Effect of Temperature on the Size of Sedimentary Remains of Littoral Chydorids
Abstract
:1. Introduction
2. Materials and Methods
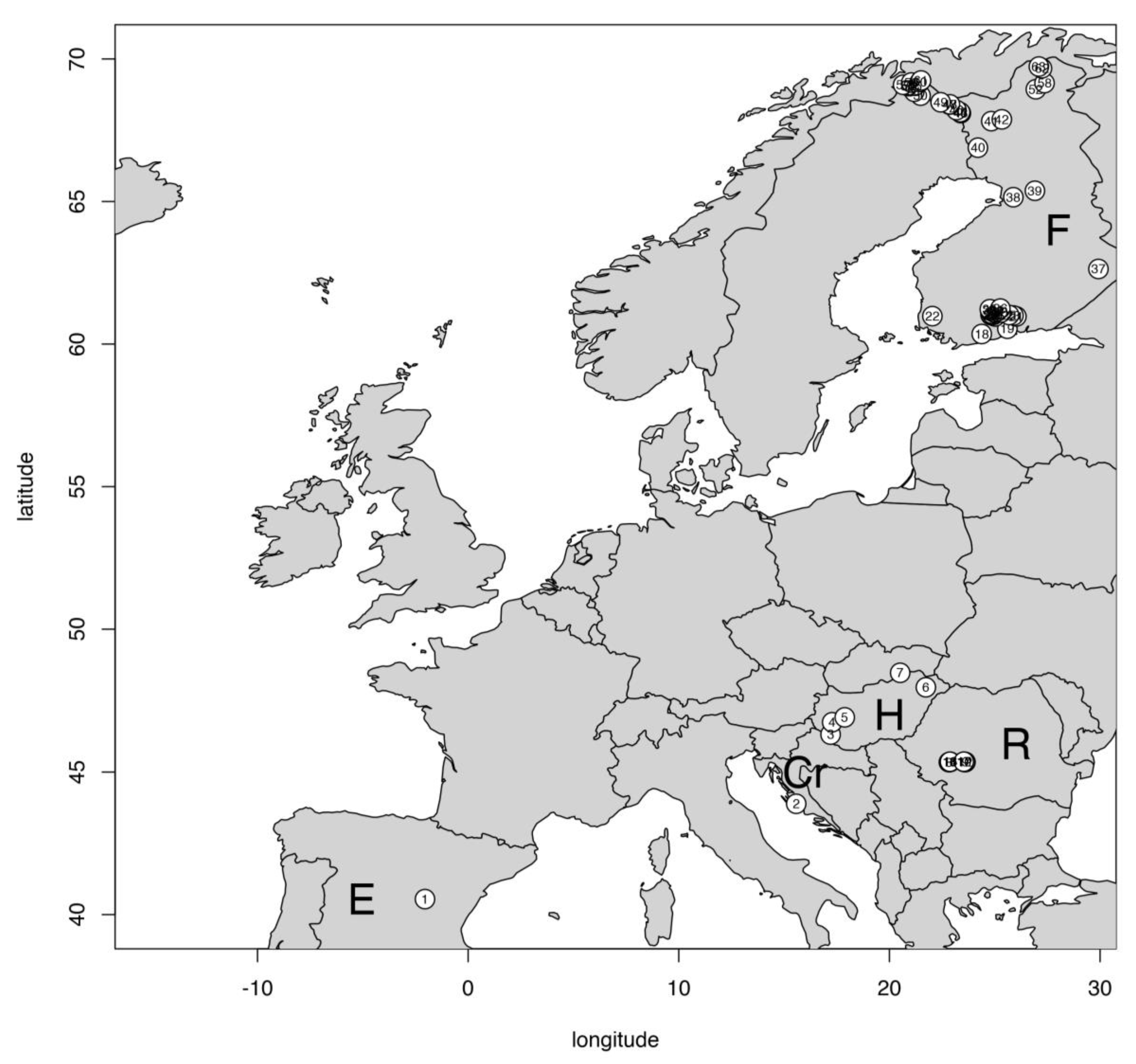
2.1. Study Sites
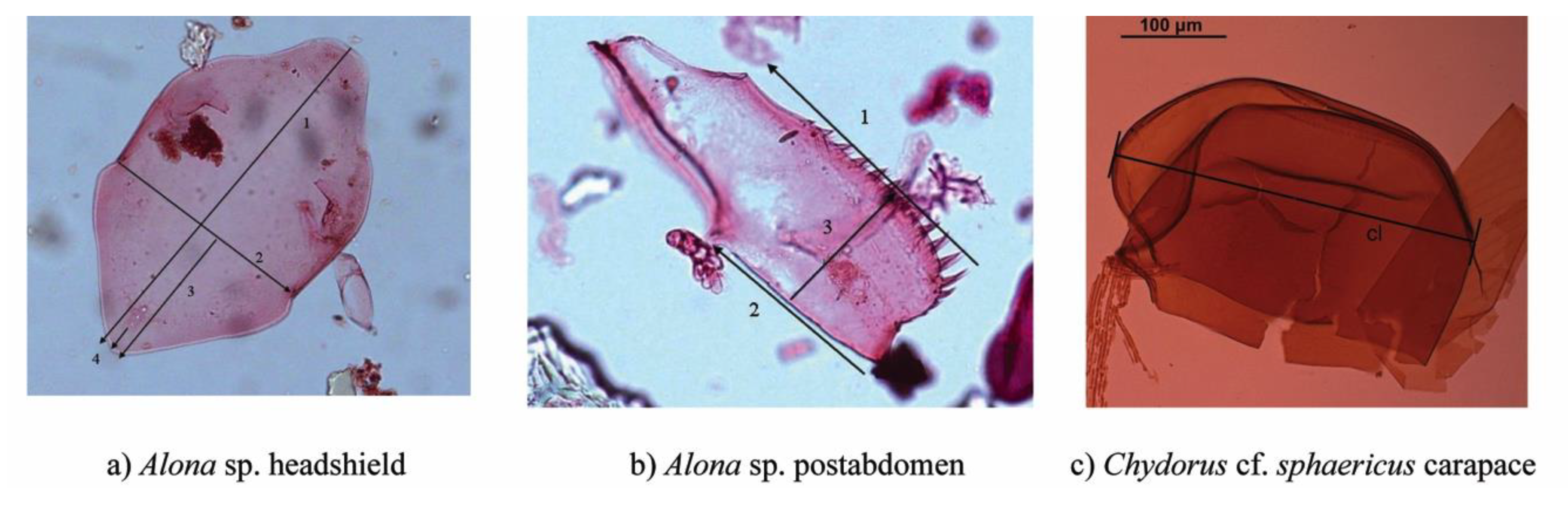
2.2. Subfossil Analyses
2.3. Data Analyses
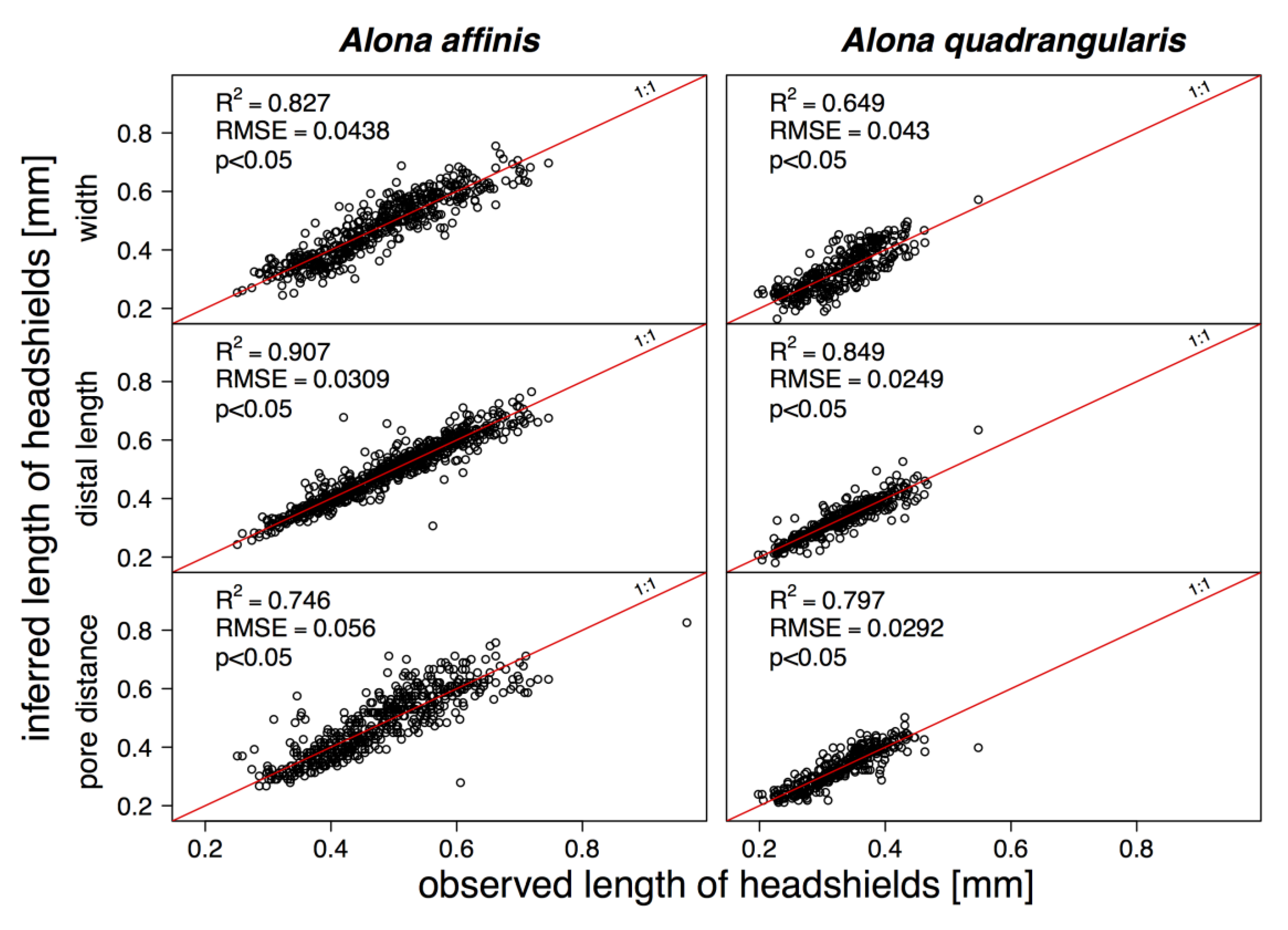
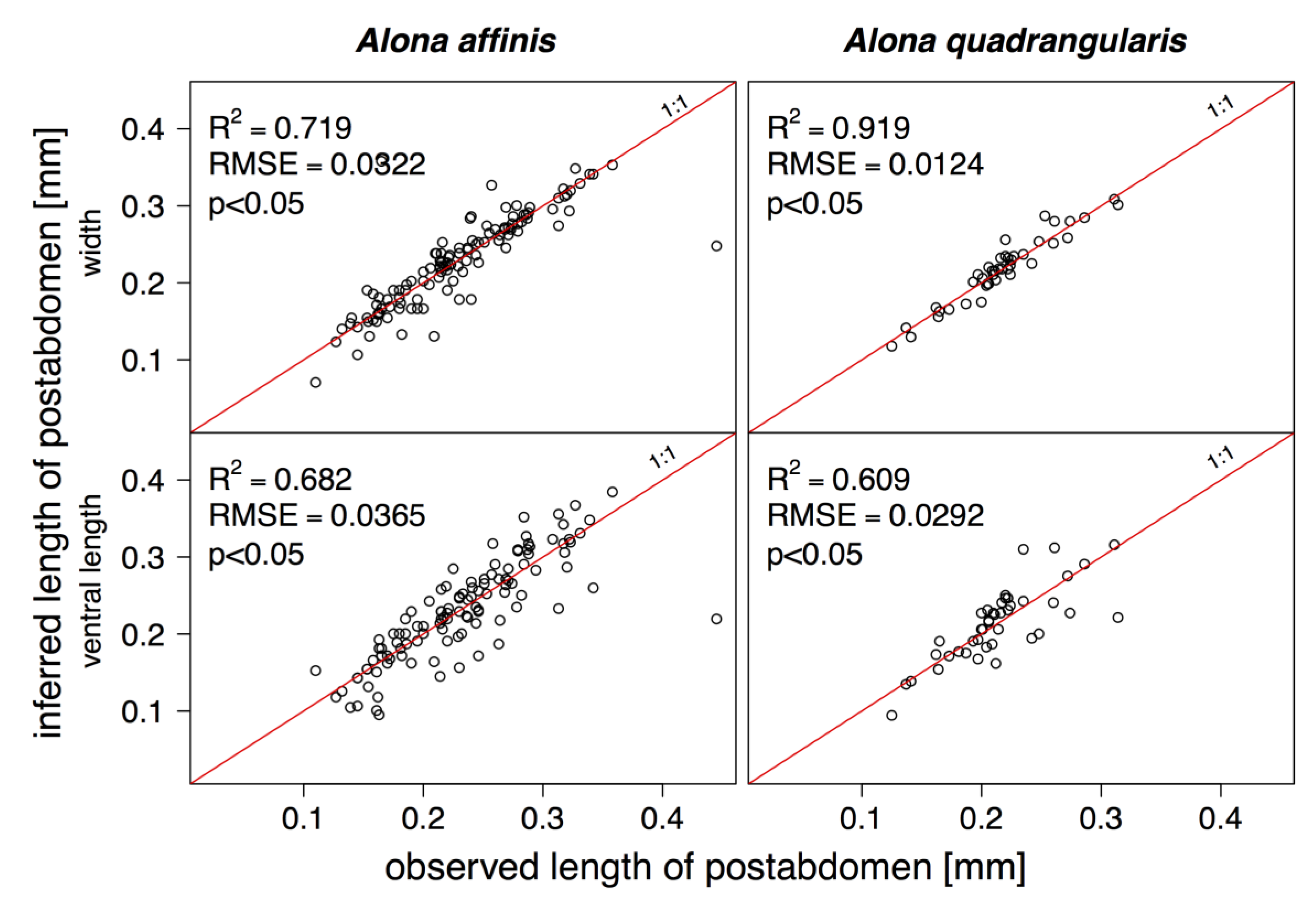
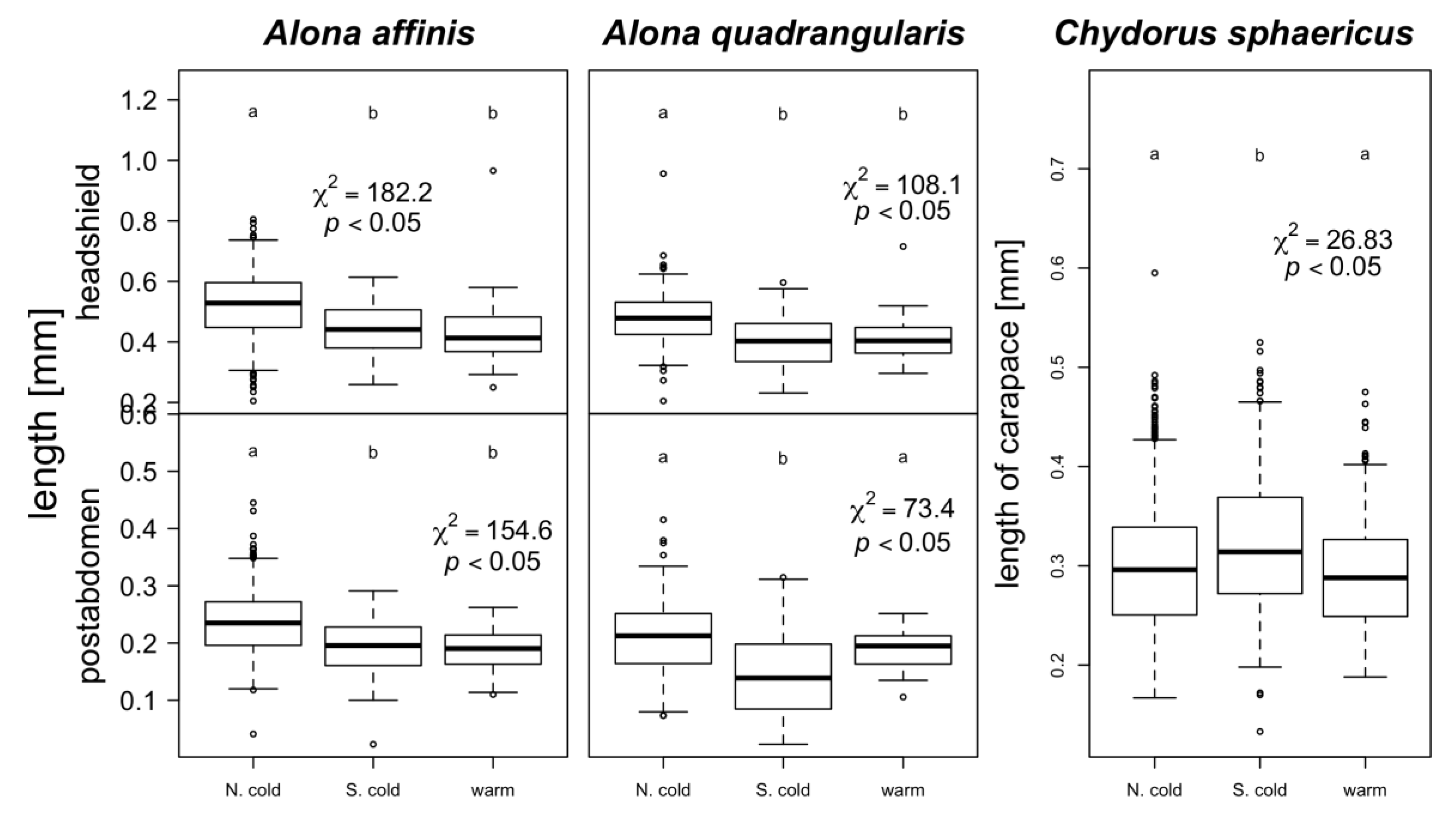
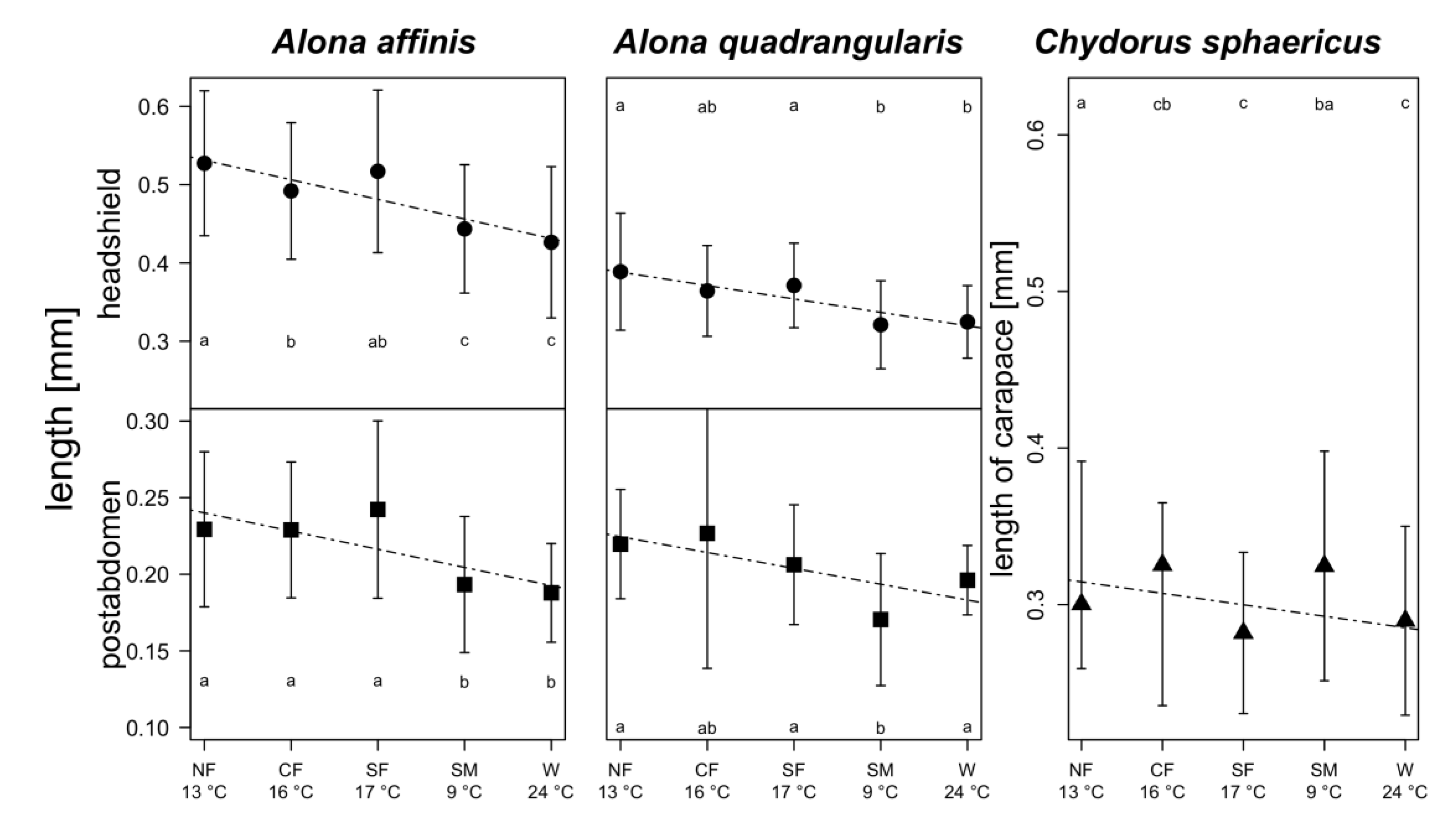
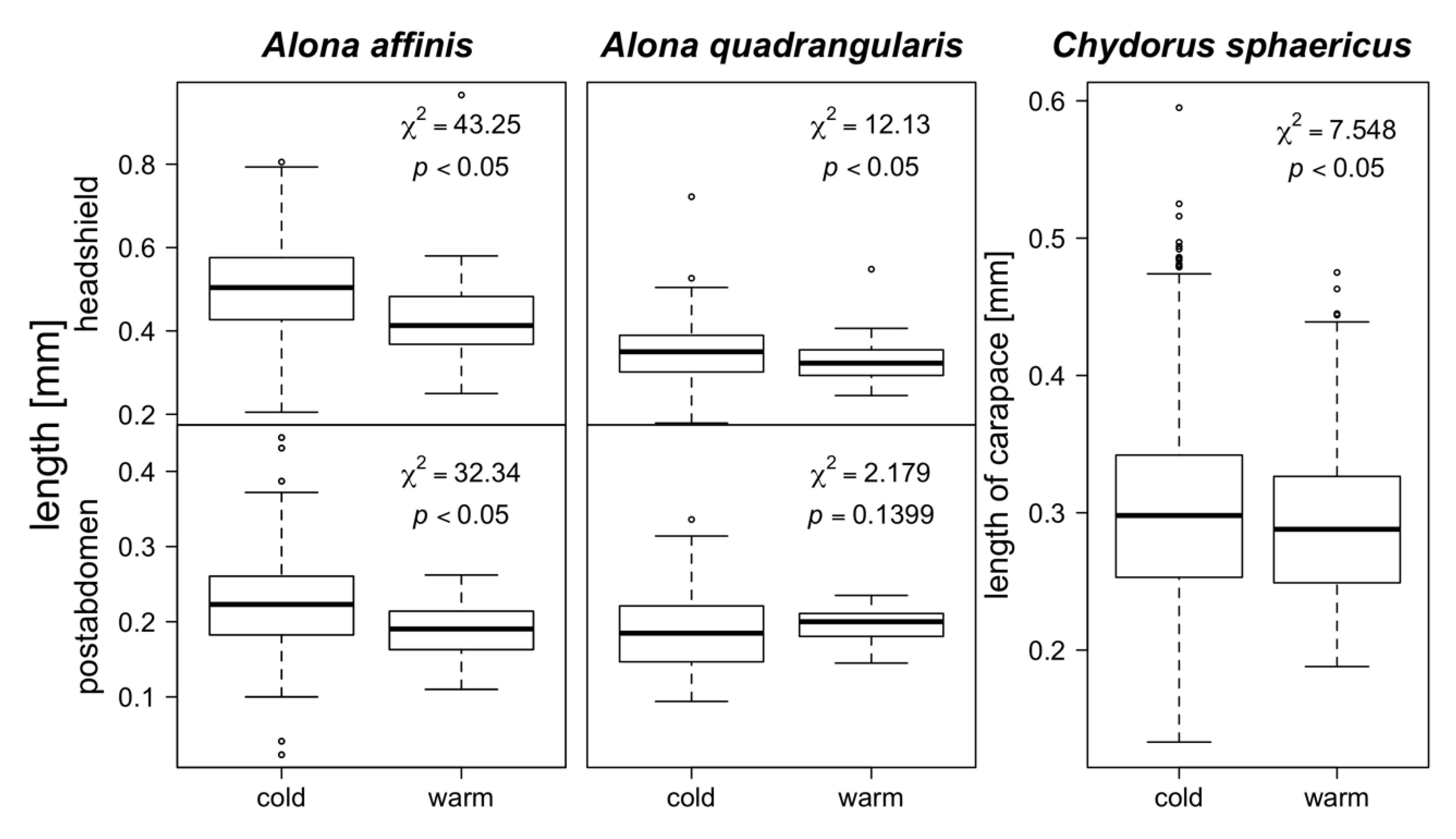
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Alona affinis | |||||
| headshield | a | b | t-value | R2 | p |
| width | 0.5465 | −0.00881 | 56.7427 | 0.8712 | <0.05 |
| distal length | 0.4232 | −0.00074 | 120.2307 | 0.9527 | <0.05 |
| pore distance | 0.0878 | −0.02046 | 46.6102 | 0.8165 | <0.05 |
| postabdomen | a | b | t-value | R2 | p |
| width | 0.4177 | 0.01548 | 32.5566 | 0.9013 | <0.05 |
| distal length | 0.5216 | 0.0205 | 20.2443 | 0.785 | <0.05 |
| Alona quadrangularis | |||||
| headshield | a | b | t-value | R2 | p |
| width | 0.5446 | 0.01065 | 26.8956 | 0.697 | <0.05 |
| distal length | 0.4074 | 0.00051 | 61.1244 | 0.9125 | <0.05 |
| pore distance | 0.151 | −0.0276 | 44.3056 | 0.8639 | <0.05 |
| postabdomen | a | b | t-value | R2 | p |
| width | 0.4192 | 0.01665 | 27.6451 | 0.9526 | <0.05 |
| ventral length | 0.5192 | 0.02206 | 10.3076 | 0.7296 | <0.05 |

| Akaike’s An Information Criterion (AIC) | |||
|---|---|---|---|
| Normal | Lognormal | Gamma | |
| Alona affinis | |||
| headshield | −2267 | −2230 | −2261 |
| postabdomen | −3150 | −3072 | −3137 |
| A. quadrangularis | |||
| headshield | −1609 | −1613 | −1620 |
| postabdomen | −1497 | −1489 | −1497 |
| Chydorus sphaericus | |||
| carapace | −4120 | −4211 | −4208 |
| Model | Parameter | Estimate | Std. Error | t Value | p-Value | AIC | |
|---|---|---|---|---|---|---|---|
| Alona affinis | |||||||
| headsield | all lake model | Intercept | 0.288 | 0.011 | 23.178 | <0.001 | −3491 |
| latitude | 0.002 | 0.000 | 13.87 | <0.001 | |||
| omit-model | Intercept | 0.291 | 0.021 | 12.075 | <0.001 | −2628 | |
| latitude | 0.002 | 0.000 | 7.473 | <0.001 | |||
| all lake model | Intercept | 0.475 | 0.008 | 47.63 | <0.001 | −3314 | |
| TJul | 0.001 | 0.001 | 1.692 | 0.091 | |||
| omit-model | Intercept | 0.637 | 0.011 | 43.813 | <0.001 | −2624 | |
| TJul | −0.005 | 0.001 | −7.19 | <0.001 | |||
| postabdomen | all lake model | Intercept | 0.120 | 0.009 | 12.202 | <0.001 | −3277 |
| latitude | 0.002 | 0.000 | 11.673 | <0.001 | |||
| omit-model | Intercept | 0.160 | 0.017 | 8.891 | <0.001 | −2280 | |
| latitude | 0.001 | 0.000 | 4.985 | <0.001 | |||
| all lake model | Intercept | 0.475 | 0.008 | 47.63 | <0.001 | −3314 | |
| TJul | 0.001 | 0.001 | 1.692 | 0.091 | |||
