First Report on Cyanotoxin (MC-LR) Removal from Surface Water by Multi-Soil-Layering (MSL) Eco-Technology: Preliminary Results
Abstract
:1. Introduction
2. Materials and Methods
2.1. MSL Pilot Description
2.2. MSL Experimental Conditions and Influent Water Quality
2.3. Samples Collection and Processing
2.4. Detection and Quantification of MC-LR by LC–ESI-MS
2.5. Statistical Analysis
3. Results
3.1. Physical and Chemical Characteristics of Influent and Effluent Waters
3.2. Quantification MC-LR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schopf, J.W. The Fossil Record: Tracing the Roots of the Cyanobacterial Lineage. In The Ecology of Cyanobacteria; Whitton, B.A., Potts, M., Eds.; Kluwer Academic: Amsterdam, The Netherlands, 2000; pp. 13–35. [Google Scholar]
- Tomitani, A.; Knoll, A.H.; Cavanaugh, C.M.; Ohno, T. The evolutionary diversification of cyanobacteria: Molecular-phylogenetic and paleontological perspectives. Proc. Natl. Acad. Sci. USA 2006, 103, 5442–5447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuad Hossain, M.; Ratnayake, R.R.; Mahbub, S.; Kumara, K.L.W.; Magana-Arachchi, D.N. Identification and culturing of cyanobacteria isolated from freshwater bodies of Sri Lanka for biodiesel production. Saudi J. Biol. Sci. 2020, 27, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G. Freshwater Cyanotoxins. In Biomarkers in Toxicology, 2nd ed.; Chapter 35; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 601–613. [Google Scholar] [CrossRef]
- Bormans, M.; Amzil, Z.; Mineaud, E.; Brient, L.; Savar, V.; Robert, E.; Lance, E. Demonstrated transfer of cyanobacteria and cyanotoxins along a freshwater-marine continuum in France. Harmful Algae 2019, 87, 101–639. [Google Scholar] [CrossRef]
- Uma, V.; Gnanasekaran, D.; Lakshmanan, U.; Dharmar, P. Survey and isolation of marine cyanobacteria from eastern coast of India as a biodiesel feedstock. Biocatal. Agric. Biotechnol. 2020, 24, 101–541. [Google Scholar] [CrossRef]
- Olokotum, M.; Mitroi, V.; Troussellier, M.; Semyalo, R.; Bernard, C.; Montuelle, B.; Okello, W.; Quiblier, C.; Humbert, J.-F. A review of the socioecological causes and consequences of cyanobacterial blooms in Lake Victoria. Harmful Algae 2020, 96, 101829. [Google Scholar] [CrossRef]
- Paerl, H.W.; Paul, V.J. Climate change: Links to global expansion of harmful cyanobacteria. Water Res. 2012, 46, 1349–1363. [Google Scholar] [CrossRef]
- Monteagudo, L.; Moreno, J.L. Benthic freshwater cyanobacteria as indicators of anthropogenic pressures. Ecol. Indic. 2016, 67, 693–702. [Google Scholar] [CrossRef]
- Ndlela, L.; Oberholster, P.; van Wyk, J.; Cheng, P. An overview of cyanobacterial bloom occurrences and research in Africa over the last decade. Harmful Algae 2016, 60, 11–26. [Google Scholar] [CrossRef]
- Coulibaly-Kalpy, J.; Soumahoro, M.-K.; Niamien-Ebrottie, J.E.; Yeo, K.; Amon, L.; Djaman, A.J.; Dosso, M. Déterminisme de la prolifération des cyanobactéries toxiques en Côte d ’Ivoire. Int. J. Biol. Chem. Sci. 2017, 11, 266–279. [Google Scholar] [CrossRef] [Green Version]
