An Overview and Evaluation of Highly Porous Adsorbent Materials for Polycyclic Aromatic Hydrocarbons and Phenols Removal from Wastewater
Abstract
1. Introduction
1.1. Sources of PAHs and Phenols
1.2. PAHs and Phenols in Wastewater Treatment Plants (WWTPs)
1.3. Toxicities of PAHs and Phenols to Humans
1.4. Environmental Regulations on PAHs and Phenols
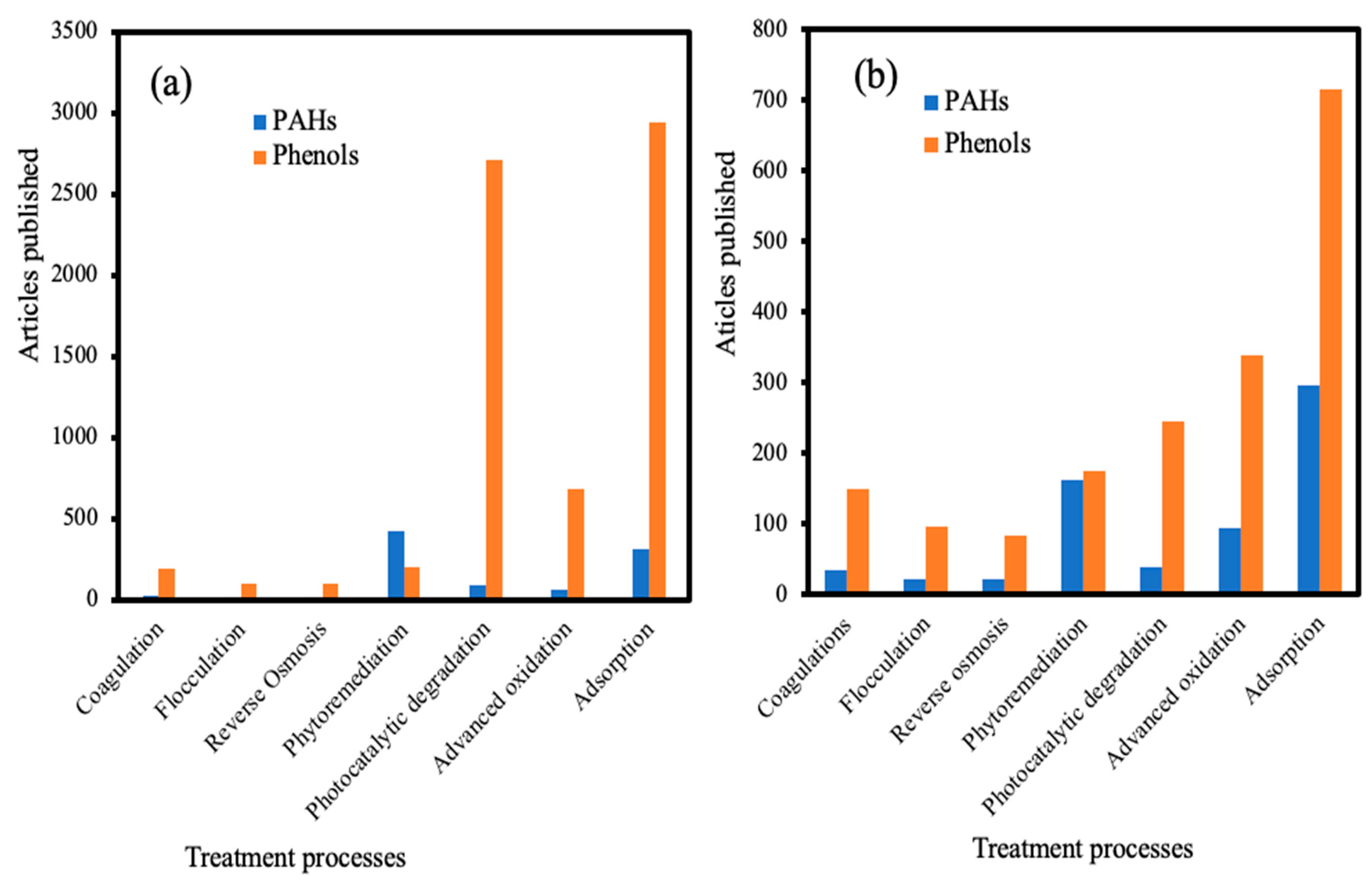
2. PAHs and Phenols Remediation in Wastewater
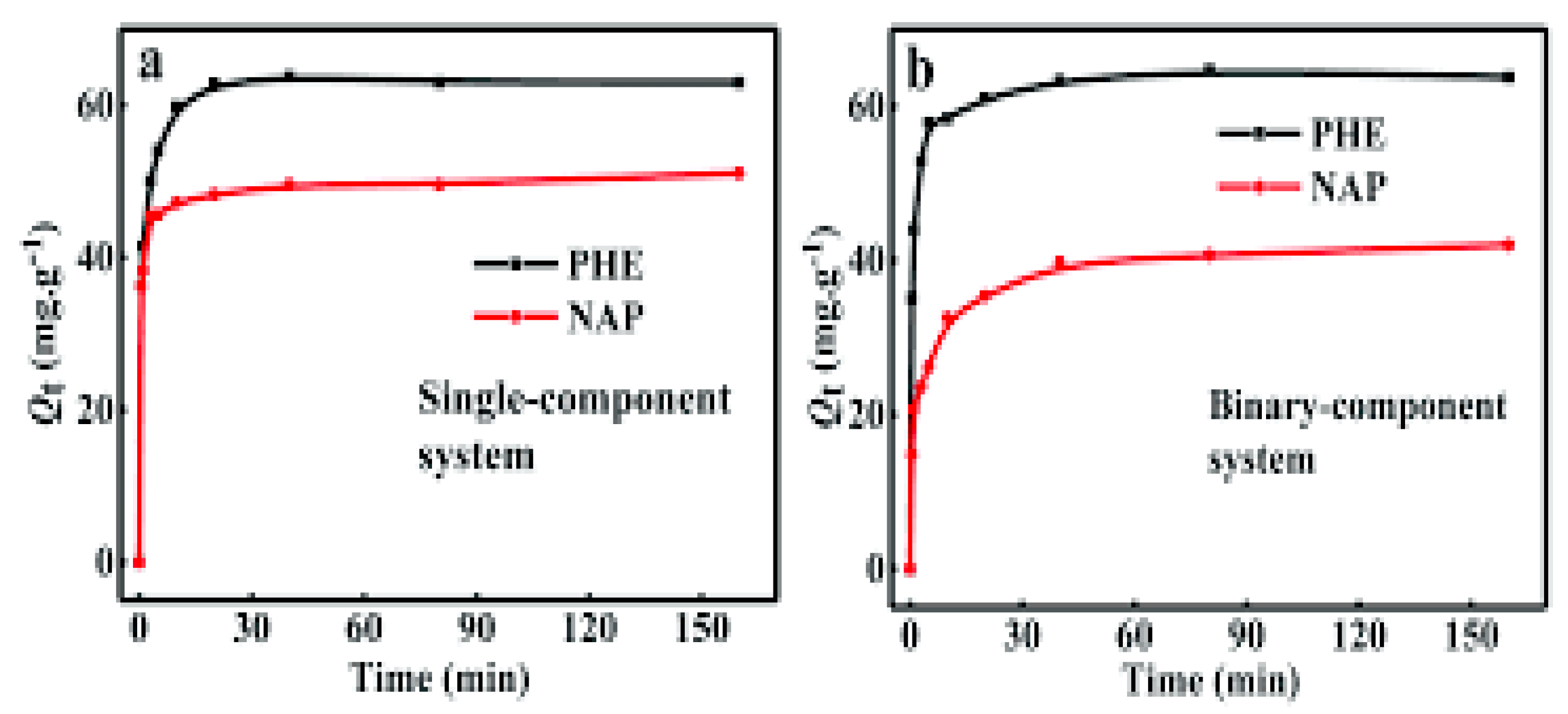
2.1. PAHs and Phenols Removal by Adsorption
2.2. Adsorption of PAHs and Phenols onto Porous Carbon Materials
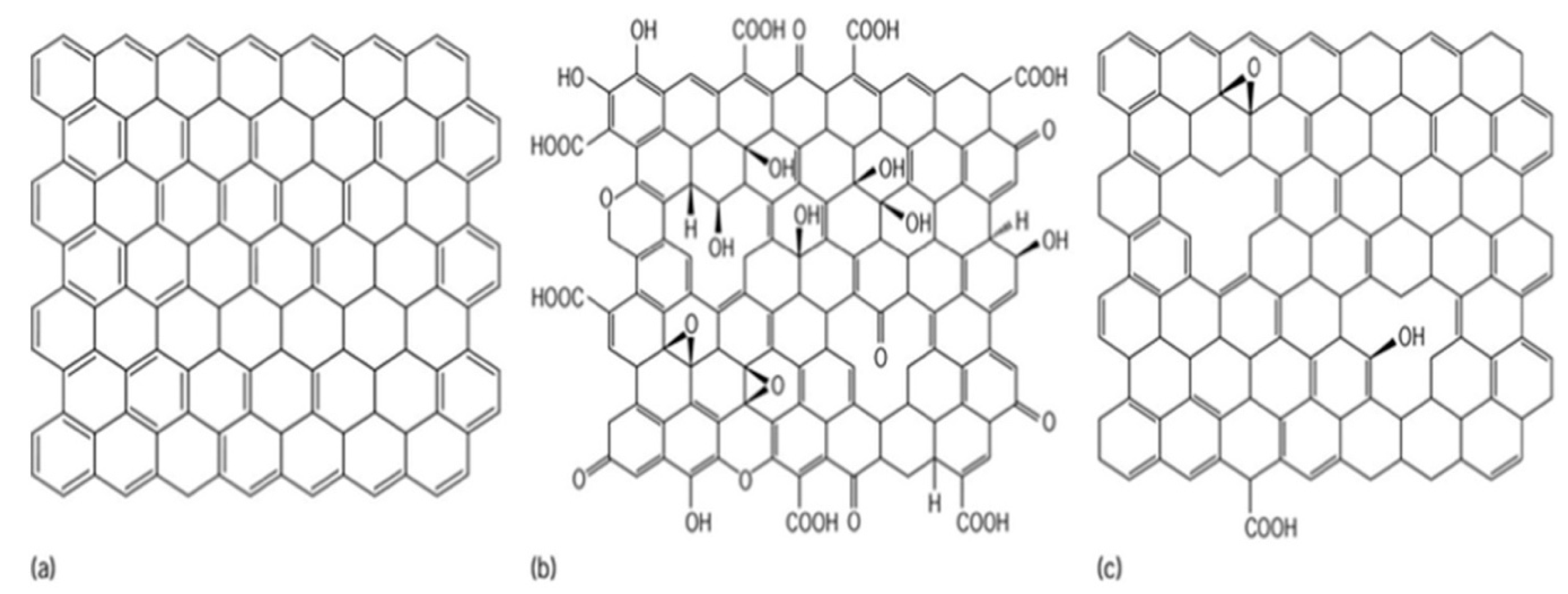
2.2.1. Biochar
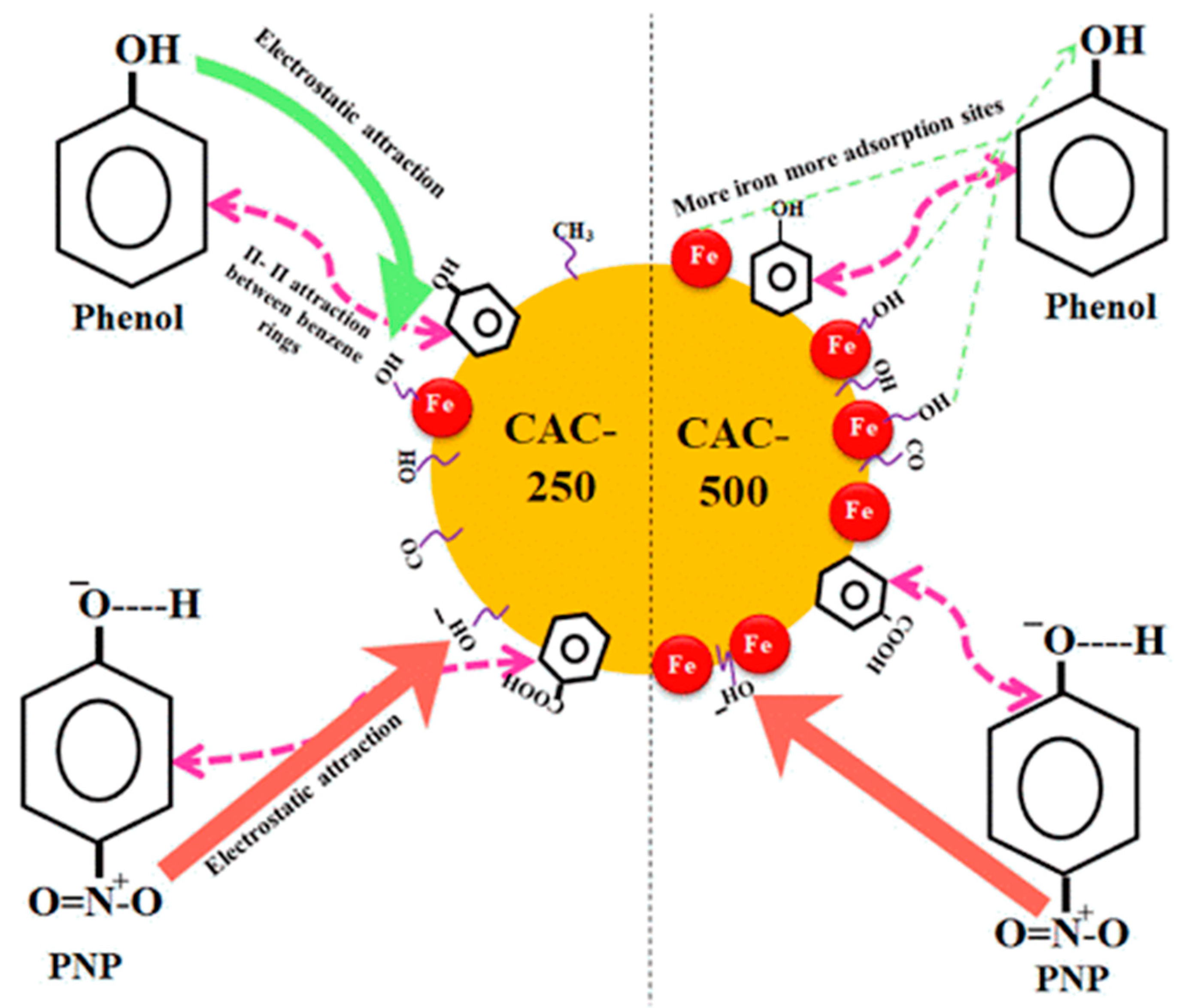
2.2.2. Activated Carbon (AC)
2.3. Other Porous Carbon Materials as Adsorbents for PAHs and Phenols
2.3.1. Graphene, Graphene Oxides, and Reduced Graphene Oxides
2.3.2. Carbon Nanotubes (CNTs)
2.4. Other Porous Materials for PAHs and Phenols Adsorption
2.4.1. Chitosan
2.4.2. Molecularly Imprinted Polymers (MIPs)
2.4.3. Mesoporous Silica
2.4.4. Metal-Organic Frameworks (MOFs)
2.5. Pilot and Industrial Scale Adsorption of PAHs and Phenol in Wastewater
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oh, J.Y.; Choi, S.D.; Kwon, H.O.; Lee, S.E. Leaching of polycyclic aromatic hydrocarbons (PAHs) from industrial wastewater sludge by ultrasonic treatment. Ultrason. Sonochem. 2016, 33, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, L.; Huang, Z.; Li, Y.; Li, J.; Wu, N.; He, J.; Zhang, Z.; Liu, Y.; Niu, Z. Pollution of polycyclic aromatic hydrocarbons (PAHs) in drinking water of China: Composition, distribution and influencing factors. Ecotoxicol. Environ. Saf. 2019, 177, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Tian, W.; Bai, J.; Liu, K.; Sun, H.; Zhao, Y. Occurrence and removal of polycyclic aromatic hydrocarbons in the wastewater treatment process. Ecotoxicol. Environ. Saf. 2012, 82, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Crisafully, R.; Milhome, M.A.L.; Cavalcante, R.M.; Silveira, E.R.; De Keukeleire, D.; Nascimento, R.F. Removal of some polycyclic aromatic hydrocarbons from petrochemical wastewater using low-cost adsorbents of natural origin. Bioresour. Technol. 2008, 99, 4515–4519. [Google Scholar] [CrossRef]
- Zhang, Y.; Tao, S. Global atmospheric emission inventory of polycyclic aromatic hydrocarbons (PAHs) for 2004. Atmos. Environ. 2009, 43, 812–819. [Google Scholar] [CrossRef]
- Liu, Y.; Zarfl, C.; Basu, N.B.; Cirpka, O.A. Turnover and legacy of sediment-associated PAH in a baseflow-dominated river. Sci. Total Environ. 2019, 671, 754–764. [Google Scholar] [CrossRef]
- Andrade, M.V.F.; Santos, F.R.; Oliveira, A.H.B.; Nascimento, R.F.; Cavalcante, R.M. Influence of sediment parameters on the distribution and fate of PAHs in an estuarine tropical region located in the Brazilian semi-arid (Jaguaribe River, Ceará coast). Mar. Pollut. Bull. 2019, 146, 703–710. [Google Scholar] [CrossRef]
- Zhao, W.; Sui, Q.; Huang, X. Removal and fate of polycyclic aromatic hydrocarbons in a hybrid anaerobic–anoxic–oxic process for highly toxic coke wastewater treatment. Sci. Total Environ. 2018, 635, 716–724. [Google Scholar] [CrossRef]
- Sun, S.; Jia, L.; Li, B.; Yuan, A.; Kong, L.; Qi, H.; Ma, W.; Zhang, A.; Wu, Y. The occurrence and fate of PAHs over multiple years in a wastewater treatment plant of Harbin, Northeast China. Sci. Total Environ. 2018, 624, 491–498. [Google Scholar] [CrossRef]
- De Gisi, S.; Lofrano, G.; Grassi, M.; Notarnicola, M. Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: A review. Sustain. Mater. Technol. 2016, 9, 10–40. [Google Scholar] [CrossRef]
- Qiao, M.; Qi, W.; Liu, H.; Qu, J. Oxygenated, nitrated, methyl and parent polycyclic aromatic hydrocarbons in rivers of Haihe River System, China: Occurrence, possible formation, and source and fate in a water-shortage area. Sci. Total Environ. 2014, 481, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Zango, Z.U.; Garba, Z.N.; Abu Bakar, N.H.H.; Tan, W.L.; Abu Bakar, M. Adsorption studies of Cu2+–Hal nanocomposites for the removal of 2,4,6-trichlorophenol. Appl. Clay Sci. 2016, 132–133, 68–78. [Google Scholar] [CrossRef]
- Lawal, I.A.; Lawal, M.M.; Azeez, M.A.; Ndungu, P. Theoretical and experimental adsorption studies of phenol and crystal violet dye on carbon nanotube functionalized with deep eutectic solvent. J. Mol. Liq. 2019, 288. [Google Scholar] [CrossRef]
- Catherine, H.N.; Ou, M.H.; Manu, B.; Shih, Y. hsin Adsorption mechanism of emerging and conventional phenolic compounds on graphene oxide nanoflakes in water. Sci. Total Environ. 2018, 635, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Bazrafshan, E.; Mostafapour, F.K.; Mahvi, A.H. Phenol removal from aqueous solutions using pistachio-nut shell ash as a low cost adsorbent. Fresenius Environ. Bull. 2012, 21, 2962–2968. [Google Scholar]
- Soni, U.; Bajpai, J.; Singh, S.K.; Bajpai, A.K. Evaluation of chitosan-carbon based biocomposite for efficient removal of phenols from aqueous solutions. J. Water Process Eng. 2017, 16, 56–63. [Google Scholar] [CrossRef]
- Guo, M.; Wang, J.; Wang, C.; Strong, P.J.; Jiang, P.; Ok, Y.S.; Wang, H. Carbon nanotube-grafted chitosan and its adsorption capacity for phenol in aqueous solution. Sci. Total Environ. 2019, 682, 340–347. [Google Scholar] [CrossRef]
- Zhang, Y.; Qin, L.; Cui, Y.; Liu, W.; Liu, X.; Yang, Y. A hydrophilic surface molecularly imprinted polymer on a spherical porous carbon support for selective phenol removal from coking wastewater. New Carbon Mater. 2020, 35, 220–231. [Google Scholar] [CrossRef]
- Salari, M.; Dehghani, M.H.; Azari, A.; Motevalli, M.D.; Shabanloo, A.; Ali, I. High performance removal of phenol from aqueous solution by magnetic chitosan based on response surface methodology and genetic algorithm. J. Mol. Liq. 2019, 285, 146–157. [Google Scholar] [CrossRef]
- Lee, C.G.; Hong, S.H.; Hong, S.G.; Choi, J.W.; Park, S.J. Production of Biochar from Food Waste and its Application for Phenol Removal from Aqueous Solution. Water Air Soil Pollut. 2019, 230. [Google Scholar] [CrossRef]
- Wang, F.; Haftka, J.J.H.; Sinnige, T.L.; Hermens, J.L.M.; Chen, W. Adsorption of polar, nonpolar, and substituted aromatics to colloidal graphene oxide nanoparticles. Environ. Pollut. 2014, 186, 226–233. [Google Scholar] [CrossRef]
- Zhou, L.C.; Meng, X.G.; Fu, J.W.; Yang, Y.C.; Yang, P.; Mi, C. Highly efficient adsorption of chlorophenols onto chemically modified chitosan. Appl. Surf. Sci. 2014, 292, 735–741. [Google Scholar] [CrossRef]
- Blanchard, M.; Teil, M.J.; Ollivon, D.; Legenti, L.; Chevreuil, M. Polycyclic aromatic hydrocarbons and polychlorobiphenyls in wastewaters and sewage sludges from the Paris area (France). Environ. Res. 2004, 95, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Mojiri, A.; Zhou, J.L.; Ohashi, A.; Ozaki, N.; Kindaichi, T. Comprehensive review of polycyclic aromatic hydrocarbons in water sources, their effects and treatments. Sci. Total Environ. 2019, 696. [Google Scholar] [CrossRef] [PubMed]
- Lamichhane, S.; Bal Krishna, K.C.; Sarukkalige, R. Polycyclic aromatic hydrocarbons (PAHs) removal by sorption: A review. Chemosphere 2016, 148, 336–353. [Google Scholar] [CrossRef] [PubMed]
- Alegbeleye, O.O.; Opeolu, B.O.; Jackson, V.A. Polycyclic Aromatic Hydrocarbons: A Critical Review of Environmental Occurrence and Bioremediation. Environ. Manag. 2017, 60, 758–783. [Google Scholar] [CrossRef]
- Ndunda, E.N.; Mizaikoff, B. Molecularly imprinted polymers for the analysis and removal of polychlorinated aromatic compounds in the environment: A review. Analyst 2016, 141, 3141–3156. [Google Scholar] [CrossRef]
- Kumar, V.; Kim, K.H.; Park, J.W.; Hong, J.; Kumar, S. Graphene and its nanocomposites as a platform for environmental applications. Chem. Eng. J. 2017, 315, 210–232. [Google Scholar] [CrossRef]
- de Souza, C.V.; Corrêa, S.M. Polycyclic aromatic hydrocarbons in diesel emission, diesel fuel and lubricant oil. Fuel 2016, 185, 925–931. [Google Scholar] [CrossRef]
- Akinpelu, A.A.; Ali, M.E.; Johan, M.R.; Saidur, R.; Qurban, M.A.; Saleh, T.A. Polycyclic aromatic hydrocarbons extraction and removal from wastewater by carbon nanotubes: A review of the current technologies, challenges and prospects. Process Saf. Environ. Prot. 2019, 122, 68–82. [Google Scholar] [CrossRef]
- Redondo, J.M.; Platonov, A.K. Self-similar distribution of oil spills in European coastal waters. Environ. Res. Lett. 2009, 4. [Google Scholar] [CrossRef]
- Lai, X.; Ning, X.-A.; He, Y.; Yuan, Y.; Sun, J.; Ke, Y.; Man, X. Treatment of a simulated sludge by ultrasonic zero-valent iron/EDTA/Air process: Interferences of inorganic salts in polyaromatic hydrocarbon removal. Waste Manag. 2019, 85, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Zheng, G.; Zhou, W.; Wang, J.; Zhou, L. Bioleaching conditioning increased the bioavailability of polycyclic aromatic hydrocarbons to promote their removal during co-composting of industrial and municipal sewage sludges. Sci. Total Environ. 2019, 665, 1073–1082. [Google Scholar] [CrossRef]
- Ihsanullah; Asmaly, H.A.; Saleh, T.A.; Laoui, T.; Gupta, V.K.; Atieh, M.A. Enhanced adsorption of phenols from liquids by aluminum oxide/carbon nanotubes: Comprehensive study from synthesis to surface properties. J. Mol. Liq. 2015, 206, 176–182. [Google Scholar] [CrossRef]
- Marañón, E.; Vázquez, I.; Rodríguez, J.; Castrillón, L.; Fernández, Y.; López, H. Treatment of coke wastewater in a sequential batch reactor (SBR) at pilot plant scale. Bioresour. Technol. 2008, 99, 4192–4198. [Google Scholar] [CrossRef]
- Bargieł, P.; Zabochnicka-Świątek, M. Technologies of Coke Wastewater Treatment in the Frame of Legislation in Force. Ochr. Sr. Zasobow Nat. 2018, 29, 11–15. [Google Scholar] [CrossRef]
