Comparison of Fish, Macroinvertebrates and Diatom Communities in Response to Environmental Variation in the Wei River Basin, China
Abstract
:1. Introduction
2. Materials and Methods
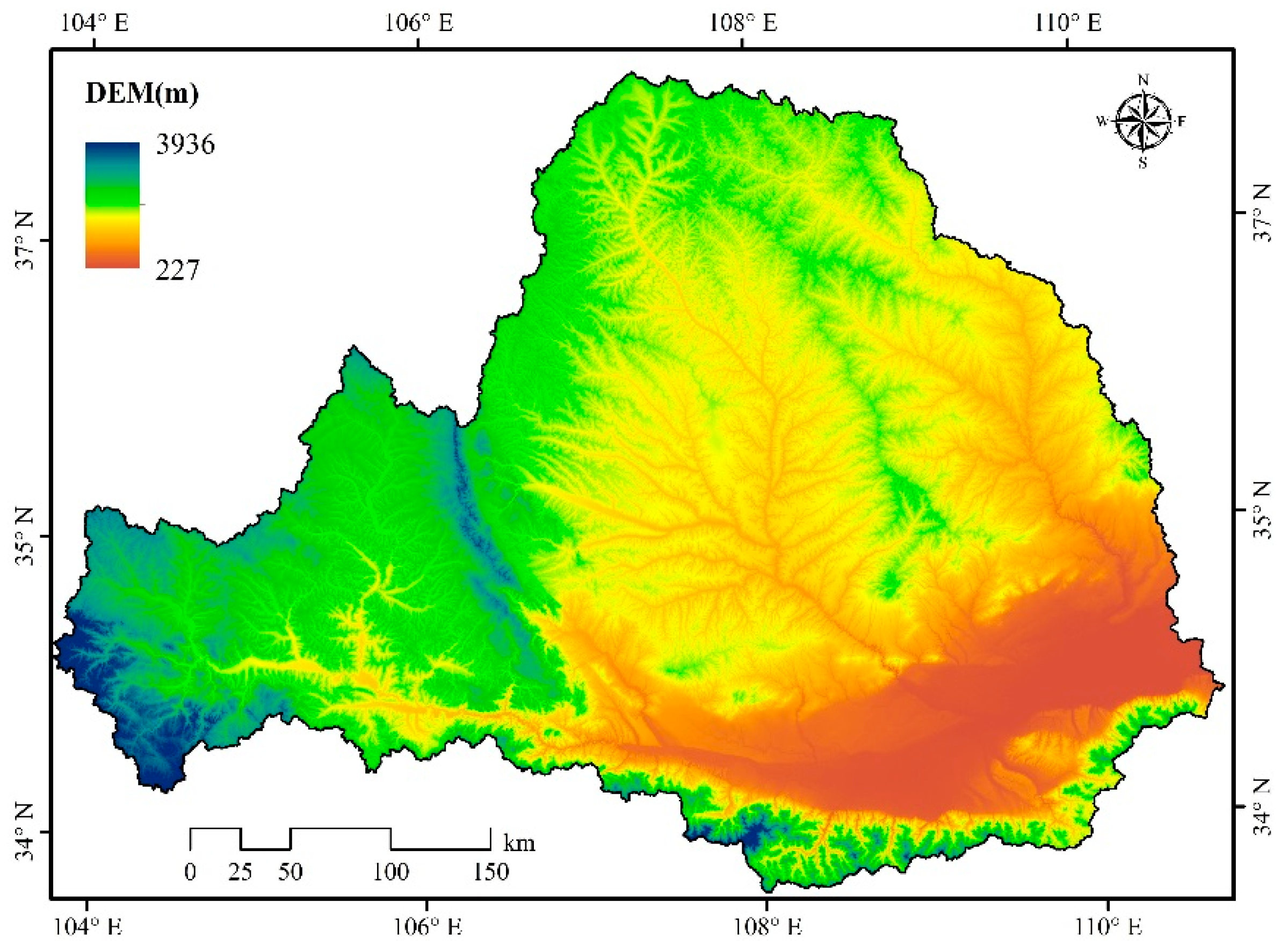
2.1. Study Area Description
2.2. Data Collection
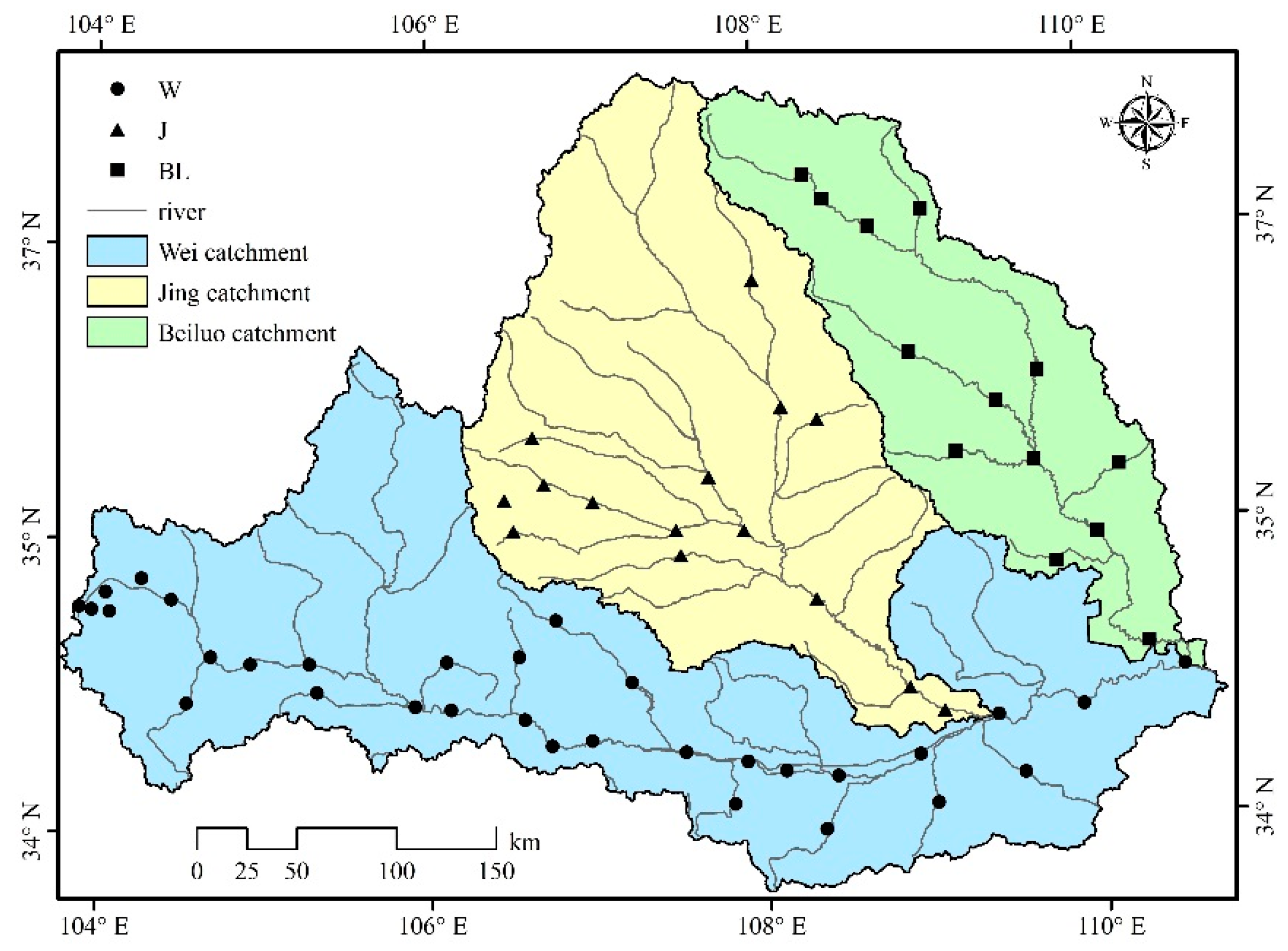
2.2.1. Sampling Sites
2.2.2. Fish Sampling
2.2.3. Macroinvertebrate Sampling
2.2.4. Epilithic Diatom Sampling
2.2.5. Biodiversity Indices
2.2.6. Physiochemical Variable
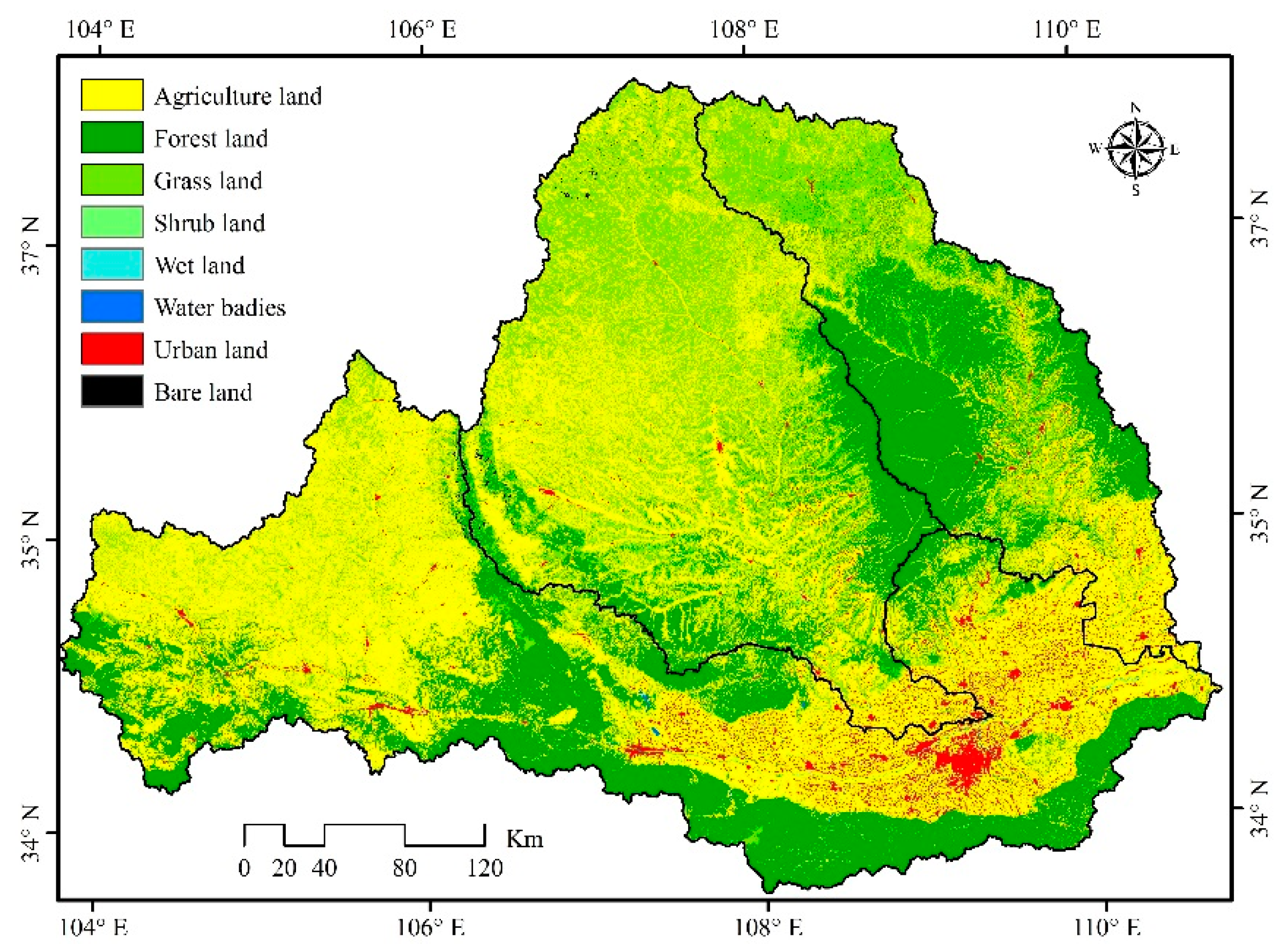
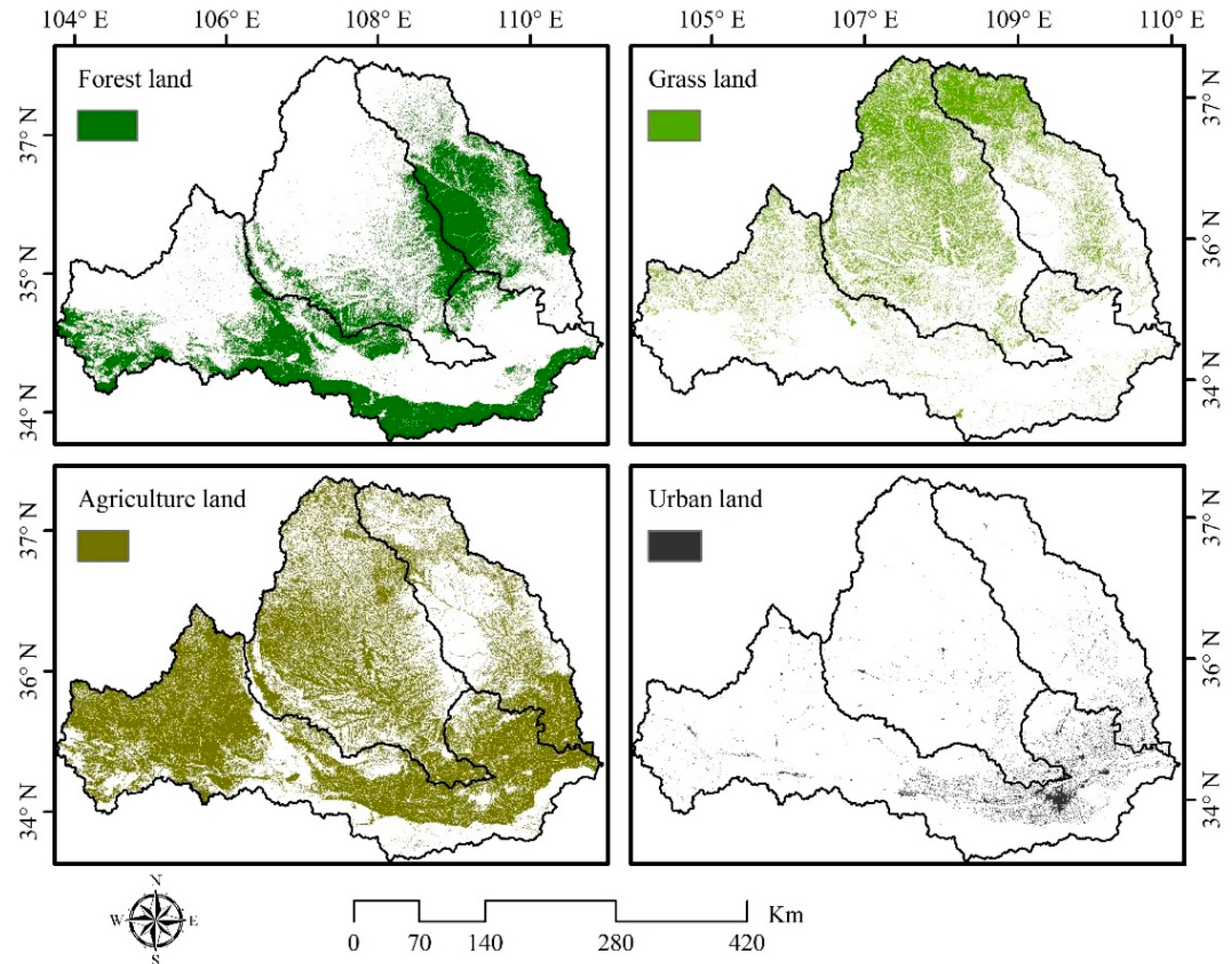
2.2.7. Land Use Type
2.3. Data Analysis
3. Results
3.1. Land Use Characteristics
3.2. Physiochemical variables
3.3. Community Structure and Biological Indices
3.4. Correlations between Biological Indices and Environmental Variables
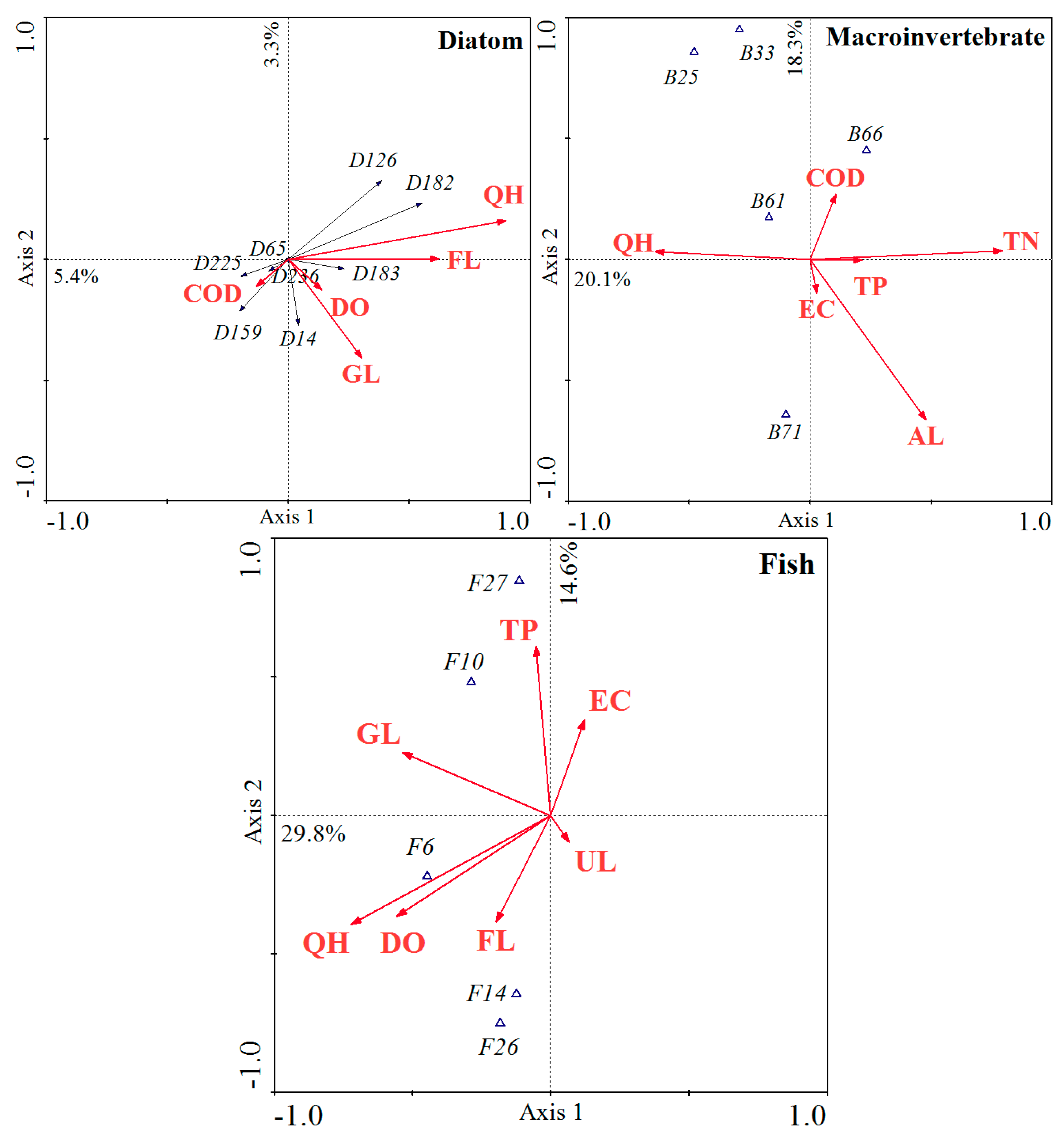
3.5. Relationships between Biological Assemblages and Environmental Variables
4. Discussion
4.1. Characteristic of Aquatic Ecosystems
4.2. Influence of Environmental Variables on Biological Indices
4.3. Response of Biological Assemblages to Environmental Variables
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. The Distribution of Diatom Assemblage in the W, J, and BL Catchments
| ID | Species | W | J | BL |
|---|---|---|---|---|
| D1 | Melosira varians | + | + | |
| D2 | Melosira granulata | + | + | + |
| D3 | Melosira granulata var. angustissima | + | ||
| D4 | Navicula lanceolata | + | + | + |
| D5 | Navicula exigua Krasske | + | + | |
| D6 | Navicula confervacea | + | + | |
| D7 | Navicula cryptocephala | + | + | + |
| D8 | Navicula cryptocephala var. intermedia | + | + | + |
| D9 | Navicula cryptocephala var. venta | + | + | + |
| D10 | Navicula cincta | + | + | + |
| D11 | Navicula cincta var. leptocephala | + | + | + |
| D12 | Navicula cincta var. heufleri | + | ||
| D13 | Navicula pusilla | + | + | |
| D14 | Chamaepinnularia begeri | + | + | + |
| D15 | Navicula pupula | + | + | + |
| D16 | Navicula pupula var. capitata | + | + | + |
| D17 | Navicula cuspidata | + | ||
| D18 | Navicula cuspidata var. heribaudii | + | + | + |
| D19 | Navicula radiosq | + | + | + |
| D20 | Navicula cari | + | + | + |
| D21 | Navicula cari var. angusta | + | ||
| D22 | Navicula salinarum | + | ||
| D23 | Navicula viridula | + | + | + |
| D24 | Navicula viridula var. capitata | + | + | + |
| D25 | Navicula viridula var. pamirensis | + | + | |
| D26 | Navicula simplex | + | + | + |
| D27 | Navicula gothlandica | + | + | + |
| D28 | Navicula accommoda | + | + | + |
| D29 | Navicula rhynchocephala | + | + | |
| D30 | Navicula virihensis | + | + | + |
| D31 | Navicula menisculus | + | + | + |
| D32 | Naviclua anglica | + | ||
| D33 | Navicula exigua Ehr | + | ||
| D34 | Navicula gracilis | + | + | + |
| D35 | Navicula gracilis var. neglecta | + | + | |
| D36 | Navicula rostellata | + | + | |
| D37 | Navicula seminulum | + | ||
| D38 | Navicula seminuloides | + | + | + |
| D39 | Navicula | + | + | + |
| D40 | Navicula muralis | + | + | + |
| D41 | Navicula notha | + | + | + |
| D42 | Navicula halophilioides | + | + | + |
| D43 | Navicula scabellum | + | ||
| D44 | Navicula omissa | + | ||
| D45 | Navicula perrostrata | + | ||
| D46 | Navicula disjuncta | + | ||
| D47 | Navicula disjuncta f. anglica | + | ||
| D48 | Navicula minuscula | + | + | |
| D49 | Navicula placentula | + | ||
| D50 | Navicula asellus | + | ||
| D51 | Navicula dicephala | + | ||
| D52 | Navicula lenzii | + | ||
| D53 | Navicula rotaenea | + | ||
| D54 | Navicula virihensis | + | + | |
| D55 | Navicula hasta Pantocsek | + | ||
| D56 | Navicula protracta | + | ||
| D57 | Navicula protracta var. elliptica | + | ||
| D58 | Navicula adversa | + | ||
| D59 | Navicula tuscula | + | + | + |
| D60 | Navicula atomus | + | + | |
| D61 | Navicula permitis | + | + | + |
| D62 | Navicula nivaloides | + | ||
| D63 | Navicula virihensis | + | ||
| D64 | Navicula virihensis | + | ||
