Zooplankton Community Dynamics in Temporary Mediterranean Wetlands: Which Drivers Are Controlling the Seasonal Species Replacement?
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sample Collection
2.2. Statistical Analysis
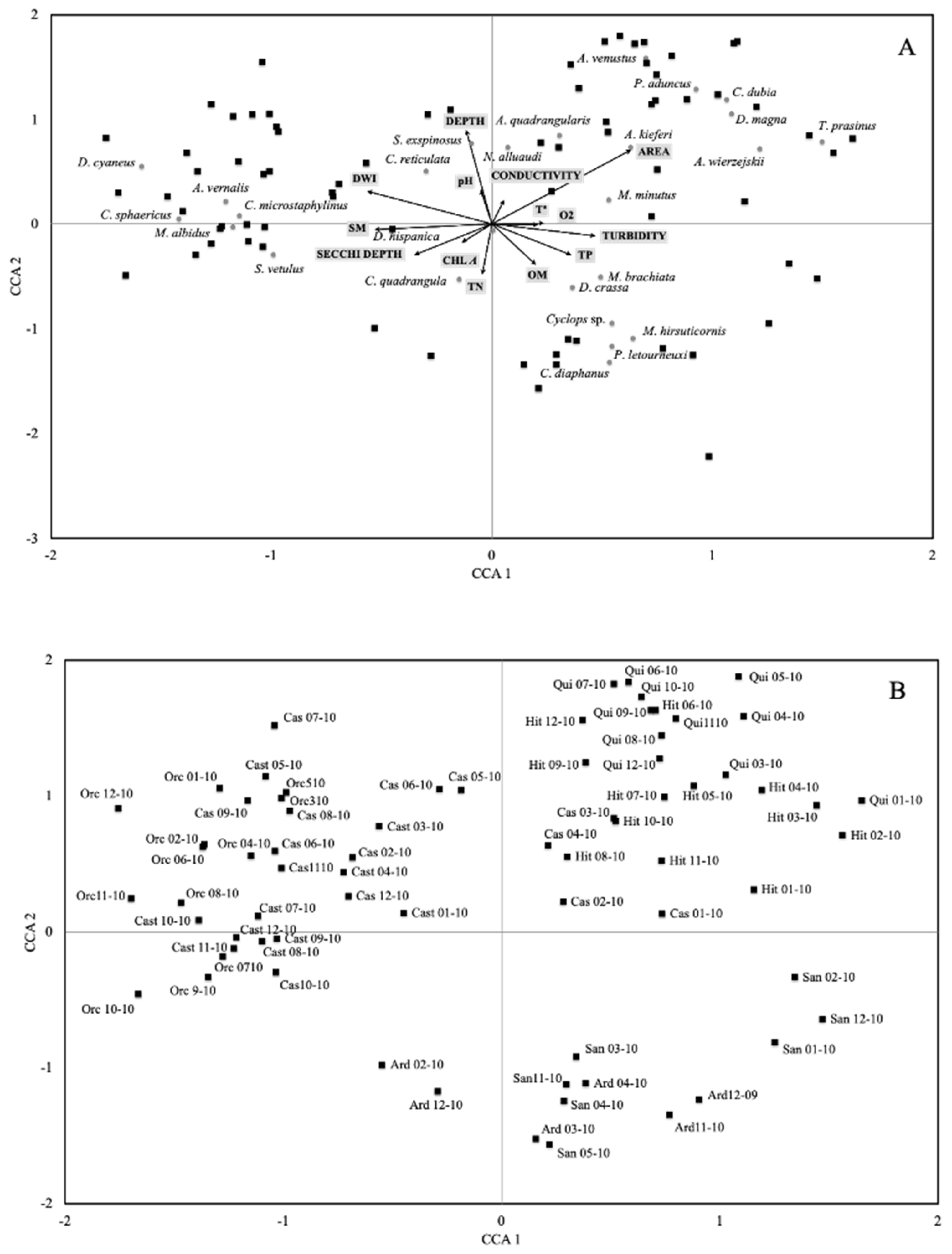
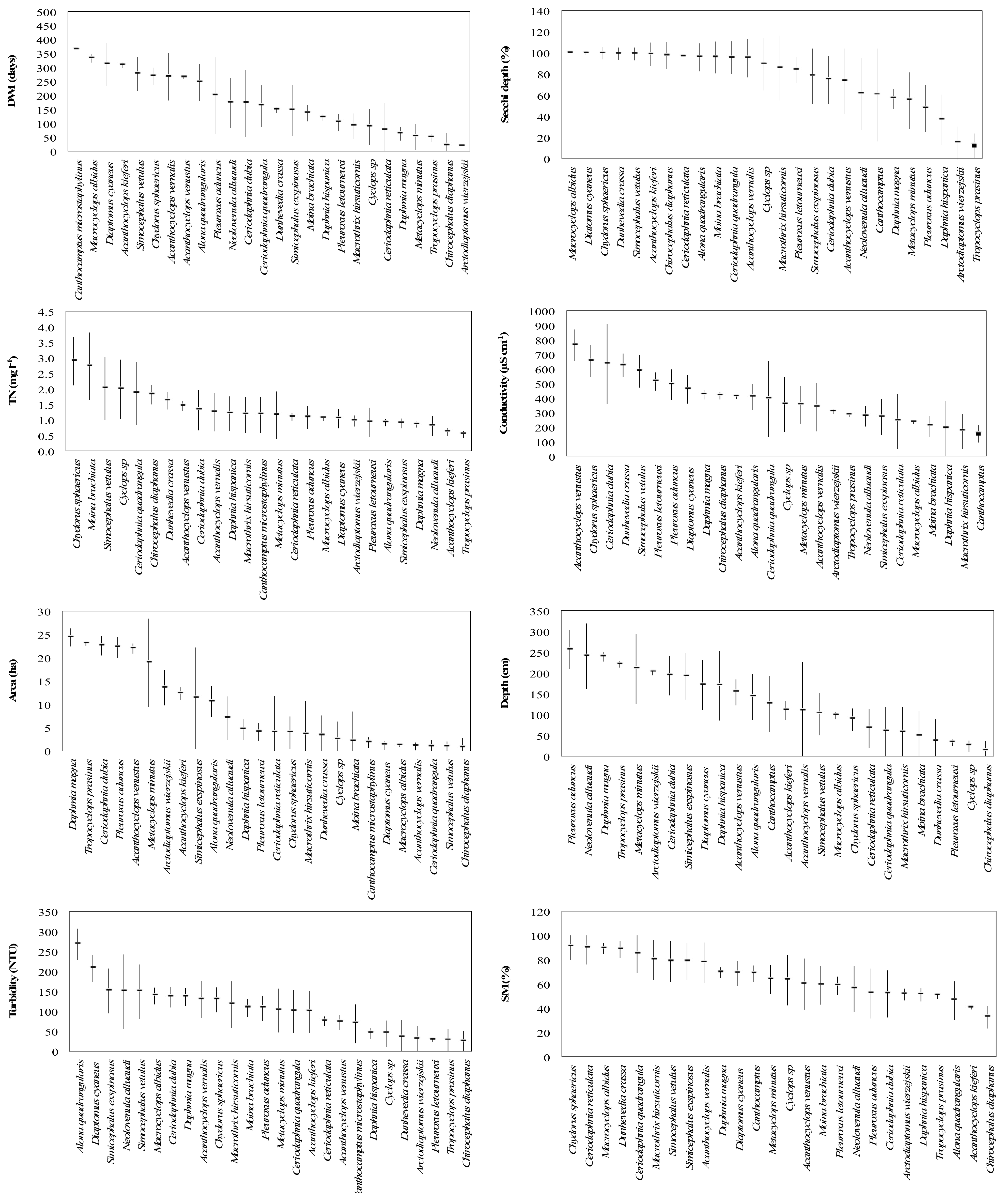
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Álvarez-Cobelas, M.; Rojo, C.; Angeler, D.G. Mediterranean limnology: Current status, gaps and the future. J. Limnol. 2005, 64, 13–29. [Google Scholar] [CrossRef]
- Gilbert, J.D.; de Vicente, I.; Ortega, F.; Jiménez-Melero, R.; Parra, G.; Guerrero, F. A comprehensive evaluation of the crustacean assemblages in southern Iberian Mediterranean wetlands. J. Limnol. 2015, 74, 169–181. [Google Scholar] [CrossRef]
- Boix, D.; Gascón, S.; Sala, J.; Badosa, A.; Brucet, S.; López-Flores, R.; Martinoy, M.; Gifre, J.; Quintana, X.D. Patterns of composition and species richness of crustaceans and aquatic insects along environmental gradients in Mediterranean water bodies. Hydrobiologia 2008, 597, 53–69. [Google Scholar] [CrossRef]
- Badosa, A.; Frisch, D.; Arechederra, A.; Serrano, L.; Green, A.J. Recovery of zooplankton diversity in a restored Mediterranean temporary marsh in Doñana National Park (SW Spain). Hydrobiologia 2010, 654, 67–82. [Google Scholar] [CrossRef]
- Gilbert, J.D.; de Vicente, I.; Jiménez-Melero, R.; Parra, G.; Guerrero, F. Selecting priority conservation areas based on zooplankton diversity: The case of Mediterranean wetlands. Mar. Freshw. Res. 2014, 65, 857–871. [Google Scholar] [CrossRef]
- Grillas, P.; Gauthier, P.; Yavercovski, N.; Perennou, C. Mediterranean Temporary Pools. Issues Relating to Conservation, Functioning and Management; Station Biologique de la Tour du Valat: Arles, France, 2004; Volume 1. [Google Scholar]
- Guerrero, F.; Parra, G.; Jiménez-Gómez, F.; Salazar, C.; Jiménez-Melero, R.; Galotti, A.; García-Muñoz, E.; Lendínez, M.L.; Ortega, F. Ecological studies in Alto Guadalquivir wetlands: A first step towards the application of conservation plans. Limnetica 2006, 25, 95–106. [Google Scholar]
- Zacharias, I.; Zamparas, M. Mediterranean temporary ponds. A disappearing ecosystem. Biodiver. Conserv. 2010, 19, 3827–3834. [Google Scholar] [CrossRef]
- Harris, R.; Jenkinson, I.; Lehman, J. The first 25 years of Journal of Plankton Research: Looking to the future. J. Plankton Res. 2004, 26, 1–3. [Google Scholar] [CrossRef]
