Conservation Paleobiology as a Tool to Define Reference Conditions in Naturally Stressed Transitional Settings: Micropaleontological Insights from the Holocene of the Po Coastal Plain (Italy)
Abstract
1. Introduction
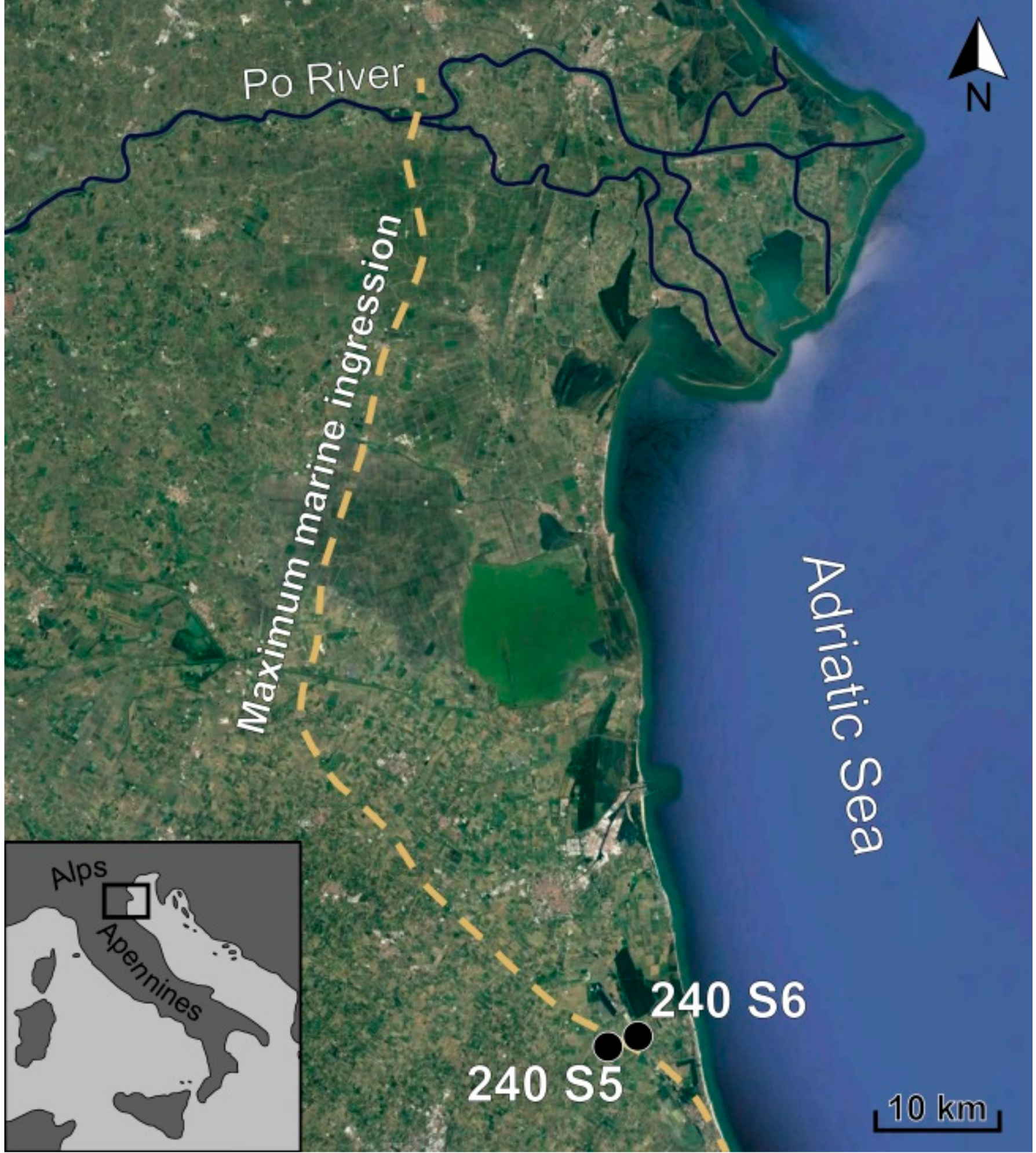
2. Geological Setting
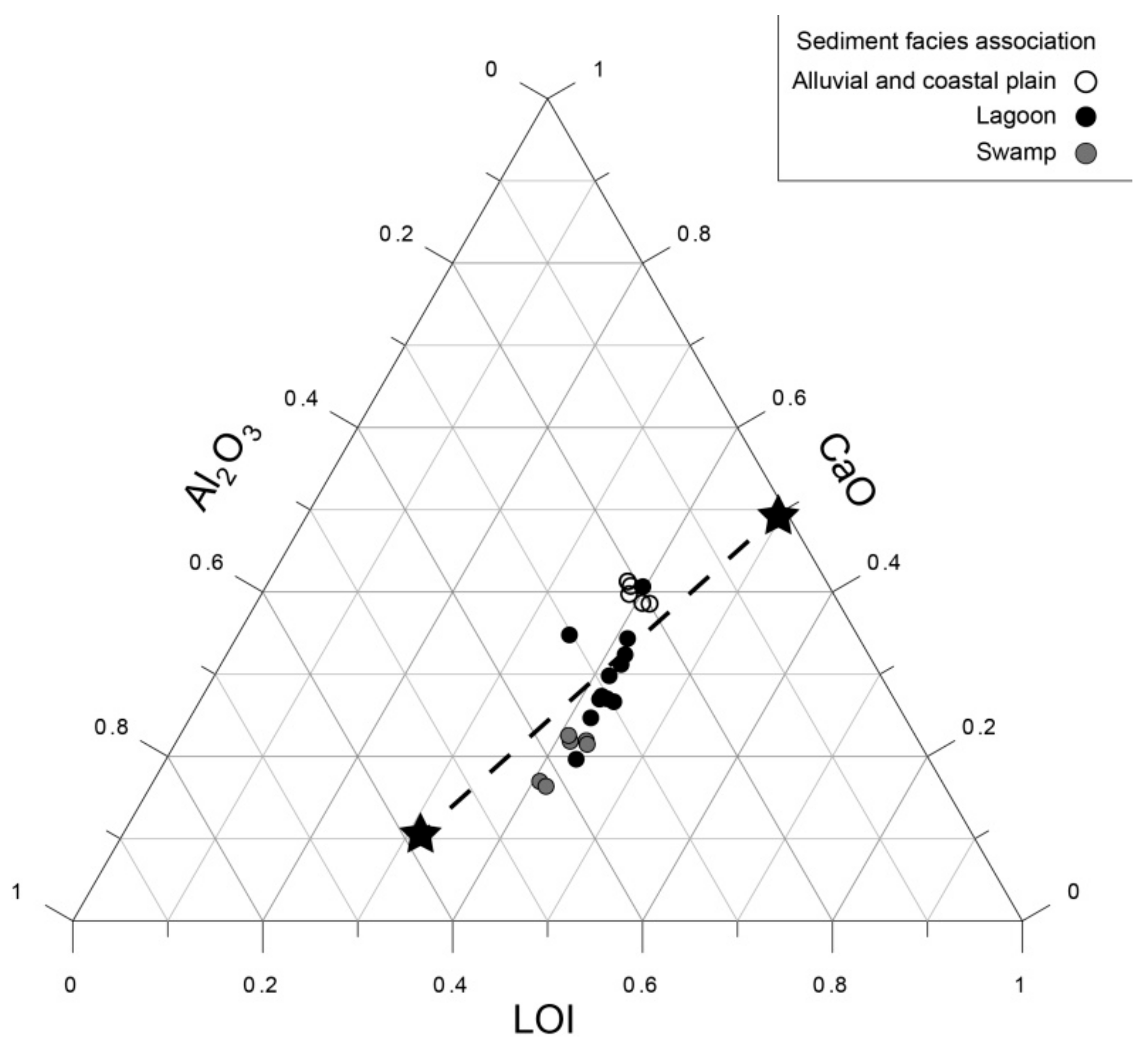
3. Materials and Methods
4. Results
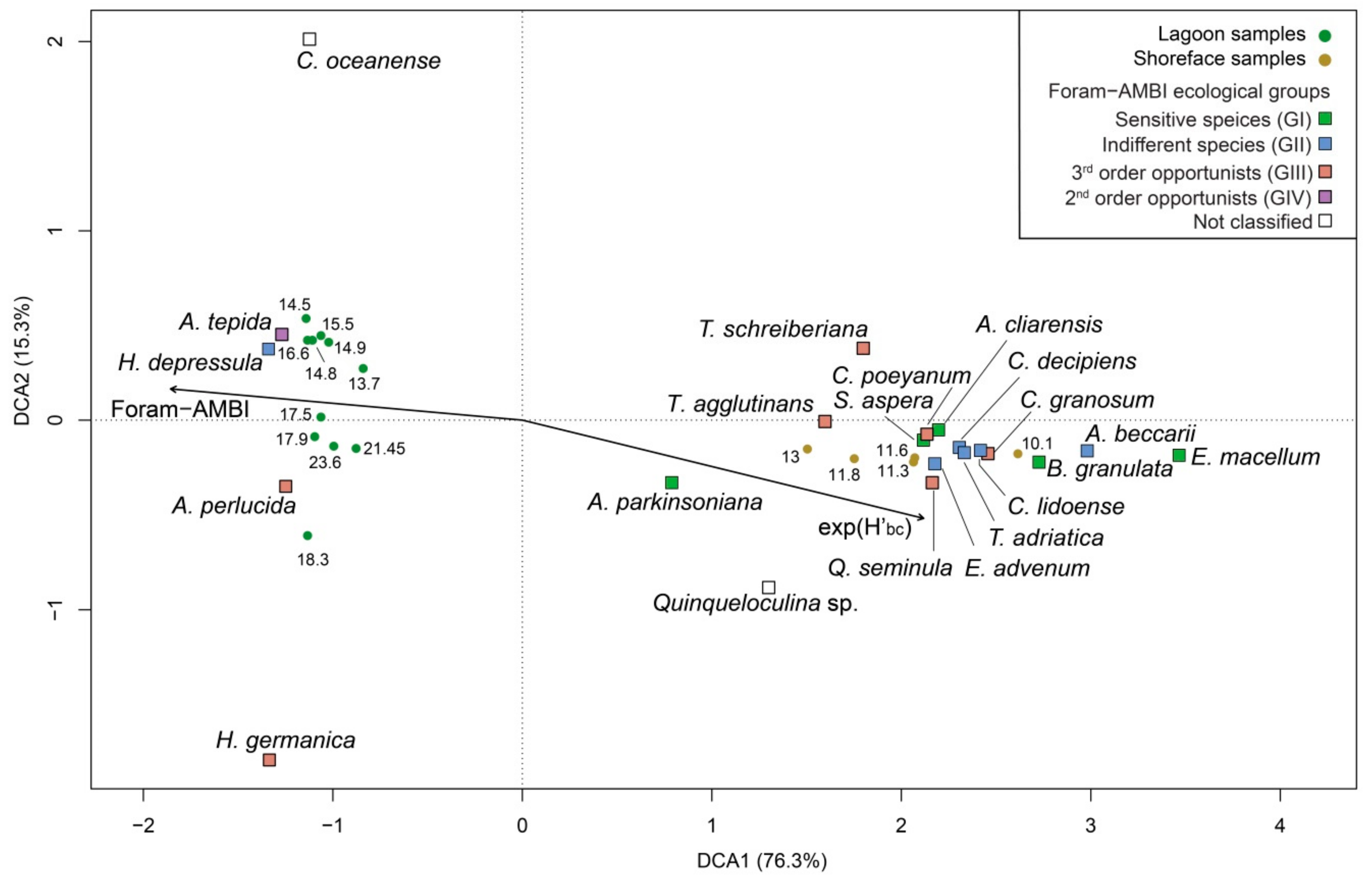
4.1. Benthic Foraminifers
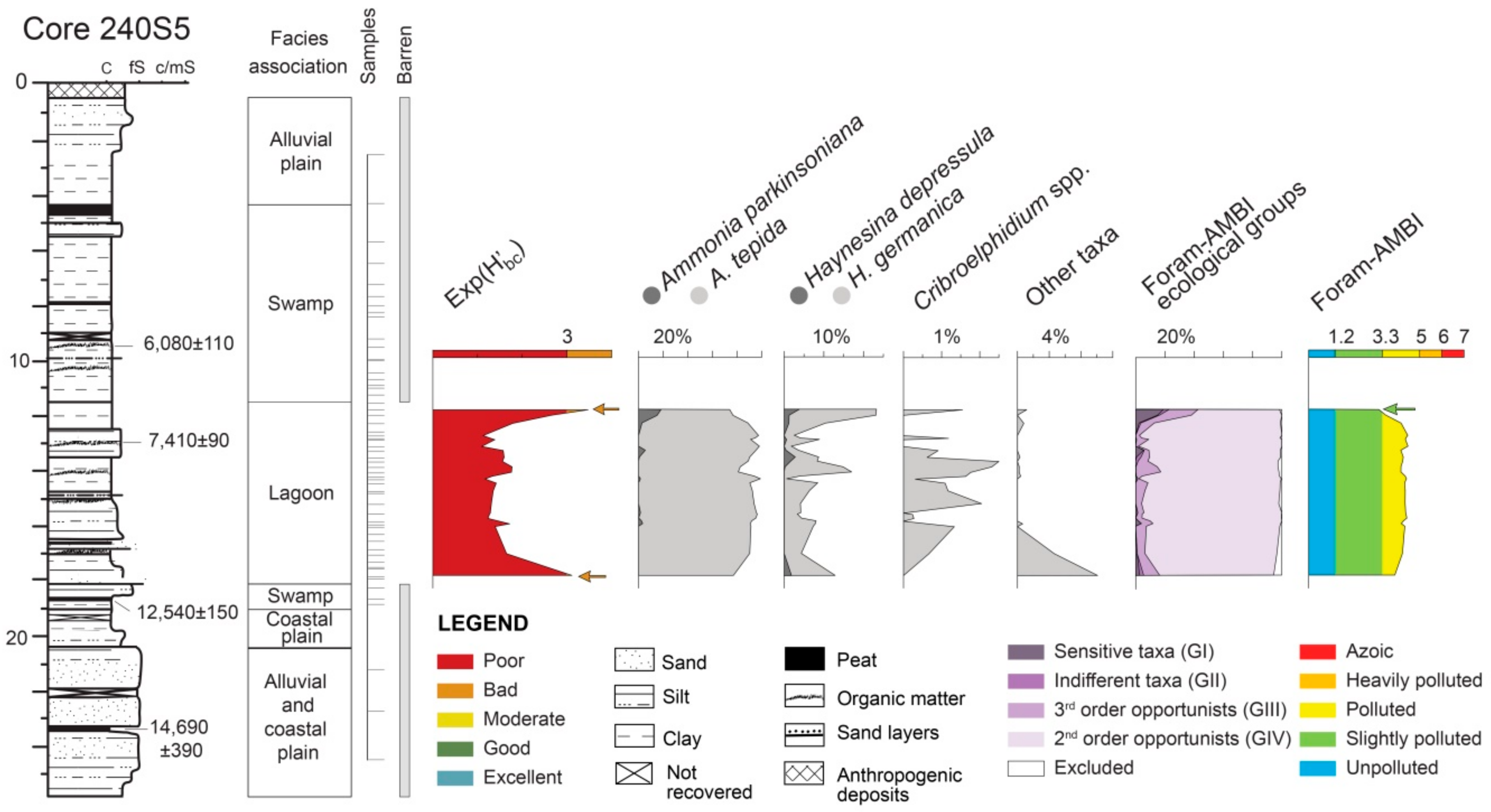
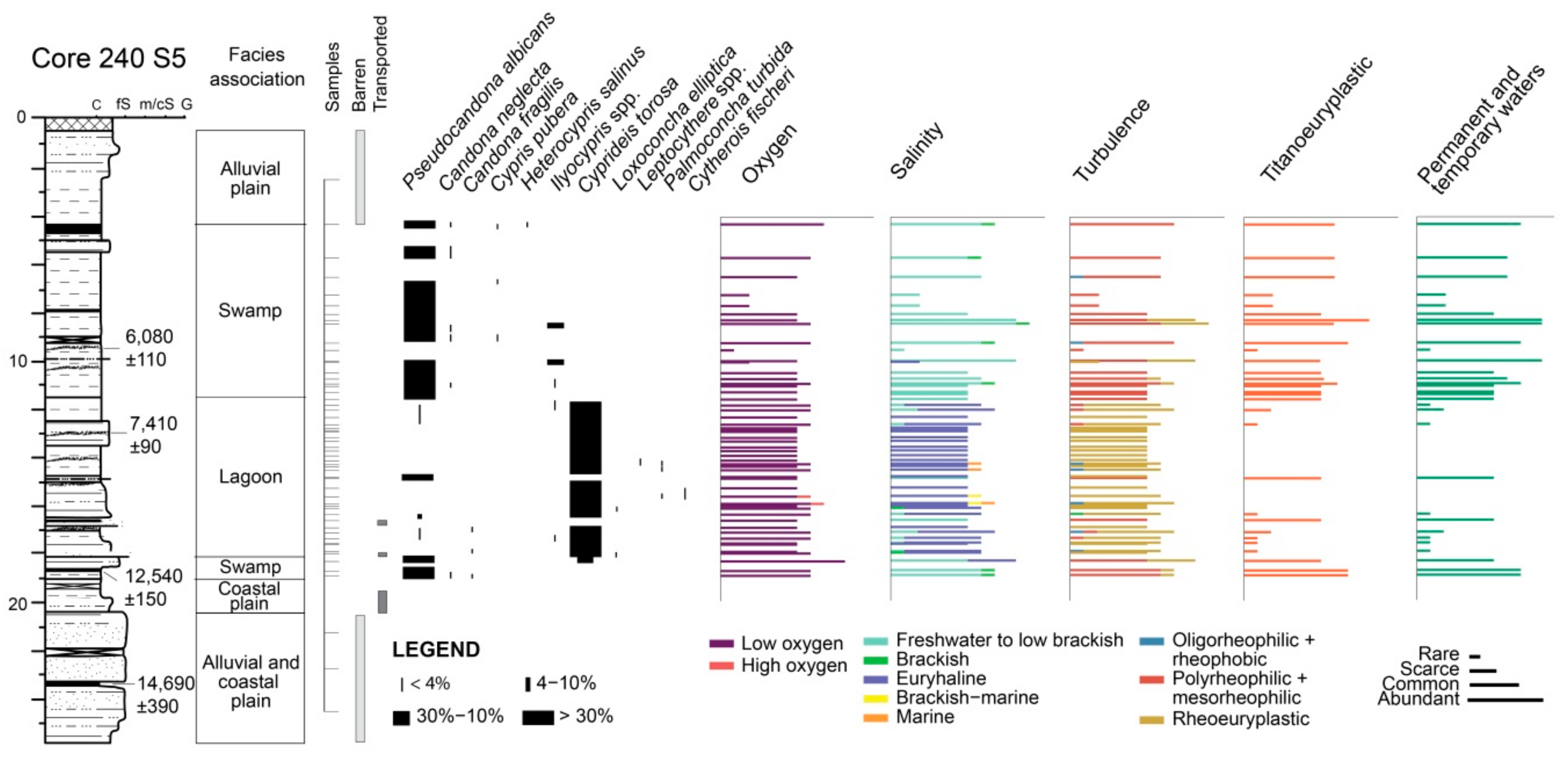
4.1.1. Core 240 S5
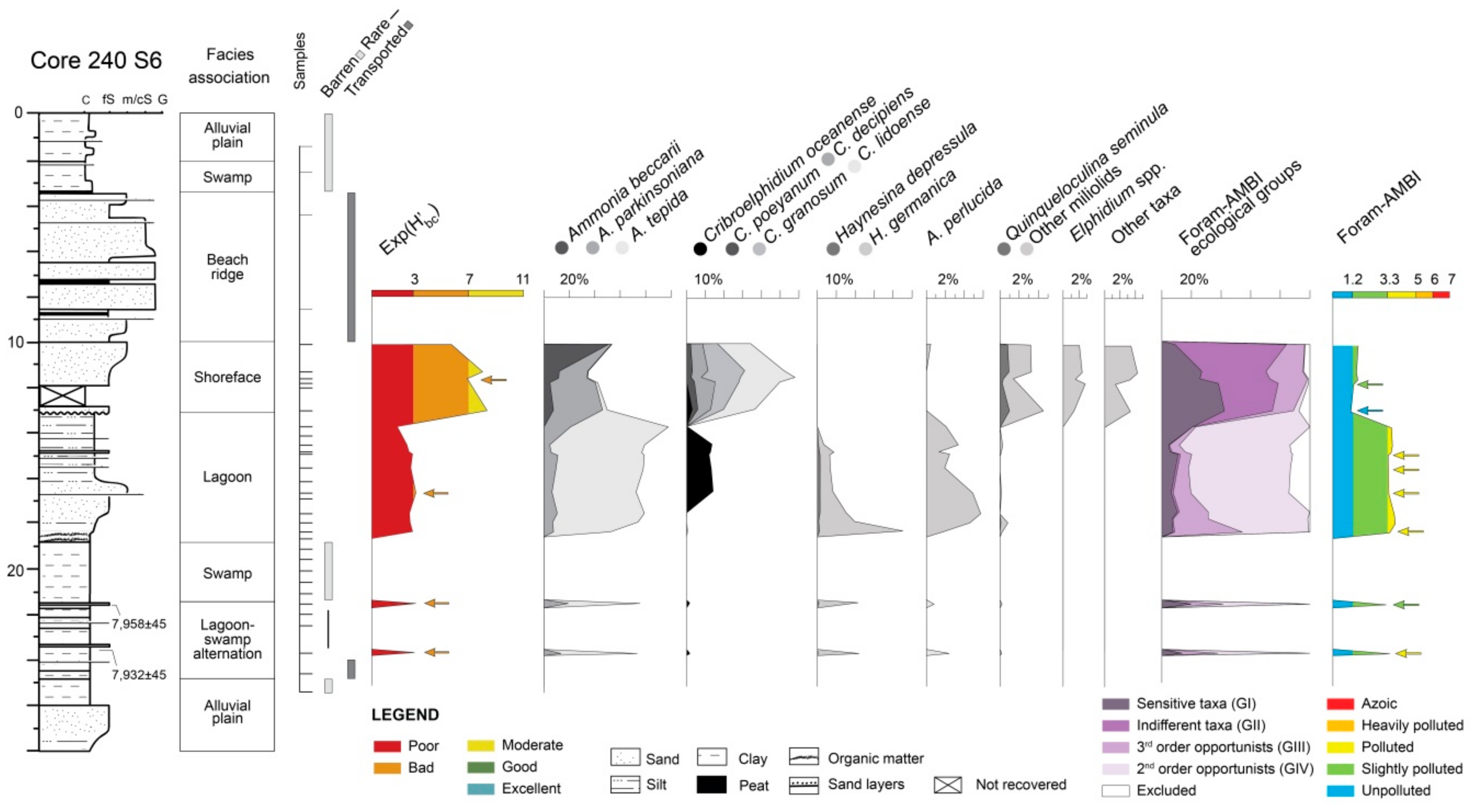
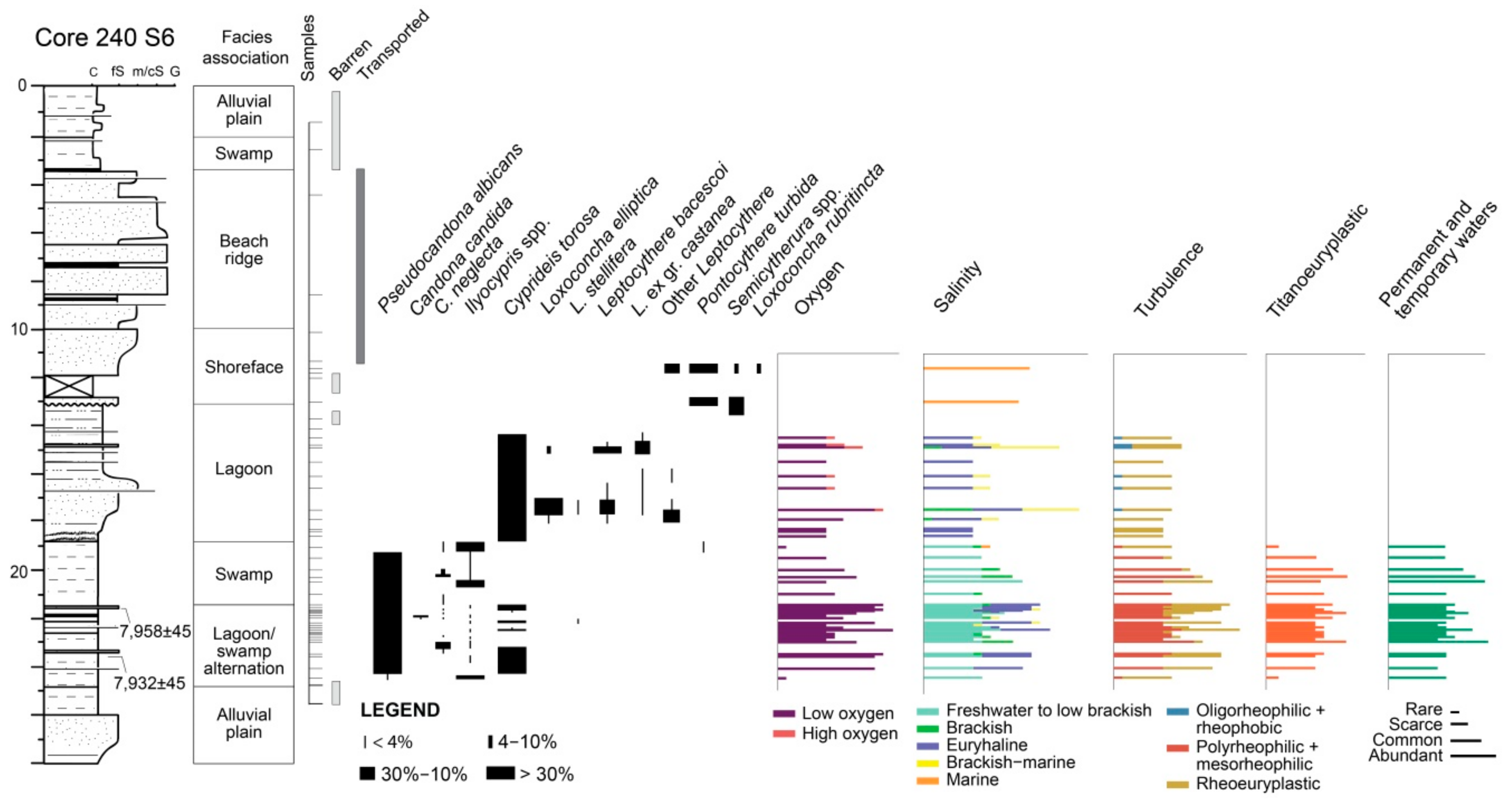
4.1.2. Core 240 S6
4.2. Ostracods
4.2.1. Core 240 S5
4.2.2. Core 240 S6
5. Discussion
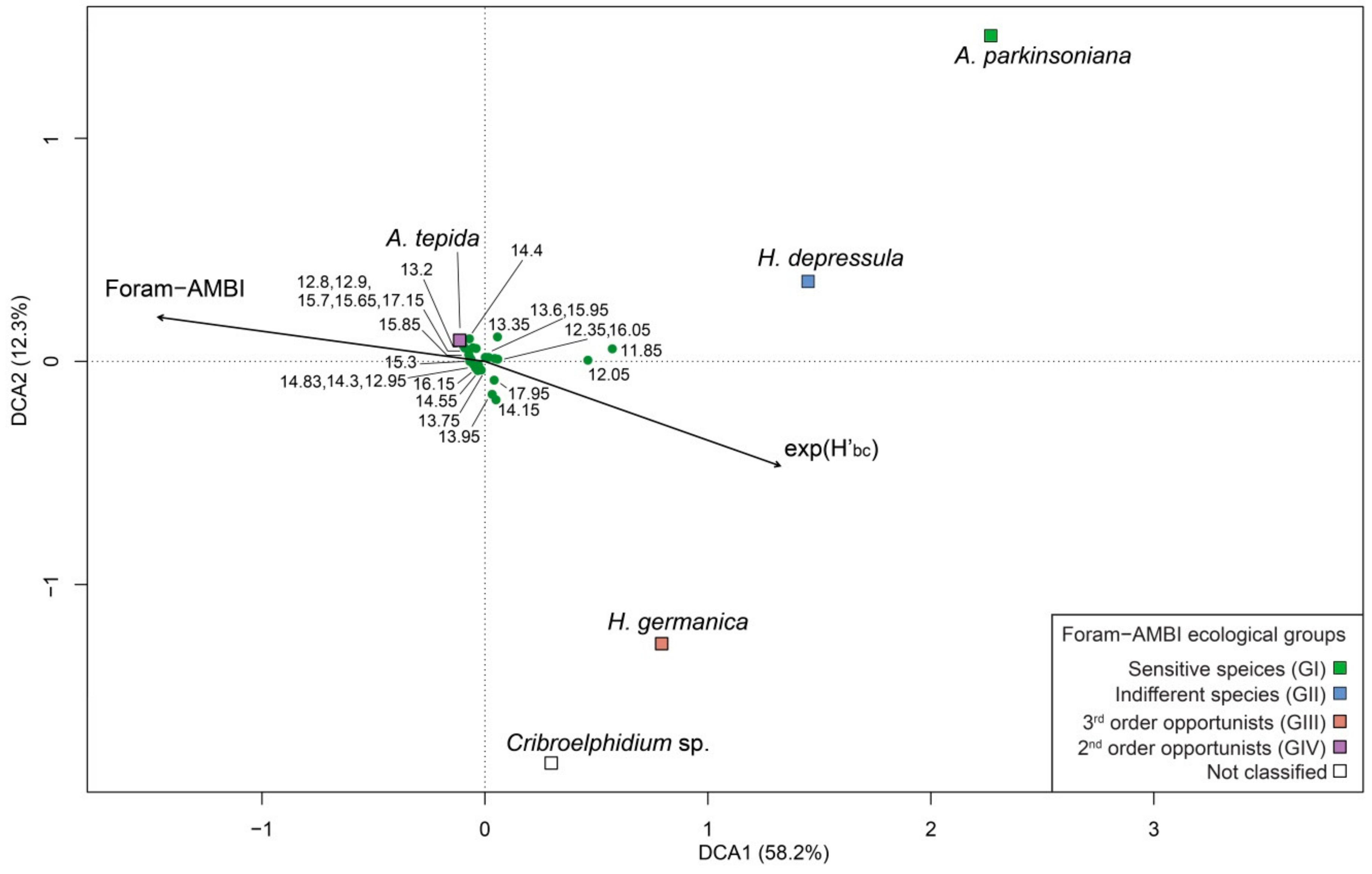
5.1. PaleoEcoQs through Short Paleoenvironmental Gradients: Restricted Back-Barrier Settings (Core 240 S5)
5.2. PaleoEcoQs through Long Paleoenvironmental Gradients: From Transitional to Coastal Settings (Core 240 S6)
5.3. Temporal Resolution of the Fossil Record
5.4. How to Define Reference Conditions?
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lloret, J.; Marín, A.; Marín-Guirao, L. Is coastal lagoon eutrophication likely to be aggravated by global climate change? Estuar. Coast. Shelf Sci. 2008, 78, 403–412. [Google Scholar] [CrossRef]
- Munari, C.; Mistri, M. The performance of benthic indicators of ecological change in Adriatic coastal lagoons: Throwing the baby with the water? Mar. Pollut. Bull. 2008, 56, 95–105. [Google Scholar] [CrossRef] [PubMed]
- European Parliament. Directive 2008/56/EC of the European Parliament and of the Council of 17 June 2008 establishing a framework for community action in the field of marine environmental policy (Marine Strategy Framework Directive). Off. J. Eur. Union 2008, L164, 1–51. [Google Scholar]
- European Commission. Common Implementation Strategy for the Water Framework Directive (2000/60/EC). Transitional and Coastal Waters: Typology, Reference Conditions and Classification Systems; Guidance Document. No 5; Office for Official Publications of the European Communities: Luxembourg, 2003. [Google Scholar]
- Dauvin, J.-C. Paradox of estuarine quality: Benthic indicators and indices, consensus or debate for the future. Mar. Pollut. Bull. 2007, 55, 271–281. [Google Scholar] [CrossRef]
- Elliott, M.; Quintino, V. The Estuarine Quality Paradox, Environmental Homeostasis and the difficulty of detecting anthropogenic stress in naturally stressed areas. Mar. Pollut. Bull. 2007, 54, 640–645. [Google Scholar] [CrossRef]
- Ruellet, T.; Dauvin, J.-C. Benthic indicators: Analysis of the threshold values of ecological quality classifications for transitional waters. Mar. Pollut. Bull. 2007, 54, 1707–1714. [Google Scholar] [CrossRef]
- Fitch, J.E.; Cooper, K.M.; Crowe, T.P.; Hall-Spencer, J.M.; Phillips, G. Response of multi-metric indices to anthropogenic pressures in distinct marine habitats: The need for recalibration to allow wider applicability. Mar. Pollut. Bull. 2014, 87, 220–229. [Google Scholar] [CrossRef]
- Dietl, G.P.; Flessa, K.W. Conservation paleobiology: Putting the dead to work. Trends Ecol. Evol. 2011, 26, 30–37. [Google Scholar] [CrossRef]
- Dietl, G.P.; Kidwell, S.M.; Brenner, M.; Burney, D.A.; Flessa, K.W.; Jackson, S.T.; Koch, P.L. Conservation paleobiology: Leveraging knowledge of the past to inform conservation and restoration. Annu. Rev. Earth Planet. Sci 2015, 43, 79–103. [Google Scholar] [CrossRef]
- Dietl, G.P.; Smith, J.A.; Durham, S.R. Discounting the past: The undervaluing of paleontological data in conservation science. Front. Ecol. Evol. 2019, 7, 108. [Google Scholar] [CrossRef]
- O’Dea, A.; Lepore, M.; Altieri, A.H.; Chan, M.; Morales-Saldaña, J.M.; Muñoz, N.-H.; Pandolfi, J.M.; Toscano, M.A.; Zhao, J.; Dillon, E.M. Defining variation in pre-human ecosystems can guide conservation: An example from a Caribbean coral reef. Sci. Rep. 2020, 10, 2922. [Google Scholar] [CrossRef] [PubMed]
