Review of Methods to Repair and Maintain Lithophilic Fish Spawning Habitat
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
4.1. Loss of Material
4.2. Sedimentation
4.3. Biofouling
4.3.1. Periphyton
4.3.2. Invertebrates
4.4. Novel Remediation Techniques
4.5. Physical Monitoring Techniques
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Family | Common Name | Scientific Name |
|---|---|---|
| Acipenseridae | Lake sturgeon | Acipenser fulvescens |
| Acipenseridae | White sturgeon | Acipenser transmontanus |
| Catostomidae | Black redhorse | Moxostoma duquesni |
| Catostomidae | Northern hogsucker | Hypentelium nigricans |
| Cyprinidae | Common barbel | Barbus barbus |
| Cyprinidae | River chub | Nocomis micropogon |
| Eleotridae | Carp gudgeon | Hypseleotris spp. |
| Eleotridae | Flathead gudgeon | Philypnodon grandiceps |
| Percichthyidae | River blackfish | Gadopsis marmoratus |
| Percidae | Walleye | Sander vitreus |
| Petromyzontidae | Brook lamprey | Lampetra planeri |
| Salmonidae | Atlantic salmon | Salmo salar |
| Salmonidae | Bonneville cutthroat trout | Oncorhynchus clarkii utah |
| Salmonidae | Brook trout | Salvelinus fontinalis |
| Salmonidae | Brown trout | Salmo trutta |
| Salmonidae | Chinook salmon | Oncorhynchus tshawytscha |
| Salmonidae | Chum salmon | Oncorhynchus keta |
| Salmonidae | Cisco | Coregonus artedi |
| Salmonidae | Coho salmon | Oncorhynchus kisutch |
| Salmonidae | Cutthroat trout | Oncorhynchus clarkii |
| Salmonidae | Grayling | Thymallus thymallus |
| Salmonidae | Lake trout | Salvelinus namaycush |
| Salmonidae | Lake whitefish | Coregonus clupeaformis |
| Salmonidae | Masu salmon | Oncorhynchus masou |
| Salmonidae | Pink salmon | Oncorhynchus gorbuscha |
| Salmonidae | Rainbow trout | Oncorhynchus mykiss |
| Salmonidae | Sockeye salmon | Oncorhynchus nerka |
References
- Brickhill, M.J.; Lee, S.Y.; Connolly, R.M. Fishes associated with artificial reefs: Attributing changes to attraction or production using novel approaches. J. Fish Biol. 2005, 67, 53–71. [Google Scholar] [CrossRef]
- Gunderson, D.R.; Parma, A.M.; Hilborn, R.; Cope, J.M.; Fluharty, D.L.; Miller, M.L.; Vetter, R.D.; Heppell, S.S.; Greene, G.H. The challenge of managing nearshore rocky reef resources. Fisheries 2008, 33, 172–179. [Google Scholar] [CrossRef]
- Kennedy, D. Year of the Reef. Science 2007, 318, 1695–1696. [Google Scholar] [CrossRef] [PubMed]
- Bouckaert, E.K.; Auer, N.A.; Roseman, E.F.; Boase, J. Verifying success of artificial spawning reefs in the St. Clair-Detroit River System for lake sturgeon (Acipenser fulvescens Rafinesque, 1817). J. Appl. Ichthyol. 2014, 30, 1393–1401. [Google Scholar] [CrossRef]
- Riley, S.C.; Marsden, J.E.; Ridgway, M.S.; Konrad, C.P.; Farha, S.A.; Binder, T.R.; Middel, T.A.; Esselman, P.C.; Krueger, C.C. A conceptual framework for the identification and characterization of lacustrine spawning habitats for native lake charr Salvelinus namaycush. Environ. Biol. Fishes 2019, 102, 1533–1557. [Google Scholar] [CrossRef]
- Roseman, E.F.; Kennedy, G.W.; Boase, J.; Manny, B.A.; Todd, T.N.; Stott, W. Evidence of lake whitefish spawning in Detroit River: Implications for habitat and population recovery. J. Gt. Lakes Res. 2007, 33, 397–406. [Google Scholar] [CrossRef]
- Shuter, B.J. Population-level indicators of stress. Am. Fish. Soc. Symp. 1990, 8, 145–166. [Google Scholar]
- Hayes, D. Issues affecting fish habitat in the Great Lakes Basin. In Great Lakes Fisheries Policy and Management: A Binational Perspective; Taylor, W.W., Ferreri, C.P., Eds.; Michigan State University Press: East Lansing, MI, USA, 1999; pp. 207–237. [Google Scholar]
- Hendry, K.; Cragg-Hine, D.; O’Grady, M.; Sambrook, H.; Stephen, A. Management of habitat for rehabilitation and enhancement of salmonid stocks. Fish. Res. 2003, 62, 171–192. [Google Scholar] [CrossRef]
- Nagata, M.; Omori, H.; Yanai, S. Restoration of spawning and rearing habitats for masu salmon, Oncorhynchus masou in a channelized stream. Fish. Sci. 2002, 68, 1707–1710. [Google Scholar] [CrossRef][Green Version]
- Baine, M. Artificial reefs: A review of their design, application, management and performance. Ocean Coast. Manag. 2001, 44, 241–259. [Google Scholar] [CrossRef]
- Bohnsack, J.A.; Sutherland, D.L. Artificial reef research: A review with recommendations for future priorities. Bull. Mar. Sci. 1985, 37, 11–39. [Google Scholar]
- Miller, R.R.; Williams, J.D.; Williams, J.E. Extinctions of North American fishes during the past century. Fisheries 1989, 14, 22–38. [Google Scholar] [CrossRef]
- Gladstone, W. Requirements for marine protected areas to conserve the biodiversity of rocky reef fishes. Aquat. Conserv. Mar. Freshw. Ecosyst. 2006, 17, 71–87. [Google Scholar] [CrossRef][Green Version]
- Airoldi, L. The effects of sedimentation on rocky coast assemblages. Oceanogr. Mar. Biol. Annu. Rev. 2003, 41, 161–236. [Google Scholar]
- Connell, S.D. Assembly and maintenance of subtidal habitat heterogeneity: Synergistic effects of light penetration and sedimentation. Mar. Ecol. Prog. Ser. 2005, 289, 53–61. [Google Scholar] [CrossRef]
- Kemp, P.; Sear, D.; Collins, A.; Naden, P.; Jones, I. The impacts of fine sediment on riverine fish. Hydrol. Process. 2011, 25, 1800–1821. [Google Scholar] [CrossRef]
- Bennion, D.H.; Manny, B.A. Construction of Shipping Channels in the Detroit River: History and Environmental Consequences; US Geological Survey: Ann Arbor, MI, USA, 2011.
- Rutherford, E.S.; Marshall, E.; Clapp, D.; Horns, W.; Gorenflo, T.; Trudeau, T. Lake Michigan Environmental Objectives; Great Lakes Fishery Commission: Ann Arbor, MI, USA, 2004; p. 81. [Google Scholar]
- Brown, A.V.; Lyttle, M.M.; Brown, K.B. Impacts of gravel mining on gravel bed streams. Trans. Am. Fish. Soc. 1998, 127, 979–994. [Google Scholar] [CrossRef]
- Kondolf, G.M. Hungry water: Effects of dams and gravel mining on river channels. Environ. Manag. 1997, 21, 533–551. [Google Scholar] [CrossRef]
- Støttrup, J.G.; Dahl, K.; Niemann, S.; Stenberg, C.; Reker, J.; Stamphøj, E.M.; Göke, C.; Svendsen, J.C. Restoration of a boulder reef in temperate waters: Strategy, methodology and lessons learnt. Ecol. Eng. 2017, 102, 468–482. [Google Scholar] [CrossRef]
- Bickel, T.O.; Closs, G.P. Impact of Didymosphenia geminata on hyporheic conditions in trout redds: Reason for concern? Mar. Freshw. Res. 2008, 59, 1028–1033. [Google Scholar] [CrossRef]
- Johnson, J.H.; LaPan, S.R.; Klindt, R.M.; Schiavone, A. Lake sturgeon spawning on artificial habitat in the St Lawrence River. J. Appl. Ichthyol. 2006, 22, 465–470. [Google Scholar] [CrossRef]
- Rutecki, T.L.; Dorr, J.A.; Jude, D.J. Preliminary analysis of colonization and succession of selected algae, invertebrates, and fish on two artificial reefs in inshore southeastern Lake Michigan. In Artificial Reefs: Marine and Freshwater Applications; D’Itri, F.M., Ed.; Lewis Publishers, Inc.: Chelsea, MI, USA, 1985; pp. 459–489. [Google Scholar]
- Leach, J.H. Biota of Lake St. Clair: Habitat evaluation and environmental assessment. Hydrobiologia 1991, 219, 187–202. [Google Scholar] [CrossRef]
- Marsden, J.E.; Chotkowski, M.A. Lake trout spawning on artificial reefs and the effect of zebra mussels: Fatal attraction? J. Gt. Lakes Res. 2001, 27, 33–43. [Google Scholar] [CrossRef]
- Evans, E.P.; Simm, J.D.; Thorne, C.R.; Arnell, N.W.; Ashley, R.M.; Hess, T.; Lane, S.; Morris, J.; Nicholls, R.; Penning-Rowsell, E.; et al. An Update of the Foresight Future Flooding 2004 Qualitative Risk Analysis; Cabinet Office: London, UK, 2008.
