Can Zooplankton Be Entangled by Microfibers in the Marine Environment?: Laboratory Studies
Abstract
:1. Introduction
2. Materials and Methods
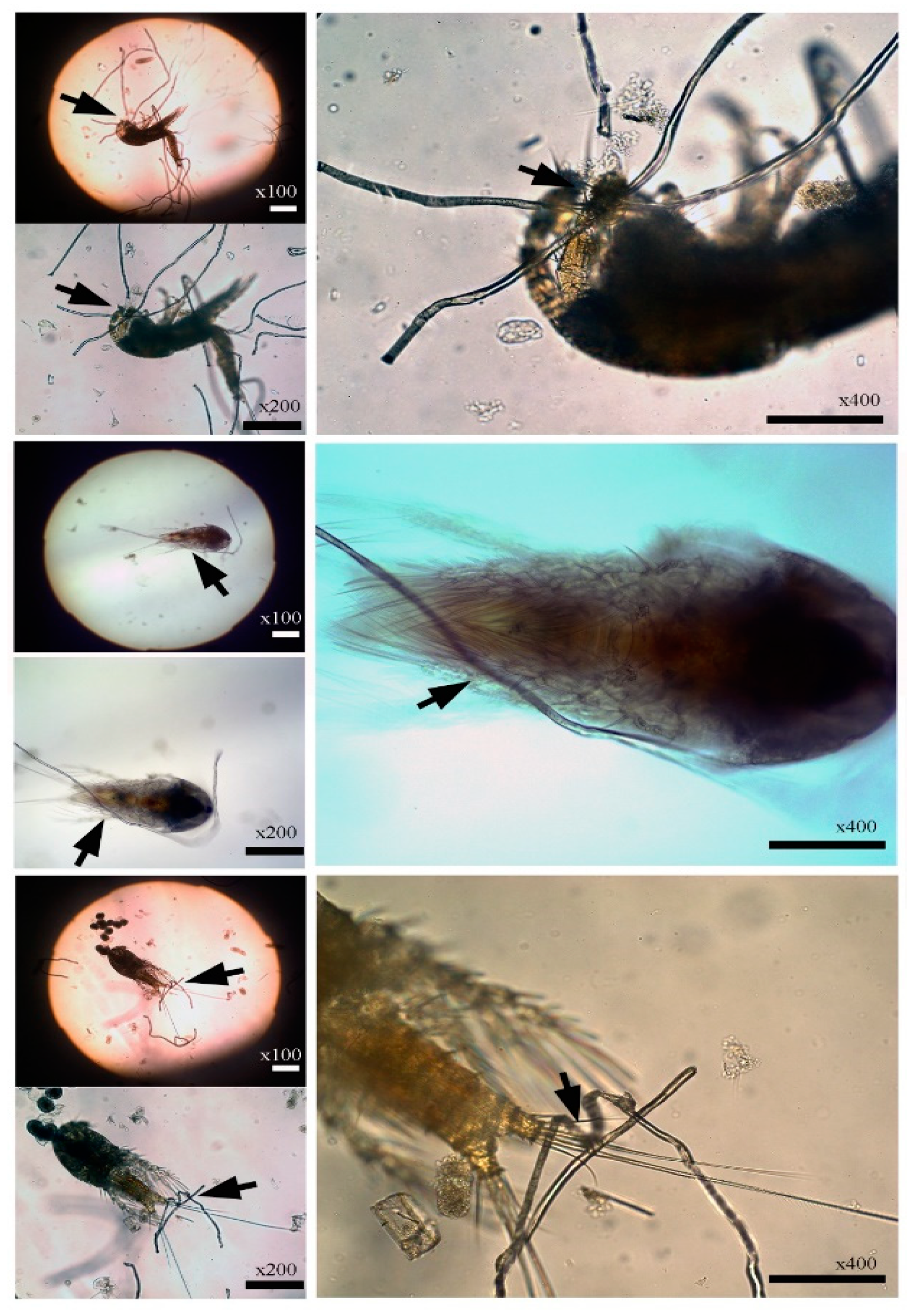
2.1. Accidental Observation of Copepods Entangled in Microfibers
2.2. Analysis for Exposure Condition Considering Environmentally Relevant Concentration
2.3. Microfiber Exposure Experiment
2.3.1. Preparation of Seawater for Exposure
2.3.2. Preparation of Copepod for Exposure
2.3.3. Preparation of Microfiber for Exposure
2.3.4. Exposure Design for Entanglement Experiment
