Microalgal-Bacterial Granular Sludge Process in Non-Aerated Municipal Wastewater Treatment under Natural Day-Night Conditions: Performance and Microbial Community
Abstract
1. Introduction
2. Materials and Methods
2.1. MBGS and Simulated Wastewater
2.2. Experimental Setup
2.3. Analytical Methods
3. Results
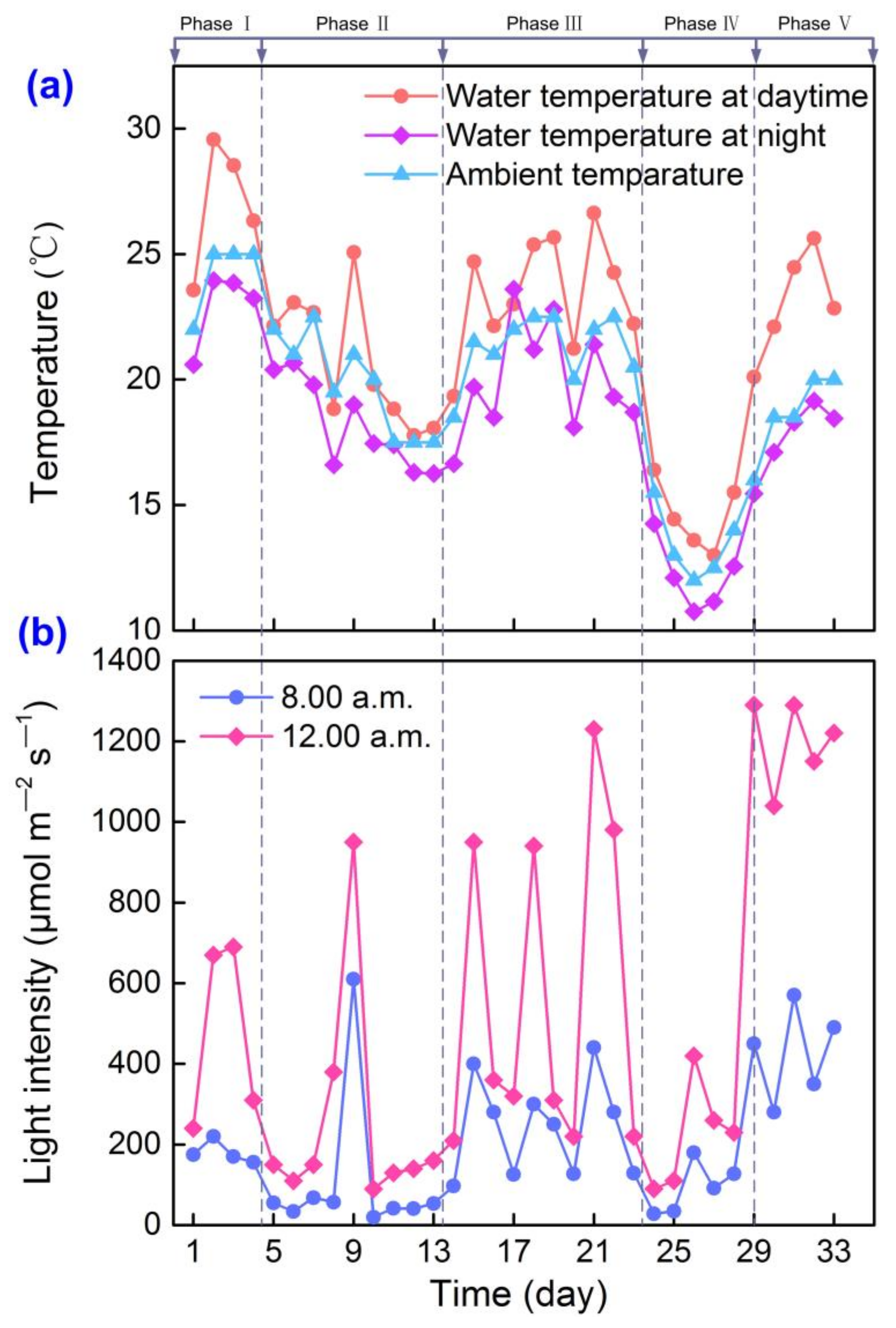
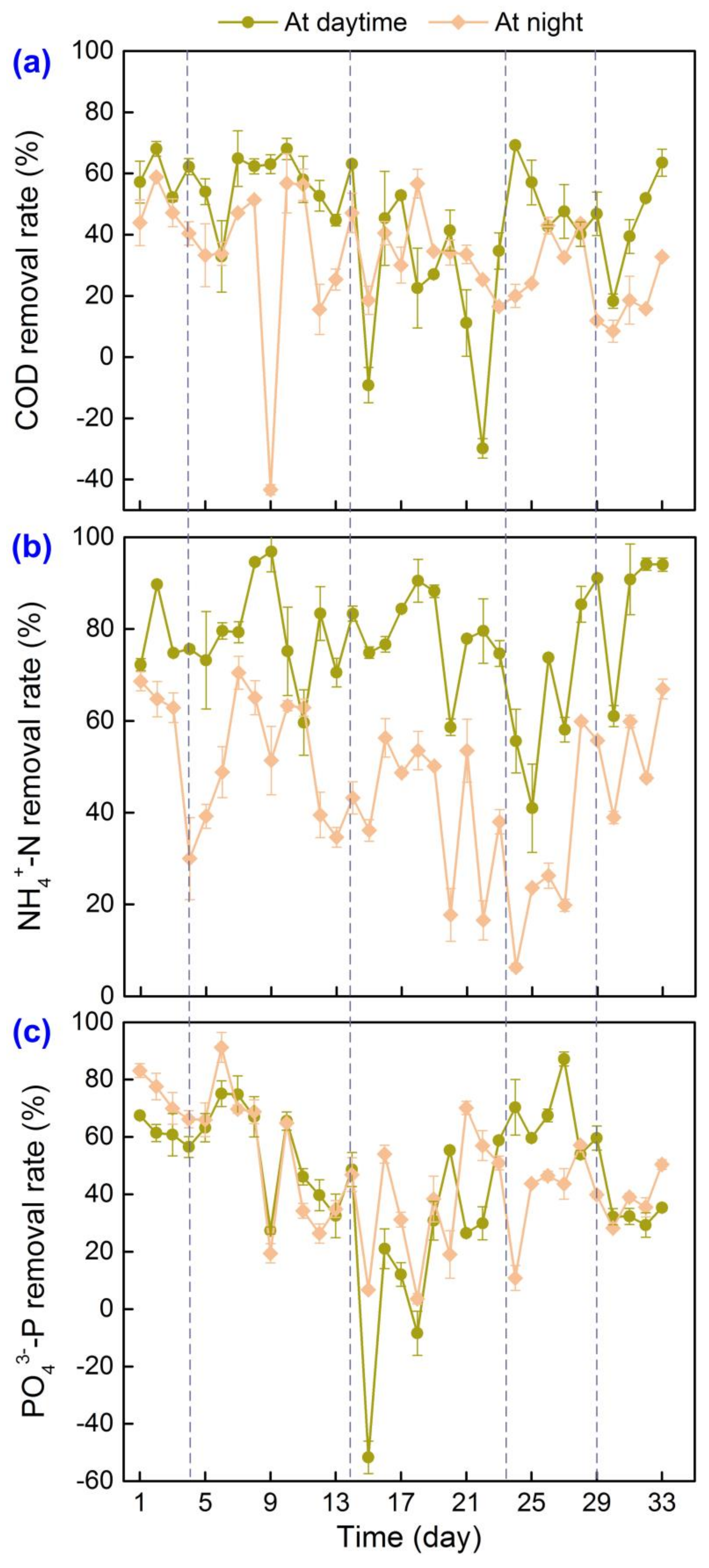
3.1. Performance of MBGS Process
3.2. Basic Parameters of MBGS
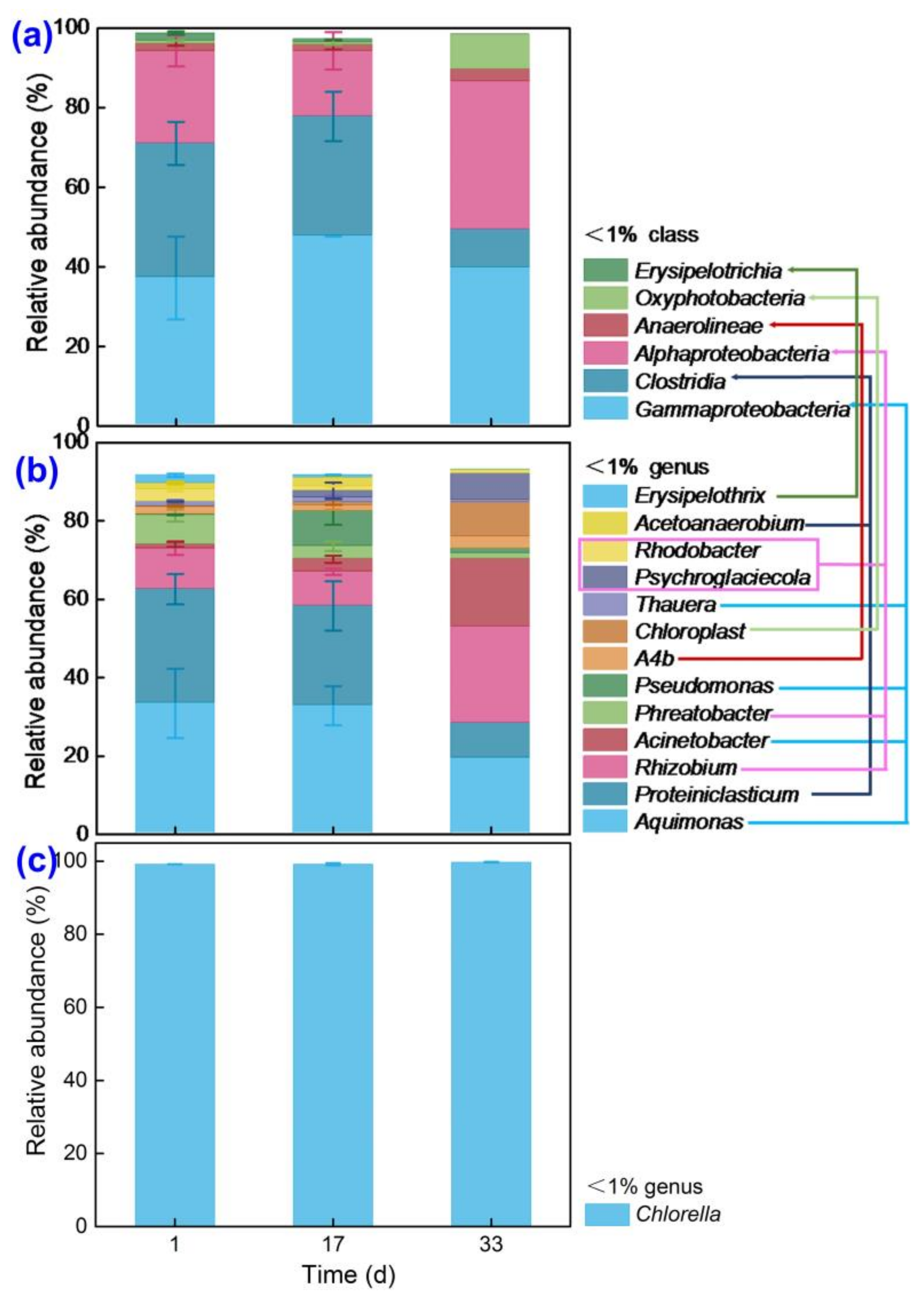
3.3. The Community Structure of MBGS
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, X.; Zhang, M.; Liu, H.; Gu, J.; Liu, Y. Environmental sustainability: A pressing challenge to biological sewage treatment processes. Curr. Opin. Environ. Sci. Health 2019, 12, 1–5. [Google Scholar] [CrossRef]
- Liu, Y.; Gu, J.; Zhang, M. AB Processes: Towards Energy Self-Sufficient Municipal Wastewater Treatment; IWA Publishing: London, UK, 2019. [Google Scholar]
- Ji, B.; Zhu, L.; Wang, S.; Qin, H.; Ma, Y.; Liu, Y. A novel micro-ferrous dosing strategy for enhancing biological phosphorus removal from municipal wastewater. Sci. Total Environ. 2020, 704, 135453. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, X.; Li, X.; Jia, S.; Peng, Y. Advanced nutrient removal from ammonia and domestic wastewaters by a novel process based on simultaneous partial nitrification-anammox and modified denitrifying phosphorus removal. Chem. Eng. J. 2018, 354, 589–598. [Google Scholar] [CrossRef]
- Pang, H.; Li, L.; He, J.; Yan, Z.; Ma, Y.; Nan, J.; Liu, Y. New insight into enhanced production of short-chain fatty acids from waste activated sludge by cation exchange resin-induced hydrolysis. Chem. Eng. J. 2020, 388, 124235. [Google Scholar] [CrossRef]
- Zhang, M.; Ji, B.; Liu, Y. Microalgal-bacterial granular sludge process: A game changer of future municipal wastewater treatment? Sci. Total Environ. 2021, 752, 141957. [Google Scholar] [CrossRef]
