Abstract
Tracer testing is a mature technology used for characterizing aquatic flow systems. To gain more insights from tracer tests a combination of conservative (non-reactive) tracers together with at least one reactive tracer is commonly applied. The reactive tracers can provide unique information about physical, chemical, and/or biological properties of aquatic systems. Although, previous review papers provide a wide coverage on conservative tracer compounds there is no systematic review on reactive tracers yet, despite their extensive development during the past decades. This review paper summarizes the recent development in compounds and compound classes that are exploitable and/or have been used as reactive tracers, including their systematization based on the underlying process types to be investigated. Reactive tracers can generally be categorized into three groups: (1) partitioning tracers, (2) kinetic tracers, and (3) reactive tracers for partitioning. The work also highlights the potential for future research directions. The recent advances from the development of new tailor-made tracers might overcome existing limitations.
1. Introduction
Tracer tests are one of the most well established techniques for site and process characterizations in the aquatic environment (i.e., in hydrology or hydrogeology). Various additives (e.g., particles, solids, solutes, and gases) and physical quantities (e.g., temperature and pressure) can be applied as tracers for interpreting hydraulic transport properties and/or reactive processes in the aquatic environment [1,2,3,4]. Some basic hydraulic properties, such as flow velocity or porosity, can be obtained by tracer tests using conservative (non-reactive) tracer compounds. The combination of a conservative tracer with at least one reactive tracer is commonly applied in order to assess additional system parameters, such as residual saturation [5,6], microbial activity [7,8], or temperature distribution [9,10]. The unique features of reactive tracers could provide valuable information on physical, chemical, and/or biological properties of the hydrological system which surpasses the capability of conservative tracers.
The application potential for tracers within the scope of advanced reservoir management, such as geothermal power generation or carbon capture and storage, has triggered the development of new tracers and tracer techniques in the past decades [11,12]. Reactive tracers used to detect specific properties and processes in the aquatic environment must generally either have distinctive physicochemical properties (e.g., sorption) or undergo specific reactions such as hydrolysis. To identify the most suitable tracer compounds for a specific system or problem, a thorough understanding of the physicochemical properties and their chemically reactive behavior in the probed system is a prerequisite.
The main objective of this overview article is to present a systematic review of existing and proposed reactive solute tracers based on current research advances conducted in different scientific fields. The focus of this work is on chemical/artificial tracers which are intentionally introduce in the tracer tests. For each subclass of tracer, the underlying process, their key properties, and possible target parameters/applications are described. Furthermore, the potential areas for the future development and exploitation of new reactive tracers are elaborated. Hereby, the new approach of producing tailor-made reactive tracers may break down currently existing limitations on the investigation potential of commercially available compounds.
2. Definition and Theoretical Background
2.1. Definition
A tracer is defined herein as a distinguishable chemical compound which is deliberately added to an aquatic system having a temporally and spatially well-known input function (e.g., pulse injection). The respective system property or information of interest is derived based on the relation of the input function to the observed response function (breakthrough curve) within the investigated system.
Two general tracer types can be defined based on the degree of interaction with the systems. First, conservative tracers show virtually no interaction with the reservoir materials, and thus they flow passively with the carrier fluids at their velocity. Furthermore, they do not suffer any chemical or biological processes. This implies that these tracers are inert under reservoir conditions. The second type of tracers can be summarized as reactive tracers. The interpretation of reactive tracers relies on their known properties, physicochemical or chemical behavior during the transport. Reactive tracers are compounds that undergo a chemical reaction or physicochemical interaction processes in a predictable way under specific boundary conditions existing in the investigated system. Consequently, using the particular features of reactive tracers could provide unique information on physicochemical properties and/or water chemistry of the hydrological system far beyond the capability of conservative tracers.
Traditionally, tracer tests were conducted using conservative tracers. These tracers can provide general physical and hydraulic parameters of the system (e.g., porosity, dispersivity, or arrival time). In order to derive these parameters with great accuracy, the compounds are desired to behave ideally. The properties of an ideal tracer are well established [2]; they (1) behave conservatively (e.g., are transported with water velocity, not degradable), (2) have a low background concentration in the system, (3) are detectable in very low concentrations, and (4) have low or no toxicological environmental impact. Nevertheless, all solute tracers are influenced to some degree by physical, chemical, and/or biological processes. This means that completely ideal tracers do not exist in reality. Therefore, some knowledge of the investigated system is required beforehand to verify the practicality of the tracer behavior and thus to avoid test failure.
2.2. Conservative Tracer Transport versus Reactive Tracer Transport
The transport behavior of a tracer compound in the aquatic environment is affected by several physical and chemical processes. These processes result in spatial and/or temporal concentration changes of the introduced tracer during its transport, which are reflected in the system response function (e.g., breakthrough curve ). Tracer transport is commonly described based on the principle of mass conservation by means of the advection-dispersion-reaction model in the three-dimensional form as follows:
 where is the retardation factor, is the tracer concentration, is the time, is the average pore water velocity, is the hydrodynamic dispersion tensor (including mechanical dispersion and molecular diffusion), and is the source/sink term accounting for the tracer transformation (degradation/generation).
where is the retardation factor, is the tracer concentration, is the time, is the average pore water velocity, is the hydrodynamic dispersion tensor (including mechanical dispersion and molecular diffusion), and is the source/sink term accounting for the tracer transformation (degradation/generation).

As described above, tracer transport in water can be classified as conservative or reactive according to its interaction within the system to be studied. A conservative tracer does not interact or alter during the transport, and thus the concentration is not changed by processes other than dilution, dispersion, and partial redirection. As such, conservative tracers are expected to mimic the transport of water without retardation and transformation. They underlie only the purely hydrodynamic transport processes: advection, diffusion, and dispersion (as terms 2 and 3 in Equation (1). It should be noted that various types of mixing always exist which should be interpreted with caution as the mixing or other dilution processes may influence the results of tracer experiments [13,14,15,16]. Therefore, conservative tracers are generally used to investigate hydraulic properties (e.g., tracking connectivities, flow pathways), analyzing travel times and flow velocities, determining recharge and discharge, and estimating hydromechanical properties (e.g., dispersivity, porosity). Common examples of conservative tracers under ambient temperatures are major anions such as bromide [17,18], stable isotopes such as 2H and 18O [4,19], dye tracers such as uranine [20,21,22], and rhodamine WT [23,24,25,26,27].
Apart from hydrodynamic transport processes, reactive tracers additionally underlie physical, chemical, and/or biological processes during their transport (terms 1 and 4 in Equation (1)). The implementation of reactive tracers with identical and well understood interactions or reactions could implicitly provide unique information on physicochemical aquifer properties (e.g., sorption capacity), water chemistry (e.g., redox condition, pH, ion concentrations), and other influencing parameters (e.g., temperatures, microbial activity) [28,29,30].
In order to benefit from the selective and process specific nature of reactive tracers, it is a prerequisite to combine them with at least one conservative reference tracer by performing a multitracer experiment to account for the purely hydrodynamic transport processes that affect both tracer types in the same way. Consequently, the reactive processes can be identified and quantified. The intended information from the tracers is gained by comparing the concentration versus time curves (breakthrough curves) of the reactive tracers with the conservative tracers (reference). This can be illustrated by the schematic breakthrough curves for a simulated tracer test having a pulse input function (Figure 1). The time shift and/or the reduction of the peak area (tracer mass) of the breakthrough curves indicate retardation and/or degradation, respectively. Measured breakthrough curves can be inversely interpreted using analytical or numerical models to estimate the values of controlling parameters, such as the distribution coefficient for the sorption process, the decay rate for the sorption process, or the decay rate for the biodegradation process.
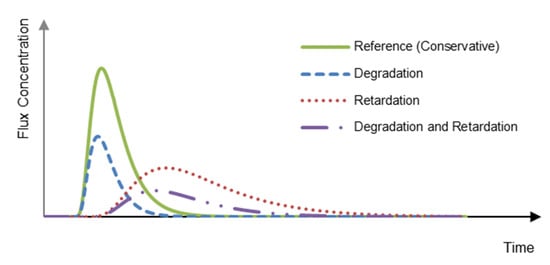
Figure 1.
Schematic breakthrough curves for conservative tracer and reactive tracers after a pulse injection.
3. Types of Reactive Tracers
A generalized classification of currently existing reactive tracers and proposed reactive tracer concepts, including their required properties, possible applications, and processes is provided. Depending on their physical, chemical, and/or biological behavior, three major subgroups are distinguished (Table 1):

Table 1.
Classification of reactive tracers.
- Partitioning tracers: These types are based on the partitioning equilibrium between two immiscible phases or at their interfaces (fluid–solid, fluid–fluid) leading to a retardation relative to the conservative tracer remaining in (one) fluid phase.
- Kinetic tracers: These types are non-equilibrium tracers in which only the reaction kinetics are used for the parameter determination. As a result of the tracer reaction, the tracer signals are decreasing (parent compound) or increasing (daughter compound) with time (degradation). These tracers usually do not show retardation (no partitioning).
- Reactive tracers for partitioning: These tracers are a hybrid form of the preceding tracers, containing features of both: chemical reaction (degradation) of the parent compound and subsequent partitioning (retardation) of the daughter products.
