Measurement and Modelling of Moisture Distribution and Water Binding Energy of Dredged Sludge
Abstract
1. Introduction
2. Water Binding Energy Calculation Model
3. Materials and Methods
3.1. Sample Preparation and Characterization
3.2. LF-NMR Analysis
3.3. TG-DTA Analysis
4. Results and Discussion
4.1. Water Distribution in Dredged Sludge Detected by LF-NMR
4.2. Model Analysis
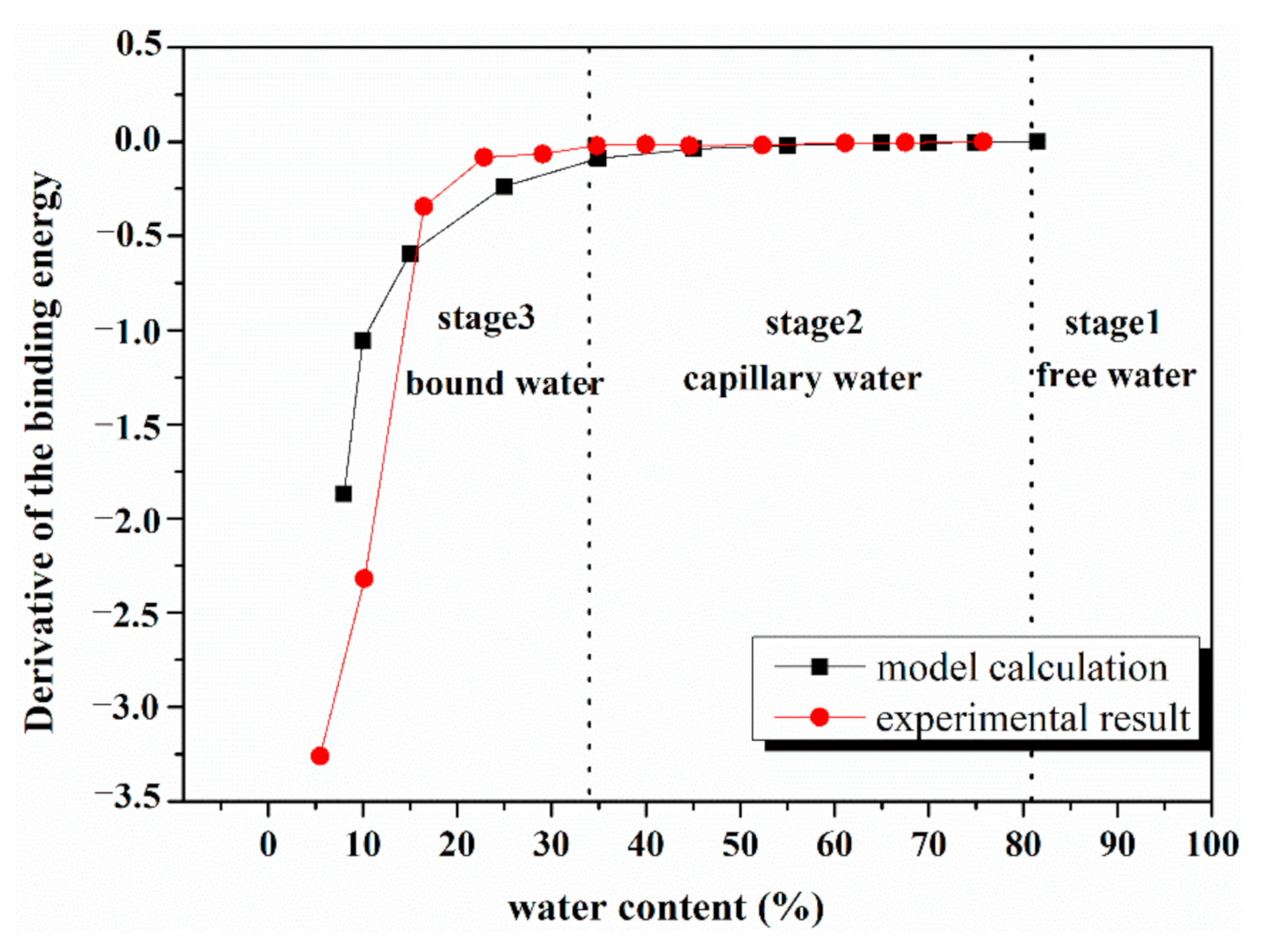
4.3. Comparison of Model Calculation and Experimental Result
4.4. Gradient of Energy Consumption Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amar, M.; Benzerzour, M.; Kleib, J.; Abriak, N.-E. From dredged sediment to supplementary cementitious material: Characterization, treatment, and reuse. Int. J. Sediment Res. 2021, 36, 92–109. [Google Scholar] [CrossRef]
- Oh, H.; Lee, J.; Banthia, N.; Talukdar, S. An Experimental Study of the Physicochemical Properties of a Cement Matrix Containing Dredged Materials. Mater. Sci. Appl. 2011, 2, 847–857. [Google Scholar] [CrossRef][Green Version]
- Bhairappanavar, S.; Liu, R.; Coffman, R. Beneficial Uses of Dredged Material in Green Infrastructure and Living Architecture to Improve Resilience of Lake Erie. Infrastructures 2018, 3, 42. [Google Scholar] [CrossRef]