- Wang, J.; Lei, Z.; Wei, Y.; Wang, Q.; Tian, C.; Shimizu, K.; Zhang, Z.; Adachi, Y.; Lee, D. Behavior of algal-bacterial granular sludge in a novel closed photo-sequencing batch reactor under no external O2 supply. Bioresour. Technol. 2020, 318, 124190. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.L.; Pires, J.C.M.; Simões, M. A review on the use of microalgal consortia for wastewater treatment. Algal Res. 2017, 24, 403–415. [Google Scholar] [CrossRef]
- Tiron, O.; Bumbac, C.; Patroescu, I.V.; Badescu, V.R.; Postolache, C. Granular activated algae for wastewater treatment. Water Sci. Technol. 2015, 71, 832–839. [Google Scholar] [CrossRef]
- Ji, B.; Zhu, L.; Wang, S.; Liu, Y. Temperature-effect on the performance of non-aerated microalgal-bacterial granular sludge process in municipal wastewater treatment. J. Environ. Manag. 2021, 282, 111955. [Google Scholar] [CrossRef]
- Liu, L.; Zeng, Z.; Bee, M.; Gibson, V.; Wei, L.; Huang, X.; Liu, C. Characteristics and performance of aerobic algae-bacteria granular consortia in a photo-sequencing batch reactor. J. Hazard. Mater. 2018, 349, 135–142. [Google Scholar] [CrossRef]
- Ji, B.; Zhang, M.; Gu, J.; Ma, Y.; Liu, Y. A self-sustaining synergetic microalgal-bacterial granular sludge process towards energy-efficient and environmentally sustainable municipal wastewater treatment. Water Res. 2020, 179, 115884. [Google Scholar] [CrossRef]
- Wang, S.; Ji, B.; Cui, B.; Ma, Y.; Guo, D.; Liu, Y. Cadmium-effect on performance and symbiotic relationship of microalgal-bacterial granules. J. Clean. Prod. 2021, 282, 125383. [Google Scholar] [CrossRef]
- Wang, S.; Ji, B.; Zhang, M.; Ma, Y.; Gu, J.; Liu, Y. Defensive responses of microalgal-bacterial granules to tetracycline in municipal wastewater treatment. Bioresour. Technol. 2020, 312, 123605. [Google Scholar] [CrossRef] [PubMed]
