Trajectories of Sediment-Water Interactions in Reservoirs as a Result of Temperature and Oxygen Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Sediment and Water Sampling
2.3. Sediment Incubation
2.4. Water Analyses
2.5. Sediment Characterization
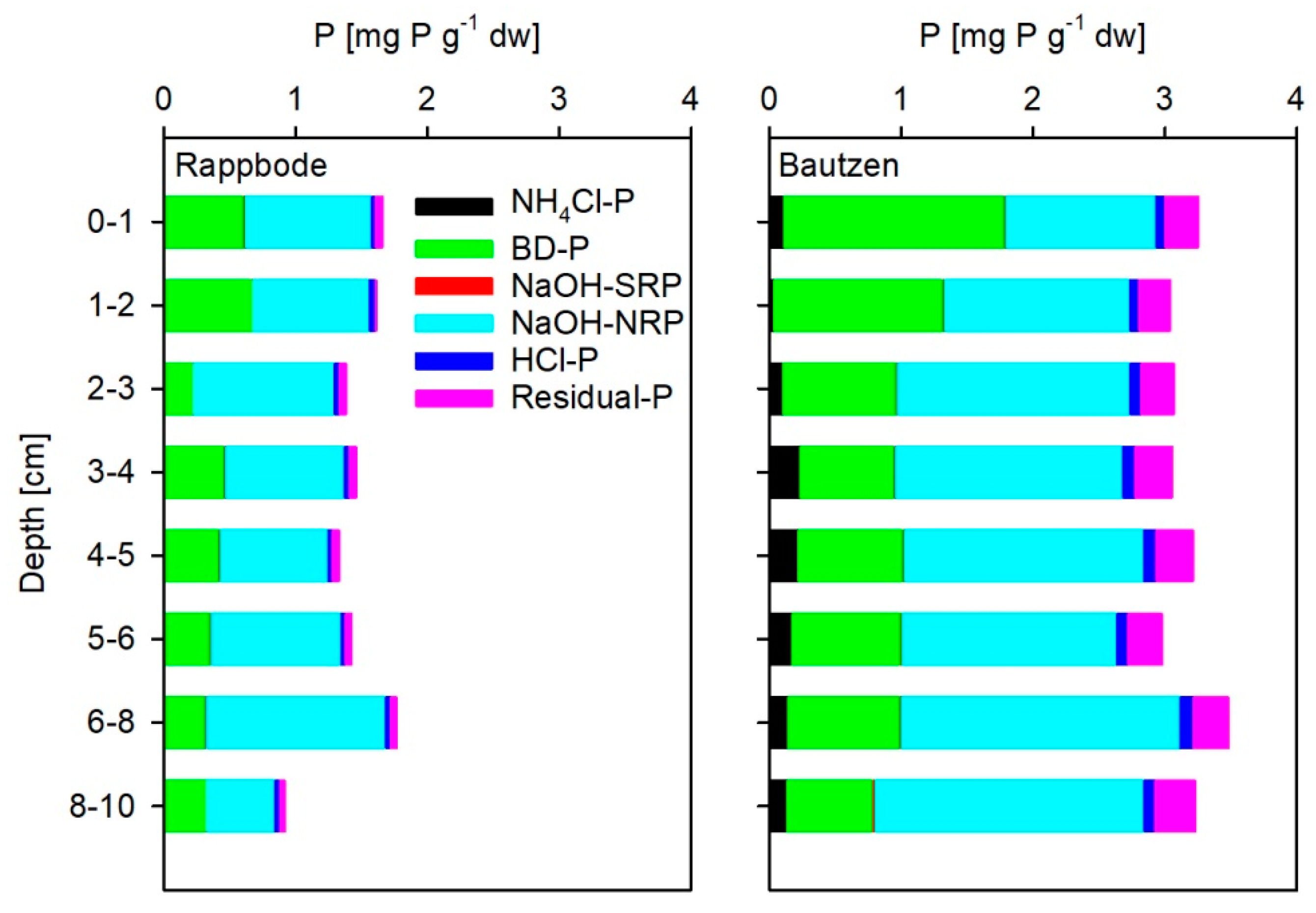
2.6. Phosphorus Fractionation
- Easily available, dissolved porewater P (NH4Cl-P) extracted by 1 M NH4Cl (ammonium chloride).
- Fe bound, redox-sensitive (BD-P) P extracted by 0.11 M NaHCO3 (sodium hydrogen carbonate)/0.11 M Na2S2O4 (sodium dithionite).
- Al-associated (NaOH-P) extracted by 1 M NaOH (sodium hydroxide), this P fraction is subdivided into NaOH-SRP (OH-exchangeable P at high pH) and NaOH-NRP (organic bound P).
- P bound by carbonates and apatite (HCl-P) extracted by 0.5 M HCl (hydrochloric acid).
- Refractory P (residual-P) determined by digestion of the remaining sediment with persulphate (K2S2O8) and sulphiric acid (H2SO4) at 110 °C for 30 min.
