Validation of Sample Preparation Methods for Microplastic Analysis in Wastewater Matrices—Reproducibility and Standardization
Abstract
1. Introduction
1.1. Sample Preparation Methods for Removing Organic Matter
1.2. Parameters Used in Microplastic Monitoring
1.3. Research Objectives and State-of-the-Art
2. Materials and Methods
2.1. Selection of Sample Preparation Protocols
2.2. Materials
2.3. Contamination Mitigation and Quality Assurance
2.4. Investigating the Discrepancies in Fenton Reactions
2.5. Investigating the Effects of Sample Preparation on Microplastics
2.5.1. Size Distribution Analysis by Laser Diffraction
2.5.2. FTIR Analysis
2.5.3. Thermal Analysis by TD-Pyr-GC/MS
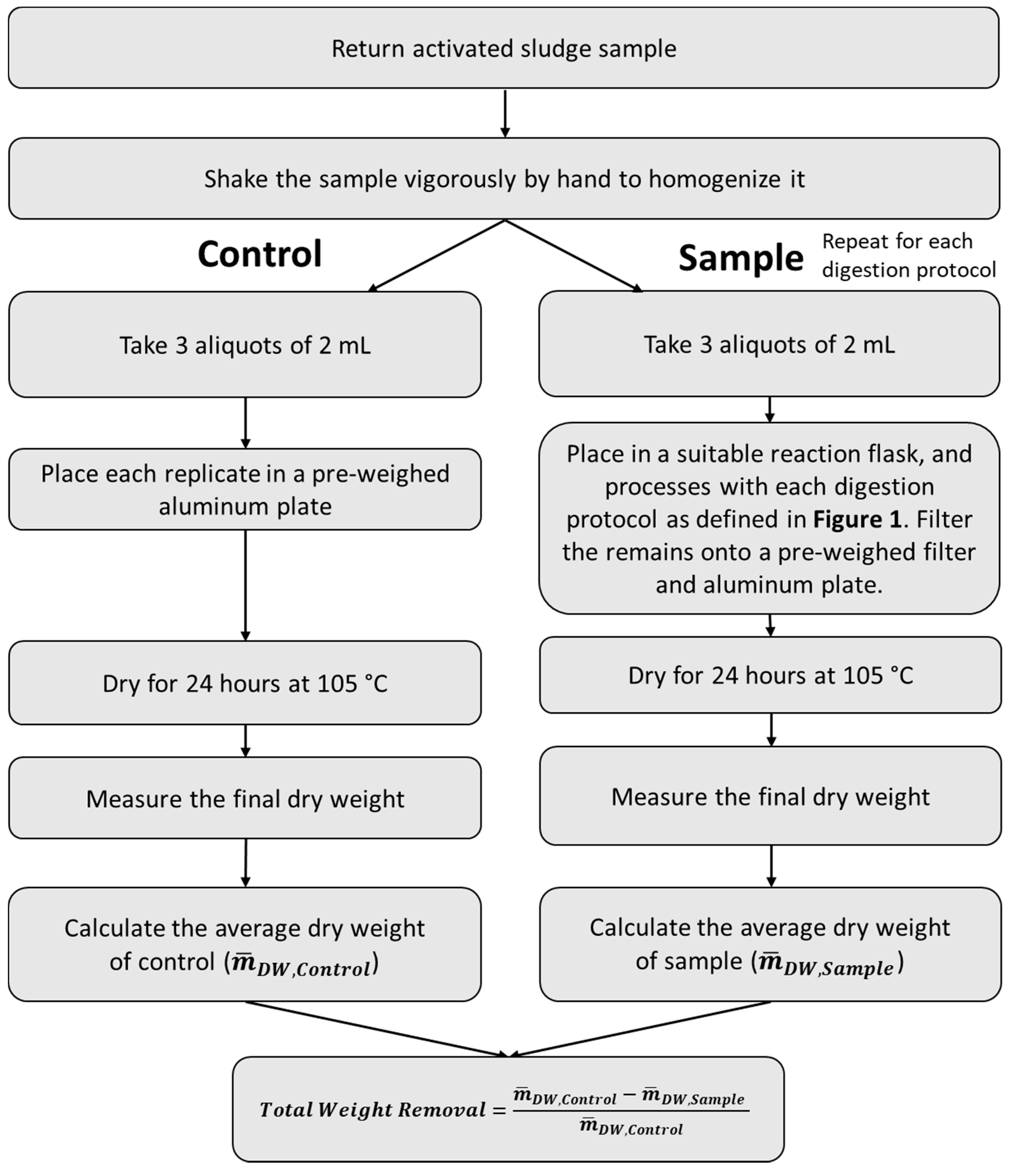
2.6. Determination of the Organic Matter Removal Efficiency from Sludge Samples
3. Results and Discussion
3.1. Discrepancies in Fenton Reactions
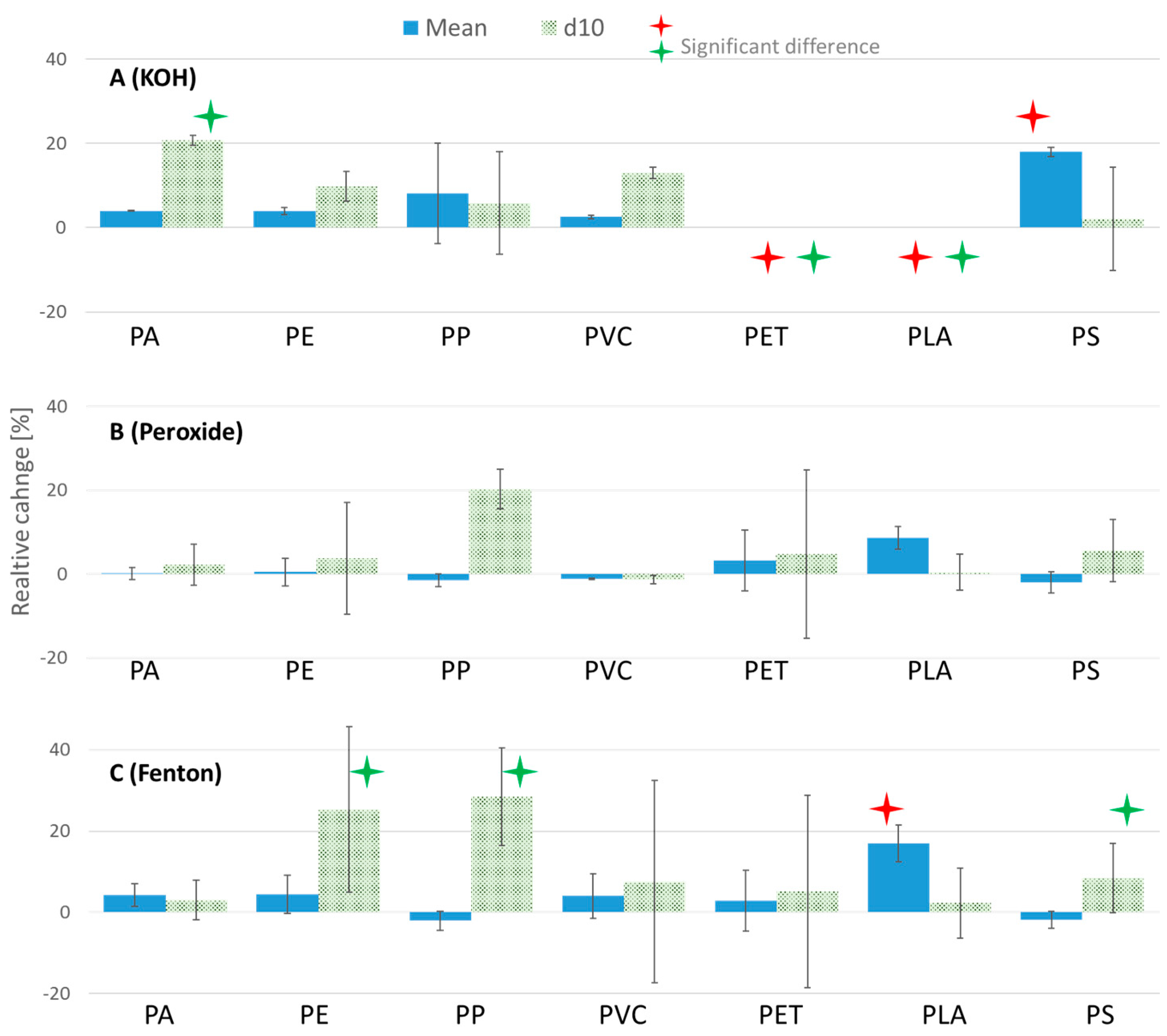
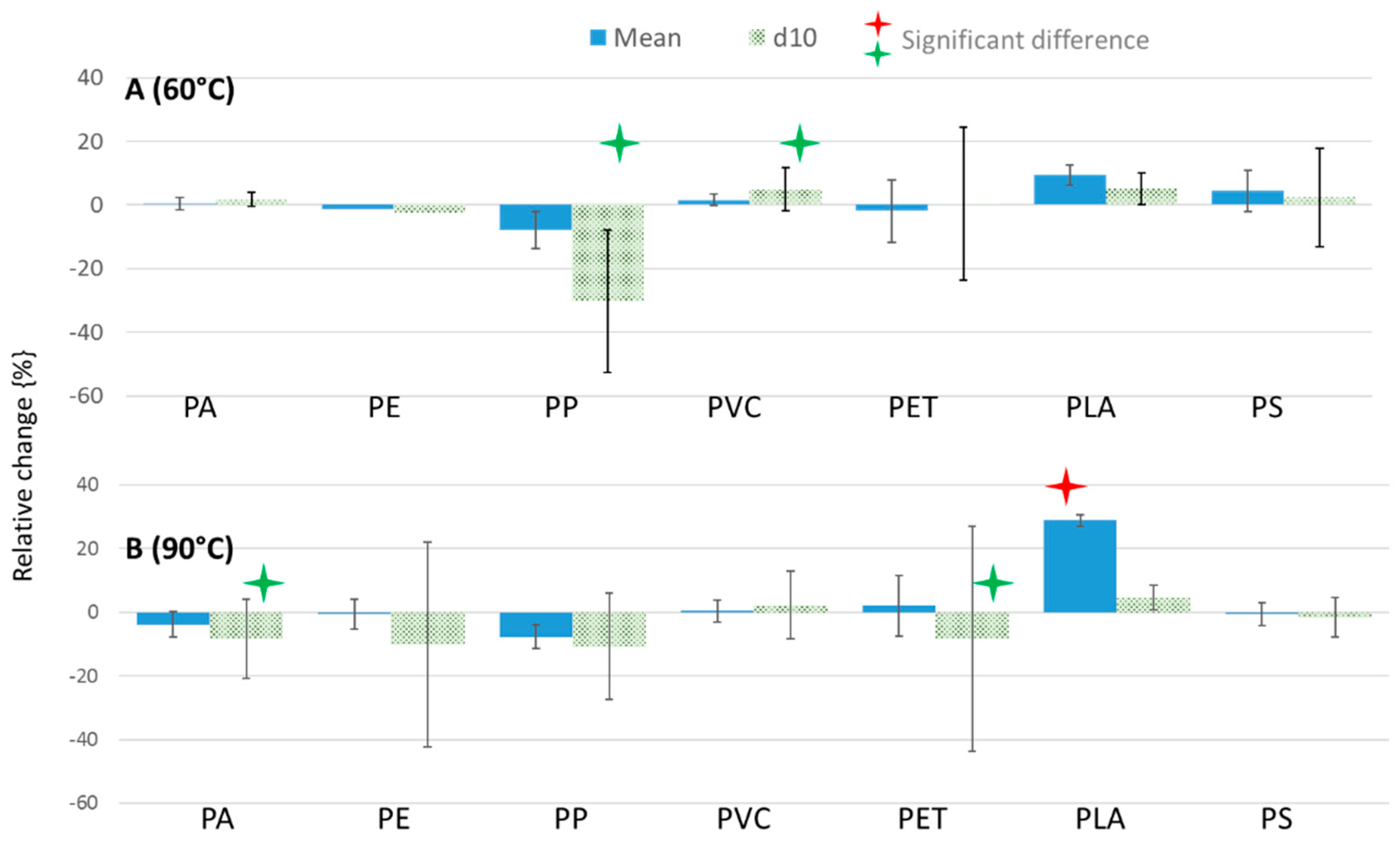
3.2. Investigating the Effects of Sample Preparation on Microplastics
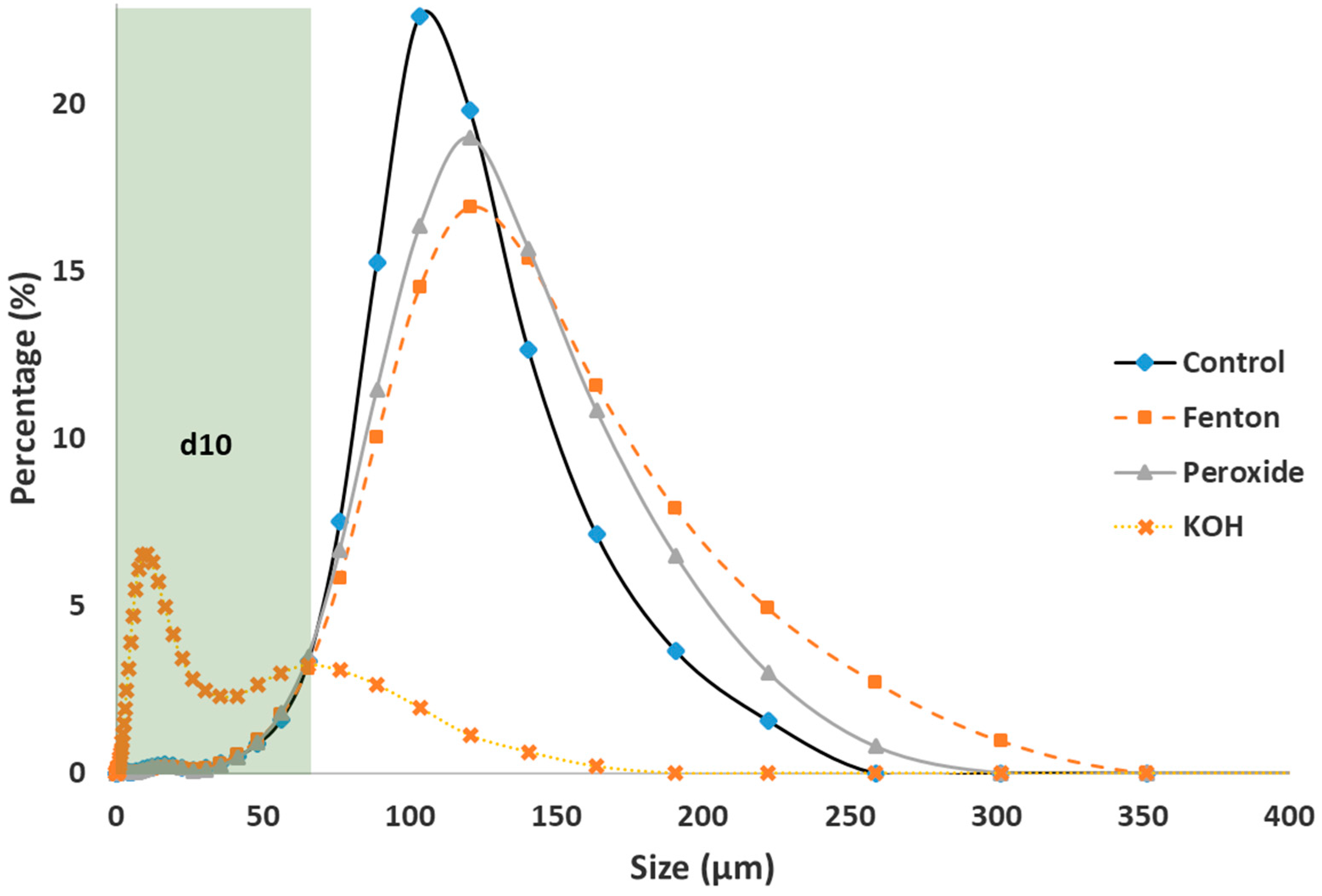
3.2.1. Variation in Size Distribution (Laser Diffraction)
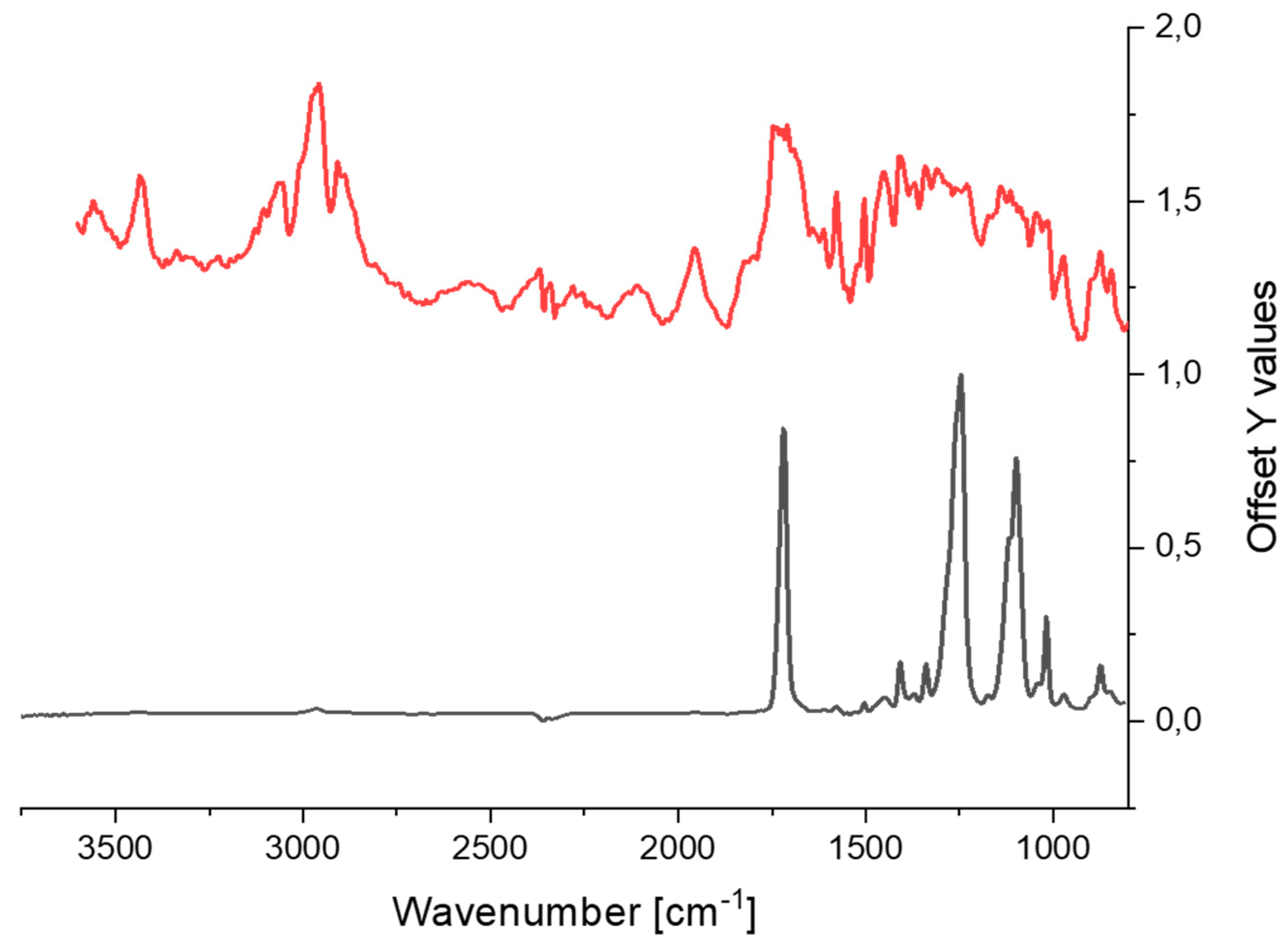
3.2.2. Variation in Infrared Spectra (FTIR)
3.2.3. Variations in Pyr-GC/MS Chromatograms
3.3. Organic Matter Removal Efficiency
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Acrylonitrile-butadiene-styrene | ABS |
| Cellulose acetate | CA |
| Crosslinked polystyrene | PSXL |
| Expanded PS | EPS |
| High-density PE | HPDE |
| Low-density PE | LDPE |
| Linear LDPE | LLDPE |
| Nylon-6 or polyamide-6 | NY6 |
| Nylon12 or polyamide-12 | NY12 |
| Nylon-66 or polyamide-66 | NY66 |
| Poly 1,4-butylene terephthalate | PBT |
| Polyamide | PA |
| Polycarbonate | PC |
| Polyethylene | PE |
| Polyethylene terephthalate | PET |
| Poly(hexamethylene nonanediamide) | Nylon 6/9 |
| Poly(methyl methacrylate) | PMMA |
| Polypropylene | PP |
| Polytetrafluoroethylene | PTFE |
| Polyurethane | PUR |
| Polyvinyl chloride | PVC |
| Styrene acrylate | SA |