- Tri, D.Q.; Kandasamy, J.; Don, N.C. Quantitative Assessment of the Environmental Impacts of Dredging and Dumping Activities at Sea. Appl. Sci. 2019, 9, 1703. [Google Scholar] [CrossRef]
- Zonta, R.; Cassin, D.; Pini, R.; Dominik, J. Substantial Decrease in Contaminant Concentrations in the Sediments of the Venice (Italy) Canal Network in the Last Two Decades—Implications for Sediment Management. Water 2020, 12, 1965. [Google Scholar] [CrossRef]
- Cappuyns, V.; Deweirt, V.; Rousseau, S. Dredged sediments as a resource for brick production: Possibilities and barriers from a consumers’ perspective. Waste Manag. 2015, 38, 372–380. [Google Scholar] [CrossRef]
- Bagarani, M.; De Vincenzo, A.; Ievoli, C.; Molino, B. The Reuse of Sediments Dredged from Artificial Reservoirs for Beach Nourishment: Technical and Economic Feasibility. Sustainability 2020, 12, 6820. [Google Scholar] [CrossRef]
- Molino, B.; Bufalo, G.; De Vincenzo, A.; Ambrosone, L. Semiempirical Model for Assessing Dewatering Process by Flocculation of Dredged Sludge in an Artificial Reservoir. Appl. Sci. 2020, 10, 3051. [Google Scholar] [CrossRef]
