A Detailed Assessment of Groundwater Quality in the Kabul Basin, Afghanistan, and Suitability for Future Development
Abstract
1. Introduction
2. Materials and Methods
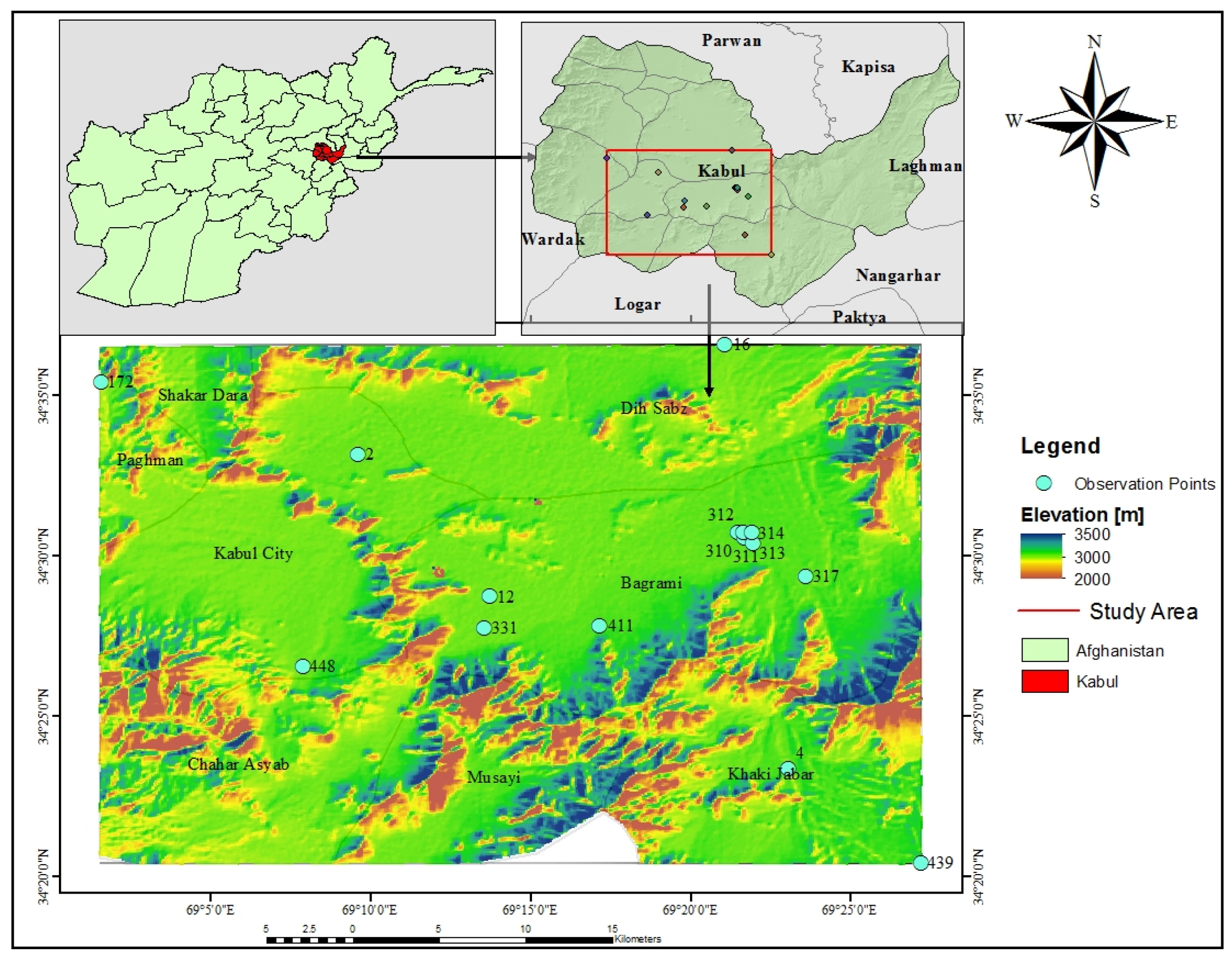
2.1. Study Area Description
2.2. Geological Setting
2.3. Sampling and Data Preparation
2.4. Laboratory Analysis
2.5. Water Quality Index (WQI) Calculation
2.6. Evaluation of Water Quality for Irrigation
3. Results and Discussion
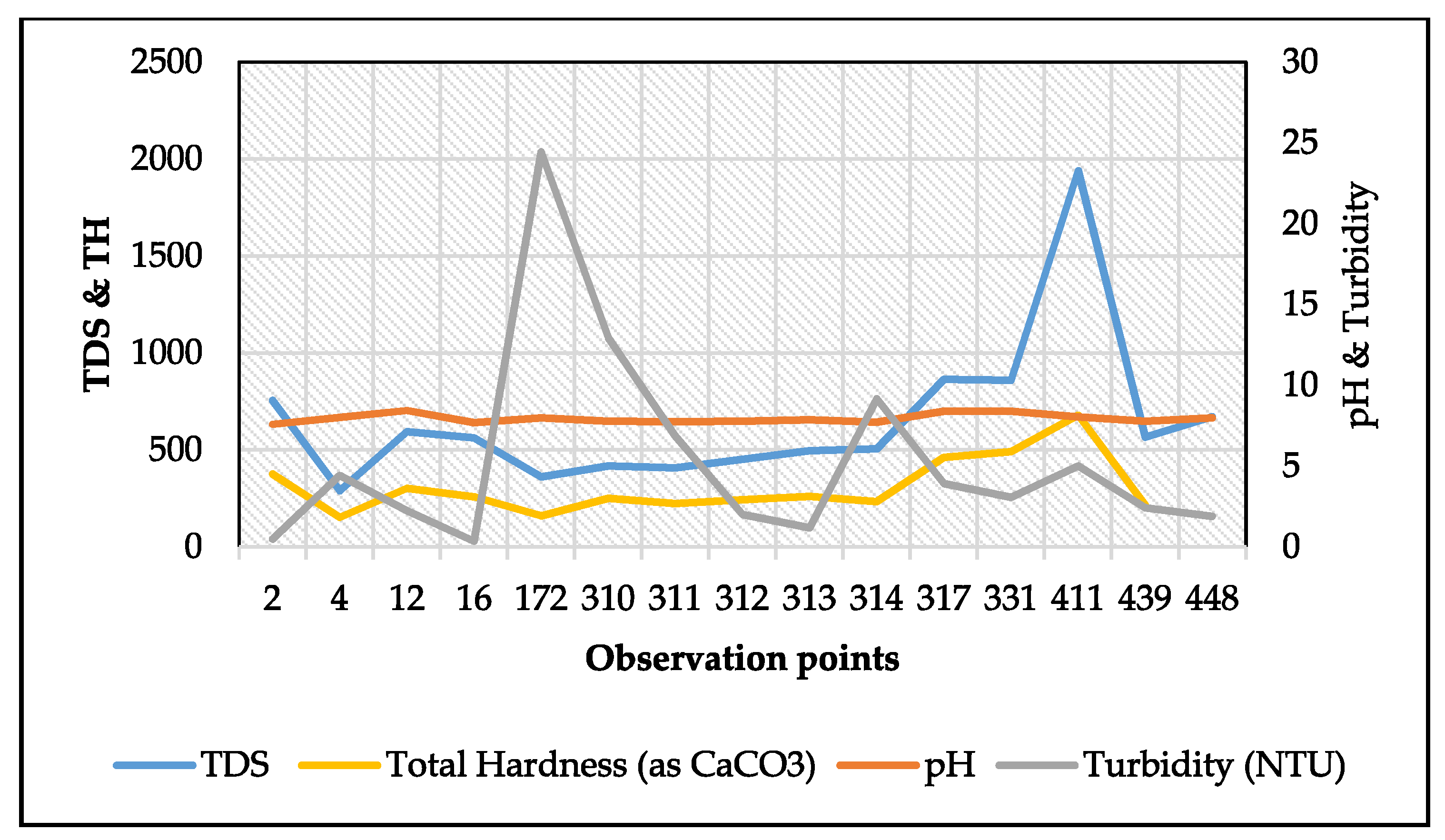
3.1. Physicochemical Parameters
3.1.1. Hydrogen Ion Potential (pH)
3.1.2. Total Dissolved Solids (TDS)
3.1.3. Total Hardness (TH)
3.1.4. Fluoride
3.1.5. Boron
3.1.6. Turbidity
3.1.7. Major Ion Chemistry
3.1.8. Total Iron
3.1.9. Nitrate
3.2. Comparison of Studied Parameters with Different Standards
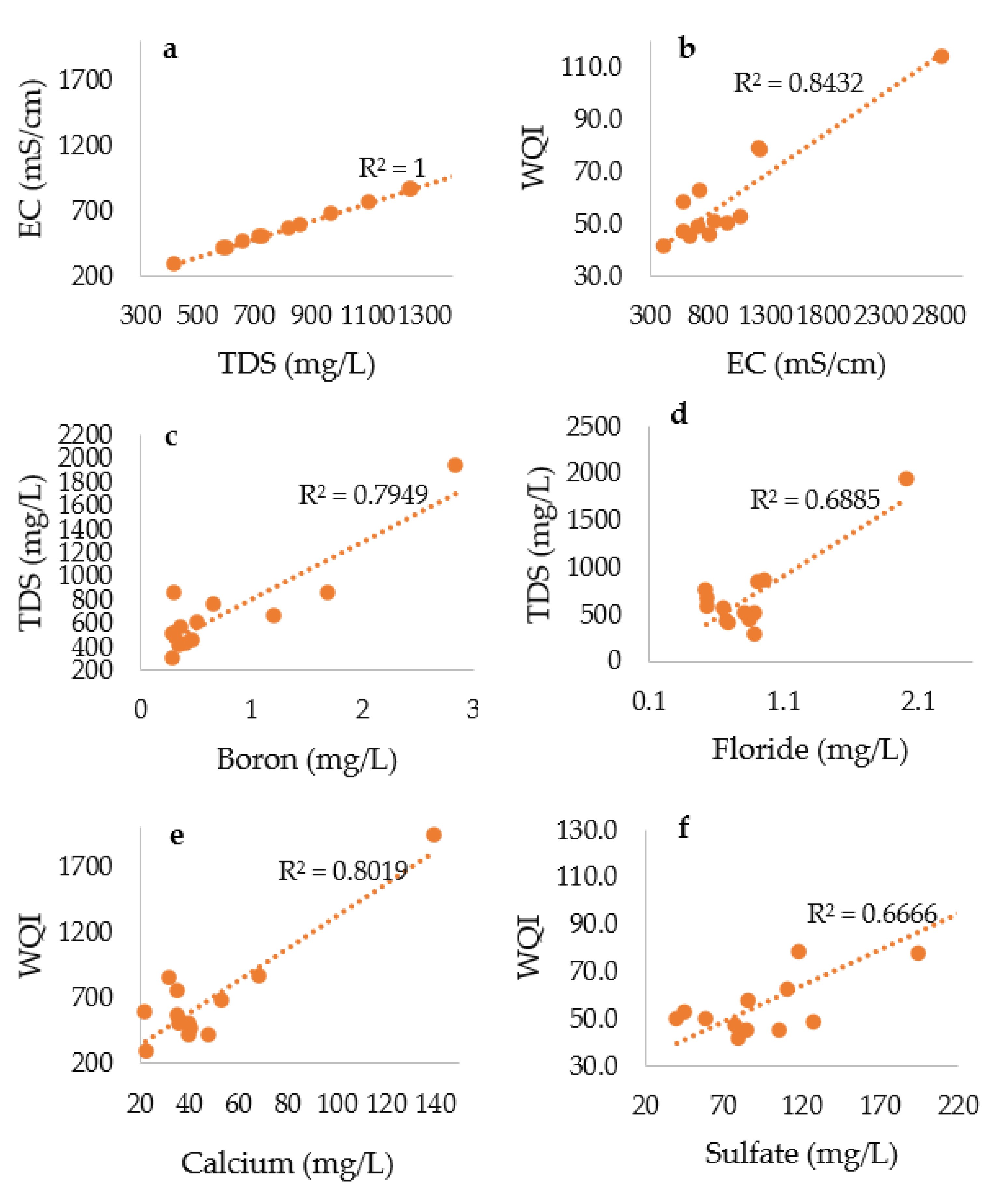
3.3. Correlation Analysis
3.4. Water Quality Index (WQI)
3.5. Interpretation of Water Quality for Irrigation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bruckmann, J.; Clauser, C. Ensemble-Based Stochastic Permeability and Flow Simulation of a Sparsely Sampled Hard-rock Aquifer Supported by High Performance Computing. Hydrogeol. J. 2020. [Google Scholar] [CrossRef]
- Danapour, M.; Hojberg, A.; Jensen, K.; Atisen, S. Assessment of Regional Inter-basin Groundwater Flow Using Both Simlpe and Highly Parametrized Optimization Schemes. Hydrogeol. J. 2019, 27, 1929–1947. [Google Scholar] [CrossRef]
- Maples, S.; Fogg, G.; Maxwell, R. Modelling Managed Aquifer Recharge Processes in a Highly Heterogeneous, Semi-confined Aquifer System. Hydrogeol. J. 2019, 27, 2869–2888. [Google Scholar] [CrossRef]
- Batabyal, A.K.; Chakraborty, S. Hydrogeochemistry and Water Quality Index in the Assessment of Groundwater Quality for Drinking Uses. Water Environ. Res. 2016, 607–617. [Google Scholar] [CrossRef]
