New Insights into the Seasonal Variation of DOM Quality of a Humic-Rich Drinking-Water Reservoir—Coupling 2D-Fluorescence and FTICR MS Measurements
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling
2.2. Characterization of Fluorescent Dissolved Organic Material
2.3. Solid-Phase Extraction (SPE) and FTICR MS Measurements
2.4. Correlation between fDOM, PARAFAC Components, and FTICR MS Components
2.5. Concomitant Parameters
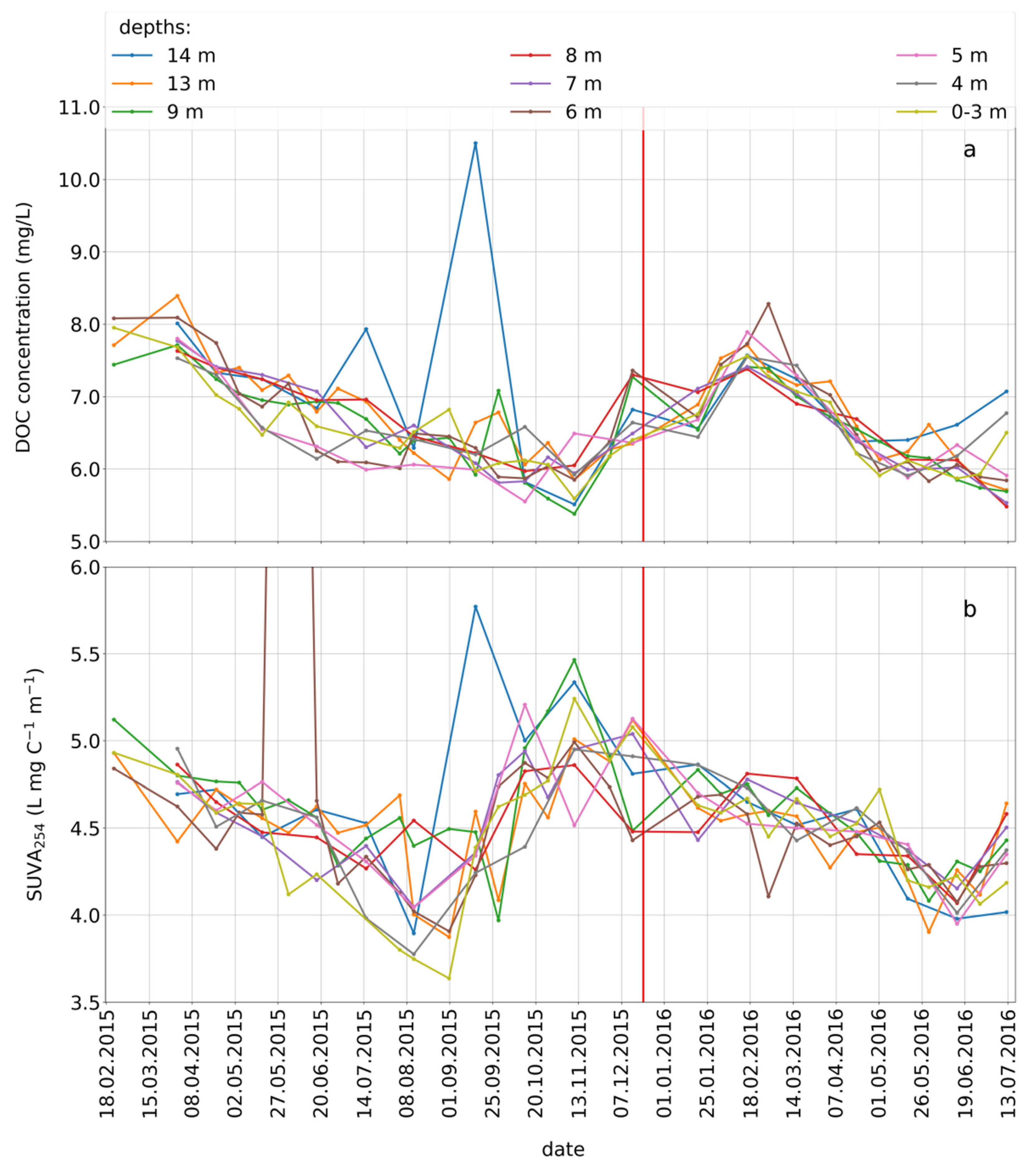
2.5.1. Concentration and Absorbance of Dissolved Organic Carbon (DOC)
2.5.2. Calculation of the Specific Ultraviolet Absorbance
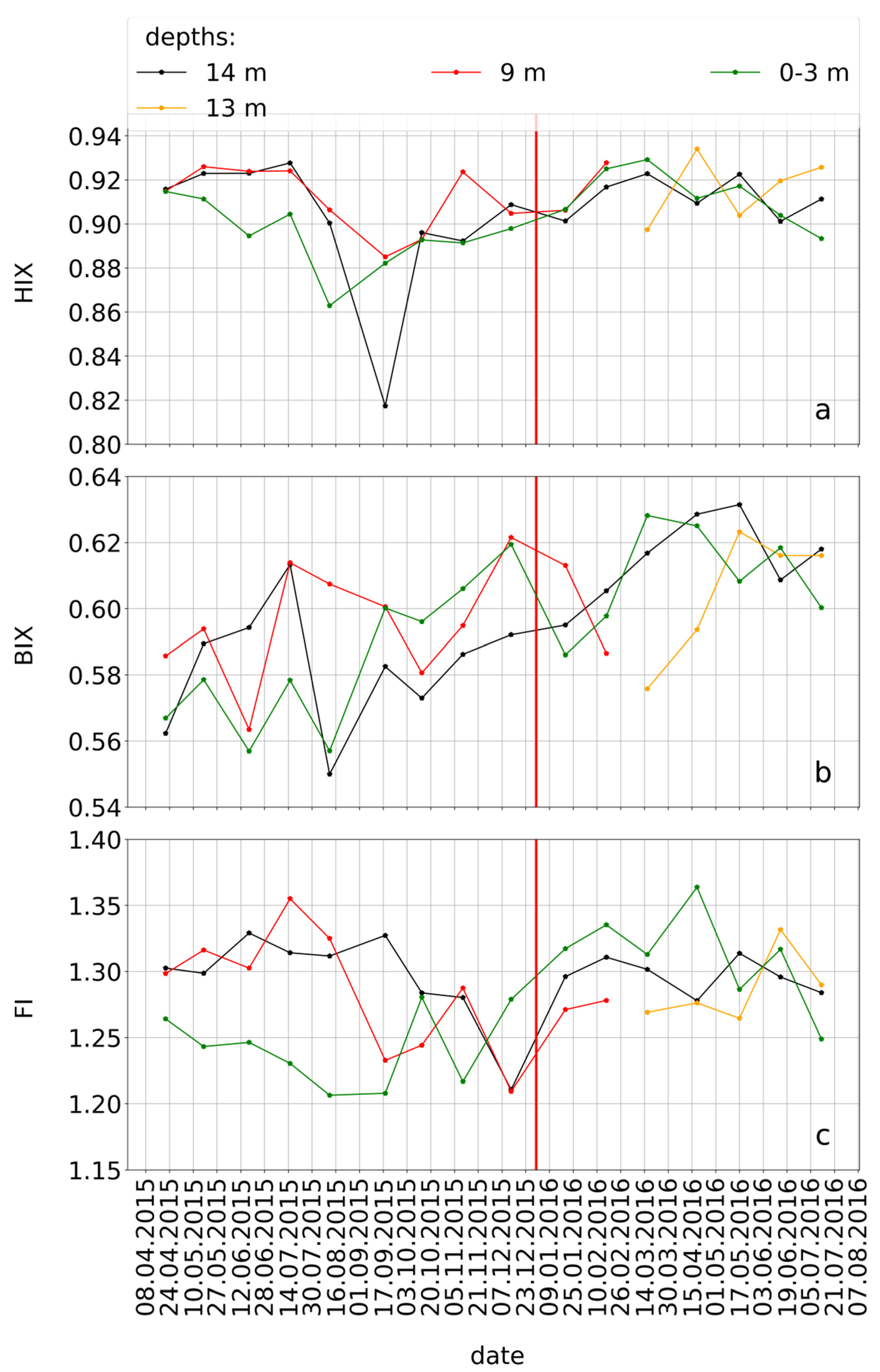
2.5.3. Calculation of the Fluorescence Indices: HIX, BIX, and FI
3. Results and Discussion
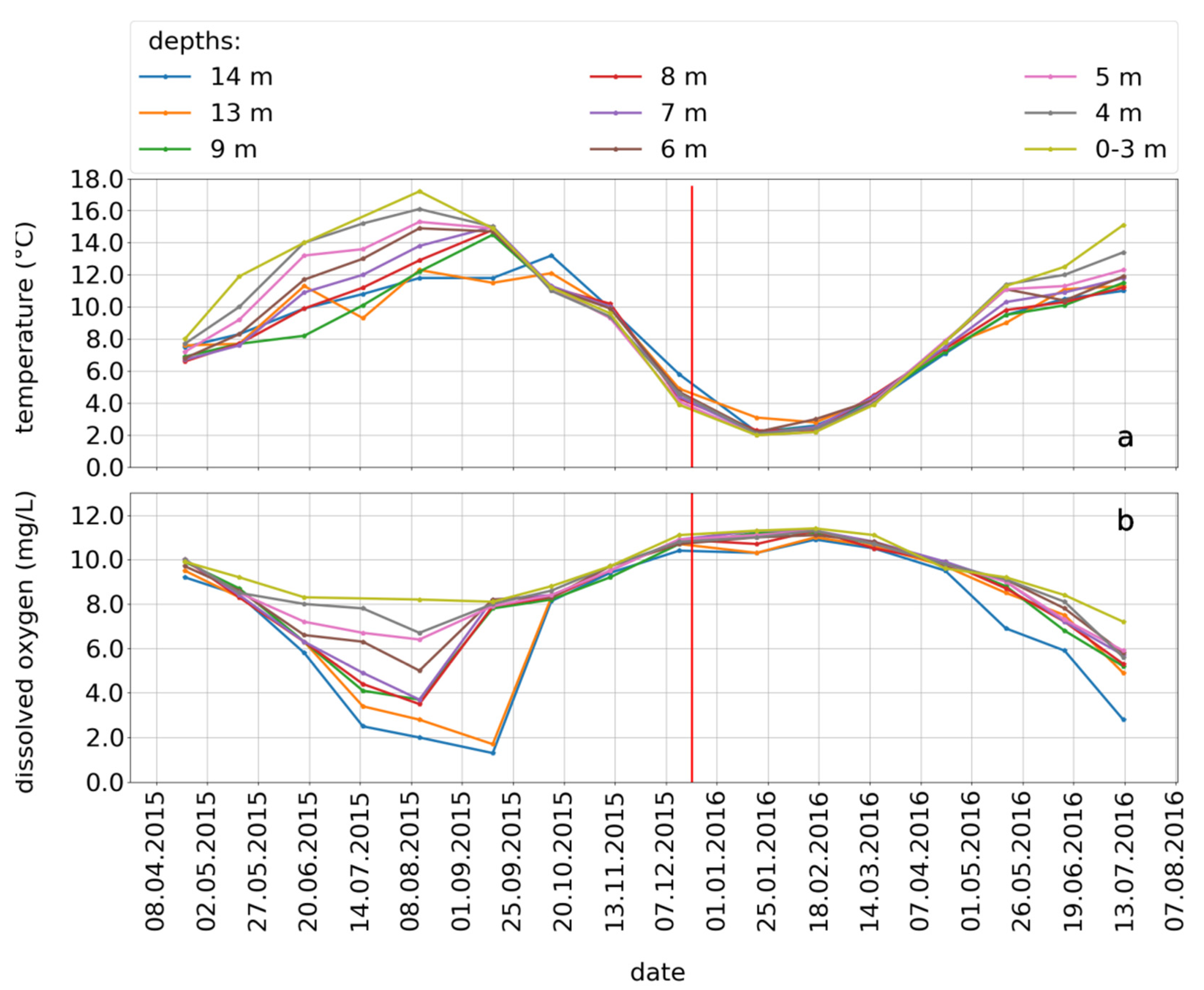
3.1. Seasonal Lake Stratification
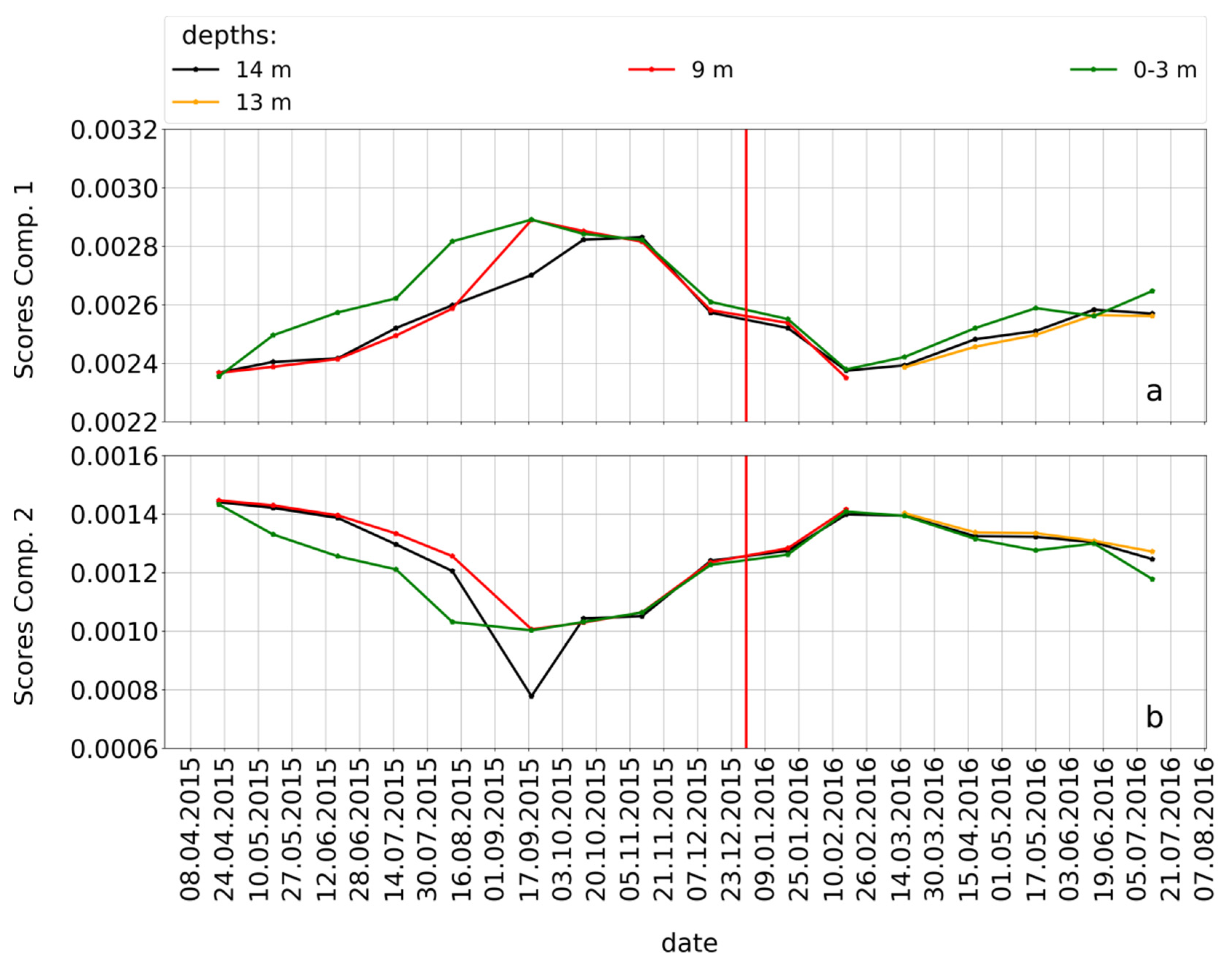
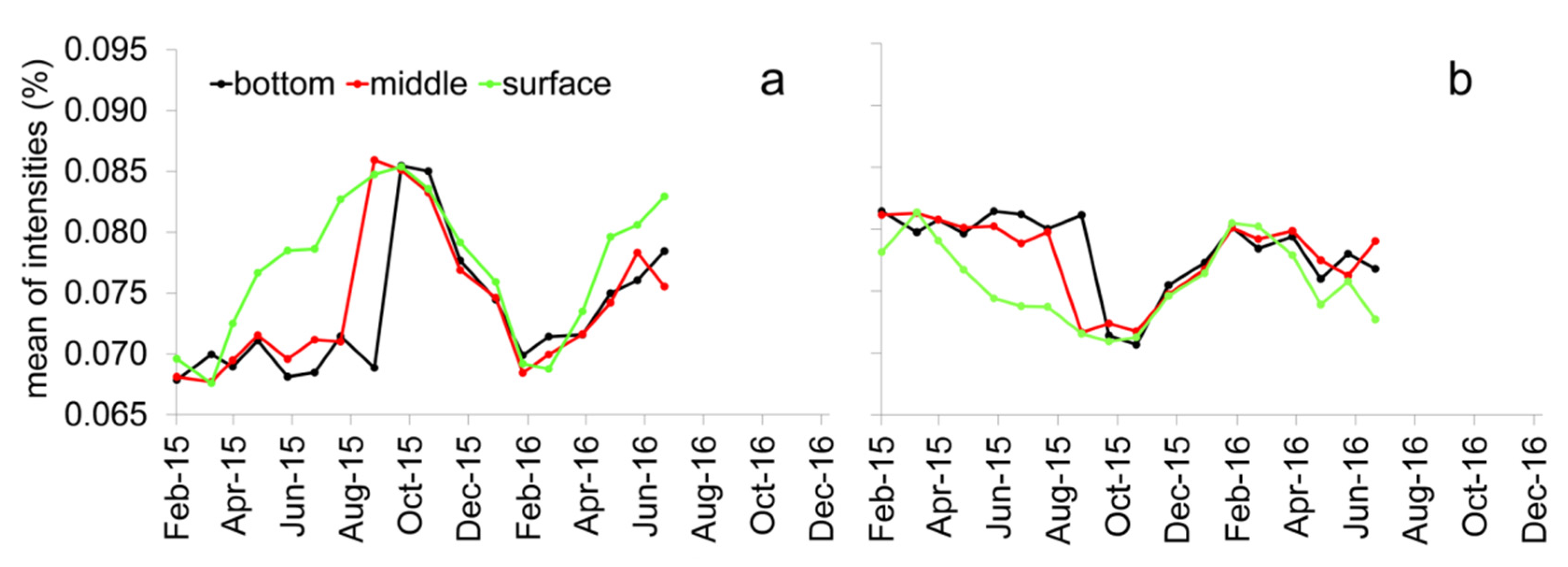
3.2. Seasonality of the PARAFAC Components within the Muldenberg Reservoir
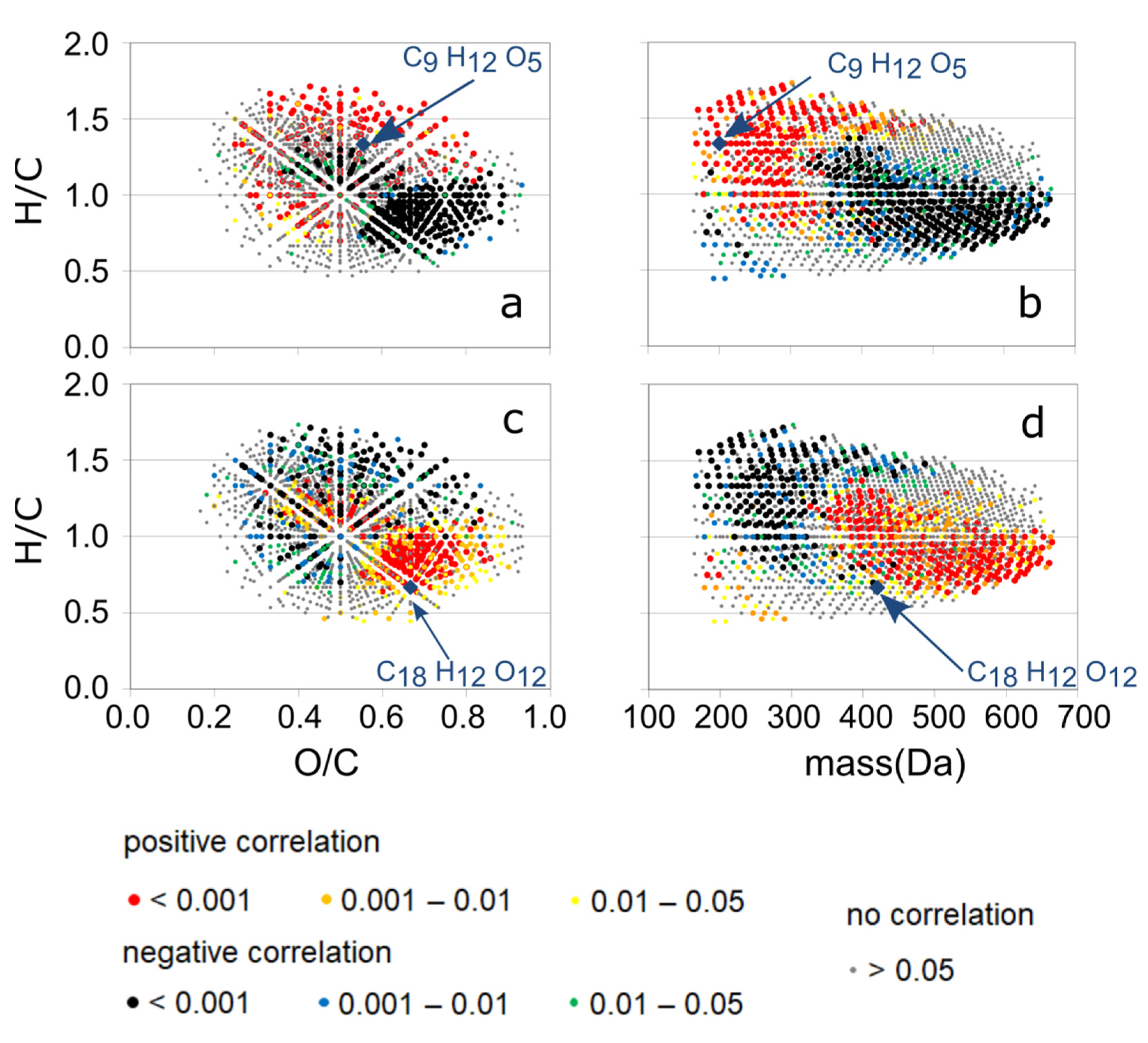
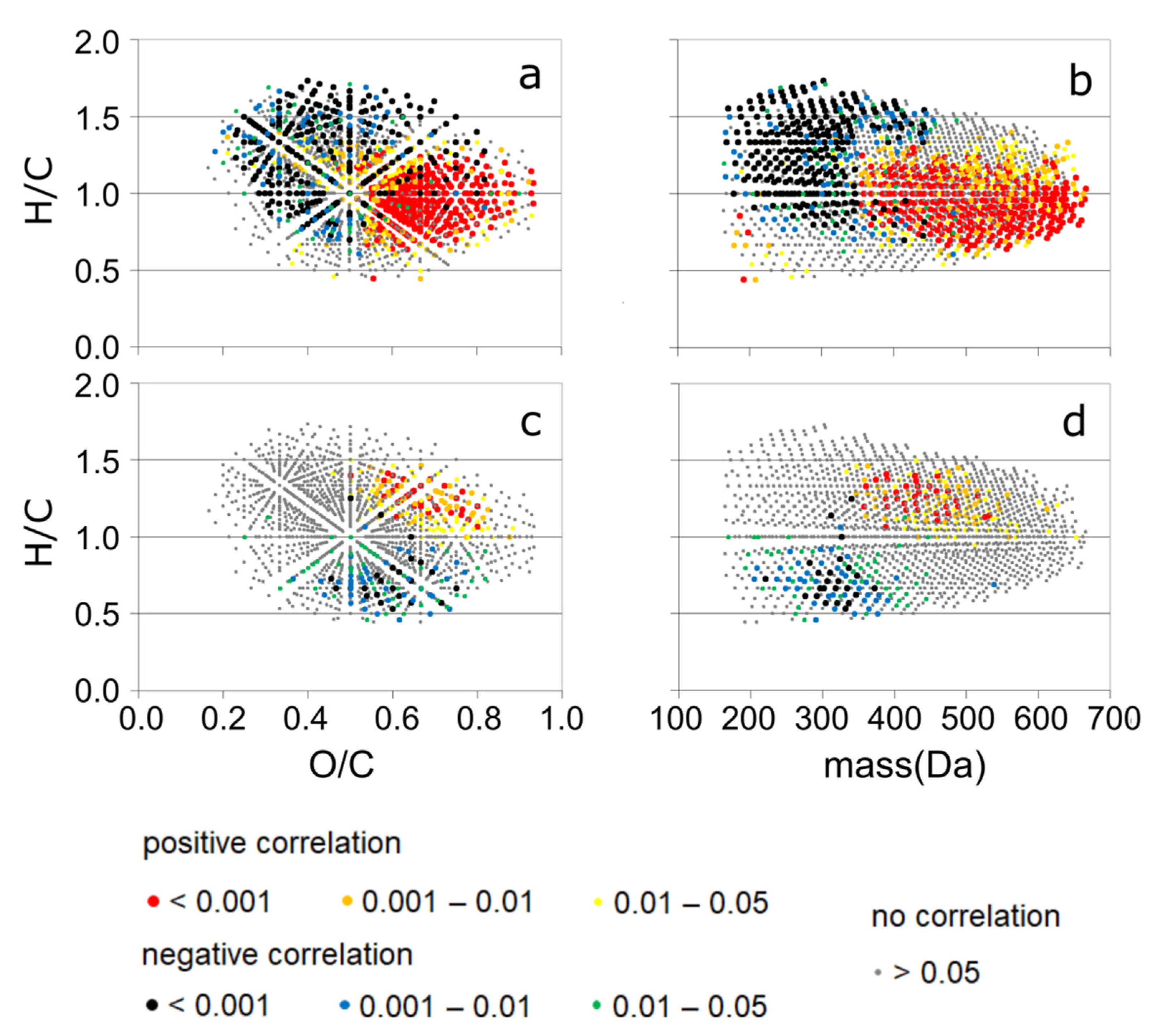
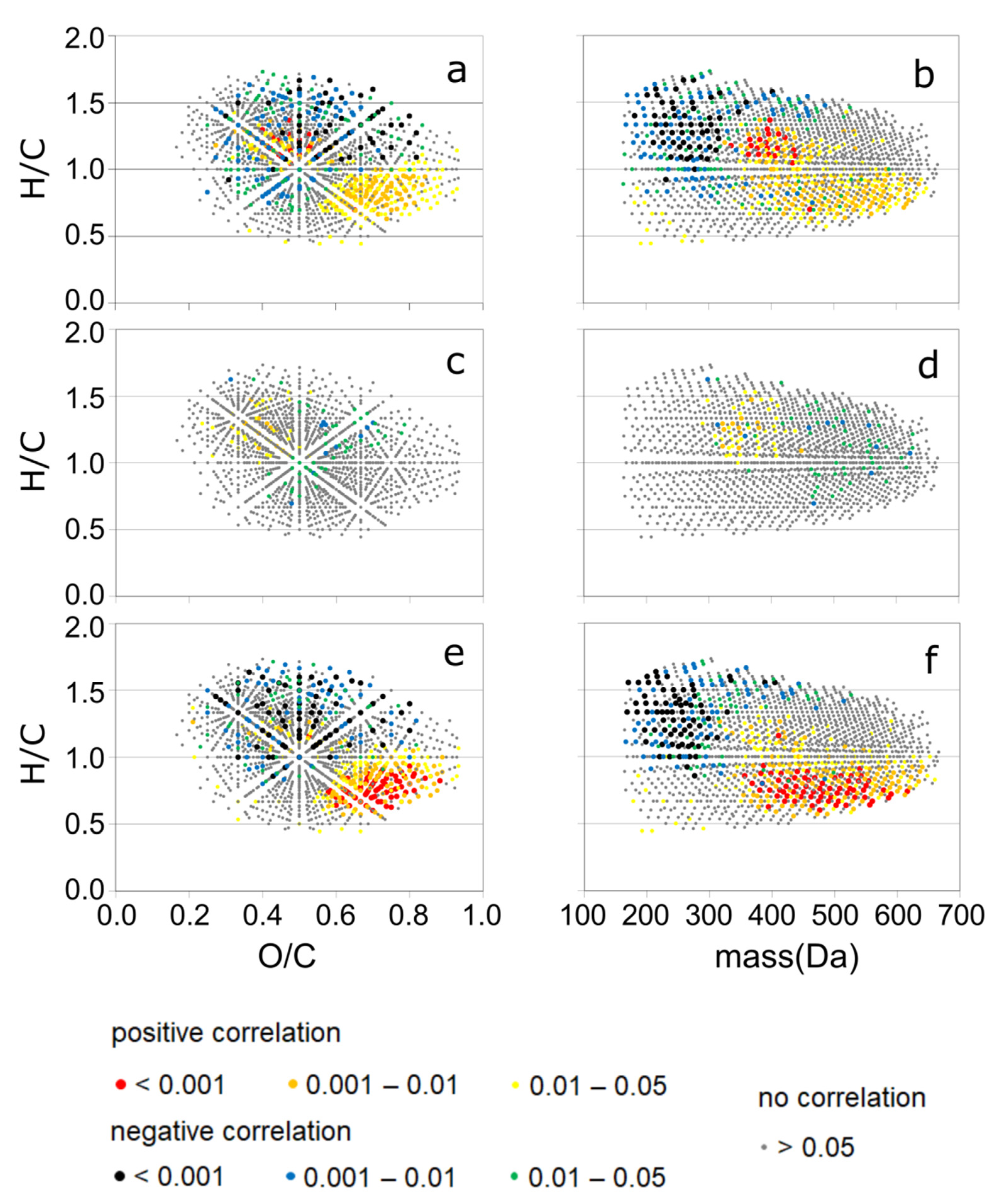
3.3. Detailed Information about DOM Seasonality from Spearman’s Rank Correlation of CHO-MF and the PARAFAC Components
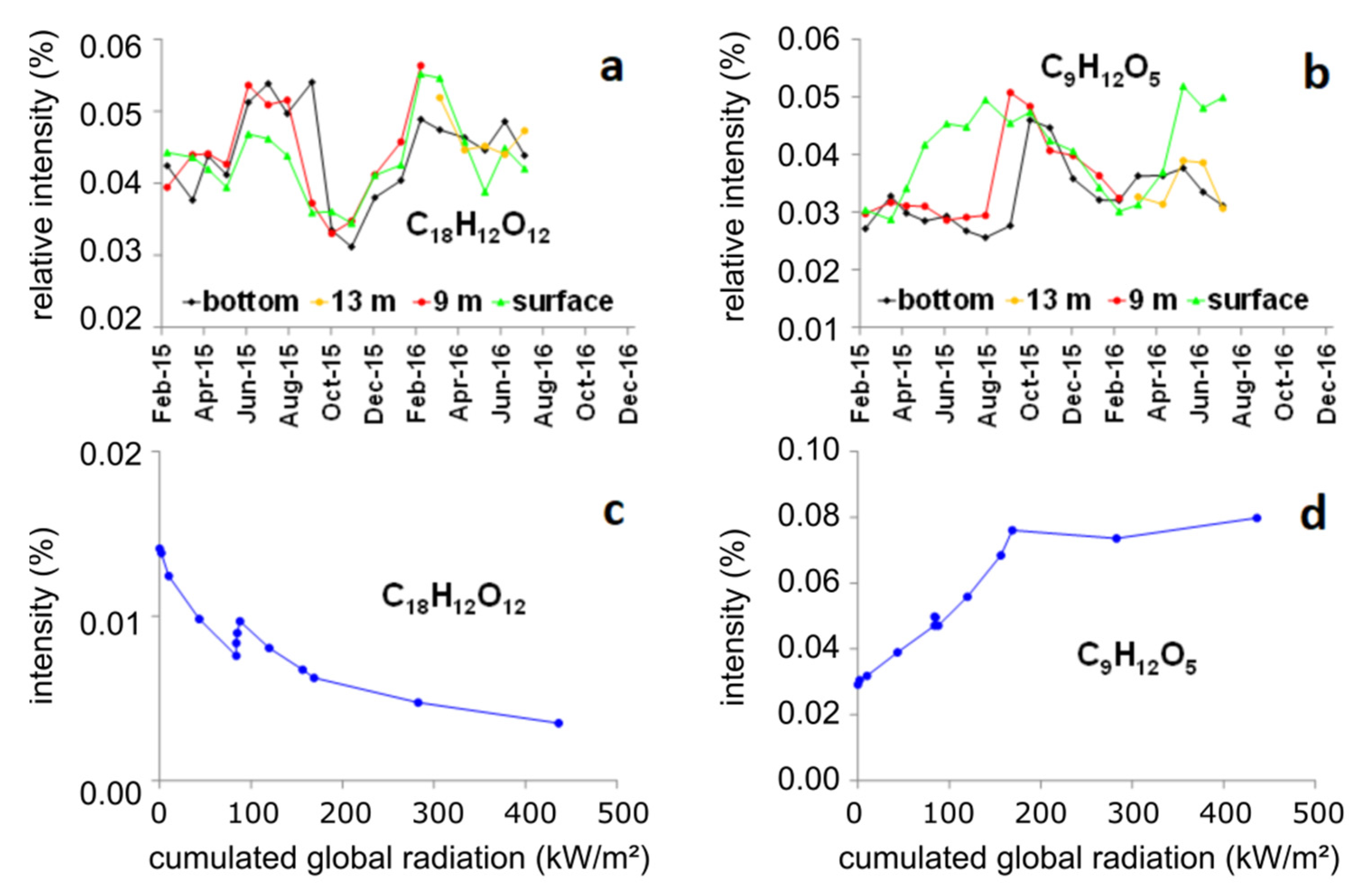
3.4. Detailed Analysis of Two Exemplary MFs and Combination with Reactivity Data from the Literature
3.5. Seasonal Variations of the Concomitant Parameters
3.6. Seasonality of the Concomitant Parameters Based on the Spearman’s Rank Correlation with the CHO-MF
3.7. Outlook in Terms of Relevance to the Waterworks
4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xu, J.; Morris, P.J.; Liu, J.; Ledesma, J.L.J.; Holden, J. Increased Dissolved Organic Carbon Concentrations in Peat-Fed UK Water Supplies Under Future Climate and Sulfate Deposition Scenarios. Water Resour. Res. 2020, 56, e2019WR025592. [Google Scholar] [CrossRef]