- Pal, P.; Kumar, R. Treatment of coke wastewater: A critical review for developing sustainable management strategies. Sep. Purif. Rev. 2014, 43, 89–123. [Google Scholar] [CrossRef]
- Ning, X.A.; Lin, M.Q.; Shen, L.Z.; Zhang, J.H.; Wang, J.Y.; Wang, Y.J.; Yang, Z.Y.; Liu, J.Y. Levels, composition profiles and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in sludge from ten textile dyeing plants. Environ. Res. 2014, 132, 112–118. [Google Scholar] [CrossRef]
- Anku, W.W.; Mamo, M.A.; Govender, P.P. Phenolic Compounds in Water: Sources, Reactivity, Toxicity and Treatment Methods. Intech Open 2016, 419–443. [Google Scholar] [CrossRef]
- Agarwal, T.; Khillare, P.S.; Shridhar, V. PAHs contamination in Bank sediment of the Yamuna River, Delhi, India. Environ. Monit. Assess. 2006, 123, 151–166. [Google Scholar] [CrossRef]
- Iwegbue, C.M.A.; Tesi, G.O.; Overah, L.C.; Emoyan, O.O.; Nwajei, G.E.; Martincigh, B.S. Effects of Flooding on the Sources, Spatiotemporal Characteristics and Human Health Risks of Polycyclic Aromatic Hydrocarbons in Floodplain Soils of the Lower Parts of the River Niger, Nigeria. Polycycl. Aromat. Compd. 2017, 6638, 1–17. [Google Scholar] [CrossRef]
- Ciesielczuk, T.; Kusza, G.; Poluszyńska, J.; Kochanowska, K. Pollution of flooded arable soils with heavy metals and polycyclic aromatic hydrocarbons (PAHs). Water Air Soil Pollut. 2014, 225. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Wang, X.C.; Fan, B. Characteristics of PAHs adsorption on inorganic particles and activated sludge in domestic wastewater treatment. Bioresour. Technol. 2011, 102, 5305–5311. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Wang, S.; Hu, J.; Wu, J.; Li, M.; Wang, Y.; Jin, J. Polychlorinated naphthalenes in human serum samples from an industrial city in Eastern China: Levels, sources, and sex differences. Ecotoxicol. Environ. Saf. 2019, 177, 86–92. [Google Scholar] [CrossRef]
- Liang, X.; Junaid, M.; Wang, Z.; Li, T.; Xu, N. Spatiotemporal distribution, source apportionment and ecological risk assessment of PBDEs and PAHs in the Guanlan River from rapidly urbanizing areas of Shenzhen, China. Environ. Pollut. 2019, 250, 695–707. [Google Scholar] [CrossRef] [PubMed]
- McDonough, C.A.; Khairy, M.A.; Muir, D.C.G.; Lohmann, R. Significance of population centers as sources of gaseous and dissolved PAHs in the lower Great Lakes. Environ. Sci. Technol. 2014, 48, 7789–7797. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wei, C.; Chai, X.; He, J.; Cai, Y.; Ren, M.; Yan, B.; Peng, P.; Fu, J. The behaviors and fate of polycyclic aromatic hydrocarbons (PAHs) in a coking wastewater treatment plant. Chemosphere 2012, 88, 174–182. [Google Scholar] [CrossRef]
- Li, J.; Dong, H.; Xu, X.; Han, B.; Li, X.; Zhu, C.; Han, C.; Liu, S.; Yang, D.; Xu, Q.; et al. Prediction of the bioaccumulation of PAHs in surface sediments of Bohai Sea, China and quantitative assessment of the related toxicity and health risk to humans. Mar. Pollut. Bull. 2016, 104, 92–100. [Google Scholar] [CrossRef]
- de Jesus, J.H.F.; Cunha, G.d.C.; Cardoso, E.M.C.; Mangrich, A.S.; Romão, L.P.C. Evaluation of waste biomasses and their biochars for removal of polycyclic aromatic hydrocarbons. J. Environ. Manag. 2017, 200, 186–195. [Google Scholar] [CrossRef]
- Qu, Y.; Qin, L.; Liu, X.; Yang, Y. Reasonable design and sifting of microporous carbon nanosphere-based surface molecularly imprinted polymer for selective removal of phenol from wastewater. Chemosphere 2020, 251. [Google Scholar] [CrossRef]
- Wu, Z.; Yuan, X.; Zhong, H.; Wang, H.; Zeng, G.; Chen, X.; Wang, H.; Zhang, L.; Shao, J. Enhanced adsorptive removal of p-nitrophenol from water by aluminum metal-organic framework/reduced graphene oxide composite. Sci. Rep. 2016, 6, 25638. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Wu, H.; Liu, L.; Zhang, F.; Preis, S.; Zhu, S.; Wei, C. Solubilization of polycyclic aromatic hydrocarbons (PAHs) with phenol in coking wastewater treatment system: Interaction and engineering significance. Sci. Total Environ. 2018, 628–629, 467–473. [Google Scholar] [CrossRef]
- Villegas, L.G.C.; Mashhadi, N.; Chen, M.; Mukherjee, D.; Taylor, K.E.; Biswas, N. A Short Review of Techniques for Phenol Removal from Wastewater. Curr. Pollut. Rep. 2016, 2, 157–167. [Google Scholar] [CrossRef]
- Bin-Dahman, O.A.; Saleh, T.A. Synthesis of carbon nanotubes grafted with PEG and its efficiency for the removal of phenol from industrial wastewater. Environ. Nanotechnol. Monit. Manag. 2020, 13. [Google Scholar] [CrossRef]
- Saleh, T.A.; Elsharif, A.M.; Asiri, S.; Mohammed, A.R.I.; Dafalla, H. Synthesis of carbon nanotubes grafted with copolymer of acrylic acid and acrylamide for phenol removal. Environ. Nanotechnol. Monit. Manag. 2020, 14. [Google Scholar] [CrossRef]
- Godlewska, P.; Siatecka, A.; Kończak, M.; Oleszczuk, P. Adsorption capacity of phenanthrene and pyrene to engineered carbon-based adsorbents produced from sewage sludge or sewage sludge-biomass mixture in various gaseous conditions. Bioresour. Technol. 2019, 280, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Giwa, A.; Daer, S.; Ahmed, I.; Marpu, P.R.; Hasan, S.W. Experimental investigation and artificial neural networks ANNs modeling of electrically-enhanced membrane bioreactor for wastewater treatment. J. Water Process Eng. 2016, 11, 88–97. [Google Scholar] [CrossRef]
- Lin, M.; Ning, X.-a.; An, T.; Zhang, J.; Chen, C.; Ke, Y.; Wang, Y.; Zhang, Y.; Sun, J.; Liu, J. Degradation of polycyclic aromatic hydrocarbons (PAHs) in textile dyeing sludge with ultrasound and Fenton processes: Effect of system parameters and synergistic effect study. J. Hazard. Mater. 2016, 307, 7–16. [Google Scholar] [CrossRef]
- Bai, H.; Zhou, J.; Zhang, H.; Tang, G. Enhanced adsorbability and photocatalytic activity of TiO2-graphene composite for polycyclic aromatic hydrocarbons removal in aqueous phase. Colloids Surf. B Biointerfaces 2017, 150, 68–77. [Google Scholar] [CrossRef]
- Huang, D.; Xu, B.; Wu, J.; Brookes, P.C.; Xu, J. Adsorption and desorption of phenanthrene by magnetic graphene nanomaterials from water: Roles of pH, heavy metal ions and natural organic matter. Chem. Eng. J. 2019, 368, 390–399. [Google Scholar] [CrossRef]
- Hwang, H.; Sahin, O.; Choi, J.W. Manufacturing a super-active carbon using fast pyrolysis char from biomass and correlation study on structural features and phenol adsorption. RSC Adv. 2017, 7, 42192–42202. [Google Scholar] [CrossRef]
- Xu, J.; Wang, L.; Zhu, Y. Decontamination of bisphenol A from aqueous solution by graphene adsorption. Langmuir 2012, 28, 8418–8425. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, Z.; Chen, B. Adsorption of polycyclic aromatic hydrocarbons by graphene and graphene oxide nanosheets. Environ. Sci. Technol. 2014, 48, 4817–4825. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, J.; Wen, T.; Ren, X.; Huang, Y.; Wang, X. Adsorption of naphthalene and its derivatives on magnetic graphene composites and the mechanism investigation. Colloids Surf. A Physicochem. Eng. Asp. 2013, 422, 118–125. [Google Scholar] [CrossRef]
- Mohammad, A.T.; Al-Obaidi, M.A.; Hameed, E.M.; Basheer, B.N.; Mujtaba, I.M. Modelling the chlorophenol removal from wastewater via reverse osmosis process using a multilayer artificial neural network with genetic algorithm. J. Water Process Eng. 2020, 33. [Google Scholar] [CrossRef]
- Yakout, S.M.; Daifullah, A.A.M.; El-Refeey, S.A. Adsorption of Naphthalene, Phenanthrene and Pyrene from Aqueous Solution Using Low-Cost Activated Carbon Derived from Agricultural. Adsorpt. Sci. Technol. 2013, 31, 293–302. [Google Scholar] [CrossRef]
- Gundogdu, A.; Duran, C.; Senturk, H.B.; Soylak, M.; Ozdes, D.; Serencam, H.; Imamoglu, M. Adsorption of phenol from aqueous solution on a low-cost activated carbon produced from tea industry waste: Equilibrium, kinetic, and thermodynamic study. J. Chem. Eng. Data 2012, 57, 2733–2743. [Google Scholar] [CrossRef]
- Rasheed, A.; Farooq, F.; Rafique, U.; Nasreen, S.; Aqeel Ashraf, M. Analysis of sorption efficiency of activated carbon for removal of anthracene and pyrene for wastewater treatment. Desalin. Water Treat. 2016, 57, 145–150. [Google Scholar] [CrossRef]
- Feng, Z.; Zhu, L. Sorption of phenanthrene to biochar modified by base. Front. Environ. Sci. Eng. 2018, 12, 1–11. [Google Scholar] [CrossRef]
- Tong, Y.; McNamara, P.J.; Mayer, B.K. Adsorption of organic micropollutants onto biochar: A review of relevant kinetics, mechanisms and equilibrium. Environ. Sci. Water Res. Technol. 2019, 5, 821–838. [Google Scholar] [CrossRef]
- Xu, L.; Wang, Z.; Ye, S.; Sui, X. Removal of p-chlorophenol from aqueous solutions by carbon nanotube hybrid polymer adsorbents. Chem. Eng. Res. Des. 2017, 123, 76–83. [Google Scholar] [CrossRef]
- Zhang, J.; Li, R.; Ding, G.; Wang, Y.; Wang, C. Sorptive removal of phenanthrene from water by magnetic carbon nanomaterials. J. Mol. Liq. 2019, 293. [Google Scholar] [CrossRef]
- Zielińska, A.; Oleszczuk, P. The conversion of sewage sludge into biochar reduces polycyclic aromatic hydrocarbon content and ecotoxicity but increases trace metal content. Biomass Bioenergy 2015, 75, 235–244. [Google Scholar] [CrossRef]
- Smith, K.M.; Fowler, G.D.; Pullket, S.; Graham, N.J.D. Sewage sludge-based adsorbents: A review of their production, properties and use in water treatment applications. Water Res. 2009, 43, 2569–2594. [Google Scholar] [CrossRef]
- Azargohar, R.; Jacobson, K.L.; Powell, E.E.; Dalai, A.K. Evaluation of properties of fast pyrolysis products obtained, from Canadian waste biomass. J. Anal. Appl. Pyrolysis 2013, 104, 330–340. [Google Scholar] [CrossRef]
- Qiu, M.; Sun, K.; Jin, J.; Gao, B.; Yan, Y.; Han, L.; Wu, F.; Xing, B. Properties of the plant- and manure-derived biochars and their sorption of dibutyl phthalate and phenanthrene. Sci. Rep. 2014, 4, 5295. [Google Scholar] [CrossRef]