- Bouchet, V.M.P.; Alve, E.; Rygg, B.; Telford, R.J. Benthic foraminifera provide a promising tool for ecological quality assessment of marine waters. Ecol. Indic. 2012, 23, 66–75. [Google Scholar] [CrossRef]
- Dimiza, M.D.; Triantaphyllou, M.V.; Koukousioura, O.; Hallock, P.; Simboura, N.; Karageorgis, A.P.; Papathanasiou, E. The Foram Stress Index: A new tool for environmental assessment of soft-bottom environments using benthic foraminifera. A case study from the Saronikos Gulf, Greece, Eastern Mediterranean. Ecol. Indic. 2016, 60, 611–621. [Google Scholar] [CrossRef]
- Alve, E.; Korsun, S.; Schönfeld, J.; Dijkstra, N.; Golikova, E.; Hess, S.; Husum, K.; Panieri, G. Foram−AMBI: A sensitivity index based on benthic foraminiferal faunas from North-East Atlantic and Arctic fjords, continental shelves and slopes. Mar. Micropaleontol. 2016, 122, 1–12. [Google Scholar] [CrossRef]
- Alve, E.; Hess, S.; Bouchet, V.M.P.; Dolven, J.K.; Rygg, B. Intercalibration of benthic foraminiferal and macrofaunal biotic indices: An example from the Norwegian Skagerrak coast (NE North Sea). Ecol. Indic. 2019, 96, 107–115. [Google Scholar] [CrossRef]
- Jorissen, F.; Nardelli, M.P.; Almogi-Labin, A.; Barras, C.; Bergamin, L.; Bicchi, E.; El Kateb, A.; Ferraro, L.; McGann, M.; Morigi, C.; et al. Developing Foram−AMBI for biomonitoring in the Mediterranean: Species assignments to ecological categories. Mar. Micropaleontol. 2018, 140, 33–45. [Google Scholar] [CrossRef]
- Melis, R.; Celio, M.; Bouchet, V.M.P.; Varagona, G.; Bazzaro, M.; Crosera, M.; Pugliese, N. Seasonal response of benthic foraminifera to anthropogenic pressure in two stations of the Gulf of Trieste (northern Adriatic Sea, Italy): The marine protected area of Miramare versus the Servola water sewage outfall. Medit. Mar. Sci. 2019, 20, 120–141. [Google Scholar] [CrossRef]
- El Kateb, A.; Stalder, C.; Martínez-Colón, M.; Mateu-Vicens, G.; Francescangeli, F.; Coletti, G.; Stainbank, S.; Spezzaferri, S. Foraminiferal-based biotic indices to assess the ecological quality status of the Gulf of Gabes (Tunisia): Present limitations and future perspectives. Ecol. Indic. 2020, 111, 105962. [Google Scholar] [CrossRef]
- Klootwijk, A.T.; Alve, E.; Hess, S.; Renaud, P.E.; Sørlie, C.; Dolven, J.K. Monitoring environmental impacts of fish farms: Comparing reference conditions of sediment geochemistry and benthic foraminifera with the present. Ecol. Indic. 2021, 120, 106818. [Google Scholar] [CrossRef]
- Bouchet, V.M.P.; Goberville, E.; Frontalini, F. Benthic foraminifera to assess Ecological Quality Statuses in Italian transitional waters. Ecol. Indic. 2018, 84, 130–139. [Google Scholar] [CrossRef]
- Frontalini, F.; Margaritelli, G.; Francescangeli, F.; Rettori, R.; Armynot du Châtelet, E.; Coccioni, R. Benthic foraminiferal assemblages and biotopes in a coastal lake: The case study of Lake Varano (Southern Italy). Acta Protozool. 2013, 52, 147–160. [Google Scholar]
- Coccioni, R.; Frontalini, F.; Marsili, A.; Mana, D. Benthic foraminifera and trace element distribution: A case-study from the heavily polluted lagoon of Venice (Italy). Mar. Pollut. Bull. 2009, 59, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Frontalini, F.; Coccioni, R.; Bucci, C. Benthic foraminiferal assemblages and trace element contents from the lagoons of Orbetello and Lesina. Environ. Monit. Assess. 2010, 170, 245–260. [Google Scholar] [CrossRef] [PubMed]
- Dinelli, E.; Ghosh, A.; Rossi, V.; Vaiani, S.C. Multiproxy reconstruction of late Pleistocene-Holocene environmental changes in coastal successions: Microfossil and geochemical evidences from the Po Plain (Northern Italy). Stratigraphy 2012, 9, 153–167. [Google Scholar]