2.7. Calculation of Solute Release Rates
2.8. Statistics
3. Results
3.1. Water, Sediment and Porewater Characteristics
3.2. Sediment Incubation Conditions
3.3. Solutes Fluxes
3.4. Phosphorus Fractionation
4. Discussion
4.1. Synergistic Temperature and Oxygen Condition Interaction
4.2. Magnitude of Solutes Fluxes
| Lake | Trophic State | Incubation Conditions | Flux (mmol m−2 day−1) | Reference | ||
|---|---|---|---|---|---|---|
| Temp. (°C) | Oxygen Condition | SRP | NH4+-N | |||
| Lake Arreso, Denmark | Eutrophic | 18 °C | Oxic | 0.13–0.39 | ND | [54] |
| Spring Lake, USA | Eutrophic | 17–25 | Oxic | −0.07–0.01 | ND | [41] |
| 17–25 | Anoxic | 0.29–0.86 | ND | |||
| Grand Lake, USA | Oligotrophic | <16 °C | Oxic | −0.002 | ND | [55] |
| >16 °C | Oxic | 0.02 | ND | |||
| Chubb Lake, Canada | Oligotrophic | 11 °C | Anoxic | 0.07 | ND | [56] |
| Onodonga Lake, USA | Hyper-eutrophic | 4 °C | Oxic | 0.04 | ND | [1] |
| 4 °C | Anoxic | 0.39 | ND | |||
| 8 °C | Oxic | 0.20–0.21 | ND | |||
| 8 °C | Anoxic | 0.62–0.71 | ND | |||
| Lake Frances, USA | Hyper-eutrophic | 22 °C | Oxic | 0.12–0.31 | ND | [57] |
| 22 °C | Anoxic | 0.50–0.73 | ND | |||
| Loch Leven Lake, Scotland | Eutrophic | 17 °C | Oxic | 0.39 | ND | [58] |
| Acton Lake (reservoir) Ohio, USA | Eutrophic | 25 °C | Oxic | 0.03 | 0.95 | [59] |
| 12 °C | Hypoxic | 0.29 | 1.39 | |||
| Lake Arreskov | Eutrophic | 16 °C | Oxic | ~0.97 | ND | [11] |
| Lake Kvind | Eutrophic | 18 °C | Oxic | 1.58 | ND | |
| Lake Søbygård | Eutrophic | 20 °C | Oxic | 5.30 | ND | |
| Lake Væng | Eutrophic | <15 °C | Oxic | 1.13 | ND | |
| Lake Tahoe, California/Nevada, USA | Ultra-oligotrophic | 5 °C | Oxic | 0 | 0 | [60] |
| Anoxic | 0.009 | 0.04 | ||||
| Upper San Leandro Reservoir, Oakland | Meso/eutrophic | 12–16 °C | Anoxic | ND | 0.64 ± 0.4 | [61] |
| Lake Mathews, Riverside Reservoir | Oligo/mesotrophic | 12–16 °C | Anoxic | ND | 0.21 ± 0.07 | |
| Walker Lake | 12–16 °C | Anoxic | ND | 1.36 | ||
| Lafayette Reservoir, Lafayette | Hypereutrophic | 12–16 °C | Anoxic | ND | 1.29 ± 0.4 | |
| Lake Bard (Reservoir), Ventura | Mesotrophic | 12–16 °C | Anoxic | ND | 0.54 | |
| Upper Crystal Springs Reservoirs, San Francisco | Mesotrophic | 12–16 °C | Anoxic | ND | 0.11 ±0.05 | |
| Lower Crystal Springs Reservoirs, San Francisco | Oligo/mesotrophic | 12–16 °C | Anoxic | ND | 0.29 ± 0.05 | |
| San Andreas Reservoir, San Francisco | Oligo/mesotrophic | 12–16 °C | Anoxic | ND | 0.14 ± 0.04 | |
| San Antonio Reservoir, San Francisco East Bay | Meso/eutrophic | 12–16 °C | Anoxic | ND | 0.31 ± 0.05 | |
| Lake Elsinore, Riverside | Hypereutrophic | 12–16 °C | Anoxic | ND | 6.43 ± 1.93 | |
| San Vicente Reservoir, San Diego | Hypereutrophic | 12–16 °C | Anoxic | ND | 2.36 ± 1.71 | |
| Lake Groß-Glienicke | Eutrophic lake | 16 °C | Oxic | 1.22 | ND | [62] |
| Anoxic | 2.77 | 0.08 | ||||
| Lake Arendsee | Eutrophic | 10 °C | Oxic | 0.08 | [63] | |
| Bautzen reservoir | Eutrophic | 6 °C | Oxic | 0.02 ± 0.01 | 0.71 ± 0.8 | This study |
| 6 °C | Anoxic | 0.04 ± 0.02 | 0.99 ± 0.36 | |||
