Can the Stream Quantification Tool (SQT) Protocol Predict the Biotic Condition of Streams in the Southeast Piedmont (USA)?
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Selection
2.2. Data Collection
2.2.1. Hydrology
2.2.2. Hydraulics
2.2.3. Geomorphology
2.2.4. Physicochemistry
2.2.5. Biology
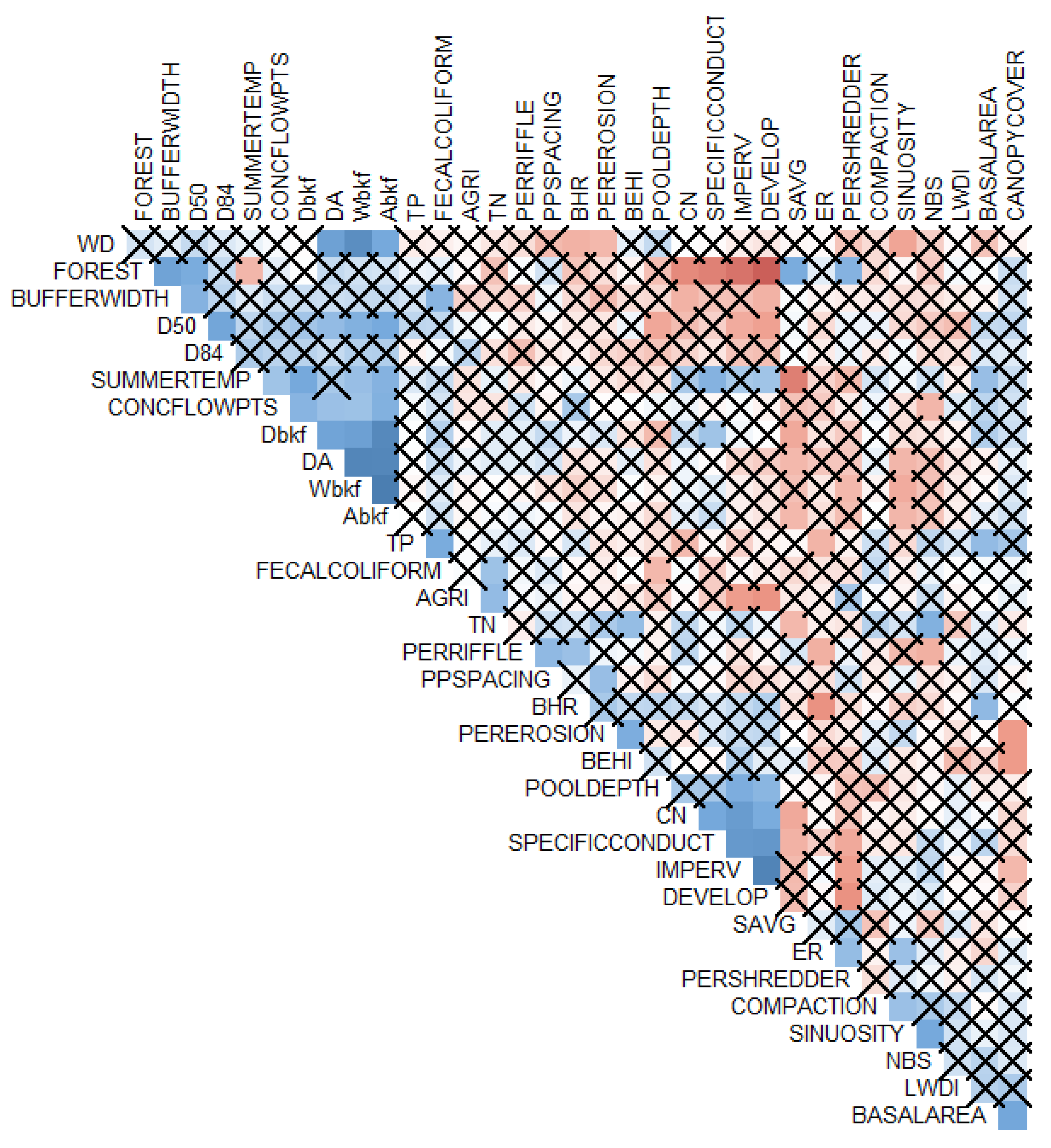
2.3. Statistical Analyses
2.3.1. Response Variable Selection
2.3.2. Statistical Approach Selection
2.3.3. Model Implementation
2.3.4. Model Selection
2.3.5. Model Performance
3. Results
3.1. Stream Functional Data for NC Piedmont Ecoregion
3.2. Model Implementation
3.2.1. SQT Models
3.2.2. SQT+ Models
3.3. Model Selection
3.4. Model Performance
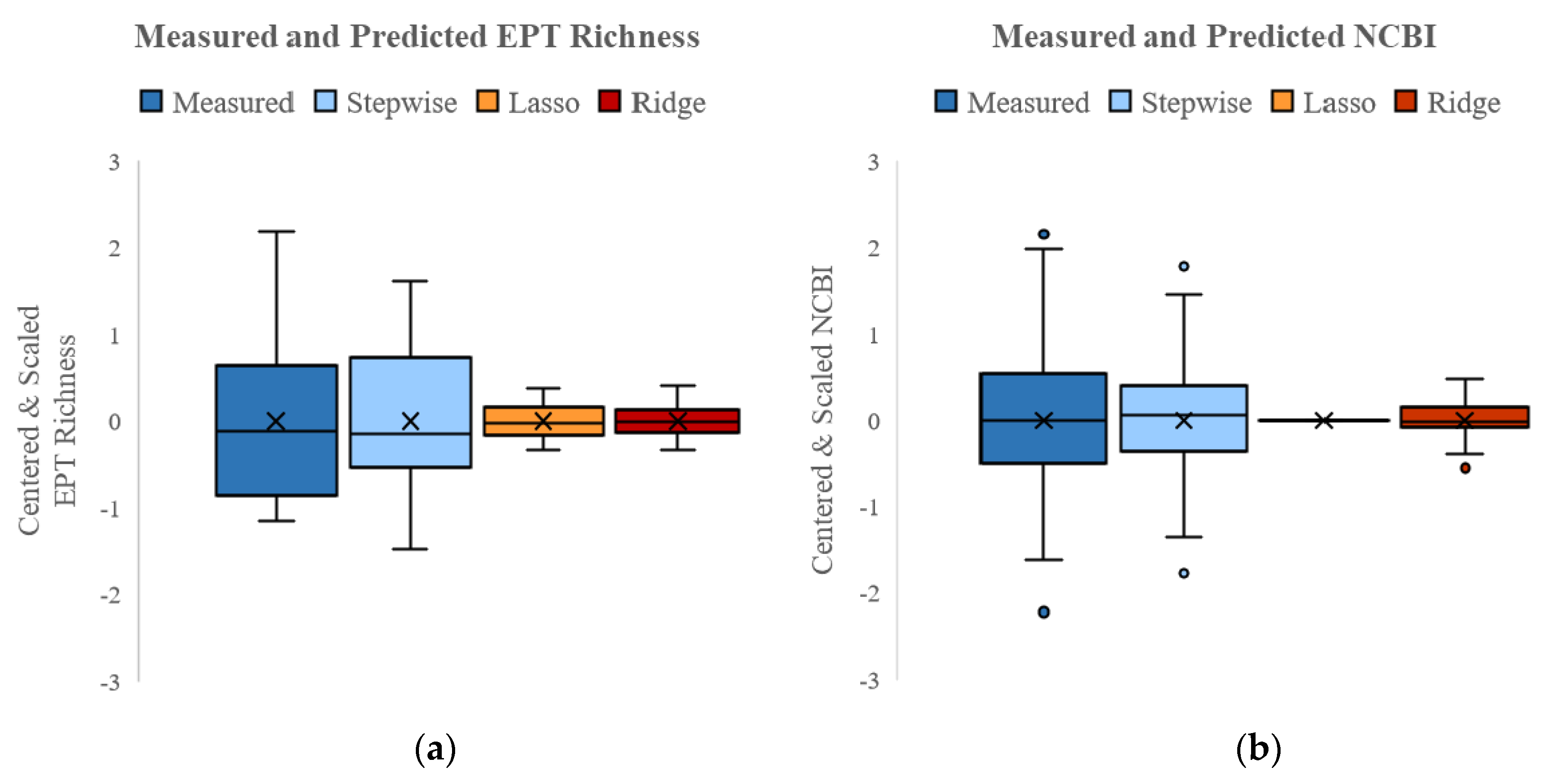
3.4.1. SQT Models
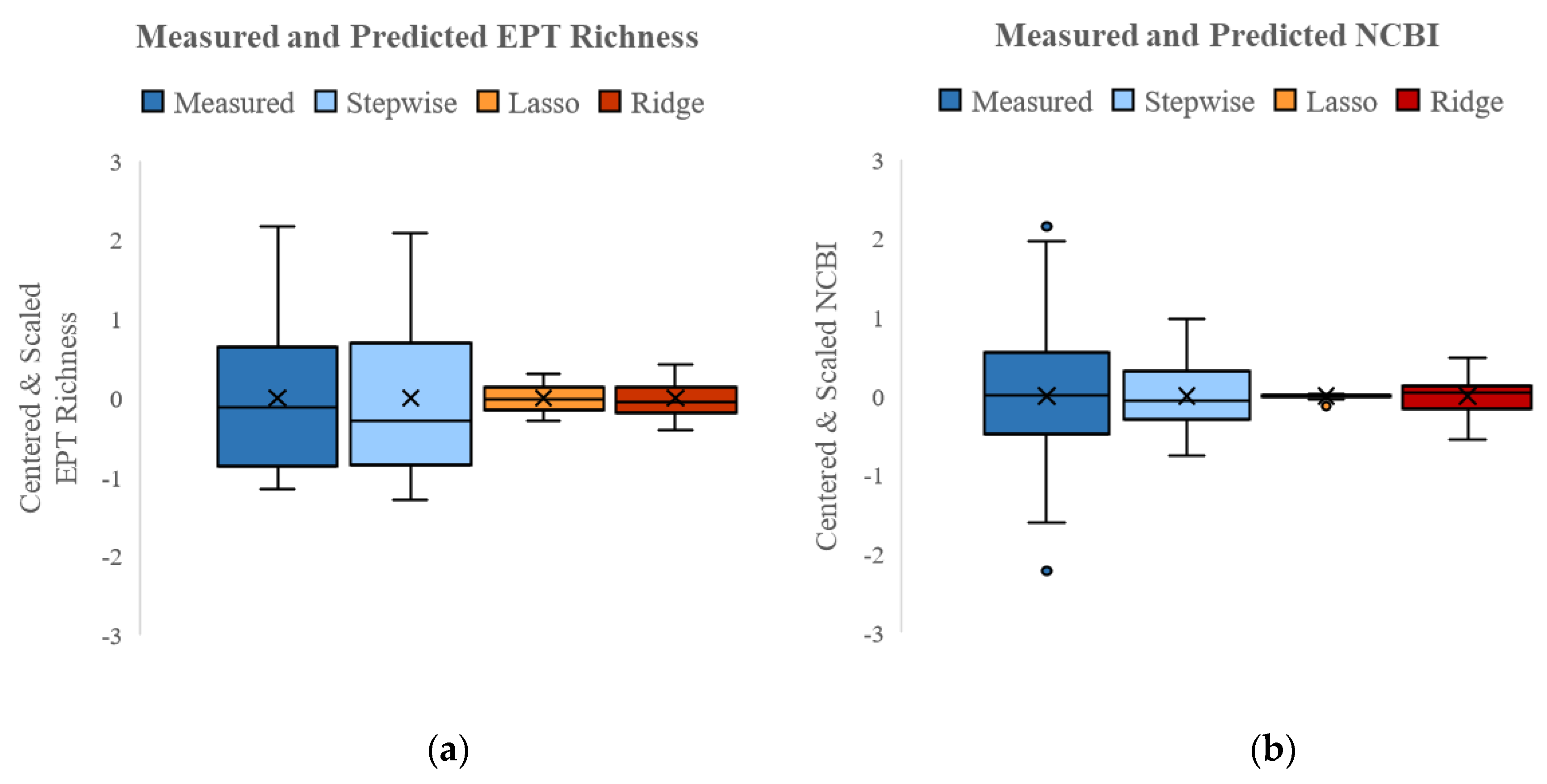
3.4.2. SQT+ Models
4. Discussion
4.1. SQT Protocol Biotic Prediction and Pyramid Framework Premise
4.2. Variables Important to Macroinvertebrates
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Kollmann, J.; Meyer, S.T.; Bateman, R.; Conradi, T.; Gossner, M.M.; de Souza Mendonça, M., Jr.; Fernandes, G.W.; Hermann, J.M.; Koch, C.; Müller, S.C.; et al. Integrating ecosystem functions into restoration ecology-recent advances and future directions. Restor. Ecol. 2016, 24, 722–730. [Google Scholar] [CrossRef]
- Palmer, M.A.; Hondula, K.L.; Koch, B.J. Ecological Restoration of Streams and Rivers: Shifting Strategies and Shifting Goals. Annu. Rev. Ecol. Evol. Syst. 2014, 45, 247–269. [Google Scholar] [CrossRef]
- Wohl, E.; Angermeier, P.L.; Bledsoe, B.; Kondolf, G.M.; MacDonnell, L.; Merritt, D.M.; Palmer, M.A.; Poff, N.L.; Tarboton, D. River restoration. Water Resour. Res. 2005, 41. [Google Scholar] [CrossRef]
- Wohl, E.; Lane, S.N.; Wilcox, A.C. The science and practice of river restoration. Water Resour. Res. 2015, 51, 5974–5997. [Google Scholar] [CrossRef]
