Predicting Lake Quality for the Next Generation: Impacts of Catchment Management and Climatic Factors in a Probabilistic Model Framework
Abstract
1. Introduction
2. Materials and Methods
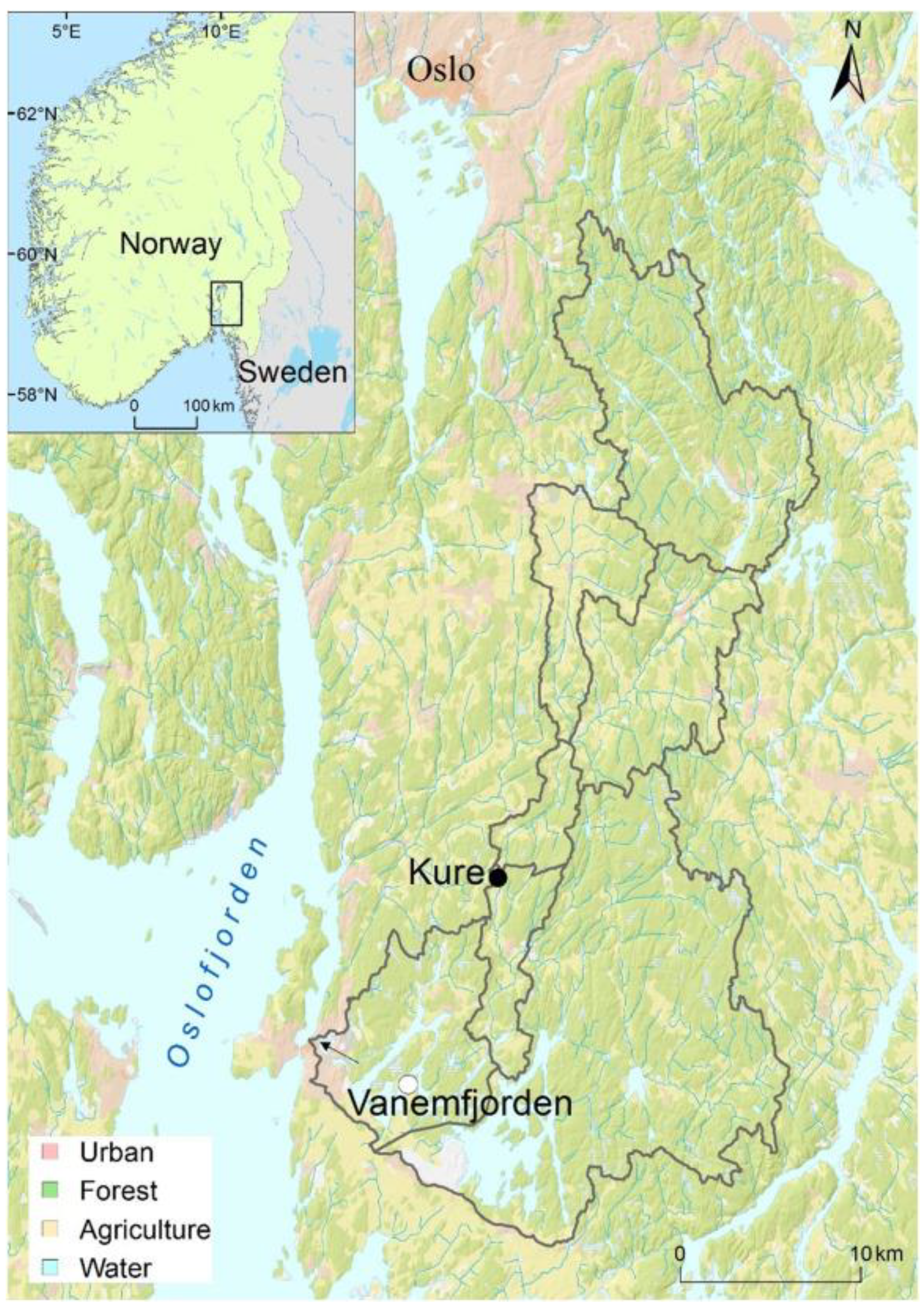
2.1. Case Study
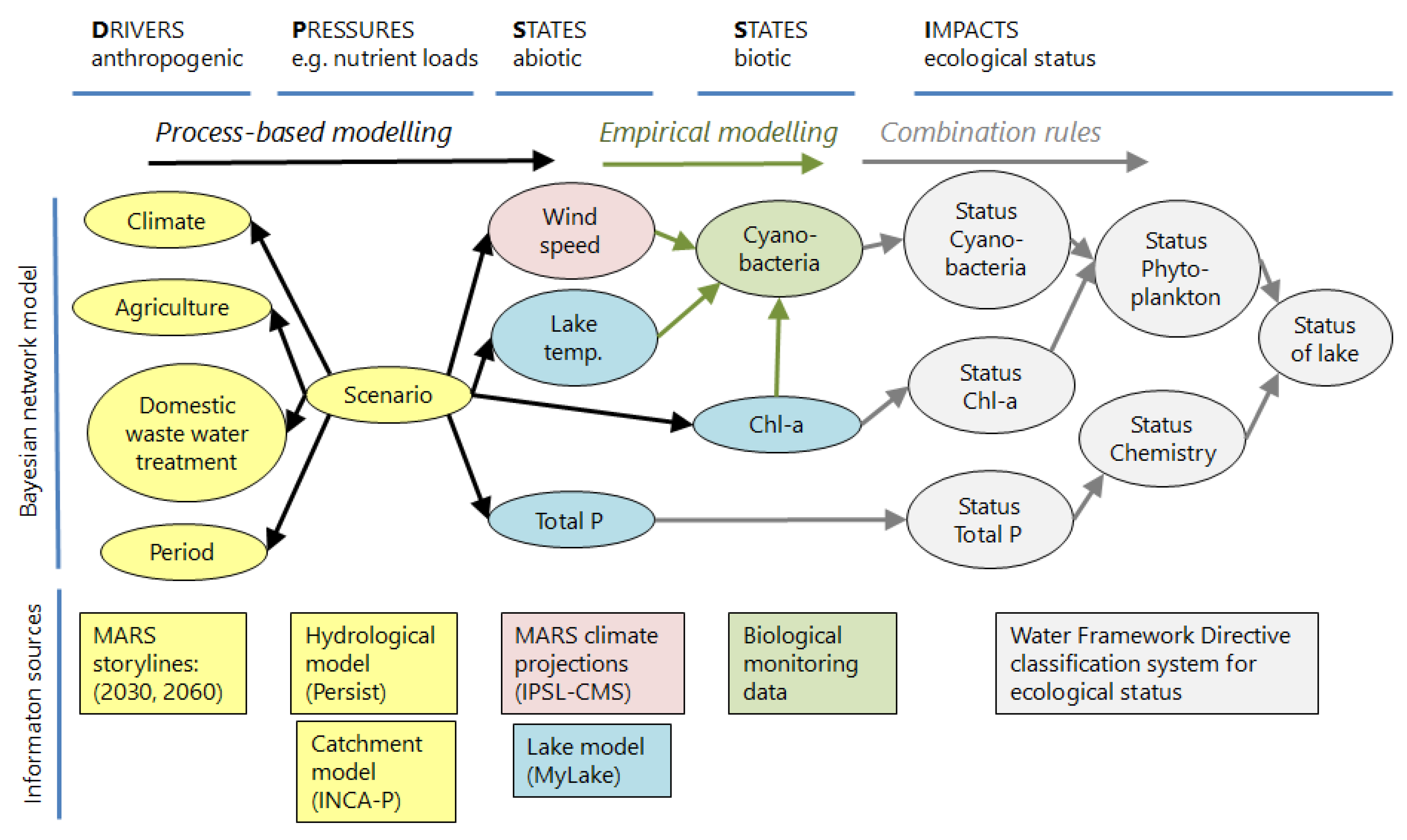
2.2. Bayesian Network as a Meta-Model for Future Storylines
- climate and management scenarios (yellow nodes),
- output from the process-based lake model MyLake (blue nodes),
- climatic data (red nodes),
- monitoring data from Lake Vansjø (green nodes), and
- the national classification system for ecological status of lakes (grey nodes).
- Consensus World (RCP4.5 and SSP2): the economy and population keep on growing, but environmental protection is prioritized. This is the best-case scenario for this case study.
- Fragmented World (RCP8.5 and SSP3) is based upon inequality: each country needs to fight for its own survival and the environment is only protected locally by rich countries.
- Techno World (RCP8.5 and SSP5) represents a future in which the world will be driven by economy. Policies are focused on enhancing trade and not on the environment. This is the worst-case scenario.
