Insecticides and Drought as a Fatal Combination for a Stream Macroinvertebrate Assemblage in a Catchment Area Exploited by Large-Scale Agriculture
Abstract
:1. Introduction
2. Materials and Methods
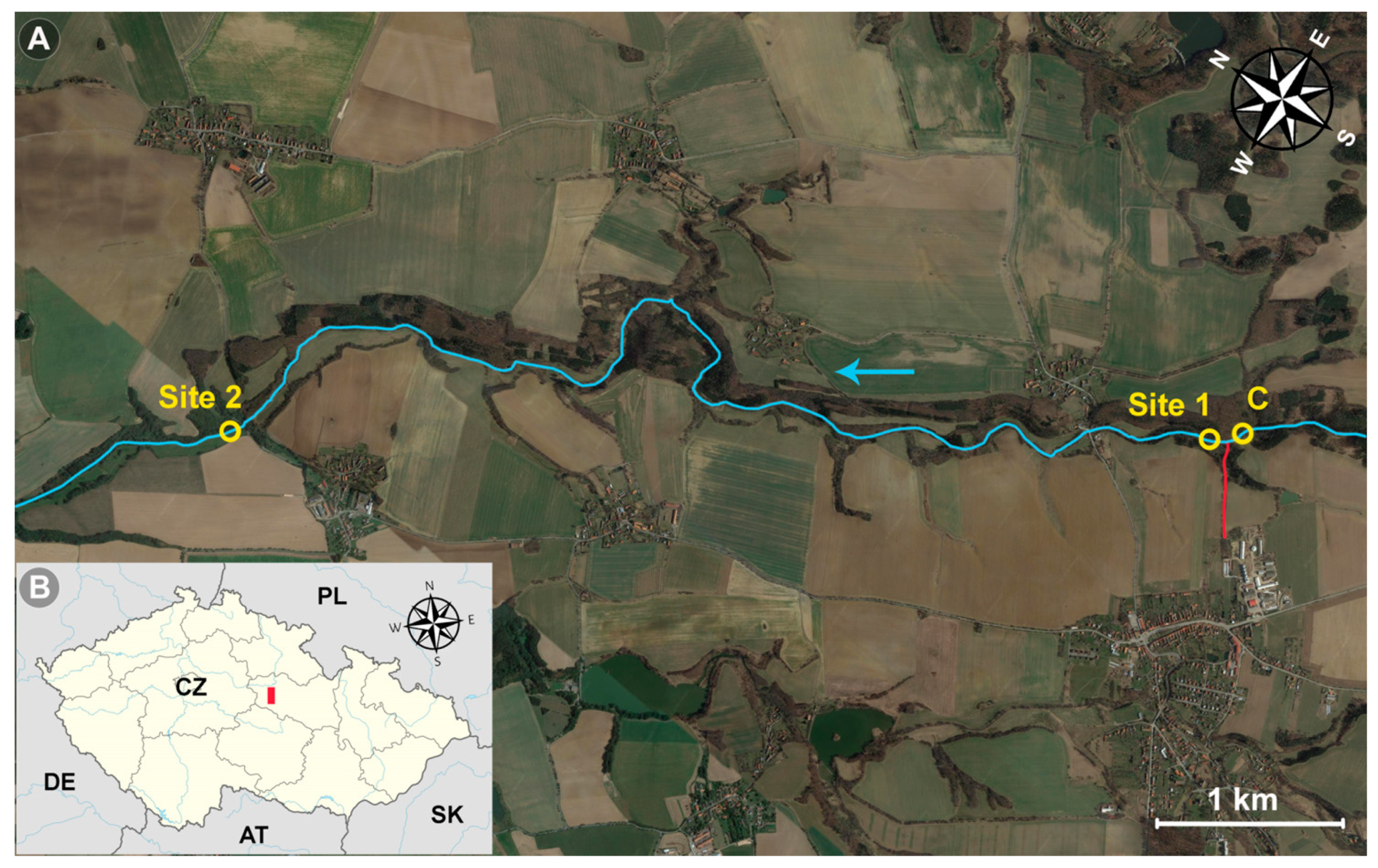
2.1. Locality Description
2.2. Insecticide Contamination
2.3. Sediment Sampling
2.4. Analysis for Pesticides
2.4.1. Reagents and Materials
2.4.2. Sample Processing and Analyses
2.4.3. LC-MS/MS Conditions
2.4.4. Validation of the Analytical Procedure
2.5. Macrozoobenthos Sampling and Analysis
2.5.1. Sampling and Determination
2.5.2. Estimation of Taxa Traits
2.5.3. Calculating Eco-Indicator Parameters
2.6. Classification of Hydrological Status
2.7. Statistical Analysis
3. Results
3.1. Recolonisation of Poisoned Stretches by Sensitive Insect Species
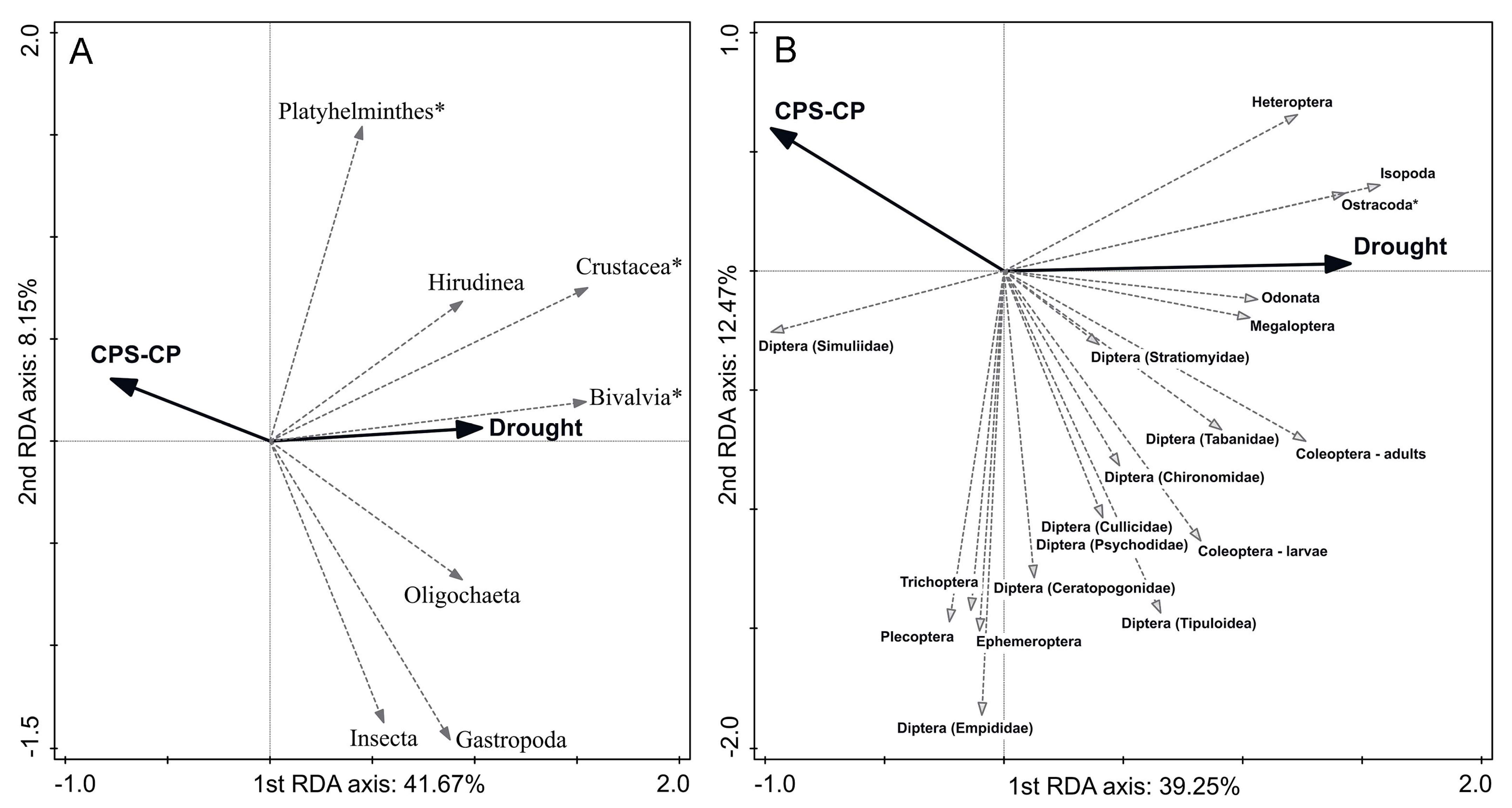
3.2. Relationship between Disturbances and Macroinvertebrate Taxonomic Composition
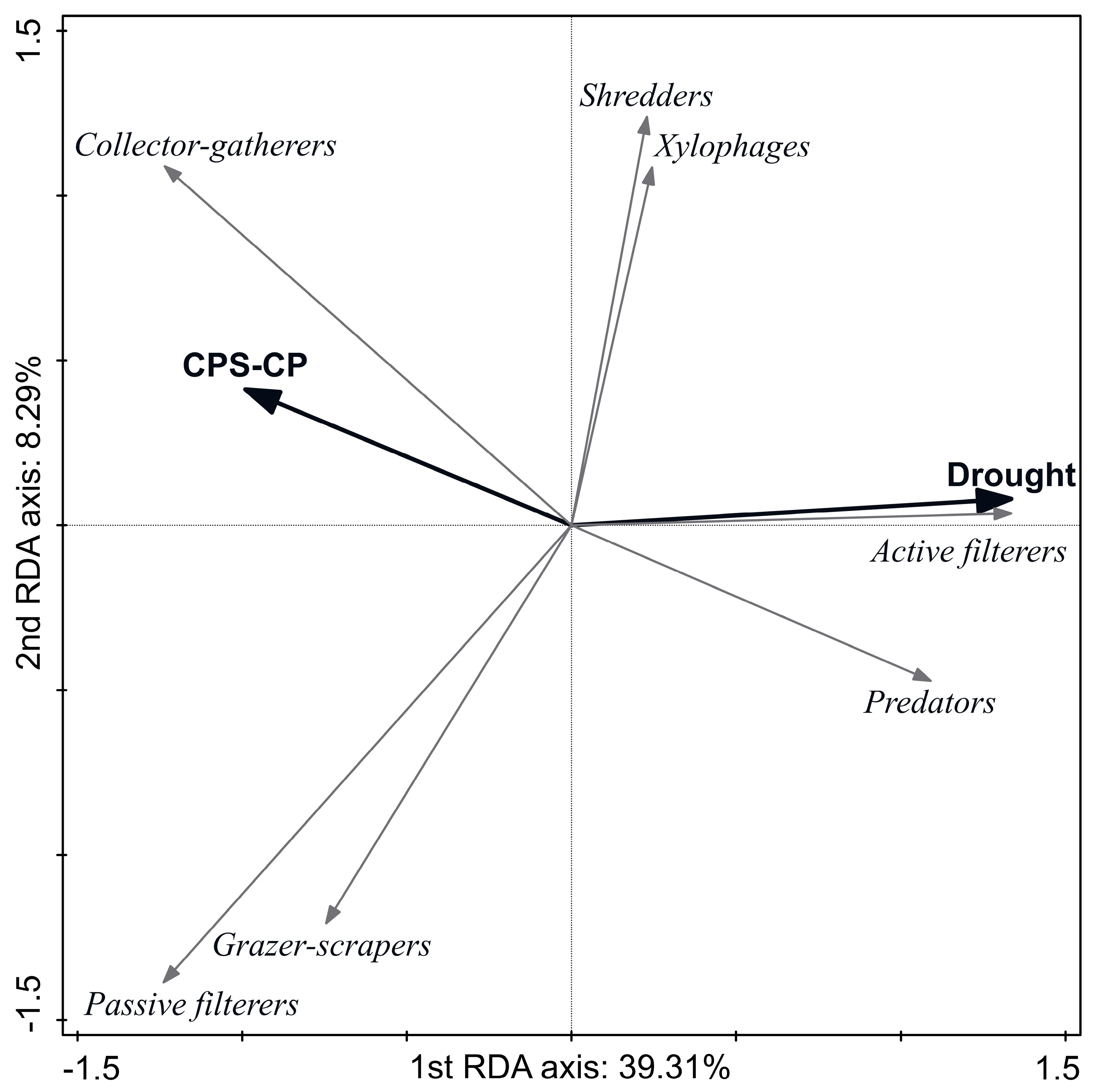
3.3. Effect of Disturbances on Ecological and Biological Species Traits
4. Discussion
4.1. Response of Macroinvertebrate Taxa to the Disturbances
4.2. Response of Species-Trait Assemblages to Disturbances
4.3. Parameters of a Macroinvertebrate Assemblage at the Studied Localities
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Měkotová, J.; Šarapatka, B.; Štěrba, O.; Harper, D. Restoration of a river landscape: Biotopes as a basis for quantification of species diversity and evaluation of landscape quality. Ecohydrol. Hydrobiol. 2006, 6, 43–51. [Google Scholar] [CrossRef]
- Bedient, P.B.; Huber, W.C.; Vieux, B.E. Hydrology and Floodplain Analysis, 4th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2008; pp. 721–722. [Google Scholar]
- Poole, G.C. Fluvial landscape ecology: Addressing uniqueness within the river discontinuum. Freshw. Biol. 2002, 47, 641–660. [Google Scholar] [CrossRef] [Green Version]
- Ward, J. Riverine landscapes: Biodiversity patterns, disturbance regimes, and aquatic conservation. Biol. Conserv. 1998, 83, 269–278. [Google Scholar] [CrossRef]
- Schäfer, R.B.; von der Ohe, P.C.; Rasmussen, J.; Kefford, B.J.; Beketov, M.A.; Schulz, R.; Liess, M. Thresholds for the effects of pesticides on invertebrate communities and leaf breakdown in stream ecosystems. Environ. Sci. Tech. 2012, 46, 5134–5142. [Google Scholar] [CrossRef] [Green Version]
- Worthington, T.; Shaw, P.; Daffern, J.; Langford, T. The effects of a thermal discharge on the macroinvertebrate community of a large British river: Implications for climate change. Hydrobiologia 2015, 753, 81–95. [Google Scholar] [CrossRef] [Green Version]
- Kennedy, T.L.; Turner, T.F. River channelization reduces nutrient flow and macroinvertebrate diversity at the aquatic terrestrial transition zone. Ecosphere 2011, 2, 1–13. [Google Scholar] [CrossRef]
- Johnson, R.C.; JIN, H.S.; Carreiro, M.M.; Jack, J.D. Macroinvertebrate community structure, secondary production and trophic-level dynamics in urban streams affected by non-point-source pollution. Freshw. Biol. 2013, 58, 843–857. [Google Scholar] [CrossRef]
- Martin, S.; Bertaux, A.; Le Ber, F.; Maillard, E.; Imfeld, G. Seasonal changes of macroinvertebrate communities in a stormwater wetland collecting pesticide runoff from a vineyard catchment (Alsace, France). AECT 2012, 62, 29–41. [Google Scholar] [CrossRef]