| Un-plasticized polyvinyl chloride | uPVC |
| Protocols | Conditions | Matrix | MPs | Effects on Organics | Effects on MP |
|---|---|---|---|---|---|
| H2O2 (30%) [7] | 60 °C for * 24/12 h or 70 °C for * 24 h | Sludge and soil | PP, LDPE, HDPE, PS, PET, NY66, PC, PMMA | Digestion > 80% for sludge and >96% for soil | Degradation of NY66 (at 70 °C), PP (at 70 °C). As for PS; less degradation at 60 °C and more at 70 °C |
| H2O2 (35%) [11] | 50 °C for 96 h | Fish | LDPE, HDPE, PP, PS, PET, PVC, NY6, NY66 | Digestion of 98% | Discoloration of PET, loss of NY6 and NY6 |
| H2O2 (30%) [12] | 55 °C for 7 days | Fish | PE, PS | ** Extraction efficiency of 70% | No report |
| Density separation then H2O2 (15%) [12] | 50 °C overnight | Fish | PE, PS | ** Extraction efficiency of 95% | No effects |
| H2O2 (30%) [13] | RT. For 7 days | Marine sediment | PVC, PET, PA, ABS, PC, PUR, PP, LDPE, LLDPE, HPDE | 50% digestion and the rest were discolored | Discoloration or degradation in PA, PC, PP, PET, LLDPE, PVC, PUR, LDPE |
| H2O2 (35%) [13] | RT. For 7 days | Marine sediment | PVC, PP, LDPE, PE, HDPE, PET, PUR, PS, PC, PA, ABS, EPS | 92% digestion and discoloration | Size reduction PP and PE |
| H2O2 (30%) then density separation [14] | 70 °C overnight | Sludge | PE | ** Extraction efficiency of 78% | No adverse effects reported |
| Protocols | Conditions | Matrix | MPs | Effects on Organics | Effects on MP | |
|---|---|---|---|---|---|---|
| H2O2 (30%) + 20 g/L FeSO4 7H2O2 1:1 (v/v) [7] | <40 °C (Ice bath) | *24 h/20 min | Sludge and soil | PP, LDPE, HDPE, PS, PET, NY66, PC, PMMA | Digestion 87% for sludge and 106% for soil | No adverse effects |
| H2O2 (30%) + 15 g/L FeSO4 7H2O2 1:1 (v/v) [15] | 50 °C for 1 h. | Plant and animal tissues | PE, PP, PS, PET, PVC, CA, PA | Digestion of 72.6% of fish tissue, 100% of algae, 26.3% driftwood, 17.5% paraffin | Degradation of CA | |
| H2O2 (30%) + 15 g/L FeSO4 7H2O2 1:1 (v/v) [16] | 75 °C for >35 min | Water, beach and sediment | None tested | No reports | No reports | |