| D65 | Pinnularia appendiculata var. budensis | + | + | + |
| D66 | Pinnularia molaris | + | + | |
| D67 | Pinnularia bogotensis | + | ||
| D68 | Hantzschia amphioxys | + | + | |
| D69 | Hantzschia amphioxys var. aequalis | + | ||
| D70 | Meridion circulare | + | + | |
| D71 | Nitzschia obtusa var. scalpelliformis | + | ||
| D72 | Nitzschia palea | + | + | + |
| D73 | Nitzschia acicularis | + | + | + |
| D74 | Nitzschia hantzschiana | + | ||
| D75 | Nitzschia frustulum | + | + | |
| D76 | Nitzschia frustulum var. perpusilla | + | ||
| D77 | Nitzschia frustulum var. perminuta | + | ||
| D78 | Nitzschia frustulum var. subsalina | + | + | + |
| D79 | Nitzschia recta | + | + | + |
| D80 | Nitzschia dissipata | + | + | + |
| D81 | Nitischia constricta | + | + | + |
| D82 | Nitzschia hungarica | + | + | |
| D83 | Nitzschia sigmoides | + | + | |
| D84 | Nitzschia linearis | + | + | + |
| D85 | Nitzschia microcephala | + | + | + |
| D86 | Nitzschia heuflerana | + | ||
| D87 | Nitzschia stagnorum | + | + | |
| D88 | Nitzschia fonticola | + | + | + |
| D89 | Nitzschia ovalis | + | + | |
| D90 | Nitzschia paleacea | + | ||
| D91 | Nitzschia | + | + | + |
| D92 | Nitzschia thermalis | + | ||
| D93 | Nitzschia tryblionella var. victorise | + | + | + |
| D94 | Nitzschia tryblionella var. levidensis | + | ||
| D95 | Nitzschia commutata | + | ||
| D96 | Nitzschia acula | + | + | |
| D97 | Nitzschia debilis | + | + | |
| D98 | Nitzschia actinastroides | + | ||
| D99 | Nitzschia communis var. abbreviata | + | + | |
| D100 | Nitzschia heidenii | + | + | |
| D101 | Nitzschia angustata var. acuta | + | ||
| D102 | Nitzschia kuetzingiana | + | ||
| D103 | Nitzschia sinuata var. tabellaria | + | + | + |
| D104 | Nitzschia gracilis | + | + | + |
| D105 | Nitzschia romana | + | ||
| D106 | Nitzschia amphbia | + | + | |
| D107 | Nitzschia clausi | + | ||
| D108 | Nitzschia sublinearis | + | ||
| D109 | Stauroneis anceps | + | ||
| D110 | Stauroneis anceps var. linearis | + | ||
| D111 | Stauroneis schroederi | + | + | |
| D112 | Stauroneis dubitabilis | + | ||
| D113 | Stauroneis kriegeri | + | ||
| D114 | Stauroneis palustris | + | ||
| D115 | Rhoicosphenia curvata | + | + | |
| D116 | Amphora ovalis | + | + | + |
| D117 | Amphora ovalis var. gracilis | + | + | + |
| D118 | Amphora perpusilla | + | + | |
| D119 | Cymbella ehrenbergii | + | + | + |
| D120 | Cymbella sinnata | + | + | + |
| D121 | Cymbella microcephala | + | + | + |
| D122 | Cymbella pusilla | + | + | + |
| D123 | Cymbella cistula | + | + | + |
| D124 | Cymbella cistula var. maculata | + | + | |
| D125 | Cymbella cistula var. caldostagnensis | + | ||
| D126 | Encyonema ventricosum | + | + | + |
| D127 | Cymbella ventricosa var. simicircularis | + | + | |
| D128 | Cymbella amphicephala | + | + | + |
| D129 | Cymbella amphicephala var. intermedia | + | ||
| D130 | Cymbella aequalis | + | + | + |
| D131 | Cymbella tumidula | + | + | |
| D132 | Cymbella turgida | + | + | + |
| D133 | Cymbella turgidula | + | ||
| D134 | Cymbella aequalis | + | ||
| D135 | Cymbella aequalis var. pisciculus | + | + | + |
| D136 | Cymbella prostrata | + | + | + |
| D137 | Cymbella gaeumanni | + | + | |
| D138 | Cymbella sphaerophora | + | + | + |
| D139 | Cymbella cymbiformis | + | + | + |
| D140 | Cymbella perpusilla | + | + | |
| D141 | Cymbella bremii | + | + | |
| D142 | Cymbella lata | + | + | |
| D143 | Cymbella gracilis | + | ||
| D144 | Cymbella lunata | + | ||
| D145 | Cymbella excisa | + | ||
| D146 | Cymbella alpina var. minuta | + | ||
| D147 | Cymbella lapponica | + | ||
| D148 | Cymbella aspera | + | ||
| D149 | Cymbella hustedtii | + | + | |
| D150 | Cymbella jolmolungnensis | + | ||
| D151 | Cymbella hauckii | + | ||
| D152 | Cymbella parva | + | ||
| D153 | Cymbella hybrida | + | + | + |
| D154 | Cymbella helvatica | + | ||
| D155 | Diatoma vulgare | + | + | + |
| D156 | Diatoma vulgare var. lineare | + | + | + |
| D157 | Diatoma vulgare var. producta | + | + | + |