- Lafta, J.G. Analysis of Water Quality Using Chemical-Physical-Biological Parameters of the Kinds of Water Used for Drinking in the Baghdad Province Al Adhamiya City. Chem. Mater. Res. 2015, 7, 1–4. [Google Scholar]
- Mehrdad, B.; Thomas, H. Effects of Upscaling Temporal Resolution of Groundwater Flow and Transport of Boundary on the Performance of Nitrate-Transport Models at the Regional Management Scale. Hydrogeol. J. 2020, 28, 1299–1322. [Google Scholar] [CrossRef]
- Beisner, K.; Solder, J.; Tillman, F.; Anderson, J.; Antwwiler, R. Geochemical Characterization of Groundwater Evolution South of Grand Canyon, Arizona (USA). Hydrogeol. J. 2020. [Google Scholar] [CrossRef]
- Apollaro, C.; Fuoco, I.; Brozzo, G.; De Rosa, R. Release and fate of Cr (VI) in the ophiolite aquifers of Italy: The role of Fe (III) as a potential oxidant of Cr (III) supported by reaction path modelling. Sci. Total Environ. 2019, 660, 1459–1471. [Google Scholar] [CrossRef]
- Teresa, C.; Marini, L.; Schott, J.; Mavromatic, V.; Apollaro, C.; Rinder, T.; Rosa, R.D.; Oelkers, E.H. Dissolution rates of actiolite and chlorite from a whole-rock experimental study of metabaslt dissolution from 2 ≤ pH ≥ 12 at 25C. Chem. Geol. 2014, 390, 100–108. [Google Scholar]
- Rao, C.S.; Rao, B.S.; Hariharan, A.V.L.N.S.H.; Bharathi, N.M. Determination of Water Quality Index of Some Areas in Guntur District of Andhra Pradesh. Int. J. Appl. Biol. Pharm. Technol. 2010, 1, 79–86. [Google Scholar]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; CRC Press: Amsterdam, The Netherlands, 2005; ISBN 04-1536-421-3. [Google Scholar]
- Fetter, C.W., Jr. Applied Hydrogeology, 4th ed.; PEARSON Press: London, UK, 2014; ISBN 1-292-02290-6. [Google Scholar]
- Merkel, B.J. Groundwater Geochemistry: A Practical Guide to Modeling of Natural and Contaminated Aquatic System; Darrell, K.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2005; ISBN 3-540-24195-7. [Google Scholar]