- Morling, K.; Herzsprung, P.; Kamjunke, N. Discharge determines production of, decomposition of and quality changes in dissolved organic carbon in pre-dams of drinking water reservoirs. Sci. Total Environ. 2017, 577, 329–339. [Google Scholar] [CrossRef]
- Herzsprung, P.; von Tumpling, W.; Hertkorn, N.; Harir, M.; Buttner, O.; Bravidor, J.; Friese, K.; Schmitt-Kopplin, P. Variations of DOM Quality in Inflows of a Drinking Water Reservoir: Linking of van Krevelen Diagrams with EEMF Spectra by Rank Correlation. Environ. Sci. Technol. 2012, 46, 5511–5518. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.E.C.; Bergamaschi, B.A.; Hernes, P.J.; Doctor, D.; Kendall, C.; Downing, B.D.; Losee, R.F. How reservoirs alter drinking water quality: Organic matter sources, sinks, and transformations. Lake Reserv. Manag. 2011, 27, 205–219. [Google Scholar] [CrossRef]
- Wilske, C.; Herzsprung, P.; Lechtenfeld, O.J.; Kamjunke, N.; von Tumpling, W. Photochemically Induced Changes of Dissolved Organic Matter in a Humic-Rich and Forested Stream. Water 2020, 12, 331. [Google Scholar] [CrossRef]
- Dadi, T.; Harir, M.; Hertkorn, N.; Koschorreck, M.; Schmitt-Kopplin, P.; Herzsprung, P. Redox Conditions Affect Dissolved Organic Carbon Quality in Stratified Freshwaters. Environ. Sci. Technol. 2017, 51, 13705–13713. [Google Scholar] [CrossRef]
- Bracchini, L.; Dattilo, A.M.; Hull, V.; Loiselle, S.A.; Nannicini, L.; Picchi, M.P.; Ricci, M.; Santinelli, C.; Seritti, A.; Tognazzi, A.; et al. Spatial and seasonal changes in optical properties of autochthonous and allochthonous chromophoric dissolved organic matter in a stratified mountain lake. Photochem. Photobiol. Sci. 2010, 9, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.; Lavonen, E.; Harir, M.; Gonsior, M.; Hertkorn, N.; Schmitt-Kopplin, P.; Kylin, H.; Bastviken, D. Selective removal of natural organic matter during drinking water production changes the composition of disinfection by-products. Environ. Sci. Water Res. Technol. 2020, 6, 779–794. [Google Scholar] [CrossRef]