- Askeland, M.; Clarke, B.; Paz-Ferreiro, J. Comparative characterization of biochars produced at three selected pyrolysis temperatures from common woody and herbaceous waste streams. PeerJ 2019, 7, e6784. [Google Scholar] [CrossRef]
- Guo, W.; Wang, S.; Wang, Y.; Lu, S.; Gao, Y. Sorptive removal of phenanthrene from aqueous solutions using magnetic and non-magnetic rice husk-derived biochars. R. Soc. Open Sci. 2018, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Qu, R.; Li, C.; Guo, W.; Han, X.; He, F.; Ma, Y.; Xing, B. Selective removal of polycyclic aromatic hydrocarbons (PAHs) from soil washing effluents using biochars produced at different pyrolytic temperatures. Bioresour. Technol. 2014, 163, 193–198. [Google Scholar] [CrossRef]
- Md Arshad, S.H.; Ngadi, N.; Wong, S.; Saidina Amin, N.; Razmi, F.A.; Mohamed, N.B.; Inuwa, I.M.; Abdul Aziz, A. Optimization of phenol adsorption onto biochar from oil palm empty fruit bunch (EFB). Malays. J. Fundam. Appl. Sci. 2019, 15, 1–5. [Google Scholar] [CrossRef]
- Bedia, J.; Peñas-Garzón, M.; Gómez-Avilés, A.; Rodriguez, J.; Belver, C. A Review on the Synthesis and Characterization of Biomass-Derived Carbons for Adsorption of Emerging Contaminants from Water. J. Carbon Res. 2018, 4, 63. [Google Scholar] [CrossRef]
- Qiao, K.; Tian, W.; Bai, J.; Dong, J.; Zhao, J.; Gong, X.; Liu, S. Preparation of biochar from Enteromorpha prolifera and its use for the removal of polycyclic aromatic hydrocarbons (PAHs) from aqueous solution. Ecotoxicol. Environ. Saf. 2018, 149, 80–87. [Google Scholar] [CrossRef]
- Tang, J.; Lv, H.; Gong, Y.; Huang, Y. Preparation and characterization of a novel graphene/biochar composite for aqueous phenanthrene and mercury removal. Bioresour. Technol. 2015, 196, 355–363. [Google Scholar] [CrossRef] [PubMed]
- de Jesus, J.H.F.; Matos, T.T.d.S.; Cunha, G.d.C.; Mangrich, A.S.; Romão, L.P.C. Adsorption of aromatic compounds by biochar: Influence of the type of tropical biomass precursor. Cellulose 2019. [Google Scholar] [CrossRef]
- Liu, X.; Sun, J.; Duan, S.; Wang, Y.; Hayat, T.; Alsaedi, A.; Wang, C.; Li, J. A Valuable Biochar from Poplar Catkins with High Adsorption Capacity for Both Organic Pollutants and Inorganic Heavy Metal Ions. Sci. Rep. 2017, 7, 10033. [Google Scholar] [CrossRef]
- Shin, W. seok Adsorption characteristics of phenol and heavy metals on biochar from Hizikia fusiformis. Environ. Earth Sci. 2017, 76, 782. [Google Scholar] [CrossRef]
- Braghiroli, F.L.; Bouafif, H.; Hamza, N.; Neculita, C.M.; Koubaa, A. Production, characterization, and potential of activated biochar as adsorbent for phenolic compounds from leachates in a lumber industry site. Environ. Sci. Pollut. Res. 2018, 25, 26562–26575. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Z.; Liu, S.; Xu, H.; Song, Z. Study on adsorption of phenol from aqueous media using biochar of Chinese herb residue. IOP Conf. Ser. Mater. Sci. Eng. 2018, 394. [Google Scholar] [CrossRef]
- Siipola, V.; Pflugmacher, S.; Romar, H.; Wendling, L.; Koukkari, P. Low-Cost Biochar Adsorbents for Water Purification Including Microplastics Removal. Appl. Sci. 2020, 10, 788. [Google Scholar] [CrossRef]
- Jin, D.F.; Xu, Y.Y.; Zhang, M.; Jung, Y.S.; Ok, Y.S. Comparative evaluation for the sorption capacity of four carbonaceous sorbents to phenol. Chem. Speciat. Bioavailab. 2016, 28, 18–25. [Google Scholar] [CrossRef]
- Mohammed, N.A.S.; Abu-Zurayk, R.A.; Hamadneh, I.; Al-Dujaili, A.H. Phenol adsorption on biochar prepared from the pine fruit shells: Equilibrium, kinetic and thermodynamics studies. J. Environ. Manag. 2018, 226, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Danish, M.; Ahmad, T. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sustain. Energy Rev. 2018, 87, 1–21. [Google Scholar] [CrossRef]
- Alade, A.O.; Amuda, O.S.; Ibrahim, A.O. Isothermal studies of adsorption of acenaphthene from aqueous solution onto activated carbon produced from rice (Oryza sativa) husk. Elixir Chem. Eng. 2012, 46, 87–95. [Google Scholar] [CrossRef]
- Alade, A.O.; Amuda, O.S.; Afolabi, T.J.; Okoya, A.A. Adsorption of naphthalene onto activated carbons derived from milk bush kernel shell and flamboyant pod. J. Environ. Chem. Ecotoxicol. 2012, 4, 124–132. [Google Scholar] [CrossRef]
- Jin, X.J.; Zhu, Y.M. Absorption of Phenol on Nitrogen-Enriched Activated Carbon from Wood Fiberboard Waste with Chemical Activation by Potassium Carbonate. J. Chem. Eng. Process Technol. 2014, 5, 4. [Google Scholar] [CrossRef]
- Ingole, R.S.; Lataye, D.H. Adsorptive removal of phenol from aqueous solution using activated carbon prepared from Babul sawdust. J. Hazard. Toxicol. Radioact. Waste 2015, 19, 1–9. [Google Scholar] [CrossRef]
- Dąbrowski, A.; Podkościelny, P.; Hubicki, Z.; Barczak, M. Adsorption of phenolic compounds by activated carbon—A critical review. Chemosphere 2005, 58, 1049–1070. [Google Scholar] [CrossRef]
- Xiao, X.; Liu, D.; Yan, Y.; Wu, Z.; Wu, Z.; Cravotto, G. Preparation of activated carbon from Xinjiang region coal by microwave activation and its application in naphthalene, phenanthrene, and pyrene adsorption. J. Taiwan Inst. Chem. Eng. 2015, 53, 160–167. [Google Scholar] [CrossRef]
- Eeshwarasinghe, D.; Loganathan, P.; Vigneswaran, S. Simultaneous removal of polycyclic aromatic hydrocarbons and heavy metals from water using granular activated carbon. Chemosphere 2019, 223, 616–627. [Google Scholar] [CrossRef]
- Rakowska, M.I.; Kupryianchyk, D.; Grotenhuis, T.; Rijnaarts, H.H.M.; Koelmans, A.A. Extraction of sediment-associated polycyclic aromatic hydrocarbons with granular activated carbon. Environ. Toxicol. Chem. 2013, 32, 304–311. [Google Scholar] [CrossRef]
- Ge, X.; Wu, Z.; Wu, Z.; Yan, Y.; Cravotto, G.; Ye, B.C. Enhanced PAHs adsorption using iron-modified coal-based activated carbon via microwave radiation. J. Taiwan Inst. Chem. Eng. 2016, 64, 235–243. [Google Scholar] [CrossRef]
- Ge, X.; Tian, F.; Wu, Z.; Yan, Y.; Cravotto, G.; Wu, Z. Adsorption of naphthalene from aqueous solution on coal-based activated carbon modi fi ed by microwave induction: Microwave power effects. Chem. Eng. Process. 2015, 91, 67–77. [Google Scholar] [CrossRef]
- Wu, Z.; Sun, Z.; Liu, P.; Li, Q.; Yang, R.; Yang, X. Competitive adsorption of naphthalene and phenanthrene on walnut shell based activated carbon and the veri fi cation via theoretical. RSC Adv. 2020, 10, 10703–10714. [Google Scholar] [CrossRef]
- Liu, J.; Chen, J.; Jiang, L.; Yin, X. Adsorption of mixed polycyclic aromatic hydrocarbons in surfactant solutions by activated carbon. J. Ind. Eng. Chem. 2014, 20, 616–623. [Google Scholar] [CrossRef]
- Alade, A.O.; Amuda, O.S.; Afolabi, A.O.; Adelowo, F.E. Adosrption of Acenaphthene unto Ativated Crabon Produced from Agricultural Wastes. J. Envion. Sci. Technol. 2012, 5, 192–209. [Google Scholar]
- Yakout, S.M.; Daifullah, A.A.M.; El-Reefy, S.A. Equilibrium and kinetic studies of sorption of polycyclic aromatic hydrocarbons from water using rice husk activated carbon. Asian J. Chem. 2013, 25, 10037–10042. [Google Scholar] [CrossRef]
- Rinawati; Hidayat, D.; Supriyanto, R.; Permana, D.F. Yunita Adsorption of Polycyclic Aromatic Hydrocarbons using Low-Cost Activated Carbon Derived from Rice Husk. J. Phys. Conf. Ser. 2019, 1338. [Google Scholar] [CrossRef]
- Awe, A.A.; Opeolu, B.O.; Fatoki, O.S.; Ayanda, O.S.; Jackson, V.A.; Snyman, R. Preparation and characterisation of activated carbon from Vitis vinifera leaf litter and its adsorption performance for aqueous phenanthrene. Appl. Biol. Chem. 2020, 63, 12. [Google Scholar] [CrossRef]
- Khare, P.; Kumar, A. Removal of phenol from aqueous solution using carbonized Terminalia chebula-activated carbon: Process parametric optimization using conventional method and Taguchi’s experimental design, adsorption kinetic, equilibrium and thermodynamic study. Appl. Water Sci. 2012, 2, 317–326. [Google Scholar] [CrossRef][Green Version]
- Lin, J.Q.; Yang, S.E.; Duan, J.M.; Wu, J.J.; Jin, L.Y.; Lin, J.M.; Deng, Q.L. The Adsorption Mechanism of Modified Activated Carbon on Phenol. MATEC Web Conf. 2016, 67. [Google Scholar] [CrossRef]
- Biglari, H.; Afsharnia, M.; Javan, N.; Sajadi, S.A. Phenol Removal from Aqueous Solutions by Adsorption on Activated Carbon of Miswak’s Root Treated with KMnO4. Iran. J. Health Sci. 2016, 42, 20–30. [Google Scholar] [CrossRef]
- Xie, B.; Qin, J.; Wang, S.; Li, X.; Sun, H.; Chen, W. Adsorption of Phenol on Commercial Activated Carbons: Modelling and Interpretation. Int. J. Environ. Res. Public Health 2020, 17, 789. [Google Scholar] [CrossRef]
- Beker, U.; Ganbold, B.; Dertli, H.; Gülbayir, D.D. Adsorption of phenol by activated carbon: Influence of activation methods and solution pH. Energy Convers. Manag. 2010, 51, 235–240. [Google Scholar] [CrossRef]
- Germain, T.; Lynda, E.; Tchirioua, E. Adsorption of Phenol on Carbon Based on Cactus and Banana Peel. Aust. J. Basic Appl. Sci. 2019, 13, 64–70. [Google Scholar] [CrossRef]
- Mojoudi, N.; Mirghaffari, N.; Soleimani, M.; Shariatmadari, H.; Belver, C.; Bedia, J. Phenol adsorption on high microporous activated carbons prepared from oily sludge: Equilibrium, kinetic and thermodynamic studies. Sci. Rep. 2019, 9, 19352. [Google Scholar] [CrossRef]
- Nouha, S.; Souad, N.S.; Abdelmottalab, O. Enhanced adsorption of phenol using alkaline modified activated carbon prepared from olive stones. J. Chil. Chem. Soc. 2019, 64, 4352–4359. [Google Scholar] [CrossRef]
- Siti Hadjar, M.A.; Astimar, A.A.; Norzita, N.; Noraishah, S.A. Phenol adsorption by activated carbon of different fibre size derived from empty fruit bunches. J. Oil Palm Res. 2012, 24, 1524–1532. [Google Scholar]