| 20 °C | Oxic | 0.01 ± 0.003 | 2.11 ± 2.24 | |||
| 20 °C | Anoxic | 0.16 ± 0.03 | 5.63 ± 1.50 | |||
| Rappbode reservoir | Oligotrophic | 6 °C | Oxic | −0.0007 ± 0.0009 | 1.11 ± 0.43 | |
| 6 °C | Anoxic | 0.001 ± 0.002 | −0.02 ± 0.08 | |||
| 20 °C | Oxic | 0.0021 ± 0.0025 | 0.32 ± 0.32 | |||
| 20 °C | Anoxic | 0.0128 ± 0.0095 | 0.94 ± 0.35 | |||
4.3. Implications
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Penn, M.R.; Auer, M.T.; Doerr, S.M.; Driscoll, C.T.; Brooks, C.M.; Effler, S.W. Seasonality in phosphorus release rates from the sediments of a hypereutrophic lake under a matrix of pH and redox conditions. Can. J. Fish. Aquat. Sci. 2000, 57, 1033–1041. [Google Scholar] [CrossRef]
- Hupfer, M.; Lewandowski, J. Oxygen controls the phosphorus release from lake sediments—A long-lasting paradigm in limnology. Int. Rev. Hydrobiol. 2008, 93, 415–432. [Google Scholar] [CrossRef]
- Dadi, T.; Friese, K.; Wendt-Potthoff, K.; Koschorreck, M. Benthic dissolved organic carbon fluxes in a drinking water reservoir. Limnol. Oceanogr. 2016, 445–459. [Google Scholar] [CrossRef] [Green Version]
- Livingstone, D.M.; Imboden, D.M. The prediction of hypolimnetic oxygen profiles: A plea for a deductive approach. Can. J. Fish. Aquat. Sci. 1996, 53, 924–932. [Google Scholar] [CrossRef]
- Seitzinger, S.P. Denitrification in freshwater and coastal marine ecosystems: Ecological and geochemical significance. Limnol. Oceanogr. 1988, 33, 702–724. [Google Scholar] [CrossRef]
- Søndergaard, M.; Jensen, J.P.; Jeppesen, E. Internal phosphorus loading in shallow Danish lakes. Hydrobiologia 1999, 408, 145–152. [Google Scholar] [CrossRef]
- Gudasz, C.; Bastviken, D.; Steger, K.; Premke, K.; Sobek, S.; Tranvik, L.J. Temperature-controlled organic carbon mineralization in lake sediments. Nature 2010, 466, 478–481. [Google Scholar] [CrossRef]
- Kerimoglu, O.; Rinke, K. Stratification dynamics in a shallow reservoir under different hydro-meteorological scenarios and operational strategies. Water Resour. Res. 2013, 49, 7518–7527. [Google Scholar] [CrossRef]
- Lepori, F.; Roberts, J.J. Past and future warming of a deep European lake (Lake Lugano): What are the climatic drivers? J. Great Lakes Res. 2015, 41, 973–981. [Google Scholar] [CrossRef]
- Dokulil, M.T.; Jagsch, A.; George, G.D.; Anneville, O.; Jankowski, T.; Wahl, B.; Lenhart, B.; Blenckner, T.; Teubner, K. Twenty years of spatially coherent deepwater warming in lakes across Europe related to the North Atlantic Oscillation. Limnol. Oceanogr. 2006, 51, 2787–2793. [Google Scholar] [CrossRef] [Green Version]
- Jensen, H.S.; Andersen, F.O. Importance of temperature, nitrate, and pH for phosphate release from aerobic sediments of four shallow, eutrophic lakes. Limnol. Oceanogr. 1992, 37, 577–589. [Google Scholar] [CrossRef]
- Rigosi, A.; Carey, C.C.; Ibelings, B.W.; Brookes, J.D. The interaction between climate warming and eutrophication to promote cyanobacteria is dependent on trophic state and varies among taxa. Limnol. Oceanogr. 2014, 59, 99–114. [Google Scholar] [CrossRef] [Green Version]
