Limnological Quality: Seasonality Assessment and Potential for Contamination of the Pindaré River Watershed, Pre-Amazon Region, Brazil
Abstract
1. Introduction
2. Materials and Methods
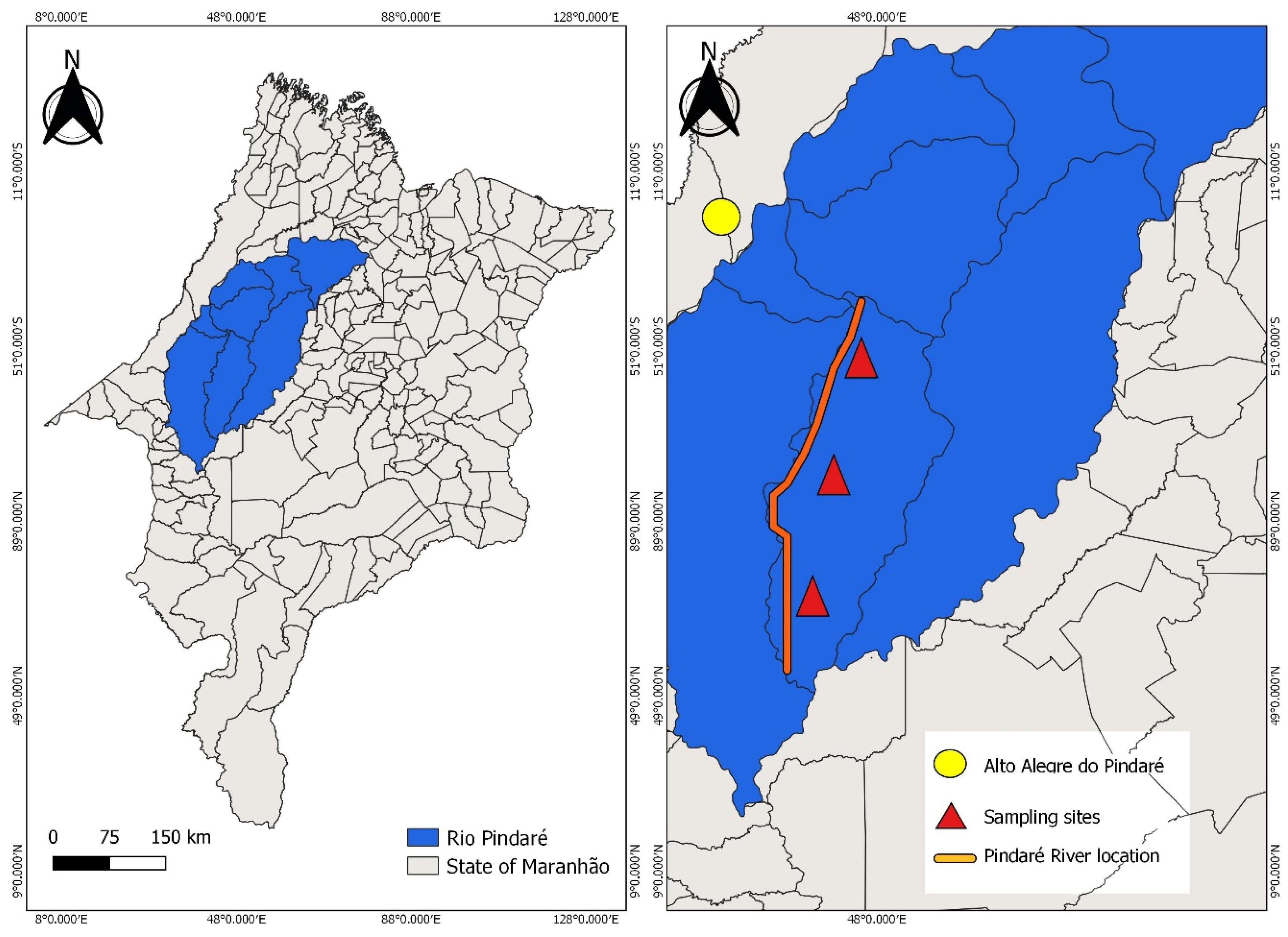
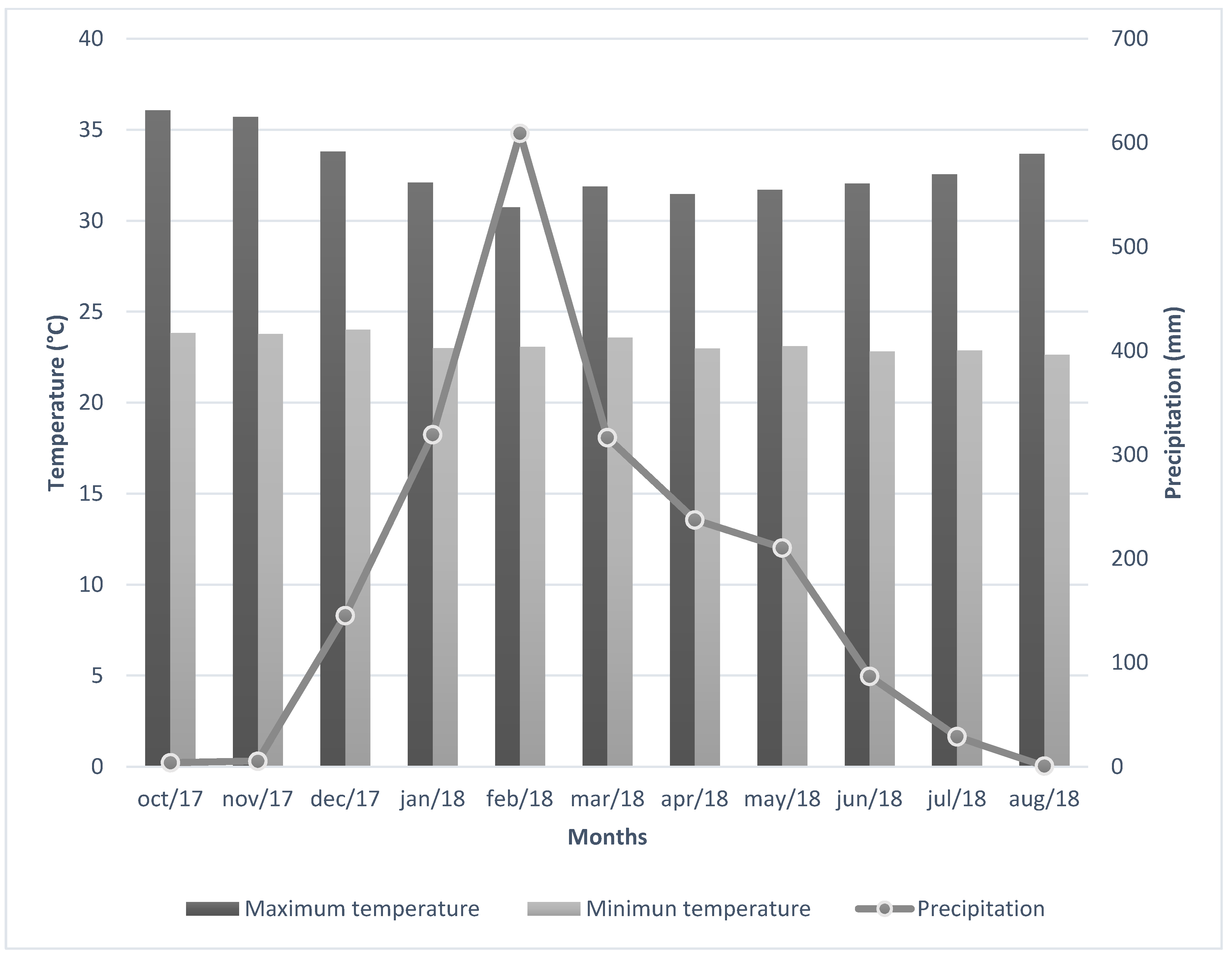
2.1. Study Area Description
2.2. Monitored Parameters
2.3. Multivariate Statistical Method: PCA
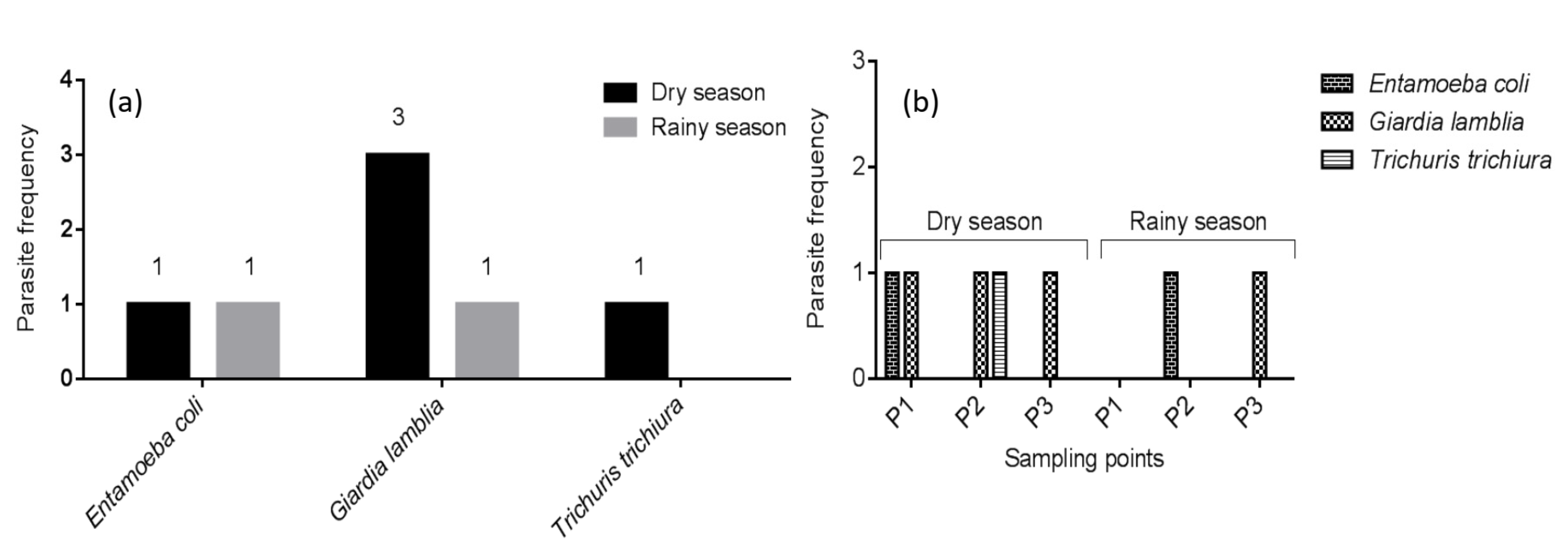
3. Results
3.1. Descriptive Measures of River Water Quality Data
3.2. Box Plots of Water Quality Parameters
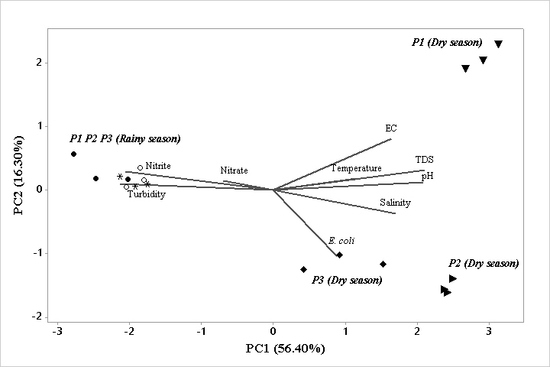
