Spatio-Temporal Variations of Hydrochemical and Microbial Characteristics in Karst Water in Samcheok, South Korea
Abstract
:1. Introduction
2. Study Areas and Methods
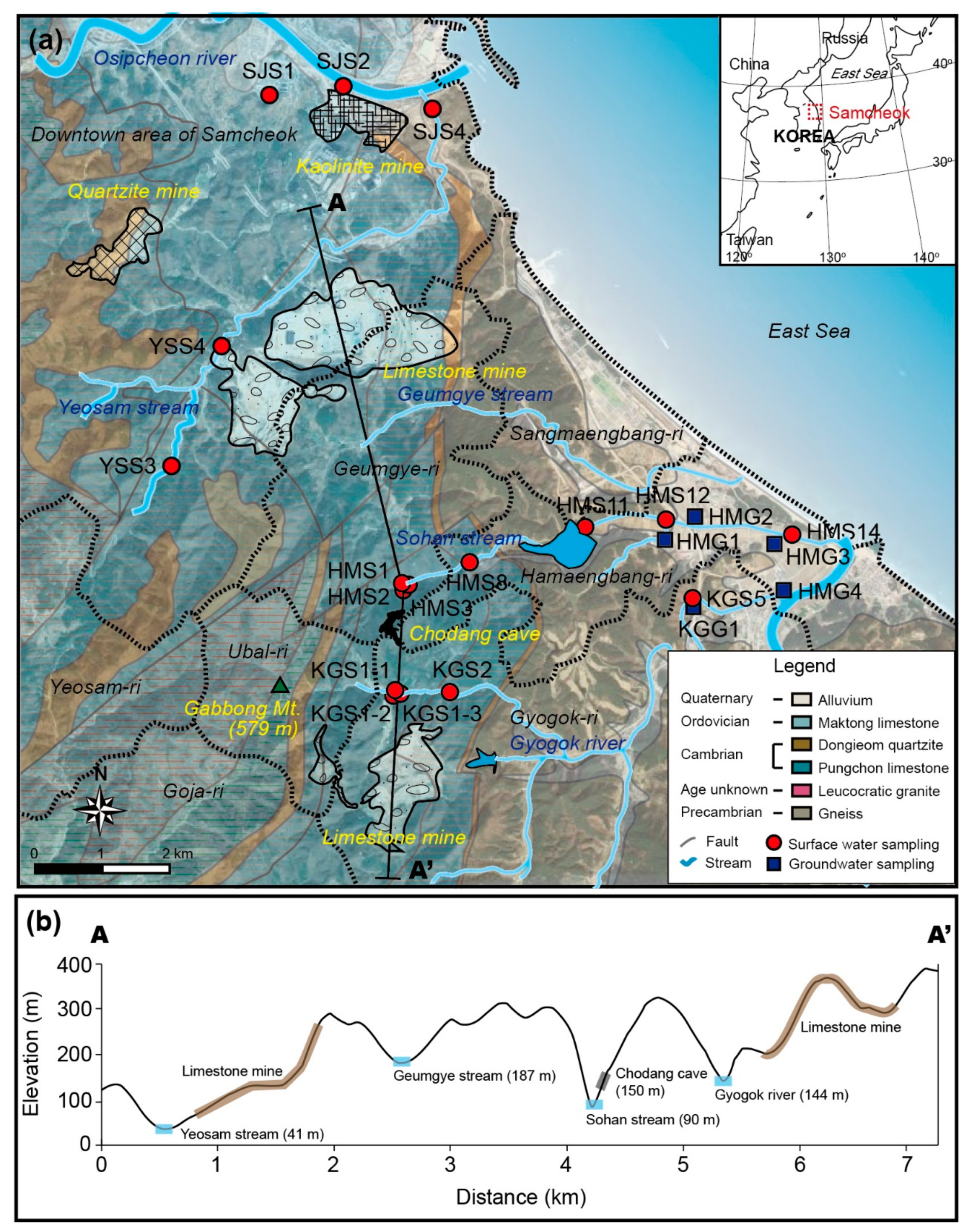
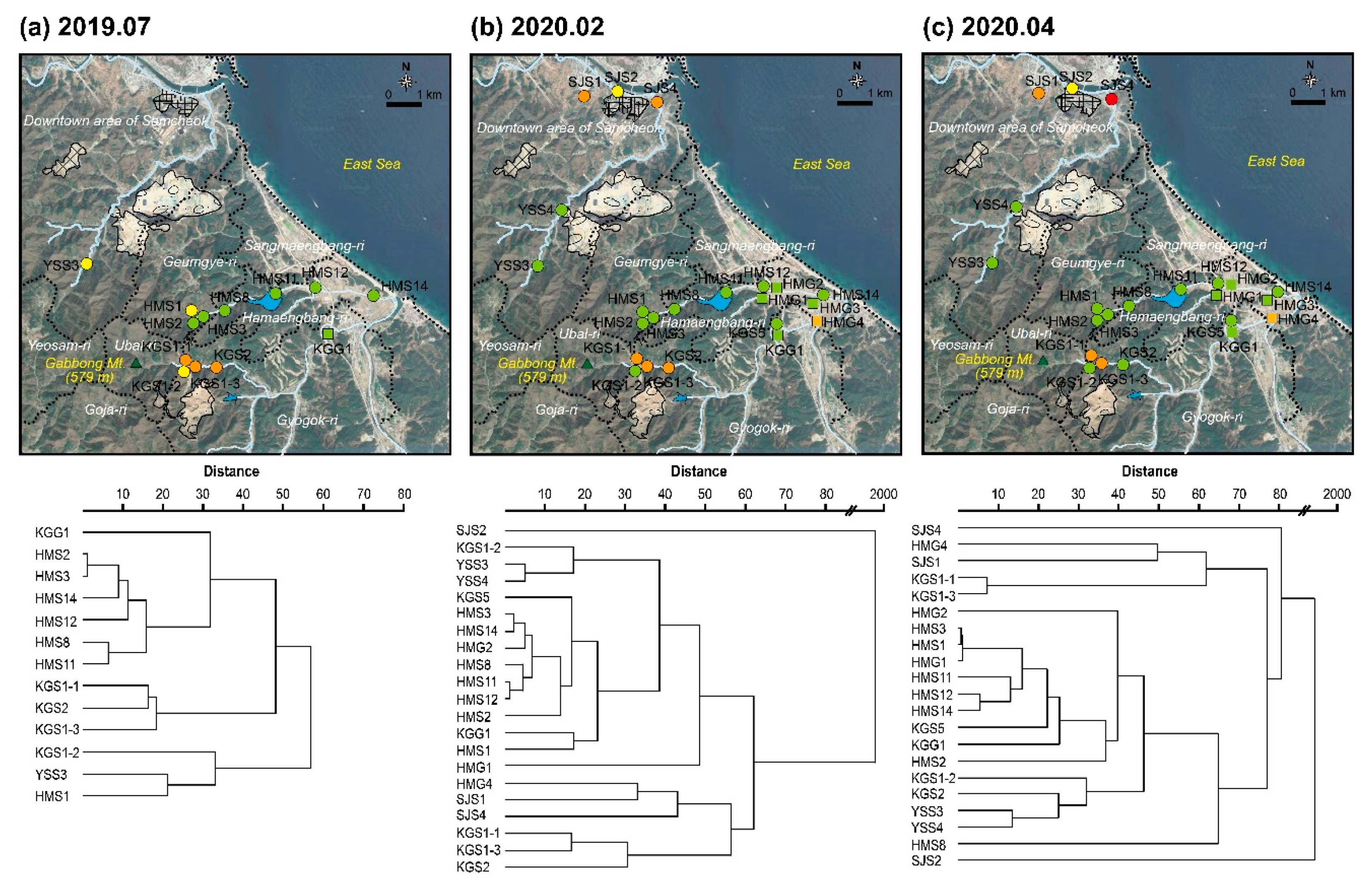
2.1. Study Areas
2.2. Methods
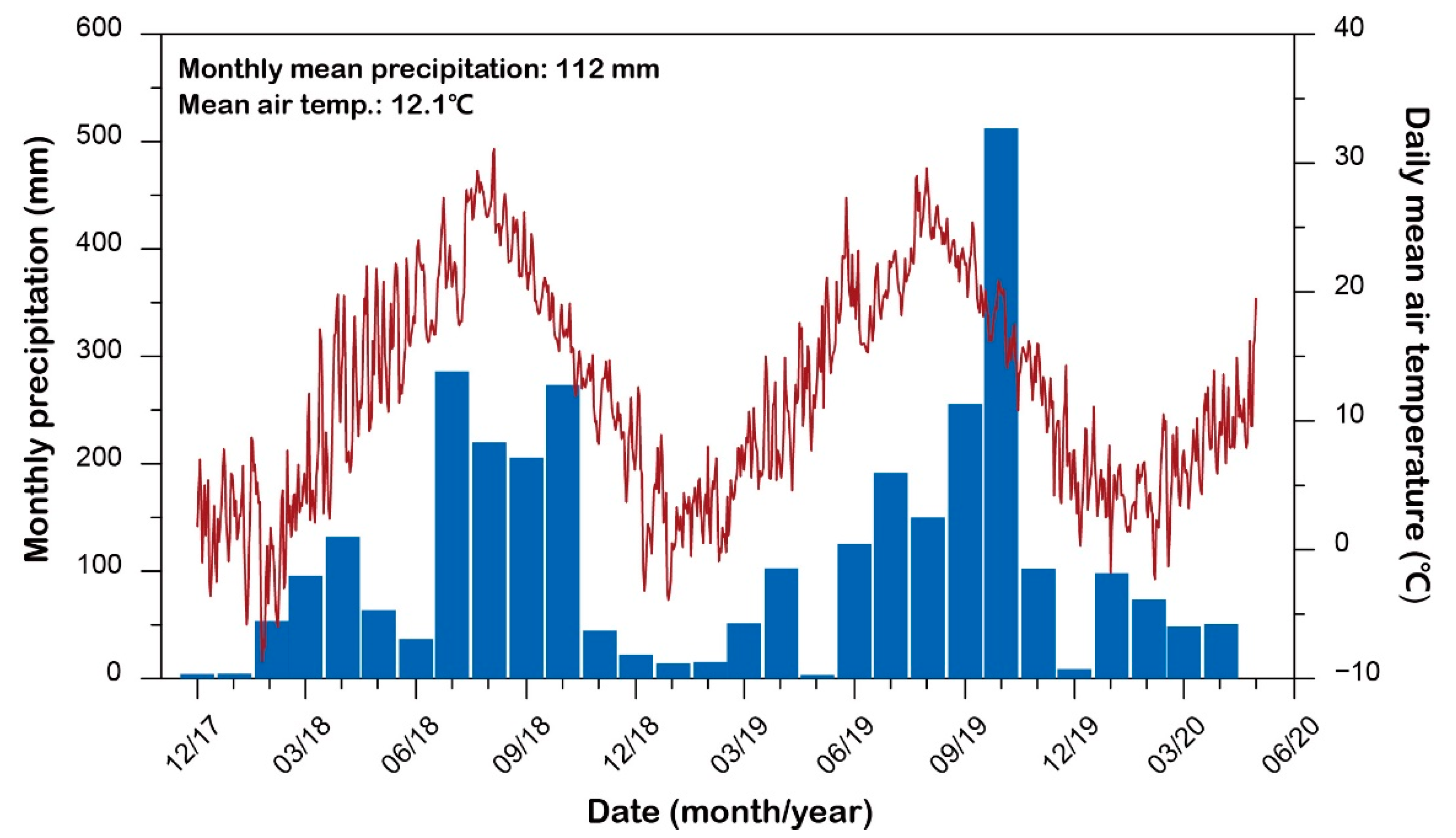
2.2.1. Field Survey
2.2.2. Cation/Anion Analysis
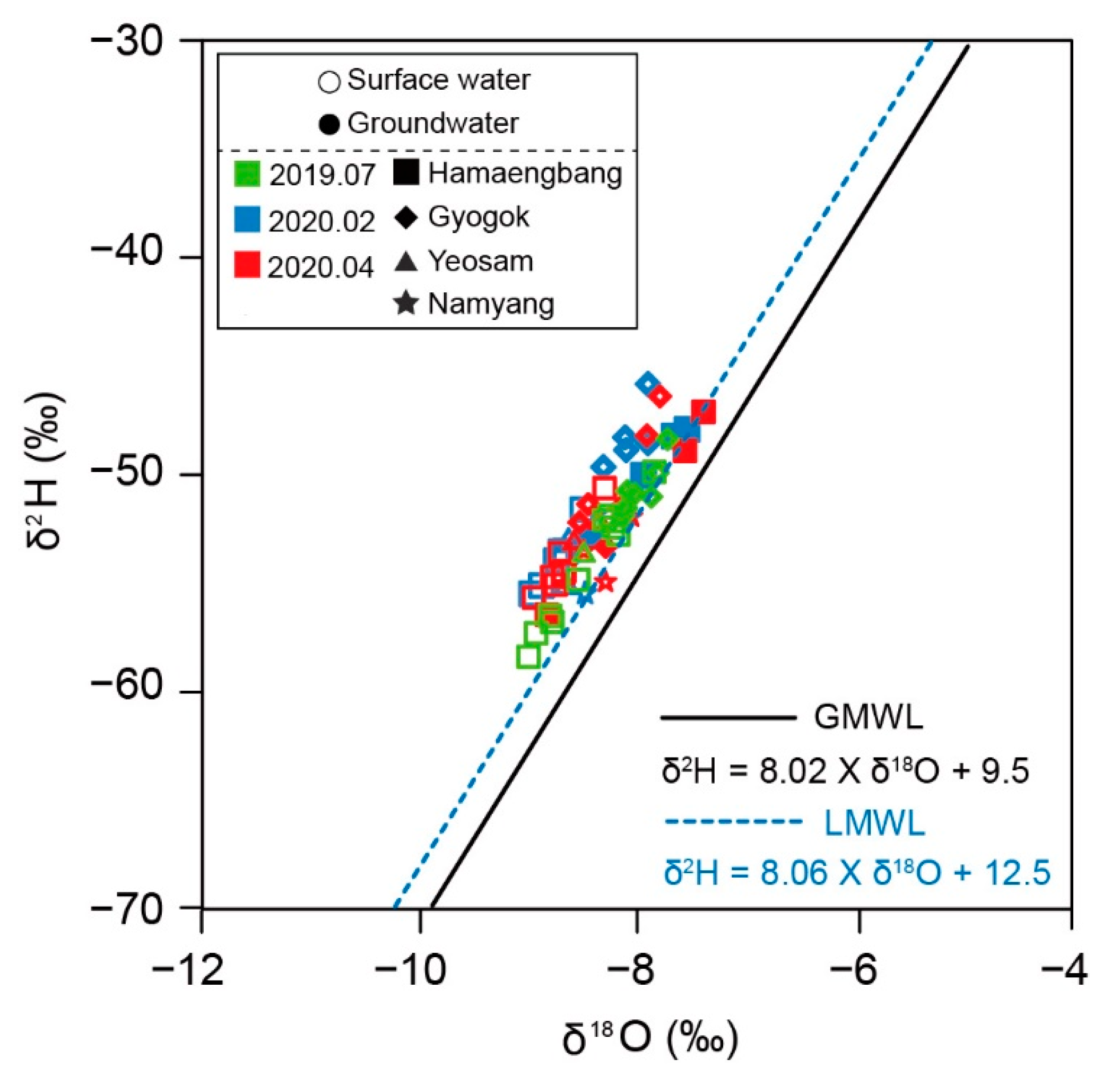