- Kosten, S.; Huszar, V.L.M.; Bécares, E.; Costa, L.S.; van Donk, E.; Hansson, L.-A.; Jeppesen, E.; Kruk, C.; Lacerot, G.; Mazzeo, N.; et al. Warmer climates boost cyanobacterial dominance in shallow lakes. Glob. Chang. Biol. 2012, 18, 118–126. [Google Scholar] [CrossRef]
- Alsterberg, C.; Hulth, S.; Sundbäck, K. Response of a shallow-water sediment system to warming. Limnol. Oceanogr. 2011, 56, 2147–2160. [Google Scholar] [CrossRef]
- Boehrer, B.; Schultze, M. Stratification of lakes. Rev. Geophys. 2008, 46, RG2005. [Google Scholar] [CrossRef] [Green Version]
- North, R.P.; North, R.L.; Livingstone, D.M.; Köster, O.; Kipfer, R. Long-term changes in hypoxia and soluble reactive phosphorus in the hypolimnion of a large temperate lake: Consequences of a climate regime shift. Glob. Chang. Biol. 2014, 20, 811–823. [Google Scholar] [CrossRef]
- Jean-Philippe, J.; Fabien, A.; Benjamin, A.; Jean-Marcel, D.; Pierre, S.; Michel, M.; Marie-Elodie, P. Inherited hypoxia: A new challenge for reoligotrophicated lakes under global warming. Glob. Biogeochem. Cycles 2014, 28, 1413–1423. [Google Scholar] [CrossRef]
- Fang, X.; Stefan, H.G. Simulations of climate effects on water temperature, dissolved oxygen, and ice and snow covers in lakes of the contiguous U.S. under past and future climate scenarios. Limnol. Oceanogr. 2009, 54, 2359–2370. [Google Scholar] [CrossRef]
- Rolighed, J.; Jeppesen, E.; Sondergaard, M.; Bjerring, R.; Janse, J.H.; Mooij, W.M.; Trolle, D. Climate Change Will Make Recovery from Eutrophication More Difficult in Shallow Danish Lake Sobygaard. Water 2016, 8, 459. [Google Scholar] [CrossRef] [Green Version]
- Einsele, W. Ueber die Beziehungen des Eisenkreislaufs zum Phosphatekreislauf im eutrophen See. Arch. Hydrobiol. 1936, 29, 664–686. [Google Scholar]
- Mortimer, C.H. The exchange of dissolved substances between mud and water in lakes. J. Ecol. 1942, 30, 147–201. [Google Scholar] [CrossRef]
- Wendt-Potthoff, K.; Kloß, C.; Schultze, M.; Koschorreck, M. Anaerobic metabolism of two hydro-morphological similar pre-dams under contrasting nutrient loading (Rappbode Reservoir System, Germany). Int. Rev. Hydrobiol. 2014, 99, 350–362. [Google Scholar] [CrossRef]
- Kelderman, P. Sediment-water exchange in Lake Grevelingen under different environmental conditions. Neth. J. Sea Res. 1984, 18, 286–311. [Google Scholar] [CrossRef]
- Rozan, T.F.; Taillefert, M.; Trouwborst, R.E.; Glazer, B.T.; Ma, S.F.; Herszage, J.; Valdes, L.M.; Price, K.S.; Luther, G.W. Iron-sulfur-phosphorus cycling in the sediments of a shallow coastal bay: Implications for sediment nutrient release and benthic macroalgal blooms. Limnol. Oceanogr. 2002, 47, 1346–1354. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Wen, Y.; Zhou, J.; Wu, Y. Phosphorus release from lake sediments: Effects of pH, temperature and dissolved oxygen. KSCE J. Civ. Eng. 2014, 18, 323–329. [Google Scholar] [CrossRef]
- Sobek, S.; Cristian, G.; Birgit, K.; Tranvik, L.J.; David, B.; María, M.-P. Temperature Dependence of Apparent Respiratory Quotients and Oxygen Penetration Depth in Contrasting Lake Sediments. J. Geophys. Res. Biogeosci. 2017, 122, 3076–3087. [Google Scholar] [CrossRef] [Green Version]
