Removal of Molybdenum(VI) from Raw Water Using Nano Zero-Valent Iron Supported on Activated Carbon
Abstract
1. Introduction
2. Experimental
2.1. NZVI/AC; Synthesis and Characterization
2.2. The Simulation Water and Raw Water Quality Parameters of a Reservoir
2.3. Adsorption Kinetics
2.4. Models
2.5. The Influence of pH on the Adsorption Behavior of NZVI/AC
2.6. Effect of Humic Acid and Coexisting Ions
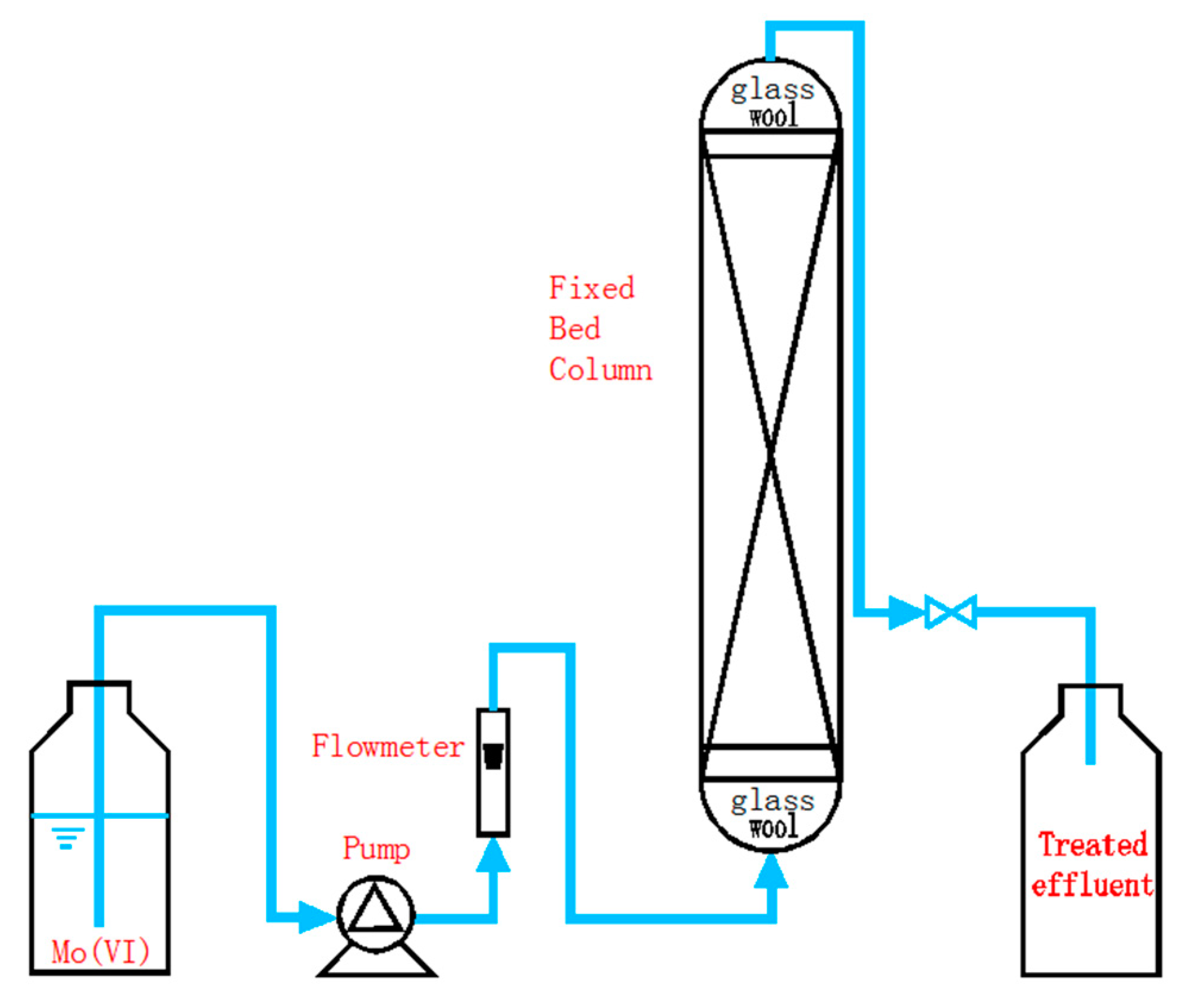
2.7. Column Experiments and Regeneration
3. Results and Discussion
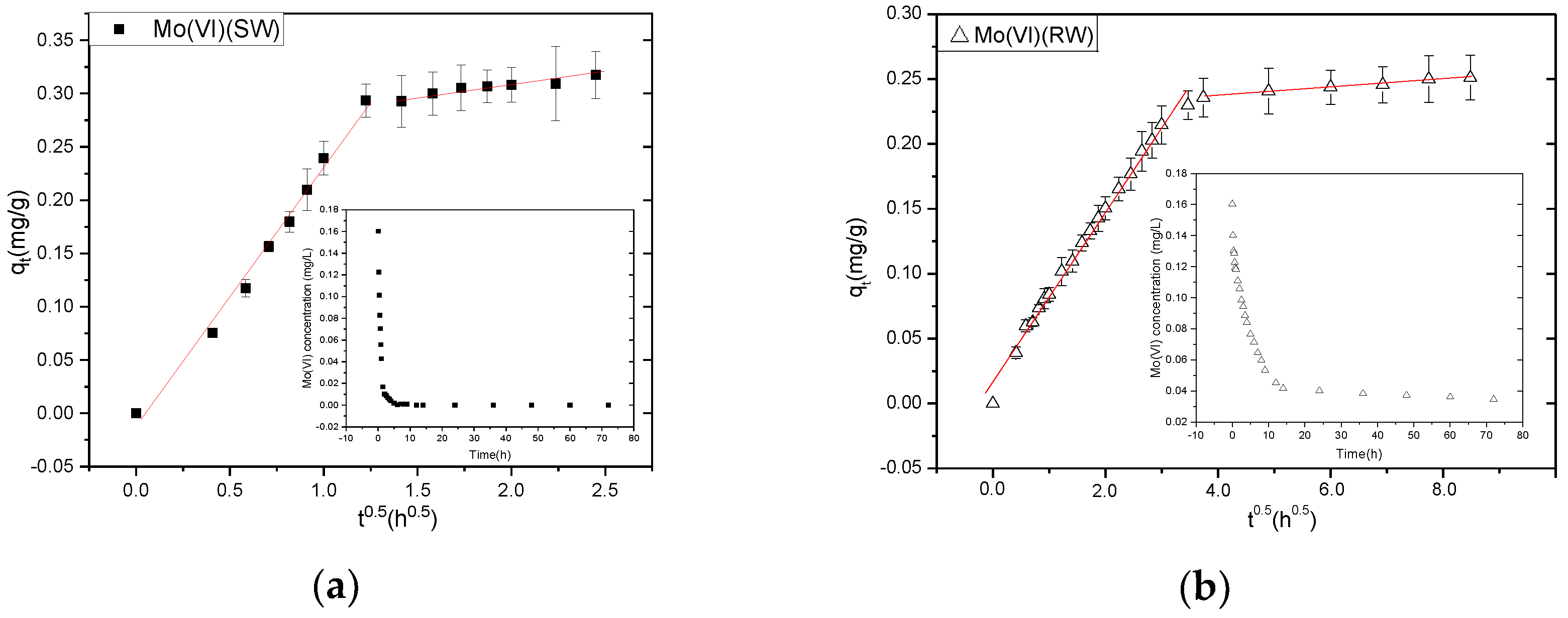
3.1. Adsorption Kinetics
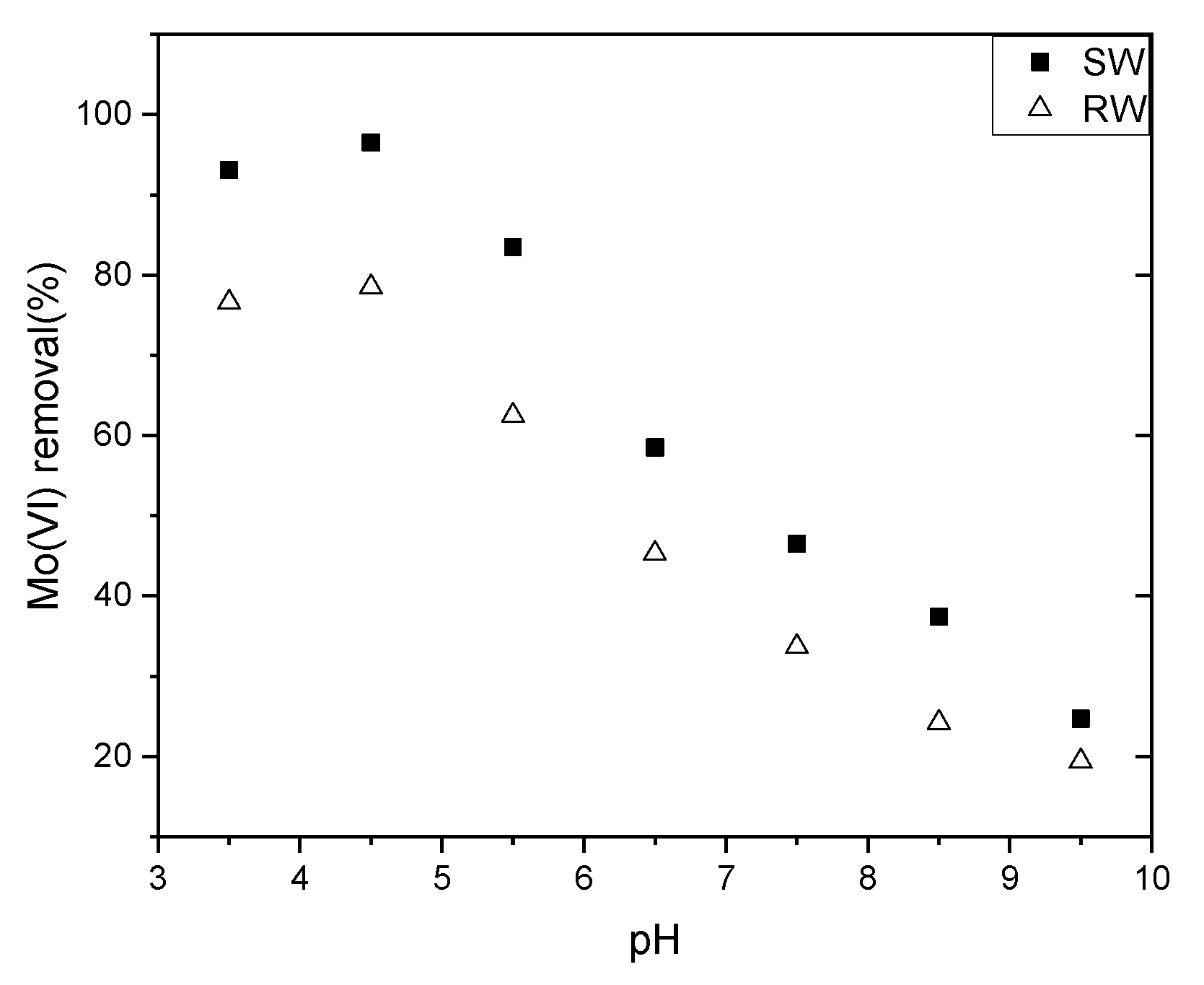
3.2. Effect of pH
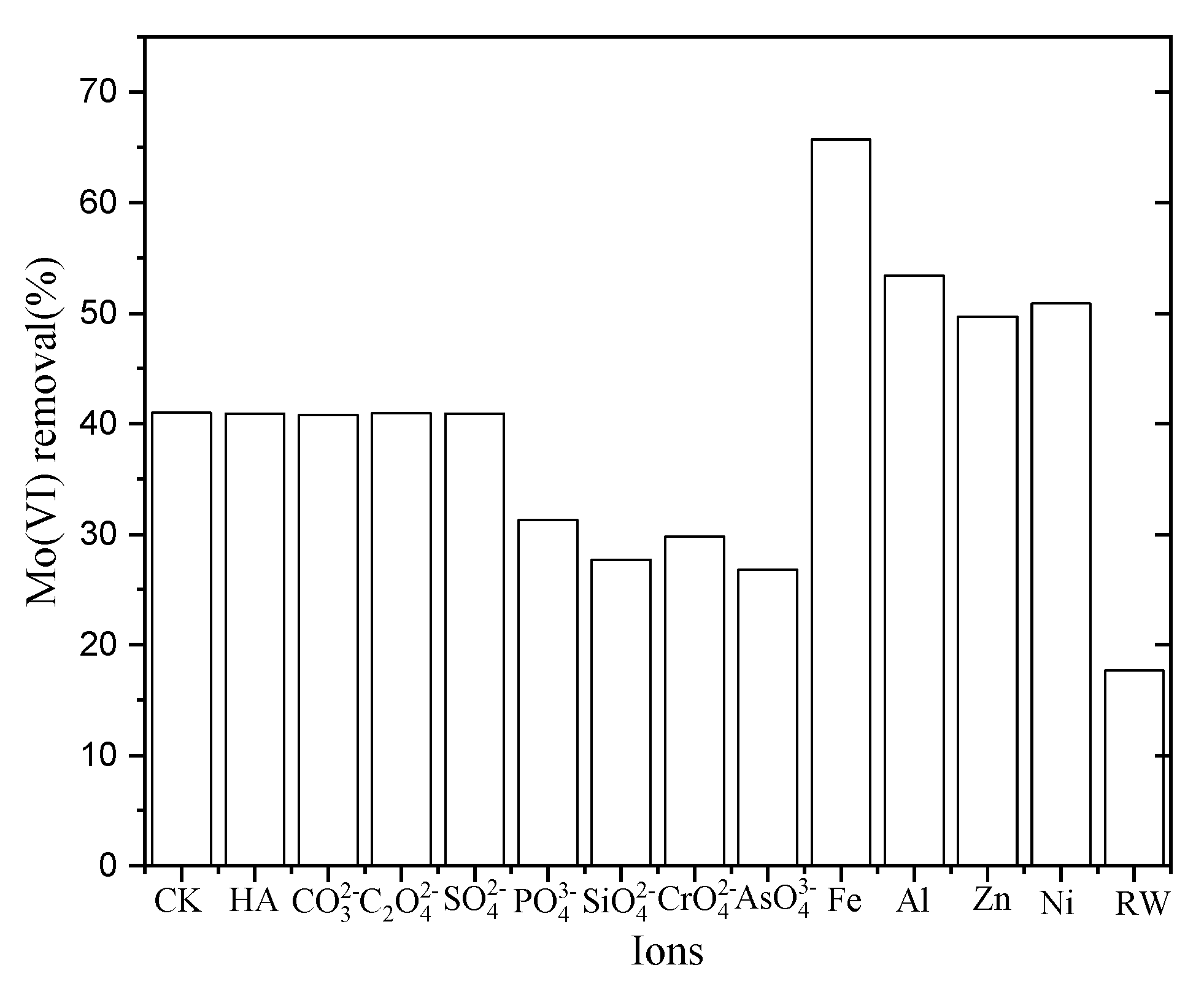
3.3. Effect of Coexisting Ions
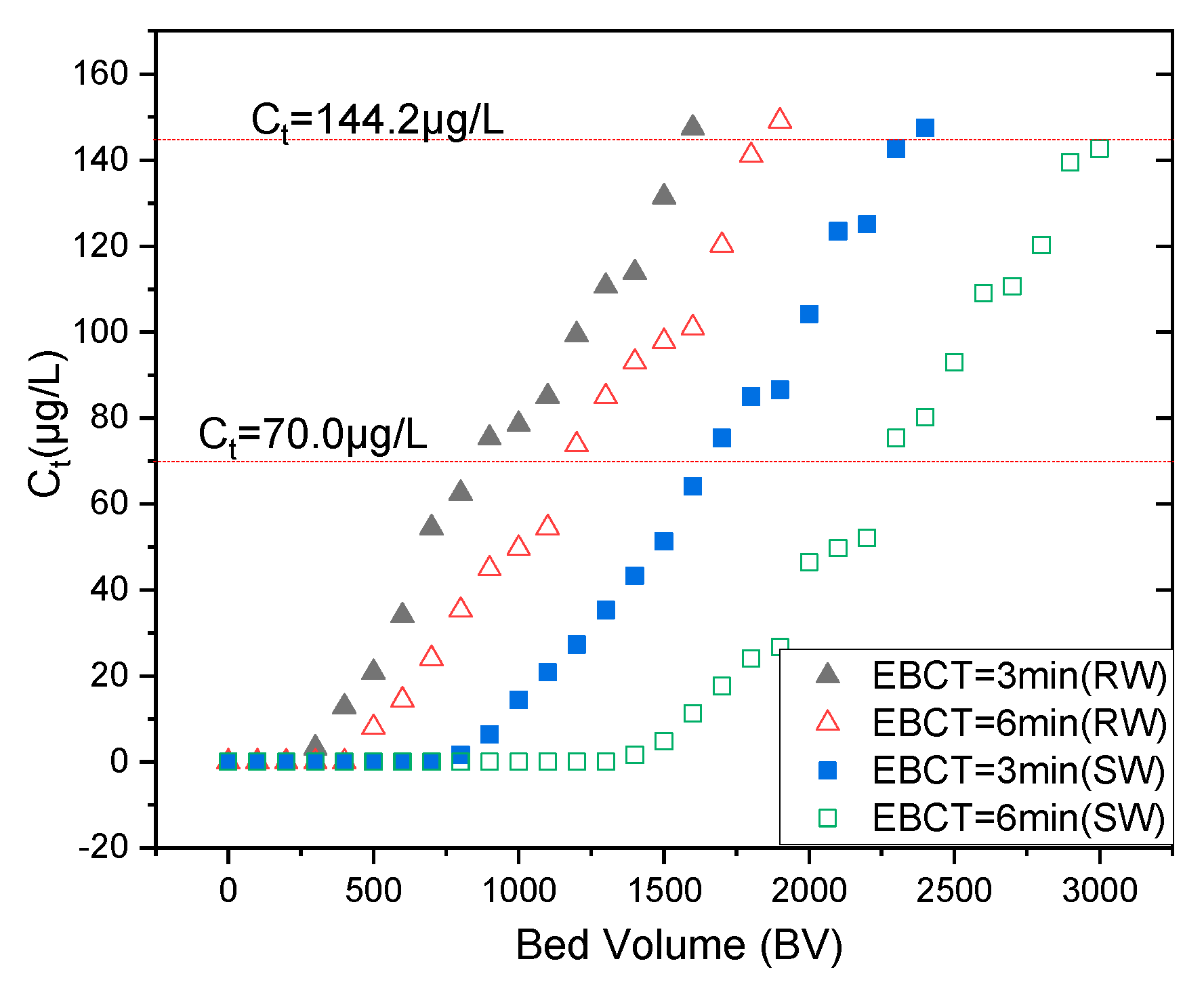
3.4. Fixed-Bed Column Runs
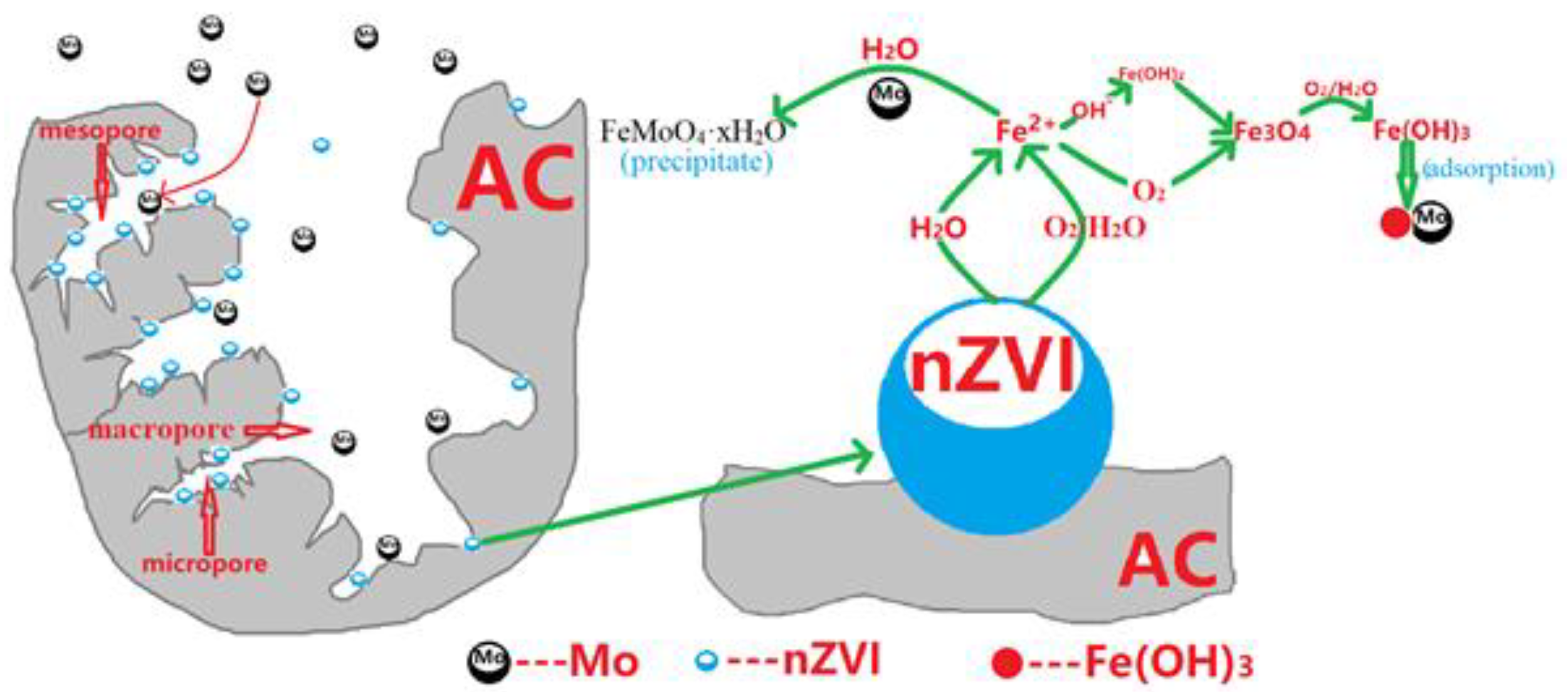
3.5. Mechanism of Removing Mo(VI) from Water by NZVI/AC
- (1)
- Fe0 reacts with water or dissolved oxygen to form Fe2+:
- (2)
- Fe2+ is further transformed into iron (hydrated) oxides by the pH of the solution and the oxidation-reduction potential and other factors:
