Removal of Diclofenac in Wastewater Using Biosorption and Advanced Oxidation Techniques: Comparative Results
Abstract
:1. Introduction
2. Materials and Methods
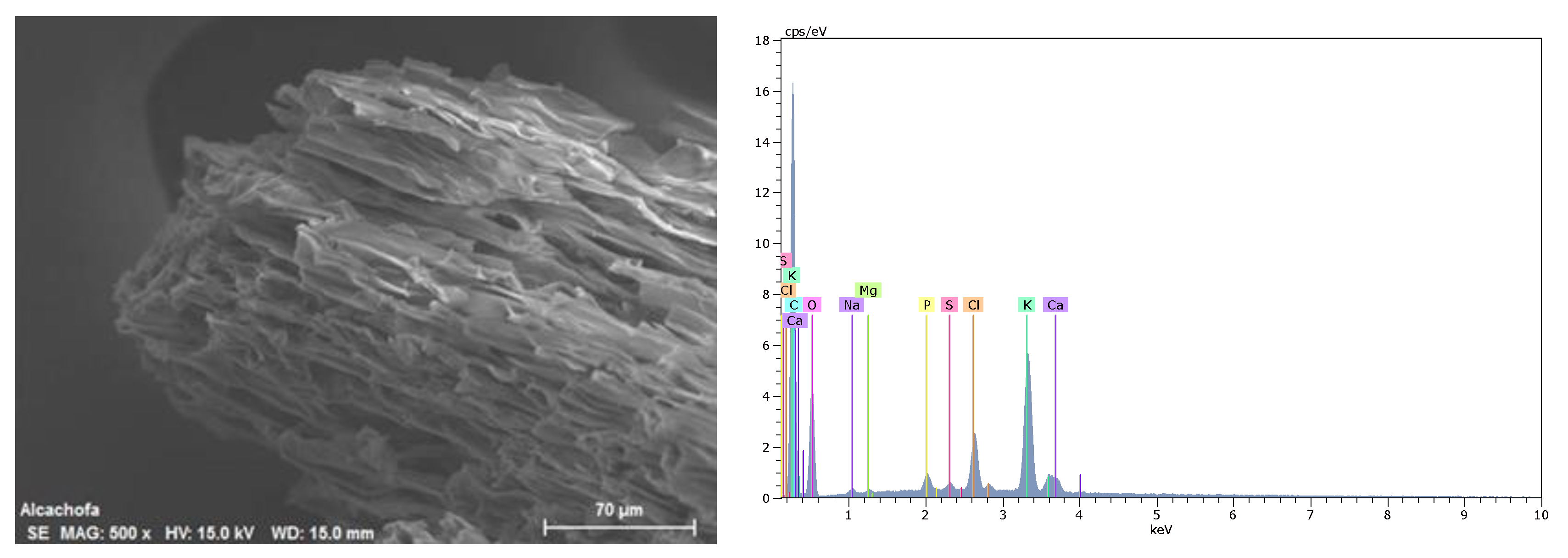
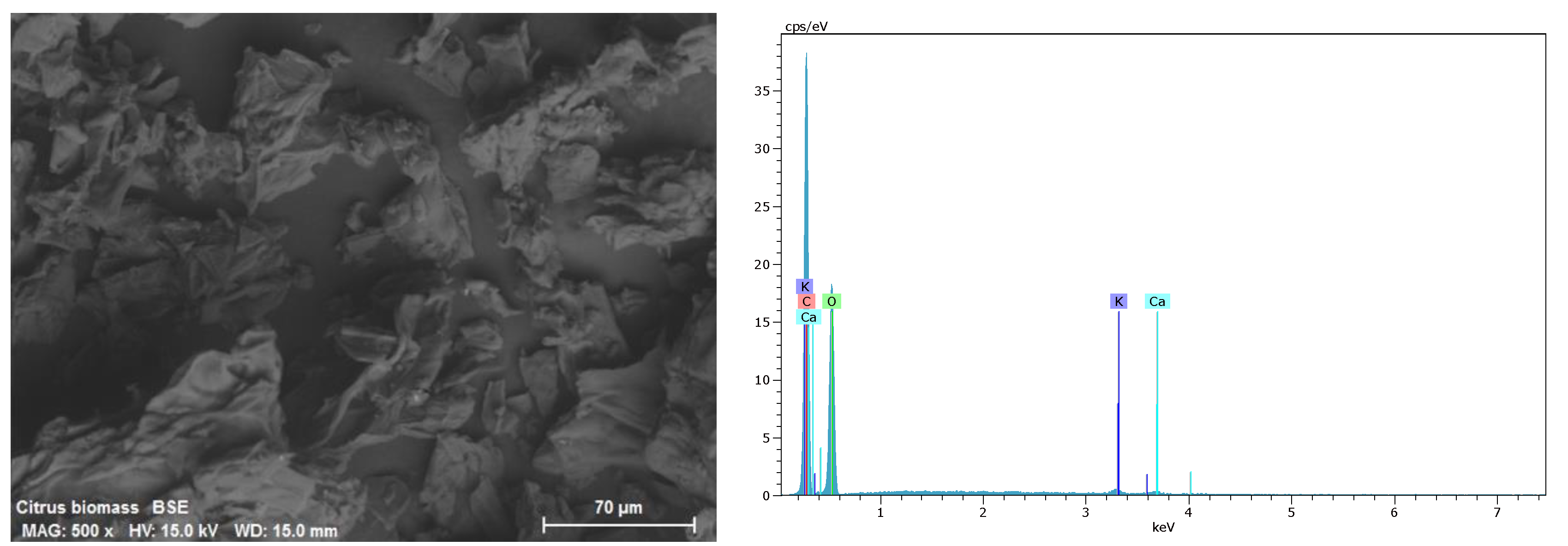
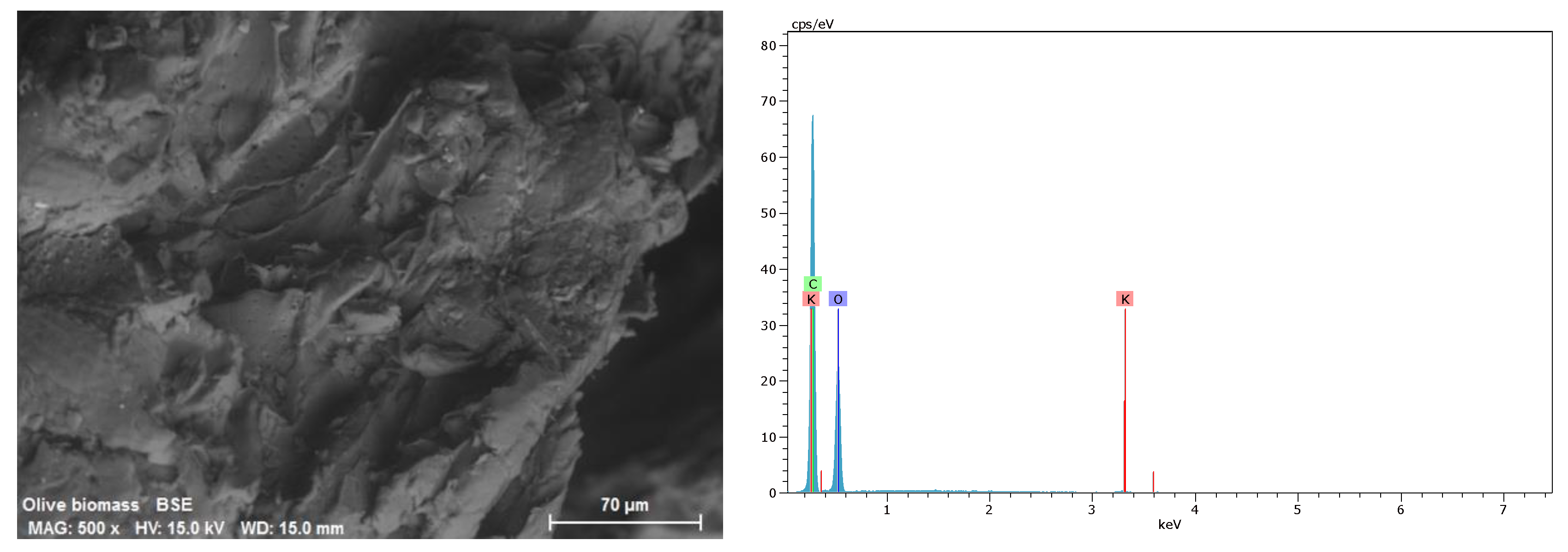
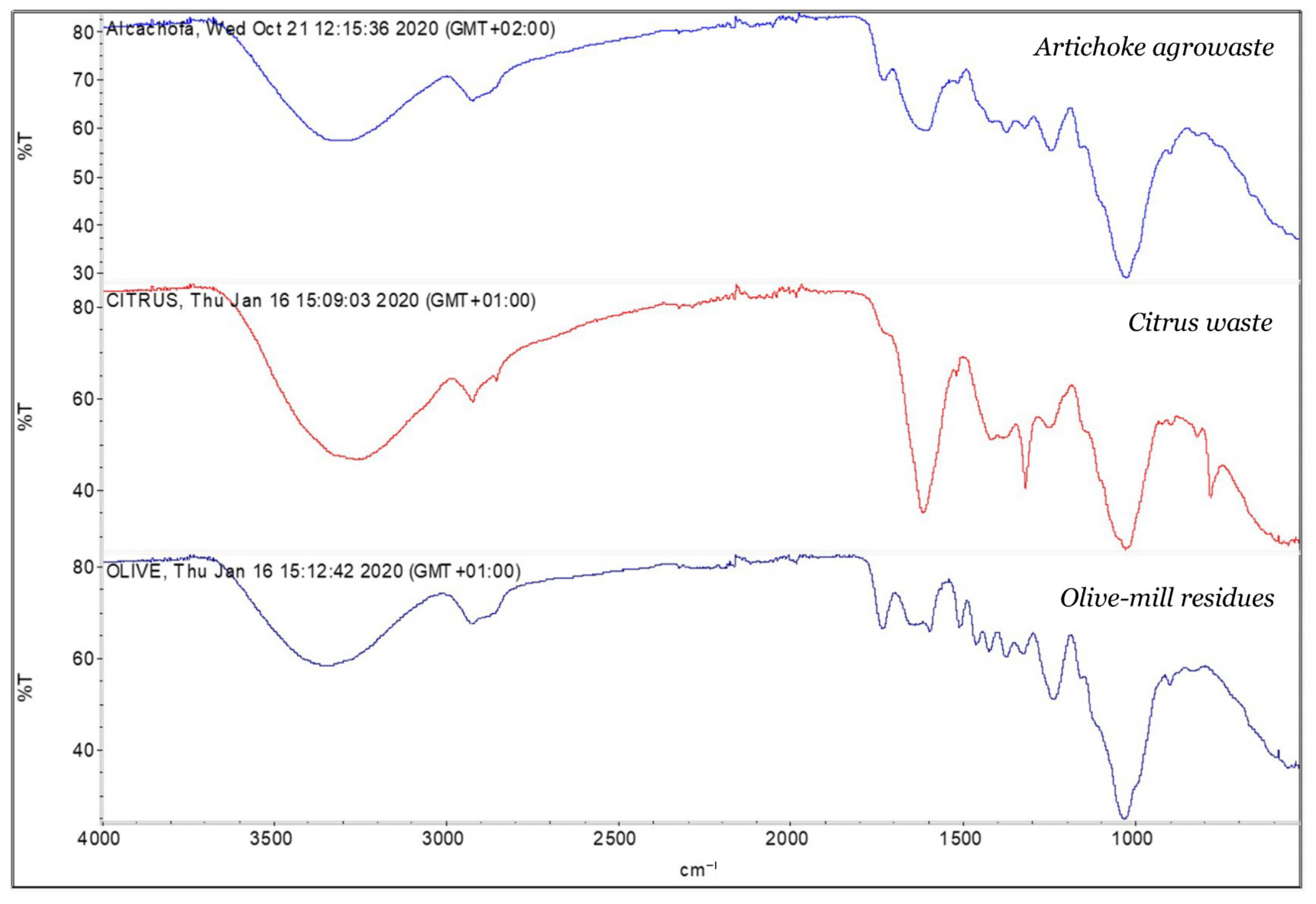
2.1. Preparation and Characterization of Agrowaste Biomass
2.2. Preparation of Diclofenac Solutions
2.3. Batch-Biomass Tests
2.4. Diclofenac Determination
2.5. Advanced Oxidation Treatments
2.5.1. Sodium Hypochlorite
2.5.2. UV Light Treatments
2.5.3. UV Radiation Combined with Sodium Hypochlorite (UV/HOCl) and Hydrogen Peroxide (UV/H2O2)
2.6. Chemical Oxygen Demand
3. Results and Discussion
3.1. Characterization of Biomass Samples
3.2. Diclofenac Biosorption
3.3. Advanced Oxidation Treatments
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dalecka, B.; Oskarsson, C.; Juhna, T.; Rajarao, G.K. Isolation of fungal strains from municipal wastewater for the removal of pharmaceutical substances. Water 2020, 12, 524. [Google Scholar] [CrossRef] [Green Version]
- Geisen, V.; Mol, H.; Klumpp, E.; Umlauf, G.; Nadal, M.; Van der Ploeg, M.; van de Zee, S.E.A.T.M.; Ritsema, C.J. Emerging pollutants in the environment: A challenge for water resource management. Int. Soil Water Conserv. Res. 2015, 3, 57–65. [Google Scholar] [CrossRef]
- Sauvé, S.; Desrosiers, M. A review of what is an emerging contaminant. Chem. Cent. J. 2014, 8, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Doval, J.C.; Montagner, C.C.; de Alburquerque, A.F.; Moschini-Carlos, V.; Umbuzeiro, G.; Pompeo, M. Nutrients, emerging pollutants and pesticides in a tropical urban reservoir: Spatial distributions and risk assessment. Sci. Total Environ. 2017, 575, 1307–1324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Margenat, A.; Matamoros, V.; Díez, S.; Cañameras, N.; Comas, J.; Bayona, J.M. Ocurrence of chemical contaminants in peri-urban agricultural irrigation waters and assesment of their phytotoxicity and crop productivity. Sci. Total Environ. 2017, 599, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.; Drogui, P.; Brar, S.K. Sonochemical techniques to degrade pharmaceutical organic pollutants. Environ. Chem. Lett. 2015, 13, 251–268. [Google Scholar] [CrossRef]