- Davidson, G.; Holt, H.; Blainey, J. Geochemical Assessment of the Degree of Isolation of Edge-of-aquifer Groundwater Along a Fringe of the Southern High Plains Aquifer, USA. Hydrogeol. J. 2019, 27, 1817–1825. [Google Scholar] [CrossRef]
- Bäumle, R.; Himmelsbach, T.; Noell, U. Hydrogeology and Geochemistry of a Tectonically Controlled, Deep-Seated and Semi-Fossil Aquifer in the Zambezi Region (Namibia). Hydrogeol. J. 2018, 27, 885–914. [Google Scholar] [CrossRef]
- Frischmann, P. Afghanistan Resource Corridor Development: Water Strategy Final Kabul River Basin Report. World Bank 2012, 1, 49–58. [Google Scholar]
- World Urbanization Prospect (WUP). Kabul Population; UN: New York, NY, USA, 2020. [Google Scholar]
- Safi, M.H. Groundwater Natural Reources and Quality Concerns in Kabul Basin, Afghanistan; DACAAR: Kabul, Afghanistan, 2011. [Google Scholar]
- Thomas, J.M. Conceptual Model of Water Resources in the Kabul Basin, Afghanistan; USGS: Reston, VA, USA, 2010. [Google Scholar]
- Robert, E.; Borshears, M.A. Inventory of Groundwater Resources in the Kabul Basin, Afghanistan; USGS: Reston, VA, USA, 2005. [Google Scholar]
- Zaryab, A.H.; Noori, A.R.; Wegerich, K.K.B. Assessment of Water quality and Quantity Trends in the Kabul Aquifers with an Outline for Future Drinking Water Supplies: Special issue on water management challenges in central Asia and Afghanistan. Cent. Asia J. Water Res. 2017, 3, 3–11. [Google Scholar]
- Thomas, J.M. Groundwater Availability in the Kabul Basin, Afghanistan; ResearchGate: Berlin, Germany, 2018. [Google Scholar]
- ANDWQS. Afghanistan National Drinking Water Quality Standard; Afghan National Standards Authority: Kabul, Afghanistan, 2012. [Google Scholar]
- Department of Agriculture, Environment and Rural Affairs (DAERA). Available online: https://www.daera-ni.gov.uk/ (accessed on 17 October 2016).