- Jax, K. Function and “functioning” in ecology: What does it mean? Oikos 2005, 111, 641–648. [Google Scholar] [CrossRef]
- Vinson, M.R.; Hawkins, C.P. Biodiversity of Stream Insects: Variation at Local, Basin, and Regional Scales. Annu. Rev. Entomol. 1998, 43, 271–293. [Google Scholar] [CrossRef] [PubMed]
- Schueler, T.R. The Importance of Imperviousness. Watershed Prot. Tech. 1995, 1, 100–111. [Google Scholar]
- Booth, D. Urbanization and the Natural Drainage System—Impacts, Solutions, and Prognoses. Northwest Environ. J. 1991, 7, 93–118. [Google Scholar]
- Jones, R.C.; Clark, C.C. Impact of Watershed Urbanization on Stream Insect Communities. JAWRA J. Am. Water Resour. Assoc. 1987, 23, 1047–1055. [Google Scholar] [CrossRef]
- Klein, R.D. Urbanization and Stream Quality Impairment. JAWRA J. Am. Water Resour. Assoc. 1979, 15, 948–963. [Google Scholar] [CrossRef]
- Limburg, K.E.; Schmidt, R.E. Patterns of Fish Spawning in Hudson River Tributaries: Response to an Urban Gradient? Ecology 1990, 71, 1238–1245. [Google Scholar] [CrossRef]
- Steedman, R.J. Modification and Assessment of an Index of Biotic Integrity to Quantify Stream Quality in Southern Ontario. Can. J. Fish. Aquat. Sci. 1988, 45, 492–501. [Google Scholar] [CrossRef]
- Allan, J.D.; Castillo, M.M. Stream Ecology—Structure and Function of Running Waters, 2nd ed.; Springer: Dordrecht, The Netherlands, 2007; ISBN 978-1-4020-5582-9. [Google Scholar]
- Sweeney, B.W. Effects of Streamside Vegetation on Macroinvertebrate Communities of White Clay Creek in Eastern North America. Proc. Acad. Nat. Sci. Phila. 1993, 144, 291–340. [Google Scholar]
- Sweeney, B.W.; Bott, T.L.; Jackson, J.K.; Kaplan, L.A.; Newbold, J.D.; Standley, L.J.; Hession, W.C.; Horwitz, R.J. Riparian deforestation, stream narrowing, and loss of stream ecosystem services. Proc. Natl. Acad. Sci. USA 2004, 101, 14132–14137. [Google Scholar] [CrossRef] [PubMed]