| D158 | Diatoma anceps | + | ||
| D159 | Diatoma elongata | + | ||
| D160 | Diatoma elongata var. tenuis | + | + | + |
| D161 | Diatoma hiemale | + | ||
| D162 | Gomphonema parvulum | + | + | + |
| D163 | Gomphonema parvulum var. subellipticum | + | + | + |
| D164 | Gomphonema parvulum var. exilissima | + | + | |
| D165 | Gomphonema angustatum | + | + | + |
| D166 | Gomphonema angustatum var. aequalis | + | ||
| D167 | Gomphonema intricatum | + | + | + |
| D168 | Gomphonema intricatum var. dichotomiformis | + | + | + |
| D169 | Gomphonema olivaceum | + | + | + |
| D170 | Gomphonema olivaceum var. minutissima | + | ||
| D171 | Gomphonema turris | + | ||
| D172 | Gomphonema gracile | + | + | + |
| D173 | Gomphonema gracile var. intricatiformis | + | + | |
| D174 | Gomphonema constrictum | + | + | + |
| D175 | Gomphonema montanum | + | + | |
| D176 | Gomphonema sphaerophorum | + | + | + |
| D177 | Gomphonema | + | ||
| D178 | Gomphonema tergestium | + | ||
| D179 | Achnanthes lanceolata | + | ||
| D180 | Achnanthes lanceolata f. ventricosa | + | + | + |
| D181 | Achnanthes linearis | + | + | |
| D182 | Achnanthidium minutissimum | + | + | + |
| D183 | Achnanthidium minutissimum var. cryptocephala | + | + | + |
| D184 | Achnanthes amphicephala | + | + | |
| D185 | Achnanthes hauckiana | + | + | + |
| D186 | Achnanthes tibetica | + | ||
| D187 | Achnanthes affinis | + | + | + |
| D188 | Achnanthes delicatula | + | + | |
| D189 | Achnanthes montana | + | + | + |
| D190 | Achnanthes crassa | + | + | + |
| D191 | Achnanthes exilis | + | ||
| D192 | Achnanthes subhudsonis | + | ||
| D193 | Achnanthes conspicua | + | ||
| D194 | Achnanthes clevei | + | ||
| D195 | Achnanthes nodosa | + | ||
| D196 | Achnanthes microcephala | + | + | |
| D197 | Cocconeis pediculus | + | + | + |
| D198 | Cocconeis placentula | + | + | + |
| D199 | Fragilaria capucina | + | + | |
| D200 | Fragilaria capucina var. mesolepta | + | ||
| D201 | Fragilaria var. subsalina | + | + | + |
| D202 | Fragilaria ungeriana | + | ||
| D203 | Fragilaria intermedia | + | ||
| D204 | Fragilaria vaucheriae var. capitellata | + | ||
| D205 | Fragilaria virescens var. mesolepta | + | ||
| D206 | Synedra acus | + | + | |
| D207 | Synedra acus var. radians | + | ||
| D208 | Synedra ulna | + | + | + |
| D209 | Synedra ulna var. danica | + | + | + |
| D210 | Synedra ulna var. contracta | + | + | + |
| D211 | Synedra ulna var. oxyrhnchus | + | + | |
| D212 | Synedra amphicephala | + | ||
| D213 | Surirella subsalsa | + | ||
| D214 | Surirella tibetica | + | + | + |
| D215 | Surirella ovalis | + | ||
| D216 | Surirella ovalis var. salina | + | + | + |
| D217 | S.brebissonii | + | + | + |
| D218 | Surirella | + | ||
| D219 | Surirella robusta | + | ||
| D220 | Surirella capronii | + | ||
| D221 | Surirella angusta | + | + | |
| D222 | Cyclotella stelligera | + | + | + |
| D223 | Cyclotella meneghiniana | + | + | + |
| D224 | Cyclotella kuetzingiana | + | + | + |
| D225 | Pantocsekiella ocellata | + | + | + |
| D226 | Cyclotella catenata | + | + | + |
| D227 | Cyclotella asterocostata | + | ||
| D228 | Gyrosigma scalproides | + | + | + |
| D229 | Gyrosigma acuminatum | + | + | |
| D230 | Gyrosigma attenuatum | + | ||
| D231 | Gyrosigma kuetzingii | + | + | + |
| D232 | Cymatopleuta solea | + | + | |
| D233 | Diploneis elliptica | + | ||
| D234 | Diploneis ovalis | + | + | |
| D235 | Diploneis pseudovalis | + | ||
| D236 | Neidium kozlowi var. elliptica | + | ||
| D237 | Neidium iridis var. ampliatum | + | ||
| D238 | Stephanodiscus minutulus | + | ||
| D239 | Ceratoneis arcus | + | + | + |
| D240 | Ceratoneis arcus var. linearis | + | + | + |
| D241 | Ceratoneis arcus var. linearis f.recta | + | ||
| D242 | Ceratoneis arcus var. amphioxys | + | ||
| D243 | Caloneis alpestris var. lanceolata | + | ||
| D244 | Caloneis bacilaria | + | ||
| D245 | Caloneis amphisbaena | + | ||
| D246 | Amphiraphia xizangensis | + | ||
