Massive Influx of Pelagic Sargassum spp. on the Coasts of the Mexican Caribbean 2014–2020: Challenges and Opportunities
Abstract
1. Introduction
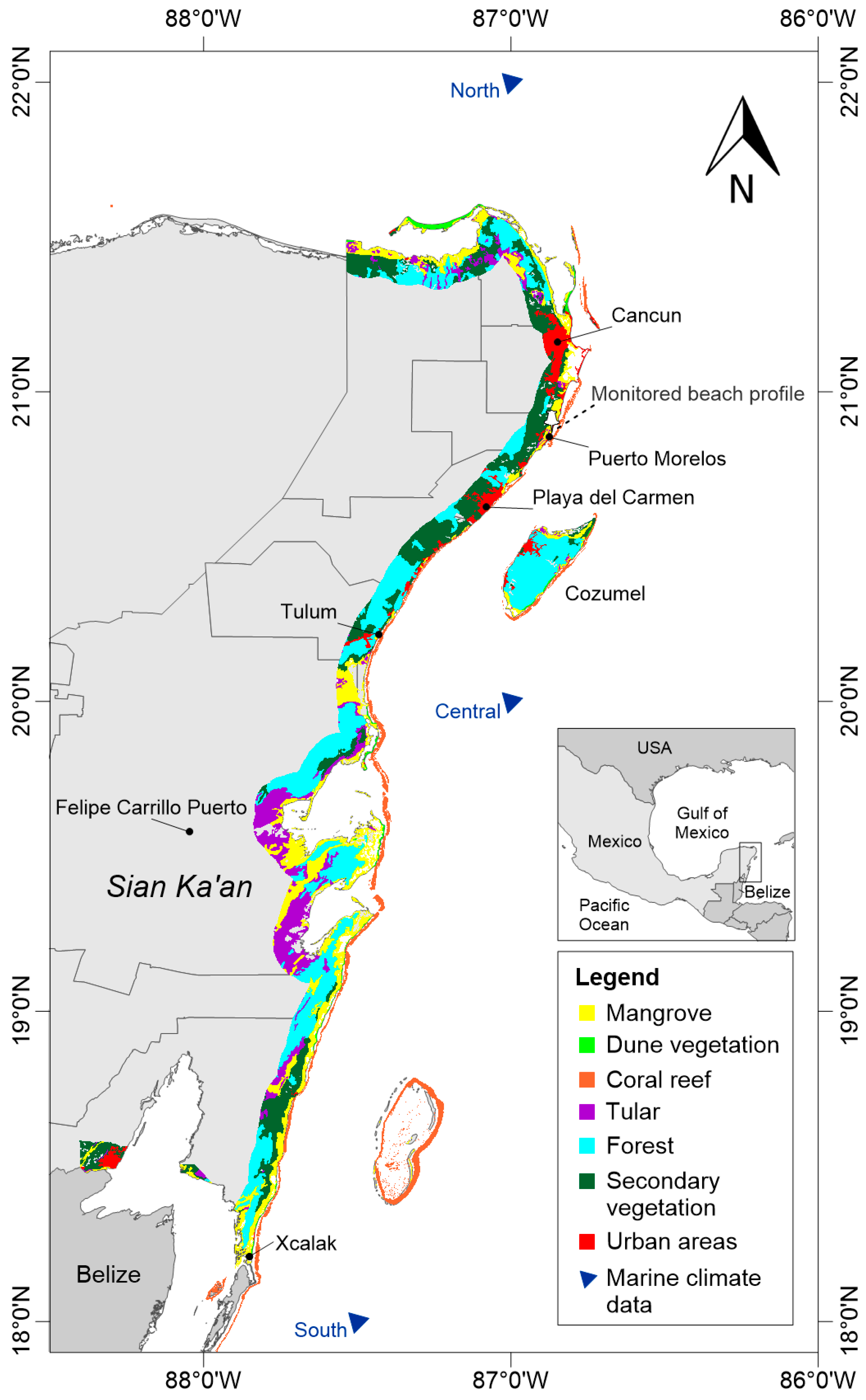
2. Study Area
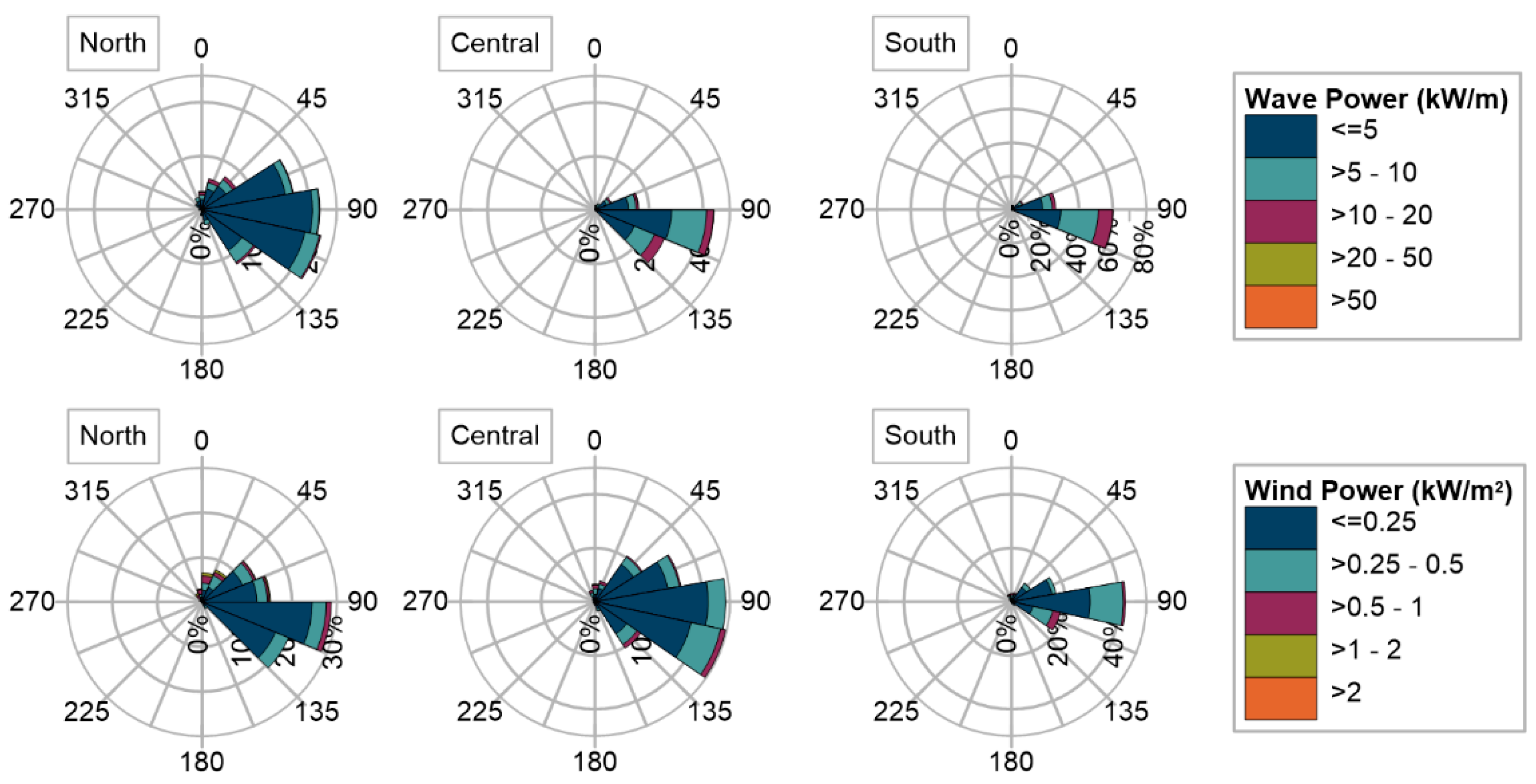
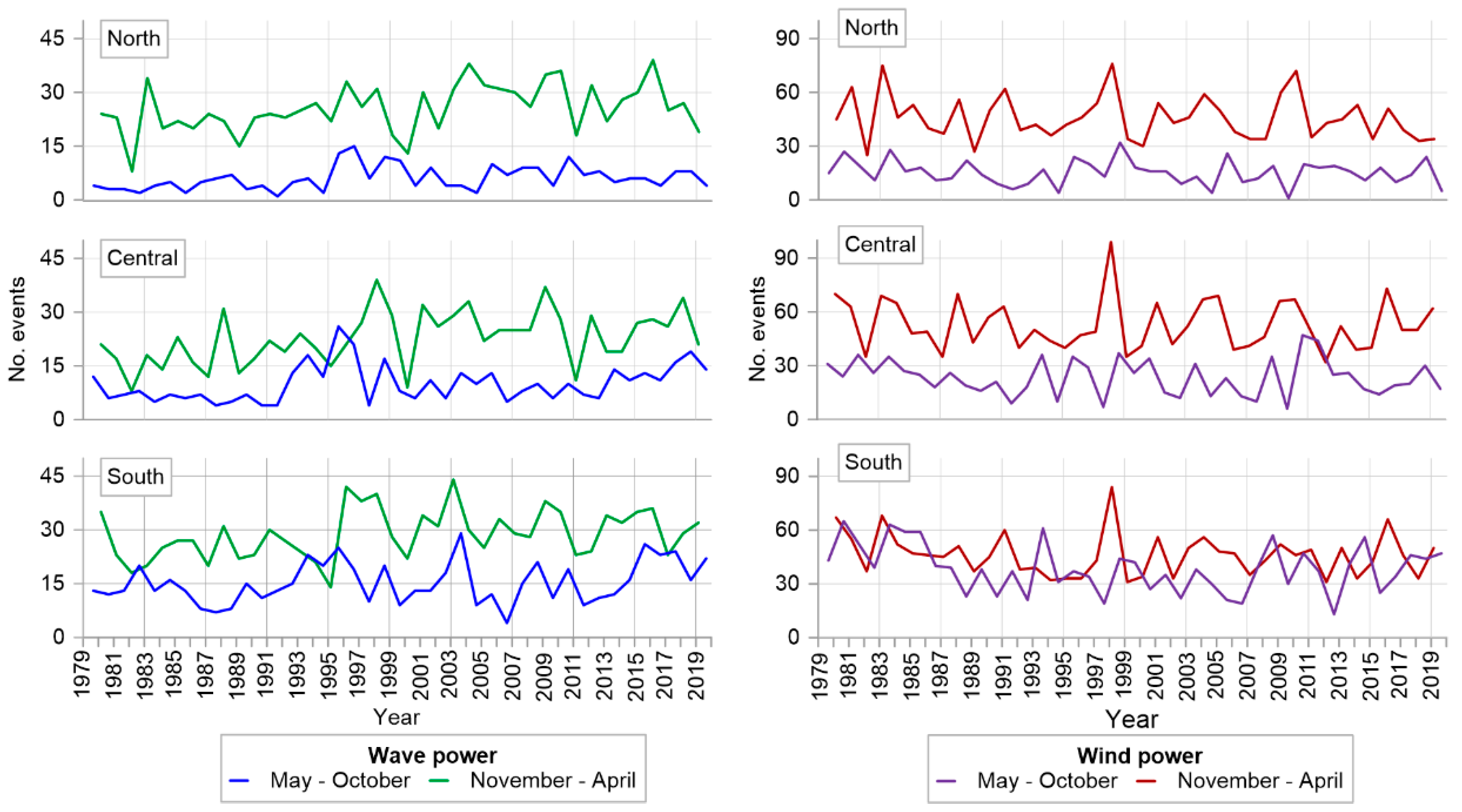
2.1. Marine and Atmospheric Climates
2.2. Geomorphological Features
2.3. Coastal Ecosystems
2.4. Socioeconomic Characteristics
3. Materials and Methods
3.1. Sargasso Dynamics
3.2. Impacts of the Massive Influx of Sargasso
4. Results and Discussion
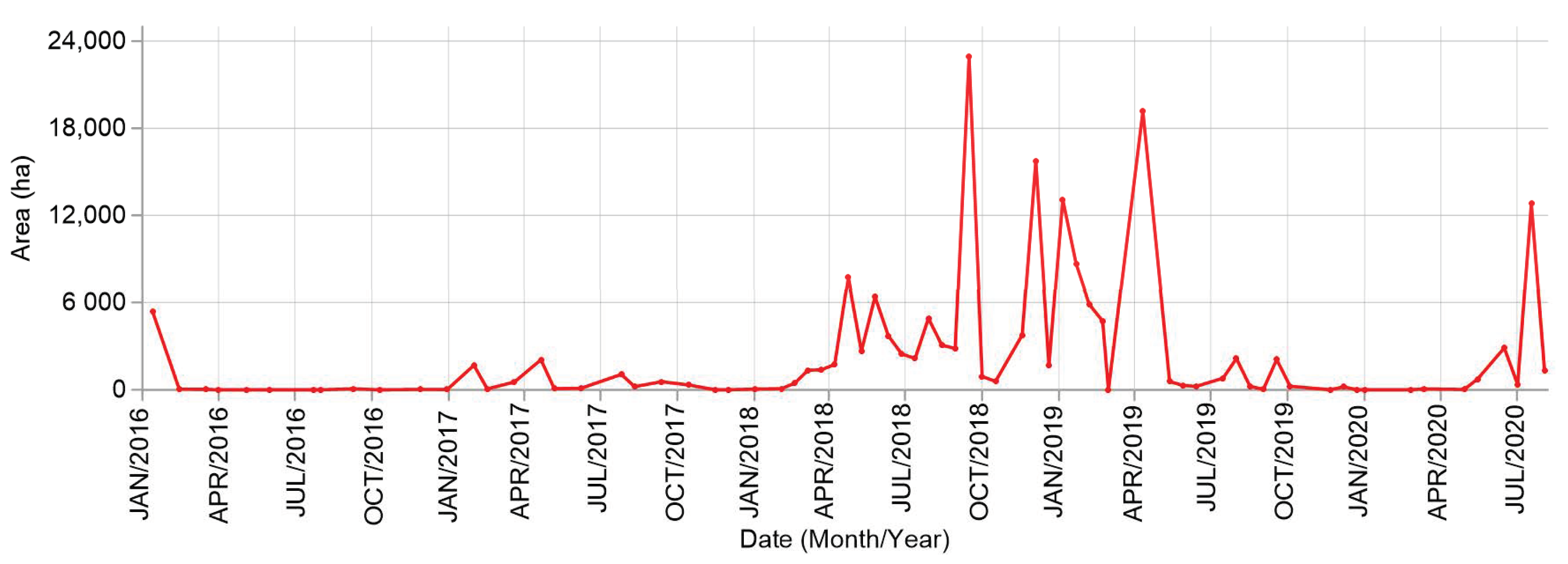
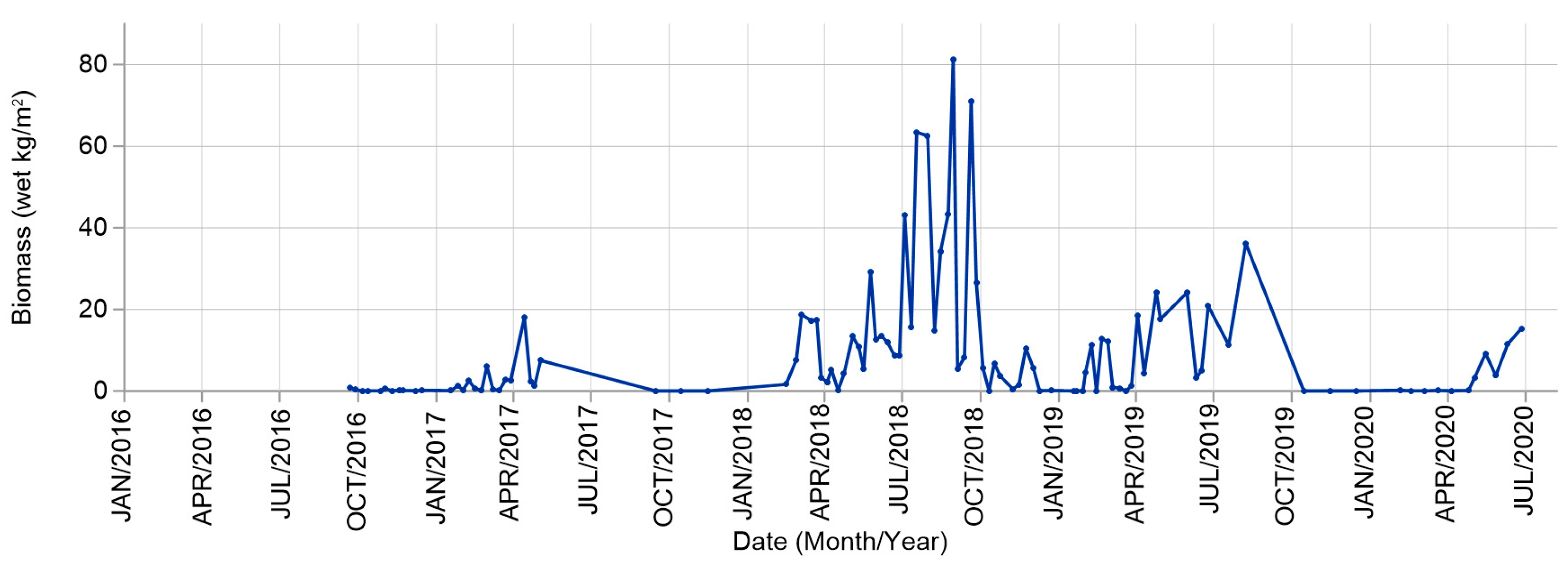
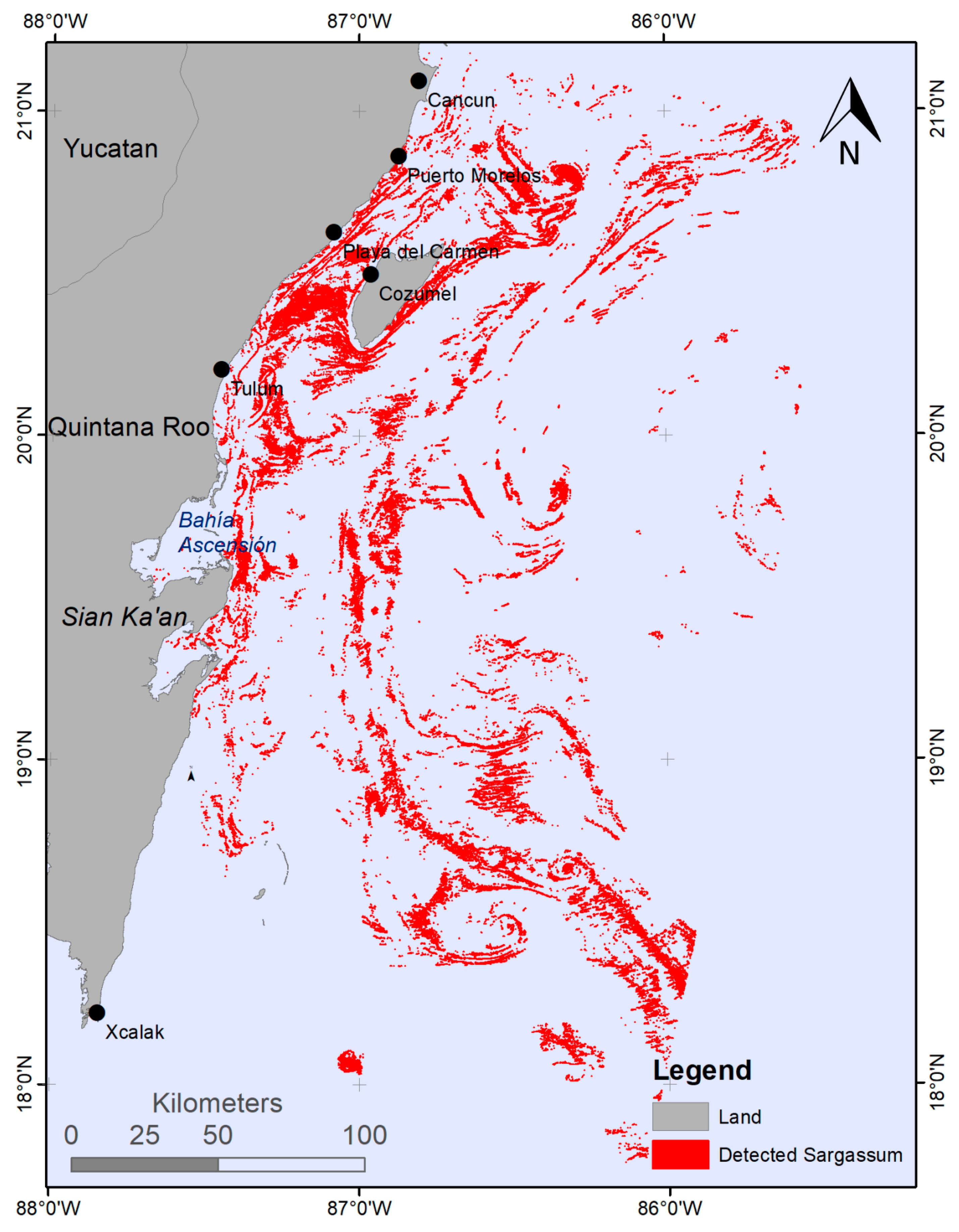
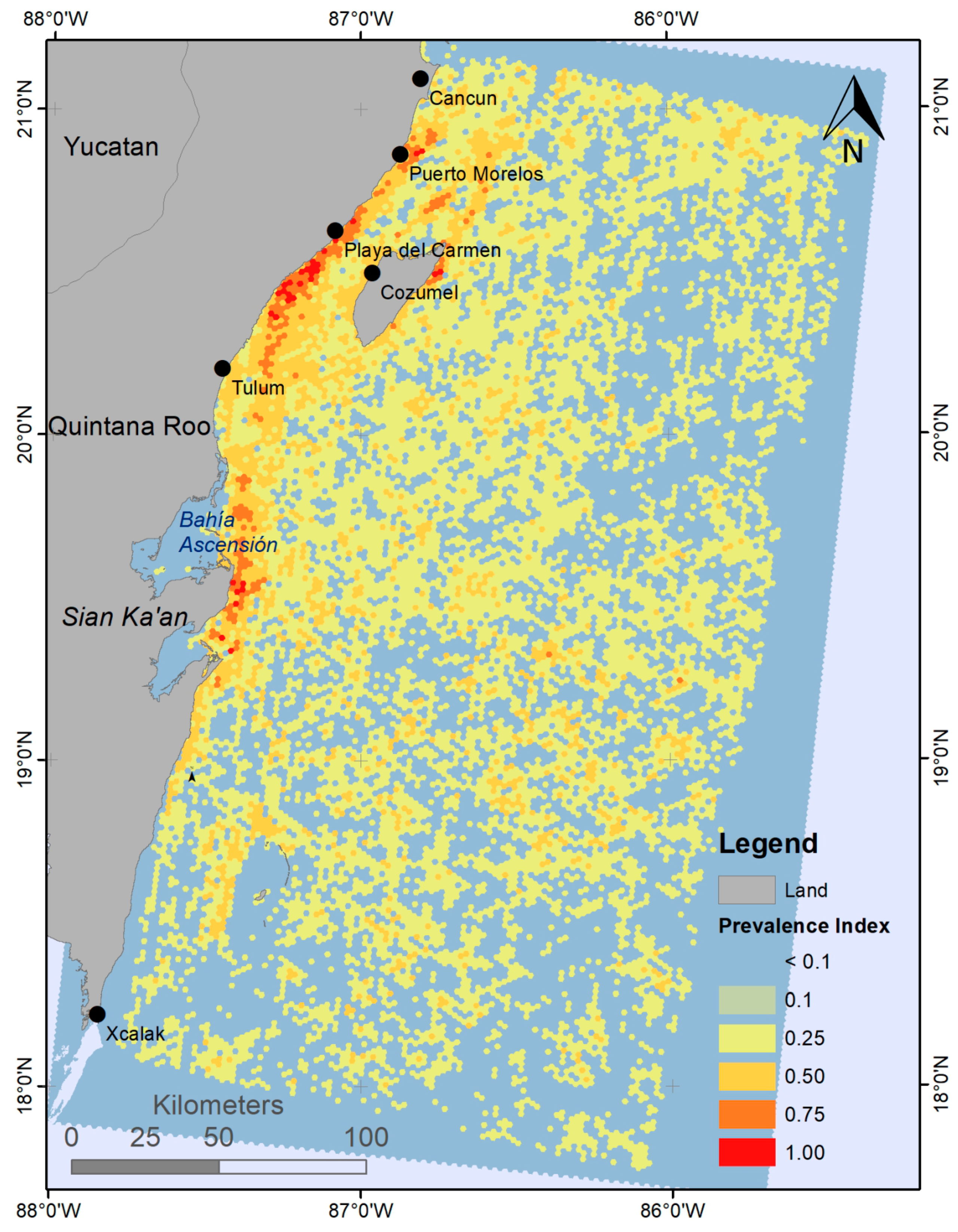
4.1. Sargasso Dynamics
4.2. Impact on Ecosystems
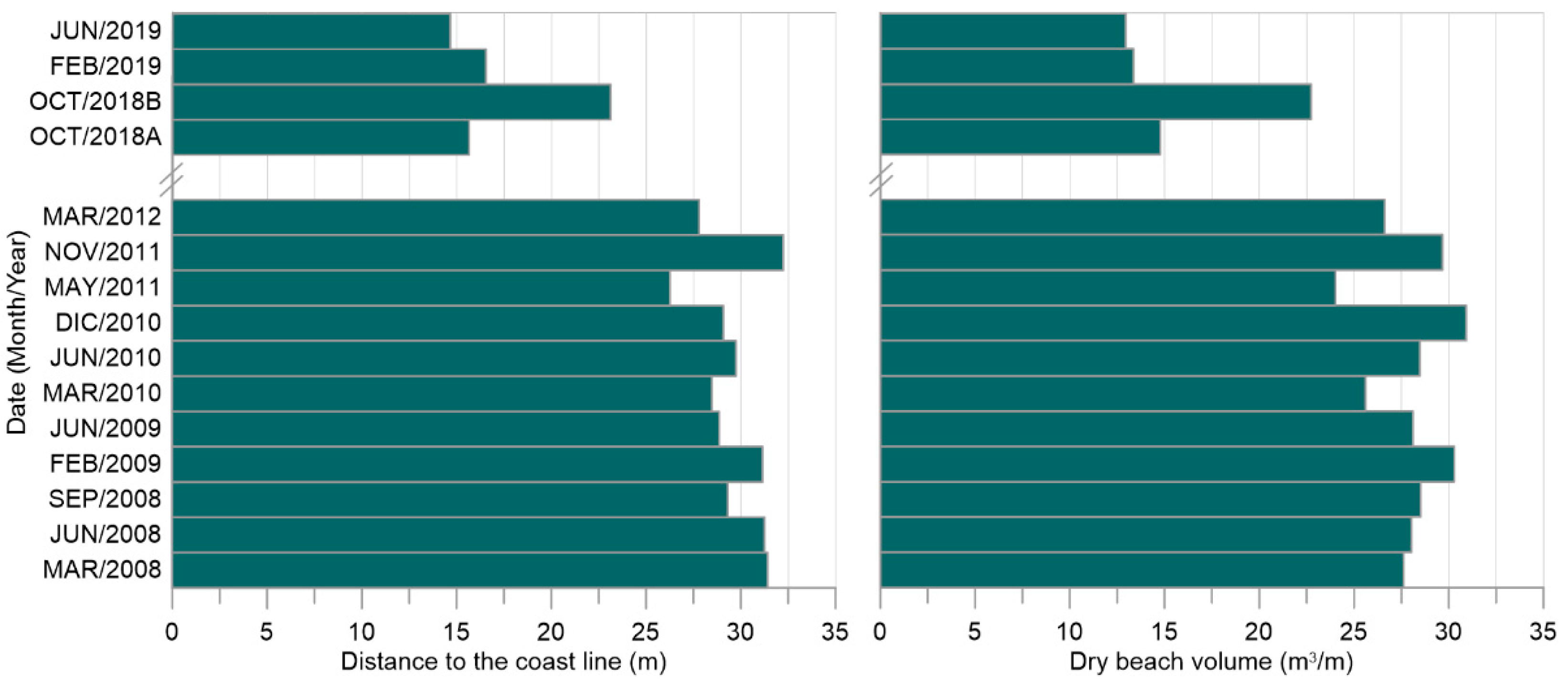
4.2.1. Beaches and Coastal Dunes
4.2.2. Nearshore Waters—Sargasso Brown Tides
4.2.3. Other Ecosystems
4.3. Impacts on Society and on Economic Activities Locally
4.3.1. Socioeconomic Impacts
4.3.2. Beach Removal and Off-Shore Collection of Sargasso
4.3.3. Uses and Valorization of Sargasso
- Fertilizer (clean and dry sargasso) and compost (contained with equal amounts of water).
- Sodium alginate for food, textile and pharmaceutical products.
- Construction blocks (40% sargasso).
- Manually produced paper.
- Beauty care products.
- Crop and livestock production.
- Bioplastics.
Biogas
Biosorption Potential of Sargasso Biomass
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gower, J.; Young, E.; King, S. Satellite images suggest a new Sargassum source region in 2011. Remote Sens. Lett. 2013, 4, 764–773. [Google Scholar] [CrossRef]
