A Field Pilot Study on Treating Groundwater Contaminated with Sulfolane Using UV/H2O2
Abstract
:1. Introduction
2. Method and Materials
2.1. Chemicals
2.2. Site Description and Contaminated Groundwater
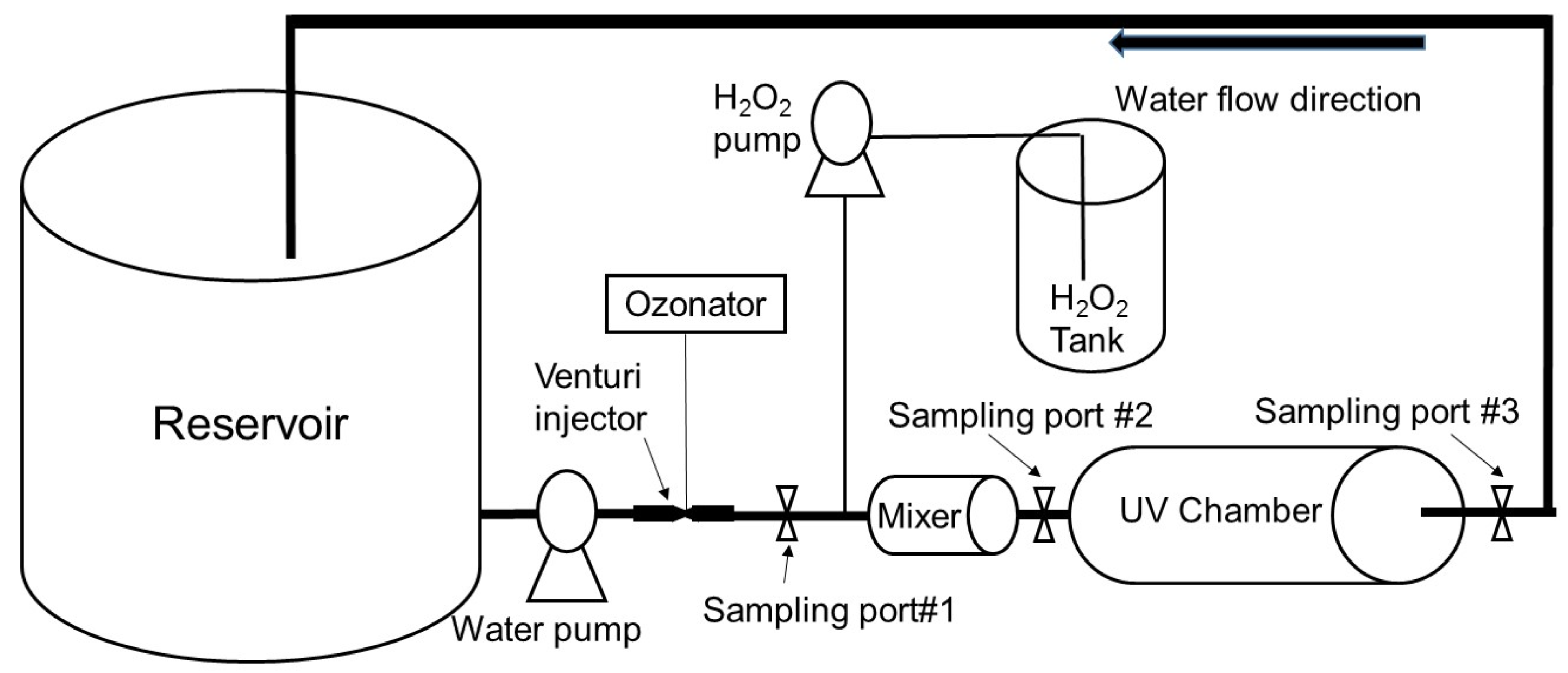