| D247 | Rhopalodia gibba | + | ||
| D248 | Amphipleura pellucida | + | + | |
| D249 | Didymosphenia geminata | + | ||
| D250 | Denticula elegans | + | ||
| D251 | Frustulia vulgaris | + |
Appendix B. The Distribution of Macroinvertebrate Assemblage in the W, J, and BL Catchments
| ID | Species | W | J | BL |
|---|---|---|---|---|
| B1 | Baetidae Analetridae | + | + | + |
| B2 | Baetis vaillanti | + | + | + |
| B3 | Serratellasp. | + | + | + |
| B4 | Leptophlebiasp. | + | + | + |
| B5 | Cinygmasp. | + | + | |
| B6 | Polymitarcyidae | + | ||
| B7 | Epeorsu curvispinosa | + | ||
| B8 | Chromarcyssp. | + | ||
| B9 | Ephemera nigroptera | + | ||
| B10 | Osobenussp. | + | + | |
| B11 | Hydropsychesp. | + | + | + |
| B12 | Brunnea larva | |||
| B13 | Dolophilodes sp. | + | ||
| B14 | Austrotinodessp. | + | ||
| B15 | Tipulasp. | + | ||
| B16 | Antochasp. | + | ||
| B17 | Tabanussp. | + | + | |
| B18 | Natarsia punctata | |||
| B19 | Ablabesmyia phatta | + | ||
| B20 | Procladius choreus | + | + | + |
| B21 | Conchapelopia sp. | + | + | + |
| B22 | Polypedilum scalaenum | + | ||
| B23 | Procladius paradouxus | + | ||
| B24 | Orthocaladius mixtus | + | ||
| B25 | Orthocladius makabensis Sasa | + | + | |
| B26 | Cricotopus albiforceps | + | ||
| B27 | Cricotopus trifasciatus | + | + | + |
| B28 | Cricotopus triannulatus | + | + | + |
| B29 | Cricotopus bicinctus | + | + | |
| B30 | Paracricotopus sp. | + | + | |
| B31 | Cricotopus anulator Goetghebuer | + | ||
| B32 | Diplocladius Kieffer | + | ||
| B33 | Rheocricotopus fuscipes | + | + | |
| B34 | Rheocricotopus effuses | + | ||
| B35 | Paratrichocladius rufivertris | + | ||
| B36 | Rheotanytarsus sp. | + | ||
| B37 | Chironomus riparius Meigen | + | + | + |
| B38 | Chironomus salinarius Kiffer | + | + | |
| B39 | Thienmanniola sp. | + | + | + |
| B40 | Chironomus sp. | + | + | + |
| B41 | Polypedilum paraviceps Niitsuma | + | + | |
| B42 | Micropesectra atrofasciata | + | + | |
| B43 | Cyphomella cornea | + | ||
| B44 | Antocha bifida Alexander | + | ||
| B45 | Sympotthastia takatensis | + | ||
| B46 | Lappodiamesa sp. | + | + | |
| B47 | Simuliumsp. | + | + | + |
| B48 | Sciomyzidae sp. | + | + | |
| B49 | Psychodasp. | + | + | + |
| B50 | Liodessussp. | + | ||
| B51 | Hydrous sp. | + | ||
| B52 | Stenelmis sp adult | + | + | + |
| B53 | Gomphussp. | + | + | |
| B54 | Gomphidae sp. | + | + | |
| B55 | Aeschna sp. | + | ||
| B56 | Pontamalota sp. | + | ||
| B57 | Epitheca.marginata | |||
| B58 | Radix clessini | + | + | |
| B59 | Radix ovata | + | + | + |
| B60 | physa acuta cf. | + | + | |
| B61 | Polypylis hemisphaerula | + | + | |
| B62 | Bellamya aeruginosa | + | ||
| B63 | Schistodesmus lampreyanus | + | + | |
| B64 | Limnodrilus hoffmeisteri | + | + | + |
| B65 | Branchiura sowerbyi | + | ||
| B66 | Limnodrilus claparedianus | + | + | |
| B67 | Tubifex sinicus | + | + | |
| B68 | Whitmania pigra | + | ||
| B69 | Barbronia weberi | + | ||
| B70 | Gammarussp. | + | + | |
| B71 | Sinopotamidae.sp | + | + | |
| B72 | Exopalaemon modestus | + | + | |
| B73 | Macrobrachium nipponense de Haan | + |
Appendix C. The Distribution of Fish Assemblage in the W, J, and BL Catchments
| ID | Species | W | J | BL |
|---|---|---|---|---|
| F1 | Protosalanx hyalocranius | + | ||
| F2 | Paracobitis variegates | + | ||
| F3 | Triplophysa dalaica | + | + | + |
| F4 | Triplophysa sellaefer | + | + | + |
| F5 | Triplophysa shaanxiensis | + | + | + |
| F6 | Triphysa stoliczkae | + | + | + |
| F7 | Triplopphysa bleekeri | + | + | + |
| F8 | Triplophysa robusta | + | + | + |
| F9 | Triplophysa stoliczkae dorsonotata | + | + | + |
| F10 | Triplophysa kungessana orientalis | + | + | |
| F11 | Triplophysa pappenheimi | + | + | + |
| F12 | Triplophysa sp. | + | ||
| F13 | Botia superciliaris | + | ||
| F14 | Cobitis granoei | + | ||
| F15 | Misgurnus anguillicaudatus | + | + | + |
| F16 | Paramisgurnus dabryyanus | + | + | |
| F17 | Opasariichthys bidens | + | + | + |