- Liikanen, A.; Murtoniemi, T.; Tanskanen, H.; Väisänen, T.; Martikainen, P.J. Effects of Temperature and Oxygen Availability on Greenhouse Gas and Nutrient Dynamics in Sediment of a Eutrophic Mid-Boreal Lake. Biogeochemistry 2002, 59, 269–286. [Google Scholar] [CrossRef]
- Wouters, R. Das Bodewerk. Wasser Abfall 2011, 9, 10–15. [Google Scholar] [CrossRef]
- Bocaniov, S.A.; Barton, D.R.; Schiff, S.L.; Smith, R.E.H. Impact of tributary DOM and nutrient inputs on the nearshore ecology of a large, oligotrophic lake (Georgian Bay, Lake Huron, Canada). Aquat. Sci. 2013, 75, 321–332. [Google Scholar] [CrossRef]
- Rinke, K.; Kuehn, B.; Bocaniov, S.; Wendt-Potthoff, K.; Buettner, O.; Tittel, J.; Schultze, M.; Herzsprung, P.; Roenicke, H.; Rink, K.; et al. Reservoirs as sentinels of catchments: The Rappbode Reservoir Observatory (Harz Mountains, Germany). Environ. Earth Sci. 2013, 69, 523–536. [Google Scholar] [CrossRef]
- Benndorf, J.; Kranich, J.; Mehner, T.; Wagner, A. Temperature impact on the midsummer decline of Daphnia galeata: An analysis of long-term data from the biomanipulated Bautzen Reservoir (Germany). Freshwat. Biol. 2001, 46, 199–211. [Google Scholar] [CrossRef]
- Hülsmann, S. Recruitment patterns of Daphnia: A key for understanding midsummer declines? Hydrobiologia 2003, 491, 35–46. [Google Scholar] [CrossRef]
- Rinke, K.; Hübner, I.; Petzoldt, T.; Rolinski, S.; König-Rinke, M.; Post, J.; Lorke, A.; Benndorf, J. How internal waves influence the vertical distribution of zooplankton. Freshwat. Biol. 2007, 52, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Al-Mukhtar, M.; Dunger, V.; Merkel, B. Runoff and sediment yield modeling by means of WEPP in the Bautzen dam catchment, Germany. Environ. Earth Sci. 2014, 72, 2051–2063. [Google Scholar] [CrossRef]
- Friese, K.; Schultze, M.; Boehrer, B.; Büttner, O.; Herzsprung, P.; Koschorreck, M.; Kuehn, B.; Rönicke, H.; Tittel, J.; Wendt-Potthoff, K.; et al. Ecological response of two hydro-morphological similar pre-dams to contrasting land-use in the Rappbode reservoir system (Germany). Int. Rev. Hydrobiol. 2014, 99, 335–349. [Google Scholar] [CrossRef]
- Wentzky, V.C.; Tittel, J.; Jäger, C.G.; Rinke, K. Mechanisms preventing a decrease in phytoplankton biomass after phosphorus reductions in a German drinking water reservoir—Results from more than 50 years of observation. Freshwat. Biol. 2018, 63, 1063–1076. [Google Scholar] [CrossRef]
- Dadi, T.; Völkner, C.; Koschorreck, M. A sediment core incubation method to measure the flux of dissolved organic carbon between sediment and water. J. Soils Sed. 2015, 15, 2350–2358. [Google Scholar] [CrossRef]
- Morgenstern, P.; Friese, K.; Wendt-Potthoff, K.; Wennrich, R. Bulk Chemistry Analysis of Sediments from Acid Mine Lakes by Means of Wavelength Dispersive X-ray Fluorescence. Mine Water Environ. 2001, 20, 105–113. [Google Scholar] [CrossRef]