| H2O2 (30%) + 20 g/L FeSO4 7H2O2 (2:1 v/v) [17] | RT. (no ice bath) | 10 min reaction + 10 min cooling | Sludge | PE, PP, PVC, PA | Effective removal (No exact details provided) | No observed effects |
| Protocols | Conditions | Matrix | MPs | Effects on Organics | Effects on MP |
|---|---|---|---|---|---|
| HNO3 (69%) [11] | 25 °C for 96 h | Fish | LDPE, HDPE, PP, PS, PET, PVC, NY6, NY66 | Removal >95% | Loss of NY6, NY66. Melting of LDPE, HDPE, PP. Recovery of PVC 69%. Discoloration of others |
| HNO3 (55%) [20] | Based on fish size: RT. For 3–10 h or 80 °C for 10–20 min | Fish | HDPE, PA, Nylon 6/9, PBT, PVC | Complete digestion of the fish | Tested at room temperature for 1 month. Loss of Nylon within 24 h, the rest showed no significant changes after 1 month. |
| HNO3 (22.5 M) [21] | RT. Overnight then boiling for 2 h | Mussels | PS, PA | Good digestion of the tissues | PA fibers (30 × 200 µm) were lost. PA particles 100 × 400 µm were 98% recovered. PS particles 30 µm and 10 µm were recovered. |
| HNO3 (22.5 M) [21] | 60 °C for 1 h. then 100 °C for 1 h | Mussels | PS, PA | Satisfactory digestion of the tissues | PS particles melting together when exposed to the reagent directly. This did not occur when PS was embedded in the tissues. PA was completely lost |
| HNO3 (22.5 M) [12] | RT. overnight then boiling for 30 min | Fish | PS, PE | ** Extraction efficiency of only 4% | Almost complete loss of PS and PE |
| HNO3 (65%) [22] | RT. overnight then 60 °C for 2 h | None | LDPE, HDPE, PP, NY12, PS | Not tested due to damaged polymers | Melting of NY12, yellowing of the rest |
| HNO3 (35%) [25] | 60 °C overnight | Mussels | PET, HDPE, PVC, PA | Complete digestion | Melting together of PET, HDPE, loss of PA |
| HCL (37%) [11] | 25 °C for 96 h | Fish | LDPE, HDPE, PP, PS, PET, PVC, NY6, NY66 | Digestion efficiency >95% | Loss of NY6, NY66. Melting and clumping of PET |
| HCL (20%) [13] | RT. for 7 days | Marine sediment | PVC, PET, PA, ABS, PC, PUR, PP, LDPE, LLDPE, HPDE | No complete dissolution of any biogenic organic matter | Not reported |