| F18 | Brachymystax lenok | + | ||
| F19 | Phoxinus lagowskii | + | + | + |
| F20 | Rhodeus sinensis | + | + | |
| F21 | Rhoaeus lighti | + | + | + |
| F22 | Hemiculter leucisculus | + | + | |
| F23 | Belligobio nummifer | + | + | |
| F24 | Oryzias latipes | + | + | |
| F25 | Pseudorasbora parva | + | + | + |
| F26 | Gnathopogon imberbis | + | + | |
| F27 | Huigobio chinssuensis | + | ||
| F28 | Gobio rivuloides | + | ||
| F29 | Abbottina rivularis | + | + | + |
| F30 | Sarcocheilichthys nigripinnis | + | ||
| F31 | Huigobio chinssuensis | + | + | + |
| F32 | Scaphesthes macrolepis | + | ||
| F33 | Gymnodiptychus pachycheilus weiheensis | + | ||
| F34 | Schizopygopsis pylzovi | + | ||
| F35 | Cyprinus carpio | + | ||
| F36 | Carassius auratus | + | + | + |
| F37 | Silurus asotus | + | + | + |
| F38 | Pelteobagrus nitidus | + | ||
| F39 | Hypseleotris swinhonis | + | + | |
| F40 | Ctenogobius cliffordpopei | + | + | + |
| F41 | Ctenogobius brunneus | + | + | + |
| F42 | Ctenogobius gymnauchen | + | + | |
| F43 | Ctenogobius shennongensis | + | + | |
| F44 | Ctenogobius giurinus | + | + | |
| F45 | Channa argus | + |
References
- Cooper, S.D.; Lake, P.S.; Sabater, S.; Melack, J.M.; Sabo, J.L. The effects of land use changes on streams and rivers in mediterranean climates. Hydrobiologia 2013, 719, 383–425. [Google Scholar] [CrossRef]
- Kratzer, B.E.; Jackson, J.K.; Arscott, D.B.; Aufdenkampe, A.K.; Dow, C.L. Macroinvertebrate distribution in relation to land use and water chemistry in New York City drinking-water-supply watersheds. J. N. Am. Benthol. Soc. 2009, 25, 954–976. [Google Scholar] [CrossRef]
- Spnseller, R.A.; Benfield, E.F.; Valett, H.M. Relationships between land use, spatial scale and stream macroinvertebrate communities. Freshw. Biol. 2001, 46, 1409–1424. [Google Scholar] [CrossRef] [Green Version]
- Vázquez, G.; Aké-Castillo, J.A.; Favila, M.E. Algal assemblages and their relationship with water quality in tropical Mexican streams with different land uses. Hydrobiologia 2011, 667, 173–189. [Google Scholar] [CrossRef]
- Dahm, V.; Hering, D.; Nemitz, D.; Graf, W.; Schmidt-Kloiber, A.; Leitner, P.; Melcher, A.; Feld, C.K. Effects of physico-chemistry, land use and hydromorphology on three riverine organism groups: A comparative analysis with monitoring data from Germany and Austria. Hydrobiologia 2013, 704, 389–415. [Google Scholar] [CrossRef]
- Miserendino, M.L.; Casaux, R.; Archangelsky, M.; di Prinzio, C.Y.; Brand, C.; Kutschker, A.M. Assessing land-use effects on water quality, in-stream habitat, riparian ecosystems and biodiversity in Patagonian northwest streams. Sci. Total Environ. 2011, 409, 612–624. [Google Scholar] [CrossRef]
- Villeneuve, B.; Souchon, Y.; Usseglio-Polatera, P.; Ferréol, M.; Valette, L. Can we predict biological condition of stream ecosystems? A multi-stressors approach linking three biological indices to physico-chemistry, hydromorphology and land use. Ecol. Indic. 2015, 48, 88–98. [Google Scholar] [CrossRef]
- Feld, C.K. Response of three lotic assemblages to riparian and catchment-scale land use: Implications for designing catchment monitoring programmes. Freshw. Biol. 2013, 58, 715–729. [Google Scholar] [CrossRef]
- Nerbonne, B.A.; Vondracek, B. Effects of local land use on physical habitat, benthic macroinvertebrates, and fish in the Whitewater River, Minnesota, USA. Environ. Manag. 2001, 28, 87–99. [Google Scholar] [CrossRef]
- Theodoropoulos, C.; Aspridis, D.; Iliopoulou-Georgudaki, J. The influence of land use on freshwater macroinvertebrates in a regulated and temporary Mediterranean river network. Hydrobiologia 2015, 751, 201–213. [Google Scholar] [CrossRef]
- Booth, D.B.; Karr, J.R.; Schauman, S.; Konrad, C.P.; Morley, S.A.; Larson, M.G.; Burges, S.J. Reviving urban streams: Land use, hydrology, biology, and human behavior. J. Am. Water Resour. Assoc. 2004, 40, 1351–1364. [Google Scholar] [CrossRef]
