Hydrochemical Parameters to Assess the Evolutionary Process of CO2-Rich Spring Water: A Suggestion for Evaluating CO2 Leakage Stages in Silicate Rocks
Abstract
:1. Introduction
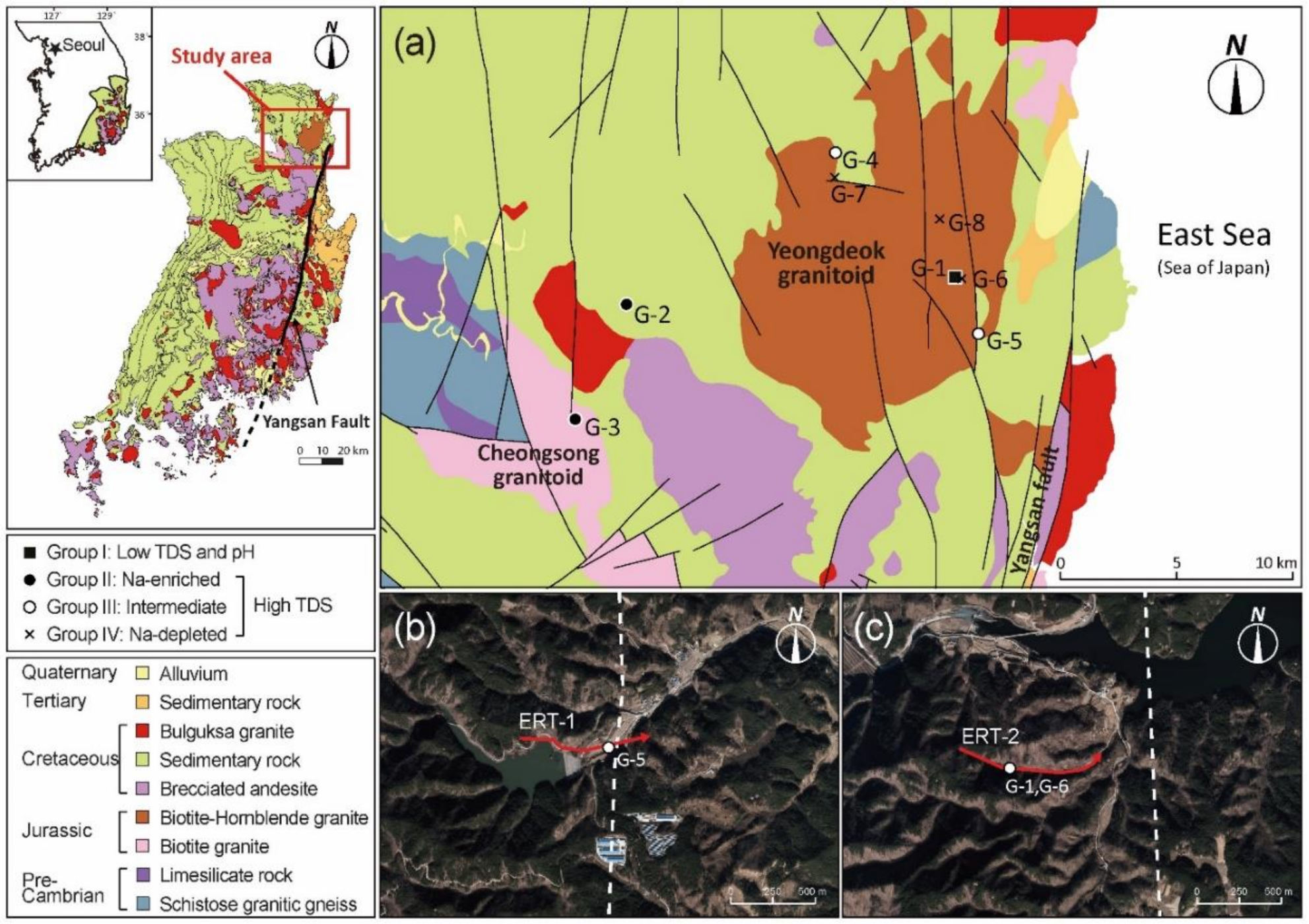
2. Study Area
3. Methods
3.1. Sampling and Analytical Methods
3.2. Geophysical Exploration
4. Results
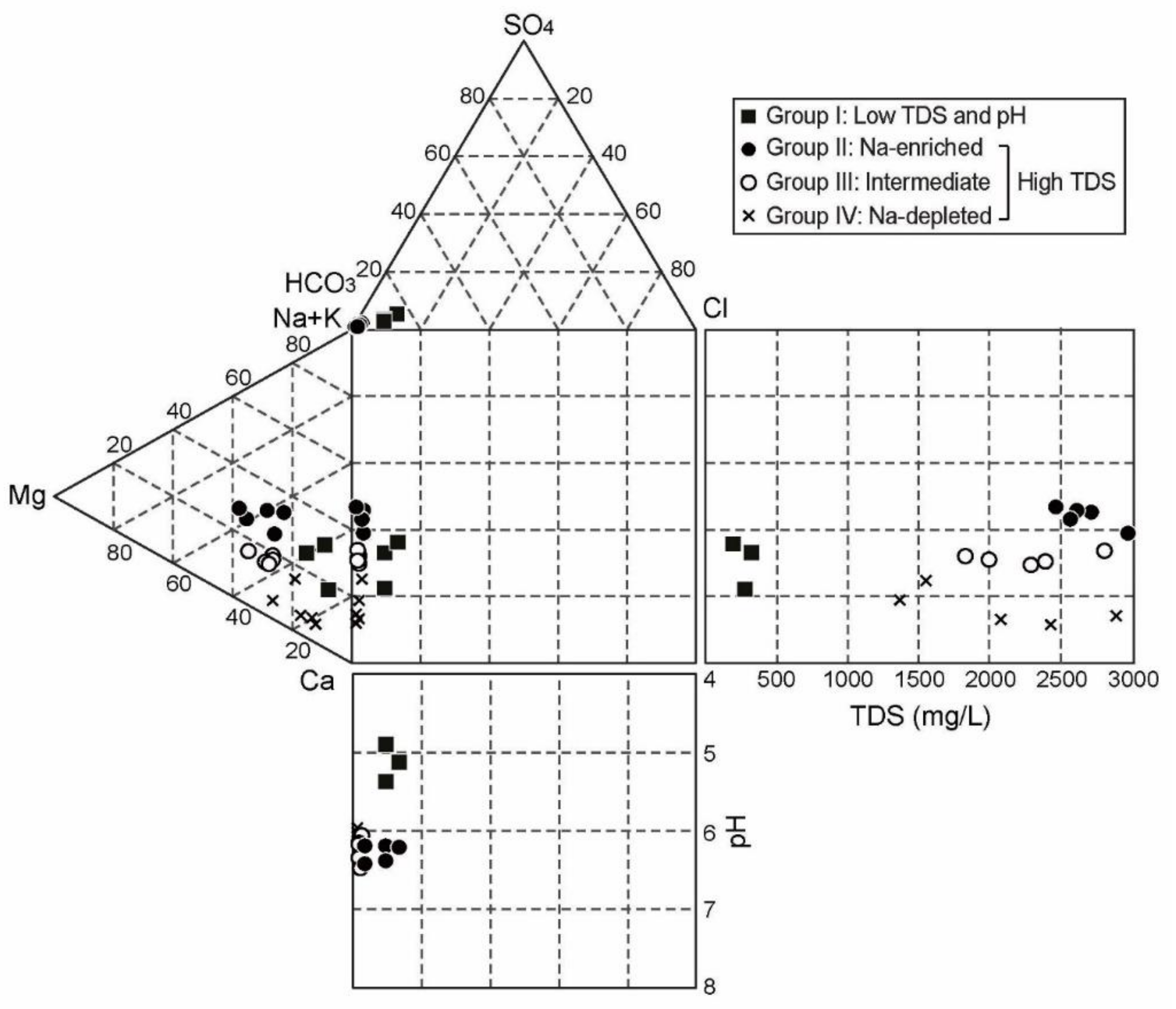
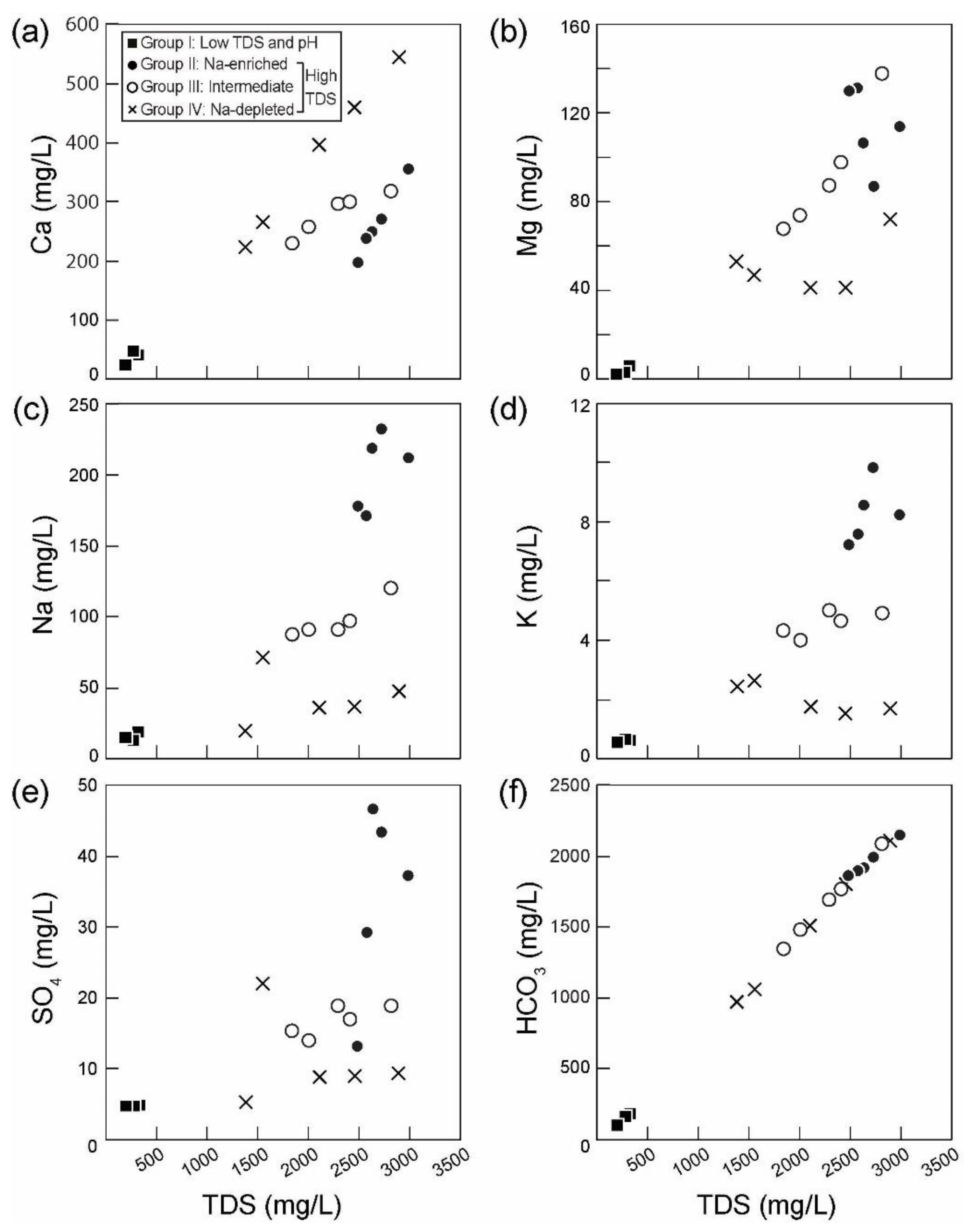
