Distribution, Drivers, and Threats of Aluminum in Groundwater in Nova Scotia, Canada
Abstract
1. Introduction
2. Materials and Methods
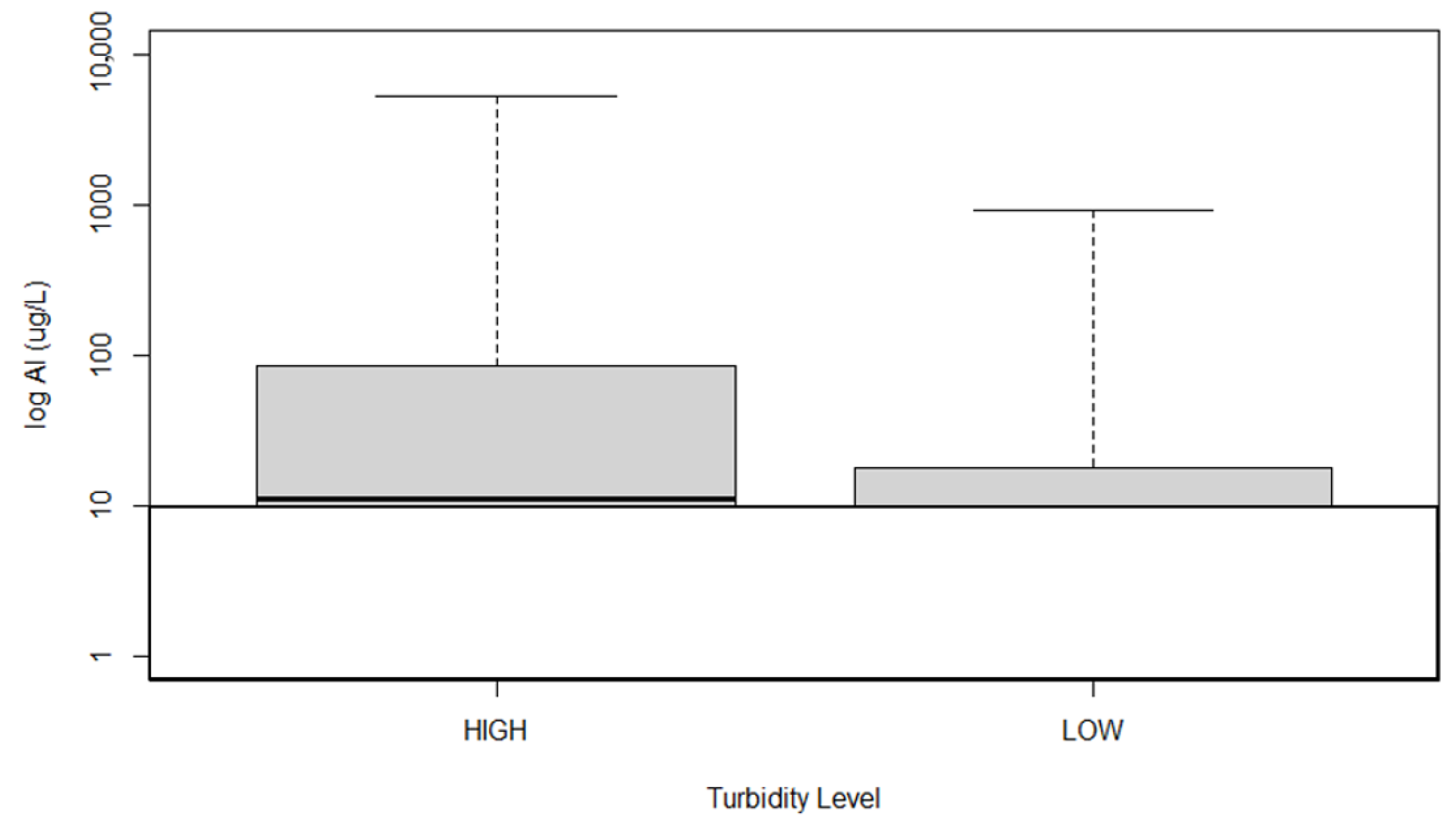
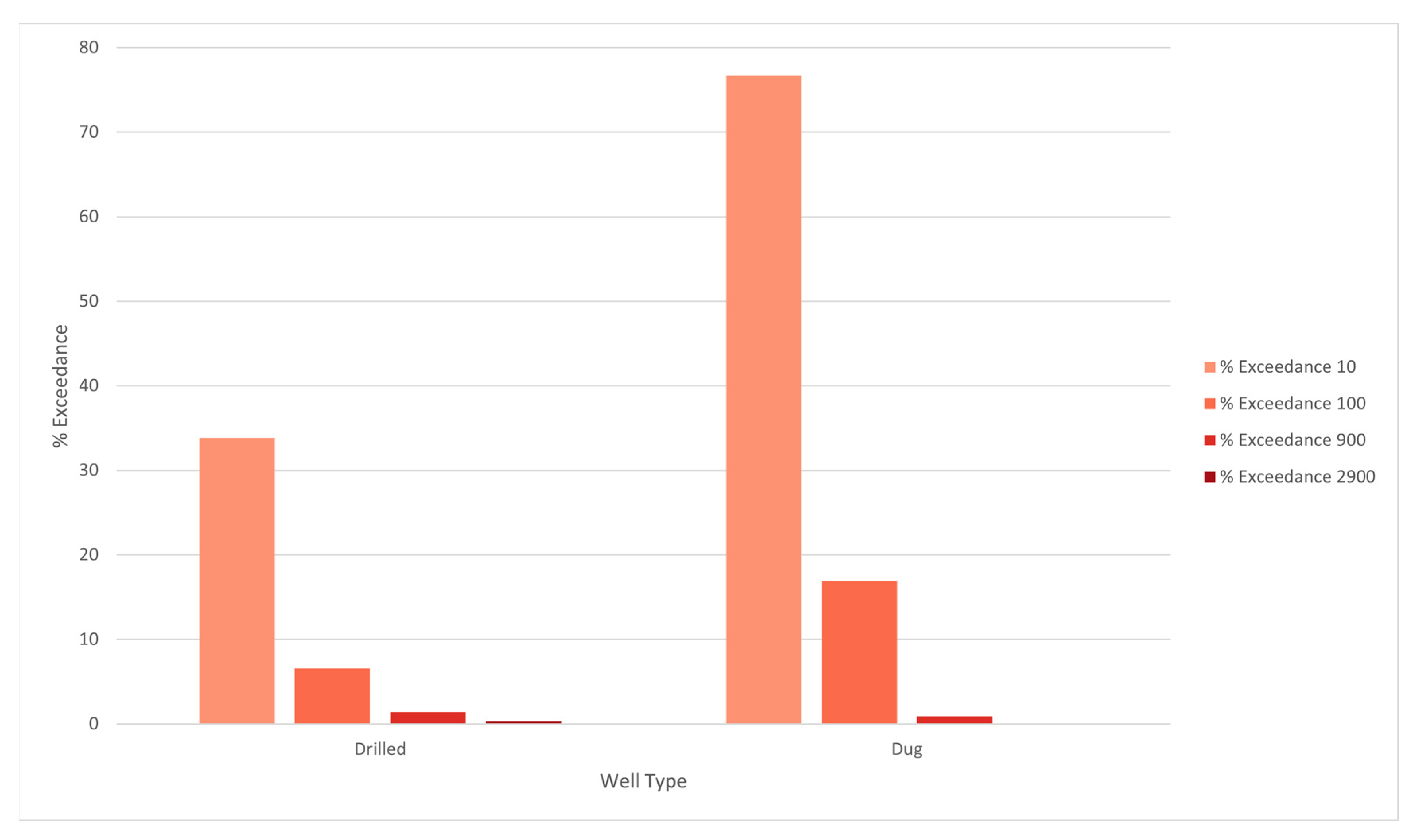
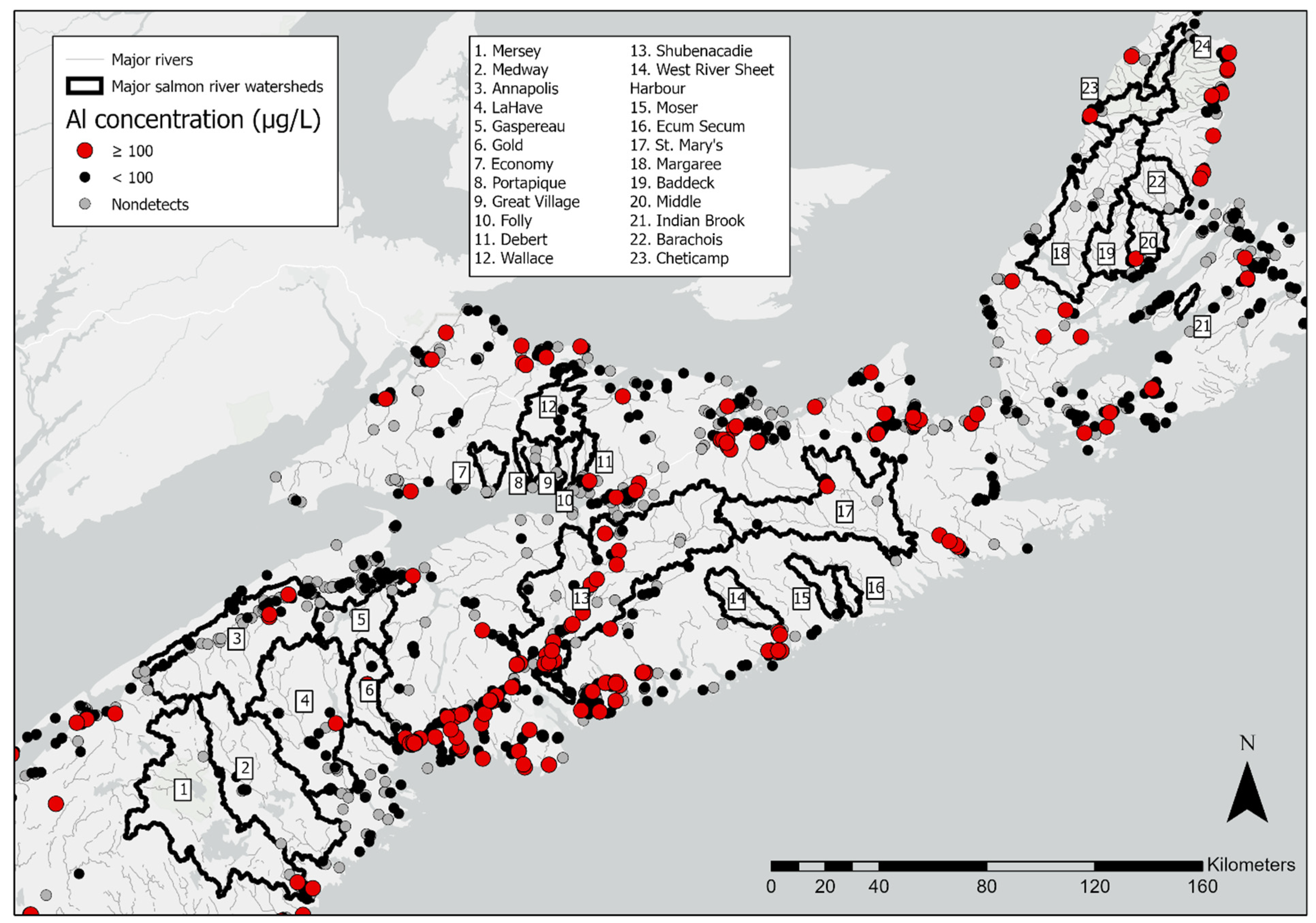
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baekken, T.; Kroglund, F.; Lindstrom, E.-A.; Carvalho, L. Acidification of rivers and lakes. In Indicators and Methods for the Ecological Status Assessment under the Water Framework Directive: Linkage between Chemical and Biological Quality of Surface Waters; Solimini, A.G., Cardoso, A.C., Heiskanen, A.-S., Eds.; Institute for Environment and Sustainability: Ispra, Italy, 2006; pp. 117–132. [Google Scholar]
- Clair, T.A.; Hindar, A. Liming for the Mitigation of Acid Rain Effects in Freshwaters: A Review of Recent Results. Environ. Rev. 2005, 13, 91–128. [Google Scholar] [CrossRef]
- Hesthagen, T.; Sevaldrud, I.H.; Berger, H.M. Assessment of Damage to Fish Populations in Norwegian Lakes Due to Acidification. Ambio 1999, 28, 112–117. [Google Scholar]
- Knutsson, G. Acidification Effects on Groundwater—Prognosis of the Risks for the Future. In Future Groundwater Resources at Risk (Proceedings of the Helsinki Conference, June 1994); IAHS Press: Wallingford, UK, 1994. [Google Scholar]