- Moss, B.; Stephen, D.; Álvarez, C.; Becares, E.; van de Bunt, W.; Collings, S.E.; van Donk, E.; de Eyto, E.; Feldmann, T.; Fernández-Aláez, C.; et al. The determination of ecological quality in shallow lakes—A tested system (ECOFRAME) for implementation of the European Water Framework Directive. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, 507–549. [Google Scholar] [CrossRef]
- Lougheed, V.; Chow-Fraser, P. Development and use of a zooplankton index of wetland quality in the Lauretian Great Lakes basin. Ecol. Appl. 2002, 12, 474–486. [Google Scholar] [CrossRef]
- Bianchi, F.; Acri, F.; Bernardi-Aubry, F.; Berton, A.; Boldrin, A.; Camatti, E.; Cassin, D.; Comaschi, A. Can plankton communities be considered as bio-indicators of water quality in the Lagoon of Venice? Mar. Pollut. Bull. 2003, 46, 964–971. [Google Scholar] [CrossRef]
- Boix, D.; Gascón, S.; Sala, J.; Martinoy, M.; Gifre, J.; Quintana, X.D. A new index of water quality assessment in Mediterranean wetlands based on crustacean and insect assemblages: The case of the Catalunya (NE Iberian Peninsula). Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 16, 441–455. [Google Scholar] [CrossRef]
- Frisch, D.; Moreno-Ostos, E.; Green, A.J. Species richness and distribution of copepods and cladocerans and their relation to hydroperiods and other environmental variables in Doñana, south-west Spain. Hydrobiologia 2006, 556, 327–340. [Google Scholar] [CrossRef]
- Wetzel, R.G. Limnology: Lake and River Ecosystems, 3rd ed.; Academic Press: London, UK, 2001. [Google Scholar]
- Armengol, X.; Antón-Pardo, M.; Atiénzar, F.; Echevarrías, J.L.; Barba, E. Limnological variables relevant to the presence of the endangered White-headed duck in southeastern Spanish wetlands during a dry period. Acta Zool. Acad. Sci. Hung. 2008, 54 (Suppl. 1), 45–60. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for determination of phosphate in natural waters. Anal. Chem. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association Inc.: New York, NY, USA, 1995. [Google Scholar]
- Dussart, B. Calanoides et Harpacticoides. Les Copépodes des eaux Continentales d´Europe Occidentale; Tome I; Boubee, N. & Cie: Paris, France, 1967. [Google Scholar]
- Dussart, B. Cyclopoides et Biologie. Les Copépodes de eaux Continentales d´Europe Occidentale; Tome II; Boubee, N. & Cie: Paris, France, 1969. [Google Scholar]
- Alonso, M. Crustacea, Branchiopoda. Fauna Ibérica; Museo Nacional de Ciencias Naturales, CSIC: Madrid, Spain, 1996. [Google Scholar]
- Gannon, J.E.; Stemberger, R.S. Zooplankton (especially crustaceans and rotifers) as indicators of water quality. Trans. Am. Microsc. Soc. 1978, 97, 16–35. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F. Unimodal Models to Relate Species to Environment; Agricultural Mathematics Group: Wageningen, The Netherlands, 1987. [Google Scholar]
