Coastal Wetlands: Ecosystems Affected by Urbanization?
Abstract
1. Introduction
2. Materials and Methods
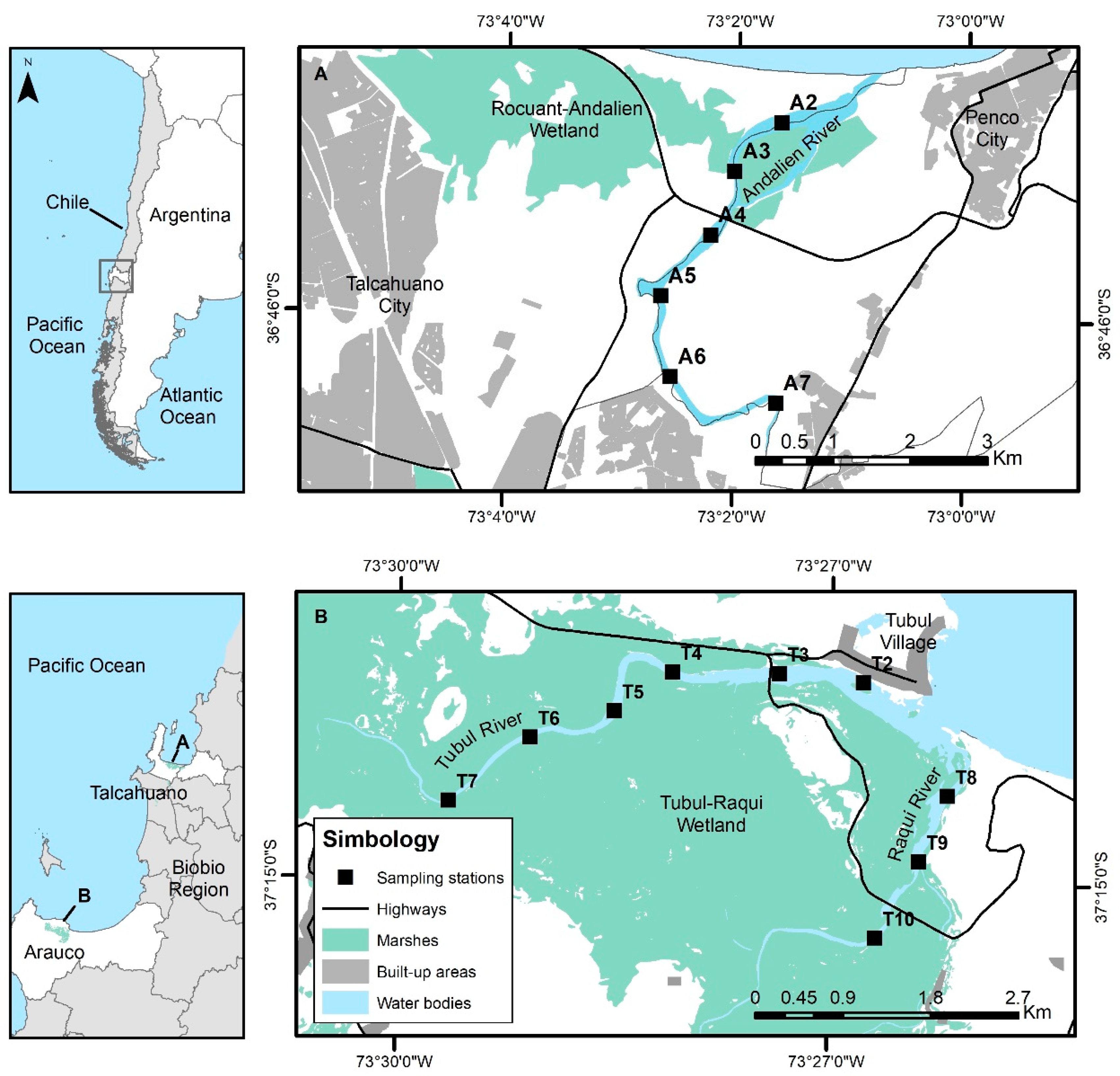
2.1. Study Areas
2.2. Physical-Chemicals Parameters of Wetland Water Quality
2.3. Biological Sampling
2.4. Macroinvertebrate Assemblages
2.5. Statistical Analysis
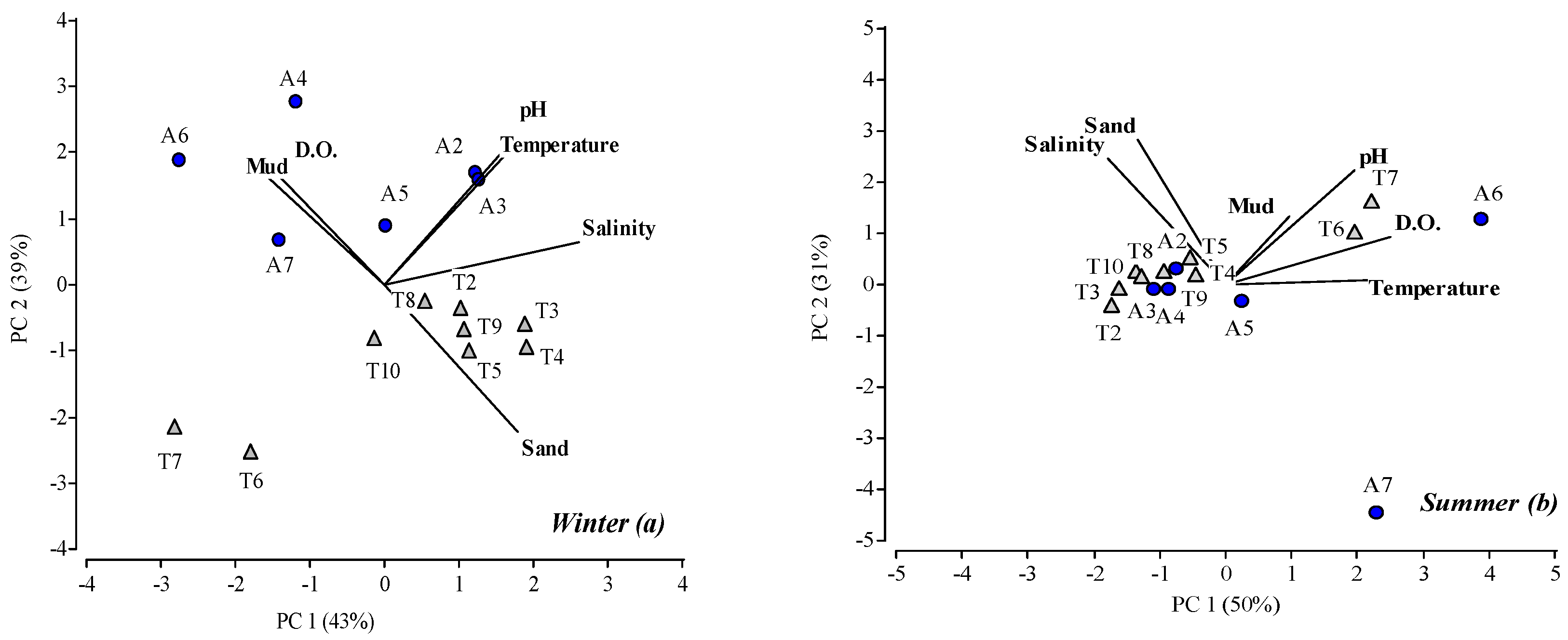
3. Results
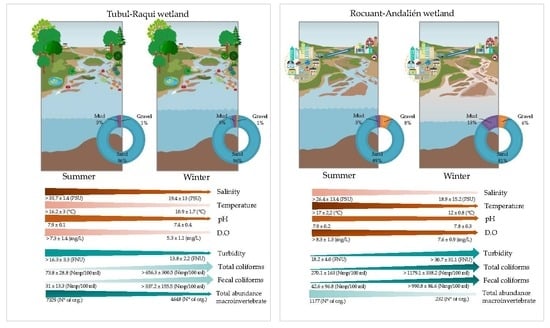
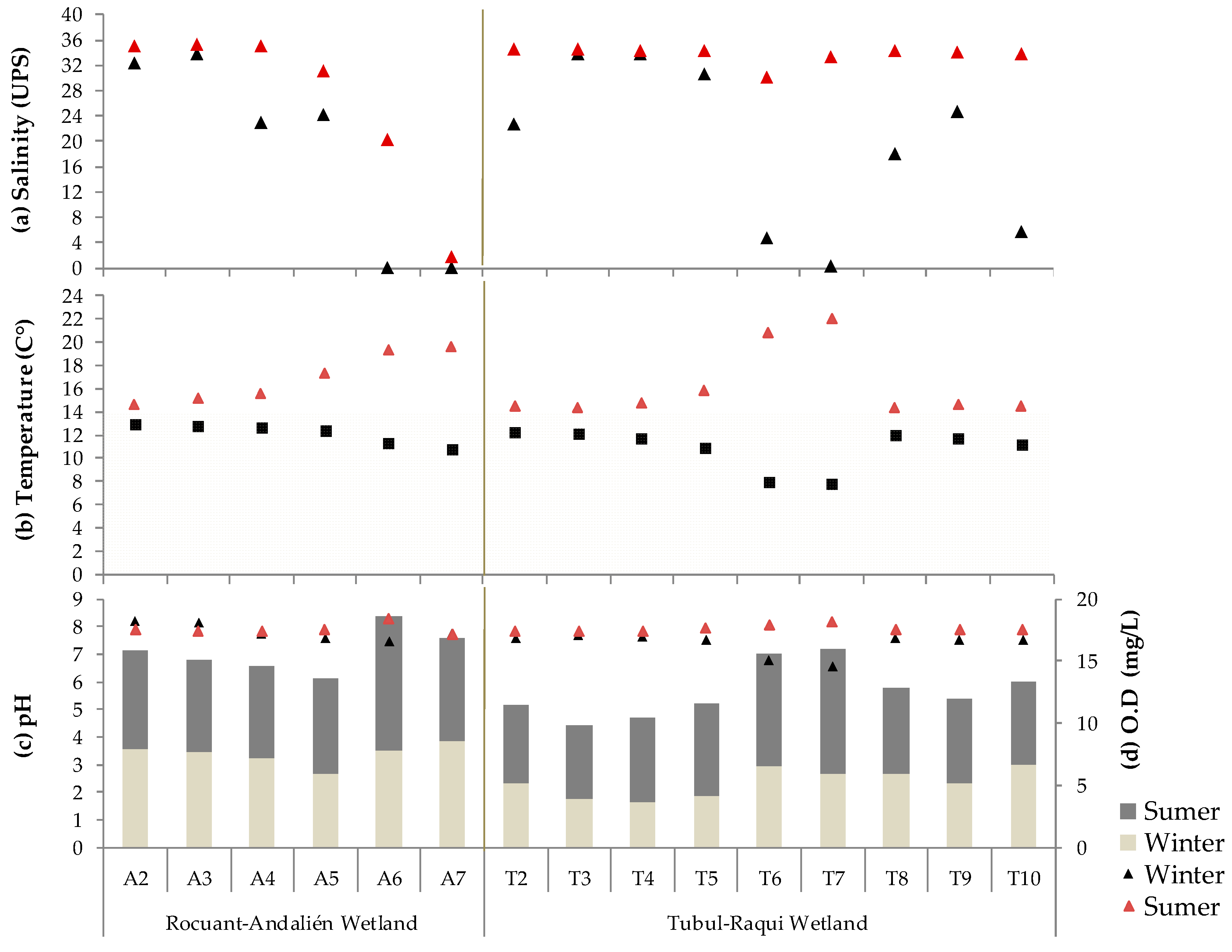
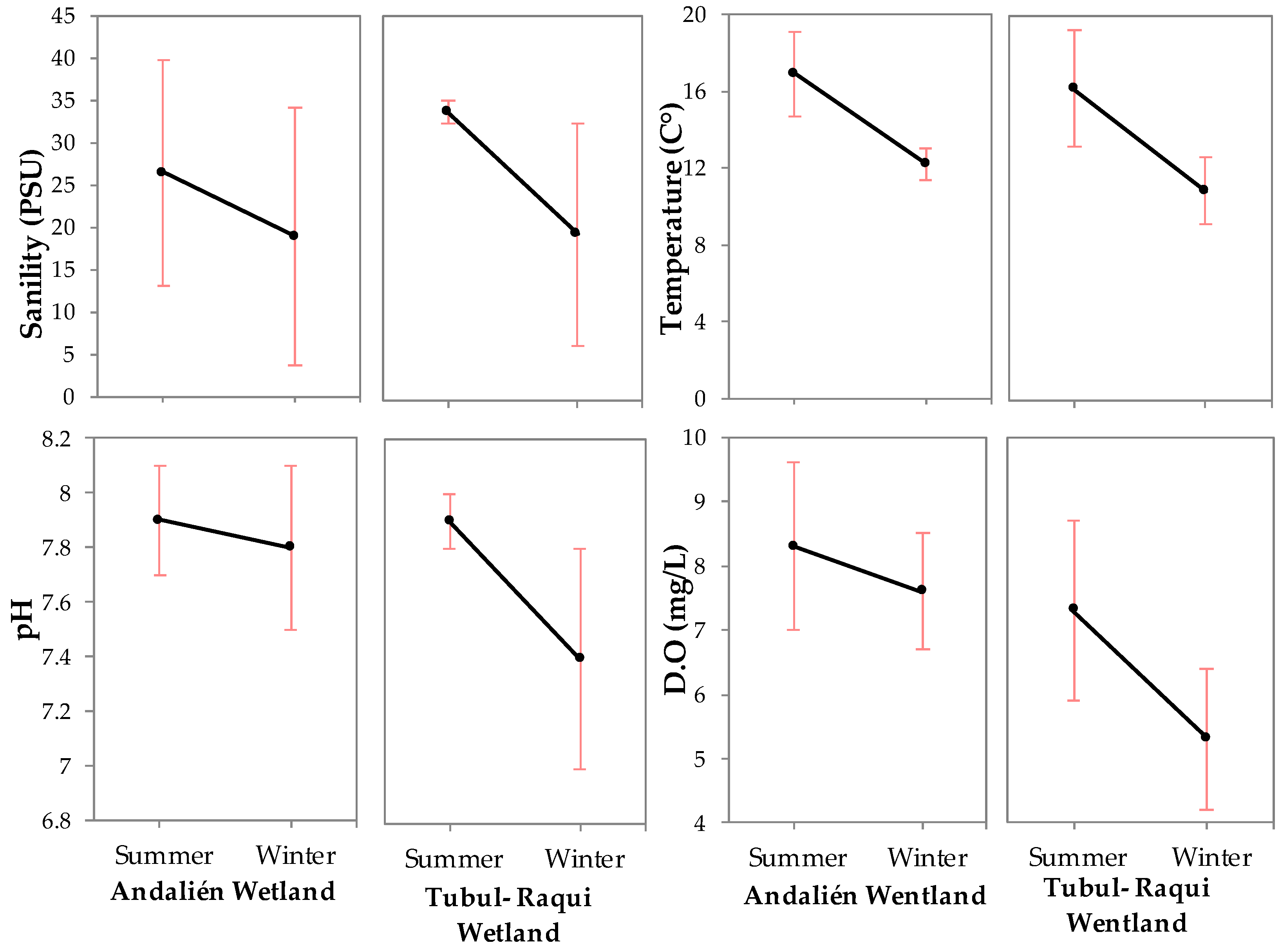
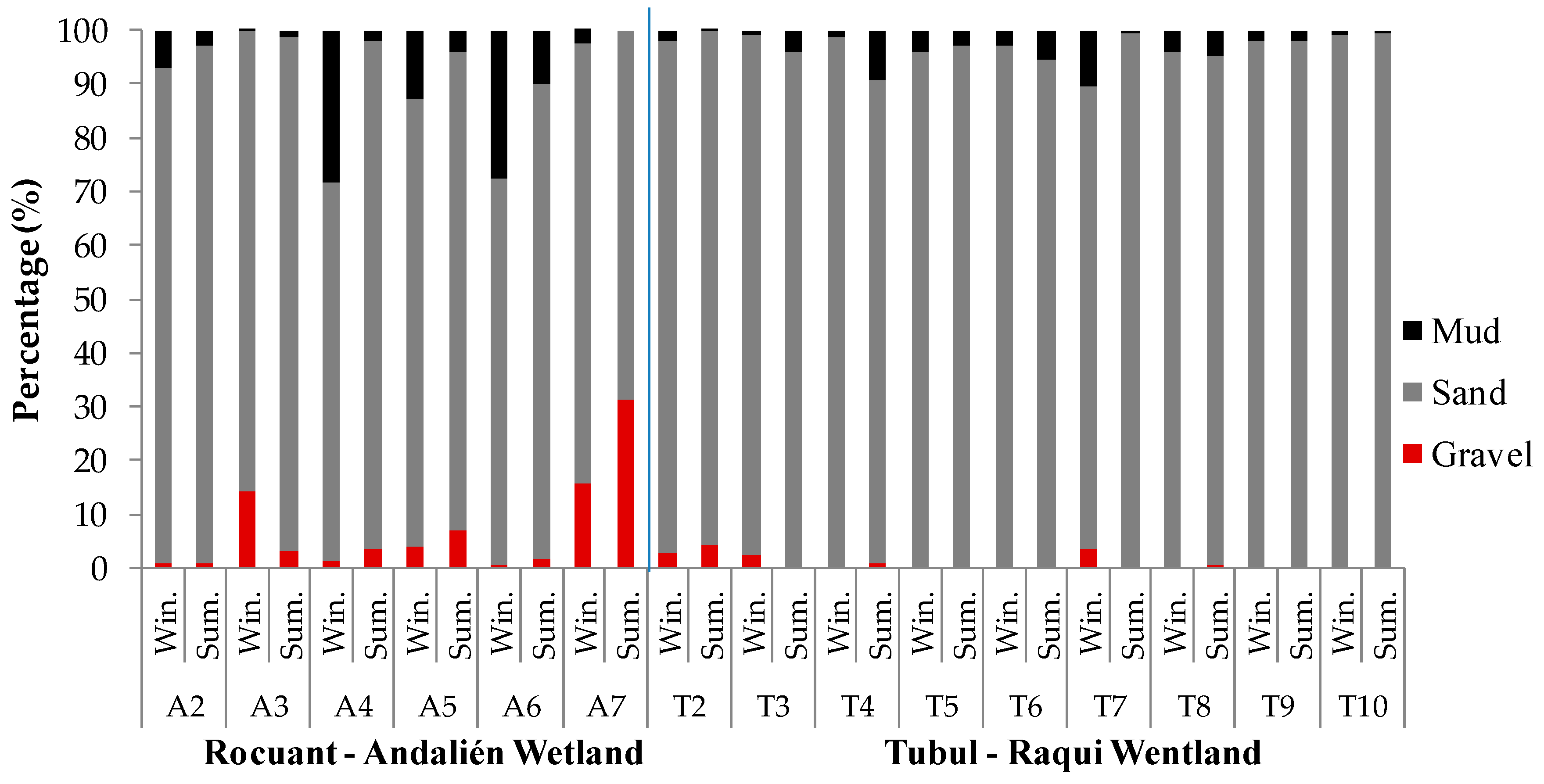
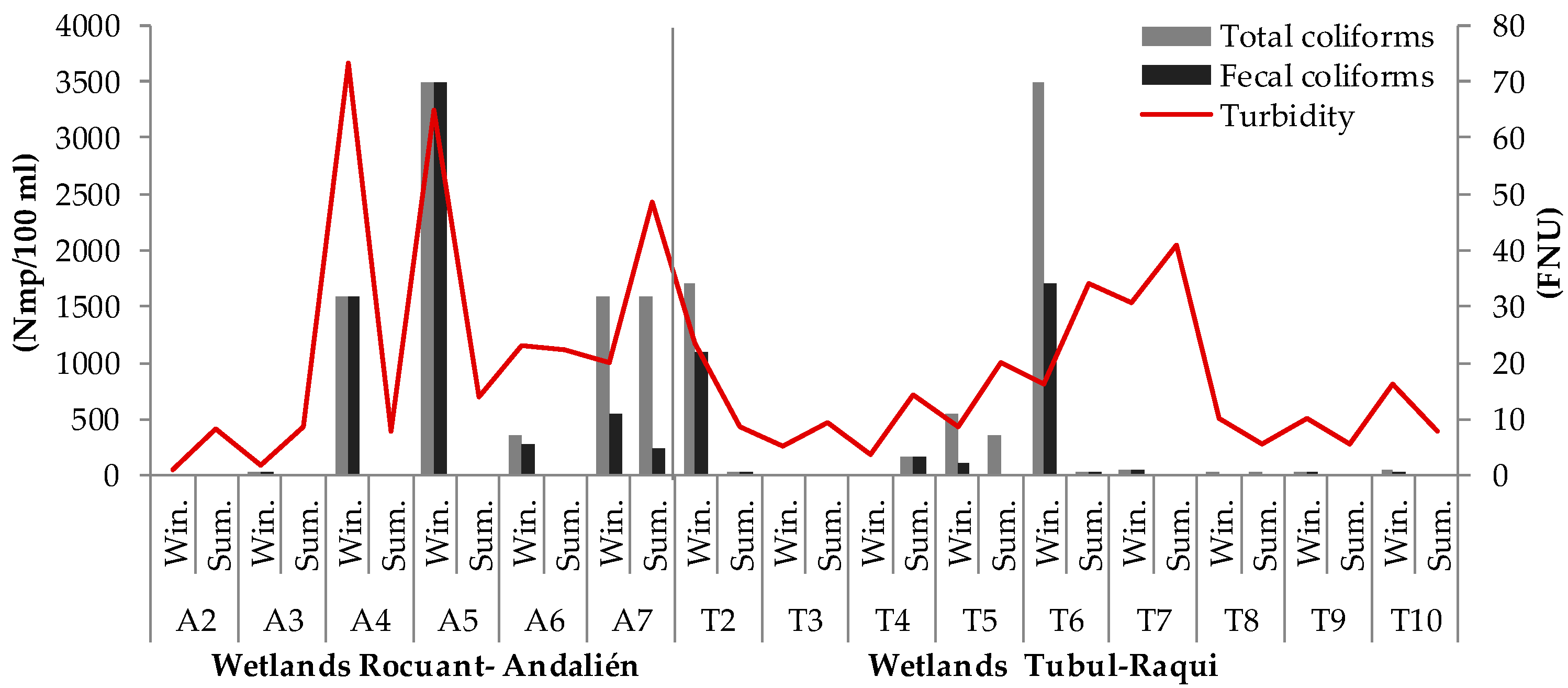
3.1. Physical-Chemical and Bacteriological Parameters of Wetland Water Quality
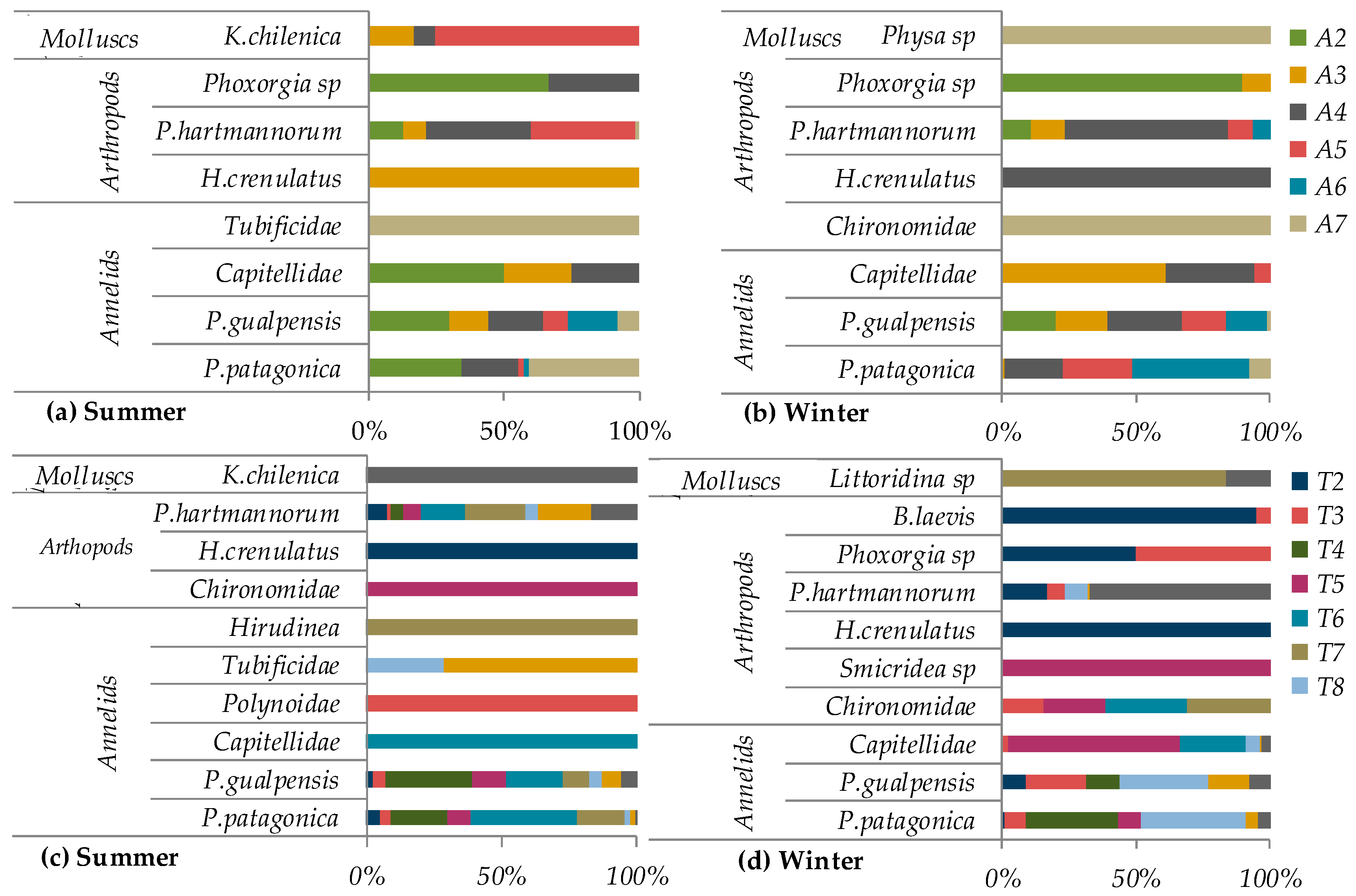
3.2. Biological Parameters
3.3. Macrovertebrate Taxonomic Composition, Abundance and Richness
4. Discussion
4.1. Physochemical Parameters
4.2. Biological Parameters
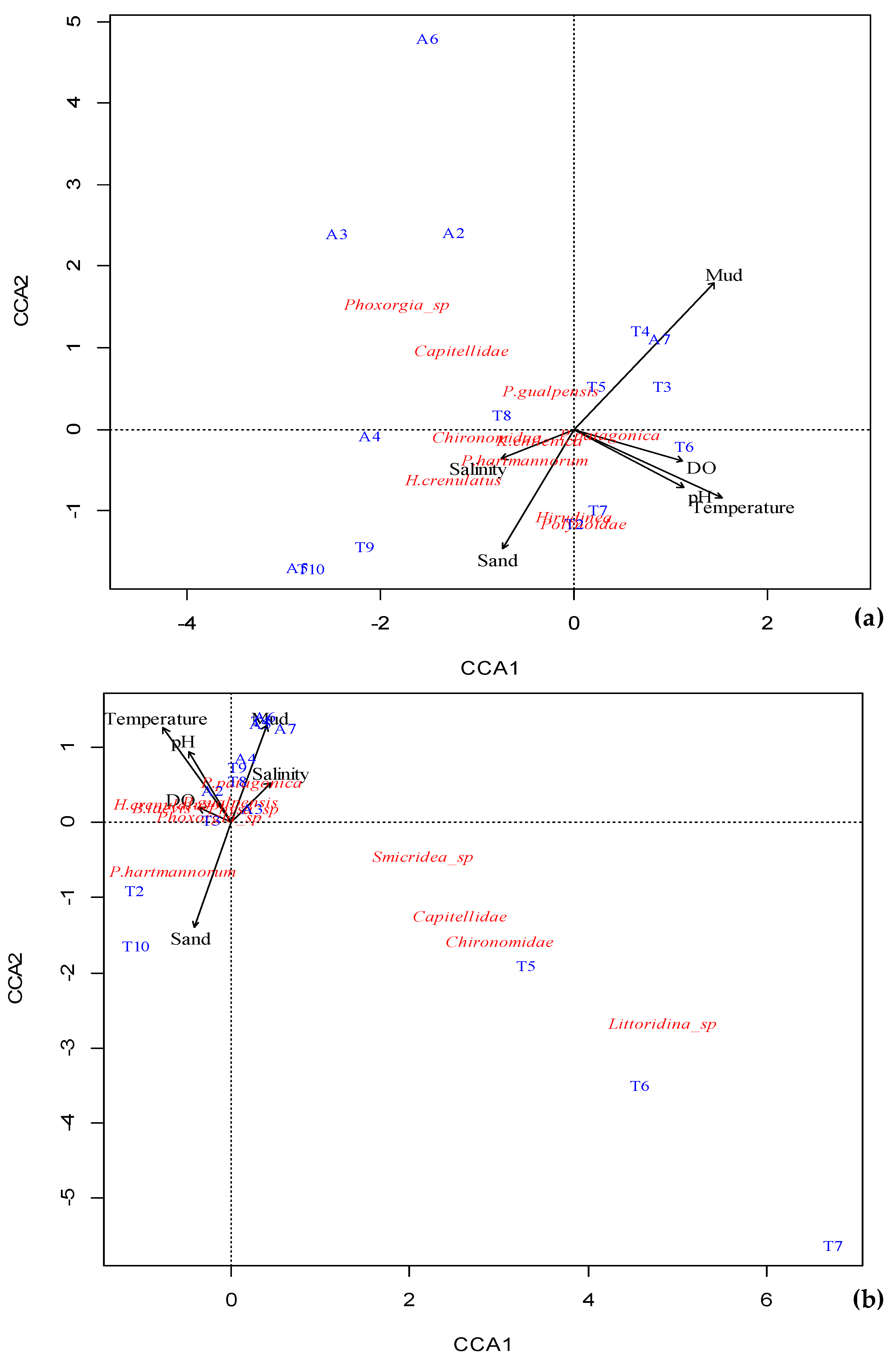
4.3. Macroinvertebrate Assemblages
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Neumann, B.; Vafeidis, A.T.; Zimmermann, J.; Nicholls, R.J. Future Coastal Population Growth and Exposure to Sea-Level Rise and Coastal Flooding—A Global Assessment. PLoS ONE 2015, 10, 1–34. [Google Scholar] [CrossRef]
- Barbier, E.B.; Hacker, S.D.; Kennedy, C.; Koch, E.W.; Stier, A.C.; Silliman, B.R. The value of estuarine and coastal ecosystem services. Ecol. Monogr. 2011, 81, 169–193. [Google Scholar] [CrossRef]
- Basso, G.; Vaudrey, J.M.P.; O’Brien, K.; Barrett, J. Advancing Coastal Habitat Resiliency Through Landscape-Scale Assessment. Coast. Manag. 2018, 46, 19–39. [Google Scholar] [CrossRef]
- Li, Y.F.; Zhang, X.X.; Zhao, X.X.; Ma, S.Q.; Cao, H.H.; Cao, J.K. Assessing spatial vulnerability from rapid urbanization to inform coastal urban regional planning. Ocean Coast. Manag. 2016, 123, 53–65. [Google Scholar] [CrossRef]
- Sengupta, D.; Chen, R.S.; Meadows, M.E. Building beyond land: An overview of coastal land reclamation in 16 global megacities. Appl. Geogr. 2018, 90, 229–238. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Megonigal, J.P. Tidal wetland stability in the face of human impacts and sea-level rise. Nature 2013, 504, 53–60. [Google Scholar] [CrossRef]
- Camacho-Valdez, V.; Ruiz-Luna, A.; Ghermandi, A.; Nunes, P. Valuation of ecosystem services provided by coastal wetlands in northwest Mexico. Ocean Coast. Manag. 2013, 78, 1–11. [Google Scholar] [CrossRef]
- Kennish, M.J.; Meixler, M.S.; Petruzzelli, G.; Fertig, B. Tuckerton Peninsula Salt Marsh System: A Sentinel Site for Assessing Climate Change Effects. Bull. N. J. Acad. Sci. 2014, 58, 1–5. [Google Scholar]
- Alam, M.Z.; Carpenter-Boggs, L.; Rahman, A.M.M.; Haque Miah, R.U.; Moniruzzaman, M.; Qayum, A.; Abdullah, H.M. Water quality and resident perceptions of declining ecosystem services at Shitalakka wetland in Narayanganj city. Sustain. Water Qual. Ecol. 2017, 9–10, 53–66. [Google Scholar] [CrossRef]
- Ericson, J.P.; Vorosmarty, C.J.; Dingman, S.L.; Ward, L.G.; Meybeck, M. Effective sea-level rise and deltas: Causes of change and human dimension implications. Glob. Planet. Chang. 2006, 50, 63–82. [Google Scholar] [CrossRef]
- Leonardi, N.; Camacina, I.; Donatelli, C.; Ganju, N.K.; Plater, A.J.; Schuerch, M.; Temmerman, S. Dynamic interactions between coastal storms and salt marshes: A review. Geomorphology 2018, 301, 92–107. [Google Scholar] [CrossRef]