- Maund, S.J.; Hamer, M.J.; Lane, M.C.; Farrelly, E.; Rapley, J.H.; Goggin, U.M.; Gentle, W.E. Partitioning, bioavailability, and toxicity of the pyrethroid insecticide cypermethrin in sediments. Environ. Toxicol. Chem. 2002, 21, 9–15. [Google Scholar] [CrossRef]
- Bláha, M.; Grabicova, K.; Shaliutina, O.; Kubec, J.; Randák, T.; Zlabek, V.; Buřič, M.; Veselý, L. Foraging behaviour of top predators mediated by pollution of psychoactive pharmaceuticals and effects on ecosystem stability. Sci. Total Environ. 2019, 662, 655–661. [Google Scholar] [CrossRef]
- Veselý, L.; Boukal, D.S.; Buřič, M.; Kozák, P.; Kouba, A.; Sentis, A. Effects of prey density, temperature and predator diversity on nonconsumptive predator-driven mortality in a freshwater food web. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Racey, P.R.; Swift, S.M.; Rydell, J.; Brodie, L. Bats and insects over two Scottish rivers with contrasting nitrate status. Anim. Conserv. 1998, 1, 195–202. [Google Scholar] [CrossRef]
- Benítez-Mora, A.; Camargo, J.A. Ecological responses of aquatic macrophytes and benthic macroinvertebrates to dams in the Henares River Basin (Central Spain). Hydrobiologia 2014, 728, 167–178. [Google Scholar] [CrossRef]
- Anderson, C.B.; Rosemond, A.D. Ecosystem engineering by invasive exotic beavers reduces in-stream diversity and enhances ecosystem function in Cape Horn, Chile. Oecologia 2007, 154, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Iñiguez-Armijos, C.; Hampel, H.; Breuer, L. Land-use effects on structural and functional composition of benthic and leaf-associated macroinvertebrates in four Andean streams. Aquat. Ecol. 2018, 52, 77–92. [Google Scholar] [CrossRef] [Green Version]
- Hawkins, C.P.; Mykrä, H.; Oksanen, J.; Vander Laan, J.J. Environmental disturbance can increase beta diversity of stream macroinvertebrate assemblages. Glob. Ecol. Biogeogr. 2015, 24, 483–494. [Google Scholar] [CrossRef]
- Chester, E.; Robson, B. Anthropogenic refuges for freshwater biodiversity: Their ecological characteristics and management. Biol. Conserv. 2013, 166, 64–75. [Google Scholar] [CrossRef]
- Oertli, B. Freshwater biodiversity conservation: The role of artificial ponds in the 21st century. Aquat. Conserv. 2018, 28, 264–269. [Google Scholar] [CrossRef]
- Harabiš, F.; Dolný, A. Odonates need natural disturbances: How human-induced dynamics affect the diversity of dragonfly assemblages. Freshw. Sci. 2015, 34, 1050–1057. [Google Scholar] [CrossRef]
- Schuldt, B.; Buras, A.; Arend, M.; Vitasse, Y.; Beierkuhnlein, C.; Damm, A.; Gharun, M.; Grams, T.E.; Hauck, M.; Hajek, P. A first assessment of the impact of the extreme 2018 summer drought on Central European forests. BAAE 2020, 45, 86–103. [Google Scholar] [CrossRef]
- Hänsel, S.; Ustrnul, Z.; Łupikasza, E.; Skalak, P. Assessing seasonal drought variations and trends over Central Europe. Adv. Water Resour. 2019, 127, 53–75. [Google Scholar] [CrossRef]
