Characterization of Flowpath Using Geochemistry and 87Sr/86Sr Isotope Ratios in the Yalahau Region, Yucatan Peninsula, Mexico
Abstract
1. Introduction
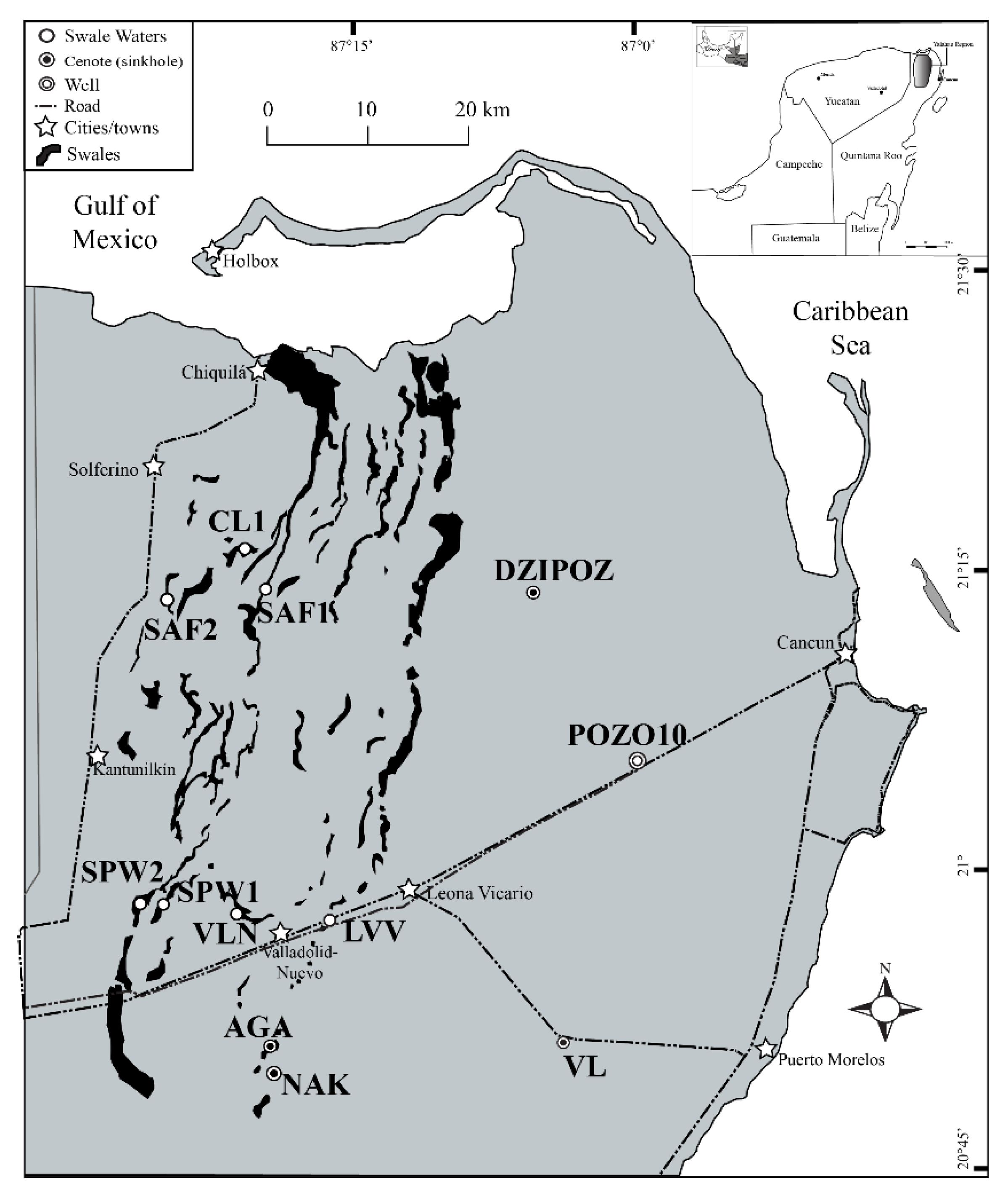
1.1. Study Area
1.1.1. Climate
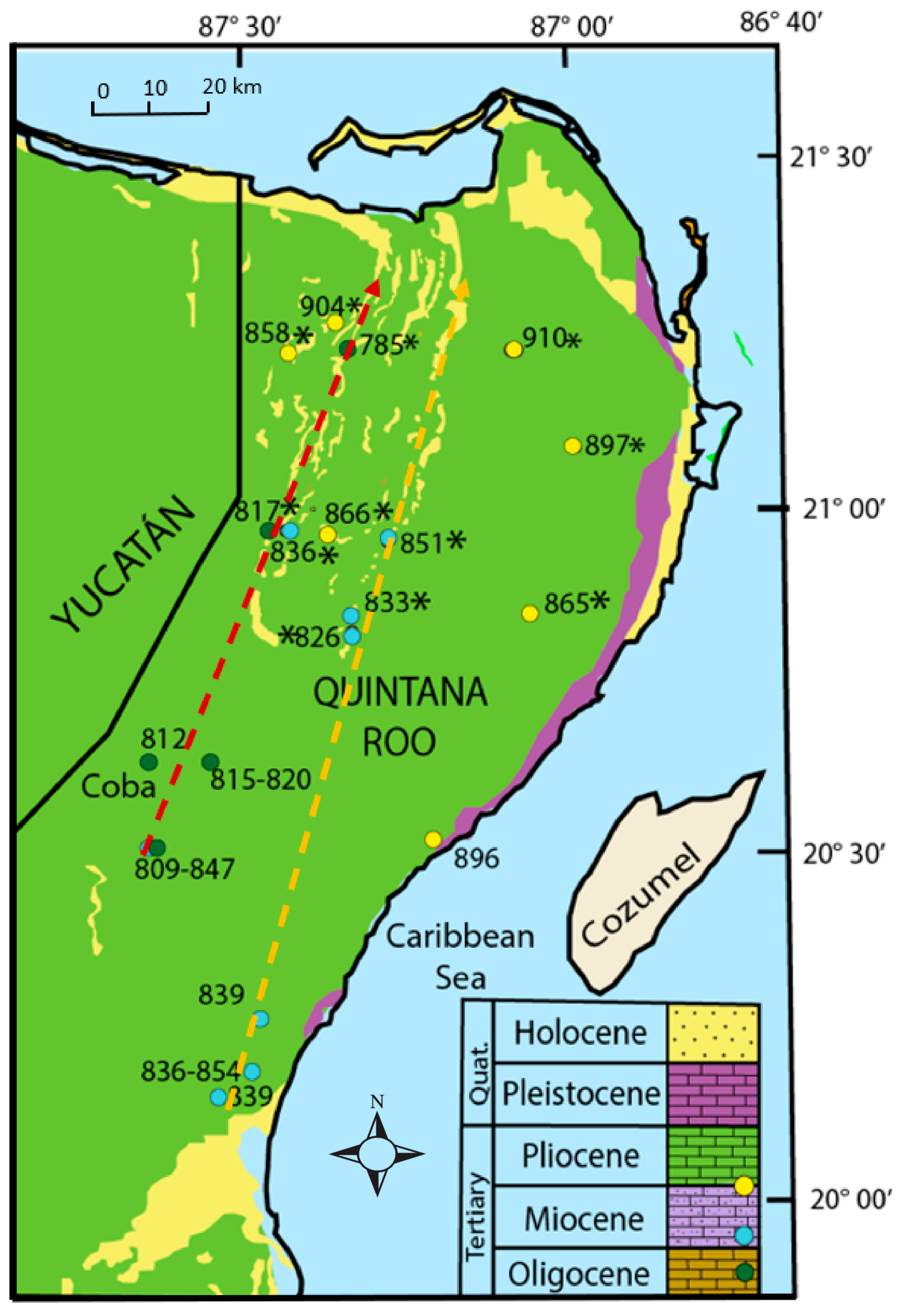
1.1.2. Geology and Hydrogeology
2. Materials and Methods
2.1. Water Sample Collection
2.2. Physiochemical Properties
2.3. Major Ion Geochemistry
2.4. Strontium
3. Results
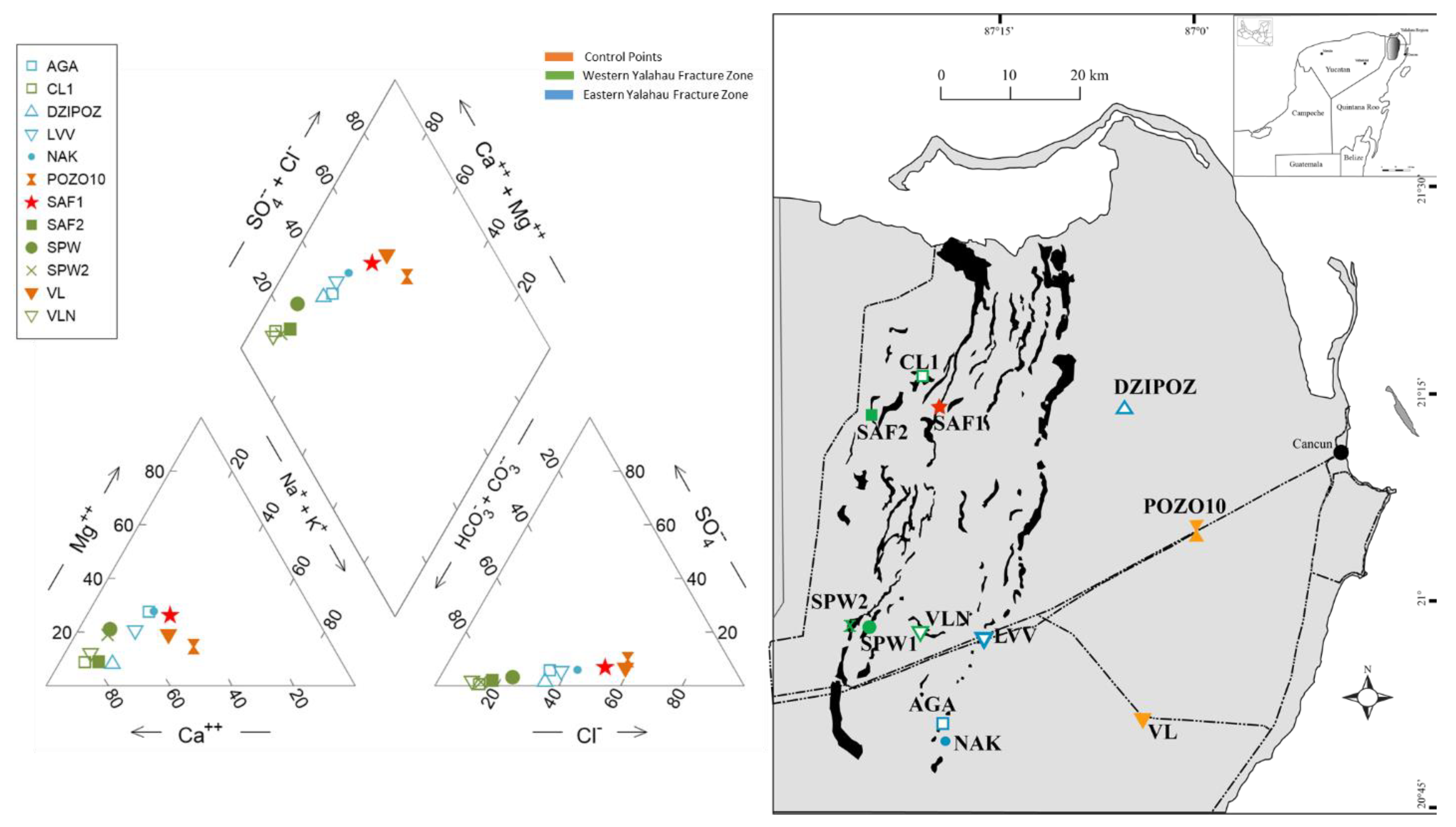
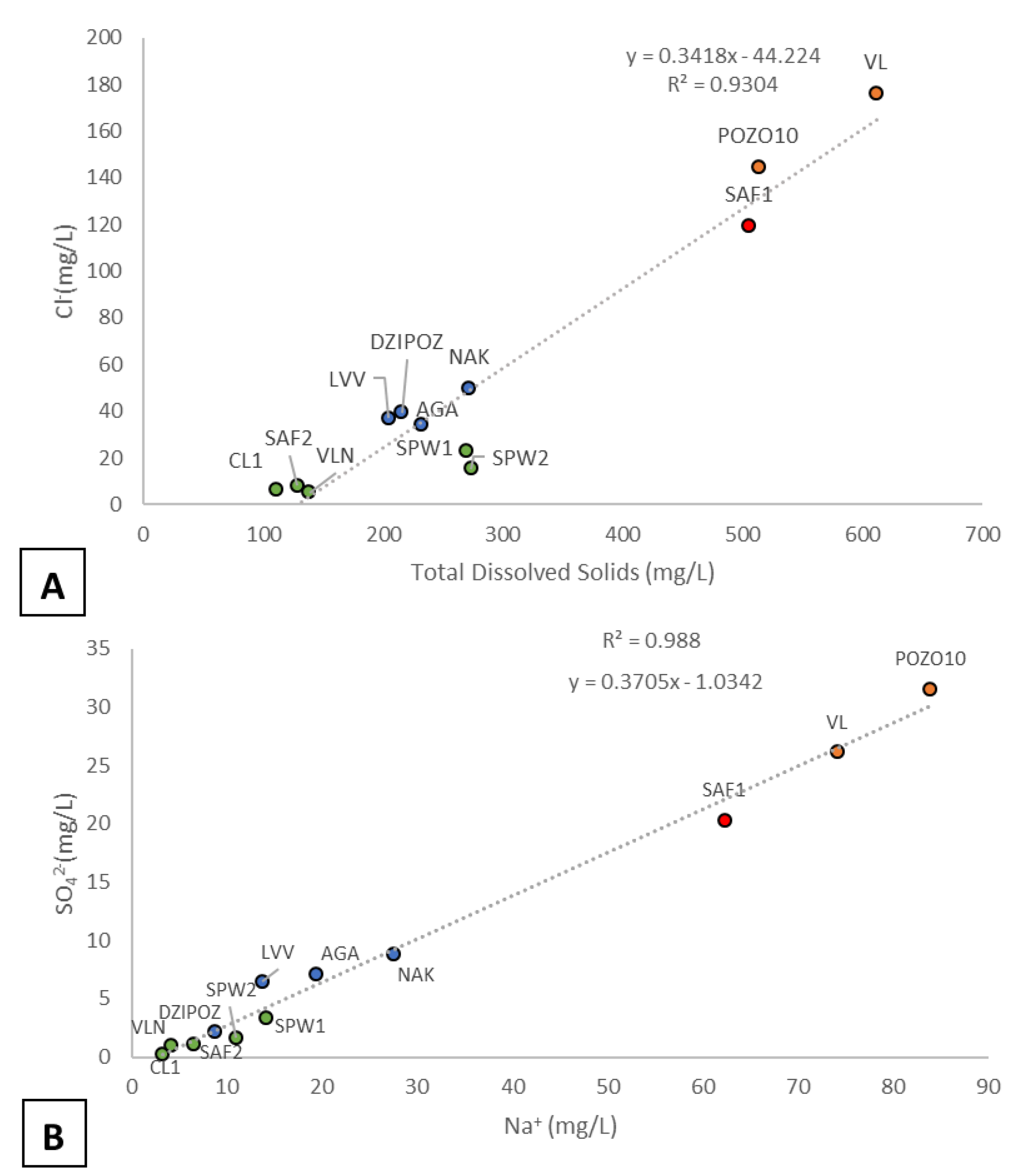
3.1. Major Ion Geochemistry
3.2. Control Points (CP)
3.3. Western Yalahau Fracture Zone (WYFZ)
3.4. Site SAF1
3.5. Eastern Yalahau Fracture Zone (EYFZ)
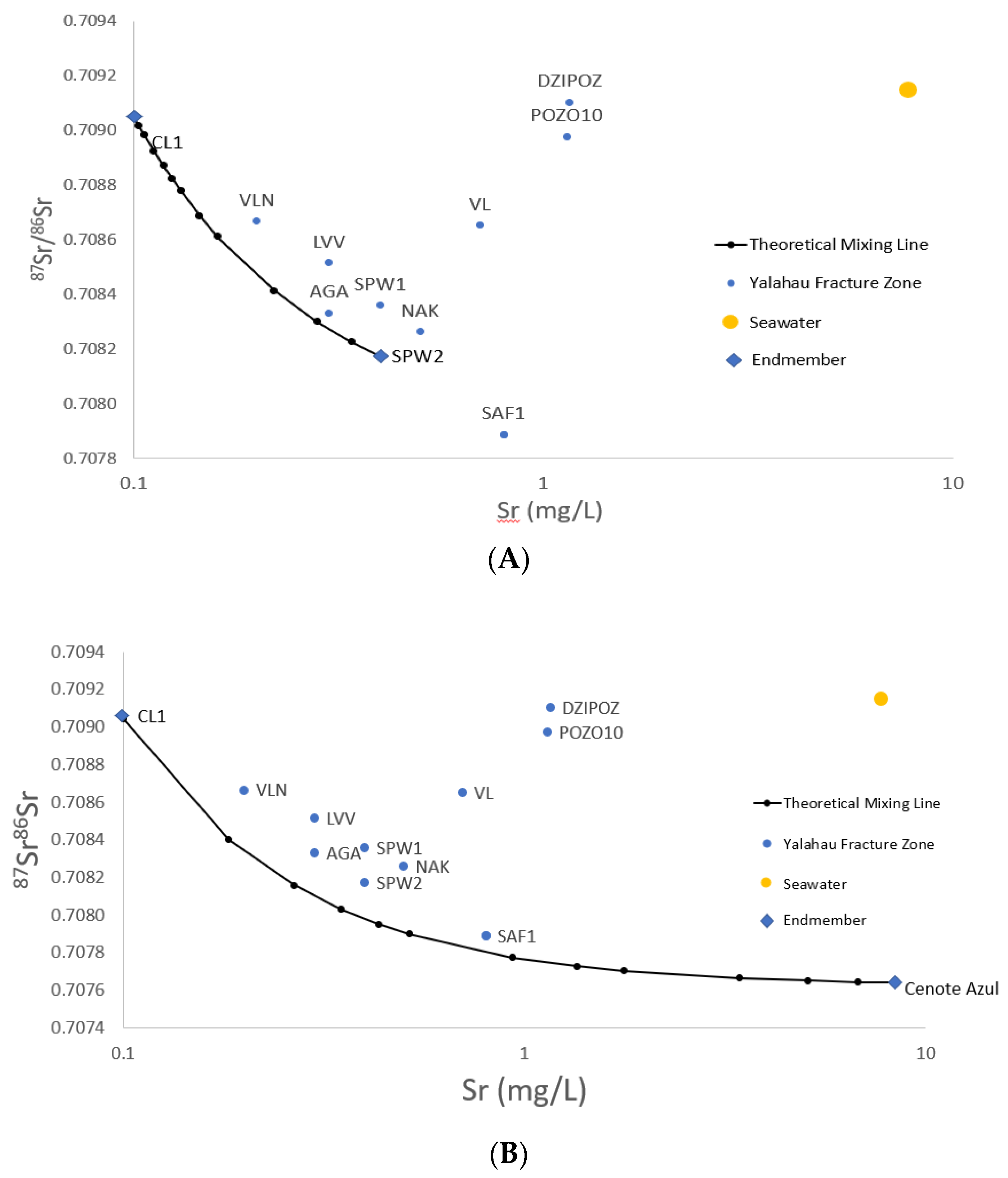
3.6. 87Sr/86Sr
4. Discussion
4.1. Hydrogeochemistry
4.2. 87Sr/86Sr
4.3. Sr Endmember Mixing Model
SrM = SrAfa + SrB(1 − fa)
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weidie, A.E. Geology of Yucatan Platform. In Geology and Hydrogeology of the Yucatan and Quaternary Geology of Northeastern Yucatan Peninsula; Ward, W.C., Weidie, A.E., Back, W., Eds.; New Orleans Geological Society: New Orleans, LA, USA, 1985. [Google Scholar]
- Weidie, A.E. (Ed.) Lineaments on the Yucatan Peninsula and Fractures of the Central Quintana Roo Coast: Road Log and Supplement to 1978; Guidebook, Field trip no. 10, Yucatan; Geological Society of America Meeting (GSA): Washington, DC, USA, 1982; pp. 21–25. [Google Scholar]
- Southworth, C.S. Applications of remote sensing data, eastern Yucatan. In Geology and Hydrogeology of the Yucatan and Quaternary Geology of Northeastern Yucatan Peninsula; Weidie, A.E., Ed.; New Orleans Geological Society: New Orleans, LA, USA, 1985; pp. 12–19. [Google Scholar]