- Richards, C.; Johnson, L.B.; Host, G.E. Landscape-scale influences on stream habitats and biota. Can. J. Fish. Aquat. Sci. 1996, 53, 17. [Google Scholar] [CrossRef]
- Walters, D.M.; Roy, A.H.; Leigh, D.S. Environmental indicators of macroinvertebrate and fish assemblage integrity in urbanizing watersheds. Ecol. Indic. 2009, 9, 1222–1233. [Google Scholar] [CrossRef]
- Richards, C.; Haro, R.; Johnson, L.; Host, G. Catchment and reach-scale properties as indicators of macroinvertebrate species traits. Freshw. Biol. 1997, 37, 219–230. [Google Scholar] [CrossRef]
- Sponseller, R.A.; Benfield, E.F.; Valett, H.M. Relationships between land use, spatial scale and stream macroinvertebrate communities. Freshw. Biol. 2001, 46, 1409–1424. [Google Scholar] [CrossRef]
- Palmer, M.A.; Filoso, S.; Fanelli, R.M. From ecosystems to ecosystem services: Stream restoration as ecological engineering. Ecol. Eng. 2014, 65, 62–70. [Google Scholar] [CrossRef]
- Bunn, S.E.; Abal, E.G.; Smith, M.J.; Choy, S.C.; Fellows, C.S.; Harch, B.D.; Kennard, M.J.; Sheldon, F. Integration of science and monitoring of river ecosystem health to guide investments in catchment protection and rehabilitation. Freshw. Biol. 2010, 55, 223–240. [Google Scholar] [CrossRef]
- Doll, B.A.; Jennings, G.D.; Spooner, J.; Penrose, D.L.; Usset, J.L. Evaluating the eco-geomorphological condition of restored streams using visual assessment and macroinvertebrate metrics. J. Am. Water Resour. Assoc. 2015, 51, 68–83. [Google Scholar] [CrossRef]
- Somerville, D.E. Stream Assessment and Mitigation Protocols: A Review of Commonalities and Differences; Contract No. GS-00F0032M, Document No. EPA 843-S-12-003; U.S. Environmental Protection Agency, Office of Wetlands, Oceans, and Watersheds: Washington, DC, USA, 2010.