- Beniston, M. The 2003 heat wave in Europe: A shape of things to come? An analysis based on Swiss climatological data and model simulations. Geophys. Res. Lett. 2004, 31, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Karger, D.N.; Schmatz, D.; Dettling, G.; Zimmermann, N.E. High resolution monthly precipitation and temperature timeseries for the period 2006–2100. Sci. Data 2020, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Štěrba, O.; Měkotová, J.; Benář, V.; Šarapatka, B.; Rychnovská, M.; Kubíček, F.; Řehořek, V. River Landscape and Its Ecosystems, 1st ed.; In Czech Republic; Univerzita Palackého: Olomouc, Czech Republic, 2008; pp. 295–335. [Google Scholar]
- Müller, C.; Cramer, W.; Hare, W.L.; Lotze-Campen, H. Climate change risks for African agriculture. Proc. Natl. Acad. Sci. USA 2011, 108, 4313–4315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhattachan, A.; Emanuel, R.E.; Ardon, M.; Bernhardt, E.S.; Anderson, S.M.; Stillwagon, M.G.; Ury, E.A.; Bendor, T.K.; Wright, J.P. Evaluating the effects of land-use change and future climate change on vulnerability of coastal landscapes to saltwater intrusion. Elementa Sci. Anthrop. 2018, 6, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Rulík, M.; White, S. The Role of Water in the Landscape. In Assessment and Protection of Water Resources in the Czech Republic; Zelenakova, M., Fialová, J., Negm, A.M., Eds.; Springer Water: Cham, Switzerland, 2020; pp. 71–90. [Google Scholar]
- Benateau, S.; Gaudard, A.; Stamm, C.; Altermatt, F. Climate Change and Freshwater Ecosystems: Impacts on Water Quality and Ecological Status; Hydro-CH2018Project; Federal Office for the Environment (FOEN): Bern, Switzerland, 2019; pp. 1–110. [Google Scholar] [CrossRef]
- Piggott, J.J.; Townsend, C.R.; Matthaei, C.D. Climate warming and agricultural stressors interact to determine stream macroinvertebrate community dynamics. Glob. Chang. Biol. 2015, 21, 1887–1906. [Google Scholar] [CrossRef] [PubMed]
- Munz, N.A.; Burdon, F.J.; De Zwart, D.; Junghans, M.; Melo, L.; Reyes, M.; Schönenberger, U.; Singer, H.P.; Spycher, B.; Hollender, J. Pesticides drive risk of micropollutants in wastewater-impacted streams during low flow conditions. Water Res. 2017, 110, 366–377. [Google Scholar] [CrossRef]
- Matsumura, F. Toxicology of Insecticides; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012; pp. 355–402. [Google Scholar]
- Carvalho, F.P. Pesticides, environment, and food safety. Food Energy Secur. 2017, 6, 48–60. [Google Scholar] [CrossRef]
- López, Ó.; Fernández-Bolaños, J.G.; Gil, M.V. Classical Insecticides: Past, Present, and Future. In Green Trends in Insect Control; López, Ó., Fernández-Bolaños, J.G., Eds.; Royal Society of Chemistry: Cambridge, UK, 2011; pp. 53–93. [Google Scholar]
- Nowell, L.H. Pesticides in Stream Sediment and Aquatic Biota: Distribution, Trends, and Governing Factors; CRC Press: Boca Raton, FL, USA, 1999; pp. 47–463. [Google Scholar]
- Beketov, M.; Foit, K.; Schäfer, R.; Schriever, C.; Sacchi, A.; Capri, E.; Biggs, J.; Wells, C.; Liess, M. SPEAR indicates pesticide effects in streams–comparative use of species-and family-level biomonitoring data. Environ. Pollut. 2009, 157, 1841–1848. [Google Scholar] [CrossRef]
