Reforestation Based on Mono-Plantation of Fast-Growing Tree Species Make It Difficult to Maintain (High) Soil Water Content in Tropics, a Case Study in Hainan Island, China
Abstract
1. Introduction
2. Materials and Methods
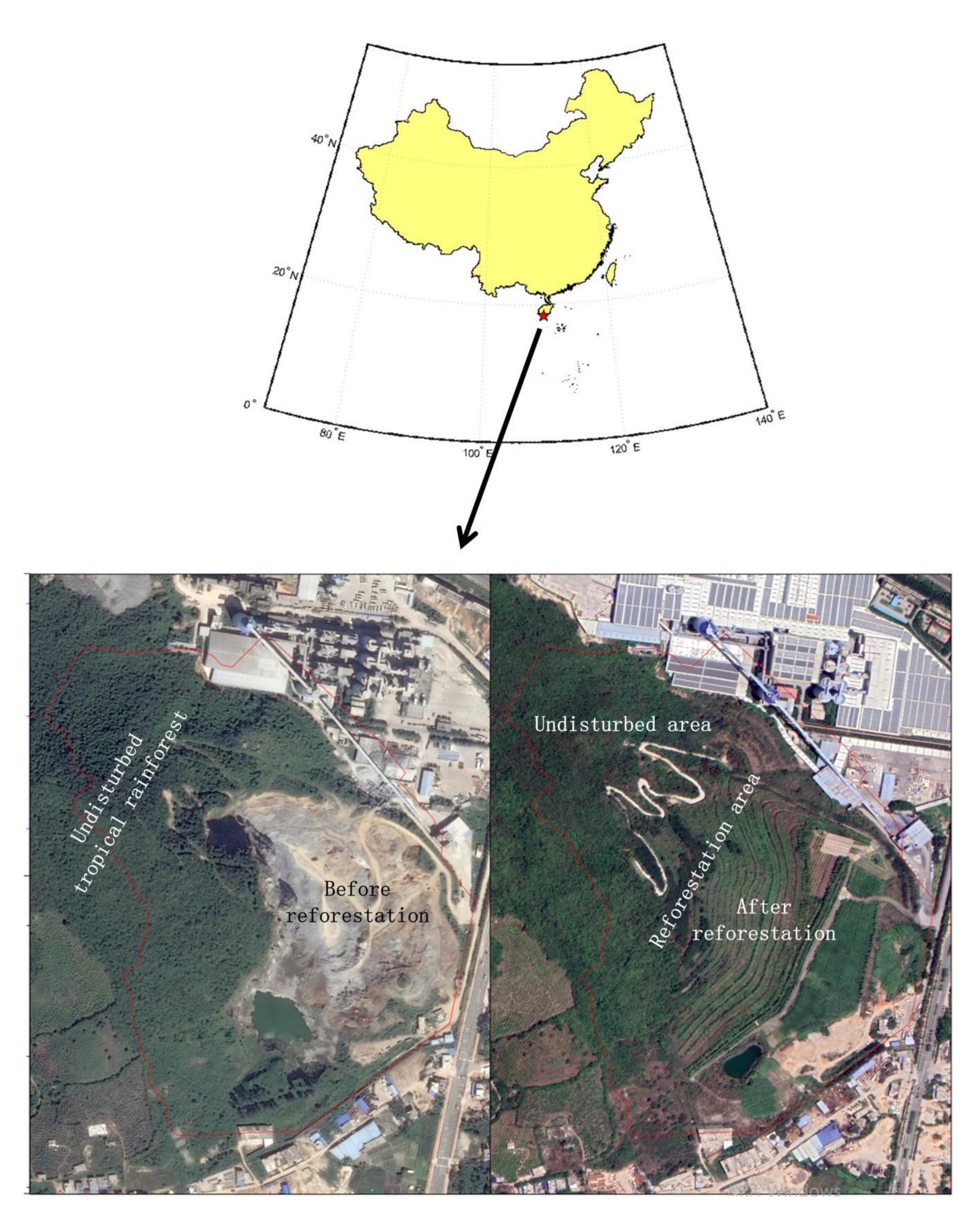
2.1. Study Sites
2.2. Sampling
2.3. Statistics Methods
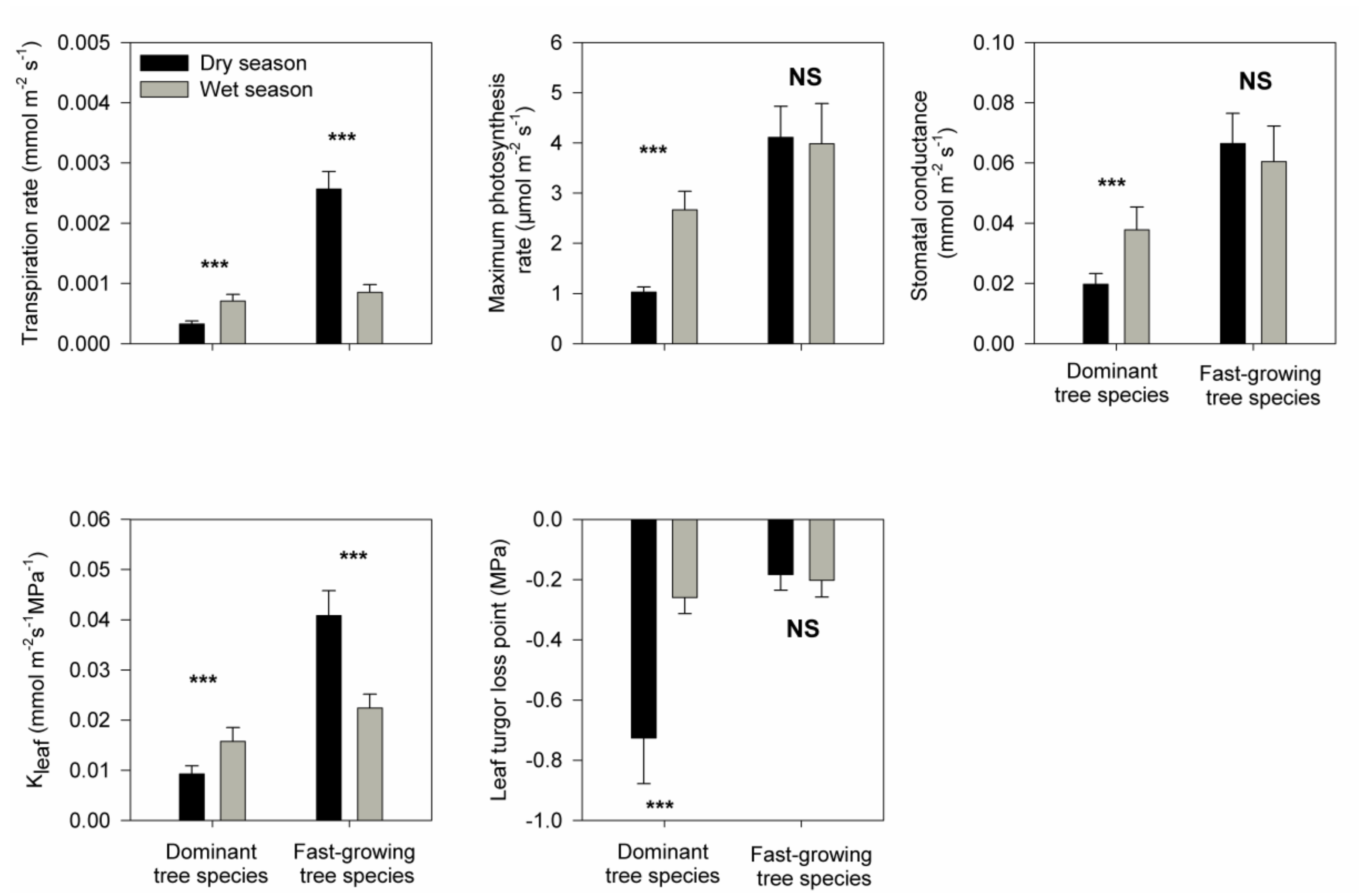
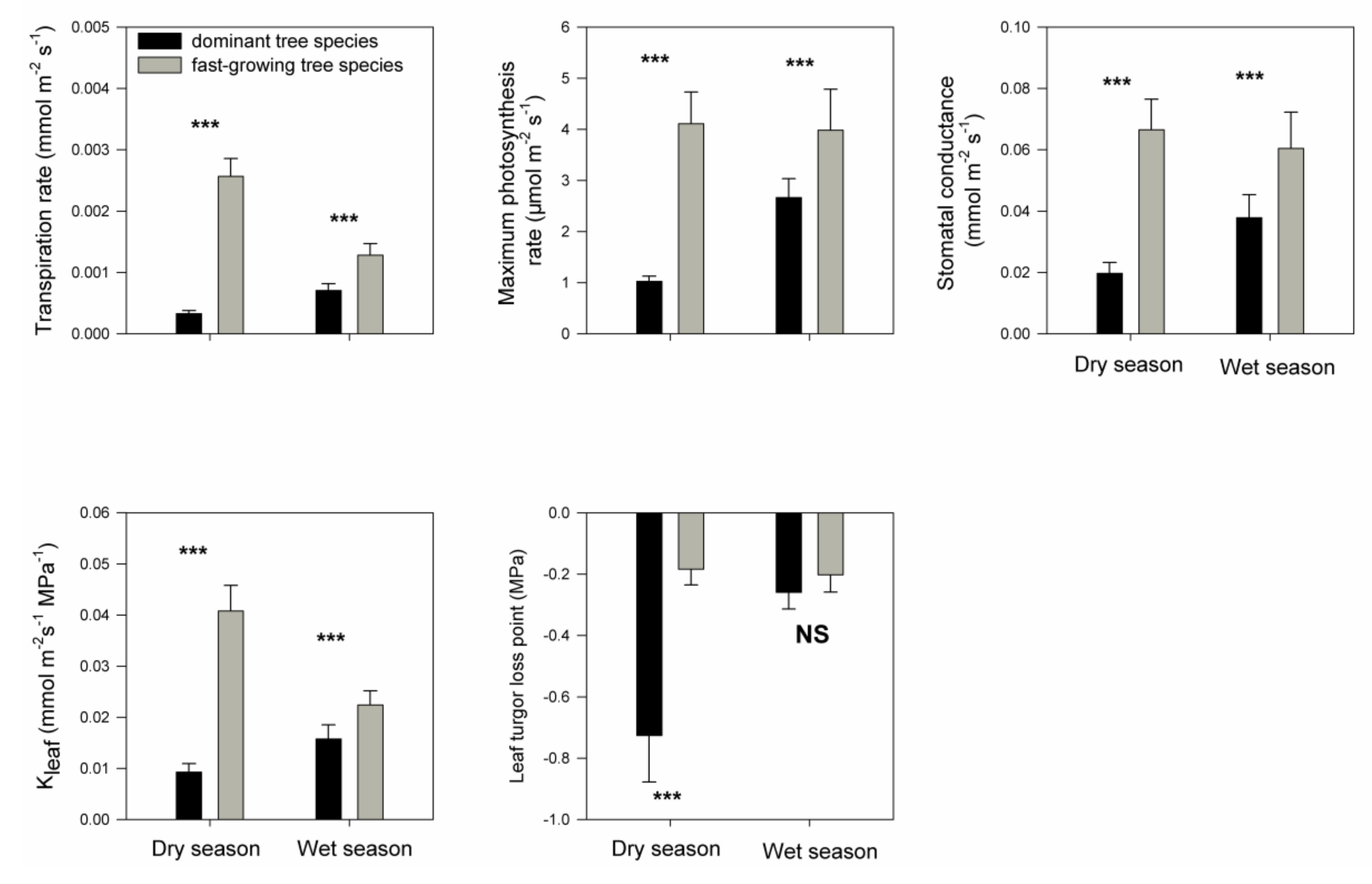
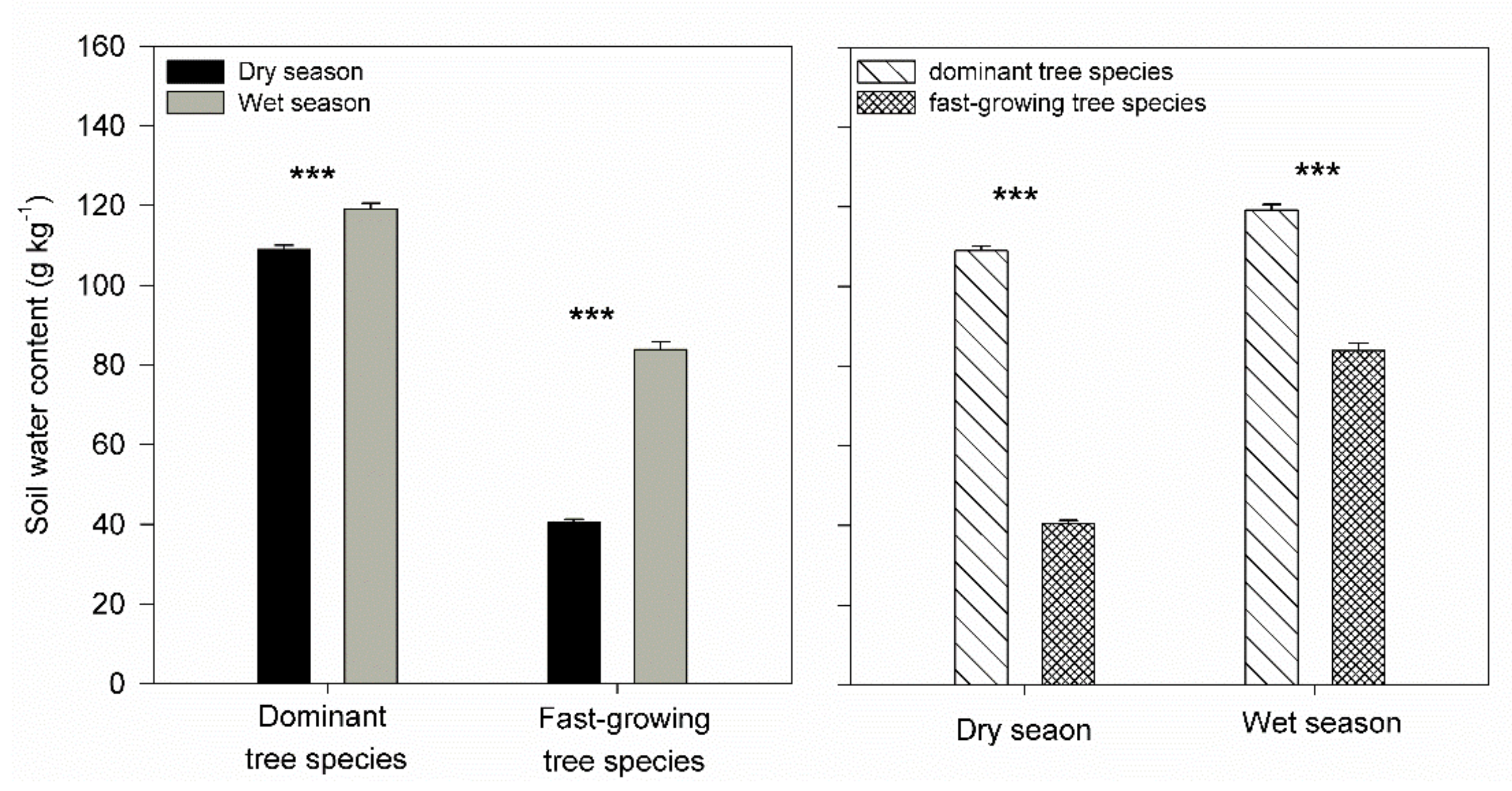
3. Results
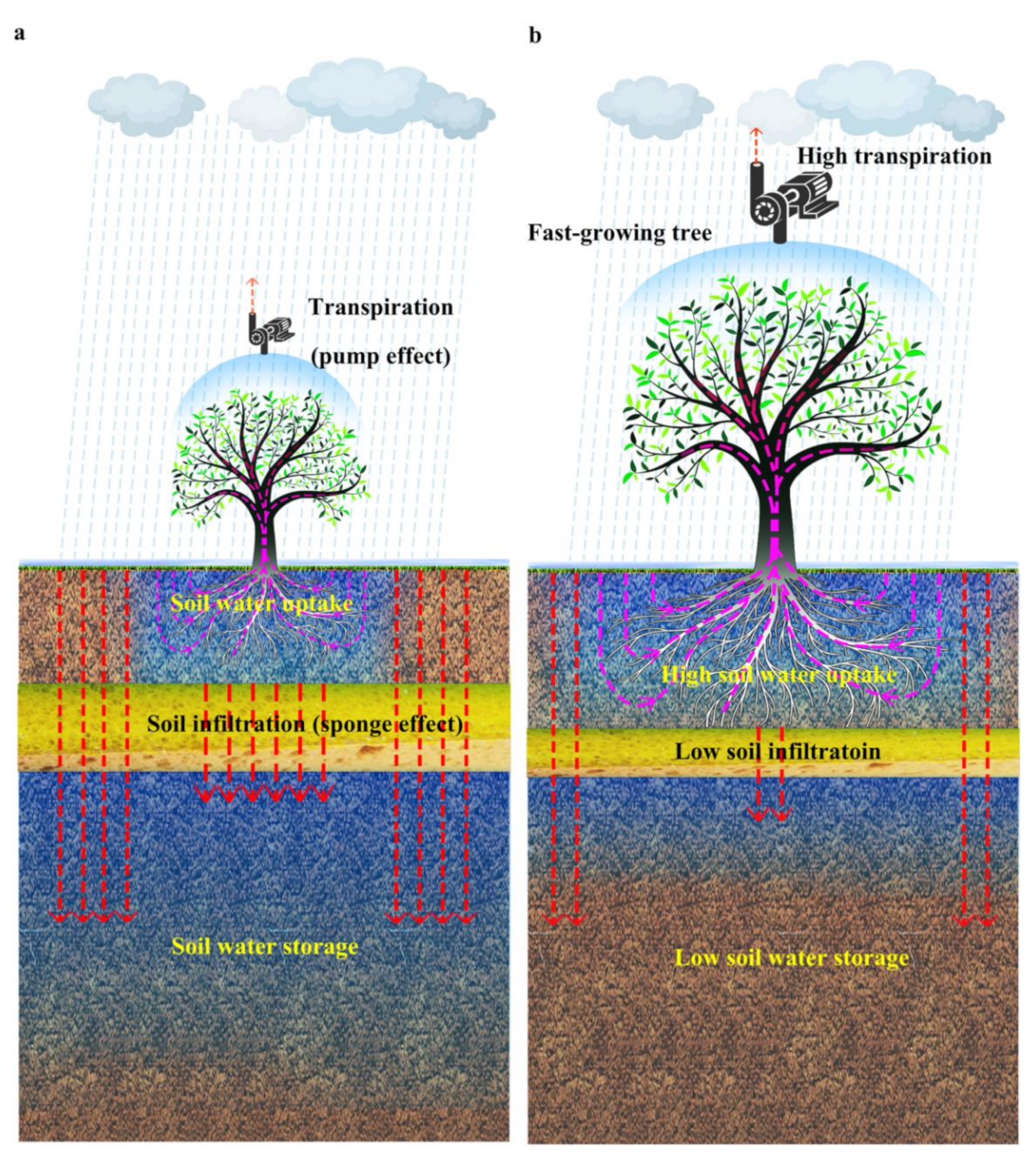
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mekonnen, M.M.; Hoekstra, A.Y. Four billion people facing severe water scarcity. Sci. Adv. 2016, 2, e1500323. [Google Scholar] [CrossRef] [PubMed]
- Famiglietti, J.S.; Rodell, M. Water in the Balance. Science 2013, 340, 1300. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Wei, X.; Chen, X.; Zhou, P.; Liu, X.; Xiao, Y.; Sun, G.; Scott, D.F.; Zhou, S.; Han, L.; et al. Global pattern for the effect of climate and land cover on water yield. Nat. Commun. 2015, 6, 5918. [Google Scholar] [CrossRef] [PubMed]
- Sahin, V.; Hall, M.J. The effects of afforestation and deforestation on water yields. J. Hydrol. 1996, 178, 293–309. [Google Scholar] [CrossRef]
- Lambin, E.F.; Meyfroidt, P. Global land use change, economic globalization, and the looming land scarcity. Proc. Natl. Acad. Sci. USA 2011, 108, 3465–3472. [Google Scholar] [CrossRef] [PubMed]