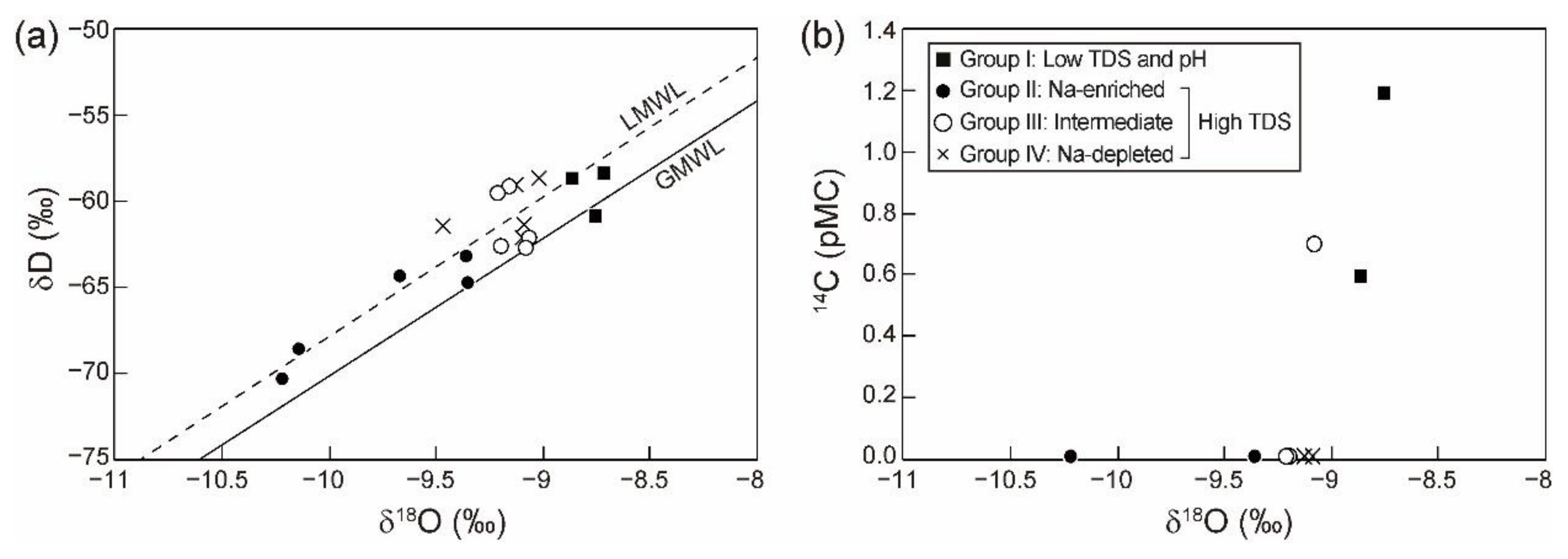
4.1. Hydrochemcial and Isotopic Compositions
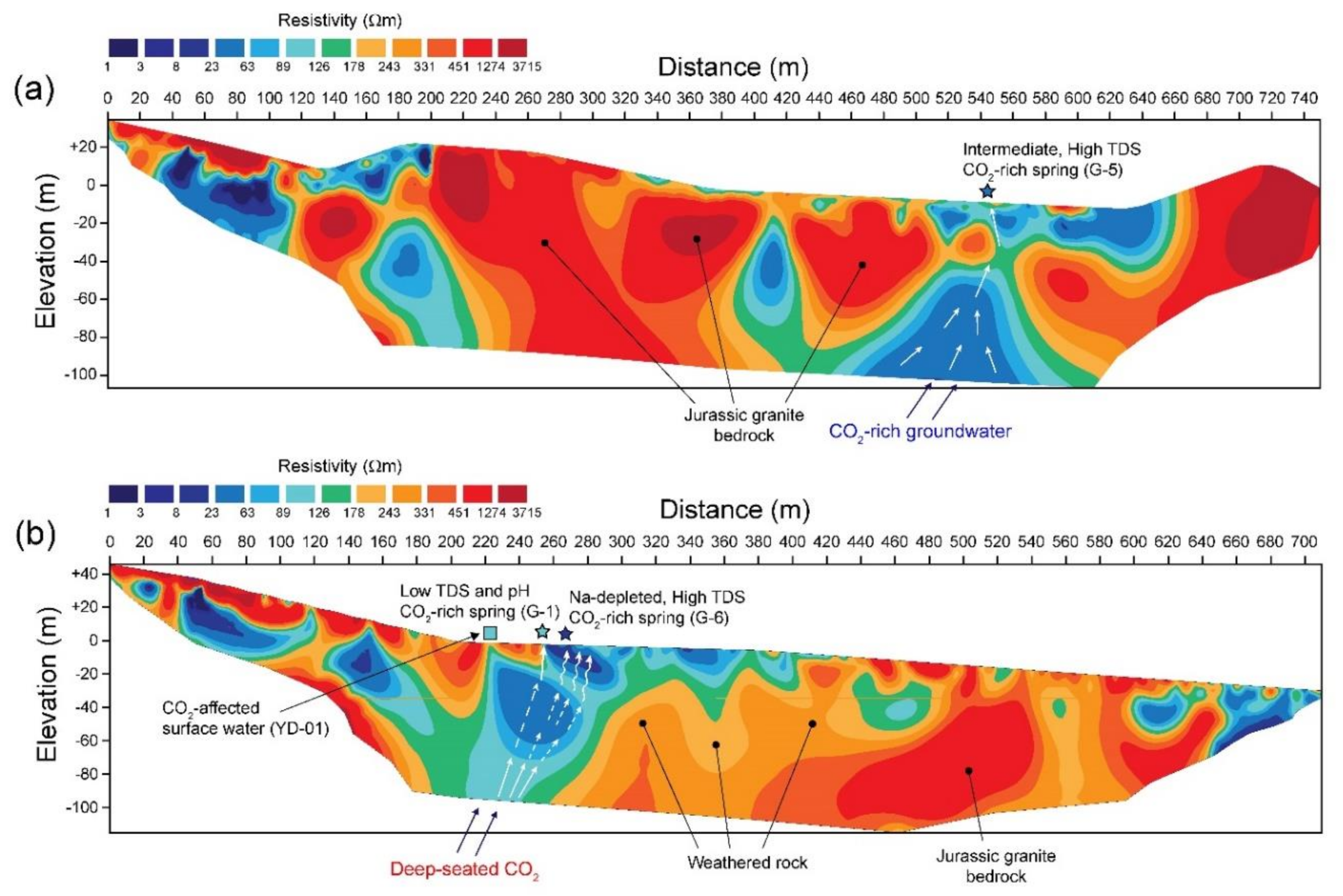
4.2. Geophysical Survey
5. Discussion
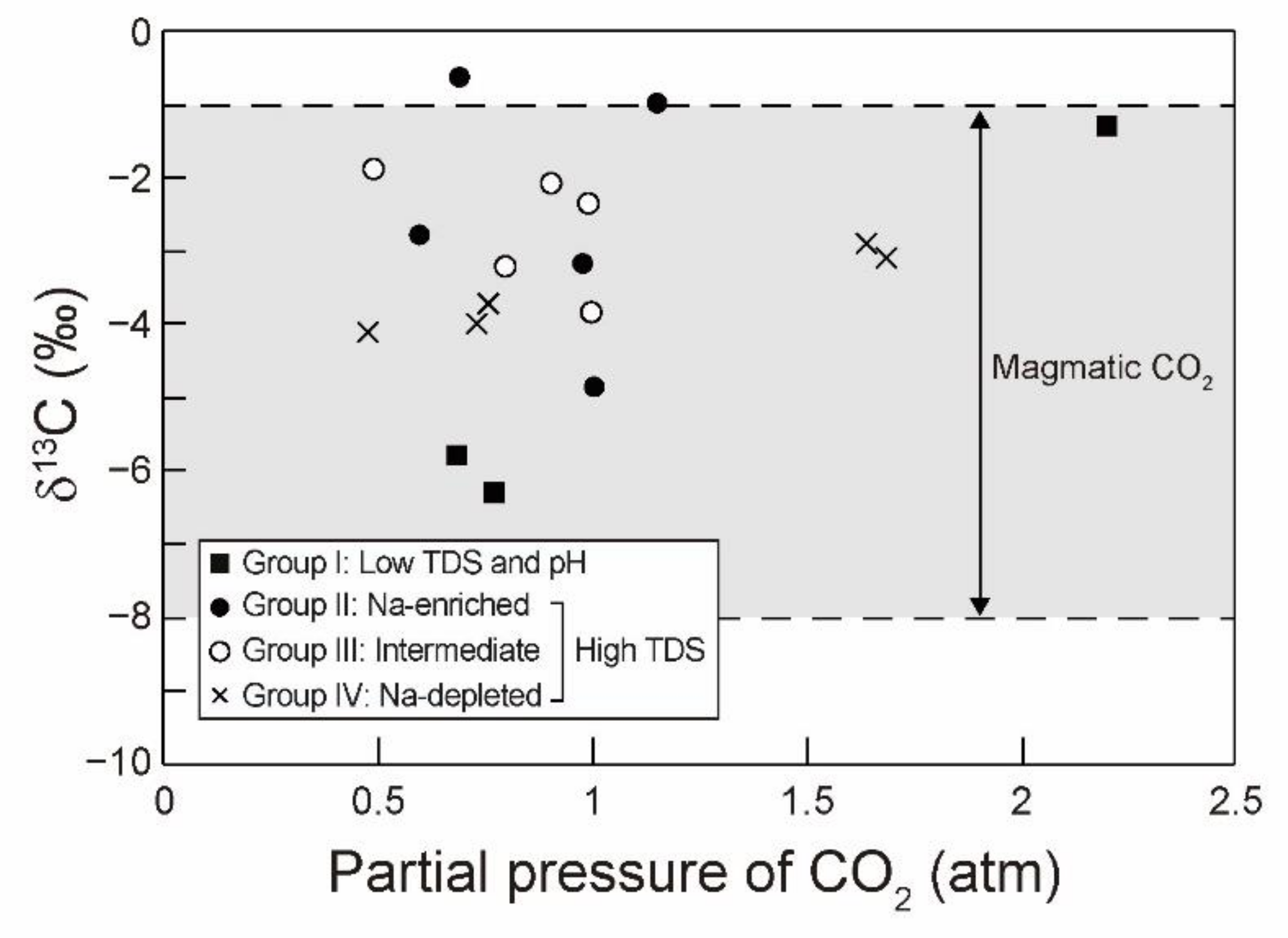
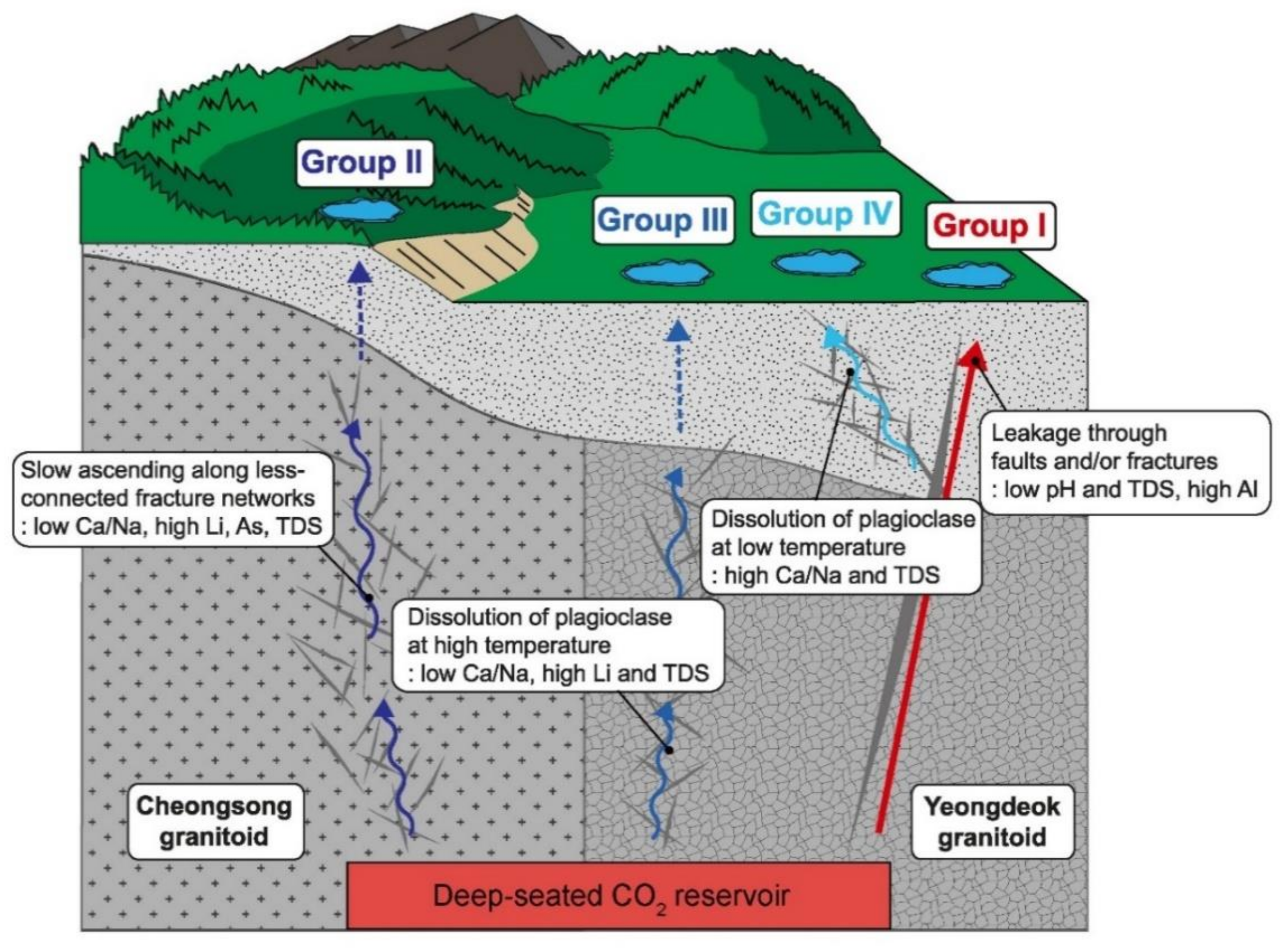
5.1. Source and Discharge Pathways of CO2
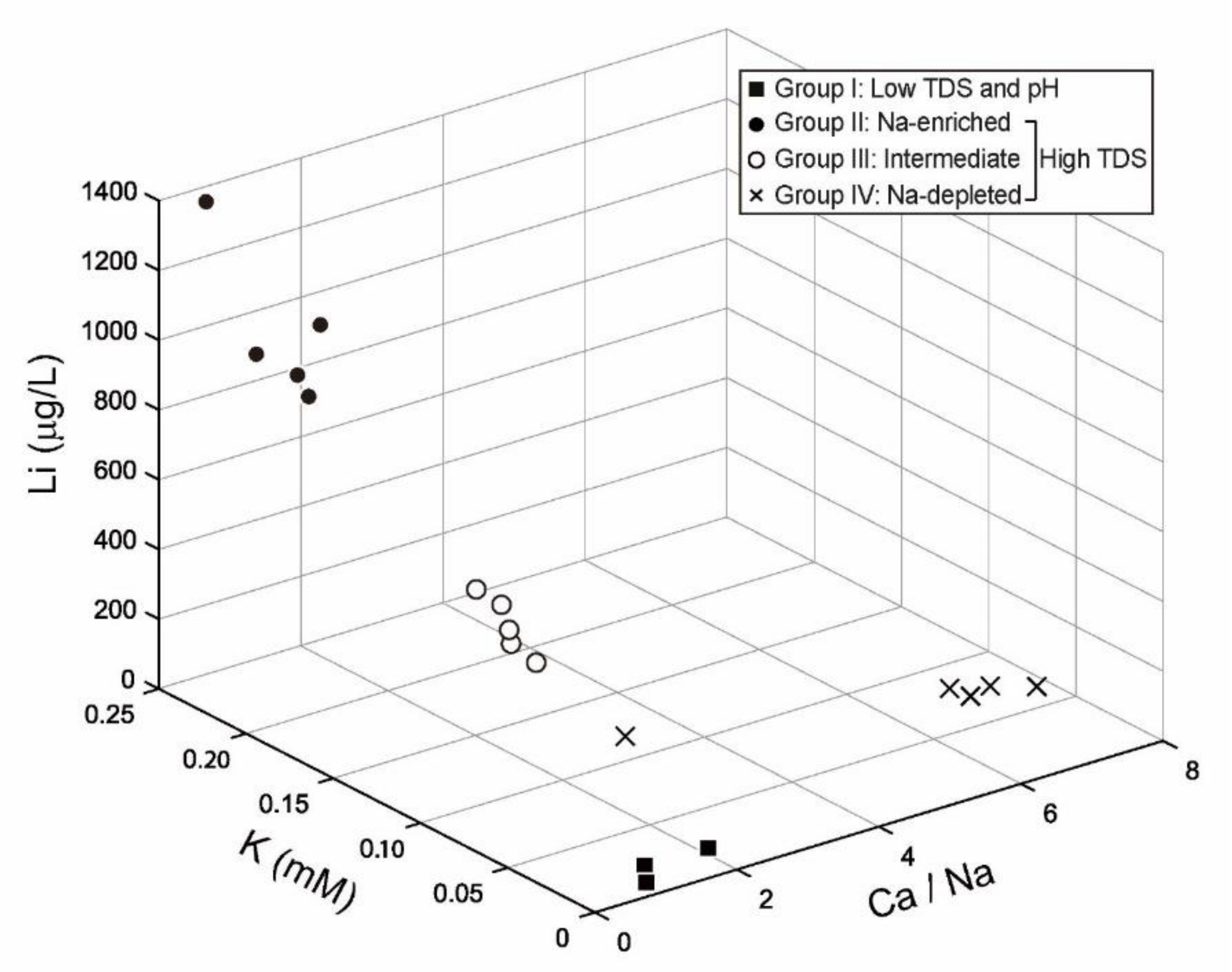
5.2. Hydrochemical Evolutionary Processes
5.2.1. Group I and II
5.2.2. Group III and IV
5.3. Useful Hydrochemical Parameters
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Metz, B.; Davidson, O.; De Coninck, H.C.; Loos, M.; Meyer, L.A. IPCC, 2005: IPCC Special Report on Carbon Dioxide Capture and Storage; Working Group III of the Intergovernmental Panel on Climate Change: Cambridge, UK; New York, NY, USA, 2005; p. 442.
- Oelkers, E.H.; Cole, D.R. Carbon dioxide sequestration a solution to a global problem. Elements 2008, 4, 305–310. [Google Scholar] [CrossRef]
- Benson, S.M.; Cole, D.R. CO2 Sequestration in Deep Sedimentary Formations. Elements 2008, 4, 325–331. [Google Scholar] [CrossRef]
- DOEFE (USDOE Office of Fossil Energy (FE) (United States)). Report of the Interagency Task Force on Carbon Capture and Storage; 2010. Available online: https://www.osti.gov/servlets/purl/985209 (accessed on 17 December 2018).
