Variability in Environmental Conditions Strongly Impacts Ostracod Assemblages of Lowland Springs in a Heavily Anthropized Area
Abstract
1. Introduction
2. Materials and Methods
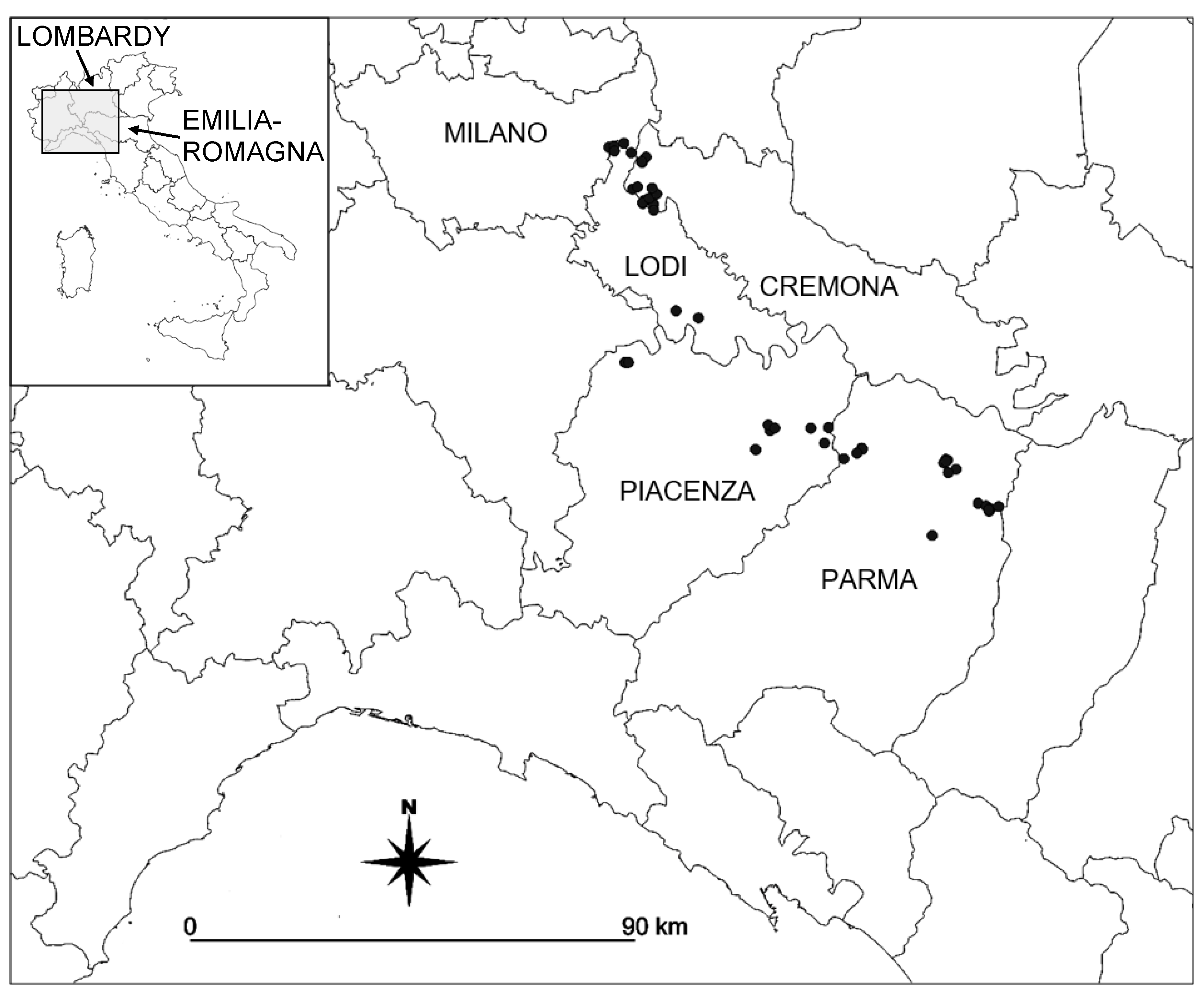
2.1. Study Area
2.2. Field and Laboratory Techniques
2.3. Data Analysis
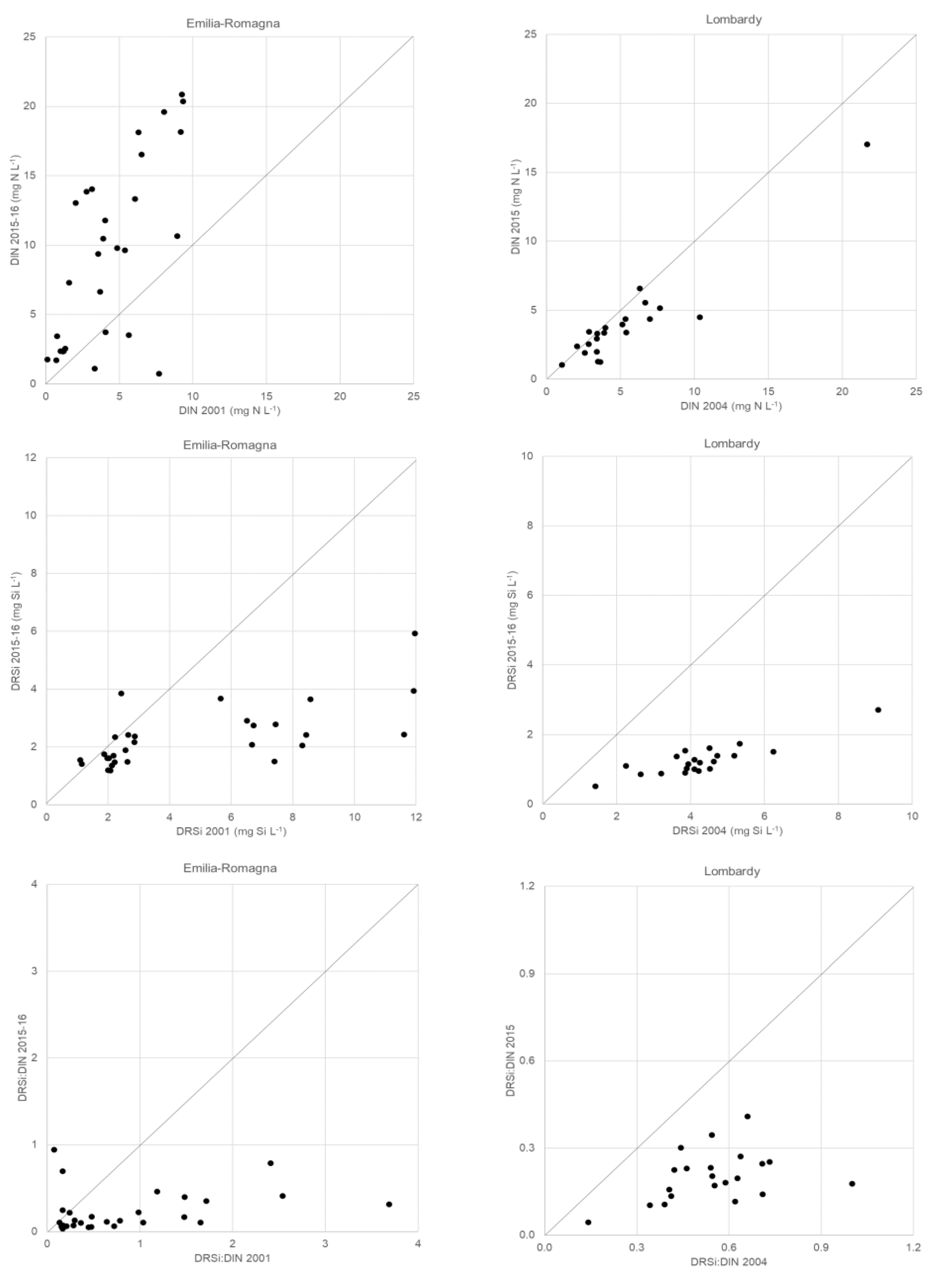
3. Results
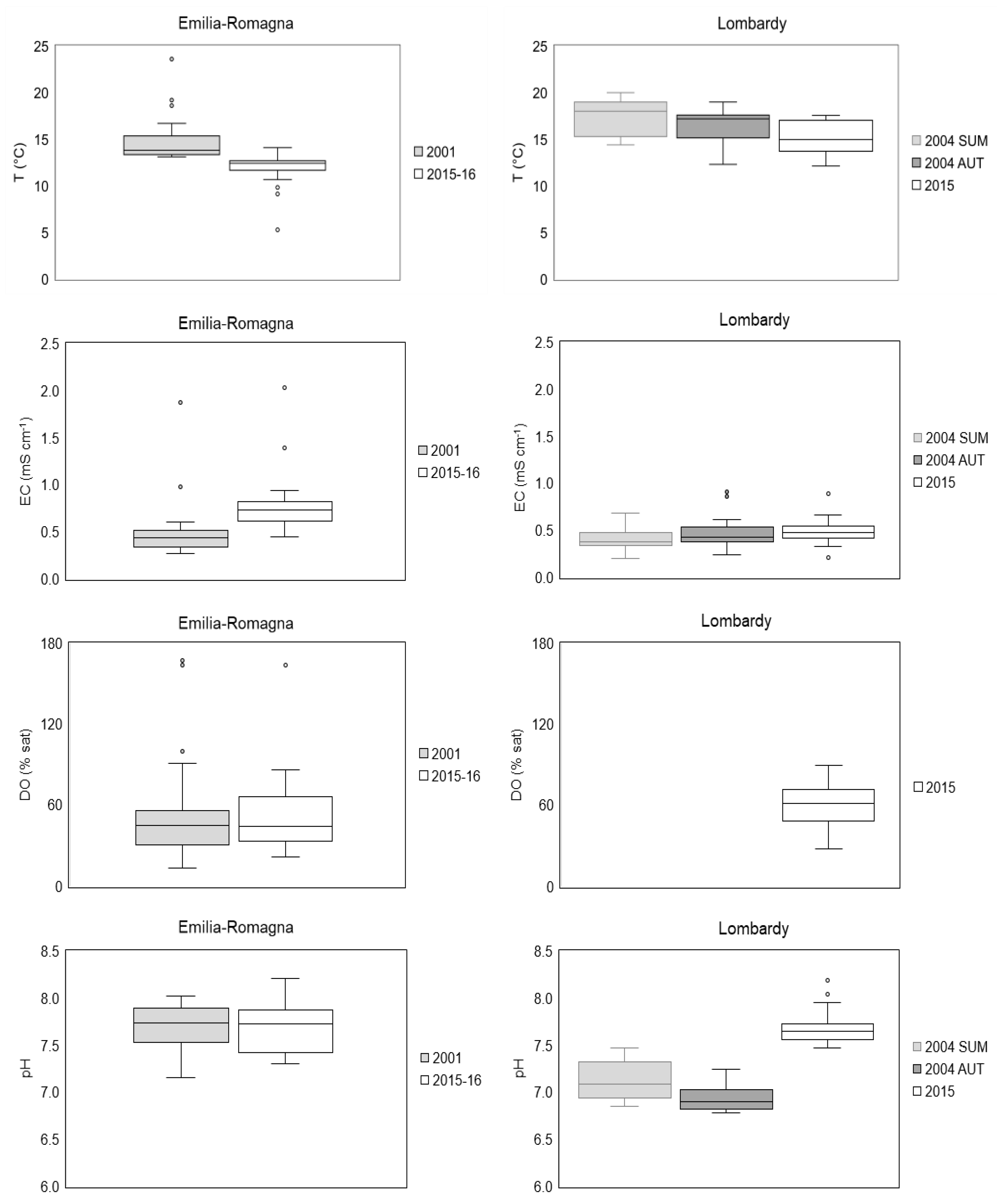
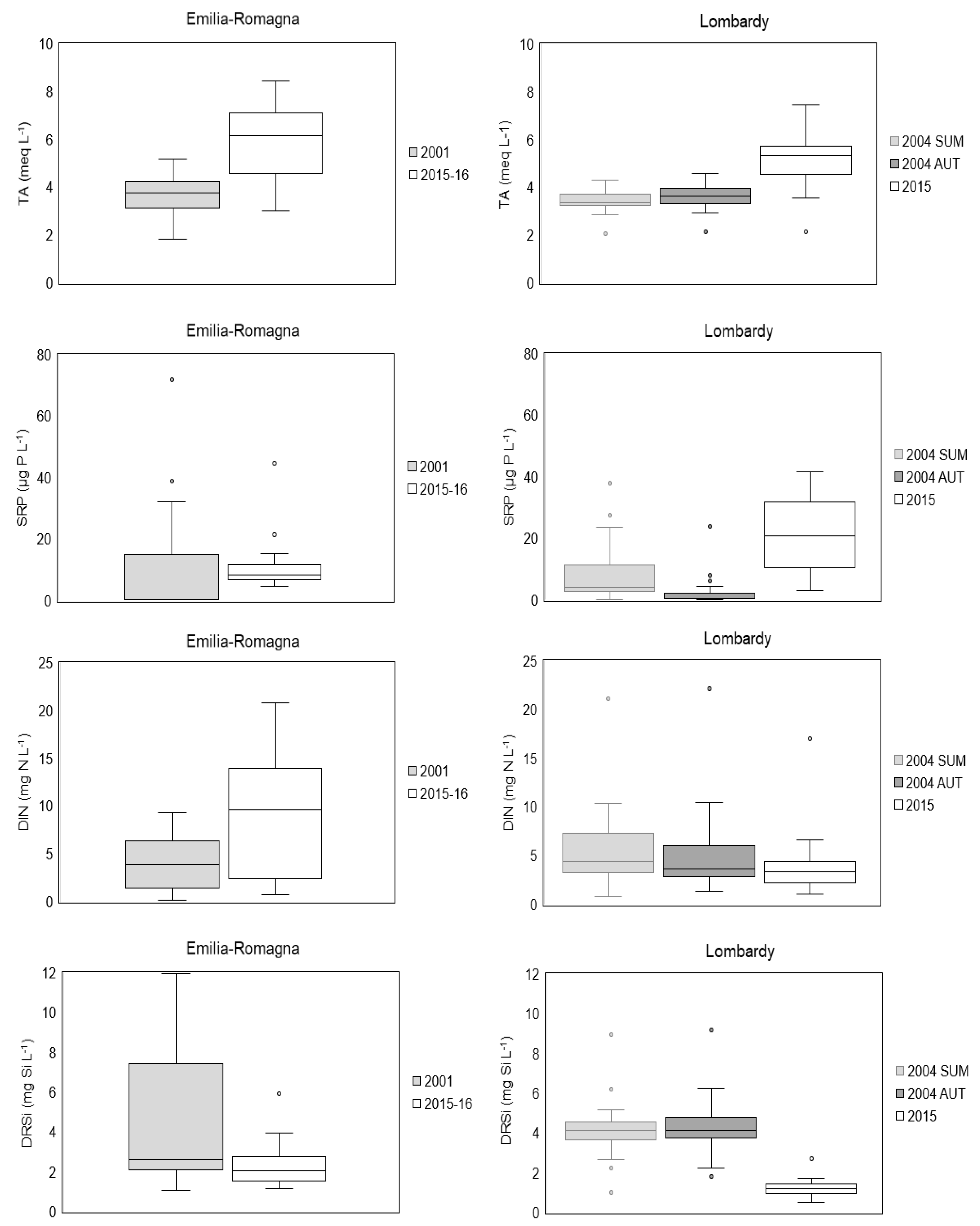
3.1. Physical and Chemical Variables
3.1.1. Water Characteristics of Springs
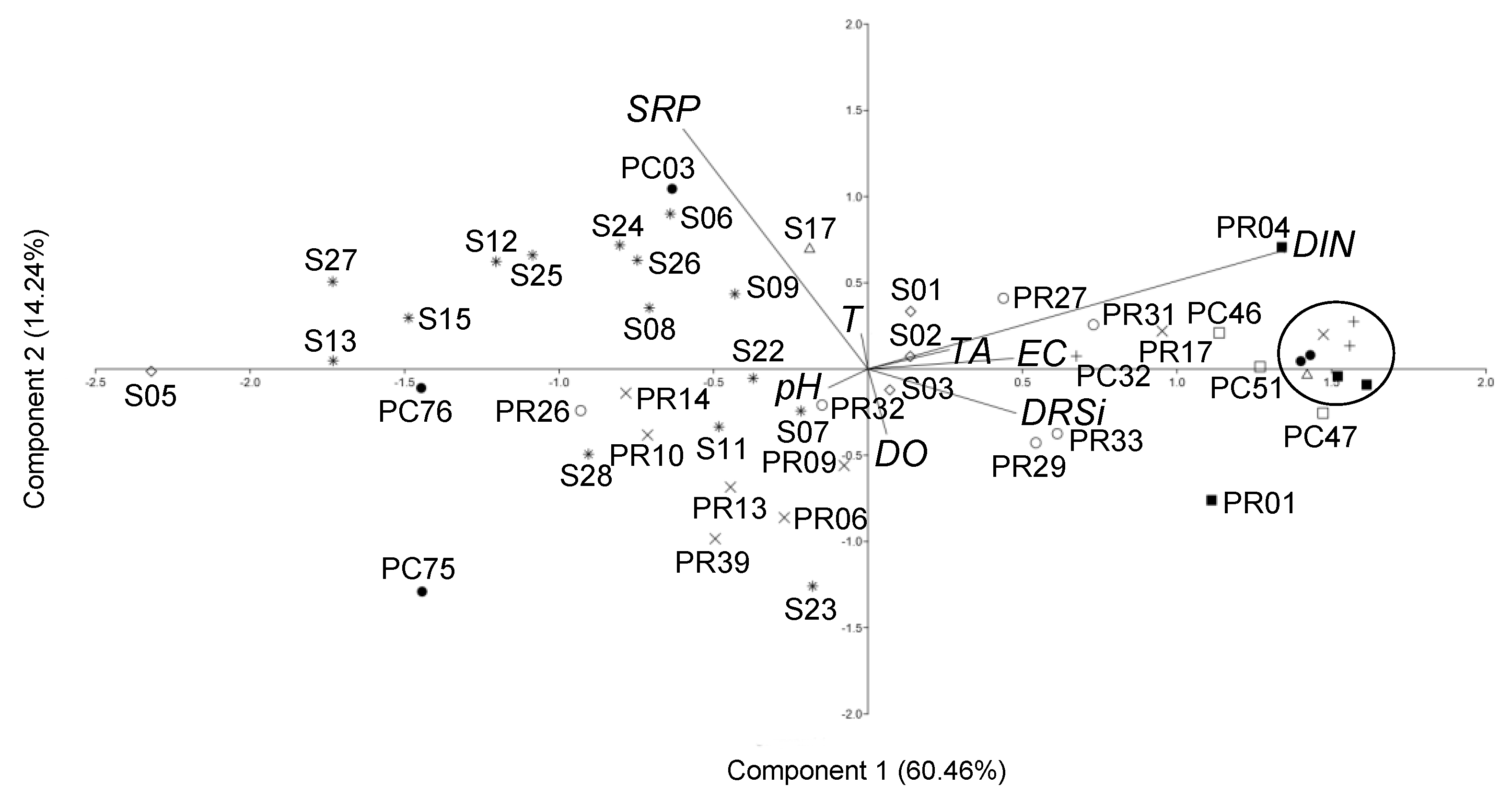
3.1.2. PCA Results
3.1.3. Comparison with Previous Studies
3.2. Ostracod Assemblages
4. Discussion
4.1. Effects of Hydrological Factors and Agricultural Practice on Water Quality
4.2. Trends in Physico-Chemical Characteristics of Spring Waters
4.3. Structure, Distribution Patterns and Evolution of Ostracod Assemblages
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kløve, B.; Ala-Aho, P.; Bertrand, G.; Boukalova, Z.; Ertürk, A.; Goldscheider, N.; Ilmonen, J.; Karakaya, N.; Kupfersberger, H.; Kvœrner, J.; et al. Groundwater dependent ecosystems. Part I: Hydroecological status and trends. Environ. Sci. Policy 2011, 14, 770–781. [Google Scholar] [CrossRef]
- De Luca, D.A.; Destefanis, E.; Forno, M.G.; Lasagna, M.; Masciocco, L. The genesis and the hydrogeological features of the Turin Po Plain fontanili, typical lowland springs in Northern Italy. Bull. Eng. Geol. Environ. 2014, 73. [Google Scholar] [CrossRef]
- Abdelahad, N.; Bolpagni, R.; Lasinio, G.J.; Vis, M.L.; Amadio, C.; Laini, A.; Keil, E.J. Distribution, morphology and ecological niche of Batrachospermum and Sheathia species (Batrachospermales, Rhodophyta) in the fontanili of the Po plain (northern Italy). Eur. J. Phycol. 2015, 50, 318–329. [Google Scholar] [CrossRef]
- Fumagalli, N.; Senes, G.; Ferrario, P.S.; Toccolini, A. A minimum indicator set for assessing fontanili (lowland springs) of the Lombardy Region in Italy. Eur. Countrys. 2017, 9, 1–16. [Google Scholar] [CrossRef]
- Cantonati, M.; Füreder, L.; Gerecke, R.; Jüttner, I.; Cox, E.J. Crenic habitats, hotspots for freshwater biodiversity conservation: Toward an understanding of their ecology. Freshw. Sci. 2012, 31, 463–480. [Google Scholar] [CrossRef]
- Zanini, A.; Petrella, E.; Sanangelantoni, A.M.; Angelo, L.; Ventosi, B.; Viani, L.; Rizzo, P.; Remelli, S.; Bartoli, M.; Bolpagni, R.; et al. Groundwater characterization from an ecological and human perspective: An interdisciplinary approach in the Functional Urban Area of Parma, Italy. Rend. Lincei Sci. Fis. Nat. 2018, 30, 93–108. [Google Scholar] [CrossRef]
- Soana, E.; Balestrini, R.; Vincenzi, F.; Bartoli, M.; Castaldelli, G. Mitigation of nitrogen pollution in vegetated ditches fed by nitrate-rich spring waters. Agric. Ecosyst. Environ. 2017, 243, 74–82. [Google Scholar] [CrossRef]