- Fedick, S.L.; Morrison, B.A.; Andersen, B.J.; Boucher, S.; Acosta, J.C.; Mathews, J.P. Wetland Manipulation in the Yalahau Region of the Northern Maya Lowlands. J. Field Archaeol. 2000, 27, 131. [Google Scholar] [CrossRef]
- Tulaczyk, S.M. Karst Geomorphology and Hydrogeology of the Northeastern Yucatán Peninsula, Mexico. Master’s Thesis, Northern Illinois University, DeKalb, IL, USA, 1993. [Google Scholar]
- Perry, E.; Velazquez-Oliman, G.; Marín, L. The Hydrogeochemistry of the Karst Aquifer System of the Northern Yucatan Peninsula, Mexico. Int. Geol. Rev. 2002, 44, 191–221. [Google Scholar] [CrossRef]
- Perry, E.; Paytan, A.; Pedersen, B.; Velazquez-Oliman, G. Groundwater geochemistry of the Yucatan Peninsula, Mexico: Constraints on stratigraphy and hydrogeology. J. Hydrol. 2009, 367, 27–40. [Google Scholar] [CrossRef]
- Gondwe, B.R.N.; Hong, S.-H.; Wdowinski, S.; Bauer-Gottwein, P. Hydrologic Dynamics of the Ground-Water-Dependent Sian Ka’an Wetlands, Mexico, Derived from InSAR and SAR Data. Wetlands 2010, 30, 1–13. [Google Scholar] [CrossRef]
- Solleiro-Rebolledo, E.; Cabadas-Báez, H.; Pi, P.; Gonzalez, A.; Fedick, S.; Chmilar, J.; Leonard, D. Genesis of hydromorphic Calcisols in wetlands of the northeast Yucatan Peninsula, Mexico. Geomorphology 2011, 135, 322–331. [Google Scholar] [CrossRef]
- Fedick, S.L.; Mathews, J.P. The Yalahau regional human ecology project: An introduction and summary of recent research. In Quintana Roo Archaeology; Shaw, J.M., Mathews, J.P., Eds.; University of Arizona Press: Tucson, AZ, USA, 2005; pp. 33–50. [Google Scholar]
- Glover, J.B. The Yalahau Regional Settlement Pattern Survey: A Study of Ancient Maya Social Organization in Northern Quintana Roo, Mexico. Ph.D. Thesis, University of California, Riverside, CA, USA, 2006. [Google Scholar]