- Cuéllar-Franca, R.M.; Azapagic, A. Carbon capture, storage and utilisation technologies: A critical analysis and comparison of their life cycle environmental impacts. J. CO2 Util. 2015, 9, 82–102. [Google Scholar] [CrossRef]
- Cai, B.; Li, Q.; Liu, G.; Liu, L.; Jin, T.; Shi, H. Environmental concern-based site screening of carbon dioxide geological storage in China. Sci. Rep. 2017, 7, 7598. [Google Scholar] [CrossRef]
- Bachu, S.; Bennion, D.B. Experimental assessment of brine and/or CO2 leakage through well cements at reservoir conditions. Int. J. Greenh. Gas Control 2009, 3, 494–501. [Google Scholar] [CrossRef]
- Cahill, A.G.; Marker, P.; Jakobsen, R. Hydrogeochemical and mineralogical effects of sustained CO2 contamination in a shallow sandy aquifer: A field-scale controlled release experiment. Water Resour. Res. 2014, 50, 1735–1755. [Google Scholar] [CrossRef]
- Spangler, L.H.; Dobeck, L.M.; Repasky, K.S.; Nehrir, A.R.; Humphries, S.D.; Barr, J.L.; Keith, C.J.; Shaw, J.A.; Rouse, J.H.; Cunningham, A.B.; et al. A shallow subsurface controlled release facility in Bozeman, Montana, USA, for testing near surface CO2 detection techniques and transport models. Environ. Earth Sci. 2010, 60, 227–239. [Google Scholar] [CrossRef]
- Ju, Y.; Gilfillan, S.M.V.; Lee, S.-S.; Kaown, D.; Hahm, D.; Lee, S.; Park, I.-W.; Ha, S.-W.; Park, K.; Do, H.-K.; et al. Application of noble gas tracers to identify the retention mechanisms of CO2 migrated from a deep reservoir into shallow groundwater. Int. J. Greenh. Gas Control 2020, 97, 103041. [Google Scholar] [CrossRef]
- Martens, S.; Liebscher, A.; Möller, F.; Henninges, J.; Kempka, T.; Lüth, S.; Norden, B.; Prevedel, B.; Szizybalski, A.; Zimmer, M.; et al. CO2 storage at the Ketzin Pilot site, Germany: Fourth year of injection, monitoring, modelling and verification. Energy Procedia 2013, 37, 6434–6443. [Google Scholar] [CrossRef] [Green Version]
- Emberley, S.; Hutcheon, I.; Shevalier, M.; Durocher, K.; Mayer, B.; Gunter, W.D.; Perkins, E.H. Monitoring of fluid–rock interaction and CO2 storage through produced fluid sampling at the Weyburn CO2-injection enhanced oil recovery site, Saskatchewan, Canada. Appl. Geochem. 2005, 20, 1131–1157. [Google Scholar] [CrossRef]
- Bickle, M.; Kampman, N.; Wigley, M. Natural Analogues. Rev. Mineral. Geochem. 2013, 77, 15–71. [Google Scholar] [CrossRef] [Green Version]
- Keating, E.H.; Fessenden, J.; Kanjorski, N.; Koning, D.J.; Pawar, R. The impact of CO2 on shallow groundwater chemistry: Observations at a natural analog site and implications for carbon sequestration. Environ. Earth Sci. 2010, 60, 521–536. [Google Scholar] [CrossRef] [Green Version]
- Lewicki, J.L.; Birkholzer, J.; Tsang, C.-F. Natural and industrial analogues for leakage of CO2 from storage reservoirs: Identification of features, events, and processes and lessons learned. Environ. Geol. 2007, 52, 457–467. [Google Scholar] [CrossRef] [Green Version]
- Menció, A.; Guasch, H.; Soler, D.; Canelles, A.; Zamorano, M.; Brusi, D. Influence of regional hydrogeological systems at a local scale: Analyzing the coupled effects of hydrochemistry and biological activity in a Fe and CO2 rich spring. Sci. Total Environ. 2016, 569–570, 700–715. [Google Scholar] [CrossRef] [PubMed]
- Lemieux, J.-M. Review: The potential impact of underground geological storage of carbon dioxide in deep saline aquifers on shallow groundwater resources. Hydrogeol. J. 2011, 19, 757–778. [Google Scholar] [CrossRef]
- Ko, K.; Lee, J.; Chung, H. Highly efficient colorimetric CO2 sensors for monitoring CO2 leakage from carbon capture and storage sites. Sci. Total Environ. 2020, 138786. [Google Scholar] [CrossRef]
- Kim, J.; Yu, S.; Yun, S.-T.; Kim, K.-H.; Kim, J.-H.; Shinn, Y.-J.; Chae, G. CO2 leakage detection in the near-surface above natural CO2-rich water aquifer using soil gas monitoring. Int. J. Greenh. Gas Control 2019, 88, 261–271. [Google Scholar] [CrossRef]
- Schroder, I.F.; Zhang, H.; Zhang, C.; Feitz, A.J. The role of soil flux and soil gas monitoring in the characterisation of a CO2 surface leak: A case study in Qinghai, China. Int. J. Greenh. Gas Control 2016, 54, 84–95. [Google Scholar] [CrossRef] [Green Version]
- Schroder, I.F.; Wilson, P.; Feitz, A.F.; Ennis-King, J. Evaluating the performance of soil flux surveys and inversion methods for quantification of CO2 leakage. Energy Procedia 2017, 114, 3679–3694. [Google Scholar] [CrossRef]
- Schütze, C.; Sauer, U.; Beyer, K.; Lamert, H.; Bräuer, K.; Strauch, G.; Flechsig, C.; Kämpf, H.; Dietrich, P. Natural analogues: A potential approach for developing reliable monitoring methods to understand subsurface CO2 migration processes. Environ. Earth Sci. 2012, 67, 411–423. [Google Scholar] [CrossRef]
- Choi, B.-Y. Potential impact of leaking CO2 gas and CO2-rich fluids on shallow groundwater quality in the Chungcheong region (South Korea): A hydrogeochemical approach. Int. J. Greenh. Gas Control 2019, 84, 13–28. [Google Scholar] [CrossRef]
- Do, H.-K.; Yun, S.-T.; Yu, S.; Ryuh, Y.-G.; Choi, H.-S. Evaluation of long-term impacts of CO2 leakage on groundwater quality using hydrochemical data from a natural analogue site in South Korea. Water 2020, 12, 1457. [Google Scholar] [CrossRef]
- Choi, B.Y.; Yun, S.T.; Mayer, B.; Hong, S.Y.; Kim, K.H.; Jo, H.Y. Hydrogeochemical processes in clastic sedimentary rocks, South Korea: A natural analogue study of the role of dedolomitization in geologic carbon storage. Chem. Geol. 2012, 306–307, 103–113. [Google Scholar] [CrossRef]
- Kim, K.-H.; Yun, S.-T.; Yu, S.; Choi, B.-Y.; Kim, M.-J.; Lee, K.-J. Geochemical pattern recognitions of deep thermal groundwater in South Korea using self-organizing map: Identified pathways of geochemical reaction and mixing. J. Hydrol. 2020, 589, 125202. [Google Scholar] [CrossRef]