- Balestrini, R.; Delconte, C.; Palumbo, M.; Buffagni, A. Biotic control of in-stream nutrient retention in nitrogen-rich springs (Po Valley, Northern Italy). Ecol. Eng. 2018, 122, 303–314. [Google Scholar] [CrossRef]
- Minelli, A. (Ed.) Springs and Spring Watercourses-Springs in the Northern Italian Plains; Italian Habitats, Italian Ministry of the Environment and Territory Protection, Friuli Museum of Natural History: Udine, Italy, 2001; 154p. [Google Scholar]
- Rossetti, G.; Pieri, V.; Martens, K. Recent ostracods (Crustacea, Ostracoda) found in lowland springs of the provinces of Piacenza and Parma (Northern Italy). Hydrobiologia 2005, 542, 287–296. [Google Scholar] [CrossRef]
- Cantonati, M.; Poikane, S.; Pringle, C.M.; Stevens, L.E.; Turak, E.; Heino, J.; Richardson, J.S.; Bolpagni, R.; Borrini, A.; Cid, N.; et al. Characteristics, Main Impacts, and Stewardship of Natural and Artificial Freshwater Environments: Consequences for Biodiversity Conservation. Water 2020, 12, 260. [Google Scholar] [CrossRef]
- Gandolfi, G.; Zerunian, S.; Torricelli, P.; Marconato, A. I Pesci Delle Acque Interne Italiane; Ministero dell’Ambiente, Unione Zoologica Italiana. Istituto Poligrafico e Zecca dello Stato: Roma, Italy, 1991. [Google Scholar]
- Barbaresi, S.; Gherardi, F. The Invasion of the Alien Crayfish Procambarus clarkii in Europe, with Particular Reference to Italy. Biol. Invasions 2000, 2, 259–264. [Google Scholar] [CrossRef]
- Bracco, F.; Paradisi, S.; Sburlino, G.; Stoch, F. Degradation and conservation. In Springs and Spring Watercourses-Springs in the Northern Italian Plains; Minelli, A., Ed.; Italian Habitats, Italian Ministry of the Environment and Territory Protection, Friuli Museum of Natural History: Udine, Italy, 2001; pp. 123–139. [Google Scholar]
- Pieri, V.; Caserini, C.; Gomarasca, S.; Martens, K.; Rossetti, G. Water quality and diversity of the Recent ostracod fauna in lowland springs from Lombardy (northern Italy). Hydrobiologia 2007, 585, 79–87. [Google Scholar] [CrossRef]
- Balderacchi, M.; Perego, A.; Lazzari, G.; Muñoz-Carpena, R.; Acutis, M.; Laini, A.; Giussani, A.; Sanna, M.; Kane, D.; Trevisan, M. Avoiding social traps in the ecosystem stewardship: The Italian Fontanile lowland spring. Sci. Total. Environ. 2016, 539, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Laini, A.; Bartoli, M.; Castaldi, S.; Viaroli, P.; Capri, E.; Trevisan, M. Greenhouse gases (CO2, CH4 and N2O) in lowland springs within an agricultural impacted watershed (Po River Plain, northern Italy). Chem. Ecol. 2011, 27, 177–187. [Google Scholar] [CrossRef]
- Balestrini, R.; Sacchi, E.; Tidili, D.; Delconte, C.; Buffagni, A. Factors affecting agricultural nitrogen removal in riparian strips: Examples from groundwater-dependent ecosystems of the Po Valley (Northern Italy). Agric. Ecosyst. Environ. 2016, 221, 132–144. [Google Scholar] [CrossRef]