- Davidson, N.C. How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar. Freshw. Res. 2014, 65, 934–941. [Google Scholar] [CrossRef]
- Li, X.; Bellerby, R.; Craft, C.; Widney, S.E. Coastal wetland loss, consequences, and challenges for restoration. Anthr. Coasts. 2018, 1, 1–15. [Google Scholar] [CrossRef]
- Lin, Q.Y.; Yu, S. Losses of natural coastal wetlands by land conversion and ecological degradation in the urbanizing Chinese coast. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Rojas, C.; Sepúlveda-Zúñiga, E.; Barbosa, O.; Rojas, O.; Martínez, C. Patrones de urbanización en la biodiversidad de humedales urbanos en Concepción metropolitano. Rev. De Geogr. Norte Gd. 2015, 61, 181–204. [Google Scholar] [CrossRef]
- Rojas, C.; Munizaga, J.; Rojas, O.; Martinez, C.; Pino, J. Urban development versus wetland loss in a coastal Latin American city: Lessons for sustainable land use planning. Land Use Policy 2019, 80, 47–56. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Bernal, B.; Hernandez, M.E. Ecosystem services of wetlands, International Journal of Biodiversity Science. Ecosyst. Serv. Manag. 2015, 11, 1–4. [Google Scholar] [CrossRef]
- Tian, B.; Wu, W.T.; Yang, Z.Q.; Zhou, Y.X. Drivers, trends, and potential impacts of long-term coastal reclamation in China from 1985 to 2010. Estuar. Coast. Shelf Sci. 2016, 170, 83–90. [Google Scholar] [CrossRef]
- Wu, W.T.; Zhou, Y.X.; Tian, B. Coastal wetlands facing climate change and anthropogenic activities: A remote sensing analysis and modelling application. Ocean Coast. Manag. 2017, 138, 1–10. [Google Scholar] [CrossRef]
- Wu, W.T.; Yang, Z.Q.; Tian, B.; Huang, Y.; Zhou, Y.X.; Zhang, T. Impacts of coastal reclamation on wetlands: Loss, resilience, and sustainable management. Estuar. Coast. Shelf Sci. 2018, 210, 153–161. [Google Scholar] [CrossRef]
- Strayer, D.L.; Findlay, S.E.G. Ecology of freshwater shore zones. Aquat. Sci. 2010, 72, 127–163. [Google Scholar] [CrossRef]
- Meixler, M.S.; Kennish, M.J.; Crowley, K.F. Assessment of Plant Community Characteristics in Natural and Human-Altered Coastal Marsh Ecosystems. Estuaries Coasts 2018, 41, 52–64. [Google Scholar] [CrossRef]
- Nakano, S.; Murakami, M. Reciprocal subsidies: Dynamic interdependence between terrestrial and aquatic food webs. Proc. Natl. Acad. Sci. USA 2001, 98, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Fierro, P.; Arismendi, I.; Hughes, R.M.; Valdovinos, C.; Jara-Flores, A. A benthic macroinvertebrate multimetric index for Chilean Mediterranean streams. Ecol. Indic. 2018, 91, 13–23. [Google Scholar] [CrossRef]
- Yang, W.; Sun, T.; Yang, Z. Effect of activities associated with coastal reclamation on the macrobenthos community in coastal wetlands of the Yellow River Delta, China: A literature review and systematic assessment. Ocean Coast. Manag. 2016, 129, 1–9. [Google Scholar] [CrossRef]
- Moller, I.; Kudella, M.; Rupprecht, F.; Spencer, T.; Paul, M.; van Wesenbeeck, B.K.; Wolters, G.; Jensen, K.; Bouma, T.J.; Miranda-Lange, M.; et al. Wave attenuation over coastal salt marshes under storm surge conditions. Nat. Geosci. 2014, 7, 727–731. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Temmerman, S.; Skeehan, E.E.; Guntenspergen, G.R.; Fagherazzi, S. Overestimation of marsh vulnerability to sea level rise. Nat. Clim. Chang. 2016, 6, 253–260. [Google Scholar] [CrossRef]