2.3. A Field Pilot-Scale UV/H2O2 System
2.4. Testing Procedure
2.5. Sulfolane and DOC Analysis
2.6. H2O2 Measurement
3. Results and Discussions
3.1. Degradation of Sulfolane in a UV/H2O2 Pilot System
3.2. Iron in the Groundwater
3.3. Influent Sulfolane Concentration
3.4. Water Flow Rate
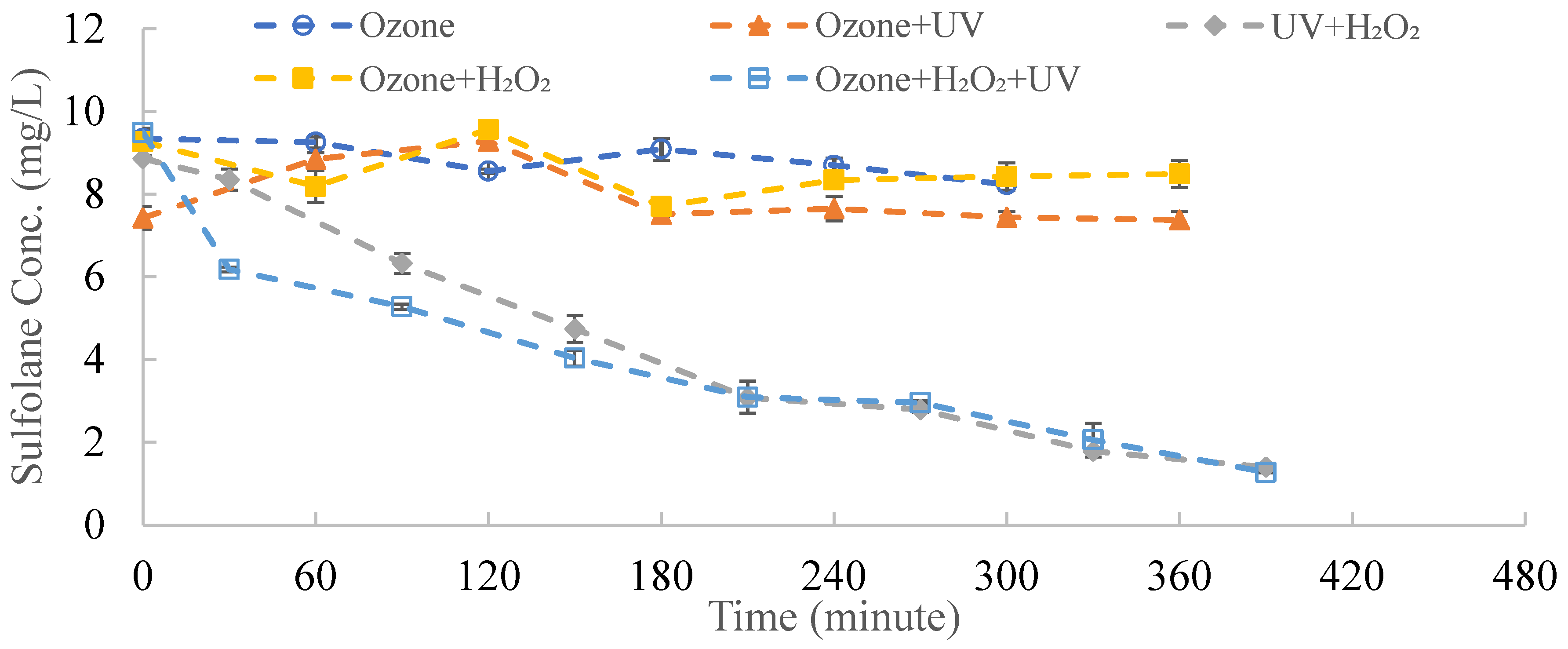
3.5. Introduction of Ozone into a UV/H2O2 System
3.6. Optimization of H2O2
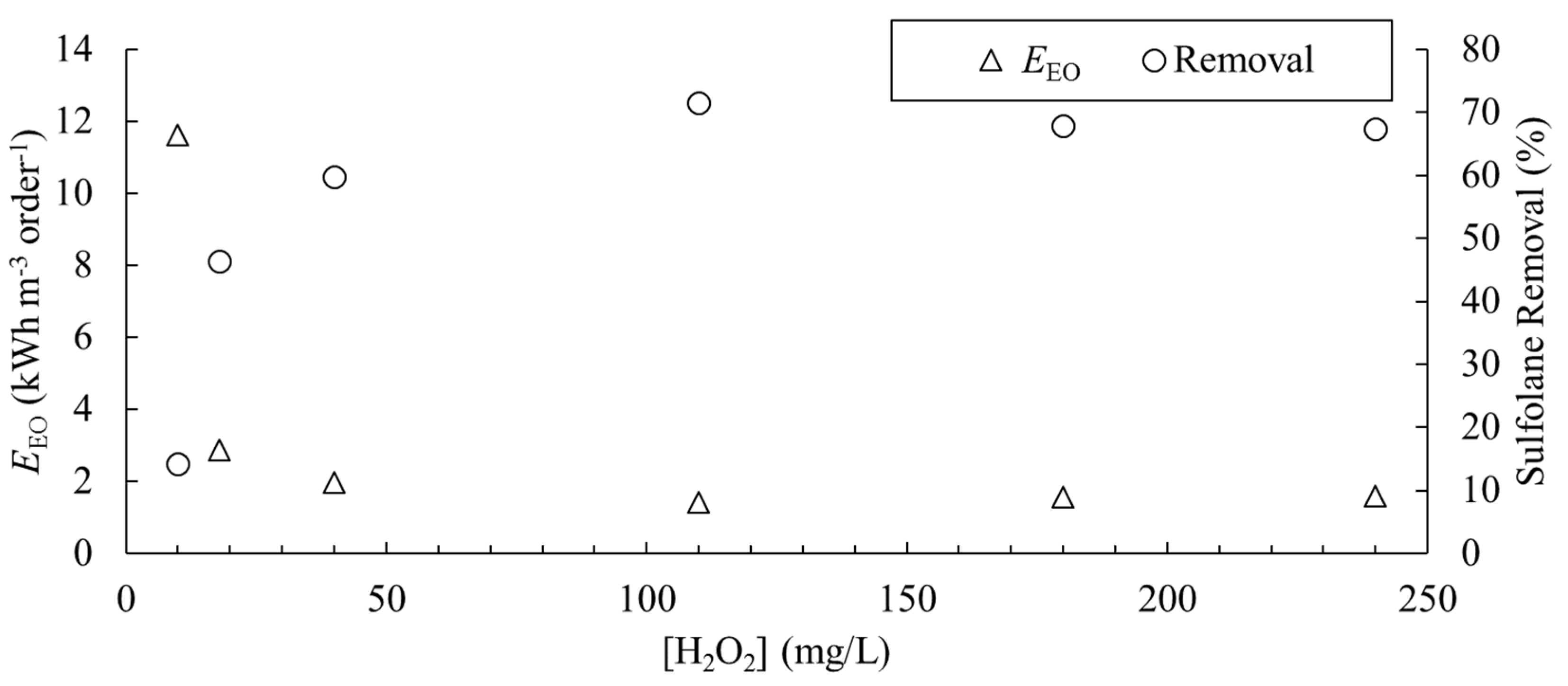
3.6.1. The Effect of H2O2 Concentration on Sulfolane Degradation
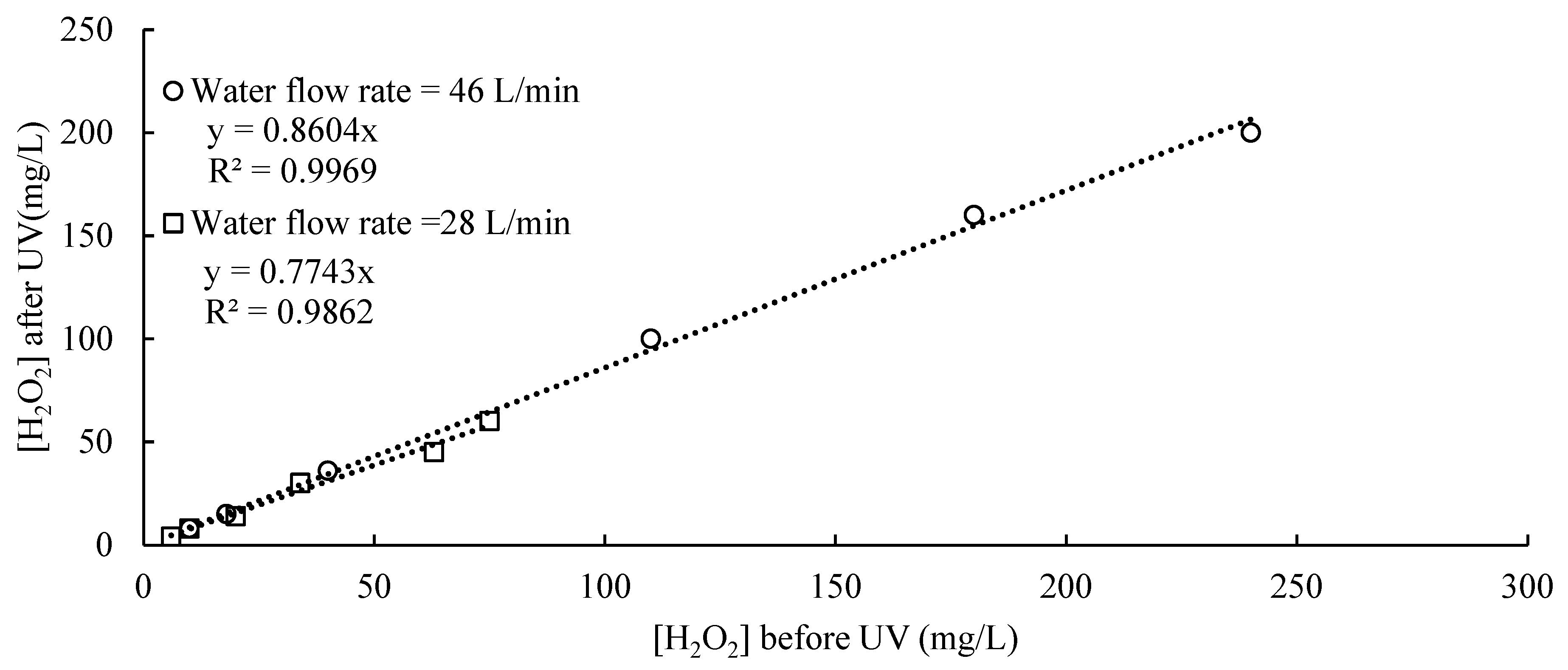