2.2.3. Stable Isotope Analysis
2.2.4. Precipitated Matter Analysis (SEM, XRD)
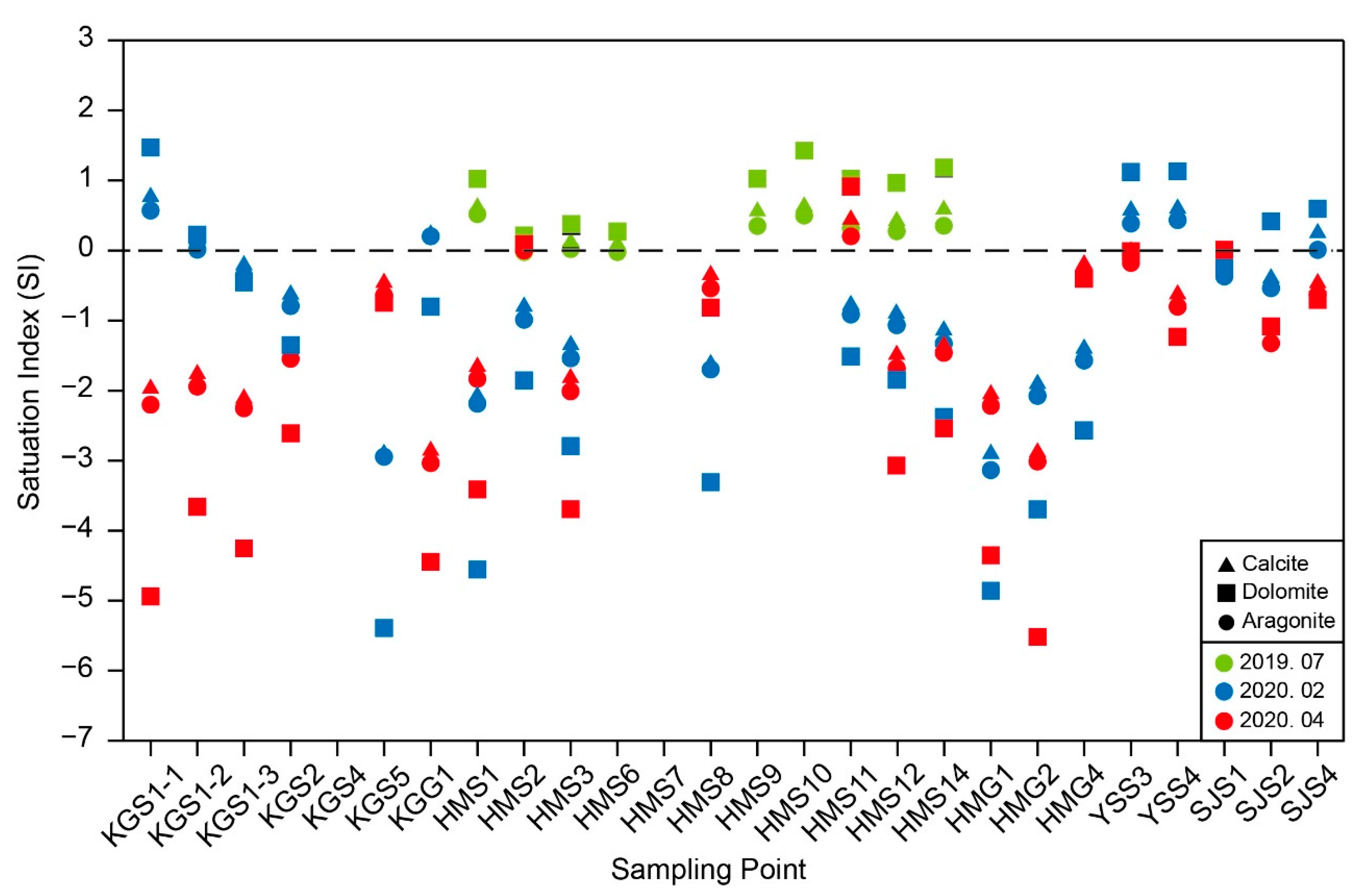
2.2.5. Saturation Index (SI)
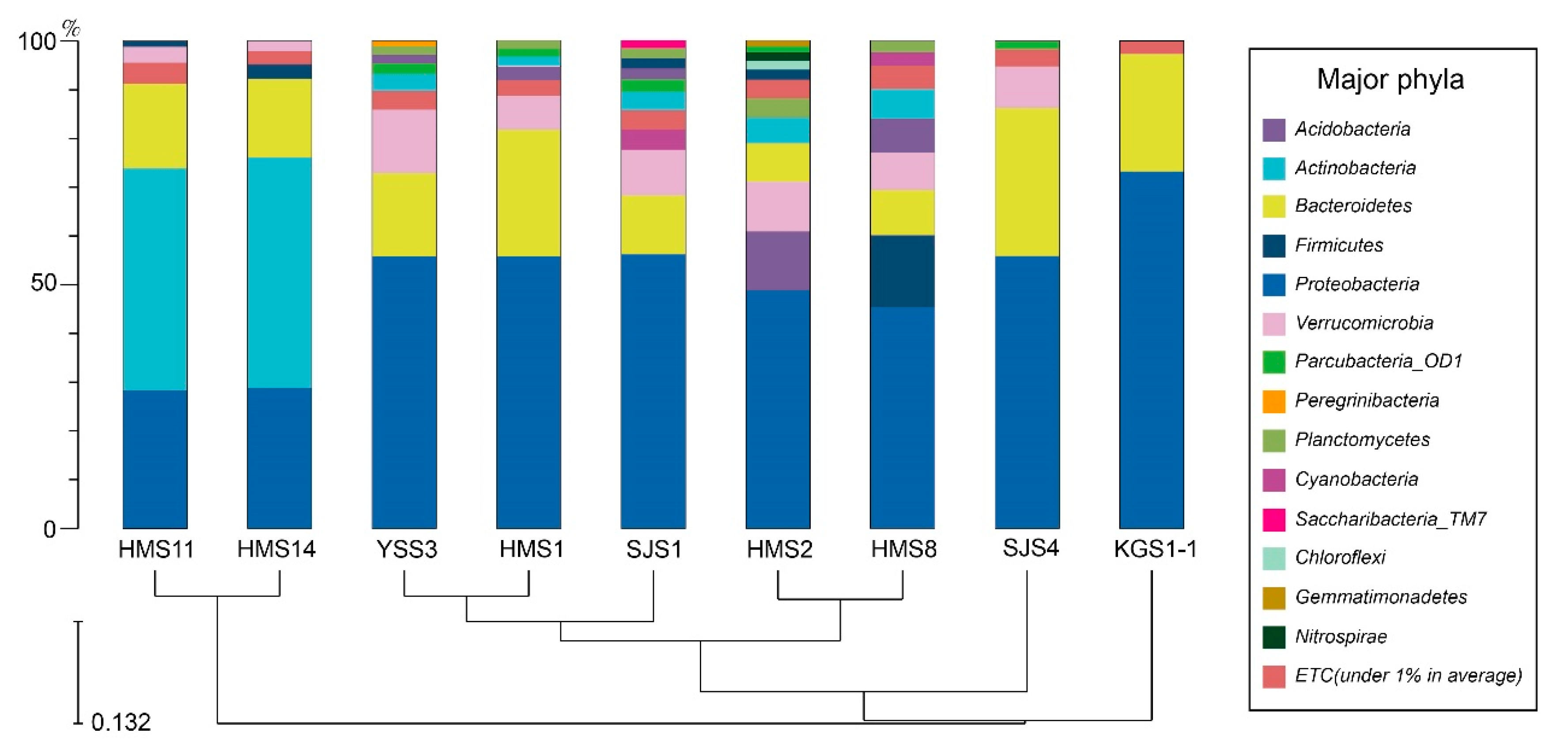
2.2.6. Microbial Cluster Analysis
2.2.7. Laboratory Analysis
3. Results and Discussion
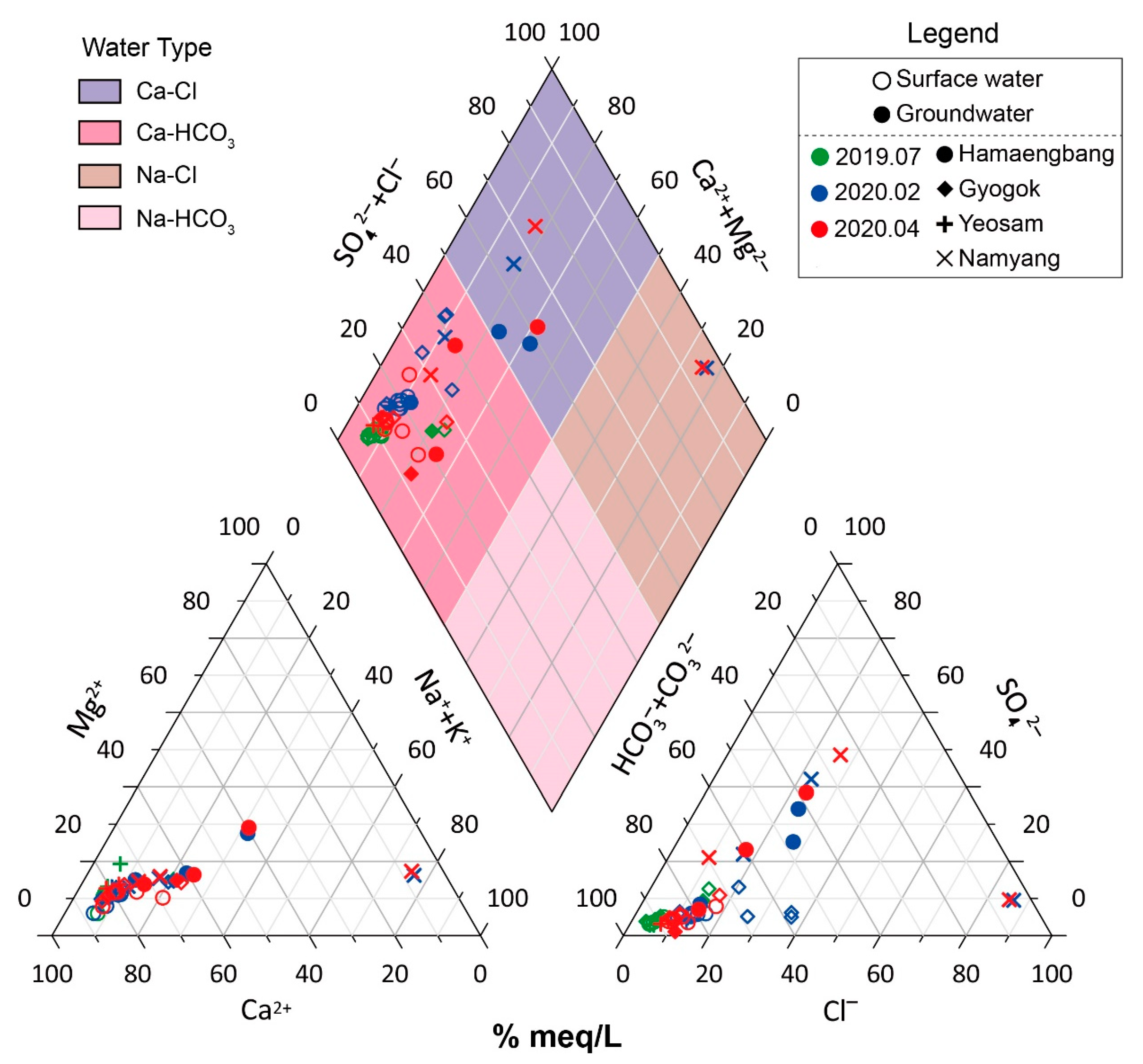
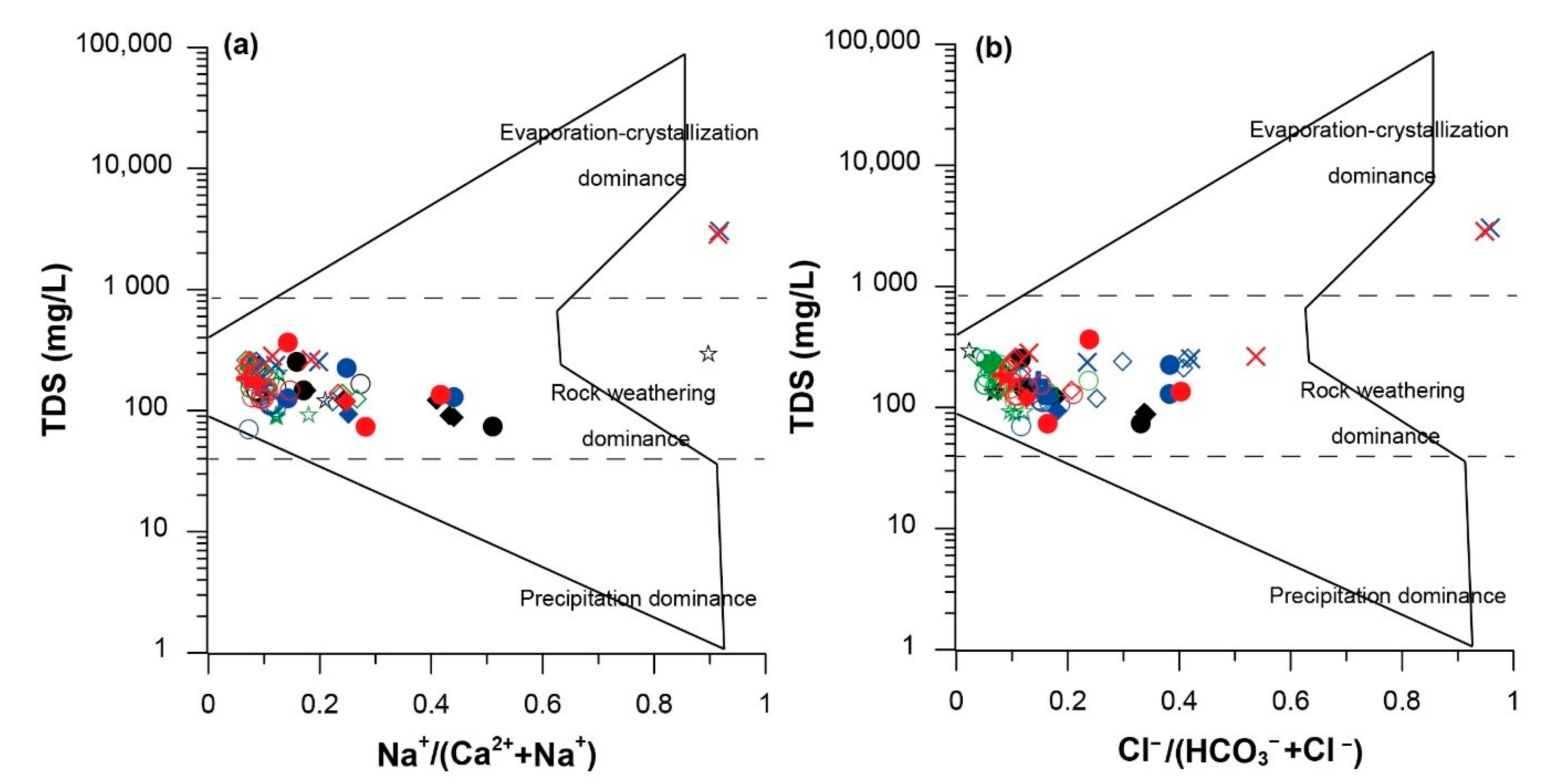
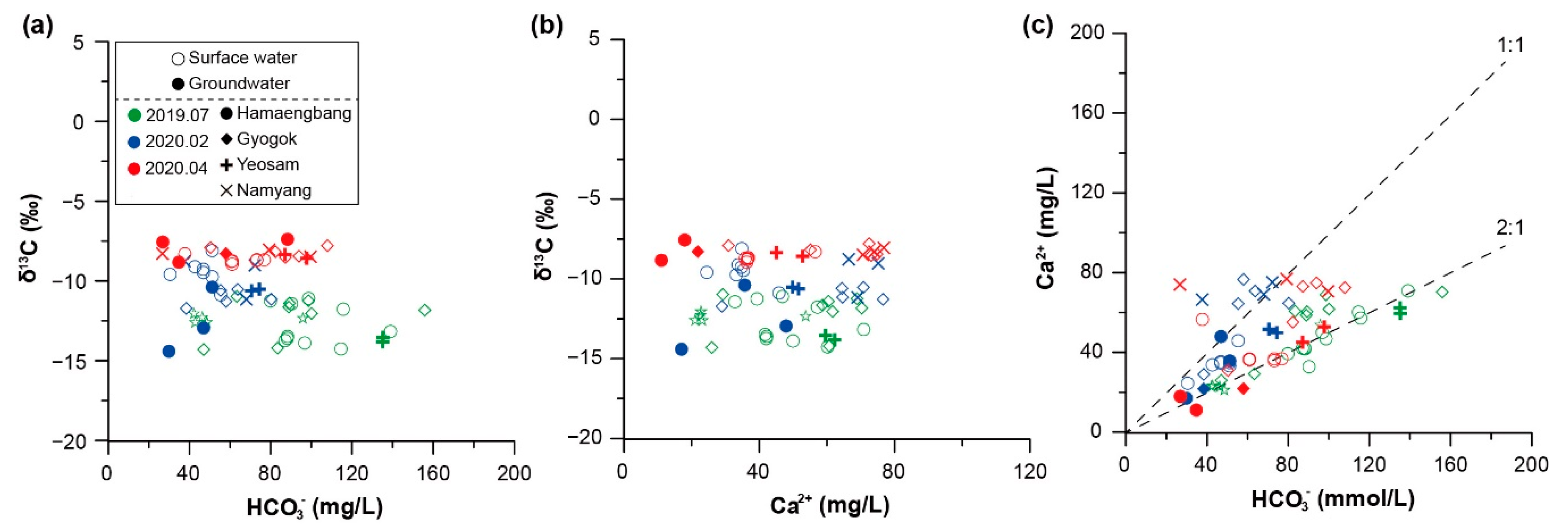