| Protocols | Conditions | Matrix | MPs | Effects on Organics | Effects on MP |
|---|---|---|---|---|---|
| KOH (10%) [7] | 60 °C for 24 h | Sludge and soil | PP, LDPE, HDPE, PS, PET, NY66, PC, PMMA | Digestion of sludge: 57% And for soil: 35% | A slight weight decrease for PC |
| KOH (10%) [11] | 25 °C for 96 h or 40 °C for 48 h or 50 °C for 36 h or 60 °C for 24 h | Fish | LDPE, HDPE, PP, PS, PET, PVC, NY6, NY66 | Removal >95% (all temperatures) | Yellowing of NY66. Reduction in PVC and PET recovery that worsens with increased temperature |
| KOH (10%) [22] | 0 °C for 24 h | Mussels, crabs, fish | CA, HDPE, LDPE, NY6, NY12, PC, PET, PMMA, PP, PS, PSXL, PTFE, PUR, uPVC, EPS | Digestion efficiency > 99.5% | Degradation of CA |
| KOH (56, 224 g/L) [26] | RT. For 14 days | None | Cera Microcrystalline, PE | ** Extraction efficiency > 95% | Discoloration of cera microcrystalline and PE (for 224 g/L) |
| KOH (10%) [28] | RT. For 2–3 weeks | Fish | No spiking: PE, PP, PET, SA | Digestion satisfactory | None tested |
| KOH (10%) [29] | 60 °C for 24 h | Vegetal | PP, PE, PVC, PUR, PET, PS | No digestion observed | No effects observed |
| NaOH (1, 10 M) [7] | 60 °C for 24 h | Sludge and soil | PP, LDPE, HDPE, PS, PET, NY66, PC, PMMA | Digestion of sludge: 61% (1M) 67% (10M) and soil: 68% (1 M) 65% (10 M) | Degradation of PET, PC (even more with 10 M) |
| NaOH (20, 30, 40, 50%) [13] | RT. For 7 days | Marine sediment | PVC, PET, PA, ABS, PC, PUR, PP, LDPE, LLDPE, HPDE | No complete dissolution of any biogenic organic matter. Strongest reaction with NaOH (20%) | Not reported |
| NaOH (10 M) [22] | 60 °C for 24 h | None | CA, HDPE, LDPE, NY6, NY12, PC, PET, PMMA, PP, PS, PSXL, PTFE, PUR, uPVC, EPS | Not tested due to damage to polymers | Degradation of CA, PC, PET |
| NaOH (1 M) [25] | 60 °C overnight | Mussels | PET, HDPE, PVC, PA | Complete digestion and extraction efficiency of 93% | No significant effects |
