Use of Larval Morphological Deformities in Chironomus plumosus (Chironomidae: Diptera) as an Indicator of Freshwater Environmental Contamination (Lake Trasimeno, Italy)
Abstract
:1. Introduction
2. Materials and Methods
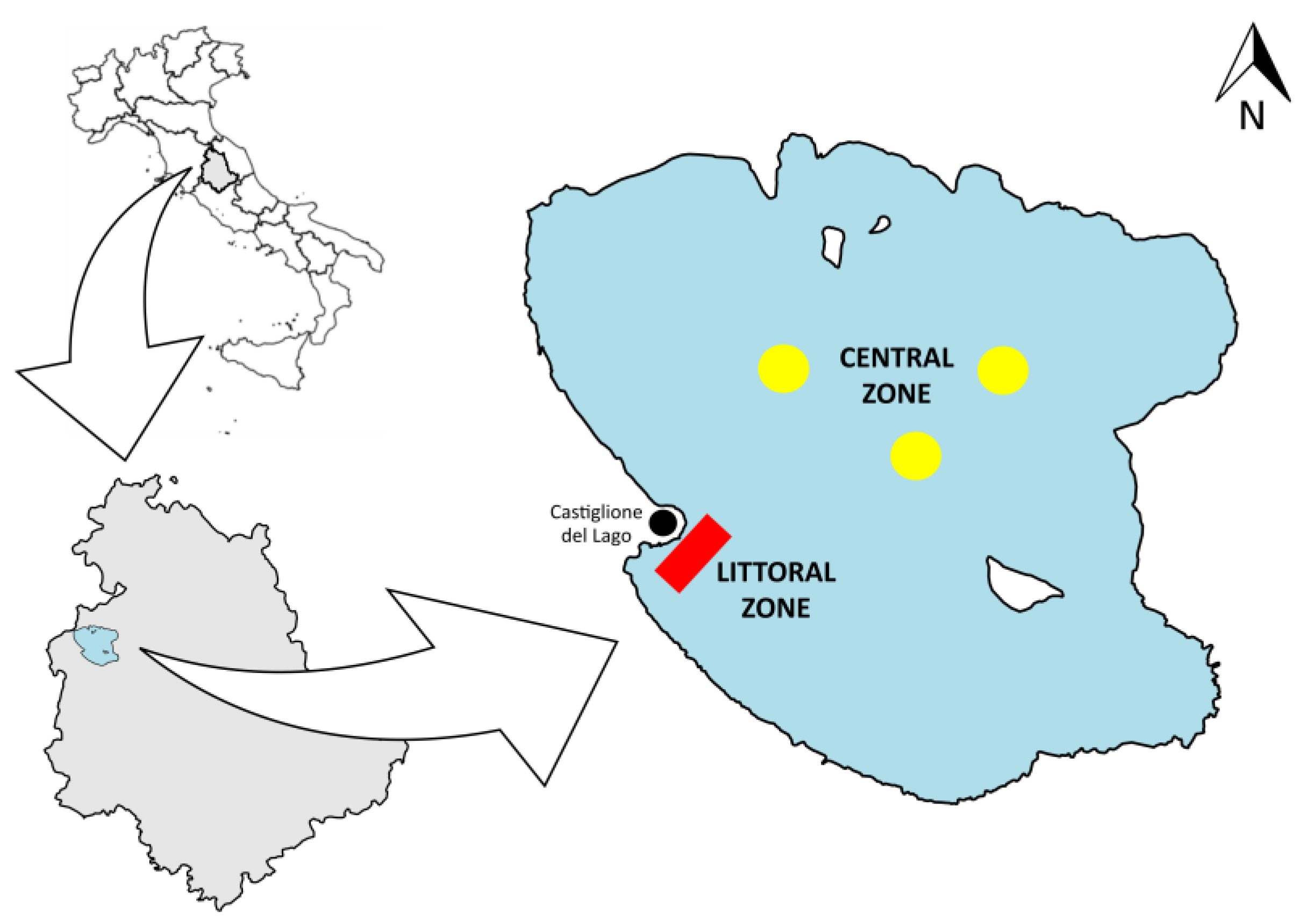
2.1. Study Area
2.2. Field Sampling Campaign
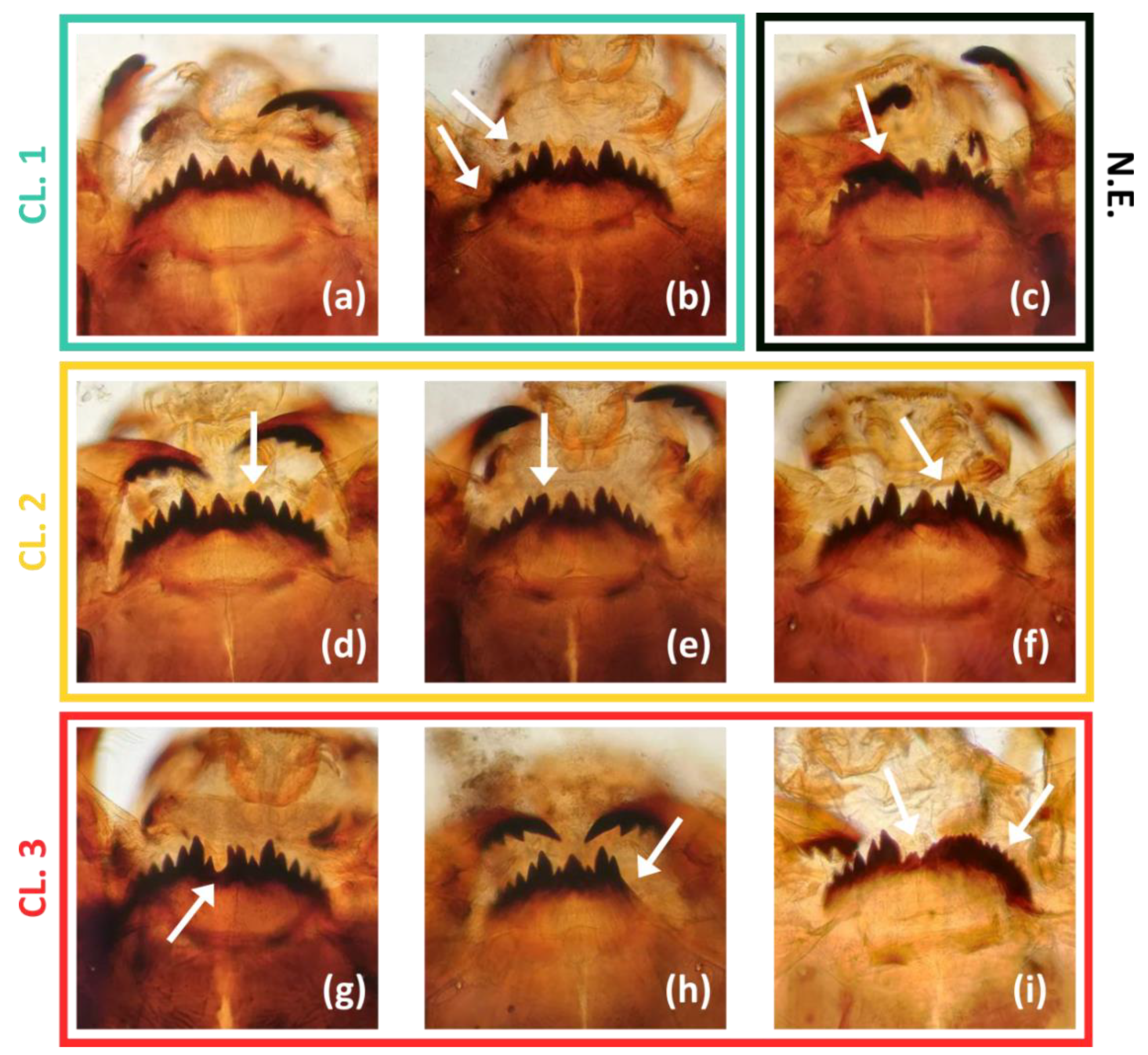
2.3. Mouthpart Deformities of Chironomus plumosus Larvae
2.4. Statistical Analysis
3. Results
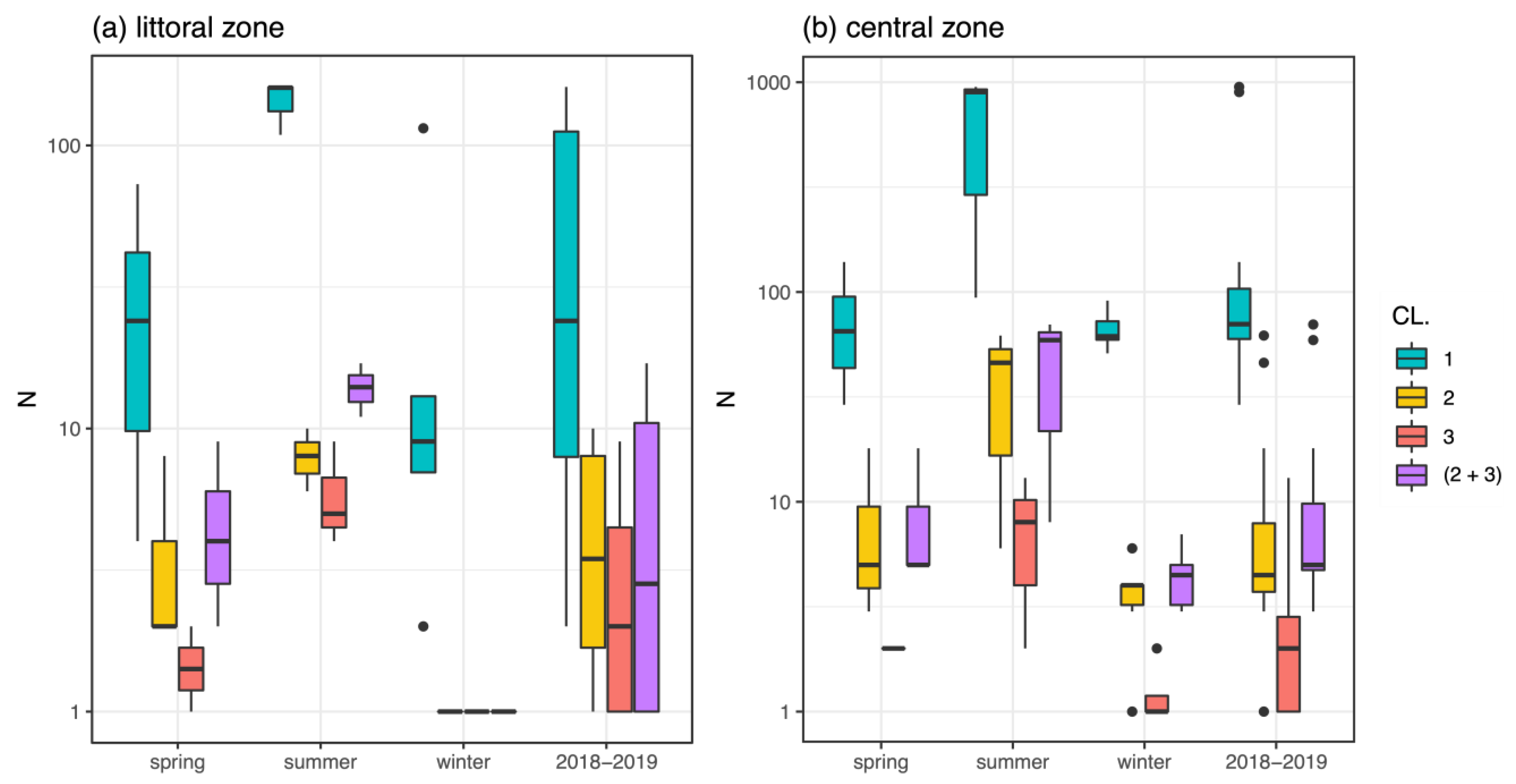
3.1. Macroinvertebrates and Chironomids
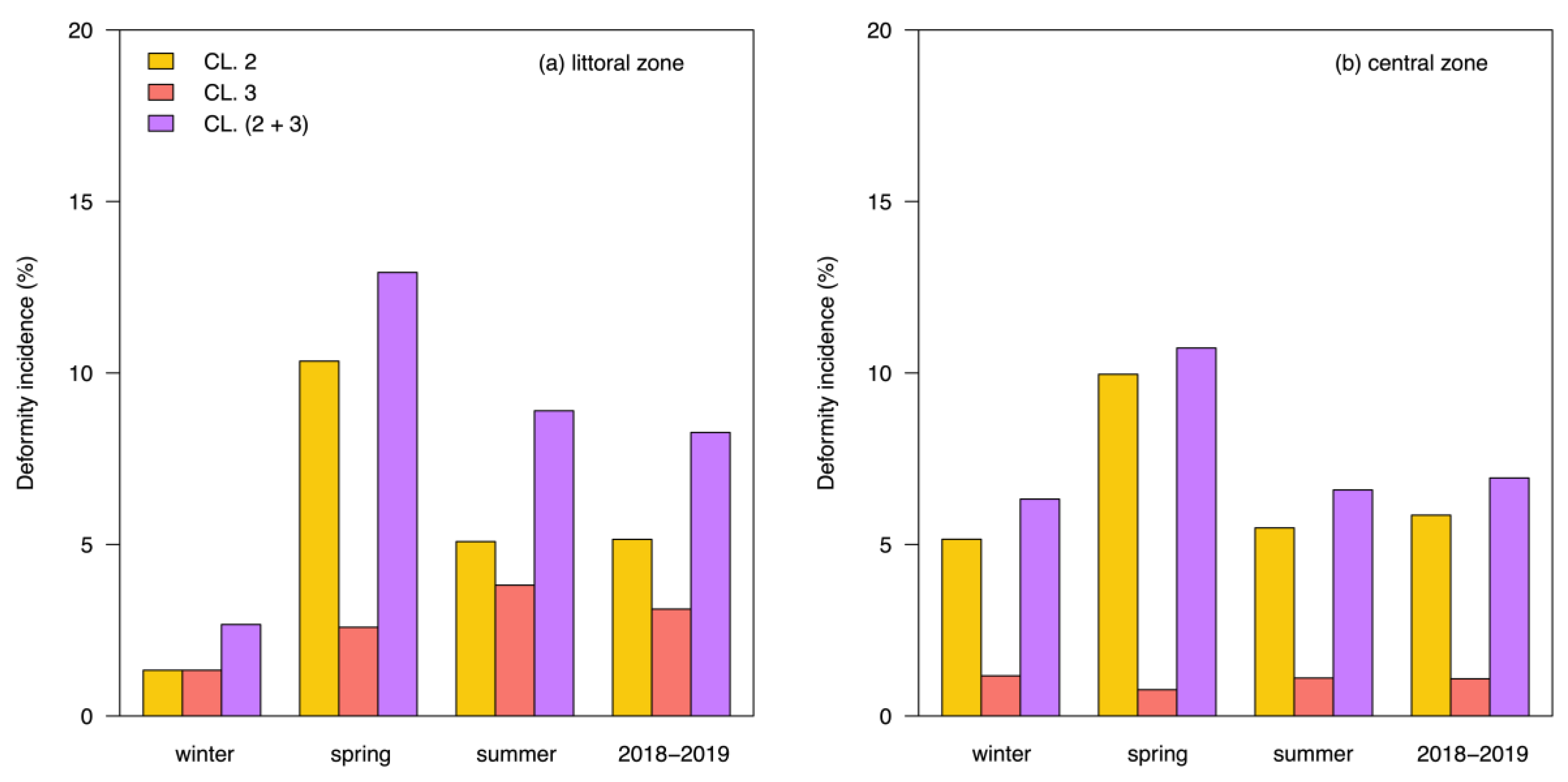
3.2. Mentum Deformity in Chironomus plumosus
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rosenberg, D.M.; Resh, V.H. Introduction to freshwater biomonitoring and benthic macroinvertebrates. In Freshwater Biomonitoring Benthic Macroinvertebrates; Chapman and Hall: New York, NY, USA, 1993. [Google Scholar]
- Maldonado, A.C.D.; Wendling, B. Manejo de ecossistemas aquáticos contaminados por Metais pesados. Revista Agropecuária Técnica 2009, 30, 21–32. [Google Scholar] [CrossRef]
- Goretti, E.; Pallottini, M.; Ricciarini, M.I.; Selvaggi, R.; Cappelletti, D. Heavy metals bioaccumulation in selected tissues of red swamp crayfish: An easy tool for monitoring environmental contamination levels. Sci. Total Environ. 2016, 559, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, A.C. Elaboration of chironomid deformities as bioindicators of toxic sediment stress: The potential application of mixture toxicity concepts. Ann. Zool. Fenn. 1995, 32, 265–285. [Google Scholar]
- Warwick, W.F. Morphological deformities in Chironomidae (Diptera) larvae as biological indicators of toxic stress. In Toxic Contaminants and Ecosystem Health; A Great Lakes Focus; Evans, M.S., Ed.; John Wiley and Sons: New York, NY, USA, 1988; pp. 281–320. [Google Scholar]
- Apostolico, F.; Vercillo, F.; La Porta, G.; Ragni, B. Long-term changes in diet and trophic niche of the European wildcat (Felis silvestris silvestris) in Italy. Mamm. Res. 2016, 61, 109–119. [Google Scholar] [CrossRef]
- Davutluoglu, O.I.; Seckin, G.; Ersu, C.B.; Ylmaz, T.; Sari, B. Heavy metal content and distribution in surface sediments of the Seyhan River, Turkey. J. Environ. Manag. 2011, 92, 2250–2259. [Google Scholar] [CrossRef]
- Goretti, E.; Pallottini, M.; Cenci Goga, B.T.; Selvaggi, R.; Petroselli, C.; Vercillo, F.; Cappelletti, D. Mustelids as bioindicators of the environmental contamination by heavy metals. Ecol. Indicat. 2018, 94, 320–327. [Google Scholar] [CrossRef]
- Goretti, E.; Pallottini, M.; Rossi, R.; La Porta, G.; Gardi, T.; Cenci Goga, B.T.; Elia, A.C.; Galletti, M.; Moroni, B.; Petroselli, C.; et al. Heavy metal bioaccumulation in honey bee matrix, an indicator to assess the contamination level in terrestrial environments. Environ. Pollut. 2019, 256, 113388. [Google Scholar] [CrossRef]
- Holt, M.S. Sources of chemical contaminants and routes into the freshwater environment. Food Chem. Toxicol. 2000, 38, 21–27. [Google Scholar] [CrossRef]
- Chapman, P.M. The sediment quality triad approach to determining pollution-induced degradation. Sci. Total Environ. 1990, 97, 815–825. [Google Scholar] [CrossRef]
- Cummins, K.W.; Wilzbach, M.A. Field Procedures for the Analysis of Functional Feeding Groups in Stream Ecosystems; Contribution 1611; Appalachian Environmental Laboratory, University of Maryland: Frostburg, MD, USA, 1985. [Google Scholar]
- Céréghino, R.; Park, Y.S.; Compin, A.; Lek, S. Predicting the species richness of aquatic insects in streams using a limited number of environmental variables. J. N. Am. Benthol. Soc. 2003, 22, 442–456. [Google Scholar] [CrossRef]