- Abouhend, A.S.; McNair, A.; Kuo-Dahab, W.C.; Watt, C.; Butler, C.S.; Milferstedt, K.; Hamelin, J.; Seo, J.; Gikonyo, G.J.; El-Moselhy, K.M.; et al. The oxygenic photogranule process for aeration-free wastewater treatment. Environ. Sci. Technol. 2018, 52, 3503–3511. [Google Scholar] [CrossRef]
- Abouhend, A.S.; Milferstedt, K.; Hamelin, J.; Ansari, A.A.; Butler, C.; Carbajal-González, B.I.; Park, C. Growth progression of oxygenic photogranules and its impact on bioactivity for aeration-free wastewater treatment. Environ. Sci. Technol. 2019, 54, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Zhang, X.; Shi, Y.; Cui, B.; Fan, J.; Ji, B.; Yuan, J. Microalgal-bacterial granular sludge process outperformed aerobic granular sludge process in municipal wastewater treatment with less carbon dioxide emissions. Environ. Sci. Pollut. Res. 2021, 28, 13616–13623. [Google Scholar] [CrossRef]
- APH Association. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Lee, C.; Lee, S.; Ko, S.; Oh, H.; Ahn, C. Effects of photoperiod on nutrient removal, biomass production, and algal-bacterial population dynamics in lab-scale photobioreactors treating municipal wastewater. Water Res. 2015, 68, 680–691. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Pan, A.; Li, Y.; Xiao, Y.; Zhou, Y.; Chen, C.; Sun, F.; Su, X. A novel strategy for enhancing anaerobic biodegradation of an anthraquinone dye reactive blue 19 with resuscitation-promoting factors. Chemosphere 2021, 263, 127922. [Google Scholar] [CrossRef]
- Zhang, B.; Lens, P.N.L.; Shi, W.; Zhang, R.; Zhang, Z.; Guo, Y.; Bao, X.; Cui, F. Enhancement of aerobic granulation and nutrient removal by an algal–bacterial consortium in a lab-scale photobioreactor. Chem. Eng. J. 2018, 334, 2373–2382. [Google Scholar] [CrossRef]
- Ji, B.; Wang, S.; Guo, D.; Pang, H. Comparative and comprehensive analysis on bacterial communities of two full-scale wastewater treatment plants by second and third-generation sequencing. Bioresour. Technol. Rep. 2020, 11, 100450. [Google Scholar] [CrossRef]