3.1. Hydrochemical Characteristics of Karst Water (Groundwater and Surface Water)
3.2. Seasonal Hydrochemical Characteristics
3.3. Regional Characteristics
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kim, H. Comparison of PCR primers for analyzing denitrifying microorganisms in the hyporheic zone. Appl. Sci. 2020, 10, 4172. [Google Scholar] [CrossRef]
- Panno, S.V.; Kelly, W.R.; Scott, J.; Zheng, W.; McNeish, R.E.; Holm, N.; Hoellein, T.J.; Baranski, E. Microplastic contamination in karst groundwater systems. Groundwater 2009, 57, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Ford, D.C.; Williams, P.W. Karst Geomorphology and Hydrology; DCFordSpringer: London, UK, 1989; p. 601. [Google Scholar]
- Michel, B. Karst groundwater: A challenge for new resources. Hydrogeol. J. 2005, 13, 148–160. [Google Scholar]
- Stevanovic, Z. Karst waters in potable water supply: A global scale overview. Environ. Earth Sci. 2019, 78, 662. [Google Scholar] [CrossRef]
- Goldscheider, N.; Drew, D. Methods in Karst Hydrogeology; Taylor & Francis: London, UK, 2007; pp. 1–9. [Google Scholar]
- Langer, W.H. Potential Environmental Impacts of Quarrying Stone in Karst—A Literature Review; USGS: Reston, VA, USA, 2001; pp. 1–35.