- Experimental consideration for exposure of microfibers to copepods
- Exposure of microfibers to copepods under environmentally relevant concentration (50 mL)
- Effects of bubbling under elevated microfibers (500 mL)
- Reduced volume of incubation under elevated microfibers (10 mL)
2.3.5. Observation and Confirmation of Entangled Copepods
2.3.6. Statistical Analysis
3. Results
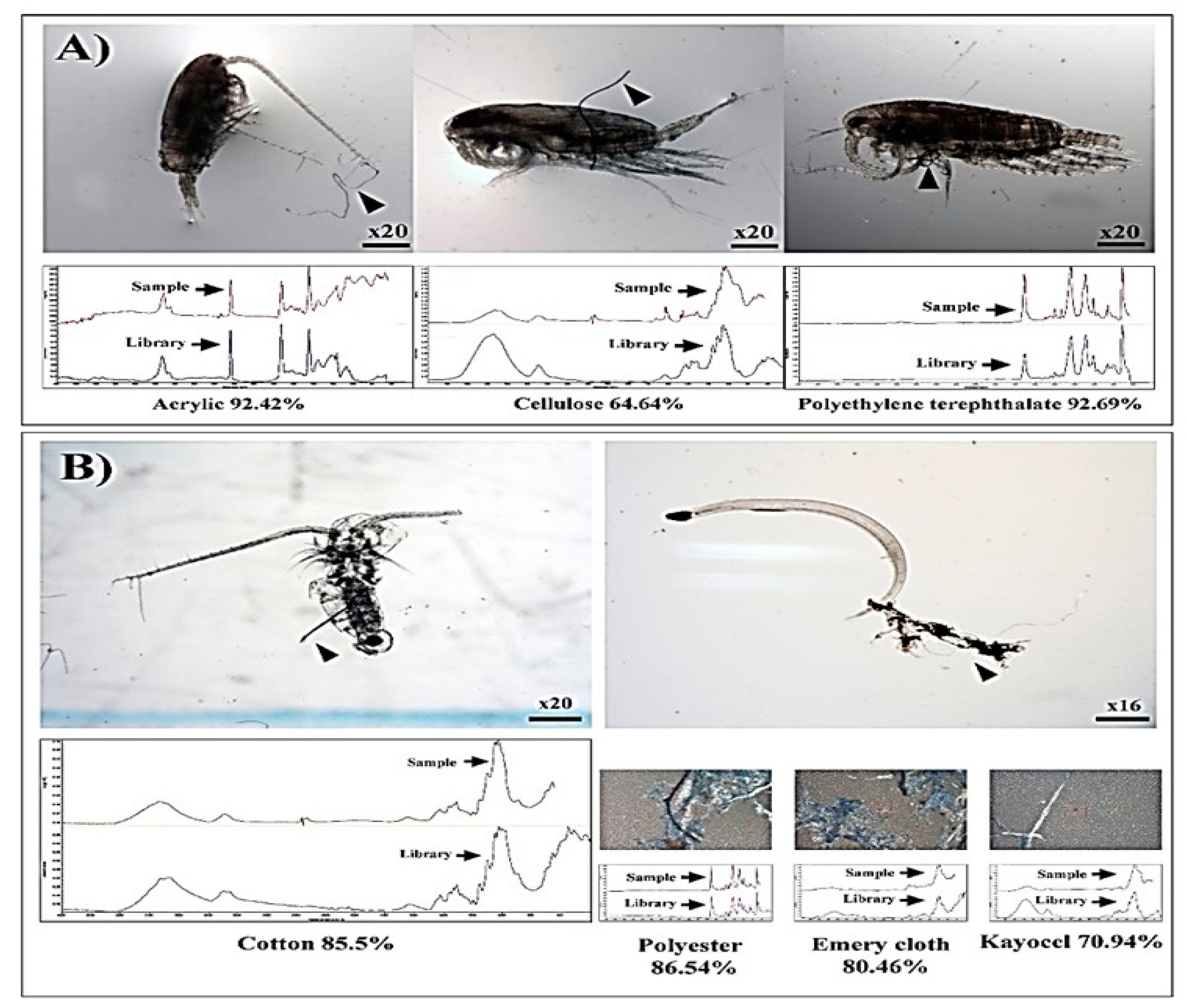
3.1. Entangled Copepods in Microfibers: On-Board Observation
3.2. Determination for Exposure Considering Environmentally Relevant Concentration
3.3. Laboratory Experiments