- World Health Organization (WHO). Guidelines for Drinking Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; 29 Press: Davis, CA, USA, 1994; ISBN 92-5-102263-1. [Google Scholar]
- Ashwani, K.T.; Prasoon, K.S.; Mukesh, K.M. GIS-Based Evaluation of Water Quality Index of Groundwater Resources in West Bokaro Coalfield, India. Curr. World Environ. 2014, 9, 843–850. [Google Scholar] [CrossRef]
- Jawad Alobaidy, A.H.; Abid, H.S.; Maulood, B.K. Application of Water Quality for Assessment of Dokan Lake Ecosystem, Kurdistan Region, Iraq. Water Resour. Prot. 2010, 792–798. [Google Scholar] [CrossRef]
- Yogendra, K.; Puttaiah, E. Determination of Water Quality Index and Suitability of an Urban Waterbody in Shimoga Town, Karmataka, Sengapta. Dep. PG Stud. Res. Environ. Sci. 2008, 342, 346. [Google Scholar]
- Etim, E.; Odoh, R.; Itodo, A.; Umoh, S.D.; Lawal, U. Water Quality Index for the Assessment of Water Quality from Different Sources in the Niger Delta Region of Nigeria. Front. Sci. 2013, 3, 89–95. [Google Scholar] [CrossRef]
- Patil, P.N.; Sawant, D.V.; Deshmuck, R.N. Physical-chemical Parameters for Testing of Water—A Riview. Int. J. Environ. Sci. 2012, 3, 1196–1207. [Google Scholar]
- Wurts, W.A. Understanding of Water Quality; ResearchGate: Berlin, Germany, 2016. [Google Scholar]
- Manitoba. Drinking Water Turbidity: Turbidity in Manitoba Water Supply; American Journal of Applied Chemistry: Dacca, Bangladesh, 2011. [Google Scholar]
- Gregory, M.H.; Michael, L.M. Turbidity as an Indicator of Water Quality in Diverse Watersheds of the Upper Pecos River Basin. Groundw. Qual. Bur. 2010, 273–284. [Google Scholar] [CrossRef]
- Indian Standard for Drinking Water-Specification. India Water Portal. Available online: http://www.indiawaterportal.org/articles/indian-standard-drinking-water-bis-specifications-10500 (accessed on 12 May 2012).
- Asuero, A.; Sayago, A.; Gonzlez, A. The Correlation Coefficients: An Overview. Crit. Rev. Anal. Chem. 2006, 36, 41–59. [Google Scholar] [CrossRef]
- Ahmad, I.K.; Salih, N.M.; Nzar, Y.H. Determination of Water Quality Index (WQI) for Qalyasan Stream in Sulaimani City/Kurdestan Region of Iraq. Int. J. Plant Anim. Environ. Sci. 2012, 2, 148–157. [Google Scholar]
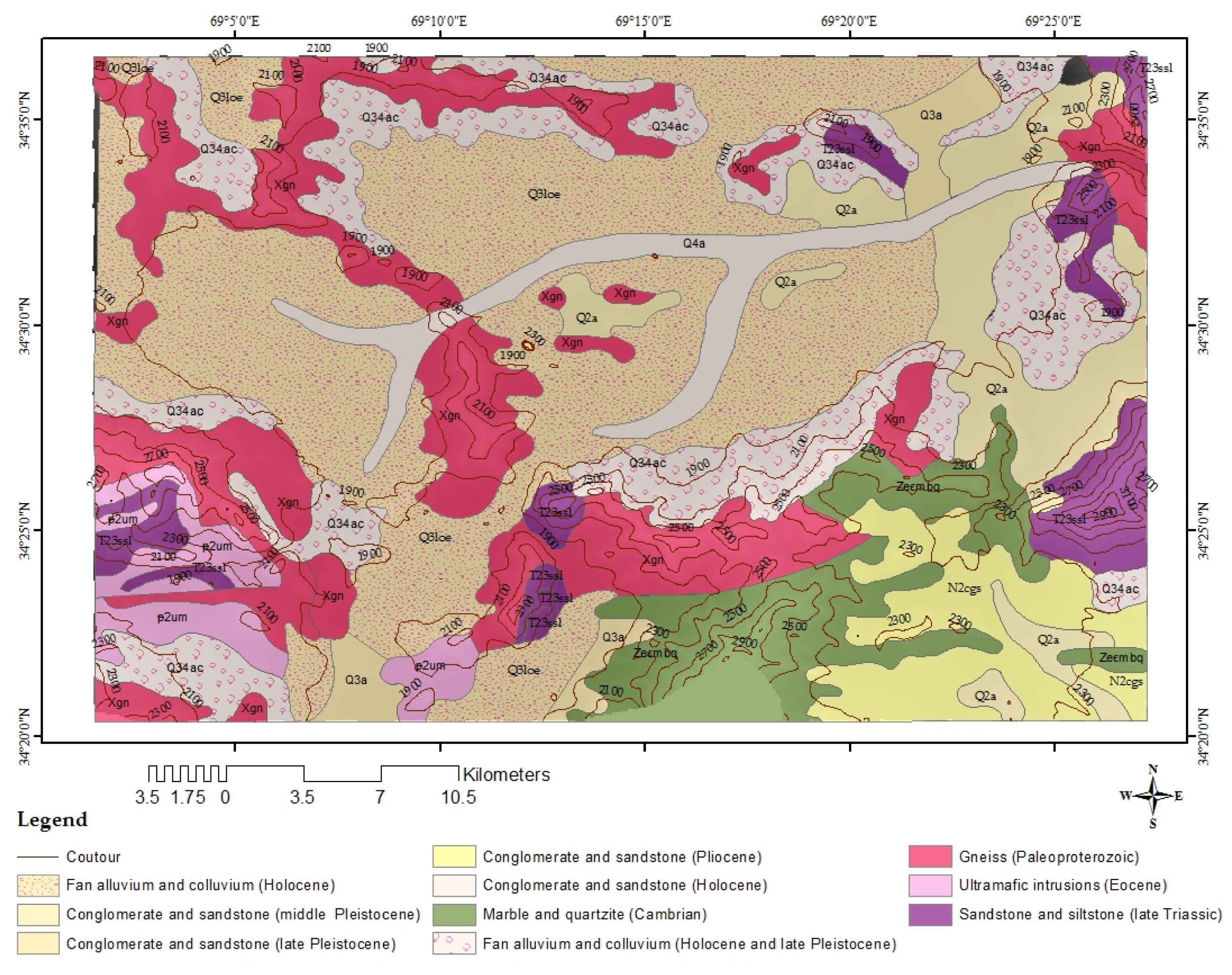

| System | Age | Lithology | Thickness Range |
|---|---|---|---|
| Quaternary | Recent | Clay, sand, gravel, pebble, and conglomerate | 600 m in total |