- Ganju, N.K.; Defne, Z.; Kirwan, M.L.; Fagherazzi, S.; D’Alpaos, A.; Carniello, L. Spatially integrative metrics reveal hidden vulnerability of microtidal salt marshes. Nat. Commun. 2017, 8, 1–7. [Google Scholar] [CrossRef]
- Luo, S.X.; Shao, D.D.; Long, W.; Liu, Y.J.; Sun, T.; Cui, B.S. Assessing ‘coastal squeeze’ of wetlands at the Yellow River Delta in China: A case study. Ocean Coast. Manag. 2018, 153, 193–202. [Google Scholar] [CrossRef]
- Feher, L.C.; Osland, M.J.; Griffith, K.T.; Grace, J.B.; Howard, R.J.; Stagg, C.L.; Enwright, N.M.; Krauss, K.W.; Gabler, C.A.; Day, R.H.; et al. Linear and nonlinear effects of temperature and precipitation on ecosystem properties in tidal saline wetlands. Ecosphere 2017, 8, 1–23. [Google Scholar] [CrossRef]
- Guo, H.; Weaver, C.; Charles, S.P.; Whitt, A.; Dastidar, S.; D’Odorico, P.; Fuentes, J.D.; Kominoski, J.S.; Armitage, A.R.; Pennings, S.C. Coastal regime shifts: Rapid responses of coastal wetlands to changes in mangrove cover. Ecology 2017, 98, 762–772. [Google Scholar] [CrossRef] [PubMed]
- Herbert, E.R.; Boon, P.; Burgin, A.J.; Neubauer, S.C.; Franklin, R.B.; Ardon, M.; Hopfensperger, K.N.; Lamers, L.P.M.; Cell, P.A. A global perspective on wetland salinization: Ecological consequences of a growing threat to freshwater wetlands. Ecosphere 2015, 6, 1–43. [Google Scholar] [CrossRef]
- Parker, V.T.; Boyer, K.E. Sea-level rise and climate change impacts on an urbanized pacific coast estuary. Wetlands 2019, 39, 1219–1232. [Google Scholar] [CrossRef]
- Zang, Z.; Zou, X.; Song, Q.; Yao, Y. Analysis of the spatiotemporal correlation between vegetation pattern and human activity intensity in Yancheng coastal wetland, China. Anthr. Coasts 2019, 2, 87–100. [Google Scholar] [CrossRef]
- Coverdale, T.C.; Brisson, C.P.; Young, E.W.; Yin, S.F.; Donnelly, J.P.; Bertness, M.D. Indirect Human Impacts Reverse Centuries of Carbon Sequestration and Salt Marsh Accretion. PLoS ONE 2014, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Meng, W.Q.; Feagin, R.A.; Hu, B.B.; He, M.X.; Li, H.Y. The spatial distribution of blue carbon in the coastal wetlands of China. Estuar. Coast. Shelf Sci. 2019, 222, 13–20. [Google Scholar] [CrossRef]
- Mao, D.H.; Wang, Z.M.; Wu, J.G.; Wu, B.F.; Zeng, Y.; Song, K.S.; Yi, K.P.; Luo, L. China’s wetlands loss to urban expansion. Land Degrad. Dev. 2018, 29, 2644–2657. [Google Scholar] [CrossRef]
- Turner, R.K.; van den Bergh, J.; Soderqvist, T.; Barendregt, A.; van der Straaten, J.; Maltby, E.; van Ierland, E.C. Ecological-economic analysis of wetlands: Scientific integration for management and policy. Ecol. Econ. 2000, 35, 7–23. [Google Scholar] [CrossRef]
- Leigh, C.; Burford, M.A.; Connolly, R.M.; Olley, J.M.; Saeck, E.; Sheldon, F.; Smart, J.C.R.; Bunn, S.E. Science to Support Management of Receiving Waters in an Event-Driven Ecosystem: From Land to River to Sea. Water 2013, 5, 780–797. [Google Scholar] [CrossRef]
- Rojas, O.; Mardones, M.; Martinez, C.; Flores, L.; Sáez, K.; Araneda, A. Flooding in Central Chile: Implications of Tides and Sea Level Increase in the 21st Century. Sustainability 2018, 10, 4335. [Google Scholar] [CrossRef]
