Vegetated Ditch Habitats Provide Net Nitrogen Sink and Phosphorus Storage Capacity in Agricultural Drainage Networks Despite Senescent Plant Leaching
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Experimental Runoff Event
2.3. Field Sample Collection and Laboratory Analysis
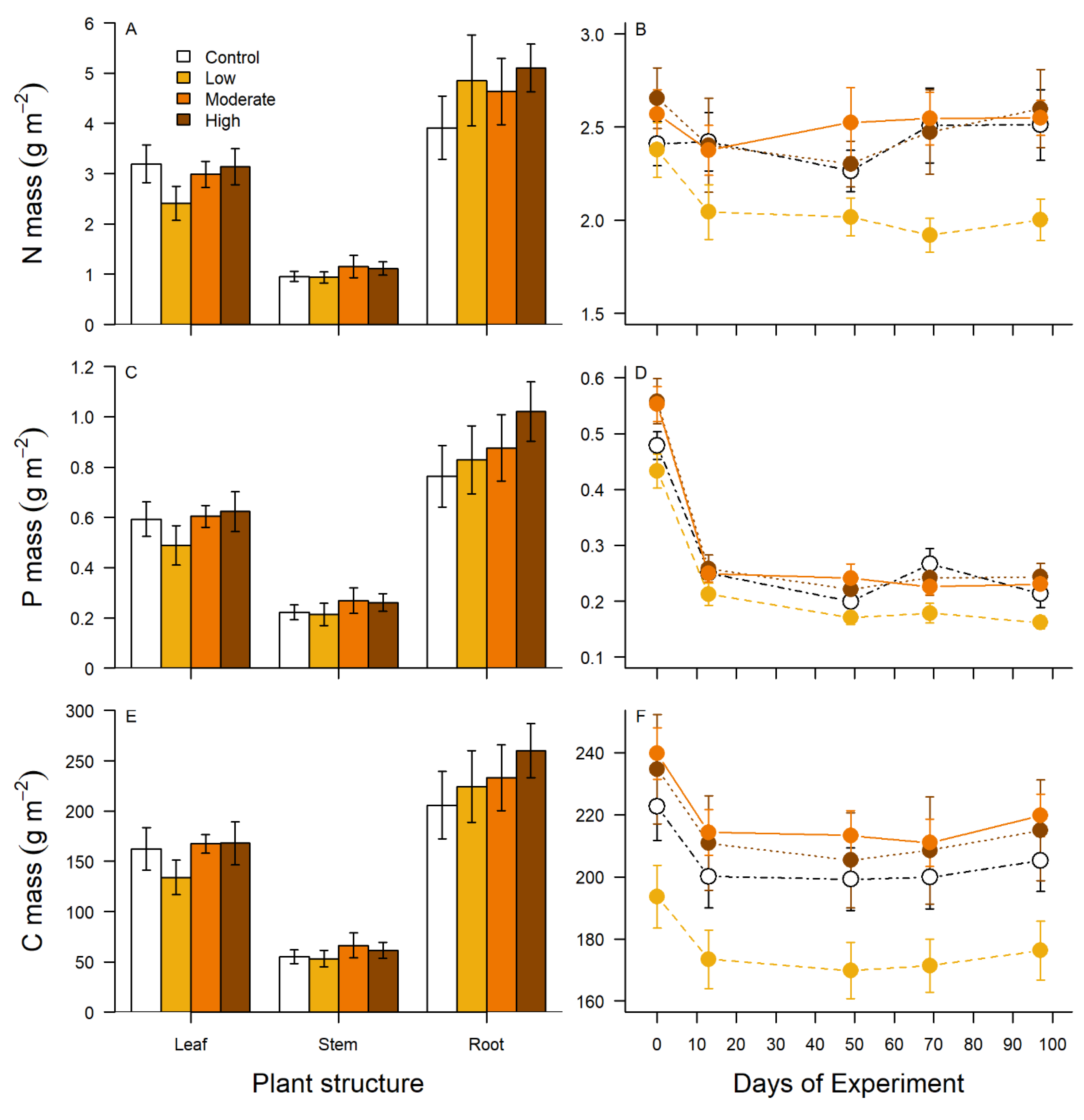
2.4. Plant Tissue Nutrient Content and Breakdown
2.5. Whole-System N and P Budgets Estimates
2.6. Statistical Analyses
3. Results
3.1. Summer Retention and Denitrification
3.2. Plant Biomass, Breakdown, and Stoichiometry
3.3. Winter Nutrient Export
3.4. Cumulative Flux
4. Discussion
4.1. The Role of Denitrification in Net Nitrogen retention in Vegetated Drainage Environments
4.2. Potential for Nutrient Remobilization from Plant Tissue Outside of Growing Season
4.3. Managing Nitrogen and Phosphorus Runoff within Vegetated Agricultural Drainage Networks
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Battye, W.; Aneja, V.P.; Schlesinger, W.H. Is nitrogen the next carbon? Earth’s Futur. 2017, 5, 894–904. [Google Scholar] [CrossRef]
- Cordell, D.; White, S. Life’s bottleneck: Sustaining the world’s phosphorus for a food secure future. Annu. Rev. Eviron. Resour. 2014, 39, 161–188. [Google Scholar] [CrossRef]
- Sarvajayakesavalu, S.; Lu, Y.; Withers, P.J.A.; Pavinato, P.S.; Pan, G.; Chareonsudjai, P. Phosphorus recovery: A need for an integrated approach. Ecosyst. Health Sust. 2018, 4, 48–57. [Google Scholar] [CrossRef]
- Stoddard, J.L.; Van Sickle, J.; Herlihy, A.T.; Brahney, J.; Paulsen, S.; Peck, D.V.; Mitchell, R.; Pollard, A.I. Continental-scale increase in lake and stream phosphorus: Are oligotrophic systems disappearing in the United States? Environ. Sci. Technol. 2016, 50, 3409–3415. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.; Acreman, M.C. Wetland nutrient removal: A review of the evidence. Hydrol. Earth Syst. Sci. 2004, 8, 673–685. [Google Scholar] [CrossRef]
- Vymazal, J. Plants used in constructed wetlands with horizontal subsurface flow: A review. Hydrobiologia 2011, 674, 133–156. [Google Scholar] [CrossRef]