- Vaiani, S.C.; Pennisi, M. Tracing freshwater provenance in palaeo-lagoons by boron isotopes and relationship with benthic foraminiferal assemblages. A comparison from late Quaternary subsurface successions in Northern and Central Italy. Boll. Soc. Paleontol. Ital. 2014, 53, 55–66. [Google Scholar] [CrossRef]
- Amorosi, A.; Bruno, L.; Campo, B.; Morelli, A.; Rossi, V.; Scarponi, D.; Hong, W.; Bohacs, K.M.; Drexler, T.M. Global sea-level control on local parasequence architecture from the Holocene record of the Po Plain, Italy. Mar. Pet. Geol. 2017, 87, 99–111. [Google Scholar] [CrossRef]
- Pint, A.; Engel, M.; Melzer, S.; Frenzel, P.; Plessen, B.; Brückner, H. How to discriminate athalassic and marginal marine microfaunas: Foraminifera and other fossils from an early Holocene continental lake in northern Saudi Arabia. J. Foraminifer. Res. 2017, 47, 175–187. [Google Scholar] [CrossRef]
- Barbieri, G.; Vaiani, S.C. Benthic foraminifera or Ostracoda? Comparing the accuracy of palaeoenvironmental indicators from a Pleistocene lagoon of the Romagna coastal plain (Italy). J. Micropalaeontol. 2018, 37, 203–230. [Google Scholar] [CrossRef]
- Pascual, A.; Rodriguez-Lazaro, J.; Weber, O.; Jouanneau, J.M. Late Holocene pollution in the Gernika estuary (southern Bay of Biscay) evidenced by the study of Foraminifera and Ostracoda. Hydrobiologia 2002, 475, 477–491. [Google Scholar]
- Ruiz, F.; Abad, M.; Bodergat, A.M.; Carbonel, P.; Rodríguez-Lázaro, J.; Yasuhara, M. Marine and brackish-water ostracods as sentinels of anthropogenic impacts. Earth Sci. Rev. 2005, 72, 89–111. [Google Scholar] [CrossRef]
- Pint, A.; Frenzel, P.; Horne, D.J.; Franke, J.; Daniel, T.; Burghardt, A.; Funai, B.; Lippold, K.; Daut, G.; Wennrich, V. Ostracoda from inland waterbodies with saline influence in Central Germany: Implications for palaeoenvironmental reconstruction. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2015, 419, 37–46. [Google Scholar] [CrossRef]
- Rossi, V.; Amorosi, A.; Sarti, G.; Mariotti, S. Late Quaternary multiple incised valley systems: An unusually well-preserved stratigraphic record of two interglacial valley-fill successions from the Arno Plain (northern Tuscany, Italy). Sedimentology 2017, 64, 1901–1928. [Google Scholar] [CrossRef]
- D’Orefice, M.; Bellotti, P.; Bertini, A.; Calderoni, G.; Censi Neri, P.; Di Bella, L.; Fiorenza, D.; Foresi, L.M.; Louvari, M.A.; Rainone, L.; et al. Holocene evolution of the Burano Paleo-Lagoon (Southern Tuscany, Italy). Water 2020, 12, 1007. [Google Scholar] [CrossRef]
- Donnini, F.; Dinelli, E.; Sangiorgi, F.; Fabbri, E. A biological and geochemical integrated approach to assess the environmental quality of a coastal lagoon (Ravenna, Italy). Environ. Int. 2007, 33, 919–928. [Google Scholar] [CrossRef]
- Migani, F.; Borghesi, F.; Dinelli, E. Geochemical characterization of surface sediments from the northern Adriatic wetlands around the Po river delta. Part I: Bulk composition and relation to local background. J. Geochem. Explor. 2015, 156, 72–88. [Google Scholar] [CrossRef]
- Zonta, R.; Cassin, D.; Pini, R.; Dominik, J. Assessment of heavy metal and As contamination in the surface sediments of Po delta lagoons (Italy). Estuar. Coast. Shelf Sci. 2019, 225, 106235. [Google Scholar] [CrossRef]
- Pitacco, V.; Mistri, M.; Ferrari, C.R.; Sfriso, A.; Sfriso, A.A.; Munari, C. Multiannual trend of micro-pollutants in sediments and benthic community response in a Mediterranean Lagoon (Sacca di Goro, Italy). Water 2020, 12, 1074. [Google Scholar] [CrossRef]
- Kusnerik, K.M.; Lockwood, R.; Grant, A.N. Using the fossil record to establish a baseline and recommendations for oyster mitigation in the Mid-Atlantic U.S. In Marine Conservation Paleobiology; Tyler, C.L., Schneider, C.L., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 75–103. ISBN 978-3-319-73795-9. [Google Scholar]