- Carmalin, A.; Lima, E. Removal of emerging contaminants from the environment by adsorption. Ecotoxicol. Environ. 2018, 150, 1–17. [Google Scholar]
- Munthe, J.; Brorstrom-Lunden, E.; Rahmberg, M.; Posthuma, L.; Alterburger, R.; Brack, W.; Bunke, D.; Engelen, G.; Gawlik, B.; Gils, J.; et al. An expanded conceptual framework for solution-focused management of chemical pollution in European waters. Environ. Sci. Eur. 2017, 29, 1072. [Google Scholar] [CrossRef] [Green Version]
- Richardson, S.D.; Kimura, S.Y. Emerging environmental contaminants: Challenges facing our next generation and potential engineering solutions. Environ. Technol. Innov. 2017, 8, 40–56. [Google Scholar] [CrossRef]
- Rodriguez-Narvaez, O.M.; Peralta-Hernandez, J.M.; Goonetilleke, A.; Bandala, E.R. Treatment technologies for emerging contaminants in water: A review. Chem. Eng. J. 2017, 323, 361–380. [Google Scholar] [CrossRef] [Green Version]
- Petrovic, M.; Solé, M.; López, M.; Barceló, D. Endocrine disruptors in sewage treatment plants, receiving river waters, and sediments: Integration of chemical analysis and biological effects of feral carp. Environ. Toxic. Chem. 2002, 21, 2146–2156. [Google Scholar] [CrossRef]
- Esplugas, S.; Bila, D.M.; Krause, L.G.T.; Dezotti, M. Ozonation and advanced oxidation technologies to remove endocrine disrupting chemicals (EDCs) and pharmaceuticals and personal care products (PPCPs) in water effluents. J. Hazard. Mater. 2007, 149, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.D. Environmental mass spectrometry: Emerging contaminants and current issues. Anal. Chem. 2012, 84, 747–778. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.D.; Kimura, S.Y. Water analysis: Emerging contaminants and current issues. Anal. Chem. 2016, 88, 546–582. [Google Scholar] [CrossRef]
- Blanco, M.; Pérez-Albadalejo, E.; Piña, B.; Kuspilic, G.; Milun, V.; Lille-Langoy, R. Assessing the environmental quality of sediments from Split coastal area (Croatia) with a battery of cell-based bioassays. Sci. Total Environ. 2018, 624, 1640–1648. [Google Scholar]