- Ter Braak, C.J.F.; Verdonschot, P.F.M. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquat. Sci. 1995, 57, 255–289. [Google Scholar] [CrossRef]
- Iturrondobeitia, J.C.; Caballero, A.I.; Arroyo, J. Avances en la utilización de los ácaros oribátidos como indicadores de las condiciones edáficas. Munibe 2004, 21, 70–91. [Google Scholar]
- Palmer, M.W. Putting things in even better order: The advantages of canonical correspondence analysis. Ecology 1993, 74, 2215–2230. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F. Canonical correspondence analysis: A new eigenvector technique for multivariate direct gradient analysis. Ecology 1986, 67, 1167–1179. [Google Scholar] [CrossRef]
- Waiser, M.J.; Robarts, R.D. Saline Inland Waters. In Encyclopedia of Inland Waters, 1st ed.; Likens, G.E., Ed.; Elsevier: Oxford, UK, 2009; Volume 2, pp. 634–644. [Google Scholar]
- Beklioglu, M.; Romo, S.; Kagalou, I.; Quintana, X.D.; Bécares, E. State of the art in the functioning of shallow Mediterranean lakes: Workshop conclusions. Hydrobiologia 2007, 584, 317–326. [Google Scholar] [CrossRef]
- Kagalou, I.I.; Kosiori, A.; Leonardos, I.D. Assessing the zooplankton community and environmental factors in a Mediterranean wetland. Environ. Monit. Assess. 2010, 170, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.D.; Guerrero, F.; Jiménez-Melero, R.; de Vicente, I. Is the bioproduction number a good index of the trophic state in Mediterranean wetlands? Knowl. Manag. Aquat. Ecosyst. 2015, 416, 5. [Google Scholar] [CrossRef][Green Version]
- Gilbert, J.D.; de Vicente, I.; Ortega, F.; García-Muñoz, E.; Jiménez-Melero, R.; Parra, G.; Guerrero, F. Linking watershed land uses and crustacean assemblages in Mediterranean wetlands. Hydrobiologia 2017, 799, 181–191. [Google Scholar] [CrossRef]
- López-González, P.; Guerrero, F.; Castro, M.C. Seasonal fluctuations in the plankton community in a hypersaline temporary lake (Honda, southern Spain). Int. J. Salt Lake Res. 1998, 6, 353–371. [Google Scholar] [CrossRef]
- Marrone, F.; Ortega, F.; Mesquita-Joanes, F.; Guerrero, F. On the occurrence of Metadiaptonus chevreuxi (Calanoida, Diaptomidae, Paradiaptomidae) in the Iberian Peninsula, with notes on the ecology and distribution of its European populations. Water 2020, 12, 1989. [Google Scholar] [CrossRef]