- Dejene, K.; Siraj, K.; Kitte, S.A. Kinetic and Thermodynamic Study of Phenol Removal from Water Using Activated Carbon Synthesizes from Avocado Kernel Seed. Int. Lett. Nat. Sci. 2016, 54, 42–57. [Google Scholar] [CrossRef]
- Banat, F.; Al-Asheh, S.; Al-Makhadmeh, L. Utilization of raw and activated date pits for the removal of phenol from aqueous solutions. Chem. Eng. Technol. 2004, 27, 80–86. [Google Scholar] [CrossRef]
- Mohtashami, S.A.; Asasian Kolur, N.; Kaghazchi, T.; Asadi-Kesheh, R.; Soleimani, M. Optimization of sugarcane bagasse activation to achieve adsorbent with high affinity towards phenol. Turk. J. Chem. 2018, 42, 1720–1735. [Google Scholar] [CrossRef]
- Girish, C.R.; Murty, V.R. Adsorption of Phenol from Aqueous Solution Using Lantana camara, Forest Waste: Packed Bed Studies and Prediction of Breakthrough Curves. Environ. Process. 2014, 2, 773–796. [Google Scholar] [CrossRef]
- Mohammadi, S.Z.; Darijani, Z.; Karimi, M.A. Fast and efficient removal of phenol by magnetic activated carbon-cobalt nanoparticles. J. Alloys Compd. 2020, 832. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Kermani, M.; Gholami, M.; Farzadkia, M. Kinetic and isotherm studies of adsorption and biosorption processes in the removal of phenolic compounds from aqueous solutions: Comparative study. J. Environ. Health Sci. Eng. 2013, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, Z.; Simitzis, J. Adsorption kinetics of phenol and 3-nitrophenol from aqueous solutions on conventional and novel carbons. J. Hazard. Mater. 2009, 171, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.; Narayan Maddheshiaya, D.; Rawat, S.; Singh, J. Adsorption and equilibrium studies of phenol and para-nitrophenol by magnetic activated carbon synthesised from cauliflower waste. Environ. Eng. Res. 2019, 25, 742–752. [Google Scholar] [CrossRef]
- Atieh, M.A. Removal of Phenol from Water Different Types of Carbon—A Comparative Analysis. APCBEE Procedia 2014, 10, 136–141. [Google Scholar] [CrossRef]
- Dehghani, M.H.; Mostofi, M.; Alimohammadi, M.; McKay, G.; Yetilmezsoy, K.; Albadarin, A.B.; Heibati, B.; AlGhouti, M.; Mubarak, N.M.; Sahu, J.N. High-performance removal of toxic phenol by single-walled and multi-walled carbon nanotubes: Kinetics, adsorption, mechanism and optimization studies. J. Ind. Eng. Chem. 2016, 35, 63–74. [Google Scholar] [CrossRef]
- Rezakazemi, M.; Kurniawan, T.A.; Albadarin, A.B.; Shirazian, S. Molecular modeling investigation on mechanism of phenol removal from aqueous media by single- and multi-walled carbon nanotubes. J. Mol. Liq. 2018, 271, 24–30. [Google Scholar] [CrossRef]
- Wang, L.C.; Ni, X.J.; Cao, Y.H.; Cao, G.Q. Adsorption behavior of bisphenol A on CTAB-modified graphite. Appl. Surf. Sci. 2018, 428, 165–170. [Google Scholar] [CrossRef]
- Chowdhury, S.; Balasubramanian, R. Recent advances in the use of graphene-family nanoadsorbents for removal of toxic pollutants from wastewater. Adv. Colloid Interface Sci. 2014, 204, 35–56. [Google Scholar] [CrossRef]
- Li, B.; Ou, P.; Wei, Y.; Zhang, X.; Song, J. Polycyclic aromatic hydrocarbons adsorption onto graphene: A DFT and AIMD study. Materials 2018, 11, 726. [Google Scholar] [CrossRef] [PubMed]
- Fraga, T.J.M.; Carvalho, M.N.; Ghislandi, M.G.; Da Motta Sobrinho, M.A. Functionalized graphene-based materials as innovative adsorbents of organic pollutants: A concise overview. Braz. J. Chem. Eng. 2019, 36, 1–31. [Google Scholar] [CrossRef]
- Ali, I.; Basheer, A.A.; Mbianda, X.Y.; Burakov, A.; Galunin, E.; Burakova, I.; Mkrtchyan, E.; Tkachev, A.; Grachev, V. Graphene based adsorbents for remediation of noxious pollutants from wastewater. Environ. Int. 2019, 127, 160–180. [Google Scholar] [CrossRef]
- Bano, Z.; Mazari, S.A.; Saeed, R.M.Y.; Majeed, M.A.; Xia, M.; Memon, A.Q.; Abro, R.; Wang, F. Water decontamination by 3D graphene based materials: A review. J. Water Process Eng. 2020, 36, 101404. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, S.; Zhao, G.; Wang, Q.; Wang, X. Adsorption of polycyclic aromatic hydrocarbons on graphene oxides and reduced graphene oxides. Chemistry 2013, 8, 2755–2761. [Google Scholar] [CrossRef]
- Adeola, A.O.; Forbes, P.B.C. Optimization of the sorption of selected polycyclic aromatic hydrocarbons by regenerable graphene wool. Water Sci. Technol. 2020, 80, 1931–1943. [Google Scholar] [CrossRef]
- Zhou, Q.; Wang, Y.; Xiao, J.; Fan, H. Fabrication and characterisation of magnetic graphene oxide incorporated Fe3O4@polyaniline for the removal of bisphenol A, t-octyl-phenol, and α-naphthol from water. Sci. Rep. 2017, 7, 11316. [Google Scholar] [CrossRef]
- Wang, X.; Hu, Y.; Min, J.; Li, S.; Deng, X.; Yuan, S.; Zuo, X. Adsorption characteristics of phenolic compounds on graphene oxide and reduced graphene oxide: A batch experiment combined theory calculation. Appl. Sci. 2018, 8, 1950. [Google Scholar] [CrossRef]
- Tian, J.; Wei, J.; Zhang, H.; Kong, Z.; Zhu, Y.; Qin, Z. Graphene oxide-functionalized dual-scale channels architecture for high-throughput removal of organic pollutants from water. Chem. Eng. J. 2019, 359, 852–862. [Google Scholar] [CrossRef]
- Apul, O.G.; Wang, Q.; Zhou, Y.; Karanfil, T. Adsorption of aromatic organic contaminants by graphene nanosheets: Comparison with carbon nanotubes and activated carbon. Water Res. 2013, 47, 1648–1654. [Google Scholar] [CrossRef]
- Das, P.; Goswami, S.; Maiti, S. Removal of naphthalene present in synthetic waste water using novel Graphene /Graphene Oxide nano sheet synthesized from rice straw: Comparative analysis, isotherm and kinetics. Front. Nanosci. Nanotechnol. 2016, 2, 38–42. [Google Scholar] [CrossRef]
- Zhao, G.; Jiang, L.; He, Y.; Li, J.; Dong, H.; Wang, X.; Hu, W. Sulfonated graphene for persistent aromatic pollutant management. Adv. Mater. 2011, 23, 3959–3963. [Google Scholar] [CrossRef]
- Mortazavi, M.; Baghdadi, M.; Seyed Javadi, N.H.; Torabian, A. The black beads produced by simultaneous thermal reducing and chemical bonding of graphene oxide on the surface of amino-functionalized sand particles: Application for PAHs removal from contaminated waters. J. Water Process Eng. 2019, 31, 100798. [Google Scholar] [CrossRef]
- Wang, J.; Chen, B. Adsorption and coadsorption of organic pollutants and a heavy metal by graphene oxide and reduced graphene materials. Chem. Eng. J. 2015, 281, 379–388. [Google Scholar] [CrossRef]
- Mehrizad, A.; Gharbani, P. Decontamination of 4-chloro-2-nitrophenol from aqueous solution by graphene adsorption: Equilibrium, kinetic, and thermodynamic studies. Pol. J. Environ. Stud. 2014, 23, 2111–2116. [Google Scholar] [CrossRef]
- De La Luz-Asunción, M.; Sánchez-Mendieta, V.; Martínez-Hernández, A.L.; Castaño, V.M.; Velasco-Santos, C. Adsorption of phenol from aqueous solutions by carbon nanomaterials of one and two dimensions: Kinetic and equilibrium studies. J. Nanomater. 2015, 2015, 405036. [Google Scholar] [CrossRef]
- Gholami-Bonabi, L.; Ziaefar, N.; Sheikhloie, H. Removal of phenol from aqueous solutions by magnetic oxide graphene nanoparticles modified with ionic liquids using the Taguchi optimization approach. Water Sci. Technol. 2020, 81, 228–240. [Google Scholar] [CrossRef]
- Li, Y.; Du, Q.; Liu, T.; Sun, J.; Jiao, Y.; Xia, Y.; Xia, L.; Wang, Z.; Zhang, W.; Wang, K.; et al. Equilibrium, kinetic and thermodynamic studies on the adsorption of phenol onto graphene. Mater. Res. Bull. 2012, 47, 1898–1904. [Google Scholar] [CrossRef]
- Wang, J.; Gao, X.; Wang, Y.; Gao, C. Novel Graphene Oxide Sponge synthesized by Freeze-Drying Process for the Removal of 2,4,6-Trichlorophenol. RSC Adv. 2014, 4, 57476–57482. [Google Scholar] [CrossRef]
- Phatthanakittiphong, T.; Seo, G.T. Characteristic evaluation of graphene oxide for bisphenol a adsorption in aqueous solution. Nanomaterials 2016, 6, 128. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Y.; Li, S.; Yu, S. Sorption and removal of tetrabromobisphenol A from solution by graphene oxide. Chem. Eng. J. 2013, 222, 94–100. [Google Scholar] [CrossRef]
- Soltani, T.; Lee, B.K. Mechanism of highly efficient adsorption of 2-chlorophenol onto ultrasonic graphene materials: Comparison and equilibrium. J. Colloid Interface Sci. 2016, 481, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhang, X.; Chen, Y. Modulated synthesis of zirconium metal—Organic framework UiO-66 with enhanced dichloromethane adsorption capacity. Mater. Lett. 2017, 197, 167–170. [Google Scholar] [CrossRef]
- Ong, Y.T.; Ahmad, A.L.; Zein, S.H.S.; Tan, S.H. A review on carbon nanotubes in an environmental protection and green engineering perspective. Braz. J. Chem. Eng. 2010, 27, 227–242. [Google Scholar] [CrossRef]
- Sarkar, B.; Mandal, S.; Tsang, Y.F.; Kumar, P.; Kim, K.H.; Ok, Y.S. Designer carbon nanotubes for contaminant removal in water and wastewater: A critical review. Sci. Total Environ. 2018, 612, 561–581. [Google Scholar] [CrossRef]
- ALOthman, Z.A.; Wabaidur, S.M. Application of carbon nanotubes in extraction and chromatographic analysis: A review. Arab. J. Chem. 2019, 12, 633–651. [Google Scholar] [CrossRef]
- Mallakpour, S.; Khadem, E. Carbon nanotube–metal oxide nanocomposites: Fabrication, properties and applications. Chem. Eng. J. 2016, 302, 344–367. [Google Scholar] [CrossRef]
- Krstić, V.; Urošević, T.; Pešovski, B. A review on adsorbents for treatment of water and wastewaters containing copper ions. Chem. Eng. Sci. 2018, 192, 273–287. [Google Scholar] [CrossRef]
- Zhang, W.; Zeng, Z.; Liu, Z.; Huang, J.; Xiao, R.; Shao, B.; Liu, Y.; Liu, Y.; Tang, W.; Zeng, G.; et al. Effects of carbon nanotubes on biodegradation of pollutants: Positive or negative? Ecotoxicol. Environ. Saf. 2020, 189. [Google Scholar] [CrossRef]
- Yu, J.G.; Zhao, X.H.; Yang, H.; Chen, X.H.; Yang, Q.; Yu, L.Y.; Jiang, J.H.; Chen, X.Q. Aqueous adsorption and removal of organic contaminants by carbon nanotubes. Sci. Total Environ. 2014, 482–483, 241–251. [Google Scholar] [CrossRef]
