Abstract
In recent years, natural thermal mineral waters have been gaining the special attention of the scientific community, namely in the prevention and treatment of some diseases, due to the microbial properties that exist in these habitats. The aim of this work was to characterize the physicochemical composition and the microbial taxonomic communities present in three thermal waters of the Galician region in Spain and two samples of the northern region in Portugal. These collected water samples were analyzed for physicochemical characterization and the respective hydrogenome of the waters using next generation sequencing together with 16S rRNA gene sequencing. The sequencing showed a high diversity of microorganisms in all analyzed waters; however, there is a clear bacterial predominance of Proteobacteria phylum, followed by Firmicutes, Deinococcus-Thermus, Aquificae and Nitrospira. The main physicochemical parameters responsible for the clustering within the Spanish waters were sulfur compounds (SO32− and S2−), CO32− and neutral pH, and in the Portuguese waters were Mg, Ca and Sr, nitrogen compounds (NO3− and NH4+), Na, Rb, conductivity and dry residue. This work will allow for a better understanding of the microbial community’s composition and how these microorganisms interfere in the physicochemical constitution of these waters often associated with medicinal properties. Furthermore, the hydrogenome may be used as an auxiliary tool in the practice of medical hydrology, increasing the likelihood of safe use of these unique water types.
1. Introduction
Water is considered the basis for biodiversity on our planet, it is part of all life processes and it has a primordial function in the existence of life as we know it [1]. The properties of water give it fundamental characteristics for the structural organization and for the three-dimensional assembly of proteins and nucleic acids [2], acting also in the accomplishment of the diverse biological functions of these molecules [3].
For thousands of years, humans have lived and seized numerous lands, grown and developed due to this invaluable resource that is water [4]. However, over time, humans began to observe the beneficial effects that some waters had on health, these waters became known as the thermal waters and some civilizations even attributed to them a strong religious and sacred character [5,6,7]. Both the ancient Greeks and the Romans surmised the beneficial properties of the thermal waters well and used them to alleviate joint pain, muscle pain or the treatment of skin diseases [8,9]. Thermal waters are all-natural mineral waters with therapeutic applications or health beneficial effects, emerging from the inside of a thermal spring, independently of the source temperature, according to Medical Hydrology [10]. According to the European Directive 2009/54/EC, natural mineral waters are defined as microbiologically wholesome and originated from an underground water table or deposit, emerging from a natural spring or bore. These waters are characterized by nature (mineral content and trace elements, among others) and original purity. In addition, these water characteristics can confer beneficial effects to health, considering the assessment of the main following parameters, such as physicochemical, microbiological (absence of pathogenic microorganisms and parasites), hydrological and geological [11].
Due to the scientific advances, the therapeutic experience of physicians and the new studies of hydrology, pharmacology and biochemistry, thermal waters have once again taken on great importance [9] in the prevention and treatment of some heart diseases [12], skin diseases [13,14,15], respiratory diseases [16,17] or rheumatic diseases, such as osteoarthritis, fibromyalgia, ankylosing spondylitis, rheumatoid arthritis and chronic back pain [18,19,20].
The specific therapeutic purposes of each thermal water are closely related to the microbial diversity and the different physical and chemical characteristics they present, such as pH, temperature, total mineralization, salts concentration and the nature of the ionic components, among others [7,10,11,21]. Based on the chemical composition, the natural mineral waters are classified as sulphated, bicarbonated, chlorinated, sulphurous, gasocarbonic and hiposaline waters. Each water chemical composition is associated with a specific therapeutic indication. For instance, sulphurous waters are indicated for reumathic and musculoskeletic, dermatologic, gynecologic and respiratory diseases; gasocarbonic waters are indicated for circulatory and digestive systems; bicarbonate thermal waters are indicated for respiratory, urinary, digestive and endocrine diseases [10,21]. These characteristics depend on their respective lithostructural and tectonic context or geographic location [7,21]. Water, when reacting with the various components present in the rocks, obtains some constituents that will modify its composition. A recent study reported that silica-rich thermal waters showed dermatologic properties with clinical benefits in case of atopic dermatitis and psoriasis, reducing the macrophages and keratinocytes proliferation and cell metabolism [15]. Additionally, the presence of microbial communities associated with thermal waters also contribute to the diversity and specialization of the hydromineral richness, according to some authors [7,13,21]. However, thermal waters with the geographic location in seismic areas should be monitored carefully, since the earthquake can show a high impact on water safety and stability, including the restoration of microbial composition [22].
There are a number of studies, where the importance of microbial communities in thermal waters has been described in obtaining new molecules of biotechnological interest [23,24,25,26]. However, only recently a few authors [13,21,27,28] have verified that the microbial composition of these waters may also play a greater role in the health benefits. Thermal waters have unique microbiomes related to the physicochemical characteristics previously described; however, the presence of some active prebiotics may stimulate the growth of bacterial species beneficial to health, especially if they are already present in its natural microbial content [13,27]. For instance, the antirheumatic properties of sulphurous-bromine-iodine thermal water have been associated with the presence of the followed bacteria belonging to genera Geothermobacterium, Thermus, Syntrophomonas, Desulfomonile, Thiofaba and Thermodesulfovibrio, according to Paduano et al. [21]. Moreover, these characteristics can contain or also allow the growth and colonization of pathogenic microorganisms like Legionella. The predominant genera identified in water samples obtained from the borehole of an Australian balneotherapy centre were Sphingobium, Agrobacterium and Thiobacillus [29].
Given the benefits presented directly related to health, there is a need to understand and study the total content and diversity of microorganisms present in this water type. With the development of the next generation sequencing (NGS) and the increase of computational capacity in bioinformatics, it is possible to have access to the total hydrogenome present in the thermal and natural mineral waters [22,26,30]. Therefore, the term hydrogenome comprises all the genomes of the microorganisms present in a given aquatic environment. This study of hydrogenome may be used as an auxiliary tool in the practice of medical hydrology, since this new approach makes possible the analysis of the composition of the taxonomic and functional profiles of the microorganisms existing in the thermal waters, including the raw material of the thermalism or balneotherapy, and increasing the safety in the used water. The use of NGS and bioinformatics tools has the main objective of assisting classical microbiology methods by allowing the direct and rapid study of the genome of the microbial communities from the most diverse environmental samples, including the identification of uncultured microorganisms [26,30].
The Iberian Peninsula is rich in hot springs with therapeutic purposes. In particular, the region of Galicia in Spain and the northern region in Portugal have a strong thermal tradition through the exploration of these resources [31,32,33,34]. For these reasons, the aim of this pilot study was to characterize the hydrogenome of five thermal waters of these two regions of the Iberian Peninsula, using NGS and the molecular technique of the 16S rRNA gene.
2. Materials and Methods
2.1. Sampling Site
This study was carried out in two regions of the Iberian Peninsula, namely in the region of Galicia (Spain) and in the northern region of Portugal.
The region of Galicia is essentially formed by a vast outcrop of metamorphic rocks (crystalline rocks) and igneous (granitic rocks). In this Spanish province, there is also a great variety of hot springs, with more than three hundred sources, of which 20 are used by spas [33]. The chemical composition of the waters emanating from this region is largely conditioned by several factors, such as atmospheric gases, aerosols, surface erosion of rocks and soils (including their biological constituents), the dissolution and precipitation of minerals from the surface and the existence of a certain “thermal anomaly” in the region [33]. The chemical composition of the Galician waters is mainly bicarbonated sodium and calcium waters, resulting from the occurrence of carbonation reactions and hydrolysis of the minerals present in the subsoil, among them feldspar [35,36]. Galician waters have as main therapeutic indications at rheumatological, respiratory, dermatological, digestive and hepatobiliary levels [37].