- Nalley, J.O.; O’Donnell, D.R.; Litchman, E. Temperature effects on growth rates and fatty acid content in freshwater algae and cyanobacteria. Algal Res. 2018, 35, 500–507. [Google Scholar] [CrossRef]
- Ettoumi, A.; El Khalloufi, F.; El Ghazali, I.; Oudra, B.; Amrani, A.; Nasri, H.; Bouaïcha, N.A. Bioaccumulation of cyanobacterial toxins in aquatic organisms and its consequences for public health. In Zooplankton and Phytoplankton: Types, Characteristics and Ecology; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 1–33. [Google Scholar]
- Chatziefthimiou, A.D.; Metcalf, J.S.; Glover, W.B.; Banack, S.A.; Dargham, S.R.; Richer, R.A. Cyanobacteria and cyanotoxins are present in drinking water impoundments and groundwater wells in desert environments. Toxicon 2016, 114, 75–84. [Google Scholar] [CrossRef]
- Bouaïcha, N.; Corbel, S. Cyanobacterial Toxins Emerging Emerging Contaminants Contaminants in Soils: A Review of Sources, Fate and Impacts on Ecosystems, Plants and Animal and Human Health. In Soil Contamination—Current Consequences and Further Solutions; BoD—Books on Demand: Norderstedt, Germany, 2016. [Google Scholar]
- Corbel, S.; Mougin, C.; Bouaïcha, N. Cyanobacterial toxins: Modes of actions, fate in aquatic and soil ecosystems, phytotoxicity and bioaccumulation in agricultural crops. Chemosphere 2014, 96, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Douma, M.; Manaut, N.; Saqrane, S.; El Khalloufi, F.; Oudra, B.; Loudiki, M. Toxicity assessment and detection of cyanobacterial toxins (Microcystins) in a Mediterranean natural lake (Dayete Aoua, Morocco). J. Mater. Environ. Sci. 2017, 8, 3247–3251. [Google Scholar]
- Metcalf, J.; Souza, N.R. Cyanobacteria and Their Toxins, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; Volume 11. [Google Scholar]
- Douma, M.; Loudiki, M.; Oudra, B.; Mouhri, K.; Ouahid, Y.; del Campo, F.F. Taxonomic diversity and toxicological assessment of Cyanobacteria in Moroccan inland waters. J. Water Sci. 2009, 22, 435–449. [Google Scholar] [CrossRef] [Green Version]
- Park, J.-A.; Yang, B.; Jang, M.; Kim, J.-H.; Kim, S.-B.; Park, H.-D.; Park, H.-M.; Lee, S.-H.; Choi, J.-W. Oxidation and molecular properties of microcystin-LR, microcystin-RR and anatoxin-a using UV-light-emitting diodes at 255 nm in combination with H2O2. Chem. Eng. J. 2019, 366, 423–432. [Google Scholar] [CrossRef]
- Greer, B.; McNamee, S.E.; Boots, B.; Cimarelli, L.; Guillebault, D.; Helmi, K.; Marcheggiani, S.; Panaiotov, S.; Breitenbach, U.; Akçaalan, R.; et al. A validated UPLC–MS/MS method for the surveillance of ten aquatic biotoxins in European brackish and freshwater systems. Harmful Algae 2016, 55, 31–40. [Google Scholar] [CrossRef]
- Turner, A.D.; Dhanji-Rapkova, M.; O’Neill, A.; Coates, L.; Lewis, A.; Lewis, K. Analysis of Microcystins in Cyanobacterial Blooms from Freshwater Bodies in England. Toxins 2018, 10, 39. [Google Scholar] [CrossRef] [Green Version]