- Boehler, S.; Strecker, R.; Heinrich, P.; Prochazka, E.; Northcott, G.L.; Ataria, J.M. Assessment of urban stream sediment pollutants entering estuaries using chemical analysis and multiple bioassays to characterize biological activities. Sci. Total Environ. 2017, 593, 498–507. [Google Scholar] [CrossRef]
- Abbas, M.; Adil, M.; Ehtisham-ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Abbas Shar, G.; Asif Tahir, M.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar]
- Schereiber, B.; Fischer, J.; Schiwy, S.; Hollert, H.; Schulz, R. Towards more ecological relevance in sediment toxicity testing with fish: Evaluation of multiple bioassays with embryos of the benthic weatherfish (Misgurnus fossilis). Sci. Total Environ. 2018, 619, 391–400. [Google Scholar] [CrossRef]
- Sang, Z.; Jiang, Y.; Tsoi, Y.-K.; Leung, K.S. Evaluating the environmental impact of artificial sweeteners: A study of their distributions, photodegradation ant toxicities. Water Res. 2014, 52, 260–274. [Google Scholar] [CrossRef]
- Funke, J.; Prasse, C.; Eversloh, C.L.; Ternes, T.A. Oxypurinol-A novel marker for wastewater contamination of the aquatic environment. Water Res. 2015, 74, 257–265. [Google Scholar]
- Lonappan, L.; Brar, S.K.; Das, R.K.; Verma, M.; Rao, Y. Diclofenac and its transformation products: Environmental occurrence and toxicity. Environ. Int. 2016, 96, 127–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grandclément, C.; Piram, A.; Petit, M.E.; Seyssiecq, I.; Laffont-Schwob, I.; Vanot, G.; Tiliacos, N.; Roche, N.; Doumenq, P. Biological removal and fate assessment of diclofenac using Bacillus subtilis and Brevibacillus laterosporus strains and ecotoxicological effects of diclofenac and 4′-hydroxy-diclofenac. J. Chem. 2020, 2020, 9789420. [Google Scholar] [CrossRef]
- Vieno, N.; Silanpää, M. Fate of diclofenac in municipal wastewater treatment plants—A review. Environ. Int. 2014, 69, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Langford, K.H.; Reid, M.; Thomas, K.V. Multi-residue screening of prioritised human pharmaceuticals, illicit drugs and bactericides in sediments and sludge. J. Environ. Monit. 2011, 13, 2284–2291. [Google Scholar] [CrossRef] [PubMed]