- Smetacek, V.; Zingone, A. Green and golden seaweed tides on the rise. Nature 2013, 504, 84–88. [Google Scholar] [CrossRef]
- Wang, M.; Hu, C. Predicting Sargassum blooms in the Caribbean Sea from MODIS observations. Geophys. Res. Lett. 2017, 44, 3265–3273. [Google Scholar] [CrossRef]
- Wang, M.; Hu, C.; Barnes, B.B.; Mitchum, G.; Lapointe, B.; Montoya, J.P. The great Atlantic Sargassum belt. Science 2019, 365, 83. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, R.E.; van Tussenbroek, B.; Jordán-Dahlgren, E. Afluencia masiva de sargazo pelágico a la costa del Caribe mexicano (2014–2015). In Florecimientos Algales Nocivos en México; García-Mendoza, E., Quijano-Scheggia, S.I., Olivos-Ortiz, A., Núñez-Vázquez, E.J., Eds.; CICESE: Ensenada, BC, Mexico, 2016; pp. 352–365. [Google Scholar]
- Silva, R.; Mendoza, E.; Mariño-Tapia, I.; Martínez, M.L.; Escalante, E. An artificial reef improves coastal protection and provides a base for coral recovery. J. Coast. Res. 2016, 467–471. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, R.E.; Medina-Valmaseda, A.E.; Blanchon, P.; Monroy-Velázquez, L.V.; Almazán-Becerril, A.; Delgado-Pech, B.; Vásquez-Yeomans, L.; Francisco, V.; García-Rivas, M.C. Faunal mortality associated with massive beaching and decomposition of pelagic Sargassum. Mar. Pollut. Bull. 2019, 146, 201–205. [Google Scholar] [CrossRef]
- García-Sánchez, M.; Graham, C.; Vera, E.; Escalante-Mancera, E.; Álvarez-Filip, L.; van Tussenbroek, B.I. Temporal changes in the composition and biomass of beached pelagic Sargassum species in the Mexican Caribbean. Aquat. Bot. 2020, 167, 103275. [Google Scholar] [CrossRef]