- Song, Z.; Zhang, W.; Gao, H.; Wang, D. Comprehensive assessment of flocculation conditioning of dredged sediment using organic polymers: Dredged sediment dewaterability and release of pollutants. Sci. Total Environ. 2020, 739, 139884. [Google Scholar] [CrossRef]
- Wang, J.; Huang, G.; Fu, H.; Cai, Y.; Hu, X.; Lou, X.; Jin, Y.; Hai, J.; Ni, J.; Zou, J. Vacuum preloading combined with multiple-flocculant treatment for dredged fill improvement. Eng. Geol. 2019, 259, 105194. [Google Scholar] [CrossRef]
- Pan, L.; Xing, J.; Luo, X.; Li, Y.; Sun, D.; Zhai, Y.; Yang, K.; Chen, Z. Influence of Electron Beam Irradiation on the Moisture and Properties of Freshly Harvested and Sun-Dried Rice. Foods 2020, 9, 1139. [Google Scholar] [CrossRef]
- Zhao, C.; Zheng, H.; Feng, L.; Wang, Y.; Liu, Y.; Liu, B.; Djibrine, B.Z. Improvement of Sludge Dewaterability by Ultrasound-Initiated Cationic Polyacrylamide with Microblock Structure: The Role of Surface-Active Monomers. Materials 2017, 10, 282. [Google Scholar] [CrossRef]
- Mao, H.; Chi, Y.; Wang, F.; Mao, F.; Liang, F.; Lu, S.; Cen, K. Effect of Ultrasonic Pre-treatment on Dewaterability and Moisture Distribution in Sewage Sludge. Waste Biomass Valoriz. 2018, 9, 247–253. [Google Scholar] [CrossRef]
- Jin, Y.; Zheng, X.; Chi, Y.; Ni, M. Rapid, Accurate Measurement of the Oil and Water Contents of Oil Sludge Using Low-Field NMR. Ind. Eng. Chem. Res. 2013, 52, 2228–2233. [Google Scholar] [CrossRef]