- Kunkel, U.; Radke, M. Fate of pharmaceuticals in rivers: Deriving a benchmark dataset at favorable attenuation conditions. Water Res. 2012, 46, 5551–5565. [Google Scholar] [CrossRef]
- Gil, A.; Santamaría, L.; Korili, S.A. Removal of caffeine and diclofenac from aqueous solution by adsorption on multiwalled carbon nanotubes. Coll. Interf. Sci. Commun. 2018, 22, 25–28. [Google Scholar] [CrossRef]
- Mabrouki, H.; Akretche, D.E. Diclofenac potassium removal from water by adsorption on natural and pillared clay. Des. Water Treatm. 2016, 57, 6033–6043. [Google Scholar] [CrossRef]
- Thi Minh Tam, N.; Liu, Y.; Bashir, H.; Yin, Z.; He, Y.; Zhou, X. Efficient removal of diclofenac from aqueous solution by potassium ferrate-activated porous graphitic biochar: Ambient condition influences and adsorption mechanism. Int. J. Environ. Res. Public Health 2020, 17, 291. [Google Scholar] [CrossRef] [Green Version]
- Coimbra, R.N.; Escapa, C.; Vázquez, N.C.; Noriega-Hevia, G.; Otero, M. Utilization of non-living microalgae biomass from two different strains for the adsorptive removal of diclofenac from water. Water 2018, 10, 1401. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Taggart, M.A.; McKenzie, C.; Zhang, Z.; Lu, Y.; Pap, S.; Gibb, S. Utilizing low-cost natural waste for the removal of pharmaceuticals from water: Mechanisms, isotherms and kinetics at low concentrations. J. Clean. Prod. 2019, 227, 88–97. [Google Scholar] [CrossRef] [Green Version]
- Yusuf, M.; Elfghi, F.M.; Zaidi, S.A.; Abdullah, E.C.; Khan, M.A. Applications of graphene and its derivatives as an adsorbent for heavy metal and dye removal: A systematic and comprehensive overview. RSC Adv. 2015, 5, 50392–50420. [Google Scholar] [CrossRef]
- Rosique, M.; Angosto, J.M.; Guibal, E.; Roca, M.J.; Fernández-López, J.A. Factorial design methodological approach for enhanced cadmium ions bioremoval by Opuntia biomass. CLEAN Soil Air Water 2016, 44, 959–966. [Google Scholar] [CrossRef]