- Vymazal, J. The use of constructed wetlands for nitrogen removal from agricultural drainage: A review. Sci. Agric. Bohem. 2017, 48, 82–91. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, D.; Dong, J.; Tan, S.K. Application of constructed wetlands for treating agricultural runoff and agro-industrial wastewater: A review. Hydrobiologia 2018, 805, 1–31. [Google Scholar] [CrossRef]
- Tournebize, J.; Chaumont, C.; Mander, Ü. Implications for constructed wetlands to mitigate nitrate and pesticide pollution in agricultural drained watersheds. Ecol. Eng. 2017, 103, 415–425. [Google Scholar] [CrossRef]
- Li, J.; Yang, X.; Wang, Z.; Shan, Y.; Zheng, Z. Comparison of four aquatic plant treatment systems for nutrient removal from eutrophied water. Bioresour. Technol. 2015, 179, 1–7. [Google Scholar] [CrossRef]
- Soana, E.; Balestrini, R.; Vincenzi, F.; Bartoli, M.; Castaldelli, G. Mitigation of nitrogen pollution in vegetated ditches fed by nitrate-rich spring waters. Agric. Ecosyst. Environ. 2017, 243, 74–82. [Google Scholar] [CrossRef]
- Lu, B.; Xu, Z.; Li, J.; Chai, X. Removal of water nutrients by different aquatic plant species: An alternative way to remediate polluted rural rivers. Ecol. Eng. 2018, 110, 18–26. [Google Scholar] [CrossRef]
- Moore, M.T.; Kroger, R.; Locke, M.A.; Cullum, R.F.; Steinriede Jr, R.W.; Testa, S., III.; Lizotte, R.E., Jr.; Bryant, C.T.; Cooper, C.M. Nutrient mitigation capacity in Mississippi Delta, USA drainage ditches. Environ. Pollut. 2010, 158, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Vzmazal, J.; Březinová, T.D. Removal of nutrients, organics and suspended solids in vegetated agricultural drainage ditch. Ecol. Eng. 2018, 118, 97–103. [Google Scholar] [CrossRef]
- Schaller, J.L.; Royer, T.V.; David, M.B.; Tank, J.L. Denitrification associated with plants and sediments in an agricultural stream. J. N. Am. Benthol. Soc. 2004, 23, 667–676. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Helton, A.M.; Poole, G.C.; Hall, R.O., Jr.; Hamilton, S.K.; Peterson, B.J.; Tank, J.L.; Ashkenas, I.R.; Cooper, I.W.; Dahm, C.N.; et al. Stream denitrification across biomes and its response to anthropogenic nitrate loading. Nature 2008, 452, 202–205. [Google Scholar] [CrossRef]
- Castaldelli, G.; Soana, E.; Racchetti, E.; Vincenzi, F.; Fano, E.A.; Bartoli, M. Vegetated canals mitigate nitrogen surplus in agricultural watersheds. Agric. Ecosyst. Environ. 2015, 212, 253–262. [Google Scholar] [CrossRef]