- Tomašových, A.; Albano, P.G.; Fuksi, T.; Gallmetzer, I.; Haselmair, A.; Kowalewski, M.; Nawrot, R.; Nerlović, V.; Scarponi, D.; Zuschin, M. Ecological regime shift preserved in the Anthropocene stratigraphic record. Proc. R. Soc. B Biol. Sci. 2020, 287, 20200695. [Google Scholar] [CrossRef]
- Kowalewski, M.; Wittmer, J.M.; Dexter, T.A.; Amorosi, A.; Scarponi, D. Differential responses of marine communities to natural and anthropogenic changes. Proc. R. Soc. B 2015, 282, 20142990. [Google Scholar] [CrossRef]
- Barmawidjaja, D.M.; van der Zwaan, G.J.; Jorissen, F.J.; Puskaric, S. 150 years of eutrophication in the northern Adriatic Sea: Evidence from a benthic foraminiferal record. Mar. Geol. 1995, 122, 367–384. [Google Scholar] [CrossRef]
- Pranovi, F.; Giovanardi, O.; Franceschini, G. Recolonization dynamics in areas disturbed by bottom fishing gears. In Recruitment, Colonization and Physical-Chemical Forcing in Marine Biological Systems; Baden, S., Pihl, L., Rosenberg, R., Strömberg, J.O., Svane, I., Tiselius, P., Eds.; Springer: Heidelberg, Germany, 1998; Volume 375/376, pp. 125–135. [Google Scholar]
- Gallmetzer, I.; Haselmair, A.; Tomašových, A.; Stachowitsch, M.; Zuschin, M. Responses of molluscan communities to centuries of human impact in the northern Adriatic Sea. PLoS ONE 2017, 12, e0180820. [Google Scholar] [CrossRef] [PubMed]
- Pitacco, V.; Mistri, M.; Munari, C. Long-term variability of macrobenthic community in a shallow coastal lagoon (Valli di Comacchio, northern Adriatic): Is community resistant to climate changes? Mar. Environ. Res. 2018, 137, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Tomašových, A.; Gallmetzer, I.; Haselmair, A.; Kaufman, D.S.; Kralj, M.; Cassin, D.; Zonta, R.; Zuschin, M. Tracing the effects of eutrophication on molluscan communities in sediment cores: Outbreaks of an opportunistic species coincide with reduced bioturbation and high frequency of hypoxia in the Adriatic Sea. Paleobiology 2018, 44, 575–602. [Google Scholar] [CrossRef]
- Albano, P.G.; Gallmetzer, I.; Haselmair, A.; Tomašových, A.; Stachowitsch, M.; Zuschin, M. Historical ecology of a biological invasion: The interplay of eutrophication and pollution determines time lags in establishment and detection. Biol. Invasions 2018, 20, 1417–1430. [Google Scholar] [CrossRef]
- Seddon, A.W.R.; Froyd, C.A.; Witkowski, A.; Willis, K.J. A quantitative framework for analysis of regime shifts in a Galápagos coastal lagoon. Ecology 2014, 95, 3046–3055. [Google Scholar] [CrossRef]
- Stegner, M.A.; Ratajczak, Z.; Carpenter, S.R.; Williams, J.W. Inferring critical transitions in paleoecological time series with irregular sampling and variable time-averaging. Quat. Sci. Rev. 2019, 207, 49–63. [Google Scholar] [CrossRef]
- Dolven, J.K.; Alve, E.; Rygg, B.; Magnusson, J. Defining past ecological status and in situ reference conditions using benthic foraminifera: A case study from the Oslofjord, Norway. Ecol. Indic. 2013, 29, 219–233. [Google Scholar] [CrossRef]
- Campo, B.; Bruno, L.; Amorosi, A. Basin-scale stratigraphic correlation of late Pleistocene-Holocene (MIS 5e-MIS 1) strata across the rapidly subsiding Po Basin (northern Italy). Quat. Sci. Rev. 2020, 237, 106300. [Google Scholar] [CrossRef]
- Jones, G.D. Paralic. In Beaches and Coastal Geology; Schwartz, M., Ed.; Encyclopedia of Earth Science Series I; Springer: Boston, MA, USA, 1984; p. 631. ISBN 978-0-387-30843-2. [Google Scholar]
- Amorosi, A.; Colalongo, M.L.; Pasini, G.; Preti, D. Sedimentary response to Late Quaternary sea-level changes in the Romagna coastal plain (northern Italy). Sedimentology 1999, 46, 99–121. [Google Scholar] [CrossRef]