- Niquette, P.; Monette, F.; Azzouz, A.; Hausler, R. Impacts of substituting aluminum-based coagulants in drinking water treatment. Water Qual. Res. J. Can. 2004, 39, 303–310. [Google Scholar] [CrossRef]
- Raeke, J.; Lechtenfeld, O.J.; Tittel, J.; Oosterwoud, M.R.; Bornmann, K.; Reemtsma, T. Linking the mobilization of dissolved organic matter in catchments and its removal in drinking water treatment to its molecular characteristics. Water Res. 2017, 113, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Awad, J.; van Leeuwen, J.; Chow, C.W.K.; Smernik, R.J.; Anderson, S.J.; Cox, J.W. Seasonal variation in the nature of DOM in a river and drinking water reservoir of a closed catchment. Environ. Pollut. 2017, 220, 788–796. [Google Scholar] [CrossRef]
- Zhang, H.F.; Zhang, Y.H.; Shi, Q.; Ren, S.Y.; Yu, J.W.; Ji, F.; Luo, W.B.; Yang, M. Characterization of low molecular weight dissolved natural organic matter along the treatment trait of a waterworks using Fourier transform ion cyclotron resonance mass spectrometry. Water Res. 2012, 46, 5197–5204. [Google Scholar] [CrossRef]
- Phungsai, P.; Kurisu, F.; Kasuga, I.; Furumai, H. Molecular characterization of low molecular weight dissolved organic matter in water reclamation processes using Orbitrap mass spectrometry. Water Res. 2016, 100, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Phungsai, P.; Kurisu, F.; Kasuga, I.; Furumai, H. Changes in Dissolved Organic Matter Composition and Disinfection Byproduct Precursors in Advanced Drinking Water Treatment Processes. Environ. Sci. Technol. 2018, 52, 3392–3401. [Google Scholar] [CrossRef]
- Phungsai, P.; Kurisu, F.; Kasuga, I.; Furumai, H. Molecular characteristics of dissolved organic matter transformed by O3 and O3/H2O2 treatments and the effects on formation of unknown disinfection by-products. Water Res. 2019, 159, 214–222. [Google Scholar] [CrossRef] [PubMed]
- ISO. DIN EN 1484: 2019-04. Water Analysis—Guidelines for the Determination of Total Organic Carbon (TOC) and Dissolved Organic Carbon (DOC); ISO: Geneva, Switherland, 2019; (German version EN 1484: 1997). [Google Scholar]