| NaOH (10 M) [27] | 60 °C for 24 h | Zooplankton | PS, PA, PET, PE, uPVC | Digestion efficiency of 91% | Partial degradation of PA, clumping of PE, yellowing of uPVC, partial loss of PS |
| NaOH (10 M) [29] | 60 °C for 24 h | Vegetal | PP, PE, PVC, PUR, PET, PS | Almost no digestion | Degradation of PET |
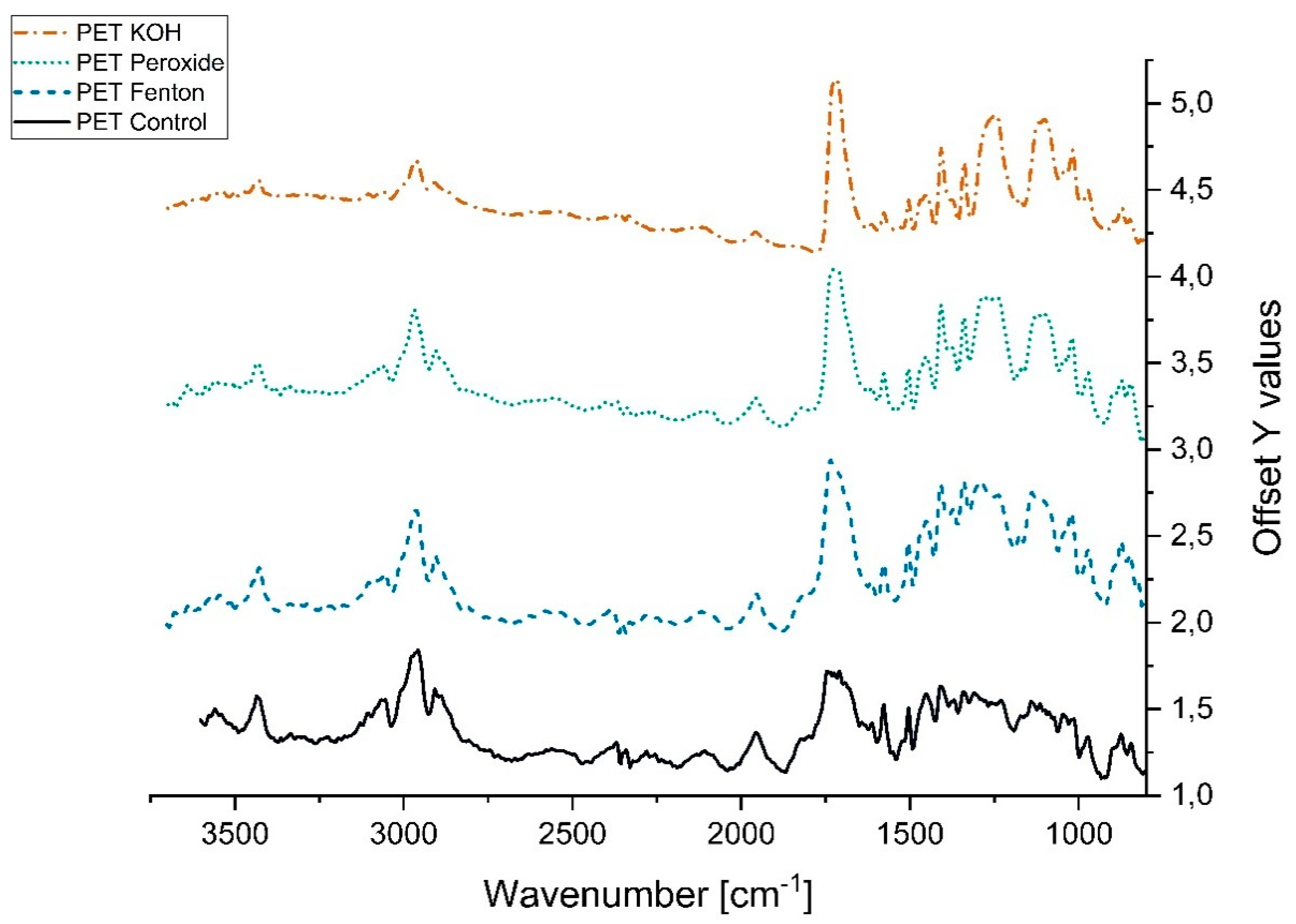
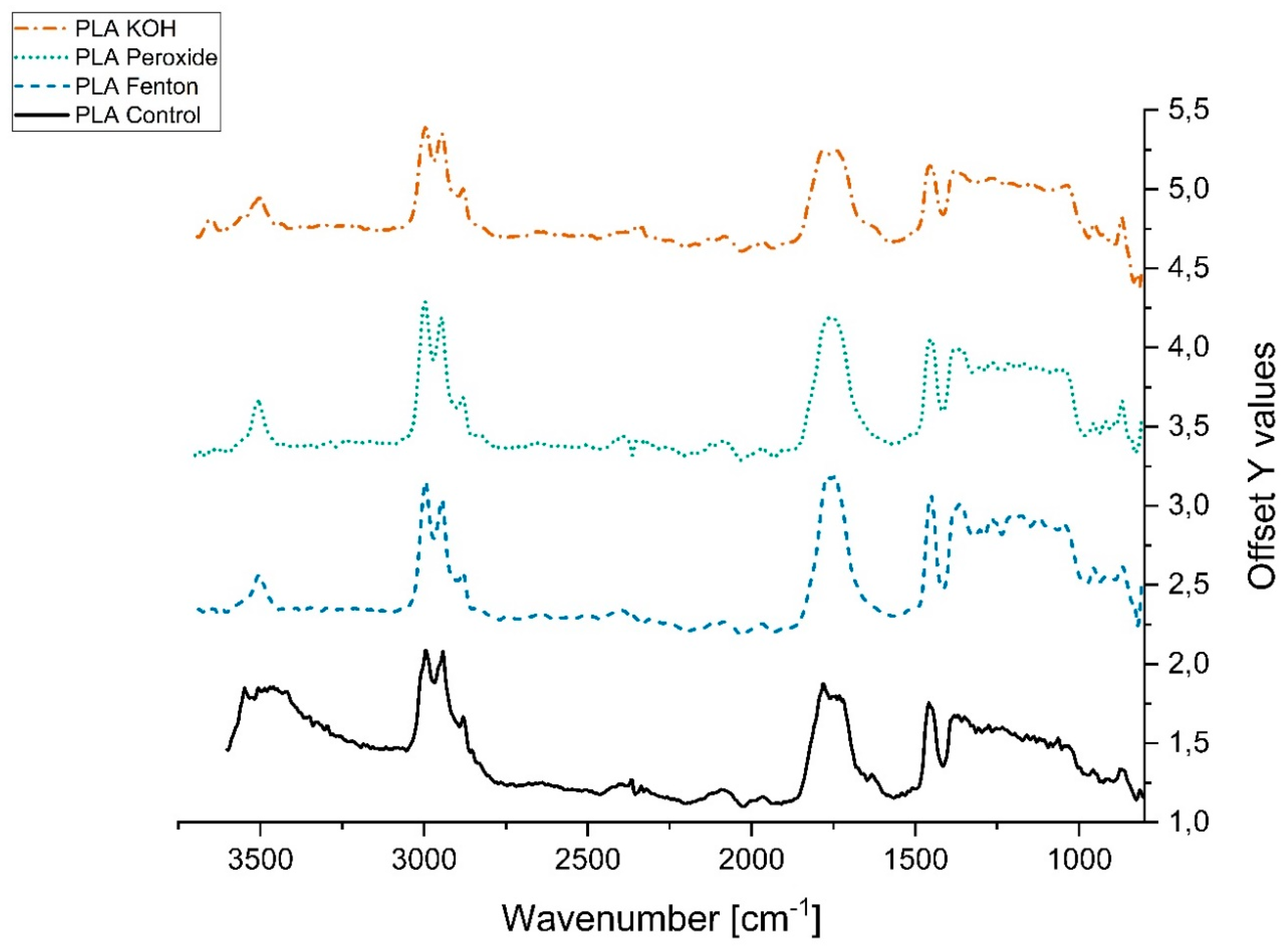
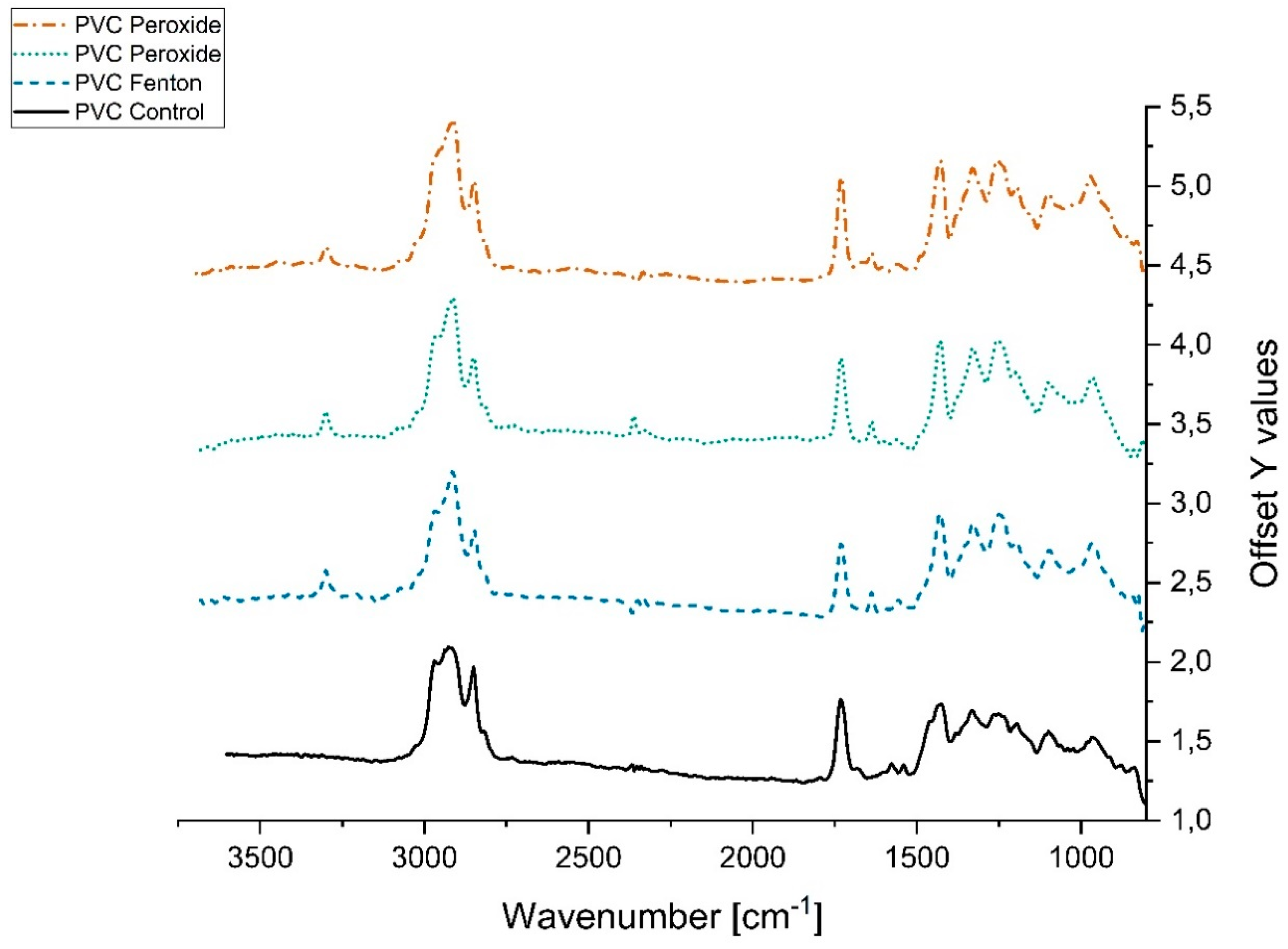
References
- Underwood, A.J.; Chapman, M.G.; Browne, M.A. Some problems and practicalities in design and interpretation of samples of microplastic waste. Anal. Methods 2017, 9, 1332–1345. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Marine pollution. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, N.B.; Hüffer, T.; Thompson, R.C.; Hassellöv, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsson, T.; Brennholt, N.; Cole, M.; et al. Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris. Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- GESAMP. Sources, Fate and Effects of Microplastics in the Marine Environment: A Global Assessment; Kershaw, P., Ed.; IMO/FAO/UNESCO-IOC/UNIDO/WMO/IAEA/UN/UNEP/UNDP Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection: Londen, UK, 2015. [Google Scholar]
- Braun, U.; Jekel, M.; Gerdts, G.; Ivleva, N.P.; Jens, R. Microplastics Analytics Sampling, Preparation and Detection Methods; German Federal Ministry of Education And Research: Berlin, Germany, 2018.
- Ivleva, N.P.; Wiesheu, A.C.; Niessner, R. Microplastic in aquatic ecosystems. Angew. Chem. Int. Ed. 2017, 56, 1720–1739. [Google Scholar] [CrossRef] [PubMed]
- Hurley, R.R.; Lusher, A.L.; Olsen, M.; Nizzetto, L. Validation of a method for extracting microplastics from complex, organic-rich, environmental matrices. Environ. Sci. Technol. 2018, 52, 7409–7417. [Google Scholar] [CrossRef]
- Tagg, A.S.; Sapp, M.; Harrison, J.P.; Ojeda, J.J. Identification and quantification of microplastics in wastewater using focal plane array-based reflectance Micro-FT-IR imaging. Anal. Chem. 2015, 87, 6032–6040. [Google Scholar] [CrossRef]