- Vörösmarty, C.J.; Hoekstra, A.Y.; Bunn, S.E.; Conway, D.; Gupta, J. Fresh water goes global. Science 2015, 349, 478. [Google Scholar] [CrossRef] [PubMed]
- Castello, L.; Macedo, M.N. Large-scale degradation of Amazonian freshwater ecosystems. Glob. Chang. Biol. 2016, 22, 990–1007. [Google Scholar] [CrossRef]
- Solange, F.; Ometto, B.M.; Weiss, K.C.B.; Palmer, M.A.; Silva, L.C.R. Impacts of forest restoration on water yield: A systematic review. PLoS ONE 2017, 12, e0183210. [Google Scholar]
- Peña-Arancibia, J.L.; Bruijnzeel, L.A.; Mulligan, M.; van Dijk, A.I.J.M. Forests as ‘sponges’ and ‘pumps’: Assessing the impact of deforestation on dry-season flows across the tropics. J. Hydrol. 2019, 574, 946–963. [Google Scholar] [CrossRef]
- Bruijnzeel, L.A. Hydrological functions of tropical forests: Not seeing the soil for the trees? Agric. Ecosyst. Environ. 2004, 104, 185–228. [Google Scholar] [CrossRef]
- Tardieu, F.; Parent, B. Predictable ‘meta-mechanisms’ emerge from feedbacks between transpiration and plant growth and cannot be simply deduced from short-term mechanisms. Plant Cell Environ. 2017, 40, 846–857. [Google Scholar] [CrossRef] [PubMed]
- Krishnaswamy, J.; Bonell, M.; Venkatesh, B.; Purandara, B.K.; Rakesh, K.N.; Lele, S.; Kiran, M.C.; Reddy, V.; Badiger, S. The groundwater recharge response and hydrologic services of tropical humid forest ecosystems to use and reforestation: Support for the “infiltration-evapotranspiration trade-off hypothesis”. J. Hydrol. 2013, 498, 191–209. [Google Scholar] [CrossRef]