- Jung, C.; Son, A.; Her, N.; Zoh, K.D.; Cho, J.; Yoon, Y. Removal of endocrine disrupting compounds, pharmaceuticals, and personal care products in water using carbon nanotubes: A review. J. Ind. Eng. Chem. 2015, 27, 1–11. [Google Scholar] [CrossRef]
- Saleh, T.A.; Sarı, A.; Tuzen, M. Carbon nanotubes grafted with poly(trimesoyl, m-phenylenediamine) for enhanced removal of phenol. J. Environ. Manag. 2019, 252. [Google Scholar] [CrossRef]
- Wu, W.; Yang, K.; Chen, W.; Wang, W.; Zhang, J.; Lin, D.; Xing, B. Correlation and prediction of adsorption capacity and affinity of aromatic compounds on carbon nanotubes. Water Res. 2016, 88, 492–501. [Google Scholar] [CrossRef]
- Selvaraj, M.; Hai, A.; Banat, F.; Haija, M.A. Application and prospects of carbon nanostructured materials in water treatment: A review. J. Water Process Eng. 2020, 33. [Google Scholar] [CrossRef]
- Apul, O.G.; Karanfil, T. Adsorption of synthetic organic contaminants by carbon nanotubes: A critical review. Water Res. 2015, 68, 34–55. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, J.; Naeem, S.; Ahmad, M.; Usman, A.R.A.; Al-Wabel, M.I. A critical review on organic micropollutants contamination in wastewater and removal through carbon nanotubes. J. Environ. Manag. 2019, 246, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Zhu, L.; Xing, B. Adsorption of polycyclic aromatic hydrocarbons by carbon nanomaterials. Environ. Sci. Technol. 2006, 40, 1855–1861. [Google Scholar] [CrossRef]
- Kah, M.; Zhang, X.; Jonker, M.T.O.; Hofmann, T. Measuring and modeling adsorption of PAHs to carbon nanotubes over a six order of magnitude wide concentration range. Environ. Sci. Technol. 2011, 45, 6011–6017. [Google Scholar] [CrossRef]
- Gotovac, S.; Yang, C.M.; Hattori, Y.; Takahashi, K.; Kanoh, H.; Kaneko, K. Adsorption of polyaromatic hydrocarbons on single wall carbon nanotubes of different functionalities and diameters. J. Colloid Interface Sci. 2007, 314, 18–24. [Google Scholar] [CrossRef]
- Pahlavan Yali, Z.; Fatemi, M.H. Prediction of the sorption coefficient for the adsorption of PAHs on MWCNT based on hybrid QSPR-molecular docking approach. Adsorption 2019, 25, 737–743. [Google Scholar] [CrossRef]
- Hu, B.; Gao, Z.; Wang, H.; Wang, J.; Cheng, M. Computational insights into the sorption mechanism of polycyclic aromatic hydrocarbons by carbon nanotube through density functional theory calculation and molecular dynamics simulation. Comput. Mater. Sci. 2020, 179. [Google Scholar] [CrossRef]
- Kragulj, M.; Tričković, J.; Kukovecz, Á.; Jović, B.; Molnar, J.; Rončević, S.; Kónya, Z.; Dalmacija, B. Adsorption of chlorinated phenols on multiwalled carbon nanotubes. RSC Adv. 2015, 5, 24920–24929. [Google Scholar] [CrossRef]
- Yang, K.; Wu, W.; Jing, Q.; Jiang, W.; Xing, B. Competitive adsorption of naphthalene with 2,4-dichlorophenol and 4-chloroaniline on multiwalled carbon nanotubes. Environ. Sci. Technol. 2010, 44, 3021–3027. [Google Scholar] [CrossRef] [PubMed]
- Tóth, A.; Törocsik, A.; Tombácz, E.; László, K. Competitive adsorption of phenol and 3-chlorophenol on purified MWCNTs. J. Colloid Interface Sci. 2012, 387, 244–249. [Google Scholar] [CrossRef]
- Asmaly, H.A.; Abussaud, B.; Ihsanullah; Saleh, T.A.; Gupta, V.K.; Atieh, M.A. Ferric oxide nanoparticles decorated carbon nanotubes and carbon nanofibers: From synthesis to enhanced removal of phenol. J. Saudi Chem. Soc. 2015, 19, 511–520. [Google Scholar] [CrossRef]
- Jiang, L.; Li, S.; Yu, H.; Zou, Z.; Hou, X.; Shen, F.; Li, C.; Yao, X. Amino and thiol modified magnetic multi-walled carbon nanotubes for the simultaneous removal of lead, zinc, and phenol from aqueous solutions. Appl. Surf. Sci. 2016, 369, 398–413. [Google Scholar] [CrossRef]
- Alves, D.C.S.; Gonçalves, J.O.; Coseglio, B.B.; Burgo, T.A.L. Adsorption of phenol onto chitosan hydrogel scaffold modified with carbon nanotubes. J. Environ. Chem. Eng. 2019, 7, 103460. [Google Scholar] [CrossRef]
- Strachowski, P.; Bystrzejewski, M. Comparative studies of sorption of phenolic compounds onto carbon-encapsulated iron nanoparticles, carbon nanotubes and activated carbon. Colloids Surf. A Physicochem. Eng. Asp. 2015, 467, 113–123. [Google Scholar] [CrossRef]
- Yi, L.; Zuo, L.; Wei, C.; Fu, H.; Qu, X.; Zheng, S.; Xu, Z.; Guo, Y.; Li, H.; Zhu, D. Enhanced adsorption of bisphenol A, tylosin, and tetracycline from aqueous solution to nitrogen-doped multiwall carbon nanotubes via cation-π and π-π electron-donor-acceptor (EDA) interactions. Sci. Total Environ. 2020, 719. [Google Scholar] [CrossRef]
- Abdel-Ghani, N.T.; El-Chaghaby, G.A.; Helal, F.S. Individual and competitive adsorption of phenol and nickel onto multiwalled carbon nanotubes. J. Adv. Res. 2015, 6, 405–415. [Google Scholar] [CrossRef]
- Zhang, Z.; Sun, D.; Li, G.; Zhang, B.; Zhang, B.; Qiu, S.; Li, Y.; Wu, T. Calcined products of Mg–Al layered double hydroxides/single-walled carbon nanotubes nanocomposites for expeditious removal of phenol and 4-chlorophenol from aqueous solutions. Colloids Surf. A Physicochem. Eng. Asp. 2019, 565, 143–153. [Google Scholar] [CrossRef]
- Chen, G.C.; Shan, X.Q.; Wang, Y.S.; Wen, B.; Pei, Z.G.; Xie, Y.N.; Liu, T.; Pignatello, J.J. Adsorption of 2,4,6-trichlorophenol by multi-walled carbon nanotubes as affected by Cu(II). Water Res. 2009, 43, 2409–2418. [Google Scholar] [CrossRef] [PubMed]
- Fathy, M.; Selim, H.; Shahawy, A.E.L. Chitosan/MCM-48 nanocomposite as a potential adsorbent for removing phenol from aqueous solution. RSC Adv. 2020, 10, 23417–23430. [Google Scholar] [CrossRef]
- Albayati, T.M.; Kalash, K.R. Polycyclic aromatic hydrocarbons adsorption from wastewater using different types of prepared mesoporous materials MCM-41in batch and fixed bed column. Process Saf. Environ. Prot. 2020, 133, 124–136. [Google Scholar] [CrossRef]
- Connolly, B.M.; Mehta, J.P.; Moghadam, P.Z.; Wheatley, A.E.H.; Fairen-Jimenez, D. From synthesis to applications: Metal–organic frameworks for an environmentally sustainable future. Curr. Opin. Green Sustain. Chem. 2018, 12, 47–56. [Google Scholar] [CrossRef]
- Jiang, N.; Shang, R.; Heijman, S.G.J.; Rietveld, L.C. High-silica zeolites for adsorption of organic micro-pollutants in water treatment: A review. Water Res. 2018, 144, 145–161. [Google Scholar] [CrossRef]
- Wen, Y.; Zhang, J.; Xu, Q.; Wu, X.; Zhu, Q. Pore surface engineering of metal—Organic frameworks for heterogeneous catalysis. Coord. Chem. Rev. 2018, 376, 248–276. [Google Scholar] [CrossRef]
- Zhang, Z.; Hou, X.; Zhang, X.; Li, H. The synergistic adsorption of pyrene and copper onto Fe(III) functionalized mesoporous silica from aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2017, 520, 39–45. [Google Scholar] [CrossRef]
- Nadar, S.S.; Varadan, N.; Suresh, S.; Rao, P.; Ahirrao, D.J.; Adsare, S. Recent progress in nanostructured magnetic framework composites (MFCs): Synthesis and applications. J. Taiwan Inst. Chem. Eng. 2018, 91, 653–677. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Anastopoulos, I. Adsorptive removal of bisphenol A (BPA) from aqueous solution: A review. Chemosphere 2017, 168, 885–902. [Google Scholar] [CrossRef]
- Mahmodi, G.; Zarrintaj, P.; Taghizadeh, A.; Taghizadeh, M.; Manouchehri, S.; Dangwal, S.; Ronte, A.; Ganjali, M.R.; Ramsey, J.D.; Kim, S.J.; et al. From microporous to mesoporous mineral frameworks: An alliance between zeolite and chitosan. Carbohydr. Res. 2020, 489. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.J.; Hameed, B.H.; Hummadi, E.H. Review on recent progress in chitosan/chitin-carbonaceous material composites for the adsorption of water pollutants. Carbohydr. Polym. 2020, 247. [Google Scholar] [CrossRef] [PubMed]
- Li, J.M.; Meng, X.G.; Hu, C.W.; Du, J. Adsorption of phenol, p-chlorophenol and p-nitrophenol onto functional chitosan. Bioresour. Technol. 2009, 100, 1168–1173. [Google Scholar] [CrossRef]
- Bibi, S.; Yasin, T.; Hassan, S.; Riaz, M.; Nawaz, M. Chitosan/CNTs green nanocomposite membrane: Synthesis, swelling and polyaromatic hydrocarbons removal. Mater. Sci. Eng. C 2015, 46, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Filho, C.M.C.; Bueno, P.V.A.; Matsushita, A.F.Y.; Rubira, A.F.; Muniz, E.C.; Durães, L.; Murtinho, D.M.B.; Valente, A.J.M. Synthesis, characterization and sorption studies of aromatic compounds by hydrogels of chitosan blended with β-cyclodextrin- and PVA-functionalized pectin. RSC Adv. 2018, 8, 14609–14622. [Google Scholar] [CrossRef]
- Bahmani, E.; Koushkbaghi, S.; Darabi, M.; ZabihiSahebi, A.; Askari, A.; Irani, M. Fabrication of novel chitosan-g-PNVCL/ZIF-8 composite nanofibers for adsorption of Cr(VI), As(V) and phenol in a single and ternary systems. Carbohydr. Polym. 2019, 224. [Google Scholar] [CrossRef]
- Karamipour, A.; Parsi, P.K.; Zahedi, P.; Moosavian, S.M.A. Using Fe3O4-coated nanofibers based on cellulose acetate/chitosan for adsorption of Cr(VI), Ni(II) and phenol from aqueous solutions. Int. J. Biol. Macromol. 2020, 154, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Lyu, H.; Hu, K.; Chu, Q.; Su, Z.; Xie, Z. Preparation of ionic liquid mediated molecularly imprinted polymer and specific recognition for bisphenol A from aqueous solution. Microchem. J. 2020, 158. [Google Scholar] [CrossRef]
- Ncube, S.; Kunene, P.; Tavengwa, N.T.; Tutu, H.; Richards, H.; Cukrowska, E.; Chimuka, L. Synthesis and characterization of a molecularly imprinted polymer for the isolation of the 16 US-EPA priority polycyclic aromatic hydrocarbons (PAHs) in solution. J. Environ. Manag. 2017, 199, 192–200. [Google Scholar] [CrossRef]