- Taylor, J.M.; Moore, M.T.; Scott, J.T. Contrasting nutrient mitigation and denitrification potential of agricultural drainage environments with different emergent aquatic macrophytes. J. Environ. Qual. 2015, 44, 1304–1314. [Google Scholar] [CrossRef]
- Speir, S.L.; Taylor, J.M.; Scott, J.T. Seasonal differences in relationships between nitrate concentration and denitrification rates in ditch sediments vegetated with rice cutgrass. J. Environ. Qual. 2017, 46, 1500–1509. [Google Scholar] [CrossRef]
- Soana, E.; Bartoli, M.; Milardi, M.; Fano, E.A.; Castaldelli, G. An ounce of prevention is worth a pound of cure: Managing macrophytes for nitrate mitigation in irrigated agricultural watersheds. Sci. Total Environ. 2019, 647, 301–312. [Google Scholar] [CrossRef]
- Tyler, H.L.; Moore, M.T.; Locke, M.A. Influence of three aquatic macrophytes on mitigation of nitrogen species from agricultural runoff. Water Air Soil Poll. 2012, 223, 3227–3236. [Google Scholar] [CrossRef]
- Tyler, H.L.; Moore, M.T.; Locke, M.A. Potential for phosphate mitigation from agricultural runoff by three aquatic macrophytes. Water Air Soil Poll. 2012, 223, 4557–4564. [Google Scholar] [CrossRef]
- Krӧger, R.; Holland, M.M.; Moore, M.T.; Cooper, C.M. Plant senescence: A mechanism for nutrient release in temperate agricultural wetlands. Environ. Pollut. 2007, 146, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Peverly, J.H. Element accumulation and release by macrophytes in a wetland stream. J. Environ. Qual. 1985, 14, 137–143. [Google Scholar] [CrossRef]
- Menon, R.; Holland, M.M. Phosphorus release due to decomposition of wetland plants. Wetlands 2014, 34, 1191–1196. [Google Scholar] [CrossRef]
- Kumwimba, M.N.; Dzakpasu, M.; Zhu, B.; Muyembe, D.K. Uptake and release of sequestered nutrient in subtropical monsoon ecological ditch plant species. Water Air Soil Poll. 2016, 227, 405. [Google Scholar] [CrossRef]
- Finlay, J.C.; Small, G.E.; Sterner, R.W. Human influences on nitrogen removal in lakes. Science 2013, 342, 247–250. [Google Scholar] [CrossRef]
- USDA Plants Database. Available online: https://plants.sc.egov.usda.gov/java/charProfile?symbol=LEOR (accessed on 22 January 2020).