2.3. Revised BN Model Structure
2.4. Parametrization of Conditional Probability Tables
2.5. Running the BN Model for Scenarios
2.5.1. Explorative Scenarios
2.5.2. Future Climate and Management Scenarios
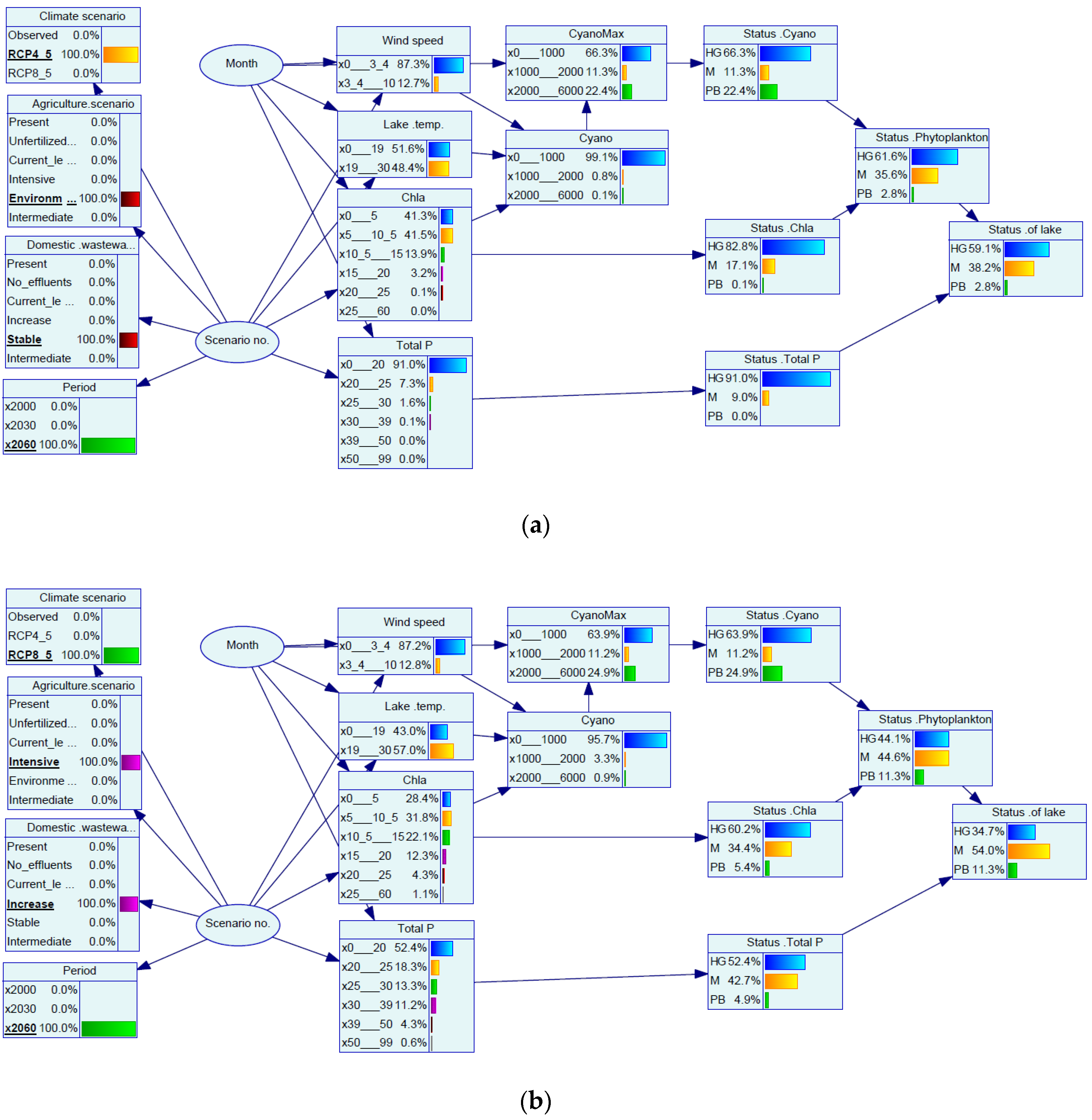
3. Results and Discussions
3.1. Effects of Explorative What-If Scenarios on Ecological Status
3.2. Predicted Lake Quality under Future Story Lines
3.3. Assessment of the Bayesian Network Modelling Approach
3.4. Conclusions and Outlook
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gozlan, R.E.; Karimov, B.K.; Zadereev, E.; Kuznetsova, D.; Brucet, S. Status, trends, and future dynamics of freshwater ecosystems in Europe and central Asia. Inland Waters 2019, 9, 78–94. [Google Scholar] [CrossRef]
- Jeppesen, E.; Kronvang, B.; Meerhoff, M.; Søndergaard, M.; Hansen, K.M.; Andersen, H.E.; Lauridsen, T.L.; Liboriussen, L.; Beklioglu, M.; Özen, A.; et al. Climate change effects on runoff, catchment phosphorus loading and lake ecological state, and potential adaptations. J. Environ. Qual. 2009, 38, 1930–1941. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Fastner, J.; Bormans, M.; Visser, P.M. Cyanobacterial blooms. Ecology, prevention, mitigation and control: Editorial to a CYANOCOST special issue. Aquat. Ecol. 2016, 50, 327–331. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Burford, M.A.; Carey, C.C.; Hamilton, D.P.; Huisman, J.; Paerl, H.W.; Wood, S.A.; Wulff, A. Perspective: Advancing the research agenda for improving understanding of cyanobacteria in a future of global change. Harmful Algae 2019. [Google Scholar] [CrossRef]
- EC (European Commission). Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy; Office for Official Publications of the European Communities: Luxembourg, 2000. [Google Scholar]
- EEA (European Environment Agency). European Waters. Assessment of Status and Pressures 2018; EEA Report No. 7/2018; Copenhagen, Denmark, 2018. [Google Scholar]
- EC (European Commission). Overall Approach to the Classification of Ecological Status and Ecological Potential;Common Implementation Strategy for the Water Framework Directive Guidance Document Number 13; Office for Official Publications of the European Communities: Luxembourg, 2005. [Google Scholar]