- Zheng, X.; Jin, Y.; Chi, Y.; Ni, M. Simultaneous Determination of Water and Oil in Oil Sludge by Low-Field 1H NMR Relaxometry and Chemometrics. Energy Fuels 2013, 27, 5787–5792. [Google Scholar] [CrossRef]
- Meiboom, S.; Gill, D.R. Modified Spin-Echo Method for Measuring Nuclear Relaxation Times. Rev. Sci. Instrum. 1958, 29, 688–691. [Google Scholar] [CrossRef]
- Yao, W.; She, A.; Yang, P. 1H-NMR relaxation and state evolvement of evaporable water in cement pastes. J. Chin. Ceram. Soc. 2009, 37, 1. [Google Scholar]
- Karr, C. Analytical Methods for Coal and Coal Products: Volume II; Academic press: Cambridge, MA, USA, 2013; Volume 2. [Google Scholar]
- Chen, G.W.; Hung, W.T.; Chang, I.L.; Lee, S.F.; Lee, D.J. Continuous Classification of Moisture Content in Waste Activated Sludges. J. Environ. Eng. 1997, 123, 253–258. [Google Scholar] [CrossRef]
- Slimanou, H.; Bouguermouh, K.; Bouzidi, N. Synthesis of geopolymers based on dredged sediment in calcined and uncalcined states. Mater. Lett. 2019, 251, 188–191. [Google Scholar] [CrossRef]
