Achieving Partial Nitrification via Intermittent Aeration in SBR and Short-Term Effects of Different C/N Ratios on Reactor Performance and Microbial Community Structure
Abstract
:1. Introduction
2. Materials and Methods
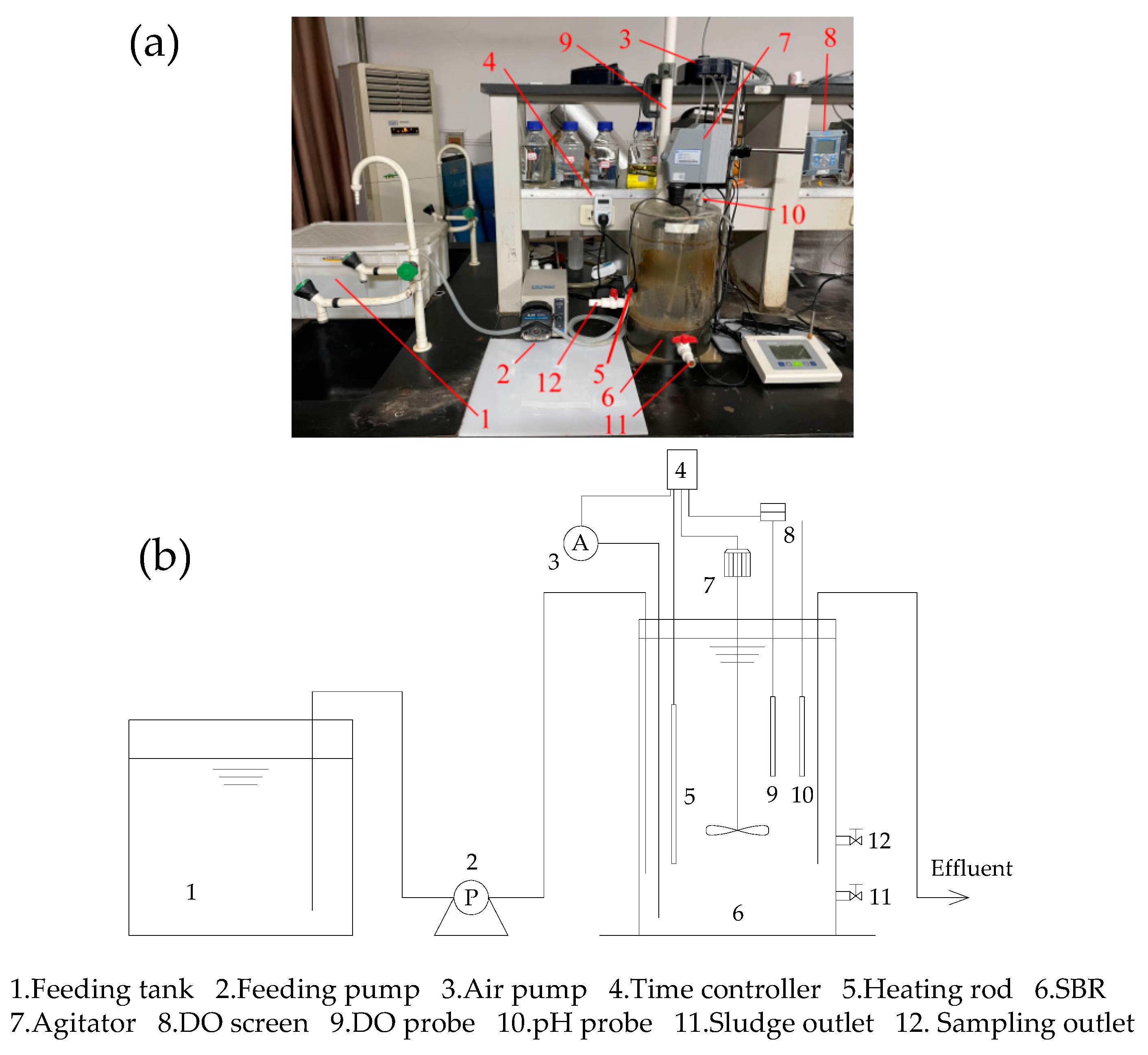
2.1. The SBR Setup and Operation Protocol
2.2. Synthetic Wastewater and Seed Sludge
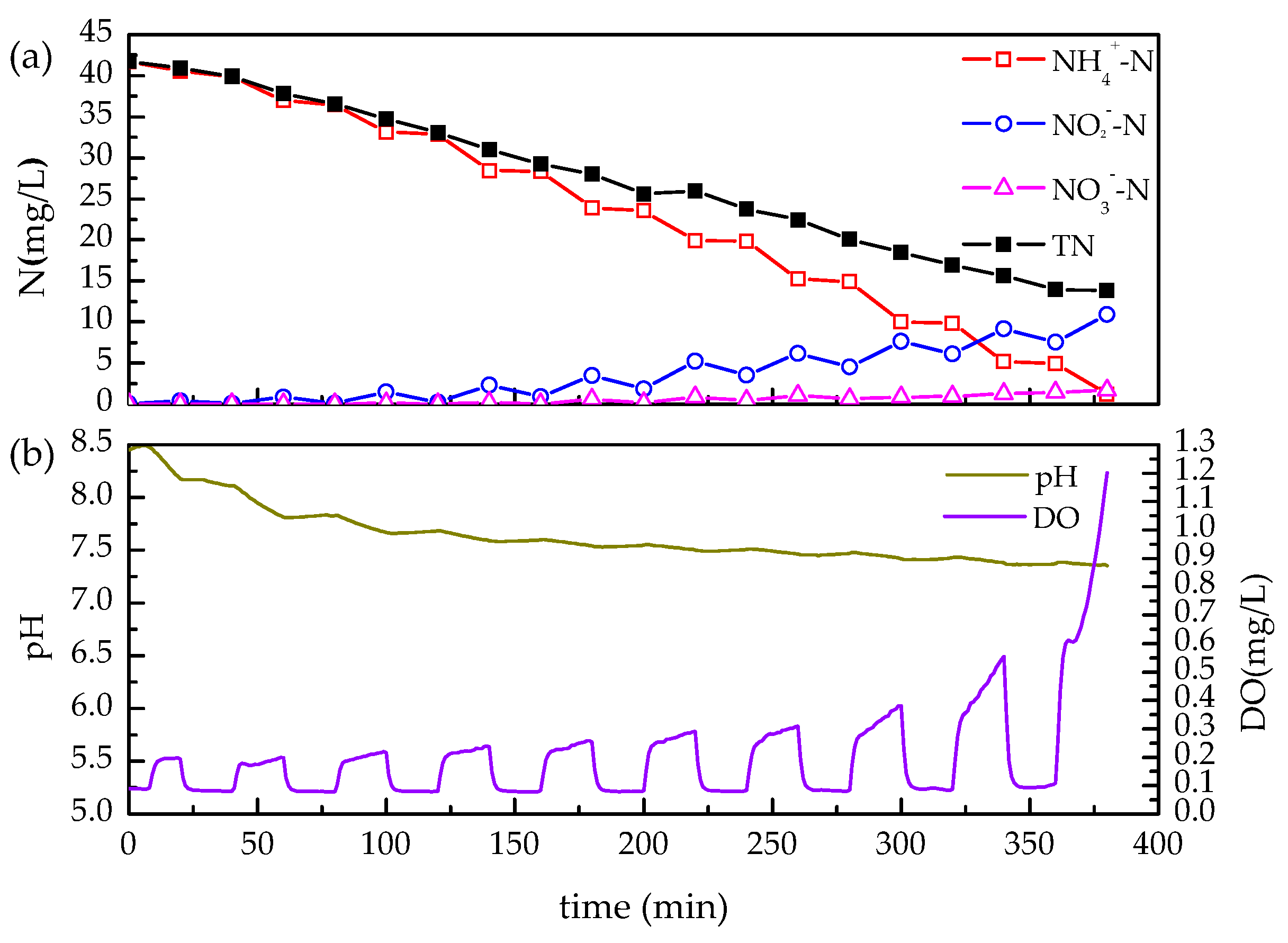
2.3. Batch Tests
2.4. Analytical Methods
2.5. DNA Extraction, PCR Amplification, and High-Throughput Sequencing Analysis
3. Results and Discussion
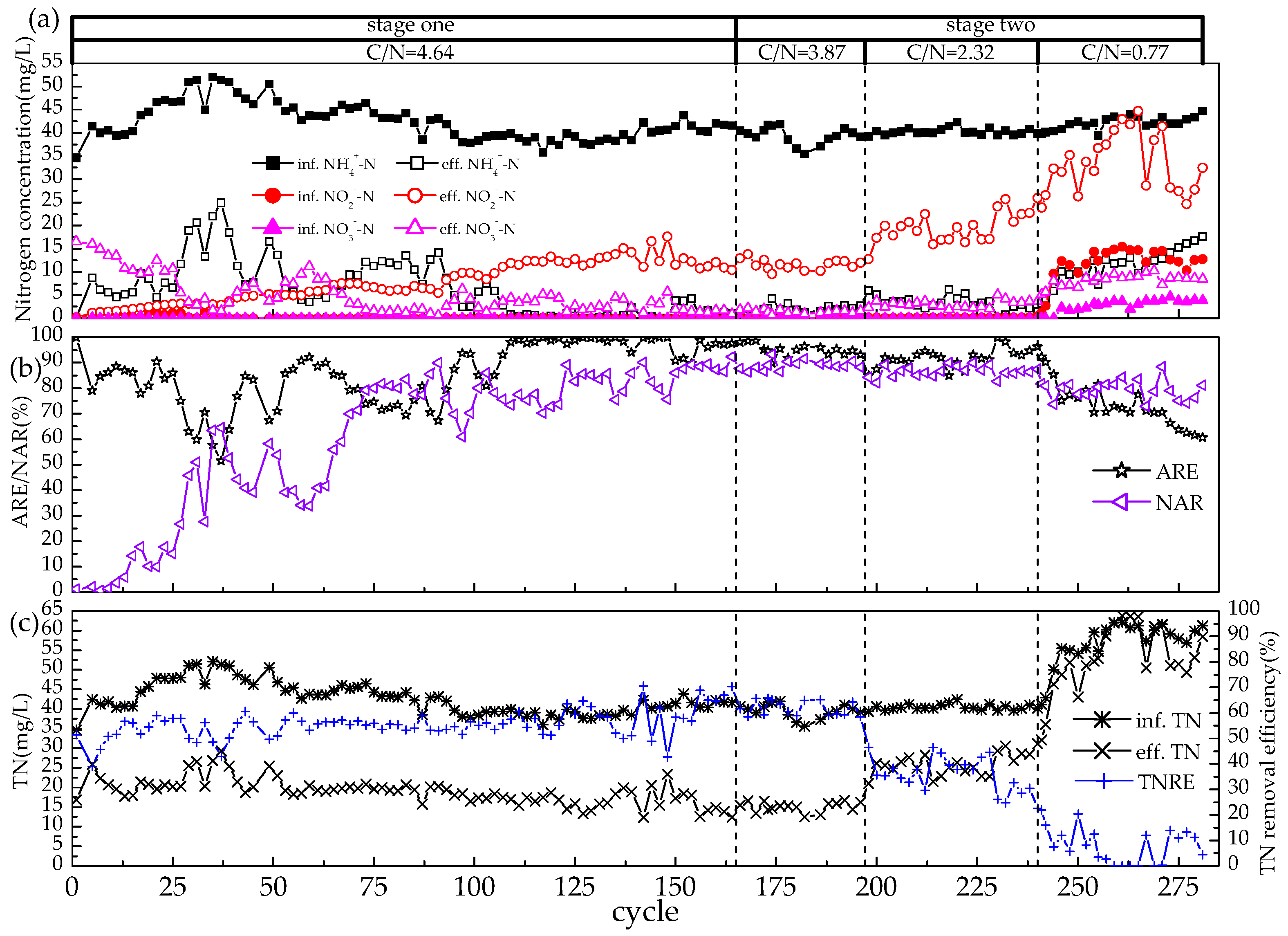
3.1. Achievement of Partial Nitrification via Intermittent Aeration
3.2. Short-Term Effects of Different C/N Ratios on the Reactor Performance
3.3. Microbial Community Analysis
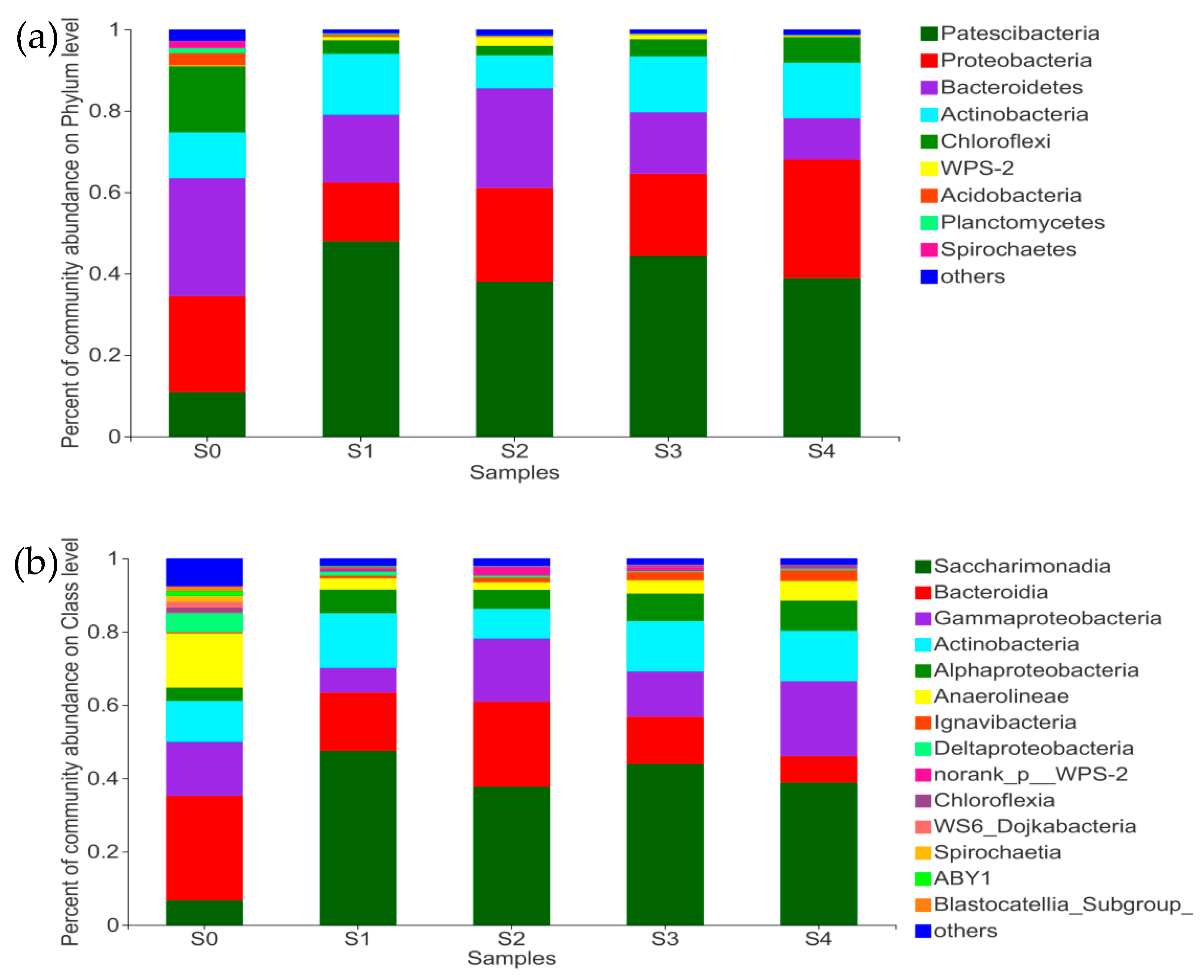
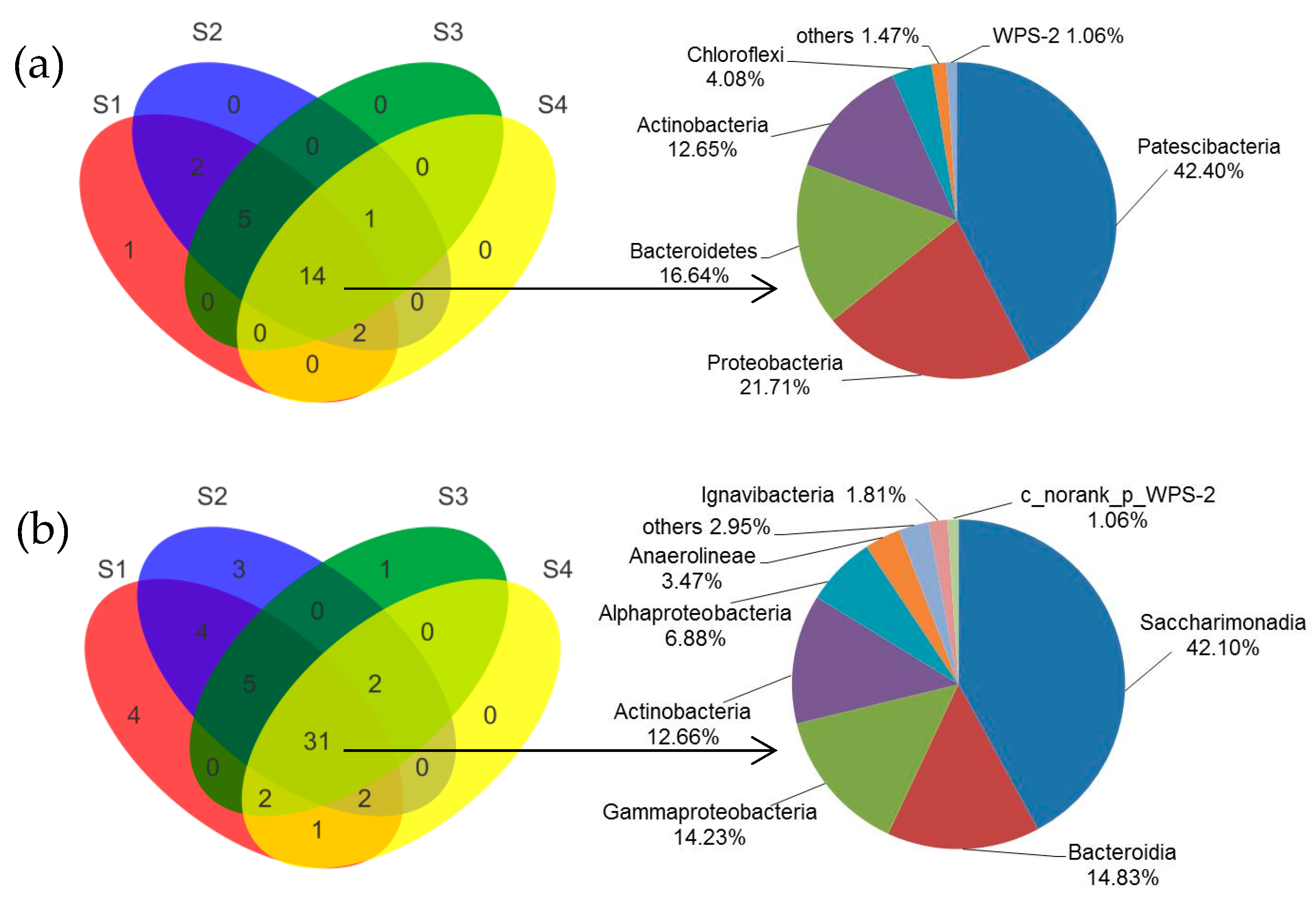
3.3.1. Changes of Microbial Community after Achieving Partial Nitrification
3.3.2. Shift of Microbial Community under Different C/N Ratios
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences—An application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.Z.; Bao, P.; Wang, B.; Zhang, Q.; Peng, Y.Z. Long-term stable simultaneous partial nitrification, anammox and denitrification (SNAD) process treating real domestic sewage using suspended activated sludge. Chem. Eng. J. 2018, 339, 180–188. [Google Scholar] [CrossRef]
- Soliman, M.; Eldyasti, A. Development of partial nitrification as a first step of nitrite shunt process in a Sequential Batch Reactor (SBR) using Ammonium Oxidizing Bacteria (AOB) controlled by mixing regime. Bioresour. Technol. 2016, 221, 85–95. [Google Scholar] [CrossRef]