3.6.2. Decomposition of Hydrogen Peroxide
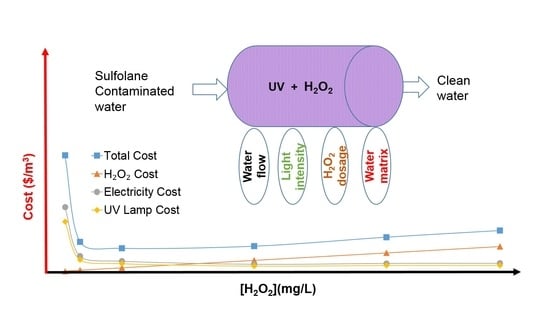
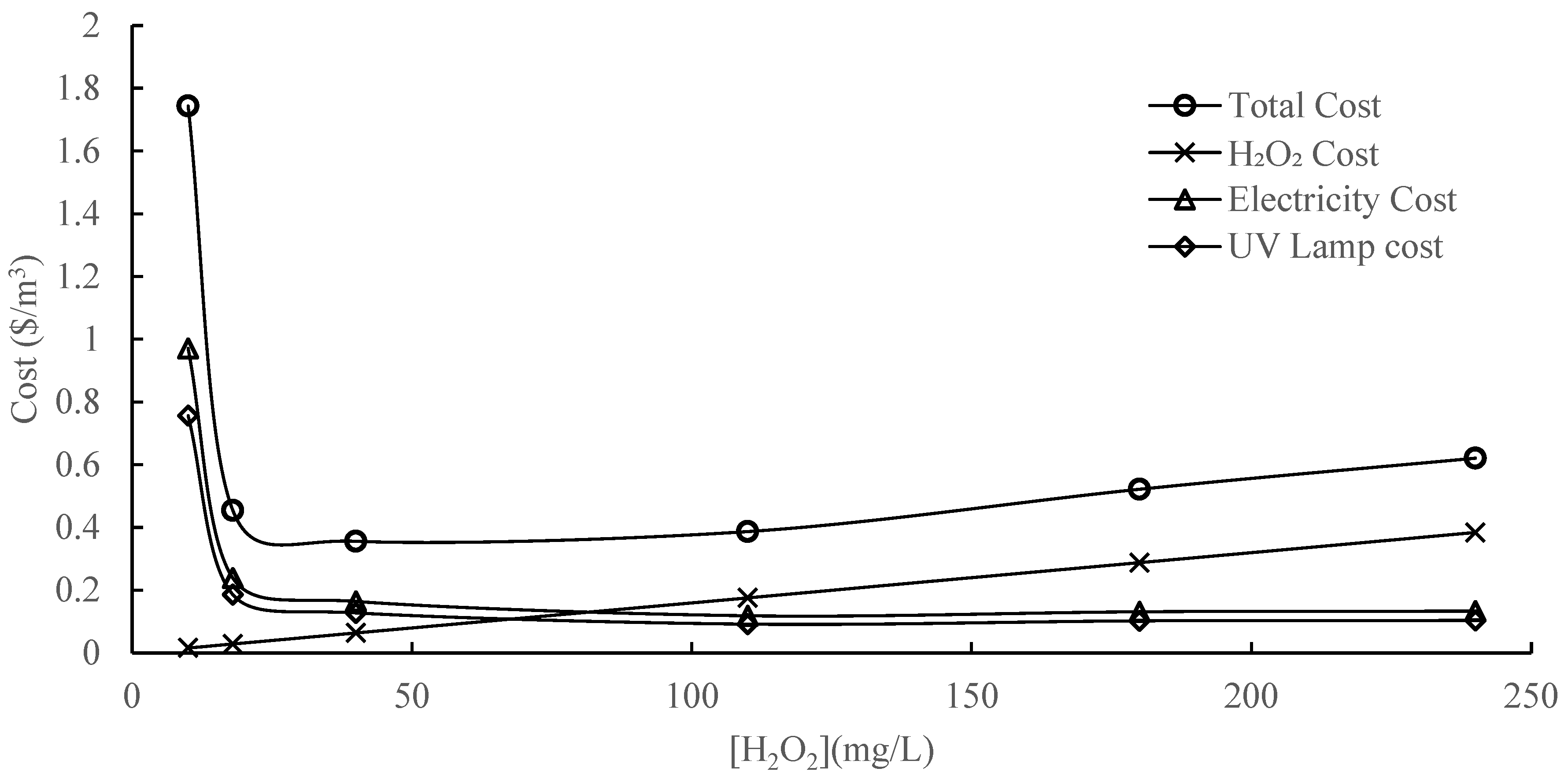
3.6.3. Operational Cost Model
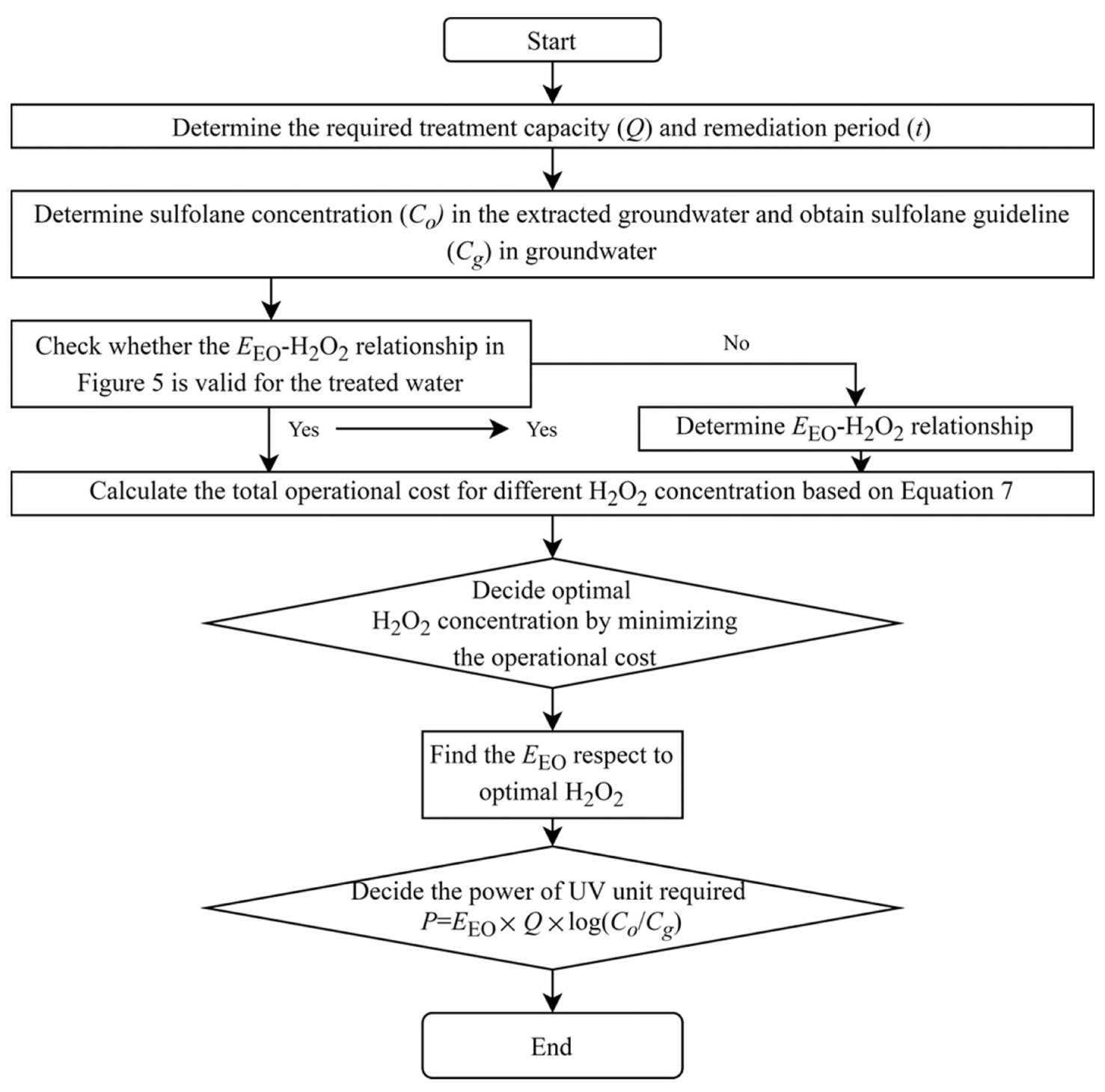
3.7. Selection of the Right Size of UV System to Treat Sulfolane in Groundwater
4. Conclusions
- The field pilot test confirmed that a pilot UV/H2O2 system can successfully treat different sulfolane-contaminated groundwaters. The lowest EEO obtained in this field pilot study was 1.4 kWh m−3 order−1.