- Psenner, R.; Pucsko, R.; Sager, M. Die Fraktionierung organischer und anorganischer Phosphorverbindungen von Sedimenten—Versuch einer Definition ökologisch wichtiger Fraktionen. [Fractionation of organic and inorganic phosphorus compounds in lake sediments: An attempt to characterize ecologically important fractions]. Arch. Hydrobiol. Suppl. 1984, 70, 111–155. [Google Scholar]
- Hupfer, M.; Gächter, R.; Giovanoli, R. Transformation of phosphorus species in settling seston and during early sediment diagenesis. Aquat. Sci. 1995, 57, 305–324. [Google Scholar] [CrossRef]
- Steinman, A.; Rediske, R.; Reddy, K.R. The reduction of internal phosphorus loading using alum in Spring Lake, Michigan. J. Environ. Qual. 2004, 33, 2040–2048. [Google Scholar] [CrossRef]
- R-Core-Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Groemping, U. Relative Importance for Linear Regression in R: The Package relaimpo. J. Stat. Softw. 2006, 17, 27. [Google Scholar] [CrossRef] [Green Version]
- Grüneberg, B.; Dadi, T.; Lindim, C.; Fischer, H. Effects of nitrogen and phosphorus load reduction on benthic phosphorus release in a riverine lake. Biogeochemistry 2014, 1–18. [Google Scholar] [CrossRef]
- Ahlgren, J.; Tranvik, L.; Gogoll, A.; Waldebäck, M.; Markides, K.; Rydin, E. Sediment Depth Attenuation of Biogenic Phosphorus Compounds Measured by 31P NMR. Environ. Sci. Technol. 2005, 39, 867–872. [Google Scholar] [CrossRef]
- Gudasz, C.; Sobek, S.; Bastviken, D.; Koehler, B.; Tranvik, L.J. Temperature sensitivity of organic carbon mineralization in contrasting lake sediments. J. Geophys. Res. Biogeosci. 2015, 120, 1215–1225. [Google Scholar] [CrossRef] [Green Version]
- Skoog, A.C.; Arias-Esquivel, V.A. The effect of induced anoxia and reoxygenation on benthic fluxes of organic carbon, phosphate, iron, and manganese. Sci. Total Environ. 2009, 407, 6085–6092. [Google Scholar] [CrossRef]
- Caraco, N.F.; Cole, J.J.; Likens, G.E. Sulfate control of phosphorus availability in lakes. Hydrobiologia 1993, 253, 275–280. [Google Scholar] [CrossRef]
- Holmer, M.; Storkholm, P. Sulphate reduction and sulphur cycling in lake sediments: A review. Freshwat. Biol. 2001, 46, 431–451. [Google Scholar] [CrossRef]
- Rapin, A.; Grybos, M.; Rabiet, M.; Mourier, B.; Deluchat, V. Phosphorus mobility in dam reservoir affected by redox oscillations: An experimental study. J. Environ. Sci. 2018. [Google Scholar] [CrossRef]
- Hinkle, M.A.G.; Wang, Z.; Giammar, D.E.; Catalano, J.G. Interaction of Fe(II) with phosphate and sulfate on iron oxide surfaces. Geochim. Cosmochim. Acta 2015, 158, 130–146. [Google Scholar] [CrossRef]
- Parfitt, R.L.; Smart, R.S.C. The Mechanism of Sulfate Adsorption on Iron Oxides1. Soil Sci. Soc. Am. J. 1978, 42, 48–50. [Google Scholar] [CrossRef]
- Geelhoed, J.S.; Hiemstra, T.; Van Riemsdijk, W.H. Phosphate and sulfate adsorption on goethite: Single anion and competitive adsorption. Geochim. Cosmochim. Acta 1997, 61, 2389–2396. [Google Scholar] [CrossRef]
- Søndergaard, M.; Kristensen, P.; Jeppesen, E. Phosphorus release from resuspended sediment in the shallow and wind-exposed Lake Arresø, Denmark. Hydrobiologia 1992, 228, 91–99. [Google Scholar] [CrossRef]