- Liu, W.L.; Yang, Q.; Ma, B.; Li, J.; Ma, L.N.; Wang, S.Y.; Peng, Y.Z. Rapid Achievement of Nitritation Using Aerobic Starvation. Environ. Sci. Technol. 2017, 51, 4001–4008. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.G.; Wang, X.J.; Yang, Y.Y.; Mirino, M.W.; Yuan, Y.L. Partial nitrification and denitrification of mature landfill leachate using a pilot-scale continuous activated sludge process at low dissolved oxygen. Bioresour. Technol. 2016, 218, 580–588. [Google Scholar] [CrossRef]
- Isanta, E.; Reino, C.; Carrera, J.; Perez, J. Stable partial nitritation for low-strength wastewater at low temperature in an aerobic granular reactor. Water Res. 2015, 80, 149–158. [Google Scholar] [CrossRef] [Green Version]
- Qian, W.T.; Peng, Y.Z.; Li, X.Y.; Zhang, Q.; Ma, B. The inhibitory effects of free ammonia on ammonia oxidizing bacteria and nitrite oxidizing bacteria under anaerobic condition. Bioresour. Technol. 2017, 243, 1247–1250. [Google Scholar] [CrossRef]
- Kong, Q.; Zhang, J.; Miao, M.S.; Tian, L.; Guo, N.; Liang, S. Partial nitrification and nitrous oxide emission in an intermittently aerated sequencing batch biofilm reactor. Chem. Eng. J. 2013, 217, 435–441. [Google Scholar] [CrossRef]
- Xu, G.J.; Xu, X.C.; Yang, F.L.; Liu, S.T.; Gao, Y. Partial nitrification adjusted by hydroxylamine in aerobic granules under high DO and ambient temperature and subsequent Anammox for low C/N wastewater treatment. Chem. Eng. J. 2012, 213, 338–345. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Chen, Z.G.; Wang, X.J.; Zheng, L.; Gu, X.Y. Partial nitrification performance and mechanism of zeolite biological aerated filter for ammonium wastewater treatment. Bioresour. Technol. 2017, 241, 473–481. [Google Scholar] [CrossRef]
- Jiang, H.C.; Liu, G.H.; Ma, Y.M.; Xu, X.L.; Chen, J.H.; Yang, Y.Q.; Liu, X.H.; Wang, H.C. A pilot-scale study on start-up and stable operation of mainstream partial nitrification-anammox biofilter process based on online pH-DO linkage control. Chem. Eng. J. 2018, 350, 1035–1042. [Google Scholar] [CrossRef]