- Connors, M.; Hildebrand, A.R.; Pilkington, M.; Ortiz-Aleman, C.; Chávez, R.E.; Urrutia-Fucugauchi, J.; Graniel-Castro, E.; Camara-Zi, A.; Vasquez, J.; Halpenny, J.F. Yucatán karst features and the size of Chicxulub crater. Geophys. J. Int. 1996, 127, F11–F14. [Google Scholar] [CrossRef]
- Rubio-Cisneros, N.; Herrera-Silveira, J.; Morales-Ojeda, S.; Moreno-Báez, M.; Montero, J.; Pech-Cárdenas, M. Water quality of inlets’ water bodies in a growing touristic barrier reef Island “Isla Holbox” at the Yucatan Peninsula. Reg. Stud. Mar. Sci. 2018, 22, 112–124. [Google Scholar] [CrossRef]
- Bauer-Gottwein, P.; Gondwe, B.R.N.; Charvet, G.; Marín, L.E.; Rebolledo-Vieyra, M.; Merediz-Alonso, G. Review: The Yucatán Peninsula karst aquifer, Mexico. Hydrogeol. J. 2011, 19, 507–524. [Google Scholar] [CrossRef]
- Reeve, A.; Perry, E.C. Hydrogeology and tidal analysis along the western north coast of the Yucatan Peninsula, Mexico. In Tropical Hydrology and Caribbean Water Resources: Proceedings of the International Symposium on Tropical Hydrology and Fourth Caribbean Islands Water Resources Congress, San Juan, PR, USA, 22–27 July 1990; Krishna, J.H., Ed.; American Water Resources Association: Bethesda, MD, USA, 2000; pp. 327–337. [Google Scholar]
- Perry, E.C.; Velazquez-Oliman, G.; Socki, R. The hydrogeology of the Yucatan Peninsula. In Hydrogeology of the Yucatan Peninsula, 21st Symposium on Plant Biology; Pompa, A.G., Fedick, S., Eds.; The Haworth Press: Binghamton, NY, USA, 2001. [Google Scholar]
- Beddows, P.A.; Smart, P.L.; Whitaker, F.F.; Smith, S.L. Decoupled fresh-saline groundwater circulation of a coastal carbonate aquifer: Spatial patterns of temperature and specific electrical conductivity. J. Hydrol. 2007, 346, 18–32. [Google Scholar] [CrossRef]