- Battegazzore, M.; Morisi, A. Environmental evaluation of springs in the intensely cultivated and industrialized inland plain of Cuneo (Northwest Italy). J. Environ. Sci. Eng. 2012, B1, 19–24. [Google Scholar]
- Shestani, L.; Morisi, A.; Fenoglio, S. Qualità ecologica dei fontanili di Cavallermaggiore nel periodo 1993-2006. St. Trent. Sc. Nat., Acta Biol. 2007, 83, 123–128. [Google Scholar]
- Viaroli, P.; Rossetti, G.; Bertolini, S.; Pieri, V.; Venturelli, G.; Bernini, F.; Mozzanica, E.; Pedrelli, C. Qualità ambientale e problemi di conservazione dei fontanili nella pianura tra Piacenza e Parma. St. Trent. Sc. Nat. Acta Biol. 2003, 80, 181–183. [Google Scholar]
- Gavazza, E.; Bolis, C.; Donelli, E.; Ravizza, L.; Camatini, M. Procedure standard ai sensi del D.Lgs. 152/99 per la valutazione dello stato di qualità dei fontanili della Provincia di Milano. Inquinamento 2003, 45, 52–56. [Google Scholar]
- Laini, A.; Bartoli, M.; Lamastra, L.; Capri, E.; Balderacchi, M.; Trevisan, M. Herbicide contamination and dispersion pattern in lowland springs. Sci. Total Environ. 2012, 438, 312–318. [Google Scholar] [CrossRef]
- Vasileiadis, S.; Puglisi, E.; Arena, M.; Cappa, F.; Van Veen, J.A.; Cocconcelli, P.S.; Trevisan, M. Soil microbial diversity patterns of a lowland spring environment. FEMS Microbiol. Ecol. 2013, 86, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Meisch, C.; Smith, R.J.; Martens, K. A subjective global checklist of the extant non-marine Ostracoda (Crustacea). Eur. J. Taxon. 2019, 492. [Google Scholar] [CrossRef]
- Pieri, V.; Martens, K.; Meisch, C.; Rossetti, G. An annotated checklist of the Recent non-marine ostracods (Ostracoda: Crustacea) from Italy. Zootaxa 2015, 3919, 271. [Google Scholar] [CrossRef] [PubMed]
- Mezquita, F.; Griffiths, H.I.; Domínguez, M.I.; Lozano-Quilis, M.A. Ostracoda (Crustacea) as ecological indicators: A case study from Iberian Mediterranean brooks. Fundam. Appl. Limnol. Arch. Hydrobiol. 2001, 150, 545–560. [Google Scholar] [CrossRef]
- Gifre, J.; Quintana, X.D.; De La Barrera, R.; Martinoy, M.; Marquèz, E. Ecological factors affecting ostracod distribution in lentic ecosystems in the Empordà Wetlands (NE Spain). Fundam. Appl. Limnol. Arch. Hydrobiol. 2002, 154, 499–514. [Google Scholar] [CrossRef]
- Külköylüoğlu, O. On the usage of ostracods (Crustacea) as bioindicator species in different aquatic habitats in the Bolu region, Turkey. Ecol. Indic. 2004, 4, 139–147. [Google Scholar] [CrossRef]
- Yilmaz, F.; Külköylüoğlu, O. Tolerance, optimum ranges and ecological requirements of freshwater Ostracoda (Crustacea) in Lake Aladag (Bolu, Turkey). Ecol. Res. 2006, 21, 165–173. [Google Scholar] [CrossRef]