- Muenze, R.; Orlinskiy, P.; Gunold, R.; Paschke, A.; Kaske, O.; Beketov, M.A.; Hundt, M.; Bauer, C.; Schüürmann, G.; Moeder, M. Pesticide impact on aquatic invertebrates identified with Chemcatcher® passive samplers and the SPEARpesticides index. Sci. Total Environ. 2015, 537, 69–80. [Google Scholar] [CrossRef]
- Belgers, J.D.M.; Van Lieverloo, R.J.; Van der Pas, L.J.; Van den Brink, P.J. Effects of the herbicide 2, 4-D on the growth of nine aquatic macrophytes. Aquat. Bot. 2007, 86, 260–268. [Google Scholar] [CrossRef]
- Stehle, S.; Knäbel, A.; Schulz, R. Probabilistic risk assessment of insecticide concentrations in agricultural surface waters: A critical appraisal. Environ. Monit. Assess. 2013, 185, 6295–6310. [Google Scholar] [CrossRef] [PubMed]
- Liess, M.; Ohe, P.C.V.D. Analyzing effects of pesticides on invertebrate communities in streams. Environ. Toxicol. Chem. 2005, 24, 954–965. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.L.; Antunes, S.C.; Castro, B.B.; Marques, C.R.; Gonçalves, A.M.; Gonçalves, F.; Pereira, R. Toxicity evaluation of three pesticides on non-target aquatic and soil organisms: Commercial formulation versus active ingredient. Ecotoxicology 2009, 18, 455–463. [Google Scholar] [CrossRef]
- Pestana, J.L.; Loureiro, S.; Baird, D.J.; Soares, A.M. Fear and loathing in the benthos: Responses of aquatic insect larvae to the pesticide imidacloprid in the presence of chemical signals of predation risk. Aquat. Toxicol. 2009, 93, 138–149. [Google Scholar] [CrossRef]
- Ankley, G.T.; Call, D.J.; Cox, J.S.; Kahl, M.D.; Hoke, R.A.; Kosian, P.A. Organic carbon partitioning as a basis for predicting the toxicity of chlorpyrifos in sediments. Environ. Toxicol. Chem. 1994, 13, 621–626. [Google Scholar] [CrossRef]
- Blann, K.L.; Anderson, J.L.; Sands, G.R.; Vondracek, B. Effects of agricultural drainage on aquatic ecosystems: A review. Crit. Rev. Environ. Sci. Tech. 2009, 39, 909–1001. [Google Scholar] [CrossRef]
- Brown, C.D.; Van Beinum, W. Pesticide transport via sub-surface drains in Europe. Environ. Pollut. 2009, 157, 3314–3324. [Google Scholar] [CrossRef]
- Matthaei, C.D.; Piggott, J.J.; Townsend, C.R. Multiple stressors in agricultural streams: Interactions among sediment addition, nutrient enrichment and water abstraction. J. Appl. Ecol. 2010, 47, 639–649. [Google Scholar] [CrossRef]
- Townsend, C.R.; Scarsbrook, M.R.; Dolédec, S. The intermediate disturbance hypothesis, refugia, and biodiversity in streams. Limnol. Oceanogr. 1997, 42, 938–949. [Google Scholar] [CrossRef] [Green Version]
- Ebert, S.; Hulea, O.; Strobel, D. Floodplain restoration along the lower Danube: A climate change adaptation case study. Clim. Dev. 2009, 1, 212–219. [Google Scholar] [CrossRef]
- Knillmann, S.; Orlinskiy, P.; Kaske, O.; Foit, K.; Liess, M. Indication of pesticide effects and recolonization in streams. Sci. Total Environ. 2018, 630, 1619–1627. [Google Scholar] [CrossRef] [PubMed]
- von der Ohe, P.C.; Goedkoop, W. Distinguishing the effects of habitat degradation and pesticide stress on benthic invertebrates using stressor-specific metrics. Sci. Total Environ. 2013, 444, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Arenas-Sánchez, A.; Rico, A.; Vighi, M. Effects of water scarcity and chemical pollution in aquatic ecosystems: State of the art. Sci. Total Environ. 2016, 572, 390–403. [Google Scholar] [CrossRef]