Portugal considered one of the richest thermal countries of the European Union, comprises more than fifty thermal centers [15]. One of the main Portuguese regions to natural thermal mineral waters is located in the Transmontano-Duriense, which presents a huge variety of hot springs [38], typologies and occurrences. These waters are mainly of meteoric origin, that is to say, they result from a deep infiltration coming from rainwater [39]. The thermal waters currently in operation in this region may have been infiltrated for about 10,000 years, at the beginning of the Holocene, placing the hydrothermal richness of the Trás-os-Montes region on a plateau of great national prominence [39]. The natural mineral waters of the Trás-os-Montes region are mainly bicarbonate, sodium, gasocarbon, fluoride and acid waters [31,40]. These waters are still geared to therapeutic practice at the level of the cardiovascular, digestive, respiratory, metabolic-endocrine, dermatological, rheumatic and musculoskeletal systems and nervous system [15,31,40,41].
2.2. Sample Collection
Water samples (50L) were collected from November 2017 to March 2018, in the following points, ES_TI (42.346583, −7.877306), ES_PR (42.254466, −8.167167) and ES_BUR (42.334706, −7.864560), which were related to the samples collected in the Galicia region in Spain, while the points PT_SA (41.855243, −7.185342) and PT_CH (41.738313, −7.473070) corresponded to the samples collected in the northern region of Portugal. The collection was performed after flushing water for 1 min, filling the bottles up to the top and closing them with caps. During the process of water collecting in the respective sources, the measurement of its emergency temperature was carried out using a digital thermometer (Table 1).

Table 1.
Physicochemical composition of the analyzed thermal waters.
2.3. Physicochemical Analysis
All the water samples were analyzed for the following physicochemical parameters: micronutrients (iron, copper, zinc, manganese, boron, cobalt, molybdenum, vanadium, iodine, fluorine and arsenic), cations (sodium, potassium, rubidium, calcium, cesium, magnesium, strontium, lead, cadmium, aluminum and ammoniacal nitrogen) and anions (chlorine, sulphites, sulphates, sulphides, carbonates, bicarbonates, nitrites, nitrates and phosphates). These analyses were carried out in the Laboratory of Soils and Plants Joaquim Quelhas dos Santos at the University of Trás-os-Montes and Alto Douro and at the Center for Scientific and Technological Support for Research of the University of Vigo, using standard methods. The values are shown in Table 1.
Taking into account the physicochemical parameters, the thermal waters collected in two regions of the Iberian Peninsula were classified chemically using the Piper diagram, indicating their similarities according to the cations and anions (expressed in mEq/L) represented by two different triangles [42,43]. Additionally, the principal component (PC) analysis and cluster analysis were performed, in order to evaluate the similarities and differences of the physicochemical composition of waters. The PC analysis was performed using the Excel XLStat statistical package (version 2.11.1, Addinsoft, Paris, France) [44]. The experimental data on the physicochemical characterization of the waters (Table 1) were first normalized, from these a correlation matrix was obtained, the own values of each main component and the percentage of the variance was explained for each main component. As a criterion for the choice of PCs, the number of components explaining most of the variability (greater than 70% of the total variance) was considered [45].
2.4. DNA Extraction and 16S Amplicon Sequencing
Water samples were filtered through 0.22 μm filters (Sartorius Stedim Biotech, Goettingen, Germany), in order to isolate the microorganisms, present in the thermal water. Total genomic DNA extraction from samples was performed using the DNeasy® PowerWater® kit (Qiagen, Hilden, Germany), following the manufacturer’s instructions. After extraction of DNA, amplification and preparation of libraries were performed according to the 16S metagenomic sequencing library preparation (Part # 15044223, see A, Illumina Inc., San Diego, CA, USA). The amplicons were obtained by the amplification of the V3-V4 regions of the 16S rRNA gene of both Bacteria and Archaea, using the following primers 341F (5′-CCTACGGGNGGCWGCAG-3′) and 785R (5′-GACTACHVGGGTATCTAATCC-3′) [46], and specific adapters of the Illumina MiSeq platform (Illumina Inc., San Diego, CA, USA) [47,48,49]. The libraries obtained were quantified and validated through a Bioanalyzer (Agilent Technologies, Santa Clara ,CA, USA). After obtaining the libraries, sequencing was performed through the Illumina MiSeq platform, using the MiSeq Reagent Kit v3, according to the manufacturer’s protocol and in a 2 × 300 bp paired-end system. All of the sequencing was performed at the Madrid Science Park.
2.5. Bioinformatic Analysis
The obtained sequencing data were analyzed using bioinformatics tools, such as FASTQC(Babraham Bioinformatics, Cambridge, UK) [50] and Metagenomic Rapid Annotations using Subsystems Technology (MG-RAST server, http://www.mg-rast.org (accessed on 12 September 2018)) [51]. The FASTQC tool was used to analyze the quality of the FASTQ files from the MiSeq sequencer (16S rRNA sequencing). This tool allows for the reporting of a wide variety of information related to the quality profile of the readings obtained, and for evaluating the guanine–cytosine (GC) content and the abundance of adapters [52], which may still be present after the initial preprocessing carried out by the company Madrid Science Park.
This MG-RAST server is a bioinformatic tool that allows the functional and taxonomic annotation of sequences of environmental samples (metagenomics) through the comparison with databases such as KEGG, M5NR, Genbank, PATRIC, RefSeq, SwissProt and SEED, among others. This tool also offers quality control, annotation and various methods for analyzing different types of data, including phylogenetic and metabolic reconstructions, and the ability to compare more than one metagenome [51].
Reads were taxonomically annotated by similarity searching against M5NR database, respectively, with default parameters (maximum e-value cutoff of 10−5, minimum identity cutoff of 60% and minimum alignment length cutoff of 15).
3. Results and Discussion
3.1. Physicochemical Classification of Natural Thermal Mineral Waters
The five water samples analyzed in this study showed distinct physicochemical and microbiota composition. The physicochemical analysis of water samples showed that the pH values vary between 6.1 and 7.5, thus indicating slightly acidic, neutral and alkaline waters (Table 1). Regarding the temperatures recorded in the various thermal waters, these were found to vary between 15 and 70 °C. Normally, higher temperatures favor the appearance of more mineralized water [7], however, water PT_SA has very high mineralization values even though it is a water with a low emergency temperature (15 °C), which may be related to the fact that emergency temperature is often much lower than the maximum temperature reached in the reservoir, as it decreases along the ascending hydraulic circuit [33,53].
According to European legislation, water with a ≤50 mg/L dry residue is classified as hyposaline or very little mineralized, 50–500 mg/L is low mineralized, 500–1500 mg/L is classified as mineralized water and water with dry residue above 1500 mg/L is classified as hypersaline, presenting strong mineralization [54]. Thus, water samples ES_PR and ES_TI can be classified as low mineralized waters, ES_BUR and PT_SA as mineralized waters and PT_CH as being hypersaline water with values of mineralization above 1500 mg/L (Table 1). These results are confirmed by the values of electrical conductivity. Since this parameter allows us to evaluate its degree of mineralization as results from the relationship between the content of dissolved minerals in the water and the resistance that it offers the passage of electric current.
Based on the Piper diagram, thermal waters of the two regions of the Iberian Peninsula were classified chemically (Figure 1), the Piper diagram analysis showed that there are mainly three water groups, two of them in a transient phase (Figure 1). The first group included the water samples ES_BUR, ES_TI and PT_CH, classifying them as bicarbonated sodium waters. In addition, these samples presented high values of sodium (Na) and potassium (K), but also of bicarbonate anions (HCO3−) (Table 1). The second group, the sample ES_PR is located in one of the transition zones, between the bicarbonated sodium waters and the sulfated or chlorinated sodium waters. The classification can be justified when analyzing these results together with Table 1, since the sample ES_PR had the highest values of sulfate anions (SO42) and sulfite (SO32−), compared to the other samples. Regarding the third group, the water sample PT_SA is also located in a transition zone, between the sodium bicarbonated waters and the bicarbonated calcium or magnesian waters. Additionally, the sample PT_SA had the highest values of magnesium (Mg) and calcium (Ca), but also had a high content of sodium (Na) and bicarbonate anions (HCO3−), thus placing it in the transition between the bicarbonated sodic waters and the bicarbonated calcium or magnesian waters.
