Leaf vs. Whole-Plant Biotic Attack: Does Vine Physiological Response Change?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design and Plant Material
2.2. Physiological Measurements
2.3. Sampling and Biomass Measurements
2.4. Nonstructural Carbohydrates (NSC) Analyses
2.5. Statistics
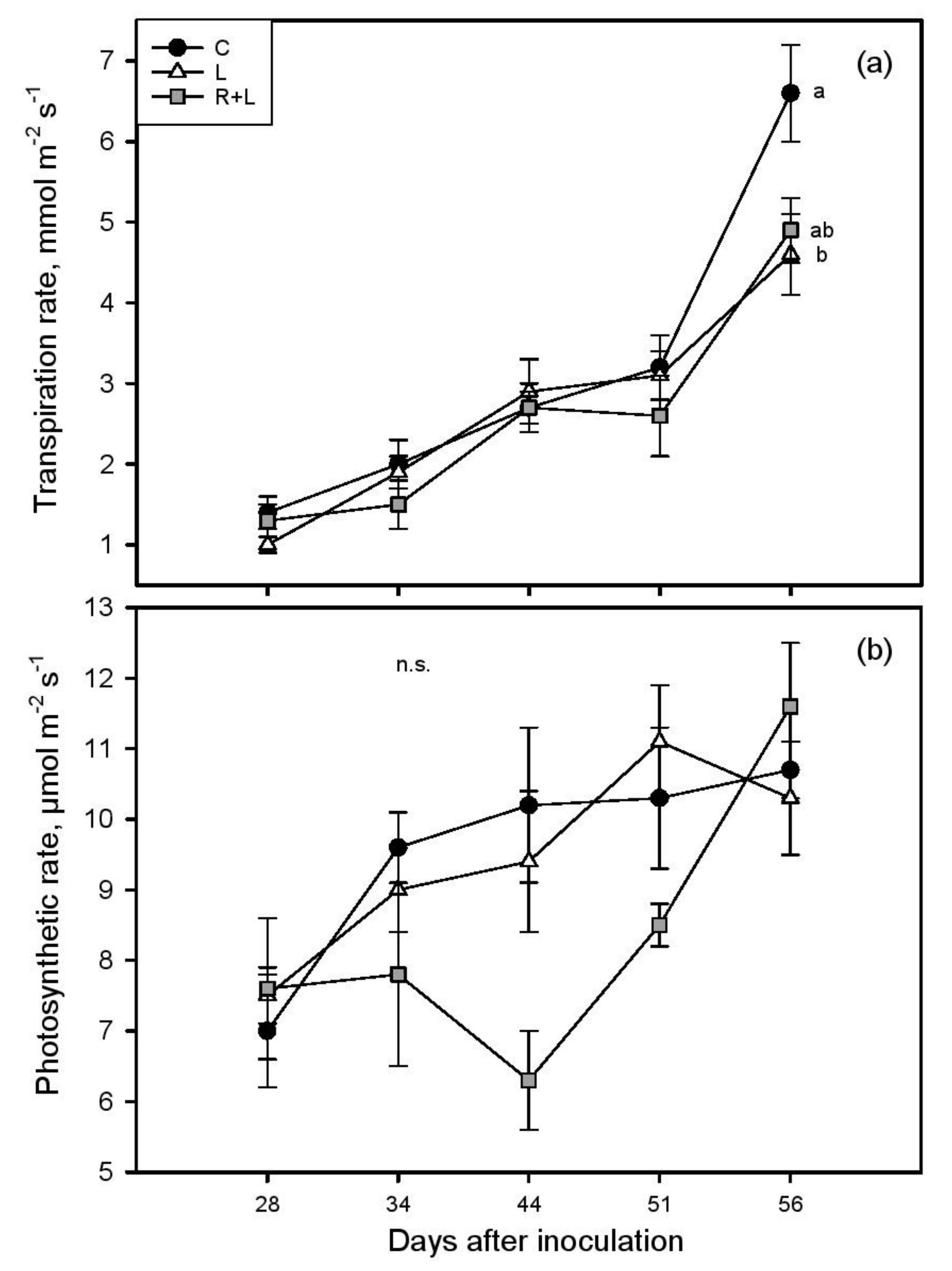
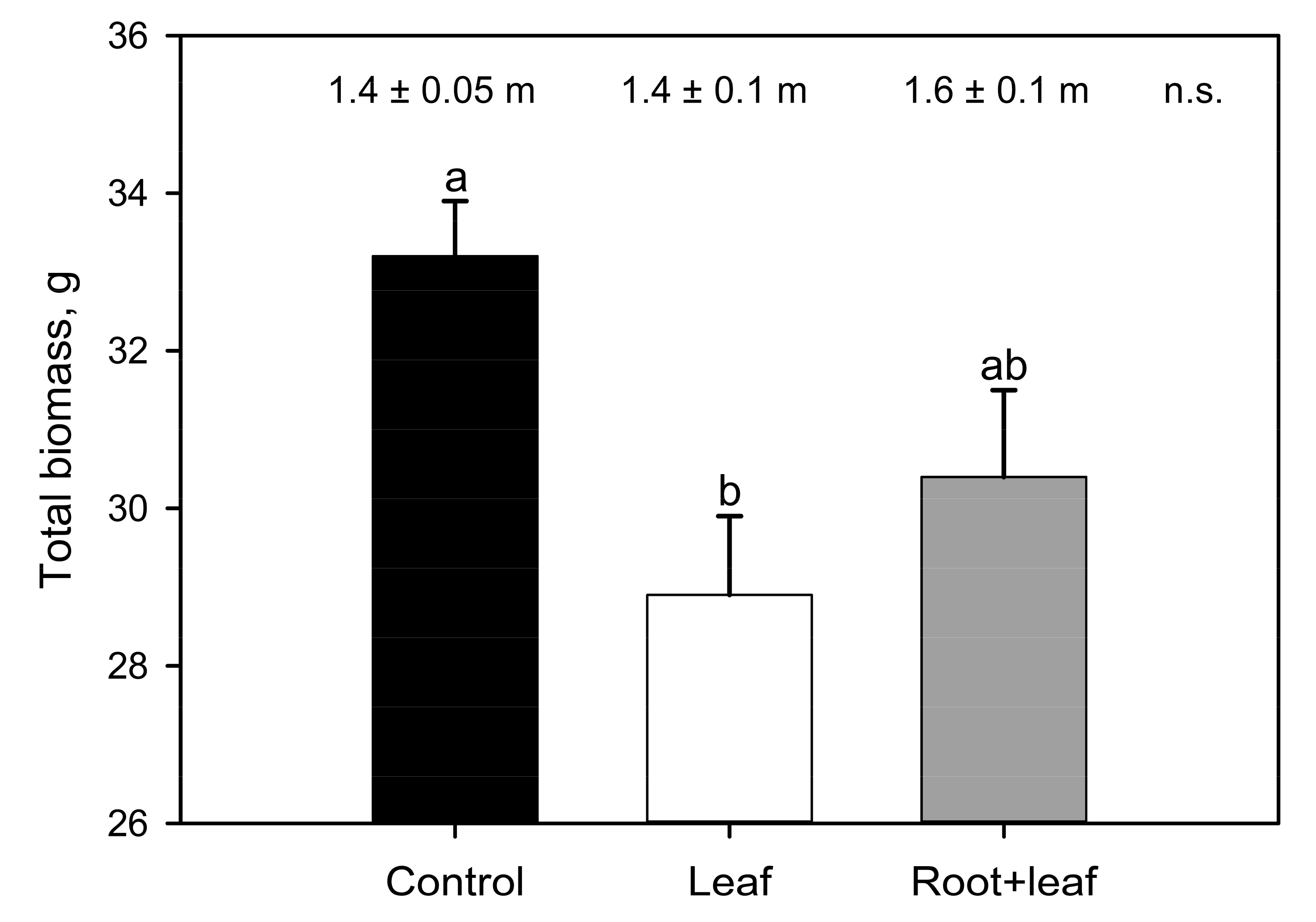
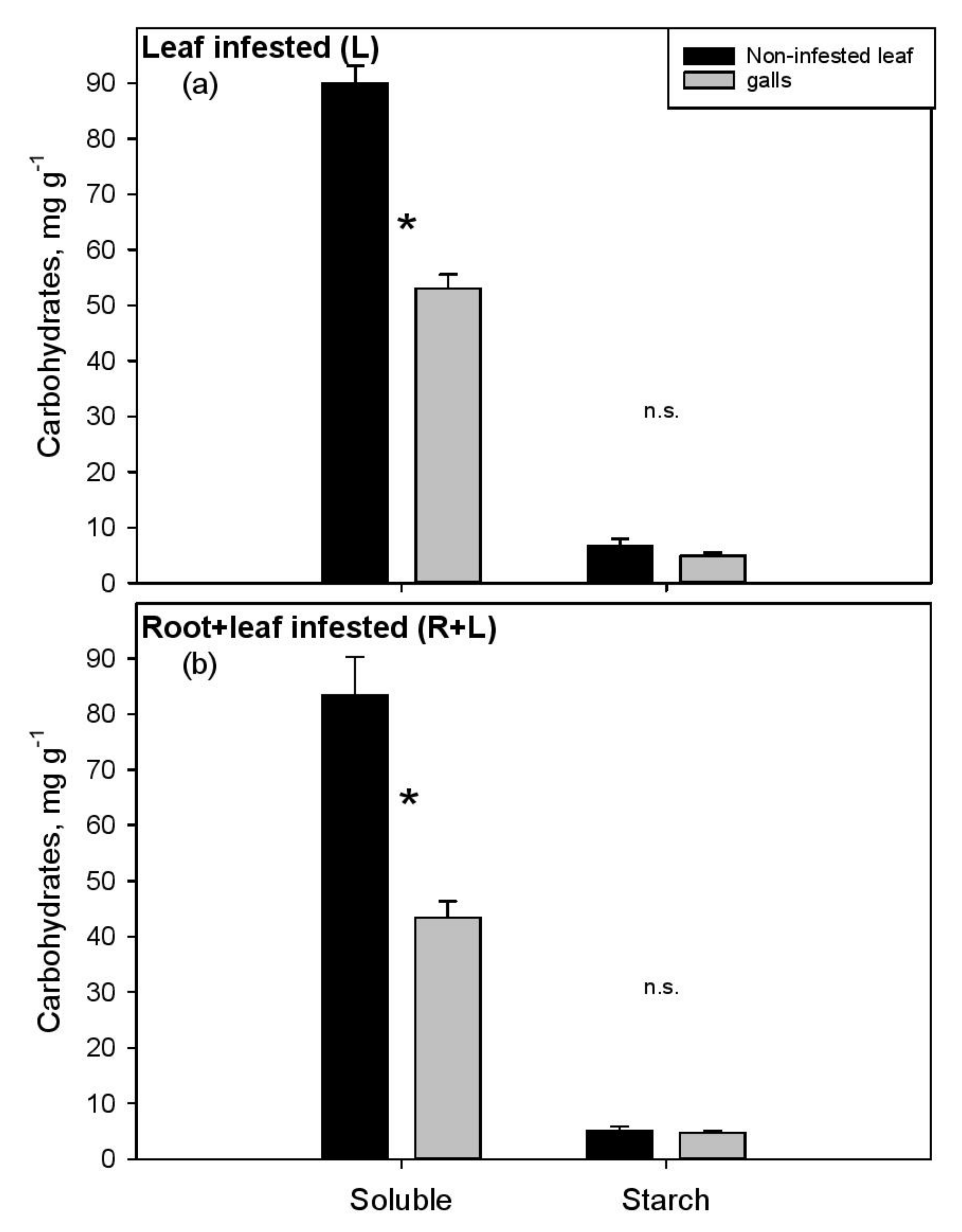
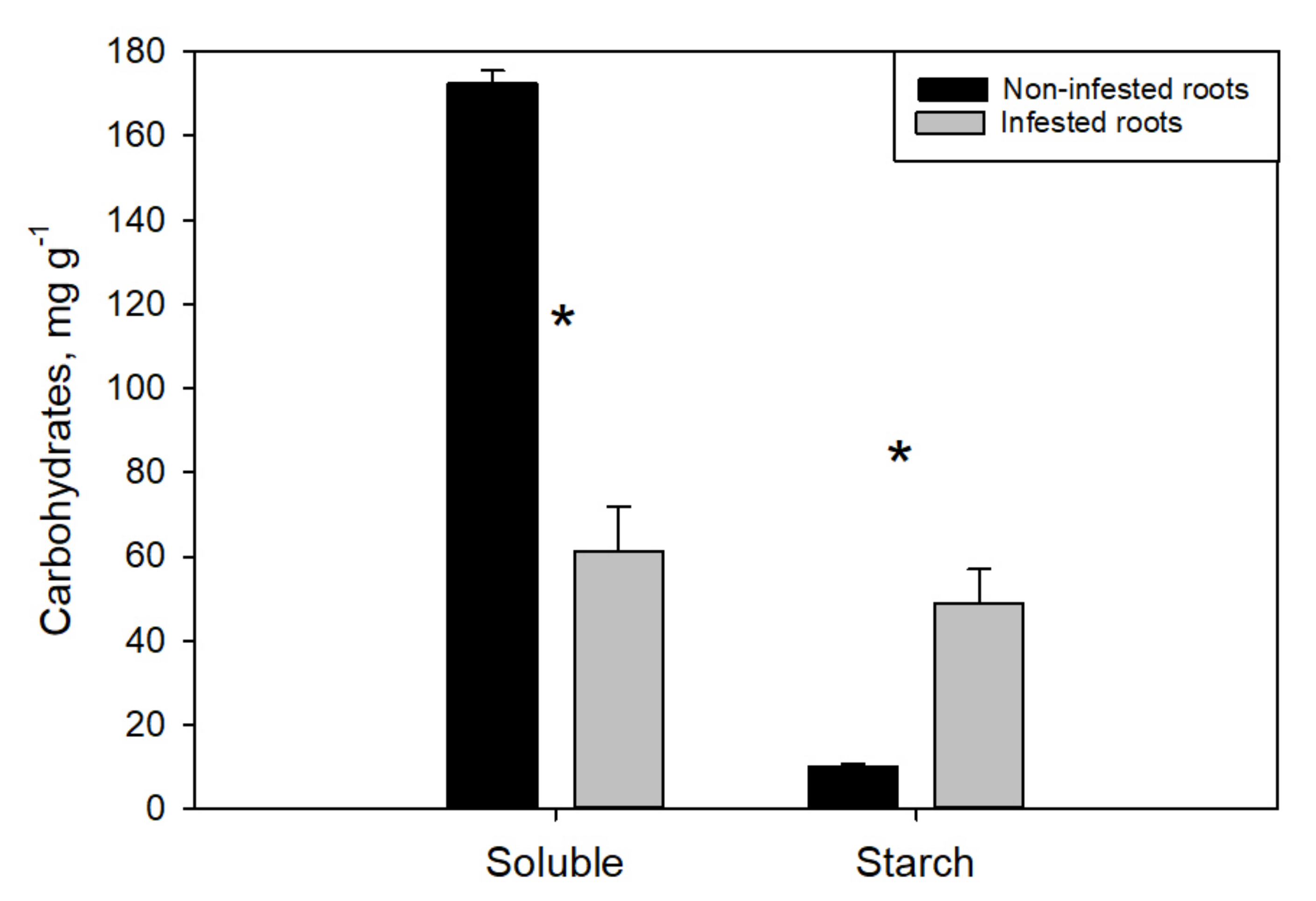
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Powell, K.S.; Cooper, P.D.; Forneck, A. The biology, physiology and host–plant interactions of grape phylloxera Daktulosphaira vitifoliae. Adv. Insect Phys. 2013, 45, 159–218. [Google Scholar] [CrossRef]
- Forneck, A.; Powell, K.S.; Walker, M.A. Scientific opinion: Improving the definition of grape phylloxera biotypes and standardizing biotype screening protocols. Am. J. Enol. Vitic. 2016, 67, 371–376. [Google Scholar] [CrossRef] [Green Version]
- Du, Y.; Wang, Z.; Yang, Y.; Zhao, Q.; Zhai, H.; Wang, Z. Nodosity formation and nutrition consumption in grape cultivars with different phylloxera-resistance and infested by grape phylloxera. Acta Entomol. Sin. 2008, 51, 1050–1054. [Google Scholar]
- Korosi, G.A.; Carmody, B.M.; Powell, K.S. Rootstock screening for phylloxera resistance under controlled conditions using selected phylloxera clonal lineages. Acta Hortic. 2011, 904, 33–40. [Google Scholar] [CrossRef]