| omit-model | Intercept | 0.637 | 0.011 | 43.813 | <0.001 | −2624 | |
| TJul | −0.005 | 0.001 | −7.19 | <0.001 | |||
| Alona quadrangularis | |||||||
| headsield | all lake model | Intercept | 0.185 | 0.014 | 13.325 | <0.001 | −1729 |
| latitude | 0.003 | 0.000 | 11.593 | <0.001 | |||
| omit-model | Intercept | 0.196 | 0.024 | 8.342 | <0.001 | −940 | |
| latitude | 0.003 | 0.000 | 7.154 | <0.001 | |||
| all lake model | Intercept | 0.310 | 0.009 | 34.854 | <0.001 | −1623 | |
| TJul | 0.002 | 0.001 | 3.955 | <0.001 | |||
| omit-model | Intercept | 0.499 | 0.019 | 25.684 | <0.001 | −939 | |
| TJul | −0.008 | 0.001 | −7.087 | <0.001 | |||
| postabdomen | all lake model | Intercept | 0.080 | 0.010 | 7.585 | <0.001 | −1728 |
| latitude | 0.002 | 0.000 | 9.258 | <0.001 | |||
| omit-model | Intercept | 0.148 | 0.022 | 6.493 | <0.001 | −722 | |
| latitude | 0.001 | 0.000 | 2.366 | 0.019 | |||
| all lake model | Intercept | 0.135 | 0.006 | 20.307 | <0.001 | −1697 | |
| TJul | 0.003 | 0.000 | 7.076 | <0.001 | |||
| omit-model | Intercept | 0.257 | 0.017 | 13.76 | <0.001 | −722 | |
| TJul | −0.002 | 0.001 | −2.423 | 0.016 | |||
| Chydorus spharicus | |||||||
| carapace | all lake model | Intercept | 0.280 | 0.011 | 24.852 | <0.001 | −4132 |
| latitude | 0.000 | 0.000 | 1.909 | 0.056 | |||
| omit-model | Intercept | 0.190 | 0.014 | 13.313 | <0.001 | −3727 | |
| latitude | 0.002 | 0.000 | 7.617 | <0.001 | |||
| all lake model | Intercept | 0.378 | 0.007 | 55.581 | <0.001 | −4256 | |
| TJul | −0.005 | 0.000 | −11.543 | <0.001 | |||
| omit-model | Intercept | 0.386 | 0.008 | 46.03 | <0.001 | −3780 | |
| TJul | −0.006 | 0.001 | −10.69 | 0.000 | |||
References
- Litchman, E.; Ohman, M.D.; Kiørboe, T. Trait-Based Approaches to Zooplankton Communities. J. Plankton Res. 2013, 35, 473–484. [Google Scholar] [CrossRef] [Green Version]
- Carpenter, S.R.; Kitchell, J.F. Consumer Control of Lake Productivity. BioScience 1988, 38, 764. [Google Scholar] [CrossRef]
- Kerfoot, W.C. Egg-Size Cycle of a Cladoceran. Ecology 1974, 55, 1259–1270. [Google Scholar] [CrossRef]
- Razak, S.A.; Saisho, T. Seasonal Variations in Egg Size, Brood Size and Body Length of Bosmina Longirostris (Crustacea: Cladocera) from Lake Ikeda, Japan. Malays. J. Sci. 2004, 23, 121–135. [Google Scholar]
- Havens, K.E.; Pinto-Coelho, R.M.; Beklioǧlu, M.; Christoffersen, K.S.; Jeppesen, E.; Lauridsen, T.L.; Mazumder, A.; Méthot, G.; Alloul, B.P.; Tavşanoğlu, U.N.; et al. Temperature Effects on Body Size of Freshwater Crustacean Zooplankton from Greenland to the Tropics. Hydrobiologia 2015, 743, 27–35. [Google Scholar] [CrossRef] [Green Version]
- Vijverberg, J.; Richter, A.F. Population Dynamics and Production of Daphnia Hyalina (Leydig) and Daphnia Cucullata Sars in Tjeukemeer. Hydrobiologia 1982, 9, 235–259. [Google Scholar] [CrossRef]