3.1. Equilibrium Tracers
3.1.1. Fluid–Solid (Sorbing Tracers)
Sensitive for Uncharged Surfaces
A tracer compound sensitive for uncharged surfaces undergoes hydrophobic sorption onto uncharged sites of the sorbent (e.g., soil, aquifer material), particularly organic matter. Hydrophobic sorption is the result from a weak solute-solvent interaction coming from a decrease in entropy of the solution and can be explained by general interactions between sorbate and sorbent, e.g., van-der-Waals forces (dipole and/or induced-dipole interactions) [31]. The organic carbon content of the aquifer material generally correlates with the sorptivity and thus the retardation of a neutral (uncharged) organic compound [32,33,34]. Therefore, it is conceivable that substances, which are sensitive to uncharged surfaces, have the potential to determine the of a system from their observed retardation factor ( assuming a linear sorption isotherm:
where ρ is bulk density, is effective porosity, and is the sorption coefficient. depends primarily on the hydrophobicity of the tracer molecules, typically characterized by the n-octanol-water partition coefficient () and the of the geological materials. From of the tracer compound, for a particular system can be estimated. According to the literature [35,36,37] can empirically be related to the organic carbon normalized sorption coefficient ( in the form:
where a and b are empirical parameters.
Thus, from known and determined , the average between the injection and observation points can be estimated. By selecting non-ionic compounds with moderate values between 1 and 3 (1H-benzotriazole, carbamazepine, diazepam, and isoproturon) from formerly published column experiments by Schaffer et al. [38,39] using correlation factors for non-hydrophobic compounds after Sabljic et al. (1995), the observed values of the columns agree very well with the independently measured ones from the bulk using total organic carbon measurements. Despite the relatively large uncertainty regarding the chosen values, all deviations of the absolute values between the measured and calculated are within one order of magnitude (less than factor 5).
To the extent of our knowledge, this tracer type has not yet been explicitly proposed, and therefore their potential could be further investigated. Some promising examples include 8:2 fluorotelomer alcohol [40], short-chained alkyl phenols [41], or pharmaceutical compounds [42,43,44].
Sensitive for Charged and Hydrophilic Surfaces
A tracer compound sensitive for charged surfaces undergoes ionic sorption between a charged moiety of a tracer molecule and an oppositely charged surface of the sorbent (e.g., soil, aquifer material). In this case, there is a strong electrostatic interaction (e.g., ion exchange, hydrogen bonding, or surface complexation) between tracer sorbate and sorbent.
Retardation of a solute due to ion sorption on natural solids can be related either to a sorbent mass (Equation (2)) or to its surface sensitivity to the surface area ( to volume ( ratio if the sorption coefficient is known [45]:
These tracers are required to be water soluble, ionized (electrically charged), and can be organic or inorganic substances. The selection of tracers for this application is based on the surface charge of the sorbents. Further, the pH condition strongly influences the charge states of organic compounds (e.g., bases, acids, and ampholytes) and the sorbent’s surface [33]; thus, pH and the point of zero charge of the surface should be considered before selecting a tracer compound.
Many laboratory tests have been conducted to demonstrate the feasibility of charged surface tracers to interrogate the surface area, e.g., using safranin [46], lithium [47,48,49], and monoamines [50]. A couple of field tests have also demonstrated the potential use of charged surface tracers for investigating the surface area, e.g., using safranin [51] and caesium [52,53]. Furthermore, this tracer type has the potential to estimate the ion exchange capacity of sediments [54].
3.1.2. Fluid–Fluid
The fluid-fluid tracers summarize liquid–liquid tracers and liquid–gas tracers due to the similarity in the underlying processes and applications.
Volume Sensitive Tracers
A volume sensitive tracer is a compound that partitions between two immiscible fluid phases (liquid–liquid or liquid–gas). A different solubility in the two fluid phases leads to the specific phase distribution and results in a retardation of the tracer. Volume sensitive tracers are very useful in estimating the volume of the immobile phase (residual saturation). For example, one common application of this type of tracer is to characterize the source zone of non-aqueous phase liquids (NAPLs) for contaminated sites. Another popular use is to evaluate the effectiveness of treatment techniques before and after the remediation of NAPLs, thereby obtaining independent estimates on the performance of the cleanup. This tracer can also be used to identify residual gas or supercritical fluid phases, such as in carbon capture and storage applications. When sorption onto solids is negligible, the retardation factor is a function of the average residual saturation within the tracer flow field [55,56]:
where is the partition coefficient between two fluid phases.
A large number of laboratory experiments and field-scale tests have been conducted to detect NAPL contaminations since the 1990s. The most commonly applied volume sensitive tracers are alcohols of varying chain length, such as 1-hexanol [57,58,59,60], 1-pentanol and 1-heptanol [61,62,63], 2-ethyl-1-butanol [5,61,64], 6-methyl-2-heptanol [65,66], 2,2-dimethyl-3-pentanol [56,65,66], 2,4-dimethyl-3-pentanol [57,63,64,67,68,69,70], substituted benzyl alcohols [6,71] and fluorotelomer alcohols [72]. Additionally, sulfur hexafluoride (SF6) [73,74,75,76,77], perfluorocarbons [61,78], radon-222 [79,80,81], and fluorescent dyes (e.g., rhodamine WT, sulforhodamine B, and eosin) [82] have also been suggested for use as volume sensitive tracers. Recently, the noble gases krypton and xenon were applied successfully in the determination of the residual CO2 saturation [83,84,85,86,87].
Interface Sensitive Tracers
An interface sensitive tracer is a compound that undergoes the accumulation (adsorption) at the interface between two immiscible fluids, typically liquid–liquid or liquid–gas, leading to the retardation of the tracer. The magnitude of adsorption at the interface is controlled by the physicochemical properties of tracer compounds and by the interfacial area, particularly the size of the specific fluid–fluid interfacial area and the interfacial adsorption coefficient . The retardation factor ( defined through porous media follows [88,89]:
where is the specific interfacial area, is the volumetric water content, and is the interfacial adsorption coefficient (ratio between the interfacial tracer concentration in the sorbed phase at the interface () and the fluid () at equilibrium).
The desired compounds for this tracer class are amphiphilic molecules (containing both hydrophobic and hydrophilic groups). Information on fluid-fluid interfacial areas, along with residual saturation (assessed by volume sensitive tracers) assists the understanding of the fate and transport of contamination in the systems.
One of the most popular interface sensitive tracers that have been successfully tested in laboratory and field scales is the anionic surfactant sodium dodecylbenzene sulfonate [67,69,88,90,91,92,93,94,95,96,97,98,99,100]. Further potential arises for other ionic and non-ionic surfactants (e.g., marlinat [101], 1-tetradecanol [102,103], sodium dihexylsulfosuccinate [104]) and for cosurfactants (e.g., n-octanol and n-nonanol [105]).
3.2. Kinetic Tracers
3.2.1. One Phase
Degradation Sensitive Tracers
Degradation sensitive tracers are compounds that undergo biotic and/or abiotic transformations. Depending upon the nature of the tracer specific (reaction controlling boundary conditions), chemical and/or biological characteristics of the flow system can be investigated. Information on the decay mechanism and the equivalent kinetic parameters is a prerequisite for their successful application. The decay mechanism is usually desired to follow a (pseudo) first order reaction to limit the number of required kinetic parameters and to avoid ambiguity. In addition, other influencing factors on kinetics should be considered before application (e.g., pH, light, and temperature). The reaction rate constant can be estimated by measuring the extent of tracer loss of the mother compound or the associated increase of a transformation product along the flow path.
This type of tracer has been studied and tested in field-scale experiments over the past 20 years. Their main purpose is to determine microbial metabolic activity (natural attenuation processes) and/or to assess redox conditions. Numerous redox-sensitive tracers have been applied for laboratory and field scale investigations, such as inorganic electron acceptors (e.g., O2, NO3−, SO42−, CO32−) [106,107,108,109,110,111,112,113,114,115,116], organic electron donors (e.g., low-molecular weight alcohols and sugars [117] and benzoate [118,119,120]), or the organic electron acceptor resazurin [8,121,122,123,124,125,126,127,128].
Thermo-Sensitive Tracers
Thermo-sensitive tracers are compounds undergoing chemical reactions that are well-defined and temperature driven, such as hydrolysis [129,130,131] or thermal decay [132,133]. Prior knowledge on their reaction mechanisms is required for each specific thermo-sensitive tracer. To avoid ambiguity, reactions following (pseudo) first order reaction are desired, and the reaction speed (expressed by the reaction rate constant )) is preferred to be solely controlled by temperature. For these reactions, the dependence of temperature on is the essential factor for estimating the thermo-sensitivity expressed by Arrhenius law:
where is the pre-exponential factor, is the activation energy, and is the ideal gas constant.
By knowing the corresponding kinetic parameters, the equivalent temperature and the cooling fraction can be obtained [134]. references the thermal state of a probed reservoir relative to an equivalent system having isothermal conditions, whereas has the potential to further estimate a spatial temperature distribution of the investigated system.
A typical application of these tracers is to investigate the temperature distribution of a georeservoir. The first field experiments using ester compounds (ethyl acetate and isopentyl acetate), however, were unable to determine a reservoir temperature [135,136,137]. The failure of the studies was attributed to the poor determination of pH dependence and the lower boiling point of the tracer compounds compared to the reservoir temperature leading to vaporization. New attempts demonstrated the successful application in the laboratory [9] and in the field [138]. Other studies using classical tracers like fluorescein [139] or Amino G [132,133] were able to identify the reservoir temperatures. Currently, extensive research has been conducted to study structure-related kinetics of defined thermo-sensitive reactions with promising results [9,10,130,131,134].
