Water Treatment Plant Prototype with pH Control Modeled on Fuzzy Logic for Removing Arsenic Using Fe(VI) and Fe(III)
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
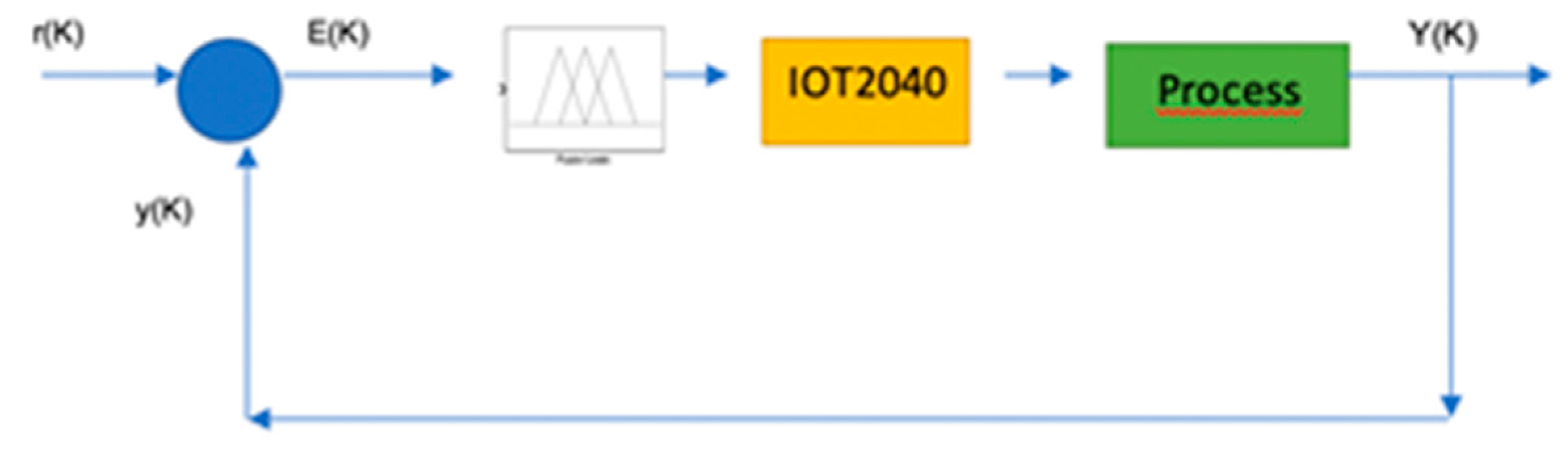
3.1. pH Control
3.2. Arsenic Removal
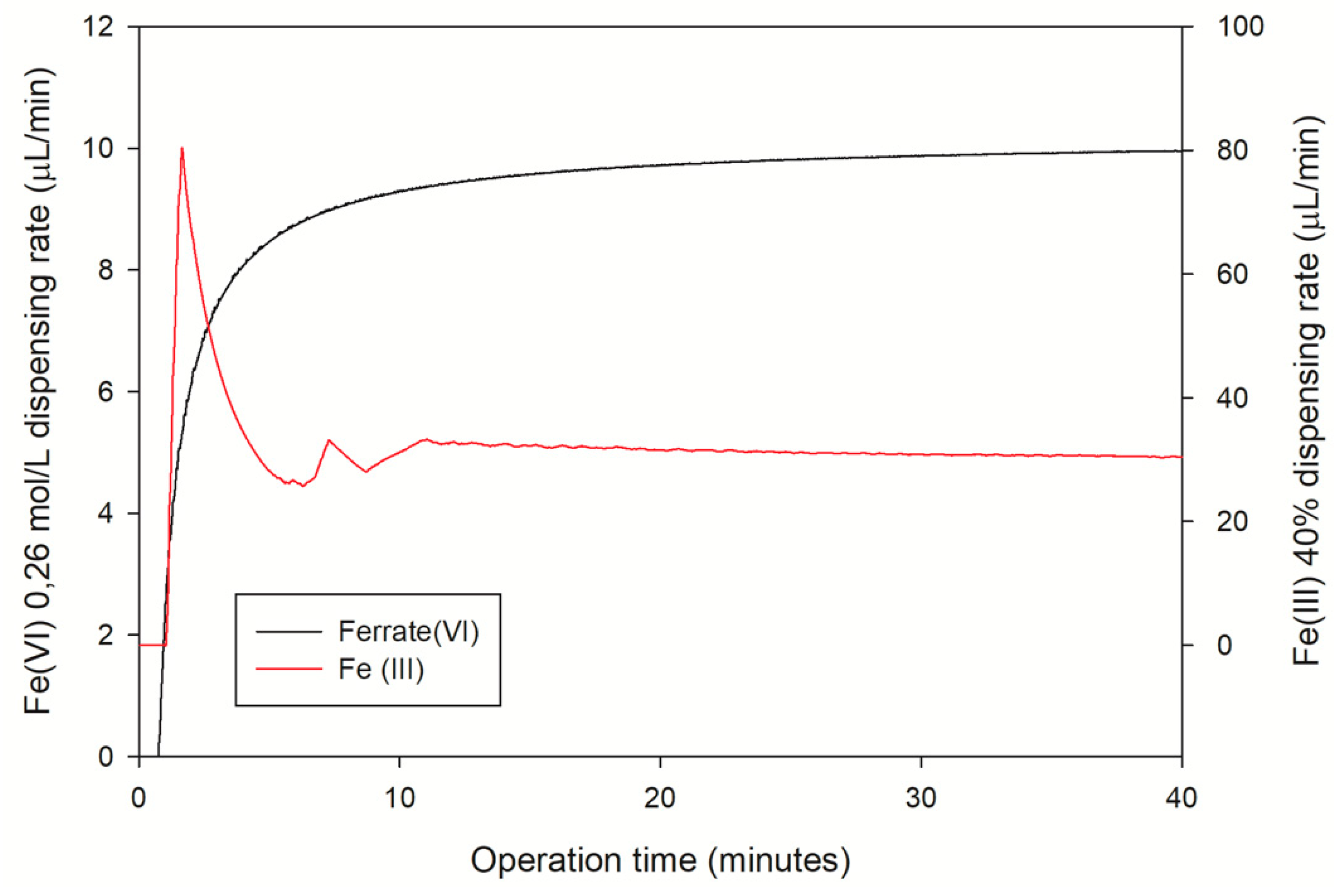
3.3. Fe(VI) and Fe(III) Instantaneous Dosing
3.4. Estimated Cost of the Treated Water
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Design and Implementation of pH Control with Fuzzy Logic
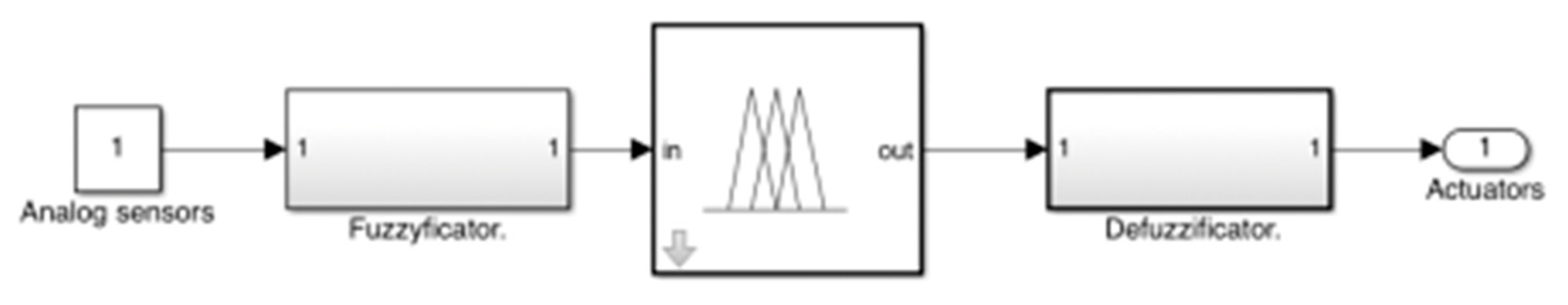
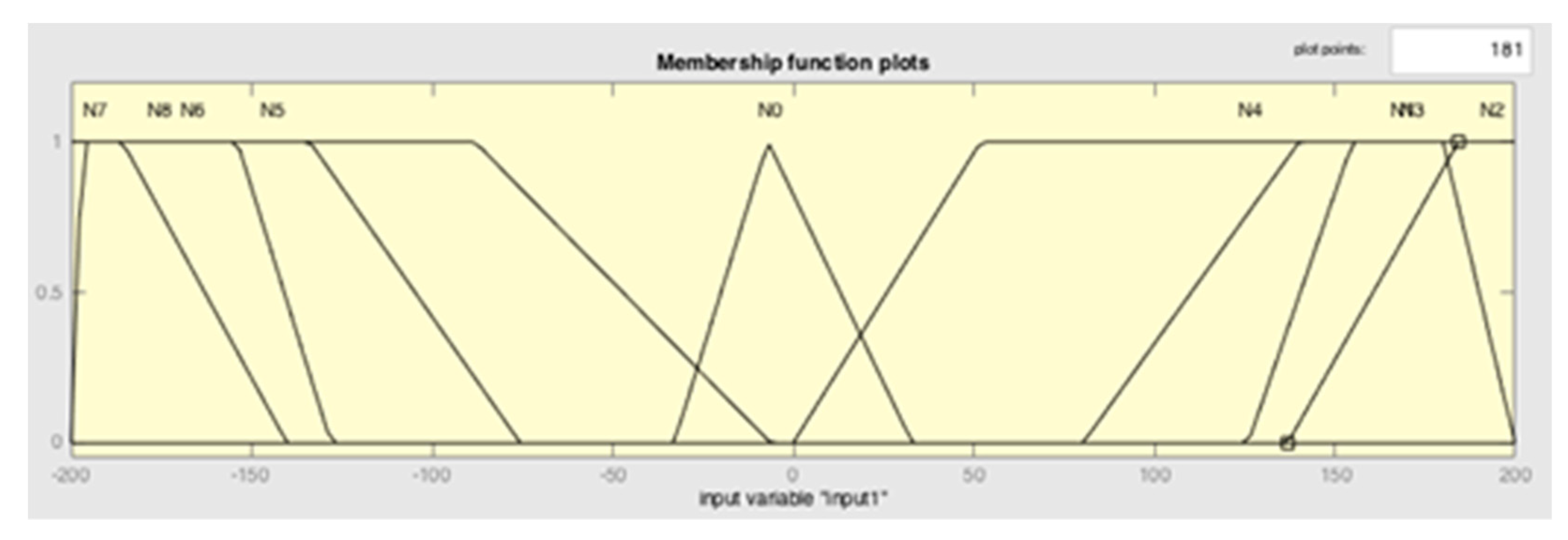
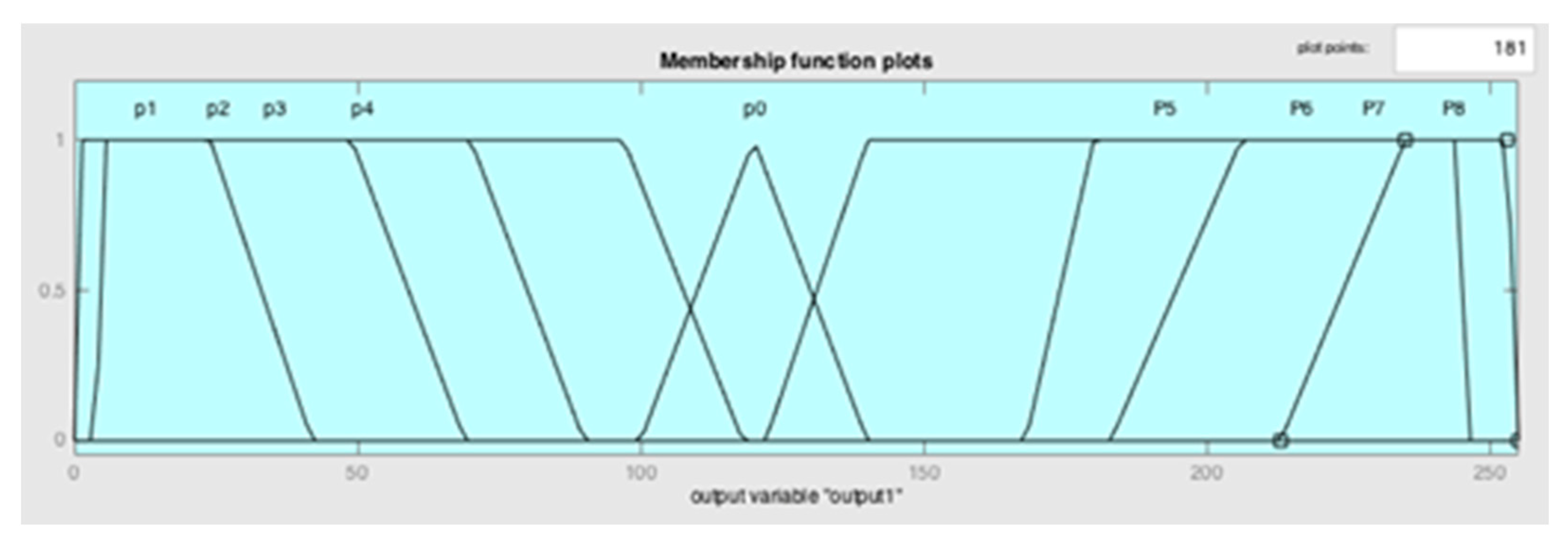
| Input | Output | ||
|---|---|---|---|
| Variable | Function Name | Variable | Function Name |
| Negative_very high | N1 | Power_very low | P1 |
| Negative_high | N2 | Power_low | P2 |
| Negative_medium | N3 | Power_medium_ low | P3 |
| Negative_very low | N4 | Power_very low | P4 |
| Balance | N0 | Power_ideal | P0 |
| Positive_very low | N5 | Power_very high | P5 |
| Positive_medium | N6 | Power_medium_high | P6 |
| Positive_high | N7 | Power_high | P7 |
| Positive_very high | N8 | Power_very high | P8 |
| Functions of the Fuzzifier (Inputs) | Fuzzy Rule System | Functions of the Defuzzifier (Outputs) |
|---|---|---|
| The pH error is N1 | If (input N0) then (output P0) | Pump is set to P1 |