- Ben-Othman, S.; Jõudu, I.; Bhat, R. Bioactives from agri-food wastes: Present insights and future challenges. Molecules 2020, 25, 510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nowicki, P.; Kazmierczak-Razna, J.; Pietrzak, R. Physico-chemical and adsorption properties of carbonaceous sorbents prepared by activation of tropical fruit skins with potassium carbonate. Mater. Des. 2016, 90, 579–585. [Google Scholar] [CrossRef]
- Castrica, M.; Rebucci, R.; Giromini, C.; Tretola, M.; Cattaneo, D.; Baldi, A. Total phenolic content and antioxidant capacity of agri-food waste and by-products. Ital. J. Anim. Sci. 2019, 18, 336–341. [Google Scholar] [CrossRef]
- Manna, S.; Toy, D.; Adhikari, B.; Thomas, S.; Das, P. Biomass for water defluoridation and current understanding on biosorption mechanisms: A review. Environ. Prog. Sustain. Energy 2018, 37, 1560–1572. [Google Scholar] [CrossRef]
- Ummartyotin, S.; Pechyen, C. Strategies for development and implementation of bio-based materials as effective renewable resources of energy: A comprehensive review on adsorbent technology. Renew. Sustain. Energy Rev. 2016, 62, 654–664. [Google Scholar] [CrossRef]
- Arief, V.O.; Trilestari, K.; Sunarso, J.; Indraswati, N.; Ismadji, S. Recent progress on biosorption of heavy metals from liquids using low cost biosorbents: Characterization, biosorption parameters and mechanism studies. CLEAN Soil Air Water 2008, 36, 937–962. [Google Scholar] [CrossRef]
- Ternes, T.; Stüber, J.; Herrmann, N.; McDowell, D.; Ried, A.; Kampmann, M.; Teiser, B. Ozonation: A tool for removal of pharmaceuticals, contrast media and musk fragrances from wastewater? Water Res. 2003, 37, 1976–1982. [Google Scholar] [CrossRef]
- Cerreta, G.; Roccamante, M.A.; Oller, I.; Malato, S.; Rizzo, L. Contaminants of emerging concern removal from real wastewater by UV/free chlorine process: A comparison with solar/free chlorine and UV/H2O2 at pilot scale. Chemosphere 2019, 236, 124354. [Google Scholar]