- Marsden, J.E.; Perkins, D.L.; Krueger, C.C. Recognition of spawning areas by lake trout: Deposition and survival of eggs on small, man-made rock piles. J. Gt. Lakes Res. 1995, 21, 330–336. [Google Scholar] [CrossRef]
- Gatch, A.J.; Koenigbauer, S.T.; Roseman, E.F.; Höök, T.O. The effect of sediment cover and female characteristics on the hatching success of walleye. N. Am. J. Fish. Manag. 2020, 40, 293–302. [Google Scholar] [CrossRef]
- Sly, P.G. Interstitial water quality of lake trout spawning habitat. J. Gt. Lakes Res. 1988, 14, 301–315. [Google Scholar] [CrossRef]
- Claramunt, R.M.; Jonas, J.L.; Fitzsimons, J.D.; Marsden, J.E. Influences of spawning habitat characteristics and interstitial predators on lake trout egg deposition and mortality. Trans. Am. Fish. Soc. 2005, 134, 1048–1057. [Google Scholar] [CrossRef]
- Bennion, D.H.; Manny, B.A. A model to locate potential areas for lake sturgeon spawning habitat construction in the St. Clair-Detroit River System. J. Gt. Lakes Res. 2014, 40, 43–51. [Google Scholar] [CrossRef]
- Binder, T.R.; Farha, S.A.; Thompson, H.T.; Holbrook, C.M.; Bergstedt, R.A.; Riley, S.C.; Bronte, C.R.; He, J.; Krueger, C.C. Fine-scale acoustic telemetry reveals unexpected lake trout, Salvelinus namaycush, spawning habitats in northern Lake Huron, North America. Ecol. Freshw. Fish 2018, 27, 594–605. [Google Scholar] [CrossRef]
- Gannon, J. International Position Statement and Evaluation Guidelines for Artificial Reef Development in the Great Lakes; Great Lakes Fishery Commission: Ann Arbor, MI, USA, 1990. [Google Scholar]
- Manny, B.A.; Roseman, E.F.; Kennedy, G.; Boase, J.C.; Craig, J.M.; Bennion, D.H.; Read, J.; Vaccaro, L.; Chiotti, J.; Drouin, R.; et al. A scientific basis for restoring fish spawning habitat in the St. Clair and Detroit Rivers of the Laurentian Great Lakes. Restor. Ecol. 2015, 23, 149–156. [Google Scholar] [CrossRef]
- Marsden, J.E.; Binder, T.R.; Johnson, J.; He, J.; Dingledine, N.; Adams, J.; Johnson, N.S.; Buchinger, T.J.; Krueger, C.C. Five-year evaluation of habitat remediation in Thunder Bay, Lake Huron: Comparison of constructed reef characteristics that attract spawning lake trout. Fish. Res. 2016, 183, 275–286. [Google Scholar] [CrossRef]
- Roseman, E.F.; McLean, M.; Pritt, J.J.; Fischer, J.; Kennedy, G. Artificial reefs and reef restoration in the Laurentian Great Lakes. In Biodiversity, Conservation, and Environmental Management in the Great Lakes Basin; Freedman, E., Neuzil, M., Eds.; Routledge: New York, NY, USA, 2017; pp. 33–46. [Google Scholar]
- Jensen, A. Artificial reefs of Europe: Perspective and future. ICES J. Mar. Sci. 2002, 59, S3–S13. [Google Scholar] [CrossRef]
- Stone, R.B. History of artificial reef use in the United States. In Artificial Reefs: Marine and Freshwater Applications; D’itri, F., Ed.; Lewis Publishers, Inc.: Chelsea, MI, USA, 1985; pp. 3–11. [Google Scholar]
- Roseman, E.F.; Manny, B.; Boase, J.; Child, M.; Kennedy, G.; Craig, J.; Soper, K.; Drouin, R. Lake sturgeon response to a spawning reef constructed in the Detroit River. J. Appl. Ichthyol. 2011, 27, 66–76. [Google Scholar] [CrossRef]
- Prichard, C.G.; Craig, J.M.; Roseman, E.F.; Fischer, J.L.; Manny, B.A.; Kennedy, G.W. Egg Deposition by Lithophilic-Spawning Fishes in the DETROIT and Saint Clair Rivers, 2005–2014; US Geological Survey: Ann Arbor, MI, USA, 2017; p. 20.
- Roni, P.; Slyke, D.V.; Miller, B.A.; Ebersole, J.L.; Pess, G. Adult coho salmon and steelhead use of boulder weirs in southwest Oregon streams. N. Am. J. Fish. Manag. 2008, 28, 970–978. [Google Scholar] [CrossRef]
- Merz, J.E.; Setka, J.D. Evaluation of a spawning habitat enhancement site for chinook salmon in a regulated California river. N. Am. J. Fish. Manag. 2004, 24, 397–407. [Google Scholar] [CrossRef]
- Merz, J.E.; Setka, J.D.; Pasternack, G.B.; Wheaton, J.M. Predicting benefits of spawning-habitat rehabilitation to salmonid (Oncorhynchus spp.) fry production in a regulated California river. Can. J. Fish. Aquat. Sci. 2004, 61, 1433–1446. [Google Scholar] [CrossRef]
- Fjeldstad, H.-P.; Barlaup, B.T.; Stickler, M.; Gabrielsen, S.-E.; Alfredsen, K. Removal of weirs and the influence on physical habitat for salmonids in a Norwegian river. River Res. Appl. 2012, 28, 753–763. [Google Scholar] [CrossRef]
- Palm, D.; Brännäs, E.; Lepori, F.; Nilsson, K.; Stridsman, S. The influence of spawning habitat restoration on juvenile brown trout (Salmo trutta) density. Can. J. Fish. Aquat. Sci. 2007, 64, 509–515. [Google Scholar] [CrossRef]
- Mitchell, L.T.N. An Assessment of Rehabilitation Gravels for Salmo trutta Spawning: A Case Study from a Small Chalk Stream, the River Stiffkey, Norfolk UK. Ph.D. Thesis, University College London, London, UK, 2016. [Google Scholar]
- Nakamura, M. Evolution of artificial fishing reef concepts in Japan. Bull. Mar. Sci. 1985, 37, 271–278. [Google Scholar]
- Kondolf, G.M.; Vick, J.C.; Ramirez, T.M. Salmon spawning habitat rehabilitation on the Merced River, California: An evaluation of project planning and performance. Trans. Am. Fish. Soc. 1996, 125, 899–912. [Google Scholar] [CrossRef]
- Larson, J.W. Essayons: A History of the Detroit District, US Army Corps of Engineers; US Army Corps of Engineers, Detroit District: Detroit, MI, USA, 1995.
- Vaccaro, L.; Bennion, D.; Boase, J.; Bohling, M.; Chiotti, J.; Craig, J.; Drouin, R.; Fischer, J.; Kennedy, G.; Manny, B.; et al. Science in Action: Lessons Learned from Fish Spawning Habitat Restoration in the St. Clair and Detroit Rivers; University of Michigan: Ann Arbor, MI, USA, 2016. [Google Scholar]
- Kelso, J.R.M.; Hartig, J.H. Methods of Modifying Habitat to Benefit the Great Lakes Ecosystem. CISTI (Canada Institute for Scientific and Technical Information) Occasional Paper No. 1; National Research Council of Canada: Ottawa, ON, Canada, 1995. [Google Scholar]
- McLean, M.; Roseman, E.F.; Pritt, J.J.; Kennedy, G.; Manny, B.A. Artificial reefs and reef restoration in the Laurentian Great Lakes. J. Gt. Lakes Res. 2015, 1, 1–8. [Google Scholar] [CrossRef]
- Fischer, J.L.; Roseman, E.F.; Mayer, C.; Wills, T. If you build it and they come, will they stay? Maturation of constructed fish spawning reefs in the St. Clair-Detroit River System. Ecol. Eng. 2020, 150, 105837. [Google Scholar] [CrossRef]
- Vehanen, T.; Huusko, A.; Mäki-Petäys, A.; Louhi, P.; Mykrä, H.; Muotka, T. Effects of habitat rehabilitation on brown trout (Salmo trutta) in boreal forest streams. Freshw. Biol. 2010, 55, 2200–2214. [Google Scholar] [CrossRef]
- Roni, P.; Bennett, T.; Morley, S.; Pess, G.R.; Hanson, K.; Slyke, D.V.; Olmstead, P. Rehabilitation of bedrock stream channels: The effects of boulder weir placement on aquatic habitat and biota. River Res. Appl. 2006, 22, 967–980. [Google Scholar] [CrossRef]
- Gatch, A.J.; Koenigbauer, S.T.; Roseman, E.F.; Höök, T.O. Assessment of two techniques for maintenance of rocky reef spawning habitat. J. Gt. Lakes Res. 2020. [Google Scholar] [CrossRef]
- Wheaton, J.M. Spawning Habitat Rehabilitation. Master’s Thesis, University of California at Davis, Davis, CA, USA, 2003. [Google Scholar]
- Taylor, J.J.; Rytwinski, T.; Bennett, J.R.; Smokorowski, K.E.; Lapointe, N.W.R.; Janusz, R.; Clarke, K.; Tonn, B.; Walsh, J.C.; Cooke, S.J. The effectiveness of spawning habitat creation or enhancement for substrate-spawning temperate fish: A systematic review. Environ. Evid. 2019, 8, 19. [Google Scholar] [CrossRef]
- Rytwinski, T.; Elmer, L.K.; Taylor, J.J.; Donaldson, L.A.; Bennett, J.R.; Smokorowski, K.E.; Winegardner, A.K.; Cooke, S.J. How Effective Are Spawning-Habitat Creation or Enhancement Measures for Substrate-Spawning Fish?: A Synthesis; Department of Fisheries and Oceans Canada: Ottawa, ON, Canada, 2019; p. 183.
- Bureau of Reclamation. Central Valley Fish and Wildlife Management Study: Fishery Problems at Red Bluff Diversion Dam and Tehama-Colusa, Canal Fish Facilities; Bureau of Reclamation Mid-Pacific Region: Sacramento, CA, USA, 1985.
- Yeo, R.R.; Dechoretz, N. Diquat and copper-ion residues in salmon-spawning channel. Weed Sci. 1976, 24, 405–409. [Google Scholar] [CrossRef]
- Louhi, P.; Vehanen, T.; Huusko, A.; Petays, A.; Muotka, T. Long-term monitoring reveals the success of salmonid habitat restoration. Can. J. Fish. Aquat. Sci. 2013, 73, 1733–1741. [Google Scholar] [CrossRef]
- Kondolf, G.M.; Matthews, W.V. Management of Coarse Sediment in Regulated Rivers of California; University of California Water Resources Center: Berkeley, CA, USA, 1991; p. 102. [Google Scholar]
- Gerke, R.J. Salmon Spawning Habitat Improvement Study; Washington Department of Fisheries: Olympia, WA, USA, 1974; p. 93.
- Wilson, D. Salmonid Spawning Habitat Improvement Study; Washington State Department of Fisheries: Olympia, WA, USA, 1976; p. 20.