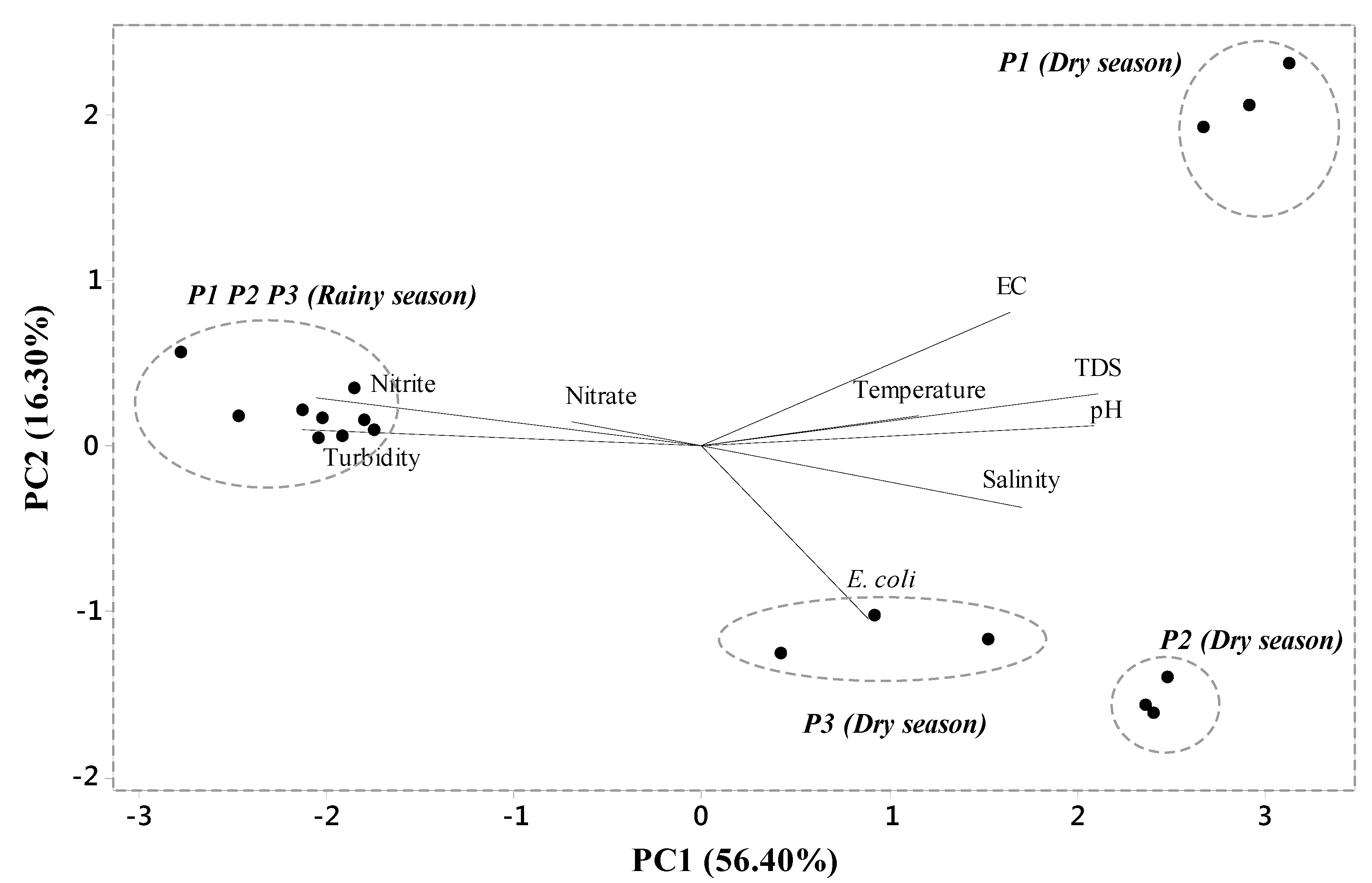
3.3. PCA of River Water Quality Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Reis, F.; Dias, C.R.; Abrahão, W.M.; Murakami, F.S. Avaliação da qualidade microbiológica de águas e superfícies de bebedouros de parques de Curitiba—PR. Visão Acadêmica 2012, 12, 55–70. [Google Scholar] [CrossRef]
- Pereira, P.R.M.; Rodrigues, T.C.S.; Viegas, J.C. Diagnóstico ambiental e caracterização morfométrica das Microbacias Hidrográficas de Pedro do Rosário, Amazônia Maranhense (Brasil). Rev. Bras. Gestão Ambient. Sustentabilidade 2016, 3, 153–163. [Google Scholar] [CrossRef][Green Version]
