Understanding the Biogeochemical Impacts of Fish Farms Using a Benthic-Pelagic Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Modeling
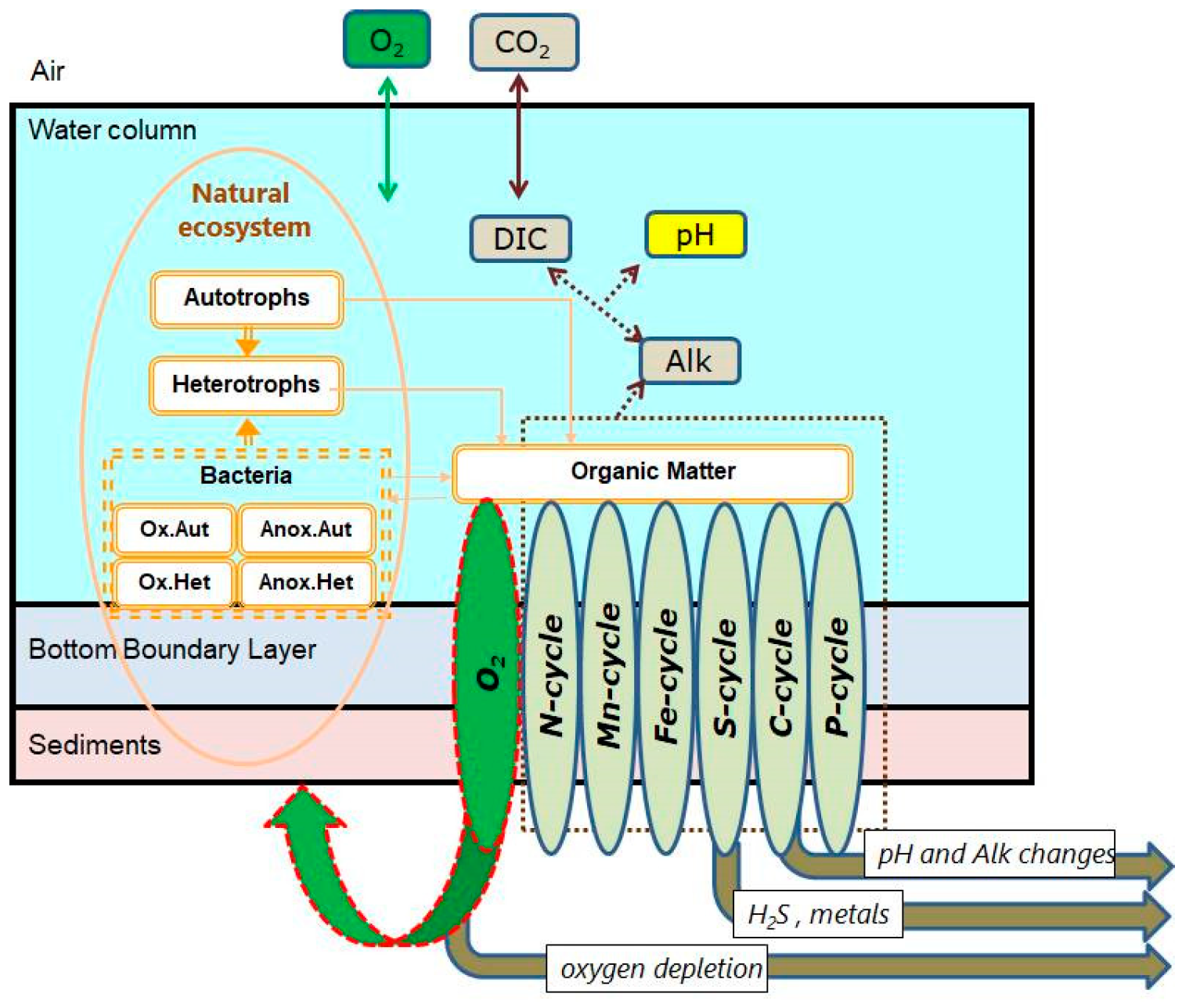
2.2. Biogeochemical Model BROM
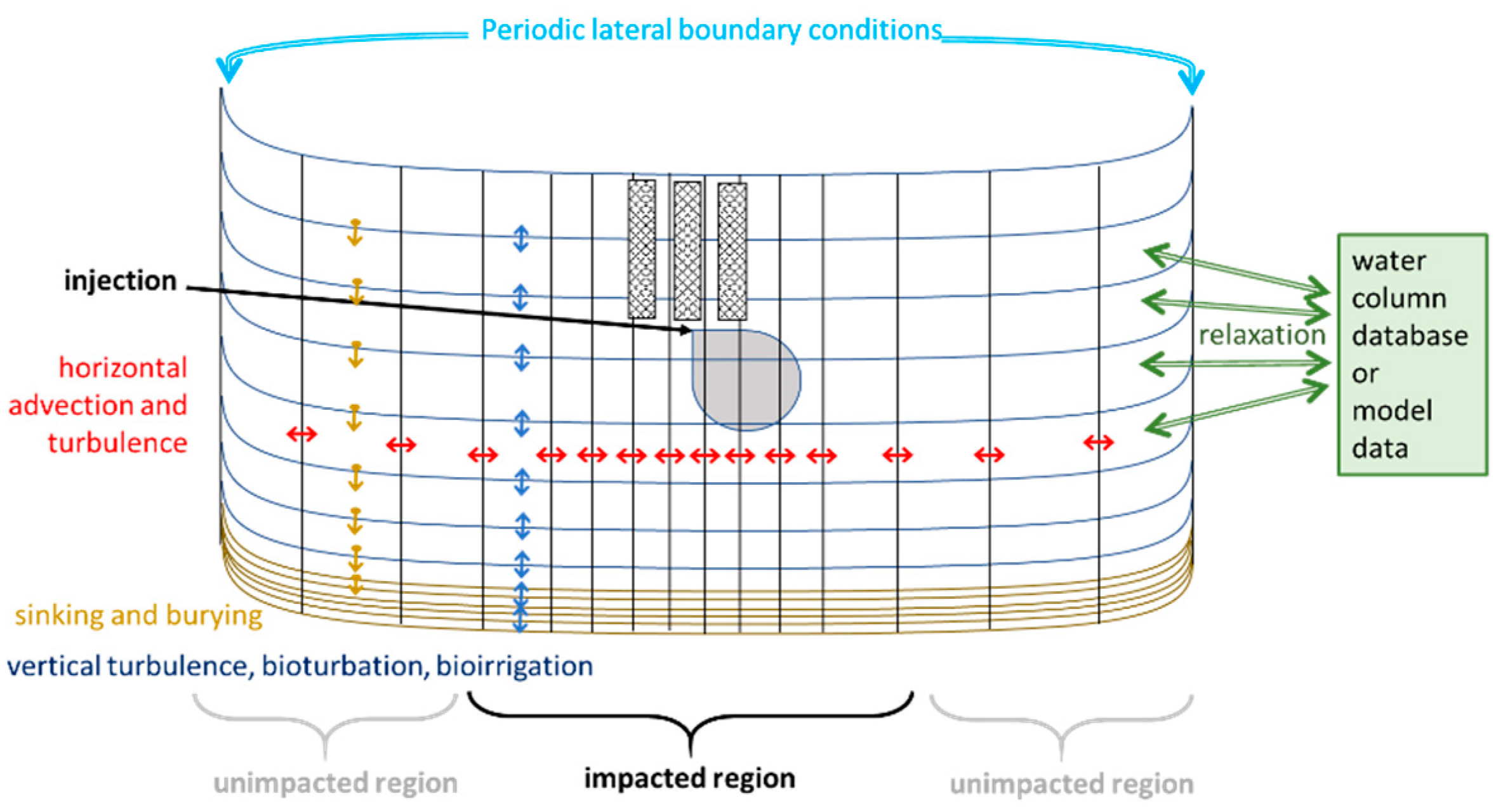
2.3. The 2DBP Transport Model
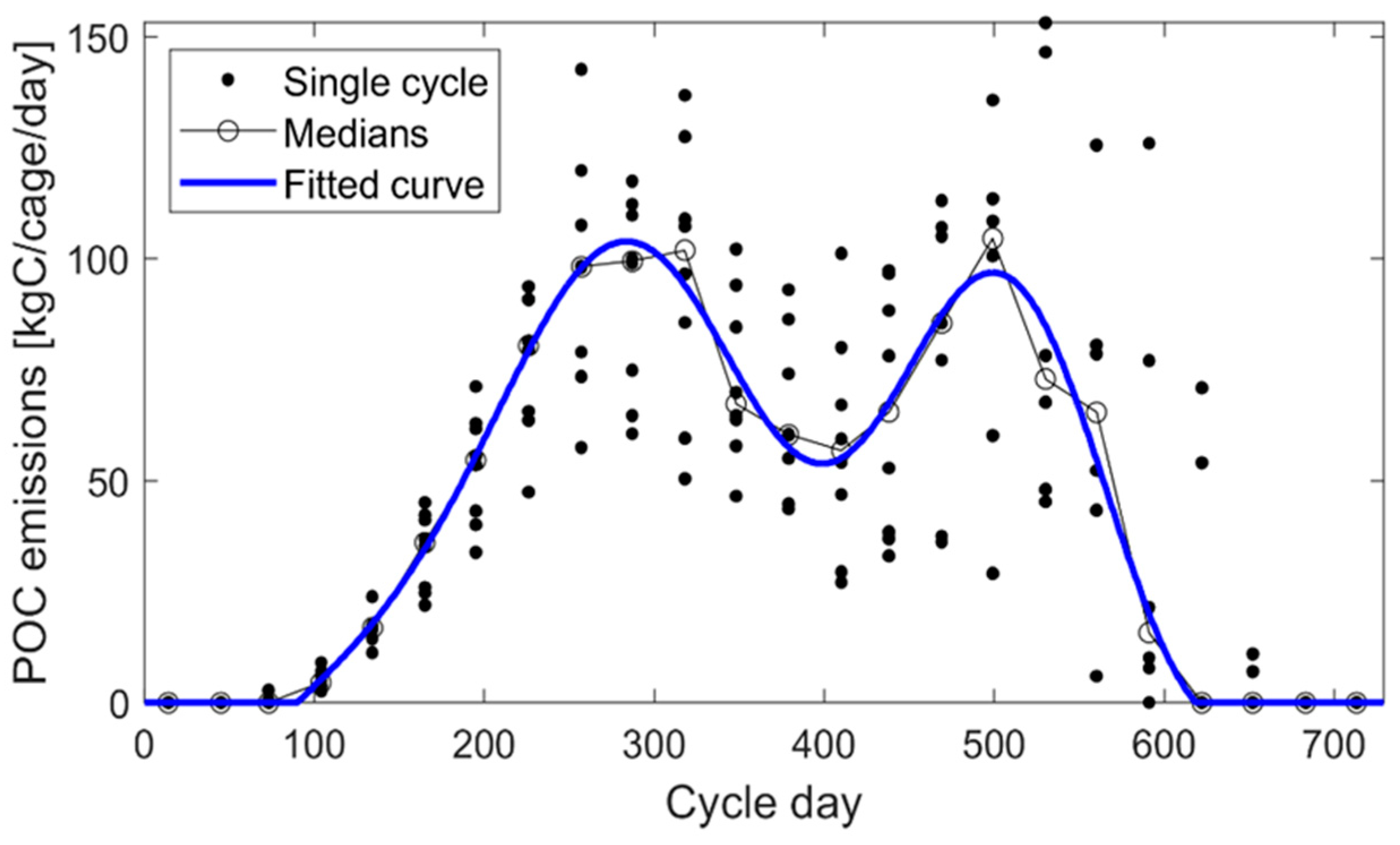
2.4. Boundary Conditions, Data for Relaxation, and Waste Deposition
2.5. Model Setup
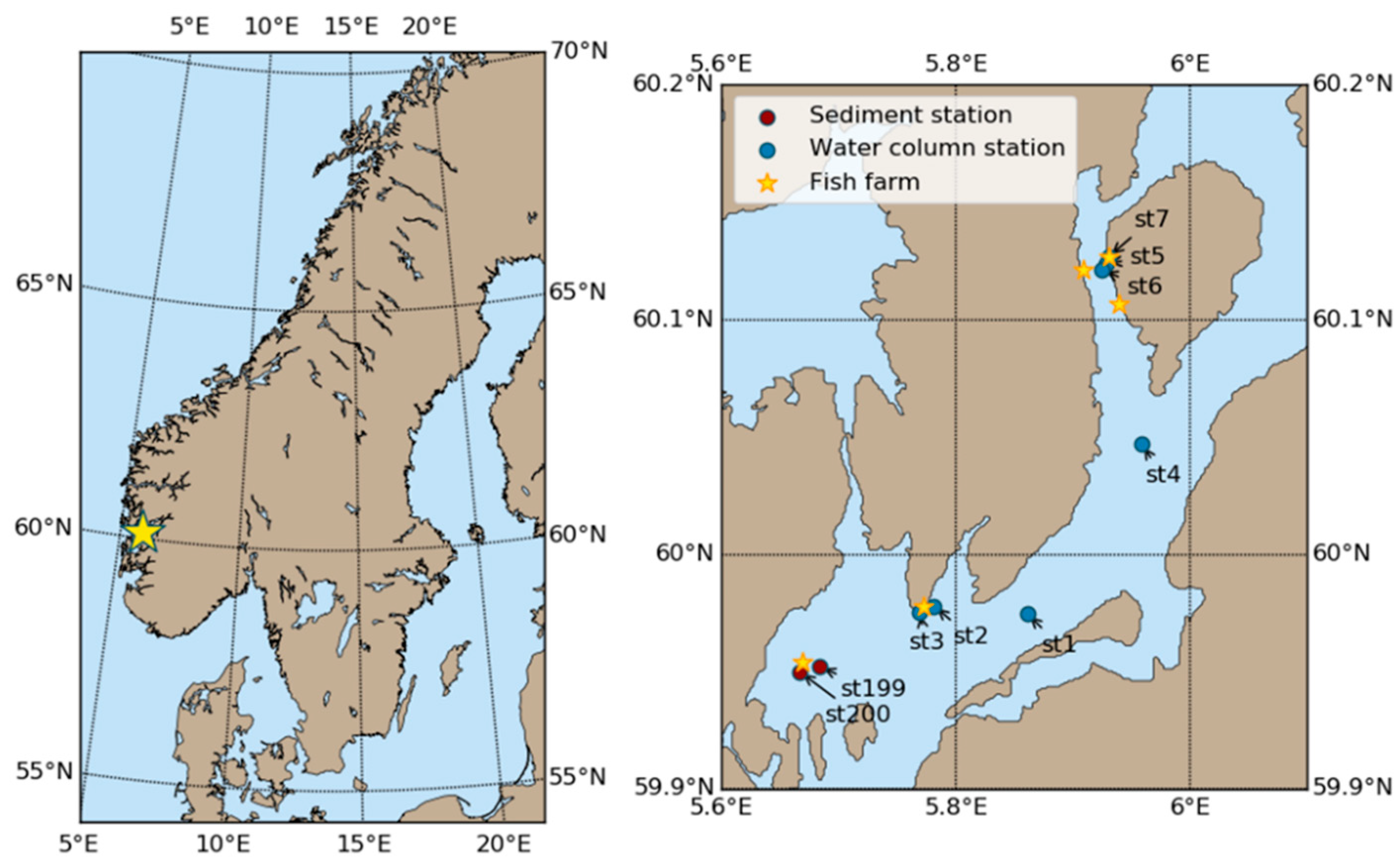
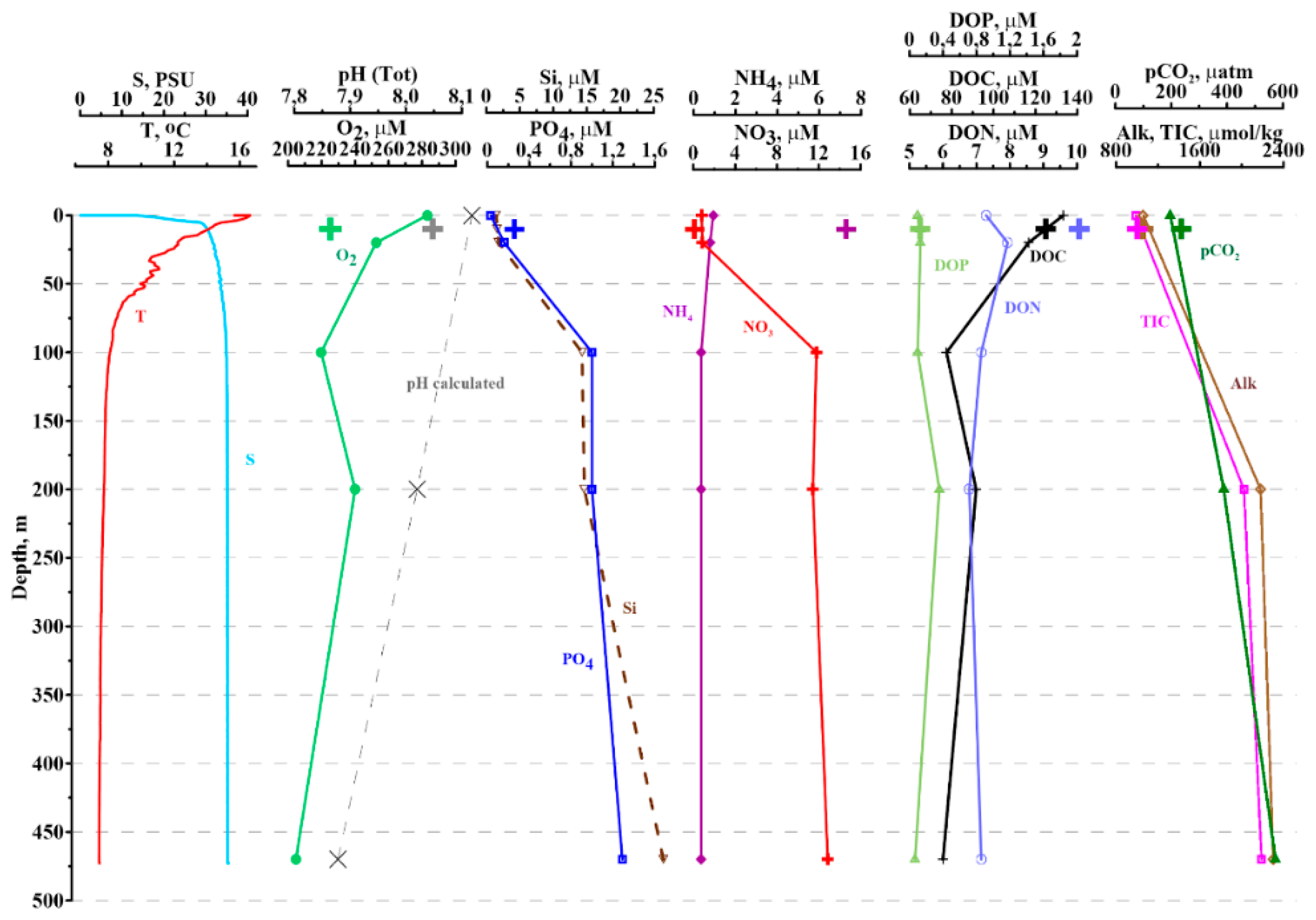
2.6. Observations from the Hardangerfjord
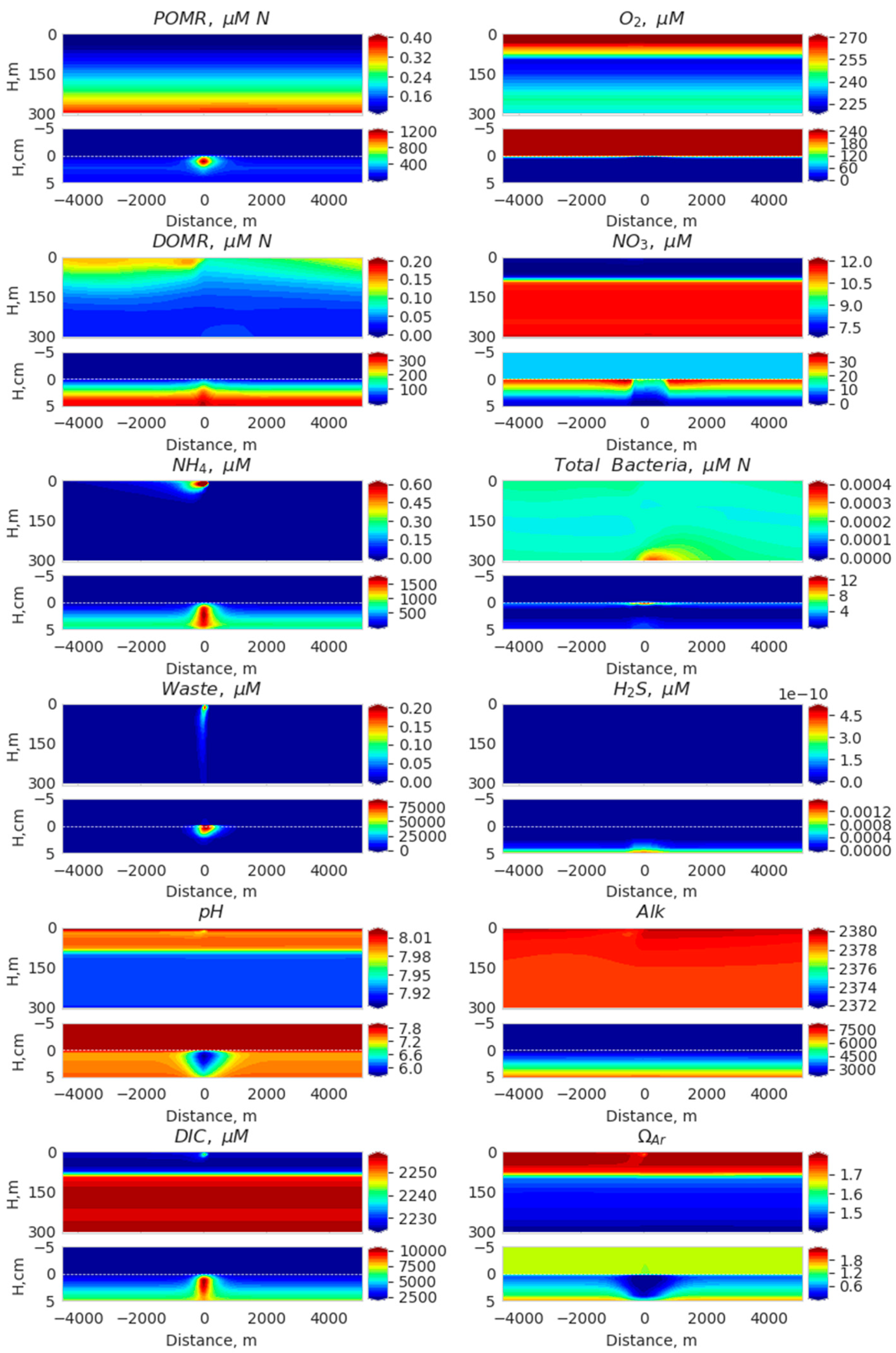
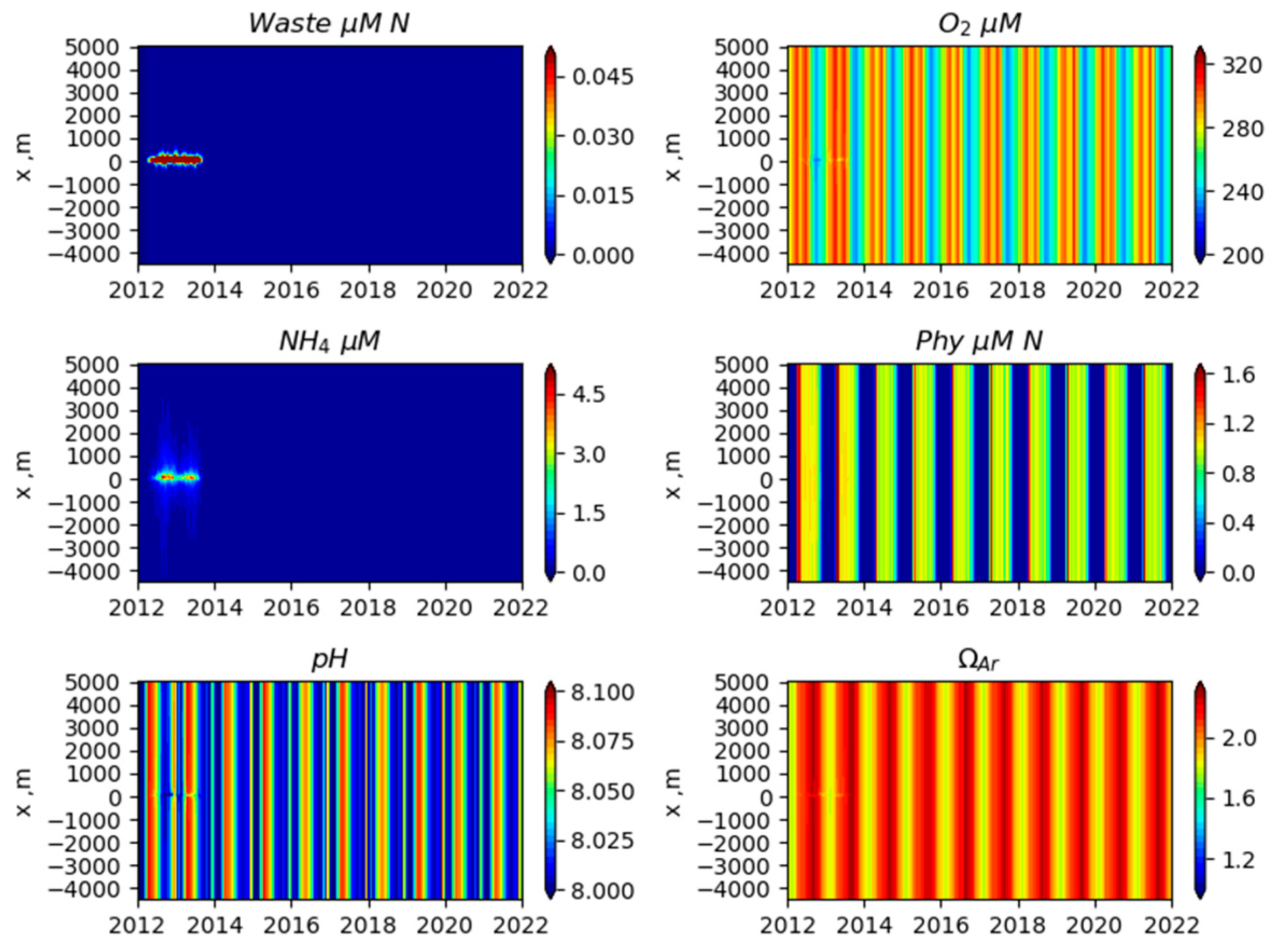
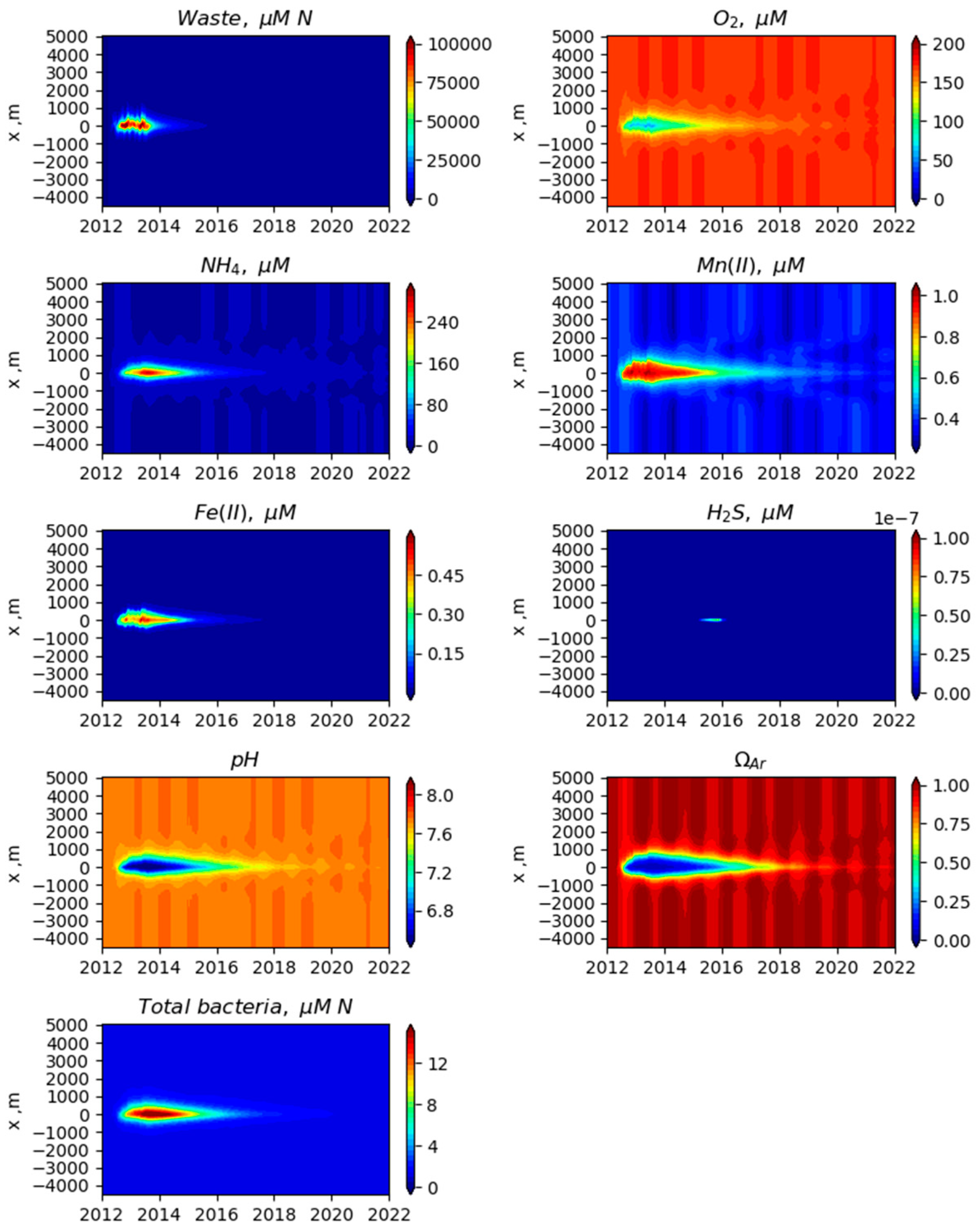
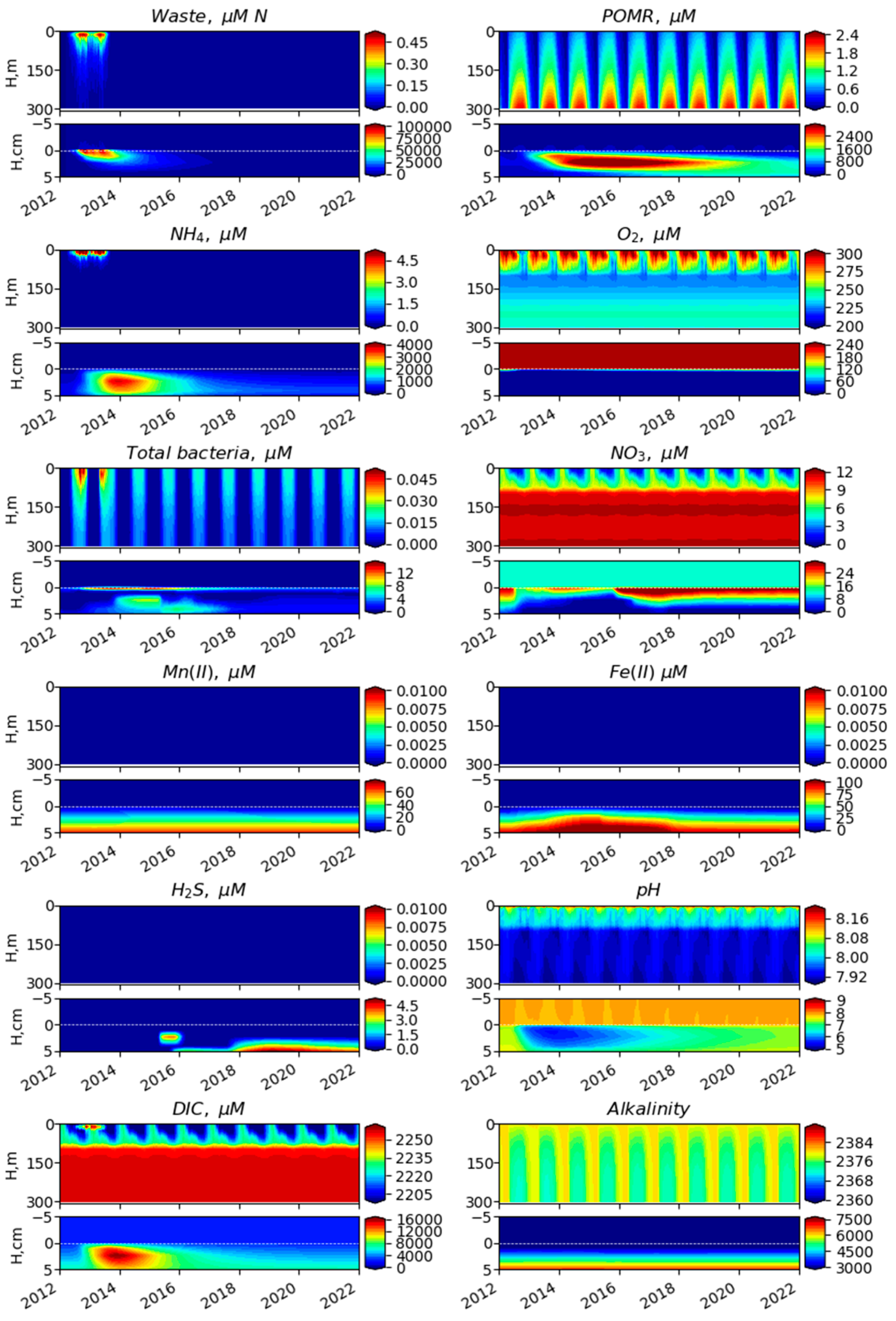
3. Results
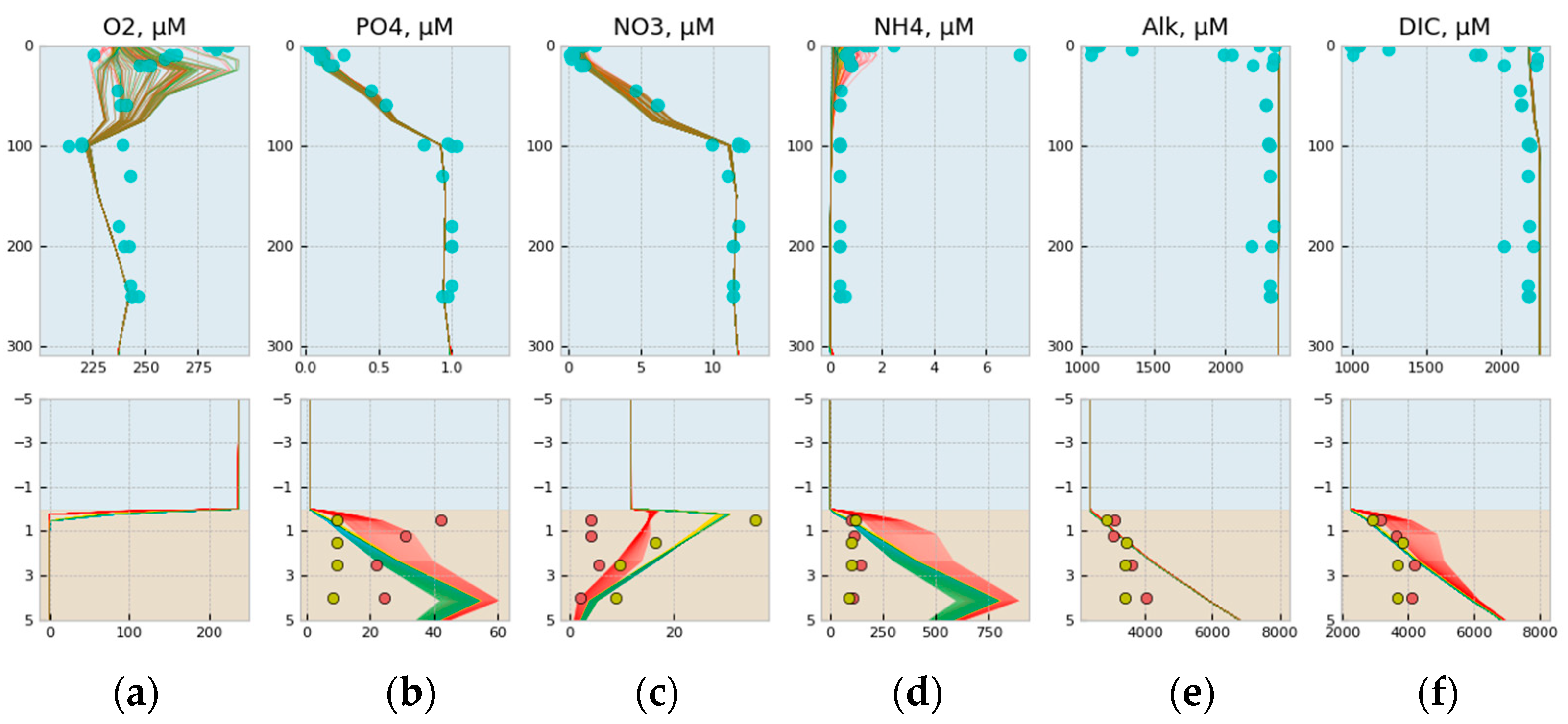
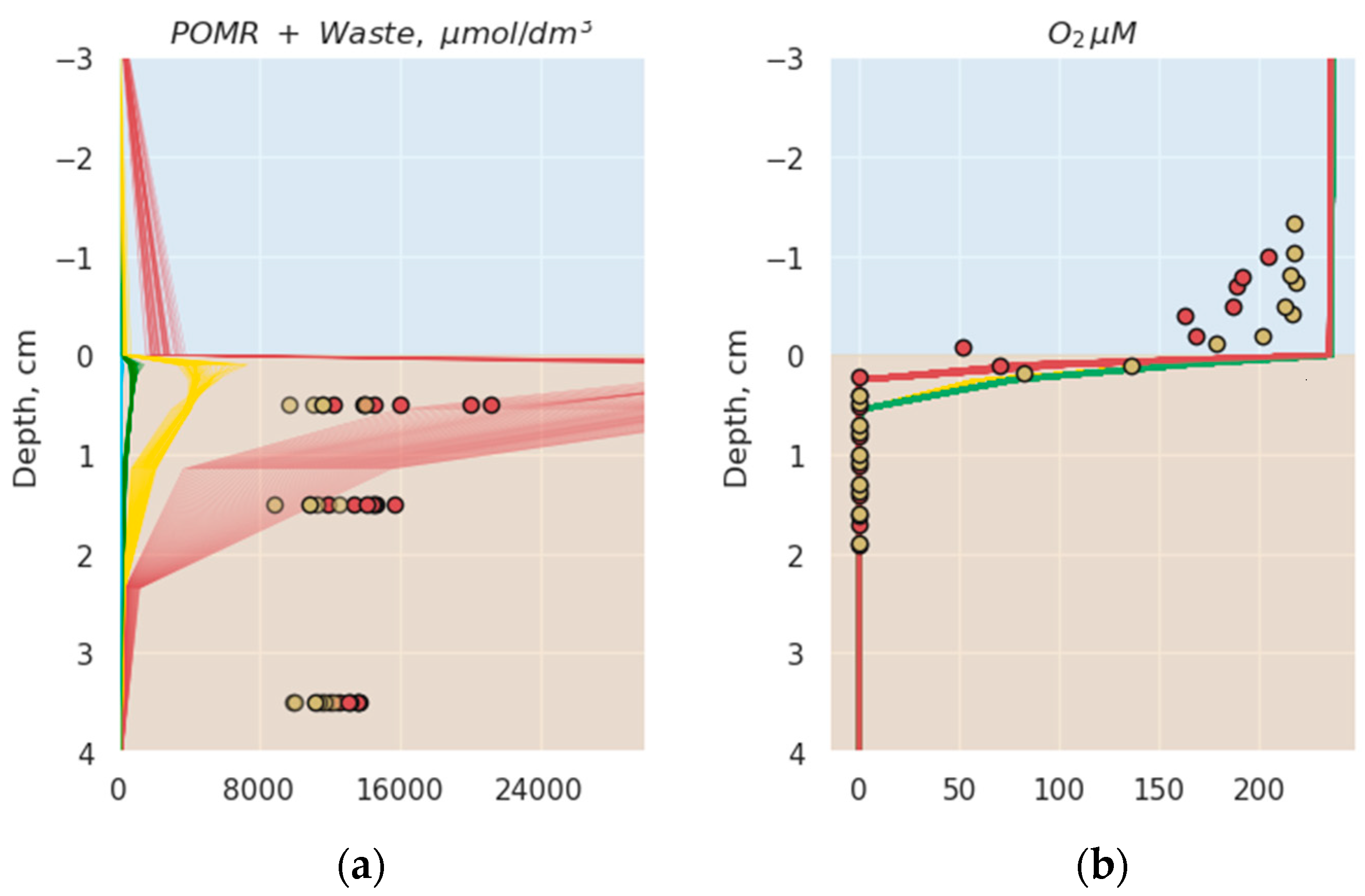
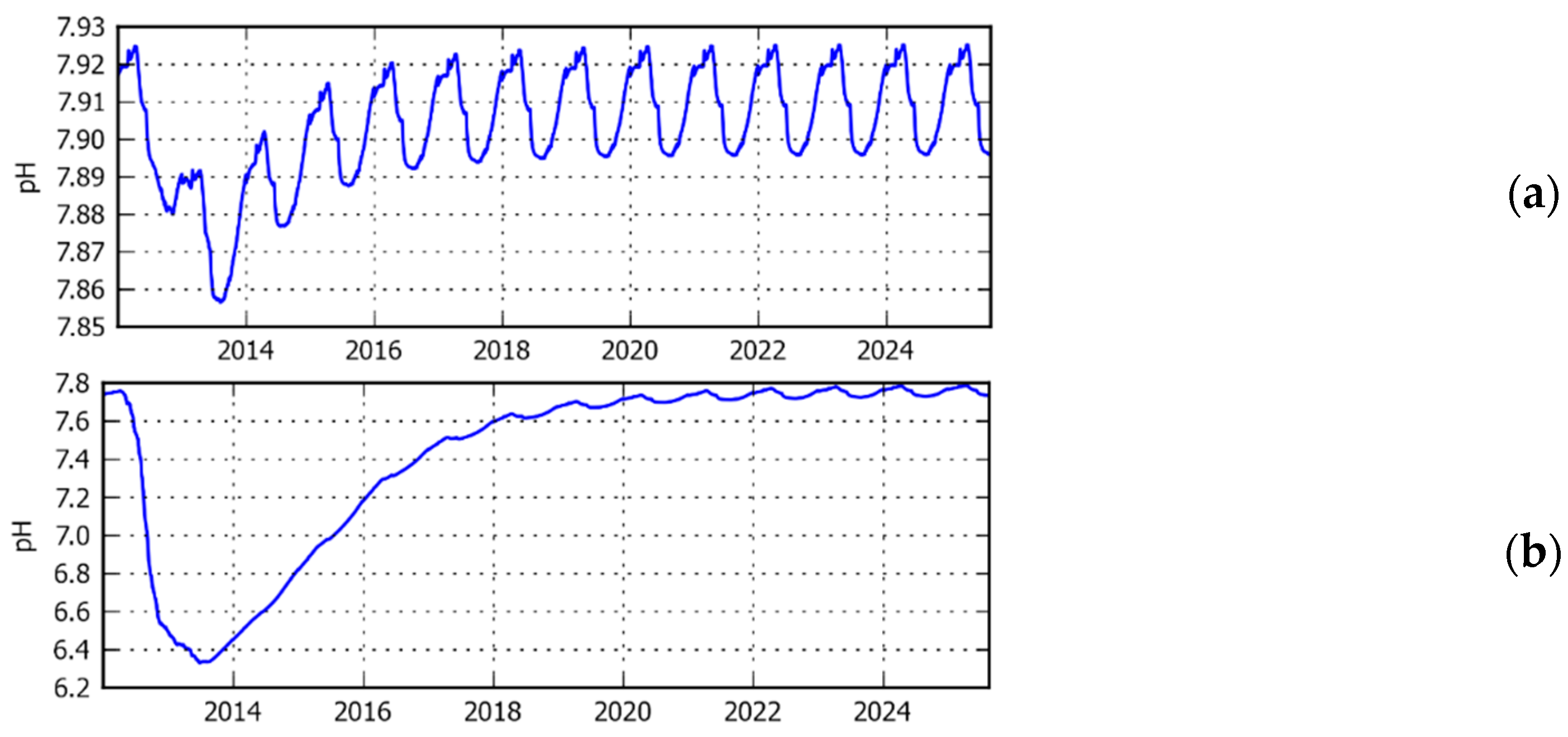
3.1. Comparison of Model Simulation with Observations
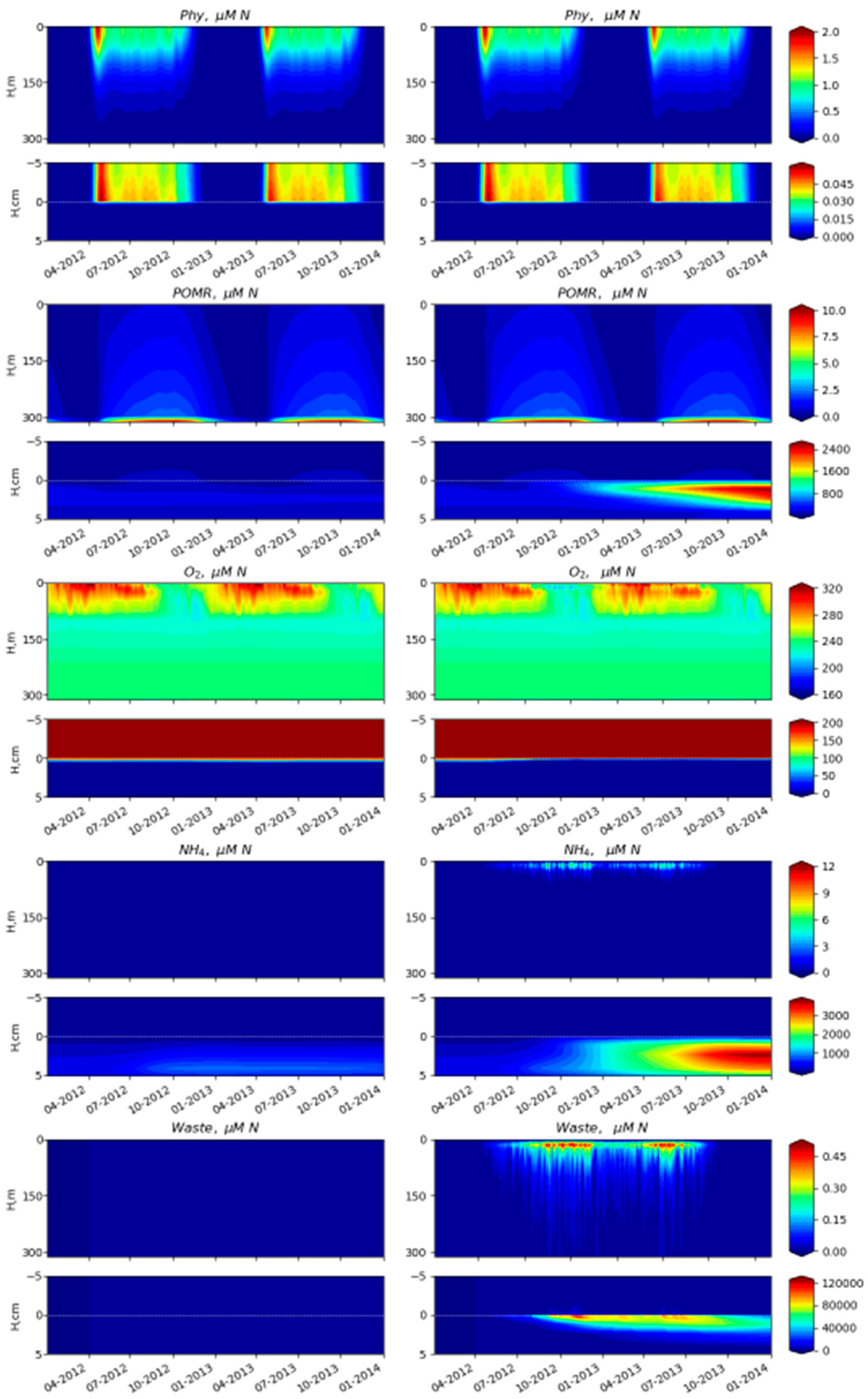
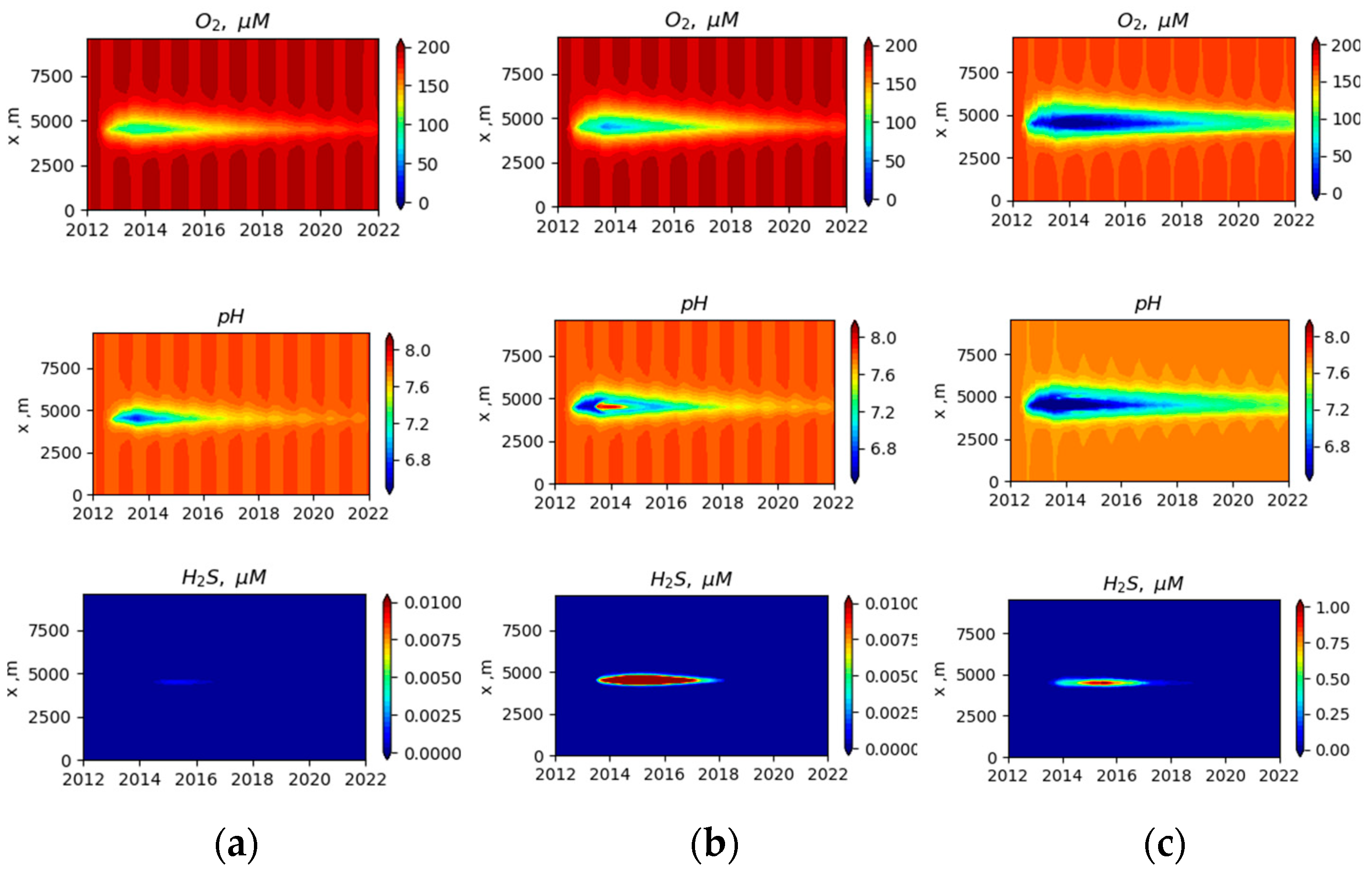
3.2. Simulated Temporal and Spatial Distributions
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bostock, J.; McAndrew, B.; Richards, R.; Jauncey, K.; Telfer, T.; Lorenzen, K.; Little, D.; Ross, L.; Handisyde, N.; Gatward, I.; et al. Aquaculture: Global status and trends. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2897–2912. [Google Scholar] [CrossRef] [PubMed]
- Bannister, R.J.; Johnsen, I.A.; Hansen, P.K.; Kutti, T.; Asplin, L. Near- and far-field dispersal modelling of organic waste from Atlantic salmon aquaculture in fjord systems. ICES J. Mar. Sci. 2016, 73, 2408–2419. [Google Scholar] [CrossRef]