3.3.1. Probability of Copepod Entanglement in Microfibers: Environmentally Relevant Concentration of Microfibers (50 mL)
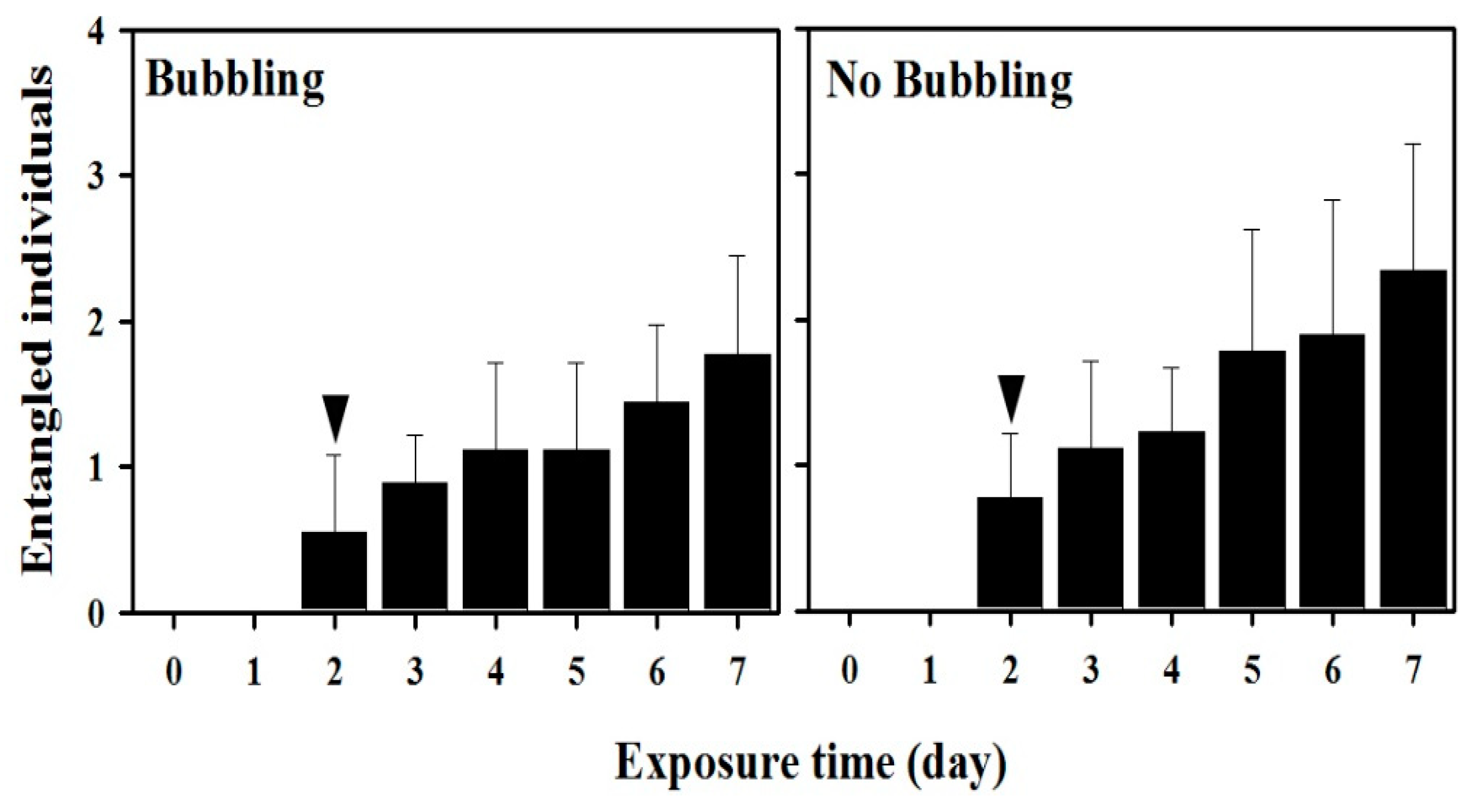
3.3.2. Probability of Copepod Entanglement in Microfibers: Effects of Bubbling Under Elevated Microfibers (500 mL)
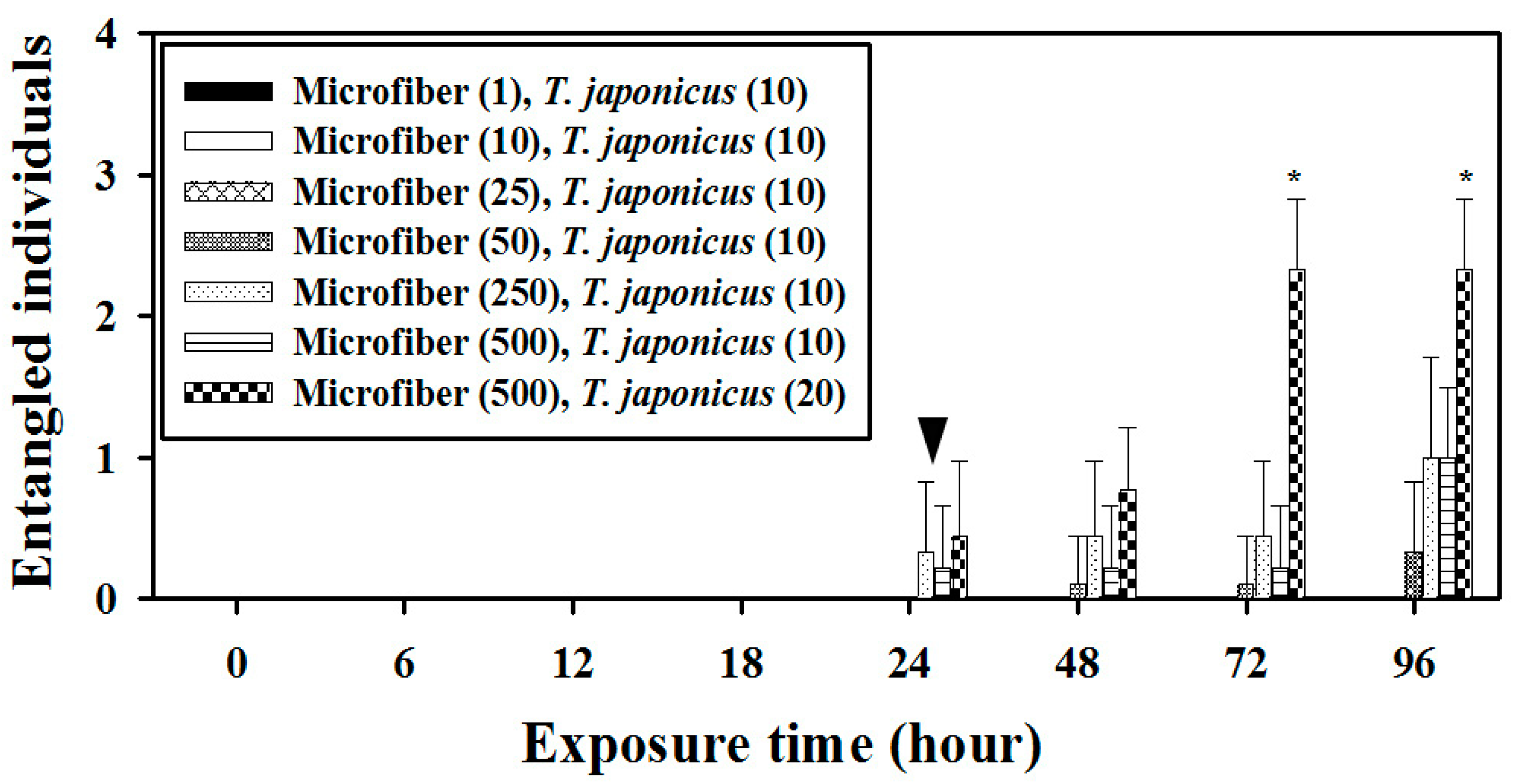
3.3.3. Probability of Zooplankton Entanglement in Microfibers: Reduced Volume of Incubation under High Microfiber Concentrations (10 mL)
3.4. Body Parts of Copepods Entangled in Microfibers
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- GESAMP. GESAMP (2010, IMO/FAO/UNESCO-IOC/UNIDO/WMO/IAEA/UN/UNEP Joint group of experts on the scientific aspects of marine environmental protection). In Micro-Plastics in the Marine Environment, Proceedings of the GESAMP International Workshop on Plastic Particles as a Vector in Transporting Persistent, Bioaccumulating and Toxic Substances in the Oceans. GESAMP Reports & Studies; Paris, France, 28–30 June 2010; Bowmer, T., Kershaw, P., Eds.; UNESCO-IOC: Paris, France, 2010; Number 82; pp. 18–27. Available online: http://www.gesamp.org/publications/proceedings-of-the-gesamp-workshop-on-microplastic-particles (accessed on 28 June 2018).