3.2.2. Two Phases
Kinetic Interface Sensitive (KIS)
KIS tracers are intended to be dissolved or mixed with a non-aqueous carrier fluid (e.g., supercritical CO2 [11]) and injected into the reservoir. The underlying process is an interface-sensitive hydrolysis reaction at the interface between the aqueous and the non-aqueous phase. Here, the tracer saturates the interface of the evolving plume due to interfacial adsorption and reacts irreversibly with water (hydrolysis with first-order kinetics). Due to the constant (adsorbed) concentration of the reactant at the interface, the reaction kinetics is simplified to (pseudo) zero order kinetics. The formed reaction products are monitored in the water phase.
In order to have minimal partitioning into the polar water phase, the potential tracers have to be non-polar in conjunction with high values. Furthermore, the KIS tracer reaction kinetics has to be adapted to the characteristics of the reservoir and the interfacial area dynamics in order to resolve the plume development. In contrast to the parent compound, at least one of the reaction products has to be highly water soluble resulting in low or even negative values. Thus, back-partitioning into the non-aqueous phase can be avoided.
This class of reactive tracers was originally intended to characterize the fluid–fluid interfacial area (e.g., between supercritical CO2 and formation brine during CO2 storage experiments [140]). Currently, only limited laboratory experiments with the supercritical CO2 analogue fluid n-octane are available [11].
3.3. Reactive Tracers for Partitioning
A reactive tracer for partitioning is a compound comprising the features of both partitioning tracers and kinetic tracers. This type of tracer undergoes in-situ decay of the parent tracer compounds with subsequent partitioning of the daughter compounds. The concentration of both parent and daughter compounds are determined. The separation of the arrival times of the two tracers indicates the residual saturation similar to volume sensitive tracers (see Section 3.1.2). The tracer compounds are hydrophilic and must be susceptible to decay leading to daughter compounds with different partitioning coefficients. Kinetic parameters should be evaluated in order to acquire suitable compounds for specific conditions of tracer tests (e.g., types and time scales). In contrast to kinetic tracers, the kinetic parameters are not used in the evaluation of the breakthrough curves for these tracers.
The most common fields for the application of these types of tracers are oilfields and carbon capture and storage. Esters like ethyl acetate have been proposed to determine the residual oil saturation according to Cooke [141]. By 1990 they have been successfully applied to oilfields [142,143] and are continued to be implemented today [144,145]. Myers et al. (2012) demonstrate the feasibility of using reactive ester tracers (i.e. triacetin, propylene glycol diacetate and tripropionin) to quantify the amount of residually trapped CO2 through an integrated program of laboratory experiments and computer simulations. Later, the research was also demonstrated successfully in field experiments [146].
4. Exploitation Potential and Further Challenges of Developing Reactive Tracers
4.1. The Necessity for New Tracers–Tracer Design Approach
The use of tracers for hydrogeological applications has a long history. The first reported tracer application was around 10 A.D. to track the connection between the spring source of the Jordan River and a nearby pond [147]. Since then, the development of technology and the advances of tracer testing with a wide selection of tracer compounds have brought effective tools for investigating different properties of the aquatic environment. In general, tracer tests could be applied to any kind of natural and engineered systems. It is especially advantageous for not directly accessible systems compared to other techniques. Nevertheless, there are still many systems in which the potential of using reactive tracers is not yet fully exploited and more attention should be paid to these, including:
- The hyporheic zone, a transition zone between surface water and subsurface water, has been recognized as a hotspot for biogeochemical reactions, making the exchange of water, nutrients, and organic matter important parameters. This zone is a mixing zone which has a complex hydrological situation and heterogeneity containing dissolved gasses, oxidized and reduced species, temperature patterns, flow rates, etc. Due to the large number of variables, the quantification of processes in the hyporheic zone is still a challenge [148,149].
- Hydraulic fracturing (fracking) in shale/tight gas reservoirs has gained growing interests in the oil and gas industry during the last decade [150]. However, fracking may pose environmental risks [151,152]. During the stimulation process, fracking fluid is injected into the reservoir to create additional flow paths for the transport of hydrocarbons. Hydraulically induced fractures may connect pre-existing natural fractures and faults leading to the creation of multiple permeable pathways which may cause groundwater contamination [153]. Therefore, there is a high demand for the application of tracers to predict the risk or to track the contamination (i.e. fracking fluid) [154].
- Other fields may include karst aquifer characterization (due to the strong system heterogeneity and variability), geothermal fluids and acid-mine-drainage (due to complex water chemistry and temperature).
The design of new innovative reactive tracers requires new strategies. Molecular design has been successfully established as a methodology for producing tailor-made molecules with desired properties or effects in several scientific disciplines, especially in life sciences, such as pharmacology, biochemistry, medicine [155], and material sciences [156]. The target-oriented combination of well-studied structural elements and molecular features (e.g., functional groups, substructures, homologues, etc.) allows the creation of novel compounds with desired structures and properties. Almost an unlimited number of compounds is imaginable and can be synthesized individually for a magnitude of applications. However, molecular target design of tracer substances for studying the aquatic environment has yet to be widely considered.
4.2. Strategy for Designing Novel Reactive Tracers
Creating tracer molecules, which react in a predictable way under given physicochemical conditions, is a relatively new and very innovative concept. By knowing exactly how certain reservoir conditions drive the tracer reaction, new insights into the controlling variables may be gained. In the following, the exemplary molecular target design of thermo-sensitive and interface-sensitive tracers is described. The prerequisite for the design (selection and modification) of molecules that are able to act as thermo-sensitive and interface-sensitive tracers in reservoir studies, respectively, is a thorough understanding of their reactive behavior. In particular, it is vital to understand the role and influence of each structural element in the molecule on its reaction kinetics and its physicochemical tracer properties (e.g., detection, acidity, solubility, sorption, etc.). In Figure 2, the main steps for a successful theoretical and practical molecular target tracer design are shown schematically.
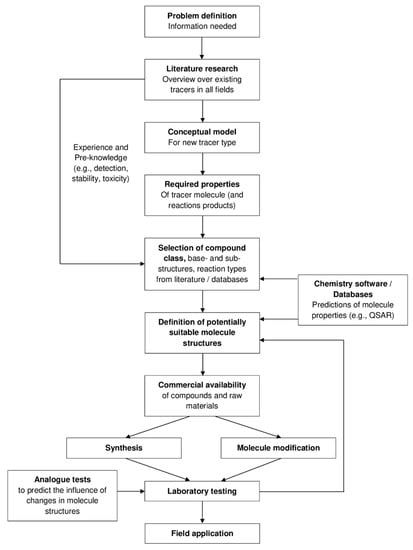
Figure 2.
Schematic overview for the design of reservoir tracers.
Based on available literature and experiences from laboratory and field tests, a promising base molecule for both tracer types is believed to be the class of naphthalenesulfonates, into which thermo- and interface-sensitive groups can be incorporated (Figure 3). Several physicochemical attributes make them convenient for the selection as the backbone structure. Naphthalenesulfonates are strong acids with corresponding low logarithmic acidity constants (pKa) of <1. Therefore, this compound class forms anions even at very low pH values and is highly water-soluble (>1000 g L−1). The resulting pH-dependent of −2.87 at pH > 5 (SciFinder, ACD (Advanced Chemistry Development)/Labs) is also very low, which implies a non-sorptive behavior and, thus, a high mobility in aquatic systems. Additionally, naphthalenesulfonates are stable under oxygen-free conditions and temperatures up to 250 °C [129,157]. The molecule’s good fluorescence with a direct detection limit in the low µg L−1 range is another important feature of naphthalenesulfonates. Hence, their detection in field tests by online determination simplifies the experimental effort needed. Furthermore, (high-pressure liquid) ion pair chromatography combined with solid phase extraction and fluorescence detection (SPE-IPC-FLD) lowers the detection limit by around one order of magnitude (<1 µg L−1) even in highly saline matrices, such as brines from deep reservoirs [158,159]. The chromatographic separation even allows the simultaneous analysis of several compounds and, therefore, the use of different isomers, derivatives, and homologues. Finally, naphthalenesulfonates are non-toxic [160], their use in groundwater studies is administratively non-restricted, and they are established conservative tracers for the characterization of geothermal reservoirs [158,161].
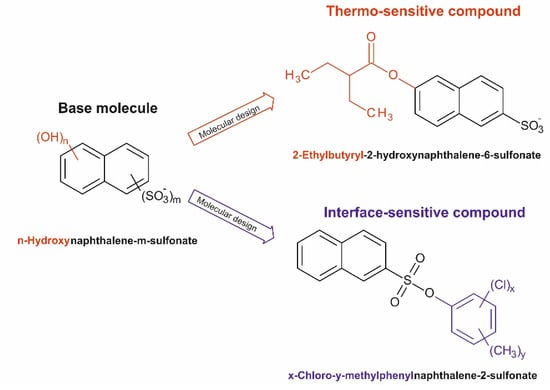
Figure 3.
Design of two different types of potential reservoir tracers based on naphthalenesulfonate as common structural element.
5. Summary and Conclusions
The selection of optimal reactive tracer compounds is main challenge that needs to be considered before conducting a tracer test. For instance, when designing a thermo-sensitive tracer test, a tracer that decays too slowly under a system’s temperature lengthens test duration needlessly and thus makes observing the differences in mean residence times difficult; too fast decay makes it challenging for the test implementation. Moreover, new reactive tracer compounds have been extensively developed in the past decades due to the demand in new advanced technologies. Therefore, a complete understanding of the physicochemical properties of reactive tracers and their occurring processes is essential. Depending on the biophysicochemical behavior, three types of reactive tracers can be distinguished, namely: equilibrium tracers, kinetic tracers and reactive tracer for partitioning. Equilibrium tracers are based on the partitioning equilibrium between two immiscible phases or at their interfaces. Kinetic tracers are non-equilibrium tracers in which only the reaction kinetics are used for the parameter determination. Reactive tracers for partitioning are a hybrid form of equilibrium tracers and kinetic tracers.