- EC (European Commission). River Basin Management in a Changing Climate. Common Implementation Strategy for the Water Framework Directive Guidance Document Number 24; Office for Official Publications of the European Communities: Luxembourg, 2009. [Google Scholar]
- Carvalho, L.; Mackay, E.B.; Cardoso, A.C.; Baattrup-Pedersen, A.; Birk, S.; Blackstock, K.L.; Borics, G.; Borja, A.; Feld, C.K.; Ferreira, M.T.; et al. Protecting and restoring Europe’s waters: An analysis of the future development needs of the water framework directive. Sci. Total Environ. 2019, 658, 1228–1238. [Google Scholar] [CrossRef]
- Hering, D.; Carvalho, L.; Argillier, C.; Beklioglu, M.; Borja, A.; Cardoso, A.C.; Duel, H.; Ferreira, T.; Globevnik, L.; Hanganu, J.; et al. Managing aquatic ecosystems and water resources under multiple stress—An introduction to the MARS project. Sci. Total Environ. 2015, 503, 10–21. [Google Scholar] [CrossRef]
- Ferreira, T.; Panagopoulos, Y.; Bloomfield, J.; Couture, R.-M.; Omerod, S.; Stefanidis, K.; Mimikou, M.; Hanganu, J.; Constantinescu, A.; Beklioğlu, M.; et al. Deliverable 4.1: Case Study Synthesis. 2016. Available online: http://www.mars-project.eu/files/download/deliverables/MARS_D4.1_case_study_synthesis.pdf. (accessed on 30 June 2019).
- Barton, D.N.; Kuikka, S.; Varis, O.; Uusitalo, L.; Henriksen, H.J.; Borsuk, M.; de la Hera, A.; Farmani, R.; Johnson, S.; Linnell, J.D.C. Bayesian networks in environmental and resource management. Integr. Environ. Assess. Manag. 2012, 8, 418–429. [Google Scholar] [CrossRef]
- Marcot, B.G.; Penman, T.D. Advances in bayesian network modelling: Integration of modelling technologies. Environ. Model. Softw. 2019, 111, 386–393. [Google Scholar] [CrossRef]
- Moe, S.J. Bayesian models in assessment and management. In Environmental Risk Assessment and Management from a Landscape Perspective; Kapustka, L., Landis, W.G., Johnson, A., Eds.; Wiley’s: Chicago, IL, USA, 2010. [Google Scholar]
- Van Geest, G.; Kramer, L.; Buijse, T.; Moe, J.; Couture, R.-M.; Lyche Solheim, A.; Molina-Navarro, E.; Andersen, H.E.; Trolle, D.; Rankinen, K.; et al. D7.2-2: Bayesian Belief Networks: Linking Abiotic and Biotic Data. 2017. Available online: http://www.mars-project.eu/files/download/deliverables/MARS_D7.2_MARS_suite_of_tools_2.pdf (accessed on 30 June 2019).