- House, R.A.; Boehne, P.L. Evaluation of instream enhancement structures for salmonid spawning and rearing in a coastal Oregon stream. N. Am. J. Fish. Manag. 1985, 5, 283–295. [Google Scholar] [CrossRef]
- Moreau, J.K. Anadromous salmonid habitat enhancement by boulder placement in Hurdygurdy Creek, California. In Proceedings of the Pacific Northwest Stream Habitat Management Workshop, Arcata, CA, USA, 10–12 October 1984; Hassler, T.J., Ed.; American Fisheries Society, Humboldt Chapter: Arcata, CA, USA, 1984. [Google Scholar]
- Klassen, H.D.; Northcote, T.G. Use of gabion weirs to improve spawning habitat for pink salmon in a small logged watershed. N. Am. J. Fish. Manag. 1988, 8, 36–44. [Google Scholar] [CrossRef]
- Klassen, H.; Northcote, T. Stream bed configuration and stability following gabion weir placement to enhance salmonid production in a logged watershed subject to debris torrents. Can. J. For. Res. 1986, 16, 197–203. [Google Scholar] [CrossRef]
- Zeh, M.; Dönni, W. Restoration of spawning grounds for trout and grayling in the river High-Rhine. Aquat. Sci. 1994, 56, 59–69. [Google Scholar] [CrossRef]
- Lychwick, T.L. Fox River Walleye Habitat Improvement. In Methods of Modifying Habitat to Benefit the Great Lakes Ecosystem. CISTI (Canada Institute for Scientific and Technical Information) Occasional Paper No. 1; Kelso, J.R.M., Hartig, J.H., Eds.; National Research Council of Canada: Ottawa, ON, Canada, 1995; pp. 272–281. [Google Scholar]
- Geiling, W.D.; Kelso, J.R.M.; Iwachewski, E. Benefits from incremental additions to walleye spawning habitat in the Current River, with reference to habitat modification as a walleye management tool in Ontario. Can. J. Fish. Aquat. Sci. 1996, 53, 79–87. [Google Scholar] [CrossRef]
- Ward, B.R.; McCubbing, D.J.F.; Slaney, P.A. Stream restoration for anadromous salmonids by the addition of habitat and nutrients. In Salmon at the Edge; Mills, D., Ed.; Blackwell Science Ltd.: Oxford, UK, 2003; pp. 235–254. [Google Scholar]
- Pedersen, M.L.; Kristensen, E.A.; Kronvang, B.; Thodsen, H. Ecological effects of re-introduction of salmonid spawning gravel in lowland Danish streams. River Res. Appl. 2009, 25, 626–638. [Google Scholar] [CrossRef]
- Pasternack, G.B.; Wang, C.L.; Merz, J.E. Application of a 2D hydrodynamic model to design of reach-scale spawning gravel replenishment on the Mokelumne River, California. River Res. Appl. 2004, 20, 205–225. [Google Scholar] [CrossRef]
- Sawyer, A.M.; Pasternack, G.B.; Merz, J.E.; Escobar, M.; Senter, A.E. Construction constraints for geomorphic-unit rehabilitation on regulated gravel-bed rivers. River Res. Appl. 2009, 25, 416–437. [Google Scholar] [CrossRef]
- Wheaton, J.M.; Pasternack, G.B.; Merz, J.E. Spawning habitat rehabilitation-II. Using hypothesis development and testing in design, Mokelumne river, California, USA. Int. J. River Basin Manag. 2004, 2, 21–37. [Google Scholar] [CrossRef]
- Pasternack, G.B. Spawning habitat rehabilitation: Advances in analysis tools. In Salmonid Spawning Habitat in Rivers: Physical Controls, Biological Responses, and Approaches to Remediation. Symposium 65; Sear, D.A., DeVries, P., Greig, S., Eds.; American Fisheries Society: Bethesda, MD, USA, 2008; Volume 65, pp. 321–348. [Google Scholar]
- Elkins, E.V.; Pasternack, G.B.; Merz, J.E. Use of slope creation for rehabilitating incised, regulated, gravel bed rivers. Water Resour. Res. 2007, 43, W05432. [Google Scholar] [CrossRef]
- Sellheim, K.L.; Watry, C.B.; Rook, B.; Zeug, S.C.; Hannon, J.; Zimmerman, J.; Dove, K.; Merz, J.E. Juvenile salmonid utilization of floodplain rearing habitat after gravel augmentation in a regulated river. River Res. Appl. 2016, 32, 610–621. [Google Scholar] [CrossRef]
- Zeug, S.C.; Sellheim, K.; Watry, C.; Rook, B.; Hannon, J.; Zimmerman, J.; Cox, D.; Merz, J. Gravel augmentation increases spawning utilization by anadromous salmonids: A case study from California, USA. River Res. Appl. 2014, 30, 707–718. [Google Scholar] [CrossRef]
- Crossman, J.A.; Hildebrand, L.R. Evaluation of spawning substrate enhancement for white sturgeon in a regulated river: Effects on larval retention and dispersal. River Res. Appl. 2014, 30, 1–10. [Google Scholar] [CrossRef]
- Utz, R.M.; Mesick, C.F.; Cardinale, B.J.; Dunne, T. How does coarse gravel augmentation affect early-stage Chinook salmon Oncorhynchus tshawytscha embryonic survivorship? J. Fish Biol. 2013, 82, 1484–1496. [Google Scholar] [CrossRef] [PubMed]
- Pander, J.; Mueller, M.; Geist, J. A comparison of four stream substratum restoration techniques concerning interstitial conditions and downstream effects. River Res. Appl. 2015, 31, 239–255. [Google Scholar] [CrossRef]
- McManamay, R.A.; Orth, D.J.; Dolloff, C.A.; Cantrell, M.A. Gravel additions as a habitat restoration technique for tailwaters. N. Am. J. Fish. Manag. 2011, 30, 1238–1257. [Google Scholar] [CrossRef]
- Merz, J.; Caldwell, L.; Beakes, M.; Hammersmark, C.; Sellheim, K. Balancing competing life-stage requirements in salmon habitat rehabilitation: Between a rock and a hard place. Restor. Ecol. 2018, 27, 661–671. [Google Scholar] [CrossRef]
- Naito, G. Lower Whatshan River Fish Habitat Enhancement Physical and Biological Effectiveness Monitoring 2015 (Year 10); Naito Environmental: Vernon, BC, Canada, 2016. [Google Scholar]
- White, H.C. Atlantic salmon redds and artificial spawning beds. J. Fish. Res. Board Can. 1942, 6, 37–45. [Google Scholar] [CrossRef]
- Meehan, W.R. Effects of gravel cleaning on bottom organisms in three southeast Alaska streams. Prog. Fish Cult. 1971, 33, 107–111. [Google Scholar] [CrossRef]
- Zeigler, E.R. An Artificially Produced Velocity Barrier for Controlling Fish Movement: Tehama-Colusa Canal; Bureau of Reclamation Hydraulics Branch: Denver, CO, USA, 1967; p. 33.
- Andrew, F.J. Gravel cleaning to increase salmon production in rivers and spawning channels. In Proceedings of the Salmon Spawning Gravel: A Renewable Resource in the Pacific Northwest, Seattle, WA, USA, 6–7 October 1980; State of Washington Water Resource Center Washington State University: Pullman, WA, USA, 1981; pp. 15–31. [Google Scholar]
- Mih, W.C.; Bailey, G.C. The development of a machine for the restoration of stream gravel for spawning and rearing of salmon. Fisheries 1981, 6, 16–20. [Google Scholar]
- Mih, W.C.; Bailey, G.C. A machine for mitigation of salmonid spawning habitat from silting. In Proceedings of the Mitigation Symposium, Fort Collins, CO, USA, 16–20 July 1979; Rocky Mountain Forest and Range Experiment Station; US Forest Service, and Colorado State University: Fort Collins, CO, USA, 1979; pp. 645–648. [Google Scholar]
- Avery, E.L. Evaluations of sediment traps and artificial gravel riffles constructed to improve reproduction of trout in three Wisconsin streams. N. Am. J. Fish. Manag. 1996, 16, 282–293. [Google Scholar] [CrossRef]
- Semple, J.R. A Simple and Effective Method of Cleaning the Gravel of Atlantic Salmon Spawning Habitat; Canada Department of Fisheries and Oceans: Halifax, NS, Canada, 1987; p. 13.
- Mundie, J.H.; Crabtree, D.G. Effects on sediments and biota of cleaning a salmonid spawning channel. Fish. Manag. Ecol. 1997, 4, 111–126. [Google Scholar] [CrossRef]
- Rubin, J.F.; Glimsater, C.G.; Jarvi, T. Characteristics and rehabilitation of the spawning habitats of the sea trout, Salmo trutta. Fish. Ecol. Manag. 2004, 11, 15–22. [Google Scholar] [CrossRef]
- Shackle, V.J.; Hughes, S.; Lewis, V.T. The influence of three methods of gravel cleaning on brown trout, Salmo trutta, egg survival. Hydrol. Process. 1999, 13, 477–486. [Google Scholar] [CrossRef]
- Dustin, D.L.; Jacobson, P.C. Evaluation of Walleye Spawning Habitat Improvement Projects in Streams; Minnesota Department of Natural Resources: St. Paul, MN, USA, 2003.
- Howson, T.J.; Robson, B.J.; Mitchell, B.D. Patch-specific spawning is linked to restoration of a sediment-disturbed lowland river, south-eastern Australia. Ecol. Eng. 2010, 36, 920–929. [Google Scholar] [CrossRef]
- Pulg, U.; Barlaup, B.T.; Sternecker, K.; Trepl, L.; Unfer, G. Restoration of spawning habitats of Brown Trout (Salmo trutta) in a regulated chalk stream. River Res. Appl. 2011, 29, 172–182. [Google Scholar] [CrossRef]
- Sternecker, K.; Wild, R.; Geist, J. Effects of substratum restoration on salmonid habitat quality in a subalpine stream. Environ. Biol. Fishes 2013, 96, 1341–1351. [Google Scholar] [CrossRef]
- Mueller, M.; Pander, J.; Geist, J. The ecological value of stream restoration measures: An evaluation on ecosystem and target species scales. Ecol. Eng. 2014, 62, 129–139. [Google Scholar] [CrossRef]
- Sepulveda, A.J.; Sechrist, J.; Marczak, L.B. Testing ecological tradeoffs of a new tool for removing fine sediment in a spring-fed stream. Ecol. Restor. 2014, 32, 68–77. [Google Scholar] [CrossRef]
- Ramezani, J.; Rennebeck, L.; Closs, G.P.; Matthaei, C.D. Effects of fine sediment addition and removal on stream invertebrates and fish: A reach-scale experiment. Freshw. Biol. 2014, 59, 2584–2604. [Google Scholar] [CrossRef]
- Sepulveda, A.J.; Layhee, M.; Sutphin, Z.A.; Sechrist, J.D. Evaluation of a fine sediment removal tool in a spring fed and snowmelt driven streams. Ecol. Restor. 2015, 33, 303–315. [Google Scholar] [CrossRef]
- Matthys, T. Estimating Fish Habitat Selection and Monitoring Stream Habitat Quality Requires More Than Simply Counting Fish. Ph.D. Dissertation, Michigan Technological University, Houghton, MI, USA, 2017. [Google Scholar]
- Basic, T.; Britton, J.R.; Rice, S.P.; Pledger, A.G. Impacts of gravel jetting on the composition of fish spawning substrates; Implications for river restoration and fisheries management. Ecol. Eng. 2017, 107, 71–81. [Google Scholar] [CrossRef]
- NHC (Northwest Hydraulic). 2016 Spawning Substrate Restoration on the Nechako River at Vanderhoof, BC; Prepared for Ministry of Forest, Lands and Natural Resource Operation: Prince George, BC, Canada, 2016; p. 35.
- Kondolf, G.M. Assessing salmonid spawning gravel quality. Trans. Am. Fish. Soc. 2000, 129, 262–281. [Google Scholar] [CrossRef]
- Wheaton, J.M.; Pasternack, G.B.; Merz, J. Spawning habitat rehabilitation—I. conceptual approach and methods. Int. J. River Basin Manag. 2004, 2, 3–20. [Google Scholar] [CrossRef]
- Argent, D.G.; Flebbe, P.A. Fine sediment effects on brook trout eggs in laboratory streams. Fish. Res. 1999, 39, 253–262. [Google Scholar] [CrossRef]
- Fudge, R.J.P.; Bodaly, R.A. Postimpoundment winter sedimentation and survival of lake whitefish (Coregonus clupeaformis) eggs in Southern Indian Lake, Manitoba. Can. J. Fish. Aquat. Sci. 1984, 41, 701–705. [Google Scholar] [CrossRef]
- Kock, T.J.; Congleton, J.L.; Anders, P.J. Effects of sediment cover on survival and development of white sturgeon embryos. N. Am. J. Fish. Manag. 2006, 26, 134–141. [Google Scholar] [CrossRef]
- Turnpenny, A.W.H.; Williams, R. Effects of sedimentation on the gravels of an industrial river system. J. Fish Biol. 1980, 17, 681–693. [Google Scholar] [CrossRef]
- Einum, S.; Hendry, A.P.; Fleming, I.A. Egg-size evolution in aquatic environments: Does oxygen availability constrain size? Proc. R. Soc. Lond. B Biol. Sci. 2002, 269, 2325–2330. [Google Scholar] [CrossRef] [PubMed]
- Saldi-Caromile, K.; Bates, K.; Skidmore, P.; Barenti, J.; Pineo, D.L. Stream Habitat Restoration Guidelines: Final Draft; Co-published by the Washington Departments of Fish and Wildlife and Ecology and the U.S. Fish and Wildlife Service: Olympia, WA, USA, 2004. [Google Scholar]
- Trombulak, S.C.; Frissell, C.A. Review of ecological effects of roads on terrestrial and aquatic communities. Conserv. Biol. 2000, 14, 18–30. [Google Scholar] [CrossRef]
- Richardson, T.W. Agitation Dredging: Lessons and Guidelines from Past Projects; US Army Corps of Engineers: Vicksburg, MS, USA, 1984; p. 145.