| The pH error is N2 | If (input N1) then (output P1) | Pump is set to P1 |
| The pH error is N3 | If (input N3) then (output P3) | Pump is set to P3 |
| The pH error is N4 | If (input N4) then (output P4) | Pump is set to P2 |
| The pH error is N0 | If (input N5) then (output P5) | Pump is set to P0 |
| The pH error is N5 | If (input N6) then (output P6) | Pump is set to P5 |
| The pH error is N6 | If (input N8) then (output P8) | Pump is set to P6 |
| The pH error is N7 | If (input N7) then (output P7) | Pump is set to P7 |
| The pH error is N8 | If (input N2) then (output P7) | Pump is set to P8 |
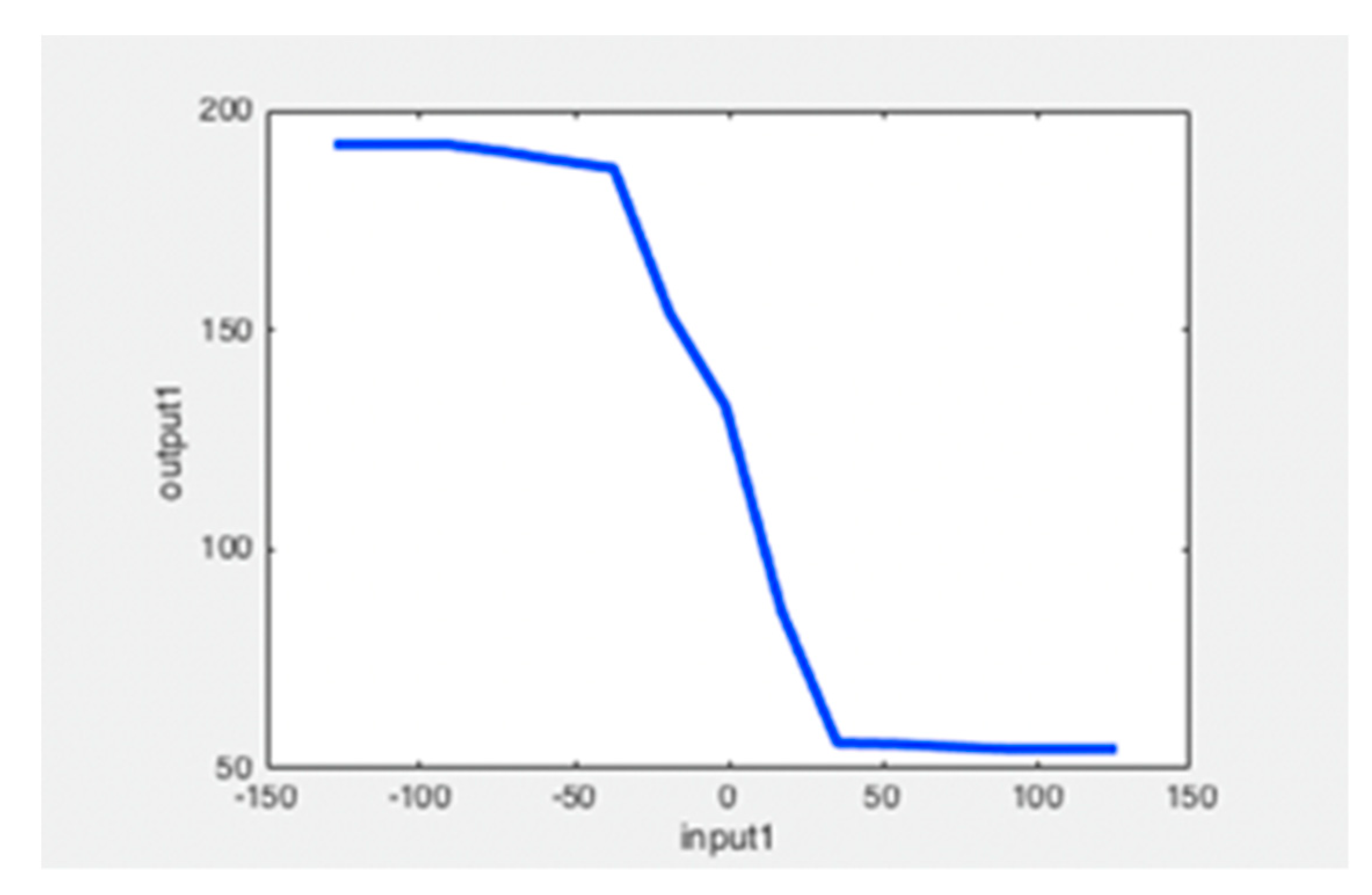
References
- Bernex, N.; Novoa, Z. Aguas y Arsénico Natural en Perú; Academia Nacional de Ciencias, Sociedad Geográfica de Lima: Lima, Peru, 2015. [Google Scholar]
- Castro de Esparza, M.L. El arsénico en los recursos hídricos del Perú. In Aguas y arsénico natural en Perú; Academia Nacional de Ciencias, Sociedad Geográfica de Lima: Lima, Peru, 2015. [Google Scholar]
- Hering, J.G.; Chen, P.Y.; Wilkie, J.A.; Elimelech, M.; Liang, S. Arsenic removal by ferric chloride. J. Am. Water Works Assoc. 1996, 88, 155–167. [Google Scholar] [CrossRef]
- Sorlini, S.; Gialdini, F. Conventional oxidation treatments for the removal of arsenic with chlorine dioxide, hypochlorite, potassium permanganate and monochloramine. Water Res. 2010, 44, 5653–5659. [Google Scholar] [CrossRef]