- Jia, H.; Yuan, Q. Nitrogen removal in photo sequence batch reactor using algae-bacteria consortium. J. Water Process Eng. 2018, 26, 108–115. [Google Scholar] [CrossRef]
- Choi, O.; Das, A.; Yu, C.; Hu, Z. Nitrifying bacterial growth inhibition in the presence of algae and cyanobacteria. Biotechnol. Bioeng. 2010, 107, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Li, B.; Zhang, C.; Zhang, Z.; Lei, Z.; Lu, B.; Zhou, B. Effect of algae growth on aerobic granulation and nutrients removal from synthetic wastewater by using sequencing batch reactors. Bioresour. Technol. 2015, 179, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhou, Y.; Xiao, W.; Ji, L.; Cao, X.; Song, C. Shifting nutrient-mediated interactions between algae and bacteria in a microcosm: Evidence from alkaline phosphatase assay. Microbiol. Res. 2012, 167, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ji, B.; Zhang, M.; Gu, J.; Ma, Y.; Liu, Y. Tetracycline-induced decoupling of symbiosis in microalgal-bacterial granular sludge. Environ. Res. 2021, 197, 111095. [Google Scholar] [CrossRef]
- Qiao, S.; Hou, C.; Wang, X.; Zhou, J. Minimizing greenhouse gas emission from wastewater treatment process by integrating activated sludge and microalgae processes. Sci. Total Environ. 2020, 732, 139032. [Google Scholar] [CrossRef]
- Ji, B.; Zhang, M.; Wang, L.; Wang, S.; Liu, Y. Removal mechanisms of phosphorus in non-aerated microalgal-bacterial granular sludge process. Bioresour. Technol. 2020, 312, 123531. [Google Scholar] [CrossRef]
- Feng, T.; Wu, J.; Chai, K.; Liu, F. Effect of Pseudomonas sp. on the degradation of aluminum/epoxy coating in seawater. J. Mol. Liq. 2018, 263, 248–254. [Google Scholar] [CrossRef]
- Han, W.; Li, T.; Cheng, L.; Liu, L.; Yu, L.; Peng, Z. Effect of adding microorganism and carbon source to substrate on nitrogen removal treating the drainage of WWTP. Water Sci. Technol. 2019, 79, 1947–1955. [Google Scholar]