- Silva, M.R.; Silva, L.V.; Barreto, L.N.C.; Rodrigues, E.H.C.; Miranda, R.C.M.; Bezerra, D.S.; Pereira, D.C.A. Qualidade da água da bacia do Rio Pindaré, nos trechos correspondentes aos municípios de Pindaré- Mirim, Tufilândia e Alto Alegre no Estado do Maranhão. Águas Subterrâneas 2017, 31, 347–354. [Google Scholar] [CrossRef]
- Bacias do Nordeste. Relatório Estatístico Hidroviário Novembro/2014. Available online: http://www.transportes.gov.br (accessed on 10 November 2017).
- Gomes, W.K.A.M.; Souza, R.F.; Crispim, M.C. Avaliação espacial e temporal da qualidade da água em rio de área costeira do nordeste brasileiro. GEOTemas 2016, 6, 3–14. [Google Scholar]
- Derisio, J.C. Introdução ao Controle de Poluição Ambiental, 4th ed.; Oficina de Texto: São Paulo, Brasil, 2012. [Google Scholar]
- Nascimento, H.S.C. Avaliação da Qualidade da Água do rio Subaé. Bachelor’s Thesis, Universidade Federal do Recôncavo, Cruz das Almas, Bahia, 2016. [Google Scholar]
- BRASIL. Resolução CONAMA 357 de 17 de Março de 2005; Ministério do Meio Ambiente: Brasília, Brasil, 2005.