Fe2+ + 2OH−→2Fe(OH)2 (s)
6Fe(OH)2(s) + O2→2Fe3O4(s) + 6H2O
Fe3O4(s) + O2(aq) + 18H2O⇌12Fe(OH)3(s)
Fe2− + MoO42− + xH2O→FeMoO4·xH2O
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Orrego, P.; Hernández, J.; Reyes, A. Uranium and molybdenum recovery from copper leaching solutions using ion exchange. Hydrometallurgy 2019, 184, 116–122. [Google Scholar] [CrossRef]
- Pradhan, D.; Kim, D.J.; Sukla, L.B.; Pattanaik, A.; Lee, S.W. Evaluation of molybdenum recovery from sulfur removed spent catalyst using leaching and solvent extraction. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ma, J. Study on the efficient removal of molybdenum ions from water by polyferric chloride. Water Supply Wastewater 2016, 42, 41–45. [Google Scholar]
- Sabullah, M.K.; Rahman, M.F.; Ahmad, S.A.; Sulaiman, M.S.; Shukor, M.S.; Shamaan, N.A.; Shukor, M.Y. Assessing Resistance and Bioremediation Ability of Enterobacter sp. Strain Saw-1 on Molybdenum in Various Heavy Metals and Pesticides. J. Math. Fundam. Sci. 2017, 49, 193–210. [Google Scholar] [CrossRef]