- Supper, R.; Motschka, K.; Ahl, A.; Bauer-Gottwein, P.; Gondwe, B.; Alonso, G.M.; Römer, A.; Ottowitz, D.; Kinzelbach, W. Spatial mapping of submerged cave systems by means of airborne electromagnetics: An emerging technology to support protection of endangered karst aquifers. Near Surf. Geophys. 2009, 7, 613–627. [Google Scholar] [CrossRef]
- Gondwe, B.R.N.; Ottowitz, D.; Supper, R.; Motschka, K.; Merediz-Alonso, G.; Bauer-Gottwein, P. Regional-scale airborne electromagnetic surveying of the Yucatan karst aquifer (Mexico): Geological and hydrogeological interpretation. Hydrogeol. J. 2012, 20, 1407–1425. [Google Scholar] [CrossRef]
- Mongelli, G.; Monni, S.; Oggiano, G.; Paternoster, M.; Sinisi, R. Tracing groundwater salinization processes in coastal aquifers: A hydrogeochemical and isotopic approach in the Na-Cl brackish waters of northwestern Sardinia, Italy. Hydrol. Earth Syst. Sci. 2013, 17, 2917–2928. [Google Scholar] [CrossRef]
- Cary, L.; Benabderraziq, H.; Elkhattabi, J.; Gourcy, L.; Parmentier, M.; Picot, J.; Khaska, M.; Laurent, A.; Négrel, P. Tracking selenium in the Chalk aquifer of northern France: Sr isotope constraints. Appl. Geochem. 2014, 48, 70–82. [Google Scholar] [CrossRef]
- Servicio Geológico Mexicano (SGM). Carta Geológico-Minera, Estados de: Campeche, Quintana Roo y Yucatán, escala 1:500000: 1 mapa. 2017. Available online: www.coremisgm.gob.mx (accessed on 1 May 2020).
- Moore, Y.H.; Stoessell, R.K.; Easley, D.H. Fresh-Water/Sea-Water Relationship Within a Ground-Water Flow System, Northeastern Coast of the Yucatan Peninsula. Ground Water 1992, 30, 343–350. [Google Scholar] [CrossRef]