- Fox, D.L. Prediction of Acid Rock Drainage (ARD) from Sulphidic Slates Using GIS Analysis of Rnineralogical, Geochemical, Magnetic and Geological Parameters: A Test Case in Southern Nova Scotia. Ph.D. Thesis, Dalhousie University, Halifax, Nova Scotia, 1999. [Google Scholar]
- Keefe, M.; Turnbull, L.; White, C.E. Acid Rock Drainage and Manganese in Rock Units Surrounding the Town of Bridgewater’s Water Supply, Lunenburg County, Nova Scotia. In Geoscience and Mines Branch, Report of Activities 2017–2018; Report ME 2018-001; Nova Scotia Department of Energy and Mines: Halifax, NS, Canada, 2018; pp. 23–31. [Google Scholar]
- Appleyard, S.; Cook, T. Reassessing the Management of Groundwater Use from Sandy Aquifers: Acidification and Base Cation Depletion Exacerbated by Drought and Groundwater Withdrawal on the Gnangara Mound, Western Australia. Hydrogeol. J. 2009, 17, 579–588. [Google Scholar] [CrossRef]
- Mosley, L.M.; Palmer, D.; Leyden, E.; Cook, F.; Zammit, B.; Shand, P.; Baker, A.W.; Fitzpatrick, R. Acidification of Floodplains Due to River Level Decline during Drought. J. Contam. Hydrol. 2014, 161, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, G.; Drage, J.; Check, G. Development of Indices to Assess the Potential Impact of Drought to Private Wells in Nova Scotia. In Proceedings of the GeoOttawa 2017, Ottawa, ON, Canada, 30 September–2 October 2017. 8p. [Google Scholar]