- The sulfolane degradation rate was not significantly impacted by the initial concentration.
- The addition of O3 at a rate of 10 g/h/m3 did not enhance sulfolane degradation by UV/H2O2.
- Consumption of hydrogen peroxide in the pilot UV/H2O2 system followed first-order kinetics.
- An operational cost model is developed. The optimal H2O2 concentration in this study was determined to be 40 mg/L, leading to an operational cost of 0.35 CAD/m3.
- A systematic procedure is presented to select the right size of UV unit.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Della Monica, M.; Jannelli, L.; Lamanna, U. Physicochemical properties of sulfolane. J. Phys. Chem. 1968, 72, 1068–1071. [Google Scholar] [CrossRef]
- Stewart, O.; Minnear, L. Sulfolane Technical Assistance and Evaluation Report; Oasis Environmental: Anchorage, AK, USA, 2010. [Google Scholar]
- Zhu, Z.H.; Sun, M.L.; Li, Z.S.; Yang, Z.C.; Zhang, T.B.; Heng, Z.C.; Xiao, B.L.; Li, Q.Y.; Peng, Q.Y.; Dong, Y.H. An investigation of the maximum allowable concentration of sulfolane in surface water. Hua Xi Yi Ke Da Xue Xue Bao 1987, 18, 376–380. [Google Scholar] [PubMed]
- Gordon, C.J.; Dyer, R.S.; Long, M.D.; Fehlner, K.S. Effect of sulfolane on behavioral and autonomic thermoregulation in the rat. J. Toxicol. Environ. Health Part A 1985, 16, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.M.; Wahba, M.; Yu, L.; Achari, G.; Habibi, H.R. Health Impact Assessment of Sulfolane on Embryonic Development of Zebrafish (Danio rerio). Toxics 2019, 7, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, L.; Mehrabani-Zeinabad, M.; Achari, G.; Langford, C.H. Application of UV based advanced oxidation to treat sulfolane in an aqueous medium. Chemosphere 2016, 160, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Mehrabani-Zeinabad, M.; Yu, L.; Achari, G.; Langford, C.H. Mineralisation of sulfolane by UV/O3/H2O2 in a tubular reactor. J. Environ. Eng. Sci. 2016, 11, 44–51. [Google Scholar] [CrossRef]
- Izadifard, M.; Achari, G.; Langford, C.H. Degradation of sulfolane using activated persulfate with UV and UV-Ozone. Water Res. 2017, 125, 325–331. [Google Scholar] [CrossRef]
- Brandão, M.; Yu, L.; Garcia, C.; Achari, G. Advanced Oxidation Based Treatment of Soil Wash Water Contaminated with Sulfolane. Water 2019, 11, 2152. [Google Scholar] [CrossRef] [Green Version]
- Khan, M.F.; Yu, L.; Achari, G.; Tay, J.H. Degradation of sulfolane in aqueous media by integrating activated sludge and advanced oxidation process. Chemosphere 2019, 222, 1–8. [Google Scholar] [CrossRef]
- Chu, X.; Xiao, Y.; Hu, J.; Quek, E.; Xie, R.; Pang, T.; Xing, Y. Pilot-scale UV/H2O2 study for emerging organic contaminants decomposition. Rev. Environ. Health 2016, 31, 71–74. [Google Scholar] [CrossRef]
- De La Cruz, N.; Esquius, L.; Grandjean, D.; Magnet, A.; Tungler, A.; De Alencastro, L.; Pulgarin, C. Degradation of emergent contaminants by UV, UV/H2O2 and neutral photo-Fenton at pilot scale in a domestic wastewater treatment plant. Water Res. 2013, 47, 5836–5845. [Google Scholar] [CrossRef]
- Miklos, D.; Hartl, R.; Michel, P.; Linden, K.; Drewes, J.E.; Hübner, U. UV/H2O2 process stability and pilot-scale validation for trace organic chemical removal from wastewater treatment plant effluents. Water Res. 2018, 136, 169–179. [Google Scholar] [CrossRef]
- Sarathy, S.R.; Stefan, M.I.; Royce, A.; Mohseni, M. Pilot-scale UV/H2O2 advanced oxidation process for surface water treatment and downstream biological treatment: Effects on natural organic matter characteristics and DBP formation potential. Environ. Technol. 2011, 32, 1709–1718. [Google Scholar] [CrossRef] [PubMed]
- Hirvonen, A.; Tuhkanen, T.; Ettala, M.; Korhonen, S.; Kalliokoski, P. Evaluation of a Field-Scale UV/H2O2-Oxidation System for the Purification of Groundwater Contaminated with PCE. Environ. Technol. 1998, 19, 821–828. [Google Scholar] [CrossRef]
- Bolton, J.R.; Bircher, K.G.; Tumas, W.; Tolman, C.A. Figures-of-merit for the technical development and application of advanced oxidation technologies for both electric- and solar-driven systems (IUPAC Technical Report). Pure Appl. Chem. 2001, 73, 627–637. [Google Scholar] [CrossRef]
- Hansen, K.M.S.; Andersen, H.R. Energy Effectiveness of Direct UV and UV/H2O2 Treatment of Estrogenic Chemicals in Biologically Treated Sewage. Available online: https://www.hindawi.com/journals/ijp/2012/270320/ (accessed on 8 January 2020).