- Moore, M.V.; Folt, C.L.; Stemberger, R.S. Consequences of Elevated Temperatures for Zooplankton Assemblages in Temperate Lakes. Arch. Hydrobiol. 1996, 135, 289–319. [Google Scholar]
- Chang, K.-H.; Hanazato, T. Seasonal and Reciprocal Succession and Cyclomorphosis of Two Bosmina Species (Cladocera, Crustacea) Co-Existing in a Lake: Their Relationship with Invertebrate Predators. J. Plankton Res. 2003, 25, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Deng, D.; Xie, P. Effect of Food and Temperature on the Growth and Development of Moina Irrasa (Cladocera: Moinidae). J. Freshw. Ecol. 2003, 18, 503–513. [Google Scholar] [CrossRef]
- Hart, R.C.; Bychek, E.A. Body Size in Freshwater Planktonic Crustaceans: An Overview of Extrinsic Determinants and Modifying Influences of Biotic Interactions. Hydrobiologia 2011, 668, 61–108. [Google Scholar] [CrossRef]
- Gillooly, J.F.; Dodson, S.I. Latitudinal Patterns in the Size Distribution and Seasonal Dynamics of New World, Freshwater Cladocerans. Limnol. Oceanogr. 2000, 45, 22–30. [Google Scholar] [CrossRef]
- Perrin, N. Why Are Offspring Born Larger When It Is Colder? Phenotypic for Offspring Plasticity Size in the Cladoceran Simocephalus Vetulus (Muller). Funct. Ecol. 1988, 2, 283–288. [Google Scholar] [CrossRef]
- Atkinson, D. Temperature and Organism Size-A Law for Ectotherms? Adv. Ecol. Res. 1994, 25, 1–58. [Google Scholar]
- Atkinson, D. Effects of Temperature on the Size of Aquatic Ectotherms: Exception to General Rule. J. Therm. Biol. 1995, 20, 61–71. [Google Scholar] [CrossRef]
- Smol, J.P. Pollution of Lakes and Rivers; Blackwell Publishing: Oxford, UK, 2002. [Google Scholar]
- Flower, R.J. Diatom Preservation: Experiments and Observations on Dissolution and Breakage in Modern and Fossil Material. Hydrobiologia 1993, 269–270, 473–484. [Google Scholar] [CrossRef]
- Battarbee, R.W.; Anderson, N.J.; Jeppesen, E.; Leavitt, P.R. Combining Palaeolimnological and Limnological Approaches in Assessing Lake Ecosystem Response to Nutrient Reduction. Freshw. Biol. 2005, 50, 1772–1780. [Google Scholar] [CrossRef]
- Kattel, G.R.; Battarbee, R.W.; Mackay, A.W.; Birks, H.J.B. Recent Ecological Change in a Remote Scottish Mountain Loch: An Evaluation of a Cladocera-Based Temperature Transfer-Function. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2008, 259, 51–76. [Google Scholar] [CrossRef]
- López-Blanco, C.; Miracle, M.R.; Vicente, E. Cladoceran Assemblages in a Karstic Lake as Indicators of Hydrological Alterations. Hydrobiologia 2011, 676, 249–261. [Google Scholar] [CrossRef]
- Korhola, A.; Rautio, M. 2. Cladocera and Other Branchiopod Crustaceans. In Tracking Environmental Change Using Lake Sediments. Volume 4: Zoological Indicators; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 5–41. [Google Scholar] [CrossRef]