- Kornaros, M.; Dokianakis, S.N.; Lyberatos, G. Partial Nitrification/Denitrification Can Be Attributed to the Slow Response of Nitrite Oxidizing Bacteria to Periodic Anoxic Disturbances. Environ. Sci. Technol. 2010, 44, 7245–7253. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.M.; Chen, S.L. Effects of organic carbon on nitrification rate in fixed film biofilters. Aquac. Eng. 2001, 25, 1–11. [Google Scholar] [CrossRef]
- Capodici, M.; Corsino, S.; Di Trapani, D.; Viviani, G. Achievement of partial nitrification under different carbon-to-nitrogen ratio and ammonia loading rate for the co-treatment of landfill leachate with municipal wastewater. Biochem. Eng. J. 2019, 149, 9. [Google Scholar] [CrossRef]
- Miao, Y.Y.; Peng, Y.Z.; Zhang, L.; Li, B.K.; Li, X.Y.; Wu, L.; Wang, S.M. Partial nitrification-anammox (PNA) treating sewage with intermittent aeration mode: Effect of influent C/N ratios. Chem. Eng. J. 2018, 334, 664–672. [Google Scholar] [CrossRef]
- Li, X.Y.; Yang, S.F. Influence of loosely bound extracellular polymeric substances (EPS) on the flocculation, sedimentation and dewaterability of activated sludge. Water Res. 2007, 41, 1022–1030. [Google Scholar] [CrossRef]
- Frolund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996, 30, 1749–1758. [Google Scholar] [CrossRef]
- Jenni, S.; Vlaeminck, S.E.; Morgenroth, E.; Udert, K.M. Successful application of nitritation/anammox to wastewater with elevated organic carbon to ammonia ratios. Water Res. 2014, 49, 316–326. [Google Scholar] [CrossRef] [Green Version]
- Park, S.; Bae, W.; Chung, J.; Baek, S.-C. Empirical model of the pH dependence of the maximum specific nitrification rate. Process Biochem. 2007, 42, 1671–1676. [Google Scholar] [CrossRef]
- He, Y.; Tao, W.; Wang, Z.; Shayya, W. Effects of pH and seasonal temperature variation on simultaneous partial nitrification and anammox in free-water surface wetlands. J. Environ. Manag. 2012, 110, 103–109. [Google Scholar] [CrossRef]