- Lekkerkerker-Teunissen, K.; Knol, A.H.; Derks, J.G.; Heringa, M.B.; Houtman, C.J.; Hofman-Caris, C.H.M.; Beerendonk, E.F.; Reus, A.; Verberk, J.Q.J.C.; Van Dijk, J. Pilot Plant Results with Three Different Types of UV Lamps for Advanced Oxidation. Ozone Sci. Eng. 2013, 35, 38–48. [Google Scholar] [CrossRef]
- Sutherland, J.; Adams, C.; Kekobad, J. Treatment of MTBE by air stripping, carbon adsorption, and advanced oxidation: Technical and economic comparison for five groundwaters. Water Res. 2004, 38, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Santoro, D.; Raisee, M.; Moghaddami, M.; Ducoste, J.J.; Sasges, M.; Liberti, L.; Notarnicola, M. Modeling Hydroxyl Radical Distribution and Trialkyl Phosphates Oxidation in UV−H2O2 Photoreactors Using Computational Fluid Dynamics. Environ. Sci. Technol. 2010, 44, 6233–6241. [Google Scholar] [CrossRef]
- Lee, Y.; Gerrity, D.; Lee, M.; Gamage, S.; Pisarenko, A.; Trenholm, R.A.; Canonica, S.; Snyder, S.A.; Von Gunten, U. Organic Contaminant Abatement in Reclaimed Water by UV/H2O2 and a Combined Process Consisting of O3/H2O2 Followed by UV/H2O2: Prediction of Abatement Efficiency, Energy Consumption, and Byproduct Formation. Environ. Sci. Technol. 2016, 50, 3809–3819. [Google Scholar] [CrossRef]
- Ijpelaar, G.F.; Harmsen, D.J.; Beerendonk, E.F.; Van Leerdam, R.C.; Metz, D.H.; Knol, A.H.; Fulmer, A.; Krijnen, S. Comparison of Low Pressure and Medium Pressure UV Lamps for UV/H2O2 Treatment of Natural Waters Containing Micro Pollutants. Ozone Sci. Eng. 2010, 32, 329–337. [Google Scholar] [CrossRef]
- Li, K.; Hokanson, D.R.; Crittenden, J.C.; Trussell, R.R.; Minakata, D. Evaluating UV/H2O2 processes for methyl tert-butyl ether and tertiary butyl alcohol removal: Effect of pretreatment options and light sources. Water Res. 2008, 42, 5045–5053. [Google Scholar] [CrossRef] [PubMed]
- Pignatello, J.J.; Oliveros, E.; Mackay, A. Advanced Oxidation Processes for Organic Contaminant Destruction Based on the Fenton Reaction and Related Chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar] [CrossRef]
- Yu, L.; Achari, G.; Langford, C.H.; Keir, I. A feasibility study on sulfolane degradation in groundwater using neutral fenton catalysts. In Proceedings of the CSCE 2016, London, ON, Canada, 1–4 June 2016. [Google Scholar]
- Do, S.-H.; Batchelor, B.; Lee, H.-K.; Kong, S.-H. Hydrogen peroxide decomposition on manganese oxide (pyrolusite): Kinetics, intermediates, and mechanism. Chemosphere 2009, 75, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Hem, J.D. Stability Field Diagrams as Aids in Iron Chemistry Studies. J. Am. Water Work. Assoc. 1961, 53, 211–232. [Google Scholar] [CrossRef]
- Peng, J.; Qiu, Y.; Gehr, R. Characterization of Permanent Fouling on the Surfaces of UV Lamps Used for Wastewater Disinfection. Water Environ. Res. 2005, 77, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Wait, I.; Johnston, C.T.; Blatchleyiii, E.; Iii, E.R.B. The influence of oxidation reduction potential and water treatment processes on quartz lamp sleeve fouling in ultraviolet disinfection reactors. Water Res. 2007, 41, 2427–2436. [Google Scholar] [CrossRef]