- Pauchard, A.; Aguayo, M.; Pena, E.; Urrutia, R. Multiple effects of urbanization on the biodiversity of developing countries: The case of a fast-growing metropolitan area (Concepcion, Chile). Biol. Conserv. 2006, 127, 272–281. [Google Scholar] [CrossRef]
- Rudolph, A.; Ahumada, R. Intercambio de nutrientes entre una marisma con una fuerte carga de contaminantes orgánicos y las aguas adyacentes. Boletín Soc. Biol. Concepción 1987, 58, 151–169. [Google Scholar]
- Correa-Araneda, F.J.; Urrutia, J.; Soto-Mora, Y.; Figueroa, R.; Hauenstein, E. Effects of the hydroperiod on the vegetative and community structure of freshwater forested wetlands, Chile. J. Freshw. Ecol. 2012, 27, 459–470. [Google Scholar] [CrossRef][Green Version]
- Rojas, O.; Zamorano, M.; Sáez, K.; Rojas, C.; Vega, C.; Arriagada, L.; Basnou, C. Social Perception of Ecosystem Services in a Coastal Wetland Post-Earthquake: A Case Study in Chile. Sustainability 2017, 9, 1983. [Google Scholar] [CrossRef]
- Ahumada, R.; Morales, R.; Rudolph, A.; Matrai, P. Efectos del afloramiento costero en la diagénesis temprana de los sedimentos en la Bahía de Concepción, Chile. Boletín De La Soc. De Biol. De Concepción 1984, 55, 135–146. [Google Scholar]
- Valdovinos, C.; Sandoval, N. Cambios ambientales del humedal Tubul-Raqui derivado del alzamiento cosísmico y tsunami, asociado al terremoto Mw 8,8; Informe técnico de la Unidad de Sistemas Acuáticos, Centro de Ciencias Ambientales EULA, Universidad de Concepción: Concepción, Chile, 2011. [Google Scholar]
- Sandoval, N.; Zarges, C.V.; Pablo, O.J.; Vasquez, D. Impacts of coseismic uplift caused by the 2010 8.8 Mw earthquake on the macrobenthic community of the Tubul-Raqui Saltmarsh (Chile). Estuar. Coast. Shelf Sci. 2019, 226, 1–11. [Google Scholar] [CrossRef]
- Martinez, C.; Rojas, O.; Aranguiz, R.; Belmonte, A.; Altamirano, A.; Flores, P. Tsunami risk in caleta Tubul. Biobio region: Extreme scenarios and territorial transformations post-earthquake. Rev. De Geogr. Norte Gd. 2012, 53, 85–106. [Google Scholar] [CrossRef]
- Sistema de Información Territorial CONAF 2016. Available online: https://sit.conaf.cl/ (accessed on 15 February 2020).
- Blott, J. Gradistat version 8.0: A Grain Size Distribution and Statistics Package for the Analysis of Unconsolidated Sediments by Sieving or Laser Granulometer; Kenneth Pye Associates Ltd.: Berkshire, UK, 2010. [Google Scholar]
- Folk, R.L. Citation classic—Brazos river bar—Study in the significance of grain-size parameters. Curr. Contents Phys. Chem. Earth Sci. 1979, 22, 12. [Google Scholar]
- Domínguez, E.; Fernández, H. Macroinvertebrados Bentónicos Sudamericanos, Sistemática y Biología; Foundation Miguel Lillo: Tucumán, Argentina, 2009; p. 656. [Google Scholar]
- Fauchald, K. The Polychaete Worms. Definitions and Keys to the Orders, Families and Genera; Natural History Museum of Los Angeles County, Science Series: Los Angeles, CA, USA, 1977; Volume 28, pp. 1–188. [Google Scholar]
- Osorio, C. Moluscos marinos en Chile: Especies de importancia económica: Guía para su identificación; Universidad de Chile: Santiago, Chile, 2002; p. 211. [Google Scholar]
- Kirwan, M.L.; Mudd, S.M. Response of salt-marsh carbon accumulation to climate change. Nature 2012, 489, 550. [Google Scholar] [CrossRef]