- Campo, B.; Amorosi, A.; Vaiani, S.C. Sequence stratigraphy and late Quaternary paleoenvironmental evolution of the Northern Adriatic coastal plain (Italy). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2017, 466, 265–278. [Google Scholar] [CrossRef]
- Amorosi, A.; Colalongo, M.L.; Fiorini, F.; Fusco, F.; Pasini, G.; Vaiani, S.C.; Sarti, G. Palaeogeographic and palaeoclimatic evolution of the Po Plain from 150-ky core records. Glob. Planet. Chang. 2004, 40, 55–78. [Google Scholar] [CrossRef]
- Bruno, L.; Amorosi, A.; Severi, P.; Costagli, B. Late Quaternary aggradation rates and stratigraphic architecture of the southern Po Plain, Italy. Basin Res. 2017, 29, 234–248. [Google Scholar] [CrossRef]
- Amorosi, A.; Barbieri, G.; Bruno, L.; Campo, B.; Drexler, T.M.; Hong, W.; Rossi, V.; Sammartino, I.; Scarponi, D.; Vaiani, S.C.; et al. Three-fold nature of coastal progradation during the Holocene eustatic highstand, Po Plain, Italy—Close correspondence of stratal character with distribution patterns. Sedimentology 2019, 66, 3029–3052. [Google Scholar] [CrossRef]
- Amorosi, A.; Sammartino, I. Influence of sediment provenance on background values of potentially toxic metals from near-surface sediments of Po coastal plain (Italy). Int. J. Earth Sci. Geol. Rundsch. 2007, 96, 389–396. [Google Scholar] [CrossRef]
- Amorosi, A.; Centineo, M.C.; Dinelli, E.; Lucchini, F.; Tateo, F. Geochemical and mineralogical variations as indicators of provenance changes in Late Quaternary deposits of SE Po Plain. Sediment. Geol. 2002, 151, 273–292. [Google Scholar] [CrossRef]
- Borghesi, F.; Migani, F.; Dinelli, E. Geochemical characterization of surface sediments from the northern Adriatic wetlands around the Po River delta. Part II: Aqua regia results. J. Geochem. Explor. 2016, 169, 13–29. [Google Scholar] [CrossRef]
- Amorosi, A. Chromium and nickel as indicators of source-to-sink sediment transfer in a Holocene alluvial and coastal system (Po Plain, Italy). Sediment. Geol. 2012, 280, 260–269. [Google Scholar] [CrossRef]
- Cibin, U.; Severi, P.; Correggiari, A.; Roveri, M. Note Illustrative della Carta Geologica d’Italia alla Scala 1:50.000—Foglio 240–241 Forlì–Cervia; ISPRA—Servizio Geologico d’Italia: Roma, Italy, 2005. [Google Scholar]
- Rossi, V.; Vaiani, S.C. Benthic foraminiferal evidence of sediment supply changes and fluvial drainage reorganization in Holocene deposits of the Po Delta, Italy. Mar. Micropaleontol. 2008, 69, 106–118. [Google Scholar] [CrossRef]
- Ellis, B.F.; Messina, A.R. Catalogue of Foraminifera; Micropaleontology Press: New York, NY, USA, 1940. [Google Scholar]
- Ellis, B.F.; Messina, A.R. Catalogue of Ostracoda; The American Museum of Natural History Special Publications: New York, NY, USA, 1952. [Google Scholar]
- Boomer, I.; Horne, D.J.; Slipper, I. The use of ostracods in palaeoenvironmental studies or what can you do with an Ostracod shell? Paleontol. Soc. Pap. 2003, 9, 153–179. [Google Scholar] [CrossRef]
- Chao, A.; Shen, T.-J. Nonparametric estimation of Shannon’s index of diversity when there are unseen species in sample. Environ. Ecol. Stat. 2003, 10, 429–443. [Google Scholar] [CrossRef]
- Shannon, C.E.; Weaver, W.W. The Mathematical Theory of Communications; University of Illinois Press: Urbana, IL, USA, 1963. [Google Scholar]
- Hess, S.; Alve, E.; Andersen, T.J.; Joranger, T. Defining ecological reference conditions in naturally stressed environments—How difficult is it? Mar. Environ. Res. 2020, 156, 104885. [Google Scholar] [CrossRef] [PubMed]
- Bergamin, L.; Romano, E. Suitable sediment fraction for paleoenvironmental reconstruction and assessment of contaminated coastal areas based on benthic foraminifera: A case study from Augusta Harbour (Eastern Sicily, Italy). Ecol. Indic. 2016, 71, 66–78. [Google Scholar] [CrossRef]