- Sahuquillo, M.; Miracle, M.R. Crustacean and rotifer seasonality in a Mediterranean temporary pond with high biodiversity (Lavajo de Abajo de Sinarcas, Eastern Spain). Limnetica 2010, 29, 75–92. [Google Scholar]
- Brucet, S.; Boix, D.; López-Flores, R.; Badosa, A.; Moreno-Amich, R.; Quintana, X.D. Zooplankton structure and dynamics in permanent and temporary Mediterranean salt marshes: Taxon-based and size-based approaches. Arch. Hydrobiol. 2005, 162, 535–555. [Google Scholar] [CrossRef]
- Florencio, M.; Fernández-Zamudio, R.; Lozano, M.; Díaz-Paniagua, C. Interannual variation in filling season affects zooplankton diversity in Mediterranean temporary ponds. Hydrobiologia 2020, 847, 1195–1205. [Google Scholar] [CrossRef]
- Boix, D.; Sala, J.; Moreno-Amich, R. The faunal composition of Espolla pond (NE Iberian peninsula): The neglected biodiversity of temporary waters. Wetlands 2001, 21, 577–592. [Google Scholar] [CrossRef]
- Badosa, A.; Boix, D.; Brucet, S.; López-Flores, R.; Quintana, X.D. Short-term variation in the ecological status of a Mediterranean coastal lagoon (NE Iberian Peninsula) after a man-made change of hydrological regime. Aquat. Conserv. Mar. Freshw. Ecosyst. 2008, 18, 1078–1090. [Google Scholar] [CrossRef]
- Pinto-Coelho, R.; Pinel-Alloul, B.; Méthot, G.; Havens, K.E. Crsutacean zooplankton in lakes and reservoirs of temperate and tropical regions: Variation with trophic status. Can. J. Fish. Aquat. Sci. 2005, 62, 348–361. [Google Scholar] [CrossRef]
- Kane, D.D.; Gordon, S.I.; Munawar, M.; Charlton, M.N.; Culver, D.A. The planktonic index of biotic integrity (P-IBI): An approach for assessing lake ecosystem health. Ecol. Indic. 2009, 9, 1234–1247. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.; Landkildehus, F. Trophic structure, species richness and biodiversity in Danish lakes: Changes along a phosphorus gradient. Freshw. Biol. 2000, 45, 201–218. [Google Scholar] [CrossRef]
- Kuczynska-Kippen, N.; Joniak, T. The impact of water chemistry on zooplankton occurrence in two types (field versus forest) of small water bodies. Int. Rev. Hydrobiol. 2010, 95, 130–141. [Google Scholar] [CrossRef]
- Kuczynska-Kippen, N.; Joniak, T. Zooplankton diversity and macrophyte biometry in shallow water bodies of various trophic state. Hydrobiologia 2016, 774, 39–51. [Google Scholar] [CrossRef]
- Jeppesen, E.; Lauridsen, T.L.; Kairesalo, T.; Perrow, M.R. Impact of submerged macrophytes on fish-zooplankton interactions in lakes. In The Structuring Role of Submerge Macrophytes in Lakes, 1st ed.; Jeppesen, E., Søndergaard, M., Søndergaard, M., Christoffersen, K., Eds.; Springer: New York, NY, USA, 1998; pp. 91–114. [Google Scholar]