- Doll, B.; Jennings, G.; Spooner, J.; Penrose, D.; Usset, J.; Blackwell, J.; Fernandez, M. Identifying watershed, landscape, and engineering design factors that influence the biotic condition of restored streams. Water 2016, 8, 151. [Google Scholar] [CrossRef]
- D’Ambrosio, J.L.; Williams, L.R.; Williams, M.G.; Witter, J.D.; Ward, A.D. Geomorphology, habitat, and spatial location influences on fish and macroinvertebrate communities in modified channels of an agriculturally-dominated watershed in Ohio, USA. Ecol. Eng. 2014, 68, 32–46. [Google Scholar] [CrossRef]
- D’Ambrosio, J.L.; Williams, L.R.; Witter, J.D.; Ward, A. Effects of geomorphology, habitat, and spatial location on fish assemblages in a watershed in Ohio, USA. Environ. Monit. Assess. 2009, 148, 325–341. [Google Scholar] [CrossRef]
- Helms, B.; Zink, J.; Werneke, D.; Hess, T.; Price, Z.; Jennings, G.; Brantley, E. Development of Ecogeomorphological (EGM) Stream Design and Assessment Tools for the Piedmont of Alabama, USA. Water 2016, 8, 161. [Google Scholar] [CrossRef]
- Fischenich, C. Functional Objectives for Stream Restoration; EMRRP Technical Notes Collection (ERDC TN-EMRRP-SR-52); U.S. Army Engineer Research and Development Center: Vicksburg, MS, USA, 2006.
- Doll, B.; Jennings, G.; Spooner, J.; Penrose, D.; Usset, J.; Blackwell, J.; Fernandez, M. Can rapid assessments predict the biotic condition of restored streams? Water 2016, 8, 143. [Google Scholar] [CrossRef]
- US Army Corps of Engineers; Environmental Protection Agency. Compensatory Mitigation for Losses of Aquatic Resources; Final Rule. In Fed. Regist.; 2008; Volume 73, pp. 19593–19705. [Google Scholar]
- Harman, W.R.; Starr, R.; Carter, M.; Tweedy, K.; Clemmons, M.; Suggs, K.; Miller, C. A Function-Based Framework for Stream Assessment and Restoration Projects; EPA 843-K12-006; US Environmental Protection Agency, Office of Wetlands, Oceans, and Watersheds: Washington, DC, USA, 2012.
- Harman, W.; Jones, C. North Carolina Stream Quantification Tool: Data Collection and Analysis Manual, NC SQT v3.0; Environmental Defense Fund: Raleigh, NC, USA, 2017. [Google Scholar]
- Minnesota Stream Quantification Tool Steering Committee (MNSQT SC). Minnesota Stream Quantification Tool and Debit Calculator (MNSQT) User Manual, Version 1.0; Contract #PC-17-001; U.S. Environmental Protection Agency, Office of Wetlands, Oceans and Watersheds: Washington, DC, USA, 2019.
- TDEC. Tennessee Stream Quantification Tool Spreadsheet User Manual TN SQT v1.0; Tennessee Department of Environment and Conservation: Nashville, TN, USA, 2017.
- US Army Corps of Engineers. Wyoming Stream Quantification Tool (WSQT) User Manual and Spreadsheet; Version 1.0; Omaha District, Wyoming Regulatory Office: Cheyenne, WY, USA, 2018.
- Public Notice: Savannah District’s 2018 Standard Operating Procedure for Compensatory Mitigation; US Army Corps of Engineers Savannah District: Savannah, GA, USA, 2018.
- Harman, W. Personal Communication, 2020.
- Strahler, A.N. Dynamic Basis of Geomorphology. Bull. Geol. Soc. Am. 1952, 63, 923–938. [Google Scholar] [CrossRef]
- Leopold, L.B. A View of the River; Harvard University Press: Cambridge, MA, USA, 1994. [Google Scholar]
- US Department of Agriculture Natural Resources Conservation Service. Soil Survey Geographic (SSURGO) Database for North Carolina and Virginia. Available online: https://websoilsurvey.sc.egov.usda.gov/App/HomePage.htm (accessed on 17 May 2019).
- Yang, L.; Jin, S.; Danielson, P.; Homer, C.; Gass, L.; Bender, S.M.; Case, A.; Costello, C.; Dewitz, J.; Fry, J.; et al. A new generation of the United States National Land Cover Database: Requirements, research priorities, design, and implementation strategies. ISPRS J. Photogramm. Remote Sens. 2018, 146, 108–123. [Google Scholar] [CrossRef]
- Cronshey, R.; McCuen, R.; Miller, N.; Rawls, W.; Robbins, S.; Woodward, D. Urban Hydrology for Small Watersheds; United States Department of Agriculture, Natural Resources Conservation Service: Washington, DC, USA, 1986.
- ArcGIS Desktop: Release 10; Environmental Systems Research Institute: Redlands, CA, USA, 2015.
- AutoCAD Civil3D; Autodesk, Inc: San Rafael, CA, USA, 2018.
- RIVERMorph 5.2.0; RIVERMorph, LLC: Louisville, KY, USA, 2018.
- Rosgen, D.L. River Stability Field Guide, 2nd ed.; Wildlands Hydrology Books: Fort Collins, CO, USA, 2014. [Google Scholar]
- David, J.C.; Minshall, G.W.; Robinson, C.T.; Landres, P. Monitoring Wilderness Stream Ecosystems; United States Department of Agriculture Forest Service: Washington, DC, USA, 2001.