- Jeppesen, E.; Leavitt, P.; De Meester, L.; Jensen, J.P. Functional Ecology and Palaeolimnology: Using Cladoceran Remains to Reconstruct Anthropogenic Impact. Trends Ecol. Evol. 2001, 16, 191–198. [Google Scholar] [CrossRef]
- Jeppesen, E.; Nõges, P.; Davidson, T.A.; Haberman, J.; Nõges, T.; Blank, K.; Lauridsen, T.L.; Søndergaard, M.; Sayer, C.; Laugaste, R.; et al. Zooplankton as Indicators in Lakes: A Scientific-Based Plea for Including Zooplankton in the Ecological Quality Assessment of Lakes According to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
- Korosi, J.B.; Paterson, A.M.; Desellas, A.M.; Smol, J.P. Linking Mean Body Size of Pelagic Cladocera to Environmental Variables in Precambrian Shield Lakes: A Paleolimnological Approach. J. Limnol. 2008, 67, 22–34. [Google Scholar] [CrossRef] [Green Version]
- Bjerring, R.; Becares, E.; Declerck, S.; Gross, E.M.; Hansson, L.-A.; Kairesalo, T.; Nykänen, M.; Halkiewicz, A.; Kornijów, R.; Conde-Porcuna, J.M.; et al. Subfossil Cladocera in Relation to Contemporary Environmental Variables in 54 Pan-European Lakes. Freshw. Biol. 2009, 54, 2401–2417. [Google Scholar] [CrossRef]
- Korponai, J.; Magyari, E.K.; Buczkó, K.; Iepure, S.; Namiotko, T.; Czakó, D.; Kövér, C.; Braun, M. Cladocera Response to Late Glacial to Early Holocene Climate Change in a South Carpathian Mountain Lake. Hydrobiologia 2011, 676, 223–235. [Google Scholar] [CrossRef]
- Nevalainen, L.; Luoto, T.P.; Kultti, S.; Sarmaja-Korjonen, K. Spatio-Temporal Distribution of Sedimentary Cladocera (Crustacea: Branchiopoda) in Relation to Climate. J. Biogeogr. 2013, 40, 1548–1559. [Google Scholar] [CrossRef]
- Nevalainen, L.; Rantala, M.V.; Luoto, T.P. Sedimentary Cladoceran Assemblages and Their Functional Attributes Record Late Holocene Climate Variability in Southern Finland. J. Paleolimnol. 2015, 1–14. [Google Scholar] [CrossRef]
- Burks, R.L.; Lodge, D.M.; Jeppesen, E.; Lauridsen, T.L. Diel Horizontal Migration of Zooplankton: Costs and Benefits of Inhabiting the Littoral. Freshw. Biol. 2002, 47, 343–365. [Google Scholar] [CrossRef]
- Sagrario, G.; de los Ángeles, M.; Balseiro, E.; Ituarte, R.; Spivak, E. Macrophytes as Refuge or Risky Area for Zooplankton: A Balance Set by Littoral Predacious Macroinvertebrates. Freshw. Biol. 2009, 54, 1042–1053. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.; Pedersen, L.J.; Jensen, L. Top-down Control in Freshwater Lakes: The Role of Nutrient State, Submerged Macrophytes and Water Depth. Hydrobiologia 1997, 342, 151–164. [Google Scholar] [CrossRef]