- Wu, C.; Peng, Y.; Wang, S.; Li, B.; Zhang, L.; Cao, S.; Du, R. Mechanisms of nitrite addition for simultaneous sludge fermentation/nitrite removal (SFNR). Water Res. 2014, 64, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Anthonisen, A.C.; Loehr, R.C.; Prakasam, T.B.S.; Srinath, E.G. Inhibition of nitrification by ammonia and nitrous-acid. J. Water Pollut. Control Fed. 1976, 48, 835–852. [Google Scholar] [PubMed]
- Du, R.; Cao, S.B.; Li, B.K.; Niu, M.; Wang, S.Y.; Peng, Y.Z. Performance and microbial community analysis of a novel DEAMOX based on partial-denitrification and anammox treating ammonia and nitrate wastewaters. Water Res. 2017, 108, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Geets, J.; Boon, N.; Verstraete, W. Strategies of aerobic ammonia-oxidizing bacteria for coping with nutrient and oxygen fluctuations. FEMS Microbiol. Ecol. 2006, 58, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Ge, S.J.; Peng, Y.Z.; Qiu, S.; Zhu, A.; Ren, N.Q. Complete nitrogen removal from municipal wastewater via partial nitrification by appropriately alternating anoxic/aerobic conditions in a continuous plug-flow step feed process. Water Res. 2014, 55, 95–105. [Google Scholar] [CrossRef]
- Ge, S.J.; Wang, S.Y.; Yang, X.; Qiu, S.; Li, B.K.; Peng, Y.Z. Detection of nitrifiers and evaluation of partial nitrification for wastewater treatment: A review. Chemosphere 2015, 140, 85–98. [Google Scholar] [CrossRef]
- Tanwar, P.; Nandy, T.; Ukey, P.; Manekar, P. Correlating on-line monitoring parameters, pH, DO and ORP with nutrient removal in an intermittent cyclic process bioreactor system. Bioresour. Technol. 2008, 99, 7630–7635. [Google Scholar] [CrossRef]