- Sullivan, N. The use of agitation dredging, water injection dredging and sidecasting: Results of a survey of ports in England and Wales. Terra Aqua 2000, 78, 11–20. [Google Scholar]
- Peterson, S.A. Lake restoration by sediment removal. J. Am. Water Resour. Assoc. 1982, 18, 423–436. [Google Scholar] [CrossRef]
- Baker, E.; Milburn, T.; Hugh, B.; Tennant, D.A. Field assessment of sediment trap efficiency under varying flow conditions. J. Mar. Res. 1988, 46, 573–592. [Google Scholar] [CrossRef]
- Gardner, W.D. Field assessment of sediment traps. J. Mar. Res. 1980, 38, 41–52. [Google Scholar]
- Parchure, T.M.; Teeter, A.M. Lessons Learned from Existing Projects on Shoaling in Harbors and Navigation Channels; US Army Corps of Engineers: Vicksburg, MS, USA, 2003.
- Herdendorf, C.E. Physical and limnological characteristics of natural spawning reefs in western Lake Erie. In Artificial Reefs Marine and Freshwater Applications; D’itri, F., Ed.; Lewis Publishers, Inc.: Chelsea, MI, USA, 1985; pp. 149–183. [Google Scholar]
- Brooks, C.; Grimm, A.; Shuchman, R.; Sayers, M.; Jessee, N. A satellite-based multi-temporal assessment of the extent of nuisance Cladophora and related submerged aquatic vegetation for the Laurentian Great Lakes. Remote Sens. Environ. 2015, 157, 58–71. [Google Scholar] [CrossRef]
- Redman, R.; Mackey, S.; Dub, J.; Czesny, S. Lake trout spawning habitat suitability at two offshore reefs in Illinois waters of Lake Michigan. J. Gt. Lakes Res. 2017, 43, 335–344. [Google Scholar] [CrossRef]
- Janssen, J.; Berg, M.B.; Lozano, S.J. Submerged terra incognita: Lake Michigan’s abundant but unknown rocky zones. State Lake Mich. Ecol. Health Manag. Ecovision World Monogr. Ser. 2005, 113–139. [Google Scholar]
- Auer, M.T.; Tomlinson, L.M.; Higgins, S.N.; Malkin, S.Y.; Howell, E.T.; Bootsma, H.A. Great Lakes Cladophora in the 21st century: Same algae—Different ecosystem. J. Gt. Lakes Res. 2010, 36, 248–255. [Google Scholar] [CrossRef]
- Blanco, S.; Ector, L. Distribution, ecology and nuisance effects of the freshwater invasive diatom Didymosphenia geminata (Lyngbye) M. Schmidt: A literature review. Nova Hedwig. 2009, 88, 347–422. [Google Scholar] [CrossRef]
- Dorr, J.A., III; O’Connor, D.V.; Foster, N.R.; Jude, D.J. Substrate conditions and abundance of lake trout eggs in a traditional spawning area in southeastern Lake Michigan. N. Am. J. Fish. Manag. 1981, 1, 165–172. [Google Scholar] [CrossRef]
- Tucker, S. Lake Sturgeon Spawning Behavior, Larval Production and Juvenile Habitat Use Within The Lower Fox River, Wisconsin. Master’s Thesis, University of Wisconsin-Green Bay, Green Bay, WI, USA, 2020. [Google Scholar]
- Da Silva, S.; Taillon, K.; McChristie, M.; Chase, M.; Berglund, E. Nipigon Bay Remedial Action Plan Report Recommending Delisting and Path Forward; North Shore of Lake Superior Remedial Action Plans: Toronto, ON, Canada, 2015; p. 80. [Google Scholar]
- Parsley, M.J.; Kofoot, E. Effects of Incubation Substrates on Hatch Timing and Success of White Sturgeon (Acipenser transmontanus) Embryos; US Department of the Interior, US Geological Survey: Reston, VA, USA, 2013; p. 16.
- Tyner, E.H. Nearshore Benthic Oxygen Dynamics in Lake Michigan. Master’s Thesis, University of Wisconsin-Milwaukee, Milwaukee, WI, USA, 2013. [Google Scholar]
- Jellyman, P.G.; Clearwater, S.J.; Clayton, J.S.; Kilroy, C.; Hickey, C.W.; Blair, N.; Biggs, B.J.F. Rapid screening of multiple compounds for control of the invasive diatom Didymosphenia geminata. J. Aquat. Plant Manag. 2010, 48, 63. [Google Scholar]
- Klauda, R.J.; Hanna, K.V. Didymosphenia Geminata Infestation in Maryland: Reactions and Responses by the Maryland Department of Natural Resources; Maryland Department of Natural Resources Resource Assessment Service Monitoring and Non-tidal Assessment Division: Annapolis, MD, USA, 2016; p. 36.
- McCuaig, L.M.; Martyniuk, C.J.; Marlatt, V.L. Morphometric and proteomic responses of early-life stage rainbow trout (Oncorhynchus mykiss) to the aquatic herbicide diquat dibromide. Aquat. Toxicol. 2020, 222, 105446. [Google Scholar] [CrossRef]
- Sesin, V.; Dalton, R.L.; Boutin, C.; Robinson, S.A.; Bartlett, A.J.; Pick, F.R. Macrophytes are highly sensitive to the herbicide diquat dibromide in test systems of varying complexity. Ecotoxicol. Environ. Saf. 2018, 165, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.B. Survival and Growth Responses of Lithobates Pipiens Tadpoles to an Herbicide and an Algaecide used to Control Aquatic Invasive Plants. Ph.D. Thesis, Bowling Green State University, Bowling Green, OH, USA, 2015. [Google Scholar]
- Clearwater, S.J.; Jellyman, P.G.; Biggs, B.J.F.; Hickey, C.W.; Blair, N.; Clayton, J.S. Pulse-dose application of chelated copper to a river for Didymosphenia geminata control: Effects on macroinvertebrates and fish. Environ. Toxicol. Chem. 2011, 30, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Boylan, J.D.; Morris, J.E. Limited effects of barley straw on algae and zooplankton in a midwestern pond. Lake Reserv. Manag. 2003, 19, 265–271. [Google Scholar] [CrossRef][Green Version]
- Peller, J.R.; Whitman, R.L.; Griffith, S.; Harris, P.; Peller, C.; Scalzitti, J. TiO2 as a photocatalyst for control of the aquatic invasive alga, Cladophora, under natural and artificial light. J. Photochem. Photobiol. Chem. 2007, 186, 212–217. [Google Scholar] [CrossRef]
- Elwell, L.C.; Gillis, C.-A.; Kunza, L.A.; Modley, M.D. Management challenges of Didymosphenia geminata. Diatom Res. 2014, 29, 303–305. [Google Scholar] [CrossRef]
- Root, S.; O’Reilly, C.M. Didymo control: Increasing the effectiveness of decontamination strategies and reducing spread. Fisheries 2012, 37, 440–448. [Google Scholar] [CrossRef]
- Nalepa, T.F. An overview of the spread, distribution, and ecological impacts of the quagga mussel, Dreissena rostriformis bugensis, with possible implications to the Colorado River system. In Proceedings of the Colorado River Basin Science and Resource Management Symposium. Coming Together, Coordination of Science and Restoration Activities for the Colorado River Ecosystem, Scottsdale, AZ, USA, 18–20 November 2008; U.S. Geological Survey: Reston, VA, USA, 2010; p. 372. [Google Scholar]
- Furgal, S.; Lantry, B.F.; Weidel, B.C.; Farrell, J.M.; Gorsky, D.; Biesinger, Z. Lake Trout Spawning and Habitat Assessment at Stony Island Reef; New York State Department of Environmental Conservation (NYSDEC): Albany, NY, USA, 2018.
- Kelch, D.O.; Snyder, F.L.; Reutter, J.M. Artificial reefs in Lake Erie: Biological impacts of habitat alteration. In Fish Habitat: Essential Fish Habitat and Rehabilitation; Benaka, L.R., Ed.; American Fisheries Society: Bethesda, MD, USA, 1999; Volume 22, pp. 335–347. [Google Scholar]
- Hecky, R.E.; Smith, R.E.H.; Barton, D.R.; Guildford, S.J.; Taylor, W.D.; Charlton, M.N.; Howell, T. The nearshore phosphorus shunt: A consequence of ecosystem engineering by dreissenids in the Laurentian Great Lakes. Can. J. Fish. Aquat. Sci. 2004, 61, 1285–1293. [Google Scholar] [CrossRef]
- Eschweiler, N.; Christensen, H.T. Trade-off between increased survival and reduced growth for blue mussels living on Pacific oyster reefs. J. Exp. Mar. Biol. Ecol. 2011, 403, 90–95. [Google Scholar] [CrossRef]
- Norling, P.; Lindegarth, M.; Lindegarth, S.; Strand, Å. Effects of live and post-mortem shell structures of invasive Pacific oysters and native blue mussels on macrofauna and fish. Mar. Ecol. Prog. Ser. 2015, 518, 123–138. [Google Scholar] [CrossRef][Green Version]
- Eshenroder, R.L.; Peck, J.W.; Olver, C.H. Research Priorities for Lake Trout Rehabilitation in the Great Lakes: A 15-Year Retrospective; Great Lakes Fishery Commission: Ann Arbor, MI, USA, 1999; Volume 64, pp. 1–40. [Google Scholar]
- Glon, M.G.; Larson, E.R.; Reisinger, L.S.; Pangle, K.L. Invasive dreissenid mussels benefit invasive crayfish but not native crayfish in the Laurentian Great Lakes. J. Gt. Lakes Res. 2017, 43, 289–297. [Google Scholar] [CrossRef]
- Levin, P.S.; Coyer, J.A.; Petrik, R.; Good, T.P. Community wide effects of nonindigenous species on temperate rocky reefs. Ecology 2002, 83, 3182–3193. [Google Scholar] [CrossRef]
- LaFrancois, B.; Bootsma, H. Manual Removal of Invasive Mussels: A Case Study from Sleeping Bear Dunes, Lake Michigan; National Park Service: Porter, IN, USA, 2018.
- Fitzsimons, J.D.; Leach, J.H.; Nepszy, S.J.; Cairns, V.W. Impacts of zebra mussel on walleye (Stizostedion vitreum) reproduction in western Lake Erie. Can. J. Fish. Aquat. Sci. 1995, 52, 578–586. [Google Scholar] [CrossRef]
- Muir, A.M.; Blackie, C.T.; Marsden, J.E.; Krueger, C.C. Lake charr Salvelinus namaycush spawning behaviour: New field observations and a review of current knowledge. Rev. Fish Biol. Fish. 2012, 22, 575–593. [Google Scholar] [CrossRef]
- Muir, A.M.; Krueger, C.C.; Hansen, M.J. Re-establishing lake trout in the Laurentian Great Lakes: Past, present, and future. In Great Lakes Fishery Policy and Management: A Binational Perspective, 2nd ed.; Michigan State University Press: East Lansing, MI, USA, 2012; pp. 533–588. [Google Scholar]
- Hargrave, J.; Jensen, D. Assessment of the Water Quality Conditions at ed Zorinsky Reservoir and the Zebra Mussel (Dreissena Polymorpha) Population Emerged after the Drawdown of the Reservoir and Management Implications for the District’s Papillion and Salt Creek Reservoirs; U.S. Army Corps of Engineers: Omaha, NE, USA, 2012.