- Moe, S.J.; Haande, S.; Couture, R.-M. Climate change, cyanobacteria blooms and ecological status of lakes: A Bayesian network approach. Ecol. Model. 2016, 337, 330–347. [Google Scholar] [CrossRef]
- Carvalho, L.; McDonald, C.; de Hoyos, C.; Mischke, U.; Phillips, G.; Borics, G.; Poikane, S.; Skjelbred, B.; Lyche Solheim, A.; Van Wichelen, J.; et al. Sustaining recreational quality of European lakes: Minimizing the health risks from algal blooms through phosphorus control. J. Appl. Ecol. 2013, 50, 315–323. [Google Scholar] [CrossRef]
- Richardson, J.; Miller, C.; Maberly, S.C.; Taylor, P.; Globevnik, L.; Hunter, P.; Jeppesen, E.; Mischke, U.; Moe, S.J.; Pasztaleniec, A.; et al. Effects of multiple stressors on cyanobacteria abundance vary with lake type. Glob. Chang. Biol. 2018, 24, 5044–5055. [Google Scholar] [CrossRef]
- Ibelings, B.W.; Vonk, M.; Los, H.F.J.; van der Molen, D.T.; Mooij, W.M. Fuzzy modeling of cyanobacterial surface waterblooms: Validation with NOAA-AVHRR satellite images. Ecol. Appl. 2003, 13, 1456–1472. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Blooms like it hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef]
- Woolway, R.I.; Meinson, P.; Nõges, P.; Jones, I.D.; Laas, A. Atmospheric stilling leads to prolonged thermal stratification in a large shallow polymictic lake. Clim. Chang. 2017, 141, 759–773. [Google Scholar] [CrossRef]
- Mack, L.; Andersen, H.E.; Beklioğlu, M.; Bucak, T.; Couture, R.-M.; Cremona, F.; Ferreira, M.T.; Hutchins, M.G.; Mischke, U.; Molina-Navarro, E.; et al. The future depends on what we do today-projecting Europe’s surface water quality into three different future scenarios. Sci. Total Environ. 2019, 668, 470–484. [Google Scholar] [CrossRef]
- Couture, R.M.; Tominaga, K.; Starrfelt, J.; Moe, S.J.; Kaste, O.; Wright, R.F. Modelling phosphorus loading and algal blooms in a Nordic agricultural catchment-lake system under changing land-use and climate. Environ. Sci. Process. Impacts 2014, 16, 1588–1599. [Google Scholar] [CrossRef]
- Skarbøvik, E.; Haande, S.; Bechmann, M.; Skjelbred, B. Vannovervåking i Morsa 2018. Innsjøer, Elver og Bekker: November 2017–Oktober 2018; NIBIO: Høgskoleveien, Norway, 2019. [Google Scholar]
- Haande, S.; Lyche Solheim, A.; Moe, J.; Brænden, R. Klassifisering av Økologisk Tilstand i Elver og Innsjøer i Vannområde Morsa iht. Vanndirektivet; Norsk Institutt for Vannforskning: Oslo, Norway, 2011; p. 39. [Google Scholar]
- Couture, R.-M.; Moe, S.J.; Lin, Y.; Kaste, Ø.; Haande, S.; Lyche Solheim, A. Simulating water quality and ecological status of Lake Vansjø, Norway, under land-use and climate change by linking process-oriented models with a Bayesian network. Sci. Total Environ. 2018, 621, 713–724. [Google Scholar] [CrossRef]
- Futter, M.N.; Erlandsson, M.A.; Butterfield, D.; Whitehead, P.G.; Oni, S.K.; Wade, A.J. Persist: The precipitation, evapotranspiration and runoff simulator for solute transport. Hydrol. Earth Syst. Sci. Discuss. 2013, 10, 8635–8681. [Google Scholar] [CrossRef]
- Wade, A.J.; Whitehead, P.G.; Butterfield, D. The integrated catchments model of phosphorus dynamics (INCA-P), a new approach for multiple source assessment in heterogeneous river systems: Model structure and equations. Hydrol. Earth Syst. Sci. Discuss. 2002, 6, 583–606. [Google Scholar] [CrossRef]
- Saloranta, T.M.; Andersen, T. MyLake—A multi-year lake simulation model code suitable for uncertainty and sensitivity analysis simulations. Ecol. Model. 2007, 207, 45–60. [Google Scholar] [CrossRef]
- Lyche Solheim, A.; Phillips, G.; Drakare, S.; Free, G.; Järvinen, M.; Skjelbred, B.; Tierney, D.; Trodd, W. Northern Lake Phytoplankton Ecological Assessment Methods. Water Framework Directive Intercalibration Technical Report; Poikane, S., Ed.; Jrc-Report eur 26503 en; Publications Office of the European Union: Brussels, Belgium, 2014. [Google Scholar]
- Elliott, J.A. The seasonal sensitivity of cyanobacteria and other phytoplankton to changes in flushing rate and water temperature. Glob. Chang. Biol. 2010, 16, 864–876. [Google Scholar] [CrossRef]
- Aguilera, P.A.; Fernández, A.; Fernández, R.; Rumí, R.; Salmerón, A. Bayesian networks in environmental modelling. Environ. Model. Softw. 2011, 26, 1376–1388. [Google Scholar] [CrossRef]
- Varis, O.; Kuikka, S. Learning Bayesian decision analysis by doing: Lessons from environmental and natural resources management. Ecol. Model. 1999, 119, 177–195. [Google Scholar] [CrossRef]
- Chen, S.H.; Pollino, C.A. Good practice in Bayesian network modelling. Environ. Model. Softw. 2012, 37, 134–145. [Google Scholar] [CrossRef]
- Direktoratsgruppen Vanndirektivet. Veileder 2:2018 Klassifisering av Miljøtilstand i Vann. Økologisk og Kjemisk Klassifiseringssystem for Kystvann, Grunnvann, Innsjøer og Elver. Available online: http://www.vannportalen.no/globalassets/nasjonalt/dokumenter/veiledere-direktoratsgruppa/Klassifisering-av-miljotilstand-i-vann-02-2018.pdf (accessed on 30 June 2019).