- González-Sagrario, M.; Balseiro, E.; Ituarte, R.B.; Spivak, E. Macrophytes as refuge or risky area for zooplankton: A balance set by littoral predacious macroinvertebrates. Freshw. Biol. 2009, 54, 1042–1053. [Google Scholar] [CrossRef]
- Romo, S.; Villena, M.J.; Sahuquillo, M.; Soria, J.M.; Giménez, M.; Alfonso, T.; Vicente, E.; Miracle, M.R. Response of a shallow Mediterranean lake to nutrient diversion: Does it follow similar patterns as in northern shallow lakes? Freshw. Biol. 2005, 50, 1706–1717. [Google Scholar] [CrossRef]
- Søndergaard, M.; Jensen, J.P.; Jeppesen, E. Role of sediments and internal loading of phosphorus in shallow lakes. Hydrobiologia 2003, 506–509, 135–145. [Google Scholar] [CrossRef]
- Gilbert, J.D.; Guerrero, F.; de Vicente, I. Sediment desiccation as a driver of phosphate availability in the water column of Mediterranean wetlands. Sci. Total Environ. 2014, 466–467, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Angeler, D.G.; Viedma, O.; Sánchez-Carrillo, S. Conservation issues of temporary wetland Branchiopoda (Anostraca, Notostraca: Crustacea) in a semiarid agricultural landscape: What spatial scales are relevant? Biol. Conserv. 2008, 141, 1224–1234. [Google Scholar] [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes, 1st ed.; Springer: New York, NY, USA, 1998. [Google Scholar]
- Kasprzak, P.; Lathrop, R.C.; Carpenter, S.R. Influence of different-sized Daphnia species on chlorophyll concentration and summer phytoplankton community structure in eutrophic Wisconsin lakes. J. Plankton Res. 1999, 21, 2161–2174. [Google Scholar] [CrossRef]
- Moreno-Ostos, E.; Paracuellos, M.; de Vicente, I.; Nevado, J.C.; Cruz-Pizarro, L. Response of waterbirds to alternating clear and turbid water phases in two shallow Mediterranean lakes. Aquat. Ecol. 2008, 42, 701–706. [Google Scholar] [CrossRef]
- Effler, S.W.; Spada, M.E.; Gelda, R.K.; Peng, F.; Matthews, D.A.; Kearns, C.M.; Hairston, N.G., Jr. Daphnia grazing, the clear water phase, and implications of minerogenic particles in Onondaga Lake. Inland Waters 2015, 5, 317–330. [Google Scholar] [CrossRef]
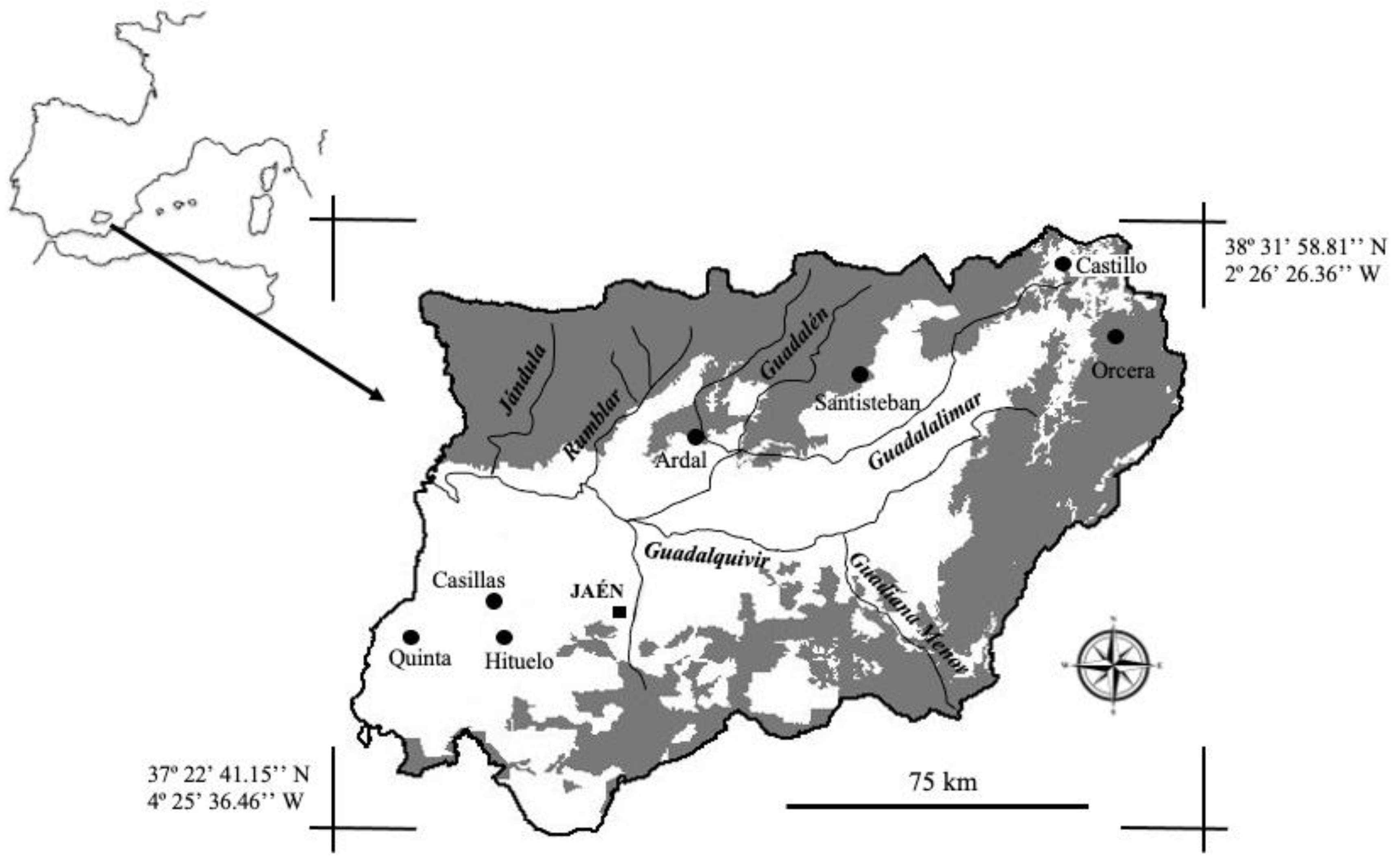
| Castillo | Orcera | Ardal | Santisteban | Hituelo | Quinta | Casillas | |
|---|---|---|---|---|---|---|---|
| Conductivity (µS cm−1) | 144 ± 55 | 444 ± 126 | 116 ± 52 | 454 ± 128 | 343 ± 74 | 561 ± 199 | 236 ± 44 |
| pH | 8.3 ± 0.6 | 8.1 ± 0.5 | 8.1 ± 0.8 | 7.8 ± 0.2 | 8.1 ± 0.4 | 8.5 ± 0.7 | 8.5 ± 0.6 |
| Turbidity (NTU) | 28 ± 67 | 6 ± 4 | 31 ± 25 | 71 ± 96 | 20 ± 24 | 11 ± 6 | 32 ± 44 |