| Upper Tertiary | Neogene | Gray conglomerate, grit, limestone, marl | |
| Crystalline rocks | / | gneisses, granitic-gneisses, amphibolite, mica, shiest, quartzite, and marbles |
| Measurement | Device/Method | HACH Method | Reference Method |
|---|---|---|---|
| pH | Electrode | 8156 | EPA 150.1, 150.2; SM 4500-H + B |
| EC | Electrode | 8160 | SM 2510 B |
| TDS | Electrode | 8277 | SM 2540 C |
| Total hardness | Digital Titration | 8213 | SM 2340 B or C |
| Turbidity | Nephelometric | 8195 | EPA 180.1 |
| Sulfate | Turbidimetry | 8051 | ASTM D516-90,02 |
| Calcium | Titration | 8222 | SM 3500-Ca B or D |
| Magnesium | Colorimetry, Calmagite | 8030 | EPA 130.1 |
| Sodium | Electrode | 8322 | ASTM D2791-07 |
| Chloride | Digital Titration | 8207 | SM 4500-Cl B |
| Fluoride | Colorimetry, SPADNS, TNTplus | TNT878 | SM 4500-F B/D |
| Boron | Colorimetry, Carmine | 8015 | SM 4500-B |
| Total Iron | Colorimetry, 1,10-Phenanthroline | 8008 | 40 CFR 136 |
| Phosphate, Ortho | Colorimetry, Molybdate | 8048 | EPA 365.1/365.2 |
| Nitrate | Colorimetry, Dimethyl phenol | TNT 835 (10206) | 40CFR 141/136 |
| Parameters | Afghan/WHO Standard | Relative Weight ( | |
|---|---|---|---|
| (…) | (mg/L) | (…) | (mg/L) |
| pH | 6.5–8.5 | 4 | 0.108 |
| TDS | 1000 | 4 | 0.108 |
| TH | 500 | 3 | 0.054 |
| Turbidity | 5NTU | 3 | 0.081 |
| Sulfate | 250 | 5 | 0.108 |
| Calcium | 75 | 2 | 0.054 |
| Sodium | 200 | 4 | 0.108 |
| Magnesium | 30 | 2 | 0.054 |
| Chloride | 250 | 3 | 0.081 |
| Fluoride | 1.5 | 4 | 0.108 |
| Boron | 2.4 | 3 | 0.081 |
| Total Iron | 0.3 | 4 | 0.108 |
| Nitrate | 50 | 2 | 0.054 |
| Total | 39 | 1.000 | |
| WQI | Ranges | Water Type |
| 0–25 | Excellent Water | |
| 26–50 | Good Water | |
| 51–75 | Poor Water | |
| 76–100 | Very Poor Water | |
| >100 | Unsuitable for drinking |
| Infiltration Rate of Water into the Soil, Evaluation Using SAR and EC Together | Degree of Restriction on Use | ||||
|---|---|---|---|---|---|
| None | Slight–Moderate | Severe | |||
| SAR = | 0–3 | EC = | >0.7 | 0.7–0.2 | <0.2 |
| 3.0–6.0 | >1.2 | 1.2–0.3 | <0.3 | ||
| 6.0–12 | >1.9 | 1.9–0.5 | <0.5 | ||
| 12.0–20 | >2.9 | 2.9–1.3 | <1.3 | ||
| 20–40 | >5.0 | 5–2.9 | <2.9 | ||
| Point Name | Hardness of Water Samples | Hardness Rate | Percentage | |
|---|---|---|---|---|
| Very Hard | Hard | |||
| (mg/L) | … | % | % | |
| 2 | 376 | Very Hard | 86.7 | 13.3 |
