Isolation and Characterization of an Aerobic Denitrifier Bacillus sp. SC16 from an Intensive Aquaculture Pond
Abstract
:1. Introduction
2. Materials and Methods
2.1. Media
2.2. Isolation and Identification
2.3. Nitrogen Removal Performance
2.4. Enzyme Assay
2.5. Amplification of the Functional Genes
2.6. Quantitative Real-Time PCR (QRT-PCR) Assay
2.7. The Performance of Strain SC16 in Aquaculture Water
2.8. Analytical Methods
2.9. Statistical Analysis
3. Results
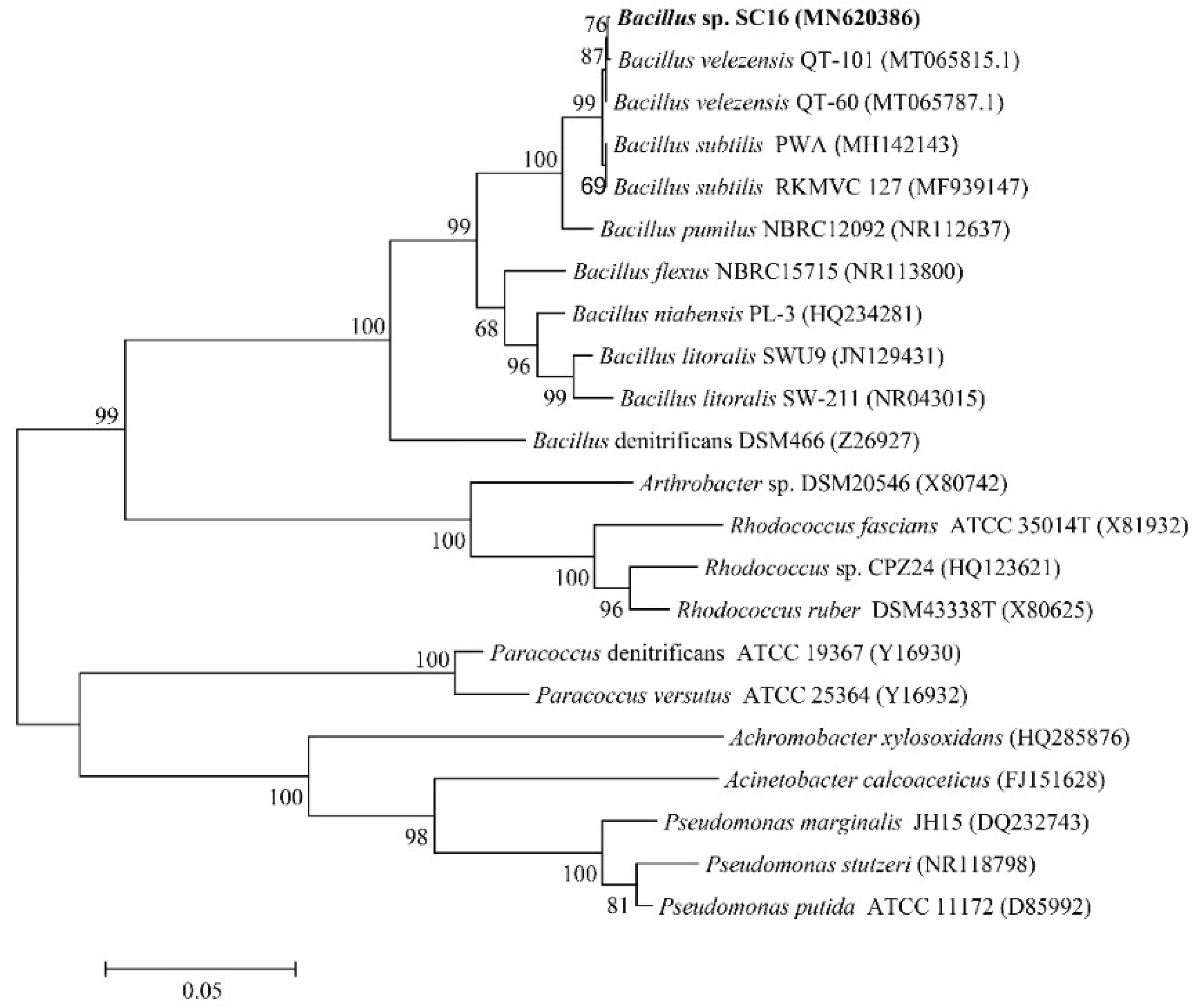
3.1. Isolation and Identification of Strain SC16
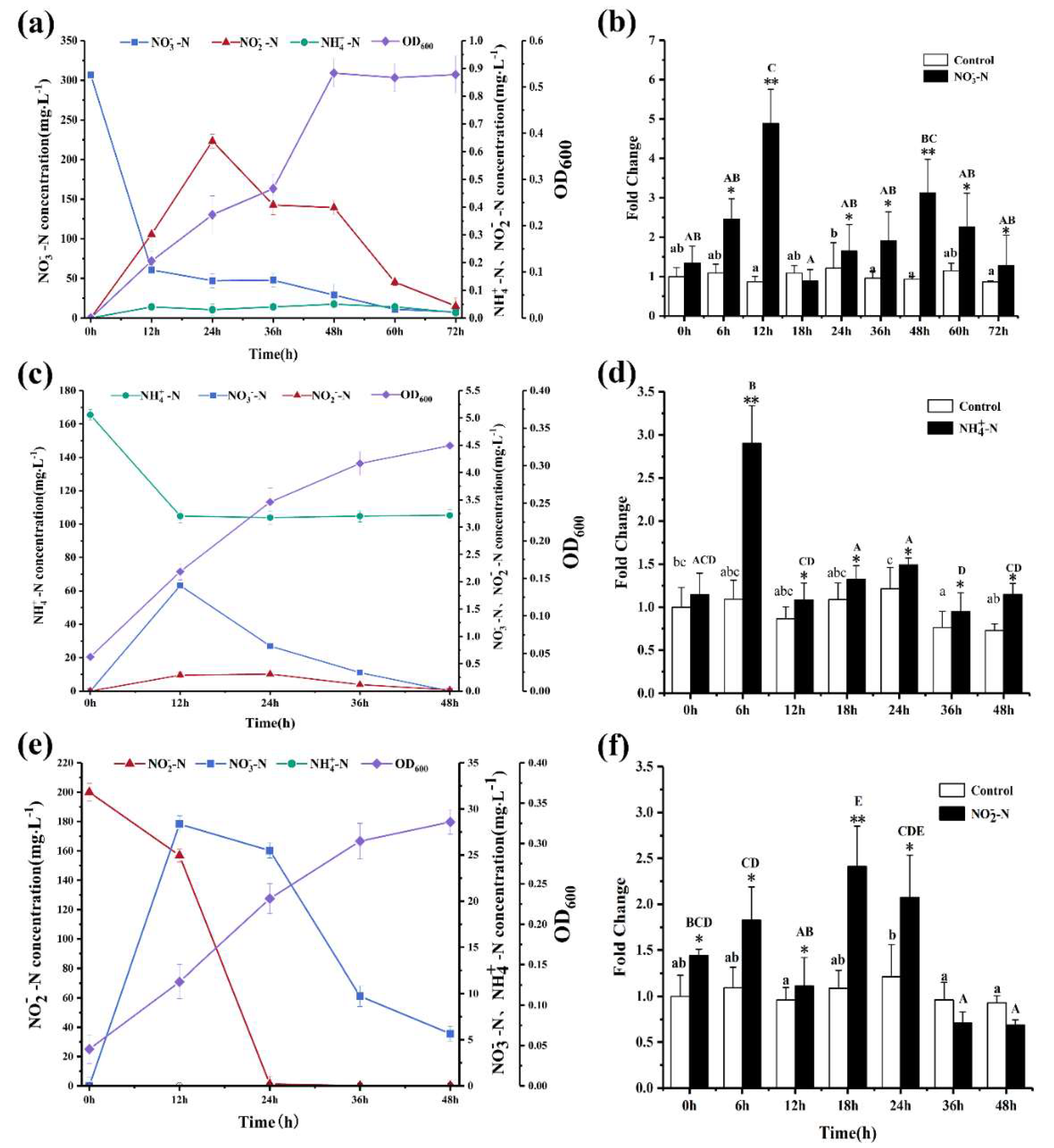
3.2. Evaluation of Nitrogen Removal Ability of Strain SC16
3.3. Assessment of Nitrite Removal Performance of SC16 under Different Nitrite Concentrations
3.4. Enzyme Assay
3.5. Amplification of the Denitrification Genes
3.6. Effects of Nitrogen Sources on the Expression Level of Nitrite Reductase
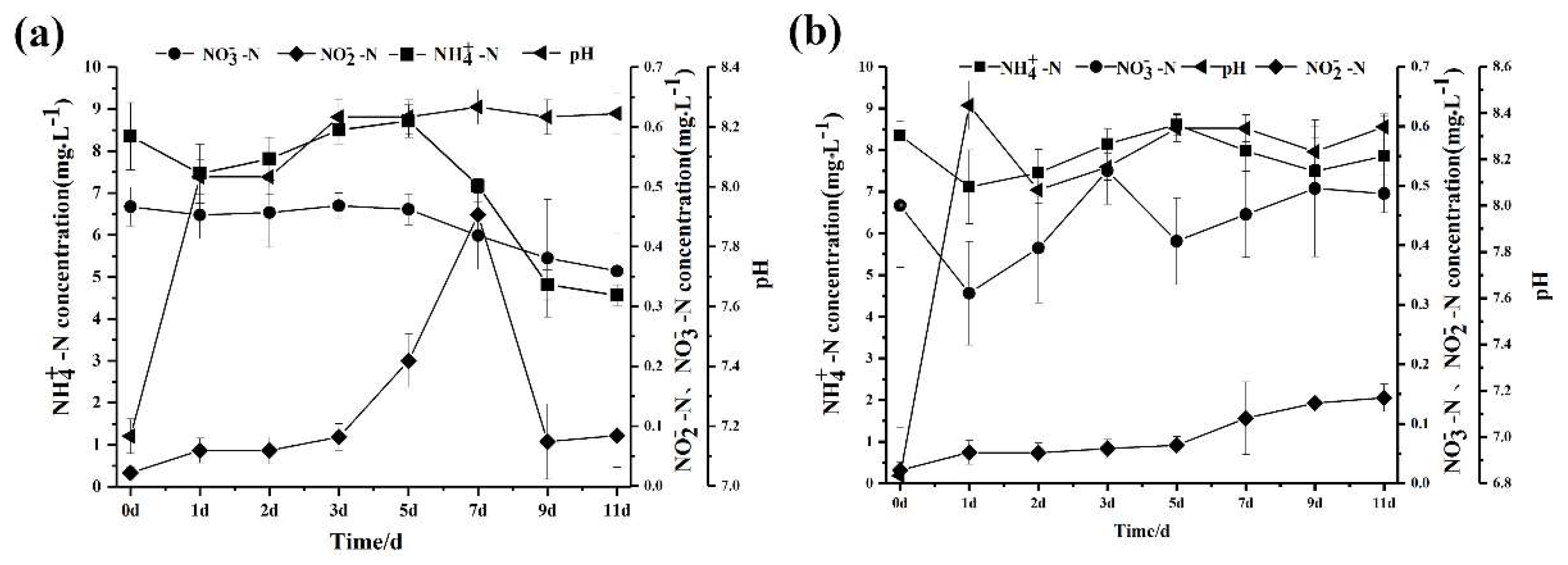
3.7. The Application of Strain SC16 in Aquaculture Water
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cao, L.; Naylor, R.; Henriksson, P.; Leadbitter, D.; Metian, M.; Troell, M.; Zhang, W. China’s aquaculture and the world’s wild fisheries. Science 2015, 347, 133. [Google Scholar] [CrossRef] [PubMed]
- Fishery Bureau, Ministry of Agriculture, People’s Republic of China. China Fisheries Yearbook 2019; China Agriculture Press: Beijing, Chian, 2019.