- Darris, D.; Bartow, A. Plant Fact Sheet for Rice Cutgrass (Leersia oryzoides); USDA-Natural Resources Conservation Service, Plant Materials Center: Corvallis, OR, USA, 2004. [Google Scholar]
- Shields, F.D., Jr.; Testa, S., III.; Cooper, C.M. Nitrogen and phosphorus levels in the Yazoo River basin, Mississippi. Ecohydrology 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Krӧger, R.; Cooper, C.M.; Moore, M.T. A preliminary study of an alternative controlled drainage strategy in surface drainage ditches: Low-grade weirs. Agric. Water Manag. 2008, 95, 678–684. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association/American Water Works Association/Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- Benfield, E.F.; Fritz, K.M.; Tiegs, S.D. Leaf-litter breakdown. In Methods in Stream Ecology, Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: London, UK, 2017; pp. 71–82. [Google Scholar]
- Halvorson, H.M.; White, G.; Scott, J.T.; Evans-White, M.A. Dietary and taxonomic controls on incorporation of microbial carbon and phosphorus by detritivorous caddisflies. Oecologia 2016, 180, 567–579. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; Heisterkamp, S.; Van Willigen, B. Nlme: Linear and Nonlinear Mixed Effects Models. R Project for Statistical Computing, Vienna, Austria. Available online: https://cran.r-project.org/web/packages/nlme (accessed on 27 July 2018).
- Nifong, R.L.; Taylor, J.M.; Adams, G.; Moore, M.T.; Farris, J.L. Recognizing both denitrification and nitrogen consumption improves performance of stream diel N2 flux models. Limno. Oceanogr. Meth. In press.
- Christensen, P.B.; Sørensen, J. Temporal variation of denitrification activity in plant-covered, littoral sediment from Lake Hampen, Denmark. Appl. Environ. Microbiol. 1986, 51, 1174–1179. [Google Scholar] [CrossRef] [PubMed]
- Zhai, X.; Piwpuan, N.; Arias, C.A.; Headley, T.; Brix, H. Can root exudates from emergent plants fuel denitrification in subsurface flow constructed wetlands. Ecol. Eng. 2013, 61, 555–563. [Google Scholar] [CrossRef]
- Brix, H.; Sorrel, B.K.; Orr, P.T. Internal pressurization and convective gas flow in some emergent freshwater macrophytes. Limnol. Oceanogr. 1992, 37, 1420–1433. [Google Scholar] [CrossRef]
- Bendix, M.T.; Tornbjerg, T.; Brix, H. Internal gas transport in Typha latifolia L. and Typha angustifolia L.: 1. Humidity-induced pressurization and convective throughflow. Aquat. Bot. 1994, 49, 75–89. [Google Scholar] [CrossRef]
- Veraart, A.J.; De Bruijne, W.J.J.; de Klein, J.J.M.; Peeters, E.T.H.M.; Scheffer, M. Effects of aquatic vegetation type on denitrification. Biogeochemistry 2011, 104, 267–274. [Google Scholar] [CrossRef]
- Sorrel, B.K.; Hawes, I. Convective gas flow development and the maximum depths achieved by helophyte vegetation in lakes. Ann. Bot. 2010, 105, 165–174. [Google Scholar] [CrossRef]
- Konnerup, D.; Sorrell, B.K.; Brix, H. Do tropical wetland plants possess convective gas flow mechanisms? New Phytol. 2011, 190, 379–386. [Google Scholar] [CrossRef]