- Taranger, G.L.; Karlsen, Ø.; Bannister, R.J.; Glover, K.A.; Husa, V.; Karlsbakk, E.; Kvamme, B.O.; Boxaspen, K.K.; Bjørn, P.A.; Finstad, B.; et al. Risk assessment of the environmental impact of Norwegian Atlantic salmon farming. ICES J. Mar. Sci. 2015, 72, 997–1021. [Google Scholar] [CrossRef]
- Det Kongelige Norske Videnskapers Selskab (DKNVS), Norges Tekniske Vitenskapsakademi (NTVA). Verdiskaping Basert pa Produktive hav i 2050. 2012. SINTEF Norway. Available online: https://www.sintef.no/globalassets/upload/fiskeri_og_havbruk/publikasjoner/verdiskaping-basert-pa-produktive-hav-i-2050.pdf (accessed on 25 August 2020). (In Norwegian).
- Corner, R.A.; Brooker, A.J.; Telfer, T.C.; Ross, L.G. A fully integrated GIS-based model of particulate waste distribution from marine fish-cage sites. Aquaculture 2006, 258, 299–311. [Google Scholar] [CrossRef]
- Findlay, R.H.; Watling, L.; Mayer, L.M. Environmental impact of salmon net-pen culture on marine benthic communities in Maine: A case study. Estuaries 1995, 18, 145–179. [Google Scholar] [CrossRef]
- Holmer, M.; Duarte, C.M.; Heilskov, A.; Olesen, B.; Terrados, J. Biogeochemical conditions in sediments enriched by organic matter from net-pen fish farms in the Bolinao area, Philippines. Mar. Pollut. Bull. 2003, 46, 1470–1479. [Google Scholar] [CrossRef]
- Macleod, C.K.; Crawford, C.M.; Moltschaniwskyj, N.A. Assessment of long term change in sediment condition after organic enrichment: Defining recovery. Mar. Pollut. Bull. 2004, 49, 79–88. [Google Scholar] [CrossRef]
- Valdemarsen, T.; Bannister, R.J.; Hansen, P.K.; Holmer, M.; Ervik, A. Biogeochemical malfunctioning in sediments beneath a deep-water fish farm. Environ. Pollut. 2012, 170, 15–25. [Google Scholar] [CrossRef]