- Ghernaout, D.; Naceur, M.W. Ferrate (VI): In situ generation and water treatment–A review. Desalin. Water Treat. 2011, 30, 319–332. [Google Scholar] [CrossRef]
- Licht, S.; Yu, X. Electrochemical Alkaline Fe(VI) Water Purification and Remediation. Environ. Sci. Technol. 2005, 39, 8071–8076. [Google Scholar] [CrossRef] [PubMed]
- Prucek, R.; Tuček, J.; Kolařík, J.; Hušková, I.; Filip, J.; Varma, R.S.; Sharma, V.K.; Zbořil, R. Ferrate (VI)-prompted removal of metals in aqueous media: Mechanistic delineation of enhanced efficiency via metal entrenchment in magnetic oxides. Environ. Sci. Technol. 2015, 49, 2319–2327. [Google Scholar] [CrossRef]
- Tiwari, D.; Lee, S.M. Ferrate (VI) in the treatment of wastewaters: A new generation green chemical. Wastewater Treat. Reutil. 2011, 12, 241–276. [Google Scholar]
- Yates, B.J.; Zbořil, R.; Sharma, V.K. Engineering Aspects of Ferrate in Water and Wastewater Treatment-a Review. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2014, 49, 1603–1614. [Google Scholar] [CrossRef]
- Farooqi, S.; Bari, A. High level disinfection of wastewaters for reuse. Environ. Technol. 1988, 9, 379–390. [Google Scholar] [CrossRef]
- Cui, J.; Zheng, L.; Deng, Y. Emergency water treatment with ferrate (VI) in response to natural disasters. Environ. Sci. Water Res. Technol. 2018, 4, 359–368. [Google Scholar] [CrossRef]