- Dolédec, S.; Statzner, B. Reponses of freshwater biota to human disturbances: Contribution of J-NABS to developments in ecological integrity assessments. J. N. Am. Benthol. Soc. 2010, 29, 286–311. [Google Scholar] [CrossRef]
- Pallottini, M.; Goretti, E.; Gaino, E.; Selvaggi, R.; Cappelletti, D.; Cereghino, R. Invertebrate diversity in relation to chemical pollution in an Umbrian stream system (Italy). C. R. Biol. 2015, 338, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Pallottini, M.; Cappelletti, D.; Fabrizi, E.; Gaino, E.; Goretti, E.; Selvaggi, R.; Cereghino, R. Macroinvertebrate functional trait responses to chemical pollution in agricultural-industrial landscapes. River Res. Appl. 2017, 33, 505–513. [Google Scholar] [CrossRef]
- Adler, P.H.; Courtney, G.W. Ecological and Societal Services of Aquatic Diptera. Insects 2019, 10, 70. [Google Scholar] [CrossRef] [Green Version]
- Armitage, P.D.; Pinder, L.C.; Cranston, P. The Chironomidae: Biology and Ecology of Non-Biting Midges; Springer Netherlands: Heidelberg, Germany, 1995. [Google Scholar]
- Goretti, E.; Coletti, A.; Di Veroli, A.; Di Giulio, A.M.; Gaino, E. Artificial light device for attracting pestiferous chironomids (Diptera): A case study at Lake Trasimeno (Central Italy). Ital. J. Zool. 2011, 78, 336–342. [Google Scholar] [CrossRef]
- OECD. Test No. 235: Chironomus sp., Acute Immobilisation Test; OECD Publishing: Paris, France, 2011. [Google Scholar] [CrossRef]
- Božanić, M.; Marković, Z.; Živić, M.; Dojčinovć, B.; Perić, A.; Stanković, M.; Živić, I. Mouthpart Deformities of Chironomus plumosus Larvae Caused by Increased Concentrations of Copper in Sediment from Carp Fish Pond. Turk. J. Fish. Aquat. Sci. 2018, 19, 251–259. [Google Scholar] [CrossRef]
- Di Veroli, A.; Goretti, E.; Leόn Paumen, M.; Kraak, M.H.S.; Admiraal, W. Mouthpart deformities in Chironomus riparius larvae exposed to toxicants. Environ. Pollut. 2012, 166, 212–217. [Google Scholar] [CrossRef]
- Martinez, E.A.; Moore, B.C.; Schaumloffel, J.; Dasgupta, N. Morphologic and Growth Responses in Chironomus tentans to Arsenic Exposure. Arch. Environ. Contam. Toxicol. 2006, 51, 529–536. [Google Scholar] [CrossRef]
- Gagliardi, B.S.; Long, S.M.; Pettigrove, V.J.; Griffin, P.C.; Hoffmann, A.A. A re-evaluation of chironomid deformities as an environmental stress response: Avoiding survivorship bias and testing noncontaminant biological factors. Environ. Toxicol. Chem. 2019, 38, 1658–1667. [Google Scholar] [CrossRef]
- Corbi, J.J.; Bernegossi, A.C.; Moura, L.; Felipe, M.C.; Issa, C.G.; Silva, M.R.L.; Gorni, G.R. Chironomus sancticaroli (Diptera, Chironomidae) as a Sensitive Test Species: Can We Rely on Its Use After Repeated Generations, Under Laboratory Conditions? Bull. Environ. Contam. Toxicol. 2019, 103, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Saether, O.A. A survey of the bottom fauna of the Okanagan valley, British Columbia. Can. Fish. Mar. Serv. Techn. Rep. 1970, 196, 1–29. [Google Scholar]
- Hamilton, A.L.; Saether, O.A. The occurrence of characteristic deformities in the chironomid larvae of several Canadian lakes. Can. Entomol. 1971, 103, 363–368. [Google Scholar] [CrossRef]
- Hare, L.; Carter, J.C.H. The distribution of Chironomus (s.s.)? Cucini (salinarius group) larvae (Diptera: Chironomidae) in Parry Sound, Georgian Bay, with particular reference to structural deformities. Can. J. Zool. 1976, 54, 2129–2134. [Google Scholar] [CrossRef]
- Janssens de Bisthoven, L.; Vermeulen, A.; Ollevier, F. Experimental Induction of Morphological Deformities in Chironomus riparius Larvae by Chronic Exposure to Copper and Lead. Arch. Environ. Contam. Toxicol. 1998, 35, 249–256. [Google Scholar] [CrossRef]