- Viehberg, F. Freshwater ostracod assemblages and their relationship to environmental variables in waters from northeast Germany. Hydrobiologia 2006, 571, 213–224. [Google Scholar] [CrossRef]
- Allen, P.E.; Dodson, S.I. Land use and ostracod community structure. Hydrobiologia 2011, 668, 203–219. [Google Scholar] [CrossRef]
- Pieri, V.; Vandekerkhove, J.; Goi, D. Ostracoda (Crustacea) as indicators for surface water quality: A case study from the Ledra River basin (NE Italy). Hydrobiologia 2010, 688, 25–35. [Google Scholar] [CrossRef]
- Coviaga, C.; Cusminsky, G.; Pérez, P. Ecology of freshwater ostracods from Northern Patagonia and their potential application in paleo-environmental reconstructions. Hydrobiologia 2017, 816, 3–20. [Google Scholar] [CrossRef]
- Akdemir, D.; Külköylüoğlu, O.; Yavuzatmaca, M.; Tanyeri, M.; Gürer, M.; Alper, A.; Dere, Ş.; Çelen, E.; Yilmaz, O.; Özcan, G. Ecological Characteristics And Habitat Preferences Of Ostracoda (Crustacea) With A New Bisexual Population Record (Muğla, Turkey). Appl. Ecol. Environ. Res. 2020, 18, 1471–1487. [Google Scholar] [CrossRef]
- Boomer, I.; Horne, D.J.; Slipper, I.J. The Use of Ostracods in Palaeoenvironmental Studies, or What can you do with an Ostracod Shell? Paléontol. Soc. Pap. 2003, 9, 153–180. [Google Scholar] [CrossRef]
- Rodriguez-Lazaro, J.; Ruiz-Muñoz, F. A General Introduction to Ostracods. Dev. Quat. Sci. 2012, 17, 1–14. [Google Scholar] [CrossRef]
- Smith, A.J.; Horne, D.J.; Martens, K.; Schön, I. Class Ostracoda. In Thorp and Covich’s Freshwater Invertebrates; Elsevier BV: Amsterdam, The Netherlands, 2015; pp. 757–780. [Google Scholar]
- APHA; AWWA; WPCF. Standard Methods for Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Koroleff, F. Direct determination of ammonia in natural waters as indophenol blue. Information on techniques and methods for seawater analysis. Inf. Tech. Methods Seawater Anal. 1970, 3, 19–22. [Google Scholar]
- Valderrama, J.C. The simultaneous analysis of total nitrogen and total phosphorus in natural waters. Mar. Chem. 1981, 10, 109–122. [Google Scholar] [CrossRef]
- González Mozo, M.E.; Martens, K.; Baltanás, Á. A taxonomic revision of European Herpetocypris Brady and Norman, 1889 (Crustacea, Ostracoda). Bull. Inst. R.Sc. Nat. Belg. Biol. 1996, 66, 93–132. [Google Scholar]
- Mazzini, I.; Gliozzi, E.; Rossetti, G.; Pieri, V. A multidisciplinary contribution to the Ilyocypris puzzle. Int. Rev. Hydrobiol. 2014, 99, 1–14. [Google Scholar] [CrossRef]
- Meisch, C. Freshwater Ostracoda of Western and Central Europe; Spektrum Academischer Verlag GmbH: Berlin/Heidelberg, Germany, 2000; 522p. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for education and data analysis. Palaeontol. Electron. 2001, 4, 2–9. [Google Scholar]