- Stehle, S.; Schulz, R. Agricultural insecticides threaten surface waters at the global scale. Proc. Natl. Acad. Sci. USA 2015, 112, 5750–5755. [Google Scholar] [CrossRef] [Green Version]
- Boulton, A.J.; Lake, P.S. Effects of Drought on Stream Insects and its Ecological Consequences. In Aquatic Insects: Challenges to Populations: Proceedings of the Royal Entomological Society’s 24th Symposium; Lancaster, J., Briers, R.A., Eds.; CABI: Wallingford, UK, 2008; pp. 81–102. [Google Scholar]
- Boulton, A.J.; Peterson, C.G.; Grimm, N.B.; Fisher, S.G. Stability of an aquatic macroinvertebrate community in a multiyear hydrologic disturbance regime. Ecology 1992, 73, 2192–2207. [Google Scholar] [CrossRef]
- Pastorino, P.; Zaccaroni, A.; Doretto, A.; Falasco, E.; Silvi, M.; Dondo, A.; Elia, A.C.; Prearo, M.; Bona, F. Functional Feeding Groups of Aquatic Insects Influence Trace Element Accumulation: Findings for Filterers, Scrapers and Predators from the Po Basin. Biology 2020, 9, 288. [Google Scholar] [CrossRef]
- Geist, J. Strategies for the conservation of endangered freshwater pearl mussels (Margaritifera margaritifera L.): A synthesis of conservation genetics and ecology. Hydrobiologia 2010, 644, 69–88. [Google Scholar] [CrossRef] [Green Version]
- Schletterer, M.; Füreder, L. The family Prosopistomatidae (Ephemeroptera): A review on its ecology and distribution, with particular emphasis on the European species Prosopistoma pennigerum Müller, 1785. Aquat. Insects 2009, 31, 603–620. [Google Scholar] [CrossRef]
- Diamond, J. Overview: Laboratory experiments, field experiments, and natural experiments. Nature 1983, 304, 586–587. [Google Scholar] [CrossRef]
- Ligeiro, R.; Hughes, R.M.; Kaufmann, P.R.; Heino, J.; Melo, A.S.; Callisto, M. Choice of field and laboratory methods affects the detection of anthropogenic disturbances using stream macroinvertebrate assemblages. Ecol. Indic. 2020, 115, 106382. [Google Scholar] [CrossRef]
- Ferenčík, M.; Schovánková, J. Occurrence of Polar Contaminants (Pesticides and Pharmaceuticals) in Suspended Matter in Povodí Labe Basin (in Czech). In Proceedings of the VII. Conference “Sedimenty vodných tokov a nádrží“ (in Slovak), Bratislava, Slovak Republic, 22–23 May 2013; Hucko, P., Tölgyessy, P., Eds.; Slovenská vodohospodárska spoločnosť člen ZSVTS pri Výskumnom ústave vodného hospodárstva: Bratislava, Slovak Republic, 2013; pp. 125–132. [Google Scholar]
- Rocha, A.A.; Monteiro, S.H.; Andrade, G.C.; Vilca, F.Z.; Tornisielo, V.L. Monitoring of pesticide residues in surface and subsurface waters, sediments, and fish in center-pivot irrigation areas. J. Braz. Chem. Soc. 2015, 26, 2269–2278. [Google Scholar] [CrossRef]
- Kokeš, J.; Němejcová, D. Metodika Odběru a Zpracování vzorků Makrozoobentosu Tekoucích vod Metodou PERLA; VÚV TGM Praha: Prague, Czech Republic, 2006; pp. 1–10. (In Czech) [Google Scholar]
- Kindt, R. Package ‘BiodiversityR’. Package for Community Ecology and Suitability Analysis. Version 2.11–12. 2019. Available online: https://CRAN.R-project.org/package=BiodiversityR (accessed on 11 March 2021).