- Uribe-Martínez, A.; Guzmán-Ramírez, A.; Arreguín-Sánchez, F.; Cuevas, E. El sargazo en el Caribe mexicano, revisión de una historia impensable. In Gobernanza y Manejo de las Costas y Mares ante la Incertidumbre; Rivera-Arriaga, E., Azuz-Adeath, I., Cervantes Rosas, O.D., Espinoza-Tenorio, A., Silva Casarín, R., Ortega-Rubio, A., et al., Eds.; Universidad Autónoma de Campeche, RICOMAR: Campeche, Mexico, 2020; pp. 743–768. [Google Scholar]
- Johnson, D.R.; Ko, D.S.; Franks, J.S.; Moreno, P.; Sanchez-Rubio, G. The Sargassum invasion of the Eastern Caribbean and dynamics of the Equatorial North Atlantic. In Proceedings of the 65th Gulf and Caribbean Fisheries Institute, Santa Marta, Colombia, 5–9 November 2012; pp. 102–103. [Google Scholar]
- Sissini, M.N.; de Barros Barreto, M.B.B.; Széchy, M.T.M.; de Lucena, M.B.; Oliveira, M.C.; Gower, J.; Liu, G.; de Oliveira Bastos, E.; Milstein, D.; Gusmão, F.; et al. The floating Sargassum (Phaeophyceae) of the South Atlantic Ocean—Likely scenarios. Phycologia 2017, 56, 321–328. [Google Scholar] [CrossRef]
- Lapointe, B.E.; West, L.E.; Sutton, T.T.; Hu, C. Ryther revisited: Nutrient excretions by fishes enhance productivity of pelagic Sargassum in the western North Atlantic Ocean. J. Exp. Mar. Biol. Ecol. 2014, 458, 46–56. [Google Scholar] [CrossRef]
- Johns, E.M.; Lumpkin, R.; Putman, N.F.; Smith, R.H.; Muller-Karger, F.E.; T. Rueda-Roa, D.; Hu, C.; Wang, M.; Brooks, M.T.; Gramer, L.J.; et al. The establishment of a pelagic Sargassum population in the tropical Atlantic: Biological consequences of a basin-scale long distance dispersal event. Prog. Oceanogr. 2020, 182, 102269. [Google Scholar] [CrossRef]