- Zafarzadeh, A.; Bina, B.; Nikaeen, M.; Attar, H.M.; Khiadani, M.H. Effect of dissolved oxygen and chemical oxygen demand to nitrogen ratios on the partial nitrification/denitrification process in moving bed biofilm reactors. Iran. J. Biotechnol. 2011, 9, 197–205. [Google Scholar]
- More, T.T.; Yadav, J.S.S.; Yan, S.; Tyagi, R.D.; Surampalli, R.Y. Extracellular polymeric substances of bacteria and their potential environmental applications. J. Environ. Manag. 2014, 144, 1–25. [Google Scholar] [CrossRef]
- Wang, B.; Peng, Y.Z.; Guo, Y.Y.; Zhao, M.Y.; Wang, S.Y. Illumina MiSeq sequencing reveals the key microorganisms involved in partial nitritation followed by simultaneous sludge fermentation, denitrification and anammox process. Bioresour. Technol. 2016, 207, 118–125. [Google Scholar] [CrossRef]
- Dorador, C.; Meneses, D.; Urtuvia, V.; Demergasso, C.; Vila, I.; Witzel, K.-P.; Imhoff, J.F. Diversity of Bacteroidetes in high-altitude saline evaporitic basins in northern Chile. J. Geophys. Res.-Biogeosci. 2009, 114. [Google Scholar] [CrossRef] [Green Version]
- Miura, Y.; Watanabe, Y.; Okabe, S. Significance of Chloroflexi in performance of submerged membrane Bioreactors (MBR) treating municipal wastewater. Environ. Sci. Technol. 2007, 41, 7787–7794. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Wang, W.Q.; Zhang, J.; Wang, S.Y.; Li, J. Dominance of Candidatus saccharibacteria in SBRs achieving partial denitrification: Effects of sludge acclimating methods on microbial communities and nitrite accumulation. RSC Adv. 2019, 9, 11263–11271. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Dong, Y.H.; Qian, G.S.; Hu, X.; Ye, L.L. Performance and microbial community analysis of bio-electrocoagulation on simultaneous nitrification and denitrification in submerged membrane bioreactor at limited dissolved oxygen. Bioresour. Technol. 2018, 258, 168–176. [Google Scholar] [CrossRef]