- Barton, D.N.; Saloranta, T.; Bakken, T.H.; Lyche Solheim, A.; Moe, J.; Selvik, J.R.; Vagstad, N. Using Bayesian network models to incorporate uncertainty in the economic analysis of pollution abatement measures under the Water Framework Directive. Water Supply 2005, 5, 99–104. [Google Scholar] [CrossRef]
- Barton, D.N.; Saloranta, T.; Moe, S.J.; Eggestad, H.O.; Kuikka, S. Bayesian belief networks as a meta-modelling tool in integrated river basin management—Pros and cons in evaluating nutrient abatement decisions under uncertainty in a Norwegian river basin. Ecol. Econ. 2008, 66, 91–104. [Google Scholar] [CrossRef]
- Barton, D.N.; Andersen, T.; Bergland, O.; Engebretsen, A.; Moe, J.; Orderud, G.; Tominaga, K.; Romstad, E.; Vogt, R. Eutropia—Integrated valuation of lake eutrophication abatement decisions using a Bayesian belief network. In Applied System Science; Routledge: London, UK, 2014. [Google Scholar]
- Faneca Sanchez, M.; Duel, H.; Sampedro, A.A.; Rankinen, K.; Holmberg, M.; Prudhomme, C. Deliverable 2.1—Four Manuscripts on the Multiple Stressor Framework; Part 4: Report on the MARS scenarios of future changes in drivers and pressures with respect to Europe’s water resources. 2015. Available online: http://www.mars-project.eu/files/download/deliverables/MARS_D2.1_Four_manuscripts_on_the_multiple_stressor_framework.pdf (accessed on 30 June 2019).
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; Scientific Research: Vienna, Austria, 2018; Available online: http://www.R-project.Org/ (accessed on 30 June 2019).
- Cleveland, W.S. LOWESS: A program for smoothing scatterplots by robust locally weighted regression. Am. Stat. 1981, 35, 54. [Google Scholar] [CrossRef]
- Nojavan A, F.; Qian, S.S.; Stow, C.A. Comparative analysis of discretization methods in Bayesian networks. Environ. Model. Softw. 2017, 87, 64–71. [Google Scholar] [CrossRef]
- Therneau, T.; Atkinson, B.; Ripley, B. Rpart: Recursive Partitioning and Regression Trees. R Package Version 4.1-9. 2015. Available online: http://cran.r-project.org/package=rpart (accessed on 30 June 2019).