- Valias, A.P.G.S.; Roqueto, M.A.; Hornink, D.G.; Koroiva, E.H.; Vieira, F.C.; Rosa, G.M. Qualidade Microbiológica de Águas de Poços Rasos e de Nascentes de Propriedades Rurais do Município de São João da Boa Vista; Associação Brasileira de Águas Subterrâneas: São Paulo, Brasil, 2000; pp. 1–13. [Google Scholar]
- Amaral, L.A.; Nader Filho, A.; Rossi Júnior, O.D.; Penha, L.H.C. Características microbiológicas da água utilizada no processo de obtenção do leite. Pesqui. Veterinária Bras. 1995, 15, 85–88. [Google Scholar]
- Macari, M.; Amaral, L.A. Importância da qualidade da água e tipos de bebedouros para frangos de corte. In Manejo de Frangos de Corte, Campinas; Fundação Apinco de Ciência e Tecnologia Avícolas—FACTA: Campinas, Brazil, 1997; pp. 101–120. [Google Scholar]
- Lechevallier, M.W.; Norton, W.D.; Lee, R.G. Occurrence of Giardia and Cryptosporidium spp. in surface water supplies. Appl. Environ. Microbiol. 1997, 57, 2610–2616. [Google Scholar] [CrossRef]
- Mons, C.; Dumètre, A.; Gosselin, S.; Galliot, C.; Moulin, L. Monitoring of Cryptosporidium and Giardia river contamination in Paris area. Water Res. 2009, 43, 211–217. [Google Scholar] [CrossRef]
- Vieira, P.B.; Brandelli, C.L.C.; Veríssimo, C.M.; Tasca, T. Mecanismos específicos de patogenicidade e protozoários de mucosa: Entamoeba istolytica, Giardia lamblia e Trichomonas vaginalis. Rev. Hcpa 2012, 32, 58–70. [Google Scholar]
- Barbosa, A.S. Detecção de parasitos veiculados pela água e solo em aldeias Guarani dos municípios de Angra dos Reis e Paraty do Estado do Rio de Janeiro, Brasil. Rev. Inst. Adolfo Lutz 2011, 70, 423. [Google Scholar]
- Shrestha, S.; Kazama, F. Assessment of surface water quality using multivariate statistical techniques: A case study of the Fuji river basin, Japan. Environ. Model. Softw. 2007, 22, 464–475. [Google Scholar] [CrossRef]
- Cho, K.H.; Park, Y.; Kang, J.-H.; Ki, S.J.; Cha, S.; Lee, S.W.; Kim, J.H. Interpretation of seasonal water quality variation in the Yeongsan Reservoir, Korea using multivariate statistical analyses. Water Sci. Technol. 2009, 59, 2219–2226. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Wang, X.; Ye, L.; Qu, X. Evaluation of spatial and temporal variation in stream water quality by multivariate statistical techniques: A case study of the Xiangxi River basin, China. Quat. Int. 2012, 282, 137–144. [Google Scholar]
- Wang, P.; Wang, Y.; Bai, Y.; Tian, Z.; Li, J.; Shao, X.; Mustavich, L.F.; Li, B.L. Assessment of surface water quality via multivariate statistical techniques: A case study of the Songhua River Harbin region, China. J. Hydro-Environ. Res. 2013, 7, 30–40. [Google Scholar] [CrossRef]
- Feher, I.-C.; Zaharie, M.; Oprean, I. Spatial and seasonal variation of organic pollutants in surface water using multivariate statistical techniques. Water Sci. Technol. 2016, 74, 1726–1735. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.R.C.; Silva, L.V.; Barreto, L.N.; Firmo, W.C.A.; Silva, D.F.; Rodrigues, E.H.C. Avaliação dos parâmetros físico-químicos do sedimento do rio Pindaré, nos municípios de Pindaré-Mirim, Tufilândia e Alto Alegre (MA). In Meio Ambiente em Foco, 1st ed.; Toledo, F.B., Ed.; Editora Poisson: Belo Horizonte, Brazil, 2019; Volume 3, pp. 104–109. [Google Scholar]
- IBGE—Instituto Brasileiro de Geografia e estatística. Área da Unidade Territorial. 2016. Available online: https://cidades.ibge.gov.br (accessed on 10 December 2019).
- IBGE—Instituto Brasileiro de Geografia e Estatística. Cidades, 2017. Available online: https://cidades.ibge.gov.br (accessed on 12 December 2019).
- INMET (Instituto Nacional de Meteorologia). BDMEP—Banco de Dados Meteorológicos para Ensino e Pesquisa, 2019. Available online: www.inmet.gov.br/portal/index.php (accessed on 14 January 2020).