- Dyachenko, A.; Mitchell, J.; Arsem, N. Extraction and identification of microplastic particles from secondary wastewater treatment plant (WWTP) effluent. Anal. Methods 2017, 9, 1412–1418. [Google Scholar] [CrossRef]
- Schwaferts, C.; Niessner, R.; Elsner, M.; Ivleva, N.P. Methods for the analysis of submicrometer- and nanoplastic particles in the environment. TrAC Trends Anal. Chem. 2019, 112, 52–65. [Google Scholar] [CrossRef]
- Karami, A.; Golieskardi, A.; Choo, C.K.; Romano, N.; Ho, Y.B.; Salamatinia, B. A high-performance protocol for extraction of microplastics in fish. Sci. Total Environ. 2017, 578, 485–494. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Regoli, F. Experimental development of a new protocol for extraction and characterization of microplastics in fish tissues: First observations in commercial species from adriatic sea. Mar. Environ. Res. 2015, 111, 18–26. [Google Scholar] [CrossRef]
- Nuelle, M.T.; Dekiff, J.H.; Remy, D.; Fries, E. A new analytical approach for monitoring microplastics in marine sediments. Environ. Pollut. 2014, 184, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Sujathan, S.; Kniggendorf, A.-K.; Kumar, A.; Roth, B.; Rosenwinkel, K.-H.; Nogueira, R. Heat and bleach: A cost-efficient method for extracting microplastics from return activated sludge. Arch. Environ. Contam. Toxicol. 2017, 73, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Prata, J.C.; da Costa, J.P.; Girão, A.V.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T. Identifying a quick and efficient method of removing organic matter without damaging microplastic samples. Sci. Total Environ. 2019, 686, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Masura, J.; Baker, J.E.; Foster, G.D.; Arthur, C.; Herring, C. Laboratory Methods for the Analysis of Microplastics in the Marine Environment: Recommendations for Quantifying Synthetic Particles in Waters and Sediments; National Oceanic and Atmospheric Administration: Silver Spring, MD, USA, 2015.