- Grazio, J.L.; Montz, G. Winter lake drawdown as a strategy for zebra mussel (Dreissena polymorpha) control: Results of pilot studies in Minnesota and Pennsylvania. In Proceedings of the 11th International Conference on Aquatic Invasive Species, Alexandria, VA, USA, 25 February–1 March 2002; pp. 207–217. [Google Scholar]
- Whitledge, G.W.; Weber, M.M.; DeMartini, J.; Oldenburg, J.; Roberts, D.; Link, C.; Rackl, S.M.; Rude, N.; Yung, A.; Bock, L.R.; et al. An evaluation Zequanox efficacy and application strategies for targeted control of zebra mussels in shallow-water habitats in lakes. Manag. Biol. Invasions 2015, 6, 71–82. [Google Scholar] [CrossRef]
- Crank, K.M.; Barnes, M.E. Zebra mussel veliger chemical control treatments do not impact rainbow trout eyed egg survival. Int. J. Innov. Stud. Aquat. Biol. Fish. 2017, 3, 15–17. [Google Scholar]
- Hillard, S.; Huysman, N.; Barnes, M.E. Impacts of zebra mussel veliger control treatments on the survival of water-hardened landlocked fall chinook salmon eggs. Nat. Resour. 2019, 10, 115–120. [Google Scholar] [CrossRef]
- Edwards, W.J.; Babcock-Jackson, L.; Culver, D.A. Field testing of protocols to prevent the spread of zebra mussels Dreissena polymorpha during fish hatchery and aquaculture activities. N. Am. J. Aquac. 2002, 64, 220–223. [Google Scholar] [CrossRef]
- Wimbush, J.; Frischer, M.E.; Zarzynski, J.W.; Nierzwicki-Bauer, S.A. Eradication of colonizing populations of zebra mussels (Dreissena polymorpha) by early detection and SCUBA removal: Lake George, NY. Aquat. Conserv. Mar. Freshw. Ecosyst. 2009, 19, 703–713. [Google Scholar] [CrossRef]
- Claudi, R.; Mackie, G.L. Practical Manual for Zebra Mussel Monitoring and Control; Lewis Publishers, CRC: Boca Raton, FL, USA, 1994. [Google Scholar]
- Wong, W.H.; Gerstenberger, S.; Watters, A. Using Pressurized Hot Water Spray to Kill and Remove Dreissenid Mussels on Watercraft: Field Testing on the Efficacy of Water Temperature, High Pressure, and Duration of Exposure; US Fish and Wildlife Service: Washington, DC, USA, 2014; p. 20.
- Culver, C.; Lahr, H.; Johnson, L.; Cassell, J. Quagga and Zebra Mussel Eradication and Control Tactics; University of California: Oakland, CA, USA, 2013. [Google Scholar]
- Waller, D.L.; Bartsch, M.R. Use of carbon dioxide in zebra mussel (Dreissena polymorpha) control and safety to a native freshwater mussel (Fatmucket, Lampsilis siliquoidea). Manag. Biol. Invasions 2018, 9, 439–450. [Google Scholar] [CrossRef]
- Cook, G.D. Marine Excavation. Encycl. Archaeol. Sci. 2018, 1–4. [Google Scholar] [CrossRef]
- Goggin, J.M. Underwater archeology: Its nature and limitations. Am. Antiq. 1960, 25, 348–354. [Google Scholar] [CrossRef]
- Viduka, A.J. Unit 10: Intrusive techniques in underwater archaeology. In Training Manual for the UNESCO Foundation Course on the Protection and Management of Underwater Cultural Heritage in Asia and the Pacific; UNESCO Bangkok: Bangkok, Thailand, 2012; p. 29. [Google Scholar]
- Bass, G.F.; Hope-Simpson, R. Archaeologists, sports divers, and treasure-hunters. J. Field Archaeol. 1985, 12, 256–260. [Google Scholar] [CrossRef]
- Liou, Y.C.; Herbich, J.B. Sediment Movement Induced by Ships in Restricted Waterways; Texas A&M University: College Station, TX, USA, 1976. [Google Scholar]
- Wang, P.; Rivera-Duarte, I.; Richter, K.; Liao, Q.; Farley, K.; Chen, H.-C.; Germano, J.; Markillie, K.; Gailani, J. Evaluation of Resuspension from Propeller Wash in DoD Harbors, Project ER-201031; SSC Pacific: San Diego, CA, USA, 2016; p. 325. [Google Scholar]
- Wood, P.J. Biological effects of fine sediment in the lotic environment. Environ. Manag. 1997, 21, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Mouton, A.M.; Buysse, D.; Stevens, M.; Neucker, T.; Coeck, J. Evaluation of riparian habitat restoration in a lowland river. River Res. Appl. 2012, 28, 845–857. [Google Scholar] [CrossRef]
- Kailing, P.J.; Tucker, R.L. An innovative device for stream bedload sediment removal and measurement. In Proceedings of the Seventh Federal Interagency Sedimentation Conference, Reno, NV, USA, 25–29 March 2001; pp. 34–40. [Google Scholar]
- Lipscomb, C.M.; Darrow, A.; Thornton, C.I. Removal Efficiency Testing of Streamside Systems’ Bedload Monitoring Collector; Engineering Research Center, Colorado State University: Fort Collins, CO, USA, 2005; p. 87. [Google Scholar]
- Thomas, R.C.; McArthur, J.; Braatz, D.; Welp, T. Sediment Management Methods to Reduce Dredging: Part 2, Sediment Collector Technology; US Army Engineer Research and Development Center: Vicksburg, MS, USA, 2017; p. 11.
- Geiling, D. Construction of self-cleaning substrate. In Methods of Modifying Habitat to Benefit the Great Lakes Ecosystem. CISTI (Canada Institute for Scientific and Technical Information) Occasional Paper No. 1; Kelso, J.R.M., Hartig, J.H., Eds.; National Research Council of Canada: Ottawa, ON, Canada, 1995; pp. 32–38. [Google Scholar]
- Sayce, C.S.; Larson, C.C. Willapa oyster studies—Use of the pasture harrow for the cultivation of oysters. Comm. Commer. Fish. Rev. 1966, 28, 21–26. [Google Scholar]
- MacKenzie, C.L. Development of an aquacultural program for rehabilitation of damaged oyster reefs in Mississippi. Mar. Fish. Rev. 1977, 39, 1–13. [Google Scholar]
- Perret, W.S.; Dugas, R.; Roussel, J.; Wilson, C.A.; Supan, J. Oyster Habitat Restoration: A Response to Hurricane Andrew. In Oyster Reef Habitat Restoration: A Synopsis and Synthesis of Approaches; Proceedings from the Symposium, Williamsburg, Virginia, April 1995; Luckenbach, M.W., Wesson, J.A., Eds.; Virginia Institute of Marine Science, College of William and Mary: Gloucester Point, VA, USA, 1999; pp. 93–99. [Google Scholar]
- Wesson, J.; Mann, R.; Luckenbach, M.W. Oyster Restoration Efforts in Virginia. In Oyster Reef Habitat Restoration: A Synopsis and Synthesis of Approaches; Proceedings from the Symposium, Williamsburg, Virginia, April 1995; Luckenbach, M.W., Wesson, J.A., Eds.; Virginia Institute of Marine Science, College of William and Mary: Gloucester Point, VA, USA, 1999; pp. 117–129. [Google Scholar]
- Binkowski, F.P. Utilization of artificial reefs in the inshore areas of Lake Michigan. In Artificial Reefs: Marine and Freshwater Applications; D’itri, F., Ed.; Lewis Publishers, Inc.: Chelsea, MI, USA, 1985; pp. 349–362. [Google Scholar]
- Kevern, N.R.; Biener, W.E.; VanDerLann, S.; Cornelius, S.D. Preliminary evaluation of an artificial reef as a fishery management strategy in Lake Michigan. In Artificial Reefs: Marine and Freshwater Applications; D’itri, F., Ed.; Lewis Publishers, Inc.: Chelsea, MI, USA, 1985; pp. 443–458. [Google Scholar]