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 2012. [Google Scholar]
- Neves, D.P. Parasitologia Humana, 11st ed.; Ateneu: São Paulo, Brasil, 2005. [Google Scholar]
- Shaw, P.J.A. Multivariate Statistics for the Environmental Sciences; Hodder-Arnold: London, UK, 2003. [Google Scholar]
- Piratoba, A.R.A.; Ribeiro, H.M.C.; Morales, G.P.; Gonçalves, W.G. Caracterização de parâmetros de qualidade da água na área portuária de Barcarena, PA, Brasil. Rev. Ambient Água. 2017, 12, 435–456. [Google Scholar] [CrossRef]
- Araujo, M.C.; Oliveira, M.B.M. Monitoramento da qualidade das águas de um riacho da Universidade Federal de Pernambuco, Brasil. Rev. Ambient Água 2013, 8, 247–257. [Google Scholar]
- Esteves, F. Fundamentos de Limnologia, 3rd ed.; Interciência: Rio de Janeiro, Brazil, 2011. [Google Scholar]
- Queiroz, T.M.; Zanini, T.S. Estudo qualitativo da água do riacho Queima pé, em Tangará da Serra—MT. Rev. Eng. Tecnol. 2017, 9, 85–94. [Google Scholar]
- Almeida, O.A. Qualidade da Água de Irrigação; EMBRAPA Mandioca e Fruticultura: Cruz das Almas, Brazil, 2010. [Google Scholar]
- Carvalho, R.A.A.; Martins, P.C.C. Caracterização da atividade de carcinicultura no vale do rio Açu, Rio Grande do Norte, Brasil. HOLOS 2017, 2, 96–107. [Google Scholar] [CrossRef][Green Version]
- ANA. Indicadores de Qualidade—Índice de Qualidade das Águas (IQA). Available online: http://pnqa.ana.gov.br/indicadores-indice-aguas.aspx (accessed on 16 September 2018).
- Ferreira, F.A.S.; De Queiroz, T.M.; Da Silva, T.V.; Andrade, A.C.O. À margem do rio e da sociedade: A qualidade da água em uma comunidade quilombola no estado de Mato Grosso. Saúde Soc. 2017, 26, 822–828. [Google Scholar] [CrossRef]
- Fregonesi, B.M.; Sampaio, C.F.; Ragazzi, M.F.; Tonani, K.A.A.; Segura-Muñoz, S.I. Cryptosporidium and Giardia: Challenges in public water supplies. O Mundo Saúde 2012, 36, 602–609. [Google Scholar] [CrossRef]
- Simões, B.S.; Machado-Coelho, G.L.L.; Pena, J.L.; Freitas, S.N. Condições ambientais e prevalência de infecção parasitária em indígenas Xukuru-Kariri, Caldas, Brasil. Rev. Panam. Salud Publica 2015, 38, 42–48. [Google Scholar]
- Vieira, E.K.; Gomes, E.A. Influência das Estações seca e Cheia na Ocorrência das Parasitoses Intestinais no Município de Tefé, Amazonas, Brasil. Bachelor’s Thesis, Universidade do Estado do Amazonas, Amazonas, Brazil, 2017. [Google Scholar]