- Tuzet, A.; Perrier, A.; Leuning, R. A coupled model of stomatal conductance, photosynthesis and transpiration. Plant Cell Environ. 2003, 26, 1097–1116. [Google Scholar] [CrossRef]
- Gebrehiwot, S.G.; Ellison, D.; Bewket, W.; Seleshi, Y.; Inogwabini, B.-I.; Bishop, K. The Nile Basin waters and the West African rainforest: Rethinking the boundaries. Wiley Interdiscip. Rev. Water 2019, 6, e1317. [Google Scholar] [CrossRef]
- Mapulanga, A.M.; Naito, H. Effect of deforestation on access to clean drinking water. Proc. Natl. Acad. Sci. USA 2019, 116, 8249. [Google Scholar] [CrossRef] [PubMed]
- Guidicini, G.; Iwasa, O.Y. Tentative correlation between rainfall and landslides in a humid tropical environment. Bull. Eng. Geol. Environ. 1977, 16, 13–20. [Google Scholar] [CrossRef]
- Chang, K.T.; Chiang, S.H.; Lei, F. Analysing the Relationship between Typhoon-Triggered Landslides and Critical Rainfall Conditions. Earth Surf. Proc. Land. 2008, 33, 1261–1271. [Google Scholar] [CrossRef]
- Yumul, J.G.P.; Servando, N.T.; Suerte, L.O.; Magarzo, M.Y.; Juguan, L.V.V.; Dimalanta, C.B. Tropical cyclone–southwest monsoon interaction and the 2008 floods and landslides in Panay island, central Philippines: Meteorological and geological factors. Nat. Hazards 2012, 62, 827–840. [Google Scholar] [CrossRef]