- Jiang, J.Q.; Stanford, C.; Alsheyab, M. The on-line generation and application of ferrate(VI) for sewage treatment—A pilot scale trial. Sep. Purif. Technol. 2009, 68, 227–231. [Google Scholar] [CrossRef]
- Ang, K.H.; Chong, G.; Li, Y. PID control system analysis, design, and technology. IEEE Trans. Control Syst. Technol. 2005, 13, 559–576. [Google Scholar]
- Abdullah, N.H.S.; Karsiti, M.N.; Ibrahim, R. A review of pH neutralization process control. In Proceedings of the 2012 4th International conference on intelligent and Advanced Systems (ICIAS2012), IEEE, Kuala Lumpur, Malaysia, 12–14 June 2012; Volume 2, pp. 594–598. [Google Scholar]
- Von Altrock, C.; Krause, B.; Zimmermann, H.J. Advanced fuzzy logic control technologies in automotive applications. In Proceedings of the IEEE International Conference on Fuzzy Systems, IEEE, San Diego, CA, USA, 8–12 March 1992; pp. 835–842. [Google Scholar]
- Sahai, N.; Lee, Y.J.; Xu, H.; Ciardelli, M.; Gaillard, J.F. Role of Fe (II) and phosphate in arsenic uptake by coprecipitation. Geochim. Cosmochim. Acta 2007, 71, 3193–3210. [Google Scholar] [CrossRef]
- Quino-Favero, J.; Eyzaguirre, R.; Mogrovejo, P.; Prieto, P.; del Pino, L.F. Electrochemical synthesis of ferrate(VI): Optimization of parameters and evaluation of their impact in production cost. Desalin. Water Treat. 2018, 113, 179–186. [Google Scholar] [CrossRef]