- Yang, C.; Wang, Q.; Simon, P.N.; Liu, J.; Liu, L.; Dai, X.; Zhang, X.; Kuang, X.; Igarashi, Y.; Pan, X.; et al. Distinct network interactions in particle-associated and free-living bacterial communities during a Microcystis aeruginosa bloom in a plateau lake. Front. Microbiol. 2017, 8, 1202. [Google Scholar] [CrossRef]
- Ji, B.; Yang, K.; Zhu, L.; Jiang, Y.; Wang, H.; Zhou, J.; Zhang, H. Aerobic denitrification: A review of important advances of the last 30 years. Biotechnol. Bioprocess Eng. 2015, 20, 643–651. [Google Scholar] [CrossRef]
- Zhao, Y.; Jiang, B.; Tang, X.; Liu, S. Metagenomic insights into functional traits variation and coupling effects on the anammox community during reactor start-up. Sci. Total Environ. 2019, 687, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Ni, B.; Zeng, R.; Fang, F.; Xie, W.; Xu, J.; Sheng, G.; Sun, Y.; Yu, H. Evaluation on factors influencing the heterotrophic growth on the soluble microbial products of autotrophs. Biotechnol. Bioeng. 2011, 108, 804–812. [Google Scholar] [CrossRef]

| Time (day) | COD (mg/L) | NH4+-N (mg/L) | PO43−-P (mg/L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Daytime | Night | Daytime | Night | Daytime | Night | |||||||
| Influent | Effluent | Influent | Effluent | Influent | Effluent | Influent | Effluent | Influent | Effluent | Influent | Effluent | |
| 1 | 415 ± 14 | 178 ± 34 | 477 ± 12 | 268 ± 29 | 40.1 ± 0.5 | 11.1 ± 0.7 | 35.1 ± 0.1 | 11.0 ± 0.7 | 6.83 ± 0.17 | 2.22 ± 0.02 | 6.15 ± 0.12 | 1.04 ± 0.17 |
| 2 | 393 ± 6 | 126 ± 11 | 510 ± 70 | 210 ± 29 | 36.9 ± 0.1 | 3.8 ± 0.1 | 63.3 ± 6.2 | 22.3 ± 10.7 | 5.45 ± 0.34 | 2.11 ± 0.03 | 6.26 ± 0.27 | 1.41 ± 0.23 |
| 3 | 352 ± 40 | 168 ± 14 | 525 ± 67 | 276 ± 12 | 36.3 ± 1.9 | 9.2 ± 0.5 | 37.2 ± 3.1 | 13.8 ± 0.1 | 6.13 ± 0.05 | 2.41 ± 0.47 | 8.37 ± 0.63 | 2.52 ± 0.27 |
| 4 | 490 ± 57 | 184 ± 9 | 442 ± 9 | 263 ± 12 | 45.7 ± 2.5 | 11.1 ± 0.9 | 35.0 ± 1.7 | 24.5 ± 1.9 | 5.77 ± 0.64 | 2.51 ± 0.48 | 4.09 ± 0.19 | 1.38 ± 0.18 |
| 5 | 417 ± 63 | 190 ± 11 | 303 ± 3 | 202 ± 29 | 26.6 ± 10.6 | 7.1 ± 0.2 | 25.8 ± 1.5 | 15.7 ± 1.6 | 3.56 ± 0.72 | 1.31 ± 0.44 | 2.23 ± 0.08 | 0.76 ± 0.10 |