- Choi, H.; Woo, N.C. Natural analogue monitoring to estimate the hydrochemical change of groundwater by the carbonating process from the introduction of CO2. J. Hydrol. 2018, 562, 318–334. [Google Scholar] [CrossRef]
- Hutcheon, I.; Abercrombie, H.J.; Krouse, H.R. Inorganic origin of carbon dioxide during low temperature thermal recovery of bitumen: Chemical and isotopic evidence. Geochim. Cosmochim. Acta 1990, 54, 165–171. [Google Scholar] [CrossRef]
- Yoshimura, K.; Liu, Z.; Cao, J.; Yuan, D.; Inokura, Y.; Noto, M. Deep source CO2 in natural waters and its role in extensive tufa deposition in the Huanglong Ravines, Sichuan, China. Chem. Geol. 2004, 205, 141–153. [Google Scholar] [CrossRef]
- Weaver, T.R.; Cartwright, I.; Tweed, S.O.; Ahearne, D.; Cooper, M.; Czapnik, K.; Tranter, J. Controls on chemistry during fracture-hosted flow of cold CO2-bearing mineral waters, Daylesford, Victoria, Australia: Implications for resource protection. Appl. Geochem. 2006, 21, 289–304. [Google Scholar] [CrossRef]
- Marques, J.M.; Andrade, M.; Carreira, P.M.; Eggenkamp, H.G.M.; GraçA, R.C.; Aires-Barros, L.; Antunes Da Silva, M. Chemical and isotopic signatures of Na/HCO3/CO2-rich geofluids, North Portugal. Geofluids 2006, 6, 273–287. [Google Scholar] [CrossRef]
- Rodrigo-Naharro, J.; Aracil, E.; del Villar, L.P. Geophysical investigations in the Gañuelas-Mazarrón Tertiary basin (SE Spain): A natural analogue of a geological CO2 storage affected by anthropogenic leakages. J. Appl. Geophys. 2018, 155, 187–198. [Google Scholar] [CrossRef]
- Sauer, U.; Watanabe, N.; Singh, A.; Dietrich, P.; Kolditz, O.; Schütze, C. Joint interpretation of geoelectrical and soil-gas measurements for monitoring CO2 releases at a natural analogue. Surf. Geophys. 2014, 12, 165–178. [Google Scholar] [CrossRef] [Green Version]
- Griffiths, D.H.; Barker, R.D. Two-dimensional resistivity imaging and modelling in areas of complex geology. J. Appl. Geophys. 1993, 29, 211–226. [Google Scholar] [CrossRef]
- Chough, S.K.; Kwon, S.-T.; Ree, J.-H.; Choi, D.K. Tectonic and sedimentary evolution of the Korean peninsula: A review and new view. Earth Sci. Rev. 2000, 52, 175–235. [Google Scholar] [CrossRef]
- Shin, Y.S.; Lee, Y.T.; Lee, Y.J.; Koh, I.S. Petrochemical characteristics of sandstones and mudrocks from the Lower Hayang Group (Cretaceous), Taegu. Korea J. Geol. Soc. Korea 2001, 37, 153–168. [Google Scholar]
- Cheong, C.S.; Kwon, S.T.; Sagong, H. Geochemical and Sr-Nd-Pb isotopic investigation of Triassic granitoids and basement rocks in the northern Gyeongsang Basin, Korea: Implications for the young basement in the East Asian continental margin. Isl. ARC 2002, 11, 25–44. [Google Scholar] [CrossRef]
- Woo, H.; Jang, Y. Petrological characteristics of the Yeongdeok granite. J. Petrol. Soc. Korea 2014, 23, 31–43. [Google Scholar] [CrossRef] [Green Version]
- Sagong, H.; Kwon, S.T.; Ree, J.H. Mesozoic episodic magmatism in South Korea and its tectonic implication. Tectonics 2005, 24, 1–18. [Google Scholar] [CrossRef]
- Jeong, C.H. Genesis of hydrochemistry of CO2-rich springs from Kyungpook Province, Korea. Econ. Environ. Geol. 2002, 35, 121–136. [Google Scholar]
- Jeong, C.H.; Kim, H.J.; Lee, S.Y. Hydrochemistry and genesis of CO2-rich springs from Mesozoic granitoids and their adjacent rocks in South Korea. Geochem. J. 2005, 39, 517–530. [Google Scholar] [CrossRef] [Green Version]
- Jeong, C.-H.; Yoo, S.-W.; Kim, K.-H.; Nagao, K. Hydrochemistry and origin of noble gases and CO2 gas within carbonated mineral waters in the Kyeoungbuk-Kangwon Province, Korea. J. Eng. Geol. 2011, 21, 65–77. [Google Scholar] [CrossRef] [Green Version]
- Jeong, C.H.; Jeong, G.Y. Geochemical water quality and genesis of carbonated Dalki mineral water in the Chungsong area, Kyungpook, Econ. Environ. Geol. 1999, 32, 455–468. [Google Scholar]
- Parkhurst, B.D.L.; Appelo, C.A.J. User’s Guide to PHREEQC (Version 2)—A Computer Program for Speciation, and Inverse Geochemical Calculations. Water Resour. Investig. Rep. 1999, 99, 312. [Google Scholar]
- Yun, J.A.; Kim, K.H. Geochemistry and stable isotopes of carbonated waters in South Korea. J. Korean Soc. Groundw. Environ. 2000, 7, 116–124. [Google Scholar]
- Jeong, C.-H.; Kim, K.-H.; Nagao, K. Hydrogeochemistry and origin of CO2 and noble gases in the Dalki carbonate waters of the Chungsong area. J. Eng. Geol. 2012, 22, 123–134. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2017; ISBN 978-92-4-154995-0. [Google Scholar]
- Clark, I.D.; Fritz, P. Environmental Isotopes in Hydrogeology; CRC Press: Boca Raton, FL, USA, 2013; ISBN 1-4822-4291-5. [Google Scholar]
- Lee, K.-S.; Kim, Y. Determining the seasonality of groundwater recharge using water isotopes: A case study from the upper North Han River basin, Korea. Environ. Geol. 2007, 52, 853–859. [Google Scholar] [CrossRef]
- Deines, P.; Langmuir, D.; Harmon, R.S. Stable carbon isotope ratios and the existence of a gas phase in the evolution of carbonate ground waters. Geochim. Cosmochim. Acta 1974, 38, 1147–1164. [Google Scholar] [CrossRef]
- Hoefs, J. Stable Isotope Geochemistry; Springer: Berlin/Heidelberg, Germany, 2009; Volume 285. [Google Scholar]
- Kang, Y.-J.; Yun, S.-T.; Yu, S.; Do, H.-K.; Chae, G. Quantitative assessment of deep-seated CO2 leakage around CO2-rich springs with low soil CO2 efflux using end-member mixing analysis and carbon isotopes. J. Environ. Manag. 2020, 276, 111333. [Google Scholar] [CrossRef]
- Sharma, P.V. Environmental and Engineering Geophysics; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Do, H.-K.; Kim, K.-H.; Yun, S.-T. Hydrochemical and isotopic characteristics of CO2-rich groundwater in the Gyeongsang Sedimentary Basin, South Korea: A natural analogue study on the potential leakage of geologically-stored CO2. Energy Procedia 2017, 114, 3805–3811. [Google Scholar] [CrossRef]