- Saien, J.; Moradi, V.; Soleymani, A.R. Investigation of a jet mixing photo-reactor device for rapid dye discoloration and aromatic degradation via UV/H2O2 process. Chem. Eng. J. 2012, 183, 135–140. [Google Scholar] [CrossRef]
- Tabrizi, G.B.; Mehrvar, M. Pilot-plant study for the photochemical treatment of aqueous linear alkylbenzene sulfonate. Sep. Purif. Technol. 2006, 49, 115–121. [Google Scholar] [CrossRef]
- Daneshvar, N.; Behnajady, M.A.; Asghar, Y.Z. Photooxidative degradation of 4-nitrophenol (4-NP) in UV/H2O2 process: Influence of operational parameters and reaction mechanism. J. Hazard. Mater. 2007, 139, 275–279. [Google Scholar] [CrossRef]


| Parameter | Units | Water I | Water II | Water III |
|---|---|---|---|---|
| Hardness (CaCO3) | mg/L | 653 | 3.6 | 310 |
| Total Dissolved Solids | mg/L | 671 | 554 | 370 |
| Conductivity | S/cm | 1.13 | 0.992 | 0.68 |
| pH | 7.75 | 9.07 | 8.52 | |
| Alkalinity (Total as CaCO3) | mg/L | 415 | 450 | 200 |
| Bicarbonate (HCO3) | mg/L | 506 | 457 | 230 |
| Sulphate (SO4) | mg/L | 113 | 21.1 | 77 |
| Chloride (Cl) | mg/L | 56.7 | 22.5 | 61 |
| Nitrate (NO3) | mg/L | 0.07 | <0.044 | 0.49 |
| Nitrite (NO2) | mg/L | <0.033 | <0.033 | 0.17 |
| Calcium (Ca) | mg/L | 177 | 1.1 | 48 |
| Magnesium (Mg) | mg/L | 51.4 | 0.22 | 47 |
| Potassium (K) | mg/L | 5.62 | 0.56 | 4.8 |
| Sodium (Na) | mg/L | 17.0 | 239 | 14 |
| Iron (Fe) | mg/L | 0.355 | <0.06 | 0.084 |
| Manganese (Mn) | mg/L | 0.678 | <0.004 | 0.018 |
| Influent Sulfolane Conc. (μg/L) | Effluent Sulfolane Conc. (μg/L) | Removal Percentage |
|---|---|---|
| 260 | 200 | 23 |
| 252 | 173 | 31 |
| 238 | 184 | 23 |
| 200 | 159 | 21 |
| 188 | 126 | 33 |
| 136 | 104 | 24 |
| 134 | 98 | 27 |
| 120 | 86 | 28 |
| 108 | 71 | 34 |
| 82 | 63 | 23 |
| 60 | 36 | 40 |
| 42 | 28 | 33 |
| [Sulfolane] μg/L | [H2O2] mg/L | Flow Rate (L/min) | Removal Percentage | Calculated EEO (kWh m−3 order−1) |
|---|---|---|---|---|
| 200 | 40 | 46 | 36 | 4.0 |
| 200 | 40 | 15.1 | 79 | 3.6 |
| 200 | 80 | 46 | 50 | 2.6 |
| 200 | 80 | 15.1 | 89 | 2.5 |
| Parameters | Value |
|---|---|
| Unit price of H2O2 (50% purity) | 0.8 CAD/kg |
| Unit price of electricity | 0.08 CAD/kWh |
| Lifetime of UV lamps | 12,000 h |
| Price of UV lamp | 200 CAD each |
| Number of UV lamp | 8 |
| Number of log reduction | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, L.; Iranmanesh, S.; Keir, I.; Achari, G. A Field Pilot Study on Treating Groundwater Contaminated with Sulfolane Using UV/H2O2. Water 2020, 12, 1200. https://doi.org/10.3390/w12041200
Yu L, Iranmanesh S, Keir I, Achari G. A Field Pilot Study on Treating Groundwater Contaminated with Sulfolane Using UV/H2O2. Water. 2020; 12(4):1200. https://doi.org/10.3390/w12041200
Chicago/Turabian StyleYu, Linlong, Sobhan Iranmanesh, Ian Keir, and Gopal Achari. 2020. "A Field Pilot Study on Treating Groundwater Contaminated with Sulfolane Using UV/H2O2" Water 12, no. 4: 1200. https://doi.org/10.3390/w12041200
APA StyleYu, L., Iranmanesh, S., Keir, I., & Achari, G. (2020). A Field Pilot Study on Treating Groundwater Contaminated with Sulfolane Using UV/H2O2. Water, 12(4), 1200. https://doi.org/10.3390/w12041200