- Licht, S.; Naschitz, V.; Halperin, L.; Halperin, N.; Lin, L.; Chen, J.; Ghosh, S.; Liu, B. Analysis of ferrate(VI) compounds and super-iron Fe(VI) battery cathodes: FTIR, ICP, titrimetric, XRD, UV/VIS, and electrochemical characterization. J. Power Sources 2001, 101, 167–176. [Google Scholar] [CrossRef]
- British Geographical Survey. Arsenic Contamination of Groundwater in Bangladesh; Survey (Technical Report, WC/00/19. 4 Volumes); British Geological Survey: Keyworth, UK, 2001. [Google Scholar]
- Amrose, S.; Gadgil, A.; Srinivasan, V.; Kowolik, K.; Muller, M.; Huang, J.; Kostecki, R. Arsenic removal from groundwater using iron electrocoagulation: Effect of charge dosage rate. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2013, 48, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- British Geographical Survey, and Department of Public Health and Engineering. Arsenic Contamination of Groundwater in Bangladesh; Kinninburg, D.G., Smedley, P.L., Eds.; British Geological Survey Technical Report WC/00/19; British Geological Survey: Keyworth, UK, 2001. [Google Scholar]
- Gallagher, P.A.; Schwegel, C.A.; Wei, X.; Creed, J.T. Speciation and preservation of inorganic arsenic in drinking water sources using EDTA with IC separation and ICP-MS detection. J. Environ. Monit. 2001, 3, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, P.A.; Schwegel, C.A.; Parks, A.; Gamble, B.M.; Wymer, L.; Creed, J.T. Preservation of As(III) and As(V) in drinking water supply samples from across the United States using EDTA and acetic acid as a means of minimizing iron-arsenic coprecipitation. Environ. Sci. Technol. 2004, 38, 2919–2927. [Google Scholar] [CrossRef]
- Smith, R.B.; Giesy, P.M. The titration of ferric chloride with sodium hydroxide, using the oxygen electrode: A proof of the non-existence on iron oxychloride. J. Am. Pharm. Assoc. 1923, 12, 855–856. [Google Scholar]
- Kelkar, B.; Postlethwaite, B. Study of pH control process using fuzzy modeling. In Proceedings of the 1994 International Conference on Control-Control’94, IET, Orlando, FL, USA, 21–24 March 1994; Volume 1, pp. 272–275. [Google Scholar]
- Velázquez-González, R.; Gómez-Lemus, T.; Rodríguez-Reséndiz, J. A pH process control embedded on a PLC using Fuzzy Logic. In Proceedings of the 2017 XIII International Engineering Congress (CONIIN), Santiago de Queretaro, Mexico, 15–19 May 2017; pp. 1–6. [Google Scholar]
- Hao, L.; Liu, M.; Wang, N.; Li, G. A critical review on arsenic removal from water using iron-based adsorbents. RSC Adv. 2018, 8, 39545–39560. [Google Scholar] [CrossRef]