- Li, Y.; Yang, F.; Miao, S.; Wang, D.; Li, Z.; Yuan, X.; Yuan, L.; Liu, Q. Achieved deep-dewatering of dredged sediments by Fe(II) activating persulfate pretreatment: Filtrating performance and mechanistic insights. Chem. Eng. J. 2021, 405, 126847. [Google Scholar] [CrossRef]
- Zhang, X.; Gu, Q.; Long, X.-E.; Li, Z.-L.; Liu, D.-X.; Ye, D.-H.; He, C.-Q.; Liu, X.-Y.; Väänänen, K.; Chen, X. Anthropogenic activities drive the microbial community and its function in urban river sediment. J. Soils Sediments 2015, 16, 716–725. [Google Scholar] [CrossRef]
- Huang, P.L. A Review of: “Modern Drying Technology, Second Enhanced Edition Editors: Yongkang Pan, Xizhong Wang and Xiangdong Liu”. Dry. Technol. 2007, 25, 2057–2058. [Google Scholar] [CrossRef]
- Yariv, S.; Cross, H. Physical Chemistry of Surfaces. Geochem. Colloid Syst. 1979, 150, 93–155. [Google Scholar] [CrossRef]
- Prausnitz, J.M.; Lichtenthaler, R.N.; De Azevedo, E.G. Molecular Thermodynamics of Fluid-Phase Equilibria; Pearson Education: Hoboken, NJ, USA, 1998. [Google Scholar]
- Hirschfelder, J.O.; Curtiss, C.F.; Bird, R.B.; Mayer, M.G. Molecular Theory of Gases and Liquids; Wiley: New York, NY, USA, 1964; Volume 165. [Google Scholar]
- Ansari, R.; Kazemi, E. Detailed investigation on single water molecule entering carbon nanotubes. Appl. Math. Mech. 2012, 33, 1287–1300. [Google Scholar] [CrossRef]