- Reynolds, S.K.; Ferrington, L.C. Differential Morphological Responses of Chironomid Larvae to Severe Heavy Metal Exposure (Diptera: Chironomidae). J. Kans. Entomol. Soc. 2002, 75, 172–184. [Google Scholar]
- Martinez, E.A.; Moore, B.C.; Schaumloffel, J.; Dasgupta, N. Morphological abnormalities in Chironomus tentans exposed to cadmium and copper-spiked sediments. Ecotoxicol. Environ. Saf. 2003, 55, 204–212. [Google Scholar] [CrossRef]
- Péry, A.R.R.; Mons, R.; Garric, J. Energy-based modeling to study population growth rate and production for the midge Chironomus riparius in ecotoxicological risk assessment. Ecotoxicology 2004, 13, 647–656. [Google Scholar] [CrossRef]
- Ebau, W.; Rawi, C.S.M.; Din, Z.; Al-Shami, S.A. Toxicity of cadmium and lead on tropical midge larvae, Chironomus kiiensis Tokunaga and Chironomus javanus Kieffer (Diptera: Chironomidae). Asian Pac. J. Trop. Biomed. 2012, 2, 631–634. [Google Scholar] [CrossRef] [Green Version]
- Gagliardi, B.; Pettigrove, V. Removal of intensive agriculture from the landscape improves aquatic ecosystem health. Agric. Ecosyst. Environ. 2013, 176, 1–8. [Google Scholar] [CrossRef]
- Grebenjuk, L.P.; Tomilina, I.I. Morphological Deformations of Hard Chitinized Mouthpart Structures in Larvae of the Genus Chironomus (Diptera, Chironomidae) as the Index of Organic Pollution in Freshwater Ecosystems. Inland Water Biol. 2014, 7, 273–285. [Google Scholar] [CrossRef]
- Beneberu, G.; Mengistou, S. Head capsule deformities in Chironomus spp. (Diptera: Chironomidae) as indicator of environmental stress in Sebeta River, Ethiopia. Afr. J. Ecol. 2014, 53, 268–277. [Google Scholar] [CrossRef]
- Arimoro, F.O.; Odume, O.N.; Meme, F.K. Environmental drivers of head capsule deformities in Chironomus spp. (Diptera: Chironomidae) in a stream in north central Nigeria. Zool. Ecol. 2015, 25, 70–76. [Google Scholar] [CrossRef]
- Odume, O.N.; Palmer, C.G.; Arimoro, F.O.; Mensaha, P.K. Chironomid assemblage structure and morphological response to pollution in an effluent-impacted river, Eastern Cape, South Africa. Ecol. Indic. 2016, 67, 391–402. [Google Scholar] [CrossRef]
- Arimoro, F.O.; Auta, Y.I.; Odume, O.N.; Keke, U.N.; Mohammed, A.Z. Mouthpart deformities in Chironomidae (Diptera) as bioindicators of heavy metals pollution in Shiroro Lake, Niger State, Nigeria. Ecotoxicol. Environ. Saf. 2018, 149, 96–100. [Google Scholar] [CrossRef]
- Akyildiz, G.K.; Bakir, R.; Polat, S.; Duran, M. Mentum Deformities of Chironomid Larvae as an Indicator of Environmental Stress in Büyük Menderes River, Turkey. Inland Water Biol. 2018, 11, 515–522. [Google Scholar] [CrossRef]
- Deliberalli, W.; Cansian, R.L.; Pereira, A.A.M.; Loureiro, R.C.; Hepp, L.U.; Restello, R.M. The effects of heavy metals on the incidence of morphological deformities in Chironomidae (Diptera). Zoologia 2018, 35. [Google Scholar] [CrossRef] [Green Version]
- Di Veroli, A.; Selvaggi, R.; Pellegrino, R.M.; Goretti, E. Sediment toxicity and deformities of chironomid larvae in Lake Piediluco (Central Italy). Chemosphere 2010, 79, 33–39. [Google Scholar] [CrossRef]
- Di Veroli, A.; Selvaggi, R.; Goretti, E. Chironomid mouthpart deformities as indicator of environmental quality: A case study in Lake Trasimeno (Italy). J. Environ. Monit. 2012, 14, 1473–1478. [Google Scholar] [CrossRef]
- Di Veroli, A.; Santoro, F.; Pallottini, M.; Selvaggi, R.; Scardazza, F.; Cappelletti, D.; Goretti, E. Deformities of chironomid larvae and heavy metal pollution: From laboratory to field studies. Chemosphere 2014, 112, 9–17. [Google Scholar] [CrossRef]