- Reisinger, A.J.; Tank, J.L.; Hoellein, T.J.; Hall, R.O., Jr. Sediment, water column, and open-channel denitrification in rivers measured using membrane-inlet mass spectrometry. J. Geophys. Res. Biogeosci. 2016, 121, 1258–1274. [Google Scholar] [CrossRef]
- Yang, J.; Udvardi, M. Senescence and nitrogen use efficiency in perennial grasses for forage and biofuel production. J. Exp. Bot. 2018, 69, 855–865. [Google Scholar] [CrossRef]
- McMillan, S.K.; Piehler, M.F.; Thompson, S.P.; Pearl, H.W. Denitrification of nitrogen released from senescing algal biomass in coastal agricultural headwater streams. J. Environ. Qual. 2010, 39, 274–281. [Google Scholar] [CrossRef]
- Soana, E.; Gavioli, A.; Tamburini, E.; Fano, E.A.; Castaldelli, G. To mow or not to mow: Reed biofilms as denitrification hotspots in drainage canals. Ecol. Eng. 2018, 113, 1–10. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Q.; Hu, C.; Liang, R.; Qiu, J.; Wang, Y. Phosphorus release during decomposition of the submerged macrophyte Potamogeton crispus. Limnology 2018, 19, 355–366. [Google Scholar] [CrossRef]
- Scott, J.T.; Doyle, R.D. Coupled photosynthesis and heterotrophic bacterial biomass production in a nutrient-limited wetland periphyton mat. Aquat. Microb. Ecol. 2006, 45, 69–77. [Google Scholar] [CrossRef]
- Scott, J.T.; Back, J.A.; Taylor, J.M.; King, R.S. Does nutrient enrichment decouple algal-bacterial production in periphyton? J. N. Am. Benthol. Soc. 2008, 27, 332–344. [Google Scholar] [CrossRef]
- Sharpley, A.N.; Krogstad, T.; Kleinman, P.J.A.; Haggard, B.; Shigaki, F.; Saporito, L.S. Managing natural processes in drainage ditches for nonpoint source phosphorus control. J. Soil Water Conserv. 2007, 62, 197–206. [Google Scholar]
- Moore, M.T.; Bennett, E.R.; Cooper, C.M.; Smith, S., Jr.; Shields, F.D., Jr.; Milam, C.D.; Farris, J.L. Transport and fate of atrazine and lambda-cyhalothrin in an agricultural drainage ditch in the Mississippi Delta, USA. Agric. Ecosyst. Environ. 2001, 87, 309–314. [Google Scholar] [CrossRef]
- Needelman, B.A.; Kleinman, P.J.A.; Strock, J.S.; Allen, A.L. Improved management of agricultural drainage ditches for water quality protection: An overview. J. Soil Water Conserv. 2007, 62, 171–178. [Google Scholar]
- Stock, J.S.; Dell, C.J.; Schmidt, J.P. Managing natural processes in drainage ditches for nonpoint source nitrogen control. J. Soil Water Conserv. 2007, 62, 188–196. [Google Scholar]
- Royer, T.V.; Tank, J.L.; David, M.B. Transport and fate of nitrate in headwater agricultural streams in Illinois. J. Environ. Qual. 2004, 33, 1296–1304. [Google Scholar] [CrossRef]
- Sheibley, R.W.; Duff, J.H.; Tesoriero, A.J. Low transient storage and uptake efficiencies in seven agricultural streams. J. Environ. Qual. 2014, 43, 1980–1990. [Google Scholar] [CrossRef][Green Version]
- Krӧger, R.; Prince Czarnecki, J.M.; Tank, J.L.; Christopher, S.F.; Witter, J.D. Implementing innovative drainage management practices in the Mississippi River Basin to enhance nutrient reductions. J. Am. Water Resour. Assoc. 2015, 51, 1020–1028. [Google Scholar] [CrossRef]