- Liu, Z.; Li, Q.; Sun, H.; Wang, J. Seasonal, diurnal and storm-scale hydrochemical variations of typical epikarst springs in subtropical karst areas of SW China: Soil CO2 and dilution effects. J. Hydrol. 2007, 337, 207–223. [Google Scholar] [CrossRef]
- Liu, Z.; Zhao, J. Contribution of carbonate rock weathering to the atmospheric CO2 sink. Environ. Geol. 2000, 39, 1053–1058. [Google Scholar] [CrossRef]
- Yoshimura, K.; Nakao, S.; Noto, M.; Inokura, Y.; Urata, K.; Chen, M.; Lin, P.W. Geochemical and stable isotope studies on natural water in the Taroko Gorge karst area, Taiwan-chemical weathering of carbonate rocks by deep source CO2 and sulfuric acid. Chem. Geol. 2001, 177, 415–430. [Google Scholar] [CrossRef]
- Ryu, H.S.; Lee, J.Y.; Lim, C.W.; Kim, K. Hydrochemical characteristics of groundwater and stream water in a karst area of Samcheok. Korea. J. Geol. Soc. Korea 2019, 55, 117–129. [Google Scholar] [CrossRef]
- Klimchouk, A. Hypogene Karst Regions and Caves of the World; Springer: Berlin, Germany, 2017; pp. 1–40. [Google Scholar]
- Assaad, F.A.; Jordan, H. Karst terranes and environmental aspects. Environ. Geol. 1994, 23, 228–237. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Z.; Cu, M.; Deng, C.; Liu, M.; Li, L. Spatial and vertical distribution and pollution assessment of soil fluorine in a lead-zinc mining area in the Karst region of Guangxi, China. Plant Soil Environ. 2010, 56, 282–287. [Google Scholar] [CrossRef] [Green Version]
- Xu, K.; Dai, G.; Duan, Z.; Xue, X. Hydrogeochemical evolution of an Ordovician limestone aquifer influenced by coal mining: A case study in the Hancheng mining area, China. Mine Water Environ. 2018, 37, 238–248. [Google Scholar] [CrossRef]
- Kim, H.; Jang, J.; Park, S.; Kim, C.S.; Ryu, H.S.; Lee, J.Y. Spatial variations in microbial compositions in a karst critical zone in Samcheok, Republic of Korea. Appl. Sci. 2020, 10, 4714. [Google Scholar] [CrossRef]
- Park, Y.; Lee, J.Y.; Lim, H.G.; Park, Y.C. Characteristics of hydraulic conductivity of carbonate aquifers in Gangwon Province. J. Eng. Geol. 2011, 21, 79–85. [Google Scholar] [CrossRef]
- Lee, J.Y.; Raza, M.; Park, Y.C. Current status and management for the sustainable groundwater resources in Korea. Episodes 2018, 41, 179–191. [Google Scholar] [CrossRef] [Green Version]
- ME (Ministry of Environment). K-Water Report of Groundwater Basic Survey in Donghae and Samcheok Area; K-Water: Gwacheon, Korea, 2018; pp. 6–80. [Google Scholar]
- Woo, K.S.; Won, C.K.; Lee, K.C.; Choi, Y.K. Comprehensive Investigation Report of Chodang Cave; Samcheok City: Samcheok, Korea, 2000; pp. 11–85. [Google Scholar]
- Won, J.K.; Park, B.K.; Lee, S.H. Geologic Report on Samcheock-Gosari Sheet; KIGAM: Daejeon, Korea, 1994; pp. 1–36. [Google Scholar]
- Beta Analytic. Available online: https://www.radiocarbon.com/ (accessed on 21 October 2020).
- Park, Y.; Kwon, K.S.; Kim, N.; Lee, J.Y.; Yoon, J.G. Change of geochemical properties of groundwater by use of open loop geothermal cooling and heating system. J. Geol. Soc. Korea 2013, 49, 289–296. [Google Scholar]
- Mothur. Available online: https://mothur.org/ (accessed on 25 October 2020).
- Kim, H.; Lee, K.K. Effect of vertical flow exchange on microbial community distributions in hyporheic zones. Episodes 2019, 42, 1–19. [Google Scholar] [CrossRef]
- Guseva, N. The origin of the natural water chemical composition in the permafrost region of the eastern slope of the Polar Urals. Water 2016, 8, 594. [Google Scholar] [CrossRef]
- Das, N.; Patel, A.K.; Deka, G.; Das, A.; Sarma, K.P.; Kumar, M. Geochemical controls and future perspective of arsenic mobilization for sustainable groundwater management: A study from Northeast India. Groundw. Sustain. Dev. 2015, 1, 92–104. [Google Scholar] [CrossRef]