- Savi, T.; García González, A.; Herrera, J.C.; Forneck, A. Gas exchange, biomass and non-structural carbohydrates dynamics in vines under combined drought and biotic stress. BMC Plant Biol. 2019, 19, 1–11. [Google Scholar] [CrossRef]
- Granett, J.; Walker, M.A.; Kocsis, L.; Omer, A.D. Biology and management of grape phylloxera. Annu. Rev. Èntomol. 2001, 46, 387–412. [Google Scholar] [CrossRef]
- Ruehl, E.; Schmid, J.; Eibach, R.; Töpfer, R. Grapevine breeding programmes in Germany. In Grapevine Breeding Programs for the Wine Industry; Woodhead Publishing: Cambridge, UK, 2015; Volume 268, pp. 77–101. [Google Scholar] [CrossRef]
- Fahrentrapp, J.; Müller, L.; Schumacher, P. Is there need for leaf-galling grape phylloxera control? Presence and distribution of Dactulosphaira vitifoliae in Swiss vineyards. Int. J. Pest Manag. 2015, 61, 1–6. [Google Scholar] [CrossRef]
- Mori, N.; Marchesini, E.; Duso, C.; Forneck, A. Nuove infestazioni di fillossera su vite europea: Tra cambiamenti climatici e nuovi biotipi. Inf. Agrar. 2018, 3, 47–52. [Google Scholar]
- Forneck, A.; MammeR + Ler, R.; Tello, J.; Breuer, M.; Müller, J.; Fahrentrapp, J. First European leaf-feeding grape phylloxera (Daktulosphaira vitifoliae Fitch) survey in Swiss and German commercial vineyards. Eur. J. Plant Pathol. 2019, 154, 1029–1039. [Google Scholar] [CrossRef] [Green Version]