- Andrady, A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Derraik, J.G.B. The pollution of the marine environment by plastic debris: A review. Mar. Pollut. Bull. 2002, 44, 842–852. [Google Scholar] [CrossRef]
- Gregory, M.R. Environmental implications of plastic debris in marine settings-entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2013–2025. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, S.; Domingo, A.; Brazeiro, A.; Defeo, O.; Phillips, R.A. Marine debris ingestion by albatrosses in the southwest Atlantic Ocean. Mar. Pollut. Bull. 2015, 96, 149–154. [Google Scholar] [CrossRef] [Green Version]
- Nicolau, L.; Marçalo, A.; Ferreira, M.; Sá, S.; Vingada, J.; Eira, C. Ingestion of marine litter by loggerhead sea turtles, Caretta caretta, in Portuguese continental waters. Mar. Pollut. Bull. 2016, 103, 179–185. [Google Scholar] [CrossRef]
- Ryan, P.G.; Nico de Bruyn, P.J.; Bester, M.N. Regional differences in plastic ingestion among Southern Ocean fur seals and albatrosses. Mar. Pollut. Bull. 2016, 104, 207–210. [Google Scholar] [CrossRef] [Green Version]
- Foley, C.J.; Feiner, Z.S.; Malinich, T.D.; Höök, T.O. A meta-analysis of the effects of exposure to microplastics on fish and aquatic invertebrates. Sci. Total Environ. 2018, 631–632, 550–559. [Google Scholar] [CrossRef] [Green Version]
- Wright, S.L.; Thompson, R.C.; Galloway, T.S. The physical impacts of microplastics on marine organisms: A review. Environ. Pollut. 2013, 178, 483–492. [Google Scholar] [CrossRef]
- Browne, M.A.; Crump, P.; Niven, S.J.; Teuten, E.; Tonkin, A.; Galloway, T.; Thompson, R. Accumulation of microplastic on shorelines worldwide: Sources and sinks. Environ. Sci. Technol. 2011, 45, 9175–9179. [Google Scholar] [CrossRef]
- Buchanan, J. Pollution by synthetic fibres. Mar. Pollut. Bull. 1971, 2, 23. [Google Scholar] [CrossRef]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.G.; McGonigle, D.; Russell, A.E. Lost at sea: Where is all the plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef] [PubMed]
- Claessens, M.; Meester, S.D.; Landuyt, L.V.; Clerck, K.D.; Janssen, C.R. Occurrence and distribution of microplastics in marine sediments along the Belgian coast. Mar. Pollut. Bull. 2011, 62, 2199–2204. [Google Scholar] [CrossRef] [PubMed]
- Desforges, J.P.W.; Galbraith, M.; Dangerfield, N.; Ross, P.S. Widespread distribution of microplastics in subsurface seawater in the NE Pacific Ocean. Mar. Pollut. Bull. 2014, 79, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Ivar do Sul, J.A.; Costa, M.F. The present and future of microplastic pollution in the marine environment. Environ. Pollut. 2014, 185, 352–364. [Google Scholar] [CrossRef]
- Kang, J.-H.; Kwon, O.-Y.; Lee, K.-W.; Song, Y.K.; Shim, W.J. Marine neustonic microplastics around the southeastern coast of Korea. Mar. Pollut. Bull. 2015, 96, 304–312. [Google Scholar] [CrossRef]
- Aytan, U.; Valente, A.; Senturk, Y.; Usta, R.; Sahin, F.B.E.; Mazlum, R.E.; Agirbas, E. First evaluation of neustonic microplastics in Black Sea waters. Mar. Environ. Res. 2016, 119, 22–30. [Google Scholar] [CrossRef]
- Gago, J.; Carretero, O.; Filgueiras, A.V.; Viñas, L. Synthetic microfibers in the marine environment: A review on their occurrence in seawater and sediments. Mar. Pollut. Bull. 2018, 127, 365–376. [Google Scholar] [CrossRef]
- Au, S.Y.; Bruce, T.F.; Bridges, W.C.; Klaine, S.J. Responses of Hyalella Azteca to acute and chronic microplastic exposures. Environ. Toxicol. Chem. 2015, 34, 2564–2572. [Google Scholar] [CrossRef]
- Murray, F.; Cowie, P.R. Plastic contamination in the decapod crustacean Nephrops norvegicus (Linnaeus, 1758). Mar. Pollut. Bull. 2011, 62, 1207–1217. [Google Scholar] [CrossRef]
- Taylor, M.L.; Gwinnett, C.; Robinson, L.F.; Woodall, L.C. Plastic microfiber ingestion by deep-sea organisms. Sci. Rep. 2016, 6, 33997. [Google Scholar] [CrossRef] [Green Version]