- Lin, D.T.W.; Chen, C.-K. A molecular dynamics simulation of TIP4P and Lennard-Jones water in nanochannel. Acta Mech. 2004, 173, 181–194. [Google Scholar] [CrossRef]
- Da Silva, L.B. Structural and dynamical properties of water confined in carbon nanotubes. J. Nanostruct. Chem. 2014, 4, 1–5. [Google Scholar] [CrossRef]
- Fort, T.; Putnam, F. The interaction of gases with solid surfaces. J. Colloid Interface Sci. 1976, 57, 190–191. [Google Scholar] [CrossRef]
- Mao, H.; Wang, F.; Mao, F.; Chi, Y.; Lu, S.; Cen, K. Measurement of water content and moisture distribution in sludge by1H nuclear magnetic resonance spectroscopy. Dry. Technol. 2016, 34, 267–274. [Google Scholar] [CrossRef]
- Ahunbay, M.G. Monte Carlo simulation of water adsorption in hydrophobic MFI zeolites with hydrophilic sites. Langmuir 2011, 27, 4986–4993. [Google Scholar] [CrossRef]
- Prost, R.; Koutit, T.; Benchara, A.; Huard, E. State and location of water adsorbed on clay minerals: Consequences of the hydration and swelling-shrinkage phenomena. Clays Clay Min. 1998, 46, 117–131. [Google Scholar] [CrossRef]
- Steele, W.A. The physical interaction of gases with crystalline solids: I. Gas-solid energies and properties of isolated adsorbed atoms. Surf. Sci. 1973, 36, 317–352. [Google Scholar] [CrossRef]