- De Luca, D.A.; Lasagna, M.; Debernardi, L. Hydrogeology of the western Po plain (Piedmont, NW Italy). J. Maps 2020, 16, 265–273. [Google Scholar] [CrossRef]
- Cantonati, M.; Segadelli, S.; Spitale, D.; Gabrieli, J.; Gerecke, R.; Angeli, N.; De Nardo, M.T.; Ogata, K.; Wehr, J.D. Geological and hydrochemical prerequisites of unexpectedly high biodiversity in spring ecosystems at the landscape level. Sci. Total. Environ. 2020, 740, 140157. [Google Scholar] [CrossRef] [PubMed]
- Balestrini, R.; Delconte, C.; Sacchi, E.; Buffagni, A. Groundwater-dependent ecosystems as transfer vectors of nitrogen from the aquifer to surface waters in agricultural basins: The fontanili of the Po Plain (Italy). Sci. Total. Environ. 2021, 753, 141995. [Google Scholar] [CrossRef] [PubMed]
- Racchetti, E.; Salmaso, F.; Pinardi, M.; Quadroni, S.; Soana, E.; Sacchi, E.; Severini, E.; Celico, F.; Viaroli, P.; Bartoli, M. Is Flood Irrigation a Potential Driver of River-Groundwater Interactions and Diffuse Nitrate Pollution in Agricultural Watersheds? Water 2019, 11, 2304. [Google Scholar] [CrossRef]
- Severini, E.; Bartoli, M.; Pinardi, M.; Celico, F. Reactive Silica Traces Manure Spreading in Alluvial Aquifers Affected by Nitrate Contamination: A Case Study in a High Plain of Northern Italy. Water 2020, 12, 2511. [Google Scholar] [CrossRef]
- Viaroli, P.; Nizzoli, D.; Pinardi, M.; Rossetti, G.; Bartoli, M. Factors Affecting Dissolved Silica Concentrations, and DSi and DIN Stoichiometry in a Human Impacted Watershed (Po River, Italy). Silicon 2012, 5, 101–114. [Google Scholar] [CrossRef]
- Bartoli, M.; Racchetti, E.; Delconte, C.A.; Sacchi, E.; Soana, E.; Laini, A.; Longhi, D.; Viaroli, P. Nitrogen balance and fate in a heavily impacted watershed (Oglio River, Northern Italy): In quest of the missing sources and sinks. Biogeosciences 2012, 9, 361–373. [Google Scholar] [CrossRef]
- Viaroli, P.; Soana, E.; Pecora, S.; Laini, A.; Naldi, M.; Fano, E.A.; Nizzoli, D. Space and time variations of watershed N and P budgets and their relationships with reactive N and P loadings in a heavily impacted river basin (Po river, Northern Italy). Sci. Total Environ. 2018, 639, 1574–1587. [Google Scholar] [CrossRef] [PubMed]
- Viaroli, P.; Bartoli, M.; Castaldelli, G.; Naldi, M.; Nizzoli, D.; Rossetti, G. Recent evolution and expected changes of nutrient loads in a heavily exploited watershed: The Po River, Italy. In Understanding Freshwater Quality Problems in a Changing World, Proceedings of H04, IAHS-IAPSO-IASPEI Assembly, Gothenburg, Sweden, 22–26 July 2013; Arheimer, B., Ed.; International Association of Hydrological Sciences: Wallingford, UK, 2013; Volume 361, pp. 175–182. [Google Scholar]
- Rosati, M.; Cantonati, M.; Primicerio, R.; Rossetti, G. Biogeography and relevant ecological drivers in spring habitats: A review on ostracods of the Western Palearctic. Int. Rev. Hydrobiol. 2014, 99, 409–424. [Google Scholar] [CrossRef]