- Saad, N.; Chaaban, M.; Patra, D.; Ghanem, A.; El-rassy, H. Molecularly imprinted phenyl-functionalized silica aerogels: Selective adsorbents for methylxanthines and PAHs. Microporous Mesoporous Mater. 2020, 292, 109759. [Google Scholar] [CrossRef]
- Azizi, A.; Bottaro, C.S. A critical review of molecularly imprinted polymers for the analysis of organic pollutants in environmental water samples. J. Chromatogr. A 2020, 1614. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Liang, R.; Wang, Z.; Qin, W. Hydrophilic molecularly imprinted polymers for selective recognition of polycyclic aromatic hydrocarbons in aqueous media. RSC Adv. 2015, 5, 2659–2662. [Google Scholar] [CrossRef]
- Bayramoglu, G.; Arica, M.Y.; Liman, G.; Celikbicak, O.; Salih, B. Removal of bisphenol A from aqueous medium using molecularly surface imprinted microbeads. Chemosphere 2016, 150, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Balbino, T.A.C.; Bellato, C.R.; da Silva, A.D.; de Marques Neto, J.O.; de Guimarães, L.M. Magnetic cross-linked chitosan modified with ethylenediamine and β-cyclodextrin for removal of phenolic compounds. Colloids Surf. A Physicochem. Eng. Asp. 2020, 602. [Google Scholar] [CrossRef]
- Heydaripour, J.; Gazi, M.; Oladipo, A.A.; Gulcan, H.O. Porous magnetic resin-g-chitosan beads for adsorptive removal of phenolic compounds. Int. J. Biol. Macromol. 2019, 123, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, B.; Delnavaz, M.; Shakeri, A. Removal of Cd(II) and phenol using novel cross-linked magnetic EDTA/chitosan/TiO2 nanocomposite. Carbohydr. Polym. 2018, 181, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Rebekah, A.; Bharath, G.; Naushad, M.; Viswanathan, C.; Ponpandian, N. Magnetic graphene/chitosan nanocomposite: A promising nano-adsorbent for the removal of 2-naphthol from aqueous solution and their kinetic studies. Int. J. Biol. Macromol. 2020, 159, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.; Chen, W.; Liao, L.; Xie, P.; Zhang, T.C.; Wu, X.; Feng, X. Comparative adsorption of emerging contaminants in water by functional designed magnetic poly(N-isopropylacrylamide)/chitosan hydrogels. Sci. Total Environ. 2019, 671, 377–387. [Google Scholar] [CrossRef]
- Nguyen, M.L.; Huang, C.; Juang, R.S. Synergistic biosorption between phenol and nickel(II) from Binary mixtures on chemically and biologically modified chitosan beads. Chem. Eng. J. 2016, 286, 68–75. [Google Scholar] [CrossRef]
- Nadavala, S.K.; Swayampakula, K.; Boddu, V.M.; Abburi, K. Biosorption of phenol and o-chlorophenol from aqueous solutions on to chitosan-calcium alginate blended beads. J. Hazard. Mater. 2009, 162, 482–489. [Google Scholar] [CrossRef]
- Krupadam, R.J.; Korde, B.A.; Ashokkumar, M.; Kolev, S.D. Novel molecularly imprinted polymeric microspheres for preconcentration and preservation of polycyclic aromatic hydrocarbons from environmental samples. Anal. Bioanal. Chem. 2014, 406, 5313–5321. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, L. Highly selective detection of polycyclic aromatic hydrocarbons using multifunctional magnetic-luminescent molecularly imprinted polymers. ACS Appl. Mater. Interfaces 2013, 5, 10502–10509. [Google Scholar] [CrossRef] [PubMed]
- Tiu, B.D.B.; Krupadam, R.J.; Advincula, R.C. Pyrene-imprinted polythiophene sensors for detection of polycyclic aromatic hydrocarbons. Sens. Actuators B Chem. 2016, 228, 693–701. [Google Scholar] [CrossRef]
- Saleh, T.A.; Tuzen, M.; Sari, A. Magnetic vermiculite-modified by poly(trimesoyl chloride-melamine) as a sorbent for enhanced removal of bisphenol A. J. Environ. Chem. Eng. 2019, 7. [Google Scholar] [CrossRef]
- An, F.; Gao, B.; Feng, X. Adsorption and recognizing ability of molecular imprinted polymer MIP-PEI/SiO2 towards phenol. J. Hazard. Mater. 2008, 157, 286–292. [Google Scholar] [CrossRef]
- Liang, W.; Lu, Y.; Li, N.; Li, H.; Zhu, F. Microwave-assisted Synthesis of Magnetic Surface Molecular Imprinted Polymer for Adsorption and Solid Phase Extraction of 4-nitrophenol in wastewater. Microchem. J. 2020, 105316. [Google Scholar] [CrossRef]
- Zhao, Z.; Fu, D.; Zhang, B. Novel molecularly imprinted polymer prepared by palygorskite as support for selective adsorption of bisphenol A in aqueous solution. Desalin. Water Treat. 2016, 57, 12433–12442. [Google Scholar] [CrossRef]
- Nasreen, S.; Rafique, U.; Ehrman, S.; Ashraf, M.A. Synthesis and characterization of mesoporous silica nanoparticles for environmental remediation of metals, PAHs and phenols. Ekoloji 2018, 27, 1625–1637. [Google Scholar]
- Costa, J.A.S.; Sarmento, V.H.V.; Romão, L.P.C.; Paranhos, C.M. Removal of polycyclic aromatic hydrocarbons from aqueous media with polysulfone/MCM-41 mixed matrix membranes. J. Membr. Sci. 2020, 601. [Google Scholar] [CrossRef]
- Balati, A.; Shahbazi, A.; Amini, M.M.; Hashemi, S.H.; Jadidi, K. Comparison of the efficiency of mesoporous silicas as absorbents for removing naphthalene from contaminated water. Eur. J. Environ. Sci. 2014, 4, 69–76. [Google Scholar] [CrossRef]
- Yuan, P.; Li, X.; Wang, W.; Liu, H.; Yang, H.; Yue, Y.; Bao, X. Tailored design of differently modified mesoporous materials to deeply understand the adsorption mechanism for PAHs. Langmuir 2018. [Google Scholar] [CrossRef] [PubMed]
- Yangui, A.; Abderrabba, M.; Sayari, A. Amine-modified mesoporous silica for quantitative adsorption and release of hydroxytyrosol and other phenolic compounds from olive mill wastewater. J. Taiwan Inst. Chem. Eng. 2017, 70, 111–118. [Google Scholar] [CrossRef]
- Zou, D.; Liu, D. Understanding the modifications and applications of highly stable porous frameworks via UiO-66. Mater. Today Chem. 2019, 12, 139–165. [Google Scholar] [CrossRef]
- Elsabawy, K.M.; Fallatah, A.M. New advanced approach of ultra-fast synthesis of ultrahigh-BET-surface area crystalline metal-organic-frameworks MOFs. Mater. Lett. 2018, 224, 71–74. [Google Scholar] [CrossRef]
- Jiang, D.; Chen, M.; Wang, H.; Zeng, G.; Huang, D.; Cheng, M.; Liu, Y.; Xue, W.; Wang, Z.W. The application of different typological and structural MOFs-based materials for the dyes adsorption. Coord. Chem. Rev. 2019, 380, 471–483. [Google Scholar] [CrossRef]
- Mahmoodi, N.M.; Oveisi, M.; Asadi, E. Synthesis of NENU metal-organic framework-graphene oxide nanocomposites and their pollutant removal ability from water using ultrasound. J. Clean. Prod. 2019, 211, 198–212. [Google Scholar] [CrossRef]
- Zango, Z.U.; Sambudi, N.S.; Jumbri, K.; Abu Bakar, N.H.H.; Abdullah, N.A.F.; Negim, E.S.M.; Saad, B. Experimental and molecular docking model studies for the adsorption of polycyclic aromatic hydrocarbons onto UiO-66(Zr) and NH2-UiO-66(Zr) metal-organic frameworks. Chem. Eng. Sci. 2020, 220, 115608. [Google Scholar] [CrossRef]
- Zango, Z.U.; Abu Bakar, N.H.H.; Sambudi, N.S.; Jumbri, K.; Abdullah, N.A.F.; Abdul Kadir, E.; Saad, B. Adsorption of chrysene in aqueous solution onto MIL-88(Fe) and NH2-MIL-88(Fe) metal-organic frameworks: Kinetics, isotherms, thermodynamics and docking simulation studies. J. Environ. Chem. Eng. 2019. [Google Scholar] [CrossRef]
- Lin, K.A.; Hsieh, Y. Copper-based metal organic framework (MOF), HKUST-1, as an efficient adsorbent to remove p-nitrophenol from water. J. Taiwan Inst. Chem. Eng. 2015, 50, 223–228. [Google Scholar]
- Xu, Z.; Wen, Y.; Tian, L.; Li, G. Efficient and selective adsorption of nitroaromatic explosives by Zr-MOF. Inorg. Chem. Commun. 2017, 77, 11–13. [Google Scholar] [CrossRef]
- Luo, Z.; Chen, H.; Wu, S.; Yang, C.; Cheng, J. Enhanced removal of bisphenol A from aqueous solution by aluminum-based MOF/sodium alginate-chitosan composite beads. Chemosphere 2019, 237. [Google Scholar] [CrossRef] [PubMed]
- Balati, A.; Shahbazi, A.; Amini, M.M.; Hashemi, S.H. Adsorption of polycyclic aromatic hydrocarbons from wastewater by using silica-based organic–inorganic nanohybrid material. J. Water Reuse Desalin. 2015, 5, 50–63. [Google Scholar] [CrossRef]
- Costa, J.A.S.; Sarmento, V.H.V.; Romão, L.P.C.; Paranhos, C.M. Synthesis of functionalized mesoporous material from rice husk ash and its application in the removal of the polycyclic aromatic hydrocarbons. Environ. Sci. Pollut. Res. 2019, 26, 25476–25490. [Google Scholar] [CrossRef] [PubMed]
- Anbia, M.; Amirmahmoodi, S. Adsorption of phenolic compounds from aqueous solutions using functionalized SBA-15 as a nano-sorbent. Sci. Iran. 2011, 18, 446–452. [Google Scholar] [CrossRef]
- Nasreen, S.; Rafique, U.; Ehrman, S.; Ashraf, M.A. Hybrid mesoporous silicates: A distinct aspect to synthesis and application for decontamination of phenols. Saudi J. Biol. Sci. 2019, 26, 1161–1170. [Google Scholar] [CrossRef]
- Asgharnia, H.; Nasehinia, H.; Rostami, R.; Rahmani, M.; Mehdinia, S.M. Phenol removal from aqueous solution using silica and activated carbon derived from rice husk. Water Pract. Technol. 2019, 14, 897–907. [Google Scholar] [CrossRef]
- Zango, Z.U.; Jumbri, K.; Sambudi, N.S.; Abu Bakar, N.H.H.; Abdullah, N.A.F.; Basheer, C.; Saad, B. Removal of anthracene in water by MIL-88(Fe), NH2-MIL-88(Fe), and mixed-MIL-88(Fe) metal–organic frameworks. RCS Adv. 2019, 9, 41490–41501. [Google Scholar] [CrossRef]
- Zango, Z.U.; Sambudi, N.S.; Jumbri, K.; Abu Bakar, N.H.H.; Saad, B. Removal of Pyrene from Aqueous Solution Using Fe-based Metal-organic Frameworks. IOP Conf. Ser. Earth Environ. Sci. 2020, 549, 012061. [Google Scholar] [CrossRef]
- Zhou, M.; Wu, Y.-n.; Qiao, J.; Zhang, J.; McDonald, A.; Li, G.; Li, F. The removal of bisphenol A from aqueous solutions by MIL-53(Al) and mesostructured MIL-53(Al). J. Colloid Interface Sci. 2013, 405, 157–163. [Google Scholar] [CrossRef]
- Tan, Y.; Sun, Z.; Meng, H.; Han, Y.; Wu, J.; Xu, J.; Xu, Y.; Zhang, X. A new MOFs/polymer hybrid membrane: MIL-68(Al)/PVDF, fabrication and application in high-efficient removal of p-nitrophenol and methylene blue. Sep. Purif. Technol. 2019, 215, 217–226. [Google Scholar] [CrossRef]