- Williams, B.J.; Cole, B. Mining monitored data for decision-making with a Bayesian network model. Ecol. Model. 2013, 249, 26–36. [Google Scholar] [CrossRef]
- Borsuk, M.E.; Stow, C.A.; Reckhow, K.H. A Bayesian network of eutrophication models for synthesis, prediction, and uncertainty analysis. Ecol. Model. 2004, 173, 219–239. [Google Scholar] [CrossRef]
- Christensen, R.H.B. Ordinal—Regression Models for Ordinal Data. R Package Version. 2015. Available online: https://cran.r-project.org/web/packages/ordinal (accessed on 30 June 2019).
- Phillips, G.; Pietiläinen, O.P.; Carvalho, L.; Solimini, A.; Lyche Solheim, A.; Cardoso, A.C. Chlorophyll–nutrient relationships of different lake types using a large European dataset. Aquat. Ecol. 2008, 42, 213–226. [Google Scholar] [CrossRef]
- Elliott, J.A. Is the future blue-green? A review of the current model predictions of how climate change could affect pelagic freshwater cyanobacteria. Water Res. 2012, 46, 1364–1371. [Google Scholar] [CrossRef]
- Wagner, C.; Adrian, R. Cyanobacteria dominance: Quantifying the effects of climate change. Limnol. Oceanogr. 2009, 54, 2460–2468. [Google Scholar] [CrossRef]
- Kehoe, M.J.; Ingalls, B.P.; Venkiteswaran, J.J.; Baulch, H.M. Successful forecasting of harmful cyanobacteria blooms with high frequency lake data. bioRxiv 2019, 674325. [Google Scholar] [CrossRef]
- Landuyt, D.; Broekx, S.; D’Hondt, R.; Engelen, G.; Aertsens, J.; Goethals, P.L.M. A review of Bayesian belief networks in ecosystem service modelling. Environ. Model. Softw. 2013, 46, 1–11. [Google Scholar] [CrossRef]
- Birk, S.; Chapman, D.; Carvalho, L.; Spears, B.M.; Andersen, H.E.; Argillier, C.; Auer, S.; Baattrup-Pedersen, A.; Banin, L.; Beklioğlu, M.; et al. Are millennial freshwater ecosystems under more complex stress? Nat. Ecol. Evol. 2019. in review. [Google Scholar]
- Kosten, S.; Huszar, V.L.M.; Bécares, E.; Costa, L.S.; Donk, E.; Hansson, L.-A.; Jeppesen, E.; Kruk, C.; Lacerot, G.; Mazzeo, N.; et al. Warmer climates boost cyanobacterial dominance in shallow lakes. Glob. Chang. Biol. 2012, 18, 118–126. [Google Scholar] [CrossRef]