- Harman, W.; Barrett, T.; Jones, C.; James, A.; Peel, H. Application of the Large Woody Debris Index: A Field User Manual Version 1; Stream Mechanics and Ecosystem Planning & Restoration: Raleigh, NC, USA, 2017. [Google Scholar]
- US Army Corps of Engineers; NC Interagency Review Team. Monitoring Requirements and Performance Standards for Compensatory Mitigation in North Carolina; Wilmington District: Wilmington, NC, USA, 2013.
- Forestry Supplies, Inc. Using Forest Densiometers; Forestry Supplies, Inc: Jackson, MS, USA, 2008. [Google Scholar]
- ESRI, DigitalGlobe, GeoEye, i-cubed, USDA FSA, USGS, AEX, Getmapping, Aerogrid, IGN, Swisstopo, GIS User, Community. World Imagery Basemap, November 2018.
- NC Department of Environmental Quality. Appendix 7: Intensive Survey Branch Standard Operating Procedures Manual: Physical and Chemical Monitoring, Version 1.2; Division of Water Resources: Raleigh, NC, USA, 2013.
- YSI Professional Plus Multiparameter Instrument; YSI Incorporated: Yellow Springs, OH, USA.
- NC Department of Environmental Quality. Standard Operating Procedures for the Collection and Analysis of Benthic Macroinvertebrates; Division of Water Resources: Raleigh, NC, USA, 2016.
- Crawford, J.K.; Lenat, D.R. Effects of Land Use on the Water Quality and Biota of Three Streams in the Piedmont Province of North Carolina; US Geological Survey: Raleigh, NC, USA, 1989.
- Eaton, L.E.; Lenat, D.R. Comparison of a Rapid Bioassessment Method with North Carolina’s Qualitative Macroinvertebrate Collection Method. J. N. Am. Benthol. Soc. 1991, 10, 335–338. [Google Scholar] [CrossRef]
- Lenat, D.R.; Penrose, D.L. History of the EPT taxa richness metric. Bull. N. Am. Benthol. Soc. 1996, 13, 305–306. [Google Scholar]
- Penrose, D. Ecological Functions of Restored Stream Systems: Benthic Macroinvertebrates; 2003 Summary Addendum to Wetland Program Development Grant; NCDENR: Raleigh, NC, USA, 2004.
- Hilsenhoff, W.L. An Improved Biotic Index of Organic Stream Pollution. Gt. Lakes Entomol. 1987, 20, 10. [Google Scholar]
- Lenat, D.R. A Biotic Index for the Southeastern United States: Derivation and List of Tolerance Values, with Criteria for Assigning Water-Quality Ratings. J. N. Am. Benthol. Soc. 1993, 12, 279–290. [Google Scholar] [CrossRef]
- Hilsenhoff, W.L. Use of Arthropods to Evaluate Water Quality of Streams; Technical Bulleting No. 100; Department of Natural Resources: Madison, WI, USA, 1997.
- Plafkin, J.L.; Barbour, M.T.; Porter, S.K.; Gross, S.K.; Hughes, R.M. Rapid Bioassessment Protocols for Use in Streams and Rivers: Benthic Macroinvertebrates and Fish; EPA: Washington, DC, USA, 1989.
- Wallace, J.B.; Grubaugh, J.W.; Whiles, M.R. Biotic Indices and Stream Ecosystem Processes: Results from an Experimental Study. Ecol. Appl. 1996, 6, 140–151. [Google Scholar] [CrossRef]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning with Applications in R, 8th ed.; Springer: New York, NY, USA, 2017; ISBN 978-1-4614-7138-7. [Google Scholar]
- Faraway, J.J. Linear Models with R, 2nd ed.; Chapman & Hall/CRC: Boca Raton, FL, USA, 2014; ISBN 978-1-4398-8734-9. [Google Scholar]
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carré, G.; Marquéz, J.R.G.; Gruber, B.; Lafourcade, B.; Leitão, P.J.; et al. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- RStudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2019. [Google Scholar]
- Kennedy, P. A Guide to Econometrics; Blackwell: Oxford, UK, 1992. [Google Scholar]
- Mason, R.L.; Gunst, R.F.; Hess, J.L. Statistical Design and Analysis of Experiments With Applications to Engineering and Science; Balding, D.J., Bloomfield, P., Cressie, N.A.C., Fisher, N.I., Johnstone, I.M., Kadane, J.B., Ryan, L.M., Scott, D.W., Smith, A.F.M., Tuegels, J.L., Eds.; Wiley: New York, NY, USA, 2003. [Google Scholar]
- Menard, S. Applied Logistic Regression Analysis: Sage University Series on Quantitative Applications in the Social Sciences, 2nd ed.; Sage: Thousand Oaks, CA, USA, 2002. [Google Scholar]
- Neter, J.; Wasserman, W.; Kutner, M.H. Applied Linear Regression Models; Irwin: Homewood, IL, USA, 1989. [Google Scholar]
- Thomas Lumley Based on Fortran Code by Alan Miller. Leaps: Regression Subset Selection, R Package Version 3.1; 2020. Available online: https://CRAN.R-project.org/package=leaps (accessed on 22 May 2020).
- Friedman, J.; Hastie, T.; Tibshirani, R. Regularization Paths for Generalized Linear Models via Coordinate Descent. J. Stat. Softw. 2010, 33, 1–22. [Google Scholar] [CrossRef]
- Wyoming Stream Technical Team. Scientific Support for the Wyoming Stream Quantification Tool, Version 1.0; US Army Corps of Engineering, Omaha District, Wyoming Regulatory Office: Cheyenne, WY, USA, 2018.