- Perrone, D.; Jasechko, S. Dry Groundwater Wells in the Western United States. Environ. Res. Lett. 2017, 12, 104002. [Google Scholar] [CrossRef]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water; U.S. Geological Survey Water-Supply Paper 2254; Department of the Interior: Alexandria, VA, USA, 1985. [Google Scholar]
- Smeltzer, D. Loon Lake Andalusite Project, Guysborough County, Nova Scotia, Till Sample Program and Outcrop Analyses; Nova Scotia Department of Natural Resources: Halifax, NS, Canada, 1996; 16p. [Google Scholar]
- MacDonald, M.A.; Horne, R.J.; Corey, M.C.; Ham, L.J. An Overview of Recent Bedrock Mapping and Follow-up Petrological Studies of the South Mountain Batholith, Southwestern Nova Scotia, Canada. Atl. Geol. 1992, 28, 7–28. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Schecher, W.D. The Chemistry of Aluminum in the Environment. Environ. Geochem. Health 1990, 12, 28–49. [Google Scholar] [CrossRef]
- Exley, C.; Chappell, J.S.; Birchall, J.D. A Mechanism for Acute Aluminium Toxicity in Fish. J. Theor. Biol. 1991, 151, 417–428. [Google Scholar] [CrossRef]
- Nilsen, T.O.; Ebbesson, L.O.; Kverneland, O.G.; Kroglund, F.; Finstad, B.; Stefansson, S.O. Effects of Acidic Water and Aluminum Exposure on Gill Na, K -ATPase α-Subunit Isoforms, Enzyme Activity, Physiology and Return Rates in Atlantic Salmon (Salmo Salar L.). Aquat. Toxicol. 2010, 97, 250–259. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. 2018 Edition of the Drinking Water Standards and Health Advisories Tables; U.S. Environmental Protection Agency: Washington, DC, USA, 2018.