- Gurbuz, F.; Uzunmehmetoğlu, O.Y.; Diler, Ö.; Metcalf, J.S.; Codd, G.A. Occurrence of microcystins in water, bloom, sediment and fish from a public water supply. Sci. Total Environ. 2016, 562, 860–868. [Google Scholar] [CrossRef]
- Zastepa, A.; Pick, F.R.; Blais, J.M. Distribution and flux of microcystin congeners in lake sediments. Lake Reserv. Manag. 2017, 33, 444–451. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Liu, J.; Zhang, D.; Luo, L.; Liao, Q.; Yuan, L.; Wu, N. Seasonal and spatial variations of microcystins in Poyang Lake, the largest freshwater lake in China. Environ. Sci. Pollut. Res. 2017, 25, 6300–6307. [Google Scholar] [CrossRef]
- Simiyu, B.M.; Oduor, S.O.; Rohrlack, T.; Sitoki, L.; Kurmayer, R. Microcystin Content in Phytoplankton and in Small Fish from Eutrophic Nyanza Gulf, Lake Victoria, Kenya. Toxins 2018, 10, 275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Major, Y.; Kifle, D.; Spoof, L.; Meriluoto, J. Cyanobacteria and microcystins in Koka reservoir (Ethiopia). Environ. Sci. Pollut. Res. 2018, 25, 26861–26873. [Google Scholar] [CrossRef] [PubMed]
- Fathalli, A.; Jenhani, A.B.R.; Moreira, C.; Welker, M.; Romdhane, M.; Antunes, A.; Vasconcelos, V. Molecular and phylogenetic characterization of potentially toxic cyanobacteria in Tunisian freshwaters. Syst. Appl. Microbiol. 2011, 34, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Oudra, B.; Loudiki, M.; Sbiyyaa, B.; Martins, R.; Vasconcelos, V.; Namikoshi, N. Isolation, characterization and quantification of microcystins (heptapeptides hepatotoxins) in Microcystis aeruginosa dominated bloom of Lalla Takerkoust lake reservoir (Morocco). Toxicon 2001, 39, 1375–1381. [Google Scholar] [CrossRef]
- Wiegand, C.; Pflugmacher, S. Ecotoxicological effects of selected cyanobacterial secondary metabolites a short review. Toxicol. Appl. Pharmacol. 2005, 203, 201–218. [Google Scholar] [CrossRef] [PubMed]
- El Khalloufi, F.; Oufdou, K.; Bertrand, M.; Lahrouni, M.; Oudra, B.; Ortet, P.; Barakat, M.; Heulin, T.; Achouak, W. Microbiote shift in the Medicago sativa rhizosphere in response to cyanotoxins extract exposure. Sci. Total Environ. 2016, 539, 135–142. [Google Scholar] [CrossRef]
- Corbel, S.; Mougin, C.; Nélieu, S.; Delarue, G.; Bouaïcha, N. Science of the Total Environment Evaluation of the transfer and the accumulation of microcystins in tomato (Solanum lycopersicum cultivar MicroTom) tissues using a cyanobacterial extract containing microcystins and the radiolabeled. Sci. Total Environ. 2016, 541, 1052–1058. [Google Scholar] [CrossRef]
- Manon, A. Analyse et Gestion du Risque Cyanobactéries/Cyanotoxines Dans les Eaux de Baignade et Les Eaux Destinées à La Consommation Humaine du Département de La Haute-Vienne. Master’s Thesis, L’école des Hautes Etudes en Santé Publique, Rennes, France, 2009. [Google Scholar]
- Meriluoto, J.; Spoof, L.; Codd, G. Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis, 1st ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017. [Google Scholar]