- Lian, J.J.; Huang, Y.G.; Chen, B.; Wang, S.S.; Wang, P.; Niu, S.P.; Liu, Z.L. Removal of molybdenum(VI) from aqueous solutions using nano zero-valent iron supported on biochar enhanced by cetyl-trimethyl ammonium bromide: Adsorption kinetic, isotherm and mechanism studies. Water Sci. Technol. 2018, 2017, 859–868. [Google Scholar] [CrossRef]
- Toli, A.; Mystrioti, C.; Xenidis, A.; Papassiopi, N. Continuous Flow Process for Cr(VI) Removal from Aqueous Solutions Using Resin Supported Zero-Valent Iron. Bull. Environ. Contam. Toxicol. 2020, 1–6. [Google Scholar] [CrossRef]
- Ito, T.; Kim, S.Y. Adsorption and separation behaviors of molybdenum from high-level liquid waste using a silica-based hydroxyoxime impregnated adsorbent. J. Radioanal. Nucl. Chem. 2018, 316, 1165–1172. [Google Scholar] [CrossRef]
- Fumihiko, O.; Takehiro, N.; Naohito, K. Adsorption capability of virgin and calcined wheat bran for molybdenum present in aqueous solution and elucidating the adsorption mechanism by adsorption isotherms, kinetics, and regeneration. J. Environ. Chem. Eng. 2018, 6, 4459–4466. [Google Scholar]