- Lacoul, P.; Freedman, B.; Clair, T. Effects of Acidification on Aquatic Biota in Atlantic Canada. Environ. Rev. 2011, 19, 429–460. [Google Scholar] [CrossRef]
- Flaten, T.P. Aluminium as a Risk Factor in Alzheimer’s Disease, with Emphasis on Drinking Water. Brain Res. Bull. 2001, 55, 187–196. [Google Scholar] [CrossRef]
- Martyn, C.N.; Osmond, C.; Edwardson, J.A.; Barker, D.; Harris, E.C.; Lacey, R.F. Geographical Relation between Alzheimer’s Disease and Aluminium in Drinking Water. Lancet 1989, 333, 61–62. [Google Scholar] [CrossRef]
- Wills, M.R.; Savory, J. Water Content of Aluminum, Dialysis Dementia, and Osteomalacia. Environ. Health Perspect. 1985, 63, 141–147. [Google Scholar] [CrossRef]
- Health Canada. Guidelines for Canadian Drinking Water Quality: Guideline Technical Document—Aluminum; Health Canada: Ottawa, ON, Canada, 2021; 72p, ISBN 978-0-660-37115-3. [Google Scholar]
- World Health Organization. Aluminium in Drinking-Water; Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO Press: Geneva, Switzerland, 2010. [Google Scholar]
- Poirier, J.; Semple, H.; Davies, J.; Lapointe, R.; Dziwenka, M.; Hiltz, M.; Mujibi, D. Double-Blind, Vehicle-Controlled Randomized Twelve-Month Neurodevelopmental Toxicity Study of Common Aluminum Salts in the Rat. Neuroscience 2011, 193, 338–362. [Google Scholar] [CrossRef]
- Panhwar, A.H.; Kazi, T.G.; Naeemullah; Afridi, H.I.; Shah, F.; Arain, M.B.; Arain, S.A. Evaluated the Adverse Effects of Cadmium and Aluminum via Drinking Water to Kidney Disease Patients: Application of a Novel Solid Phase Microextraction Method. Environ. Toxicol. Pharmacol. 2016, 43, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.M.; Oli, H.; Walker, G.S.; Lewins, A.M. Water Supply aluminium concentration, dialysis dementia, and effect of reverse-osmosis water treatment. Lancet 1982, 320, 785–787. [Google Scholar] [CrossRef]
- Goodsell, T.H.; Carling, G.T.; Aanderud, Z.T.; Nelson, S.T.; Fernandez, D.P.; Tingey, D.G. Thermal Groundwater Contributions of Arsenic and Other Trace Elements to the Middle Provo River, Utah, USA. Environ. Earth Sci. 2017, 76, 268. [Google Scholar] [CrossRef]
- Hagerthey, S.E.; Kerfoot, W.C. Groundwater Flow Influences the Biomass and Nutrient Ratios of Epibenthic Algae in a North Temperate Seepage Lake. Limnol. Oceanogr. 1998, 43, 1227–1242. [Google Scholar] [CrossRef]
- Hoehn, E. Solute Exchange between River Water and Groundwater in Headwater Environments. Hydrol. Water Resour. Ecol. Headwaters 1998, 7, 165–172. [Google Scholar]
- Shaw, G.D.; White, E.S.; Gammons, C.H. Characterizing Groundwater–Lake Interactions and Its Impact on Lake Water Quality. J. Hydrol. 2013, 492, 69–78. [Google Scholar] [CrossRef]
- Hansen, B.K.; Postma, D. Acidification, Buffering, and Salt Effects in the Unsaturated Zone of a Sandy Aquifer, Klosterhede, Denmark. Water Resour. Res. 1995, 31, 2795–2809. [Google Scholar] [CrossRef]
- Kennedy, G. Potential Corrosivity of Groundwater in Nova Scotia and Its Association with Lead in Private Well Water; Open File Report ME 2019-002; Nova Scotia Department of Energy and Mines: Halifax, Nova Scotia, 2019; 22p. [Google Scholar]
- Bondu, R.; Cloutier, V.; Rosa, E.; Roy, M. An Exploratory Data Analysis Approach for Assessing the Sources and Distribution of Naturally Occurring Contaminants (F, Ba, Mn, As) in Groundwater from Southern Quebec (Canada). Appl. Geochem. 2020, 114, 104500. [Google Scholar] [CrossRef]
- Government of New Brunswick. New Brunswick Groundwater Chemistry Atlas: 1994-2007; Environmental Reporting Series; New Brunswick Department of Environment: Fredericton, NB, Canada, 2008. [Google Scholar]
- de Meyer, C.M.C.; Rodríguez, J.M.; Carpio, E.A.; García, P.A.; Stengel, C.; Berg, M. Arsenic, Manganese and Aluminum Contamination in Groundwater Resources of Western Amazonia (Peru). Sci. Total Environ. 2017, 607–608, 1437–1450. [Google Scholar] [CrossRef]
- Briggs, M.A.; Lane, J.W.; Snyder, C.D.; White, E.A.; Johnson, Z.C.; Nelms, D.L.; Hitt, N.P. Shallow Bedrock Limits Groundwater Seepage-Based Headwater Climate Refugia. Limnologica 2018, 68, 142–156. [Google Scholar] [CrossRef]
- Ebersole, J.L.; Liss, W.J.; Frissell, C.A. Thermal Heterogeneity, Stream Channel Morphology, and Salmonid Abundance in Northeastern Oregon Streams. Can. J. Fish. Aquat. Sci. 2003, 60, 1266–1280. [Google Scholar] [CrossRef]
- Kurylyk, B.L.; MacQuarrie, K.T.B.; Linnansaari, T.; Cunjak, R.A.; Curry, R.A. Preserving, Augmenting, and Creating Cold-Water Thermal Refugia in Rivers: Concepts Derived from Research on the Miramichi River, New Brunswick (Canada). Ecohydrology 2015, 8, 1095–1108. [Google Scholar] [CrossRef]
- Berman, C.H.; Quinn, T.P. Behavioural Thermoregulation and Homing by Spring Chinook Salmon, Oncorhynchus Tshawytscha (Walbaum), in the Yakima River. J. Fish. Biol. 1991, 39, 301–312. [Google Scholar] [CrossRef]
- Sutton, R.J.; Deas, M.L.; Tanaka, S.K.; Soto, T.; Corum, R.A. Salmonid Observations at a Klamath River Thermal Refuge under Various Hydrological and Meteorological Conditions. River Res. Appl. 2007, 23, 775–785. [Google Scholar] [CrossRef]
- de Graaf, I.E.M.; Gleeson, T.; (Rens) van Beek, L.P.H.; Sutanudjaja, E.H.; Bierkens, M.F.P. Environmental Flow Limits to Global Groundwater Pumping. Nature 2019, 574, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Howells, G.; Dalziel, T.; Reader, J.P.; Solbe, J.F. EIFAC Water Quality Criteria for European Freshwater Fish: Report on Aluminium. Chem. Ecol. 1990, 4, 117–173. [Google Scholar] [CrossRef]
- Nova Scotia Department of Energy and Mines Nova Scotia Groundwater Atlas Well Water Chemistry. 2021. Available online: https://fletcher.novascotia.ca/DNRViewer/?viewer=Groundwater (accessed on 27 May 2021).