- Paisley, M.; Trigg, D.; Walley, W. Revision of the biological monitoring working party (BMWP) score system: Derivation of present-only and abundance-related scores from field data. River Res. Appl. 2014, 30, 887–904. [Google Scholar] [CrossRef] [Green Version]
- Lepš, J.; Šmilauer, P. Multivariate Analysis of Ecological Data Using CANOCO; Cambridge University Press: Cambridge, UK, 2003; pp. 60–75. [Google Scholar]
- Friberg, N.; Lindstrøm, M.; Kronvang, B.; Larsen, S.E. Macroinvertebrate/sediment relationships along a pesticide gradient in Danish streams. Hydrobiologia 2003, 493, 103–110. [Google Scholar] [CrossRef]
- Cuffney, T.F.; Wallace, J.B.; Webster, J.R. Pesticide manipulation of a headwater stream: Invertebrate responses and their significance for ecosystem processes. Freshw. Invertebr. Biol. 1984, 3, 153–171. [Google Scholar] [CrossRef]
- Fritz, K.M.; Dodds, W.K. Resistance and resilience of macroinvertebrate assemblages to drying and flood in a tallgrass prairie stream system. Hydrobiologia 2004, 527, 99–112. [Google Scholar] [CrossRef]
- Stanley, E.H.; Buschman, D.L.; Boulton, A.J.; Grimm, N.B.; Fisher, S. Invertebrate resistance and resilience to intermittency in a desert stream. Amer. Midl. Nat. 1994, 288–300. [Google Scholar] [CrossRef] [Green Version]
- Clarke, A.; Mac Nally, R.; Bond, N.; Lake, P.S. Macroinvertebrate diversity in headwater streams: A review. Freshw. Biol. 2008, 53, 1707–1721. [Google Scholar] [CrossRef]
- Buřič, M.; University of South Bohemia, České Budějovice, Czech Republic; Kouba, A.; University of South Bohemia, České Budějovice, Czech Republic. Personal communication, 2018.
- Reynolds, J.; Souty-Grosset, C.; Richardson, A. Ecological roles of crayfish in freshwater and terrestrial habitats. Freshwater Crayfish 2013, 19, 197–218. [Google Scholar] [CrossRef]
- Watts, M. Ecotoxicity. In Chlorpyrifos as a Possible Global POP; Pesticide Action Network North America: Oakland, CA, USA, 2012; pp. 1–34. Available online: http://www.panna.org/sites/default/files/Chlorpyrifos_as_POP_final_2013_0.pdf (accessed on 12 May 2021).
- European Commission. Review report for the active substance alpha-cypermethrin. In Health and Consumer Protection Directorate-General; European Commission: Brussels, Belgium, 2004; pp. 1–70. Available online: https://epdfs.co.uk/doc/304998/european-commission-alpha-cypermethrin-sanco/4335/2000- (accessed on 12 May 2021).
- Rubach, M.N.; Ashauer, R.; Maund, S.J.; Baird, D.J.; Van den Brink, P.J. Toxicokinetic variation in 15 freshwater arthropod species exposed to the insecticide chlorpyrifos. Environ. Toxicol. Chem. 2010, 29, 2225–2234. [Google Scholar] [CrossRef]
- Plahuta, M.; Tišler, T.; Toman, M.J.; Pintar, A. Toxic and endocrine disrupting effects of wastewater treatment plant influents and effluents on a freshwater isopod Asellus aquaticus (Isopoda, Crustacea). Chemosphere 2017, 174, 342–353. [Google Scholar] [CrossRef]
- Spänhoff, B.; Bischof, R.; Böhme, A.; Lorenz, S.; Neumeister, K.; Nöthlich, A.; Küsel, K. Assessing the impact of effluents from a modern wastewater treatment plant on breakdown of coarse particulate organic matter and benthic macroinvertebrates in a lowland river. Water Air Soil Pollut. 2007, 180, 119–129. [Google Scholar] [CrossRef]
- Iversen, T.M.; Wiberg-Larsen, P.; Hansen, S.B.; Hansen, F.S. The effect of partial and total drought on the macroinvertebrate communities of three small Danish streams. Hydrobiologia 1978, 60, 235–242. [Google Scholar] [CrossRef]
- Extence, C. The effect of drought on benthic invertebrate communities in a lowland river. Hydrobiologia 1981, 83, 217–224. [Google Scholar] [CrossRef]
- Finelli, C.M.; Hart, D.D.; Merz, R. Stream insects as passive suspension feeders: Effects of velocity and food concentration on feeding performance. Oecologia 2002, 131, 145–153. [Google Scholar] [CrossRef]
- Currie, D.C.; Craig, D.A. Feeding strategies of larval black flies. In Black Flies: Ecology, Population Management, and Annotated World List; Kim, K.C., Merritt, R.W., Eds.; Pennsylvania State University Press: University Park, PA, USA, 1987; pp. 155–170. [Google Scholar]