- De Los Ángeles, M.; Sagrario, G.; Balseiro, E. The Role of Macroinvertebrates and Fish in Regulating the Provision by Macrophytes of Refugia for Zooplankton in a Warm Temperate Shallow Lake. Freshw. Biol. 2010, 55, 2153–2166. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Søndergaard, M.; Christoffersen, K. The Structuring Role of Submerged Macrophytes in Lakes; Jeppesen, E., Søndergaard, M., Søndergaard, M., Christoffersen, K., Eds.; Ecological Studies; Springer: New York, NY, USA, 1998; Volume 131. [Google Scholar] [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes; Springer: Dordrecht, The Netherlands, 2004. [Google Scholar] [CrossRef]
- Płaska, W.; Mieczan, T. Effects of Water Bugs on Crustacean Zooplankton in a Shallow Littoral Zone. Knowl. Manag. Aquat. Ecosyst. 2018. [Google Scholar] [CrossRef] [Green Version]
- Adamczuk, M. The Effect of Habitat Complexity on the Contribution of Some Littoral-Benthic Cladocera to the Pelagic Food Web. Mar. Freshw.Res. 2013, 64, 1049–1057. [Google Scholar] [CrossRef]
- Adamczuk, M. Niche Separation by Littoral-Benthic Chydoridae (Cladocera, Crustacea) in a Deep Lake—Potential Drivers of Their Distribution and Role in Littoral-Pelagic Coupling. J. Limnol. 2014, 73, 490–501. [Google Scholar] [CrossRef] [Green Version]
- Choedchim, W.; Damme, K.; Van Maiphae, S. Spatial and Temporal Variation of Cladocera in a Tropical Shallow Lake. Ann. Limnol. 2017, 53, 233–252. [Google Scholar] [CrossRef] [Green Version]
- Basińska, A.M.; Antczak, M.; Świdnicki, K.; Jassey, V.E.J.; Kuczyńska-Kippen, N. Habitat Type as Strongest Predictor of the Body Size Distribution of Chydorus Sphaericus (O. F. Müller) in Small Water Bodies. Int. Rev. Hydrobiol. 2014, 99, 382–392. [Google Scholar] [CrossRef]
- Rivera-De la Parra, L.; Sarma, S.S.S.; Nandini, S. Effects of Predation by Hydra (Cnidaria) on Cladocerans (Crustacea: Cladocera). J. Limnol. 2016, 75 (Suppl. 1), 39–47. [Google Scholar] [CrossRef] [Green Version]
- Rabus, M.; Söllradl, T.; Clausen-Schaumann, H.; Laforsch, C. Uncovering Ultrastructural Defences in Daphnia Magna—An Interdisciplinary Approach to Assess the Predator-Induced Fortification of the Carapace. PLoS ONE 2013, 8, e67856. [Google Scholar] [CrossRef] [Green Version]
- Laforsch, C.; Ngwa, W.; Grill, W.; Tollrian, R. An Acoustic Microscopy Technique Reveals Hidden Morphological Defenses in Daphnia. Proc. Natl. Acad. Sci. USA 2004, 101, 15911–15914. [Google Scholar] [CrossRef] [Green Version]
- Rizo, E.Z.; Xu, S.; Tang, Q.; Papa, R.D.S.; Dumont, H.J.; Qian, S.S.; Han, B.P. A Global Analysis of Cladoceran Body Size and Its Variation Linking to Habitat, Distribution and Taxonomy. Zool. J. Linn. Soc. 2019, 187, 1119–1130. [Google Scholar] [CrossRef]
- Pirinen, P.; Simola, H.; Aalto, J.; Kaukoranta, J.-P.; Karlsson, P.; Ruuhela, R. Tilastoja Suomen Ilmastosta 1981–2010; Ilmatieteen laitos: Helsinki, Finland, 2012; Volume 1.