- Lee, Y.; von Gunten, U. Oxidative transformation of micropollutants during municipal wastewaters treatment: Comparison of kinetic aspects of selective (chlorine, chlorine dioxide, ferrate VI and ozone) and non-selective oxidants (hydroxyl radicals). Water Res. 2010, 39, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Kanakaraju, D.; Glass, B.D.; Oelgemöller, M. Titanium dioxide photocatalysis for pharmaceutical wastewater treatment. Environ. Chem. Lett. 2014, 12, 27–47. [Google Scholar] [CrossRef]
- Jiang, J.; Pang, S.Y.; Ma, J.; Liu, H. Oxidation of phenolic endocrine disrupting chemicals by potassium permanganate in synthetic and real waters. Environ. Sci. Technol. 2012, 46, 1774–1781. [Google Scholar] [CrossRef]
- Fiol, N.; Villaescusa, I. Determination of sorbent point zero charge: Usefulness in sorption studies. Environ. Chem. Lett. 2009, 7, 79–84. [Google Scholar] [CrossRef]
- Eaton, A.D.; American Public Health Association; American Water Works Association; Water Environment Federation. Standard Methods for the Examination of Water and Wastewater, 21st ed.; APHA-AWWA-WEF: Washington, DC, USA, 2005. [Google Scholar]
- Amiri, H.; Karimi, K. Improvement of acetone, butanol, and ethanol production from woody biomass using organosolv pretreatment. Bioproc. Biosyst. Eng. 2015, 38, 1959–1972. [Google Scholar] [CrossRef] [PubMed]
- Saavedra, M.I.; Doval Miñarro, M.; Angosto, J.M.; Fernández-López, J.A. Reuse potential of residues of artichoke (Cynara scolymus L.) from industrial canning processing as sorbent of heavy metals in multimetallic effluents. Ind. Crops Prod. 2019, 141, 111751. [Google Scholar] [CrossRef]
- Mahato, N.; Sharma, K.; Sinha, M.; Baral, E.R.; Koteswararao, R.; Dhyani, A.; Cho, M.H.; Cho, S. Bio-sorbents, industrially important chemicals and novel materials from citrus processing waste as a sustainable and renewable bioresource: A review. J. Adv. Res. 2020, 23, 61–82. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.; Esteves, V.I.; Guégan, R.; Crespo, J.S.; Fernandes, A.N.; Giovanela, M. Removal of diclofenac sodium from aqueous solution by Isabel grape bagasse. Chem. Eng. J. 2012, 192, 114–121. [Google Scholar] [CrossRef] [Green Version]
- Larous, S.; Meniai, A.-H. Adsorption of diclofenac from aqueous solution using activated carbon prepared from olive stones. Int. J. Hydrog. Energy 2016, 41, 10380–10390. [Google Scholar] [CrossRef]