- Zhou, Q.; Takenaka, S.; Murakami, S.; Seesuriyachan, P.; Kuntiya, A.; Aoki, K. Screening and characterization of bacteria that can utilize ammonium and nitrate ions simultaneously under controlled cultural conditions. J. Biosci. Bioeng. 2007, 103, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Camargo, J.A.; Alonso, A. Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Environ. Int. 2006, 32, 831–849. [Google Scholar] [CrossRef] [PubMed]
- Crab, R.; Avnimelech, Y.; Defoirdt, T.; Bossier, P.; Verstraete, W. Nitrogen removal techniques in aquaculture for a sustainable production. Aquaculture 2007, 270, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Chen, P.; Li, J.; Li, Q.X.; Wang, Y.; Li, S.; Ren, T.; Wang, L. Simultaneous heterotrophic nitrification and aerobic denitrification by bacterium Rhodococcus sp. CPZ24. Bioresour. Technol. 2012, 116, 266–270. [Google Scholar] [CrossRef]
- Robertson, L.A.; Kuenen, J.G. Combined heterotrophic nitrification and aerobic denitrification in Thiosphaera pantotropha and other bacteria. Antonie Leeuwenhoek 1990, 57, 139–152. [Google Scholar] [CrossRef]
- Padhi, S.K.; Tripathy, S.; Mohanty, S.; Maiti, N.K. Aerobic and heterotrophic nitrogen removal by Enterobacter cloacae CF-S27 with efficient utilization of hydroxylamine. Bioresour. Technol. 2017, 232, 285–296. [Google Scholar] [CrossRef]
- Kim, M.; Jeong, S.Y.; Yoon, S.J.; Cho, S.J.; Kim, Y.H.; Kim, M.J.; Ryu, E.Y.; Lee, S.J. Aerobic denitrification of Pseudomonas putida AD-21 at different C/N ratios. J. Biosci. Bioeng. 2008, 106, 498–502. [Google Scholar] [CrossRef]
- Joo, H.S.; Hirai, M.; Shoda, M. Piggery wastewater treatment using Alcaligenes faecalis strain No. 4 with heterotrophic nitrification and aerobic denitrification. Water Res. 2006, 40, 3029–3036. [Google Scholar] [CrossRef]
- Su, J.J.; Liu, B.Y.; Liu, C.Y. Comparison of aerobic denitrification under high oxygen atmosphere by Thiosphaera pantotropha ATCC 35512 and Pseudomonas stutzeri SU2 newly isolated from the activated sludge of a piggery wastewater treatment system. J. Appl. Microbiol. 2001, 90, 457–462. [Google Scholar] [CrossRef]
- Yang, X.P.; Wang, S.M.; Zhang, D.W.; Zhou, L.X. Isolation and nitrogen removal characteristics of an aerobic heterotrophic nitrifying-denitrifying bacterium, Bacillus subtilis A1. Bioresour. Technol. 2011, 102, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Gao, D.; Liu, H.; Cai, J.; Zhang, J.; Qi, Z. Biopotentiality of high efficient aerobic denitrifier Bacillus megaterium S379 for intensive aquaculture water quality management. J. Environ. Manag. 2018, 222, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Zumft, W.G. Cell biology and molecular basis of denitrification. Microbiol. Mol. Biol. R. 1997, 61, 533–616. [Google Scholar] [CrossRef]