- Han, T.; Li, C.; Guo, X.; Huang, H.; Liu, D.; Zhong, C. In-situ synthesis of SiO2@MOF composites for high-efficiency removal of aniline from aqueous solution. Appl. Surf. Sci. 2016, 390, 506–512. [Google Scholar] [CrossRef]
- Abazari, R.; Salehi, G.; Mahjoub, A.R. Ultrasound-assisted preparation of a nanostructured zinc(II) amine pillar metal-organic framework as a potential sorbent for 2,4-dichlorophenol adsorption from aqueous solution. Ultrason. Sonochem. 2018, 46, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Abazari, R.; Mahjoub, A.R. Ultrasound-assisted synthesis of Zinc(II)-based metal organic framework nanoparticles in the presence of modulator for adsorption enhancement of 2,4-dichlorophenol and amoxicillin. Ultrason. Sonochem. 2018, 42, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Xiao, Y.; Tong, M.; Huang, H.; Liu, D.; Wang, L.; Zhong, C. Synthesis of CNT@MIL-68(Al) composites with improved adsorption capacity for phenol in aqueous solution. Chem. Eng. J. 2015, 275, 134–141. [Google Scholar] [CrossRef]
- Jung, B.K.; Hasan, Z.; Jhung, S.H. Adsorptive removal of 2,4-dichlorophenoxyacetic acid (2,4-D) from water with a metal–organic framework. Chem. Eng. J. 2013, 234, 99–105. [Google Scholar] [CrossRef]
- Guo, H.; Niu, B.; Wu, X.; Zhang, Y.; Ying, S. Effective removal of 2,4,6-trinitrophenol over hexagonal metal–organic framework NH2-MIL-88B(Fe). Appl. Organomet. Chem. 2018, 33, e4580. [Google Scholar] [CrossRef]
- Giraldo, L.; Bastidas-Barranco, M.; Húmpola, P.; Moreno-Piraján, J.C. Design, synthesis and characterization of MOF-199 and ZIF-8: Applications in the adsorption of phenols derivatives in aqueous solution. Eur. J. Chem. 2017, 8, 293–304. [Google Scholar] [CrossRef]
- Ahsan, M.A.; Jabbari, V.; Islam, M.T.; Turley, R.S.; Dominguez, N.; Kim, H.; Castro, E.; Hernandez-Viezcas, J.A.; Curry, M.L.; Lopez, J.; et al. Sustainable synthesis and remarkable adsorption capacity of MOF/graphene oxide and MOF/CNT based hybrid nanocomposites for the removal of Bisphenol A from water. Sci. Total Environ. 2019, 673, 306–317. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, L.; Han, J.; Wu, J.; Li, C.; Ni, L.; Wang, Y. Improving laccase activity and stability by HKUST-1 with cofactor via one-pot encapsulation and its application for degradation of bisphenol A. J. Hazard. Mater. 2020, 383. [Google Scholar] [CrossRef]
- Kamali, M.; Persson, K.M.; Costa, M.E.; Capela, I. Sustainability criteria for assessing nanotechnology applicability in industrial wastewater treatment: Current status and future outlook. Environ. Int. 2019, 125, 261–276. [Google Scholar] [CrossRef]
- El-Naas, M.H.; Surkatti, R.; Al-Zuhair, S. Petroleum refinery wastewater treatment: A pilot scale study. J. Water Process Eng. 2016, 14, 71–76. [Google Scholar] [CrossRef]
- Kalmykova, Y.; Moona, N.; Strömvall, A.M.; Björklund, K. Sorption and degradation of petroleum hydrocarbons, polycyclic aromatic hydrocarbons, alkylphenols, bisphenol A and phthalates in landfill leachate using sand, activated carbon and peat filters. Water Res. 2014, 56, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Frascari, D.; Bacca, A.E.M.; Zama, F.; Bertin, L.; Fava, F.; Pinelli, D. Olive mill wastewater valorisation through phenolic compounds adsorption in a continuous flow column. Chem. Eng. J. 2016, 283, 293–303. [Google Scholar] [CrossRef]
| Compound | Formula | Abbreviation | Structure | Molecular Weight (g/mol) | Partition Coefficient |
|---|---|---|---|---|---|
| Naphthalene | C10H8 | NAP | 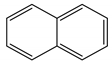 | 128.17 | 3.37 |
| Acenaphthylene | C12H8 | ACE | 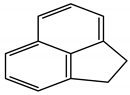 | 152.1 | 4.0 |
| Acenaphthene | C1H10 | ACE | 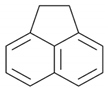 | 154.21 | 3.92 |
| Fluorene | C13H10 | FLU |  | 166.22 | 4.18 |
| Phenanthrene | C14H10 | PHE | 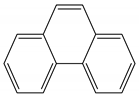 | 178.23 | 4.57 |
| Anthracene | C14H10 | ANT |  | 178.23 | 4.54 |
| Fluoranthene | C16H10 | FL | 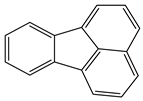 | 202.25 | 4.58 |
| Pyrene | C16H10 | PYR |  | 202.25 | 4.58 |
| Benz[a]anthracene | C18H12 | B[a]A |  | 228.3 | 6.14 |
| Chrysene | C18H12 | CRY | 228.29 | 5.30 | |
| Benzo[b]fluoranthene | C20H12 | BbF | 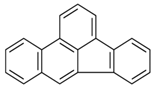 | 252.3 | 5.74 |
| Benz[k]fluoranthene | C20H12 | BkF | 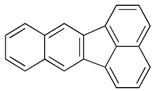 | 252.3 | 5.74 |
| Benz[a]pyrene | C20H12 | BaP | 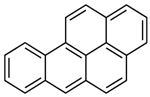 | 252.3 | 6.74 |
| Benzo[ghi]perylene | C22H12 | BhP | 276.3 | 6.52 | |
| Dibenz[a,h]anthracene | C22H14 | DahA | 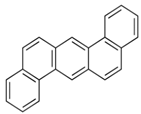 | 278.4 | 6.20 |
| Indeno[1,2,3-c,d]pyrene | C22H12 | IP |  | 276.3 | 6.20 |
| Compound | Formula | Structure | Molecular Weight (g/mol) | Partition Coefficient |
|---|---|---|---|---|
| Phenol | C6H6O |  | 94.11 | 10.0 |
| Cresol | C7H8O |  | 108.14 | 10.28 |
| Resorcinol | C6H6O2 | 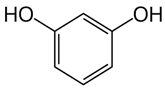 | 110.1 | 19.15 |
| Hydroquinone | C6H6O2 | 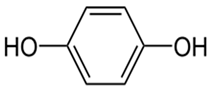 | 110.11 | 9.9 |
| Pyrogallol | C6H6O3 |  | 126.11 | 9.03 |
| Naphthol | C6H8O | 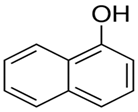 | 144.17 | 9.51 |
| Salicylic acid | C7H6O3 | 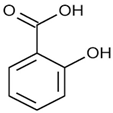 | 178.23 | 2.97 |
| Picric acid | C6H3N3O7 |  | 229.1 | 0.38 |
| Bisphenol A | C15H16O2 | 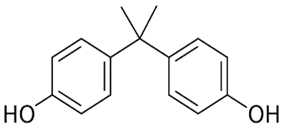 | 228.29 | 10.29 |
| Chlorophenol | C6H5ClO | 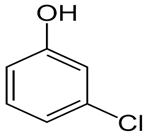 | 128.6 | 9.12 |
| Nitrophenol | C6H5NO3 |  | 139.10 | 7.15 |
| Method | Pros | Cons |
|---|---|---|
| Coagulation |
|
|
| Flocculation |
|
|
| Bioremediation; including the use of microbes such as bacteria, fungi, and algae. |
|
|
| Reverse Osmosis |
|
|
| Chemical oxidations |
|
|
| Solvent extraction and ion exchange process |
|
|
| Photocatalytic degradation |
|
|
| Adsorption process |
|
|
| Adsorbent | Pollutant | Concentration (mg/L) | Qe (mg/g) | Equilibrium Time | Ref |
|---|---|---|---|---|---|
| RHB | PHE | 5 | 17 | 1 h | [78] |
| Magnetic modified RHB | 42 | ||||
| Biochar | PHE | 1 | 2 | 3 days | [83] |
| graphene/biochar composite | 3 | ||||
| Rice-350-M | PHE | 1 | 43 | 24 h | [69] |
| Elephant grass biochar | BaA | 0.1 | 19 | 250 min | [84] |
| BkF | 19 | ||||
| BaP | 18 | ||||
| DahA | 8 | ||||
| Poplar Catkins biochar | PHE | 30 | 384 | 1120 min | [85] |
| Sewage sludge char | PHE | 0.8 | - | 24 h | [56] |
| PYR | - | ||||
| Wood biochar | PYR | ||||
| Wheat straw biochar | PHE | 9.1 | - | 48 h | [79] |
| FLU | 10.0 | - | |||
| PYR | 11 | - | |||
| Chitin biochar | phenol | 500 | 184 | 12 h | |
| 2-nitrphenol | 206 | ||||
| Hizikia fusiformis biochar | phenol | 50 | 30 | 1440 min | [86] |
| Black spruce biochar | phenol | 200 | 233 | 18 h | [87] |
| White birch biochar | 150 | 4 h | |||
| Chinese herb biochar | phenol | 50 | - | 360 min | [88] |
| scots pine bark biochar | phenol | 500 | 149 | 24 h | [89] |
| spruce bark biochar | 84 | ||||
| Bamboo biochar | phenol | 10 | 17 | 24 h | [90] |
| Oak wood biochar | 13 | ||||
| Food waste biochar | phenol | 10 | 3 | 30 min | [20] |
| Japanese red pine char | Phenol | 200 | 6 | 30 min | [61] |
| Yellow poplar char | Phenol | 7 | |||
| Pine fruit shell biochar | Phenol | 50 | 27 | 24 h | [91] |
| Adsorbent | Pollutant | Concentration (mg/g) | Qe (mg/g) | Equilibrium Time | Ref |
|---|---|---|---|---|---|
| Commercial PAC | ANT | - | 143 | 4 h, 5 min | [68] |
| PYR | 142 | 4 h, 10 min | |||
| Commercial AC | PHE | 120 | 46 | 24 h | [104] |
| FLA | 80 | 36 | |||
| BaA | 12 | 9.7 | |||
| Rice husk AC | NAP | 8 | 14 | 24 h | [66] |
| PHE | 17 | ||||
| PYR | 18 | ||||
| Flamboyant pod back AC | ACE | 50 | 4.7 | 2 h | [105] |
| Milk bush kernel AC | 4 | ||||
| Rice husk AC | 5.6 | ||||
| Rice husk AC | NAP | 8 | 15 | 24 h | [106] |
| PHE | 17.5 | ||||
| PYR | 18 | ||||
| Rice husk AC | PHE | 10 | - | 60 min | [107] |
| FLU | - | 30 min | |||
| Rice husk AC | ACE | 50 | 46 | 3 h | [93] |
| Vitis vinifera leaf AC (Zncl2) | PHE | 1 | 94 | 180 min | [108] |
| Vitis vinifera leaf AC (H3PO4) | 89 | ||||
| Walnut shell AC | NAP | 10 | 49.6 | 40 min | [103] |
| PHE | 10 | 63.8 | |||
| flamboyant pod AC | NAP | 50 | 294.2 | 2 h | [94] |
| milk bush kernel shell AC | 63.3 | ||||
| Coal-based AC | NAP | 200 | 78.2 | 20 min | [98] |
| PHE | 111.4 | ||||
| PYR | 117.2 | ||||
| Fe-modified Coal-based AC | NAP | 30 | 167.8 | 40 min | [101] |
| PHE | 190.4 | ||||
| PYR | 20.3 | ||||
| Terminalia chebula AC | phenol | 100 | 26.2 | 24 h | [109] |
| Commercial AC | phenol | 1000 | 132.3 | 360 min | [110] |
| Commercial AC | phenol | ||||
| Miswak root AC | phenol | 100 | 142.2 | 120 min | [111] |
| Coal-based GAC | phenol | 300 | 169.9 | 180 min | [112] |
| Coal-based PAC | 176.6 | ||||
| Coconut shell PAC | 213 | ||||
| Cherry stone AC | phenol | 25 | 80 | 24 h | [113] |
| Agave Utahensis AC | Phenol | 14 | 1.7 | 3h | [114] |
| Euphorbia Resinifira AC | 1.9 | ||||
| Banana peel AC | 2.0 | ||||
| Oily sludge AC | phenol | 100 | 434 | 30 min | [115] |
| Tea waste AC | phenol | 100 | 132.5 | 4 h | [67] |
| Babul sawdust AC | phenol | 50 | 15.3 | 60 min | [96] |
| Olive stone AC | phenol | 800 | 44,269 | 4 h | [116] |
| Palm oil bunch AC | phenol | 40 | 55.3 | 2 h | [117] |
| Wood fiberboard AC | phenol | 250 | 208 | 120 min | [95] |
| Avocado kernel AC | phenol | 0.7 | 3.3 | 100 min | [118] |
| Date pit AC | Phenol | 50 | 46 | 1 h | [119] |
| Sugar cane bagasse AC | Phenol | 100 | 27 | 30 min | [120] |
| Lantana camara AC | Phenol | 150 | 112.5 | 7 h | [121] |
| Magnetic AC | phenol | 80 | 107.5 | 15 min | [122] |
| Granular activated carbon (GAC) | Phenol, | 50 | 4.9 | 60 min | [123] |
| 2-chlorophenol | 4.3 | ||||
| 4-chlorophenol | 4.5 | ||||
| Red pine AC | Phenol | 200 | 454.5 | 30 min | [61] |