- Rosati, M.; Cantonati, M.; Pieri, V.; Roca, J.R.; Mezquita, F.; Rossetti, G. Are aquatic assemblages from small water bodies more stochastic in dryer climates? An analysis of ostracod spring metacommunities. Hydrobiologia 2016, 793, 199–212. [Google Scholar] [CrossRef]
- Fea, G.; Nardi, P.A.; Ghia, D.; Spairani, M.; Manenti, R.; Rossi, S.; Moroni, M.; Bernini, F. Dati preliminari sulla distribuzione in dei gamberi d’acqua dolce autoctoni e alloctoni. Atti Soc. ital. sci. nat., Mus. civ. stor. nat. Milano 2006, 147, 201–210. [Google Scholar]
- Delmastro, G.B.; Balma, G.A.; Deandrea, G. L’ittiofauna Delle Risorgive in Piemonte Occidentale. The fishes of Lowland Springs in the Western Piedmont (North-Western Italy); Quaderni ETP-Ente Tutela Pesca del Friuli Venezia Giulia: Udine, Italy, 2006; Volume 34, pp. 9–19. [Google Scholar]
- Pezzoli, E. Notes on new or rare taxa of Crustaceans and Molluscs from a “fontanile” in Arzago d’Adda, Bergamo, Italy (Crustacea, Mollusca). Biodivers. J. 2010, 1, 45–55. [Google Scholar]
- Bischetti, G.; Fumagalli, N.; Gomarasca, S.; Pillitter, T.; Piantanida, E.; Senes, G.; Negri, G.; Marziali, L.; Toccolini, A. Tutela e valorizzazione dei fontanili del territorio lombardo. In Progetto FonTe; Quaderni della Ricerca n. 144; Regione Lombardia, D.G. Agricoltura: Milano, Italy, 2012; 102p. [Google Scholar]
- Stefani, F.; Schiavon, A.; Tirozzi, P.; Gomarasca, S.; Marziali, L. Functional response of fish communities in a multistressed freshwater world. Sci. Total Environ. 2020, 740, 139902. [Google Scholar] [CrossRef] [PubMed]

| EMILIA-ROMAGNA | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EC (mS cm−1) | TA (meq L−1) | DIN (mg N L−1) | DRSi (mg Si L−1) | ||||||||||
| N | 2001 | This study | SL | 2001 | This study | SL | 2001 | This study | SL | 2001 | This study | SL | |
| Arda-Ongina | 3 | 0.47 ± 0.09 | 0.74 ± 0.08 | * | 4.07 ± 0.06 | 6.57 ± 0.42 | ** | 7.33 ± 3.27 | 16.12 ± 6.01 | NS | 2.62 ± 0.22 | 2.70 ± 1.02 | NS |
| Chiavenna | 5 | 0.42 ± 0.04 | 0.70 ± 0.13 | ** | 3.39 ± 0.33 | 6.90 ± 1.06 | ** | 5.89 ± 1.62 | 8.00 ± 8.59 | NS | 1.68 ± 0.53 | 1.64 ± 0.44 | NS |
| Staffora-Luria-Versa-Coppa | 3 | 0.52 ± 0.03 | 0.86 ± 0.03 | ** | 4.93 ± 0.33 | 7.52 ± 0.34 | ** | 2.65 ± 0.56 | 13.64 ± 0.54 | ** | 5.29 ± 2.31 | 2.38 ± 0.78 | NS |
| Enza | 6 | 0.43 ± 0.11 | 0.70 ± 0.04 | ** | 3.80 ± 0.68 | 4.78 ± 0.65 | * | 3.13 ± 1.61 | 6.60 ± 3.40 | * | 2.08 ± 0.10 | 1.49 ± 0.19 | ** |
| Parma | 8 | 0.34 ± 0.09 | 0.60 ± 0.18 | ** | 3.03 ± 0.67 | 4.57 ± 1.14 | ** | 2.99 ± 3.81 | 5.72 ± 6.59 | NS | 6.19 ± 2.31 | 2.54 ± 0.78 | ** |
| Taro | 4 | 0.98 ± 0.63 | 1.27 ± 0.58 | NS | 4.11 ± 0.72 | 7.20 ± 0.31 | ** | 5.89 ± 1.67 | 13.58 ± 4.29 | * | 10.98 ± 1.71 | 3.68 ± 1.66 | ** |
| LOMBARDY | |||||||||||||