- Denkova, A.G.; Terpstra, B.E.; Steinbach, O.M.; Dam, J.; Wolterbeek, H.T. Adsorption of Molybdenum on Mesoporous Aluminum Oxides for Potential Application in Nuclear Medicine. Sep. Sci. Technol. 2013, 48, 1331–1338. [Google Scholar] [CrossRef]
- Chen, Y.C.; Lu, C. Kinetics, thermodynamics and regeneration of molybdenum adsorption in aqueous solutions with NaOCl-oxidized multiwalled carbon nanotubes. J. Ind. Eng. Chem. 2014, 20, 2521–2527. [Google Scholar] [CrossRef]
- Pang, X.; Yang, J.; Pang, M.; He, J.; Yang, W.; Qin, H.; Zhan, Y. Adsorption and migration behavior of molybdenum atom on graphite (0001) surface. Appl. Surf. Sci. 2019, 470, 1064–1070. [Google Scholar] [CrossRef]
- Lian, J.; Zhou, F.; Chen, B.; Yang, M.; Wang, S.; Liu, Z.; Niu, S. Enhanced adsorption of molybdenum(VI) onto drinking water treatment residues modified by thermal treatment and acid activation. J. Clean. Prod. 2020, 244, 118719. [Google Scholar] [CrossRef]
- Sun, W.; Selim, M. Kinetic Modeling of pH-Dependent Molybdenum (VI) Adsorption and Desorption on Iron Oxide-Coated Sand. Soil Sci. Soc. Am. J. 2019, 83, 357–365. [Google Scholar] [CrossRef]