- Wang, L.; Kanehl, P. Influences of Watershed Urbanization and Instream Habitat on Macroinvertebrates in Cold Water Streams. JAWRA J. Am. Water Resour. Assoc. 2003, 39, 1181–1196. [Google Scholar] [CrossRef]
- Nerbonne, B.A.; Vondracek, B. Effects of Local Land Use on Physical Habitat, Benthic Macroinvertebrates, and Fish in the Whitewater River, Minnesota, USA. Environ. Manag. N. Y. 2001, 28, 87–99. [Google Scholar] [CrossRef]
- Paller, M.H.; Specht, W.L.; Dyer, S.A. Effects of stream size on taxa richness and other commonly used benthic bioassessment metrics. Hydrobiologia 2006, 568, 309–316. [Google Scholar] [CrossRef]
- Florsheim, J.L.; Mount, J.L.; Chin, A. Bank Erosion as a Desirable Attribute of Rivers. BioScience 2008, 58, 519–529. [Google Scholar] [CrossRef]
- Roy, A.H.; Rosemond, A.D.; Leigh, D.S.; Paul, M.J.; Wallace, J.B. Habitat-specific responses of stream insects to land cover disturbance: Biological consequences and monitoring implications. J. N. Am. Benthol. Soc. 2003, 22, 292–307. [Google Scholar] [CrossRef]
- Simpson, A.; Turner, I.; Brantley, E.; Helms, B. Bank erosion hazard index as an indicator of near-bank aquatic habitat and community structure in a southeastern Piedmont stream. Ecol. Indic. 2014, 43, 19–28. [Google Scholar] [CrossRef]
- Wenner, D.B.; Ruhlman, M.; Eggert, S. The Importance of Specific Conductivity for Assessing Environmentally Impacted Streams. In Proceedings of the 2003 Georgia Water Resources Conference, Athens, GA, USA, 23–24 April 2003. [Google Scholar]
- Johnson, R.C.; Jin, H.-S.; Carreiro, M.M.; Jack, J.D. Macroinvertebrate community structure, secondary production and trophic-level dynamics in urban streams affected by non-point-source pollution. Freshw. Biol. 2013, 58, 843–857. [Google Scholar] [CrossRef]
- Boehme, E.A.; Zipper, C.E.; Schoenholtz, S.H.; Soucek, D.J.; Timpano, A.J. Temporal dynamics of benthic macroinvertebrate communities and their response to elevated specific conductance in Appalachian coalfield headwater streams. Ecol. Indic. 2016, 64, 171–180. [Google Scholar] [CrossRef]
- Pond, G.J.; Passmore, M.E.; Borsuk, F.A.; Reynolds, L.; Rose, C.J. Downstream effects of mountaintop coal mining: Comparing biological conditions using family- and genus-level macroinvertebrate bioassessment tools. J. N. Am. Benthol. Soc. 2008, 27, 717–737. [Google Scholar] [CrossRef]

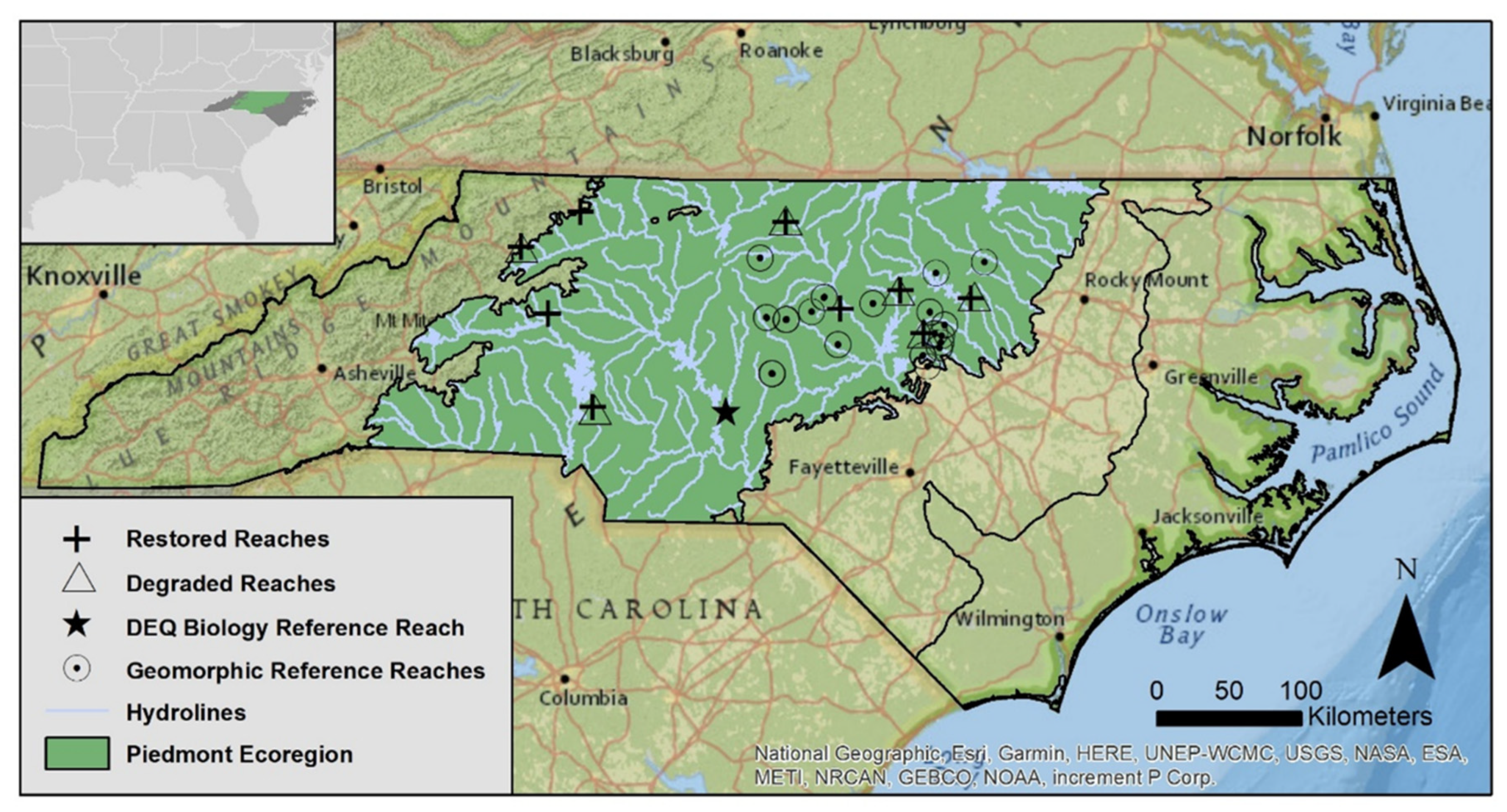
| Attribute | Restored | Degraded | Geomorphic Ref. | Biological Ref. | |||
|---|---|---|---|---|---|---|---|
| (Range; Arithmetic mean) | (Value) | ||||||
| Drainage Area (km2) | 1.0–22.0; | 6.2 | 0.5–9.8; | 3.4 | 0.2–21.3; | 4.3 | 8.9 |
| Impervious Cover (%) | 0.0–40.1; | 12.0 | 0.2–47.8; | 21.4 | 0.1–18.1; | 4.7 | 0.0 |
| Developed Cover (%) | 0.0–98.3; | 40.3 | 2.1–98.9; | 61.1 | 0.3–96.9; | 23.2 | 1.1 |
| Forested Cover (%) | 1.7–98.2; | 38.5 | 0.7–76.9; | 26.6 | 3.1–94.8; | 53.3 | 96.4 |
| Agricultural Cover (%) | 0.3–55.6; | 16.6 | 0.0–23.4; | 8.4 | 0.0–51.4; | 16.5 | 0.6 |
| Reach slope, Savg (%) | 0.2–4.6; | 1.0 | 0.3–2.1; | 0.9 | 0.4–2.3; | 0.9 | 0.9 |
| Median Substrate Diameter, D50 (mm) | 0.6–19.3; | 5.0 | 0.6–19.3; | 6.2 | 0.4–77.0; | 26.9 | 52.5 |