- Plauta, M.; Tisler, T.; Toman, M.J.; Pintar, A. Efficiency of advanced oxidation processes in lowering bisphenol A toxicity and oestrogenic activity in aqueous samples. Arch. Ind. Hyg. Toxicol. 2014, 65, 77–87. [Google Scholar]
- Zhao, Q.; Shang, C.; Zhang, X.; Ding, G.; Yang, X. Formation of halogenated organic byproducts during medium-pressure UV and chlorine coexposure of model compounds, NOM and bromide. Water Res. 2011, 45, 6545–6554. [Google Scholar] [CrossRef] [PubMed]
- Stasinakis, A. Use of selected advanced oxidation processes (AOPs) for wastewater treatment—A mini review. Glob. NEST J. 2008, 10, 376–385. [Google Scholar]
- De la Cruz, N.; Giménez, J.; Esplugas, S.; Grandjean, D.; de Alencastro, L.F.; Pulgarín, C. Degradation of 32 emergent contaminants by UV and neutral photo-fenton in domestic wastewater effluent previously treated by activated sludge. Water Res. 2012, 46, 1947–1957. [Google Scholar] [CrossRef] [PubMed]
- Vogna, D.; Marotta, R.; Napolitano, A.; Andreozzi, R.; d’Ischia, M. Advanced oxidation of the pharmaceutical drug diclofenac with UV/H2O2 and ozone. Water Res. 2004, 38, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Luongo, G.; Guida, M.; Siciliano, A.; Libralato, G.; Saviano, L.; Amoresano, A.; Previtera, L.; Di Fabio, G.; Zarrelli, A. Oxidation of diclofenac in water by sodium hypochlorite: Identification of new degradation by-products and their ecotoxicological evaluation. J. Pharmac. Biomed. Anal. 2020, 113762. [Google Scholar] [CrossRef] [PubMed]
- Chong, S.; Zhang, G.; Zhang, N.; Liu, Y.; Huang, T.; Chang, H. Diclofenac degradation in water by FeCeOx catalyzed H2O2: Influencing factors, mechanism and pathways. J. Hazard. Mat. 2017, 334, 150–159. [Google Scholar] [CrossRef]
| Parameter | Value |
|---|---|
| pH | 6.6 ± 0.2 * |
| Conductivity (µS·cm−1) | 12.5 ± 2.5 |
| TSS (mg/L) | 98.2 ± 8.6 |
| Turbidity (NTU) | 4.7 ± 0.3 |
| NO3− (mg·L−1) | 16.8 ± 2.7 |
| PO43− (mg·L−1) | 5.6 ± 1.1 |
| BOD (mg O2·L−1) | 113.3 ± 10.2 |
| COD (mg O2·L−1) | 282.0 ± 15.0 |
| Diclofenac initial (mg·L−1) | 37.6 |