- Arts, R.J.; Baradello, L.; Girard, J.F.; Kirby, G.; Lombardi, S.; Williamson, P.; Zaja, A. Results of geophysical monitoring over a “leaking” natural analogue site in Italy. Energy Procedia 2009, 1, 2269–2276. [Google Scholar] [CrossRef]
- Choi, H.S.; Yun, S.T.; Koh, Y.K.; Mayer, B.; Park, S.S.; Hutcheon, I. Geochemical behavior of rare earth elements during the evolution of CO2-rich groundwater: A study from the Kangwon district, South Korea. Chem. Geol. 2009, 262, 334–343. [Google Scholar] [CrossRef]
- Chae, G.; Yu, S.; Jo, M.; Choi, B.-Y.; Kim, T.; Koh, D.-C.; Yun, Y.-Y.; Yun, S.-T.; Kim, J.-C. Monitoring of CO2-rich waters with low pH and low EC: An analogue study of CO2 leakage into shallow aquifers. Environ. Earth Sci. 2016, 75, 1–15. [Google Scholar] [CrossRef]
- Kim, K.; Jeong, D.H.; Kim, Y.; Koh, Y.; Kim, S.; Park, E. The geochemical evolution of very dilute CO2-rich water in Chungcheong Province, Korea: Processes and pathways. Geofluids 2008, 8, 3–15. [Google Scholar] [CrossRef]
- Kim, K.; Jeong, G.Y. Factors influencing natural occurrence of fluoride-rich groundwaters: A case study in the southeastern part of the Korean Peninsula. Chemosphere 2005, 58, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Chae, G.-T.; Yun, S.-T.; Kim, K.; Mayer, B. Hydrogeochemistry of sodium-bicarbonate type bedrock groundwater in the Pocheon spa area, South Korea: Water–rock interaction and hydrologic mixing. J. Hydrol. 2006, 321, 326–343. [Google Scholar] [CrossRef]
- Koh, Y.K.; Choi, B.Y.; Yun, S.T.; Choi, H.S.; Mayer, B.; Ryoo, S.W. Origin and evolution of two contrasting thermal groundwaters (CO2-rich and alkaline) in the Jungwon area, South Korea: Hydrochemical and isotopic evidence. J. Volcanol. Geotherm. Res. 2008, 178, 777–786. [Google Scholar] [CrossRef]
- Faure, G. Principles and Applications of Inorganic Geochemistry: A Comprehensive Textbook for Geology Students; Maxwell Macmillan International: New York, NY, USA, 1991; ISBN 0-02-336441-6. [Google Scholar]
- White, W.M. Geochemistry; John Wiley & Sons: Hoboken, NJ, USA, 2013; ISBN 1-118-48527-0. [Google Scholar]
- Shankar, S.; Shanker, U. Shikha arsenic contamination of groundwater: A review of sources, prevalence, health risks, and strategies for mitigation. Sci. World J. 2014, 2014, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Gascoyne, M.; Kamineni, D. The hydrogeochemistry of fractured plutonic rocks in the Canadian Shield. Appl. Hydrogeol. 1994, 2, 43–49. [Google Scholar] [CrossRef]
- Stober, I.; Bucher, K. Deep groundwater in the crystalline basement of the Black Forest region. Appl. Geochem. 1999, 14, 237–254. [Google Scholar] [CrossRef]
- Aiuppa, A.; Allard, P.; D’Alessandro, W.; Michel, A.; Parello, F.; Treuil, M.; Valenza, M. Mobility and fluxes of major, minor and trace metals during basalt weathering and groundwater transport at Mt. Etna volcano (Sicily). Geochim. Cosmochim. Acta 2000, 64, 1827–1841. [Google Scholar] [CrossRef]
- Bernhard, G.; Geipel, G.; Reich, T.; Brendler, V.; Amayri, S.; Nitsche, H. Uranyl(VI) carbonate complex formation: Validation of the Ca2UO2(CO3)3(aq.) species. Radiochim. Acta 2001, 89, 511–518. [Google Scholar] [CrossRef]
- Dong, W.; Brooks, S.C. Determination of the formation constants of ternary complexes of uranyl and carbonate with alkaline earth metals (Mg2+, Ca2+, Sr2+, and Ba2+) using anion exchange method. Environ. Sci. Technol. 2006, 40, 4689–4695. [Google Scholar] [CrossRef] [PubMed]
- Shin, W.; Oh, J.; Choung, S.; Cho, B.-W.; Lee, K.-S.; Yun, U.; Woo, N.-C.; Kim, H.K. Distribution and potential health risk of groundwater uranium in Korea. Chemosphere 2016, 163, 108–115. [Google Scholar] [CrossRef] [PubMed]
| Group | Sample ID | Sampling Date | Temp. (°C) | pH | Eh (mV) | EC (µS/cm) | DO (mg/L) | PCO2 * (atm) | TDS (mg/L) | Concentration (mg/L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca | Mg | Na | K | SiO2 | F | Cl | NO3 | SO4 | HCO3 | ||||||||||
| I | G-1 | February-2013 | 10.8 | 4.92 | 517.6 | 214 | 2.3 | 2.2 | 328.9 | 40.19 | 6.15 | 19.16 | 0.63 | 63.14 | 0.30 | 9.54 | 1.64 | 4.89 | 182.08 |
| February-2014 | 8.0 | 5.38 | 378.5 | 217 | 3.6 | 0.7 | 280.1 | 47.43 | 2.95 | 13.07 | 0.67 | 38.52 | 0.36 | 8.31 | 1.43 | 4.76 | 161.70 | ||
| March-2015 | 11.4 | 5.13 | 424.9 | 222 | 3.2 | 0.8 | 200.3 | 25.36 | 2.33 | 14.88 | 0.57 | 41.04 | 0.35 | 7.63 | 1.38 | 4.71 | 100.68 | ||
| Average | 10.1 | 5.14 | 440.3 | 217.6 | 3.0 | 1.2 | 269.8 | 37.7 | 3.8 | 15.7 | 0.6 | 47.6 | 0.3 | 8.5 | 1.5 | 4.8 | 148.2 | ||
| II | G-2 | February-2013 | 14.3 | 6.20 | 490.9 | 2100 | 1.0 | 1.1 | 2983.8 | 355.82 | 114.19 | 212.51 | 8.26 | 72.15 | 1.37 | 31.76 | 0.34 | 37.19 | 2144.43 |
| February-2014 | 8.7 | 6.40 | 299.2 | 2725 | 2.2 | 0.7 | 2727.5 | 271.70 | 87.24 | 232.93 | 9.87 | 57.58 | 1.27 | 28.30 | 0.19 | 43.35 | 1989.17 | ||
| March-2015 | - | 6.23 | 296.0 | 2588 | 1.6 | 1.0 | 2632.5 | 249.70 | 106.55 | 219.33 | 8.58 | 47.56 | 0.76 | 25.53 | 0.17 | 46.59 | 1912.95 | ||
| G-3 | February-2014 | 9.5 | 6.44 | 297.1 | 2816 | 1.6 | 0.6 | 2575.9 | 238.25 | 131.65 | 171.46 | 7.60 | 69.19 | 1.46 | 23.75 | 1.64 | 29.20 | 1897.72 | |
| March-2015 | - | 6.21 | 285.5 | 2858 | 0.9 | 1.0 | 2485.2 | 196.83 | 130.16 | 178.17 | 7.25 | 73.16 | 0.41 | 9.15 | 0.96 | 13.14 | 1861.10 | ||
| Average | 10.8 | 6.30 | 333.7 | 2617.4 | 1.4 | 0.9 | 2681.0 | 262.5 | 114.0 | 202.9 | 8.3 | 63.9 | 1.1 | 23.7 | 0.7 | 33.9 | 1961.1 | ||
| III | G-4 | February-2014 | 12.2 | 6.08 | 300.8 | 1979 | 1.2 | 1.0 | 1838.1 | 230.14 | 67.76 | 87.73 | 4.36 | 63.84 | 0.49 | 13.92 | 0.09 | 15.40 | 1348.52 |
| March-2015 | - | 6.16 | 340.4 | 1984 | 0.8 | 0.9 | 2008.5 | 257.27 | 74.07 | 91.06 | 4.01 | 66.50 | 0.01 | 9.31 | 0.03 | 14.02 | 1479.67 | ||