| Yellow poplar AC | 625 | ||||
| Commercial AC | 500 |
| Adsorbent | Pollutant | Concentration (mg/L) | Qe (mg/g) | Equilibrium Time | Ref |
|---|---|---|---|---|---|
| Graphene wool | PHE | 0.8 | 5 | 24 h | [126] |
| PYR | 5 | 200 | |||
| rGO | NAP | 30 | 16 | 7 days | [125] |
| ANT | 0.05 | 247 | |||
| PYR | 0.1 | 369 | |||
| GO/FeO⋅Fe2O3 | NAP | 0.1 | 6 | 48 h | [64] |
| Graphene GO | NAP | 4.6 | 1 66 | 4 h | [63] |
| Graphene GO | NAP | - | 9 8 | 10 min | [141] |
| Sulfonated graphene | NAP | 0.39 | 2 | 5 days | [142] |
| rGO@Sand | NAP ACE | 0.8 | 4 6 | 120 min | [143] |
| Magnetic graphene nanosheet | PHE | 0.5 | 0.02 | 1 h | [72] |
| Magnetic GO Magnetic chemically GO Magnetic rG | PHE | 1 | 14 30 26 | 24 h | [60] |
| TiO2-Graphene | PHE | 4.2 | 1.2 | 180 min | [59] |
| FLAN | 4.5 | 0.9 | |||
| BaP | 2.5 | 0.5 | |||
| GO rG Reduced graphene | NAP | 20 | 21 52 145 | 24 h | [144] |
| Fe3O4@PANI-GO | Bisphenol A | 5 | 14 | 5 min | [137] |
| t-octyl phenol | 5 | 31 | 5 min | ||
| α-naphthol | 10 | 23 | 300 min | ||
| rGO | 2-phenylphenol | 0.470 | 12 | 60 min | [138] |
| bisphenol A | 0.350 | 1.56 | |||
| 4-isopropylphenol | 0.587 | 1.117 | |||
| 4-methylphenol | 0.550 | 0.841 | |||
| phenol | 0.850 | 0.483 | |||
| CTAB-modified graphite | bisphenol A | 300 | 125 | 10 min | [129] |
| Amino-functionalized GO | Bisphenol A | 1 | 2.3 | 3.4 s | [139] |
| rGO GO | phenol | 50 | 16 23 | ||
| GO | bisphenol A 4-nonylphenol tetrabromobisphenol A 2,4,6-trichlorophenol | 20 | 21 10 11 18 | 1400 min | [14] |
| Graphene | 4-chloro-2-nitrophenol | 10 | 25 | 60 min | [145] |
| GO rGO | Phenol | 50 | 23 16.32 | 100 min | [146] |
| MGO-IL | phenol | 30 | 741 | 12 min | [147] |
| Graphene | Bisphenol A | 10 | 87 | 5 h | [62] |
| Sulfonated graphene | 1-naphthol | 0.7 | 2.41 | 5 days | [142] |
| Graphene | phenol | 50 | 28 | 400 min | [148] |
| GO sheet GO sponge | 2,4,6-trichlorophenol | 5 | 10 21 | 30 min 12 min | [149] |
| GO | Bisphenol A | - | 49 | 30 min | [150] |
| GO | tetrabromobisphenol A | 1 | 41 | 120 min | [151] |
| GO | p-nitrophenol | 200 | 138 | 24 h | [51] |
| GO Chemically GO rG | 1-naphthol | 50 | 21 52 145 | 24 h | [144] |
| GO Ultrasonic GO Conventional GO Ultrasonic GO | 2-chlorophenol | 50 | 32 134 50 209 | 50 min | [152] |
| Adsorbent | Pollutant | Concentration (mg/L) | Qe (mg/g) | Equilibrium Time | Ref |
|---|---|---|---|---|---|
| MWCNT | NAP | 200 | 69.2 | 5 days | [173] |
| Commercial MWCNT | NAP PHE PYR | - | 10.9 10.6 10.6 | 5 days | [167] |
| Magnetic SWCNT Magnetic MWCNT | PHE | 500 | 24.82 19.37 | 30 min | [72] |
| MWCNT | phenol 3-chlorophenol | 0.147 1.25 | 0.13 0.38 | 3 days | [174] |
| CNT-polymer bead | p-chlorophenol | 100 | 24 | 1200 min | [71] |
| CNT-polyethylene glycol | phenol | 20 | 27 | 40 min | [54] |
| CNT-polymer composite | phenol | 30 | 262 | 50 min | [162] |
| SWCNT MWCNT | phenol | 50 | 51 65 | 54 min 36 min | [127] |
| CNT CNT-Fe2O3 | phenol | 2 | 1.1 2.8 | 2 h | [175] |
| Magnetic-N2H4-SH/MWCNT | phenol | 40 | 39 | 700 min | [176] |
| Chitosan hydrogel-CNT | phenol | 200 | 404 | 120 min | [177] |
| CNT CNT-Al2O3 | phenol 4-chlorophenol phenol 4-chlorophenol | 2 | 1.1 1.3 2.8 2.8 | 120 min | [34] |
| CNT CNT-deep eutectic solvent | phenol | 50 | 62 120 | 24 h | [13] |
| Chitosan-MWCNT | phenol | 150 | 88 | 24 h | [17] |
| CNT CNT-activated | phenol 2-chlorophenol 4-chlorophenol phenol 2-chlorophenol 4-chlorophenol | 150 | 24 86 52 64 240 105 | 50 min 50 min 30 min 50 min 50 min 30 min | [178] |
| MWCNT Nitrogen doped-MWCNT | bisphenol A | 0.37 | 111 440 | 3 days | [179] |
| MWCNT | phenol | 25 | 32 | 300 min | [180] |
| SWCNT SWCNTO MWCNT MCNTO | phenol | 50 | 20 21 18 16 | 100 min | [146] |
| CNT-Mg-Al hydroxide | phenol 4-chlorophenol | 50 | 13 14 | [181] | |
| MWCNT | 2,4,6-trichlorophenol | 120 | 2.3 | 48 h | [182] |
| Adsorbent | Pollutant | Concentration (mg/L) | Qe (mg/g) | Equilibrium Time | Ref |
|---|---|---|---|---|---|
| Chitosan | |||||
| Pec-β-CD/Chitosan | PYR B[b]F B[a]P | 2 | 0.1 0.2 0.2 | 1200 min | [195] |
| Chitosan | phenol | ||||
| Chitosan/CNT | phenol | 400 | 404 | 120 min | [177] |
| Chitosan-cyclodextrin | phenol | 150 | 34 | - | [193] |
| p-chlorophenol | 157 | 180 | |||
| p-nitrophenol | 150 | 21 | |||
| chitosan-g-PNVCL/ZIF-8 | Phenol | 20 | 152 | 30 min | [196] |
| CA/Chitosan Fe2O3-CA/Chitosan | phenol | 100 | 97 163 | 5 h | [197] |
| Chitosan MWCNT-Chitosan | phenol | 150 | 62 87 | 24 h | [16] |
| Fe-EDA/β-CD/Chitosan | phenol p-nitrophenol p-cresol | 50 | 243 274 298 | 120 min | [204] |
| Magnetic chitosan | phenol | 15 | 52 | 50 min | [19] |
| Resin-chitosan | phenol 4-chlorophenol | 50 | 189 99 | 200 min 350 min | [205] |
| EDTA/Chitosan/TiO2 | phenol | 50 | 209 | 240 min | [206] |
| Chitosan-Carbon composite | phenol | 10 | 34 | 1 h | |
| Magnetic graphene-Chitosan | 2-napthol | 40 | 169 | 60 min | [207] |
| β-cyclodextrin-chitosan | phenol 2-chlorophenol 4-chlorophenol 2,4-dichlorophenol 2,4,6-trichlorophenol | 120 | 60 71 96 315 376 | 3 h | [22] |
| Magnetic-Chitosan | bisphenol A | 100 | 55 | 120 min | [208] |
| Glutaraldehyde-Chitosan | phenol | 50 | 21 | 60 min | [209] |
| phenol o-chlorophenol | 100 | 71 51 | 240 min | [210] | |
| MIPs | |||||
| MIP | BaP PYR CHR | 1 | 75.9 7 7 | 3 h | [211] |
| MIP | PHE | 0.6 | 16 | 90 min | [212] |
| MIP | PYR | 1 | 11 | 90 min | [213] |
| MIP | bisphenol A | 150 | 77 | 120 min | [203] |
| vermiculite MP magnetic vermiculite MP | bisphenol A | 1000 | 217 274 | 90 min | [214] |
| Surface-MIP | phenol p-nitrophenol p-tert-butylphenol | 0.75 | 86 8 7 | 4 h | [50] |
| MIP/PES/SiO2 | phenol p-nitrophenol | 4000 | 47 37 | 8 h | [215] |
| AMPS-Am-MIP | Phenol | 50 | 97 | 180 min | [18] |
| MIP IL-MIP | Bisphenol A | 100 | 87 116 | 20 min | [198] |
| Fe3O4@SiO2@PNP-SMIP | 4-nitophenol | 210 | 134 | 60 min | [216] |
| MIP SMIP | bisphenol-A | 20 | 24 23 | 240 min | [217] |
| Adsorbent | Pollutant | Concentration (mg/L) | Qe (mg/g) | Equilibrium Time | Ref |
|---|---|---|---|---|---|
| Mesoporous Silica | |||||
| MCM-41 | NAP | 15 | 61 | 20 min | [184] |
| Periodic mesoporous organosilica | NAP ACE FLU FLUO PYR | 10 | 1.5 0.9 0.9 0.7 1.3 | 24 h | |
| Fe-SBA-15 | PYR | 0.1 | 0.034 | 20 min | [188] |
| NH2-SBA-15 | NAP ACE PHE | 1 | 1.9 1.4 0.8 | 24 h | [232] |
| MCM-41-PABA | NAP B[b]FLUO B[k]FLUO B[b]PYR | 200 μg/L | 17.83 20 19 19 | 24 h | [233] |
| MCM-48 SBA-15 | NAP | 7 | 0.7 0.4 | 24 h | [220] |
| NH2-SBA-15 | 4-chlorophenol 4-bromophenol 4-iodophenol | 250 | 141 115 72 | 6 h | [234] |
| MCM-41- | phenol o-chlorophenol | 50 | 12 30 | 24 h | [222] |
| MCM-48-Chitosan | Phenol | 500 | 149 | 24 h | [183] |
| SBA-15 | 4-aminophenol 4-nitrophenol | - | 10 8 | 20 min | [235] |
| Rice husk silica | phenol | 15 | 4 | 60 min | [236] |
| MCM-41 | phenol | 60 | |||
| MOFs | |||||
| UiO-66(Zr) NH2-UiO-66(Zr) | ANT CRY ANT CRY | 4 | 24 24 22 19 | 30 min 25 min 30 min 25 min | [227] |
| MIL-88(Fe) NH2-MIL-88(Fe) | CRY | 4 | 24 22 | 25 min | [228] |
| MIL-88(Fe) Mixed-MIL-88(Fe) NH2-MIL-88(Fe) | ANT | 4 | 24 23 21 | 30 min | [237] |
| MIL-88(Fe) NH2-MIL-88(Fe) | PYR | 4 | 24 23 | 40 min | [238] |
| MIL-53(Al) MIL-53(Al)-F127 | bisphenol A | 250 | 329 473 | 90 min 30 min | [239] |
| MIL-68(Al)/PVDF | p-nitrophenol | 10 | 126 | 720 min | [240] |
| HKUST-1(Cu) | p-nitrophenol | 200 | 400 | 30 min | [229] |
| SiO2@MIL-68(Al) | aniline | 3000 | 532 | 40 s | [241] |
| [Zn(ATA)(BPD)] MOF-VII | 2,4-dichlorophenol | 60 | - - | 90 min | [242] |
| [Zn(TDC) MOF | 2, 4-dichloropheno | 60 | - | 180 min | [243] |
| MIL-68(Al) CNT@MIL-68(Al) | phenol phenol | 1000 | 118 257 | 120 min | [244] |
| NH2-UiO-66(Zr) | 2,4,6-trinitrophenol Styphnic acid 2,4-dinitrophenol 2,4,6-tri- nitrotoluene 2,4-dinitrotoluene | 100 | 23 24 30 0.5 2 | 36 h | [230] |
| MIL-53(Al) | dichlorophenoxyacetic acid | 150 | 556 | 1 h | [245] |
| MIL–68(Al) MIL–68(Al)/GO | p–nitrophenol | 300 | 271 332 | 17 h 17 h | [51] |
| NH2-MIL-88(Fe) | 2,4,6-trinitrophenol | 35 | 164 | 40 min | [246] |
| MOF-199(Cu) | Phenol p-nitro phenol | 500 | 58 68 | 300 min 30 min | [247] |
| Al-MOF/SA-CS | bisphenol A | 50 | 137 | 18 h | [231] |
| Cu-BDC MOF Cu-BDC@GrO Cu-BDC@CNT | bisphenol A | 200 | 60 182 164 | 40 min | [248] |
| laccase@HKUST-1 | bisphenol A | 200 | - | 4 h | [249] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zango, Z.U.; Sambudi, N.S.; Jumbri, K.; Ramli, A.; Abu Bakar, N.H.H.; Saad, B.; Rozaini, M.N.H.; Isiyaka, H.A.; Osman, A.M.; Sulieman, A. An Overview and Evaluation of Highly Porous Adsorbent Materials for Polycyclic Aromatic Hydrocarbons and Phenols Removal from Wastewater. Water 2020, 12, 2921. https://doi.org/10.3390/w12102921
Zango ZU, Sambudi NS, Jumbri K, Ramli A, Abu Bakar NHH, Saad B, Rozaini MNH, Isiyaka HA, Osman AM, Sulieman A. An Overview and Evaluation of Highly Porous Adsorbent Materials for Polycyclic Aromatic Hydrocarbons and Phenols Removal from Wastewater. Water. 2020; 12(10):2921. https://doi.org/10.3390/w12102921
Chicago/Turabian StyleZango, Zakariyya Uba, Nonni Soraya Sambudi, Khairulazhar Jumbri, Anita Ramli, Noor Hana Hanif Abu Bakar, Bahruddin Saad, Muhammad Nur’ Hafiz Rozaini, Hamza Ahmad Isiyaka, Abubaker Mohammed Osman, and Abdelmoneim Sulieman. 2020. "An Overview and Evaluation of Highly Porous Adsorbent Materials for Polycyclic Aromatic Hydrocarbons and Phenols Removal from Wastewater" Water 12, no. 10: 2921. https://doi.org/10.3390/w12102921
APA StyleZango, Z. U., Sambudi, N. S., Jumbri, K., Ramli, A., Abu Bakar, N. H. H., Saad, B., Rozaini, M. N. H., Isiyaka, H. A., Osman, A. M., & Sulieman, A. (2020). An Overview and Evaluation of Highly Porous Adsorbent Materials for Polycyclic Aromatic Hydrocarbons and Phenols Removal from Wastewater. Water, 12(10), 2921. https://doi.org/10.3390/w12102921