- Samuelsen, O.B.; Lunestad, B.T.; Hannisdal, R.; Bannister, R.; Olsen, S.; Tjensvoll, T.; Farestveit, E.; Ervik, A. Distribution and persistence of the anti sea-lice drug teflubenzuron in wild fauna and sediments around a salmon farm, following a standard treatment. Sci. Total Environ. 2015, 508, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Sweetman, A.K.; Norling, K.; Gunderstad, C.; Haugland, B.T.; Dale, T. Benthic ecosystem functioning beneath fish farms in different hydrodynamic environments. Limnol. Oceanogr. 2014, 59, 1139–1151. [Google Scholar] [CrossRef]
- Broch, O.J.; Daae, R.L.; Ellingsen, I.H.; Nepstad, R.; Bendiksen, E.A.; Reed, J.L.; Senneset, G. Spatiotemporal dispersal and deposition of fish farm wastes: A model study from central Norway. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Stigebrandt, A. Carrying capacity: General principles of model construction. Aquac. Res. 2011, 42, 41–50. [Google Scholar] [CrossRef]
- Pérez, O.M.; Telfer, T.C.; Beveridge, M.C.M.; Ross, L.G. Geographical Information Systems (GIS) as a simple tool to aid modelling of particulate waste distribution at marine fish cage sites. Estuar. Coast. Shelf Sci. 2002, 54, 761–768. [Google Scholar] [CrossRef]
- Jusup, M.; Geček, S.; Legović, T. Impact of aquacultures on the marine ecosystem: Modelling benthic carbon loading over variable depth. Ecol. Modell. 2007, 200, 459–466. [Google Scholar] [CrossRef]
- Brigolin, D.; Meccia, V.L.; Venier, C.; Tomassetti, P.; Porrello, S.; Pastres, R. Modelling biogeochemical fluxes across a Mediterranean fish cage farm. Aquac. Environ. Interact. 2014, 5, 71–88. [Google Scholar] [CrossRef]