- Peacock, M.; Evans, C.D.; Fenner, N.; Freeman, C.; Gough, R.; Jones, T.G.; Lebron, I. UV-visible absorbance spectroscopy as a proxy for peatland dissolved organic carbon (DOC) quantity and quality: Considerations on wavelength and absorbance degradation. Environ. Sci. Process. Impacts 2014, 16, 1445–1461. [Google Scholar] [CrossRef]
- Lin, H.J.; Dai, Q.Y.; Zheng, L.L.; Hong, H.C.; Deng, W.J.; Wu, F.Y. Radial basis function artificial neural network able to accurately predict disinfection by-product levels in tap water: Taking haloacetic acids as a case study. Chemosphere 2020, 248, 125999. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.C.; Zhang, Z.Y.; Guo, A.D.; Shen, L.G.; Sun, H.J.; Liang, Y.; Wu, F.Y.; Lin, H.J. Radial basis function artificial neural network (RBF ANN) as well as the hybrid method of RBF ANN and grey relational analysis able to well predict trihalomethanes levels in tap water. J. Hydrol. 2020, 591, 125574. [Google Scholar] [CrossRef]
- Deng, Y.; Zhou, X.L.; Shen, J.; Xiao, G.; Hong, H.C.; Lin, H.J.; Wu, F.Y.; Liao, B.Q. New methods based on back propagation (BP) and radial basis function (RBF) artificial neural networks (ANNs) for predicting the occurrence of haloketones in tap water. Sci. Total Environ. 2021, 772, 145534. [Google Scholar] [CrossRef] [PubMed]
- Howard, D.W.; Hounshell, A.G.; Lofton, M.E.; Woelmer, W.M.; Hanson, P.C.; Carey, C.C. Variability in fluorescent dissolved organic matter concentrations across diel to seasonal time scales is driven by water temperature and meteorology in a eutrophic reservoir. Aquat. Sci. 2021, 83, 1–18. [Google Scholar] [CrossRef]
- Wang, X.C.; Zhang, H.; Bertone, E.; Stewart, R.A.; O’Halloran, K. Analysis of the Mixing Processes in a Shallow Subtropical Reservoir and Their Effects on Dissolved Organic Matter. Water 2019, 11, 737. [Google Scholar] [CrossRef]