- Ding, J.; Jiang, Y.; Liu, Q.; Hou, Z.; Liao, J.; Fu, L.; Peng, Q. Influences of the land use pattern on water quality in low-order streams of the Dongjiang River basin, China: A multi-scale analysis. Sci. Total Environ. 2016, 551–552, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Castello, L.; Macedo, M.N. Large-scale degradation of Amazonian freshwater ecosystems. Glob. Chang. Biol. 2016, 22, 990–1007. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.T.Y.; Chen, W. Modeling the relationship between land use and surface water quality. J. Environ. Manag. 2002, 66, 377–393. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, E.; Carmona-Catot, G.; Moyle, P.B.; García-Berthou, E. Development and evaluation of a fish-based index to assess biological integrity of Mediterranean streams. Aquat. Conserv. Mar. Freshw. Ecosyst. 2011, 21, 324–337. [Google Scholar] [CrossRef]
- Tan, X.; Ma, P.; Bunn, S.E.; Zhang, Q. Development of a benthic diatom index of biotic integrity (BD-IBI) for ecosystem health assessment of human dominant subtropical rivers, China. J. Environ. Manag. 2015, 151, 286–294. [Google Scholar] [CrossRef]
- Van Ael, E.; de Cooman, W.; Blust, R.; Bervoets, L. Use of a macroinvertebrate based biotic index to estimate critical metal concentrations for good ecological water quality. Chemosphere 2015, 119, 138–144. [Google Scholar] [CrossRef]
- Almeida, D.; Alcaraz-Hernández, J.D.; Merciai, R.; Benejam, L.; García-Berthou, E. Relationship of fish indices with sampling effort and land use change in a large Mediterranean river. Sci. Total Environ. 2017, 605–606, 1055–1063. [Google Scholar] [CrossRef]
- Urbanič, G.; Mihaljević, Z.; Petkovska, V.; Pavlin Urbanić, M. Disentangling the effects of multiple stressors on large rivers using benthic invertebrates—A study of Southeastern European large rivers with implications for management. Water 2020, 12, 621. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.L.; Wan, A.; Zhang, X.K. The effect of land use type on the macroinvertebrate assemblage in Wannan mountain area, China. J. Suzhou Univ. 2015, 30, 110–113. [Google Scholar]
- Li, J.L.; Jin, W.; Wang, B.H.; Xiang, Z.L.; Yin, X.W.; Xu, Z.X.; Zhang, Y. Relationship between land use types within riparian zones and community structure of diatom in Taizi River, China. Res. Environ. Sci. 2015, 28, 1662–1669. [Google Scholar]
- Wu, W.; Xu, Z.; Kennard, M.J.; Yin, X.; Zuo, D. Do human disturbance variables influence more on fish community structure and function than natural variables in the Wei River basin, China? Ecol. Indic. 2016, 61, 438–446. [Google Scholar] [CrossRef]
- Yu, S.; Xu, Z.; Wu, W.; Zuo, D. Effect of land use types on stream water quality under seasonal variation and topographic characteristics in the Wei River basin, China. Ecol. Indic. 2016, 60, 202–212. [Google Scholar] [CrossRef]
- Zuo, D.; Xu, Z.; Wu, W.; Zhao, J.; Zhao, F. Identification of Streamflow Response to Climate Change and Human Activities in the Wei River Basin, China. Water Resour. Manag. 2014, 28, 833–851. [Google Scholar] [CrossRef]
- Zuo, D.; Xu, Z.; Peng, D.; Song, J.; Cheng, L.; Wei, S.; Abbaspour, K.C.; Yang, H. Simulating spatiotemporal variability of blue and green water resources availability with uncertainty analysis. Hydrol. Process. 2015, 29, 1942–1955. [Google Scholar] [CrossRef]
- Mangadze, T.; Bere, T.; Mwedzi, T. Choice of biota in stream assessment and monitoring programs in tropical streams: A comparison of diatoms, macroinvertebrates and fish. Ecol. Indic. 2016, 63, 128–143. [Google Scholar] [CrossRef]
- Lin, G.Y. Fauna Sinica: Phylum Mollusca, Class Gastropoda, Subclass Opisthobranchia, Order Cephalaspidea; Science Press: Beijing, China, 1997. [Google Scholar]
- Wang, J.C.; Wang, X.H. Chironomus Larvae in Northern China; China Yan Shi Press: Beijing, China, 2011. [Google Scholar]
- Yang, T. Fauna Sinica: Annelida, Hirudinea; Science Press: Beijing, China, 2002. [Google Scholar]