- Falconer, L.; Telfer, T.C.; Ross, L.G. Investigation of a novel approach for aquaculture site selection. J. Environ. Manag. 2016, 181, 791–804. [Google Scholar] [CrossRef] [PubMed]
- Bravo, F.; Grant, J. Modelling sediment assimilative capacity and organic carbon degradation efficiency at marine fish farms. Aquac. Environ. Interact. 2018, 10, 309–328. [Google Scholar] [CrossRef]
- Aure, J.; Stigebrandt, A. Quantitative estimates of the eutrophication effects of fish farming on fjords. Aquaculture 1990, 90, 135–156. [Google Scholar] [CrossRef]
- Ferreira, J.G.; Hawkins, A.J.S.; Bricker, S.B. Management of productivity, environmental effects and profitability of shellfish aquaculture—The Farm Aquaculture Resource Management (FARM) model. Aquaculture 2007, 264, 160–174. [Google Scholar] [CrossRef]
- Skogen, M.D.; Eknes, M.; Asplin, L.C.; Sandvik, A.D. Modelling the environmental effects of fish farming in a Norwegian fjord. Aquaculture 2009, 298, 70–75. [Google Scholar] [CrossRef]
- Yakushev, E.V.; Protsenko, E.A.; Bruggeman, J.; Wallhead, P.; Pakhomova, S.V.; Yakubov, S.K.; Bellerby, R.G.J.; Couture, R.-M. Bottom RedOx Model (BROM v.1.1): A coupled benthic-pelagic model for simulation of water and sediment biogeochemistry. Geosci. Model Dev. 2017, 10, 453–482. [Google Scholar] [CrossRef]
- Griffiths, J.R.; Kadin, M.; Nascimento, F.J.A.; Tamelander, T.; Törnroos, A.; Bonaglia, S.; Bonsdorff, E.; Brüchert, V.; Gårdmark, A.; Järnström, M.; et al. The importance of benthic-pelagic coupling for marine ecosystem functioning in a changing world. Glob. Chang. Biol. 2017, 23, 2179–2196. [Google Scholar] [CrossRef]