- Saadi, I.; Borisover, M.; Armon, R.; Laor, Y. Monitoring of effluent DOM biodegradation using fluorescence, UV and DOC measurements. Chemosphere 2006, 63, 530–539. [Google Scholar] [CrossRef]
- Wang, Z.W.; Wu, Z.C.; Tang, S.J. Characterization of dissolved organic matter in a submerged membrane bioreactor by using three-dimensional excitation and emission matrix fluorescence spectroscopy. Water Res. 2009, 43, 1533–1540. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.Y.; Hur, J.; Zhuang, W.N. Occurrence and behaviors of fluorescence EEM-PARAFAC components in drinking water and wastewater treatment systems and their applications: A review. Environ. Sci. Pollut. Res. 2015, 22, 6500–6510. [Google Scholar] [CrossRef] [PubMed]
- Ishii, S.K.L.; Boyer, T.H. Behavior of Reoccurring PARAFAC Components in Fluorescent Dissolved Organic Matter in Natural and Engineered Systems: A Critical Review. Environ. Sci. Technol. 2012, 46, 2006–2017. [Google Scholar] [CrossRef]
- Gao, J.K.; Shi, Z.Y.; Wu, H.M.; Lv, J.L. Fluorescent characteristics of dissolved organic matter released from biochar and paddy soil incorporated with biochar. RSC Adv. 2020, 10, 5785–5793. [Google Scholar] [CrossRef]
- Zhao, Y.; Song, K.S.; Wen, Z.D.; Li, L.; Zang, S.Y.; Shao, T.T.; Li, S.J.; Du, J. Seasonal characterization of CDOM for lakes in semiarid regions of Northeast China using excitation-emission matrix fluorescence and parallel factor analysis (EEM-PARAFAC). Biogeosciences 2016, 13, 1635–1645. [Google Scholar] [CrossRef]
- Wang, W.W.; Zheng, B.H.; Jiang, X.; Chen, J.Y.; Wang, S.H. Characteristics and Source of Dissolved Organic Matter in Lake Hulun, A Large Shallow Eutrophic Steppe Lake in Northern China. Water 2020, 12, 953. [Google Scholar] [CrossRef]
- Herzsprung, P.; Wentzky, V.; Kamjunke, N.; von Tümpling, W.; Wilske, C.; Friese, K.; Boehrer, B.; Reemtsma, T.; Rinke, K.; Lechtenfeld, O.J. Improved Understanding of Dissolved Organic Matter Processing in Freshwater Using Complementary Experimental and Machine Learning Approaches. Environ. Sci. Technol. 2020, 54, 13556–13565. [Google Scholar] [CrossRef] [PubMed]