- Kellow, A.V.; Sedgley, M.; Van Heeswijck, R. Interaction Between Vitis vinifera and grape phylloxera: Changes in root tissue during nodosity formation. Ann. Bot. 2004, 93, 581–590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, Y.; Wang, F.; Ji, X.; Jiang, E.; Zhai, H. Phylloxera infestation and the uptake and distribution of 13C and 15N tracers in grape vines. Vitis 2014, 53, 227–231. [Google Scholar]
- Griesser, M.; Lawo, N.C.; Crespo Martinez, S.; Schoedl-Hummel, K.; Wieczorek, K.; Gorecka, M.; Liebner, F.; Zweckmair, T.; Pavese, N.S.; Kreil, D.; et al. Phylloxera (Daktulosphaira vitifoliae Fitch) alters the carbohydrate metabolism in root galls to allowing the compatible interaction with grapevine (Vitis ssp.) roots. Plant Sci. 2015, 234, 38–49. [Google Scholar] [CrossRef] [Green Version]
- Cadle-Davidson, L. Variation within and between Vitis spp. for foliar Resistance to the downy mildew pathogen Plasmopara viticola. Plant Dis. 2008, 92, 1577–1584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cadle-Davidson, L.; Chicoine, D.R.; Consolie, N.H. Variation within and among Vitis spp. for foliar resistance to the powdery mildew pathogen Erysiphe necator. Plant Dis. 2011, 95, 202–211. [Google Scholar] [CrossRef] [Green Version]
- Vezzulli, S.; Vecchione, A.; Stefanini, M.; Zulini, L. Downy mildew resistance evaluation in 28 grapevine hybrids promising for breeding programs in Trentino region (Italy). Eur. J. Plant Pathol. 2017, 150, 485–495. [Google Scholar] [CrossRef]
- Granett, J.; Kocsis, L. Populations of grape phylloxera gallicoles on rootstock foliage in Hungary. Vitis 2000, 39, 37–41. [Google Scholar] [CrossRef]