| Chl-a (µg L−1) | 8.91 ± 7.46 | 7.64 ± 4.57 | 7.23 ± 3.55 | 8.55 ± 2.97 | 7.76 ± 10.08 | 8.82 ± 8.56 | 6.98 ± 5.06 |
| SM (%) | 80 ± 19 | 88 ± 14 | 65 ± 17 | 56 ± 27 | 54 ± 14 | 66 ± 23 | 64 ± 28 |
| Area (ha) | 1.94 ± 1.24 | 1.68 ± 1.24 | 0.14 ± 0.09 | 5.10 ± 5.20 | 16.82 ± 4.82 | 22.81 ± 3.45 | 3.62 ± 1.14 |
| Depth (cm) | 148 ± 61 | 172 ± 78 | 29 ± 9 | 27 ± 17 | 179 ± 52 | 20 0 ± 51 | 242 ± 87 |
| Secchi depth (cm) | 94 ± 54 | 172 ± 78 | 29 ± 9 | 15 ± 6 | 111 ± 65 | 146 ± 69 | 113 ± 76 |
| Temperature (°C) | 17.1 ± 8.2 | 14.4 ± 7.6 | 16.3 ± 6.5 | 14.7 ± 5.9 | 20.5 ± 7.8 | 20.4 ± 7.0 | 17.8 ± 7.1 |
| O2 (%) | 87.8 ± 17.9 | 77.6 ± 17.1 | 116.4 ± 32.8 | 74.5 ± 14.4 | 92.0 ± 10.0 | 82.7 ± 15.6 | 82.6 ± 21.7 |
| OM (mg L−1) | 3.9 ± 2.8 | 3.5 ± 2.5 | 8.2 ± 5.4 | 8.5 ± 3.7 | 4.2 ± 2.0 | 6.1 ± 4.9 | 3.8 ± 3.9 |
| TP (µg L−1) | 57.8 ± 44.1 | 35.4 ± 23.5 | 150.9 ± 87.0 | 122.3 ± 80.4 | 55.5 ± 33.6 | 49.1 ± 22.1 | 206.7 ± 155.3 |
| TN (mg L−1) | 1.1 ± 0.3 | 1.1 ± 0.8 | 2.0 ± 1.0 | 1.6 ± 0.7 | 0.7 ± 0.3 | 1.0 ± 0.6 | 1.0 ± 0.3 |
| DWI (days) | 414 | 353 | 192 | 171 | 343 | 340 | 343 |
| Coordinates (UTM) | 30SWH2357 | 30SWH3542 | 30SVH4721 | 30SVH8134 | 30SVG0679 | 30SUG8679 | 30SVG1084 |
| Altitude (m) | 780 | 1270 | 400 | 637 | 476 | 289 | 442 |
| Total Cumulative Richness | Copepod Abundance (ind L−1) | Branchiopod’s Abundance (ind L−1) | Shannon-Wiener (H’) | Equitability (J) | Simpson (D) | Cal:Cycl+Cla | GranCla:TotCla | Total Biomass (mg L−1) | |
|---|---|---|---|---|---|---|---|---|---|
| Castillo | 9 | 47.06 ± 127.21 | 1.51 ± 4.16 | 0.71 ± 0.29 | 0.49 ± 0.20 | 0.61 ± 0.16 | 0.0023 ± 0.0070 | 0.411 ± 0.420 | 0.104 ± 0.149 |
| Orcera | 8 | 65.73 ± 148.89 | 22.07 ± 67.51 | 0.60 ± 0.37 | 0.44 ± 0.27 | 0.67 ± 0.21 | 0.2299 ± 0.5309 | 0.151 ± 0.205 | 0.123 ± 0.201 |
| Ardal | 6 | 419.93 ± 678.49 | 45.62 ± 86.90 | 0.58 ± 0.22 | 0.50 ± 0.28 | 0.64 ± 0.18 | --- | 0.002 | 0.001 ± 0.001 |
| Santisteban | 9 | 809.82 ± 956.02 | 18.36 ± 34.63 | 0.30 ± 0.33 | 0.20 ± 0.19 | 0.85 ± 0.20 | --- | 0.0347 ± 0.0482 | 0.303 ± 0.195 |
| Hituelo | 13 | 39.72 ± 100.37 | 0.97 ± 2.67 | 0.46 ± 0.43 | 0.29 ± 0.27 | 0.74 ± 0.27 | 52.9825 ± 118.8370 | 0.059 ± 0.092 | 0.074 ± 0.119 |
| Quinta | 12 | 289.70 ± 938.60 | 11.56 ± 32.24 | 0.56 ± 0.43 | 0.40 ± 0.34 | 0.70 ± 0.25 | 0.001 ± 0.001 | 0.234 ± 0.259 | 0.015 ± 0.014 |
| Casillas | 12 | 21.36 ± 51.69 | 1.96 ± 4.00 | 0.47 ± 0.35 | 0.32 ± 0.26 | 0.75 ± 0.19 | 11.9280 ± 23.0303 | 0.078 ± 0.143 | 0.039 ± 0.048 |
| Pond | Season | Copepod Abundance (ind. L−1) | Branchiopoda Abundance (ind. L−1) | Cal:Cycl+Cla | GranCla:TotCla | Total Biomass |