- Tian, R.M.; Ning, D.L.; He, Z.L.; Zhang, P.; Spencer, S.J.; Gao, S.H.; Shi, W.L.; Wu, L.W.; Zhang, Y.; Yang, Y.F.; et al. Small and mighty: Adaptation of superphylum Patescibacteria to groundwater environment drives their genome simplicity. Microbiome 2020, 8, 15. [Google Scholar] [CrossRef] [Green Version]
- Herrmann, M.; Wegner, C.E.; Taubert, M.; Geesink, P.; Lehmann, K.; Yan, L.J.; Lehmann, R.; Totsche, K.U.; Kusel, K. Predominance of Cand. Patescibacteria in Groundwater Is Caused by Their Preferential Mobilization from Soils and Flourishing under Oligotrophic Conditions. Front. Microbiol. 2019, 10, 15. [Google Scholar] [CrossRef] [Green Version]
- Sowell, S.M.; Wilhelm, L.J.; Norbeck, A.D.; Lipton, M.S.; Nicora, C.D.; Barofsky, D.F.; Carlson, C.A.; Smith, R.D.; Giovanonni, S.J. Transport functions dominate the SAR11 metaproteome at low-nutrient extremes in the Sargasso Sea. ISME J. 2009, 3, 93–105. [Google Scholar] [CrossRef] [Green Version]
- Mao, Y.P.; Xia, Y.; Zhang, T. Characterization of Thauera-dominated hydrogen-oxidizing autotrophic denitrifying microbial communities by using high-throughput sequencing. Bioresour. Technol. 2013, 128, 703–710. [Google Scholar] [CrossRef]
- Huang, W.Y.; She, Z.L.; Gao, M.C.; Wang, Q.; Jin, C.J.; Zhao, Y.G.; Guo, L. Effect of anaerobic/aerobic duration on nitrogen removal and microbial community in a simultaneous partial nitrification and denitrification system under low salinity. Sci. Total Environ. 2019, 651, 859–870. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, J.; Zhou, S.; Wang, X.; Chen, Y.; Lin, X.; Yan, Y.; Ma, X.; Wu, M.; Han, H. 16S rRNA gene high-throughput sequencing reveals shift in nitrogen conversion related microorganisms in a CANON system in response to salt stress. Chem. Eng. J. 2017, 317, 512–521. [Google Scholar] [CrossRef]