- Acosta, L.A.; Eugenio, E.A.; Macandog, P.B.M.; Macandog, D.B.M.; Lin, E.K.H.; Abucay, E.R.; Cura, A.L.; Primavera, M.G. Loss and damage from typhoon-induced floods and landslides in the Philippines: Community Perceptions on climate impacts and adaptation options. Int. J. Glob. Warm. 2016, 9, 33–65. [Google Scholar] [CrossRef]
- Gao, Q.; Yu, M. Reforestation-induced changes of landscape composition and configuration modulate freshwater supply and flooding risk of tropical watersheds. PLoS ONE 2017, 12, e0181315. [Google Scholar] [CrossRef] [PubMed]
- Villamayor, B.M.R.; Rollon, R.N.; Samson, M.S.; Albano, G.M.G.; Primavera, J.H. Impact of Haiyan on Philippine mangroves: Implications to the fate of the widespread monospecific Rhizophora plantations against strong typhoons. Ocean Coast. Manag. 2016, 132, 1–14. [Google Scholar] [CrossRef]
- Stokes, A.; Atger, C.; Bengough, A.G.; Fourcaud, T.; Sidle, R.C. Desirable plant root traits for protecting natural and engineered slopes against landslides. Plant Soil 2009, 324, 1–30. [Google Scholar] [CrossRef]
- Walker, L.R.; Velázquez, E.; Shiels, A.B. Applying lessons from ecological succession to the restoration of landslides. Plant. Soil. 2009, 324, 157–168. [Google Scholar] [CrossRef]