- Morrison, S.; Mushovic, P.; Niesen, P. Early breakthrough of molybdenum and uranium in a permeable reactive barrier. Environ. Sci. Technol. 2006, 40, 2018–2024. [Google Scholar] [CrossRef]
- Zhu, H.; Jia, Y.; Wu, X.; Wang, H. Removal of arsenic from water by supported nano zero-valent iron on activated carbon. J. Hazard. Mater. 2009, 172, 1591–1596. [Google Scholar] [CrossRef]
- Hameed, B.H.; Ahmad, A.A.; Aziz, N. Isotherms, kinetics and thermodynamics of acid dye adsorption on activated palm ash. Chem. Eng. J. 2007, 133, 195–203. [Google Scholar] [CrossRef]
- Ong, S.A.; Seng, C.E.; Lim, P. Kinetics of adsorption of Cu (II) and Cd (II) from aqueous solution on rice husk and modified rice husk. Electron. J. Environ. Agric. Food Chem. 2007, 6, 1764–1774. [Google Scholar]
- Shi, L.; Zhou, Y.; Chen, Z. Simultaneous adsorption and degradation of Zn2+ and Cu2+ from wastewaters using nanoscale zero-valent iron impregnated with clays. Environ. Sci. Pollut. Res. 2013, 20, 3639–3648. [Google Scholar] [CrossRef]