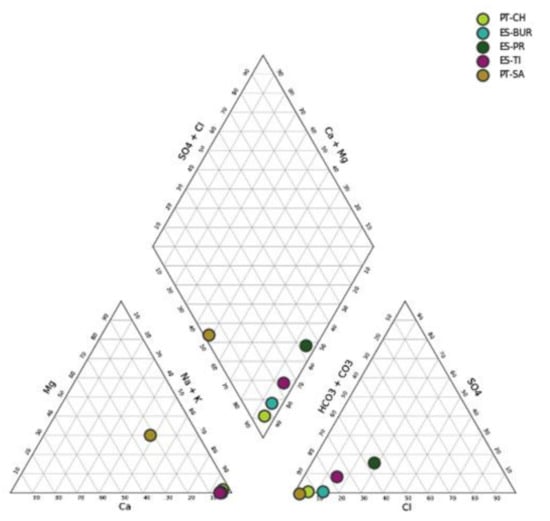
Figure 1.
Classification of the thermal waters analyzed in this study, based on their anions and cations composition, using the Piper diagram.
The analysis of principal components based on the all analyzed physicochemical parameters (Table 1) showed that considering the first two main components (PC1 and PC2), the obtained results can explain 71% of the total system information (Table 2). However, the addition of a third major component (PC3) can explain 95% of the cumulative variance. Therefore, only the first three main components were selected for the next analysis.

Table 2.
Obtained values of the correlation matrix of the thermal waters analyzed, for the four principal components (PCs).
The projection of the first two main components (PC1 and PC2) and the main components (PC1 and PC3) are demonstrated in Figure 2, respectively. Regardless of the PC1 and PC2 components or the PC1 and PC3 components can be observed that the five samples of thermal waters in both projections were always grouped in four groups, although a displacement occurs in the positioning of the waters in the quadrants. Considering this reason and that the PC1 and PC2 components present more than 70% of the variance in comparison to 66.74% for the PC1 and PC3; the thermal waters were clustered into the following groups: (1) ES_BUR, (2) PT_CH, (3) PT_SA and (4) ES_PR and ES_TI.
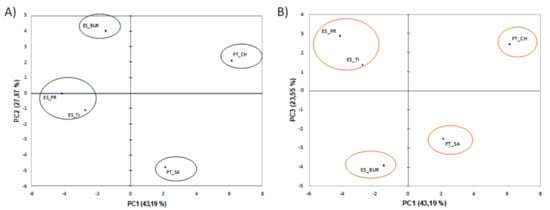
Figure 2.
Projection of the first two main components (PC1 and PC2), showing 71.05% of cumulative variance (A) and projection of the main components (PC1 and PC3), showing 66.74% of cumulative variance (B).
The results of the PC analysis represented graphically using the “biplot” function with the scale of species and localities (Figure 3), allowed visualizing the variables that most influence the classification of the thermal waters. The variables with greater weight in each component are represented (square cosine > 0.5).
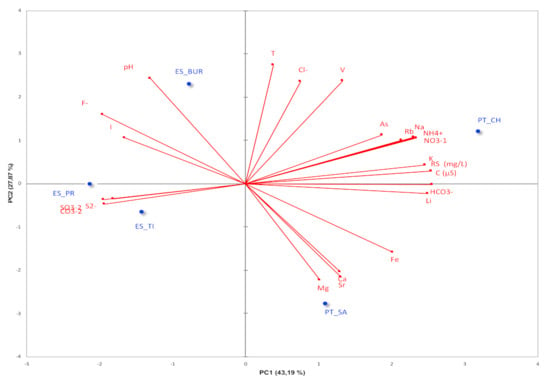
Figure 3.
Results of the PC analysis of the main components PC1 and PC2 represented graphically, using the “biplot” function with 71.05% of cumulative variance.
Considering the water samples into four groups, among which ES_PR and ES_TI formed an independent cluster located in the opposite (and therefore negatively correlated) quadrant relative to water PT_CH. In turn, the PC analysis positions the water ES_BUR in the quadrant opposite PT_SA. The water samples ES_PR and ES_TI demonstrated a negative correlation with the PC1 and PC2 components, and the ions that allowed them to be grouped were mainly composed of sulfur (SO32− and S2−) and CO32−. This fact was confirmed by physicochemical results, in which these two waters showed the richest in sulfur compounds (Table 1). This type of water exerts a number of beneficial effects on health, namely anti-inflammatory, keratoplastic and antipruritic properties, also having antibacterial and antifungal properties due to the presence of sulfur [10,55]. The sulfur may also interact with oxygen radicals present in the deepest layers of the epidermis, generating sulfur and sulfur dioxide, which can be transformed into pentaionic acid (H2S5O6), acid responsible for the antibacterial and antifungal activity of sulfur water [55]. In relation to the other waters, the variables that most contribute to their grouping in different groups are the cations (Mg, Ca and Sr) for the water sample PT_SA and the nitrogen compounds (NO3− and NH4+), conductivity, dry residue, Na and Rb for the sample PT_CH. For water ES_BUR, the pH is the only variable that contributes to classify it, considering the components PC1 and PC2.
3.2. Characterization of Microbiota Communities of Natural Thermal Mineral Waters
A total of 804,158 sequence reads were obtained from the five water samples by the sequencing of the 16S rRNA gene, with an average length of 426 base pairs (bp) (Table 3). About 99.95% of the reads submitted from the thermal water samples were maintained after the quality control step of the server itself. In addition, evaluation of the GC content demonstrated good global GC contents (>50%) in all water samples. These sequences were deposited in the NCBI Short Read Archive (GenBank accession numbers SRX5099959-SRX5099963, https://www.ncbi.nlm.nih.gov/bioproject/?term=PRJNA508462 (accessed on 20 May 2021)). According to Browne et al. [56], the optimal coverage for GCs content should be range between 45 and 65% in the metagenome dataset. While GC content outside of this interval can show coverage biases, indicating difficulty on genome sequencing.

Table 3.
General metagenomic annotation data on the MG-RAST server.
For each analyzed water sample, the rarefaction curve was calculated through the MG-RAST server. These rarefaction curves represent the richness of the different species annotated as a function of the number of reads presented, demonstrated that practically all samples tend to approach an asymptote before 100,000 reads, that is, most species present in the samples have already been discovered (Figure 4). Taking into account that all samples reached a stable plateau, obtaining more than 100,000 reads after the quality control of the MG-RAST server (Table 3) and a larger number of reads would not increase the count of species obtained [21,57]. These results demonstrated a reliable sequencing of the microbiota community and there is still a great diversity of unknown microorganisms.
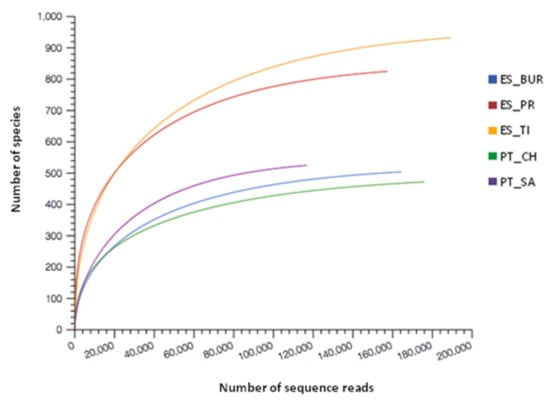
Figure 4.
Rarefaction curves of the bacterial 16S rRNA gene sequences for each water sample obtained from the MG-RAST server. The total number of sequence reads analysed is plotted against the number of species observed in the same community.