- Rajta, A.; Bhatia, R.; Setia, H.; Pathania, P. Role of heterotrophic aerobic denitrifying bacteria in nitrate removal from wastewater. J. Appl. Microbiol. 2020, 128, 1261–1278. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yang, J.; Wang, X.; Wang, E.; Li, B.; He, R.; Yuan, H. Removal of nitrogen by heterotrophic nitrification-aerobic denitrification of a phosphate accumulating bacterium Pseudomonas stutzeri YG-24. Bioresour. Technol. 2015, 182, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Jia, Y.; Chen, Y.; Hu, Y. Simultaneous nitrification and denitrification without nitrite accumulation by a novel isolated Ochrobactrum anthropic LJ81. Bioresour. Technol. 2019, 272, 442–450. [Google Scholar] [CrossRef]
- Wittorf, L.; Jones, C.M.; Bonilla-Rosso, G.; Hallin, S. Expression of nirK and nirS genes in two strains of Pseudomonas stutzeri harbouring both types of NO-forming nitrite reductases. Res. Microbiol. 2018, 169, 343–347. [Google Scholar] [CrossRef]
- Song, Z.F.; An, J.; Fu, G.H.; Yang, X.L. Isolation and characterization of an aerobic denitrifying Bacillus sp. YX-6 from shrimp culture ponds. Aquaculture 2011, 319, 188–193. [Google Scholar] [CrossRef]
- Zhao, B.; He, Y.L.; Huang, J.; Taylor, S.; Hughes, J. Heterotrophic nitrogen removal by Providencia rettgeri strain YL. J. Ind. Microbiol. Biotechnol. 2010, 37, 609–616. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Yang, L.; Ren, Y.X.; Zhao, S.Q.; Liang, X.; Wang, J.P. Isolation and characterization of three heterotrophic nitrifying-aerobic denitrifying bacteria from a sequencing batch reactor. Ann. Microbiol. 2015, 66, 737–747. [Google Scholar] [CrossRef]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 19th ed.; American Public Health Association: Washington, DC, USA, 1985. [Google Scholar]
- He, T.; Li, Z.; Sun, Q.; Xu, Y.; Ye, Q. Heterotrophic nitrification and aerobic denitrification by Pseudomonas tolaasii Y-11 without nitrite accumulation during nitrogen conversion. Bioresour. Technol. 2016, 200, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Padhi, S.K.; Tripathy, S.; Sen, R.; Mahapatra, A.S.; Mohanty, S.; Maiti, N.K. Characterisation of heterotrophic nitrifying and aerobic denitrifying Klebsiella pneumoniae CF-S9 strain for bioremediation of wastewater. Int. Biodeter. Biodegr. 2013, 78, 67–73. [Google Scholar] [CrossRef]
- Sun, Y.; Li, A.; Zhang, X.; Ma, F. Regulation of dissolved oxygen from accumulated nitrite during the heterotrophic nitrification and aerobic denitrification of Pseudomonas stutzeri T13. Appl. Microbiol. Biot. 2015, 99, 3243–3248. [Google Scholar] [CrossRef]
- He, X.; Sun, Q.; Xu, T.; Dai, M.; Wei, D. Removal of nitrogen by heterotrophic nitrification-aerobic denitrification of a novel halotolerant bacterium Pseudomonas mendocina TJPU04. Bioproc. Biosyst. Eng. 2019, 42, 853–866. [Google Scholar] [CrossRef]
- He, T.; Xie, D.; Li, Z.; Ni, J.; Sun, Q. Ammonium stimulates nitrate reduction during simultaneous nitrification and denitrification process by Arthrobacter arilaitensis Y-10. Bioresour. Technol. 2017, 239, 66–73. [Google Scholar] [CrossRef]