- Smith, L.S.; Lee, L.L. The Lennard-Jones 9:3 adsorptive system I: The Percus–Yevick and hypernetted chain theories and their modifications. J. Chem. Phys. 1979, 71, 4085. [Google Scholar] [CrossRef]
- Huang, Q.; Zhou, G.; Yu, B.; Wang, S.; Chi, Y.; Yan, J. Quantitative model for predicting the desorption energy of water contained in lignite. Fuel 2015, 157, 202–207. [Google Scholar] [CrossRef]
- Bushuev, Y.G.; Sastre, G. Atomistic Simulation of Water Intrusion—Extrusion in ITQ-4 (IFR) and ZSM-22 (TON): The Role of Silanol Defects. J. Phys. Chem. C 2011, 115, 21942–21953. [Google Scholar] [CrossRef]
- Fitts, T.G.; Brown, K. Stress-induced smectite dehydration: Ramifications for patterns of freshening and fluid expulsion in the N. Barbados accretionary wedge. Earth Planet. Sci. Lett. 1999, 172, 179–197. [Google Scholar] [CrossRef]
- Allardice, D.; Evans, D. The-brown coal/water system: Part 2. Water sorption isotherms on bed-moist Yallourn brown coal. Fuel 1971, 50, 236–253. [Google Scholar] [CrossRef]
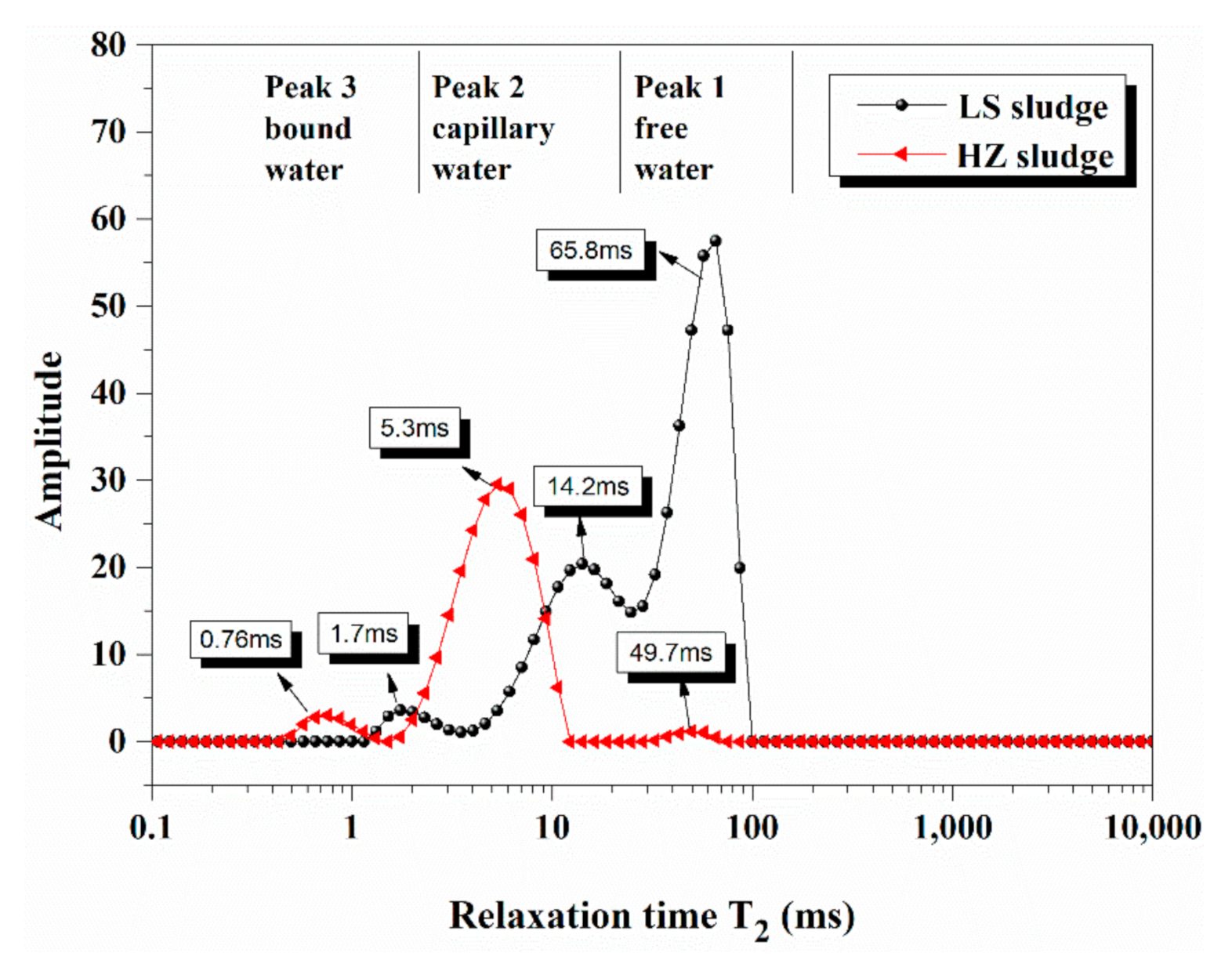
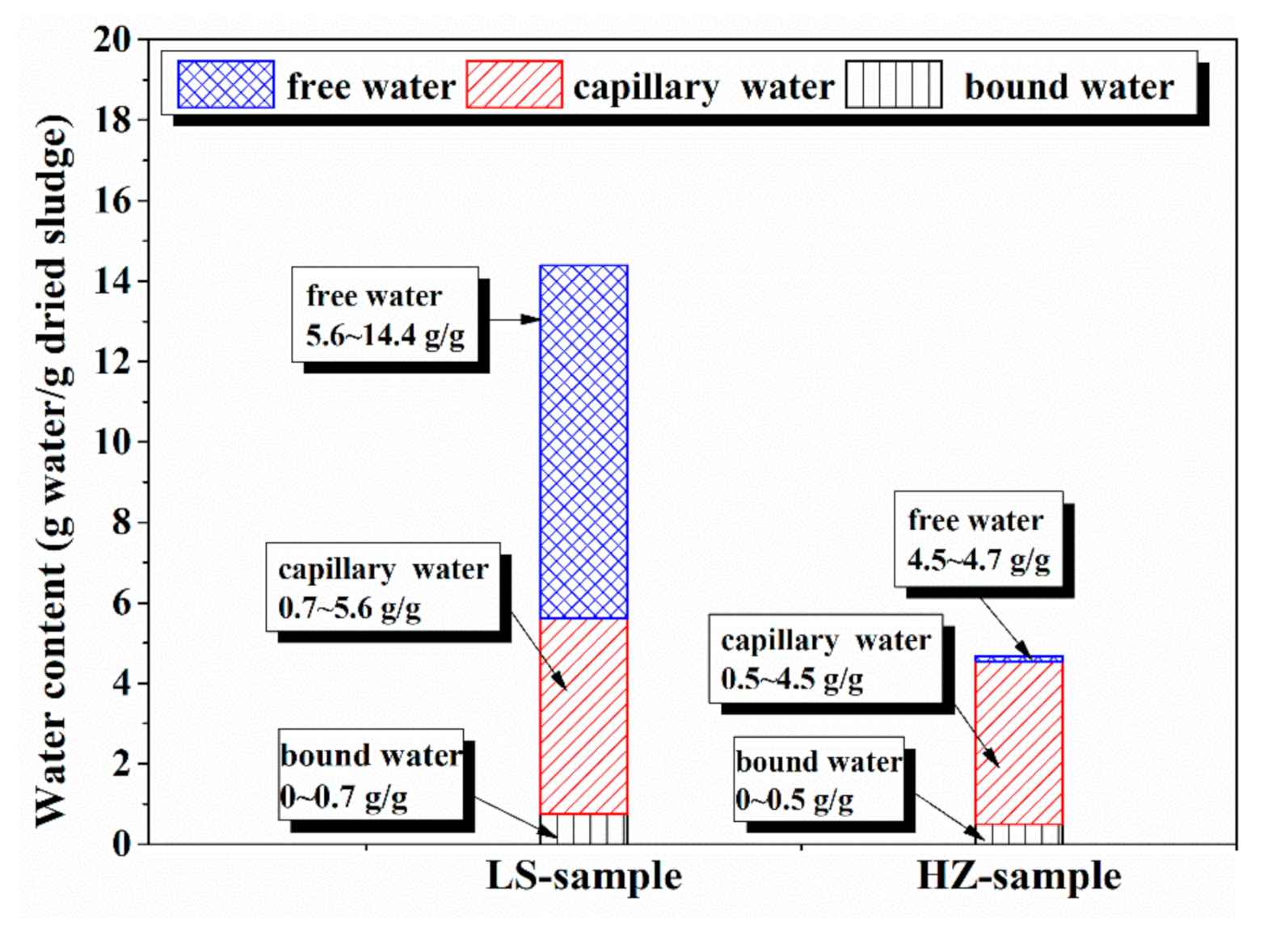
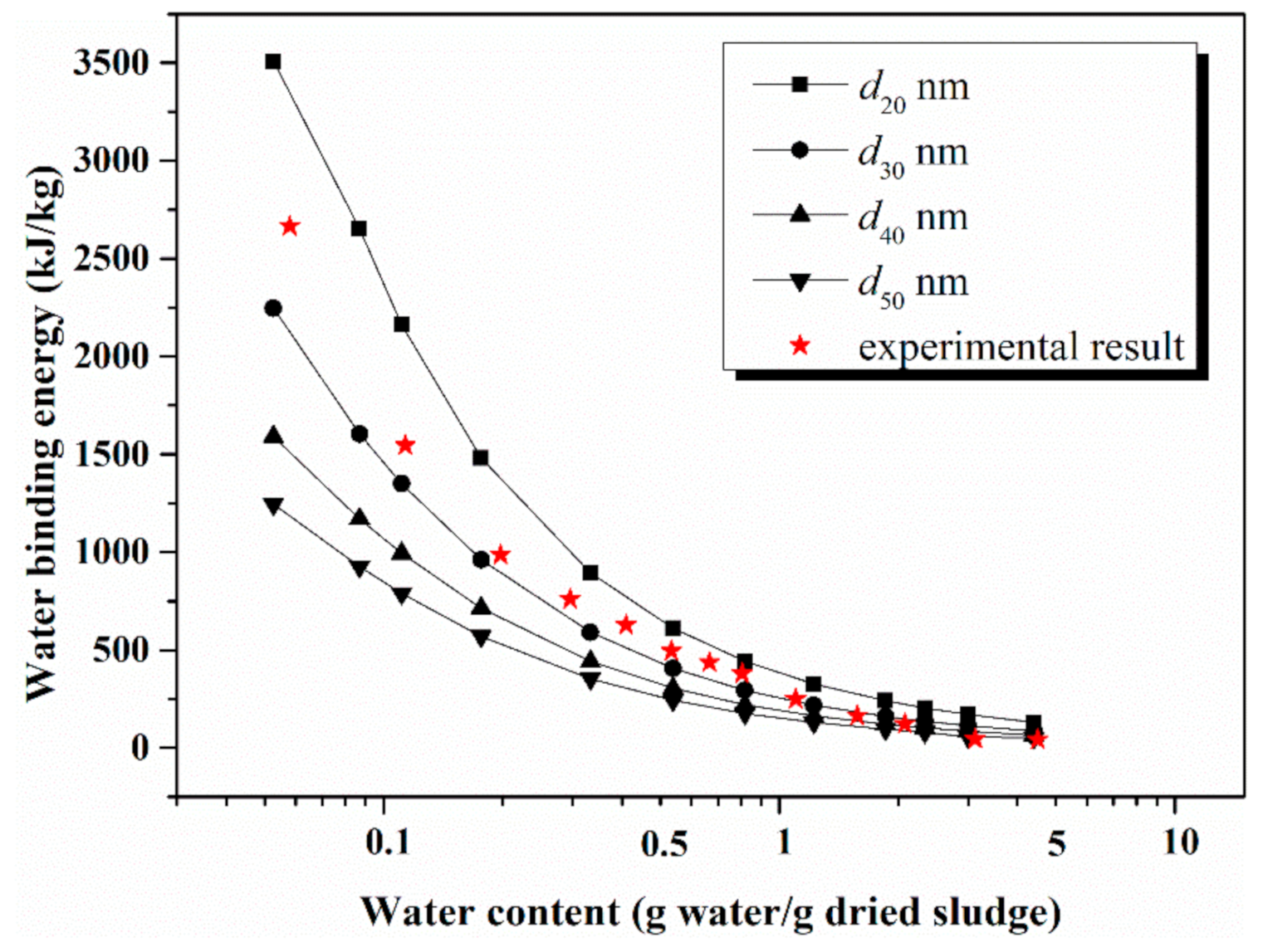
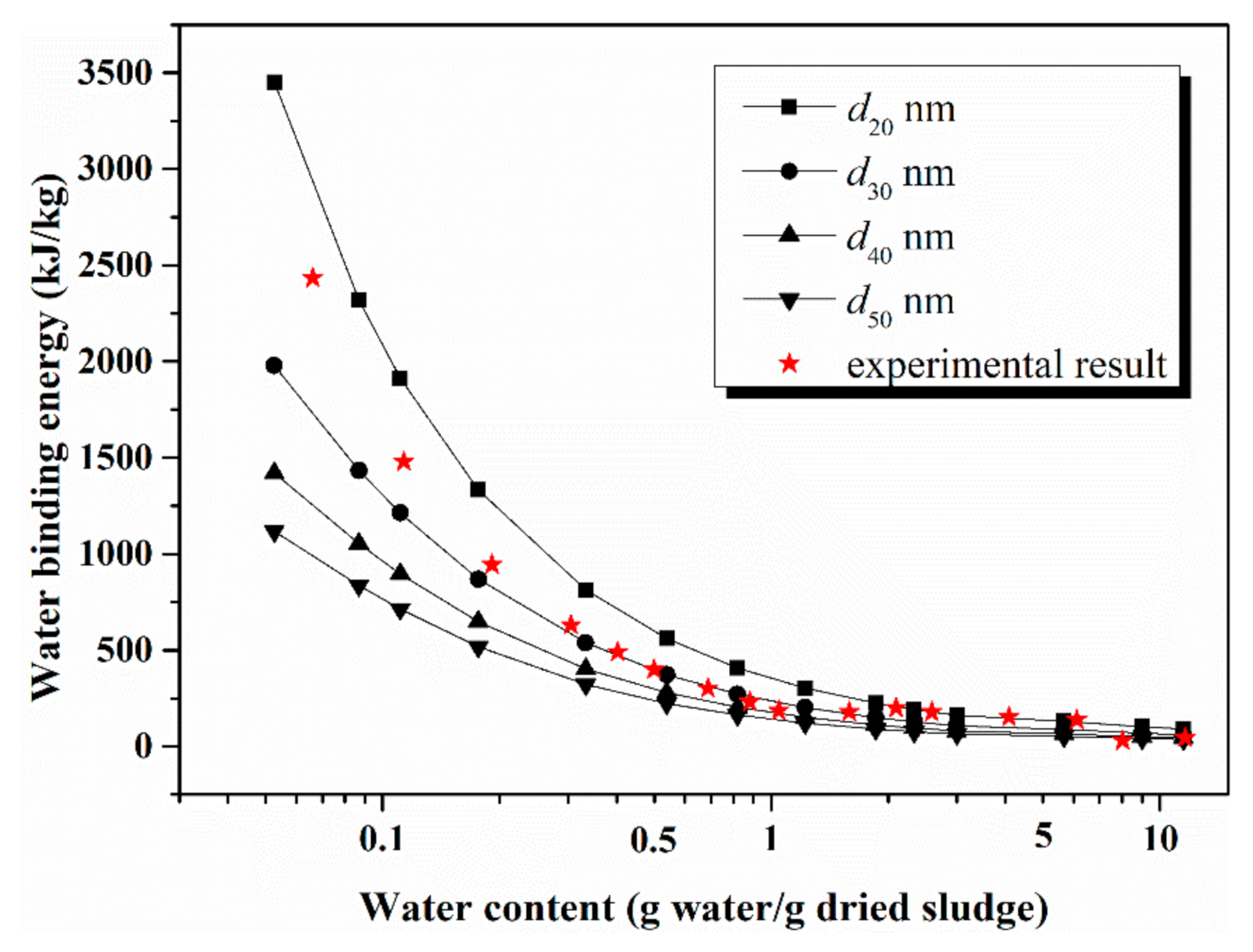