Regarding the number of species, the samples tended to cluster into two distinct groups, ES_BUR, PT_SA and PT_CH formed a first group, and ES_PR and ES_TI formed a second group (Figure 4). This may be related to the different temperatures recorded in their waters. The temperature is one of the factors that most affects the microbial growth, as these increases accelerate the enzymatic reactions, and the growth is faster [58]. This process occurs until reaching a maximum temperature from which some of the cellular components can begin to denature irreversibly (proteins, nucleic acids, etc.), thus stopping the microbial growth [58,59]. Therefore, each microorganism has its own optimum growth temperature in which the enzymatic reactions occur at the maximum possible speed. Paduano et al. [21] reported the highest bacterial diversity and complexity of the thermal waters in the lowest temperatures, varying between 36 and 44 °C. In the case of mesophilic microorganisms, their temperatures can vary between 15 and 60 °C [23]. Taking into account that in this study, the water samples of group two (ES_TI and ES_PR) had temperatures between 40 and 50.4 °C, this could be one of the main factors that contributed to the appearance of a higher species richness (number of species) compared to group one. Group one included the water samples (ES_BUR, PT_CH and PT_SA) with more extreme temperatures, which probably could be related to a lesser amount of microbial diversity. In addition, the extreme environments present numerous challenges for the microorganisms that inhabit them, challenges that may require an adaptation of all cellular machinery for both psychrophilic microorganisms and for thermophilic and hyperthermophilic microorganisms [60]. Thus, given that both low and high temperatures affect the structure and function of microbial cells [23,61]. Into group two is expected that the existence of microorganisms in these waters is strongly conditioned by the type of adaptations they have, showing a lower degree of species richness compared to the samples of another group (Figure 4). Some studies demonstrated that few microbial communities are adapted to conditions of high (>60 °C) or low temperatures (<20 °C), resulting, therefore, in a low diversity compared to mesophilic environments [62,63]. For instance, Everroad et al. [59] reported that the only Aquificales members, in particular Sulfurihydrogenibium, grew above 67 °C from a slightly alkaline sulfide-containing hot spring in Japan. In addition, these authors observed that with the decrease of temperature, the primary productivity increased through the anoxygenic and oxygenic phototrophs, and consequently, the community diversification.
Comparison of Microbial Taxonomic Profiles
The taxonomic classification based on 16S rRNA gene sequences analysis allowed determining the composition of the microbial communities associated to the different waters analyzed in the two regions of the Iberian Peninsula, through the MG-RAST server, according to Ribosomal Database Project (RDP) classification by interactive graphs (Krona) created to each sample (Figure 5 and Figure 6). Krona graphs represent an important metagenomic visualization tool, facilitating the study and access to bioinformatic data [64]. In this study, the taxonomic profile showed that between 99.0% and 99.9% of assigned reads were from the Bacteria realm and the remainder from Archaea.
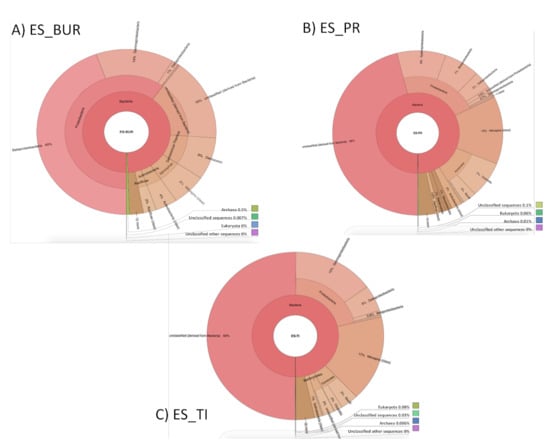
Figure 5.
Analysis of the taxonomic distribution of the microbial community in the Spanish thermal waters, based on the 16S rRNA gene sequences.
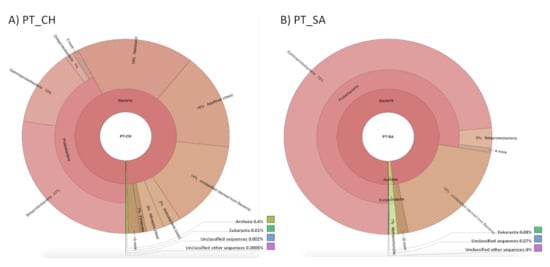
Figure 6.
Analysis of the taxonomic distribution of the microbial community in the Portuguese thermal waters, based on the 16S rRNA gene sequences.
The water sample ES_BUR showed that 99.95% of the sequences belong to the realm of Bacteria, of which 60% were of the Proteobacteria (45% Betaproteobacterias, 14% Gammaproteobacterias and 1% Deltaproteobacterias), 8% Deinococci, 6% Nitrospira, 4% Actinobacteria, 2% Aquificae and 4% belonged to minority classes. However, 16% of these bacterial sequences were not classified (Figure 5).
The sample ES_PR, 99.99% of its sequences belonging to the Bacteria kingdom, of these sequences, 22% belong to the phylum of Proteobacteria (9% Epsilonproteobacteria, 7% Betaproteobacterias and the remaining were distributed among the other classes, such as Deltaproteobacteria), 13% belong to the Nitrospira class, 10% to the Firmicutes class (3% Bacilli and 7% Clostridia class), 2% to the Actinobacteria class, 1% to the phylum Verrucomicrobia and Bacteroidetes, and the remaining 5% belong to minority classes. While 46% were unclassified bacterial sequences.
The sample ES_TI had 99.99% of obtained sequences belonging to the Bacteria kingdom, of which 50% were unclassified, 22% belong to the phylum of Proteobacteria (15% Gammaproteobacterias, 5% Deltaproteobacterias and the remaining 2% were distributed among the other classes, such as Betaproteobacteria), 17% belongs to the Nitrospira class, 5% to the Firmicutes class (3% Bacilli class and 2% Clostridia), 2% Bacteroidetes, 1% to the Actinobacteria classes and the remaining 3% belong to the minority classes.
In the water sample PT_CH, the major of sequences were associated to the realm of Bacteria (99.95%), of which 42% belongs to the phylum Proteobacteria (27% Betaproteobacterias, 13% Gammaproteobacterias, 1% Deltaproteobacterias and the remaining 1% is distributed among the other classes, as is the case of Alphaproteobacteria), 18% to the Deinococci class, 16% to the Aquificae class, 14% were not classified and the remaining 10% belong to the minority classes, such as Actinobacteria and Nitrospira classes (Figure 6).
In relation to the sample PT_SA (Figure 6), 99% of the sequences belongs to the realm of Bacteria, of these sequences, 78% were of the Proteobacteria phylum (74% Epsilonproteobacteria, 3% Betaproteobacterias and the remaining 1% was distributed by other classes, such as Gammaproteobacteria), 19% are unclassified and the remaining 2% are distributed by the minority phyla, such as the phylum Firmicutes. In the case of this water sample, in particular, it is verified that 1% of the total sequences obtained were classified as belonging to the realm of Archaea, of these sequences, 100% are of the Euryarchaeota phylum.
In all water samples of this study, the phylum Proteobacteria (22% to 78%) was the most abundant, following the phylum Deinococcus-Thermus (8% to 18%), Nitrospira (5% to 17%), Aquificae (2% to 16%) and Firmicutes (2% to 10%). In addition, the Proteobacteria phylum was clearly the most representative phyla in the following water samples PT_CH, ES_BUR and PT_SA, compressing the class Betaproteobacteria (ES_BUR and PT_CH), Epsilonproteobacteria (PT_SA and ES_PR) and the class Gammaproteobacteria (ES_BUR, ES_TI and PT_CH). The phyla and classes with a representation ≥10% are shown in Figure 7.