- Narihiro, T.; Terada, T.; Ohashi, A.; Kamagata, Y.; Nakamura, K.; Sekiguchi, Y. Quantitative detection of previously characterized syntrophic bacteria in anaerobic wastewater treatment systems by sequence-specific rRNA cleavage method. Water Res. 2012, 46, 2167–2175. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.C.; Lu, X.; Ding, X.F.; Wu, B.; Wang, D.P.; Huang, L.; Wang, X.L.; Tan, Y.F.; Zhang, X.X.; Liu, B. The total and functional bacterial community of nitrogen removal in the SND ditches. Int. Biodeterior. Biodegrad. 2017, 118, 102–109. [Google Scholar] [CrossRef]
- Chen, Y.S.; Zhao, Z.; Peng, Y.K.; Li, J.; Xiao, L.; Yang, L.Y. Performance of a full-scale modified anaerobic/anoxic/oxic process: High-throughput sequence analysis of its microbial structures and their community functions. Bioresour. Technol. 2016, 220, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Gong, B.Z.; Huang, W.; Wang, Y.M.; Zhou, J. Bacterial community structure in simultaneous nitrification, denitrification and organic matter removal process treating saline mustard tuber wastewater as revealed by 16S rRNA sequencing. Bioresour. Technol. 2017, 228, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Fang, D.X.; Zhao, G.; Xu, X.Y.; Zhang, Q.; Shen, Q.S.; Fang, Z.Y.; Huang, L.P.; Ji, F.Y. Microbial community structures and functions of wastewater treatment systems in plateau and cold regions. Bioresour. Technol. 2018, 249, 684–693. [Google Scholar] [CrossRef]

| C/N | Cycle | SAOB | SNOB |
|---|---|---|---|
| (mg N/(g MLSS·h)) | (mg N/(g MLSS·h)) | ||
| 4.64 | 29 | 5.25 | 3.48 |
| 113 | 3.89 | 0.96 | |
| 160 | 4.29 | 0.83 | |
| 3.87 | 196 | 5.51 | 1.01 |
| 2.32 | 238 | 6.29 | 1.04 |
| 0.77 | 281 | 2.04 | Almost zero |
| Samples | Sobs | Shannon | Simpson | Ace | Chao | Coverage |
|---|---|---|---|---|---|---|
| S0 | 913 | 5.25 | 0.020 | 967.5 | 968.3 | 0.9968 |
| S1 | 627 | 3.73 | 0.091 | 754.9 | 742.9 | 0.9953 |