- Van Tussenbroek, B.I.; Hernández Arana, H.A.; Rodríguez-Martínez, R.E.; Espinoza-Avalos, J.; Canizales-Flores, H.M.; González-Godoy, C.E.; Barba-Santos, M.G.; Vega-Zepeda, A.; Collado-Vides, L. Severe impacts of brown tides caused by Sargassum spp. on near-shore Caribbean seagrass communities. Mar. Pollut. Bull. 2017, 122, 272–281. [Google Scholar] [CrossRef]
- Maurer, A.; Stapleton, S.; Layman, C. Impacts of the Caribbean Sargassum influx on sea turtle nesting. In Proceedings of the 71st Gulf and Caribbean Fisheries Institute, San Andres, Colombia, 5–9 November 2018. [Google Scholar]
- Rioja-Nieto, R.; Garza-Pérez, R.; Álvarez-Filip, L.; Mariño-Tapia, I.; Enríquez, C. The Mexican Caribbean: From Xcalak to Holbox. In World Seas: An Environmental Evaluation, 2nd ed.; Sheppard, C., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 637–653. [Google Scholar] [CrossRef]
- Escudero, M.; Silva, R.; Mendoza, E. Beach erosion driven by natural and numan activity at Isla del Carmen barrier island, Mexico. J. Coast. Res. 2014, 62–74. [Google Scholar] [CrossRef]
- Cerdeira-Estrada, S.; Heege, T.; Kolb, M.; Ohlendorf, S.; Uribe, A.; Müller, A.; Garza, R.; Ressl, R.; Aguirre, R.; Mariño, I.; et al. Benthic habitat and bathymetry mapping of shallow waters in Puerto morelos reefs using remote sensing with a physics based data processing. In Proceedings of the International Geoscience and Remote Sensing Symposium, Munich, Germany, 22–27 July 2012; pp. 4383–4386. [Google Scholar]
- UNEP-WCMC, UN Environment World Conservation Monitoring Centre. Global Distribution of Coral Reefs. Available online: http://data.unepwcmc.org/datasets/1 (accessed on 4 June 2018).