| 4 | 152 | Hard | ||
| 12 | 302 | Very Hard | ||
| 16 | 259 | Very Hard | ||
| 172 | 160 | Hard | ||
| 310 | 252 | Very Hard | ||
| 311 | 223 | Very Hard | ||
| 312 | 242 | Very Hard | ||
| 313 | 261 | Very Hard | ||
| 314 | 234 | Very Hard | ||
| 317 | 461 | Very Hard | ||
| 331 | 492 | Very Hard | ||
| 411 | 679 | Very Hard | ||
| 439 | 216 | Very Hard | ||
| 448 | 338 | Very Hard | ||
| Observation Points | pH | EC | T | TDS | Total Hardness (as CaCO3) | Turbidity (NTU) | Sulphates | Calcium | Magnesium | Sodium | Chloride | Fluoride | Boron | Total Iron | Phosphate | Nitrate |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (…) | (µS/cm) | (°C) | (mg/L) | |||||||||||||
| 2 | 7.59 | 1100 | 17.5 | 757 | 430 | 0.48 | 46 | 35 | 70 | 104 | 100 | 0.52 | 0.67 | 0.05 | 0.27 | 58.40 |
| 4 | 8.01 | 420 | 14.8 | 289 | 190 | 4.41 | 80 | 23 | 23 | 81 | 33 | 0.8l9 | 0.28 | 0.04 | 0.05 | 25.00 |
| 12 | 8.44 | 863 | 19.3 | 594 | 380 | 2.23 | 60 | 22 | 60 | 140 | 57 | 0.53 | 0.51 | 0.07 | 0.13 | 11.16 |
| 16 | 7.7 | 815 | 19.4 | 561 | 390 | 0.35 | 64 | 36 | 41 | 87 | 58 | 0.57 | 0.41 | 0.07 | 0.17 | 40.80 |
| 172 | 7.97 | 523 | 16.4 | 360 | 270 | 24.44 | 18 | 36 | 17 | 78 | 25 | 0.83 | 0.35 | 0.05 | 0.13 | 21.40 |
| 310 | 7.77 | 606 | 19.8 | 417 | 300 | 12.95 | 87 | 48 | 32 | 59 | 29 | 0.68 | 0.4 | 0.05 | 0.09 | 15.30 |
| 311 | 7.75 | 593 | 21.2 | 408 | 290 | 6.85 | 78 | 40 | 30 | 149 | 25 | 0.69 | 0.36 | 0.05 | 0.08 | 20.40 |
| 312 | 7.77 | 657 | 18.3 | 452 | 460 | 2.00 | 106 | 41 | 34 | 121 | 22 | 0.85 | 0.47 | 0.06 | 0.11 | 20.06 |
| 313 | 7.87 | 723 | 16.7 | 497 | 290 | 1.17 | 128 | 40 | 39 | 157 | 32 | 0.89 | 0.28 | 0.06 | 0.02 | 54.00 |
| 314 | 7.72 | 735 | 17.1 | 506 | 350 | 9.15 | 112 | 36 | 35 | 197 | 88 | 0.82 | 0.31 | 0.09 | 0.05 | 60.20 |
| 317 | 8.4 | 1256 | 19.2 | 864 | 390 | 3.92 | 195 | 69 | 70 | 97 | 155 | 0.96 | 0.3 | 0.05 | 0.12 | 63.00 |
| 331 | 8.39 | 1249 | 19.5 | 859 | 420 | 3.05 | 120 | 32 | 100 | 103 | 105 | 0.92 | 1.7 | 0.07 | 0.15 | 55.00 |
| 411 | 8.04 | 2820 | 15.8 | 1940 | 700 | 5.00 | 224 | 140 | 80 | 19 | 145 | 2.02 | 2.84 | 0.09 | 0.58 | 29.52 |
| 439 | 7.78 | 820 | 17.8 | 564 | 390 | 2.41 | 85 | 35.2 | 31 | 120 | 31 | 0.66 | 0.37 | 0.07 | 0.09 | 40.18 |
| 448 | 7.99 | 974 | 15.7 | 670 | 520 | 1.90 | 40 | 53 | 50 | 131 | 82 | 0.54 | 1.21 | 0.02 | 0.19 | 12.18 |
| Parameters | Standards | Current Samples | ||||
|---|---|---|---|---|---|---|