The complexities of natural systems, along with the large number of requirements for the tracers, make the selection and use of reactive tracers not a simple task, but an art. Based on the knowledge of tracer properties, tailor-made tracer compounds are being developed with the required properties or effects in hydrogeology. The target-oriented combination of well-studied structural elements and molecular features (e.g., functional groups, substructures, homologues) allows for the creation of novel compounds with desired structures and properties. Nearly an unlimited number of compounds can be synthesized individually for specific applications. This innovative concept can expand the potential application of tracers in different fields (e.g., quantification of processes in the hyporheic zone, prediction of environmental risks of hydraulic fracturing). Molecular design assists the preselected properties (e.g., fluorescence) of both reactants and products. This allows a mass balance, and thereby opens the opportunity of a tracer test design without an additional conservative tracer.
Author Contributions
Conceptualization, V.C., M.S., and T.L.; methodology, V.C., M.S., and T.L.; formal analysis, V.C.; writing-original draft preparation, V.C. and M.S.; writing-review and editing, V.C., M.S., R.T. and T.L.; visualization, V.C., R.T; funding acquisition, R.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ministry of Education and Training of Vietnam (MOET, DA-911), the European Community’s 7th Framework Programme FP7/2007−2013, within the MUSTANG project (grant agreement no. 227286), the German Ministry for Environment (BMU) within the project REAKTHERM (grant no. 0325417) and the German Research Foundation (DFG) within the GEOCAT project (project no. LI 1314/3-1 and 1314/3-2.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chrysikopoulos, C.V. Artificial tracers for geothermal reservoir studies. Environ. Geol. 1993, 22, 60–70. [Google Scholar] [CrossRef]
- Flury, M.; Wai, N.N. Dyes as tracers for vadose zone hydrology. Rev. Geophys. 2003, 41, 2.1–2.37. [Google Scholar] [CrossRef]
- Serres-Piole, C.; Preud’homme, H.; Moradi-Tehrani, N.; Allanic, C.; Jullia, H.; Lobinski, R. Water tracers in oilfield applications: Guidelines. J. Pet. Sci. Eng. 2012, 98–99, 22–39. [Google Scholar] [CrossRef]
- Abbott, B.W.; Baranov, V.; Mendoza-Lera, C.; Nikolakopoulou, M.; Harjung, A.; Kolbe, T.; Balasubramanian, M.N.; Vaessen, T.N.; Ciocca, F.; Campeau, A.; et al. Using multi-tracer inference to move beyond single-catchment ecohydrology. Earth-Sci. Rev. 2016, 160, 19–42. [Google Scholar] [CrossRef]
- Rhee, S.; Kang, J.; Park, J. Partitioning tracer method for quantifying the residual saturation of refined petroleum products in saturated soil. Environ. Earth Sci. 2011, 64, 2059–2066. [Google Scholar] [CrossRef]
- Silva, M.; Stray, H.; Bjornstad, T. Studies on new chemical tracers for determination of residual oil saturation in the inter-well region. In Proceedings of the SPE Oklahoma City Oil Gas Symposium, Oklahoma City, OK, USA, 27–31 March 2017; pp. 1–14. [Google Scholar]
- Hillebrand, O.; Noedler, K.; Sauter, M.; Licha, T. Multitracer experiment to evaluate the attenuation of selected organic micropollutants in a karst aquifer. Sci. Total Environ. 2015, 506–507, 338–343. [Google Scholar] [CrossRef]
- Knapp, J.L.A.; González-Pinzón, R.; Drummond, J.D.; Larsen, L.G.; Cirpka, O.A.; Harvey, J.W. Tracer-based characterization of hyporheic exchange and benthic biolayers in streams. Water Resour. Res. 2017, 53, 1575–1594. [Google Scholar] [CrossRef]
- Maier, F.; Schaffer, M.; Licha, T. Temperature determination using thermo-sensitive tracers: Experimental validation in an isothermal column heat exchanger. Geothermics 2015, 53, 533–539. [Google Scholar] [CrossRef]
- Schaffer, M.; Idzik, K.R.; Wilke, M.; Licha, T. Amides as thermo-sensitive tracers for investigating the thermal state of geothermal reservoirs. Geothermics 2016, 64, 180–186. [Google Scholar] [CrossRef]
- Schaffer, M.; Maier, F.; Licha, T.; Sauter, M. A new generation of tracers for the characterization of interfacial areas during supercritical carbon dioxide injections into deep saline aquifers: Kinetic interface-sensitive tracers (KIS tracer). Int. J. Greenh. Gas Control 2013, 14, 200–208. [Google Scholar] [CrossRef]
- Myers, M.; Stalker, L.; Ross, A.; Dyt, C.; Ho, K.-B. Method for the determination of residual carbon dioxide saturation using reactive ester tracers. Appl. Geochem. 2012, 27, 2148–2156. [Google Scholar] [CrossRef]
- Cornaton, F.J.; Park, Y.J.; Deleersnijder, E. On the biases affecting water ages inferred from isotopic data. J. Hydrol. 2011, 410, 217–225. [Google Scholar] [CrossRef][Green Version]
- Delhez, É.J.M.; Deleersnijder, É.; Mouchet, A.; Beckers, J.M. A note on the age of radioactive tracers. J. Mar. Syst. 2003, 38, 277–286. [Google Scholar] [CrossRef]
- Deleersnijder, E.; Delhez, E.J.M.; Crucifix, M.; Beckers, J.M. On the symmetry of the age field of a passive tracer released into a one-dimensional fluid flow by a point-source. Bull. la Soc. R. des Sci. Liege 2001, 70, 5–21. [Google Scholar]
- White, L.; Legat, V.; Deleersnijder, E. Tracer conservation for three-dimensional, finite-element, free-surface, ocean modeling on moving prismatic meshes. Mon. Weather Rev. 2008, 136, 420–442. [Google Scholar] [CrossRef]
- Reimus, P.W.; Pohll, G.; Mihevc, T.; Chapman, J.; Haga, M.; Lyles, B.; Kosinski, S.; Niswonger, R.; Sanders, P. Testing and parameterizing a conceptual model for solute transport in a fractured granite using multiple tracers in a forced-gradient test. Water Resour. Res. 2003, 39. [Google Scholar] [CrossRef]
- Yakirevich, A.; Shelton, D.; Hill, R.; Kiefer, L.; Stocker, M.; Blaustein, R.; Kuznetsov, M.; McCarty, G.; Pachepsky, Y. Transport of conservative and “smart” tracers in a first-order creek: Role of transient storage type. Water 2017, 9, 485. [Google Scholar] [CrossRef]
- Moeck, C.; Radny, D.; Popp, A.; Brennwald, M.; Stoll, S.; Auckenthaler, A.; Berg, M.; Schirmer, M. Characterization of a managed aquifer recharge system using multiple tracers. Sci. Total Environ. 2017, 609, 701–714. [Google Scholar] [CrossRef]
- Hillebrand, O.; Nödler, K.; Licha, T.; Sauter, M.; Geyer, T. Caffeine as an indicator for the quantification of untreated wastewater in karst systems. Water Res. 2012, 46, 395–402. [Google Scholar] [CrossRef]
- Field, M.S.; Wilhelm, R.G.; Quinlan, J.F.; Aley, T.J. An assessment of the potential adverse properties of fluorescent tracer dyes used for groundwater tracing. Environ. Monit. Assess. 1995, 38, 75–96. [Google Scholar] [CrossRef]
- Wolkersdorfer, C.; Göbel, J.; Hasche-Berger, A. Assessing subsurface flow hydraulics of a coal mine water bioremediation system using a multi-tracer approach. Int. J. Coal Geol. 2016, 164, 58–68. [Google Scholar] [CrossRef]
- Zhu, Z.; Motta, D.; Jackson, P.R.; Garcia, M.H. Numerical modeling of simultaneous tracer release and piscicide treatment for invasive species control in the Chicago Sanitary and Ship Canal, Chicago, Illinois. Environ. Fluid Mech. 2017, 17, 211–229. [Google Scholar] [CrossRef]
- Battaglia, D.; Birindelli, F.; Rinaldi, M.; Vettraino, E.; Bezzi, A. Fluorescent tracer tests for detection of dam leakages: The case of the Bumbuna dam—Sierra Leone. Eng. Geol. 2016, 205, 30–39. [Google Scholar] [CrossRef]
- Guo, C.; Cui, Y.; Dong, B.; Liu, F. Tracer study of the hydraulic performance of constructed wetlands planted with three different aquatic plant species. Ecol. Eng. 2017, 102, 433–442. [Google Scholar] [CrossRef]
- Olson, J.C.; Marcarelli, A.M.; Timm, A.L.; Eggert, S.L.; Kolka, R.K. Evaluating the effects of culvert designs on ecosystem processes in Northern Wisconsin Streams. River Res. Appl. 2017, 33, 777–787. [Google Scholar] [CrossRef]
- Shih, S.S.; Zeng, Y.Q.; Lee, H.Y.; Otte, M.L.; Fang, W.T. Tracer experiments and hydraulic performance improvements in a treatment pond. Water 2017, 9, 137. [Google Scholar] [CrossRef]