- Savić-Zdravković, D.; Jovanović, B.; Ðurđević, A.; Stojković-Piperac, M.; Savić, A.; Vidmar, J.; Milošević, D. An environmentally relevant concentration of titanium dioxide (TiO2) nanoparticles induces morphological changes in the mouthparts of Chironomus tentans. Chemosphere 2018, 211, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Tomilina, I.I.; Grebenjuk, L.P. Induction of Deformities of the Hard-Chitinized Mouthpart Structures of Larvae Chironomus riparius Meigen under Various Contents of Persistent Organic Substances in Bottom Sediments. Inland Water Biol. 2019, 12, 346–355. [Google Scholar] [CrossRef]
- Park, K.; Kwak, I.S. Disrupting effects of antibiotic sulfathiazole on developmental process during sensitive life-cycle stage of Chironomus riparius. Chemosphere 2018, 190, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Pallottini, M.; Goretti, E.; Selvaggi, R.; Cappelletti, D.; Dedieu, N.; Cereghino, R. An efficient semi-quantitative macroinvertebrate multimetric index forthe assessment of water and sediment contamination in streams. Inland Waters 2017, 7, 314–322. [Google Scholar] [CrossRef]
- Arambourou, H.; Planelló, R.; Llorente, L.; Fuertes, I.; Barata, C.; Delorme, N.; Noury, P.; Herrero, Ó.; Villeneuve, A.; Bonnineau, C. Chironomus riparius exposure to field-collected contaminated sediments: From subcellular effect to whole-organism response. Sci. Total Environ. 2019, 671, 874–882. [Google Scholar] [CrossRef]
- He, H.; Xi, G.; Lu, X. Molecular cloning, characterization, and expression analysis of an ecdysone receptor homolog in Teleogryllus emma (Orthoptera: Gryllidae). J. Insect Sci. 2015, 15. [Google Scholar] [CrossRef] [Green Version]
- Vermeulen, A.C.; Liberloo, G.; Dumont, P.; Ollevier, F.; Goddeeris, B.R. Exposure of Chironomus riparius larvae (Diptera) to lead, mercury, and β-sitosterol: Effects on mouthpart deformation and moulting. Chemosphere 2000, 41, 1581–1591. [Google Scholar] [CrossRef]
- Marchegiano, M.; Francke, A.; Gliozzi, E.; Wagner, B.; Arizteguia, D. High-resolution palaeohydrological reconstruction of central Italy during the Holocene. Holocene 2018, 29, 481–492. [Google Scholar] [CrossRef]
- Frondini, F.; Dragoni, W.; Morgantini, N.; Donnini, M.; Cardellini, C.; Caliro, S.; Melillo, M.; Chiodini, G. An Endorheic Lake in a Changing Climate: Geochemical Investigations at Lake Trasimeno (Italy). Water 2019, 11, 1319. [Google Scholar] [CrossRef] [Green Version]
- Ludovisi, A.; Gaino, E. Meteorological and water quality changes in Lake Trasimeno (Umbria, Italy) during the last fifty years. J. Limnol. 2010, 69, 174–188. [Google Scholar] [CrossRef] [Green Version]
- Marchegiano, M.; Francke, A.; Gliozzi, E.; Arizteguia, D. Arid and humid phases in central Italy during the Late Pleistocene revealed by the Lake Trasimeno ostracod record. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2018, 490, 55–69. [Google Scholar] [CrossRef]
- Mancinelli, G.; Papadia, P.; Ludovisi, A.; Migoni, D.; Bardelli, R.; Fanizzi, F.P.; Vizzini, S. Beyond the mean: A comparison of trace- and macroelement correlation profiles of two lacustrine populations of the crayfish Procambarus clarkii. Sci. Total Environ. 2018, 624, 1455–1466. [Google Scholar] [CrossRef] [PubMed]
- Tachet, H.; Richoux, P.; Bournaud, M.; Usseglio-Polatera, P. Invertébrés d’eau douce. Systématique, biologie, écologie; CNRS Éditions: Paris, France, 2010. [Google Scholar]
- Ferrarese, U.; Rossaro, B. Chironomidi, 1. In Guida Per Il Riconoscimento Delle Specie Animali Delle Acque Interne Italiane; CNR AQ/1/129; CNR: Roma, Italy, 1981. [Google Scholar]
- Rossaro, B. Chironomidi, 2. In Guida Per Il Riconoscimento Delle Specie Animali Delle Acque Interne Italiane; CNR AQ/1/171; CNR: Roma, Italy, 1982. [Google Scholar]
- Ferrarese, U. Chironomidi, 3. In Guida Per Il Riconoscimento Delle Specie Animali Delle Acque Interne Italiane; CNR AQ/1/204; CNR: Roma, Italy, 1983. [Google Scholar]