- Moore, C.J.; Moore, S.L.; Leecaster, M.K.; Weisberg, S.B. A comparison of plastic and plankton in the North Pacific central gyre. Mar. Pollut. Bull. 2001, 42, 1297–1300. [Google Scholar] [CrossRef]
- Song, Y.K.; Hong, S.H.; Jang, M.; Kang, J.-H.; Kwon, O.Y.; Han, G.M.; Shim, W.J. Large accumulation of micro-sized synthetic polymer particles in the sea surface microlayer. Environ. Sci. Technol. 2014, 48, 9014–9021. [Google Scholar] [CrossRef] [PubMed]
- Lusher, A.L.; Hernandez-Milian, G.; O’Brien, J.; Berrow, S.; O’Connor, I.; Officer, R. Microplastic and macroplastic ingestion by a deep diving, oceanic cetacean: The True’s beaked whale Mesoplodon mirus. Environ. Pollut. 2015, 199, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Tagg, A.S.; Sapp, M.; Harrison, J.P.; Ojeda, J.J. Identification and quantification of microplastics in wastewater using focal plane array-based reflectance micro-FT-IR Imaging. Anal. Chem. 2015, 87, 6032–6040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raisuddin, S.; Kwok, K.W.H.; Leung, M.Y.; Schlenk, D.; Lee, J.S. The copepod Tigriopus: A promising marine model organism for ecotoxicology and environmental genomics. Aqut. Toxicol. 2007, 83, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.J.; Watson, W.; Bowlin, N.M.; Sheavly, S.B. Plastic particles in coastal pelagic ecosystems of the Northeast Pacific ocean. Mar. Environ. Res. 2011, 71, 41–52. [Google Scholar] [CrossRef]
- Frias, J.P.G.L.; Otero, V.; Sobral, P. Evidence of microplastics in samples of zooplankton from Portuguese coastal waters. Mar. Environ. Res. 2014, 95, 89–95. [Google Scholar] [CrossRef]
- Lee, K.-W.; Shim, W.J.; Kwon, O.Y.; Kang, J.-H. Size-dependent effects of micro polystyrene particles in the marine copepod Tigriopus japonicus. Environ. Sci. Technol. 2013, 47, 11278–11283. [Google Scholar] [CrossRef]
- Davison, P.; Asch, R.G. Plastic ingestion by mesopelagic fishes in the North Pacific Subtropical Gyre. Mar. Ecol. Prog. Ser. 2011, 432, 173–180. [Google Scholar] [CrossRef] [Green Version]
- Collignon, A.; Hecq, J.H.; Galgani, F.; Collard, F.; Goffart, A. Annual variation in neustonic micro- and meso-plastic particles and zooplankton in the Bay of Calvi (Mediterranean-Corsica). Mar. Pollut. Bull. 2014, 79, 293–298. [Google Scholar] [CrossRef] [Green Version]
- Kang, J.-H.; Kwon, O.-Y.; Shin, W.J. Potential threat of microplastics to zooplanktivores in the surface waters of the southern sea of Korea. Arch. Environ. Contam. Toxicol. 2015, 69, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Botterell, Z.L.R.; Beaumont, N.; Dorrington, T.; Steinke, M.; Thompson, R.C.; Lindeque, P.K. Bioavailability and effects of microplastics on marine zooplankton: A review. Environ. Pollut. 2019, 245, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.J. Synthetic polymers in the marine environment: A rapidly increasing, long-term threat. Environ. Res. 2008, 108, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Law, K.L.; Moret-Ferguson, S.; Maximenko, N.A.; Proskurowski, G.; Peacock, E.E.; Hafner, J.; Reddy, C.M. Plastic accumulation in the North Atlantic Subtropical Gyre. Science 2010, 329, 1185–1188. [Google Scholar] [CrossRef] [Green Version]
- Carson, H.S.; Nerheim, M.S.; Carroll, K.A.; Eriksen, M. The plastic-associated microorganisms of the North Pacific Gyre. Mar. Pollut. Bull. 2013, 75, 126–132. [Google Scholar] [CrossRef]
- Nel, H.A.; Froneman, P.W. A quantitative analysis of microplastic pollution along the south-eastern coastline of South Africa. Mar. Pollut. Bull. 2015, 101, 274–279. [Google Scholar] [CrossRef]
- Mauchline, J.; Blaxter, J.H.S.; Southward, A.J.; Tyler, P.A. Advances in Marine Biology: The Biology of Calanoid Copepods, 1st ed.; Academic Press: San Diego, CA, USA, 1998; pp. 140–174. Available online: https://www.sciencedirect.com/bookseries/advances-in-marine-biology/vol/33 (accessed on 28 June 2018).