- Ptak, T.; Piepenbrink, M.; Martac, E. Tracer tests for the investigation of heterogeneous porous media and stochastic modelling of flow and transport—A review of some recent developments. J. Hydrol. 2004, 294, 122–163. [Google Scholar] [CrossRef]
- Divine, C.E.; McDonnell, J.J. The future of applied tracers in hydrogeology. Hydrogeol. J. 2005, 13, 255–258. [Google Scholar] [CrossRef]
- Luhmann, A.J.; Covington, M.D.; Alexander, S.C.; Chai, S.Y.; Schwartz, B.F.; Groten, J.T.; Alexander, E.C., Jr. Comparing conservative and nonconservative tracers in karst and using them to estimate flow path geometry. J. Hydrol. 2012, 448–449, 201–211. [Google Scholar] [CrossRef]
- Hassett, J.J.; Means, J.C.; Banwart, W.L.; Wood, S.G. Sorption Properties of Sediments and Energy-Related Pollutants; Environmental Research Laboratory, Office of Research and Development, US Environmental Protection Agency: Athens, GA, USA, 1980. [Google Scholar]
- Schwarzenbach, R.P.; Giger, W.; Hoehn, E.; Schneider, J.K. Behavior of organic compounds during infiltration of river water to groundwater. Field studies. Environ. Sci. Technol. 1983, 17, 472–479. [Google Scholar] [CrossRef]
- Schaffer, M.; Licha, T. A framework for assessing the retardation of organic molecules in groundwater: Implications of the species distribution for the sorption-influenced transport. Sci. Total Environ. 2015, 524–525, 187–194. [Google Scholar] [CrossRef]
- Maeng, S.K.; Sharma, S.K.; Lekkerkerker-Teunissen, K.; Amy, G.L. Occurrence and fate of bulk organic matter and pharmaceutically active compounds in managed aquifer recharge: A review. Water Res. 2011, 45, 3015–3033. [Google Scholar] [CrossRef] [PubMed]
- Karickhoff, S.; Brown, D.; Scott, T. Sorption of hydrophobic pollutants on natural sediments. Water Res. 1979, 13, 241–248. [Google Scholar] [CrossRef]
- Sabljic, A.; Gusten, H.; Verhaar, H.; Hermens, J. Corrigendum: QSAR modelling of soil sorption. Improvements and systematics of log k(OC) vs. log k(OW) correlations. Chemosphere 1995, 33, 2577. [Google Scholar]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2002; ISBN 9780471649649. [Google Scholar]
- Schaffer, M.; Boxberger, N.; Börnick, H.; Licha, T.; Worch, E. Sorption influenced transport of ionizable pharmaceuticals onto a natural sandy aquifer sediment at different pH. Chemosphere 2012, 87, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, M.; Kröger, K.F.; Nödler, K.; Ayora, C.; Carrera, J.; Hernández, M.; Licha, T. Influence of a compost layer on the attenuation of 28 selected organic micropollutants under realistic soil aquifer treatment conditions: Insights from a large scale column experiment. Water Res. 2015, 74, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lee, L.S. Solubility and sorption by soils of 8:2 fluorotelomer alcohol in water and cosolvent systems. Environ. Sci. Technol. 2005, 39, 7535–7540. [Google Scholar] [CrossRef]
- Fischer, S.; Licha, T.; Markelova, E. Transportverhalten kurzkettiger Alkylphenole (SCAP) im Grundwasser und in der Umwelt. Grundwasser 2014, 19, 119–126. [Google Scholar] [CrossRef]
- Burke, V.; Treumann, S.; Duennbier, U.; Greskowiak, J.; Massmann, G. Sorption behavior of 20 wastewater originated micropollutants in groundwater—Column experiments with pharmaceutical residues and industrial agents. J. Contam. Hydrol. 2013, 154, 29–41. [Google Scholar] [CrossRef]
- Hebig, K.H.; Groza, L.G.; Sabourin, M.J.; Scheytt, T.J.; Ptacek, C.J. Transport behavior of the pharmaceutical compounds carbamazepine, sulfamethoxazole, gemfibrozil, ibuprofen, and naproxen, and the lifestyle drug caffeine, in saturated laboratory columns. Sci. Total Environ. 2017, 590–591, 708–719. [Google Scholar] [CrossRef]
- Nham, H.T.T.; Greskowiak, J.; Nödler, K.; Rahman, M.A.; Spachos, T.; Rusteberg, B.; Massmann, G.; Sauter, M.; Licha, T. Modeling the transport behavior of 16 emerging organic contaminants during soil aquifer treatment. Sci. Total Environ. 2015, 514, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Freeze, R.A.; Cherry, J.A. Groundwater; Prentice-Hall, Inc.: Englewood Cliffs, NJ, USA, 1979. [Google Scholar]
- Leecaster, K.; Ayling, B.; Moffitt, G.; Rose, P.E. Use of safranin T as a reactive tracer for geothermal reservoir characterization. In Proceedings of the 37th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 30 January–1 February 2012. [Google Scholar]
- Dean, C.; Reimus, P.W.; Newell, D.; Diagnostics, C.; Observations, E.S.; Alamos, L. Evaluation of a Cation Exchanging Tracer to Interrogate Fracture Surface Area in Egs Systems. In Proceedings of the 37th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 30 January–1 February 2012. [Google Scholar]
- Reimus, P.W.; Williams, M.; Vermeul, V.; Rose, P.E.; Leecaster, K.; Ayling, B.; Sanjuan, R.; Ames, M.; Dean, C.; Benoit, D. Use of Tracers to Interrogate Fracture Surface Area in Single-well Tracer Tests in Egs Systems. In Proceedings of the 37th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 30 January–1 February 2012. [Google Scholar]
- Dean, C.; Reimus, P.W.; Oates, J.; Rose, P.E.; Newell, D.; Petty, S. Laboratory experiments to characterize cation-exchanging tracer behavior for fracture surface area estimation at Newberry Crater, OR. Geothermics 2015, 53, 213–224. [Google Scholar] [CrossRef]
- Schaffer, M.; Warner, W.; Kutzner, S.; Börnick, H.; Worch, E.; Licha, T. Organic molecules as sorbing tracers for the assessment of surface areas in consolidated aquifer systems. J. Hydrol. 2017, 546, 370–379. [Google Scholar] [CrossRef]
- Rose, P.E.; Leecaster, K.; Clausen, S.; Sanjuan, R.; Ames, M.; Reimus, P.W.; Williams, M.; Vermeul, V.; Benoit, D. A tracer test at the Soda Lake, Nevada geothermal field using a sorbing tracer. In Proceedings of the 37th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 30 January–1 February 2012; pp. 30–33. [Google Scholar]
- Neretnieks, I. A stochastic multi-channel model for solute transport-analysis of tracer tests in fractured rock. J. Contam. Hydrol. 2002, 55, 175–211. [Google Scholar] [CrossRef]
- Hawkins, A.; Fox, D.; Zhao, R.; Tester, J.W.; Cathles, L.; Koch, D.; Becker, M. Predicting Thermal Breakthrough from Tracer Tests: Simulations and Observations in a Low-Temperature Field Laboratory. In Proceedings of the Fortieth Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 26–28 January 2015; pp. 1–15. [Google Scholar]
- Wilson, R.D. Reactive tracers to characterize pollutant distribution and behavior in aquifers. In Handbook of Hydrocarbon and Lipid Microbiology; Springer: Berlin, Germany, 2010; pp. 2465–2471. ISBN 978-3-540-77584-3. [Google Scholar]
- Jin, M.; Delshad, M.; Dwarakanath, V.; Mckinney, D.C.; Pope, G.A.; Sepehmoori, K.; Tilburg, C.E.; Jackson, R.E.; Cherry, N. Partitioning tracer test for detection, estimation, and remediation performance assessment of subsurface nonaqueous phase liquids. Water Resour. Res. 1995, 31, 1201–1211. [Google Scholar] [CrossRef]
- Annable, M.D.; Rao, P.S.C.; Hatfield, K.; Graham, W.D.; Wood, A.L.; Enfield, C.G. Partitioning Tracers for Measuring Residual NAPL: Field-Scale Test Results. J. Environ. Eng. 1998, 124, 498–503. [Google Scholar] [CrossRef]
- Jawitz, J.W.; Sillan, R.K.; Annable, M.D.; Rao, P.S.C.; Warner, K. In-situ alcohol flushing of a DNAPL source zone at a dry cleaner site. Environ. Sci. Technol. 2000, 34, 3722–3729. [Google Scholar] [CrossRef]
- Johnston, C.D.; Davis, G.B.; Bastow, T.P.; Annable, M.D.; Trefry, M.G.; Furness, A.; Geste, Y.; Woodbury, R.J.; Rao, P.S.C.; Rhodes, S. The use of mass depletion-mass flux reduction relationships during pumping to determine source zone mass of a reactive brominated-solvent DNAPL. J. Contam. Hydrol. 2013, 144, 122–137. [Google Scholar] [CrossRef]
- Cápiro, N.L.; Granbery, E.K.; Lebrón, C.A.; Major, D.W.; McMaster, M.L.; Pound, M.J.; Löffler, F.E.; Pennell, K.D. Liquid-liquid mass transfer of partitioning electron donors in chlorinated solvent source zones. Environ. Sci. Technol. 2011, 45, 1547–1554. [Google Scholar] [CrossRef]