- Mastrocicco, M.; Busico, G.; Colombani, N.; Vigliotti, M.; Ruberti, D. Modelling Actual and Future Seawater Intrusion in the Variconi Coastal Wetland (Italy) Due to Climate and Landscape Changes. Water 2019, 11, 1502. [Google Scholar] [CrossRef]
- Delaney, J.; Shiel, R.J.; Storey, A.W. Prioritising wetlands subject to secondary salinisation for ongoing management using aquatic invertebrate assemblages: A case study from the Wheatbelt Region of Western Australia. Wetl. Ecol. Manag. 2016, 24, 15–32. [Google Scholar] [CrossRef]
- Mabidi, A.; Bird, M.S.; Perissinotto, R. Increasing salinity drastically reduces hatching success of crustaceans from depression wetlands of the semi-arid Eastern Cape Karoo region, South Africa. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Stagg, C.L.; Baustian, M.M.; Perry, C.L.; Carruthers, T.J.B.; Hall, C.T. Direct and indirect controls on organic matter decomposition in four coastal wetland communities along a landscape salinity gradient. J. Ecol. 2018, 106, 655–670. [Google Scholar] [CrossRef]
- Ro, H.M.; Kim, P.G.; Park, J.S.; Yun, S.I.; Han, J. Nitrogen removal through N cycling from sediments in a constructed coastal marsh as assessed by N-15-isotope dilution. Mar. Pollut. Bull. 2018, 129, 275–283. [Google Scholar] [CrossRef]
- Krauss, K.W.; Noe, G.B.; Duberstein, J.A.; Conner, W.H.; Stagg, C.L.; Cormier, N.; Jones, M.C.; Bernhardt, C.E.; Lockaby, B.G.; From, A.S.; et al. The Role of the Upper Tidal Estuary in Wetland Blue Carbon Storage and Flux. Glob. Biogeochem. Cycles 2018, 32, 817–839. [Google Scholar] [CrossRef]
- Karimian, N.; Johnston, S.G.; Burton, E.D. Iron and sulfur cycling in acid sulfate soil wetlands under dynamic redox conditions: A review. Chemosphere 2018, 197, 803–816. [Google Scholar] [CrossRef]
- Van Dijk, G.; Smolders, A.J.P.; Loeb, R.; Bout, A.; Roelofs, J.G.M.; Lamers, L.P.M. Salinization of coastal freshwater wetlands; effects of constant versus fluctuating salinity on sediment biogeochemistry. Biogeochemistry 2015, 126, 71–84. [Google Scholar] [CrossRef]
- Volik, O.; Petrone, R.M.; Wells, C.M.; Price, J.S. Impact of Salinity, Hydrology and Vegetation on Long-Term Carbon Accumulation in a Saline Boreal Peatland and its Implication for Peatland Reclamation in the Athabasca Oil Sands Region. Wetlands 2018, 38, 373–382. [Google Scholar] [CrossRef]
- Rojas, O.; Carrillo, K.S.; Reyes, C.M.; Castillo, E.J. Post-Catastrophe social-environmental effects in vulnerable coastal areas affected by the Tsunami of 02/27/2010 in Chile. Interciencia 2014, 39, 383–390. [Google Scholar]
- Huang, X.D.; Deng, J.; Wang, W.; Feng, Q.S.; Liang, T.G. Impact of climate and elevation on snow cover using integrated remote sensing snow products in Tibetan Plateau. Remote Sens. Environ. 2017, 190, 274–288. [Google Scholar] [CrossRef]
- Meng, W.Q.; He, M.X.; Hu, B.B.; Mo, X.Q.; Li, H.Y.; Liu, B.Q.; Wang, Z.L. Status of wetlands in China: A review of extent, degradation, issues and recommendations for improvement. Ocean Coast. Manag. 2017, 146, 50–59. [Google Scholar] [CrossRef]