- Zhang, Q.L.; Liu, Y.; Ai, G.M.; Miao, L.L.; Zheng, H.Y.; Liu, Z.P. The characteristics of a novel heterotrophic nitrification–aerobic denitrification bacterium, Bacillus methylotrophicus strain L7. Bioresour. Technol. 2012, 108, 35–44. [Google Scholar] [CrossRef]
- Huang, F.; Pan, L.; Lv, N.; Tang, X. Characterization of novel Bacillus strain N31 from mariculture water capable of halophilic heterotrophic nitrification-aerobic denitrification. J. Biosci. Bioeng. 2017, 124, 564–571. [Google Scholar] [CrossRef]
- Guo, L.; Chen, Q.; Fang, F.; Hu, Z.; Wu, J.; Miao, A.; Xiao, L.; Chen, X.; Yang, L. Application potential of a newly isolated indigenous aerobic denitrifier for nitrate and ammonium removal of eutrophic lake water. Bioresour. Technol. 2013, 142, 45–51. [Google Scholar] [CrossRef]
- Huang, X.; Li, W.; Zhang, D.; Qin, W. Ammonium removal by a novel oligotrophic Acinetobacter sp. Y16 capable of heterotrophic nitrification-aerobic denitrification at low temperature. Bioresour. Technol. 2013, 146, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, S.; Zhou, L. Effect of carbon source, C/N ratio, nitrate and dissolved oxygen concentration on nitrite and ammonium production from denitrification process by Pseudomonas stutzeri D6. Bioresour. Technol. 2012, 104, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, H.F.; Tiedje, J.M.; Firestone, R.B. Denitrification and dissimilatory nitrate reduction to ammonium in digested sludge. Can. J. Microbiol. 1981, 27, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Joo, H.S.; Hirai, M.; Shoda, M. Characteristics of ammonium removal by heterotrophic nitrification-aerobic denitrification by Alcaligenes faecalis No. 4. J. Biosci. Bioeng. 2005, 100, 184–191. [Google Scholar] [CrossRef]
- Doi, Y.; Takaya, N.; Takizawa, N. Novel denitrifying bacterium Ochrobactrum anthropi YD50.2 tolerates high levels of reactive nitrogen oxides. Appl. Environ. Microb. 2009, 75, 5186–5194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brittain, T.; Blackmore, R.; Greenwood, C.; Thomson, A.J. Bacterial nitrite-reducing enzymes. Eur. J. Biochem. 1992, 209, 793–802. [Google Scholar] [CrossRef]
- Vaccaro, B.J.; Thorgersen, M.P.; Lancaster, W.A.; Price, M.N.; Wetmore, K.M.; Poole, F.L.; Deutschbauer, A.; Arkin, A.P.; Adams, M.W. Determining roles of accessory genes in denitrification by mutant fitness analyses. Appl. Environ. Microb. 2016, 82, 51–61. [Google Scholar] [CrossRef] [Green Version]
- Gao, H.; Li, C.; Ramesh, B.; Hu, N. Cloning, purification and characterization of novel Cu-containing nitrite reductase from the Bacillus firmus GY-49. World J. Microb. Biot. 2017, 34, 10. [Google Scholar] [CrossRef]
- Braker, G.; Zhou, J.; Wu, L.; Devol, A.H.; Tiedje, J.M. Nitrite reductase genes (nirK and nirS) as functional markers to investigate diversity of denitrifying bacteria in pacific northwest marine sediment communities. Appl. Environ. Microb. 2000, 66, 2096–2104. [Google Scholar] [CrossRef] [Green Version]