- Ruíz-Rentería, F.; van Tussenbroek, B.I.; Jordán-Dahlgren, E. Puerto Morelos, Quintana Roo, Mexico; Special Report; UNESCO: Paris, France, 1998; pp. 57–66. [Google Scholar]
- Rodríguez-Martínez, R.E.; Ruíz-Rentería, F.; Tussenbroek, B.v.; Barba-Santos, G.; Escalante-Mancera, E.; Jordán-Garza, G.; Jordán-Dahlgren, E. Environmental state and tendencies of the Puerto Morelos CARICOMP site, Mexico. Rev. Biol. Trop. 2010, 58, 23–43. [Google Scholar]
- Climate Reanalysis. Available online: https://climate.copernicus.eu/climate-reanalysis (accessed on 5 June 2020).
- CONAGUA. Programa Hidráulico Regional 2002–2006, Región XII Península de Yucatán; SEMARNAT: Mexico City, Mexico, 2003.
- Null, K.A.; Knee, K.L.; Crook, E.D.; de Sieyes, N.R.; Rebolledo-Vieyra, M.; Hernández-Terrones, L.; Paytan, A. Composition and fluxes of submarine groundwater along the Caribbean coast of the Yucatan Peninsula. Cont. Shelf Res. 2014, 77, 38–50. [Google Scholar] [CrossRef]
- Hernández-Terrones, L.M.; Null, K.A.; Ortega-Camacho, D.; Paytan, A. Water quality assessment in the Mexican Caribbean: Impacts on the coastal ecosystem. Cont. Shelf Res. 2015, 102, 62–72. [Google Scholar] [CrossRef]
- Van Tussenbroek, B.I. Dynamics of seagrasses and associated algae in coral reef lagoons. Hidrobiológica 2011, 21, 293–310. [Google Scholar]
- Jordán-Dahlgren, E.; Rodríguez-Martínez, R.E. The Atlantic coral reefs of Mexico. In Latin American Coral Reefs; Cortés, J., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2003; pp. 131–158. [Google Scholar]
- Rioja-Nieto, R.; Álvarez-Filip, L. Coral reef systems of the Mexican Caribbean: Status, recent trends and conservation. Mar. Pollut. Bull. 2019, 140, 616–625. [Google Scholar] [CrossRef]
- Harvell, D.; Jordán-Dahlgren, E.; Merkel, S.; Rosenberg, E.; Raymundo, L.; Smith, G.; Weil, E.; Willis, B. Coral disease, environmental drivers, and the balance between coral and microbial associates. Oceanography 2007, 20, 172–195. [Google Scholar] [CrossRef]
- Contreras-Silva, A.I.; Tilstra, A.; Migani, V.; Thiel, A.; Pérez-Cervantes, E.; Estrada-Saldívar, N.; Elias-Ilosvay, X.; Mott, C.; Alvarez-Filip, L.; Wild, C. A meta-analysis to assess long-term spatiotemporal changes of benthic coral and macroalgae cover in the Mexican Caribbean. Sci. Rep. 2020, 10, 8897. [Google Scholar] [CrossRef]
- Estrada-Saldívar, N.; Jordán-Dalhgren, E.; Rodríguez-Martínez, R.E.; Perry, C.; Alvarez-Filip, L. Functional consequences of the long-term decline of reef-building corals in the Caribbean: Evidence of across-reef functional convergence. R. Soc. Open Sci. 2019, 6, 190298. [Google Scholar] [CrossRef]
- Van Tussenbroek, B.I.; Cortés, J.; Collin, R.; Fonseca, A.C.; Gayle, P.M.H.; Guzmán, H.M.; Jácome, G.E.; Juman, R.; Koltes, K.H.; Oxenford, H.A.; et al. Caribbean-wide, long-term study of seagrass beds reveals local variations, shifts in community structure and occasional collapse. PLoS ONE 2014, 9, e90600. [Google Scholar] [CrossRef]
- Cruz-Palacios, V.; van Tussenbroek, B.I. Simulation of hurricane-like disturbances on a Caribbean seagrass bed. J. Exp. Mar. Biol. Ecol. 2005, 324, 44–60. [Google Scholar] [CrossRef]
- Van Tussenbroek, B.I.; van Dijk, J.K. Spatial and temporal variability in biomass and production of psammophytic Halimeda incrassata (bryopsidales, chlorophyta) in a Caribbean reef lagoon. J. Phycol. 2007, 43, 69–77. [Google Scholar] [CrossRef]
- James, R.K.; Silva, R.; van Tussenbroek, B.I.; Escudero-Castillo, M.; Mariño-Tapia, I.; Dijkstra, H.A.; van Westen, R.M.; Pietrzak, J.D.; Candy, A.S.; Katsman, C.A.; et al. Maintaining Tropical Beaches with Seagrass and Algae: A Promising Alternative to Engineering Solutions. BioScience 2019, 69, 136–142. [Google Scholar] [CrossRef]
- López-Mendoza, P.G.; Ruiz-Fernández, A.C.; Sanchez-Cabeza, J.A.; van Tussenbroek, B.I.; Cuellar-Martinez, T.; Pérez-Bernal, L.H. Temporal trends of organic carbon accumulation in seagrass meadows from the northern Mexican Caribbean. CATENA 2020, 194, 104645. [Google Scholar] [CrossRef]
- Moreno-Casasola, P. Beaches and Dunes of the Gulf of Mexico: A view of the current situation. In Environmental analysis of the Gulf of Mexico; Caso, M., Pisanty, I., Exequiel Ezcurra, E., Eds.; Harte Research Institute of the Gulf of Mexico: Houston, TX, USA, 2007; pp. 302–313. [Google Scholar]
- Silva-Casarin, R.; Mariño-Tapia, I.; Enriquez-Ortiz, C.; Mendoza-Baldwin, E.; Escalante-Mancera, E.; Ruiz-Rentería, F. Monitoring shoreline changes at Cancun beach, Mexico: Effects of hurricane Wilma. In Proceedings of the 30th International Conference on Coastal Engineering, San Diego, CA, USA, 3–8 September 2006; pp. 3491–3503. [Google Scholar]
- De Alegria-Arzaburu, A.R.; Mariño-Tapia, I.; Enriquez, C.; Silva, R.; González-Leija, M. The role of fringing coral reefs on beach morphodynamics. Geomorphology 2013, 198, 69–83. [Google Scholar] [CrossRef]
- Elizondo, N.C.; Islebe, G.A.; Infante Mata, D.; Valdéz, M.; Weissenberger, H.; García, J.R.; Troche Souza, C.H.; Camacho Sandoval, T.; Weissenberger, H.; López, M. Diagnóstico Sobre el Estado Actual del Ecosistema de Duna Costera en la Costa Norte, Centro y Sur de la Reserva de la Biosfera Sian Ka’an; CONANP: Felipe Carrillo Puerto, QR, Mexico, 2016; p. 254.