- Perry, E.C.; Velazquez-Oliman, G. The hydrogeology of the northern Yucatan Peninsula, Mexico, with special reference to coastal processes. In Ground Water Discharge in the Coastal Zone: Proceedings of an International Symposium; Buddemeier, R.W., Ed.; LOICZ (Land Ocean Interactions in the Coastal Zone): Den Burg, The Netherlands, 1996; pp. 92–97. [Google Scholar]
- Neumann, K.; Dreiss, S. Strontium 87/strontium 86 rations as tracers in groundwaters and surface waters in Mono Basin, California. Water Resour. Res. 1995, 23, 3183–3193. [Google Scholar] [CrossRef]
- Katz, B.G.; Bullen, T.D. The combined use of 87Sr/86Sr and carbon and water isotopes to study the hydrogeochemical interaction between groundwater and lakewater in mantled karst. Geochim. Cosmochim. Atca 1996, 60, 5075–5087. [Google Scholar] [CrossRef]
- Dogramaci, S.; Herczeg, A.L. Strontium and carbon isotope constraints on carbonate-solution interactions and inter-aquifer mixing in groundwaters of the semi-arid Murray Basin, Australia. J. Hydrol. 2002, 262, 50–67. [Google Scholar] [CrossRef]
- Frost, C.D.; Toner, R.N. Strontium Isotopic Identification of Water-Rock Interaction and Ground Water Mixing. Ground Water 2004, 42, 418–432. [Google Scholar] [CrossRef]
- Negrel, P.; Petelet-Giraud, E.; Widory, D. Strontium isotope geochemistry of alluvial groundwater: A tracer for groundwater resources characterisation. Hydrol. Earth Syst. Sci. 2004, 8, 959–972. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, Q.; Su, C.; Ma, T. Strontium isotope characterization and major ion geochemistry of karst water flow, Shentou, northern China. J. Hydrol. 2006, 328, 592–603. [Google Scholar] [CrossRef]