- Nabity, P.D.; Haus, M.J.; Berenbaum, M.R.; DeLucia, E.H. Leaf-galling phylloxera on grapes reprograms host metabolism and morphology. Proc. Natl. Acad. Sci. 2013, 110, 16663–16668. [Google Scholar] [CrossRef] [Green Version]
- Savi, T.; Petruzzellis, F.; Martellos, S.; Stenni, B.; Dal Borgo, A.; Zini, L.; Lisjak, K.; Nardini, A. Vineyard water relations in a karstic area: Deep roots and irrigation management. Agric. Ecosyst. Environ. 2018, 263, 53–59. [Google Scholar] [CrossRef]
- Smith, A.M.; Zeeman, S.C. Quantification of starch in plant tissues. Nat. Protoc. 2006, 1, 1342–1345. [Google Scholar] [CrossRef]
- Landhäusser, S.M.; Chow, P.S.; Dickman, L.; E Furze, M.; Kuhlman, I.; Schmid, S.; Wiesenbauer, J.; Wild, B.; Gleixner, G.; Hartmann, H.; et al. Standardized protocols and procedures can precisely and accurately quantify non-structural carbohydrates. Tree Physiol. 2018, 38, 1764–1778. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, T.R.; Tanner, C.B.; Bennett, J.M. Water-use efficiency in crop production. Bioscience 1984, 34, 36–40. [Google Scholar] [CrossRef]
- Brodribb, T.J.; Holbrook, N.M.; Edwards, E.J.; Gutiérrez, M.V. Relations between stomatal closure, leaf turgor and xylem vulnerability in eight tropical dry forest trees. Plant Cell Environ. 2003, 26, 443–450. [Google Scholar] [CrossRef] [Green Version]