|---|---|---|---|---|---|---|
| Castillo | Winter | 13.86 ± 28.91 | 1.35 ± 3.07 | 0.0003 ± 0.0002 | 0.77 ± 0.37 | 0.08 ± 0.13 |
| Spring | 5.74 ± 7.91 | 0.57 ± 0.78 | 0.009 ± 0.01 | 0.55 ± 0.38 | 0.01 ± 0.01 | |
| Summer | 115.12 ± 238.43 | 0.26 ± 0.32 | 0.0021 | 0.45 ± 0.47 | 0.10 ± 0.11 | |
| Autumn | 47.75 ± 49.63 | 5.92 ± 10.25 | --- | 0.014 | 0.19 ± 0.23 | |
| Orcera | Winter | 1.32 ± 1.20 | 0.08 ± 0.08 | 0.82 ± 0.92 | 0.071 | 0.02 ± 0.02 |
| Spring | 8.05 ± 7.13 | 1.75 ± 3.94 | --- | 0.041 ± 0.030 | 0.004 ± 0.003 | |
| Summer | 312.95 ± 302.73 | 79.39 ± 126.66 | --- | 0.12 ± 0.01 | 0.23 ± 0.30 | |
| Autumn | 58.11 ± 74.21 | 6.51 ± 8.76 | 0.103 ± 0.080 | 0.47 ± 0.10 | 0.24 ± 0.24 | |
| Ardal | Winter | 135.1 ± 156.12 | 90.74 ± 134.91 | --- | --- | 0.0004 ± 0.0001 |
| Spring | 1171.47 ± 1656.33 | 71.73 ± 49.78 | --- | 0.002 | 0.001 | |
| Summer | --- | --- | --- | --- | --- | |
| Autumn | 359.31 ± 392.70 | 5.92 ± 7.19 | --- | --- | 0.001 ± 0.001 | |
| Santisteban | Winter | 709.65 ± 881.71 | 11.97 ± 28.41 | --- | 0.07 ± 0.07 | 0.28 ± 0.15 |
| Spring | 1580.43 ± 833.22 | 28.17 ± 40.01 | --- | 0.03 ± 0.01 | 0.20 ± 0.04 | |
| Summer | --- | --- | --- | --- | --- | |
| Autumn | 603.03 ± 1040.81 | 25.06 ± 47.76 | --- | --- | 0.40 ± 0.54 | |
| Hituelo | Winter | 136.87 ± 192.97 | 1.28 ± 1.48 | 1.18 ± 0.03 | 0.23 ± 0.06 | 0.21 ± 0.19 |
| Spring | 7.98 ± 13.59 | 1.41 ± 3.87 | 38.31 ± 33.30 | 0.010 ± 0.005 | 0.07 ± 0.06 | |
| Summer | 4.08 ± 6.18 | 0.01 ± 0.003 | 172.21 ± 216.15 | --- | 0.009 ± 0.01 | |
| Autumn | 28.37 ± 40.41 | 0.87 ± 2.81 | 0.24 ± 0.34 | 0.08 ± 0.08 | 0.009 ± 0.009 | |
| Quinta | Winter | 989.41 ± 1790.86 | 27.23 ± 52.55 | 0.001 ± 0.001 | 0.60 ± 0.05 | 0.017 ± 0.02 |
| Spring | 4.67 ± 9.92 | 0.24 ± 0.25 | 0.003 | 0.37 ± 0.10 | 0.02 ± 0.02 | |
| Summer | 93.66 ± 132.21 | 1.57 ± 1.78 | --- | --- | 0.006 ± 0.008 | |
| Autumn | 57.50 ± 69.46 | 9.90 ± 20.42 | 0.0003 | 0.02 ± 0.02 | 0.02 ± 0.008 | |
| Casillas | Winter | 45.52 ± 87.95 | 3.08 ± 5.93 | 0.53 ± 0.57 | 0.04 ± 0.06 | 0.08 ± 0.05 |
| Spring | 21.83 ± 26.58 | 1.67 ± 3.21 | 5.40 ± 0.36 | 0.24 ± 0.32 | 0.08 ± 0.04 | |
| Summer | 0.46 ± 0.59 | 1.81 ± 3.29 | 0.32 ± 0.22 | --- | 0.005 ± 0.006 | |
| Autumn | 6.56 ± 12.03 | 0.85 ± 1.94 | 41.75 ± 33.35 | 0.12 ± 0.11 | 0.0003 ± 0.0002 |
| Conductivity | pH | Turbidity | Chl-a | SM | Area | Depth | Secchi | Temperature | O2 | MO | TP | TN | DWI | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total richness | −0.290 * | −0.318 ** | 0.256 * | 0.284 * | −0.350 ** | −0.250 * | ||||||||