- Horak, C.N.; Assef, Y.A.; Grech, M.G.; Miserendino, M.L. Agricultural practices alter function and structure of macroinvertebrate communities in Patagonian piedmont streams. Hydrobiologia 2020, 847, 3659–3676. [Google Scholar] [CrossRef]
- Lenat, D.R.; Crawford, J.K. Effects of land use on water quality and aquatic biota of three North Carolina Piedmont streams. Hydrobiologia 1994, 294, 185–199. [Google Scholar] [CrossRef]
- Aspin, T.W.; Hart, K.; Khamis, K.; Milner, A.M.; O’Callaghan, M.J.; Trimmer, M.; Wang, Z.; Williams, G.M.; Woodward, G.; Ledger, M.E. Drought intensification alters the composition, body size, and trophic structure of invertebrate assemblages in a stream mesocosm experiment. Freshw. Biol. 2019, 64, 750–760. [Google Scholar] [CrossRef]
- Vallenduuk, H.J.; Pillot, H.K.M. Chironomidae Larvae, Vol. 1: Tanypodinae: General Ecology and Tanypodinae; KNNV Publishing: Zeist, The Netherlands, 2007; pp. 74–132. [Google Scholar]
- Pillot, H.K.M. Chironomidae Larvae, Vol. 2: Chironomini: Biology and Ecology of the Chironomini; KNNV Publishing: Zeist, The Netherlands, 2009; pp. 6–247. [Google Scholar]
- Pillot, H.K.M. Chironomidae Larvae, Vol. 3: Orthocladiinae: Biology and Ecology of the Aquatic Orthocladiinae; KNNV Publishing: Zeist, The Netherlands, 2014; Volume 3, pp. 7–287. [Google Scholar]
- Tokeshi, M. Life cycles and population dynamics. In The Chironomidae: The Biology and Ecology of Non-Biting Midges; Armitage, P.D., Cranston, P.S., Pinder, L.C.V., Eds.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1995; pp. 225–268. [Google Scholar]
- Barron, M.G.; Woodburn, K.B. Ecotoxicology of chlorpyrifos. Rev. Environ. Contam. Toxicol. 1995, 1–93. [Google Scholar] [CrossRef]
- Mehler, W.T.; Du, J.; Lydy, M.J.; You, J. Joint toxicity of a pyrethroid insecticide, cypermethrin, and a heavy metal, lead, to the benthic invertebrate Chironomus dilutus. Environ. Toxicol. Chem. 2011, 30, 2838–2845. [Google Scholar] [CrossRef]
- Lancaster, J.; Ledger, M.E. Population-level responses of stream macroinvertebrates to drying can be density-independent or density-dependent. Freshw. Biol. 2015, 60, 2559–2570. [Google Scholar] [CrossRef]
- Marcogliese, D. The impact of climate change on the parasites and infectious diseases of aquatic animals. Rev. Sci. Tech. 2008, 27, 467–484. [Google Scholar] [CrossRef]
- Hutchens, J.J., Jr.; Chung, K.; Wallace, J.B. Temporal variability of stream macroinvertebrate abundance and biomass following pesticide disturbance. J. N. Am. Benthol. Soc. 1998, 17, 518–534. [Google Scholar] [CrossRef]
- Johnson, R.C.; Carreiro, M.M.; Jin, H.-S.; Jack, J.D. Within-year temporal variation and life-cycle seasonality affect stream macroinvertebrate community structure and biotic metrics. Ecol. Indic. 2012, 13, 206–214. [Google Scholar] [CrossRef]
- Graf, W.; Murphy, J.; Dahl, J.; Zamora-Munoz, C.; Lopez-Rodriguez, M.J. Distribution and Ecological Preferences of European Freshwater Organisms. Volume 1. Trichoptera; Pensoft Publishers: Sofia, Bulgaria, 2008; p. 284. [Google Scholar]
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The river continuum concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Wallace, J.B.; Eggert, S.L.; Meyer, J.L.; Webster, J.R. Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 1997, 277, 102–104. [Google Scholar] [CrossRef] [Green Version]
- Herbst, D.B.; Cooper, S.D.; Medhurst, R.B.; Wiseman, S.W.; Hunsaker, C.T. Drought ecohydrology alters the structure and function of benthic invertebrate communities in mountain streams. Freshw. Biol. 2019, 64, 886–902. [Google Scholar] [CrossRef]
- Zilli, F.L.; Montalto, L.; Marchese, M.R. Benthic invertebrate assemblages and functional feeding groups in the Paraná River floodplain (Argentina). Limnologica 2008, 38, 159–171. [Google Scholar] [CrossRef] [Green Version]
- Adámek, Z.; Helešic, J.; Maršálek, B.; Rulík, M. Applied Hydrobiology; University South Bohemia in České Budějovice, Faculty of Fisheries and Protection of Waters: Vodňany, Czech Republic, 2014; pp. 205–244. [Google Scholar]
- Wallace, J.B.; Vogel, D.S.; Cuffney, T. Recovery of a headwater stream from an insecticide-induced community disturbance. J. N. Am. Benthol. Soc. 1986, 5, 115–126. [Google Scholar] [CrossRef]
- Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32000L0060 (accessed on 23 February 2021).