| 6 | 302 ± 37 | 205 ± 60 | 455 ± 26 | 300 ± 1 | 24.0 ± 0.8 | 4.9 ± 0.3 | 29.4 ± 2.1 | 15.0 ± 0.6 | 2.26 ± 0.86 | 0.56 ± 0.31 | 3.86 ± 0.29 | 0.34 ± 0.23 |
| 7 | 458 ± 29 | 162 ± 52 | 362 ± 6 | 191 ± 3 | 29.7 ± 2.3 | 6.1 ± 1.1 | 23.4 ± 0.2 | 6.9 ± 0.9 | 2.72 ± 0.02 | 0.69 ± 0.17 | 2.01 ± 0.05 | 0.61 ± 0.04 |
| 8 | 369 ± 74 | 140 ± 37 | 385 ± 84 | 187 ± 44 | 28.2 ± 1.9 | 1.5 ± 0.1 | 29.7 ± 0.8 | 10.4 ± 1.4 | 2.71 ± 0.63 | 0.90 ± 0.39 | 7.61 ± 0.12 | 2.37 ± 0.35 |
| 9 | 373 ± 1 | 138 ± 11 | 305 ± 12 | 436 ± 12 | 30.3 ± 0.3 | 0.9 ± 1.3 | 29.5 ± 1.2 | 14.3 ± 2.8 | 6.83 ± 0.50 | 4.96 ± 0.28 | 6.72 ± 0.03 | 5.41 ± 0.25 |
| 10 | 340 ± 34 | 109 ± 23 | 473 ± 31 | 189 ± 87 | 25.1 ± 0.6 | 6.2 ± 2.3 | 30.8 ± 2.0 | 11.3 ± 0.4 | 5.89 ± 0.23 | 2.03 ± 0.27 | 7.96 ± 0.18 | 2.80 ± 0.07 |
| 11 | 450 ± 6 | 188 ± 32 | 342 ± 47 | 150 ± 38 | 27.5 ± 0.4 | 11.1 ± 2.3 | 20.8 ± 0.1 | 7.7 ± 0.4 | 6.74 ± 0.03 | 3.63 ± 0.18 | 5.72 ± 0.36 | 3.76 ± 0.09 |
| 12 | 253 ± 9 | 120 ± 9 | 241 ± 15 | 204 ± 32 | 16.9 ± 0.9 | 2.8 ± 0.8 | 21.5 ± 0.9 | 13.0 ± 0.5 | 5.01 ± 0.23 | 3.02 ± 0.13 | 5.54 ± 0.29 | 4.08 ± 0.02 |
| 13 | 334 ± 37 | 184 ± 14 | 237 ± 20 | 177 ± 23 | 17.2 ± 0.4 | 5.1 ± 0.6 | 30.7 ± 1.7 | 18.7 ± 0.4 | 6.03 ± 0.53 | 4.07 ± 0.10 | 5.22 ± 0.27 | 3.40 ± 0.02 |
| 14 | 642 ± 117 | 237 ± 49 | 469 ± 35 | 247 ± 12 | 40.5 ± 1.4 | 6.8 ± 0.4 | 35.1 ± 1.5 | 19.9 ± 0.4 | 8.20 ± 0.64 | 4.22 ± 0.16 | 6.40 ± 0.28 | 3.40 ± 0.22 |
| 15 | 298 ± 32 | 324 ± 17 | 354 ± 6 | 288 ± 12 | 29.3 ± 0.2 | 7.4 ± 0.3 | 27.3 ± 5.6 | 17.5 ± 2.9 | 4.18 ± 0.13 | 6.33 ± 0.04 | 4.92 ± 0.02 | 4.58 ± 0.05 |
| 16 | 338 ± 20 | 186 ± 63 | 486 ± 41 | 288 ± 6 | 28.9 ± 1.1 | 6.7 ± 0.7 | 27.6 ± 1.1 | 12.1 ± 1.2 | 4.44 ± 0.19 | 3.51 ± 0.16 | 8.02 ± 0.23 | 3.69 ± 0.14 |
| 17 | 472 ± 37 | 223 ± 23 | 268 ± 64 | 185 ± 29 | 26.4 ± 0.2 | 4.1 ± 0.1 | 22.7 ± 3.3 | 11.7 ± 1.9 | 6.72 ± 0.03 | 5.91 ± 0.25 | 6.33 ± 0.58 | 4.36 ± 0.24 |