- Nocentini, A.M. Chironomidi, 4. In Guida Per Il Riconoscimento Delle Specie Animali Delle Acque Interne Italiane; CNR AQ/1/233; CNR: Roma, Italy, 1985. [Google Scholar]
- Janssens De Bisthoven, L.; Yts, P.; Goddeeris, B.; Ollevier, F. Sublethal parameters in morphologically deformed Chironomus larvae: Clues to understand their bioindicator value. Freshw. Biol. 1998, 39, 179–191. [Google Scholar] [CrossRef]
- Madden, C.P.; Austin, A.D.; Sutter, P.J. Pollution monitoring using chironomid larvae: What is a deformity? In Chironomids from Genes to Ecosystems; Craston, P., Ed.; CSRIO Publication: East Melbourne, Australia, 1995; pp. 89–94. [Google Scholar]
- Nazarova, L.B.; Riss, H.W.; Kahlheber, A.; Werding, B. Some observations of buccal deformities in chironomid larvae (Diptera: Chironomidae) from the Ciénaga Grande de Santa Marta, Colombia. Caldasia 2004, 26, 275–290. [Google Scholar]
- Cao, Y.; Zhang, E.; Tang, H.; Langdon, P.; Ning, D.; Zheng, W. Combined effects of nutrients and trace metals on chironomid composition and morphology in a heavily polluted lake in central China since the early 20th century. Hydrobiologia 2016, 779, 147–159. [Google Scholar] [CrossRef] [Green Version]
- Newcombe, R.G. Interval estimation for the difference between independent proportions: Comparison of eleven methods. Stat. Med. 1998, 17, 873–890. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 13 December 2019).
- Warwick, W.F. Paleolimnology of the Bay of Quinte, Lake Ontario: 2800 years of cultural influence. Can. Bull. Fish. Aquat. Sci. 1980, 206, 1–118. [Google Scholar]
- Wiederholm, T. Incidence of deformed chironomid larvae (Diptera Chironomidae) in Swedish Lakes. Hydrobiologia 1984, 109, 243–249. [Google Scholar] [CrossRef]
- Warwick, W.F. Morphological abnormalities in Chironomidae (Diptera) larvae as measures of toxic stress in freshwater ecosystems: Indexing antennal deformities in Chironomus Meigen. Can. J. Fish. Aquat. Sci. 1985, 42, 1881–1914. [Google Scholar] [CrossRef]
- Bird, G.A. Use of chironomid deformities to assess environmental degradation in the Yamaska River, Quebec. Environ. Monit. Assess. 1994, 30, 163–175. [Google Scholar] [CrossRef]
- Vogt, C.; Langer-Jaesrich, M.; Elsässer, O.; Schmitt, C.; Van Dongen, S.; Köhler, H.R.; Oehlmann, J.; Nowak. Effects of inbreeding on mouthpart deformities of Chironomus riparius under sublethal pesticide exposure. Environ. Toxicol. Chem. 2013, 32, 423–425. [Google Scholar] [CrossRef] [PubMed]
- Arambourou, H.; Beisel, J.N.; Branchu, P.; Debat, V. Exposure to sediments from polluted rivers has limited phenotypic effects on larvae and adults of Chironomus riparius. Sci. Total Environ. 2014, 484, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Salmelin, J.; Vuori, K.M.; Hämäläinen, H. Inconsistency in the analysis of morphological deformities in Chironomidae (Insecta: Diptera) larvae. Environ. Toxicol. Chem. 2015, 34, 1891–1898. [Google Scholar] [CrossRef]
- Salmelin, J.; Karjalainen, A.K.; Hämäläinen, H.; Leppänen, M.T.; Kiviranta, H.; Kukkonen, J.V.K.; Vuori, K.M. Biological responses of midge (Chironomus riparius) and lamprey (Lampetra fluviatilis) larvae in ecotoxicity assessment of PCDD/F-, PCB- and Hg-contaminated river sediments. Environ. Sci. Pollut. Res. 2016, 23, 18379–18393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gagliardi, B.S.; Pettigrove, V.J.; Long, S.M.; Hoffmann, A.A. A Meta-Analysis Evaluating the Relationship between Aquatic Contaminants and Chironomid Larval Deformities in Laboratory Studies. Environ. Sci. Technol. 2016, 50, 12903–12911. [Google Scholar] [CrossRef] [PubMed]