- Kim, H.-J.; Yamade, T.; Iwasaki, K.; Marcial, H.S.; Hagiwara, A. Phototactic behavior of the marine harpacticoid copepod Tigriopus japonicus related to developmental stages under various light conditions. J. Exp. Mar. Biol. Ecol. 2019, 518, 151183. [Google Scholar] [CrossRef]
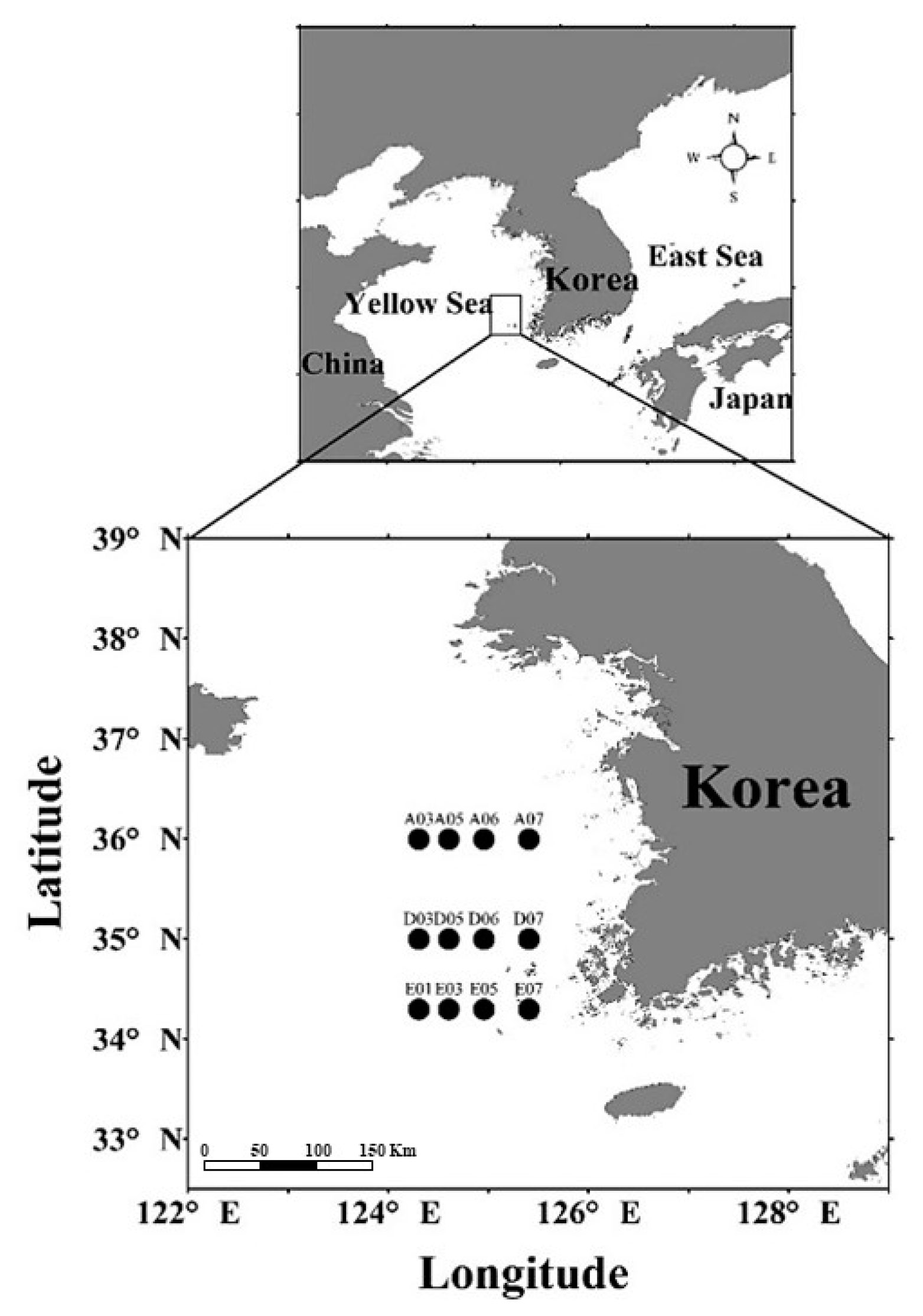
| Year | Station | Sampling Strata | Filtered Volume (m3/sample) | Subsampling for Analysis of Entanglement (%) | Subsampling for Analysis of MP (%) | |||
|---|---|---|---|---|---|---|---|---|
| Spring | Summer | Spring | Summer | Spring | Summer | |||
| 2013 | A03 | 10 m to surface (surface mixed layer) | 5.0 | 3.1 | 3.5 | 1.8 | 50 | 50 |
| A05 | 3.9 | 6.6 | 1.9 | 1.8 | 50 | 50 | ||
| A06 | 4.1 | 4.4 | 3.8 | 1.6 | 50 | 50 | ||
| A07 | 4.0 | 4.7 | 4.1 | 1.7 | 50 | 50 | ||
| D03 | 3.9 | 4.2 | 1.8 | 1.7 | 50 | 50 | ||
| D05 | 3.4 | 5.5 | 100.0 | 2.5 | 50 | 50 | ||
| D06 | 3.8 | 3.9 | 100.0 | 2.0 | 50 | 50 | ||
| D07 | 4.5 | 4.7 | 5.8 | 1.7 | 50 | 50 | ||
| E01 | 4.9 | 3.5 | 100.0 | 3.8 | 50 | 50 | ||
| E03 | 5.5 | 5.8 | 7.3 | 0.9 | 50 | 50 | ||
| E05 | 4.6 | 8.3 | 100.0 | 1.9 | 50 | 50 | ||
| E07 | 4.6 | 5.7 | 3.7 | 0.9 | 50 | 50 | ||
| Condition | Zooplankton | Body Length of Entangled Zooplankton (mm) | Length of Entangled Microfibers (mm) | Thickness of Entangled Microfibers (µm) | Polymer Types of Entangled Microfibers |
|---|---|---|---|---|---|
| Archived samples | Calanus sinicus, Unknown copepod, Sagitta sp. | 2.94 ± 1.81 (n = 5) | 2.14 ± 0.76 (n = 5) | 41.5 ± 10.7 (n = 4) | Acrylic, Cellulose, Polyethylene terephthalate, Cotton, Polyester, Emery cloth, Kayocel |
| Laboratory Studies | Tigriopus japonicus | 0.85 ± 0.02 (n = 20) | 0.83 ± 0.13 (n = 20) | 27.1 ± 0.3 (n = 20) | Polyester |
| Information of Study | Month/Year | Station Which Entangled Zooplankton Is Observed | Ambient Condition | Abundance (Individuals/m3) and Proportion of Entangled Zooplankton | Microfiber/Zooplankton Ratio | |
| Microfiber (number/m3) | Zooplankton (individuals/m3) | |||||
| In-situ study (Yellow Sea) | 08/2013 | A03 | 8.00 | 3258 | 37/1.1% | 0.002 |