- Moreno-Casasola, P.; Castillo, S. 19—Dune ecology on the eastern coast of Mexico. In Coastal Plant Communities of Latin America; Seeliger, U., Ed.; Academic Press: San Diego, CA, USA, 1992; pp. 309–321. [Google Scholar]
- Feller, I.C. Preliminary Survey of the Mangrove Ecosystem at Puerto Morelos, Quintana Roo, Mexico; Smithsonian Environmental Research Center: Edgewater, MD, USA, 1997; p. 10. [Google Scholar]
- Flores, J.S.; Espejel, I. Tipos de vegetación de la Península de Yucatán. Etnoflora Yucatanense. In Etnoflora Yucatanense; Universidad Autónoma de Yucatán: Yucatán, México, 1994. [Google Scholar]
- Pérez-Salicrup, D. Forest types and their implications. In Integrated Land-Change Science and Tropical Deforestation in the Southern Yucatan: Final Frontiers; Turner, B.L., II, Geoghegan, J., Foster, D., Eds.; Oxford University Press: Oxford, MI, USA, 2004; pp. 63–80. [Google Scholar]
- Hernandez-Stefanoni, J.L.; Pineda, J.B.; Valdes-Valadez, G. Comparing the use of indigenous knowledge with classification and ordination techniques for assessing the species composition and structure of vegetation in a tropical forest. Environ. Manag. 2006, 37, 686–702. [Google Scholar] [CrossRef]
- Gutiérrez-Granados, G.; Pérez-Salicrup, D.R.; Dirzo, R. Differential diameter-size effects of forest management on tree species richness and community structure: Implications for conservation. Biodivers. Conserv. 2011, 20, 1571–1585. [Google Scholar] [CrossRef]
- Escudero-Castillo, M.; Felix-Delgado, A.; Silva, R.; Mariño-Tapia, I.; Mendoza, E. Beach erosion and loss of protection environmental services in Cancun, Mexico. Ocean Coastal Manag. 2018, 156, 183–197. [Google Scholar] [CrossRef]
- INEGI. Encuesta Intercensal. 2015. Available online: https://www.inegi.org.mx/programas/intercensal/2015/ (accessed on 4 June 2020).
- DATATUR. Quintana Roo. Available online: https://www.datatur.sectur.gob.mx/ITxEF/ITxEF_QROO.aspx (accessed on 4 June 2020).
- OECD. Tourism Trends and Policies 2020; OECD: Paris, France, 2020. Available online: https://doi.org/10.1787/6b47b985-en (accessed on 5 August 2020).
- Cruz, C.J.; Mendoza, E.; Silva, R.; Chávez, V. Assessing degrees of anthropization on the coast of Mexico from ecosystem conservation and population growth data. J. Coast. Res. 2019, 92, 136–144. [Google Scholar] [CrossRef]
- Hu, C. A novel ocean color index to detect floating algae in the global oceans. Remote Sens. Environ. 2009, 113, 2118–2129. [Google Scholar] [CrossRef]
- Wang, M.; Hu, C. Mapping and quantifying Sargassum distribution and coverage in the Central West Atlantic using MODIS observations. Remote Sens. Environ. 2016, 183, 350–367. [Google Scholar] [CrossRef]
- Cuevas, E.; Uribe-Martínez, A.; Liceaga-Correa, M.D.L.Á. A satellite remote-sensing multi-index approach to discriminate pelagic Sargassum in the waters of the Yucatan Peninsula, Mexico. Int. J. Remote. Sens. 2018, 39, 3608–3627. [Google Scholar] [CrossRef]
- USGS Earth Explorer. Available online: https://earthexplorer.usgs.gov/ (accessed on 5 August 2020).
- Prat, H. Remarques sur la faune et la flore associees aux Sargasses flottantes. Nat. Can. 1935, 62, 120–129. [Google Scholar]
- Fine, M.L. Faunal variation on pelagic Sargassum. Mar. Biol. 1970, 7, 112–122. [Google Scholar] [CrossRef]
- Coston-Clements, L.; Settle, L.R.; Hoss, D.E.; Cross, F.A. Utilization of the Sargassum Habitat by Marine Invertebrates and Vertebrates—A Review; NOAA Technical Memorandum NMFA-SEFSC-296; US Department of Commerce, National Oceanic and Atmospheric Administration: Washington, DC, USA, 1991; p. 32.
- Butler, J.N.; Morris, B.F.; Cadwallader, J.; Stoner, A.W. Studies of Sargassum and the Sargassum Community; Bermuda Biological Station for Research: St. Georges, Bermuda, UK, 1983; Volume 22. [Google Scholar]
- Poore, A.G.B.; Gallagher, K.M. Strong consequences of diet choice in a talitrid amphipod consuming seagrass and algal wrack. Hydrobiologia 2013, 701, 117–127. [Google Scholar] [CrossRef]
- INECC. Lineamientos Técnicos y de Gestión Para la Atención de la Contingencia Ocasionada Por Sargazo en el Caribe Mexicano y el Golfo de Mexico; Instituto Nacional de Ecología y Cambio Climático: Mexico City, Mexico; Unpublished work; 2019.
- Williams, A.; Feagin, R. Sargassum as a natural solution to enhance dune plant growth. Environ. Manag. 2010, 46, 738–747. [Google Scholar] [CrossRef]
- Sigren, J.M.; Figlus, J.; Armitage, A.R. Coastal sand dunes and dune vegetation: Restoration, erosion, and storm protection. Shore Beach 2014, 82, 5–12. [Google Scholar]
- Figlus, J.; Sigren, J.; Webster, R.; Linton, T. Innovative Technology Seaweed Prototype: Dunes Demonstration Project; Texas A & M University: Galveston, TX, USA, 2015. [Google Scholar]
- Eckert, K.L.; Eckert, E. An Atlas of Sea Turtle Nesting Habitat for the Wider Caribbean Region, Revised ed.; WIDECAST Technical Report 19; Wider Caribbean Sea Turtle Conservation Network: Godfrey, IL, USA, 2019; p. 232. [Google Scholar]
- Piniak, W.E.D.; Eckert, K.L. Sea turtle nesting habitat in the Wider Caribbean Region. Endanger Species Res. 2011, 15, 129–141. [Google Scholar] [CrossRef][Green Version]
- Tzeek-Tuz, M.; Herrera-Pavón, R.; Quintana-Pali, G.P.; Barragán-Zepeda, A.; Gómez-Nieto, L. Programa de conservación de tortugas marinas Riviera Maya-Tulum: Resultado de más de tres décadas de protección. In El Uso del Conocimiento de las Tortugas Marinas Como Herramienta Para la Restauración de Sus Poblaciones y Hábitats Asociados; Cuevas Flores, E.A., Guzmán Hernández, V., Guerra Santos, J.J., Rivas Hernández, G.A., Eds.; Universidad Autónoma del Carmen: Campeche, Mexico, 2020. [Google Scholar]
- Santos, K.C.; Livesey, M.; Fish, M.; Lorences, A.C. Climate change implications for the nest site selection process and subsequent hatching success of a green turtle population. Mitig. Adapt. Strateg. Glob. Change 2017, 22, 121–135. [Google Scholar] [CrossRef]
- CONANP. Informe de la Operación del Campamento Tortuguero “Parque Nacional Arrecife de Puerto Morelos” Temporada 2015; Comisión Nacional de Áreas Naturales Protegidas: Felipe Carrillo Puerto, QR, Mexico, 2015; p. 27.