| S2 | 481 | 3.62 | 0.055 | 639.7 | 626.0 | 0.9955 |
| S3 | 462 | 3.72 | 0.067 | 595.2 | 605.2 | 0.9961 |
| S4 | 416 | 3.69 | 0.069 | 508.2 | 506.0 | 0.9969 |
| Denitrifying Bacteria | S1 | S2 | S3 | S4 |
|---|---|---|---|---|
| Candidatus_Competibacter | 0.8% | 0.5% | 0.7% | 0.3% |
| Dechloromonas | 1.0% | 10.3% | 2.7% | 0.9% |
| Denitratisoma | 0.0% | 0.0% | 0.2% | 0.3% |
| Dokdonella | 0.0% | 0.0% | 0.0% | 0.2% |
| Hyphomicrobium | 0.2% | 0.4% | 0.4% | 0.6% |
| Pseudomonas | 0.0% | 0.0% | 0.0% | 0.0% |
| Rhodobacter | 0.3% | 1.3% | 2.9% | 3.1% |
| Terrimonas | 0.1% | 0.0% | 0.1% | 0.1% |
| Thauera | 0.1% | 0.0% | 0.0% | 0.0% |
| Zoogloea | 1.8% | 0.3% | 0.1% | 0.0% |
| total | 4.3% | 12.9% | 7.0% | 5.5% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Z.; Qi, M.; Wang, H. Achieving Partial Nitrification via Intermittent Aeration in SBR and Short-Term Effects of Different C/N Ratios on Reactor Performance and Microbial Community Structure. Water 2020, 12, 3485. https://doi.org/10.3390/w12123485
Zhou Z, Qi M, Wang H. Achieving Partial Nitrification via Intermittent Aeration in SBR and Short-Term Effects of Different C/N Ratios on Reactor Performance and Microbial Community Structure. Water. 2020; 12(12):3485. https://doi.org/10.3390/w12123485
Chicago/Turabian StyleZhou, Zhengyao, Meixia Qi, and Hongping Wang. 2020. "Achieving Partial Nitrification via Intermittent Aeration in SBR and Short-Term Effects of Different C/N Ratios on Reactor Performance and Microbial Community Structure" Water 12, no. 12: 3485. https://doi.org/10.3390/w12123485
APA StyleZhou, Z., Qi, M., & Wang, H. (2020). Achieving Partial Nitrification via Intermittent Aeration in SBR and Short-Term Effects of Different C/N Ratios on Reactor Performance and Microbial Community Structure. Water, 12(12), 3485. https://doi.org/10.3390/w12123485