- Environnement Illimité Inc. Investigation of Lake Sturgeon Spawning Activities at Xxxxxxxxxxxxx on the St. Lawrence River in 2008—Final Report; White Plains: New York, NY, USA, 2009; p. 33. [Google Scholar]
- Peck, J.W. Dynamics of reproduction by hatchery lake trout on a man-made spawning reef. J. Gt. Lakes Res. 1986, 12, 293–303. [Google Scholar] [CrossRef]
- Kim, C.G.; Kim, H.S. Post-placement management of artificial reefs in Korea. Fisheries 2008, 33, 61–68. [Google Scholar] [CrossRef]
- Yoon, H.S.; Dongha, K.; Na, W. Estimation of effective usable and burial volumes of artificial reefs and the prediction of cost effective management. Ocean Coast. Manag. 2016, 120, 135–147. [Google Scholar] [CrossRef]
- Sheng, Y.P. Physical characteristics and engineering at reef sites. In Artificial Reef Evaluation with Application to Natural Marine Habitats; CRC Press: Boca Raton, FL, USA, 2000; pp. 51–94. [Google Scholar]
- Mumby, P.J.; Harborne, A.R.; Raines, P.S.; Ridley, J.M. A critical assessment of data derived from Coral Cay Conservation volunteers. Bull. Mar. Sci. 1995, 56, 737–751. [Google Scholar]
- Toft, J.D.; Ogston, A.S.; Heerhartz, S.M.; Cordell, J.R.; Flemer, E.E. Ecological response and physical stability of habitat enhancements along an urban armored shoreline. Ecol. Eng. 2013, 57, 97–108. [Google Scholar] [CrossRef]
- Collier, J.S.; Brown, C.J. Correlation of sidescan backscatter with grain size distribution of surficial seabed sediments. Mar. Geol. 2005, 214, 431–449. [Google Scholar] [CrossRef]
- Dumont, P.; D’Amours, J.; Thibodeau, S.; Dubuc, N.; Verdon, R.; Garceau, S.; Bilodeau, P.; Mailhot, Y.; Fortin, R. Effects of the development of a newly created spawning ground in the Des Prairies River (Quebec, Canada) on the reproductive success of lake sturgeon (Acipenser fulvescens). J. Appl. Ichthyol. 2011, 27, 394–404. [Google Scholar] [CrossRef]
- Foster, N.R.; Kennedy, G.W. Patterns of egg deposition by lake trout and lake whitefish at Tawas artificial reef, Lake Huron, 1990-1993. In The Lake Huron Ecosystem: Ecology, Fisheries, and Management; Leach, J.H., Edsall, T.A., Munawar, M., Eds.; SPB Academic Publishing: Amsterdam, The Netherlands, 1995; pp. 191–206. [Google Scholar]
- Diegues, A.; Pinto, J.; Ribeiro, P.; Frias, R. Others Automatic habitat mapping using convolutional neural networks. In Proceedings of the 2018 IEEE OES Autonomous Underwater Vehicle Symposium, Porto, Portugal, 6–9 November 2018; pp. 1–6. [Google Scholar]
- Bonin-Font, F.; Burguera, A.; Lisani, J.-L. Visual discrimination and large area mapping of Posidonia oceanica using a lightweight AUV. IEEE Access 2017, 5, 24479–24494. [Google Scholar] [CrossRef]
- Brown, C.J.; Smith, S.J.; Lawton, P.; Anderson, J.T. Benthic habitat mapping: A review of progress towards improved understanding of the spatial ecology of the seafloor using acoustic techniques. Estuar. Coast. Shelf Sci. 2011, 92, 502–520. [Google Scholar] [CrossRef]
- Kaeser, A.J.; Litts, T.L. A novel technique for mapping habitat in navigable streams using low-cost side scan sonar. Fisheries 2010, 35, 163–174. [Google Scholar] [CrossRef]
- Katz, S.; Barnas, K.; Hicks, R.; Cowen, J.; Jenkinson, R. Freshwater Habitat Restoration Actions in the. Pacific Northwest: A Decade’s Investment in Habitat Improvement. Restor. Ecol. 2007, 15, 494–505. [Google Scholar] [CrossRef]
- Jensen, A.; Ken, C.; Peter, L. Current issues relating to artificial reefs in European seas. In Artificial Reefs in European Seas; Springer: Berlin, Germany, 2000; pp. 489–499. [Google Scholar]

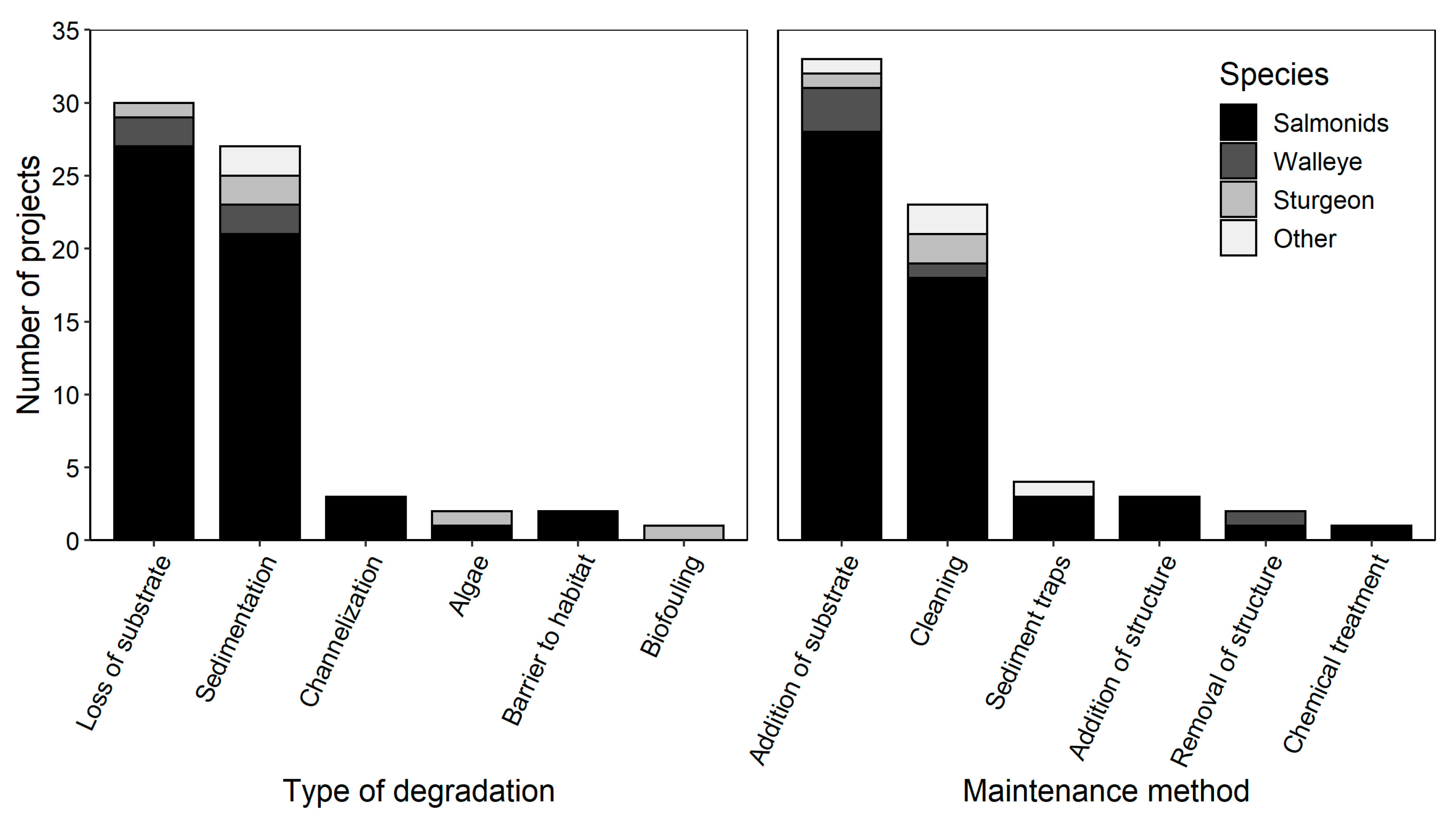
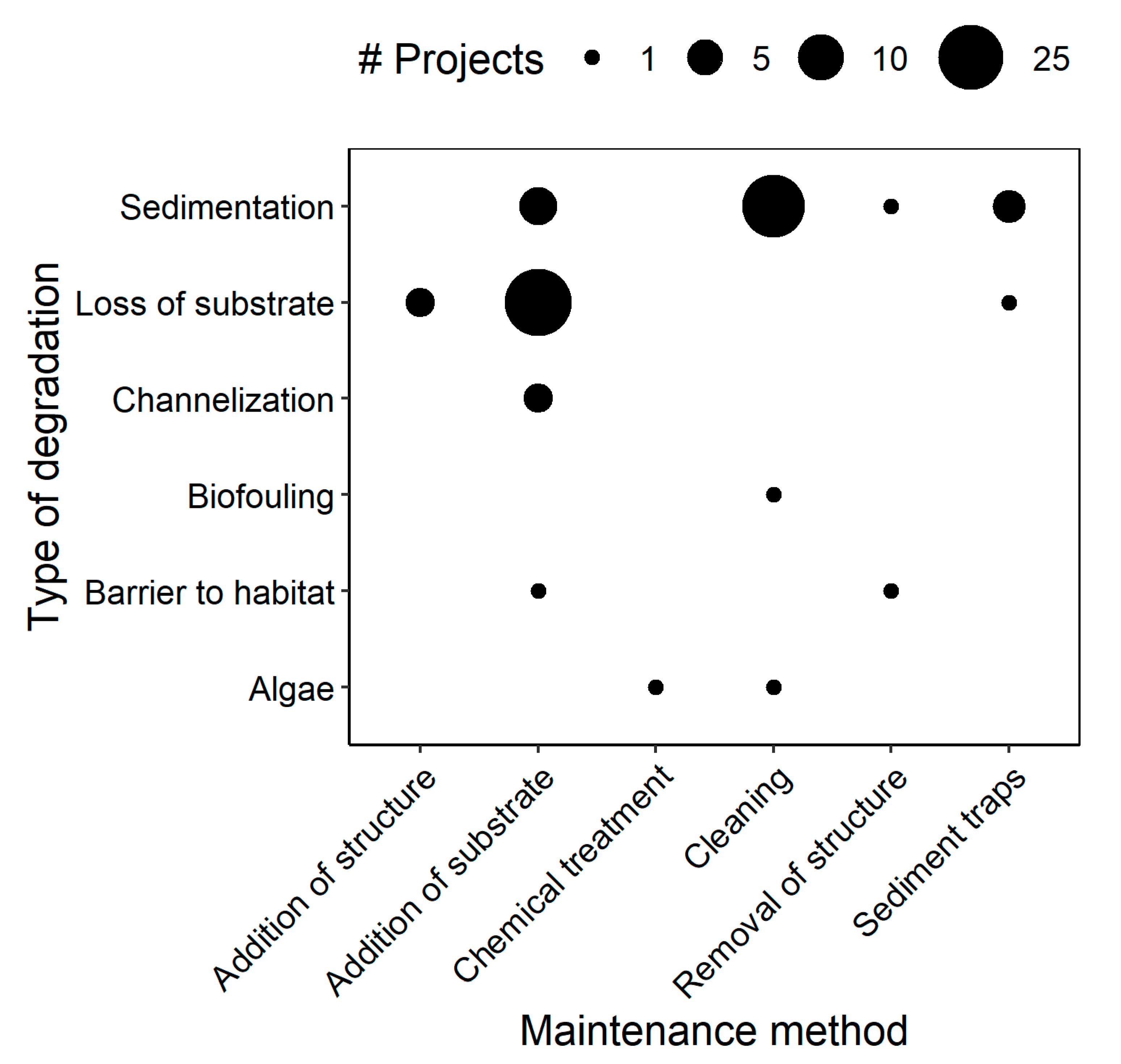
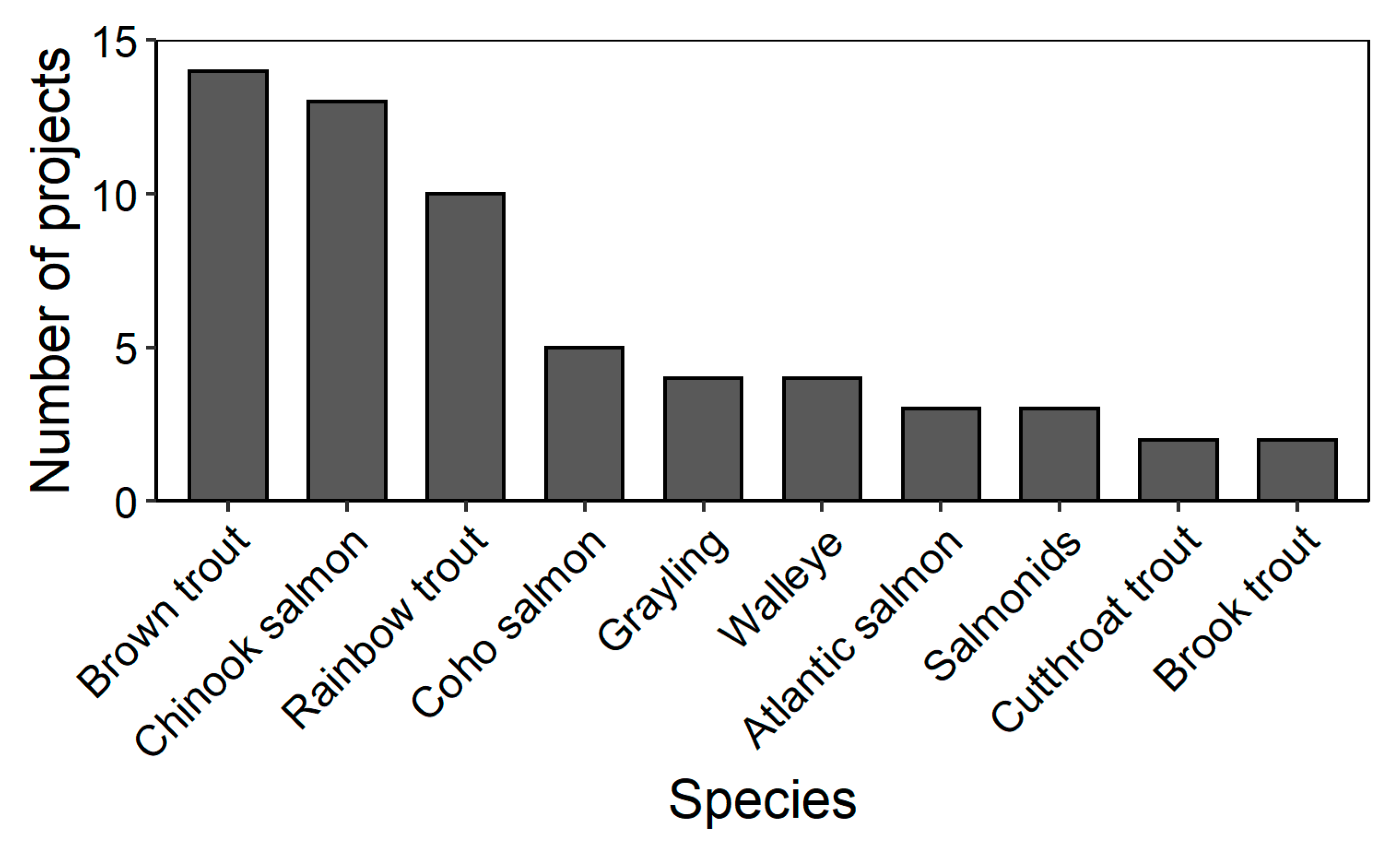
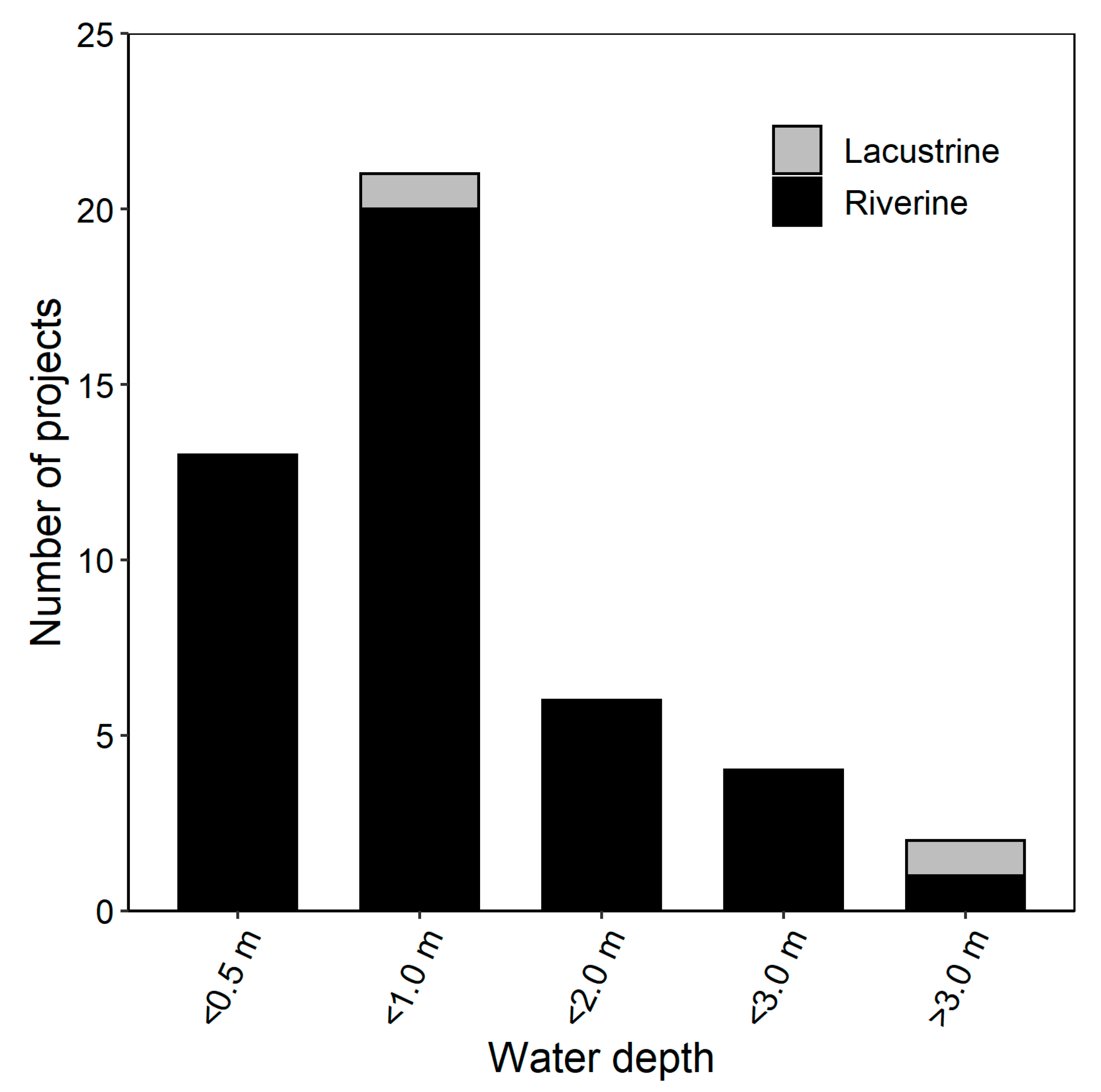

| Type of Degradation | Remedial Action | Target Species | Study Year(s) | Location | Latitude, Longitude | Specific Remedial Method | References |
|---|---|---|---|---|---|---|---|
| Algae | Chemical treatments/Cleaning | Chinook salmon | 1972–1982 | Tehama-Colusa Canal on Sacramento River, California, USA | 39.9660° N, 122.1350° W | Applied diquat, copper sulfate pentahydrate, and acrolein compounds to artificial spawning channels to control Cladophora algae, later transitioned to manual cleaning by dragging chains/scraping devices | [62,63] |
| Barrier to habitat/Loss of gravel | Addition of substrate | Chinook salmon, rainbow trout | 2000–2003 | Mokelumne River, California, USA | 38.0960° N, 121.5699° W | Addition of gravel bars, berms, and riffles; 11,000 m3 of gravel at 12 sites | [45] |
| Barrier to habitat/Loss of substrate | Removal of structure/Addition of substrate | Atlantic salmon, brown trout | 2002–2009 | Nidelva River, Norway | 63.2582° N, 10.4637° E | Removed weirs and added six 52–270 m2 gravel beds | [46] |
| Channelization/Loss of substrate | Addition of substrate | Masu salmon | 1994–1998 | Shakotan River, Japan | 43.3310° N, 140.4823° E | Gabions removed, added log dams (3 sets) and gravel (5–10 cm, area unknown) | [10] |
| Channelization/Loss of substrate | Addition of substrate | Brown trout | 1999–2013 | Oulujoki River streams, Finland | 64.9781° N, 25.6053° E | Addition of 77 boulder weirs, 141 log weirs, and boulders. Placed gravel beds (1.38 m2 gravel per 100 m2 of rehabilitated stream) | [56,64] |
| Channelization/Loss of substrate | Addition of substrate | Coho salmon, rainbow trout | 2001–2005 | Smith River reaches, Oregon, USA | 43.7370° N, 124.0787° W | Added 46 boulder weirs/deflectors to six rivers | [43,57] |
| Loss of substrate | Addition of substrate | Salmonids | 1962–1991 | Multiple rivers, California, USA | 36.7783° N, 119.4179° W | Added ~800,000 m3 of gravel to 12 different rivers/reservoirs | [65] |
| Loss of substrate/Sedimentation | Addition of structure/Addition of substrate/Cleaning | Chinook salmon, chum salmon | 1973–1975 | Four rivers and creeks, Washington, USA | 47.7511° N, 120.7401° W | Cleaned gravel with a bulldozer, replaced gravel, and constructed 10 gabion dams to retain gravel | [66,67] |
| Loss of substrate | Addition of structure | Coho salmon, rainbow trout, cutthroat trout | 1981–1983 | East Fork Lobster Creek, Oregon, USA | 44.3027° N, 123.7255° W | Installed gabion weirs (7 sites), boulders (1 site), and log sills (3 sites) to retain gravel | [68] |