| EC (mS cm−1) | TA (meq L−1) | DIN (mg N L−1) | DRSi (mg Si L−1) | ||||||||||
| N | 2004 | This study | SL | 2004 | This study | SL | 2004 | This study | SL | 2004 | This study | SL | |
| Adda | 15 | 0.39 ± 0.04 | 0.45 ± 0.05 | ** | 3.53 ± 0.32 | 4.89 ± 0.68 | ** | 3.33 ± 0.93 | 2.94 ± 1.02 | NS | 2.83 ± 0.74 | 1.14 ± 0.23 | ** |
| Lambro-Olona meridionale | 4 | 0.43 ± 0.14 | 0.52 ± 0.21 | NS | 3.56 ± 0.97 | 5.06 ± 1.94 | NS | 5.22 ± 2.63 | 4.58 ± 2.44 | NS | 4.40 ± 1.74 | 1.25 ± 0.52 | * |
| Po | 2 | 0.52 ± 0.03 | 0.86 ± 0.03 | NA | 4.51 ± 0.08 | 6.91 ± 0.77 | NA | 16.28 ± 8.33 | 10.76 ± 8.85 | NA | 7.73 ± 2.09 | 2.10 ± 0.85 | NA |
| Site Code | Candona candida (O. F. Müller, 1776) | Cyclocypris laevis (O.F. Müller, 1776) | Cyclocypris ovum (Jurine, 1820) | Cypria ophthalmica (Jurine, 1820) | Cypridopsis vidua (O. F. Müller, 1776) | Herpetocypris brevicaudata Kaufmann, 1900 | Herpetocypris reptans (Baird, 1835) | Herpetocypris sp. | Heterocypris reptans (Kaufmann, 1900) | Heterocypris salina (Brady, 1868) | Ilyocypris bradyi Sars, 1890 | Ilyocypris gibba (Ramdohr, 1808) | Ilyocypris inermis Kaufmann, 1900 | Ilyocypris salebrosa Stepanaitys, 1960 | Neglecandona neglecta (Sars, 1887) | Neglecandona gr. neglecta | Notodromas persica Gurney, 1921 | Potamocypris smaragdina (Vávra, 1891) | Prionocypris zenkeri (Chyzer & Toth, 1858) | Pseudocandona albicans (Brady, 1864) | Pseudocandona compressa (Koch, 1838) | Pseudocandona lobipes (Hartwig, 1900) | Pseudocandona pratensis (Hartwig, 1901) | Pseudocandona sp. | Scottia pseudobrowniana Kempf, 1971 | Taxonomic Richness 2015–2016 | Variation in Taxonomic Richness |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PC03 | B | C | A | 2 | = | ||||||||||||||||||||||
| PC10 | A | A | 2 | = | |||||||||||||||||||||||
| PC18 | B | A | 1 | –1 | |||||||||||||||||||||||
| PC24 | A | 1 | = | ||||||||||||||||||||||||
| PC32 | B | B | 0 | −2 | |||||||||||||||||||||||
| PC44 | A | A | C | 3 | +1 | ||||||||||||||||||||||
| PC46 | A | A | 2 | = | |||||||||||||||||||||||
| PC47 | A | C | B | 2 | = | ||||||||||||||||||||||
| PC51 | A | C | B | 2 | = | ||||||||||||||||||||||
| PC75 | A | 1 | = | ||||||||||||||||||||||||
| PC76 | A | C | 2 | +1 | |||||||||||||||||||||||
| PR01 | A | 1 | = | ||||||||||||||||||||||||
| PR02 | A | 1 | = | ||||||||||||||||||||||||
| PR03 | A | C | C | C | 4 | +3 | |||||||||||||||||||||
| PR04 | A | 1 | = | ||||||||||||||||||||||||
| PR06 | C | A | B | C | 3 | +1 | |||||||||||||||||||||
| PR09 | B | C | A | 3 | = | ||||||||||||||||||||||
| PR10 | B | A | B | C | C | A | 4 | = | |||||||||||||||||||
| PR13 | C | A | A | 3 | +1 | ||||||||||||||||||||||
| PR14 | B | C | A | A | C | A | 5 | +1 | |||||||||||||||||||
| PR17 | A | B | C | A | C | C | 5 | +2 | |||||||||||||||||||
| PR20 | A | B | B | C | B | 3 | −1 | ||||||||||||||||||||