- Sala, A.; Piper, F.; Hoch, G. Physiological mechanisms of drought-induced tree mortality are far from being resolved. New Phytol. 2010, 186, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Gambetta, G.A.; Herrera, J.C.; Dayer, S.; Feng, Q.; Hochberg, U.; Castellarin, S.D. The physiology of drought stress in grapevine: Towards an integrative definition of drought tolerance. J. Exp. Bot. 2020, 71, 4658–4676. [Google Scholar] [CrossRef]
- Savi, T.; Casolo, V.; Luglio, J.; Bertuzzi, S.; Trifilo’, P.; Lo Gullo, M.A.; Nardini, A. Species-specific reversal of stem xylem embolism after a prolonged drought correlates to endpoint concentration of soluble sugars. Plant Physiol. Biochem. 2016, 106, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.W.; Wang, L.J.; Zhao, J.Y.; Fan, P.G.; Li, S.H. Effect and after-effect of water stress on the distribution of newly-fixed 14C-photoassimilate in micropropagated apple plants. Environ. Exp. Bot. 2007, 60, 484–494. [Google Scholar] [CrossRef]
- Frioni, T.; Acimovic, D.; Tombesi, S.; Sivilotti, P.; Palliotti, A.; Poni, S.; Sabbatini, P. Changes in within-shoot carbon partitioning in pinot noir grapevines subjected to eaR + Ly basal leaf removal. Front. Plant Sci. 2018, 9, 1122. [Google Scholar] [CrossRef]
- Warick, R.P.; Hildebrandt, A.C. Free amino acid contents of stem and phylloxera gall tissue cultures of grape. Plant Physiol. 1966, 41, 573–578. [Google Scholar] [CrossRef] [Green Version]