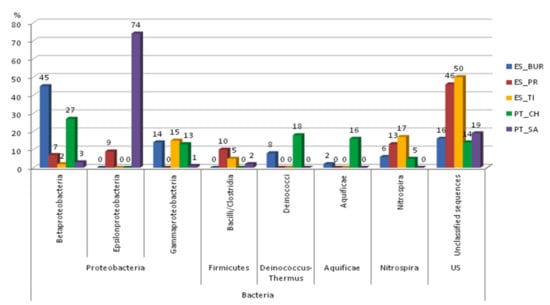
Figure 7.
Comparison of the most representative bacterial diversity communities at the phylum and class level (≥10% of the sequences) of the analyzed natural thermal mineral water samples in this study, including the unclassified bacterial sequences (US).
Proteobacteria have relevant biological importance since this phylum is mostly constituted by Gram-negative bacteria and these have a high medical, veterinary, industrial and agricultural interest [65,66]. In general, most Proteobacteria are mesophilic bacteria, that is, with optimal growth temperatures between 15 and 60 °C, however, there are some bacteria that may be thermophilic, as in the case of Thiomonas thermosulfata or psychrophiles, such as Polaromonas [65]. The possibility of these bacteria being present in both psychrophilic, mesophilic and thermophilic environments confirms our results obtained in Figure 7, where there is also the existence of Proteobacteria in water with temperatures between 15 and 70 °C.
Some studies have demonstrated that Proteobacteria represent an important component of the skin microbiota [13,40,67,68]. However, although this phylum has already been detected in several metagenomic studies, the microbiota of cutaneous Proteobacteria still remains little described [69]. The microbial communities of the human skin have strong capacities to interact with their environment and can thus establish a relationship between skin and environmental ecotypes in the same species of Proteobacteria [70]. Considering the various therapeutical properties associated with natural thermal mineral waters, such as psoriasis, acne, alopecia, eczema and (chronic) atopic dermatitis, among others [55]; some studies have demonstrated that the microbial composition of these waters may also play a greater role in the health benefits [21,27,28,71]. The One Health concept recognizes that human health is strongly linked to animal health and the environment [69].
In relation to the Firmicutes phylum in this study, the samples that presented a greater amount of sequences were ES_PR and ES_TI (Figure 5). This phylum has some interesting characteristics for scientific research, in particular by including some genera of aerobic or anaerobic bacteria that produce endospores. These endospores are responsible for conferring resistance to conditions of high temperatures and other environmental stresses [72]. The Firmicutes phylum also presents genera such as Staphylococcus, Enterococcus, Streptococcus, Clostridium, Bacillus and Lactobacillus, being these genera are quite used in biotechnological processes. The appearance of microorganisms of the Firmicutes phylum in thermal waters with temperatures around 50 °C, has been frequently reported worldwide [73,74,75].
The species belonging to the Deinococcus-Thermus phylum show a great biochemical, physiological and phenotypic diversity [76], being these bacteria able to survive high doses of ionizing radiation, vacuum, high temperatures, desiccation, hydrogen peroxide and several other agents capable of cause DNA damage [77,78]. The presence of microorganisms of this phylum in the samples of water with higher temperatures (PT_CH and ES_BUR) corroborates the obtained results by several authors [78,79].
The microorganisms belonging to the Aquificae phylum are Gram-negative, do not form spores and are strictly thermophilic with an ideal growth usually occurring above 65 °C [80,81]. This optimum growth temperature corroborates our results, in which the Aquificae phylum is mostly present in the water sample PT_CH (with a temperature of 70 °C). Regarding their metabolism, most of the microorganisms belonging to the Aquificae phylum are bacteria that can oxidize hydrogen, but alternatively, thiosulfate or sulfur can also be used as energy sources [80]. The thermal stability found in many of the enzymes belonging to the genus Aquifex and other thermophilic bacteria of the Aquificae phylum, are of particular interest in industrial and biotechnological applications [80,81].
The Nitrospira Phylum is composed of bacteria capable of oxidizing nitrite, being the most abundant and diverse group of bacteria that act on nitrification [82]. Some studies have demonstrated its wide distribution in natural habitats, in soils [83], oceans [84] and hot springs [85]. Members of the genus Nitrospira belong to the most interesting microorganisms for biotechnology [86].
4. Conclusions
This study demonstrated that the thermal waters of the Galician region of Spain and the northern region of Portugal could be clustered into four distinct groups based on their physical–chemical composition. The waters ES_PR and ES_TI were high content in the sulfur compounds (SO32− and S2−) and CO32−. For the sample ES_BUR, the variable that most contributed to its grouping was the pH. The sample PT_SA was rich in the cations (Mg, Ca and Sr) and the sample PT_CH was constituted by the nitrogen compounds (NO3− and NH4+), conductivity, dry residue, Na and Rb.
Based on the sequencing of the 16S rRNA gene, the water samples revealed a high diversity and a bacterial predominance of the Proteobacteria phylum (in water samples PT_CH, ES_BUR and PT_SA), followed by the phylum Firmicutes (in waters ES_PR and ES_TI), Deinococcus-Thermus (in waters PT_CH and ES_BUR), Aquificae (in waters ES_BUR and PT_CH) and Nitrospira (in waters PT_CH, ES_TI, ES_PR and ES_BUR); the abundance of Archaea was higher in water PT_SA (1%).
This innovative work allows for the first time to study the taxonomic diversity of microbial communities in the waters of the northern region of Portugal, and it also contributes to increasing the existing knowledge of the microbial diversity of the thermal waters of the Galician region. Although it was possible to identify most of the microbial composition of the studied waters used for therapeutic purposes; there are samples (ES_PR and ES_TI) that showed more than 40% of the obtained sequences as unclassified, still demonstrating an enormous gap in the knowledge of the microbial communities of these waters.
The study of these microorganisms that exist in these unique habitats was able to occur mainly due to the metagenomics and the new technologies of sequencing (NGS), allowing it to accede to the most diverse microbiomes. This knowledge has the possibility of leading to the discovery of new drugs, namely antibiotics, immunosuppressants, enzymes and new compounds of great importance for both medicine and biotechnology. Additionally, this work can contribute to increasing the safe use of these unique natural thermal mineral waters. The continuity of the work is essential, for instance analyzing the metabolic pathways of the microorganisms present in the waters and identifying how these metabolic pathways may be related to the diversity and specialization of the hydromineral richness. In addition, the elaboration of hydrogen OTEC will be allowed to group all hydrogenomes of the natural thermal minerals waters of the Iberian Peninsula. Finally, correlate the physical–chemical composition of the waters with the diverse microbial groups found in these habitats is essential to amplify the knowledge of these thermal waters.