- Guo, X.; Chen, F. Removal of arsenic by bead cellulose loaded with iron oxyhydroxide from groundwater. Environ. Sci. Technol. 2005, 39, 6808–6818. [Google Scholar] [CrossRef]
- Jia, Y.; Demopoulos, G.P. Adsorption of arsenate onto ferrihydrite from aqueous solution: Influence of media (sulfate vs nitrate), added gypsum, and pH alteration. Environ. Sci. Technol. 2005, 39, 9523–9527. [Google Scholar] [CrossRef]
- Giasuddin, A.B.M.; Kanel, S.R.; Choi, H. Adsorption of humic acid onto nanoscale zerovalent iron and its effect on arsenic removal. Environ. Sci. Technol. 2007, 41, 2022–2027. [Google Scholar] [CrossRef] [PubMed]
- Wilkie, J.A.; Hering, J.G. Adsorption of arsenic onto hydrous ferric oxide: Effects of adsorbate/adsorbent ratios and co-occurring solutes. Colloids Surf. A Physicochem. Eng. Asp. 1996, 107, 97–110. [Google Scholar] [CrossRef]
- Jiang, Z.X.; Zhan, J.Q.; Song, Z.X. Ion Exchanger Engineering; Tianjin University Press: Tianjin, China, 1992; p. 206. [Google Scholar]
- Manning, B.A.; Hunt, M.L.; Amrhein, C.; Yarmoff, J.A. Arsenic (III) and arsenic (V) reactions with zerovalent iron corrosion products. Environ. Sci. Technol. 2002, 36, 5455–5461. [Google Scholar] [CrossRef]