- Shand, P.; Love, A.J.; Darbyshire, D.P.F.; Edmunds, W.M. Sr isotopes in natural waters: Applications to source characterization and water–rock interaction in contrasting landscapes. In Water-Rock Interaction; Bullen, T.D., Wang, Y., Eds.; Taylor and Francis: London, UK, 2007; pp. 27–35. [Google Scholar]
- Beddows, P.A. Groundwater Hydrology of a Coastal Conduit Carbonate Aquifer: Caribbean Coast of the Yucatán Peninsula, México. Ph.D. Thesis, University of Bristol, Bristol, UK, 2004. [Google Scholar]
- Peterman, Z.E.; Wallin, B. Synopsis of strontium isotope variations in groundwater at Äspö, southern Sweden. Appl. Geochem. 1999, 14, 939–951. [Google Scholar] [CrossRef]
- Curtis, J.B., Jr.; Stueber, A.M. Sr/Sr Ratios and Toal Strontium Concentrations in Surface Waters of the Scioto River Drainage Basin, Ohio. Ohio J. Sci. 1973, 73, 166–175. [Google Scholar]
- Santoni, S.; Huneau, F.; Garel, E.; Aquilina, L.; Vergnaud-Ayraud, V.; Labasque, T.; Celle-Jeanton, H. Strontium isotopes as tracers of water-rocks interactions, mixing processes and residence time indicator of groundwater within the granite-carbonate coastal aquifer of Bonifacio (Corsica, France). Sci. Total. Environ. 2016, 573, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Glover, J.B.; Rissolo, D.; Mathews, J.P. The Hidden World of the Maritime Maya: Lost Landscapes along the North Coast of Quintana Roo, Mexico. In The Archaeology of Maritime Landscapes; Ford, B., Ed.; Springer: New York, NY, USA, 2011; Volume 2, pp. 195–216. [Google Scholar]
- SEMARNAT. 2019. Available online: http://dgeiawf.semarnat.gob.mx:8080/ibi_apps/WFServlet?IBIF_ex=D3_AGUA01_01&IBIC_user=dgeia_mce&IBIC_pass=dgeia_mce&NOMBREENTIDAD=*&NOMBREANIO=* (accessed on 5 August 2020).