- Lauber, C.L.; Hamady, M.; Knight, R.; Fierer, N. Pyrosequencing-Based Assessment of Soil pH as a Predictor of Soil Bacterial Community Structure at the Continental Scale. Appl. Environ. Microbiol. 2009, 75, 5111–5120. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.Q.; Bai, J.H.; Zhang, G.L.; Jia, J.; Wang, W.; Wang, X. Effects of water and salinity regulation measures on soil carbon sequestration in coastal wetlands of the Yellow River Delta. Geoderma 2018, 319, 219–229. [Google Scholar] [CrossRef]
- Kemmitt, S.J.; Wright, D.; Goulding, K.W.T.; Jones, D.L. pH regulation of carbon and nitrogen dynamics in two agricultural soils. Soil Biol. Biochem. 2006, 38, 898–911. [Google Scholar] [CrossRef]
- Rousk, J.; Brookes, P.C.; Baath, E. Contrasting Soil pH Effects on Fungal and Bacterial Growth Suggest Functional Redundancy in Carbon Mineralization. Appl. Environ. Microbiol. 2009, 75, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Debnath, M.; Ray, S.; Ghosh, P.B.; Roy, M. Dynamic modelling of dissolved oxygen in the creeks of Sagar island, Hooghly-Matla estuarine system, West Bengal, India. Appl. Math. Model. 2012, 36, 5952–5963. [Google Scholar] [CrossRef]
- Knight, J.M.; Griffin, L.; Dale, P.E.R.; Sheaves, M. Short-term dissolved oxygen patterns in sub-tropical mangroves. Estuar. Coast. Shelf Sci. 2013, 131, 290–296. [Google Scholar] [CrossRef]
- Baumann, H.; Wallace, R.B.; Tagliaferri, T.; Gobler, C.J. Large Natural pH, CO2 and O-2 Fluctuations in a Temperate Tidal Salt Marsh on Diel, Seasonal, and Interannual Time Scales. Estuaries Coasts 2015, 38, 220–231. [Google Scholar] [CrossRef]
- Dubuc, A.; Waltham, N.; Malerba, M.; Sheaves, M. Extreme dissolved oxygen variability in urbanised tropical wetlands: The need for detailed monitoring to protect nursery ground values. Estuar. Coast. Shelf Sci. 2017, 198, 163–171. [Google Scholar] [CrossRef]
- Bai, J.H.; Zhao, Q.Q.; Lu, Q.Q.; Wang, J.J.; Reddy, K.R. Effects of freshwater input on trace element pollution in salt marsh soils of a typical coastal estuary, China. J. Hydrol. 2015, 520, 186–192. [Google Scholar] [CrossRef]
- Phillips, J.D. Coastal wetlands, sea level, and the dimensions of geomorphic resilience. Geomorphology 2018, 305, 173–184. [Google Scholar] [CrossRef]
- Davis, J.; Sim, L.; Chambers, J. Multiple stressors and regime shifts in shallow aquatic ecosystems in antipodean landscapes. Freshw. Biol. 2010, 55, 5–18. [Google Scholar] [CrossRef]
- de Nijs, M.A.J.; Pietrzak, J.D. Saltwater intrusion and ETM dynamics in a tidally-energetic stratified estuary. Ocean Model. 2012, 49–50, 60–85. [Google Scholar] [CrossRef]
- Grant, S.B.; Sanders, B.F.; Boehm, A.B.; Redman, J.A.; Kim, J.H.; Mrše, R.D.; Chu, A.K.; Gouldin, M.; McGee, C.D.; Gardiner, N.A.; et al. Generation of enterococci bacteria in a coastal saltwater marsh and its impact on surf zone water quality. Environ. Sci. Technol. 2001, 35, 2407–2416. [Google Scholar] [CrossRef]
- O’Mullan, G.D.; Juhl, A.R.; Reichert, R.; Schneider, E.; Martinez, N. Patterns of sediment-associated fecal indicator bacteria in an urban estuary: Benthic-pelagic coupling and implications for shoreline water quality. Sci. Total Environ. 2019, 656, 1168–1177. [Google Scholar] [CrossRef]
- Sanders, B.F.; Arega, F.; Sutula, M. Modeling the dry-weather tidal cycling of fecal indicator bacteria in surface waters of an intertidal wetland. Water Res. 2005, 39, 3394–3408. [Google Scholar] [CrossRef]