- Korponai, J.; Varga, K.A.; Lengré, T.; Papp, I.; Tóth, A.; Braun, M. Paleolimnological Reconstruction of the Trophic State in Lake Balaton (Hungary) Using Cladocera Remains. Hydrobiologia 2011, 676, 237–248. [Google Scholar] [CrossRef]
- Livingstone, D.M.; Lotter, A.F. The Relationship between Air and Water Temperatures in Lakes of the Swiss Plateau: A Case Study with Palæolimnological Implications. J. Paleolimnol. 1998, 19, 181–198. [Google Scholar] [CrossRef]
- Livingstone, D.M.; Padisák, J. Large-Scale Coherence in the Response of Lake Surface-Water Temperatures to Synoptic-Scale Climate Forcing during Summer. Limnol. Oceanogr. 2007, 52, 896–902. [Google Scholar] [CrossRef] [Green Version]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very High Resolution Interpolated Climate Surfaces for Global Land Areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Phillips, S.; Leathwick, J.; Elith, J. Dismo: Species Distribution Modeling; 2016. [Google Scholar]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-Km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Szeroczyńska, K.; Sarmaja-Korjonen, K. Atlas of Subfossil Cladocera from Central and Northern Europe; Friends of Lower Vistula Society: Swiecie, Poland, 2007. [Google Scholar]
- Pohlert, T. Package ‘PMCMRplus’: Calculate Pairwise Cultiple Comparisons of Mean Rank Sums Extended; R-Project: Vienna, Austria, 2018. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2015. [Google Scholar]
- Gamble, A.E.; Lloyd, R.; Aiken, J.; Johannsson, O.E.; Mills, E.L. Using Zooplankton Biomass Size Spectra to Assess Ecological Change in a Well-Studied Freshwater Lake Ecosystem: Oneida Lake, New York. Can. J. Fish. Aquat. Sci. 2006, 63, 2687–2699. [Google Scholar] [CrossRef]
- Brooks, J.L.; Dodson, S.I. Predation, Body Size, and Composition of Plankton. Science 1965, 150, 28–35. [Google Scholar] [CrossRef]
- Cousyn, C.; De Meester, L.; Colbourne, J.K.; Brendonck, L.; Verschuren, D.; Volckaert, F. Rapid, Local Adaptation of Zooplankton Behavior to Changes in Predation Pressure in the Absence of Neutral Genetic Changes. Proc. Natl. Acad. Sci. USA 2001, 98, 6256–6260. [Google Scholar] [CrossRef] [Green Version]
- Sweetman, J.N.; Finney, B.P. Differential Responses of Zooplankton Populations (Bosmina Longirostris) to Fish Predation and Nutrient-Loading in an Introduced and a Natural Sockeye Salmon Nursery Lake on Kodiak Island, Alaska, USA. J. Paleolimnol. 2003, 30, 183–193. [Google Scholar] [CrossRef]
- Lotter, A.F.; Birks, H.J.B.; Hofmann, W.; Marchetto, A. Modern Diatom, Cladocera, Chironomid, and Chrysophyte Cyst Assemblages as Quantitative Indicators for the Reconstruction of Past Environmental Conditions in the Alps. I. Climate. J. Paleolimnol. 1997, 18, 395–420. [Google Scholar] [CrossRef] [Green Version]
- Manca, M.; Comoli, P. Reconstructing Long-Term Changes in Daphnia’s Body Size from Subfossil Remains in Sediments of a Small Lake in the Himalayas. J. Paleolimnol. 2004, 32, 95–107. [Google Scholar] [CrossRef]
- Atkinson, D.; Morley, S.A.; Hughes, R.N. From Cells to Colonies: At What Levels of Body Organization Does the “temperature-Size Rule” Apply? Evol. Dev. 2006, 8, 202–214. [Google Scholar] [CrossRef]
- Whiteside, M.C.; Williams, J.B.; White, C.P. Seasonal Abundance and Pattern of Chydorid, Cladocera in Mud and Vegetative Habitats. Ecology 1978, 59, 1177–1188. [Google Scholar] [CrossRef]
- Tremel, B.; Frey, S.E.L.; Yan, N.D.; Somers, K.M.; Pawson, T.W. Habitat Specificity of Littoral Chydoridae (Crustacea, Branchiopoda, Anomopoda) in Plastic Lake, Ontario, Canada. Hydrobiologia 2000, 432, 195–205. [Google Scholar] [CrossRef]
- Smirnov, N. The Chydorinae and Sayciinae (Chydoridae) of the World; Guides to the Identification of the Microinvertebrates of the Continental Waters of the World; SPB Academic: Amsterdam, The Netherlands, 1996. [Google Scholar]
- Alonso, M. Crustacea, Branchiopoda, Fauna Iberica. Vol. 7; Museo Nacional de Ciencias Naturales, Consejo Superior de Investigaciones Cientificas: Madrid, Spain, 1996. [Google Scholar] [CrossRef] [Green Version]
- Fryer, G. Evolution and Adaptive Radiation in the Chydoridae (Crustacea: Cladocera): A Study in Comparative Functional Morphology and Ecology. Philos. Trans. R. Soc. B Biol. Sci. 1968, 254, 224–387. [Google Scholar]
- Van Damme, K.; Brancelj, A.; Dumont, H.J. Adaptations to the Hyporheic in Aloninae (Crustacea: Cladocera): Allocation of Alona Protzi Hartwig, 1900 and Related Species to Phreatalona Gen. Nov. Hydrobiologia 2009, 618, 1–34. [Google Scholar] [CrossRef]