- Vasconcelos, V.; Sivonen, K.; Evans, W.; Carmichael, W.; Namikoshi, M. Hepatotoxic microcystin diversity in cyanobacterial blooms collected in portuguese freshwaters. Water Res. 1996, 30, 2377–2384. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Loudiki, M.; Oudra, B.; Sabour, B.; Sbiyyaa, B.; Vasconcelos, V. Taxonomy and geographic distribution of potential toxic cyanobacterial strains in Morocco. Ann. Limnol. Int. J. Limnol. 2002, 38, 101–108. [Google Scholar] [CrossRef] [Green Version]
- Oudra, B.; Loudiki, M.; Vasconcelos, V.; Sabour, B.; Sbiyyaa, B.; Oufdou, K.; Mezrioul, N.; Mezrioui, N. Detection and quantification of microcystins from cyanobacteria strains isolated from reservoirs and ponds in Morocco. Environ. Toxicol. 2002, 17, 32–39. [Google Scholar] [CrossRef]
- El Ghazali, I.; Saqrane, S.; Saker, M.; Youness, O.; Oudra, B.; Vasconcelos, V.; del Campo, F.F. Caractérisation biochimique et moléculaire d’efflorescences à cyanobactéries toxiques dans le réservoir Lalla Takerkoust (Maroc). J. Water Sci. 2011, 24, 117–128. [Google Scholar] [CrossRef] [Green Version]
- Oudra, B.; Andaloussi, M.D.-E.; Vasconcelos, V.M. Identification and quantification of microcystins from a Nostoc muscorum bloom occurring in Oukaïmeden River (High-Atlas mountains of Marrakech, Morocco). Environ. Monit. Assess. 2009, 149, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Wannicke, N.; Herrmann, A.; Gehringer, M.M. Atmospheric CO2 availability induces varying responses in net photosynthesis, toxin production and N2 fixation rates in heterocystous filamentous Cyanobacteria (Nostoc and Nodularia). Aquat. Sci. 2021, 83, 1–17. [Google Scholar] [CrossRef]
- Chen, W.; Jia, Y.; Liu, A.; Zhou, Q.; Song, L. Simultaneous elimination of cyanotoxins and PCBs via mechanical collection of cyanobacterial blooms: An application of “green-bioadsorption concept”. J. Environ. Sci. 2017, 57, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-A.; Jung, S.-M.; Yi, I.-G.; Choi, J.-W.; Kim, S.-B.; Lee, S.-H. Adsorption of microcystin-LR on mesoporous carbons and its potential use in drinking water source. Chemosphere 2017, 177, 15–23. [Google Scholar] [CrossRef]
- Chen, G.; Ding, X.; Zhou, W. Study on ultrasonic treatment for degradation of Microcystins (MCs). Ultrason. Sonochem. 2020, 63, 104900. [Google Scholar] [CrossRef]
- An, J.; Li, N.; Wang, S.; Liao, C.; Zhou, L.; Li, T.; Wang, X.; Feng, Y. A novel electro-coagulation-Fenton for energy efficient cyanobacteria and cyanotoxins removal without chemical addition. J. Hazard. Mater. 2019, 365, 650–658. [Google Scholar] [CrossRef]
- Chae, S.; Noeiaghaei, T.; Oh, Y.; Kim, I.S.; Park, J.-S. Effective removal of emerging dissolved cyanotoxins from water using hybrid photocatalytic composites. Water Res. 2019, 149, 421–431. [Google Scholar] [CrossRef]
- Serràa, A.; Pipabc, P.; Gómezde, E.; Philippea, L. Efficient magnetic hybrid ZnO-based photocatalysts for visible-light-driven removal of toxic cyanobacteria blooms and cyanotoxins. Appl. Catal. B Environ. 2020, 268, 118745. [Google Scholar] [CrossRef]