- Lopes, A.M.M.B.; Gomes, L.N.L.; Martins, F.C.; Cerqueira, D.A.; Filho, C.R.M.; Sperling, E.V.; Pádua, V.L. Dinâmica de protozoários patogênicos e cianobactérias em um reservatório de abastecimento público de água no sudeste do Brasil. Rev. Eng. Sanitária Ambient. 2017, 22, 25–43. [Google Scholar] [CrossRef]
- Nunes, L.G.P. Qualidade da água no Município de São Mateus: Análise Parasitológica, Microbiológica e Físico-Química. Master’s Thesis, Universidade Federal do Espírito Santo, Espírito Santo, Brazil, 2018. [Google Scholar]
- Zhao, J.; Fu, G.; Lei, K.; Li, Y. Multivariate analysis of surface water quality in the Tree Gorges area of China and implications for water management. J. Environ. Sci. 2011, 23, 1460–1471. [Google Scholar] [CrossRef]
- Boyacioglu, H.; Boyacioglu, H. Water pollution sources assessment by multivariate statistical methods in the Tahtali Basin, Turkey. Environ. Geol. 2008, 54, 275–282. [Google Scholar] [CrossRef]
- Chounlamany, V.; Tanchuling, M.A.; Inoue, T. Spatial and temporal variations of water of a segment Marikina River using multivariate statistical methods. Water Sci. Technol. 2017, 76, 1510–1522. [Google Scholar] [CrossRef]
- Liu, C.W.; Lin, K.H.; Kuo, Y.M. Application of factor analysis in the assessment of groundwater quality in a Blackfoot disease area in Taiwan. Sci. Total Environ. 2003, 313, 77–89. [Google Scholar] [CrossRef]
- Oliveira, T.W.S.; Silva, S.H.P.; Silva, D.F.; Viana, J.F.C.; Firmo, W.C.A.; Silva, M.R.C. Aspecto da sazonalidade nos parâmetros físico-químicos da água dos poços, Raposa—Maranhão, Brasil. Águas Subterrâneas 2019, 33, 1–9. [Google Scholar] [CrossRef]
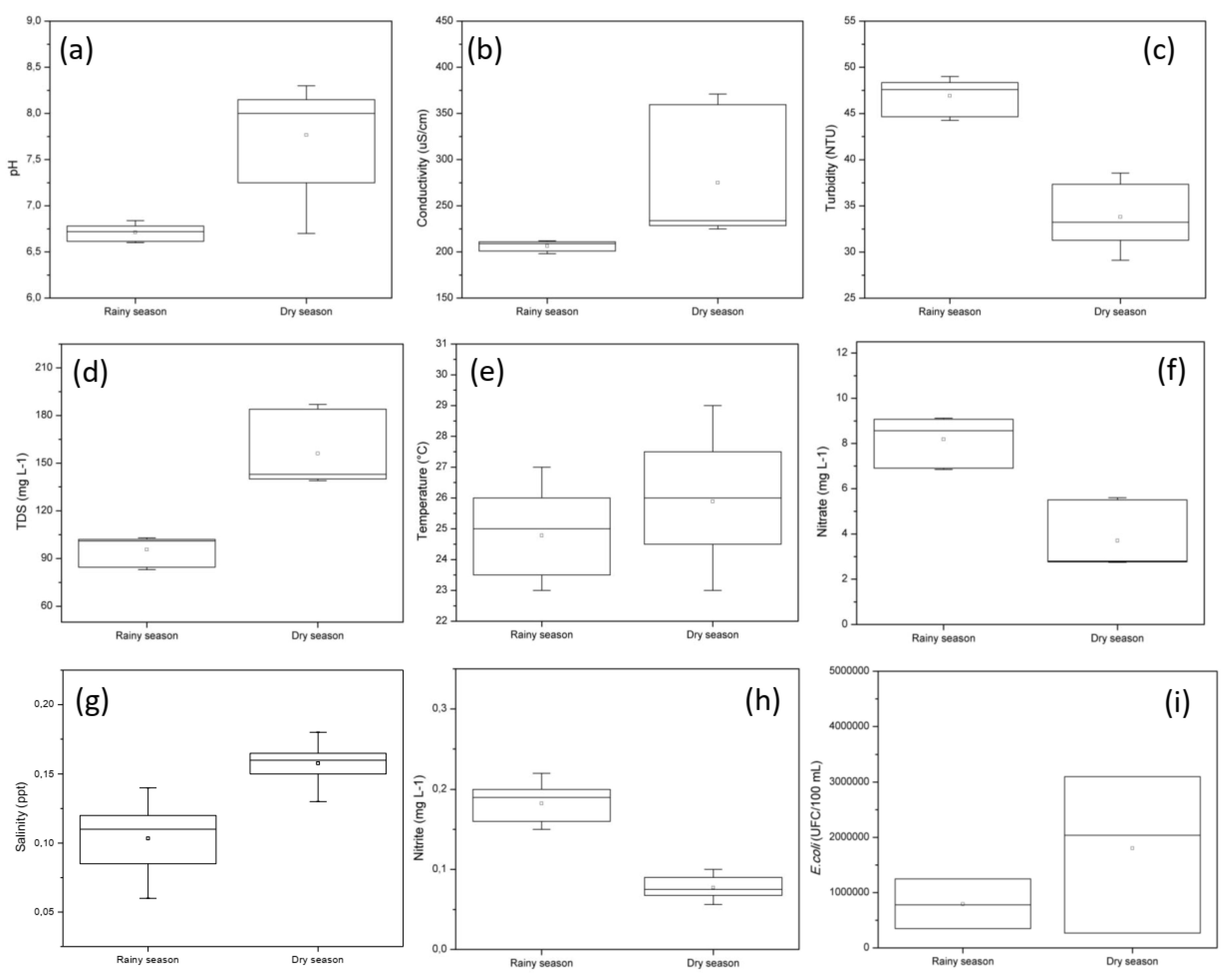
| Parameters | P1 | P2 | P3 |
|---|---|---|---|
| pH | 8.13 ± 0.15a | 8.10 ± 0.10a | 7.06 ± 0.40b |
| EC (µS cm−1) | 363.33 ± 6.65a | 230.33 ± 3.51b | 231.00 ± 6.55b |
| Turb. (NTU) | 32.34 ± 1.50a | 31.30 ± 2.06a | 37.74 ± 0.70b |
| Sal. (ppt) | 0.14 ± 0.010a | 0.15 ± 0.005a | 0.17 ± 0.010a |
| TDS (mg L−1) | 185.00 ± 2.00a | 140.66 ± 2.08b | 142.66 ± 2.51b |
| Temp (°C) | 26.66 ± 2.08a | 27.00 ± 1.00a | 24.00 ± 1.00a |
| NO3− (mg L−1) | 5.54 ± 0.05a | 2.79 ± 0.03b | 2.78 ± 0.02b |
| NO2− (mg L−1) | 0.08 ± 0.015a | 0.07 ± 0.005a | 0.07 ± 0.017a |
| Escherichia coli (UFC/100 mL) | 2.68e−5a | 3.10e−6b | 2.04e−6c |
| Parameters | P1 | P2 | P3 |
|---|---|---|---|
| pH | 6.80 ± 0.040a | 6.61 ± 0.010b | 6.72 ± 0.025c |
| EC (µS cm−1) | 210.66 ± 1.52a | 208.66 ± 2.51a | 200.00 ± 2.00b |