- Pang, C.C.; Ma, X.K.-K.; Lo, J.P.-L.; Hung, T.T.-H.; Hau, B.C.-H. Vegetation succession on landslides in Hong Kong: Plant regeneration, survivorship and constraints to restoration. Glob. Ecol. Conserv. 2018, 15, e00428. [Google Scholar] [CrossRef]
- Miyashita, K.; Tanakamaru, S.; Maitani, T.; Kimura, K. Recovery responses of photosynthesis, transpiration, and stomatal conductance in kidney bean following drought stress. Environ. Exp. Bot. 2005, 53, 205–214. [Google Scholar] [CrossRef]
- Fisher, R.A.; Williams, M.; Da Costa, A.L.; Malhi, Y.; Da Costa, R.F.; Almeida, S.; Metr, P. The response of an Eastern Amazonian rain forest to drought stress: Results and modelling analyses from a throughfall exclusion experiment. Global Change Biol. 2007, 13, 2361–2378. [Google Scholar] [CrossRef]
- Maherali, H.; Sherrard, M.E.; Clifford, M.H.; Latta, R.G. Leaf hydraulic conductivity and photosynthesis are genetically correlated in an annual grass. New Phytol. 2008, 180, 240–247. [Google Scholar] [CrossRef]
- Santos, V.A.H.F.d.; Ferreira, M.J.; Rodrigues, J.V.F.C.; Garcia, M.N.; Ceron, J.V.B.; Nelson, B.W.; Saleska, S.R. Causes of reduced leaf-level photosynthesis during strong El Niño drought in a Central Amazon forest. Global Change Biol. 2018, 24, 4266–4279. [Google Scholar] [CrossRef]
- White, D.A.; McGrath, J.F.; Ryan, M.G.; Battaglia, M.; Mendham, D.S.; Kinal, J.; Downes, G.M.; Crombie, D.S.; Hunt, M.E. Managing for water-use efficient wood production in Eucalyptus globulus plantations. Forest Ecol. Manag. 