- Nova Scotia Environment and Climate Change Nova Scotia Well Logs Database. 2020. Available online: https://novascotia.ca/natr/meb/geoscience-online/groundwater_about.asp (accessed on 1 June 2021).
- Kennedy, G.W.; Drage, J. Groundwater Regions Map of Nova Scotia; Nova Scotia Department of Natural Resources: Halifax, NS, Canada, 2008. [Google Scholar]
- Keppie, J.D. Geological Map of the Province of Nova Scotia; Nova Scotia Department of Natural Resource: Halifax, NS, Canada, 2000. [Google Scholar]
- ESRI. ArcGIS Desktop: Release 10; Environmental Systems Research Institute: Redlands, CA, USA, 2011. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 1 June 2021).
- Kennedy, G.; Polegato, A. Where Does Our Tap Water Come From? An Analysis of Domestic Water Source and Supply Demographics in Nova Scotia; Open File Report ME 2016-006; Nova Scotia Department of Natural Resources: Halifax, NS, Canada, 2017; 18p. [Google Scholar]
- Utting, D.; Goodwin, T. Lake-Sediment Geochemistry and Its Influence on Lake Alkalinity, Southwest Nova Scotia. In Mineral Resources Branch, Report of Activities 2007; Report ME 2008-1; Nova Scotia Department of Natural Resources: Halifax, NS, Canada, 2008; pp. 105–111. [Google Scholar]
- Dyck, W.; Chatterjee, A.K.; Gemmell, D.E.; Murricane, K. Well Water Trace Element Reconnaissance, Eastern Maritime Canada. J. Geochem. Explor. 1976, 6, 139–162. [Google Scholar] [CrossRef]
- Sterling, S.M.; MacLeod, S.; Rotteveel, L.; Hart, K.; Clair, T.A.; Halfyard, E.A.; O’Brien, N.L. Ionic Aluminium Concentrations Exceed Thresholds for Aquatic Health in Nova Scotian Rivers, Even during Conditions of High Dissolved Organic Carbon and Low Flow. Hydrol. Earth Syst. Sci. 2020, 24, 4763–4775. [Google Scholar] [CrossRef]
- Nova Scotia Department of Energy and Mines, Relative Risk of Corrosive Groundwater in Drilled Water Wells. 2021. Available online: https://fletcher.novascotia.ca/DNRViewer/index.html?viewer=Drilled_Corrosive_Groundwater_NS.Relative_Corrosivity_of_Groundwater_in_Drilled_Water_Wells (accessed on 1 June 2021).