- Tagg, A.S.; Harrison, J.P.; Ju-Nam, Y.; Sapp, M.; Bradley, E.L.; Sinclair, C.J.; Ojeda, J.J. Fenton’s reagent for the rapid and efficient isolation of microplastics from wastewater. Chem. Commun. 2017, 53, 372–375. [Google Scholar] [CrossRef]
- Zhang, H.; Heung, J.C.; Huang, C.P. Optimization of fenton process for the treatment of landfill leachate. J. Hazard. Mater. 2005, 125, 166–174. [Google Scholar] [CrossRef]
- Flotron, V.; Delteil, C.; Padellec, Y.; Camel, V. Removal of sorbed polycyclic aromatic hydrocarbons from soil, sludge and sediment samples using the Fenton’s reagent process. Chemosphere 2005, 59, 1427–1437. [Google Scholar] [CrossRef]
- Naidoo, T.; Goordiyal, K.; Glassom, D. Are nitric acid (HNO3) digestions efficient in isolating microplastics from juvenile fish? Water. Air. Soil Pollut. 2017, 228, 1–11. [Google Scholar] [CrossRef]
- Claessens, M.; Van Cauwenberghe, L.; Vandegehuchte, M.B.; Janssen, C.R. New techniques for the detection of microplastics in sediments and field collected organisms. Mar. Pollut. Bull. 2013, 70, 227–233. [Google Scholar] [CrossRef]
- Dehaut, A.; Cassone, A.-L.; Frère, L.; Hermabessiere, L.; Himber, C.; Rinnert, E.; Rivière, G.; Lambert, C.; Soudant, P.; Huvet, A.; et al. Microplastics in seafood: Benchmark protocol for their extraction and characterization. Environ. Pollut. 2016, 215, 223–233. [Google Scholar] [CrossRef]
- Rocha-Santos, T.; Duarte, A.C. A critical overview of the analytical approaches to the occurrence, the fate and the behavior of microplastics in the environment. TrAC Trends Anal. Chem. 2015, 65, 47–53. [Google Scholar] [CrossRef]
- Qiu, Q.; Tan, Z.; Wang, J.; Peng, J.; Li, M.; Zhan, Z. Extraction, enumeration and identification methods for monitoring microplastics in the environment. Estuar. Coast. Shelf Sci. 2016, 176, 102–109. [Google Scholar] [CrossRef]
- Catarino, A.I.; Thompson, R.; Sanderson, W.; Henry, T.B. Development and optimization of a standard method for extraction of microplastics in mussels by enzyme digestion of soft tissues. Environ. Toxicol. Chem. 2017, 36, 947–951. [Google Scholar] [CrossRef] [PubMed]
- Munno, K.; Helm, P.A.; Jackson, D.A.; Rochman, C.; Sims, A. Impacts of temperature and selected chemical digestion methods on microplastic particles. Environ. Toxicol. Chem. 2018, 37, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.; Webb, H.; Lindeque, P.K.; Fileman, E.S.; Halsband, C.; Galloway, T.S. Isolation of microplastics in biota-rich seawater samples and marine organisms. Sci. Rep. 2014, 4, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Foekema, E.M.; De Gruijter, C.; Mergia, M.T.; Van Franeker, J.A.; Murk, A.J.; Koelmans, A.A. Plastic in north sea fish. Environ. Sci. Technol. 2013, 47, 8818–8824. [Google Scholar] [CrossRef]
- Herrera, A.; Garrido-Amador, P.; Martínez, I.; Samper, M.D.; López-Martínez, J.; Gómez, M.; Packard, T.T. Novel methodology to isolate microplastics from vegetal-rich samples. Mar. Pollut. Bull. 2018, 129, 61–69. [Google Scholar] [CrossRef]
- Mintenig, S.M.; Int-Veen, I.; Löder, M.G.J.; Primpke, S.; Gerdts, G. Identification of microplastic in effluents of waste water treatment plants using focal plane array-based micro-fourier-transform infrared imaging. Water Res. 2017, 108, 365–372. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, L.; Li, D. Microplastic in three urban estuaries, China. Environ. Pollut. 2015, 206 (Suppl. C), 597–604. [Google Scholar] [CrossRef]
- Gewert, B.; Ogonowski, M.; Barth, A.; MacLeod, M. Abundance and composition of near surface microplastics and plastic debris in the stockholm archipelago, Baltic Sea. Mar. Pollut. Bull. 2017, 120, 292–302. [Google Scholar] [CrossRef]
- Van Cauwenberghe, L.; Vanreusel, A.; Mees, J.; Janssen, C.R. Microplastic pollution in deep-sea sediments. Environ. Pollut. 2013, 182 (Suppl. C), 495–499. [Google Scholar] [CrossRef]
- Renner, G.; Schmidt, T.C.; Schram, J. Characterization and quantification of microplastics by infrared spectroscopy. Compr. Anal. Chem. 2017, 75, 67–118. [Google Scholar] [CrossRef]
- Imhof, H.K.; Ivleva, N.P.; Schmid, J.; Niessner, R.; Laforsch, C. Contamination of beach sediments of a subalpine lake with microplastic particles. Curr. Biol. 2013, 23, R867–R868. [Google Scholar] [CrossRef] [PubMed]
- Carr, S.A.; Liu, J.; Tesoro, A.G. Transport and fate of microplastic particles in wastewater treatment plants. Water Res. 2016, 91, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Cabernard, L.; Durisch-Kaiser, E.; Vogel, J.C.; Rensch, D.; Niederhauser, P.; Gewässerschutz, A.W.E.L. Mikroplastik in Abwasser Und Gewässern; Aqua & Gas N°7/8: Zurich, Switzerland, 2016. [Google Scholar]
- Aramyan, S.M. Advances in fenton and fenton based oxidation processes for industrial effluent contaminants Control-A review. Int. J. Environ. Sci. Nat. Resour. 2017, 2, 1–18. [Google Scholar] [CrossRef]
- ISO—ISO 13320:2009—Particle Size Analysis—Laser Diffraction Methods; International Organization for Standardization: Geneva, Switzerland, 2009.
- Ochiai, N.; Sasamoto, K.; Kanda, H.; Yamagami, T.; David, F.; Tienpont, B.; Sandra, P. Optimization of a multi-residue screening method for the determination of 85 pesticides in selected food matrices by stir bar sorptive extraction and thermal desorption GC-MS. J. Sep. Sci. 2005, 28, 1083–1092. [Google Scholar] [CrossRef]
- DIN 38409-1—1987-01, German Standard Methods for the Examination of Water, Waste Water and Sludge; Parameters Characterizing Effects and Substances (Group H); Determination of Total Dry Residue, Filtrate Dry Residue and Residue on Ignition (H 1); German Institute for Standardisation: Berlin, Germany, 1987.