| Variable | Units | Arithmetic Mean ± SD | Range |
|---|---|---|---|
| Hydrology | |||
| Runoff curve number (CN) | - | 69.1 ± 8.1 | 51–84 |
| Concentrated flow points | #/305 m | 1.1 ± 1.8 | 0–5.9 |
| Soil compaction | cm | 24.8 ± 12.5 | 6.8–53.4 |
| Drainage area (DA) | km2 | 4.8 ± 5.5 | 0.2–22.0 |
| Impervious cover | % | 9.4 ± 12.5 | 0.0–47.8 |
| Developed cover | % | 33.8 ± 36.4 | 0.0–98.9 |
| Forested cover | % | 45.9 ± 30.8 | 0.7–98.2 |
| Agricultural cover | % | 14.6 ± 17.1 | 0.0–55.6 |
| Hydraulics | |||
| Bank height ratio (BHR) | - | 1.4 ± 0.5 | 1.0–2.8 |
| Entrenchment ratio (ER) | - | 6.2 ± 4.4 | 1.2–19.3 |
| Bankfull channel width (Wbkf) | m | 5.4 ± 2.6 | 2.2–13.8 |
| Mean bankfull depth (dbkf) | m | 0.4 ± 0.1 | 0.2–0.9 |
| Bankfull area (Abkf) | m2 | 2.5 ± 1.8 | 0.6–9.1 |
| Average channel slope (Savg) | % | 1.0 ± 0.8 | 0.2–4.6 |
| Width-to-depth ratio (W/D) | - | 12.8 ± 4.7 | 6.9–22.6 |
| Geomorphology | |||
| Riffle & run extent (% Riffle) | % | 59.3 ± 14.9 | 33.9–95.5 |
| Pool spacing ratio | - | 6.3 ± 4.2 | 0–23.08 |
| Pool depth ratio | - | 2.8 ± 0.9 | 1.4–5.4 |
| Sinuosity | - | 1.3 ± 0.2 | 1.0–1.7 |
| Riparian buffer width | m | 113.7 ± 131.2 | 0–619 |
| Streambank erosion | % | 16.5 ± 14.3 | 0–55 |
| Bank erosion hazard index (BEHI) | Index | 29.6 ± 6.7 | 20–45 |
| Near-bank stress (NBS) | Index | 2.9 ± 1.2 | 1–5 |
| Basal area | m2/ha | 20.5 ± 13.5 | 0–58 |
| Large woody debris index (LWDI) | Index | 184.6 ± 147.1 | 10–542 |
| Canopy cover | % | 84.4 ± 22.9 | 2–97 |
| D50 | mm | 18.2 ± 21.8 | 0.4–77.0 |
| D84 | mm | 75.3 ± 106.9 | 3.0–618.0 |
| Physicochemistry | |||
| Total nitrogen (TN) | mg/L | 1.1 ± 1.0 | 0.18–5.85 |
| Total phosphorus (TP) | mg/L | 0.1 ± 0.0 | 0.01–0.20 |
| Summer temperature | deg. C | 22.1 ± 2.1 | 17.1–25.7 |
| Fecal coliform | cfu/100 mL | 964.7 ± 1498.4 | 10–5909 |
| Shredder taxa (% Shredders) | % | 9.8 ± 11.7 | 0.0–50.0 |
| Specific conductivity * | uS/cm | 122.3 ± 72.2 | 34.1–370.6 |
| Biology | |||
| EPT richness | # & Index | 11.1 ± 9.5 | 0–32 |
| NC biotic index (NCBI) | Index | 5.3 ± 1.4 | 2.03–8.43 |
| Total taxa richness | # | 29.1 ± 13.9 | 6–54 |
| Pyramid Level | EPT Richness | NCBI | ||||
|---|---|---|---|---|---|---|
| Metric | Stepwise1 | Lasso | Ridge | Stepwise1 | Lasso | Ridge |
| Hydrology | ||||||
| CN | - | - | −0.03 | - | - | 0.05 |
| Conc. flow points | - | - | 0.02 | - | - | −0.01 |
| Soil compaction | - | - | −0.01 | - | - | −0.02 |
| Hydraulics | ||||||
| BHR | - | - | −0.03 | - | - | 0.04 |
| ER | - | - | 0.02 | - | - | −0.02 |
| Geomorphology | ||||||
| NBS | −0.48 *** | −0.19 | −0.06 | 0.24 * | - | 0.05 |
| BEHI | 0.23 * | - | 0.01 | - | - | −0.03 |
| % Streambank erosion | −0.33 ** | - | −0.01 | - | - | 0.01 |
| LWDI | - | - | −0.01 | - | - | 0.00 |
| Buffer width | - | - | 0.03 | - | - | −0.02 |
| Basal area | - | - | −0.02 | - | - | 0.04 |
| Canopy cover | - | - | 0.00 | - | - | 0.00 |
| Pool depth ratio | −0.49 *** | −0.02 | −0.05 | 0.52 *** | - | 0.07 |
| % Riffle | - | - | 0.03 | - | - | −0.05 |
| P-P spacing | - | - | 0.01 | - | - | −0.01 |
| Sinuosity | - | - | −0.03 | - | - | 0.00 |
| Physicochemistry | ||||||
| TN | - | - | −0.02 | - | - | 0.02 |
| TP | - | - | −0.02 | 0.20 | - | 0.04 |