2014, 331, 272–280. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, H.Y.H.; Lian, J.; John, R.; Li, R.; Liu, H.; Ye, W.; Berninger, F.; Ye, Q. Using functional trait diversity patterns to disentangle the scale-dependent ecological processes in a subtropical forest. Func. Ecol. 2018, 32, 1379–1389. [Google Scholar] [CrossRef]
- Zhang, H.; John, R.; Zhu, S.; Liu, H.; Xu, Q.; Qi, W.; Liu, K.; Chen, H.Y.H.; Ye, Q. Shifts in functional trait–species abundance relationships over secondary subalpine meadow succession in the Qinghai-Tibetan Plateau. Oecologia 2018, 188, 547–557. [Google Scholar] [CrossRef]
- Luo, J.H.; Cui, J.; Shree, P.P.; Jiang, K.; Tan, Z.Y.; He, Q.F.; Zhang, H.; Long, W.X. Seasonally distinctive growth and drought stress functional traits enable Leucaena Leucocephala to successfully invade a Chinese tropical forest. Trop. Conserv. Sci. 2020, 9, 1–7. [Google Scholar]
- Bartlett, M.K.; Scoffoni, C.; Sack, L. The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: A global meta-analysis. Ecology 2016, 97, 503–504. [Google Scholar] [CrossRef] [PubMed]
- Guan, K.; Pan, M.; Li, H.; Wolf, A.; Wu, J.; Medvigy, D.; Caylor, K.K.; Sheffield, J.; Wood, E.F.; Liang, M.; et al. Photosynthetic seasonality of global tropical forests constrained by hydroclimate. Nature Geosci. 2015, 8, 284–289. [Google Scholar] [CrossRef]