| 08/2013 | D06 | 6.94 | 1359 | 26/1.9% | 0.005 | |
| Laboratory Study (Beaker) | Exposure Time (h) | Treatment | Experimental condition | Mean abundance (n = 3) (individuals) and Proportion of Entangled T. japonicus | Microfiber/Copepod Ratio | |
| Microfiber (number) | T. japonicus(individuals) | |||||
| 50-mL beaker | 168 | No bubbling | 1 | 20 | 0/0.0% | 0.05 |
| 1 | 50 | 0/0.0% | 0.02 | |||
| Instruments for Exposure | Exposure Time (h) | Treatment | Number of Microfibers Treated | Inserted T. japonicus | Mean Abundance (n = 9) and Proportion of Entangled T. japonicus | Microfiber/Copepod Ratio |
|---|---|---|---|---|---|---|
| Beaker (500 mL) | 168 | Bubbling | 50 * | 30 | 0.0/0.0% | 1.6 |
| No bubbling | 50 * | 30 | 0.3/1.0% | 1.6 | ||
| Bubbling | 5000 *** | 30 | 1.8/6.0% | 166.6 | ||
| No bubbling | 5000 *** | 30 | 2.3/7.7% | 166.6 | ||
| 6-well plate (10 mL) | 96 | No bubbling | 1 * | 10 | 0.0/0.0% | 0.1 |
| 10 ** | 10 | 0.0/0.0% | 1.0 | |||
| 25 ** | 10 | 0.0/0.0% | 2.5 | |||
| 50 ** | 10 | 0.3/3.0% | 5.0 | |||
| 250 *** | 10 | 1.0/10.0% | 25.0 | |||
| 500 *** | 10 | 1.0/10.0% | 50.0 | |||
| 500 *** | 20 | 2.3/11.5% | 25.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.-H.; Kwon, O.-Y.; Hong, S.H.; Shim, W.J. Can Zooplankton Be Entangled by Microfibers in the Marine Environment?: Laboratory Studies. Water 2020, 12, 3302. https://doi.org/10.3390/w12123302
Kang J-H, Kwon O-Y, Hong SH, Shim WJ. Can Zooplankton Be Entangled by Microfibers in the Marine Environment?: Laboratory Studies. Water. 2020; 12(12):3302. https://doi.org/10.3390/w12123302
Chicago/Turabian StyleKang, Jung-Hoon, Oh-Youn Kwon, Sang Hee Hong, and Won Joon Shim. 2020. "Can Zooplankton Be Entangled by Microfibers in the Marine Environment?: Laboratory Studies" Water 12, no. 12: 3302. https://doi.org/10.3390/w12123302
APA StyleKang, J.-H., Kwon, O.-Y., Hong, S. H., & Shim, W. J. (2020). Can Zooplankton Be Entangled by Microfibers in the Marine Environment?: Laboratory Studies. Water, 12(12), 3302. https://doi.org/10.3390/w12123302