- Bruggeman, J.; Bolding, K. A general framework for aquatic biogeochemical models. Environ. Model. Softw. 2014, 61, 249–265. [Google Scholar] [CrossRef]
- Butenschön, M.; Clark, J.; Aldridge, J.N.; Allen, J.I.; Artioli, Y.; Blackford, J.; Bruggeman, J.; Cazenave, P.; Ciavatta, S.; Kay, S.; et al. ERSEM 15.06: A generic model for marine biogeochemistry and the ecosystem dynamics of the lower trophic levels. Geosci. Model Dev. Discuss. 2015, 8, 7063–7187. [Google Scholar] [CrossRef]
- Vindenes, H.; Orvik, K.A.; Søiland, H.; Wehde, H. Analysis of tidal currents in the North Sea from shipboard acoustic Doppler current profiler data. Cont. Shelf Res. 2018, 162, 1–12. [Google Scholar] [CrossRef]
- Yakushev, E.V.; Kuznetsov, I.S.; Podymov, O.I.; Burchard, H.; Neumann, T.; Pollehne, F. Modeling the influence of oxygenated inflows on the biogeochemical structure of the Gotland Sea, central Baltic Sea: Changes in the distribution of manganese. Comput. Geosci. 2011, 37, 398–409. [Google Scholar] [CrossRef]
- Shchepetkin, A.F.; McWilliams, J.C. The regional oceanic modeling system (ROMS): A split-explicit, free-surface, topography-following-coordinate oceanic model. Ocean Model. 2005, 9, 347–404. [Google Scholar] [CrossRef]
- Okubo, A. Remarks on the use of “diffusion diagrams” in modeling scale-dependent diffusion. Deep. Res. Oceanogr. Abstr. 1976, 23, 1213–1214. [Google Scholar] [CrossRef]
- Hansen, P.K.; Pittman, K.; Ervik, A. Organic waste from marine fish farms—Effects on the seabed. Mar. Aquac. Environ. 1991, 22, 105–119. [Google Scholar]