| Afghan | WHO | Indian | Max | Min | Percent above WHO Standard | |
| pH | 6.5–8.5 | 6.5–8.5 | 6.5–8.5 | 8.44 | 7.59 | 0 |
| TDS | 1000 | 259–500 | 500–2000 | 1940 | 289 | 61.5 |
| TH | 500 | 200 | 300–600 | 679 | 152 | 15 |
| Turbidity | 5NTU | 5–25 | 10–25 | 24.44 | 0.35 | 30.7 |
| Sulfate | 250 | 200–400 | 200–400 | 224 | 18 | 0 |
| Calcium | 75 | 75–200 | 75–200 | 140 | 22 | 7.7 |
| Magnesium | 30 | 30–150 | 30–100 | 100 | 17 | 92.3 |
| Sodium | 200 | 200 | 200 | 197 | 19 | 0 |
| Chloride | 250–1000 | 200–600 | 250–1000 | 155 | 22 | 0 |
| Fluoride | <1.5 | / | 1–1.5 | 2.02 | 0.52 | 0 |
| Boron | 2.4 | 2.4 | 2.84 | 0.28 | 7.6 | |
| Total Iron | 0.3 | 0.1–1.0 | 0.3–1.0 | 0.09 | 0.02 | 0 |
| Nitrate | 50 | 50 | 45 | 63 | 11.16 | 39 |
| R2 | Interpretation |
|---|---|
| 1–0.9 | Very high correlation |
| 0.89–0.70 | High correlation |
| 0.69–0.50 | Moderate correlation |
| 0.49–0.30 | Low correlation |
| 0.29–0.00 | Little if any correlation |
| Observation Points | WQI | Water Type | Percentage | ||||
|---|---|---|---|---|---|---|---|
| Excellent | Good | Poor | Very Poor | U.F. Drinking | |||
| 2 | 52.36 | Poor water | 0% | 40% | 40% | 13.33% | 6.66% |
| 4 | 41.40 | Good water | |||||
| 12 | 50.59 | Poor water | |||||
| 16 | 42.91 | Good water | |||||
| 172 | 71.89 | Poor water | |||||
| 310 | 57.88 | Poor water | |||||
| 311 | 46.68 | Good water | |||||
| 312 | 45.03 | Good water | |||||
| 313 | 48.47 | Good water | |||||
| 314 | 62.53 | Poor water | |||||
| 317 | 77.52 | Very Poor water | |||||
| 331 | 78.47 | Very Poor water | |||||
| 411 | 113.51 | Unsuitable for drinking | |||||
| 439 | 45.47 | Good water | |||||
| 448 | 50.27 | Poor water | |||||
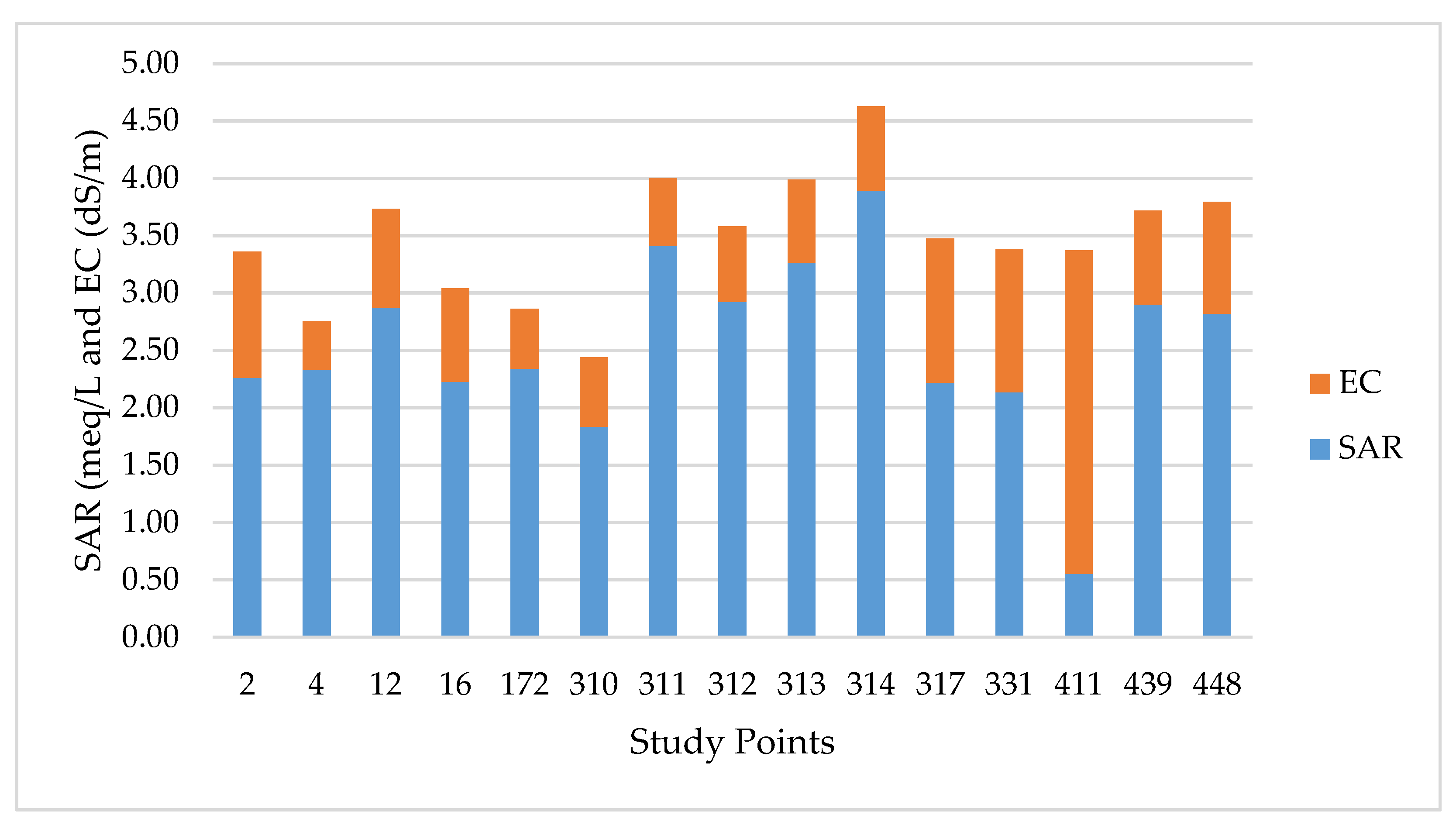
| Parameters | Unit | Degree of Restriction on Use | Study Points | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| None | Slight–Moderate | Severe | 2 | 4 | 12 | 16 | 172 | 310 | 311 | 312 | 313 | 314 | 317 | 331 | 411 | 439 | 448 | |||
| EC | dS/m | <0.7 | 0.7-3 | >3 | 1.1 | 0.42 | 0.86 | 0.81 | 0.52 | 0.6 | 0.59 | 0.65 | 0.72 | 0.73 | 1.25 | 1.24 | 2.82 | 0.82 | 0.97 | |
| TDS | mg/L | <450 | 450–2000 | >2000 | 757 | 289 | 594 | 561 | 360 | 417 | 408 | 452 | 497 | 506 | 864 | 859 | 1940 | 564 | 670 | |
| Ca2+ | meq/L | 0–20 | 1.75 | 1.15 | 1.10 | 1.80 | 1.80 | 2.40 | 2.00 | 2.05 | 2.00 | 1.80 | 3.45 | 1.60 | 7.00 | 1.76 | 2.65 | |||