- Jáuregui, E. Climatology and landfalling hurricanes and tropical storms in Mexico. Atmósfera 2003, 16, 193–204. [Google Scholar]
- Hodell, D.; Brenner, M.; Curtis, J.H. Climate and cultural history of the Northeastern Yucatan Peninsula, Quintana Roo, Mexico. Clim. Chang. 2007, 83, 215–240. [Google Scholar] [CrossRef]
- Beddows, P.A. An Introduction to the Yucatan Peninsula Hydrogeology: A World Class Example of a Coastal Carbonate Density Stratified Aquifer. In Cavern Guide Training Program; Asociación de Prestadores de Servícios Acuáticos: Riviera Maya, Mexico, 2003. [Google Scholar]
- Perry, E.; Marin, L.; McClain, J.; Velazquez, G. Ring of Cenotes (sinkholes), northwest Yucatan, Mexico: Its hydrogeologic characteristics and possible association with the Chicxulub impact crater. Geology 1995, 23. [Google Scholar] [CrossRef]
- López-Ramos, E. Estudio geológico de la península de Yucatán. In Boletín de la Asociación Mexicana de Geólogos Petroleros; Asociación Mexicana de Geólogos Petroleros: San Bartolo, Mexico, 1973; Volume 25, pp. 23–76. [Google Scholar]
- Schönian, F.; Tagle, R.; Stöffler, D.; Kenkmann, T. Geology of Southern Quintana Roo (Mexico) and the Chicxulub Ejecta Blanket. In Proceedings of the 36th Lunar and Planetary Science Conference, League City, TX, USA, 14–18 March 2005; p. 2389. Available online: http://www.liber-lapidum.net/frank/frank.html (accessed on 1 May 2020).
- CONAGUA. Determinación de La Disponibilidad Del Agua En El Acuífero de La Península de Yucatán [Determination of the Water Availability at the Yucatan Peninsula Aquifer]; CONAGUA: Ciudad de México, México, 2002; pp. 1–20. [Google Scholar]
- Rebolledo-Vieyra, M.; Urrutia-Fucugauchi, J.; Trejo-Garcia, A.; Marin, L.; Sharpton, V.; Soler, A. UNAM’s Scientific Shallow Drilling Program into the Chicxulub impact crater. Int. Geol. Rev. 2000, 42, 928–940. [Google Scholar] [CrossRef]
- Housh, T.; McMahon, T.P. Sr, Nd and Pb Isotopic Characteristics of Late Miocene Pleistocene Collision-Related Igneous Rocks in Western New Guinea (Irian Jaya, Indonesia). Lithos 2000, 50, 217–222. [Google Scholar]
- Ghosh, M.J.; Padhy, P.K. Characterization and Classification of Hydrochemistry using Mulitvariate Graphical and Hydrostatistical Techniques. Res. J. Chem. Sci. 2013, 3, 32–42. [Google Scholar]
- McArthur, J.M.; Howarth, R.J.; Bailey, T. Strontium Isotope Stratigraphy: LOWESS Version 3: Best Fit to the Marine Sr-Isotope Curve for 0–509 Ma and Accompanying Look-up Table for Deriving Numerical Age. J. Geol. 2001, 109, 155–170. [Google Scholar] [CrossRef]
- Han, G.; Liu, C.-Q. Water geochemistry controlled by carbonate dissolution: A study of the river waters draining karst-dominated terrain, Guizhou Province, China. Chem. Geol. 2004, 204, 1–21. [Google Scholar] [CrossRef]
- Fitts, C.R. Groundwater Science, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Faure, G.; Mensing, T.M. Isotope Principle and Applications, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Tran, K.C.; Valdés, D.; Euan, J.; Real, E.; Gil, E. Status of water quality at Holbox Island, Quintana Roo State, Mexico. Aquat. Ecosyst. Heal. Manag. 2002, 5, 173–189. [Google Scholar] [CrossRef]