- Hu, H.J.; Wei, Y.X. The Freshwater Algae of China: Systematics, Taxonomy and Ecology; Science Press: Beijing, China, 2006. [Google Scholar]
- Zhu, H.Z.; Chen, J.Y. China Tibet Diatoms; Science Press: Beijing, China, 2000. [Google Scholar]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; Stribling, J.B. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates and Fish; US Environmental Protection Agency, Office of Water: Washington, DC, USA, 1999.
- Wu, Z.; Wang, X.; Chen, Y.; Cai, Y.; Deng, J. Assessing river water quality using water quality index in Lake Taihu Basin, China. Sci. Total Environ. 2018, 612, 914–922. [Google Scholar] [CrossRef]
- Fonseca, D.G.; Tanaka, M.O. Influence of an exotic grass on benthic macroinvertebrate communities in a tropical rural landscape. Hydrobiologia 2015, 762, 239–251. [Google Scholar] [CrossRef]
- Hlúbiková, D.; Novais, M.H.; Dohet, A.; Hoffmann, L.; Ector, L. Effect of riparian vegetation on diatom assemblages in headwater streams under different land uses. Sci. Total Environ. 2014, 475, 234–247. [Google Scholar] [CrossRef]
- Longyang, Q. Assessing the effects of climate change on water quality of plateau deep-water lake-A study case of Hongfeng Lake. Sci. Total Environ. 2019, 647, 1518–1530. [Google Scholar] [CrossRef]
- Pardo, I.; Delgado, C.; Abraín, R.; Gómez-Rodríguez, C.; García-Roselló, E.; García, L.; Reynoldson, T.B. Apredictive diatom-based model to assess the ecological status of streams and rivers of Northern Spain. Ecol. Indic. 2018, 90, 519–528. [Google Scholar] [CrossRef]
- Petersen, C.R.; Jovanovic, N.Z.; Grenfell, M.C.; Oberholster, P.J.; Cheng, P. Responses of aquatic communities to physical and chemical parameters in agriculturally impacted coastal river systems. Hydrobiologia 2018, 813, 157–175. [Google Scholar] [CrossRef]
- Shen, R.; Ren, H.; Yu, P.; You, Q.; Pang, W.; Wang, Q. Benthic Diatoms of the Ying River (Huaihe River Basin, China) and Their Application in Water Trophic Status Assessment. Water 2018, 10, 1013. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Pan, Y.; Cao, Y.; Li, B.; Wang, Q.; Wang, B.; Pang, W.; Zhang, J.; Zhu, Z.; Deng, G. Detecting early signs of environmental degradation in protected areas: An example of Jiuzhaigou Nature Reserve, China. Ecol. Indic. 2018, 91, 287–298. [Google Scholar] [CrossRef]
- Szczepocka, E.; Krawczyk, P.N.; Kruk, A. Deceptive ecological status of urban streams and rivers—evidence from diatom indices. Ecosphere 2018, 9, e02310. [Google Scholar] [CrossRef]
- Zhang, Y.; Cheng, L.; Tolonen, K.E.; Yin, H.; Gao, J.; Zhang, Z.; Li, K.; Cai, Y. Substrate degradation and nutrient enrichment structuring macroinvertebrate assemblages in agriculturally dominated Lake Chaohu Basins, China. Sci. Total Environ. 2018, 627, 57–66. [Google Scholar] [CrossRef]
- Maceda-Veiga, A.; Nally, R.M.; de Sostoa, A. Water-quality impacts in semi-arid regions: Can natural ‘green filters’ mitigate adverse effects on fish assemblages? Water Res. 2018, 144, 628–641. [Google Scholar] [CrossRef]
- Maceda-Veiga, A.; Nally, R.M.; de Sostoa, A. Environmental correlates of food-chain length, mean trophic level and trophic level variance in invaded riverine fish assemblages. Sci. Total Environ. 2018, 644, 420–429. [Google Scholar] [CrossRef]
- Delgado, C.; Pardo, I. Comparison of benthic diatoms from Mediterranean and Atlantic Spanish streams: Community changes in relation to environmental factors. Aquat. Bot. 2015, 120, 304–314. [Google Scholar] [CrossRef]
- Meador, M.R.; Frey, J.W. Relative Importance of Water-Quality Stressors in Predicting Fish Community Responses in Midwestern Streams. J. Am. Water Resour. Assoc. 2018, 54, 708–723. [Google Scholar] [CrossRef]
- Xu, M.; Wang, Z.; Duan, X.; Pan, B. Effects of pollution on macroinvertebrates and water quality bio-assessment. Hydrobiologia 2014, 729, 247–259. [Google Scholar] [CrossRef]