- Jasim, S.Y.; Saththasivam, J. Advanced oxidation processes to remove cyanotoxins in water. Desalination 2017, 406, 83–87. [Google Scholar] [CrossRef]
- Bakheet, B.; Islam, M.A.; Beardall, J.; Zhang, X.; McCarthy, D. Electrochemical inactivation of Cylindrospermopsis raciborskii and removal of the cyanotoxin cylindrospermopsins. J. Hazard. Mater. 2018, 344, 41–248. [Google Scholar] [CrossRef] [PubMed]
- León, C.; Boix, C.; Beltrán, E.; Peñuela, G.; López, F.; Sancho, J.V.; Hernández, F. Study of cyanotoxin degradation and evaluation of their transformation products in surface waters by LC-QTOF MS. Chemosphere 2019, 229, 538–548. [Google Scholar] [CrossRef]
- Bai, M.; Zheng, Q.; Zheng, W.; Li, H.; Lin, S.; Huang, L.; Zhang, Z. •OH Inactivation of Cyanobacterial Blooms and Degradation of Toxins in Drinking Water Treatment System. Water Res. 2019, 154, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Chintalapati, P.; Mohseni, M. Degradation of cyanotoxin microcystin-LR in synthetic and natural waters by chemical-free UV/VUV radiation. J. Hazard. Mater. 2020, 381, 120921. [Google Scholar] [CrossRef]
- González-Jartín, J.M.; Alves, L.D.C.; Alfonso, A.; Piñeiro, Y.; Vilar, S.Y.; Rodríguez, I.; Gomez, M.G.; Osorio, Z.V.; Sainz, M.J.; Vieytes, M.R.; et al. Magnetic nanostructures for marine and freshwater toxins removal. Chemosphere 2020, 256, 127019. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Zhang, F.; Liu, C.; Wang, M. Biodegradation of Microcystins by Bacillus sp. strain EMB. Energy Procedia 2012, 16, 2054–2059. [Google Scholar] [CrossRef] [Green Version]
- Yang, F.; Zhou, Y.; Sun, R.; Wei, H.; Li, Y.; Yin, L.; Pu, Y. Biodegradation of microcystin-LR and-RR by a novel microcystin-degrading bacterium isolated from Lake Taihu. Biodegradation 2014, 25, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Bourne, D.G.; Blakeley, R.L.; Riddles, P.; Jones, G.J. Biodegradation of the cyanobacterial toxin microcystin LR in natural water and biologically active slow sand filters. Water Res. 2006, 40, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yang, X.; Yang, L.; Xiao, B.; Wu, X.; Wang, J.; Wan, H. An effective pathway for the removal of microcystin LR via anoxic biodegradation in lake sediments. Water Res. 2010, 44, 1884–1892. [Google Scholar] [CrossRef]
- Nybom, S.; Dziga, D.; Heikkilä, J.; Kull, T.; Salminen, S.; Meriluoto, J. Characterization of microcystin-LR removal process in the presence of probiotic bacteria. Toxicon 2012, 59, 171–181. [Google Scholar] [CrossRef]
- Thees, A.; Atari, E.; Birbeck, J.; Westrick, J.A.; Huntley, J.F. Isolation and characterization of Lake Erie bacteria that degrade the cyanobacterial microcystin toxin MC-LR. J. Great Lakes Res. 2019, 45, 138–149. [Google Scholar] [CrossRef]
- Ndlela, L.L.; Oberholster, P.J.; van Wyk, J.H.; Cheng, P.H. Bacteria as biological control agents of freshwater cyanobacteria: Is it feasible beyond the laboratory? Appl. Microbiol. Biotechnol. 2018, 102, 9911–9923. [Google Scholar] [CrossRef]