- Rotteveel, L.; Sterling, S.M. Five Aluminum Seasonality Regimes Identified in Chronically Acidified Rivers of Nova Scotia. Environ. Sci. Technol. 2020, 54, 807–817. [Google Scholar] [CrossRef] [PubMed]
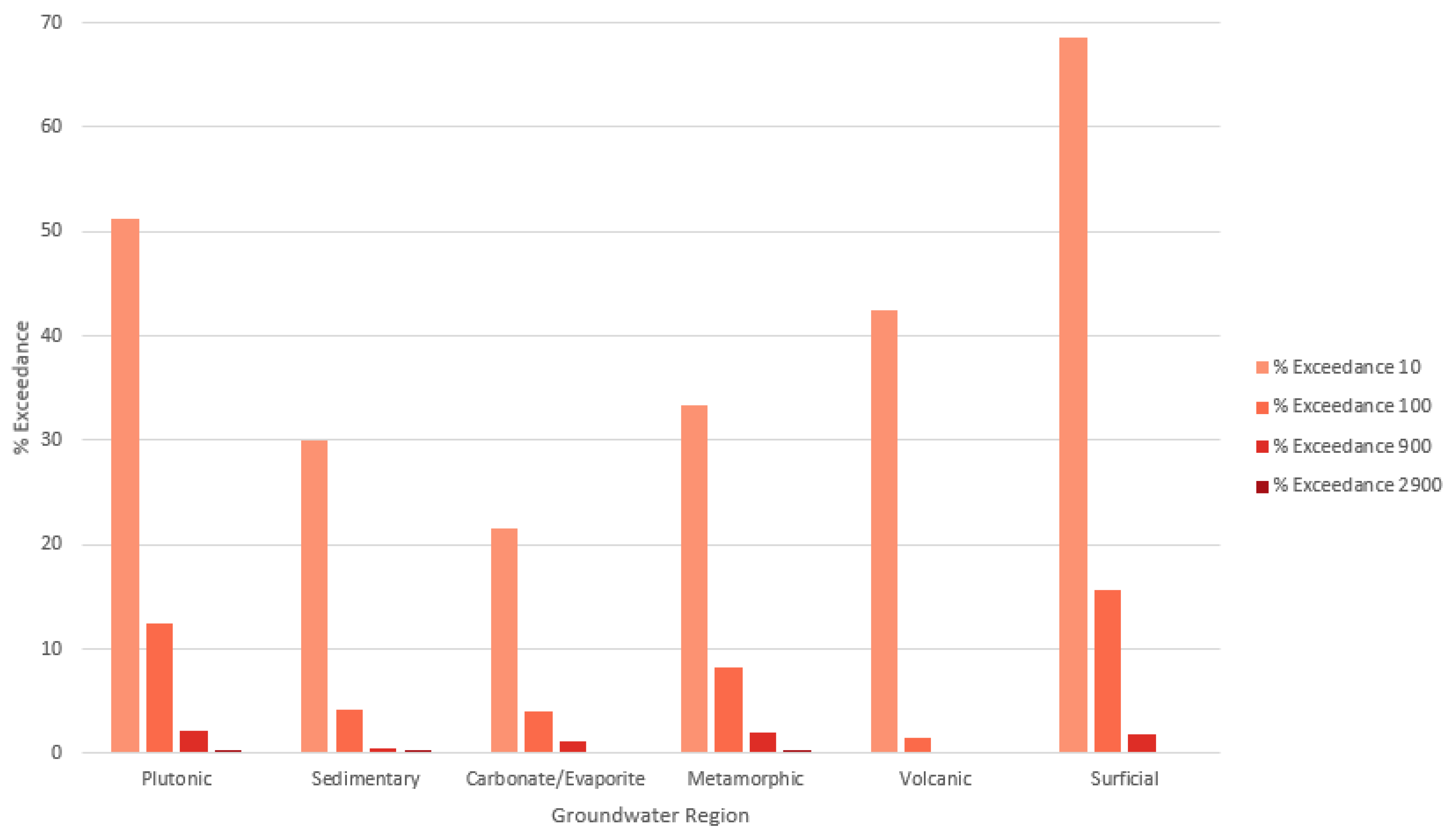
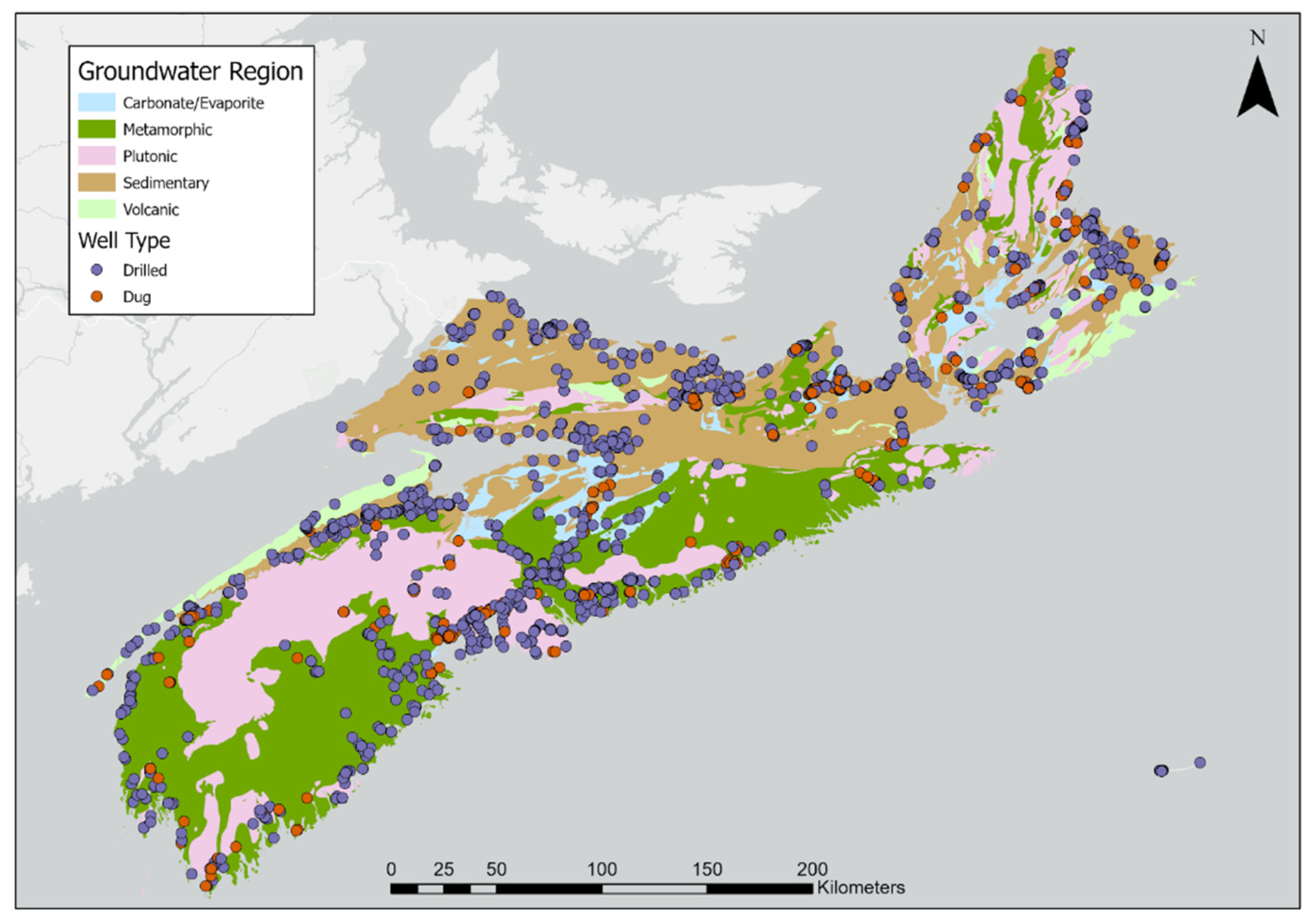
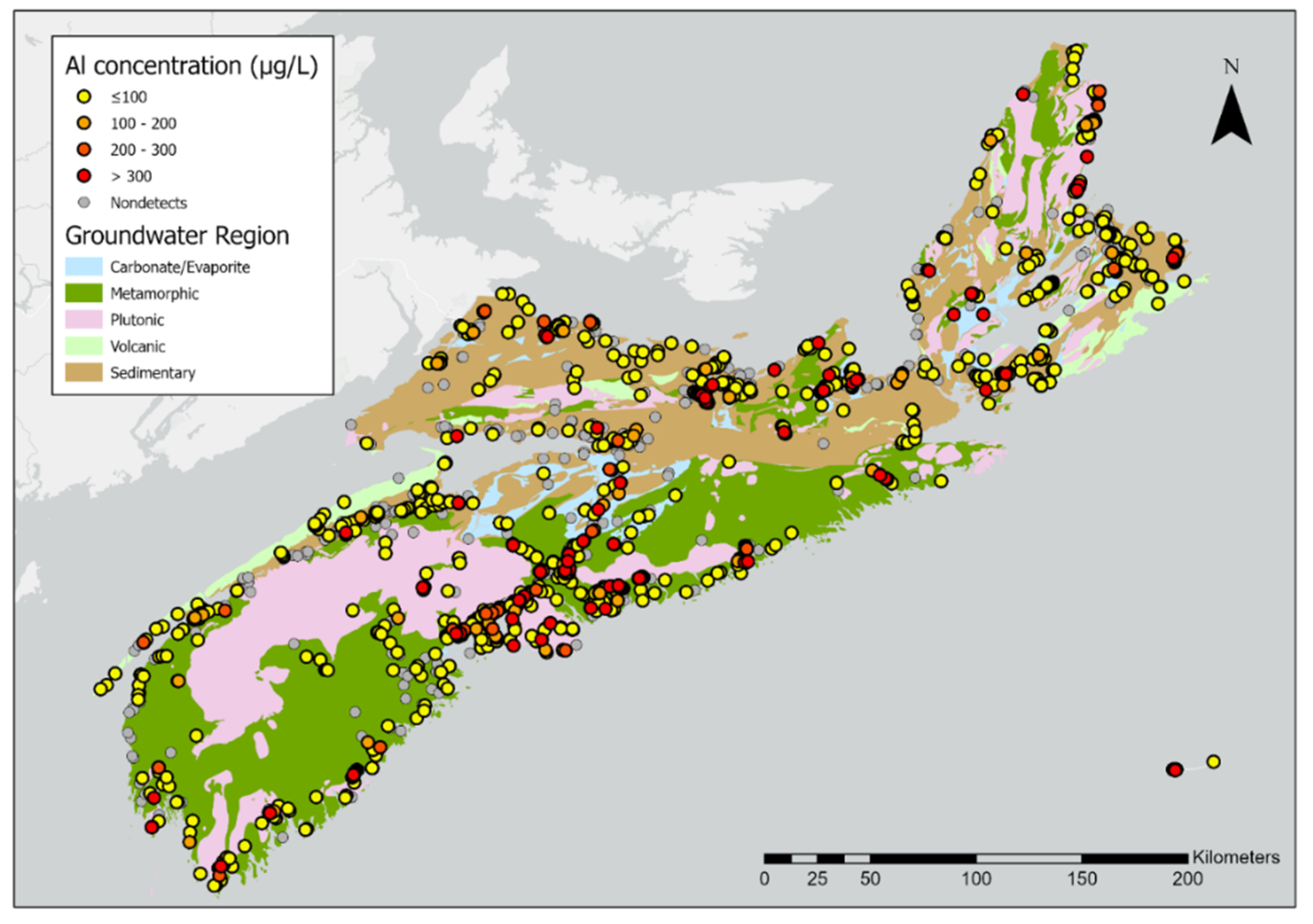
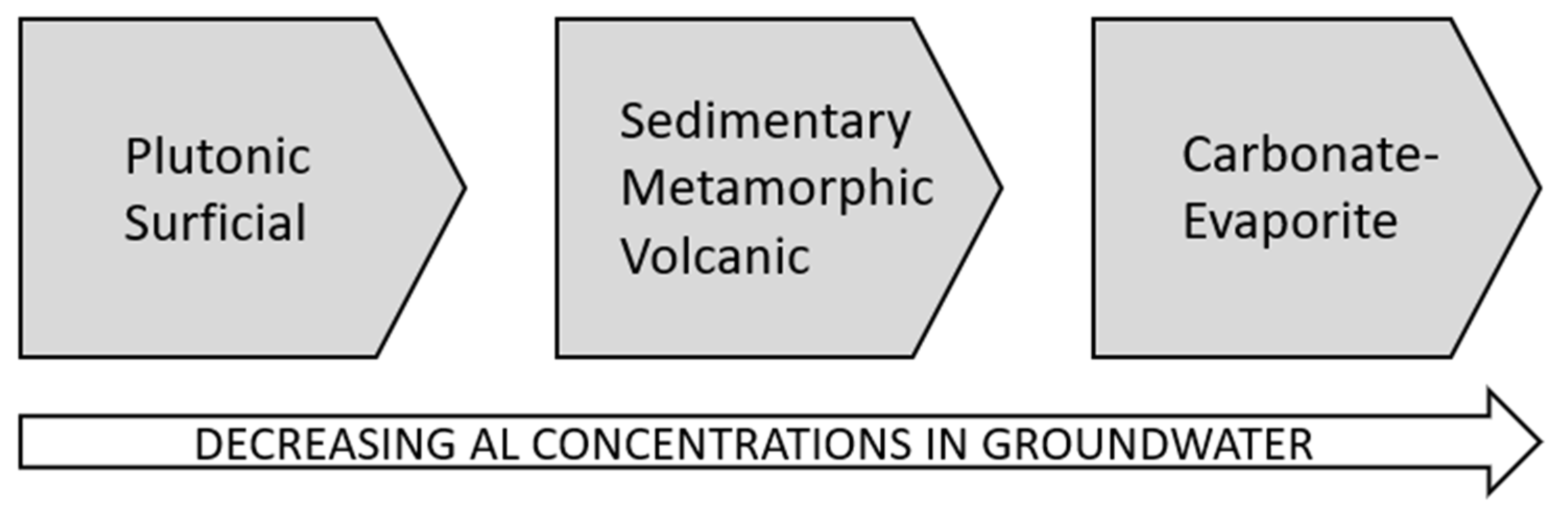
| (a) | |||||||
| Groundwater Region | Count Observations | Count Detects 1 | % Detects | % Exceed 10 | % Exceed 100 | % Exceed 900 | % Exceed 2900 |
| Plutonic | 320 | 230 | 71.9% | 51.2% | 12.5% | 2.19% | 0.31% |
| Sedimentary | 954 | 439 | 46.0% | 30.0% | 4.19% | 0.52% | 0.31% |
| Carbonate/Evaporite | 176 | 57 | 32.4% | 21.6% | 3.98% | 1.14% | 0.00% |
| Metamorphic | 458 | 250 | 54.6% | 33.4% | 8.30% | 1.96% | 0.22% |
| Volcanic | 66 | 41 | 62.1% | 42.4% | 1.51% | 0.00% | 0.00% |
| Surficial | 544 | 435 | 80.0% | 68.6% | 15.6% | 1.84% | 0.18% |
| TOTAL | 2518 | 1452 | 57.7% | 41.2% | 8.34% | 1.31% | 0.24% |