- Imhoff, P.T.; Pirestani, K.; Jafarpour, Y.; Spivey, K.M. Tracer interaction effects during partitioning tracer tests for NAPL detection. Environ. Sci. Technol. 2003, 37, 1441–1447. [Google Scholar] [CrossRef]
- Jin, M.; Jackson, R.E.; Pope, G.A.; Taffinder, S. Development of Partitioning Tracer Tests for Characterization of Nonaqueous-Phase Liquid-Contaminated Aquifers. In Proceedings of the SPE Annual Technical Conference and Exhibition; Society of Petroleum Engineers, San Antonio, TX, USA, 5–8 October 1997. [Google Scholar]
- Young, C.M.; Jackson, R.E.; Jin, M.; Londergan, J.T.; Mariner, P.E.; Pope, G.A.; Anderson, F.J.; Houk, T. Characterization of a TCE DNAPL Zone in Alluvium by Partitioning Tracers. Groundw. Monit. Remediat. 1999, 19, 84–94. [Google Scholar] [CrossRef]
- Brooks, M.C.; Annable, M.D.; Rao, P.S.C.; Hatfield, K.; Jawitz, J.W.; Wise, W.R.; Wood, A.L.; Enfield, C.G. Controlled release, blind tests of DNAPL characterization using partitioning tracers. J. Contam. Hydrol. 2002, 59, 187–210. [Google Scholar] [CrossRef]
- Divine, C.E.; McCray, J.E.; Wolf Martin, L.M.; Blanford, W.J.; Blitzer, D.J.; Brusseau, M.L.; Boving, T.B. Partitioning tracer tests as a remediation metric: Case study at naval amphibious base little creek, Virginia Beach, Virginia. Remediat. J. 2004, 14, 7–31. [Google Scholar] [CrossRef]
- Cain, R.B.; Johnson, G.R.; McCray, J.E.; Blanford, W.J.; Brusseau, M.L. Partitioning tracer tests for evaluating remediation performance. Ground Water 2000, 38, 752–761. [Google Scholar] [CrossRef]
- Jawitz, J.W.; Annable, M.D.; Clark, C.J.; Puranik, S. Inline Gas Chromatographic Tracer Analysis: An Alternative To Conventional Sampling and Laboratory Analysis for Partitioning Tracer Tests. Instrum. Sci. Technol. 2002, 30, 415–426. [Google Scholar] [CrossRef]
- Noordman, W.H.; De Boer, G.J.; Wietzes, P.; Volkering, F.; Janssen, D.B. Assessment of the Use of Partitioning and Interfacial Tracers To Determine the Content and Mass Removal Rates of Nonaqueous Phase Liquids. Environ. Sci. Technol. 2000, 34, 4301–4306. [Google Scholar] [CrossRef][Green Version]
- Annable, M.D.; Hatfield, K.; Cho, J.; Klammler, H.; Parker, B.L.; Cherry, J.A.; Rao, P.S.C. Field-scale evaluation of the passive flux meter for simultaneous measurement of groundwater and contaminant fluxes. Environ. Sci. Technol. 2005, 39, 7194–7201. [Google Scholar] [CrossRef]
- Hartog, N.; Cho, J.; Parker, B.L.; Annable, M.D. Characterization of a heterogeneous DNAPL source zone in the Borden aquifer using partitioning and interfacial tracers: Residual morphologies and background sorption. J. Contam. Hydrol. 2010, 115, 79–89. [Google Scholar] [CrossRef]
- Wang, F.; Annable, M.D.; Schaefer, C.E.; Ault, T.D.; Cho, J.; Jawitz, J.W. Enhanced aqueous dissolution of a DNAPL source to characterize the source strength function. J. Contam. Hydrol. 2014, 169, 75–89. [Google Scholar] [CrossRef]
- Jessheim, B.; Viig, S.; Dugstad, O.; Stray, H. Tracers. WO2014096459A1, 23 December 2013. [Google Scholar]
- Dean, R.M.; Walker, D.L.; Dwarakanath, V.; Malik, T.; Spilker, K. Use of partitioning tracers to estimate oil saturation distribution in heterogeneous reservoirs. In Proceedings of the SPE Improved Oil Recover Conference, Tulsa, OK, USA, 11–13 April 2016. [Google Scholar]
- Wilson, R.D.; Mackay, D.M. Direct Detection of Residual Nonaqueous Phase Liquid in the Saturated Zone Using SF6 as a Partitioning Tracer. Environ. Sci. Technol. 1995, 29, 1255–1258. [Google Scholar] [CrossRef]
- Werner, D.; Höhener, P. Diffusive Partitioning Tracer Test for Nonaqueous Phase Liquid (NAPL) Detection in the Vadose Zone. Environ. Sci. Technol. 2002, 36, 1592–1599. [Google Scholar] [CrossRef] [PubMed]
- Vulava, V.M.; Perry, E.B.; Romanek, C.S.; Seaman, J.C. Dissolved gases as partitioning tracers for determination of hydrogeological parameters. Environ. Sci. Technol. 2002, 36, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.M.; Istok, J.D.; Semprini, L. Push-pull partitioning tracer tests using radon-222 to quantify non-aqueous phase liquid contamination. J. Contam. Hydrol. 2002, 58, 129–146. [Google Scholar] [CrossRef]
- Reid, M.C.; Jaffé, P.R. A push-pull test to measure root uptake of volatile chemicals from wetland soils. Environ. Sci. Technol. 2013, 47, 3190–3198. [Google Scholar] [CrossRef] [PubMed]
- Deeds, N.; Pope, G.A.; Mckinney, D.C. Vadose Zone Characterization at a Contaminated Field Site Using Partitioning Interwell Tracer Technology. Environ. Sci. Technol. 1999, 33, 2745–2751. [Google Scholar] [CrossRef]
- Hunkerler, D.; Hoehn, E.; Höhener, P.; Zeyer, J. 222Rn as a Partitioning Tracer To Detect Diesel Fuel Contamination in Aquifers: Laboratory Study and Field Observations. Environ. Sci. Technol. 1997, 31, 3180–3187. [Google Scholar] [CrossRef][Green Version]
- Schubert, M.; Paschke, A.; Lau, S.; Geyer, W.; Knöller, K. Radon as a naturally occurring tracer for the assessment of residual NAPL contamination of aquifers. Environ. Pollut. 2007, 145, 920–927. [Google Scholar] [CrossRef]
- Ponsin, V.; Chablais, A.; Dumont, J.; Radakovitch, O.; Höhener, P. 222Rn as Natural Tracer for LNAPL Recovery in a Crude Oil-Contaminated Aquifer. Groundw. Monit. Remediat. 2015, 35, 30–38. [Google Scholar] [CrossRef]
- Ghanem, A.; Soerens, T.S.; Adel, M.M.; Thoma, G.J. Investigation of Fluorescent Dyes as Partitioning Tracers for Subsurface Nonaqueous Phase Liquid (NAPL) Characterization. J. Environ. Eng. 2003, 129, 740–744. [Google Scholar] [CrossRef]
- LaForce, T.; Ennis-King, J.; Boreham, C.; Paterson, L. Residual CO2 saturation estimate using noble gas tracers in a single-well field test: The CO2CRC Otway project. Int. J. Greenh. Gas Control 2014, 26, 9–21. [Google Scholar] [CrossRef]
- Stalker, L.; Boreham, C.; Underschultz, J.; Freifeld, B.; Perkins, E.; Schacht, U.; Sharma, S. Application of tracers to measure, monitor and verify breakthrough of sequestered CO2 at the CO2CRC Otway Project, Victoria, Australia. Chem. Geol. 2015, 399, 2–19. [Google Scholar] [CrossRef]
- Rasmusson, K.; Rasmusson, M.; Fagerlund, F.; Bensabat, J.; Tsang, Y.; Niemi, A. Analysis of alternative push-pull-test-designs for determining in situ residual trapping of carbon dioxide. Int. J. Greenh. Gas Control 2014, 27, 155–168. [Google Scholar] [CrossRef]
- Zhang, Y.; Freifeld, B.; Finsterle, S.; Leahy, M.; Ennis-King, J.; Paterson, L.; Dance, T. Single-well experimental design for studying residual trapping of supercritical carbon dioxide. Int. J. Greenh. Gas Control 2011, 5, 88–98. [Google Scholar] [CrossRef]
- Roberts, J.J.; Gilfillan, S.M.V.; Stalker, L.; Naylor, M. Geochemical tracers for monitoring offshore CO2 stores. Int. J. Greenh. Gas Control 2017, 65, 218–234. [Google Scholar] [CrossRef]
- Saripalli, K.P.; Kim, H.; Annable, M.D. Measurement of Specific Fluid—Fluid Interfacial Areas of Immiscible Fluids in Porous Media. Environ. Sci. Technol. 1997, 31, 932–936. [Google Scholar] [CrossRef]
- Saripalli, K.P.; Rao, P.S.C.; Annable, M.D. Determination of specific NAPL–water interfacial areas of residual NAPLs in porous media using the interfacial tracers technique. J. Contam. Hydrol. 1998, 30, 375–391. [Google Scholar] [CrossRef]
- Zhong, H.; El Ouni, A.; Lin, D.; Wang, B.; Brusseau, M.L. The two-phase flow IPTT method for measurement of nonwetting-wetting liquid interfacial areas at higher nonwetting saturations in natural porous media. Water Resour. Res. 2016, 52, 5506–5515. [Google Scholar] [CrossRef]
- Araujo, J.B.; Mainhagu, J.; Brusseau, M.L. Measuring air-water interfacial area for soils using the mass balance surfactant-tracer method. Chemosphere 2015, 134, 199–202. [Google Scholar] [CrossRef]
- Schaefer, C.E.; DiCarlo, D.A.; Blunt, M.J. Determination of Water-Oil Interfacial Area during 3-Phase Gravity Drainage in Porous Media. J. Colloid Interface Sci. 2000, 221, 308–312. [Google Scholar] [CrossRef]