- Herzsprung, P.; Osterloh, K.; von Tumpling, W.; Harir, M.; Hertkorn, N.; Schmitt-Kopplin, P.; Meissner, R.; Bernsdorf, S.; Friese, K. Differences in DOM of rewetted and natural peatlands—Results from high-field FT-ICR-MS and bulk optical parameters. Sci. Total Environ. 2017, 586, 770–781. [Google Scholar] [CrossRef] [PubMed]
- Stubbins, A.; Lapierre, J.F.; Berggren, M.; Prairie, Y.T.; Dittmar, T.; del Giorgio, P.A. What’s in an EEM? Molecular Signatures Associated with Dissolved Organic Fluorescence in Boreal Canada. Environ. Sci. Technol. 2014, 48, 10598–10606. [Google Scholar] [CrossRef] [PubMed]
- Hertkorn, N.; Frommberger, M.; Witt, M.; Koch, B.P.; Schmitt-Kopplin, P.; Perdue, E.M. Natural organic matter and the event horizon of mass spectrometry. Anal. Chem. 2008, 80, 8908–8919. [Google Scholar] [CrossRef] [PubMed]
- Gesetz zur Ordnung des Wasserhaushaltsgesetz—WHG. § 51 Festsetzung von Wasserschutzgebieten, § 52 Besondere Anforderungen in Wasserschutzgebieten. 2019. Available online: https://www.gesetze-im-internet.de/whg_2009/WHG.pdf (accessed on 8 May 2021).
- Bro, R.; Kiers, H.A.L. A new efficient method for determining the number of components in PARAFAC models. J. Chemom. 2003, 17, 274–286. [Google Scholar] [CrossRef]
- Harshman, R.A.; Lundy, M.E. PARAFAC—Parallel Factor-Analysis. Comput. Stat. Data Anal. 1994, 18, 39–72. [Google Scholar] [CrossRef]
- Carroll, J.D.; Chang, J.J. Analysis of Individual Differences in Multidimensional Scaling via an N-way Generalization of Eckart-Young Decomposition. Psychometrika 1970, 35, 283–319. [Google Scholar] [CrossRef]
- Harshman., R.A. Foundations of the PARAFAC procedure: Models and conditions for an “explanatory” multi-modal factor analysis. UCLA Work. Pap. Phon. 1970, 16, 1–84. [Google Scholar]
- Stedmon, C.A.; Markager, S.; Bro, R. Tracing dissolved organic matter in aquatic environments using a new approach to fluorescence spectroscopy. Mar. Chem. 2003, 82, 239–254. [Google Scholar] [CrossRef]