Author Contributions
Methodology, P.P.-F., D.G.-P. and G.I.; validation, P.P., J.C., F.F.-R., A.T.-A., M.L.R. and G.I.; investigation, P.P.-F. and G.I.; resources, P.P.-F., M.L.R. and G.I.; draft of the manuscript, P.P.-F., C.M. and G.I.; data analysis and supervision, P.P., J.C., F.F.-R., A.T.-A., M.L.R. and G.I.; funding acquisition, M.L.R.R. and G.I. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financial supported by the Associate Laboratory for Green Chemistry—LAQV, which is financed by national funds from FCT/MCTES (UIDB/50006/2020 and UIDP/50006/2020).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
This project was supported by the European Union Seventh Framework Programme (FP7/2007–2013) under grant agreement No. 324439. D.G.-P. and F.F.-R. were supported by the Consellería de Educación, Universidades e Formación Profesional (Xunta de Galicia) under the scope of the strategic funding of ED431C2018/55-GRC Competitive Reference Group and the “Centro singular de investigación de Galicia” (accreditation 2019–2022) funded by the European Regional Development Fund (ERDF)-Ref. ED431G2019/06. We also acknowledge the support of Xunta de Galicia, Spain (CITACA Strategic Partnership, Reference: ED431E 2018/07) and Project PRODIXOS funded by the Spanish Ministry of Science and Innovation (Project RTI2018-099249-B-I00). The authors are grateful for the financial support provided by Project CAREBIO2—Comparative assessment of antimicrobial resistance in environmental biofilms through proteomics—toward innovative theranostic biomarkers, with reference NORTE-01-0145FEDER-030101 and PTDC/SAU-INF/ 30101/2017, financed by the European Regional Development Fund (ERDF) through the Northern Regional Operational Program (NORTE 2020) and the Foundation for Science and Technology (FCT). P.P.-F. was supported by Erasmus grant financed by the European Commission’s Erasmus + program.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lozán, J.L.; Meyer, S.; Karbe, L. Water As the Basis of Life. In Global Change: Enough Water for All? Lozán, J.L., Grassl, H., Hupfer, L., Menzel, L., Schönwiese, C.-D., Eds.; Wissenschaftliche Auswertungen: Hamburg, Germany, 2007. [Google Scholar]
- Chaplin, M. Do we underestimate the importance of water in cell biology? Nat. Rev. Mol. Cell Biol. 2006, 7, 861–866. [Google Scholar] [CrossRef]
- Chaplin, M.F. Water: Its importance to life. Biochem. Mol. Biol. Educ. 2001, 29, 54–59. [Google Scholar] [CrossRef]
- Bacci, D.C.; Pataca, E.M. Educação para a água. Estud. Avançados 2008, 22, 211–226. Available online: http://www.scielo.br/pdf/ea/v22n63/v22n63a14.pdf (accessed on 15 March 2018). [CrossRef]
- Lund, J.W. Balneological Use of Thermal Waters. 1998. Available online: http://www.sld.cu/galerias/pdf/sitios/rehabilitacion-bal/lundwj2.pdf (accessed on 15 March 2018).
- Quintela, M.M. Saberes e práticas termais: Uma perspectiva comparada em Portugal (Termas de S. Pedro do Sul) e no Brasil (Caldas da Imperatriz). História Ciências Saúde-Manguinhos 2004, 11 (Suppl. 1), 239–260. [Google Scholar] [CrossRef]
- Oliveira, A.S.; Almeida, S.M.S.; Reis, A.R.; Alencoão, A.M.P.; Sousa, L.M.O.; Lourenço, J.M.M. Águas Minerais de Trás-os-Montes e Alto Douro (NE de Portugal): Sistematização e Aproveitamentos . In Para Conhecer a Terra: Memórias e Notícias de Geociências no Espaço Lusófono; Lopes, F.C., Andrade, A.I., Henriques, M.H., Quinta-Ferreira, M., Barata, M.T., Pena dos Reis, R., Eds.; Imprensa daniversidade de Coimbra: Coimbra, Portugal, 2012; pp. 409–420. [Google Scholar]
- Ramos, A.R.C.C.V. O Termalismo em Portugal: Dos Factores de Obstrução à Revitalização pela Dimensão Turística. Ph.D. Thesis, Universidade de Aveiro, Aveiro, Portugal, 2005. [Google Scholar]
- Gianfaldoni, S.; Tchernev, G.; Wollina, U.; Roccia, M.G.; Fioranelli, M.; Gianfaldoni, R.; Lotti, T. History of the baths and thermal medicine. Open Access Maced. J. Med. Sci. 2017, 5, 566–568. [Google Scholar] [CrossRef]
- Silva, A.; Oliveira, A.S.; Vaz, C.V.; Correia, S.; Ferreira, R.; Breitenfeld, L.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, R.; Pereira, C.M.F.; Palmeira-de-Oliveira, A.; et al. Anti-inflammatory potential of Portuguese thermal waters. Sci. Rep. 2020, 10, 22313. [Google Scholar] [CrossRef]
- Directive 2009/54/EC of the European Parliament and of the Council. Off. J. Eur. Union 2009, 164, 45–58.
- Pagourelias, E.D.; Zorou, P.G.; Tsaligopoulos, M.; Athyros, V.G.; Karagiannis, A.; Efthimiadis, G.K. Carbon dioxide balneotherapy and cardiovascular disease. Int. J. Biometeorol. 2011, 55, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, H.E.; Bhatia, N.D.; Friedman, A.; Eng, R.M.; Seité, S. The role of cutaneous microbiota harmony in maintaining a functional skin barrier. J. Drugs Dermatol. 2017, 16, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Nunes, F.; Rodrigues, M.; Ribeiro, M.P.; Ugazio, E.; Cavalli, R.; Abollino, O.; Coutinho, P.; Araujo, A.R.T.S. Incorporation of Cró thermal water in a dermocosmetic formulation: Cytotoxicity effects, characterization and stability studies and efficacy evaluation. Int. J. Cosmet. Sci. 2019, 41, 604–612. [Google Scholar] [CrossRef]
- Oliveira, A.S.; Vaz, C.V.; Silva, A.; Correia, S.; Ferreira, R.; Breitenfeld, L.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, R.; Pereira, C.; Cruz, M.T.; et al. In vitro evaluation of potential benefits of a silica-rich thermal water (Monfortinho Thermal Water) in hyperkeratotic skin conditions. Int. J. Biometeorol. 2020, 64, 1957–1968. [Google Scholar] [CrossRef]
- Viegas, J.; Esteves, A.F.; Cardoso, E.M.; Arosa, F.A.; Vitale, M.; Taborda-Barata, L. Biological Effects of Thermal Water-Associated Hydrogen Sulfide on Human Airways and Associated Immune Cells: Implications for Respiratory Diseases. Front. Public Health 2019, 7, 128. [Google Scholar] [CrossRef] [PubMed]
- Khaltaev, N.; Solimene, U.; Vitale, F.; Zanasi, A. Balneotherapy and hydrotherapy in chronic respiratory disease. J. Thorac. Dis. 2020, 12, 4459–4468. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Zarkadoulia, E.; Rafailidis, P.I. The therapeutic effect of balneotherapy: Evaluation of the evidence from randomised controlled trials. Int. J. Clin. Pract. 2009, 63, 1068–1084. [Google Scholar] [CrossRef]
- Karagülle, M.; Karagülle, M.Z. Effectiveness of balneotherapy and spa therapy for the treatment of chronic low back pain: A review on latest evidence. Clin. Rheumatol. 2015, 34, 207–214. [Google Scholar] [CrossRef]
- Varzaityte, L.; Kubilius, R.; Rapoliene, L.; Bartuseviciute, R.; Balcius, A.; Ramanauskas, K.; Nedzelskiene, I. The effect of balneotherapy and peloid therapy on changes in the functional state of patients with knee joint osteoarthritis: A randomized, controlled, single-blind pilot study. Int. J. Biometeorol. 2020, 64, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Paduano, S.; Valeriani, F.; Romano-Spica, V.; Bargellini, A.; Borella, P.; Marchesi, I. Microbial biodiversity of thermal water and mud in an Italian spa by metagenomics: A pilot study. Water Sci. Technol. Water Supply 2018, 18, 1456–1465. [Google Scholar] [CrossRef]