- Young, R.B.; Latch, D.E.; Mawhinney, D.B.; Nguyen, T.H.; Davis, J.C.C.; Borch, T. Direct Photodegradation of Androstenedione and Testosterone in Natural Sunlight: Inhibition by Dissolved Organic Matter and Reduction of Endocrine Disrupting Potential. Environ. Sci. Technol. 2013, 47, 8416–8424. [Google Scholar] [CrossRef]
- Bertrán, C.; Arenas, J.; Parra, O. Macrofauna of the lower reach and estuary of Biobío river (Chile): Changes associated to seasonal changes of the river flow. Rev. Chil. De Hist. Nat. 2001, 7, 45–64. [Google Scholar] [CrossRef]
- Jaramillo, E.; Bertran, C.; Bravo, A. Community structure of the subtidal macroinfauna in an estuarine mussel bed in southern Chile. Mar. Ecol. Pubbl. Della Stn. Zool. Di Napoli I 1992, 13, 317–331. [Google Scholar] [CrossRef]
- Fierro, P.; Valdovinos, C.; Arismendi, I.; Diaz, G.; Jara-Flores, A.; Habit, E.; Vargas-Chacoff, L. Examining the influence of human stressors on benthic algae, macroinvertebrate, and fish assemblages in Mediterranean streams of Chile. Sci. Total Environ. 2019, 686, 26–37. [Google Scholar] [CrossRef]
- Gaston, K.J. Global patterns in biodiversity. Nature 2000, 405, 220–227. [Google Scholar] [CrossRef]
- Kovalenko, K.E.; Brady, V.J.; Ciborowski, J.J.H.; Ilyushkin, S.; Johnson, L.B. Functional Changes in Littoral Macroinvertebrate Communities in Response to Watershed-Level Anthropogenic Stress. PLoS ONE 2014, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sabetta, L.; Barbone, E.; Giardino, A.; Galuppo, N.; Basset, A. Species-area patterns of benthic macro-invertebrates in Italian lagoons. Hydrobiologia 2007, 577, 127–139. [Google Scholar] [CrossRef]
- Gleason, J.E.; Rooney, R.C. Aquatic macroinvertebrates are poor indicators of agricultural activity in northern prairie pothole wetlands. Ecol. Indic. 2017, 81, 333–339. [Google Scholar] [CrossRef]
- Begum, S. Macrobenthic Assemblage in the Rupsha-Pasur River System of the Sundarbans Ecosystem (Bangladesh) for the Sustainable Management of Coastal Wetlands. In Coastal Wetlands: Alteration and Remediation. Coastal Research Library; Finkl, C., Makowski, C., Eds.; Springer: Cham, Switzerland, 2017; Volume 21, pp. 751–776. [Google Scholar]
- Lee, S.Y.; Dunn, R.J.K.; Young, R.A.; Connolly, R.M.; Dale, P.E.R.; Dehayr, R.; Lemckert, C.J.; McKinnon, S.; Powell, B.; Teasdale, P.P.; et al. Impact of urbanization on coastal wetland structure and function. Austral Ecol. 2006, 31, 149–163. [Google Scholar] [CrossRef]
- Zhao, N.; Xu, M.Z.; Li, Z.W.; Wang, Z.Y.; Zhou, H.M. Macroinvertebrate distribution and aquatic ecology in the Ruoergai (Zoige) Wetland, the Yellow River source region. Front. Earth Sci. 2017, 11, 554–564. [Google Scholar] [CrossRef]
- Chen, H.S. Establishment and Application of Wetlands Ecosystem Services and Sustainable Ecological Evaluation Indicators. Water 2017, 9, 197. [Google Scholar] [CrossRef]
- Arriagada, L.; Rojas, O.; Arumi, J.L.; Munizaga, J.; Rojas, C.; Farias, L.; Vega, C. A new method to evaluate the vulnerability of watersheds facing several stressors: A case study in mediterranean Chile. Sci. Total Environ. 2019, 77, 114–121. [Google Scholar] [CrossRef]
| Land-Use or Land-Cover Category | Description | Andalién River Watershed | Tubul-Raqui River Watershed | ||
|---|---|---|---|---|---|
| Area (ha) | % | Area (ha) | % | ||
| Native forest | Forests of native species | 11440 | 16 | 2685 | 11.6 |