- Wu, J.; Serbin, S.P.; Ely, K.; Wolfe, B. The response of stomatal conductance to seasonal drought in tropical forests. Global Change Biol. 2020, 26, 823–839. [Google Scholar] [CrossRef]
- Bartlett, M.K.; Zhang, Y.; Yang, J.; Kreidler, N.; Sun, S.-W.; Lin, L.; Hu, Y.-H.; Cao, K.-F.; Sack, L. Drought tolerance as a driver of tropical forest assembly: Resolving spatial signatures for multiple processes. Ecology 2016, 97, 503–514. [Google Scholar] [CrossRef]
- Kirschbaum, M.U. Does enhanced photosynthesis enhance growth? Lessons learned from CO2 enrichment studies. Plant Physiol. 2011, 155, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.H.; Zhang, Y.P.; Song, Q.H.; Liu, W.J.; Deng, X.B.; Tang, J.W.; Yun, D.; Zhou, W.J.; Yang, L.Y.; Yu, G.R.; et al. Rubber plantations act as water pumps in tropical China. Geophys. Res. Lett. 2011, 38, 24406. [Google Scholar] [CrossRef]
- Wang, Y.; Shao, M.A.; Zhu, Y.; Liu, Z. Impacts of land use and plant characteristics on dried soil layers in different climatic regions on the Loess Plateau of China. Agric. Forest Meteorol. 2011, 151, 437–448. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, H.; Jian, S.; Liu, N. Tree plantations influence the abundance of ammonia-oxidizing bacteria in the soils of a coral island. Appl. Soil Ecol. 2019, 138, 220–222. [Google Scholar] [CrossRef]