- Gliozzi, E.; Grossi, F. Late Messinian lago-mare ostracod palaeoecology: A correspondence analysis approach. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2008, 264, 288–295. [Google Scholar] [CrossRef]
- Patzkowsky, M.E.; Holland, S.M. Stratigraphic Paleobiology: Understanding the Distribution of Fossil Taxa in Time and Space; University of Chicago Press: Chicago, IL, USA, 2012. [Google Scholar]
- Francescangeli, F.; Portela, M.; Armynot du Chatelet, E.; Billon, G.; Andersen, T.J.; Bouchet, V.M.P.; Trentesaux, A. Infilling of the Canche Estuary (eastern English Channel, France): Insight from benthic foraminifera and historical pictures. Mar. Micropaleontol. 2018, 142, 1–12. [Google Scholar] [CrossRef]
- The R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Oksanen, J.; Blanchet, G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.5-6 2019. Available online: https://rdrr.io/cran/vegan/ (accessed on 10 May 2020).
- Meisch, C. Freshwater Ostracoda of Western and Central Europe (Süsswasserfauna von Mitteleuropa); Spektrum Akademischer Verlag: Heidelberg, Germany, 2000; Volume 3. [Google Scholar]
- Mazzini, I.; Anadon, P.; Barbieri, M.; Castorina, F.; Ferreli, L.; Gliozzi, E.; Mola, M.; Vittori, E. Late Quaternary sea-level changes along the Tyrrhenian coast near Orbetello (Tuscany, central Italy): Palaeoenvironmental reconstruction using ostracods. Mar. Micropaleontol. 1999, 37, 289–311. [Google Scholar] [CrossRef]
- Frenzel, P.; Keyser, D.; Viehberg, F.A. An illustrated key and (palaeo)ecological primer for Postglacial to Recent Ostracoda (Crustacea) of the Baltic Sea. Boreas 2010, 39, 567–575. [Google Scholar] [CrossRef]
- Salel, T.; Bruneton, H.; Lefèvre, D. Ostracods and environmental variability in lagoons and deltas along the north-western Mediterranean coast (Gulf of Lions, France and Ebro delta, Spain). Rev. Micropaléontol. 2016, 59, 425–444. [Google Scholar] [CrossRef]
- Bronk Ramsey, C. Bayesian Analysis of Radiocarbon Dates. Radiocarbon 2009, 51, 337–360. [Google Scholar] [CrossRef]
- Langone, L.; Asioli, A.; Correggiari, A.; Trincardi, F. Age-depth modelling through the late Quaternary deposits of the central Adriatic basin. Mem. Ist. Ital. Idrobiol. 1996, 55, 177–196. [Google Scholar]
- Coccioni, R. Benthic foraminifera as bioindicators of heavy metal pollution—A case study from the Goro Lagoon (Italy). In Environmental Micropaleontology: The Application of Microfossils to Environmental Geology; Martin, R.E., Ed.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2000; pp. 71–103. [Google Scholar]
- Debenay, J.-P.; Guillou, J.-J. Ecological transitions indicated by foraminiferal assemblages in paralic environments. Estuaries 2002, 25, 1107–1120. [Google Scholar] [CrossRef]
- Mojtahid, M.; Geslin, E.; Coynel, A.; Gorse, L.; Vella, C.; Davranche, A.; Zozzolo, L.; Blanchet, L.; Bénéteau, E.; Maillet, G. Spatial distribution of living (Rose Bengal stained) benthic foraminifera in the Loire estuary (western France). J. Sea Res. 2016, 118, 1–16. [Google Scholar] [CrossRef]
- Debenay, J.-P. Distribution trends of foraminiferal assemblages in paralic environments: A base for using foraminifera as bioindicators. In Environmental Micropaleontology: The Application of Microfossils to Environmental Geology; Martin, R.E., Ed.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2000; pp. 39–67. [Google Scholar]
- Melis, R.; Covelli, S. Distribution and morphological abnormalities of recent foraminifera in the Marano and Grado Lagoon (North Adriatic Sea, Italy). Medit. Mar. Sci. 2013, 14, 432–450. [Google Scholar] [CrossRef]
- Wukovits, J.; Bukenberger, P.; Enge, A.J.; Gerg, M.; Wanek, W.; Watzka, M.; Heinz, P. Food supply and size class depending variations in phytodetritus intake in the benthic foraminifer Ammonia tepida. Biol. Open 2018, 7, bio030056. [Google Scholar] [CrossRef]