- Roberts, L.C.; Hug, S.J.; Ruettimann, T.; Billah, M.; Khan, A.W.; Rahman, M.T. Arsenic removal with Iron(II) and Iron(III) in waters with high silicate and phosphate concentrations. Environ. Sci. Technol. 2004, 38, 307–315. [Google Scholar] [CrossRef]
- Lee, Y.; Um, I.H.; Yoon, J. Arsenic (III) oxidation by iron (VI)(ferrate) and subsequent removal of arsenic (V) by iron (III) coagulation. Environ. Sci. Technol. 2003, 37, 5750–5756. [Google Scholar] [CrossRef] [PubMed]
- Samadzadeh Yazdi, M.R.; Khodadadi Darban, A. Effect of Arsenic Speciation on Remediation of Arsenic- Contaminated Soils and Waters. In Proceedings of the 15th International Conference on Heavy Metals in the Environment, Gdańsk, Poland, 19–23 September 2010; pp. 492–495. [Google Scholar]
- Puigdomenech, I. Hydra/Medusa Chemical Equilibrium Database and Plotting Software; KTH Royal Institute of Technology: Stockholm, Sweden, 2004. [Google Scholar]
- Stenerson, J.; Deeg, D. Programming Siemens Step 7 (TIA Portal), a Practical and Understandable Approach; CreateSpace Independent Publishing Platform: Scotts Valley, CA, USA, 2015. [Google Scholar]
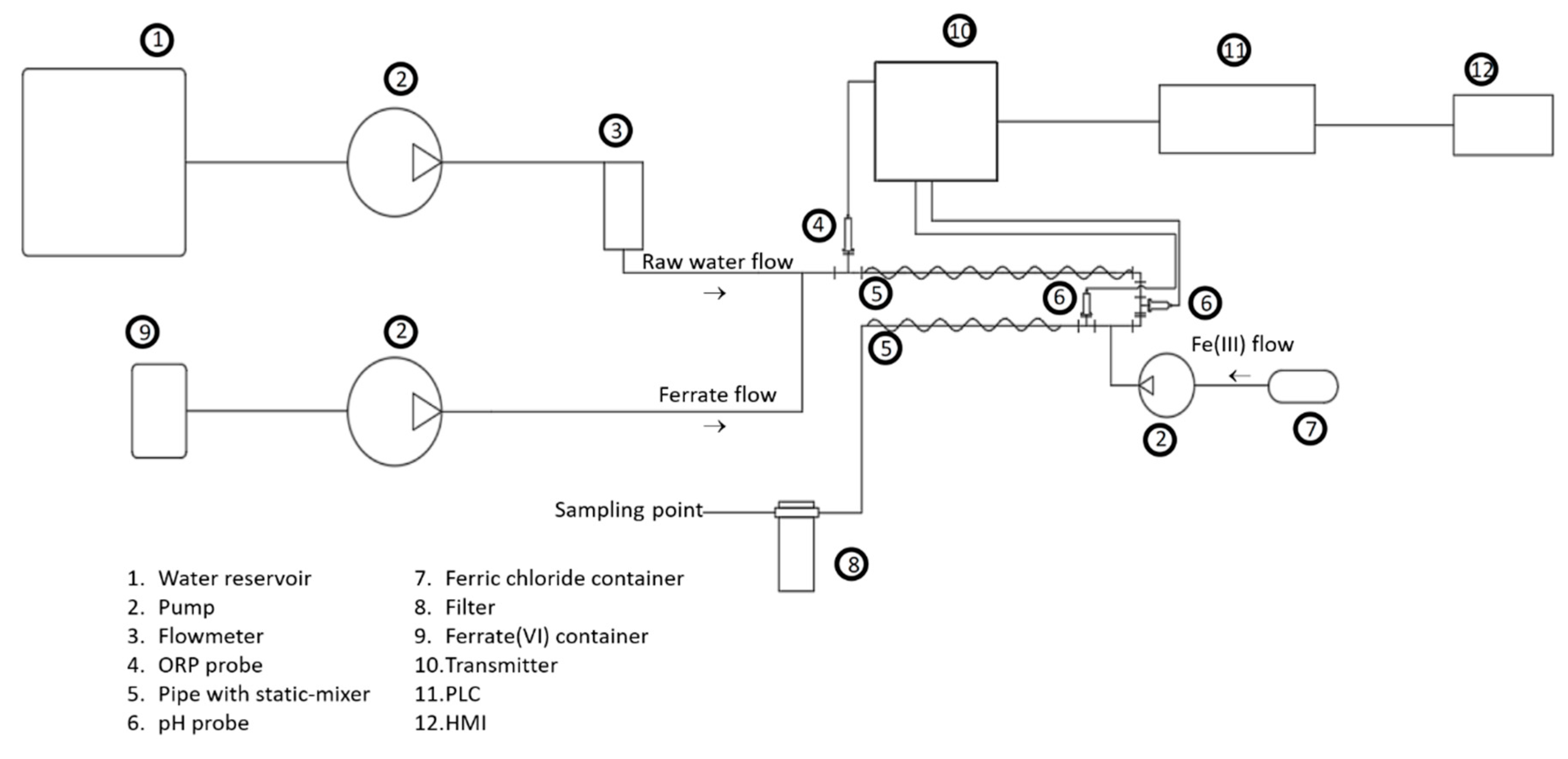
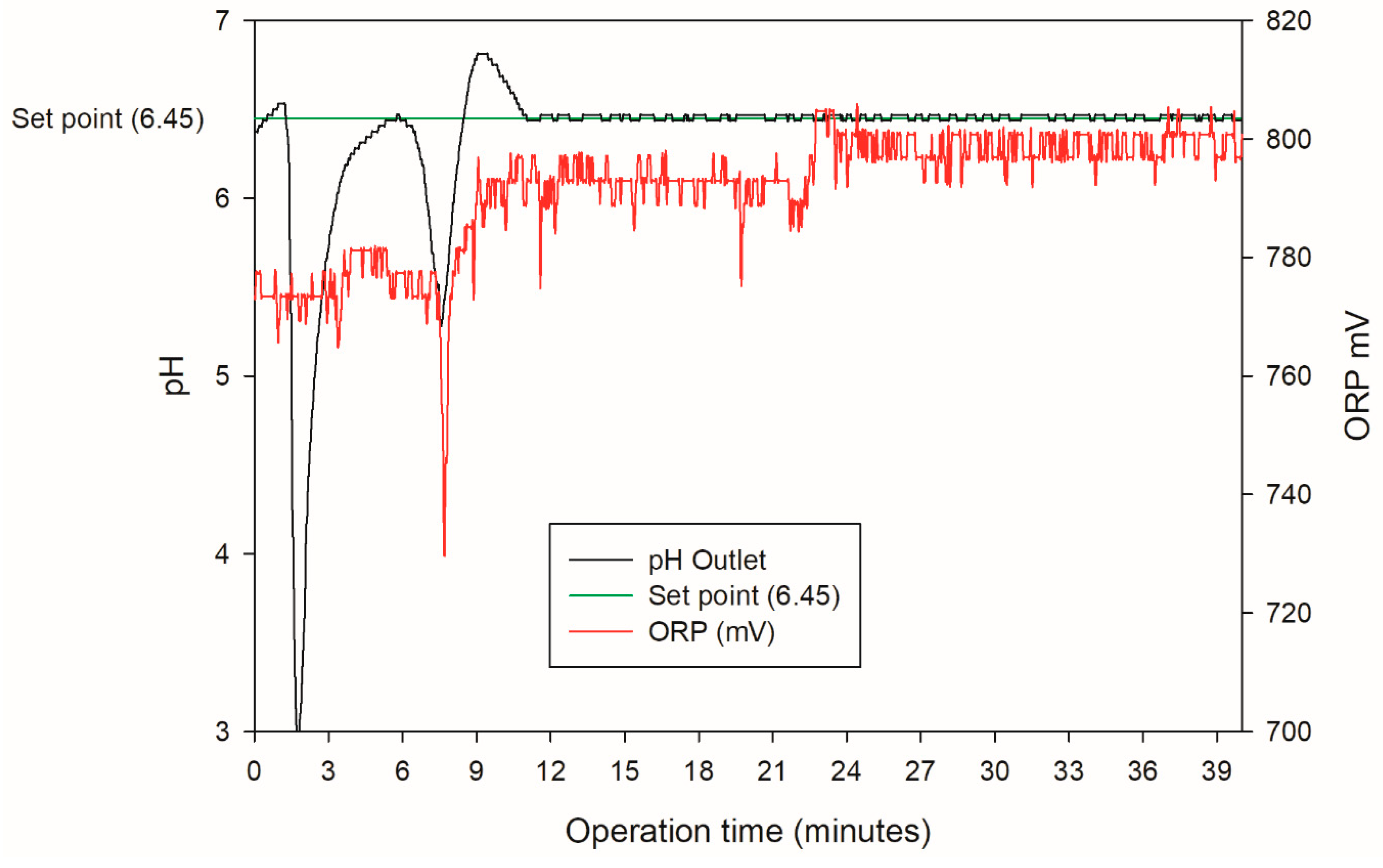

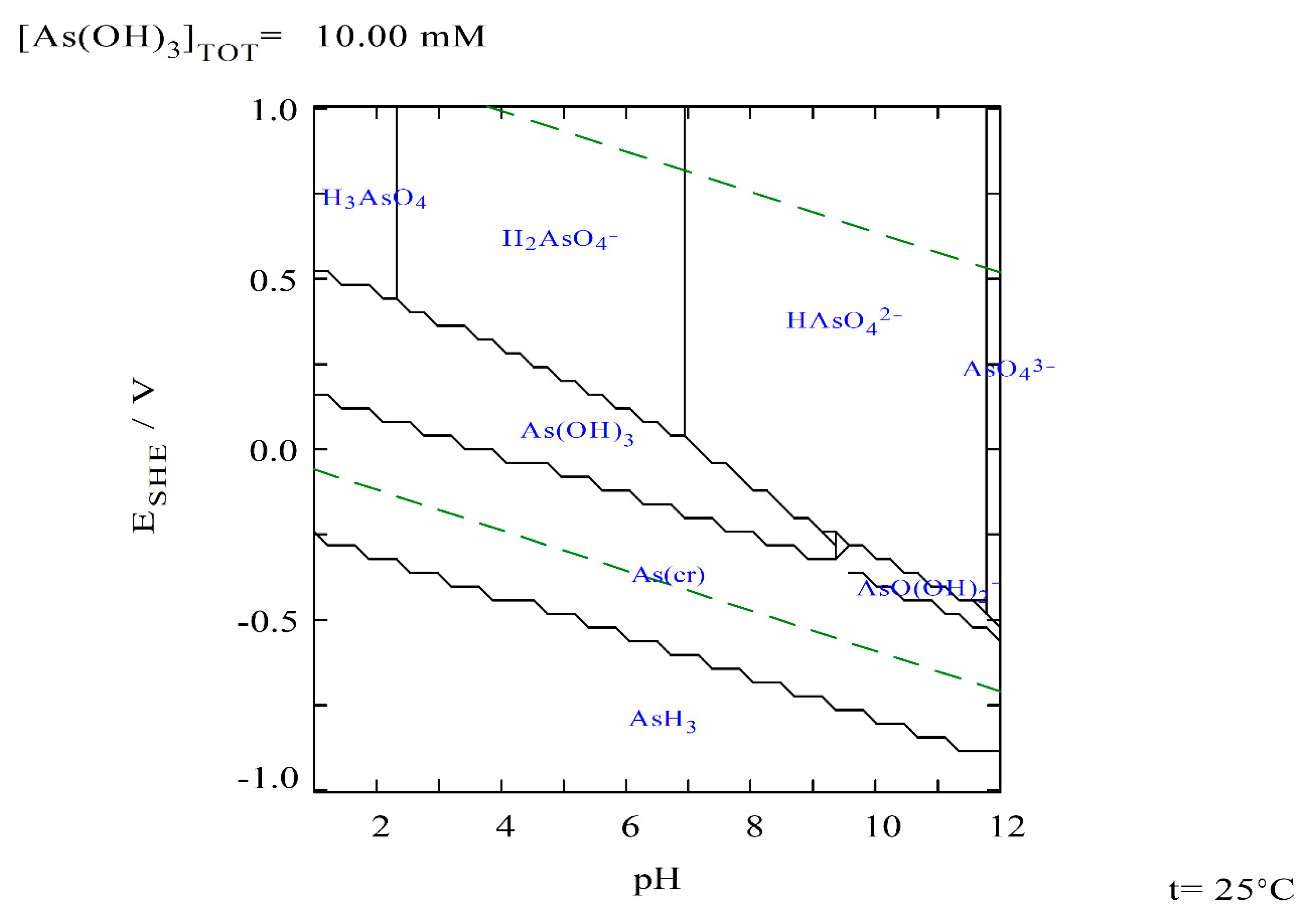