| Density g/cm3 | Specific Surface Area m2/g | Particle Size Distribution nm | |
|---|---|---|---|
| HZ sample | 1.42 | 32.5 | 25~130 |
| LS sample | 1.58 | 25.4 | 18~85 |
| Free Water % | Capillary Water % | Bound Water % | |
|---|---|---|---|
| HZ sample | 82.0~82.4% | 33.6~82.0 | 0~33.6% |
| LS sample | 84.9~93.5% | 43.3~84.9 | 0~43.3% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mao, F.; Zhao, Y.; Zhang, Y.; Chen, Z.; Yin, L. Measurement and Modelling of Moisture Distribution and Water Binding Energy of Dredged Sludge. Water 2020, 12, 3395. https://doi.org/10.3390/w12123395
Mao F, Zhao Y, Zhang Y, Chen Z, Yin L. Measurement and Modelling of Moisture Distribution and Water Binding Energy of Dredged Sludge. Water. 2020; 12(12):3395. https://doi.org/10.3390/w12123395
Chicago/Turabian StyleMao, Feiyan, Yingjie Zhao, Yiping Zhang, Zhou Chen, and Lu Yin. 2020. "Measurement and Modelling of Moisture Distribution and Water Binding Energy of Dredged Sludge" Water 12, no. 12: 3395. https://doi.org/10.3390/w12123395
APA StyleMao, F., Zhao, Y., Zhang, Y., Chen, Z., & Yin, L. (2020). Measurement and Modelling of Moisture Distribution and Water Binding Energy of Dredged Sludge. Water, 12(12), 3395. https://doi.org/10.3390/w12123395