- Dittmar, T.; Koch, B.; Hertkorn, N.; Kattner, G. A simple and efficient method for the solid-phase extraction of dissolved organic matter (SPE-DOM) from seawater. Limnol. Oceanogr. Methods 2008, 6, 230–235. [Google Scholar] [CrossRef]
- Raeke, J.; Lechtenfeld, O.J.; Wagner, M.; Herzsprung, P.; Reemtsma, T. Selectivity of solid phase extraction of freshwater dissolved organic matter and its effect on ultrahigh resolution mass spectra. Environ. Sci. Process. Impacts 2016, 18, 918–927. [Google Scholar] [CrossRef]
- Lechtenfeld, O.J.; Kattner, G.; Flerus, R.; McCallister, S.L.; Schmitt-Kopplin, P.; Koch, B.P. Molecular transformation and degradation of refractory dissolved organic matter in the Atlantic and Southern Ocean. Geochim. Cosmochim. Acta 2014, 126, 321–337. [Google Scholar] [CrossRef]
- Koch, B.P.; Kattner, G.; Witt, M.; Passow, U. Molecular insights into the microbial formation of marine dissolved organic matter: Recalcitrant or labile? Biogeosciences 2014, 11, 4173–4190. [Google Scholar] [CrossRef]
- Herzsprung, P.; Hertkorn, N.; von Tumpling, W.; Harir, M.; Friese, K.; Schmitt-Kopplin, P. Understanding molecular formula assignment of Fourier transform ion cyclotron resonance mass spectrometry data of natural organic matter from a chemical point of view. Anal. Bioanal. Chem. 2014, 406, 7977–7987. [Google Scholar] [CrossRef] [PubMed]
- Flerus, R.; Lechtenfeld, O.J.; Koch, B.P.; McCallister, S.L.; Schmitt-Kopplin, P.; Benner, R.; Kaiser, K.; Kattner, G. A molecular perspective on the ageing of marine dissolved organic matter. Biogeosciences 2012, 9, 1935–1955. [Google Scholar] [CrossRef]
- Hartung, J.E.B.; Klösener, K.H. Statistik-Lehr-und Handbuch de angewandten Statistik. Oldenbourg 2002, 546. Available online: https://www.amazon.de/Statistik-Lehr-Handbuch-angewandten/dp/3486578901 (accessed on 16 June 2021). ISBN 9783486259056.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 13 March 2019).