| Turb. (NTU) | 44.51 ± 0.41a | 48.56 ± 0.39b | 47.63 ± 0.06c |
| Sal. (ppt) | 0.10 ± 0.020a | 0.08 ± 0.025a | 0.12 ± 0.015a |
| TDS (mg L−1) | 84.00 ± 1.00a | 102.00 ± 1.00b | 101.00 ± 1.00b |
| Temp (°C) | 24.00 ± 1.00a | 26.33 ± 0.57b | 24.00 ± 1.00a |
| NO3− (mg L−1) | 8.57 ± 0.02a | 9.09 ± 0.03b | 6.89 ± 0.03c |
| NO2− (mg L−1) | 0.20 ± 0.015a | 0.17 ± 0.02a | 0.16 ± 0.020a |
| E. coli (UFC/100 mL) | 7.80e−5a | 1.20e−6b | 3.50e−5c |
| Variables | PC1 * | PC2 * | PC3 * |
|---|---|---|---|
| pH | 0.41 | 0.08 | -0,03 |
| Conductivity | 0.32 | 0.55 | 0,07 |
| Turbidity | −0.42 | 0.06 | −0,17 |
| Salinity | 0.33 | −0.25 | 0,36 |
| TDS | 0.41 | 0.21 | 0,06 |
| Temperature | 0.22 | 0.12 | −0,68 |
| Nitrate | −0.13 | 0.09 | 0,57 |
| Nitrite | −0.40 | 0.20 | −0,06 |
| Escherichia coli | 0.17 | −0.71 | −0,13 |
| Eigenvalue | 5.07 | 1.04 | 1.00 |
| Total variance (%) | 56.40 | 16.30 | 11.10 |
| Cumulative variance (%) | 56.40 | 72.70 | 83.80 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muniz, J.N.; Duarte, K.G.; Braga, F.H.R.; Lima, N.S.; Silva, D.F.; Firmo, W.C.A.; Batista, M.R.V.; Silva, F.M.A.M.; Miranda, R.d.C.M.; Silva, M.R.C. Limnological Quality: Seasonality Assessment and Potential for Contamination of the Pindaré River Watershed, Pre-Amazon Region, Brazil. Water 2020, 12, 851. https://doi.org/10.3390/w12030851
Muniz JN, Duarte KG, Braga FHR, Lima NS, Silva DF, Firmo WCA, Batista MRV, Silva FMAM, Miranda RdCM, Silva MRC. Limnological Quality: Seasonality Assessment and Potential for Contamination of the Pindaré River Watershed, Pre-Amazon Region, Brazil. Water. 2020; 12(3):851. https://doi.org/10.3390/w12030851
Chicago/Turabian StyleMuniz, Jamilly N., Klinger G. Duarte, Fábio H. Ramos Braga, Neuriane S. Lima, Darlan F. Silva, Wellyson C. A. Firmo, Márcia R. V. Batista, Flor M. A. M. Silva, Rita de C. M. Miranda, and Maria Raimunda C. Silva. 2020. "Limnological Quality: Seasonality Assessment and Potential for Contamination of the Pindaré River Watershed, Pre-Amazon Region, Brazil" Water 12, no. 3: 851. https://doi.org/10.3390/w12030851
APA StyleMuniz, J. N., Duarte, K. G., Braga, F. H. R., Lima, N. S., Silva, D. F., Firmo, W. C. A., Batista, M. R. V., Silva, F. M. A. M., Miranda, R. d. C. M., & Silva, M. R. C. (2020). Limnological Quality: Seasonality Assessment and Potential for Contamination of the Pindaré River Watershed, Pre-Amazon Region, Brazil. Water, 12(3), 851. https://doi.org/10.3390/w12030851