- Kühn, S.; van Werven, B.; van Oyen, A.; Meijboom, A.; Bravo Rebolledo, E.L.; van Franeker, J.A. The use of potassium hydroxide (KOH) solution as a suitable approach to isolate plastics ingested by marine organisms. Mar. Pollut. Bull. 2017, 115, 86–90. [Google Scholar] [CrossRef]
- Lunt, J. Large-Scale production, properties and commercial applications of poly lactic acid polymers. Polym. Degrad. Stab. 1998, 59, 145–152. [Google Scholar] [CrossRef]
- Zhang, H.; Rankin, A.; Ward, I.M. Determination of the end-group concentration and molecular weight of poly (Ethylene Naphthalene-2,6-Dicarboxylate) using infra-red spectroscopy. Polymer (Guildf) 1996, 37, 1079–1085. [Google Scholar] [CrossRef]
- Myungwan, H. Chemical Depolymerization Of Pet Bottles Via Methanolysis And Hydrolysis Myungwan Han. In Recycling of Polyethylene Terephthalate Bottles; Sabu, T., Ajay, V.R., Krishnan, K., Abitha, V., Martin, G.T., Eds.; William Andrew Publishing: New York, NY, USA, 2018. [Google Scholar]
- von der Esch, E.; Lanzinger, M.; Kohles, A.J.; Schwaferts, C.; Weisser, J.; Hofmann, T.; Glas, K.; Elsner, M.; Ivleva, N.P. Simple generation of suspensible secondary microplastic reference particles via ultrasound treatment. Front. Chem. 2020, 8, 169. [Google Scholar] [CrossRef]
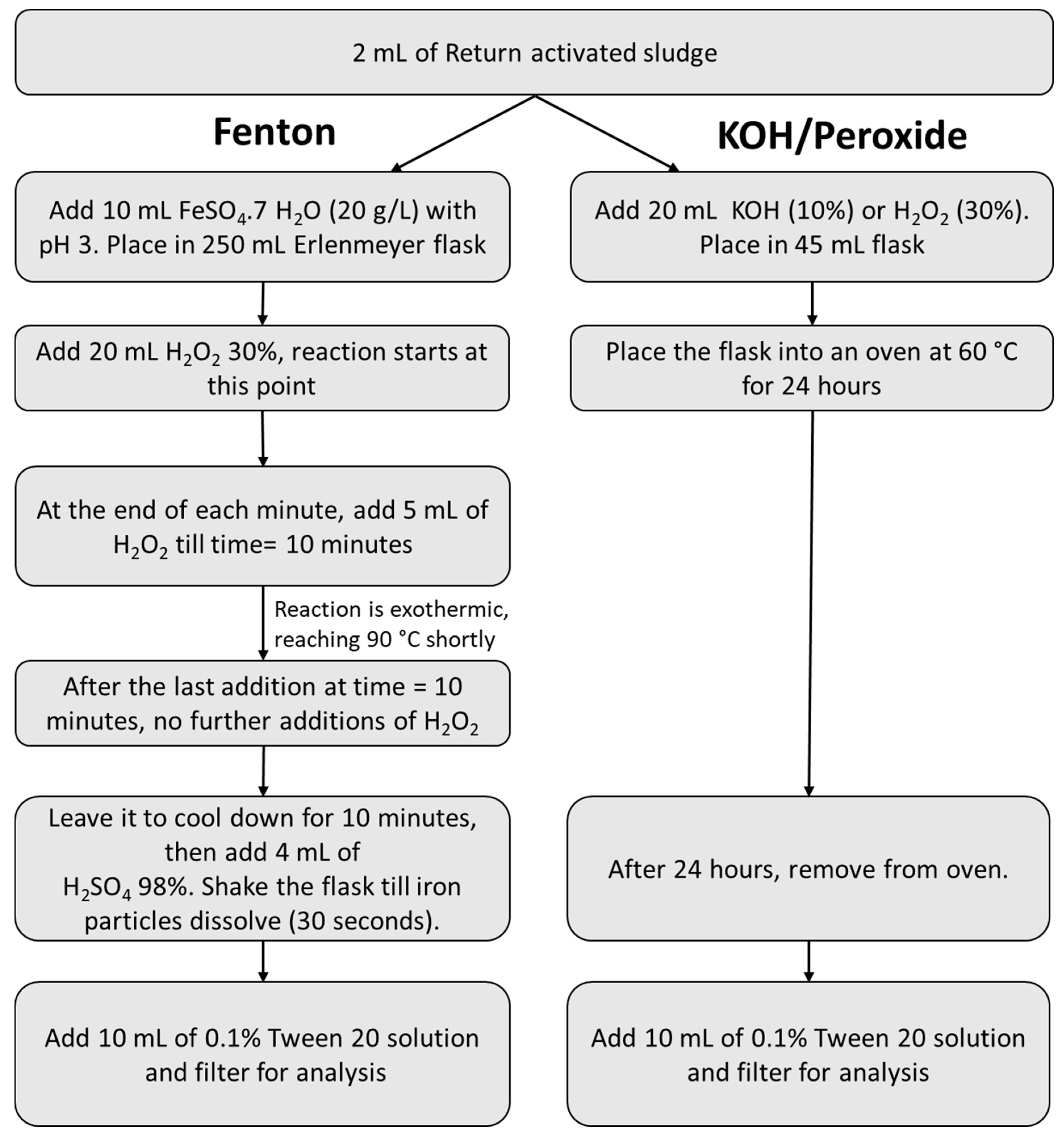



| Protocols | Temperature | Time |
|---|---|---|
| Fenton (30% H2O2 + 20 g/L FeSO4) [17] | Unregulated | 10 min + 10 min cooling |
| KOH (10%) [22] | 60 °C | 24 h |
| H2O2 (30%) [7] | 60 °C | 24 h |
| Protocols | Description |
|---|---|
| Control | 5 mL UPW @ Room Temperature |
| Fenton | As described in Figure 1 (2 mL FeSO4 as a starting volume) |
| H2O2 | As described in Figure 1 (5 mL H2O2) |
| KOH | As described in Figure 1 (5 mL KOH) |
| ** Temperature control 60 °C | 5 mL UPW @ 60 °C and 24 h |
| ** Temperature control 90 °C | 5 mL UPW @ 90 °C and 20 Minutes |
| H2O2 Batches | Tmax [°C] | pH @ Tmax [-] | Time Till Tmax [sec] |
|---|---|---|---|
| (i) | 86.9 ± 0.9 | 1.62 ± 0.03 | 157 ± 4 |
| (ii) | 87.2 ± 0.2 | 1.67 ± 0.06 | 167 ± 19 |
| (iii) | 70.4 ± 23 | 1.72 ± 0.23 | 510 ± 127 |
| (iv) | 85 ± 0.4 | 1.67 ± 0.05 | 173 ± 11 |
| (v) | 84.9 ± 1.7 | 1.69 ± 0.04 | 192 ± 5 |
| Reaction Type | Tmax [°C] | pH @ Tmax [-] | Time to Tmax [sec] |
|---|---|---|---|
| Type (I) (batch (ii)) | 84.1 ± 3.4 | 1.65 ± 0.09 | 228 ± 3 |
| Type (II) (batch (iii)) | 60.8 ± 29.3 | 1.7 ± 0.25 | 554 ± 190 |
| Polymer Type | Particle Treatment | Pyrolysis Products in TD [%]/Standard Deviation [%] |
|---|---|---|
| PA | untreated | 74.5/19.0 |
| Fenton | 17.1/12.65 | |
| H2O2 | 3.0/3.67 | |
| PS | untreated | 79.3/12.5 |
| Fenton | 78.8/16.9 | |
| H2O2 | 71.9/10.9 |
| Protocol | Total Weight Removal | Corresponding Organic Matter Removal * |
|---|---|---|
| Fenton | 83.5% ± 1.8% | 118.4% ± 2.6% |
| H2O2 | 71.3% ± 1.2% | 101.1% ± 1.6% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Azzawi, M.S.M.; Kefer, S.; Weißer, J.; Reichel, J.; Schwaller, C.; Glas, K.; Knoop, O.; Drewes, J.E. Validation of Sample Preparation Methods for Microplastic Analysis in Wastewater Matrices—Reproducibility and Standardization. Water 2020, 12, 2445. https://doi.org/10.3390/w12092445
Al-Azzawi MSM, Kefer S, Weißer J, Reichel J, Schwaller C, Glas K, Knoop O, Drewes JE. Validation of Sample Preparation Methods for Microplastic Analysis in Wastewater Matrices—Reproducibility and Standardization. Water. 2020; 12(9):2445. https://doi.org/10.3390/w12092445
Chicago/Turabian StyleAl-Azzawi, Mohammed S. M., Simone Kefer, Jana Weißer, Julia Reichel, Christoph Schwaller, Karl Glas, Oliver Knoop, and Jörg E. Drewes. 2020. "Validation of Sample Preparation Methods for Microplastic Analysis in Wastewater Matrices—Reproducibility and Standardization" Water 12, no. 9: 2445. https://doi.org/10.3390/w12092445
APA StyleAl-Azzawi, M. S. M., Kefer, S., Weißer, J., Reichel, J., Schwaller, C., Glas, K., Knoop, O., & Drewes, J. E. (2020). Validation of Sample Preparation Methods for Microplastic Analysis in Wastewater Matrices—Reproducibility and Standardization. Water, 12(9), 2445. https://doi.org/10.3390/w12092445