- Hall, P.; Anderson, L.; Holby, O.; Kollberg, S.; Samuelsson, M.-O. Chemical fluxes and mass balances in a marine fish cage farm. I Carbon. Mar. Ecol. Prog. Ser. 1990, 61, 61–73. [Google Scholar] [CrossRef]
- Wang, X.; Olsen, L.M.; Reitan, K.I.; Olsen, Y. Discharge of nutrient wastes from salmon farms: Environmental effects, and potential for integrated multi-trophic aquaculture. Aquac. Environ. Interact. 2012, 2, 267–283. [Google Scholar] [CrossRef]
- Grasshoff, K.; Kremling, K.; Ehrhardt, M. Methods of Seawater Analysis; Grasshoff, K., Kremling, K., Ehrhardt, M., Eds.; Wiley: Weinheim, Germany, 1999; ISBN 9783527295890. [Google Scholar]
- Mintrop, L.; Pérez, F.F.; González-Dávila, M.; Santana-Casiano, J.M.; Körtzinger, A. Alkalinity determination by potentiometry: Intercalibration using three different methods. Ciencias Mar. 2000, 26, 23–37. [Google Scholar] [CrossRef]
- Van Heuven, S.; Pierrot, D.; Rae, J.W.B.; Lewis, E.; Wallace, D.W.R. MATLAB Program Developed for CO2 System Calculations. ORNL/CDIAC-105b. Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, US Department of Energy, Oak Ridge, Tennessee. 2011; p. 530. Available online: https://cdiac.ess-dive.lbl.gov/ftp/co2sys/CO2SYS_calc_MATLAB_v1.1/?C=N;O=A (accessed on 25 August 2020).
- Lewis, E.; Wallace, D. Program Developed for CO2 System Calculations; Carbon Dioxide Information Analysis Center, CDIAC-105, Lockheed Martin Energy Research Corporation for the US Department of Energy: Oak Ridge, Tennessee. 1998; pp. 1–21. Available online: https://www.osti.gov/dataexplorer/biblio/dataset/1464255 (accessed on 25 August 2020).