| Fecal coliform | - | - | 0.01 | - | - | 0.01 |
| Summer temp. | −0.38 *** | −0.03 | −0.05 | 0.44 *** | - | 0.06 |
| % Shredders | - | - | 0.03 | - | - | −0.05 |
| R2 | 0.64 | 0.37 | 0.64 | 0.53 | - | 0.63 |
| Pyramid Level | EPT Richness | NCBI | ||||
|---|---|---|---|---|---|---|
| Metric | Stepwise 1 | Lasso | Ridge | Stepwise 1 | Lasso | Ridge |
| Hydrology | ||||||
| % Agricultural cover | - | - | 0.02 | −0.25 | - | −0.04 |
| DA | N/A | - | 0.03 | N/A | - | −0.01 |
| CN | - | - | −0.03 | 0.31 * | - | 0.04 |
| Conc. flow points | - | - | 0.02 | −0.09 | - | −0.01 |
| Soil compaction | - | - | −0.01 | −0.05 | - | −0.02 |
| Hydraulics | ||||||
| ER | 0.37 *** | - | 0.02 | - | - | −0.02 |
| W/D | 0.40 *** | - | 0.03 | - | - | −0.02 |
| Savg | N/A | - | 0.04 | N/A | −0.03 | −0.06 |
| dbkf | 0.27 *** | - | 0.02 | 0.06 | - | 0.01 |
| Geomorphology | ||||||
| NBS | N/A | −0.15 | −0.05 | N/A | - | 0.04 |
| BEHI | 0.24 *** | - | 0.01 | - | - | −0.03 |
| % Streambank erosion | N/A | - | −0.01 | N/A | - | 0.00 |
| LWDI | - | - | −0.01 | - | - | 0.00 |
| Buffer width | 0.28 *** | - | 0.03 | - | - | −0.02 |
| Basal area | - | - | −0.02 | - | - | 0.04 |
| Canopy cover | - | - | 0.00 | - | - | 0.00 |
| Pool depth ratio | −0.25 *** | - | −0.04 | - | - | 0.06 |
| % Riffle | 0.49 *** | - | 0.03 | - | - | −0.04 |
| P-P spacing | - | - | 0.01 | - | - | −0.01 |
| Sinuosity | - | - | −0.03 | - | - | 0.00 |
| D50 | - | 0.03 | 0.04 | - | - | −0.04 |
| D84 | 0.41 *** | - | 0.03 | - | - | −0.02 |
| Physicochemistry | ||||||
| TN | N/A | - | −0.02 | N/A | - | 0.02 |
| TP | - | - | −0.02 | - | - | 0.03 |
| Fecal coliform | - | - | 0.01 | - | - | 0.01 |
| Summer temp. | −0.59 *** | −0.02 | −0.05 | - | - | 0.05 |
| % Shredders | - | - | 0.03 | - | - | −0.05 |
| R2 | 0.88 | 0.38 | 0.75 | 0.18 | 0.21 | 0.70 |
| EPT Richness | NCBI | ||||||
|---|---|---|---|---|---|---|---|
| Stepwise | Lasso | Ridge | Stepwise | Lasso | Ridge | ||
| SQT | # of predictor variables | 5 | 3 | 21 | 4 | 0 | 21 |
| Test MSE | 1.24 | 1.40 | 1.26 | 1.46 | 1.26 | 1.26 | |
| SQT+ | # of predictor variables | 9 | 3 | 22 | 5 | 1 | 27 |
| Test MSE | 0.79 | 1.33 | 1.18 | 1.17 | 1.26 | 1.01 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donatich, S.; Doll, B.; Page, J.; Nelson, N. Can the Stream Quantification Tool (SQT) Protocol Predict the Biotic Condition of Streams in the Southeast Piedmont (USA)? Water 2020, 12, 1485. https://doi.org/10.3390/w12051485
Donatich S, Doll B, Page J, Nelson N. Can the Stream Quantification Tool (SQT) Protocol Predict the Biotic Condition of Streams in the Southeast Piedmont (USA)? Water. 2020; 12(5):1485. https://doi.org/10.3390/w12051485
Chicago/Turabian StyleDonatich, Sara, Barbara Doll, Jonathan Page, and Natalie Nelson. 2020. "Can the Stream Quantification Tool (SQT) Protocol Predict the Biotic Condition of Streams in the Southeast Piedmont (USA)?" Water 12, no. 5: 1485. https://doi.org/10.3390/w12051485
APA StyleDonatich, S., Doll, B., Page, J., & Nelson, N. (2020). Can the Stream Quantification Tool (SQT) Protocol Predict the Biotic Condition of Streams in the Southeast Piedmont (USA)? Water, 12(5), 1485. https://doi.org/10.3390/w12051485