- Eitle, M.; Cargnoni, M.; Acar, A.; Crespo Martinez, S.; Failla, O.; Kaul, H.-P.; Griesser, M.; Forneck, A. Phylloxeration effects on the sink activity and assimilation rate in phylloxera (Daktulosphaira vitifoliae Fitch) infested grapevines (Vitis spp.). Acta Hortic. 2017, 1188, 291–298. [Google Scholar] [CrossRef]
- Eitle, M.W.; Griesser, M.; Vankova, R.; Dobrev, P.; Aberer, S.; Forneck, A. Grape phylloxera (D. vitifoliae) manipulates SA/JA concentrations and signalling pathways in root galls of Vitis spp. Plant Physiol. Biochem. 2019, 144, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Eitle, M.W.; Loacker, J.; Meng-Reiterer, J.; Schuhmacher, R.; Griesser, M.; Forneck, A. Polyphenolic profiling of roots (Vitis spp.) under grape phylloxera (D. vitifoliae Fitch) attack. Plant Physiol. Biochem. 2019, 135, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Nalam, V.; Louis, J.; Shah, J. Plant defense against aphids, the pest extraordinaire. Plant Sci. 2019, 279, 96–107. [Google Scholar] [CrossRef] [PubMed]
| Glucose, mg g−1 | Fructose, mg g−1 | Sucrose, mg g−1 | Starch, mg g−1 | |
|---|---|---|---|---|
| Category | Apex | |||
| C | 25.7 ± 3.1 | 6.8 ± 1.0 | 59.0 ± 2.7 | 2.6 ± 0.3 |
| L | 20.7 ± 2.5 | 5.1 ± 0.9 | 57.4 ± 4.2 | 2.7 ± 0.2 |
| R+L | 26.2 ± 3.9 | 7.4 ± 1.4 | 49.8 ± 6.4 | 2.9 ± 0.4 |
| Noninfested leaf | ||||
| C | 8.1 ± 0.8 | 4.0 ± 0.5 | 81.1 ± 5.2 | 6.2 ± 0.5 |
| L | 6.5 ± 0.9 | 3.8 ± 0.9 | 79.7 ± 3.5 | 6.6 ± 1.4 |
| R+L | 5.5 ± 0.6 | 2.8 ± 0.5 | 75.1 ± 5.9 | 5.1 ± 0.7 |
| Leaf galls | ||||
| L | 9.1 ± 0.5 | 4.1 ± 0.3 | 39.9 ± 2.2 * | 4.9 ± 0.6 |