- Valeriani, F.; Gianfranceschi, G.; Romano Spica, V. The microbiota as a candidate biomarker for SPA pools and SPA thermal spring stability after seismic events. Environ. Int. 2020, 137, 105595. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, L.J.; Mancinelli, R.L. Life in extreme environments. Nature. 2001, 409, 1092–1101. [Google Scholar] [CrossRef]
- Rampelotto, P.H. Extremophiles and Extreme Environments. Life 2013, 3, 482–485. [Google Scholar] [CrossRef]
- Raddadi, N.; Cherif, A.; Daffonchio, D.; Neifar, M.; Fava, F. Biotechnological applications of extremophiles, extremozymes and extremolytes. Appl. Microbiol. Biotechnol. 2015, 99, 7907–7913. [Google Scholar] [CrossRef]
- Mirete, S.; Morgante, V.; González-Pastor, J.E. Functional metagenomics of extreme environments. Curr. Opin. Biotechnol. 2016, 38, 143–149. [Google Scholar] [CrossRef]
- Nicoletti, G.; Corbella, M.; Jaber, O.; Marone, P.; Scevola, D.; Faga, A. Non-pathogenic microflora of a spring water with regenerative properties. Biomed. Rep. 2015, 3, 758–762. [Google Scholar] [CrossRef]
- Seité, S.; Flores, G.E.; Henley, J.B.; Sarrazin, P.; Ainouche, R.; Fierer, N.; Martin, R. Skin microbiome in patients with psoriasis before and after balneotherapy at the thermal care center of La Roche-Posay. J. Am. Acad. Dermatol. 2016, 74, AB276. [Google Scholar]
- Aburto-Medina, A.; Shahsavari, E.; Cohen, M.; Mantri, N.; Ball, A.S. Analysis of the Microbiome (Bathing Biome) in Geothermal Waters from an Australian Balneotherapy Centre. Water 2020, 12, 1705. [Google Scholar] [CrossRef]
- Handelsman, J. Metagenomics: Application of genomics to uncultured microorganisms. Microbiol. Mol. Biol. Rev. 2004, 68, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S. Águas minerais naturais de Trás-os-Montes e Alto Douro: Património legado pela natureza. Bol. Cult. Esc. Secundária Camilo Castelo Branco 2011, 17, 18–29. [Google Scholar]
- Oliveira, A.S. Águas subterrâneas do setor norte de Portugal continental: Enquadramentos geográfico e geológico, tipologias e aproveitamentos. Bol. Soc. Española Hidrol. Med. 2015, 30, 57–71. [Google Scholar] [CrossRef][Green Version]
- Meijide-Faílde, R.; Juncosa, R.; Delgado, J. Caracterización Hidrogeoquímica y Terapéutica de las Aguas Mineromedicinales y Minerales Naturales de Galicia. In Proceedings of the Libro de Actas del I Congreso Internacional del Agua “Termalismo y Calidad de Vida”, Ourense, Spain, 23–24 September 2015; pp. 61–72. [Google Scholar]
- Meijide-Faílde, R.; Leira, M.; Torres, E.; Rodríguez, M.C.L.; Carballeira, R. Estudio del componente biológico de las aguas mineromedicinales y termales de Ourense: Burgas y Outariz. In Proceedings of the Libro de Actas del I Congreso Internacional del Agua “Termalismo y Calidad de Vida”, Ourense, Spain, 23–24 September 2015; pp. 519–524. [Google Scholar]
- Michard, G.; Beaucaire, C. Les eaux thermales des granites de Galice (Espagne): Deseaux carbogazeuses aux eaux alcalines (Thermal waters from granites of Galicia (Spain): From CO2-rich to high-pH waters. Chem. Geol. 1993, 110, 345–360. [Google Scholar] [CrossRef]
- Ramírez Ortega, A. Revisión geoquímica de las aguas minero-medicinales de Galicia. An. Real Acad. Farm. 2001, 76, 1–45. [Google Scholar]
- Maraver, F. Vademecun de Aguas Mineromedicinales Españolas; Instituto de Salud Carlos III—Universidad Complutense de Madrid: Madrid, Spain, 2002. [Google Scholar]
- Pedro, A.; Cantista, P. O termalismo em Portugal. An. Hidrol. Med. 2010, 3, 79–107. [Google Scholar]
- Oliveira, A.S. Hidrogeologia dos Sistemas Gasocarbónicos da Província Hidromineral Transmontana: Ribeirinha (Mirandela), Sandim (Vinhais), Segirei e Salgadela (Chaves). Ph.D. Thesis, Universidade de Trás-os-Montes e Alto Douro, Vila Real, Spain, 2001. [Google Scholar]
- Machado, J.M.G.S. Ocorrências Hidrominerais do NE de Portugal Continental: Inventariação, Sistematização e Aproveitamentos didácticos. Master’s Thesis, Universidade de Trás-os-Montes e Alto Douro, Vila Real, Spain, 2009. [Google Scholar]
- Pacheco, F.A.L. Hidrogeologia em Maciços de Rochas Cristalinas (Morais-Chacim-Macedo de Cavaleiros). Bases Para a Gestão Integrada dos Recursos Hídricos da Região. Ph.D. Thesis, Universidade de Trás-os-Montes e Alto Douro, Vila Real, Spain, 2000. [Google Scholar]
- Piper, A.M. A graphic procedure in the geochemical interpretation of water analysis. Am. Geophys. Union Trans. 1944, 25, 914–923. [Google Scholar] [CrossRef]
- Back, W.; Hanshaw, B.B. Chemical geohydrology. Adv. Hydrosci. 1965, 2, 49–109. [Google Scholar]
- Malan, G. XLSTAT 2017: Data Analysis and Statistical Solution for Microsoft Excel; Addinsoft: Paris, France, 2017. [Google Scholar]
- Vieira, A.F.C.S. Aplicação de Métodos Quimiométricos aos Resultados de Técnicas Hifenadas Para Caracterização da Aguardente de Cana-de-Açúcar de Cabo Verde. Master’s Thesis, Universidade de Aveiro, Aveiro, Portugal, 2007. [Google Scholar]
- Thijs, S.; De Beeck, M.O.; Beckers, B.; Truyens, S.; Stevens, V.; Van Hamme, J.D.; Weyens, N.; Vangronsveld, J. Comparative evaluation of four bacteria-specific primer pairs for 16S rRNA gene surveys. Front. Microbiol. 2017, 8, 494. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Smith, D.P.; Peay, K.G. Sequence depth, not PCR replication, improves ecological inference from Next Generation DNA Sequencing. PLoS ONE 2014, 9, e90234. [Google Scholar] [CrossRef]
- Ison, S.A.; Delannoy, S.; Bugarel, M.; Nagaraja, T.G.; Renter, D.G.; den Bakker, H.C.; Nightingale, K.K.; Fach, P.; Loneragan, G.H. Targeted amplicon sequencing for SNP genotyping of attaching and effacing Escherichia coli O26: H11 cattle strains using a high-throughput library preparation technique. Appl. Environ. Microbiol. 2015, 82, 640–649. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc (accessed on 12 September 2018).
- Wilke, A.; Gerlach, W.; Harrison, T.; Paczian, T.; Trimble, W.L.; Meyer, F. MG-RAST Manual for Version 4, Revision 3. 2017. Available online: http://ftp.metagenomics.anl.gov/data/manual/mg-rast-manual.pdf (accessed on 12 September 2018).
- Leggett, R.M.; Ramirez-Gonzalez, R.H.; Clavijo, B.J.; Waite, D.; Davey, R.P. Sequencing quality assessment tools to enable data-driven informatics for high throughput genomics. Front. Genet. 2013, 4, 288. [Google Scholar] [CrossRef]
- Marques, J.M.V.V.B. Geoquimica dos Fluidos e da Interacção Água-Rocha: Os Casos das Águas Mineralizadas Quentes e Frias de Chaves, Vilarelho da Raia, Vidago e Pedras Salgadas. Ph.D. Thesis, Universidade Técnica de Lisboa, Instituto Superior Técnico, Lisboa, Portugal, 1999. [Google Scholar]
- Branco, L.; Águas Minerais Naturais e Águas de Nascente. Associação Portuguesa dos Industriais de Águas Minerais Naturais e de Nascente. 2015. Available online: https://www.apiam.pt/images/newsconteudo/ficheiro1/158_LIVROBRANCO2015.pdf (accessed on 12 September 2018).