| Loss of substrate | Addition of substrate | Chinook salmon, rainbow trout | 1981–1984 | Hurdygurdy Creek, Indiana, USA | 41.6842° N, 123.9036° W | Added 156 boulder clusters/weirs and 25 boulder/rock deflectors across 1280 m of stream | [69] |
| Loss of substrate | Addition of structure | Coho salmon, rainbow trout, pink salmon | 1982 | Sachs Creek, British Columbia, Canada | 53.2527° N, 132.0900° W | Installed three pairs of gabion weirs to retain gravel | [70,71] |
| Loss of substrate | Addition of substrate | Grayling, brown trout, rainbow trout | 1990–1992 | Rhine River impoundment, Germany | 47.6674° N, 9.1484°E | Added 10 m3 of gravel | [72] |
| Loss of substrate | Addition of substrate | Chinook salmon | 1990–1994 | Merced River, California, USA | 37.3491° N, 120.9754°W | Addition of gravel to ~6500 m2 | [50] |
| Loss of substrate | Addition of substrate | Walleye | 1990 | Fox River, Wisconsin, USA | 44.5399° N, 88.0045° W | Added 907 tons of rock (0.6–1.8 m) across 3066 m2 | [73] |
| Loss of substrate | Addition of substrate | Walleye | 1991–1993 | Current River, Ontario, Canada | 48.4693°N, 89.1940° W | Added gravel and cobble in 3 areas (1575 m2 total), randomly placed boulders in same areas | [74] |
| Loss of substrate/Sedimentation | Addition of substrate/Cleaning | Brown trout | 1992–2003 | Hartijokki Stream, Sweden | 66.6930° N, 22.0641° E | Added boulders (600 m3) and restored gravel beds by removing armored layer and raking rocks to remove sediment | [47] |
| Loss of substrate | Addition of substrate | Rainbow trout, coho salmon | 1997–2001 | Keogh River, British Columbia, Canada | 50.6797° N, 127.3484° W | Added >450 boulder and log habitat structures, 905 kg inorganic nutrients as briquettes | [75] |
| Loss of substrate | Addition of substrate | Brown trout | 1999–2000 | Various rivers, Denmark | 56.2639° N, 9.5018° E | Assessed condition of 32 gravel projects 5–14 years post construction (area not specified) | [76] |
| Loss of substrate | Addition of substrate | Chinook salmon | 1999 | Mokelumne River, California, USA | 38.0960°N, 121.5699° W | Addition of gravel bars and boulders (2450 m3 gravel) | [77] |
| Loss of substrate | Addition of substrate | Chinook salmon | 2000–2002 | Mokelumne River, California, USA | 38.0960° N, 121.5699° W | Addition of gravel bars (976 m3 gravel) | [44] |
| Loss of substrate | Addition of substrate | Chinook salmon | 2001 | Mokelumne River, California, USA | 38.0960° N, 121.5699° W | Addition of gravel bars (650 m3 gravel over 152 m) and 10 450–680-kg boulders | [59,78,79] |
| Loss of substrate | Addition of substrate | Chinook salmon | 2002 | Mokelumne River, California, USA | 38.0960° N, 121.5699° W | Addition of gravel (1410 m3) and 3 boulder complexes | [78,80] |
| Loss of substrate | Addition of substrate | Chinook salmon | 2003–2005 | Mokelumne River, California, USA | 38.0960° N, 121.5699° W | Addition of gravel (3522 m3) to create sloped beds; extended by 2359 m3 in 2005 | [78,81] |
| Loss of substrate | Addition of substrate | Chinook salmon, rainbow trout | 2007–2012 | American River, California, USA | 38.6394° N, 120.5839° W | 3 gravel/cobble augmentation sites, each 6350–9707 metric tons | [82,83] |
| Loss of substrate | Addition of substrate | White Sturgeon | 2010 | Columbia River, British Columbia, Canada | 50.2170° N, 115.8500° W | Addition of boulders and armored bed of pebble/cobble (600 m3) | [84] |
| Loss of substrate | Addition of substrate | Chinook salmon | 2010 | Merced River, California, USA | 37.3491° N, 120.9754° W | Added 1.5 million tons of gravel to river | [85] |
| Loss of substrate/Sedimentation | Addition of substrate/Cleaning | Brown trout, grayling, brook lamprey | 2010–2011 | 6 streams in Bavaria, Germany | 4807904° N, 11.4979° E | Added 20 m3 of gravel, raked 50 m2 with excavator, added boulder constrictors, each to six rivers | [86] |
| Loss of substrate | Addition of substrate | River chub, northern hogsucker, black redhorse | 2011 | Cheoah River, North Carolina, USA | 35.4481° N, 83.9396° W | Addition of gravel at 4 sites: 17–64 tons/site | [87] |
| Loss of substrate | Addition of substrate | Chinook salmon, rainbow trout | 2012–2013 | American River, California, USA | 38.6394° N, 120.5839° W | Addition of ~50,000 metric tons of various-sized gravel | [88] |
| Loss of substrate | Addition of substrate | Rainbow trout | 2016 | Lower Whatshan River, British Columbia, Canada | 49.9144° N, 118.1160° W | Added gravel (amount not specified), 18 log jams, 11 boulder placements, 2 log placements along 1.3 km stretch | [89] |
| Sedimentation | Cleaning | Atlantic salmon | 1939–1941 | Moser River, Nova Scotia, Canada | 44.9755° N, 62.2562° W | Cleaned gravel beds by raking, built stone barriers to flush redds (area and number of structures not quantified) | [90] |
| Sedimentation | Cleaning | Salmonids | 1967 | Fish, Slocum, and Lover’s Cove creeks, Alaska, USA | 58.1253° N, 134.0461° W | Used “Riffle Sifter” machine to clean fine sediment from streams (area not quantified) | [91] |
| Sedimentation | Cleaning | Chinook salmon | 1970s–1980s | Tehama-Colusa Canal on Sacramento River, California, USA | 39.9660° N, 122.1350° W | A movable ~43-m baffle was lowered into the channel and created high flows to dislodge sediment. A rotary screen gravel washer was also used to clean gravel by collecting sediment <40 mm and suctioning it to a settling basin | [62,92] |
| Sedimentation/Algae | Cleaning | Sockeye salmon, pink salmon | 1972–1980 | Fraser River spawning channels and three rivers, British Columbia, Canada | 49.1485° N, 122.0878° W | Removal of algae and sediment with a toothed blade on heavy equipment, tilled or sifted gravel with bulldozer, used water jets with air injection to clean gravel in channels and rivers (up to 0.5 m deep), proposed use of dry-gravel cleaner | [93] |
| Sedimentation | Cleaning | Salmonids | Late 1970s–Early 1980s | Palouse River, Idaho and Kennedy Creek and Cedar River, Washington, USA | 46.6215° N, 118.1993° W; 47.5006° N, 122.2162° W | Cleaned river gravel (area not quantified) with prototype machine (“Gravel Gertie”) that cleaned 15–30 mm deep with water jets and extracted sediment to the streambank | [94,95] |
| Sedimentation | Addition of substrate/Sediment traps | Brown trout, brook trout | 1984–1991 | Hay, Waupee, and Chaffee creeks, Wisconsin, USA | 43.94776° N, 89.3215° W | Added sediment traps (3 creeks), rock sills (2 creeks), and gravel beds (2 creeks; 23–50 m long) | [96] |