| Planted forest | Forests of exotic species | 39325 | 55 | 10782 | 46.4 |
| Farmland | Agricultural crops | 7865 | 11 | 298 | 1.3 |
| Meadows and scrubland | Dense herbaceous cover | 10010 | 14 | 6572 | 28.3 |
| Wetland | Coastal area covered by vegetation (salt marshes) and occasionally flooded | 715 | 1 | 2551 | 11.0 |
| Urban and industrial areas | Land covered with structures, urban areas with buildings and infrastructure | 2860 | 4 | 33 | 0.1 |
| Water bodies | Continental water bodies such as streams, lakes and lagoons and marine water bodies such as seas, oceans and estuaries | 215 | 0.3 | 242 | 1.0 |
| Rocuant-Andalién Wetland | Tubul-Raqui Wetland | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Station | Period | Total Abundance | Shannon | Simpson | Margalef | Menhinick | Station | Period | Total Abundance | Shannon | Simpson | Margalef | Menhinick |
| N° of org. | (H’) | (D) | (Dmg) | (Dmn) | N° of org. | (H’) | (D) | (Dmg) | (Dmn) | ||||
| A2 | Summer | 270 | 1.6 | 0.4 | 0.7 | 0.3 | T2 | Summer | 388 | 1.4 | 0.4 | 0.5 | 0.2 |
| Winter | 157 | 1.1 | 0.5 | 0.4 | 0.2 | Winter | 577 | 1.6 | 0.4 | 0.9 | 0.3 | ||
| A3 | Summer | 113 | 1.3 | 0.5 | 0.8 | 0.5 | T3 | Summer | 253 | 1.2 | 0.5 | 0.5 | 0.3 |
| Winter | 144 | 1.3 | 0.6 | 0.8 | 0.4 | Winter | 432 | 1.9 | 0.3 | 1.0 | 0.3 | ||
| A4 | Summer | 282 | 1.6 | 0.4 | 0.9 | 0.4 | T4 | Summer | 1432 | 1.2 | 0.5 | 0.3 | 0.1 |
| Winter | 585 | 1.4 | 0.4 | 0.6 | 0.2 | Winter | 529 | 0.8 | 0.7 | 0.3 | 0.1 | ||
| A5 | Summer | 203 | 1.2 | 0.5 | 0.6 | 0.3 | T5 | Summer | 669 | 1.4 | 0.4 | 0.5 | 0.2 |
| Winter | 517 | 0.8 | 0.7 | 0.5 | 0.2 | Winter | 319 | 1.1 | 0.5 | 0.5 | 0.2 | ||
| A6 | Summer | 95 | 0.3 | 0.9 | 0.4 | 0.3 | T6 | Summer | 2220 | 1.1 | 0.6 | 0.4 | 0.1 |
| Winter | 784 | 0.6 | 0.8 | 0.3 | 0.1 | Winter | 86 | 0.6 | 0.8 | 0.7 | 0.4 | ||
| A7 | Summer | 214 | 1.6 | 0.4 | 0.6 | 0.3 | T7 | Summer | 1234 | 1.4 | 0.4 | 0.4 | 0.1 |
| Winter | 133 | 0.6 | 0.8 | 0.6 | 0.3 | Winter | 10 | 1.4 | 0.4 | 0.9 | 0.9 | ||
| Summer | 1177 | T8 | Summer | 236 | 1.6 | 0.3 | 0.5 | 0.3 | |||||
| Winter | 2320 | Winter | 937 | 1.6 | 0.4 | 0.4 | 0.1 | ||||||
| T9 | Summer | 512 | 1.4 | 0.5 | 0.5 | 0.2 | |||||||
| Winter | 194 | 1.3 | 0.5 | 0.6 | 0.3 | ||||||||
| T10 | Summer | 385 | 1.1 | 0.6 | 0.5 | 0.2 | |||||||
| Winter | 1564 | 0.5 | 0.8 | 0.5 | 0.1 | ||||||||
| Summer | 7329 | ||||||||||||
| Winter | 4648 | ||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novoa, V.; Rojas, O.; Ahumada-Rudolph, R.; Sáez, K.; Fierro, P.; Rojas, C. Coastal Wetlands: Ecosystems Affected by Urbanization? Water 2020, 12, 698. https://doi.org/10.3390/w12030698
Novoa V, Rojas O, Ahumada-Rudolph R, Sáez K, Fierro P, Rojas C. Coastal Wetlands: Ecosystems Affected by Urbanization? Water. 2020; 12(3):698. https://doi.org/10.3390/w12030698
Chicago/Turabian StyleNovoa, Vanessa, Octavio Rojas, Ramón Ahumada-Rudolph, Katia Sáez, Pablo Fierro, and Carolina Rojas. 2020. "Coastal Wetlands: Ecosystems Affected by Urbanization?" Water 12, no. 3: 698. https://doi.org/10.3390/w12030698
APA StyleNovoa, V., Rojas, O., Ahumada-Rudolph, R., Sáez, K., Fierro, P., & Rojas, C. (2020). Coastal Wetlands: Ecosystems Affected by Urbanization? Water, 12(3), 698. https://doi.org/10.3390/w12030698