- Matz, H.; Orion, E.; Wolf, R. Balneotherapy in dermatology. Dermat. Ther. 2003, 16, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Browne, P.D.; Nielsen, T.K.; Kot, W.; Aggerholm, A.; Gilbert, M.T.P.; Puetz, L.; Rasmussen, M.; Zervas, A.; Hansen, L.H. GC bias affects genomic and metagenomic reconstructions, underrepresenting GC-poor organisms. Gigascience 2020, 9, giaa008. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.; Wu, L.; Qin, Y.; Van Nostrand, J.D.; Ning, D.; Sun, B.; Zhou, J. Evaluation of the reproducibility of amplicon sequencing with Illumina MiSeq platform. PLoS ONE 2017, 12, e0176716. [Google Scholar] [CrossRef]
- Fuciños González, P. Um Estúdio Integrado Das Lipasas de Thermus Thermophilus HB27: Condicións de Producción, Localización Celular, Purificación e Caracterización. Ph.D. Tesis, Universidad de Vigo, Vigo, Spain, 2007. [Google Scholar]
- Everroad, R.C.; Otaki, H.; Matsuura, K.; Haruta, S. Diversification of bacterial community composition along a temperature gradient at a thermal spring. Microbes Environ. 2012, 27, 374–381. [Google Scholar] [CrossRef]
- Lewin, A.; Wentzel, A.; Valla, S. Metagenomics of microbial life in extreme temperature environments. Curr. Opin. Biotechnol. 2013, 24, 516–525. [Google Scholar] [CrossRef]
- Stetter, K.O. Extremophiles and their adaptation to hot environments. FEBS Lett. 1999, 452, 22–25. [Google Scholar] [CrossRef]
- Wang, S.; Hou, W.; Dong, H.; Jiang, H.; Huang, L.; Wu, G.; Zhang, L. Control of temperature on microbial community structure in hot springs of the tibetan plateau. PLoS ONE 2013, 8, e62901. [Google Scholar] [CrossRef]
- Sharp, C.E.; Brady, A.L.; Sharp, G.H.; Grasby, S.E.; Stott, M.B.; Dunfield, P.F. Humboldt’s spa: Microbial diversity is controlled by temperature in geothermal environments. ISME J. 2014, 8, 1166–1174. [Google Scholar] [CrossRef] [PubMed]
- Ondov, B.D.; Bergman, N.H.; Phillippy, A.M. Interactive metagenomic visualization in a Web browser. BMC Bioinform. 2011, 12, 385. [Google Scholar] [CrossRef] [PubMed]
- Kersters, K.; De Vos, P.; Gillis, M.; Swings, J.; Vandamme, P.; Stackebrandt, E. Introduction to the Proteobacteria. In The Prokaryotes, 3rd ed.; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 3–37. [Google Scholar]
- Madigan, M.T.; Martinko, J.M. Brock Biology of Microorganisms, 11th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2006. [Google Scholar]
- HMPC. Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef]
- Zeeuwen, P.L.; Boekhorst, J.; van den Bogaard, E.H.; de Koning, H.D.; van de Kerkhof, P.M.; Saulnier, D.M.; van Swam, I.I.; van Hijum, S.A.; Kleerebezem, M.; Schalkwijk, J.; et al. Microbiome dynamics of huma nepidermis folowing skin barrier disruption. Genome Biol. 2012, 13, R101. [Google Scholar] [CrossRef] [PubMed]
- Cosseau, C.; Romano-Bertrand, S.; Duplan, H.; Lucas, O.; Ingrassia, I.; Ingrassia, I.; Roques, C.; Jumas-Bilak, E. Proteobacteria from the human skin microbiota: Species-level diversity and hypotheses. One Health 2016, 2, 33–41. [Google Scholar] [CrossRef]
- Mathieu, A.; Delmont, T.O.; Vogel, T.M.; Robe, P.; Nalin, R.; Simonet, P. Life on human sur-faces: Skin metagenomics. PLoS ONE 2013, 8, e65288. [Google Scholar] [CrossRef]
- Nakatsuji, T.; Gallo, R.L. Dermatological therapy by topical application of nonpathogenic bacteria. J. Investig. Dermatol. 2014, 134, 11–14. [Google Scholar] [CrossRef]
- Madigan, M.T.; Martinko, J.M.; Bender, K.S.; Buckley, D.H.; Stahl, D.A. Microbiologia de Brock, 14th ed.; Artmed: São Paulo, Brazil, 2016. [Google Scholar]
- Mangrola, A.; Dudhagara, P.; Koringa, P.; Joshi, C.G.; Parmar, M.; Patel, R. Deciphering the microbiota of Tuwa hot spring, India using shotgun metagenomic sequencing approach. Genom. Data 2015, 4, 153–155. [Google Scholar] [CrossRef]
- Panda, A.K.; Bisht, S.S.; De Mandal, S.; Kumar, N.S. Bacterial and archeal community composition in hot springs from Indo-Burma region, North-east India. AMB Express 2016, 6, 111. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, B.; Chowdhury, T.; Chattopadhyay, B. Comparative analysis of microbial diversity in two hot springs of Bakreshwar, West Bengal, India. Genom. Data 2017, 12, 122–129. [Google Scholar] [CrossRef]
- Garrity, G.M.; Holt, J.G. Phylum BIV. “Deinococcus–Thermus”. In Bergey’s Manual of Systematic Bacteriology, 2nd ed.; Boone, D., Castenholz, R., Eds.; Springer: New York, NY, USA, 2001; Volume 1, pp. 395–420. [Google Scholar]
- Zahradka, K.; Slade, D.; Bailone, A.; Sommer, S.; Averbeck, D.; Petranovic, M.; Lindner, A.B.; Radman, M. Reassembly of shattered chromosomes in Deinococcus radiodurans. Nature 2006, 443, 569–573. [Google Scholar] [CrossRef]
- Theodorakopoulos, N.; Bachar, D.; Christen, R.; Alain, K.; Chapon, V. Exploration of Deinococcus-Thermus molecular diversity by novel group-specific PCR primers. Microbiol. Open 2013, 2, 862–872. [Google Scholar]
- Sayeh, R.; Birrien, J.L.; Alain, K.; Barbier, G.; Hamdi, M.; Prieur, D. Microbial diversity in Tunisian geothermal springs as detected by molecular and culture-based approaches. Extremophiles 2010, 14, 501–514. [Google Scholar] [CrossRef]
- Huber, R.; Eder, W. Aquificales. In The Prokaryotes: An Evolving Electronic Resource for the Microbiological Community; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Griffiths, E.; Gupta, R.S. Molecular signatures in protein sequences that are characteristics of the phylum Aquificae. Int. J. Syst. Evol. Microbiol. 2006, 56, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Daims, H.; Nielsen, J.L.; Nielsen, P.H.; Schleifer, K.H.; Wagner, M. In situ characterization of Nitrospira-like nitrite-oxidizing bacteria active in wastewater treatment plants. Appl. Environ. Microbiol. 2001, 67, 5273–5284. [Google Scholar] [CrossRef]
- Freitag, T.E.; Chang, L.; Clegg, C.D.; Prosser, J.I. Influence of inorganic nitrogen management regime on the diversity of nitrite-oxidizing bacteria in agricultural grassland soils. Appl. Environ. Microbiol. 2005, 71, 8323–8334. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.W.; Bock, E.; Valois, F.W.; Waterbury, J.B.; Schlosser, U. Nitrospira marina gen. nov. sp. nov.: A chemolithotrophic nitrite-oxidizing bacterium. Arch. Microbiol. 1986, 144, 1–7. [Google Scholar] [CrossRef]
- Lebedeva, E.V.; Alawi, M.; Fiencke, C.; Namsaraev, B.; Bock, E.; Spieck, E. Moderately thermophilic nitrifying bacteria from a hot spring of the Baikal rift zone. FEMS Microbiol. Ecol. 2005, 54, 297–306. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lucker, S.; Wagner, M.; Maixner, F.; Pelletier, E.; Koch, H.; Vacherie, B.; Daims, H. A Nitrospira metagenome illuminates the physiology and evolution of globally important nitrite-oxidizing bacteria. Proc. Natl. Acad. Sci. USA 2010, 107, 13479–13484. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).