- Huang, T.; Guo, L.; Zhang, H.; Su, J.; Wen, G.; Zhang, K. Nitrogen-removal efficiency of a novel aerobic denitrifying bacterium, Pseudomonas stutzeri strain ZF31, isolated from a drinking-water reservoir. Bioresour. Technol. 2015, 196, 209–216. [Google Scholar] [CrossRef]
- Wan, C.; Yang, X.; Lee, D.-J.; Du, M.; Wan, F.; Chen, C. Aerobic denitrification by novel isolated strain using NO2–N as nitrogen source. Bioresour. Technol. 2011, 102, 7244–7248. [Google Scholar] [CrossRef] [PubMed]
- Jin, P.; Chen, Y.; Yao, R.; Zheng, Z.; Du, Q. New insight into the nitrogen metabolism of simultaneous heterotrophic nitrification-aerobic denitrification bacterium in mRNA expression. J. Hazard. Mater. 2019, 371, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Li, W.; Deng, Y.; Ruan, Y.; Chen, G.; Yu, J.; Lin, F. Denitrification potential evaluation and nitrate-removal-pathway analysis of aerobic denitrifier strain Marinobacter hydrocarbonoclasticus RAD-2. Water 2018, 10, 1298. [Google Scholar] [CrossRef] [Green Version]


| Time (h) | Nitrite Concentration (mg·L−1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.1 | 2 | 50 | 100 | 200 | 250 | 300 | 500 | 1000 | |
| 0 | 0.091 ± 0.03 | 1.98 ± 0.088 | 49.52 ± 0.29 | 99.71 ± 0.19 | 199.76 ± 6.39 | 248.57 ± 3.29 | 297.25 ± 5.58 | 499.46 ± 17.26 | 992.96 ± 11.17 |
| 12 | 0.031 ± 0.05 | 0.043 ± 0.027 | 0.2 ± 0.15 | 0.71 ± 0.19 | 156.84 ± 4.35 | 200.48 ± 5.89 | 249.65 ± 9.19 | 438.24 ± 18.19 | 934.81 ± 14.07 |
| 24 | 0.02 ± 0.05 | 0.043 ± 0.058 | 0.1 ± 0.17 | 0.071 ± 0.17 | 1.29 ± 1.19 | 176.78 ± 1.58 | 246.89 ± 7.13 | 435.2 ± 16.52 | 933.62 ± 13.32 |
| 36 | 0.015 ± 0.02 | 0.04 ± 0.02 | 0.02 ± 0.01 | 0.07 ± 0.05 | 0.11 ± 0.18 | 138.76 ± 3.19 | 245.53 ± 6.73 | 434.34 ± 9.8 | 932.42 ± 12.3 |
| 48 | 0.01 ± 0.03 | 0.03 ± 0.02 | 0.015 ± 0.011 | 0.04 ± 0.02 | 0.039 ± 0.02 | 106.78 ± 2.79 | 240.89 ± 9.87 | 430.25 ± 11.23 | 932.23 ± 10.02 |
| Removal a | 0.09 ± 0.03 | 1.97 ± 0.02 | 49.985 ± 0.011 | 99.96 ± 0.02 | 199.98 ± 0.02 | 143.22 ± 2.79 | 59.11 ± 9.87 | 69.75 ± 11.23 | 67.77 ± 10.02 |
| Removal percentage (%) b | 90 ± 0.3 | 98.5 ± 0.01 | 99.97 ± 0.00 | 99.96 ± 0.00 | 99.99 ± 0.00 | 57.29 ± 0.01 | 19.7 ± 0.03 | 13.95 ± 0.02 | 6.78 ± 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sang, C.-G.; Fu, Y.-W.; Guo, S.-Q.; Luo, J.-J.; Zhang, Q.-Z. Isolation and Characterization of an Aerobic Denitrifier Bacillus sp. SC16 from an Intensive Aquaculture Pond. Water 2020, 12, 3559. https://doi.org/10.3390/w12123559
Sang C-G, Fu Y-W, Guo S-Q, Luo J-J, Zhang Q-Z. Isolation and Characterization of an Aerobic Denitrifier Bacillus sp. SC16 from an Intensive Aquaculture Pond. Water. 2020; 12(12):3559. https://doi.org/10.3390/w12123559
Chicago/Turabian StyleSang, Chi-Guo, Yao-Wu Fu, Shu-Quan Guo, Jia-Ju Luo, and Qi-Zhong Zhang. 2020. "Isolation and Characterization of an Aerobic Denitrifier Bacillus sp. SC16 from an Intensive Aquaculture Pond" Water 12, no. 12: 3559. https://doi.org/10.3390/w12123559