| (b) | |||||||
| Groundwater Region | K—M 2 Mean | Min | Median | 95th Percentile | Max | Median Ca (mg/L) | Median pH |
| Plutonic | 92.66 | 1.0 | 10.0 | 370 | 5270 | 17.3 | 7.32 |
| Sedimentary | 36.56 | 1.0 | <10 | 75 | 4700 | 37.0 | 7.9 |
| Carbonate/Evaporite | 30.98 | 1.0 | <10 | 72 | 1300 | 66.9 | 8.00 |
| Metamorphic | 73.28 | 0.25 | <10 | 347 | 3060 | 24.1 | 7.68 |
| Volcanic | 18.08 | 1.0 | <10 | 60 | 220 | 25.9 | 7.77 |
| Surficial | 90.58 | 1.0 | 19.0 | 330 | 3400 | 18 | 6.92 |
| Carbonate/Evaporite | Metamorphic | Plutonic | Sedimentary | Surficial | |
|---|---|---|---|---|---|
| Metamorphic | <0.0001 | - | - | - | - |
| Plutonic | <0.0001 | 0.0009 | - | - | - |
| Sedimentary | <0.0001 | 0.3480 | <0.0001 | - | - |
| Surficial | <0.0001 | <0.0001 | <0.0001 | <0.0001 | - |
| Volcanic | <0.0001 | 0.3170 | 0.0490 | 0.0690 | <0.0001 |
| Parameter | Slope | p-Value |
|---|---|---|
| pH | −1.059 | 2.15 × 10−49 |
| TOC | 0.199 | 2.19 × 10−20 |
| Ca | −0.0059 | 4.50 × 10−12 |
| Turbidity | 0.00515 | 3.01 × 10−8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hart, K.A.; Kennedy, G.W.; Sterling, S.M. Distribution, Drivers, and Threats of Aluminum in Groundwater in Nova Scotia, Canada. Water 2021, 13, 1578. https://doi.org/10.3390/w13111578
Hart KA, Kennedy GW, Sterling SM. Distribution, Drivers, and Threats of Aluminum in Groundwater in Nova Scotia, Canada. Water. 2021; 13(11):1578. https://doi.org/10.3390/w13111578
Chicago/Turabian StyleHart, Kristin A., Gavin W. Kennedy, and Shannon M. Sterling. 2021. "Distribution, Drivers, and Threats of Aluminum in Groundwater in Nova Scotia, Canada" Water 13, no. 11: 1578. https://doi.org/10.3390/w13111578
APA StyleHart, K. A., Kennedy, G. W., & Sterling, S. M. (2021). Distribution, Drivers, and Threats of Aluminum in Groundwater in Nova Scotia, Canada. Water, 13(11), 1578. https://doi.org/10.3390/w13111578