| 18 | 324 ± 29 | 249 ± 20 | 278 ± 44 | 121 ± 32 | 21.6 ± 0.8 | 2.0 ± 0.9 | 20.6 ± 0.9 | 9.6 ± 0.4 | 6.04 ± 0.27 | 6.55 ± 0.17 | 5.61 ± 0.05 | 5.41 ± 0.15 |
| 19 | 278 ± 37 | 203 ± 29 | 292 ± 29 | 191 ± 20 | 36.1 ± 0.8 | 4.2 ± 0.4 | 25.1 ± 2.2 | 12.5 ± 1.0 | 7.43 ± 0.37 | 5.14 ± 0.24 | 5.11 ± 0.45 | 3.15 ± 0.13 |
| 20 | 393 ± 103 | 227 ± 34 | 259 ± 6 | 171 ± 15 | 26.7 ± 0.7 | 11.0 ± 0.8 | 27.8 ± 1.4 | 15.1 ± 0.6 | 4.64 ± 0.07 | 2.08 ± 0.01 | 2.42 ± 0.35 | 1.96 ± 0.08 |
| 21 | 257 ± 32 | 227 ± 1 | 226 ± 6 | 150 ± 3 | 38.4 ± 0.3 | 8.5 ± 0.2 | 34.4 ± 1.3 | 16.0 ± 3.0 | 5.51 ± 0.02 | 4.05 ± 0.04 | 4.68 ± 0.08 | 1.40 ± 0.13 |
| 22 | 203 ± 29 | 263 ± 31 | 292 ± 41 | 218 ± 29 | 41.3 ± 12.6 | 8.4 ± 0.2 | 23.3 ± 0.4 | 19.4 ± 0.7 | 3.34 ± 0.19 | 2.34 ± 0.30 | 5.22 ± 0.97 | 2.25 ± 0.69 |
| 23 | 340 ± 22 | 221 ± 6 | 286 ± 14 | 239 ± 14 | 40.7 ± 1.3 | 10.3 ± 1.5 | 33.2 ± 0.7 | 20.6 ± 0.4 | 5.98 ± 0.75 | 2.47 ± 0.31 | 5.64 ± 0.19 | 2.77 ± 0.04 |
| 24 | 385 ± 3 | 197 ± 11 | 251 ± 64 | 199 ± 42 | 33.3 ± 3.2 | 14.8 ± 0.9 | 26.6 ± 0.4 | 24.9 ± 0.6 | 4.20 ± 0.19 | 1.25 ± 0.35 | 3.09 ± 0.25 | 2.75 ± 0.09 |
| 25 | 349 ± 53 | 148 ± 3 | 197 ± 6 | 150 ± 6 | 27.2 ± 1.2 | 16.1 ± 1.9 | 39.2 ± 0.4 | 29.9 ± 0.4 | 3.07 ± 0.23 | 1.24 ± 0.11 | 5.12 ± 0.21 | 2.89 ± 0.15 |
| 26 | 310 ± 3 | 178 ± 0 | 207 ± 20 | 118 ± 17 | 43.3 ± 1.3 | 11.4 ± 0.4 | 32.9 ± 1.5 | 24.2 ± 0.2 | 5.52 ± 0.23 | 1.80 ± 0.20 | 4.50 ± 0.29 | 2.41 ± 0.23 |
| 27 | 326 ± 25 | 172 ± 42 | 176 ± 8 | 118 ± 6 | 36.6 ± 1.9 | 15.3 ± 0.2 | 28.1 ± 1.2 | 22.5 ± 0.6 | 4.42 ± 0.09 | 0.57 ± 0.10 | 3.01 ± 0.01 | 1.70 ± 0.17 |
| 28 | 318 ± 31 | 190 ± 6 | 249 ± 1 | 140 ± 3 | 26.1 ± 0.1 | 3.8 ± 1.0 | 20.8 ± 0.8 | 8.3 ± 0.2 | 5.85 ± 0.08 | 2.70 ± 0.05 | 5.96 ± 0.01 | 2.55 ± 0.07 |
| 29 | 379 ± 56 | 199 ± 3 | 381 ± 20 | 336 ± 17 | 23.1 ± 0.3 | 2.1 ± 0.1 | 21.7 ± 0.1 | 9.6 ± 0.2 | 5.46 ± 0.19 | 2.21 ± 0.30 | 6.01 ± 0.12 | 3.61 ± 0.15 |
| 30 | 152 ± 20 | 124 ± 20 | 397 ± 3 | 363 ± 17 | 2.5 ± 0.1 | 1.0 ± 0.1 | 20.1 ± 0.5 | 12.3 ± 0.6 | 6.53 ± 0.50 | 4.42 ± 0.52 | 6.46 ± 0.18 | 4.64 ± 0.14 |