- Wang, Y. Research on the Removal of Molybdenum from Water by Zero-Valent Iron; Dalian University of Technology: Dalian, China, 2006. (In Chinese) [Google Scholar]
- Huang, Y.H.; Tang, C.; Zeng, H. Removing molybdate from water using a hybridized zero-valent iron/magnetite/Fe(II) treatment system. Chem. Eng. J. 2012, 200–202, 257–263. [Google Scholar] [CrossRef]
| Thickness | Shape | Diameter | Fe Content | Total Pore Volume | BET Surface Area |
|---|---|---|---|---|---|
| ~20 nm | flakes | <100 nm | ~8.2% | 0.45 cm3/g | 821.7 m2/g |
| Element | Mo | As | Cd | Cr | Cu | Fe | Hg | Mn | Al |
|---|---|---|---|---|---|---|---|---|---|
| Average value | 160.3 | 3.29 | 0.742 | 8.01 | 4.12 | 38.9 | 0.029 | 1.53 | 157.8 |
| Element | Ni | Pb | Zn | pH | NH3-N | TN | TP | CODCr | BOD5 |
| Average value | 6.90 | 0.59 | 8.69 | 8.13 | 186 | 2043 | 19.2 | 2027 | 983 |
| Parameter | Weber–Morris Diffusion | |||
|---|---|---|---|---|
| 1st Step | 2nd Step | |||
| C0 (0.1603mg/L) | kid1 | R2 | kid2 | R2 |