- Yoshimura, C.; Tockner, K.; Omura, T.; Moog, O. Species diversity and functional assessment of macroinvertebrate communities in Austrian rivers. Limnology 2006, 7, 63–74. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, L.; Cheng, L.; Cai, Y.; Yin, H.; Gao, J.; Gao, Y. Macroinvertebrate assemblages in streams and rivers of a highly developed region (Lake Taihu Basin, China). Aquat. Biol. 2014, 23, 15–28. [Google Scholar] [CrossRef] [Green Version]
- Wu, W.; Xu, Z.X.; Yin, X.W.; Yu, S.Y. Fish community structure and the effect of environmental factors in the Wei River basin. Acta Sci. Circumstantiae 2014, 34, 1298–1308. [Google Scholar]
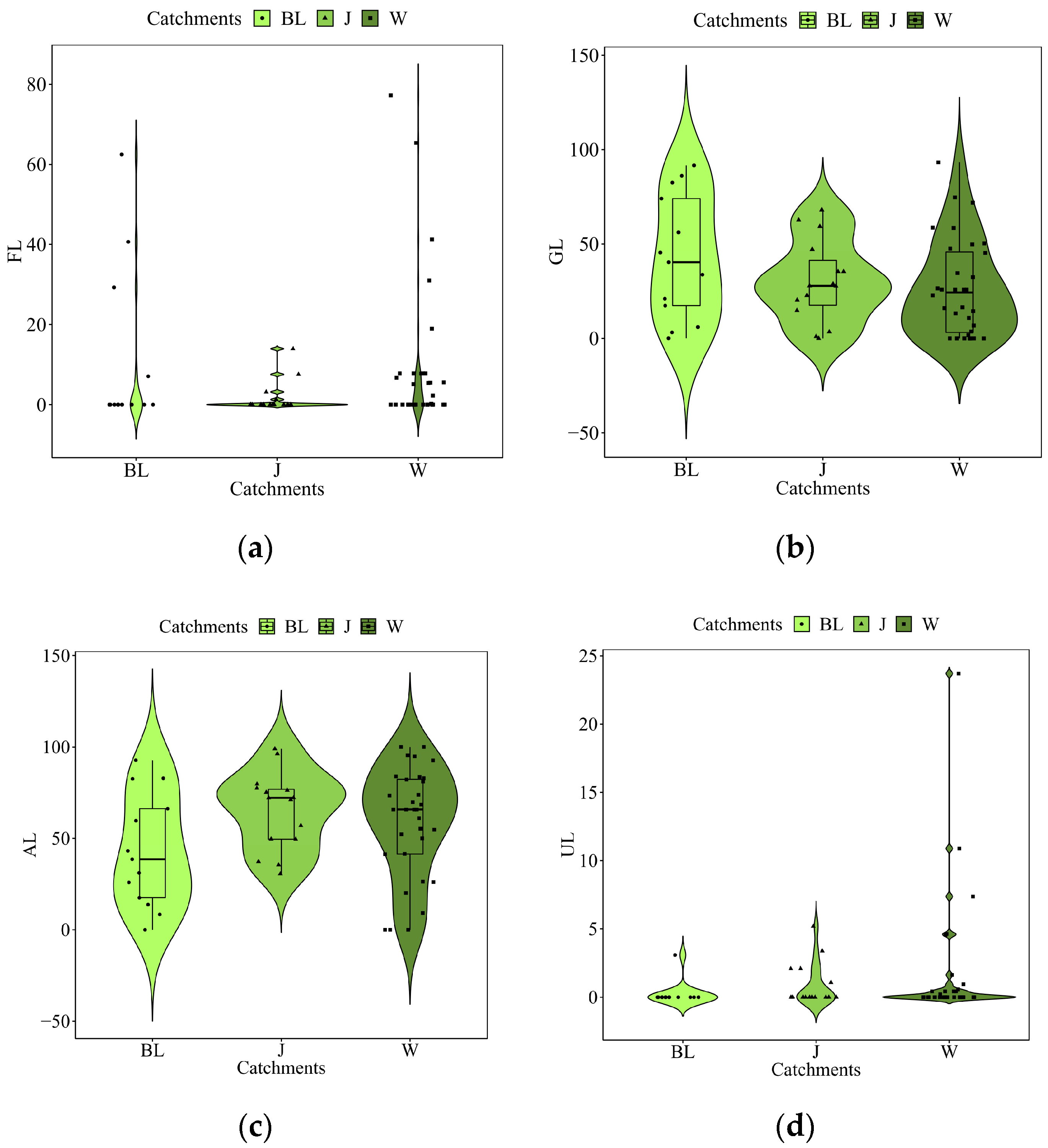
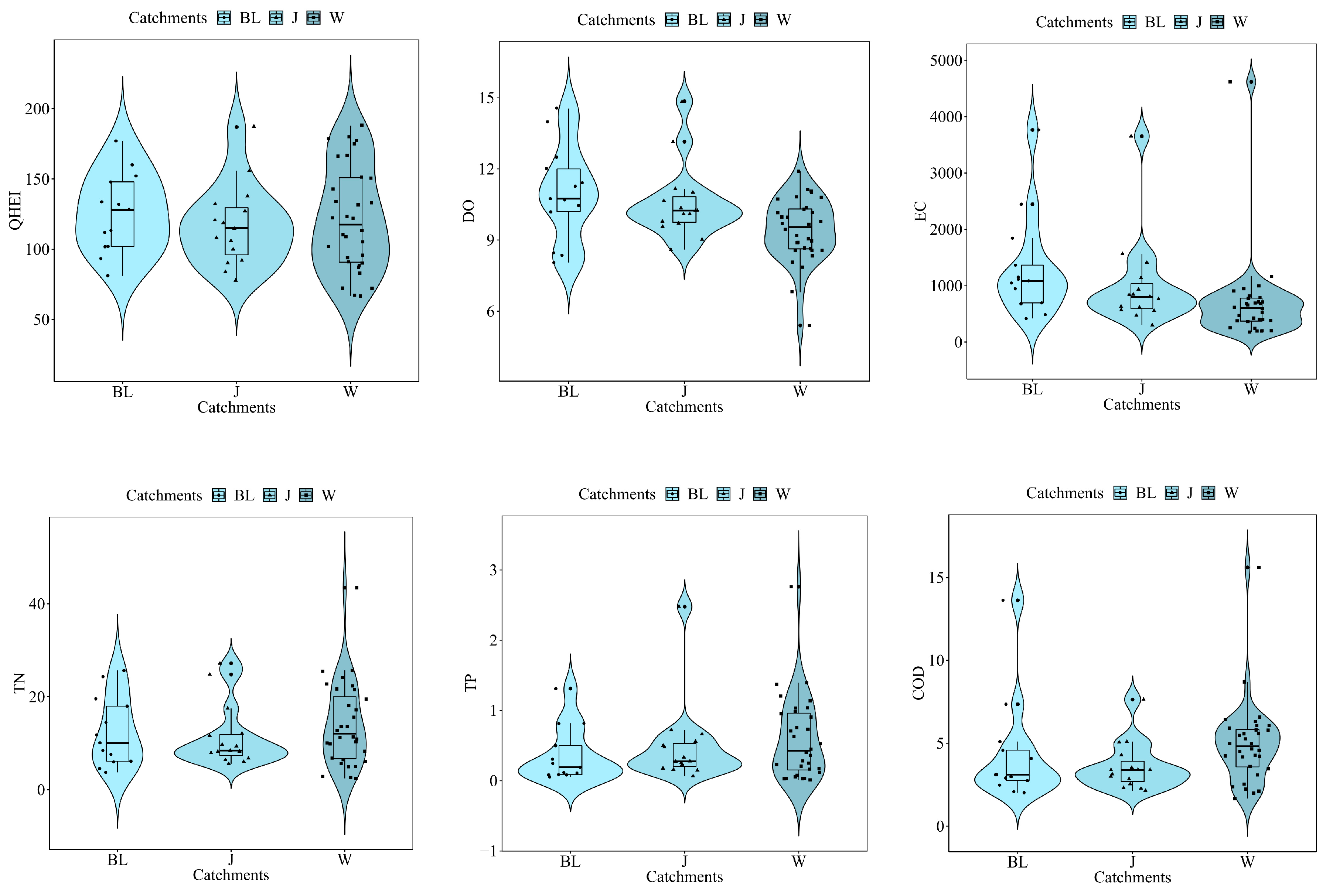
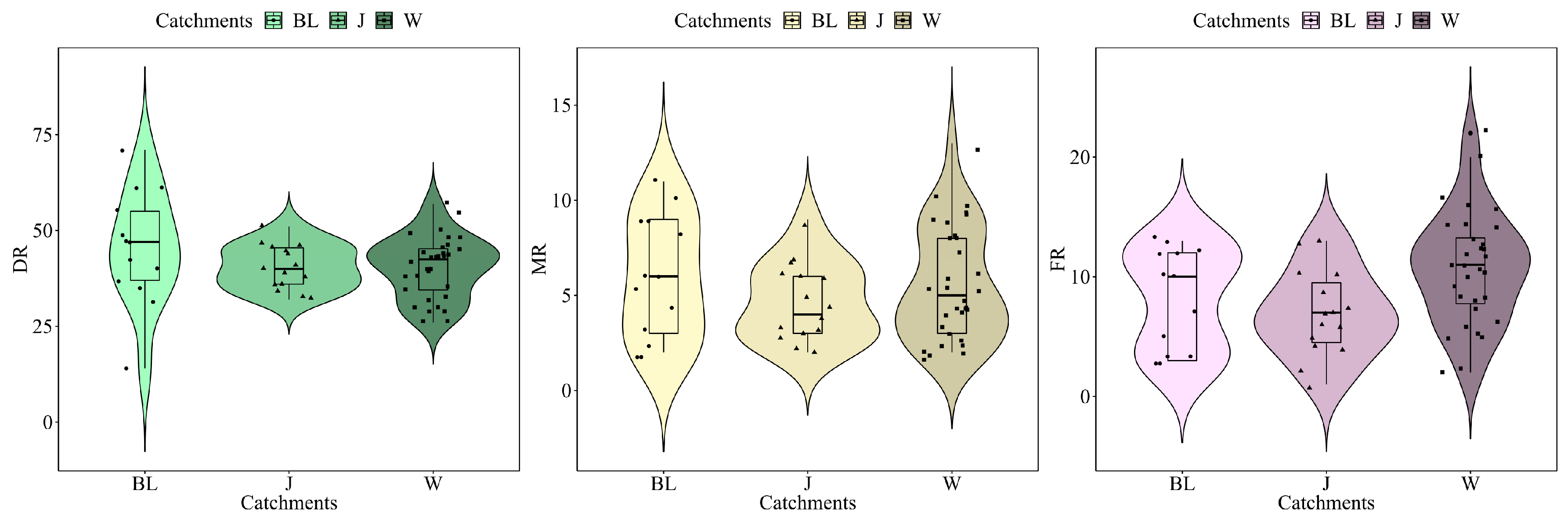
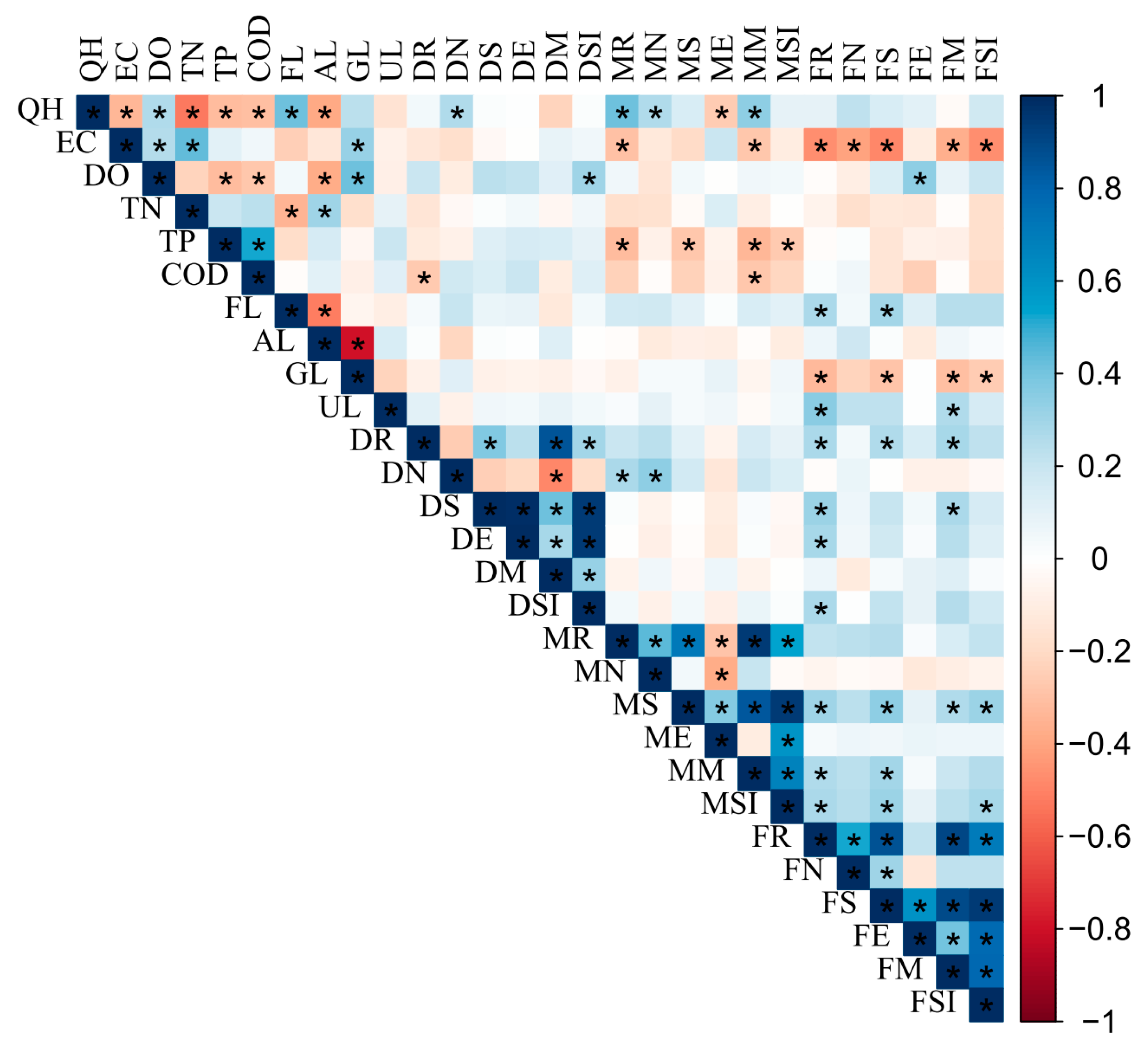
| Catchments | ID | Species | Value | P |
|---|---|---|---|---|
| W Catchment | D126 | Encyonema ventricosum | 41.4 | 0.003 |
| D182 | Achnanthidium minutissimum | 41.1 | 0.004 | |
| F6 | Triplophysa minxianensis | 37.9 | 0.008 | |
| F14 | Cobitis granoei | 28.1 | 0.016 | |
| F27 | Huigobio chinssuensis | 25.0 | 0.019 | |
| F33 | Gobio coriparoides | 24.3 | 0.042 | |
| J Catchment | D225 | Pantocsekiella ocellata | 41.0 | 0.002 |
| F10 | Triplophysa kungessana orientalis | 37.3 | 0.004 | |
| BL Catchment | D159 | Diatoma elongata | 53.8 | 0.001 |
| D183 | Achnanthidium minutissimum var. cryptocephala | 43.6 | 0.001 | |
| D14 | Chamaepinnularia begeri | 42.6 | 0.003 | |
| D65 | Caloneis budensis | 29.2 | 0.005 | |
| D236 | Neidium kozlowi var. elliptica | 30.8 | 0.002 | |
| B25 | Orthocladius makabensis Sasa | 42.8 | 0.001 | |
| B33 | Rheocricotopus fuscipes | 27.5 | 0.005 | |
| B61 | Polypylis hemisphaerula | 17.7 | 0.038 | |
| B66 | Limnodrilus.claparedianus | 31.3 | 0.012 | |
| B71 | Sinopotamidae | 13.3 | 0.042 | |
| F26 | Gnathopogon imberbis | 30.9 | 0.035 |
| Axis | Diatom | Macroinvertebrate | Fish | |||
|---|---|---|---|---|---|---|
| Eigenvalues | Correlations | Eigenvalues | Correlations | Eigenvalues | Correlations | |
| Axis 1 | 0.054 | 0.816 | 0.201 | 0.751 | 0.298 | 0.763 |
| Axis 2 | 0.033 | 0.786 | 0.183 | 0.748 | 0.146 | 0.698 |
| Axis 3 | 0.026 | 0.753 | 0.124 | 0.757 | 0.092 | 0.660 |
| Axis 4 | 0.022 | 0.842 | 0.105 | 0.724 | 0.061 | 0.674 |
| Variable | Diatom | Macroinvertebrate | Fish | |||
|---|---|---|---|---|---|---|
| F | P | F | P | F | P | |
| QH | 2.88 | 0.001 | 1.15 | 0.251 | 3.84 | 0.001 |
| DO | 0.99 | 0.438 | 1.34 | 0.091 | 2.14 | 0.006 |
| EC | 0.93 | 0.596 | 0.91 | 0.596 | 1.03 | 0.405 |
| TN | 1.04 | 0.416 | 1.85 | 0.007 | 1.20 | 0.261 |
| TP | 0.67 | 0.886 | 1.05 | 0.401 | 2.05 | 0.028 |
| COD | 1.23 | 0.168 | 0.93 | 0.550 | 0.62 | 0.870 |
| FL | 1.51 | 0.024 | 1.08 | 0.366 | 1.17 | 0.240 |
| GL | 1.38 | 0.039 | 0.75 | 0.807 | 1.63 | 0.032 |
| AL | 1.20 | 0.172 | 1.51 | 0.044 | 1.10 | 0.326 |
| UL | 0.88 | 0.593 | 1.02 | 0.393 | 0.79 | 0.660 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Xu, Z.; Yang, F.; Yin, X.; Wu, W.; Li, J. Comparison of Fish, Macroinvertebrates and Diatom Communities in Response to Environmental Variation in the Wei River Basin, China. Water 2020, 12, 3422. https://doi.org/10.3390/w12123422
Liu L, Xu Z, Yang F, Yin X, Wu W, Li J. Comparison of Fish, Macroinvertebrates and Diatom Communities in Response to Environmental Variation in the Wei River Basin, China. Water. 2020; 12(12):3422. https://doi.org/10.3390/w12123422
Chicago/Turabian StyleLiu, Linfei, Zongxue Xu, Fan Yang, Xuwang Yin, Wei Wu, and Jisheng Li. 2020. "Comparison of Fish, Macroinvertebrates and Diatom Communities in Response to Environmental Variation in the Wei River Basin, China" Water 12, no. 12: 3422. https://doi.org/10.3390/w12123422
APA StyleLiu, L., Xu, Z., Yang, F., Yin, X., Wu, W., & Li, J. (2020). Comparison of Fish, Macroinvertebrates and Diatom Communities in Response to Environmental Variation in the Wei River Basin, China. Water, 12(12), 3422. https://doi.org/10.3390/w12123422