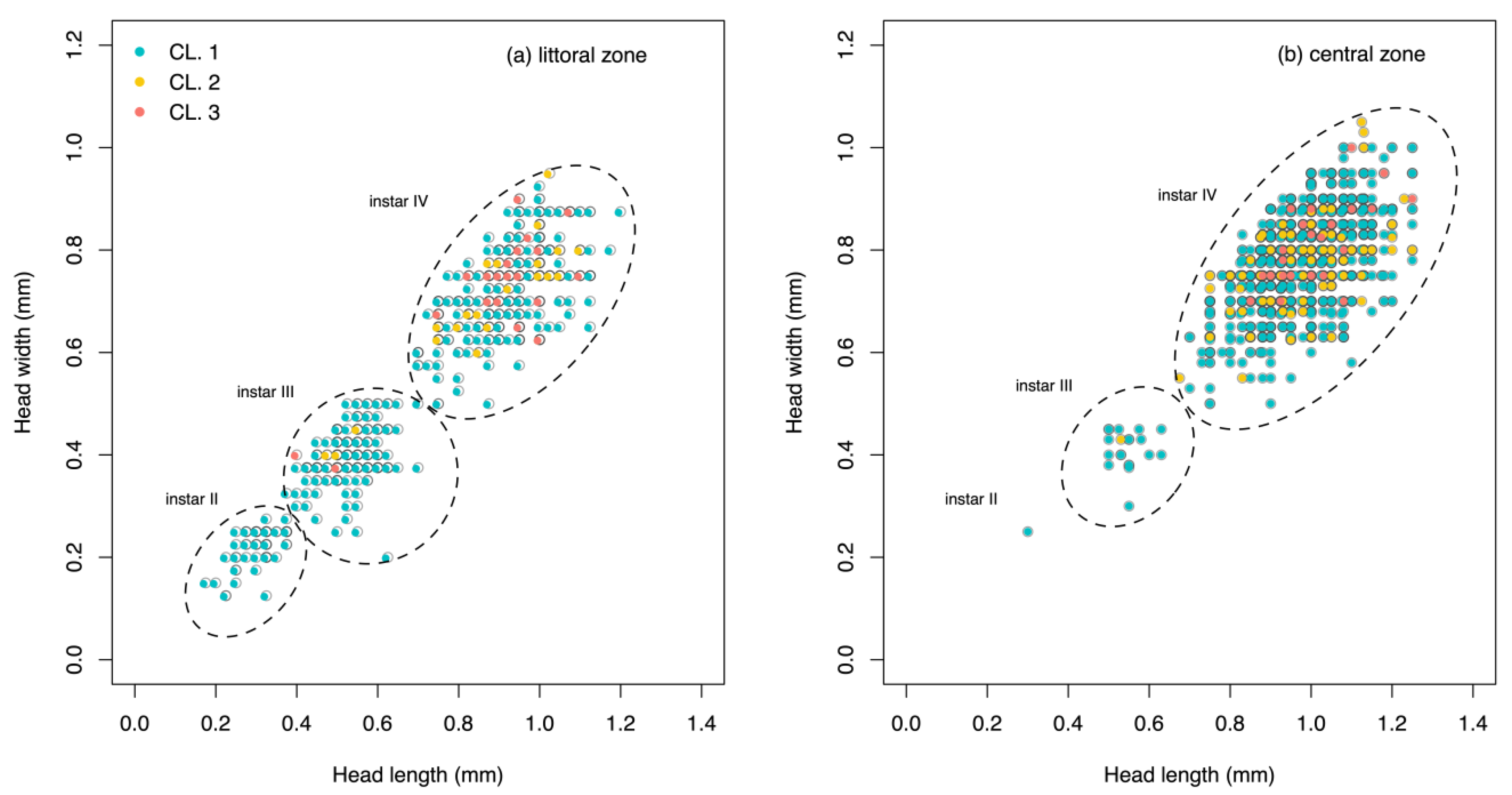
| (a) Littoral Zone | |||||||||
| Date | C. plumosus | C. plumosus | Chironomidae | Chironomidae | Naididae | Naididae | Other Taxa | Other Taxa | Macroinvertebrates |
| (N) | (% of Chironomidae) | (ind. m−2) | (%) | (ind. m−2) | (%) | (ind. m−2) | (%) | (ind. m−2) | |
| 11/05/2018 | 2 | 2.94 | 183 | 9.24 | 1793 | 90.40 | 7 | 0.36 | 1983 |
| 18/05/2018 | 6 | 4.44 | 469 | 28.08 | 1188 | 71.17 | 12 | 0.74 | 1669 |
| 25/05/2018 | 4 | 2.26 | 646 | 35.67 | 1154 | 63.68 | 12 | 0.65 | 1812 |
| 01/06/2018 | 16 | 11.85 | 518 | 35.13 | 935 | 63.36 | 22 | 1.51 | 1476 |
| 14/06/2018 | 7 | 3.87 | 599 | 42.62 | 578 | 41.12 | 228 | 16.26 | 1405 |
| 22/06/2018 | 2 | 1.60 | 426 | 39.29 | 536 | 49.41 | 122 | 11.29 | 1084 |
| 28/06/2018 | 6 | 8.33 | 239 | 23.76 | 706 | 70.23 | 60 | 6.01 | 1006 |
| 11/07/2018 | 16 | 23.53 | 242 | 23.29 | 670 | 64.56 | 126 | 12.15 | 1037 |
| 19/07/2018 | 12 | 29.27 | 122 | 9.51 | 1099 | 86.02 | 57 | 4.47 | 1277 |
| 25/07/2018 | 8 | 19.05 | 152 | 14.32 | 814 | 76.98 | 92 | 8.70 | 1058 |
| 01/08/2018 | 35 | 51.47 | 242 | 18.11 | 993 | 74.41 | 100 | 7.48 | 1334 |
| 09/08/2018 | 28 | 20.90 | 452 | 26.55 | 1182 | 69.48 | 68 | 3.97 | 1702 |
| 22/08/2018 | 21 | 24.42 | 376 | 20.10 | 1451 | 77.51 | 45 | 2.39 | 1872 |
| 03/09/2018 | 72 | 46.15 | 490 | 27.51 | 1250 | 70.20 | 41 | 2.29 | 1781 |
| 18/09/2018 | 102 | 76.69 | 439 | 35.76 | 743 | 60.60 | 45 | 3.64 | 1226 |
| 27/09/2018 | 12 | 37.50 | 179 | 13.77 | 1103 | 85.02 | 16 | 1.21 | 1297 |
| 10/10/2018 | 15 | 12.93 | 567 | 25.65 | 1641 | 74.23 | 3 | 0.12 | 2211 |
| 17/10/2018 | 33 | 13.25 | 1079 | 34.09 | 2066 | 65.27 | 20 | 0.64 | 3166 |
| 24/10/2018 | 75 | 35.55 | 843 | 47.95 | 903 | 51.34 | 12 | 0.71 | 1758 |
| 06/11/2018 | 8 | 6.11 | 457 | 48.29 | 490 | 51.71 | 0 | 0 | 947 |
| 04/12/2018 | 9 | 6.47 | 472 | 58.18 | 339 | 41.82 | 0 | 0 | 811 |
| 16/01/2019 | 3 | 1.75 | 533 | 53.00 | 462 | 45.95 | 11 | 1.04 | 1006 |
| 12/02/2019 | 16 | 10.53 | 505 | 51.70 | 472 | 48.30 | 0 | 0.00 | 977 |
| 15/03/2019 | 0 | 0 | 258 | 34.90 | 469 | 63.42 | 12 | 1.68 | 739 |
| 04/04/2019 | 6 | 4.17 | 463 | 32.93 | 944 | 67.07 | 0 | 0 | 1407 |
| 02/05/2019 | 13 | 3.75 | 1008 | 41.73 | 1375 | 56.91 | 33 | 1.36 | 2415 |
| 21/05/2019 | 7 | 7.95 | 304 | 25.70 | 829 | 70.19 | 48 | 4.10 | 1181 |
| 04/06/2019 | 16 | 7.69 | 722 | 32.59 | 1409 | 63.61 | 84 | 3.81 | 2215 |
| 18/06/2019 | 33 | 17.19 | 623 | 38.20 | 952 | 58.45 | 55 | 3.35 | 1629 |
| 27/06/2019 | 11 | 14.47 | 252 | 42.67 | 328 | 55.56 | 11 | 1.78 | 591 |
| 09/07/2019 | 68 | 83.95 | 262 | 48.66 | 262 | 48.66 | 14 | 2.67 | 539 |
| 23/07/2019 | 24 | 55.81 | 130 | 13.97 | 760 | 81.64 | 41 | 4.38 | 931 |
| 06/08/2019 | 72 | 40.22 | 656 | 24.74 | 1885 | 71.13 | 110 | 4.14 | 2651 |
| 22/08/2019 | 38 | 17.33 | 880 | 15.13 | 4655 | 80.01 | 283 | 4.86 | 5818 |
| Min | 0 | 0.00 | 122 | 9.24 | 262 | 41.12 | 0 | 0.00 | 539 |
| Median | 14 | 13.09 | 460 | 32.76 | 939 | 64.91 | 37 | 2.34 | 1369 |
| Mean | 23 | 20.69 | 464 | 31.55 | 1072 | 64.98 | 53 | 3.46 | 1589 |
| SD | 25 | 21.41 | 246 | 13.17 | 782 | 12.90 | 64 | 3.83 | 955 |
| Max | 102 | 83.95 | 1079 | 58.18 | 4655 | 90.40 | 283 | 16.26 | 5818 |
| (b) Central Zone | |||||||||
| Date | C. plumosus | C. plumosus | Chironomidae | Chironomidae | Naididae | Naididae | Other Taxa | Other Taxa | Macroinvertebrates |
| (N) | (% of Chironomidae) | (ind. m−2) | (%) | (ind. m−2) | (%) | (ind. m−2) | (%) | (ind. m−2) | |
| 29/05/2018 | 107 | 81.68 | 252 | 16.76 | 1250 | 83.24 | 0 | 0 | 1502 |
| 18/07/2018 | 396 | 98.02 | 685 | 75.69 | 220 | 24.31 | 0 | 0 | 905 |
| 08/08/2018 | 274 | 90.43 | 577 | 50.96 | 548 | 48.45 | 7 | 0.59 | 1132 |