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate—A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Stat. Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Spencer, R.G.M.; Butler, K.D.; Aiken, G.R. Dissolved organic carbon and chromophoric dissolved organic matter properties of rivers in the USA. J. Geophys. Res. Biogeosci. 2012, 117. [Google Scholar] [CrossRef]
- Weishaar, J.L.; Aiken, G.R.; Bergamaschi, B.A.; Fram, M.S.; Fujii, R.; Mopper, K. Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ. Sci. Technol. 2003, 37, 4702–4708. [Google Scholar] [CrossRef] [PubMed]
- Edzwald, J.K.; Tobiason, J.E. Enhanced coagulation: US requirements and a broader view. Water Sci. Technol. 1999, 40, 63–70. [Google Scholar] [CrossRef]
- Ohno, T. Fluorescence inner-filtering correction for determining the humification index of dissolved organic matter. Environ. Sci. Technol. 2002, 36, 742–746. [Google Scholar] [CrossRef]
- Cory, R.M.; McKnight, D.M. Fluorescence spectroscopy reveals ubiquitous presence of oxidized and reduced quinones in dissolved organic matter. Environ. Sci. Technol. 2005, 39, 8142–8149. [Google Scholar] [CrossRef]
- Parlanti, E.; Worz, K.; Geoffroy, L.; Lamotte, M. Dissolved organic matter fluorescence spectroscopy as a tool to estimate biological activity in a coastal zone submitted to anthropogenic inputs. Org. Geochem. 2000, 31, 1765–1781. [Google Scholar] [CrossRef]
- Lavonen, E.E.; Kothawala, D.N.; Tranvik, L.J.; Gonsior, M.; Schmitt-Kopplin, P.; Kohler, S.J. Tracking changes in the optical properties and molecular composition of dissolved organic matter during drinking water production. Water Res. 2015, 85, 286–294. [Google Scholar] [CrossRef]
- Nürnberg, G.K. Quantified hypoxia and anoxia in lakes and reservoirs. Sci. World J. 2004, 4, 42–54. [Google Scholar] [CrossRef]
- Miller, M.P.; McKnight, D.M. Comparison of seasonal changes in fluorescent dissolved organic matter among aquatic lake and stream sites in the Green Lakes Valley. J. Geophys. Res. Biogeosci. 2010, 115. [Google Scholar] [CrossRef]
- Hur, J.; Lee, B.M.; Lee, S.; Shin, J.K. Characterization of chromophoric dissolved organic matter and trihalomethane formation potential in a recently constructed reservoir and the surrounding areas—Impoundment effects. J. Hydrol. 2014, 515, 71–80. [Google Scholar] [CrossRef]
- Markechova, D.; Tomkova, M.; Sadecka, J. Fluorescence Excitation-Emission Matrix Spectroscopy and Parallel Factor Analysis in Drinking Water Treatment: A Review. Pol. J. Environ. Stud. 2013, 22, 1289–1295. [Google Scholar]
| DOC | SUVA | HIX | BIX | FI | Comp1 | Comp2 | |
|---|---|---|---|---|---|---|---|
| DOC | 1 | ||||||
| SUVA | 0.021 | 1 | |||||
| HIX | 0.421 | −0.005 | 1 | ||||
| BIX | −0.065 | −0.160 | 0.260 | 1 | |||
| FI | 0.357 | −0.059 | 0.347 | 0.134 | 1 | ||
| Comp1 | −0.677 | −0.034 | −0.719 | −0.060 | −0.437 | 1 | |
| Comp2 | 0.583 | −0.035 | 0.728 | 0.093 | 0.467 | −0.970 | 1 |
| MF | Mass | Byprod. Precursor | Reactivity | Rank Correlation Coefficient | ||||
|---|---|---|---|---|---|---|---|---|
| Score 1 | Score 2 | HIX | BIX | DOC | ||||
| C9H12O5 | 200.068 | - | Chla+ Rad + | +0.62 | −0.56 | −0.68 | 0 | −0.69 |
| C18H12O12 | 420.033 | - | Rad− | −0.54 | +0.48 | +0.63 | +0.20 | +0.34 |
| C9H12O6 | 216.063 | Yes | Chla+ Rad+ | +0.64 | −0.59 | −0.69 | −0.02 | −0.67 |
| C10H14O7 | 246.074 | Yes | Chla+ Rad+ | +0.72 | −0.67 | −0.72 | −0.05 | −0.68 |
| C16H12O10 | 364.043 | Yes | Chla− Rad− | −0.34 | +0.29 | +0.35 | −0.003 | +0.24 |
| C14H16O8 | 312.085 | Yes | Chla+ Rad0 | +0.27 | −0.90 | −0.94 | −0.51 | −0.97 |
| C19H28O8 | 384.178 | Yes | Chla+ Rad0 | +0.26 | −0.23 | −0.12 | +0.45 | −0.36 |
| C18H14O12 | 422.049 | - | Chla− Rad− | −0.65 | +0.55 | +0.63 | +0.04 | +0.58 |
| C18H14O13 | 438.043 | - | Rad− | −0.61 | +0.52 | +0.64 | +0.22 | +0.54 |
| C18H14O14 | 454.038 | - | - | −0.57 | +0.47 | +0.63 | +0.22 | +0.53 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilske, C.; Herzsprung, P.; Lechtenfeld, O.J.; Kamjunke, N.; Einax, J.W.; von Tümpling, W. New Insights into the Seasonal Variation of DOM Quality of a Humic-Rich Drinking-Water Reservoir—Coupling 2D-Fluorescence and FTICR MS Measurements. Water 2021, 13, 1703. https://doi.org/10.3390/w13121703
Wilske C, Herzsprung P, Lechtenfeld OJ, Kamjunke N, Einax JW, von Tümpling W. New Insights into the Seasonal Variation of DOM Quality of a Humic-Rich Drinking-Water Reservoir—Coupling 2D-Fluorescence and FTICR MS Measurements. Water. 2021; 13(12):1703. https://doi.org/10.3390/w13121703
Chicago/Turabian StyleWilske, Christin, Peter Herzsprung, Oliver J. Lechtenfeld, Norbert Kamjunke, Jürgen W. Einax, and Wolf von Tümpling. 2021. "New Insights into the Seasonal Variation of DOM Quality of a Humic-Rich Drinking-Water Reservoir—Coupling 2D-Fluorescence and FTICR MS Measurements" Water 13, no. 12: 1703. https://doi.org/10.3390/w13121703
APA StyleWilske, C., Herzsprung, P., Lechtenfeld, O. J., Kamjunke, N., Einax, J. W., & von Tümpling, W. (2021). New Insights into the Seasonal Variation of DOM Quality of a Humic-Rich Drinking-Water Reservoir—Coupling 2D-Fluorescence and FTICR MS Measurements. Water, 13(12), 1703. https://doi.org/10.3390/w13121703