- Krӧger, R.; Moore, M.T.; Farris, J.L.; Gopalan, M. Evidence for the use of flow-grade weirs in drainage ditches to improve nutrient reductions from agriculture. Water Air Soil Pollut. 2011, 221, 223–234. [Google Scholar] [CrossRef]
- Usborne, E.L.; Krӧger, R.; Pierce, S.C.; Brandt, J.; Goetz, D. Preliminary evidence of sediment and phosphorus dynamics behind newly installed low-grade weirs in agricultural drainage ditches. Water Air Soil Pollut. 2013, 224, 1–11. [Google Scholar] [CrossRef]
- Roley, S.S.; Tank, J.L.; Stephen, M.L.; Johnson, L.T.; Beaulieu, J.J.; Witter, J.D. Floodplain restoration enhances denitrification and reach-scale nitrogen removal in an agricultural stream. Ecol. App. 2012, 22, 281–297. [Google Scholar] [CrossRef]
- Mahl, U.H.; Tank, J.L.; Roley, S.S.; Davis, R.T. Two-stage ditch floodplains enhance N-removal capacity and reduce turbidity and dissolved P in agricultural streams. J. Am. Water Resour. Assoc. 2015, 51, 923–940. [Google Scholar] [CrossRef]
- Hanrahan, B.R.; Tank, J.L.; Dee, M.M.; Trentman, M.T.; Berg, E.M.; McMillan, S.K. Restored floodplains enhance denitrification compared to naturalized floodplains in agricultural streams. Biogeochemistry 2018, 141, 419–437. [Google Scholar] [CrossRef]
- Lizotte, R.E., Jr.; Yasarer, L.M.W.; Locke, M.A.; Bingner, R.L.; Knight, S.S. Lake nutrient responses to integrated conservation practices in an agricultural watershed. J. Environ. Qual. 2017, 46, 330–338. [Google Scholar] [CrossRef]
- Yasarer, L.M.W.; Bingner, R.L.; Garbrecht, J.D.; Locke, M.A.; Lizotte, R., Jr.; Momm, H.G.; Busteed, P.R. Climate change impacts on runoff, sediment, and nutrient loads in an agricultural watershed in the lower Mississippi River Basin. App. Eng. Agric. 2017, 33, 379–392. [Google Scholar] [CrossRef]
- Hanrahan, B.R.; Tank, J.L.; Christopher, S.F.; Mahl, U.H.; Trentman, M.T.; Royer, T.V. Winter cover crops reduce nitrate loss in an agricultural watershed in the central U.S. Agric. Ecosyst. Environ. 2018, 265, 513–523. [Google Scholar] [CrossRef]




| Factor | F | P |
|---|---|---|
| Nitrogen retention | ||
| P enrichment 3, 16 | 0.75 | 0.5401 |
| Month 2, 32 | 55.67 | <0.0001 |
| P enrichment × month 6, 32 | 1.40 | 0.2439 |
| Estimated denitrification | ||
| P enrichment 3, 16 | 0.86 | 0.4835 |
| Month 2, 32 | 5.10 | 0.0119 |
| P enrichment × month 6, 32 | 0.57 | 0.7478 |
| Phosphorus retention | ||
| P enrichment 3, 16 | 38.50 | <0.0001 |
| Month 2, 32 | 6.81 | 0.0034 |
| P enrichment × month 6, 32 | 10.71 | <0.0001 |
| Source of Variation | F | P | LS Means |
|---|---|---|---|
| Nitrogen mass by structure | |||
| P enrichment 3, 16 | 0.74 | 0.5454 | |
| Plant structure 2, 32 | 108.24 | <0.0001 | Stem < Leaf < Root |
| P enrichment × plant structure 6, 32 | 0.66 | 0.6841 | |
| Phosphorus mass by structure | |||
| P enrichment 3, 16 | 0.95 | 0.4403 | |
| Plant structure 2, 32 | 78.08 | <0.0001 | Stem < Leaf < Root |