| PR26 | A | C | A | 3 | +1 | ||||||||||||||||||||||
| PR27 | B | A | 1 | −1 | |||||||||||||||||||||||
| PR29 | B | A | C | 1 | = | ||||||||||||||||||||||
| PR31 | C | A | 2 | +1 | |||||||||||||||||||||||
| PR32 | B | C | A | 2 | = | ||||||||||||||||||||||
| PR33 | B | C | A | 2 | = | ||||||||||||||||||||||
| PR39 | A | B | B | 1 | -2 | ||||||||||||||||||||||
| S01 | C | C | 2 | +2 | |||||||||||||||||||||||
| S02 | B | A | C | B | C | B | 3 | −1 | |||||||||||||||||||
| S03 | A | B | B | A | 2 | −2 | |||||||||||||||||||||
| S05 | C | A | A | B | B | 3 | −1 | ||||||||||||||||||||
| S06 | B | B | C | C | B | C | B | 3 | −1 | ||||||||||||||||||
| S07 | B | B | C | B | A | 2 | −2 | ||||||||||||||||||||
| S08 | B | B | B | C | B | B | 1 | −4 | |||||||||||||||||||
| S09 | B | A | C | B | C | C | B | A | 5 | = | |||||||||||||||||
| S11 | B | C | B | B | B | 1 | –3 | ||||||||||||||||||||
| S12 | B | B | A | A | B | A | 3 | −3 | |||||||||||||||||||
| S13 | B | C | A | C | 3 | +1 | |||||||||||||||||||||
| S15 | C | C | 2 | +2 | |||||||||||||||||||||||
| S17 | B | B | C | B | 1 | −2 | |||||||||||||||||||||
| S21 | B | A | A | B | A | 3 | = | ||||||||||||||||||||
| S22 | A | B | B | 1 | –2 | ||||||||||||||||||||||
| S23 | A | A | A | C | 4 | +1 | |||||||||||||||||||||
| S24 | A | A | C | B | C | 4 | +1 | ||||||||||||||||||||
| S25 | B | A | A | 2 | −1 | ||||||||||||||||||||||
| S26 | A | B | A | A | 3 | −1 | |||||||||||||||||||||
| S27 | C | B | B | B | B | C | B | C | 3 | −2 | |||||||||||||||||
| S28 | B | A | B | B | 1 | −3 | |||||||||||||||||||||
| Σ | 1 | 4 | 5 | 37 | 14 | 5 | 7 | 1 | 1 | 1 | 7 | 2 | 4 | 0 | 0 | 3 | 5 | 1 | 11 | 0 | 0 | 2 | 0 | 0 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossetti, G.; Pieri, V.; Bolpagni, R.; Nizzoli, D.; Viaroli, P. Variability in Environmental Conditions Strongly Impacts Ostracod Assemblages of Lowland Springs in a Heavily Anthropized Area. Water 2020, 12, 3276. https://doi.org/10.3390/w12113276
Rossetti G, Pieri V, Bolpagni R, Nizzoli D, Viaroli P. Variability in Environmental Conditions Strongly Impacts Ostracod Assemblages of Lowland Springs in a Heavily Anthropized Area. Water. 2020; 12(11):3276. https://doi.org/10.3390/w12113276
Chicago/Turabian StyleRossetti, Giampaolo, Valentina Pieri, Rossano Bolpagni, Daniele Nizzoli, and Pierluigi Viaroli. 2020. "Variability in Environmental Conditions Strongly Impacts Ostracod Assemblages of Lowland Springs in a Heavily Anthropized Area" Water 12, no. 11: 3276. https://doi.org/10.3390/w12113276
APA StyleRossetti, G., Pieri, V., Bolpagni, R., Nizzoli, D., & Viaroli, P. (2020). Variability in Environmental Conditions Strongly Impacts Ostracod Assemblages of Lowland Springs in a Heavily Anthropized Area. Water, 12(11), 3276. https://doi.org/10.3390/w12113276