- Nevalainen, L.; Helama, S.; Luoto, T.P. Hydroclimatic Variations over the Last Millennium in Eastern Finland Disentangled by Fossil Cladocera. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 378, 13–21. [Google Scholar] [CrossRef]
- Kingsolver, J.G.; Huey, R.B. Size, Temperature, and Fitness: Three Rules. Evol. Ecol. Res. 2008, 10, 251–268. [Google Scholar]
- Pop, M. Mechanisms of the Filtering Area Adaptation in Daphnia. Hydrobiologia 1991, 225, 169–176. [Google Scholar] [CrossRef]
- Macháček, J.; Seda, J. Over-Wintering Daphnia: Uncoupling the Effects of Temperature and Food on Offspring Size and Filtering Screen Morphology in D. Galeata. J. Plankton Res. 2013, 35, 1069–1079. [Google Scholar] [CrossRef] [Green Version]
- Gliwicz, Z.M. Food Thresholds and Body Size in Cladocerans. Nature 1990, 343, 638–640. [Google Scholar] [CrossRef]
- Lampert, W. Phenotypic Plasticity of the Filter Screen in Daphnia: Adapatation to a Low-Food Environment. Limnol. Oceanogr. 1994, 39, 997–1006. [Google Scholar] [CrossRef]
- Repka, S.; Veen, A.; Vijverberg, J. Morphological Adaptations in Filtering Screens of Daphnia Galeata to Food Quantity and Food Quality. J. Plankton Res. 1999, 21, 971–989. [Google Scholar] [CrossRef] [Green Version]
- Boersma, M.; Boriss, H.; Mitchell, S.E. Maternal Effects after Sexual Reproduction in Daphnia Magna. J. Plankton Res. 2000, 22, 279–285. [Google Scholar] [CrossRef] [Green Version]
- Bergmann, C. Über die Verhältnisse der Wärmeökonomie der Thiere zu ihrer Grösse; Verlagsort: Göttingen, Germany, 1848. [Google Scholar]
- Forster, J.; Hirst, A.G.; Atkinson, D. Warming-Induced Reductions in Body Size Are Greater in Aquatic than Terrestrial Species. Proc. Natl. Acad. Sci. USA 2012, 109, 19310–19314. [Google Scholar] [CrossRef] [Green Version]
- Horne, C.R.; Hirst, A.G.; Atkinson, D. Temperature-Size Responses Match Latitudinal-Size Clines in Arthropods, Revealing Critical Differences between Aquatic and Terrestrial Species. Ecol. Lett. 2015. [Google Scholar] [CrossRef] [PubMed]
- Angilletta, M.J.; Steury, T.D.; Sears, M.W. Temperature, Growth Rate, and Body Size in Ectotherms: Fitting Pieces of a Puzzle. Integr. Comp. Biol. 2004, 44, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Karpowicz, M.; Ejsmont-Karabin, J.; Kozłowska, J.; Feniova, I.; Dzialowski, A.R. Zooplankton Community Responses to Oxygen Stress. Water 2020, 12, 706. [Google Scholar] [CrossRef] [Green Version]
- Verberk, W.C.E.P.; Bilton, D.T.; Calosi, P.; Spicer, J.I. Oxygen Supply in Aquatic Ectotherms: Partial Pressure and Solubility Together Explain Biodiversity and Size Patterns. Ecology 2011, 92, 1565–1572. [Google Scholar] [CrossRef] [Green Version]
- Verberk, W.C.E.P.; Atkinson, D. Why Polar Gigantism and Palaeozoic Gigantism Are Not Equivalent: Effects of Oxygen and Temperature on the Body Size of Ectotherms. Funct. Ecol. 2013, 27, 1275–1285. [Google Scholar] [CrossRef] [Green Version]
- Fang, X.; Stefan, G. Temperature Variability in Lake Sediments Depth. Water Resour. 1998, 34, 717–729. [Google Scholar] [CrossRef]
- Salmaso, N. Effects of Climatic Fluctuations and Vertical Mixing on the Interannual Trophic Variability of Lake Garda, Italy. Limnol. Oceanogr. 2005, 50, 553–565. [Google Scholar] [CrossRef]
- Luoto, T.P. Hydrological Change in Lakes Inferred from Midge Assemblages through Use of an Intralake Calibration Set. Ecol. Monogr. 2010, 80, 303–329. [Google Scholar] [CrossRef]
- Luoto, T.P. Intra-Lake Patterns of Aquatic Insect and Mite Remains. J. Paleolimnol. 2012, 47, 141–157. [Google Scholar] [CrossRef]
- Nykänen, M.; Kairesalo, T.; Mäkelä, S.; Huitu, E.; Ala-Opas, P.; Mannio, J. A Typology and Ecological Classification System for Finnish Lakes: Applicability of the ECOFRAME Scheme. Boreal Environ. Res. 2005, 10, 159–179. [Google Scholar]
- Decei, P. Lacuri de Munte. Drumetie Si Pescuit; Ditura Sport Turism: Bucharest, Romania, 1981. [Google Scholar]
- Frost, W.E. The Natural History of the Minnow, Phoxinus Phoxinus. J. Anim. Ecol. 1943, 12, 139–162. [Google Scholar] [CrossRef]
- Straskraba, M.; Chiar, J.; Frank, S.; Hruska, V. Contribution to the Problem of Food Competition among the Sculpin, Minnow and Brown-Trout. J. Anim. Ecol. 1966, 35, 303–311. [Google Scholar] [CrossRef]
- Museth, J.; Borgstrøm, R.; Brittain, J.E. Diet Overlap between Introduced European Minnow (Phoxinus Phoxinus) and Young Brown Trout (Salmo Trutta) in the Lake, Øvre Heimdalsvatn: A Result of Abundant Resources or Forced Niche Overlap? Hydrobiologia 2010, 642, 93–100. [Google Scholar] [CrossRef]