| Sedimentation | Cleaning | Atlantic salmon | 1984 | LaHave River, Nova Scotia, Canada | 44.3669° N, 64.4698° W | Centrifugal pump used to hydraulically clean gravel (864 m2) | [97] |
| Sedimentation | Cleaning | Coho salmon | 1992 | Little Qualicum River, British Columbia, Canada | 49.3587° N, 124.4845° W | Drained and dredged a constructed spawning channel, then scarified by bulldozer (82 tons of sediment removed) | [98] |
| Sedimentation/Loss of substrate | Sediment traps | Brown trout | 1992–1999 | Four streams, Sweden | 60.1282° N, 18.6435° E | Added 242 artificial spawning grounds (v-shaped deflector of large stones, log weir at narrowest point, gravel placed upstream) and sediment traps | [99] |
| Sedimentation/Algae/Biofouling | Cleaning | Lake sturgeon | 1995–1998 | St. Lawrence River, USA/Canada | 44.5583° N, 75.6440°W | Hand tools used to clear 9 m2 of artificial habitat, pressurized water used to clear 200 m2 | [24] |
| Sedimentation | Cleaning | Brown trout | 1999 | River Kennet, England, UK | 51.4557° N, 0.9570° W | Cleaned ~2520 m2 of substrate with 3 different methods: pump-washing, tractor rotovating, and high-pressure washing | [100] |
| Sedimentation | Addition of substrate/Removal of structure | Walleye | 1999–2003 | Pelican River and Ada Brook, Minnesota, USA | 46.2956° N, 96.1523° W | Added U-shaped gravel/cobble riffles and removed beaver dams | [101] |
| Sedimentation | Addition of substrate | Brown trout | 2003, 2009 | River Stiffkey, England, UK | 52.9574° N, 0.9610° E | Added 13 stone piles (gravel, cobble, and boulders) each 75–200 m2 | [48] |
| Sedimentation | Addition of substrate/Cleaning/Sediment traps | River blackfish, flathead gudgeon, carp gudgeon | 2003–2004 | Glenelg River, Australia | 37.2491° S, 141.8676° E | Sediment was extracted to lower bed height of runs, created/enlarged pools, and constructed sediment traps | [102] |
| Sedimentation | Cleaning/Addition of substrate | Brown trout, grayling | 2004–2008 | Moosach River, Germany | 48.3796° N, 11.7001° E | Sediment was cleaned by sifting with an excavator (3500 m2, 12.5% of study area) and new gravel (amount not specified) was introduced | [103] |
| Sedimentation | Cleaning | Brown trout | 2008–2009 | Moosach River, Germany | 48.3796° N, 11.7001° E | A walking excavator was used to sift gravel (30 m2 cleaned) | [104] |
| Sedimentation | Addition of substrate/Cleaning | Brown trout, grayling | 2010–2011 | Six rivers in Danube, Main/Rhine, and Elbe drainages, Germany | 51.1657° N, 10.4515° E | In each river, gravel was added (20 m3), substratum was raked with an excavator (50 m2), and boulders weirs were placed to constrict current | [105] |
| Sedimentation | Cleaning | Bonneville cutthroat trout | 2010s | Kackley Springs of Bear River, Idaho, USA | 42.5331° N, 111.7925° W | Removed 44 m3 of sediment from 6 reaches (480 m total length) with a Sand Wand | [106] |
| Sedimentation | Cleaning | Brown trout | 2011–2012 | Two streams in Pomahaka River catchment, New Zealand | 45.9065° S, 169.2251° E | Towable water blaster washed out sediment from 50-m stretches in two streams | [107] |
| Sedimentation | Cleaning | Bonneville cutthroat trout | 2011–2014 | Six creeks of Bear River, Idaho, USA | 42.4277° N, 111.7380° W | Removed 14 metric tons of sediment from 6 creeks with a Sand Wand | [108] |
| Sedimentation | Cleaning/Sediment trap | Brook trout | 2012–2014 | Salmon Trout River, Michigan, USA | 46.8614°N, 87.7788° W | Removed sediment with a Sand Wand from a 33 m reach, placed an active sediment collector above the reach | [109] |
| Sedimentation | Cleaning | Common barbel | 2014–2015 | River Great Ouse, England, UK | 52.3178° N, 0.2107° W | Cleaned 6 riffle sites (26.9–98.0 m2) and patches (0.25 m2) with gravel jetting | [110] |
| Sedimentation | Cleaning | White sturgeon | 2016 | Nechako River, British Columbia, Canada | 53.9689° N, 123.7139° W | 7260 m2 (75%) of degraded spawning habitat raked, sifted with mechanical excavator | [111] |
| Sedimentation | Cleaning | Walleye, lake whitefish | 2018–2019 | Saginaw Bay, Lake Huron, Michigan | 43.8453° N, 83.6774° W | Cleaned 14 50-m plots on two reefs using two different experimental cleaning machines: a propulsion sled and a hydro-jet sled | [58] |
| Type of Degradation | Remediation Measures | Scale of Remediation * | Cost * | Considerations |
|---|---|---|---|---|
| Sedimentation | Dredging | Moderate | $$$ | Water depth, permitting, and disposal |
| Pumping | Moderate | $$ | Water depth | |
| Scarification | Small | $ | Water depth, substrate | |
| Increasing flows (dams) | Large | $$$$ | Cost, non-target impacts | |
| Jetting | Small | $ | Water depth, area degraded | |
| Collection devices/traps | Moderate | $$ | Amount of sedimentation, water depth | |
| Loss of substrate | Addition of substrate | Moderate | $$ | Potential fouling, flow regimes |
| Manage for natural flow (dams) | Large | $$$$ | Societal impacts, non-target impacts | |
| Channel modification (weirs) | Moderate | $$$ | Geomorphic impacts | |
| Periphyton growth | Scarification | Small | $$ | Water depth, substrate |
| Jetting | Small | $$ | Water depth, substrate, area degraded | |
| Chemical treatment | Large | $$ | Non-target impacts | |
| Biofouling by invertebrates | Hand removal (divers) | Small | $$$ | Time, cost, area affected |
| Chemical treatment | Large | $$ | Non-target impacts | |
| Jetting | Small | $$ | Water depth, substrate | |
| Biological methods | Moderate–Large | $$ | Non-target impacts |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baetz, A.; Tucker, T.R.; DeBruyne, R.L.; Gatch, A.; Höök, T.; Fischer, J.L.; Roseman, E.F. Review of Methods to Repair and Maintain Lithophilic Fish Spawning Habitat. Water 2020, 12, 2501. https://doi.org/10.3390/w12092501
Baetz A, Tucker TR, DeBruyne RL, Gatch A, Höök T, Fischer JL, Roseman EF. Review of Methods to Repair and Maintain Lithophilic Fish Spawning Habitat. Water. 2020; 12(9):2501. https://doi.org/10.3390/w12092501
Chicago/Turabian StyleBaetz, Audrey, Taaja R. Tucker, Robin L. DeBruyne, Alex Gatch, Tomas Höök, Jason L. Fischer, and Edward F. Roseman. 2020. "Review of Methods to Repair and Maintain Lithophilic Fish Spawning Habitat" Water 12, no. 9: 2501. https://doi.org/10.3390/w12092501
APA StyleBaetz, A., Tucker, T. R., DeBruyne, R. L., Gatch, A., Höök, T., Fischer, J. L., & Roseman, E. F. (2020). Review of Methods to Repair and Maintain Lithophilic Fish Spawning Habitat. Water, 12(9), 2501. https://doi.org/10.3390/w12092501