| R+L | 8.7 ± 0.6 | 3.4 ± 0.2 | 31.2 ± 2.3 * | 4.7 ± 0.4 |
| Glucose, mg g−1 | Fructose, mg g−1 | Sucrose, mg g−1 | Starch, mg g−1 | |
|---|---|---|---|---|
| Noninfested roots | Root tips | |||
| C | 170.2 ± 11.9 a | 52.9 ± 5.8 a | 73.0 ± 6.7 a | 8.0 ± 1.0 b |
| L | 139.9 ± 19.6 a | 46.1 ± 6.3 a | 75.1 ± 6.4 a | 9.0 ± 1.3 b |
| R+L | 92.5 ± 5.6 b | 28.3 ± 1.2 b | 52.3 ± 2.2 b | 10.1 ± 0.7 b |
| Infested roots | ||||
| R+L | 26.3 ± 10.0 c | 4.2 ± 1.3 c | 30.8 ± 1.5 c | 49.0 ± 8.0 c |
| Old roots | ||||
| C | 3.2 ± 2.1 | 4.4 ± 2.5 | 23.7 ± 5.0 | 74.7 ± 10.6 a |
| L | 3.1 ± 1.8 | 3.7 ± 1.1 | 27.4 ± 4.0 | 28.6 ± 8.2 b |
| R+L | 1.6 ± 0.4 | 3.1 ± 1.0 | 24.9 ± 1.3 | 47.3 ± 6.6 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savi, T.; Herrera, J.C.+.L.; Forneck, A. Leaf vs. Whole-Plant Biotic Attack: Does Vine Physiological Response Change? Water 2021, 13, 1429. https://doi.org/10.3390/w13101429
Savi T, Herrera JC+L, Forneck A. Leaf vs. Whole-Plant Biotic Attack: Does Vine Physiological Response Change? Water. 2021; 13(10):1429. https://doi.org/10.3390/w13101429
Chicago/Turabian StyleSavi, Tadeja, Jose CaR + Los Herrera, and Astrid Forneck. 2021. "Leaf vs. Whole-Plant Biotic Attack: Does Vine Physiological Response Change?" Water 13, no. 10: 1429. https://doi.org/10.3390/w13101429
APA StyleSavi, T., Herrera, J. C. +. L., & Forneck, A. (2021). Leaf vs. Whole-Plant Biotic Attack: Does Vine Physiological Response Change? Water, 13(10), 1429. https://doi.org/10.3390/w13101429