- Hargrave, B. Empirical relationships describing benthic impacts of salmon aquaculture. Aquac. Environ. Interact. 2010, 1, 33–46. [Google Scholar] [CrossRef]
- Wilkie, M.P. Mechanisms of ammonia excretion across fish gills. Comp. Biochem. Physiol. Part A Physiol. 1997, 118, 39–50. [Google Scholar] [CrossRef]
- Piedecausa, M.A.; Aguado-Giménez, F.; García-García, B.; Telfer, T.C. Total ammonia nitrogen leaching from feed pellets used in salmon aquaculture. J. Appl. Ichthyol. 2010, 26, 16–20. [Google Scholar] [CrossRef]
- Dong, X.; Qin, J.; Zhang, X. Fish adaptation to oxygen variations in aquaculture from hypoxia to hyperoxia. J. Fish. Aquac. 2011, 2, 23–28. [Google Scholar]
- Abo, K.; Yokoyama, H. Assimilative capacity of fish farm environments as determined by the benthic oxygen uptake rate: Studies using a numerical model. Bull. Fish. Res. Agen. 2007, 19, 79–87. [Google Scholar]
- Keeley, N.B.; Forrest, B.M.; Macleod, C.K. Benthic recovery and re-impact responses from salmon farm enrichment: Implications for farm management. Aquaculture 2015, 435, 412–423. [Google Scholar] [CrossRef]
- Wheatcroft, R.A.; Jumars, P.A.; Smith, C.R.; Nowell, A.R.M. A mechanistic view of the particulate biodiffusion coefficient: Step lengths, rest periods and transport directions. J. Mar. Res. 1990, 48, 177–207. [Google Scholar] [CrossRef]
- Keeley, N.B.; Macleod, C.K.; Taylor, D.; Forrest, R. Comparison of three potential methods for accelerating seabed recovery beneath salmon farms. Aquaculture 2017, 479, 652–666. [Google Scholar] [CrossRef]
- Keeley, N.; Valdemarsen, T.; Woodcock, S.; Holmer, M.; Husa, V.; Bannister, R. Resilience of dynamic coastal benthic ecosystems in response to large-scale finfish farming. Aquac. Environ. Interact. 2019, 11, 161–179. [Google Scholar] [CrossRef]