| 31 | 490 ± 17 | 296 ± 17 | 395 ± 89 | 318 ± 42 | 18.2 ± 6.8 | 1.7 ± 0.7 | 22.2 ± 0.2 | 8.9 ± 0.4 | 7.08 ± 0.16 | 4.79 ± 0.31 | 6.80 ± 0.04 | 4.14 ± 0.06 |
| 32 | 217 ± 11 | 52 ± 1 | 499 ± 20 | 421 ± 14 | 21.4 ± 0.1 | 1.3 ± 0.3 | 24.1 ± 0.3 | 12.6 ± 0.3 | 5.69 ± 0.01 | 4.02 ± 0.25 | 6.88 ± 0.03 | 4.44 ± 0.21 |
| 33 | 154 ± 17 | 64 ± 4 | 458 ± 56 | 308 ± 39 | 28.1 ± 0.3 | 1.7 ± 0.4 | 23.7 ± 0.6 | 7.8 ± 0.7 | 7.41 ± 0.17 | 4.79 ± 0.14 | 6.21 ± 0.01 | 3.08 ± 0.11 |
| Time (d) | SVI5 (mL/g) | Chlorophyll a (mg/g) | Chlorophyll b (mg/g) | Chlorophyll a/b | Granular Size (µm) |
|---|---|---|---|---|---|
| 1 | 73.6 ± 10.1 | 20.5 ± 0.7 | 10.7 ± 0.6 | 1.89 ± 0.04 | 710.2 ± 18.6 |
| 33 | 83.1 ± 0.1 | 1.2 ± 0.2 | 0.4 ± 0.1 | 2.99 ± 0.87 | 625.6 ± 28.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Zhu, L.; Ji, B.; Hou, H.; Ma, Y. Microalgal-Bacterial Granular Sludge Process in Non-Aerated Municipal Wastewater Treatment under Natural Day-Night Conditions: Performance and Microbial Community. Water 2021, 13, 1479. https://doi.org/10.3390/w13111479
Wang S, Zhu L, Ji B, Hou H, Ma Y. Microalgal-Bacterial Granular Sludge Process in Non-Aerated Municipal Wastewater Treatment under Natural Day-Night Conditions: Performance and Microbial Community. Water. 2021; 13(11):1479. https://doi.org/10.3390/w13111479
Chicago/Turabian StyleWang, Shulian, Lin Zhu, Bin Ji, Huan Hou, and Yingqun Ma. 2021. "Microalgal-Bacterial Granular Sludge Process in Non-Aerated Municipal Wastewater Treatment under Natural Day-Night Conditions: Performance and Microbial Community" Water 13, no. 11: 1479. https://doi.org/10.3390/w13111479
APA StyleWang, S., Zhu, L., Ji, B., Hou, H., & Ma, Y. (2021). Microalgal-Bacterial Granular Sludge Process in Non-Aerated Municipal Wastewater Treatment under Natural Day-Night Conditions: Performance and Microbial Community. Water, 13(11), 1479. https://doi.org/10.3390/w13111479