| Copepod richness | −0.403 *** | 0.438 *** | −0.270 * | −0.272 * | −0.387 ** | |||||||||
| Branchiopod’s richness | −0.316 ** | −0.318 ** | 0.304 ** | 0.279 * | −0.321 ** | |||||||||
| Shannon-Wiener (H’) | −0.246 * | |||||||||||||
| Equitability (J) | −0.250 * | |||||||||||||
| Simpson (D) | −0.231 * | |||||||||||||
| Cal:Cycl+cla | −0.252 * | 0.350 * | 0.535 *** | −0.270 * | −0.301 * | −0.403 *** | ||||||||
| GranCla:TotCla | 0.270 * | |||||||||||||
| Total biomass |
| Axis1 | Axis 2 | Axis 3 | |
|---|---|---|---|
| Axis summary statistics | |||
| Eigenvalue | 0.677 | 0.594 | 0.401 |
| p-value | 0.001 | 0.001 | 0.001 |
| % of variance explained | 10.3 | 9 | 6.1 |
| Cumulative % explained | 10.3 | 19.3 | 25.4 |
| Pearson Correlation, Spp-Evnt* | 0.920 | 0.884 | 0.842 |
| p-value | 0.001 | 0.001 | 0.001 |
| Correlations of environmental variables and canonical axes (Intraset correlations of terBraak, 1986) | |||
| Conductivity | 0.056 | 0.232 | −0.490 |
| pH | −0.052 | 0.331 | 0.140 |
| Turbidity | 0.470 | −0.114 | 0.195 |
| Chl-a | −0.135 | −0.173 | −0.384 |
| SM | −0.531 | −0.051 | 0.207 |
| Area | 0.633 | 0.711 | −0.038 |
| Depth | −0.117 | 0.898 | 0.252 |
| Secchi depth | −0.350 | −0.293 | −0.431 |
| Temperature | 0.209 | 0.007 | −0.091 |
| O2 | 0.235 | 0.007 | 0.319 |
| OM | 0.199 | −0.383 | −0.430 |
| TP | 0.361 | −0.298 | −0.039 |
| TN | −0.044 | −0.481 | −0.287 |
| DWI | −0.565 | 0.318 | −0.476 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gilbert, J.D.; de Vicente, I.; Ortega, F.; Guerrero, F. Zooplankton Community Dynamics in Temporary Mediterranean Wetlands: Which Drivers Are Controlling the Seasonal Species Replacement? Water 2021, 13, 1447. https://doi.org/10.3390/w13111447
Gilbert JD, de Vicente I, Ortega F, Guerrero F. Zooplankton Community Dynamics in Temporary Mediterranean Wetlands: Which Drivers Are Controlling the Seasonal Species Replacement? Water. 2021; 13(11):1447. https://doi.org/10.3390/w13111447
Chicago/Turabian StyleGilbert, Juan Diego, Inmaculada de Vicente, Fernando Ortega, and Francisco Guerrero. 2021. "Zooplankton Community Dynamics in Temporary Mediterranean Wetlands: Which Drivers Are Controlling the Seasonal Species Replacement?" Water 13, no. 11: 1447. https://doi.org/10.3390/w13111447
APA StyleGilbert, J. D., de Vicente, I., Ortega, F., & Guerrero, F. (2021). Zooplankton Community Dynamics in Temporary Mediterranean Wetlands: Which Drivers Are Controlling the Seasonal Species Replacement? Water, 13(11), 1447. https://doi.org/10.3390/w13111447