- Barzegar, R.; Moghaddam, A.A.; Nazemi, A.H.; Adamowski, J. Evidence for the occurrence of hydrogeochemical processes in the groundwater of Khoy plain, northwestern Iran, using ionic ratios and geochemical modeling. Environ. Earth Sci. 2018, 77, 597. [Google Scholar] [CrossRef]
- Sampyo, K.N.U. Groundwater Impact Investigation Report (1st Stage) on High Quality Limestone Development of Dongyang 2nd Mine; Sampyo: Samcheok, Korea, 2017; pp. 42–47. [Google Scholar]
- Xu, P.; Feng, W.; Qian, H.; Zhang, Q. Hydrogeochemical characterization and irrigation quality assessment of shallow groundwater in the central-western Guanzhong basin, China. J. Environ. Res. Public Health 2019, 16, 1492. [Google Scholar] [CrossRef] [Green Version]
- Gao, X.; Li, X.; Wang, W.; Li, C. Human activity and hydrogeochemical processes relating to groundwater quality degradation in the Yuncheng basin, northern China. J. Environ. Res. Public Health 2020, 17, 867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grobe, M.; Machel, H.G.; Heuser, H. Origin and evolution of saline groundwater in the Munsterland Cretaceous basin, Germany: Oxygen, hydrogen, and strontium isotope evidence. J. Geochem. Explor. 2000, 69–70, 5–9. [Google Scholar] [CrossRef]
- Brian, H. Geochemistry and origin of formation waters in the western Canada sedimentary basin-I. Stable isotopes of hydrogen and oxygen. Geochim. Cosmochim. Acta 1969, 33, 1321–1349. [Google Scholar]
- Park, Y.; Lee, K.S.; Yu, J.Y. Seasonal variations of dissolved ions and oxygen and hydrogen isotopic compositions of precipitation in Chuncheon, Korea. J. Geol. Soc. Korea 2006, 42, 283–292. [Google Scholar]
- Noh, J.H.; Oh, S.J. Hydrothermal Alteration of the Pungchon Limestone and the Formation of High Ca Limestone. J. Geol. Soc. Korea 2005, 41, 175–197. [Google Scholar]
- Kim, H.; Kaown, D.; Mayer, B.; Lee, J.Y.; Lee, K.K. Combining pyrosequencing and isotopic approaches to assess denitrification in a hyporheic zone. Sci. Total Environ. 2018, 631–632, 755–764. [Google Scholar] [CrossRef] [PubMed]

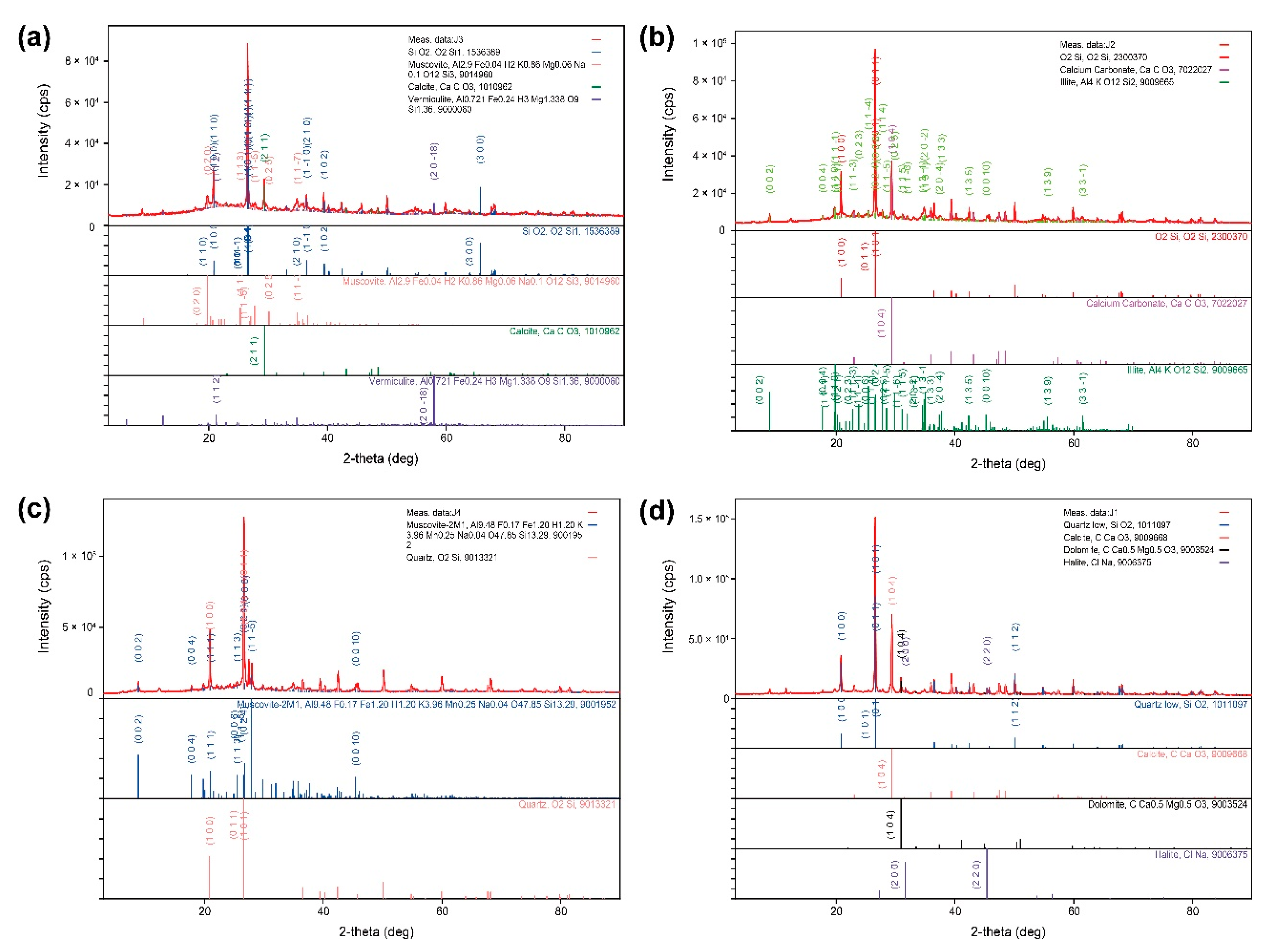
| Parameter | Hamaengbang-ri | Gyogok-ri | Yeosam-ri | Downtown Area | Groundwater | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Max. | Min. | Mean | SD * | CV. ** | Max. | Min. | Mean | SD* | CV. ** | Max. | Min. | Mean | SD * | CV. ** | Max. | Min. | Mean | SD * | CV. ** | Max. | Min. | Mean | SD * | CV. ** | |
| Temperature (°C) | 20.0 | 4.4 | 12.1 | 5.0 | 41.5 | 21.0 | 5.0 | 13.9 | 5.1 | 36.9 | 16.0 | 7.7 | 12.3 | 3.4 | 27.8 | 15.4 | 8.9 | 12.3 | 2.3 | 19.1 | 17.1 | 9.5 | 12.9 | 2.6 | 20.1 |
| pH | 8.4 | 6.5 | 7.5 | 0.7 | 9.2 | 8.6 | 5.9 | 7.4 | 1.0 | 13.7 | 8.5 | 7.2 | 8.1 | 0.6 | 7.2 | 8.3 | 6.9 | 7.7 | 0.4 | 5.8 | 7.3 | 5.5 | 6.5 | 0.6 | 9.1 |
| EC (μS/cm) | 541.0 | 183.7 | 235.2 | 76.2 | 32.4 | 504.0 | 213.0 | 340.8 | 68.5 | 20.1 | 305.0 | 255.0 | 283.8 | 18.6 | 6.5 | 4960.0 | 423.0 | 1534.8 | 1832.6 | 119.4 | 688.0 | 96.6 | 298.7 | 165.4 | 55.4 |
| Ca2+ (mg/L) | 60.2 | 24.5 | 39.6 | 8.4 | 21.1 | 76.6 | 29.0 | 62.2 | 15.0 | 24.2 | 59.6 | 45.1 | 51.8 | 5.2 | 10.1 | 76.8 | 66.4 | 72.0 | 4.0 | 5.5 | 113.2 | 11.1 | 35.1 | 31.3 | 89.3 |
| Mg2+ (mg/L) | 4.6 | 1.0 | 3.1 | 0.7 | 21.1 | 6.4 | 3.8 | 5.1 | 0.8 | 15.2 | 5.9 | 4.8 | 5.1 | 0.4 | 8.2 | 107.7 | 7.3 | 40.5 | 49.0 | 147.9 | 13.1 | 1.9 | 5.8 | 3.5 | 60.7 |
| Na+ (mg/L) | 6.3 | 1.9 | 4.3 | 0.9 | 20.5 | 9.4 | 4.8 | 6.2 | 1.3 | 21.6 | 5.4 | 3.8 | 4.7 | 0.6 | 12.8 | 837.0 | 9.2 | 285.3 | 421.9 | 147.9 | 18.9 | 4.4 | 10.5 | 4.9 | 46.6 |