- Vesper, B. Zur Morphologie und Ökologie von Cyprideis Torosa (Jones, 1850) (Crustacea, Ostracoda, Cytheridae) unter Besonderer Berücksichtigung Seiner Biometrie; Mitteilungen Hamburger Zoologisches Museum und Institut: Hamburg, Germany, 1972; pp. 21–77. [Google Scholar]
- Athersuch, J.; Horne, D.J.; Whittaker, J.E. Marine and Brackish Water Ostracods; Kermack, D.M., Barnes, R.S.K., Eds.; Synopses of the British Fauna (New Series); Brill E. J.: Leiden, The Netherlands, 1989. [Google Scholar]
- Arndt, S.; Jørgensen, B.B.; LaRowe, D.E.; Middelburg, J.J.; Pancost, R.D.; Regnier, P. Quantifying the degradation of organic matter in marine sediments: A review and synthesis. Earth Sci. Rev. 2013, 123, 53–86. [Google Scholar] [CrossRef]
- Pitacco, V.; Reizopoulou, S.; Sfriso, A.; Sfriso, A.; Mistri, M.; Munari, C. The difficulty of disentangling natural from anthropogenic forcing factors makes the evaluation of ecological quality problematic: A case study from Adriatic lagoons. Mar. Environ. Res. 2019, 150, 104756. [Google Scholar] [CrossRef]
- Pérez-Asensio, J.N.; Rodríguez-Ramírez, A. Benthic foraminiferal salinity index in marginal-marine environments: A case study from the Holocene Guadalquivir estuary, SW Spain. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2020, 560, 110021. [Google Scholar] [CrossRef]
- Carboni, M.G.; Succi, M.C.; Bergamin, L.; Bella, L.D.; Frezza, V.; Landini, B. Benthic foraminifera from two coastal lakes of southern Latium (Italy). Preliminary evaluation of environmental quality. Mar. Pollut. Bull. 2009, 59, 268–280. [Google Scholar] [CrossRef]
- Jorissen, F.J. Benthic Foraminifera from the Adriatic Sea; Principles of Phenotypic Variation; Utrecht Micropaleontological Bulletins: Utrecht, The Netherlands, 1988; Volume 37. [Google Scholar]
- Di Bella, L.; Bellotti, P.; Frezza, V.; Bergamin, L.; Carboni, M.G. Benthic foraminiferal assemblages of the imperial harbor of Claudius (Rome): Further paleoenvironmental and geoarcheological evidences. Holocene 2011, 21, 1245–1259. [Google Scholar] [CrossRef]
- Barbieri, G.; Rossi, V.; Vaiani, S.C.; Horton, B.P. Benthic Ostracoda and foraminifera from the North Adriatic Sea (Italy, Mediterranean Sea): A proxy for the depositional characterisation of river-influenced shelves. Mar. Micropaleontol. 2019, 153, 101772. [Google Scholar] [CrossRef]
- Montenegro, M.E.; Pugliese, N. Autecological remarks on the ostracod distribution in the Marano and Grado Lagoons (Northern Adriatic Sea, Italy). Boll. Soc. Paleontol. Ital. 1996, 3, 123–132. [Google Scholar]
- Tomašových, A.; Kidwell, S.M. Predicting the effects of increasing temporal scale on species composition, diversity, and rank-abundance distributions. Paleobiology 2010, 36, 672–695. [Google Scholar] [CrossRef]
- Johnson, R.G. Models and methods for analysis of the mode of formation of fossil assemblages. Bull. Geol. Soc. Am. 1960, 71, 1075–1086. [Google Scholar] [CrossRef]
- Davies, D.J.; Powell, E.N.; Stanton, R.J. Relative rates of shell dissolution and net sediment accumulation—A commentary: Can shell beds form by the gradual accumulation of biogenic debris on the sea floor? Lethaia 1989, 22, 207–212. [Google Scholar] [CrossRef]
- Martin, R.E. Taphonomy and temporal resolution of foraminiferal assemblages. In Modern Foraminifera; Sen Gupta, B.K., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999; pp. 281–298. [Google Scholar]
- Martin, R.E.; Harris, M.S.; Liddell, W.D. Taphonomy and time-averaging of foraminiferal assemblages in Holocene tidal flat sediments, Bahia la Choya, Sonora, Mexico (northern Gulf of California). Mar. Micropaleontol. 1995, 26, 187–206. [Google Scholar] [CrossRef]