| G-5 | February-2013 | 13.6 | 6.35 | 277.7 | 3240 | 1.2 | 0.8 | 2817.5 | 318.55 | 137.89 | 120.24 | 4.93 | 110.20 | 0.80 | 16.33 | 0.73 | 18.85 | 2085.38 | |
| February-2014 | 11.4 | 6.48 | 262.8 | 3190 | 3.0 | 0.5 | 2295.5 | 297.74 | 87.60 | 91.36 | 5.03 | 80.06 | 1.10 | 16.89 | 0.71 | 18.82 | 1693.41 | ||
| March-2015 | 14.1 | 6.19 | 259.9 | 2980 | 1.8 | 1.0 | 2409.3 | 301.15 | 97.82 | 97.02 | 4.66 | 92.11 | 0.52 | 12.37 | 0.27 | 16.93 | 1769.67 | ||
| Average | 12.8 | 6.25 | 288.3 | 2674.6 | 1.6 | 0.8 | 2273.8 | 281.0 | 93.0 | 97.5 | 4.6 | 82.5 | 0.6 | 13.8 | 0.4 | 16.8 | 1675.3 | ||
| IV | G-6 | February-2013 | 11.6 | 6.03 | 489.0 | 2046 | 1.3 | 1.6 | 2893.9 | 544.48 | 72.28 | 47.62 | 1.71 | 94.37 | 1.14 | 12.83 | 0.05 | 9.36 | 2103.73 |
| February-2014 | 11.0 | 6.24 | 321.4 | 2125 | 2.4 | 0.7 | 2108.3 | 396.64 | 41.39 | 36.18 | 1.76 | 69.16 | 1.33 | 13.71 | 0.37 | 8.88 | 1510.17 | ||
| March-2015 | 12.5 | 5.96 | 258.8 | 2007 | 0.8 | 1.7 | 2458.0 | 460.05 | 41.31 | 36.67 | 1.55 | 71.61 | 0.61 | 9.18 | 0.05 | 8.91 | 1800.01 | ||
| G-7 | February-2014 | 8.2 | 6.08 | 361.7 | 1500 | 2.6 | 0.7 | 1381.1 | 223.73 | 53.12 | 19.84 | 2.47 | 79.45 | 0.23 | 8.96 | 0.50 | 5.31 | 970.17 | |
| G-8 | February-2013 | 5.0 | 6.30 | 349.9 | 908 | 1.1 | 0.5 | 1554.3 | 266.16 | 46.91 | 71.42 | 2.64 | 52.75 | 0.42 | 11.47 | 0.57 | 21.99 | 1057.12 | |
| Average | 9.7 | 6.12 | 356.2 | 1717.2 | 1.6 | 1.1 | 2079.1 | 378.2 | 51.0 | 42.3 | 2.0 | 73.5 | 0.7 | 11.2 | 0.3 | 10.9 | 1488.2 | ||
| Group | Sample ID | Sampling Date | Concentration (µg/L) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fe | Mn | Al | Sr | Li | Zn | As | U | Ba | |||
| I | G-1 | February-2013 | n.d. | 133 | 197.0 | 226 | 9.7 | 16.5 | n.a. | n.a. | 166.0 |
| February-2014 | 12.6 | 51 | 237.2 | 381 | 8.2 | 22.0 | n.a. | n.a. | 174.2 | ||
| March-2015 | 1.8 | 91 | 171.7 | 429 | 0.0 | 14.9 | 0.3 | 1.0 | 189.0 | ||
| Average | 7.2 | 91.7 | 202.0 | 345.3 | 6.0 | 17.8 | 0.3 | 1.0 | 176.4 | ||
| II | G-2 | February-2013 | - | 1367 | 2.7 | 3542 | 945.0 | 17.5 | n.a. | n.a. | 126.0 |
| February-2014 | - | 99 | 32.9 | 4107 | 1356.3 | 30.7 | n.a. | n.a. | 164.9 | ||
| March-2015 | 6842.0 | 1023 | 6.5 | 5236 | 997.0 | 12.1 | 10.4 | 15.4 | 134.1 | ||
| G-3 | February-2014 | - | 464 | 29.3 | 3047 | 1147.3 | 16.9 | n.a. | n.a. | 253.7 | |
| March-2015 | 9581.0 | 869 | 18.0 | 3795 | 955.0 | 7.8 | 55.5 | 18.4 | 235.3 | ||
| Average | 8211.5 | 764.4 | 17.9 | 3945.4 | 1080.1 | 17.0 | 32.9 | 16.9 | 182.8 | ||
| III | G-4 | February-2014 | - | 845 | 23.9 | 4114 | 437.0 | 51.7 | n.a. | n.a. | 326.9 |
| March-2015 | 5390.0 | 1122 | 0.7 | 4290 | 359.0 | 8.9 | 4.5 | 5.0 | 303.9 | ||
| G-5 | February-2013 | - | - | 1.1 | 3496 | 501.0 | 13.4 | n.a. | n.a. | 306.0 | |
| February-2014 | - | - | 73.1 | 2100 | 435.8 | 6.4 | n.a. | n.a. | 294.2 | ||
| March-2015 | 11,260.0 | 924 | 2.5 | 3520 | 353.0 | 13.8 | 4.0 | 14.1 | 426.3 | ||
| Average | 8325.0 | 963.7 | 20.3 | 3504.0 | 417.2 | 18.8 | 4.3 | 9.5 | 331.5 | ||
| IV | G-6 | February-2013 | - | 2106 | 10.8 | 3237 | 146.0 | 45.6 | n.a. | n.a. | 459.0 |
| February-2014 | 22,651.4 | 2082 | 55.5 | 3313 | 131.8 | 57.1 | n.a. | n.a. | 565.9 | ||
| March-2015 | 22,070.0 | 1870 | 6.3 | 3278 | 104.0 | 24.7 | 1.1 | 40.2 | 591.3 | ||
| G-7 | February-2014 | 13,564.0 | 442 | 58.0 | 1650 | 93.2 | 25.9 | n.a. | n.a. | 327.6 | |
| G-8 | Feb-2013 | 7949.0 | 1228 | 0.0 | 12,758 | 193.0 | 69.0 | n.a. | n.a. | 231.0 | |
| Average | 16,558.6 | 1545.6 | 26.1 | 4847.2 | 133.6 | 44.5 | 1.1 | 40.2 | 435.0 | ||
| Surface water in the study area (1) | 24~300 | 1~51 | 1.4~554 | 32~450 | 0.6~49.9 | 0.2~4.2 | 0.6~1.3 | 0.1~1.0 | 5.0~307.0 | ||
| Shallow groundwater in the study area (2) | 19~100 | n.d. | 3.5~9 | 50~94 | 0.6~108 | 2.1~6.3 | 50.1 | n.d. | 19.6~28.0 | ||
| Guidelines for Drinking-Water Quality in Chemical Aspects (WHO) (3) | - | - | - | - | - | - | 10 | 30 | 1300 | ||
| Guidelines for Drinking-Water Quality in Acceptability Aspects (WHO) (4) | 300 | 100 | 100 | - | - | 4000 | - | - | - | ||
| Group | Sample ID | δ18O (‰) | δD (‰) | δ13C (‰) | 14C (pMC) | Age (BP) |
|---|---|---|---|---|---|---|
| I | G-1 | −8.8 | −60.8 | −1.3 | 1.2 | 35,720 ± 330 |
| −8.7 | −58.3 | −5.8 | n.a. | n.a. | ||
| −8.9 | −58.6 | −6.3 | 0.6 | 41,190 ± 560 | ||
| II | G-2 | −9.4 | −64.7 | −1.0 | n.a. | n.a. |
| −9.7 | −64.3 | −0.6 | n.a. | n.a. | ||
| −9.4 | −63.1 | −3.2 | 0.0 | >43,500 | ||
| G-3 | −10.2 | −68.6 | −2.8 | n.a. | n.a. | |
| −10.2 | −70.3 | −4.8 | 0.0 | >43,500 | ||
| III | G-4 | −9.1 | −62.0 | −3.8 | n.a. | n.a. |
| −9.1 | −62.6 | −2.1 | 0.0 | >43,500 | ||
| G-5 | −9.2 | −62.5 | −3.2 | 0.7 | 40,010 ± 500 | |
| −9.2 | −59.0 | −1.8 | n.a. | n.a. | ||
| −9.2 | −59.5 | −2.3 | 0.0 | >43,500 | ||
| IV | G-6 | −9.1 | −61.3 | −2.9 | 0.0 | >43,500 |
| −9.0 | −58.7 | −3.7 | n.a. | n.a. | ||
| −9.1 | −59.0 | −3.1 | 0.0 | >43,500 | ||
| G-7 | −9.5 | −61.4 | −4.0 | n.a. | n.a. | |
| G-8 | −9.1 | −62.1 | −4.1 | n.a. | n.a. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Do, H.-K.; Yu, S.; Yun, S.-T. Hydrochemical Parameters to Assess the Evolutionary Process of CO2-Rich Spring Water: A Suggestion for Evaluating CO2 Leakage Stages in Silicate Rocks. Water 2020, 12, 3421. https://doi.org/10.3390/w12123421
Do H-K, Yu S, Yun S-T. Hydrochemical Parameters to Assess the Evolutionary Process of CO2-Rich Spring Water: A Suggestion for Evaluating CO2 Leakage Stages in Silicate Rocks. Water. 2020; 12(12):3421. https://doi.org/10.3390/w12123421
Chicago/Turabian StyleDo, Hyun-Kwon, Soonyoung Yu, and Seong-Taek Yun. 2020. "Hydrochemical Parameters to Assess the Evolutionary Process of CO2-Rich Spring Water: A Suggestion for Evaluating CO2 Leakage Stages in Silicate Rocks" Water 12, no. 12: 3421. https://doi.org/10.3390/w12123421