| 22/08/2018 | 274 | 89.54 | 542 | 55.75 | 430 | 44.25 | 0 | 0 | 972 |
| 21/09/2018 | 117 | 100 | 202 | 16.40 | 1028 | 83.60 | 0 | 0 | 1230 |
| 16/10/2018 | 68 | 100 | 120 | 21.24 | 445 | 78.76 | 0 | 0 | 565 |
| 28/11/2018 | 66 | 100 | 113 | 12.88 | 767 | 87.12 | 0 | 0 | 880 |
| 20/12/2018 | 107 | 98.17 | 208 | 22.12 | 732 | 77.70 | 2 | 0.18 | 942 |
| 31/01/2019 | 82 | 100 | 143 | 22.34 | 498 | 77.66 | 0 | 0 | 642 |
| 21/02/2019 | 61 | 96.83 | 112 | 27.57 | 293 | 72.43 | 0 | 0 | 405 |
| 08/03/2019 | 86 | 100 | 145 | 27.44 | 383 | 72.56 | 0 | 0 | 528 |
| 12/04/2019 | 38 | 97.44 | 65 | 25.49 | 190 | 74.51 | 0 | 0 | 255 |
| 17/05/2019 | 69 | 100 | 123 | 28.57 | 308 | 71.43 | 0 | 0 | 432 |
| 13/06/2019 | 78 | 100 | 145 | 21.22 | 538 | 78.78 | 0 | 0 | 683 |
| 11/07/2019 | 74 | 98.67 | 137 | 37.61 | 225 | 61.93 | 2 | 0.46 | 363 |
| 31/07/2019 | 598 | 99.50 | 1115 | 37.54 | 1853 | 62.40 | 2 | 0 | 2970 |
| 08/08/2019 | 567 | 100 | 1008 | 43.94 | 1287 | 56.06 | 0 | 0 | 2295 |
| Min | 38 | 81.68 | 65 | 12.88 | 190 | 24.31 | 0 | 0.00 | 255 |
| Median | 86 | 99.50 | 145 | 27.44 | 498 | 72.56 | 0 | 0.00 | 880 |
| Mean | 180 | 97.07 | 335 | 31.97 | 647 | 67.95 | 1 | 0.08 | 982 |
| SD | 180 | 5.11 | 330 | 16.57 | 461 | 16.62 | 2 | 0.18 | 715 |
| Max | 598 | 100.00 | 1115 | 75.69 | 1853 | 87.12 | 7 | 0.59 | 2970 |
| (a) Littoral Zone | (b) Central Zone | (c) Lake Trasimeno | ||||||
|---|---|---|---|---|---|---|---|---|
| CLASS | N | % | CLASS | N | % | CLASS | N | % |
| CL. 1 | 676 | 91.72 | CL. 1 | 2575 | 93.1 | CL. 1 | 3251 | 92.78 |
| CL. 2 | 38 | 5.16 | CL. 2 | 162 | 5.85 | CL. 2 | 200 | 5.71 |
| CL. 3 | 23 | 3.12 | CL. 3 | 30 | 1.08 | CL. 3 | 53 | 1.51 |
| CL. (2 + 3) | 61 | 8.28 | CL. (2 + 3) | 192 | 6.94 | CL. (2 + 3) | 253 | 7.22 |
| (a) Littoral Zone | (b) Central Zone | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Head Lenght (mm) | Head Width (mm) | Head Lenght (mm) | Head Width (mm) | ||||||||||||||
| CL | 1 | 2 | 3 | 2 + 3 | 1 | 2 | 3 | 2 + 3 | 1 | 2 | 3 | 2 + 3 | 1 | 2 | 3 | 2 + 3 | |
| All Instars | N | 676 | 38 | 23 | 61 | 676 | 38 | 23 | 61 | 2575 | 162 | 30 | 192 | 2575 | 162 | 30 | 192 |
| Min | 0.18 | 0.48 | 0.40 | 0.40 | 0.13 | 0.38 | 0.38 | 0.38 | 0.30 | 0.53 | 0.85 | 0.53 | 0.25 | 0.43 | 0.70 | 0.43 | |
| Median | 0.83 | 0.91 | 0.95 | 0.93 | 0.65 | 0.75 | 0.75 | 0.75 | 1.00 | 1.00 | 1.00 | 1.00 | 0.78 | 0.78 | 0.79 | 0.78 | |
| Mean | 0.75 | 0.88 | 0.90 | 0.89 | 0.58 | 0.69 | 0.72 | 0.70 | 0.98 | 0.98 | 1.00 | 0.98 | 0.78 | 0.78 | 0.80 | 0.78 | |
| SD | 0.24 | 0.17 | 0.16 | 0.17 | 0.20 | 0.13 | 0.12 | 0.13 | 0.09 | 0.10 | 0.09 | 0.10 | 0.08 | 0.08 | 0.08 | 0.08 | |
| Max | 1.20 | 1.10 | 1.10 | 1.10 | 0.93 | 0.95 | 0.90 | 0.95 | 1.25 | 1.25 | 1.25 | 1.25 | 1.00 | 1.05 | 1.00 | 1.05 | |
| Instar IV | N | 402 | 33 | 21 | 54 | 402 | 33 | 21 | 54 | 2555 | 161 | 30 | 191 | 2555 | 161 | 30 | 191 |
| Min | 0.70 | 0.75 | 0.75 | 0.75 | 0.50 | 0.60 | 0.63 | 0.60 | 0.70 | 0.68 | 0.85 | 0.68 | 0.50 | 0.55 | 0.70 | 0.55 | |
| Median | 0.93 | 0.93 | 0.95 | 0.95 | 0.75 | 0.75 | 0.75 | 0.75 | 1.00 | 1.00 | 1.00 | 1.00 | 0.78 | 0.78 | 0.79 | 0.78 | |
| Mean | 0.93 | 0.93 | 0.95 | 0.94 | 0.73 | 0.73 | 0.75 | 0.74 | 0.98 | 0.98 | 1.00 | 0.98 | 0.78 | 0.78 | 0.80 | 0.79 | |
| SD | 0.10 | 0.09 | 0.08 | 0.08 | 0.08 | 0.07 | 0.07 | 0.07 | 0.08 | 0.09 | 0.09 | 0.09 | 0.07 | 0.07 | 0.08 | 0.07 | |
| Max | 1.20 | 1.10 | 1.10 | 1.10 | 0.93 | 0.95 | 0.90 | 0.95 | 1.25 | 1.25 | 1.25 | 1.25 | 1.00 | 1.05 | 1.00 | 1.05 | |
| Instar III | N | 214 | 5 | 2 | 7 | 214 | 5 | 2 | 7 | 19 | 1 | - | 1 | 19 | 1 | - | 1 |
| Min | 0.40 | 0.48 | 0.40 | 0.40 | 0.20 | 0.38 | 0.38 | 0.38 | 0.50 | 0.53 | - | 0.53 | 0.30 | 0.43 | - | 0.43 | |