- Orlinskiy, P.; Münze, R.; Beketov, M.; Gunold, R.; Paschke, A.; Knillmann, S.; Liess, M. Forested headwaters mitigate pesticide effects on macroinvertebrate communities in streams: Mechanisms and quantification. Sci. Total Environ. 2015, 524, 115–123. [Google Scholar] [CrossRef]
- Michael, J.L. Pesticides used in forestry and their impacts on water quality. South. Weed Sci. Soc. 2000, 53, 81–91. [Google Scholar]
- Schulz, R.; Liess, M. A field study of the effects of agriculturally derived insecticide input on stream macroinvertebrate dynamics. Aquat. Toxicol. 1999, 46, 155–176. [Google Scholar] [CrossRef]
- Hunt, L.; Bonetto, C.; Marrochi, N.; Scalise, A.; Fanelli, S.; Liess, M.; Lydy, M.J.; Chiu, M.-C.; Resh, V.H. Species at Risk (SPEAR) index indicates effects of insecticides on stream invertebrate communities in soy production regions of the Argentine Pampas. Sci. Total Environ. 2017, 580, 699–709. [Google Scholar] [CrossRef] [Green Version]
- Lake, P.S. Disturbance, patchiness, and diversity in streams. J. N. Am. Benthol. Soc. 2000, 19, 573–592. [Google Scholar] [CrossRef] [Green Version]
| Sampling Date | Site C (µg kg−1 d.w.) | Site 1 (µg kg−1 d.w.) | Site 2 (µg kg−1 d.w.) | Time Passed after Accident (Days) |
|---|---|---|---|---|
| 4th IV 2014 | <LOQ | 13,000 | 350 | 5 |
| 20th VI 2014 * | <LOQ | 128 | 51 | 83 |
| 17th IV 2015 * | <LOQ | 87 | <LOQ | 384 |
| 26th VII 2016 * | <LOQ | 33 | <LOQ | 850 |
| 19th IX 2017 * | <LOQ | 25 | <LOQ | 1476 |
| Description: | Score: |
|---|---|
| Fast-flowing | 1 |
| Loss of fast-flowing habitats | 2 |
| Loss of lateral connectivity to stream-edge habitats | 3 |
| Loss of longitudinal connectivity, flow stops, isolated pools form | 4 |
| Pools shrink water quality deteriorates | 5 |
| Total absence of water | 6 |
| 2014 | 2015 | 2016 | 2017 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | C | 1 | 2 | C | 1 | 2 | C | 1 | 2 | C | 1 | 2 |
| Abundance | 1142 | 228 | 2752 | 644 | 966 | 4472 | 2842 | 2313 | 7227 | 3308 | 776 | 1432 |
| Richness | 26 | 9 | 15 | 39 | 26 | 23 | 34 | 22 | 21 | 43 | 23 | 26 |
| H‘ | 2.40 | 1.44 | 1.51 | 3.10 | 2.06 | 1.69 | 2.05 | 2.44 | 1.53 | 2.99 | 2.17 | 2.77 |
| SPEARpesticides index | 0.23 | 0.15 | 0.02 | 0.11 | 0.13 | 0.09 | 0.20 | 0.06 | 0.05 | 0.16 | 0 | 0.08 |
| SPEARrefuge index | 0.25 | 0.29 | 0.05 | 0.14 | 0.15 | 0.02 | 0.06 | 0 | 0 | 0.12 | 0.08 | 0.10 |
| SPEARpesticides index (old) | 0.48 | 0.44 | 0.07 | 0.25 | 0.29 | 0.11 | 0.26 | 0.06 | 0.05 | 0.27 | 0.08 | 0.18 |
| N SPEARpesticides | 6 | 2 | 1 | 5 | 4 | 3 | 9 | 3 | 1 | 9 | 0 | 3 |
| N SPEARrefuge | 7 | 3 | 1 | 6 | 5 | 1 | 2 | 0 | 0 | 5 | 2 | 3 |
| N SPEARpesticides (old) | 13 | 5 | 2 | 11 | 9 | 4 | 11 | 3 | 1 | 14 | 2 | 6 |
| Abu SPEARpesticides | 306 | 8 | 1 | 40 | 30 | 26 | 94 | 9 | 68 | 136 | 0 | 24 |
| Abu SPEARrefuge | 142 | 44 | 6 | 58 | 36 | 2 | 40 | 0 | 0 | 280 | 16 | 72 |
| Abu SPEARpesticides (old) | 448 | 52 | 7 | 98 | 66 | 28 | 134 | 9 | 68 | 416 | 16 | 96 |
| N EPT | 14 | 5 | 2 | 12 | 10 | 4 | 7 | 0 | 1 | 11 | 2 | 4 |
| Abu EPT | 448 | 52 | 7 | 148 | 94 | 28 | 100 | 0 | 68 | 396 | 16 | 80 |
| EPT% | 42.73 | 22.81 | 0.25 | 22.98 | 9.73 | 0.63 | 3.52 | 0 | 0.94 | 11.97 | 2.06 | 5.59 |
| Original BMWP score | 100 | 42 | 26 | 107 | 77 | 42 | 102 | 49 | 38 | 141 | 65 | 72 |
| ASPT index (±SEM) | 6.67 ± 0.77 | 6.00 ± 1.35 | 3.71 ± 0.78 | 6.29 ± 0.78 | 5.50 ± 0.79 | 4.20 ± 0.63 | 5.10 ± 0.62 | 4.08 ± 0.53 | 3.80 ± 0.79 | 5.88 ± 0.63 | 4.64 ± 0.70 | 4.80 ± 0.76 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Let, M.; Špaček, J.; Ferenčík, M.; Kouba, A.; Bláha, M. Insecticides and Drought as a Fatal Combination for a Stream Macroinvertebrate Assemblage in a Catchment Area Exploited by Large-Scale Agriculture. Water 2021, 13, 1352. https://doi.org/10.3390/w13101352
Let M, Špaček J, Ferenčík M, Kouba A, Bláha M. Insecticides and Drought as a Fatal Combination for a Stream Macroinvertebrate Assemblage in a Catchment Area Exploited by Large-Scale Agriculture. Water. 2021; 13(10):1352. https://doi.org/10.3390/w13101352
Chicago/Turabian StyleLet, Marek, Jan Špaček, Martin Ferenčík, Antonín Kouba, and Martin Bláha. 2021. "Insecticides and Drought as a Fatal Combination for a Stream Macroinvertebrate Assemblage in a Catchment Area Exploited by Large-Scale Agriculture" Water 13, no. 10: 1352. https://doi.org/10.3390/w13101352
APA StyleLet, M., Špaček, J., Ferenčík, M., Kouba, A., & Bláha, M. (2021). Insecticides and Drought as a Fatal Combination for a Stream Macroinvertebrate Assemblage in a Catchment Area Exploited by Large-Scale Agriculture. Water, 13(10), 1352. https://doi.org/10.3390/w13101352






