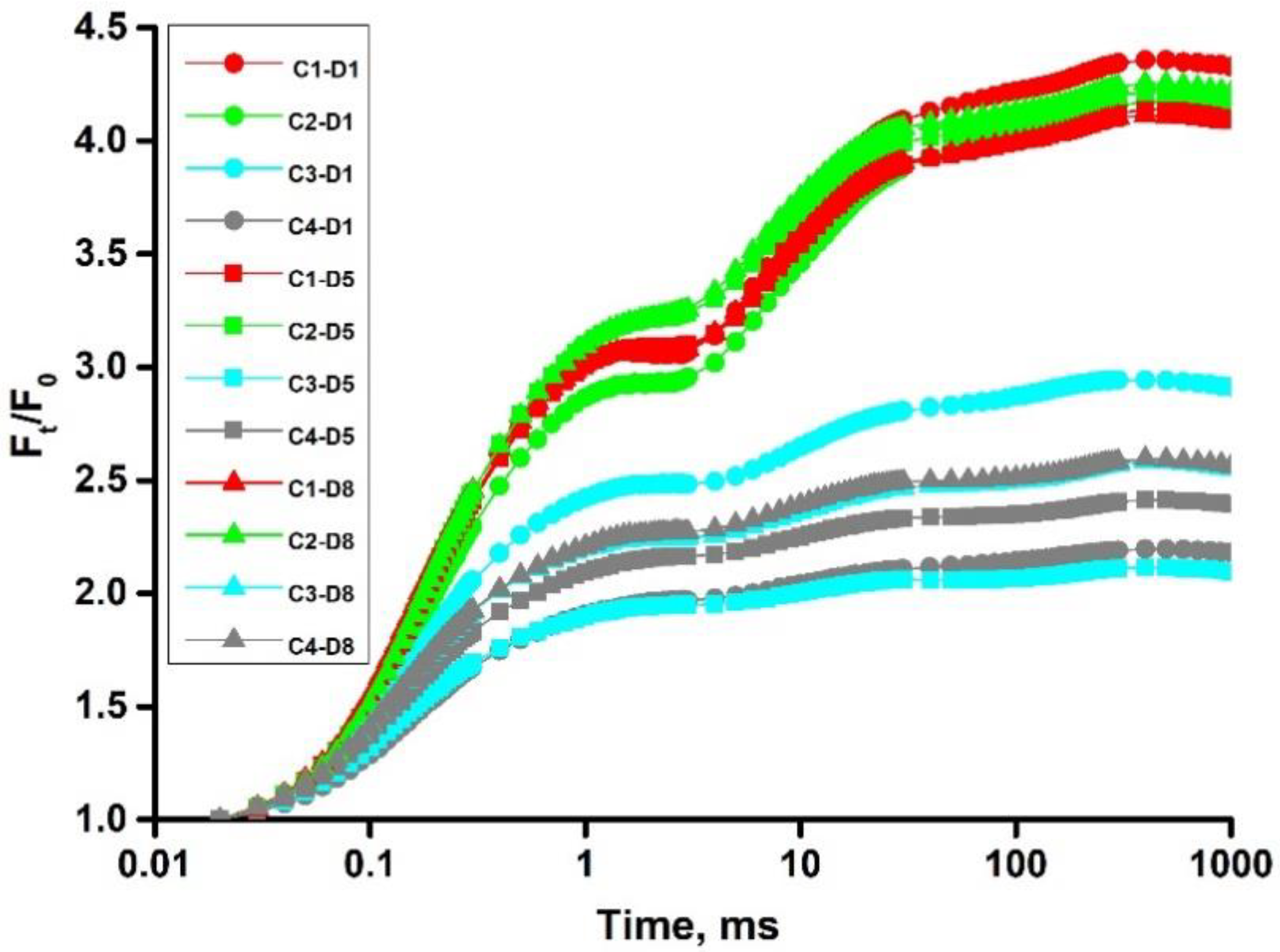
| DATA EXTRACTED FROM THE RECORDED FLUORESCENCE TRANSIENT OJIP | |
| Ft (or, simply, F) | fluorescence at time t after onset of actinic illumination |
| F20μs | minimal reliable recorded fluorescence, at 20 μs |
| F50μs | fluorescence at 50 μs (for the calculation of the slopes) |
| F100μs | fluorescence at 100 μs |
| F300μs | fluorescence at 300 μs |
| FJ ≡ F2ms | fluorescence at the J-step (2 ms) of OJIP |
| FI ≡ F30ms | fluorescence at the I-step (30 ms) of OJIP |
| FP | maximal recorded fluorescence, at the peak P of OJIP |
| BASIC PARAMETERS CALCULATED FROM THE EXTRACTED DATA | |
| F0 ≅ F20μs | fluorescence when all PSII RCs are open (≅ to the minimal reliable recorded fluorescence) |
| FM (= FP) | maximal fluorescence, when all PSII RCs are closed (= FP when the actinic light intensity is above 500 [μmol(photon) m−2 s−1]) |
| Vt ≡ (Ft − F0)/(FM − F0) | relative variable fluorescence at time t |
| M0 and M0’ ≡ [(ΔF/Δt)0]/(FM − F0) | approximated initial slopes (in ms−1) of the Vt = f(t) kinetics |
| M0 ≡ 4 × [( F300μs − F50μs)/(FM − F0)]/(Δt)0 M0’ ≡ 20 × [( F100μs − F50μs)/(FM − F0)]/(Δt)0 | with (Δt)0 = (300 − 50) μs = 0.25 ms with (Δt)0 = (100 − 50) μs = 0.05 ms |
| BIOPHYSICAL PARAMETERS DERIVED FROM THE BASIC PARAMETERS BY THE JIP-TEST | |
| Quantum yields and efficiencies/probabilities | |
| φPt ≡ TRt/ABS = [1 − (Ft/FM)] = ΔFt/FM | quantum yield for primary photochemistry, leading to QA reduction (i.e. trapped energy flux TR per absorption flux ABS), at any time t |
| φP0 ≡ TR0/ABS = [1 − (F0/FM)] | maximum quantum yield for primary photochemistry |
| φE0 ≡ ET0/ABS = [1 − (F0/FM)] × (1 − VJ) | quantum yield for electron transport (ET) further than QA− |
| φR0 ≡ RE0/ABS = [1 − (F0/FM)] × (1 − VI) | quantum yield for reduction of end electron acceptors (RE) at the PSI acceptor side |
| ψE0 ≡ ET0/TR0 = (1 − VJ) | efficiency/probability that an electron moves further than QA− |
| δR0 ≡ RE0/ET0 = (1 − VI)/(1 − VJ) | efficiency/probability that an electron from the intersystem electron carriers is transferred to reduce end electron acceptors at the PSI acceptor side |
| Specific energy flux (per active, i.e., per QA-reducing PSII reaction centre - RC), in ms−1 | |
| TR0/RC = M0 × (1/VJ) | trapped energy flux, per RC |
| DI0/RC = ABS/RC − TR0/RC | specific energy flux for dissipation per RC |
| Density of active RCs | |