| Biomass | Sorbent Dosage (g·L−1) | Removal % | qe (mg·g−1) |
|---|---|---|---|
| Artichoke agrowaste | 1 | 18.7 ± 2.4 * | 3.9 ± 0.5 |
| 2 | 19.9 ± 3.1 | 4.2 ± 0.4 | |
| Olive-mill residues | 1 | 46.4 ± 4.4 | 4.2 ± 0.6 |
| 2 | 46.3 ± 5.8 | 4.2 ± 0.8 | |
| Citrus-waste | 1 | 87.1 ± 5.7 | 7.8 ± 1.1 |
| 2 | 99.7 ± 0.5 | 9.0 ± 0.3 |
| Contact Time (min) | Diclofenac (mg·L−1) | Removed Diclofenac % | COD Reduction % |
|---|---|---|---|
| 2 | 33.9 ± 2.8 * | 9.8 ± 2.4 | 44.3 ± 3.6 |
| 5 | 16.6 ± 2.1 | 55.9 ± 3.9 | 62.1 ± 3.5 |
| 10 | 6.1 ± 1.1 | 83.8 ± 4.6 | 62.4 ± 4.1 |
| 15 | 4.3 ± 0.9 | 88.5 ± 3.8 | 66.3 ± 3.9 |
| Hypochlorite Concentration (mg·L−1) | Diclofenac (mg·L−1) | Removed Diclofenac % | COD Reduction % |
|---|---|---|---|
| 1 | 14.4 ± 3.3 * | 61.7 ± 4.7 | 24.1 ± 2.3 |
| 2 | 4.4 ± 1.2 | 88.4 ± 5.1 | 38.3 ± 2.8 |
| 3 | 3.3 ± 0.9 | 91.3 ± 4.4 | 44.0 ± 2.6 |
| 4 | n.d. † | 100 | 44.7 ± 1.8 |
| Contact Time (min) | Diclofenac (mg·L−1) | Removed Diclofenac % | COD Reduction % |
|---|---|---|---|
| 2 | 29.1 ± 2.2 * | 22.6 ± 2.8 | 14.2 ± 1.9 |
| 5 | 13.5 ± 1.8 | 64.2 ± 3.1 | 22.0 ± 2.4 |
| 10 | 2.4 ± 0.5 | 93.6 ± 4.8 | 45.0 ± 3.8 |
| 15 | n.d. † | 100 | 47.9 ± 3.4 |
| Contact Time (min) | Diclofenac (mg·L−1) | Removed Diclofenac % | COD Reduction % |
|---|---|---|---|
| 2 | 0.3 ± 0.1 * | 99.2 ± 4.4 | 23.4 ± 1.7 |
| 5 | n.d. † | 100 | 25.9 ± 2.3 |
| 10 | n.d. | 100 | 36.5 ± 2.0 |
| 15 | n.d. | 100 | 40.1 ± 3.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Angosto, J.M.; Roca, M.J.; Fernández-López, J.A. Removal of Diclofenac in Wastewater Using Biosorption and Advanced Oxidation Techniques: Comparative Results. Water 2020, 12, 3567. https://doi.org/10.3390/w12123567
Angosto JM, Roca MJ, Fernández-López JA. Removal of Diclofenac in Wastewater Using Biosorption and Advanced Oxidation Techniques: Comparative Results. Water. 2020; 12(12):3567. https://doi.org/10.3390/w12123567
Chicago/Turabian StyleAngosto, José M., María J. Roca, and José A. Fernández-López. 2020. "Removal of Diclofenac in Wastewater Using Biosorption and Advanced Oxidation Techniques: Comparative Results" Water 12, no. 12: 3567. https://doi.org/10.3390/w12123567