| simulation water | 0.2405 | 0.9908 | 0.02006 | 0.9906 |
| raw water | 0.0654 | 0.9908 | 0.00292 | 0.9950 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, H.; Huang, Q.; Fu, S.; Zhang, X.; Shi, M.; Liu, B. Removal of Molybdenum(VI) from Raw Water Using Nano Zero-Valent Iron Supported on Activated Carbon. Water 2020, 12, 3162. https://doi.org/10.3390/w12113162
Zhu H, Huang Q, Fu S, Zhang X, Shi M, Liu B. Removal of Molybdenum(VI) from Raw Water Using Nano Zero-Valent Iron Supported on Activated Carbon. Water. 2020; 12(11):3162. https://doi.org/10.3390/w12113162
Chicago/Turabian StyleZhu, Huijie, Qiang Huang, Shuai Fu, Xiuji Zhang, Mingyan Shi, and Bo Liu. 2020. "Removal of Molybdenum(VI) from Raw Water Using Nano Zero-Valent Iron Supported on Activated Carbon" Water 12, no. 11: 3162. https://doi.org/10.3390/w12113162
APA StyleZhu, H., Huang, Q., Fu, S., Zhang, X., Shi, M., & Liu, B. (2020). Removal of Molybdenum(VI) from Raw Water Using Nano Zero-Valent Iron Supported on Activated Carbon. Water, 12(11), 3162. https://doi.org/10.3390/w12113162