- Keeley, N.; Valdemarsen, T.; Strohmeier, T.; Pochon, X.; Dahlgren, T.; Bannister, R. Mixed-habitat assimilation of organic waste in coastal environments—It’s all about synergy! Sci. Total Environ. 2020, 699, 134281. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yakushev, E.V.; Wallhead, P.; Renaud, P.E.; Ilinskaya, A.; Protsenko, E.; Yakubov, S.; Pakhomova, S.; Sweetman, A.K.; Dunlop, K.; Berezina, A.; et al. Understanding the Biogeochemical Impacts of Fish Farms Using a Benthic-Pelagic Model. Water 2020, 12, 2384. https://doi.org/10.3390/w12092384
Yakushev EV, Wallhead P, Renaud PE, Ilinskaya A, Protsenko E, Yakubov S, Pakhomova S, Sweetman AK, Dunlop K, Berezina A, et al. Understanding the Biogeochemical Impacts of Fish Farms Using a Benthic-Pelagic Model. Water. 2020; 12(9):2384. https://doi.org/10.3390/w12092384
Chicago/Turabian StyleYakushev, Evgeniy V., Philip Wallhead, Paul E. Renaud, Alisa Ilinskaya, Elizaveta Protsenko, Shamil Yakubov, Svetlana Pakhomova, Andrew K. Sweetman, Kathy Dunlop, Anfisa Berezina, and et al. 2020. "Understanding the Biogeochemical Impacts of Fish Farms Using a Benthic-Pelagic Model" Water 12, no. 9: 2384. https://doi.org/10.3390/w12092384
APA StyleYakushev, E. V., Wallhead, P., Renaud, P. E., Ilinskaya, A., Protsenko, E., Yakubov, S., Pakhomova, S., Sweetman, A. K., Dunlop, K., Berezina, A., Bellerby, R. G. J., & Dale, T. (2020). Understanding the Biogeochemical Impacts of Fish Farms Using a Benthic-Pelagic Model. Water, 12(9), 2384. https://doi.org/10.3390/w12092384