| P enrichment × plant structure 6, 32 | 0.42 | 0.8581 | |
| Carbon mass by structure | |||
| P enrichment 3, 16 | 0.88 | 0.4702 | |
| Plant structure 2, 32 | 89.54 | < 0.0001 | Stem < Leaf < Root |
| P enrichment × plant structure 6, 32 | 0.35 | 0.9026 |
| Factor | F | P |
|---|---|---|
| N mass over time | ||
| P enrichment 3, 16 | 0.92 | 0.4545 |
| Day 4, 163 | 9.36 | <0.0001 |
| P enrichment × day 12, 163 | 3.16 | 0.0004 |
| P mass over time | ||
| P enrichment 3, 16 | 1.49 | 0.2555 |
| Day 4, 163 | 96.20 | <0.0001 |
| P enrichment × day 12, 163 | 1.11 | 0.3560 |
| C mass over time | ||
| P enrichment 3, 16 | 0.96 | 0.4341 |
| Day 4, 163 | 577.65 | <0.0001 |
| P enrichment × day 12, 163 | 4.02 | <0.0001 |
| Factor | F | P |
|---|---|---|
| Total Kjeldahl nitrogen export | ||
| P enrichment 3, 16 | 0.13 | 0.9420 |
| Day 13, 208 | 62.07 | <0.0001 |
| P enrichment × day 39, 208 | 1.18 | 0.2265 |
| Total phosphorus export | ||
| P enrichment 3, 16 | 1.03 | 0.4048 |
| Day 13, 208 | 104.24 | <0.0001 |
| P enrichment × day 39, 208 | 0.97 | 0.5193 |
| Total dissolved carbon export | ||
| P enrichment 3, 16 | 0.15 | 0.9271 |
| Day 13, 208 | 230.83 | <0.0001 |
| P enrichment × day 39, 208 | 0.89 | 0.6661 |
| P Treat | Retention | Denitrification | Export | Net Retention |
|---|---|---|---|---|
| Nitrogen | ||||
| Control | 1575.2 ± 107.1 | 619.9 ± 50.7 | 120.5 ± 26.6 | 1454.8 ± 95.5 |
| Low | 1468.9 ± 80.1 | 563.5 ± 78.4 | 134.0 ± 17.8 | 1334.9 ± 81.4 |
| Moderate | 1412.0 ± 84.5 | 608.9 ± 52.5 | 136.6 ± 13.0 | 1275.4 ± 95.7 |
| High | 1553.8 ± 92.0 | 578.8 ± 50.6 | 163.6 ± 35.9 | 1390.2±104.4 |
| Phosphorus | ||||
| Control | 55.6 ± 4.0a | NA | 41.3 ± 3.6 | 14.3 ± 4.3a |
| Low | 181.0 ± 26.9b | NA | 35.6 ± 4.7 | 145.4 ± 26.8b |
| Moderate | 221.9 ± 8.9c | NA | 38.5 ± 3.4 | 183.4 ± 10.8c |
| High | 404.9 ± 39.3d | NA | 42.7 ± 7.4 | 362.2 ± 42.2d |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taylor, J.; Moore, M.; Speir, S.L.; Testa, S., III. Vegetated Ditch Habitats Provide Net Nitrogen Sink and Phosphorus Storage Capacity in Agricultural Drainage Networks Despite Senescent Plant Leaching. Water 2020, 12, 875. https://doi.org/10.3390/w12030875
Taylor J, Moore M, Speir SL, Testa S III. Vegetated Ditch Habitats Provide Net Nitrogen Sink and Phosphorus Storage Capacity in Agricultural Drainage Networks Despite Senescent Plant Leaching. Water. 2020; 12(3):875. https://doi.org/10.3390/w12030875
Chicago/Turabian StyleTaylor, Jason, Matthew Moore, Shannon L. Speir, and Sam Testa, III. 2020. "Vegetated Ditch Habitats Provide Net Nitrogen Sink and Phosphorus Storage Capacity in Agricultural Drainage Networks Despite Senescent Plant Leaching" Water 12, no. 3: 875. https://doi.org/10.3390/w12030875
APA StyleTaylor, J., Moore, M., Speir, S. L., & Testa, S., III. (2020). Vegetated Ditch Habitats Provide Net Nitrogen Sink and Phosphorus Storage Capacity in Agricultural Drainage Networks Despite Senescent Plant Leaching. Water, 12(3), 875. https://doi.org/10.3390/w12030875