- Anthony, J.L.; Lewis, W.M. Low boundary layer response and temperature dependence of nitrogen and phosphorus releases from oxic sediments of an oligotrophic lake. Aquat. Sci. 2012, 74, 611–617. [Google Scholar] [CrossRef]
- Nürnberg, G.K.; Shaw, M.; Dillon, P.J.; McQueen, D.J. Internal Phosphorus Load in an Oligotrophic Precambrian Shield Lake with an Anoxic Hypolimnion. Can. J. Fish. Aquat. Sci. 1986, 43, 574–580. [Google Scholar] [CrossRef]
- Haggard, B.E.; Soerens, T.S. Sediment phosphorus release at a small impoundment on the Illinois River, Arkansas and Oklahoma, USA. Ecol. Eng. 2006, 28, 280–287. [Google Scholar] [CrossRef]
- Spears, B.M.; Carvalho, L.; Perkins, R.; Kirika, A.; Paterson, D.M. Sediment phosphorus cycling in a large shallow lake: Spatio-temporal variation in phosphorus pools and release. Hydrobiologia 2007, 584, 37–48. [Google Scholar] [CrossRef]
- Nowlin, W.H.; Evarts, J.L.; Vanni, M.J. Release rates and potential fates of nitrogen and phosphorus from sediments in a eutrophic reservoir. Freshwat. Biol. 2005, 50, 301–322. [Google Scholar] [CrossRef]
- Beutel, M.W.; Horne, A.J. Nutrient Fluxes From Profundal Sediment of Ultra-Oligotrophic Lake Tahoe, California/Nevada: Implications for Water Quality and Management in a Changing Climate. Water Resour. Res. 2018, 54, 1549–1559. [Google Scholar] [CrossRef]
- Beutel, M.W. Inhibition of ammonia release from anoxic profundal sediments in lakes using hypolimnetic oxygenation. Ecol. Eng. 2006, 28, 271–279. [Google Scholar] [CrossRef]
- Kleeberg, A.; Herzog, C.; Hupfer, M. Redox sensitivity of iron in phosphorus binding does not impede lake restoration. Water Res. 2013, 47, 1491–1502. [Google Scholar] [CrossRef] [PubMed]
- Lewandowski, J.; Laskov, C.; Hupfer, M. The relationship between Chironomus plumosus burrows and the spatial distribution of pore-water phosphate, iron and ammonium in lake sediments. Freshwat. Biol. 2007, 52, 331–343. [Google Scholar] [CrossRef]
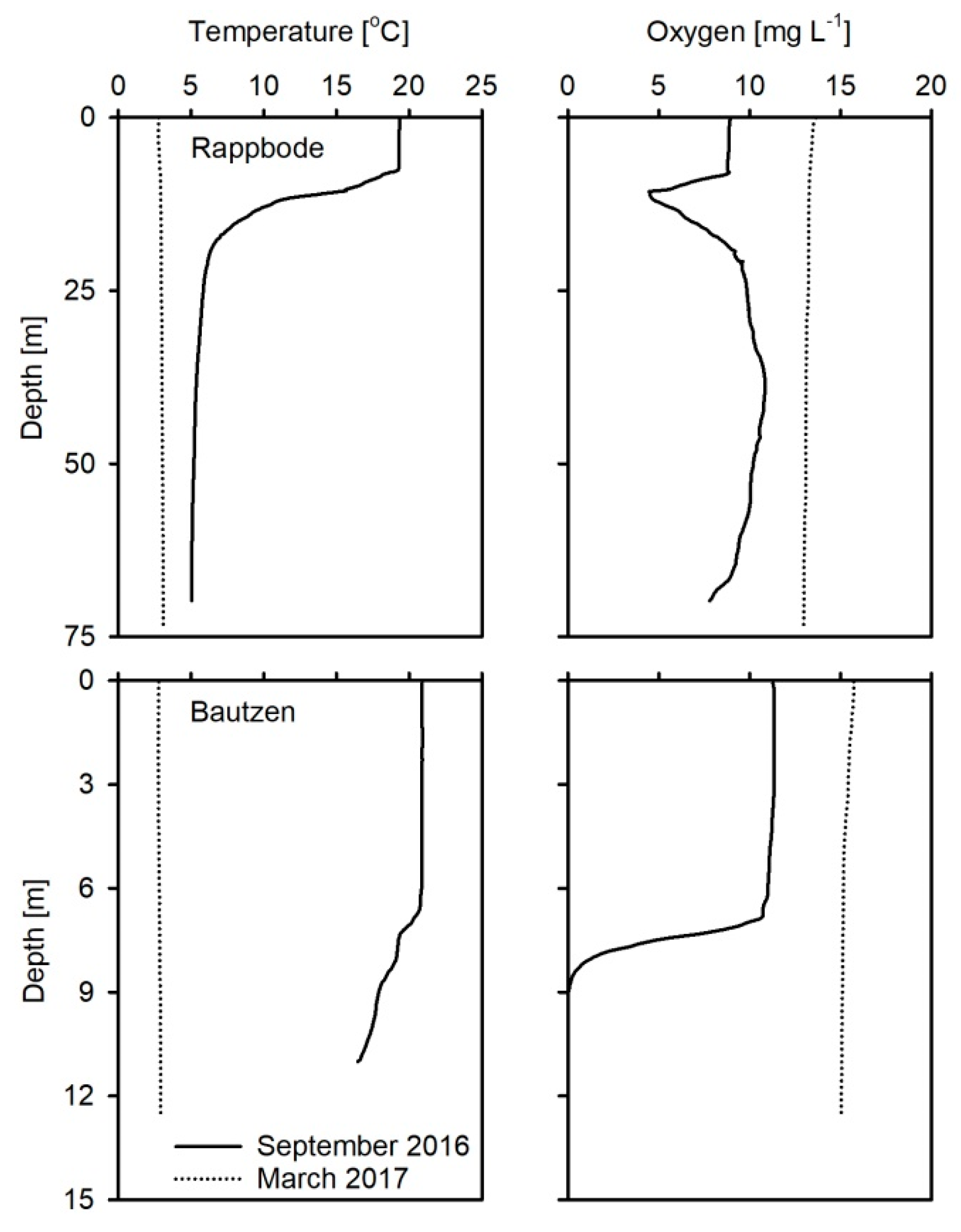
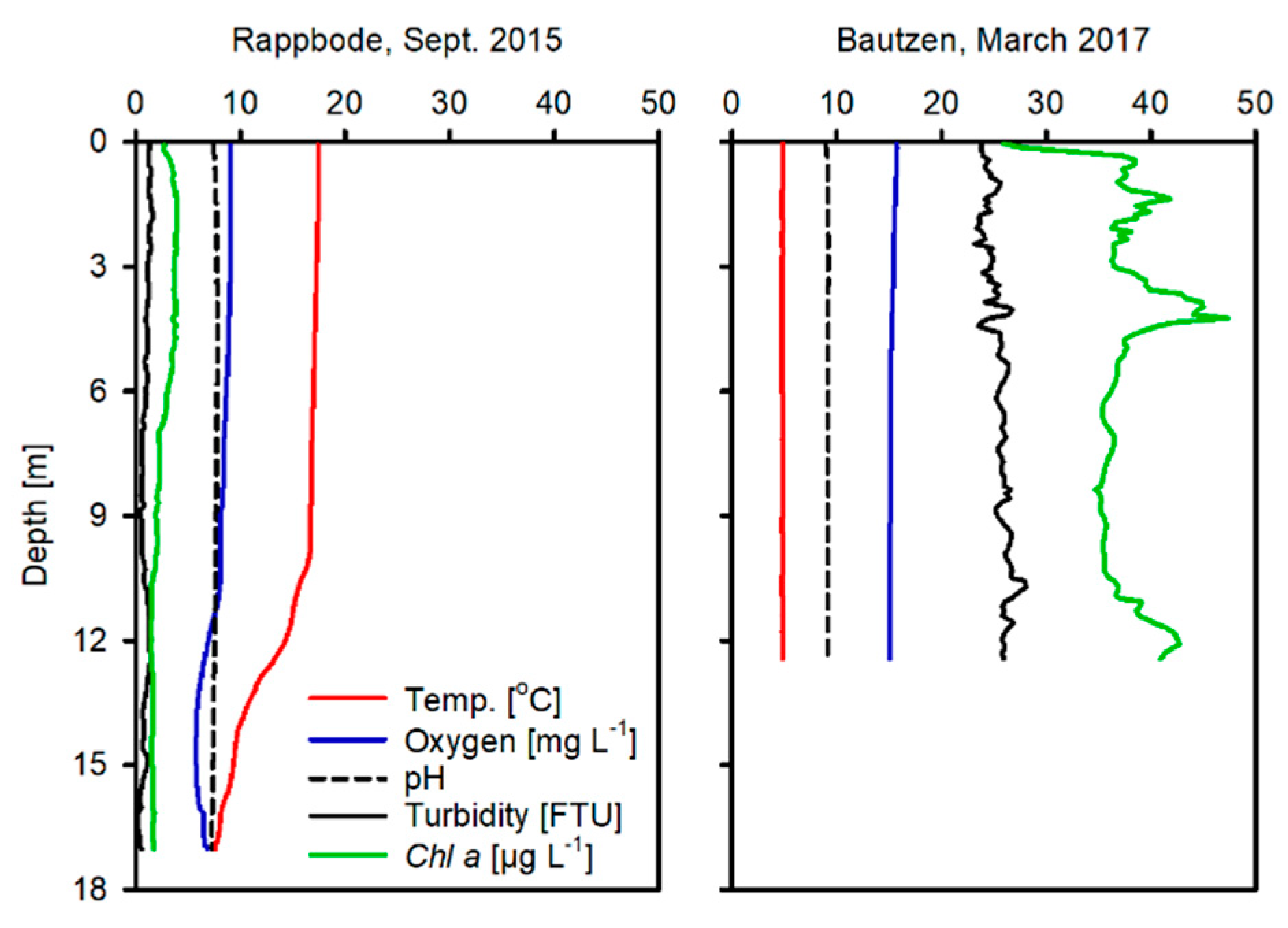
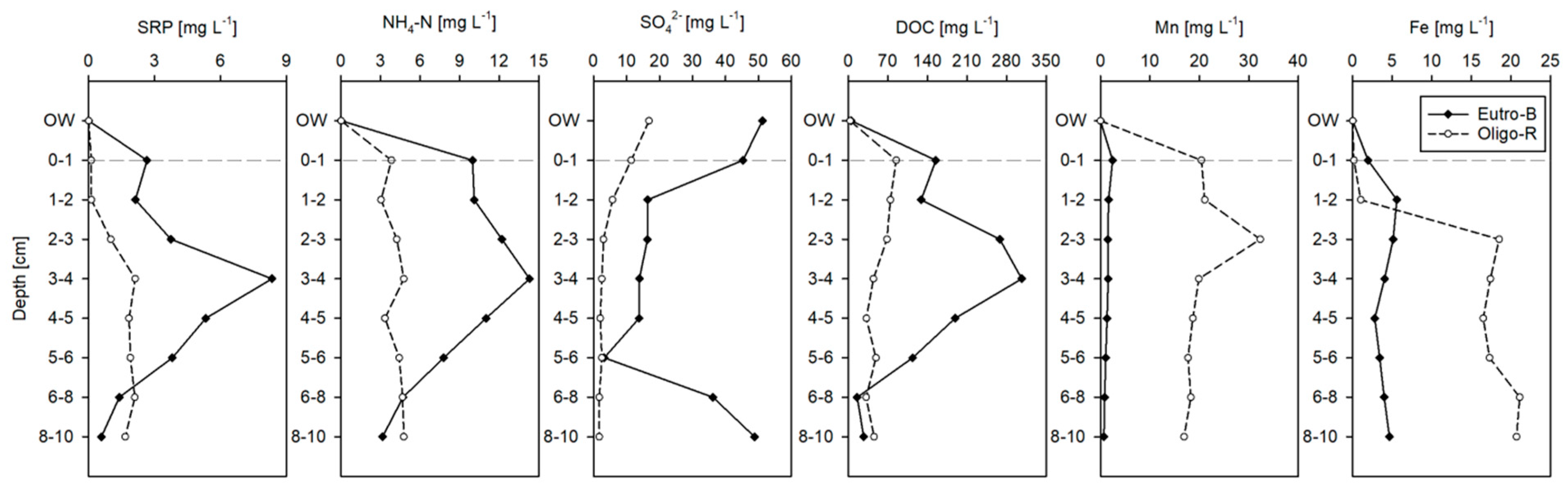

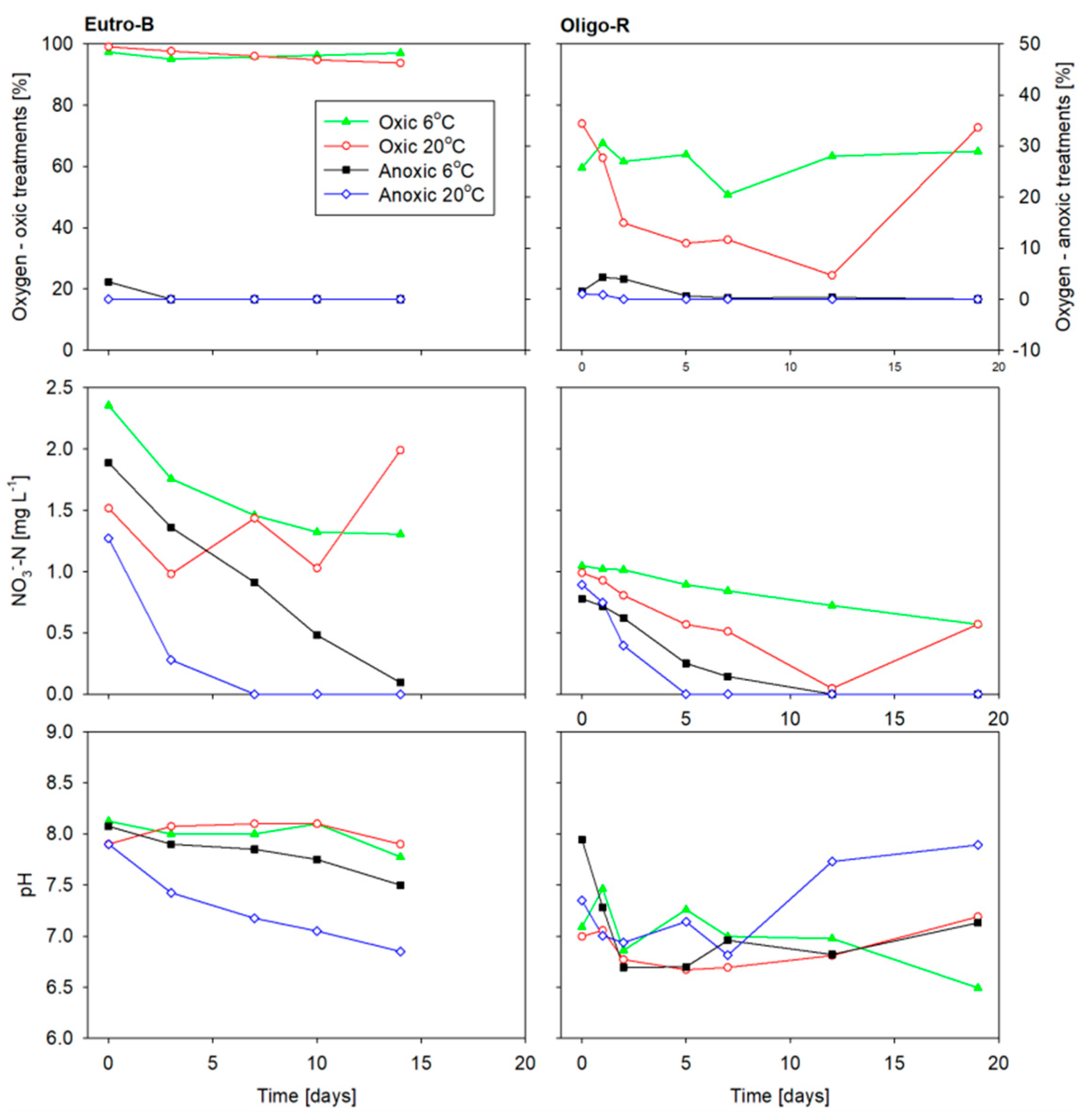
| Parameter | Oligo-R | Eutro-B |
|---|---|---|
| Volume (million m3) | 113 | 39 |
| Surface area (km2) | 3.95 | 5.33 |
| Catchment area (km2) | 116 | 310 |
| Catchment land use (%) | ||
| Forest | 75 | 16 |
| Grasslands | - | 38 |
| Agriculture | 20 | 34 |
| Urban settlements | 5 | 12 |
| Dam height (m) | 106 | 19.4 |
| Elevation (m a.s.l) | 423.6 | 167.5 |
| Mean depth (m) | 28.6 | 7.4 |
| Maximum depth (m) | 89 | 13.5 |
| Retention time (day) | 344 | 164 |
| TP (µg L−1) | <5 | 60–100 |
| TN (mg L−1) | <1 | 2 |
| Chlorophyll a (µg L−1) | <10 | 50–80 |
| Trophic state | Oligotrophic | Eutrophic |
| Parameter (mmol m−2 day−1) | Oligo-R | Eutro-B | ||||||
|---|---|---|---|---|---|---|---|---|
| Oxic 6 °C n = 3 | Oxic 20 °C n = 3 | Anoxic 6 °C n = 3 | Anoxic 20 °C n = 3 | Oxic 6 °C n = 3 | Oxic 20 °C n = 4 | Anoxic 6 °C n = 4 | Anoxic 20 °C n = 4 | |