- Ndlela, L.; Oberholster, P.; Van Wyk, J.; Cheng, P. A laboratory based exposure of Microcystis and Oscillatoria cyanobacterial isolates to heterotrophic bacteria. Toxicon 2019, 165, 1–12. [Google Scholar] [CrossRef]
- Zeng, G.; Gao, P.; Wang, J.; Zhang, J.; Zhang, M.; Sun, D. Algicidal Molecular Mechanism and Toxicological Degradation of Microcystis aeruginosa by White-Rot Fungi. Toxins 2020, 12, 406. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Ma, H.; Ren, S.; Gao, X.; He, X.; Zhu, S.; Deng, R.; Zhang, S. Insights into the mechanism of cyanobacteria removal by the algicidal fungi Bjerkandera adusta and Trametes versicolor. MicrobiologyOpen 2020, 9, e1042. [Google Scholar] [CrossRef]
- Sedmak, B.; Carmeli, S.; Eleršek, T. “Non-Toxic” Cyclic Peptides Induce Lysis of Cyanobacteria—An Effective Cell Population Density Control Mechanism in Cyanobacterial Blooms. Microb. Ecol. 2007, 56, 201–209. [Google Scholar] [CrossRef]
- El Bouaidi, W.; Essalhi, S.; Douma, M.; Tazart, Z.; Ounas, A.; Enaime, G.; Yaacoubi, A. Evaluation of the potentiality of Vicia faba and Opuntia ficus indica as eco-friendly coagulants to mitigate Microcystis aeruginosa blooms. Desalination Water Treat. 2020, 195, 198–213. [Google Scholar] [CrossRef]
- Álvarez, X.; Jiménez, A.; Cancela, Á.; Valero, E.; Sánchez, Á. Harvesting freshwater algae with tannins from the bark of forest species: Comparison of methods and pelletization of the biomass obtained. Chemosphere 2021, 268, 129313. [Google Scholar] [CrossRef]
- Bavithra, G.; Azevedo, J.; Oliveira, F.; Morais, J.; Pinto, E.; Ferreira, I.M.; Vasconcelos, V.; Campos, A.; Almeida, C.M.R. Assessment of Constructed Wetlands’ Potential for the Removal of Cyanobacteria and Microcystins (MC-LR). Water 2019, 12, 10. [Google Scholar] [CrossRef] [Green Version]
- Latrach, L.; Ouazzani, N.; Hejjaj, A.; Zouhir, F.; Mahi, M.; Masunaga, T.; Mandi, L. Optimization of hydraulic efficiency and wastewater treatment performances using a new design of vertical flow Multi-Soil-Layering (MSL) technology. Ecol. Eng. 2018, 117, 140–152. [Google Scholar] [CrossRef]
- Chen, X.; Luo, A.C.; Sato, K.; Wakatsuki, T.; Masunaga, T. An introduction of a multi-soil-layering system: A novel green technology for wastewater treatment in rural areas. Water Environ. J. 2009, 23, 255–262. [Google Scholar] [CrossRef]
- Sbahi, S.; Ouazzani, N.; Latrach, L.; Hejjaj, A.; Mandi, L. Predicting the concentration of total coliforms in treated rural domestic wastewater by multi-soil-layering (MSL) technology using artificial neural networks. Ecotoxicol. Environ. Saf. 2020, 204, 111118. [Google Scholar] [CrossRef] [PubMed]
- Luanmanee, S.; Attanandana, T.; Masunaga, T.; Wakatsuki, T. The efficiency of a multi-soil-layering system on domestic wastewater treatment during the ninth and tenth years of operation. Ecol. Eng. 2001, 18, 185–199. [Google Scholar] [CrossRef]