| Median | 0.53 | 0.50 | 0.45 | 0.50 | 0.40 | 0.40 | 0.39 | 0.40 | 0.55 | 0.53 | - | 0.53 | 0.40 | 0.43 | - | 0.43 | |
| Mean | 0.53 | 0.51 | 0.45 | 0.49 | 0.40 | 0.40 | 0.39 | 0.40 | 0.55 | 0.53 | - | 0.53 | 0.41 | 0.43 | - | 0.43 | |
| SD | 0.05 | 0.03 | 0.07 | 0.05 | 0.05 | 0.03 | 0.02 | 0.03 | 0.04 | - | - | - | 0.04 | - | - | - | |
| Max | 0.70 | 0.55 | 0.50 | 0.55 | 0.50 | 0.45 | 0.40 | 0.45 | 0.63 | 0.53 | - | 0.53 | 0.45 | 0.43 | - | 0.43 | |
| Instar II | N | 60 | - | - | - | 60 | - | - | - | 1 | - | - | - | 1 | - | - | - |
| Min | 0.18 | - | - | - | 0.13 | - | - | - | 0.30 | - | - | - | 0.25 | - | - | - | |
| Median | 0.33 | - | - | - | 0.23 | - | - | - | 0.30 | - | - | - | 0.25 | - | - | - | |
| Mean | 0.31 | - | - | - | 0.22 | - | - | - | 0.30 | - | - | - | 0.25 | - | - | - | |
| SD | 0.05 | - | - | - | 0.04 | - | - | - | - | - | - | - | - | - | - | - | |
| Max | 0.40 | - | - | - | 0.33 | - | - | - | 0.30 | - | - | - | 0.25 | - | - | - | |
| Class and Deformity Type | (a) Littoral Zone | (b) Central Zone | (c) Lake Trasimeno | |||
|---|---|---|---|---|---|---|
| (N) | (%) | (N) | (%) | (N) | (%) | |
| CL. 2 | 38 | 162 | 200 | |||
| One or Two Round/Filed Teeth | 31 | 81.58 | 113 | 69.75 | 144 | 72.00 |
| One Missing Tooth | 1 | 2.63 | 29 | 17.90 | 30 | 15.00 |
| One Additional Tooth | 2 | 5.26 | 5 | 3.09 | 7 | 3.50 |
| One Bifid Tooth | 0 | 0 | 8 | 4.94 | 8 | 4.00 |
| One Serrate Tooth | 4 | 10.53 | 6 | 3.70 | 10 | 5.00 |
| Two Joined Teeth | 0 | 0 | 1 | 0.62 | 1 | 0.50 |
| Weak Asymmetry | 0 | 0 | 0 | 0 | 0 | 0 |
| CL. 3 | 23 | 30 | 53 | |||
| Very Round/Filed Teeth | 9 | 39.13 | 10 | 33.33 | 19 | 35.85 |
| Two or More Missing Teeth | 5 | 21.74 | 5 | 16.67 | 10 | 18.87 |
| Two or More Additional Teeth | 2 | 8.70 | 0 | 0 | 2 | 3.77 |
| Two or More Bifid Teeth | 0 | 0 | 0 | 0 | 0 | 0 |
| Serrate Teeth | 0 | 0 | 0 | 0 | 0 | 0 |
| Three or More Joined Teeth | 0 | 0 | 0 | 0 | 0 | 0 |
| Severe Asymmetry | 0 | 0 | 0 | 0 | 0 | 0 |
| Köhn Gap | 0 | 0 | 6 | 20.00 | 6 | 11.32 |
| Combination of Different Deformities | 7 | 30.43 | 9 | 30.00 | 16 | 30.19 |
| CL. (2 + 3) | 61 | 192 | 253 | |||
| Round/Filed Teeth | 40 | 65.57 | 123 | 64.06 | 163 | 64.43 |
| Missing Teeth | 6 | 9.84 | 34 | 17.71 | 40 | 15.81 |
| Additional Teeth | 4 | 6.56 | 5 | 2.60 | 9 | 3.56 |
| Bifid Teeth | 0 | 0 | 8 | 4.17 | 8 | 3.16 |
| Serrate Teeth | 4 | 6.56 | 6 | 3.13 | 10 | 3.95 |
| Joined Teeth | 0 | 0 | 1 | 0.52 | 1 | 0.40 |
| Asymmetry | 0 | 0 | 0 | 0 | 0 | 0 |
| Köhn Gap | 0 | 0 | 6 | 3.13 | 6 | 2.37 |
| Combination of Different Deformities | 7 | 11.48 | 9 | 4.69 | 16 | 6.32 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goretti, E.; Pallottini, M.; Pagliarini, S.; Catasti, M.; La Porta, G.; Selvaggi, R.; Gaino, E.; Di Giulio, A.M.; Ali, A. Use of Larval Morphological Deformities in Chironomus plumosus (Chironomidae: Diptera) as an Indicator of Freshwater Environmental Contamination (Lake Trasimeno, Italy). Water 2020, 12, 1. https://doi.org/10.3390/w12010001
Goretti E, Pallottini M, Pagliarini S, Catasti M, La Porta G, Selvaggi R, Gaino E, Di Giulio AM, Ali A. Use of Larval Morphological Deformities in Chironomus plumosus (Chironomidae: Diptera) as an Indicator of Freshwater Environmental Contamination (Lake Trasimeno, Italy). Water. 2020; 12(1):1. https://doi.org/10.3390/w12010001
Chicago/Turabian StyleGoretti, Enzo, Matteo Pallottini, Sarah Pagliarini, Marianna Catasti, Gianandrea La Porta, Roberta Selvaggi, Elda Gaino, Alessandro Maria Di Giulio, and Arshad Ali. 2020. "Use of Larval Morphological Deformities in Chironomus plumosus (Chironomidae: Diptera) as an Indicator of Freshwater Environmental Contamination (Lake Trasimeno, Italy)" Water 12, no. 1: 1. https://doi.org/10.3390/w12010001