- Kowalewski, M. The youngest fossil record and conservation paleobiology: Holocene shells as eco-environmental recorders. In Conservation Paleobiology: Using the Past to Manage for the Future, Paleontological Society Short Course, Portland, OR, USA, 17 October 2009; Dietl, G.P., Flessa, K.W., Eds.; The Paleontological Society: New York, NY, USA, 2009; Volume 15, pp. 1–25. [Google Scholar]
- Horton, B.P.; Edwards, R.J. Quantifying Holocene Sea-Level Change Using Intertidal Foraminifera: Lessons from the British Isles; Special Publication No. 40.; Cushman Foundation for Foraminiferal Research: Lawrence, KS, USA, 2006. [Google Scholar]
- Levinton, J.S. The paleoecological significance of oppurtunistic species. Lethaia 1970, 3, 69–78. [Google Scholar] [CrossRef]
- Uffenorde, H. Ökologie und jahreszeitliche Verteilung rezenter benthonischer Ostracoden des Limski kanal bei Rovinj (nördliche Adria). Göttinger Arb. Geol. Paläont. 1972, 13, 142. [Google Scholar]
- Mazzini, I.; Rossi, V.; Da Prato, S.; Ruscito, V. Ostracods in archaeological sites along the Mediterranean coastlines: Three case studies from the Italian peninsula. In The Archaeological and Forensic Applications of Microfossils: A Deeper Understanding of Human History; Williams, M., Hill, T., Boomer, I., Wilkinson, I.P., Eds.; The Micropalaeontological Society Special Publications; Geological Society: London, UK, 2017; pp. 121–142. [Google Scholar]
- Gobert, S. Freshwater Ostracods as Palaeoevironmental Proxies in the Moervaart Depression, a Palaeolake in Sandy Flanders (NW Belgium). Master’s Thesis, University of Ghent, Ghent, Belgium, 2012. [Google Scholar]
- Ruiz, F.; González-Regalado, M.L.; Baceta, J.I.; Menegazzo-Vitturi, L.; Pistolato, M.; Rampazzo, G.; Molinaroli, E. Los ostrácodos actualesde la laguna de Venecia (NE de Italia). Geobios 2000, 33, 447–454. [Google Scholar] [CrossRef]
| Core | Core Depth | Material | Conventional Age | Calibrated 2σ Age | Calibrated Age Mean (yr BP) | σ | Dating Laboratory | Reference |
|---|---|---|---|---|---|---|---|---|
| 240 S5 | 9.5 | Organic clay | 5290 ± 100 | 6300–5770 | 6080 | 110 | ENEA (Bologna, Italy) | [62] |
| 240 S5 | 12.9 | Organic clay | 6770 ± 105 | 7600–7230 | 7410 | 90 | “ | “ |
| 240 S5 | 18.5 | Organic clay | 10650 ± 130 | 12770–12140 | 12540 | 150 | “ | “ |
| 240 S5 | 23.4 | Organic clay | 12500 ± 220 | 15470–13960 | 14690 | 390 | “ | “ |
| 240 S5 | 31.6 | Organic clay | 19050 ± 230 | 23520–22450 | 22980 | 280 | “ | “ |
| 240 S6 | 21.55 | Mollusk shell | 7384 ± 45 | 8037–7856 | 7958 | 45 | CEDAD (Lecce, Italy) | [54] |
| 240 S6 | 23.6 | Mollusk shell | 7358 ± 45 | 8013–7843 | 7932 | 45 | “ | “ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbieri, G.; Rossi, V.; Ghosh, A.; Vaiani, S.C. Conservation Paleobiology as a Tool to Define Reference Conditions in Naturally Stressed Transitional Settings: Micropaleontological Insights from the Holocene of the Po Coastal Plain (Italy). Water 2020, 12, 3420. https://doi.org/10.3390/w12123420
Barbieri G, Rossi V, Ghosh A, Vaiani SC. Conservation Paleobiology as a Tool to Define Reference Conditions in Naturally Stressed Transitional Settings: Micropaleontological Insights from the Holocene of the Po Coastal Plain (Italy). Water. 2020; 12(12):3420. https://doi.org/10.3390/w12123420
Chicago/Turabian StyleBarbieri, Giulia, Veronica Rossi, Anupam Ghosh, and Stefano Claudio Vaiani. 2020. "Conservation Paleobiology as a Tool to Define Reference Conditions in Naturally Stressed Transitional Settings: Micropaleontological Insights from the Holocene of the Po Coastal Plain (Italy)" Water 12, no. 12: 3420. https://doi.org/10.3390/w12123420
APA StyleBarbieri, G., Rossi, V., Ghosh, A., & Vaiani, S. C. (2020). Conservation Paleobiology as a Tool to Define Reference Conditions in Naturally Stressed Transitional Settings: Micropaleontological Insights from the Holocene of the Po Coastal Plain (Italy). Water, 12(12), 3420. https://doi.org/10.3390/w12123420