| RC/ABS = (TR0/ABS) × (TR0/RC)−1 | RCs per PSII antenna Chl a |
| ABS/RC = M0 × (1/VJ) × (1/φP0) | absorption flux (exciting PSII antenna Chl a molecules) per RC (also used as a unit-less measure of PSII apparent antenna size) |
| Energetic connectivity of PSII units | |
| M0/M0’ | grouping or connectivity among PSII units (the higher is the ratio indicate the less of connectivity) |
| Axis 1 | Axis 2 | Axis 3 | Axis 4 | |
| φP0 | −0.829 | −0.165 | −0.521 | 0.011 |
| TR0/RC | 0.214 | 0.468 | 0.025 | 0.220 |
| DI0/RC | 0.881 | 0.177 | 0.423 | 0.023 |
| RC/ABS | −0.803 | −0.279 | −0.471 | −0.052 |
| ABS/RC | 0.811 | 0.266 | 0.492 | 0.044 |
| M0’/M0 | 0.750 | 0.301 | 0.398 | 0.198 |
| φR0 | −0.386 | −0.297 | −0.235 | −0.699 |
| φE0 | −0.889 | −0.167 | −0.398 | −0.066 |
| ψE0 | −0.892 | −0.168 | −0.334 | −0.101 |
| δR0 | 0.827 | 0.026 | 0.350 | −0.345 |
| Chl a | 0.331 | −0.936 | 0.042 | 0.021 |
| Chl b | 0.410 | −0.896 | 0.135 | 0.026 |
| Chl a/b | −0.365 | −0.037 | −0.379 | −0.021 |
| RGR | 0.285 | −0.076 | 0.136 | −0.768 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malea, L.; Nakou, K.; Papadimitriou, A.; Exadactylos, A.; Orfanidis, S. Physiological Responses of the Submerged Macrophyte Stuckenia pectinata to High Salinity and Irradiance Stress to Assess Eutrophication Management and Climatic Effects: An Integrative Approach. Water 2021, 13, 1706. https://doi.org/10.3390/w13121706
Malea L, Nakou K, Papadimitriou A, Exadactylos A, Orfanidis S. Physiological Responses of the Submerged Macrophyte Stuckenia pectinata to High Salinity and Irradiance Stress to Assess Eutrophication Management and Climatic Effects: An Integrative Approach. Water. 2021; 13(12):1706. https://doi.org/10.3390/w13121706
Chicago/Turabian StyleMalea, Lamprini, Konstantinia Nakou, Apostolos Papadimitriou, Athanasios Exadactylos, and Sotiris Orfanidis. 2021. "Physiological Responses of the Submerged Macrophyte Stuckenia pectinata to High Salinity and Irradiance Stress to Assess Eutrophication Management and Climatic Effects: An Integrative Approach" Water 13, no. 12: 1706. https://doi.org/10.3390/w13121706
APA StyleMalea, L., Nakou, K., Papadimitriou, A., Exadactylos, A., & Orfanidis, S. (2021). Physiological Responses of the Submerged Macrophyte Stuckenia pectinata to High Salinity and Irradiance Stress to Assess Eutrophication Management and Climatic Effects: An Integrative Approach. Water, 13(12), 1706. https://doi.org/10.3390/w13121706