| Species | PO43− | SiO32− | SO42− | Ca2+ | Mg2+ | Cl− | Na+ | HCO3− | Fe | As (III) | As (V) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| mg/L | 1.3 | 19.5 | 8 | 61 | 8 | 125 | 138 | 275 | 0 | 0.20 | 0.20 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larroca, F.P.; Olschewski, E.S.; Quino-Favero, J.; Huamaní, J.R.; Castillo Sequera, J.L. Water Treatment Plant Prototype with pH Control Modeled on Fuzzy Logic for Removing Arsenic Using Fe(VI) and Fe(III). Water 2020, 12, 2834. https://doi.org/10.3390/w12102834
Larroca FP, Olschewski ES, Quino-Favero J, Huamaní JR, Castillo Sequera JL. Water Treatment Plant Prototype with pH Control Modeled on Fuzzy Logic for Removing Arsenic Using Fe(VI) and Fe(III). Water. 2020; 12(10):2834. https://doi.org/10.3390/w12102834
Chicago/Turabian StyleLarroca, Fabricio Paredes, Erich Saettone Olschewski, Javier Quino-Favero, Jimmy Rosales Huamaní, and José Luis Castillo Sequera. 2020. "Water Treatment Plant Prototype with pH Control Modeled on Fuzzy Logic for Removing Arsenic Using Fe(VI) and Fe(III)" Water 12, no. 10: 2834. https://doi.org/10.3390/w12102834
APA StyleLarroca, F. P., Olschewski, E. S., Quino-Favero, J., Huamaní, J. R., & Castillo Sequera, J. L. (2020). Water Treatment Plant Prototype with pH Control Modeled on Fuzzy Logic for Removing Arsenic Using Fe(VI) and Fe(III). Water, 12(10), 2834. https://doi.org/10.3390/w12102834