- Annable, M.D.; Jawitz, J.W.; Rao, P.S.C.; Dai, D.P.; Kim, H.; Wood, A.L. Field Evaluation of Interfacial and Partitioning Tracers for Characterization of Effective NAPL-Water Contact Areas. Ground Water 1998, 36, 495–502. [Google Scholar] [CrossRef]
- Kim, H.; Rao, P.S.C.; Annable, M.D. Determination of effective air-water interfacial area in partially saturated porous media using surfactant adsorption. Water Resour. Res. 1997, 33, 2705–2711. [Google Scholar] [CrossRef]
- Kim, H.; Rao, P.S.C.; Annable, M.D. Gaseous Tracer Technique for Estimating Air-Water Interfacial Areas and Interface Mobility. Soil Sci. Soc. Am. J. 1999, 63, 1554–1560. [Google Scholar] [CrossRef]
- Jain, V.; Bryant, S.; Sharma, M. Influence of Wettability and Saturation on Liquid—Liquid Interfacial Area in Porous Media. Environ. Sci. Technol. 2003, 37, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Narter, M.; Brusseau, M.L. Comparison of Interfacial Partitioning Tracer Test and High-Resolution Microtomography Measurements of Fluid-Fluid Interfacial Areas for an Ideal Porous Medium. Water Resour. Res. 2010, 46. [Google Scholar] [CrossRef] [PubMed]
- Brusseau, M.L.; Narter, M.; Janousek, H. Interfacial partitioning tracer test measurements of organic-liquid/water interfacial areas: Application to soils and the influence of surface roughness. Environ. Sci. Technol. 2010, 44, 7596–7600. [Google Scholar] [CrossRef] [PubMed]
- Anwar, F.A.H.M.; Bettahar, M.; Matsubayashi, U. A method for determining air–water interfacial area in variably saturated porous media. J. Contam. Hydrol. 2000, 43, 129–146. [Google Scholar] [CrossRef]
- Brusseau, M.L.; Peng, S.; Schnaar, G.; Murao, A. Measuring Air—Water Interfacial Areas with X-ray Microtomography and Interfacial Partitioning Tracer Tests. Environ. Sci. Technol. 2007, 41, 1956–1961. [Google Scholar] [CrossRef]
- Setarge, B.; Danze, J.; Klein, R.; Grathwohl, P. Partitioning and Interfacial Tracers to Characterize Non-Aqueous Phase Liquids (NAPLs) in Natural Aquifer Material. Phys. Chem. Earth Part B Hydrol. Oceans Atmos. 1999, 24, 501–510. [Google Scholar] [CrossRef]
- Karkare, M.V.; Fort, T. Determination of the air-water interfacial area in wet “unsaturated” porous media. Langmuir 1996, 12, 2041–2044. [Google Scholar] [CrossRef]
- Silverstein, D.L.; Fort, T. Studies in air-water interfacial area for wet unsaturated particulate porous media systems. Langmuir 1997, 7463, 4758–4761. [Google Scholar] [CrossRef]
- Dobson, R.; Schroth, M.H.; Oostrom, M.; Zeyer, J. Determination of NAPL-water interfacial areas in well-characterized porous media. Environ. Sci. Technol. 2006, 40, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Annable, M.D.; Rao, P.S.C. Influence of Air—Water Interfacial Adsorption and Gas-Phase Partitioning on the Transport of Organic Chemicals in Unsaturated Porous Media. Environ. Sci. Technol. 1998, 32, 1253–1259. [Google Scholar] [CrossRef]
- Istok, J.D.; Humphrey, M.D.; Schroth, M.H.; Hyman, M.R.; O’Reilly, K.T. Single-well, “push-pull” test for in situ determination of microbial activities. Ground Water 1997, 35, 619–631. [Google Scholar] [CrossRef]
- Schroth, M.H.; Istok, J.D.; Conner, G.T.; Hyman, M.R.; Haggerty, R.; O’Reilly, K.T. Spatial variability in in situ aerobic respiration and denitrification rates in a petroleum-contaminated aquifer. Ground Water 1998, 36, 924–937. [Google Scholar] [CrossRef]
- Goldhammer, T.; Einsiedl, F.; Blodau, C. In situ determination of sulfate turnover in peatlands: A down-scaled push-pull tracer technique. J. Plant Nutr. Soil Sci. 2008, 171, 740–750. [Google Scholar] [CrossRef]
- Michalsen, M.M.; Weiss, R.; King, A.; Gent, D.; Medina, V.F.; Istok, J.D. Push-pull tests for estimating rdx and tnt degradation rates in groundwater. Groundw. Monit. Remediat. 2013, 33, 61–68. [Google Scholar] [CrossRef]
- Henson, W.R.; Huang, L.; Graham, W.D.; Ogram, A. Nitrate reduction mechanisms and rates in an unconfined eogenetic karst aquifer in two sites with different redox potential. J. Geophys. Res. Biogeosci. 2017, 122, 1062–1077. [Google Scholar] [CrossRef]
- Cho, Y.; Han, K.; Kim, N.; Park, S.; Kim, Y. Estimating in situ biodegradation rates of petroleum hydrocarbons and microbial population dynamics by performing single-well push-pull tests in a fractured bedrock aquifer. Water. Air. Soil Pollut. 2013, 224, 1364. [Google Scholar] [CrossRef]
- Schroth, M.H.; Kleikemper, J.; Bolliger, C.; Bernasconi, S.M.; Zeyer, J. In situ assessment of microbial sulfate reduction in a petroleum-contaminated aquifer using push-pull tests and stable sulfur isotope analyses. J. Contam. Hydrol. 2001, 51, 179–195. [Google Scholar] [CrossRef]
- Burbery, L.F.; Flintoft, M.J.; Close, M.E. Application of the re-circulating tracer well test method to determine nitrate reaction rates in shallow unconfined aquifers. J. Contam. Hydrol. 2013, 145, 1–9. [Google Scholar] [CrossRef]
- Kleikemper, J.; Schroth, M.H.; Sigler, W.V.; Schmucki, M.; Bernasconi, S.M.; Zeyer, J. Activity and diversity of sulfate-reducing bacteria in a petroleum hydrocarbon-contaminated aquifer. Appl. Environ. Microbiol. 2002, 68, 1516–1523. [Google Scholar] [CrossRef] [PubMed]
- Urmann, K.; Gonzalez-Gil, G.; Schroth, M.H.; Hofer, M.; Zeyer, J. New Field Method: Gas Push−Pull Test for the In-Situ Quantification of Microbial Activities in the Vadose Zone. Environ. Sci. Technol. 2005, 39, 304–310. [Google Scholar] [CrossRef]
- Addy, K.; Kellogg, D.Q.; Gold, A.J.; Groffman, P.M.; Ferendo, G.; Sawyer, C. In situ push-pull method to determine ground water denitrification in riparian zones. J. Environ. Qual. 2002, 31, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S.C.; Annable, M.D.; Kim, H. NAPL source zone characterization and remediation technology performance assessment: Recent developments and applications of tracer techniques. J. Contam. Hydrol. 2000, 45, 63–78. [Google Scholar] [CrossRef]
- Sandrin, S.K.; Brusseau, M.L.; Piatt, J.J.; Bodour, A.A.; Blanford, W.J.; Nelson, N.T. Spatial variability of in situ microbial activity: Biotracer tests. Ground Water 2004, 42, 374–383. [Google Scholar] [CrossRef]
- Brusseau, M.L.; Nelson, N.T.; Zhang, Z.; Blue, J.E.; Rohrer, J.; Allen, T. Source-zone characterization of a chlorinated-solvent contaminated Superfund site in Tucson, AZ. J. Contam. Hydrol. 2007, 90, 21–40. [Google Scholar] [CrossRef] [PubMed]
- Alter, S.R.; Brusseau, M.L.; Piatt, J.J.; Ray-Maitra, A.; Wang, J.M.; Cain, R.B. Use of tracer tests to evaluate the impact of enhanced-solubilization flushing on in-situ biodegradation. J. Contam. Hydrol. 2003, 64, 191–202. [Google Scholar] [CrossRef]
- González-Pinzón, R.; Haggerty, R.; Myrold, D.D. Measuring aerobic respiration in stream ecosystems using the resazurin-resorufin system. J. Geophys. Res. 2012, 117, G00N06. [Google Scholar] [CrossRef]
- Stanaway, D.; Haggerty, R.; Benner, S.; Flores, A.; Feris, K. Persistent metal contamination limits lotic ecosystem heterotrophic metabolism after more than 100 years of exposure: A novel application of the Resazurin Resorufin Smart Tracer. Environ. Sci. Technol. 2012, 46, 9862–9871. [Google Scholar] [CrossRef]
- Lemke, D.; González-Pinzón, R.; Liao, Z.; Wöhling, T.; Osenbrück, K.; Haggerty, R.; Cirpka, O.A. Sorption and transformation of the reactive tracers resazurin and resorufin in natural river sediments. Hydrol. Earth Syst. Sci. 2014, 18, 3151–3163. [Google Scholar] [CrossRef]
- Haggerty, R.; Ribot, M.; Singer, G.A.; Marti, E.; Argerich, A.; Agell, G.; Battin, T.J. Ecosystem respiration increases with biofilm growth and bed forms: Flume measurements with resazurin. J. Geophys. Res. Biogeosci. 2014, 119, 2220–2230. [Google Scholar] [CrossRef]
- González-Pinzón, R.; Ward, A.S.; Hatch, C.E.; Wlostowski, A.N.; Singha, K.; Gooseff, M.N.; Haggerty, R.; Harvey, J.W.; Cirpka, O.A.; Brock, J.T. A field comparison of multiple techniques to quantify groundwater—Surface-water interactions. Freshw. Sci. 2015, 34, 139–160. [Google Scholar] [CrossRef]