- Gavio, B.; Santos-Martínez, A. Floating Sargassum in Serranilla Bank, Caribbean Colombia, may jeopardize the race to the ocean of baby sea turtles. Acta Biol. Col. 2018, 23, 311–314. [Google Scholar] [CrossRef]
- Azanza-Ricardo, J.; Pérez-Martín, R. Impacto de la acumulación de sargazo del verano del 2015 sobre las tortugas marinas de playa de La Barca, península de Guanahacabibes. Rev. Invest. Mar. 2016, 36, 54–62. [Google Scholar]
- Davenport, J.; Davenport, J.L. The impact of tourism and personal leisure transport on coastal environments: A review. Estuar. Coast. Shelf Sci. 2006, 67, 280–292. [Google Scholar] [CrossRef]
- Gray, J.S.; Wu, R.S.-S.; Or, Y.Y. Effects of hypoxia and organic enrichment on the coastal marine environment. Mar. Ecol. Prog. Ser. 2002, 238, 249–279. [Google Scholar] [CrossRef]
- Truebano, M.; Tills, O.; Collins, M.; Clarke, C.; Shipsides, E.; Wheatley, C.; Spicer, J.I. Short-term acclimation in adults does not predict offspring acclimation potential to hypoxia. Sci. Rep. 2018, 8, 3174. [Google Scholar] [CrossRef]
- Alvarez-Filip, L.; Estrada-Saldívar, N.; Pérez-Cervantes, E.; Molina-Hernández, A.; González-Barrios, F.J. A rapid spread of the stony coral tissue loss disease outbreak in the Mexican Caribbean. PeerJ 2019, 7, e8069. [Google Scholar] [CrossRef]
- Dooley, J.K. Fishes associated with the pelagic Sargassum complex, with a discussion of the Sargassum community. Contrib. Mar. Sci. 1972, 16, 1–32. [Google Scholar]
- Thiel, M.; Gutow, L. The Ecology of Rafting in the Marine Environment. II: The Rafting Organisms and Community; Gibson, R., Atkinson, R., Gordon, J., Eds.; Taylor & Francis: Philadelphia, PA, USA, 2005; Volume 43, pp. 279–418. [Google Scholar]
- Trott, T.M.; Mckenna, S.A.; Pitt, J.M.; Hemphill, A.; Ming, F.W.; Rouja, P.; Gjerde, K.M.; Causey, B.; Earle, S.A. Efforts to enhance protection of the Sargasso Sea. In Proceedings of the 63rd Gulf and Caribbean Fisheries Institute, San Juan, Puerto Rico, 1–5 November 2010; pp. 282–288. [Google Scholar]
- Witherington, B.; Hirama, S.; Hardy, R. Young sea turtles of the pelagic Sargassum-dominated drift community: Habitat use, population density, and threats. Mar. Ecol. Prog. Ser. 2012, 463, 1–22. [Google Scholar] [CrossRef]
- Hardy, R.F.; Hu, C.; Witherington, B.; Lapointe, B.; Meylan, A.; Peebles, E.; Meirose, L.; Hirama, S. Characterizing a sea turtle developmental habitat using Landsat observations of surface-pelagic drift communities in the eastern Gulf of Mexico. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2018, 11, 3646–3659. [Google Scholar] [CrossRef]
- Lopez, P.J.; Hervé, V.; Lambourdière, J.; René-Trouillefou, M.; Devault, D. From the sea to the land: Dynamic of the Sargassum tide holobiont in the Caribbean islands. Res. Square 2020. [Google Scholar] [CrossRef]
- Monroy-Velázquez, L.V.; Rodríguez-Martínez, R.E.; van Tussenbroek, B.I.; Aguiar, T.; Solís-Weiss, V.; Briones-Fourzán, P. Motile macrofauna associated with pelagic Sargassum in a Mexican reef lagoon. J. Environ. Manag. 2019, 252, 109650. [Google Scholar] [CrossRef]
- Monroy-Velázquez, L.V. Fauna móvil asociada a Sargassum spp. en el Parque Nacional Arrecife de Puerto Morelos. Unpublished work. 2020. [Google Scholar]
- Cabanillas-Terán, N.; Hernández-Arana, H.A.; Ruiz-Zárate, M.-Á.; Vega-Zepeda, A.; Sanchez-Gonzalez, A. Sargassum blooms in the Caribbean alter the trophic structure of the sea urchin Diadema antillarum. PeerJ 2019, 7, e7589. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Martínez, R.E.; Roy, P.D.; Torrescano-Valle, N.; Cabanillas-Terán, N.; Carrillo-Domínguez, S.; Collado-Vides, L.; García-Sánchez, M.; van Tussenbroek, B.I. Element concentrations in pelagic Sargassum along the Mexican Caribbean coast in 2018–2019. PeerJ 2020, 8, e8667. [Google Scholar] [CrossRef]
- Carruthers, T.J.B.; van Tussenbroek, B.I.; Dennison, W.C. Influence of submarine springs and wastewater on nutrient dynamics of Caribbean seagrass meadows. Estuar. Coast. Shelf Sci. 2005, 64, 191–199. [Google Scholar] [CrossRef]
- Metcalfe, C.D.; Beddows, P.A.; Bouchot, G.G.; Metcalfe, T.L.; Li, H.; Van Lavieren, H. Contaminants in the coastal karst aquifer system along the Caribbean coast of the Yucatan Peninsula, Mexico. Environ. Pollut. 2011, 159, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Vinagre, F.; Zamacona, A.; Casseb, T. Sargassum: Another Bump in Tourism; Credit Suisse: Mexico City, Mexico, 2019; p. 12. [Google Scholar]
- Espinosa, L.A.; Ng, J.J.L. El Riesgo del Sargazo para la Economía y Turismo de Quintana Roo y México; BBVA Research: Mexico City, Mexico, 2020; p. 35. [Google Scholar]
- Salter, M.A.; Rodríguez-Martínez, R.E.; Álvarez-Filip, L.; Jordán-Dahlgren, E.; Perry, C.T. Pelagic Sargassum as an emerging vector of high rate carbonate sediment import to tropical Atlantic coastlines. Glob. Planet. Change 2020, 103332. [Google Scholar] [CrossRef]
- Chirinas, J.; Ortíz, A.; Jáuregui, D.; Luna, A.D.; Cardeña, L.; May, R.; (Hoteliers and Businessman, Puerto Morelos, Quintana Roo, Mexico). Personal communication, 2020.
- Resiere, D.; Mehdaoui, H.; Névière, R.; Mégarbane, B. Sargassum invasion in the Caribbean: The role of medical and scientific cooperation. Rev. Panam. Salud Publica 2019, 43, e52. [Google Scholar] [CrossRef]
- Kuyucak, N.; Volesky, B. Biosorbents for recovery of metals from industrial solutions. Biotechnol. Lett. 1988, 10, 137–142. [Google Scholar] [CrossRef]
- Davis, T.A.; Volesky, B.; Vieira, R.H.S.F. Sargassum seaweed as biosorbent for heavy metals. Water Res. 2000, 34, 4270–4278. [Google Scholar] [CrossRef]
- Alzate-Gaviria, L.; Domínguez-Maldonad, O.J.; Chablé-Villacís, R.; Leal-Bautista, R.; Canche-Escamilla, R.; Caballero-Vázquez, A.; Hernández-Zepeda, C.F.A.B.-P.; Tapia-Tussell, R.L. Presence of polyphenols complex aromatic lignin in Sargassum spp. from Mexican Caribbean. Environ. Sci. Pollut. Res. 2020, in press. [Google Scholar]
- ADEME. Monitoring and Evaluation of Sargassum Collection Operations; ADEME: Angers, France, 2019. [Google Scholar]
- De Luna, A.; (Manufacturas Industriales Dp, S.A. De C.V., Puerto Morelos, Quitana Roo, Mexico). Personal communication, 2020.