- Wei, C.-J.; Wu, W.-Z. Performance of single-pass and by-pass multi-step multi-soil-layering systems for low-(C/N)-ratio polluted river water treatment. Chemosphere 2018, 206, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Sato, K.; Wakatsuki, T.; Masunaga, T. Effect of aeration and material composition in soil mixture block on the removal of colored substances and chemical oxygen demand in livestock wastewater using multi-soil-layering systems. Soil Sci. Plant Nutr. 2007, 53, 509–516. [Google Scholar] [CrossRef]
- Pattnaik, R.R.; Yost, S.; Porter, G.; Masunaga, T.; Attanandana, T. Removing the N and P in Dairy Effluent using Multi-Soil-Layer (MSL) Systems. Appl. Eng. Agric. 2008, 24, 431–438. [Google Scholar] [CrossRef]
- Yidong, G.; Xin, C.; Shuai, Z.; Ancheng, L. Performance of multi-soil-layering system (MSL) treating leachate from rural unsanitary landfills. Sci. Total Environ. 2012, 420, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Sy, S.; Sofyan, S.; Kasman, M. Reduction of Pollutant Parameters in Textile Dyeing Wastewater by Gambier (Uncaria gambir Roxb) Using the Multi Soil Layering (MSL) Bioreactor. Mater. Sci. Eng. 2019, 546, 22–32. [Google Scholar] [CrossRef]
- Ait-hmane, A.; Ouazzani, N.; Latrach, L.; Hejjaj, A.; Assabbane, A.; Belkouadssi, M.; Mandi, L. Feasibility of Olive Mill Wastewater treatment by Multi-Soil-Layering Ecotechnology. J. Mater. Environ. Sci. 2018, 9, 1223–1233. [Google Scholar] [CrossRef]
- Fastner, J.; Flieger, I.; Neumann, U. Optimised extraction of microcystins from field samples—A comparison of different solvents and procedures. Water Res. 1998, 32, 3177–3181. [Google Scholar] [CrossRef]
- Triantis, T.M.; Kaloudis, T.; Zervou, S.-K.; Hiskia, A. Solid-Phase Extraction of Microcystins and Nodularin from Drinking Water. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2017; pp. 354–357. [Google Scholar]
- Kumar, P.; Hegde, K.; Brar, S.K.; Cledon, M.; Kermanshahi-Pour, A. Potential of biological approaches for cyanotoxin removal from drinking water: A review. Ecotoxicol. Environ. Saf. 2019, 172, 488–503. [Google Scholar] [CrossRef]
- Santos, A.; Rachid, C.; Pacheco, A.B.; Magalhães, V. Biotic and abiotic factors affect microcystin-LR concentrations in water/sediment interface. Microbiol. Res. 2020, 236, 126452. [Google Scholar] [CrossRef] [PubMed]
- Terin, U.; Sabogal-Paz, L. Microcystis aeruginosa and microcystin-LR removal by household slow sand filters operating in continuous and intermittent flows. Water Res. 2019, 150, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Latrach, L.; Ouazzani, N.; Hejjaj, A.; Mahi, M.; Masunaga, T.; Mandi, L. Two-stage vertical flow multi-soil-layering (MSL) technology for efficient removal of coliforms and human pathogens from domestic wastewater in rural areas under arid climate. Int. J. Hyg. Environ. Health 2018, 221, 64–80. [Google Scholar] [CrossRef] [PubMed]
- Hyenstrand, P.; Metcalf, J.; Beattie, K.; Codd, G. Effects of adsorption to plastics and solvent conditions in the analysis of the cyanobacterial toxin microcystin-LR by high performance liquid chromatography. Water Res. 2001, 35, 3508–3511. [Google Scholar] [CrossRef]