- González-Pinzón, R.; Peipoch, M.; Haggerty, R.; Martí, E.; Fleckenstein, J.H. Nighttime and daytime respiration in a headwater stream. Ecohydrology 2016, 9, 93–100. [Google Scholar] [CrossRef]
- Haggerty, R.; Argerich, A.; Martí, E. Development of a “smart” tracer for the assessment of microbiological activity and sediment-water interaction in natural waters: The resazurin-resorufin system. Water Resour. Res. 2008, 44. [Google Scholar] [CrossRef]
- Haggerty, R.; Martí, E.; Argerich, A.; von Schiller, D.; Grimm, N.B. Resazurin as a “smart” tracer for quantifying metabolically active transient storage in stream ecosystems. J. Geophys. Res. 2009, 114, G03014. [Google Scholar] [CrossRef]
- Nottebohm, M.; Licha, T.; Ghergut, I.; Nödler, K.; Sauter, M. Development of Thermosensitive Tracers for Push-Pull Experiments in Geothermal Reservoir Characterization. In Proceedings of the World Geothermal Congress, Bali, Indonesia, 25–29 April 2010; pp. 25–29. [Google Scholar]
- Nottebohm, M.; Licha, T.; Sauter, M. Tracer design for tracking thermal fronts in geothermal reservoirs. Geothermics 2012, 43, 37–44. [Google Scholar] [CrossRef]
- Cao, V.; Schaffer, M.; Licha, T. The feasibility of using carbamates to track the thermal state in geothermal reservoirs. Geothermics 2018, 72, 301–306. [Google Scholar] [CrossRef]
- Rose, P.E.; Clausen, S. The Use of Amino G as a Thermally Reactive Tracer for Geothermal Applications. In Proceedings of the 39th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 24–26 February 2014; pp. 1–5. [Google Scholar]
- Rose, P.; Clausen, S. The use of amino-substituted naphthalene sulfonates as tracers in geothermal reservoirs. In Proceedings of the 42nd Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 3–15 February 2017; pp. 1–7. [Google Scholar]
- Maier, F.; Schaffer, M.; Licha, T. Determination of temperatures and cooled fractions by means of hydrolyzable thermo-sensitive tracers. Geothermics 2015, 58, 87–93. [Google Scholar] [CrossRef]
- Batchelor, A. Reservoir behaviour in a stimulated hot dry rock system. In Proceedings of the Eleventh Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 21–23 January 1986; pp. 35–41. [Google Scholar]
- Kwakwa, K.A. Tracer measurements during long-term circulation of the Rosemanowes HDR geothermal system. In Proceedings of the Thirteenth Workshop on Geothermal Reservoir Engineering, Standford, CA, USA, 19–21 January 1988; pp. 245–252. [Google Scholar]
- Tester, J.W.; Robinson, B.A.; Ferguson, J.H. Inert and Reacting Tracers for Reservoir Sizing in Fractured, Hot Dry Rock Systems. In Proceedings of the Eleventh Workshop on Geothermal Reservoir Engineering, Standford, CA, USA, 21–23 January 1986; pp. 149–159. [Google Scholar]
- Hawkins, A.J.; Fox, D.B.; Becker, M.W.; Tester, J.W. Measurement and simulation of heat exchange in fractured bedrock using inert and thermally degrading tracers. Water Resour. Res. 2017, 53, 1210–1230. [Google Scholar] [CrossRef]
- Adams, M.C.; Davis, J. Kinetics of fluorescein decay and its application as a geothermal tracer. Geothermics 1991, 20, 53–66. [Google Scholar] [CrossRef]
- Tatomir, A.B.; Schaffer, M.; Kissinger, A.; Hommel, J.; Nuske, P.; Licha, T.; Helmig, R.; Sauter, M. Novel approach for modeling kinetic interface-sensitive (KIS) tracers with respect to time-dependent interfacial area change for the optimization of supercritical carbon dioxide injection into deep saline aquifers. Int. J. Greenh. Gas Control 2015, 33, 145–153. [Google Scholar] [CrossRef]
- Cooke, C.E.J. Method of Determining Fluid Saturations in Reservoirs. U.S. Patent No. 3,590,923, 06 July 1971. [Google Scholar]
- Tang, J.; Harker, B. Mass Balance Method to Determine Residual Oil Saturation from Single Well Tracer Test Data. J. Can. Pet. Technol. 1990, 29. [Google Scholar] [CrossRef]
- Tang, J.; Zhang, P. Determination of Residual Oil Saturation in A Carbonate Reservoir. In Proceedings of the SPE Asia Pacific Improved Oil Recovery Conference, Kuala Lumpur, Malaysia, 6–9 October 2001. [Google Scholar]
- Pathak, P.; Fitz, D.; Babcock, K.; Wachtman, R.J. Residual Oil Saturation Determination for EOR Projects in Means Field, a Mature West Texas Carbonate Field. SPE Reserv. Eval. Eng. 2012, 15, 541–553. [Google Scholar] [CrossRef]
- Khaledialidusti, R.; Kleppe, J.; Skrettingland, K. Numerical interpretation of Single Well Chemical Tracer (SWCT) tests to determine residual oil saturation in Snorre Reservoir. In Proceedings of the SPE Asia Pacific Improved Oil Recovery Conference, Kuala Lumpur, Malaysia, 6–9 October 2001; pp. 1–4. [Google Scholar]
- Myers, M.; Stalker, L.; La Force, T.; Pejcic, B.; Dyt, C.; Ho, K.B.; Ennis-King, J. Field measurement of residual carbon dioxide saturation using reactive ester tracers. Chem. Geol. 2015, 399, 20–29. [Google Scholar] [CrossRef]
- Käss, W. Tracing Technique in Geohydrology; Balkema: Rotterdam, The Netherlands, 1998. [Google Scholar]
- Sophocleous, M. Interactions between groundwater and surface water: The state of the science. Hydrogeol. J. 2002, 10, 52–67. [Google Scholar] [CrossRef]
- Palmer, M.A. Experimentation in the hyporheic Zon: Challenges and prospectus. J. N. Am. Benthol. Soc. 1993, 12, 84–93. [Google Scholar] [CrossRef]
- Kissinger, A.; Helmig, R.; Ebigbo, A.; Class, H.; Lange, T.; Sauter, M.; Heitfeld, M.; Klünker, J.; Jahnke, W. Hydraulic fracturing in unconventional gas reservoirs: Risks in the geological system, part 2. Environ. Earth Sci. 2013, 70, 3855–3873. [Google Scholar] [CrossRef]
- Taherdangkoo, R.; Tatomir, A.; Taylor, R.; Sauter, M. Numerical investigations of upward migration of fracking fluid along a fault zone during and after stimulation. Energy Procedia 2017, 125, 126–135. [Google Scholar] [CrossRef]
- Tatomir, A.; Mcdermott, C.; Bensabat, J.; Class, H.; Edlmann, K.; Taherdangkoo, R.; Sauter, M. Conceptual model development using a generic Features, Events, and Processes (FEP) database for assessing the potential impact of hydraulic fracturing on groundwater aquifers. Adv. Geosci. 2018, 45, 185–192. [Google Scholar] [CrossRef]
- Taherdangkoo, R.; Tatomir, A.; Anighoro, T.; Sauter, M. Modeling fate and transport of hydraulic fracturing fluid in the presence of abandoned wells. J. Contam. Hydrol. 2019, 221, 58–68. [Google Scholar] [CrossRef]
- Kurose, S. Requiring the use of tracers in hydraulic fracturing fluid to trace alleged contamination. Sustain. Dev. Law Policy 2014, 14, 43–54. [Google Scholar]
- Kuntz, I.D.; Meng, E.C.; Shoichet, B.K. Structure-Based Molecular Design. Acc. Chem. Res. 1994, 27, 117–123. [Google Scholar] [CrossRef]
- Kang, E.T.; Zhang, Y. Surface Modification of Fluoropolymers via Molecular Design. Adv. Mater. 2000, 12, 1481–1494. [Google Scholar] [CrossRef]
- Rose, P.E.; Benoit, W.R.; Kilbourn, P.M. The application of the polyaromatic sulfonates as tracers in geothermal reservoirs. Geothermics 2001, 30, 617–640. [Google Scholar] [CrossRef]
- Rose, P.E.; Johnson, S.D.; Kilbourn, P.M. Tracer testing at dixie valley, nevada, using 2-naphthalene sulfonate and 2,7-naphthalene disulfonate. In Proceedings of the 26th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 29–31 January 2001; pp. 1–6. [Google Scholar]
- Nottebohm, M.; Licha, T. Detection of Naphthalene Sulfonates from Highly Saline Brines with High-Performance Liquid Chromatography in Conjunction with Fluorescence Detection and Solid-Phase Extraction. J. Chromatogr. Sci. 2012, 50, 477–481. [Google Scholar] [CrossRef][Green Version]
- Greim, H.; Ahlers, J.; Bias, R.; Broecker, B.; Hollander, H.; Gelbke, H.-P.; Klimisch, H.-J.; Mangelsdorf, I.; Paetz, A.; Schön, N.; et al. Toxicity and ecotoxicity of sulfonic acids: Structure-activity relationship. Chemosphere 1994, 28, 2203–2236. [Google Scholar]
- Sanjuan, B.; Pinault, J.-L.; Rose, P.E.; Gérard, A.; Brach, M.; Braibant, G.; Crouzet, C.; Foucher, J.-C.; Gautier, A.; Touzelet, S. Tracer testing of the geothermal heat exchanger at Soultz-sous-Forêts (France) between 2000 and 2005. Geothermics 2006, 35, 622–653. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).