| K+ (mg/L) | 13.2 | 0.3 | 1.5 | 2.7 | 181.0 | 5.3 | 0.8 | 2.3 | 1.1 | 49.0 | 2.1 | 0.7 | 1.2 | 0.6 | 48.1 | 60.5 | 3.0 | 20.1 | 236 | 117.2 | 11.5 | 0.9 | 3.7 | 3.6 | 98.3 |
| HCO3− (mg/L) | 114.6 | 30.5 | 67.8 | 22.6 | 33.3 | 155.9 | 38.4 | 81.7 | 293 | 35.8 | 135.3 | 70.5 | 93.0 | 25.9 | 27.9 | 99.6 | 26.7 | 63.9 | 27.1 | 42.4 | 88.3 | 26.8 | 48.6 | 19.4 | 39.9 |
| SO42− (mg/L) | 8.0 | 2.8 | 5.6 | 1.2 | 20.7 | 11.8 | 6.2 | 8.7 | 1.7 | 19.9 | 7.0 | 5.4 | 6.1 | 0.7 | 12.0 | 257.0 | 37.9 | 121.1 | 97.6 | 80.6 | 52.9 | 0.0 | 21.9 | 20.5 | 93.6 |
| Cl− (mg/L) | 13.2 | 4.0 | 8.0 | 2.2 | 27.6 | 41.2 | 5.5 | 15.2 | 11.6 | 76.7 | 12.9 | 7.7 | 10.1 | 2.3 | 22.3 | 1652.1 | 14.9 | 533.8 | 793.0 | 148.5 | 29.2 | 6.8 | 15.5 | 8.4 | 54.2 |
| NO3− (mg/L) | 9.5 | 3.4 | 5.4 | 1.5 | 26.8 | 55.4 | 4.8 | 31.9 | 21.6 | 67.9 | 8.2 | 5.9 | 6.9 | 1.0 | 14.6 | 19.9 | 7.4 | 14.4 | 5.2 | 36.2 | 33.4 | 0.0 | 8.2 | 12.2 | 148.7 |
| CO32− (mg/L) | 1.8 | 0.0 | 0.5 | 0.5 | 114.6 | 2.2 | 0.0 | 0.6 | 0.7 | 118.3 | 2.3 | 0.1 | 1.3 | 1.0 | 73.0 | 0.7 | 0.1 | 0.3 | 0.2 | 70.8 | 0.2 | 0.0 | 0.0 | 0.0 | 152.1 |
| δ13C (‰) | −8.1 | −14.3 | −10.3 | 2.0 | −19.6 | −7.8 | −14.2 | −10.4 | 1.9 | −18.4 | −8.4 | −13.5 | −10.3 | 2.1 | −20.2 | −8.1 | −11.2 | −9.0 | 1.1 | −12.6 | 4.0 | −14.4 | −8.5 | 5.3 | −61.7 |
| δ18O (‰) | −7.8 | −9.0 | −8.6 | 0.3 | −3.4 | −7.8 | −8.5 | −8.1 | 0.2 | −2.8 | −8.4 | −8.7 | −8.5 | 0.1 | −1.4 | −8.1 | −8.5 | −8.3 | 0.2 | −2.2 | −7.4 | −8.8 | −8.0 | 0.5 | −6.1 |
| δD (‰) | −49.9 | −57.3 | −57.3 | 1.9 | −3.5 | −45.8 | −52.2 | −49.6 | 2.0 | −4.0 | −52.0 | −55.0 | −53.3 | 1.1 | −2.0 | −51.5 | −55.5 | −53.2 | 1.7 | −3.2 | −47.1 | −56.4 | −50.7 | 3.1 | −6.1 |
| CBE *** | −3.00 | 1.29 | −4.05 | −0.11 | 3.70 | ||||||||||||||||||||
| Sampling Point | Target Reads | OTUs | ACE | CHAO | Jackknife | NPShannon | Shannon | Phylogenetic Diversity | Good’s Library Coverage (%) |
|---|---|---|---|---|---|---|---|---|---|
| SJS4 | 63,288 | 1990 | 2072.5 | 2031.7 | 2180.0 | 5.1 | 5.1 | 2617 | 99.70 |
| HMS1 | 35,085 | 3054 | 3591.9 | 3362.2 | 3786.0 | 6.4 | 6.3 | 3776 | 97.91 |
| YSS3 | 47,840 | 4082 | 4558.3 | 4299.3 | 4830.0 | 6.8 | 6.6 | 4576 | 98.44 |
| HMS14 | 17,577 | 1167 | 1486.4 | 1350.8 | 1520.0 | 4.7 | 4.6 | 1842 | 97.99 |
| SJS1 | 34,284 | 4001 | 4594.5 | 4299.8 | 4871.0 | 7.0 | 6.8 | 4969 | 97.46 |
| HMS2 | 37,307 | 5392 | 6100.3 | 5744.5 | 6487.0 | 7.6 | 7.4 | 6222 | 97.06 |
| HMS11 | 20,099 | 1359 | 1774.7 | 1583.0 | 1808.0 | 4.7 | 4.6 | 2285 | 97.77 |
| HMS8 | 33,959 | 4184 | 4918.9 | 4582.0 | 5201.0 | 6.7 | 6.5 | 5195 | 97.01 |
| KGS1–1 | 38,534 | 992 | 1117.7 | 1041.9 | 1185.0 | 3.6 | 3.6 | 1309 | 99.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryu, H.-S.; Kim, H.; Lee, J.-Y.; Jang, J.; Park, S. Spatio-Temporal Variations of Hydrochemical and Microbial Characteristics in Karst Water in Samcheok, South Korea. Water 2020, 12, 3407. https://doi.org/10.3390/w12123407
Ryu H-S, Kim H, Lee J-Y, Jang J, Park S. Spatio-Temporal Variations of Hydrochemical and Microbial Characteristics in Karst Water in Samcheok, South Korea. Water. 2020; 12(12):3407. https://doi.org/10.3390/w12123407
Chicago/Turabian StyleRyu, Han-Sun, Heejung Kim, Jin-Yong Lee, Jiwook Jang, and Sangwook Park. 2020. "Spatio-Temporal Variations of Hydrochemical and Microbial Characteristics in Karst Water in Samcheok, South Korea" Water 12, no. 12: 3407. https://doi.org/10.3390/w12123407
APA StyleRyu, H.-S., Kim, H., Lee, J.-Y., Jang, J., & Park, S. (2020). Spatio-Temporal Variations of Hydrochemical and Microbial Characteristics in Karst Water in Samcheok, South Korea. Water, 12(12), 3407. https://doi.org/10.3390/w12123407