| Mg2+ | meq/L | 0–5 | 3.5 | 1.1 | 3.0 | 2.0 | 0.8 | 1.6 | 1.5 | 1.7 | 1.9 | 1.7 | 3.5 | 5.0 | 4.0 | 1.5 | 2.5 | |||
| Na+ | meq/L | <3 | 3–9 | >9 | 4.5 | 3.5 | 6.1 | 3.8 | 3.4 | 2.6 | 6.5 | 5.3 | 6.8 | 8.6 | 4.2 | 4.5 | 0.8 | 5.2 | 5.7 | |
| CO3−2 | meq/L | 0–0.1 | 0.67 | 0.33 | 1.33 | 0.67 | 0.67 | 0.33 | 1.00 | 0.67 | 1.00 | 0.67 | 0.33 | 0.00 | 1.00 | 0.67 | 1.33 | |||
| HCO3− | meq/L | <1.5 | 1.5–8.5 | >8.5 | 6.56 | 3.20 | 7.70 | 4.59 | 4.43 | 4.34 | 7.21 | 6.23 | 6.39 | 6.64 | 2.79 | 7.21 | 4.02 | 5.41 | 7.70 | |
| Cl− | meq/L | <4 | 4.0–10 | >10 | 2.82 | 0.93 | 1.61 | 1.64 | 0.71 | 0.82 | 0.71 | 0.62 | 0.90 | 2.48 | 4.37 | 2.96 | 4.09 | 0.87 | 2.31 | |
| SO42− | meq/L | 0.2 | 0.96 | 1.67 | 1.25 | 1.33 | 0.38 | 1.81 | 1.63 | 2.21 | 2.67 | 2.33 | 4.06 | 2.50 | 4.67 | 1.77 | 0.83 | |||
| NO3− N | mg/L | <5 | 5.0–30 | >30 | 58.4 | 25 | 11.2 | 40.8 | 21.4 | 15.3 | 20.4 | 20.1 | 54 | 60.2 | 63 | 55 | 29.5 | 40.2 | 12.2 | |
| PO4-P | mg/L | 0–2 | 0.27 | 0.05 | 0.13 | 0.17 | 0.13 | 0.09 | 0.08 | 0.11 | 0.02 | 0.05 | 0.12 | 0.15 | 0.58 | 0.09 | 0.19 | |||
| B | mg/L | <0.7 | 0.7–3 | >3.0 | 0.67 | 0.28 | 0.51 | 0.41 | 0.35 | 0.4 | 0.36 | 0.47 | 0.28 | 0.31 | 0.3 | 1.7 | 2.84 | 0.37 | 1.21 | |
| pH | 6.5–8.4 | 7.59 | 8.01 | 8.44 | 7.7 | 7.97 | 7.77 | 7.75 | 7.77 | 7.87 | 7.72 | 8.4 | 8.39 | 8.04 | 7.78 | 7.99 | ||||
| Legend | Non-Classified | No Restriction on Use | Slight–Moderate Restriction on Use | Severe Restriction on Use | ||||||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jawadi, H.A.; Sagin, J.; Snow, D.D. A Detailed Assessment of Groundwater Quality in the Kabul Basin, Afghanistan, and Suitability for Future Development. Water 2020, 12, 2890. https://doi.org/10.3390/w12102890
Jawadi HA, Sagin J, Snow DD. A Detailed Assessment of Groundwater Quality in the Kabul Basin, Afghanistan, and Suitability for Future Development. Water. 2020; 12(10):2890. https://doi.org/10.3390/w12102890
Chicago/Turabian StyleJawadi, Hussain Ali, Jay Sagin, and Daniel D. Snow. 2020. "A Detailed Assessment of Groundwater Quality in the Kabul Basin, Afghanistan, and Suitability for Future Development" Water 12, no. 10: 2890. https://doi.org/10.3390/w12102890
APA StyleJawadi, H. A., Sagin, J., & Snow, D. D. (2020). A Detailed Assessment of Groundwater Quality in the Kabul Basin, Afghanistan, and Suitability for Future Development. Water, 12(10), 2890. https://doi.org/10.3390/w12102890