- Latrach, L.; Ouazzani, N.; Masunaga, T.; Hejjaj, A.; Bouhoum, K.; Mahi, M.; Mandi, L. Domestic wastewater disinfection by combined treatment using multi-soil-layering system and sand filters (MSL–SF): A laboratory pilot study. Ecol. Eng. 2016, 91, 294–301. [Google Scholar] [CrossRef]
- Wang, H.; Ho, L.; Lewis, D.M.; Brookes, J.D.; Newcombe, G. Discriminating and assessing adsorption and biodegradation removal mechanisms during granular activated carbon filtration of microcystin toxins. Water Res. 2007, 41, 4262–4270. [Google Scholar] [CrossRef]
- Grützmacher, G.; Böttcher, G.; Chorus, I.; Bartel, H. Removal of microcystins by slow sand filtration. Environ. Toxicol. 2002, 17, 386–394. [Google Scholar] [CrossRef]
- Ho, L.; Meyn, T.; Keegan, A.; Hoefel, D.; Brookes, J.; Saint, C.P.; Newcombe, G. Bacterial degradation of microcystin toxins within a biologically active sand filter. Water Res. 2006, 40, 768–774. [Google Scholar] [CrossRef]
- Ho, L.; Hoefel, D.; Saint, C.P.; Newcombe, G. Degradation of Microcystin-LR through Biological Sand Filters. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2007, 11, 191–196. [Google Scholar] [CrossRef]

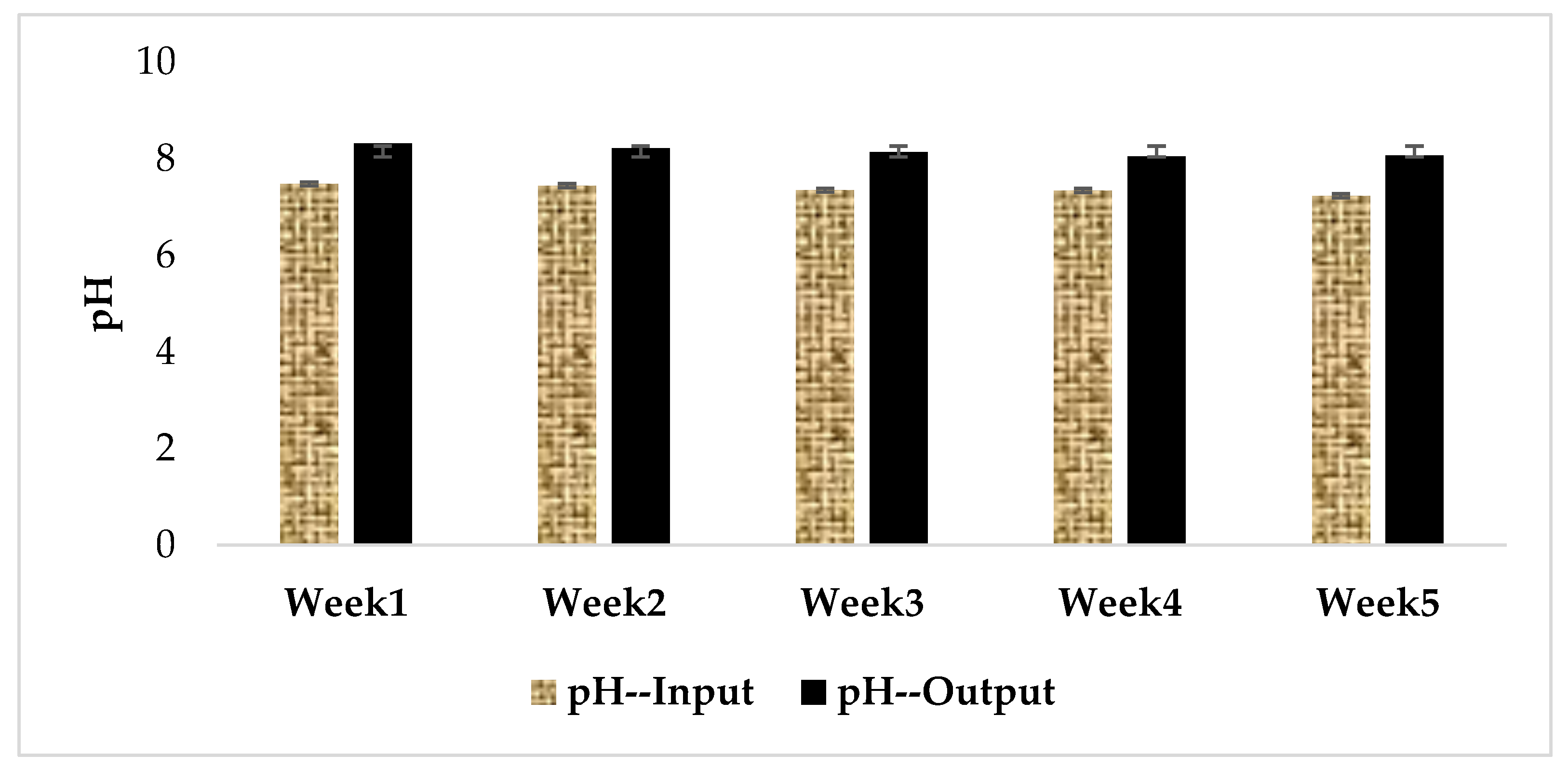
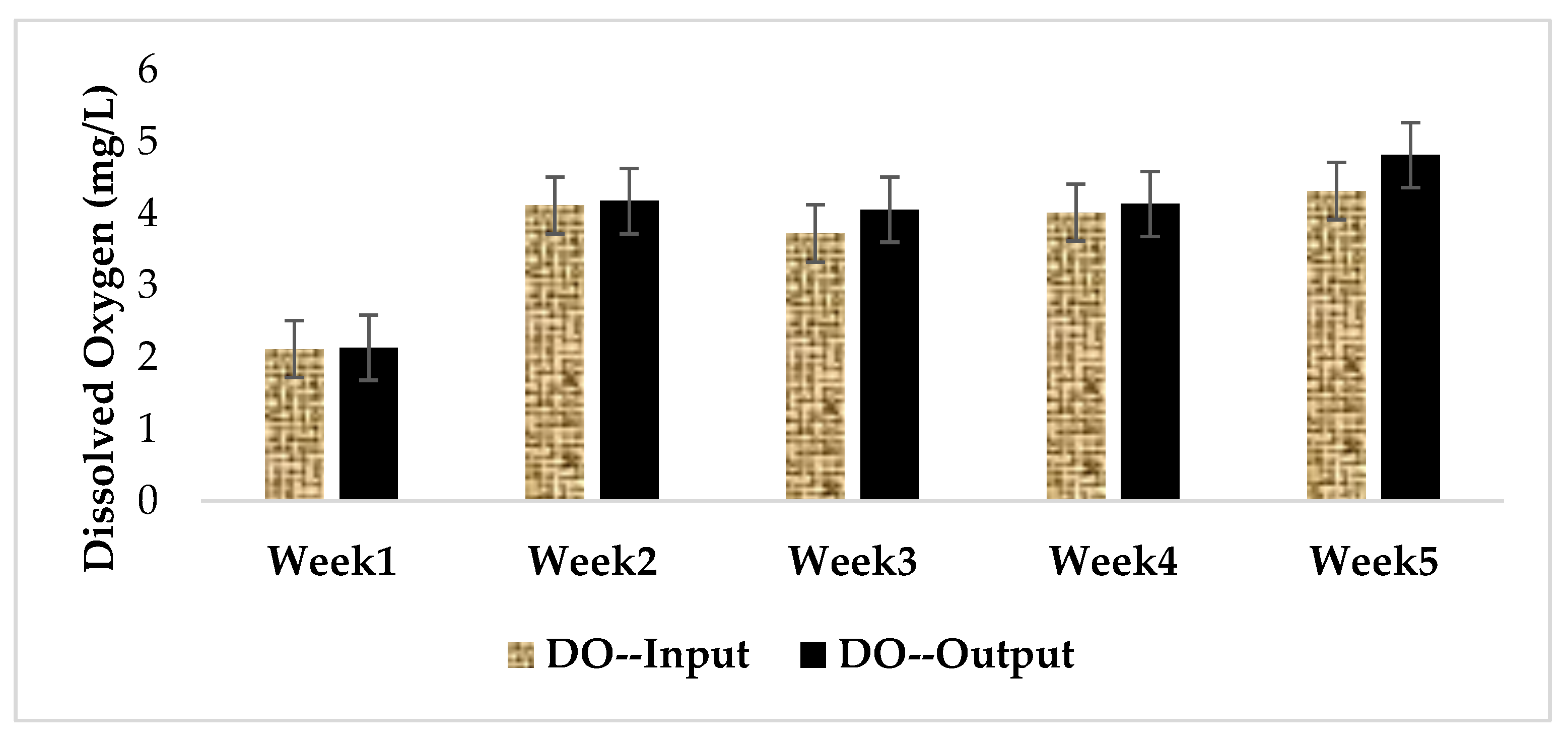
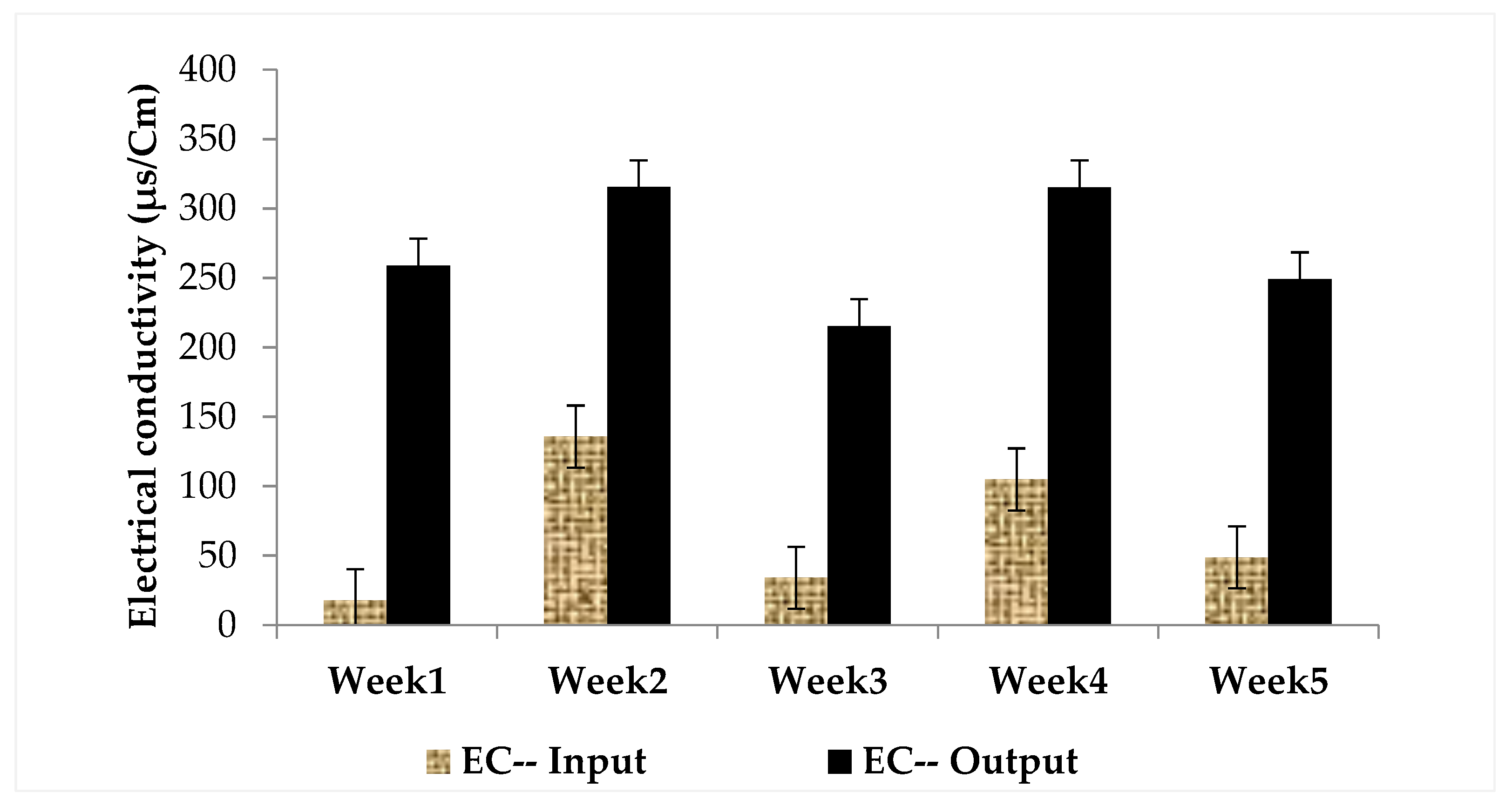
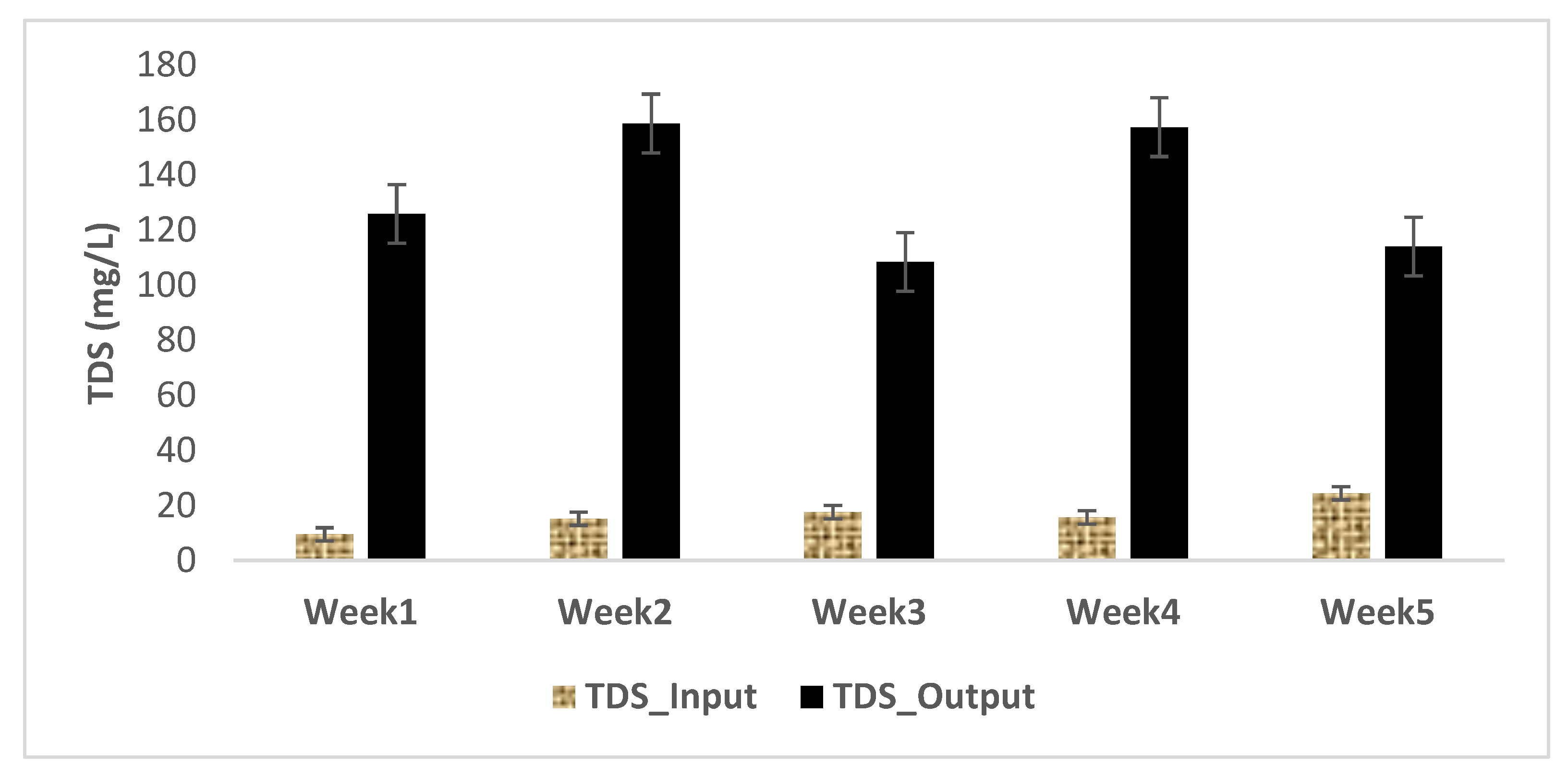
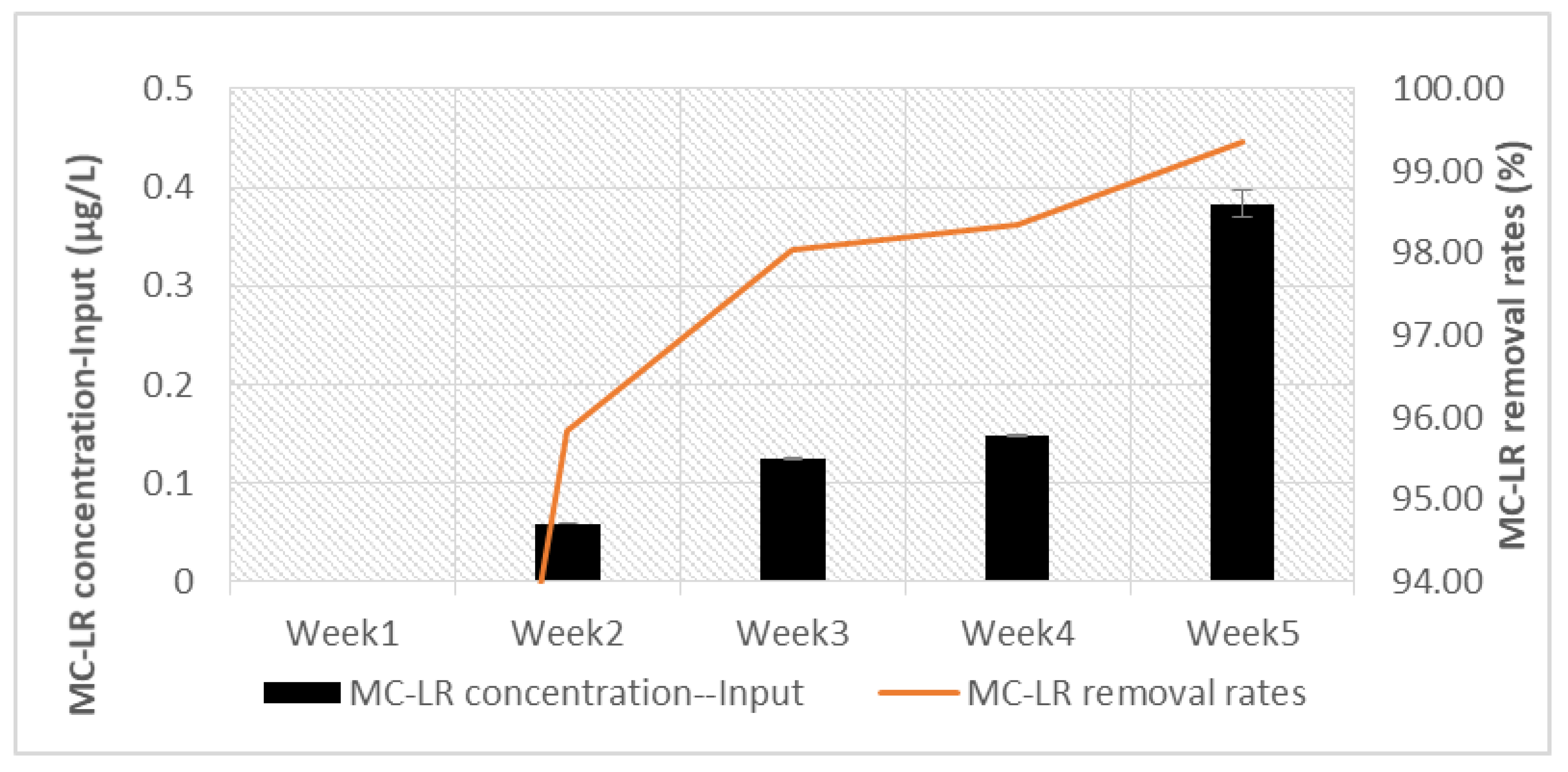
| Week | Concentration of the Extract in MC Equivalent (µg/L) at Input | Concentration of Extract in MC-LR (µg/L) at Input | MC-LR Concentration (µg/L) at Output | MC-LR Removal Rates (%) |
|---|---|---|---|---|
| 1 | 0.18 | nd * | nd * | - |
| 2 | 0.91 | 0.0585 (0.0001) | 0.0024 (0.0001) | 95.83% |
| 3 | 2.5 | 0.1248 (0.0001) | 0.0024 (0.0001) | 98.05% |
| 4 | 5 | 0.1479 (0.0014) | 0.0024 (0.0001) | 98.35% |
| 5, the first day of the week | 10 | 0.3832 (0.014) | 0.0024 (0.0001) | 99.36% |
| 5, the seventh day of the week | 0.3765 (0.0012) | 0.0024 (0.0001) | 99.35% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aba, R.P.; Mugani, R.; Hejjaj, A.; Brugerolle de Fraissinette, N.; Oudra, B.; Ouazzani, N.; Campos, A.; Vasconcelos, V.; Carvalho, P.N.; Mandi, L. First Report on Cyanotoxin (MC-LR) Removal from Surface Water by Multi-Soil-Layering (MSL) Eco-Technology: Preliminary Results. Water 2021, 13, 1403. https://doi.org/10.3390/w13101403
Aba RP, Mugani R, Hejjaj A, Brugerolle de Fraissinette N, Oudra B, Ouazzani N, Campos A, Vasconcelos V, Carvalho PN, Mandi L. First Report on Cyanotoxin (MC-LR) Removal from Surface Water by Multi-Soil-Layering (MSL) Eco-Technology: Preliminary Results. Water. 2021; 13(10):1403. https://doi.org/10.3390/w13101403
Chicago/Turabian StyleAba, Roseline Prisca, Richard Mugani, Abdessamad Hejjaj, Nelly Brugerolle de Fraissinette, Brahim Oudra, Naaila Ouazzani, Alexandre Campos, Vitor Vasconcelos, Pedro N. Carvalho, and Laila Mandi. 2021. "First Report on Cyanotoxin (MC-LR) Removal from Surface Water by Multi-Soil-Layering (MSL) Eco-Technology: Preliminary Results" Water 13, no. 10: 1403. https://doi.org/10.3390/w13101403