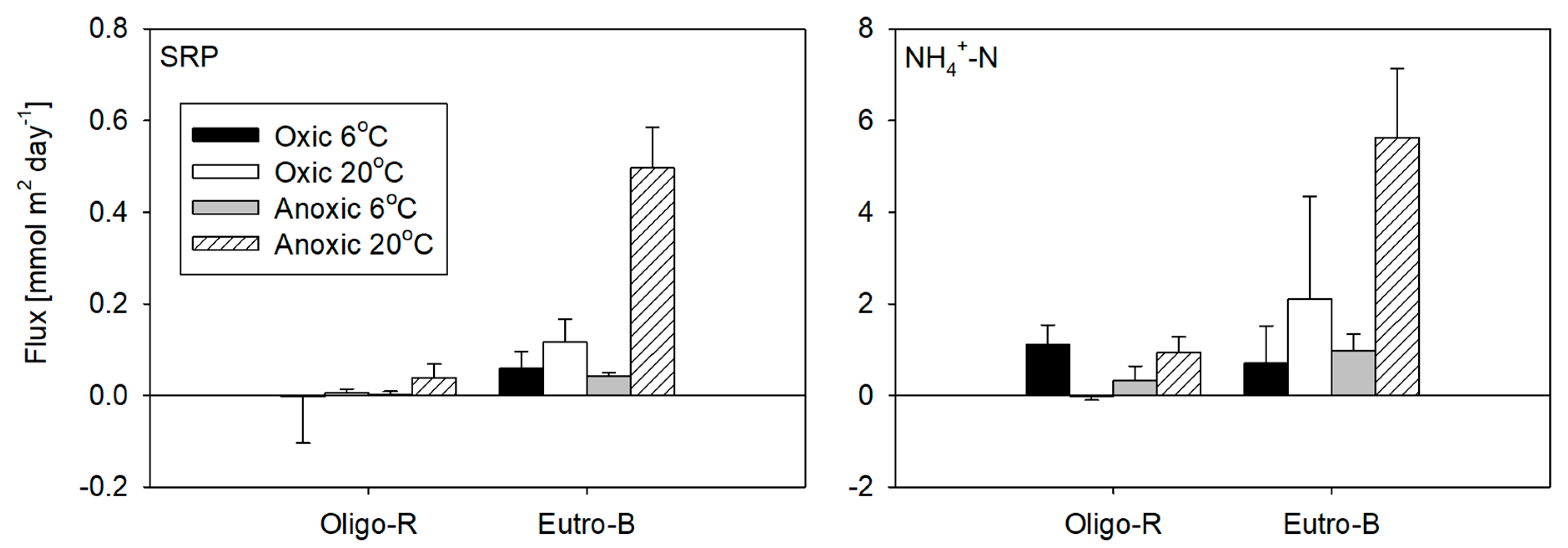
| DP | −0.003 ± 0.004 | 0.008 ± 0.008 | 0.002 ± 0.004 | 0.045 ± 0.026 | 0.06 ± 0.03 | 0.14 ± 0.07 | 0.05 ± 0.01 | 0.59 ± 0.13 |
| SRP | −0.002 ± 0.102 | 0.006 ± 0.007 | 0.003 ±0.006 | 0.039 ± 0.029 | 0.06 ± 0.04 | 0.12 ± 0.05 | 0.04 ± 0.01 | 0.50 ± 0.09 |
| NH4+-N | 1.11 ± 0.43 | 0.32 ± 0.32 | −0.02 ± 0.08 | 0.94 ± 0.35 | 0.71 ± 0.8 | 2.11 ± 2.24 | 0.99 ± 0.36 | 5.63 ± 1.50 |
| NO3−-N | −0.53 ± 0.1 | −0.54 ± 0.3 | −0.51 ± 0.23 | −0.94 ± 0.16 | −1.54 ± 0.34 | −0.24 ± 0.97 | −2.47 ± 0.16 | −1.24 ± 0.39 |
| DOC | −0.75 ± 0.27 | 0.94 ± 0.93 | 1.26 ± 1.14 | 10.39 ± 6.68 | 0.10 ± 0.18 | 1.89 ± 0.78 | 0.29 ± 0.33 | 1.78 ± 0.49 |
| Fe | −0.01 ± 0.01 | −0.01 ± 0.01 | 0.03 ± 0.06 | 0.20 ± 0.12 | 0.01 ± 0.01 | 0.001 ± 0.002 | 0.03 ± 0.02 | 1.09 ± 0.54 |
| Mn | −0.29 ± 0.19 | 0.22 ± 0.56 | 1.4 ± 1.05 | 5.16 ± 1.01 | −0.002 ± 0.03 | −0.02 ± 0.01 | 0.17 ± 0.06 | 0.15 ± 0.04 |
| SO42− | 0.24 ± 0.10 | 0.034 ± 0.12 | −0.05 ± 0.09 | −1.15 ± 0.08 | −1.88 ± 1.48 | −3.14 ± 2.39 | −1.78 ± 1.08 | −4.28 ± 1.78 |
| O2 | NA | NA | NA | NA | −28.53 ± 4.28 | −69.24 ± 23.14 | NA | NA |
| Variable | Temperature p-Value (Partial R2) | Oxygen Condition p-Value (Partial R2) | Interaction p-Value (Partial R2) | Overall R2 |
|---|---|---|---|---|
| Bautzen Reservoir (n = 15) | ||||
| SRP | 0.003 (0.380) | 0.025 (0.167) | 0.003 (0.153) | 0.70 |
| DP | 0.004 (0.366) | 0.031 (0.164) | 0.050 (0.144) | 0.67 |
| Fe | 0.007 (0.241) | 0.005 (0.272) | 0.009 (0.230) | 0.74 |
| Mn | 0.476 (0.018) | <0.001 (0.777) | 0.887 (0.000) | 0.80 |
| DOC | <0.001 (0.720) | 0.841 (0.001) | 0.556 (0.009) | 0.73 |
| NO3−-N | <0.001 (0.528) | <0.001 (0.368) | 0.007 (0.052) | 0.95 |
| NH4+-N | 0.002 (0.243) | <0.001 (0.347) | 0.003 (0.231) | 0.82 |
| SO42− | 0.05 (0.287) | 0.405 (0.038) | 0.800 (0.004) | 0.33 |
| Rappbode Reservoir (n = 12) | ||||
| SRP | 0.100 (0.231) | 0.155 (0.166) | 0.347 (0.067) | 0.46 |
| DP | 0.039 (0.292) | 0.065 (0.219) | 0.177 (0.105) | 0.62 |
| Fe | 0.132 (0.138) | 0.030 (0.341) | 0.147 (0.127) | 0.61 |
| Mn | 0.003 (0.224) | <0.001 (0.539) | 0.014 (0.130) | 0.89 |
| DOC | 0.056 (0.238) | 0.045 (0.268) | 0.163 (0.113) | 0.62 |
| NO3−-N | 0.185 (0.151) | 0.232 (0.120) | 0.179 (0.155) | 0.43 |
| NH4+-N | 0.716 (0.006) | 0.285 (0.054) | 0.005 (0.613) | 0.67 |
| SO42− | <0.001 (0.350) | <0.001 (0.453) | <0.001 (0.165) | 0.97 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dadi, T.; Rinke, K.; Friese, K. Trajectories of Sediment-Water Interactions in Reservoirs as a Result of Temperature and Oxygen Conditions. Water 2020, 12, 1065. https://doi.org/10.3390/w12041065
Dadi T, Rinke K, Friese K. Trajectories of Sediment-Water Interactions in Reservoirs as a Result of Temperature and Oxygen Conditions. Water. 2020; 12(4):1065. https://doi.org/10.3390/w12041065
Chicago/Turabian StyleDadi, Tallent, Karsten Rinke, and Kurt Friese. 2020. "Trajectories of Sediment-Water Interactions in Reservoirs as a Result of Temperature and Oxygen Conditions" Water 12, no. 4: 1065. https://doi.org/10.3390/w12041065