- Gouvêa, L.P.; Assis, J.; Gurgel, C.F.D.; Serrão, E.A.; Silveira, T.C.L.; Santos, R.; Duarte, C.M.; Peres, L.M.C.; Carvalho, V.F.; Batista, M.; et al. Golden carbon of Sargassum forests revealed as an opportunity for climate change mitigation. Sci. Total Environ. 2020, 729, 138745. [Google Scholar] [CrossRef]
- Doyle, E.; Franks, J. Sargassum Fact Sheet; Gulf and Caribbean Fisheries Institute: Fort Pierce, FL, USA, 2015. [Google Scholar]
- CAST Association. Sargassum: A Resource Guide for the Caribbean; CAST Association: Coral Gables, FL, USA, 2015; p. 14. [Google Scholar]
- Desrochers, A.; Cox, S.-A.; Oxenford, H.A.; van Tussenbroek, B. Sargassum Uses Guide: A Resource for Caribbean Researchers, Entrepreneurs and Policy Makers; Centre for Resource Management and Environmental Studies (CERMES), University of the West Indies, Cave Hill Campus: Bridgetown, Barbados, 2020; p. 125. [Google Scholar]
- Milledge, J.J.; Maneein, S.; Arribas López, E.; Bartlett, D. Sargassum inundations in Turks and Caicos: Methane potential and proximate, ultimate, lipid, amino acid, metal and metalloid analyses. Energies 2020, 13, 1523. [Google Scholar] [CrossRef]
- Thompson, T.M.; Young, B.R.; Baroutian, S. Advances in the pretreatment of brown macroalgae for biogas production. Fuel Process. Technol. 2019, 195, 106151. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Kyzas, G.Z. Progress in batch biosorption of heavy metals onto algae. J. Mol. Liq. 2015, 209, 77–86. [Google Scholar] [CrossRef]
- Pappas, C.P.; Randall, S.T.; Sneddon, J. An atomic-emission study of the removal and recovery of chromium from solution by an algal biomass (Chlorella vulgaris). Talanta 1990, 37, 707–710. [Google Scholar] [CrossRef]
- Zeraatkar, A.K.; Ahmadzadeh, H.; Talebi, A.F.; Moheimani, N.R.; McHenry, M.P. Potential use of algae for heavy metal bioremediation, a critical review. J. Environ. Manag. 2016, 181, 817–831. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, A.; Saremnia, B.; Kalantari, M. Removal of mercury (II) from aqueous solutions by biosorption on the biomass of Sargassum glaucescens and Gracilaria corticata. Arabian J. Chem. 2015, 8, 506–511. [Google Scholar] [CrossRef]
- Seolatto, A.A.; Martins, T.D.; Bergamasco, R.; Tavares, C.R.G.; Cossich, E.S.; da Silva, E. Biosorption study of Ni2+ and Cr3+ by Sargassum filipendula: Kinetics and equilibrium. Braz. J. Chem. Eng. 2014, 31, 211–227. [Google Scholar] [CrossRef]
- Da Young Kang, Y.W.J. Sargassum fusiforme extract for heavy metal removal from waste solution. Glob. Chem. Eng. Process Technol. 2018, 5, 2. [Google Scholar]
- Niad, M.; Rasoolzadeh, L.; Zarei, F. Biosorption of copper (II) on Sargassum angostifolium C. Agardh phaeophyceae biomass. Chem. Speciat. Bioavailab. 2014, 26, 176–183. [Google Scholar] [CrossRef]
- Al–Rashdi, N.; Rajamohan, N.; Ramachandran, K.P. Synthesis and application of Sargassum ilicifolium based biomass for the selective removal of phenol. Biocatal. Agric. Biotechnol. 2017, 9, 236–239. [Google Scholar] [CrossRef]
- Hannachi, Y.; Hafidh, A. Biosorption potential of Sargassum muticum algal biomass for methylene blue and lead removal from aqueous medium. Int. J. Environ. Sci. Technol. 2020, 17, 3875–3890. [Google Scholar] [CrossRef]
- Arumugam, S.; Tvn, P.; Krishnan, S. Removal of nickel and copper from aqueous solution by Sargassum sp. to remediates metal aontaminated industrial effluents. Int. J. Appl. Sci. Eng. 2009, 3, 26–30. [Google Scholar] [CrossRef]
- Mahmood, Z.; Zahra, S.; Iqbal, M.; Raza, M.A.; Nasir, S. Comparative study of natural and modified biomass of Sargassum sp. for removal of Cd2+ and Zn2+ from wastewater. Appl. Water Sci. 2017, 7, 3469–3481. [Google Scholar] [CrossRef]
- Ghasemi, F.F.; Dobaradaran, S.; Raeisi, A.; Esmaili, A.; Mohammadi, M.J.; Keshtkar, M.; Nasab, S.G.; Soleimani, F. Data on Fe (II) biosorption onto Sargassum hystrix algae obtained from the Persian Gulf in Bushehr Port, Iran. Data Brief 2016, 9, 823–827. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Teo, T.T.; Balasubramanian, R.; Joshi, U.M. Application of Sargassum biomass to remove heavy metal ions from synthetic multi-metal solutions and urban storm water runoff. J. Hazard. Mater. 2009, 164, 1019–1023. [Google Scholar] [CrossRef]
- Thompson, T.M.; Young, B.R.; Baroutian, S. Pelagic Sargassum for energy and fertiliser production in the Caribbean: A case study on Barbados. Renew. Sust. Energ. Rev. 2020, 118, 109564. [Google Scholar] [CrossRef]
- Coração, A.C.D.S.; Santos, F.S.D.; Duarte, J.A.D.; Lopes-Filho, E.A.P.; De-Paula, J.C.; Rocha, L.M.; Krepsky, N.; Fiaux, S.B.; Teixeira, V.L. What do we know about the utilization of the Sargassum species as biosorbents of trace metals in Brazil? J. Environ. Chem. Eng. 2020, 8, 103941. [Google Scholar] [CrossRef]
- Pohl, P.; Schimmack, W. Adsorption of radionuclides (134Cs, 85Sr, 226Ra, 241Am) by extracted biomasses of cyanobacteria (Nostoc Carneum, N. Insulare, Oscillatoria Geminata and Spirulina Laxis-Sima) and phaeophyceae (Laminaria Digitata and L. Japonica; waste products from alginate production) at different pH. J. Appl. Phycol. 2006, 18, 135–143. [Google Scholar] [CrossRef]
- Mata, Y.N.; Torres, E.; Blázquez, M.L.; Ballester, A.; González, F.; Muñoz, J.A. Gold (III) biosorption and bioreduction with the brown alga Fucus vesiculosus. J. Hazard. Mater. 2009, 166, 612–618. [Google Scholar] [CrossRef]
- Navarro, A.E.; Hernandez-Vega, A.; Masud, M.E.; Roberson, L.M.; Diaz-Vázquez, L.M. Bioremoval of phenol from aqueous solutions using native Caribbean seaweed. Environments 2017, 4, 1. [Google Scholar] [CrossRef]
- Rubín, E.; Rodríguez, P.; Herrero, R.; Sastre de Vicente, M.E. Biosorption of phenolic compounds by the brown alga Sargassum muticum. J. Chem. Technol. Biotechnol. 2006, 81, 1093–1099. [Google Scholar] [CrossRef]
- Putman, N.F.; Lumpkin, R.; Olascoaga, M.J.; Trinanes, J.; Goni, G.J. Improving transport predictions of pelagic Sargassum. J. Exp. Mar. Biol. Ecol. 2020, 529, 151398. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chávez, V.; Uribe-Martínez, A.; Cuevas, E.; Rodríguez-Martínez, R.E.; van Tussenbroek, B.I.; Francisco, V.; Estévez, M.; Celis, L.B.; Monroy-Velázquez, L.V.; Leal-Bautista, R.; et al. Massive Influx of Pelagic Sargassum spp. on the Coasts of the Mexican Caribbean 2014–2020: Challenges and Opportunities. Water 2020, 12, 2908. https://doi.org/10.3390/w12102908
Chávez V, Uribe-Martínez A, Cuevas E, Rodríguez-Martínez RE, van Tussenbroek BI, Francisco V, Estévez M, Celis LB, Monroy-Velázquez LV, Leal-Bautista R, et al. Massive Influx of Pelagic Sargassum spp. on the Coasts of the Mexican Caribbean 2014–2020: Challenges and Opportunities. Water. 2020; 12(10):2908. https://doi.org/10.3390/w12102908
Chicago/Turabian StyleChávez, Valeria, Abigail Uribe-Martínez, Eduardo Cuevas, Rosa E. Rodríguez-Martínez, Brigitta I. van Tussenbroek, Vanessa Francisco, Miriam Estévez, Lourdes B. Celis, L. Verónica Monroy-Velázquez, Rosa Leal-Bautista, and et al. 2020. "Massive Influx of Pelagic Sargassum spp. on the Coasts of the Mexican Caribbean 2014–2020: Challenges and Opportunities" Water 12, no. 10: 2908. https://doi.org/10.3390/w12102908
APA StyleChávez, V., Uribe-Martínez, A., Cuevas, E., Rodríguez-Martínez, R. E., van Tussenbroek, B. I., Francisco, V., Estévez, M., Celis, L. B., Monroy-Velázquez, L. V., Leal-Bautista, R., Álvarez-Filip, L., García-Sánchez, M., Masia, L., & Silva, R. (2020). Massive Influx of Pelagic Sargassum spp. on the Coasts of the Mexican Caribbean 2014–2020: Challenges and Opportunities. Water, 12(10), 2908. https://doi.org/10.3390/w12102908