- Leal-Bautista, R.M.; Lenczewski, M.; Morgan, C.; Gahala, A.; McLain, J.E.; Ma., L.-B.R.; Melissa, L.; Cheyenne, M.; Gahala, A.; McLain, J.E. Assessing Fecal Contamination in Groundwater from the Tulum Region, Quintana Roo, Mexico. J. Environ. Prot. 2013, 4, 1272–1279. [Google Scholar] [CrossRef][Green Version]
| Site Name | Location (UTM) | T °C | pH | EC | DO | ORP | HCO3− | SO42− | Cl− | Ca2+ | Mg2+ | K+ | Na+ | Sr2+ | TDS | 87Sr/86Sr | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Western Yalahau Fracture Zone | CL1 | 463748.72 2352285.01 | 28.6 | 7.67 | 159.3 | 2.83 | 146.3 | 67.7 | 0.215 | 6.439049 | 30.047 | 1.824 | 0.376 | 3.26 | 0.1 | 111.4 | 0.709046 |
| SAF1 | 465700.33 2348530.16 | 30.9 | 7.83 | 481 | 6.26 | 107.6 | 166.3 | 20.284 | 119.0887 | 95.713 | 32.428 | 1.939 | 62.342 | 0.8 | 505.4 | 0.707888 | |
| SAF2 | 456503.27 2347660.33 | 32.5 | 7.74 | 120.2 | 3.25 | 89.6 | 64 | 1.029 | 8.124794 | 42.241 | 2.889 | 2.205 | 6.535 | <0.1 | 128.7 | 0.70858 | |
| SPW1 | 456140.19 2319407.87 | 29.5 | 7.56 | 156.9 | 2.31 | 132.3 | 123.2 | 3.303 | 23.05512 | 85.467 | 15.323 | 1.003 | 14.237 | 0.4 | 269.4 | 0.70836 | |
| SPW2 | 454049.82 2319652.57 | 28.7 | 7.51 | 240 | 1.81 | 99.8 | 156.5 | 1.654 | 15.40646 | 71.514 | 11.461 | 2.239 | 10.96 | 0.4 | 69 | 0.708175 | |
| VLN | 463089.33 2318736.03 | 29.7 | 7.56 | 156.9 | 2.51 | 132.3 | 69 | 0.976 | 5.060984 | 49.568 | 4.44 | 3.196 | 4.19 | 0.2 | 138.4 | 0.708667 | |
| Eastern Yalahau Fracture Zone | AGA | 466097.06 2306386 | 28.1 | 8.23 | 234 | 7.3 | 128.3 | 103.4 | 7.072 | 34.13449 | 48.096 | 15.008 | 1.1916 | 19.451 | 0.3 | 232.3 | 0.708331 |
| LVV | 471528.2 2317978.6 | 30.5 | 7.67 | 245 | 2.51 | 77.5 | 94.9 | 6.445 | 36.92464 | 40.564 | 8.119 | 0.898 | 13.719 | 0.3 | 204.5 | 0.708515 | |
| NAK | 466324.54 2303992.03 | 28.7 | 7.83 | 291 | 5.79 | 74.6 | 102.1 | 8.752 | 49.7406 | 59.55 | 19.497 | 0.974 | 27.574 | 0.5 | 272.1 | 0.708263 | |
| DZIPOZ | 490805.17 23458004.94 | 21.3 | 7.75 | 275 | 4.77 | 106.6 | 124.4 | 2.148 | 39.3466 | 34.204 | 2.303 | 0.504 | 8.851 | 1.16 | 215.6 | 0.709103 | |
| Control Points | POZO10 | 500618.52 2332227.56 | 27.5 | 7.54 | 496 | 5.88 | 192.8 | 142.8 | 31.546 | 144.3393 | 83.272 | 16.045 | 4.02 | 83.94 | 1.14 | 513.7 | 0.708976 |
| VL | 493131.22 2306713.65 | 26.5 | 7.56 | 591 | 2.63 | 162.3 | 184.8 | 26.148 | 176.0915 | 112.941 | 25.131 | 3.833 | 74.177 | 0.7 | 611.7 | 0.709667 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McKay, J.; Lenczewski, M.; Leal-Bautista, R.M. Characterization of Flowpath Using Geochemistry and 87Sr/86Sr Isotope Ratios in the Yalahau Region, Yucatan Peninsula, Mexico. Water 2020, 12, 2587. https://doi.org/10.3390/w12092587
McKay J, Lenczewski M, Leal-Bautista RM. Characterization of Flowpath Using Geochemistry and 87Sr/86Sr Isotope Ratios in the Yalahau Region, Yucatan Peninsula, Mexico. Water. 2020; 12(9):2587. https://doi.org/10.3390/w12092587
Chicago/Turabian StyleMcKay, Jessica, Melissa Lenczewski, and Rosa Maria Leal-Bautista. 2020. "Characterization of Flowpath Using Geochemistry and 87Sr/86Sr Isotope Ratios in the Yalahau Region, Yucatan Peninsula, Mexico" Water 12, no. 9: 2587. https://doi.org/10.3390/w12092587
APA StyleMcKay, J., Lenczewski, M., & Leal-Bautista, R. M. (2020). Characterization of Flowpath Using Geochemistry and 87Sr/86Sr Isotope Ratios in the Yalahau Region, Yucatan Peninsula, Mexico. Water, 12(9), 2587. https://doi.org/10.3390/w12092587