- Rautio, M.; Vincent, W.F. Benthic and Pelagic Food Resources for Zooplankton in Shallow High-Latitude Lakes and Ponds. Freshw. Biol. 2006, 51, 1038–1052. [Google Scholar] [CrossRef]
- Milardi, M.; Siitonen, S.; Lappalainen, J.; Liljendahl, A.; Weckström, J. The Impact of Trout Introductions on Macro- and Micro-Invertebrate Communities of Fishless Boreal Lakes. J. Paleolimnol. 2016, 55, 273–287. [Google Scholar] [CrossRef]
- Helminen, H.; Karjalainen, J.; Kurkilahti, M.; Rask, M.; Sarvala, J. Eutrophication and Fish Biodiversity in Finnish Lakes. Verh. Int. Ver. Theor. Angew. Limnol. 2000, 27, 194–199. [Google Scholar] [CrossRef]
- Moss, B.; Stephen, D.; Alvarez, C.; Becares, E.; Van De Bund, W.; Collings, S.E.; Van Donk, E.; De Eyto, E.; Feldmann, T.; Fernández-Aláez, C.; et al. The Determination of Ecological Status in Shallow Lakes—A Tested System (ECOFRAME) for Implementation of the European Water Framework Directive. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, 507–549. [Google Scholar] [CrossRef]
- Belyaeva, M.; Taylor, D.J. Cryptic Species within the Chydorus Sphaericus Species Complex (Crustacea: Cladocera) Revealed by Molecular Markers and Sexual Stage Morphology. Mol. Phylogenet. Evol. 2009, 50, 534–546. [Google Scholar] [CrossRef] [PubMed]
- Frey, D.G. On the Plurality of Chydorus Sphaericus (0. F. Müller) (Cladocera, Chydoridae), and Designation of a Neotype from Sjaelsø, Denmark. Hydrobiologia 1980, 69, 83–123. [Google Scholar] [CrossRef]
- Van Damme, K.; Eggermont, H. The Afromontane Cladocera (Crustacea: Branchiopoda) of the Rwenzori (Uganda–D. R. Congo): Taxonomy, Ecology and Biogeography. Hydrobiologia 2011, 676, 57–100. [Google Scholar] [CrossRef]
 warm lakes,
warm lakes,  Southern mountain lakes,
Southern mountain lakes,  Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
 warm lakes,
warm lakes,  Southern mountain lakes,
Southern mountain lakes,  Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).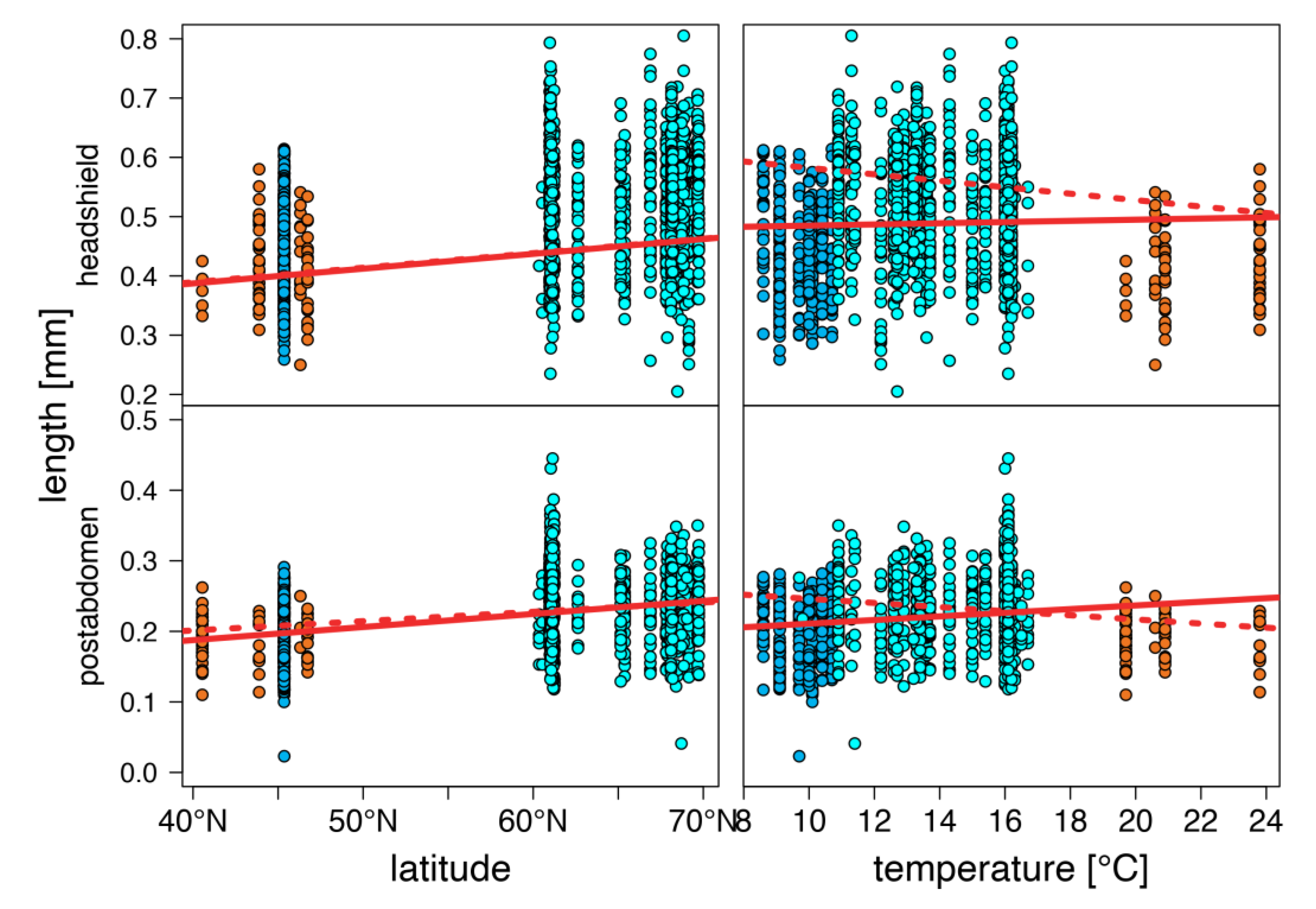
 warm lakes,
warm lakes,  Southern mountain lakes,
Southern mountain lakes,  Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
 warm lakes,
warm lakes,  Southern mountain lakes,
Southern mountain lakes,  Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).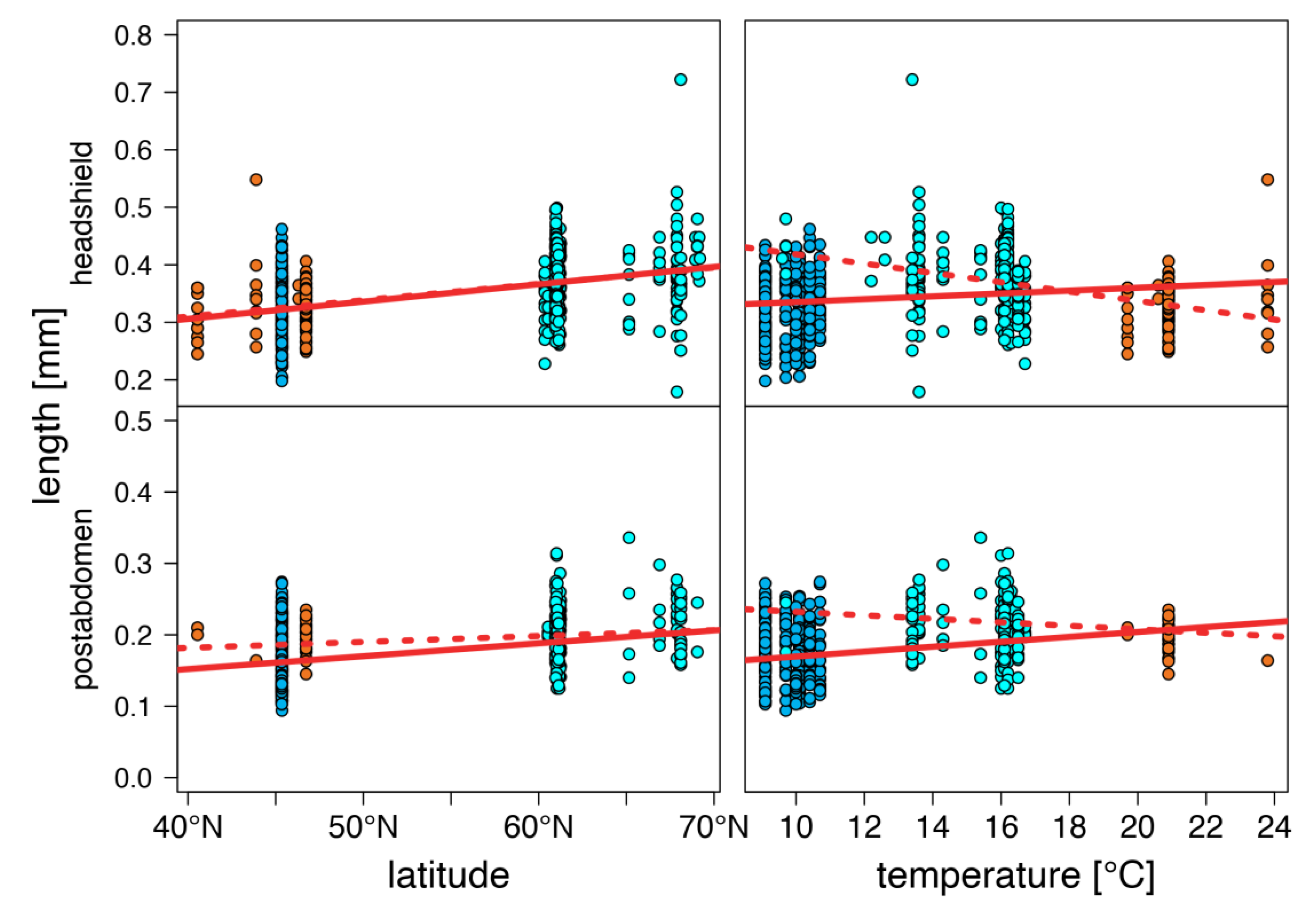
 warm lakes,
warm lakes,  Southern mountain lakes,
Southern mountain lakes,  Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
 warm lakes,
warm lakes,  Southern mountain lakes,
Southern mountain lakes,  Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).
Finnish lakes, solid line: all-lake model, dashed line: omit-SM model; Table A3).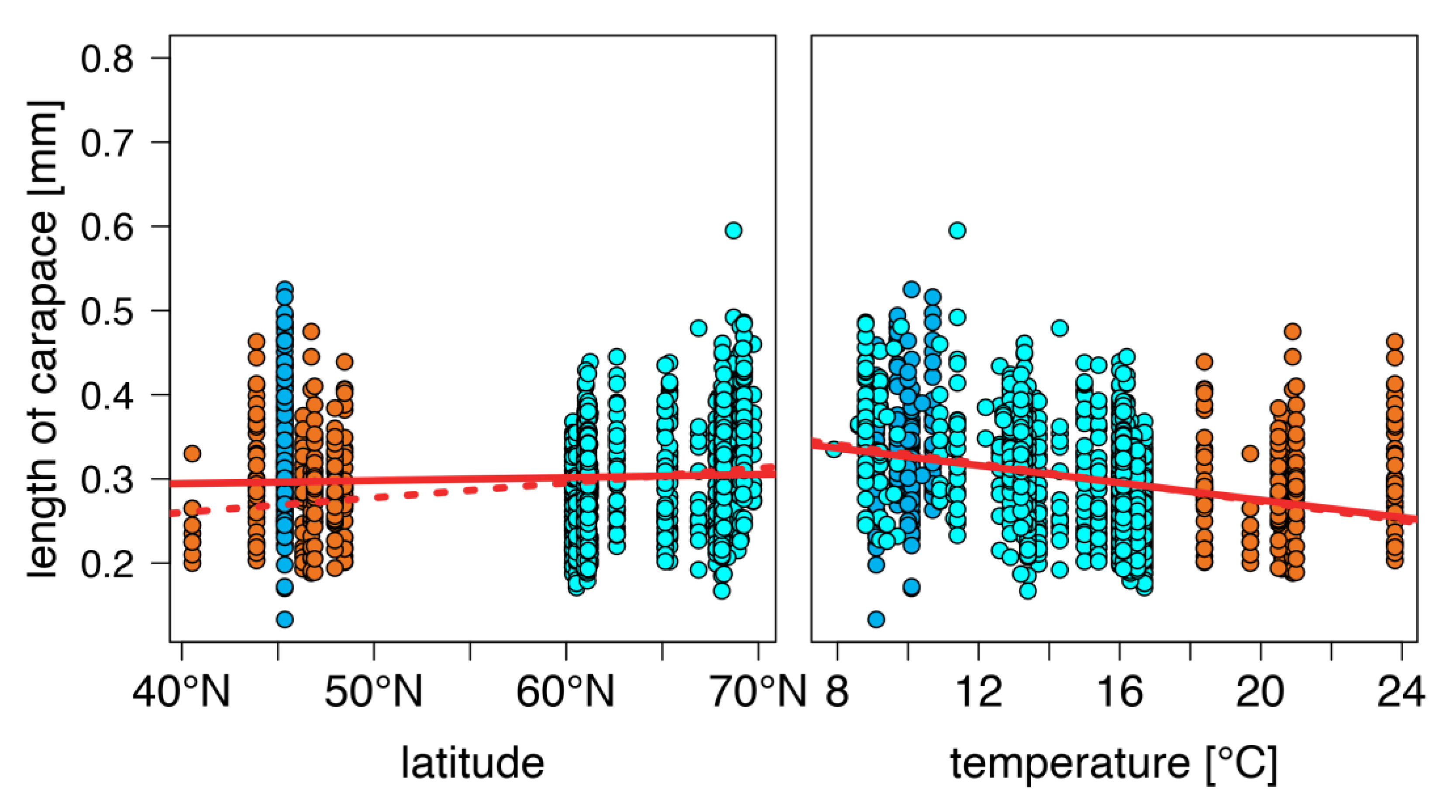
| Lakes | Latitude | Altitude (m a.s.l.) | No. of Lakes * | Depth (m) | Region | Temp. Regime |
|---|---|---|---|---|---|---|
| Finland | 60–70° N | 39–1009 | 46 | 0.5–25 | Nordic | cold |
| Hungary | 46° N | 104–384 | 5 | 3.5 | Southern | warm |
| Romania (South-Carpathians) | 45.34–45.36° N | 1909–2129 | 10 | 2.5–30 | Southern Mountain | cold |
| Croatia (Lake Vrana) | 43° N | 0 | 1 | 2.8 | Southern | warm |
| Spain (El Tobar) | 40.5° N | 1250 | 1 | 20 | Southern | warm |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korponai, J.L.; Kövér, C.; López-Blanco, C.; Gyulai, I.; Forró, L.; Katalinic, A.; Ketola, M.; Nevalainen, L.; Luoto, T.P.; Sarmaja-Korjonen, K.; et al. Effect of Temperature on the Size of Sedimentary Remains of Littoral Chydorids. Water 2020, 12, 1309. https://doi.org/10.3390/w12051309
Korponai JL, Kövér C, López-Blanco C, Gyulai I, Forró L, Katalinic A, Ketola M, Nevalainen L, Luoto TP, Sarmaja-Korjonen K, et al. Effect of Temperature on the Size of Sedimentary Remains of Littoral Chydorids. Water. 2020; 12(5):1309. https://doi.org/10.3390/w12051309
Chicago/Turabian StyleKorponai, János L., Csilla Kövér, Charo López-Blanco, István Gyulai, László Forró, Ana Katalinic, Mirva Ketola, Liisa Nevalainen, Tomi P. Luoto, Kaarina Sarmaja-Korjonen, and et al. 2020. "Effect of Temperature on the Size of Sedimentary Remains of Littoral Chydorids" Water 12, no. 5: 1309. https://doi.org/10.3390/w12051309
APA StyleKorponai, J. L., Kövér, C., López-Blanco, C., Gyulai, I., Forró, L., Katalinic, A., Ketola, M., Nevalainen, L., Luoto, T. P., Sarmaja-Korjonen, K., Magyari, E. K., Weckström, J., Urák, I., Vadkerti, E., & Buczkó, K. (2020). Effect of Temperature on the Size of Sedimentary Remains of Littoral Chydorids. Water, 12(5), 1309. https://doi.org/10.3390/w12051309