- Mason, N.W.; Richardson, S.J.; Peltzer, D.A.; de Bello, F.; Wardle, D.A.; Allen, R.B. Changes in coexistence mechanisms along a long-term soil chronosequence revealed by functional trait diversity. J. Ecol. 2012, 100, 678–689. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, W.; Yang, J.; Luo, J.; Jiang, K.; Xu, J.; Zhang, H. Reforestation Based on Mono-Plantation of Fast-Growing Tree Species Make It Difficult to Maintain (High) Soil Water Content in Tropics, a Case Study in Hainan Island, China. Water 2020, 12, 3077. https://doi.org/10.3390/w12113077
Hong W, Yang J, Luo J, Jiang K, Xu J, Zhang H. Reforestation Based on Mono-Plantation of Fast-Growing Tree Species Make It Difficult to Maintain (High) Soil Water Content in Tropics, a Case Study in Hainan Island, China. Water. 2020; 12(11):3077. https://doi.org/10.3390/w12113077
Chicago/Turabian StyleHong, Wenjun, Jindian Yang, Jinhuan Luo, Kai Jiang, Junze Xu, and Hui Zhang. 2020. "Reforestation Based on Mono-Plantation of Fast-Growing Tree Species Make It Difficult to Maintain (High) Soil Water Content in Tropics, a Case Study in Hainan Island, China" Water 12, no. 11: 3077. https://doi.org/10.3390/w12113077
APA StyleHong, W., Yang, J., Luo, J., Jiang, K., Xu, J., & Zhang, H. (2020). Reforestation Based on Mono-Plantation of Fast-Growing Tree Species Make It Difficult to Maintain (High) Soil Water Content in Tropics, a Case Study in Hainan Island, China. Water, 12(11), 3077. https://doi.org/10.3390/w12113077