- Rigosi, A.; Hanson, P.; Hamilton, D.P.; Hipsey, M.; Rusak, J.A.; Bois, J.; Sparber, K.; Chorus, I.; Watkinson, A.J.; Qin, B.; et al. Determining the probability of cyanobacterial blooms: The application of Bayesian networks in multiple lake systems. Ecol. Appl. 2015, 25, 186–199. [Google Scholar] [CrossRef]
- Lehikoinen, A.; Helle, I.; Klemola, E.; Mäntyniemi, S.; Kuikka, S.; Pitkänen, H. Evaluating the impact of nutrient abatement measures on the ecological status of coastal waters: A Bayesian network for decision analysis. Int. J. Multicriteria Decis. Mak. 2014, 4, 114–134. [Google Scholar] [CrossRef]
- Jiang, P.; Liu, X.; Zhang, J.; Te, S.H.; Gin, K.Y.H. Latent variable structured Bayesian network for cyanobacterial risk pre-control. In Proceedings of the 2018 IEEE International Conference on Industrial Engineering and Engineering Management (IEEM), Bangkok, Thailand, 16–19 December 2018. [Google Scholar]
- Manning, N.F.; Wang, Y.-C.; Long, C.M.; Bertani, I.; Sayers, M.J.; Bosse, K.R.; Shuchman, R.A.; Scavia, D. Extending the forecast model: Predicting Western Lake Erie harmful algal blooms at multiple spatial scales. J. Gt. Lakes Res. 2019, 45, 587–595. [Google Scholar] [CrossRef]
- Uusitalo, L. Advantages and challenges of Bayesian networks in environmental modelling. Ecol. Model. 2007, 203, 312–318. [Google Scholar] [CrossRef]
- Marcot, B.G.; Steventon, J.D.; Sutherland, G.D.; McCann, R.K. Guidelines for developing and updating Bayesian belief networks applied to ecological modeling and conservation. Can. J. For. Res. 2006, 36, 3063–3074. [Google Scholar] [CrossRef]
- Poikane, S.; Birk, S.; Böhmer, J.; Carvalho, L.; de Hoyos, C.; Gassner, H.; Hellsten, S.; Kelly, M.; Lyche Solheim, A.; Olin, M.; et al. A hitchhiker’s guide to European lake ecological assessment and intercalibration. Ecol. Indic. 2015, 52, 533–544. [Google Scholar] [CrossRef]
- Qian, S.S.; Miltner, R.J. A continuous variable Bayesian networks model for water quality modeling: A case study of setting nitrogen criterion for small rivers and streams in Ohio, USA. Environ. Model. Softw. 2015, 69, 14–22. [Google Scholar] [CrossRef]
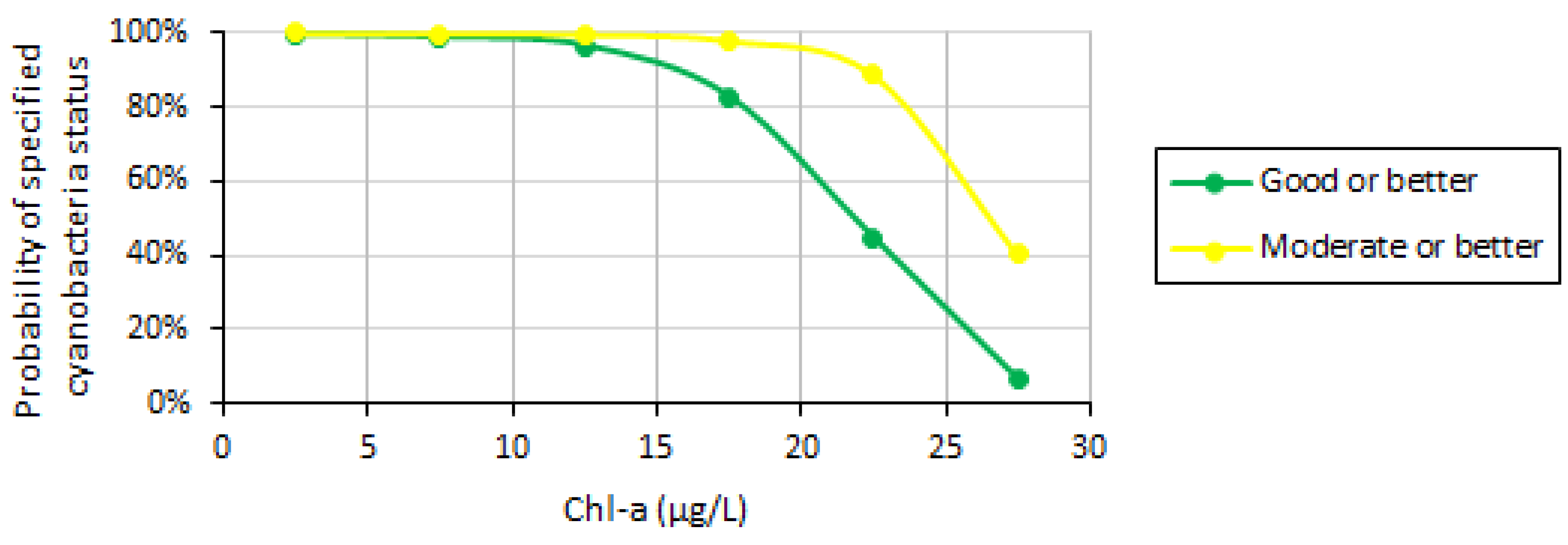
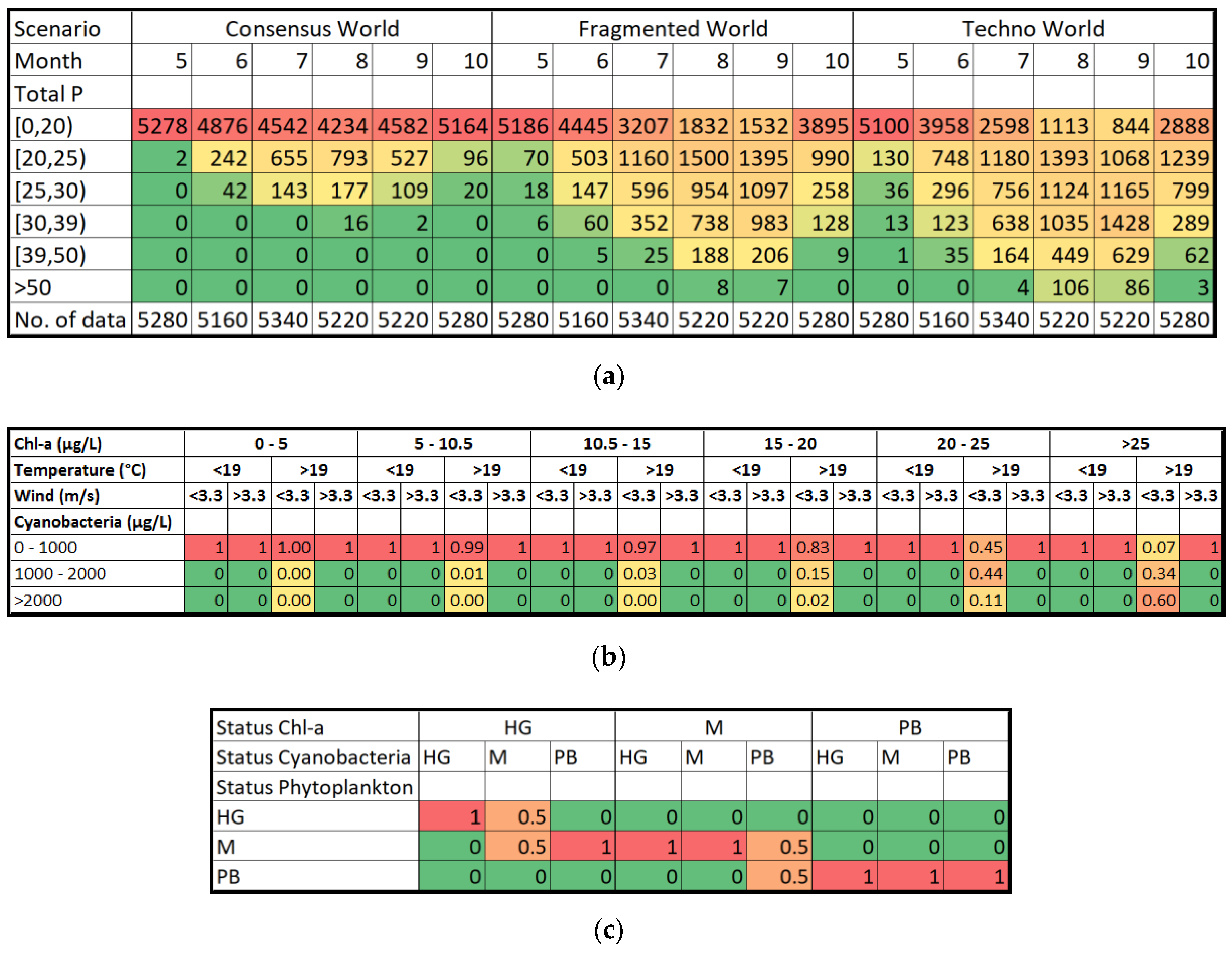
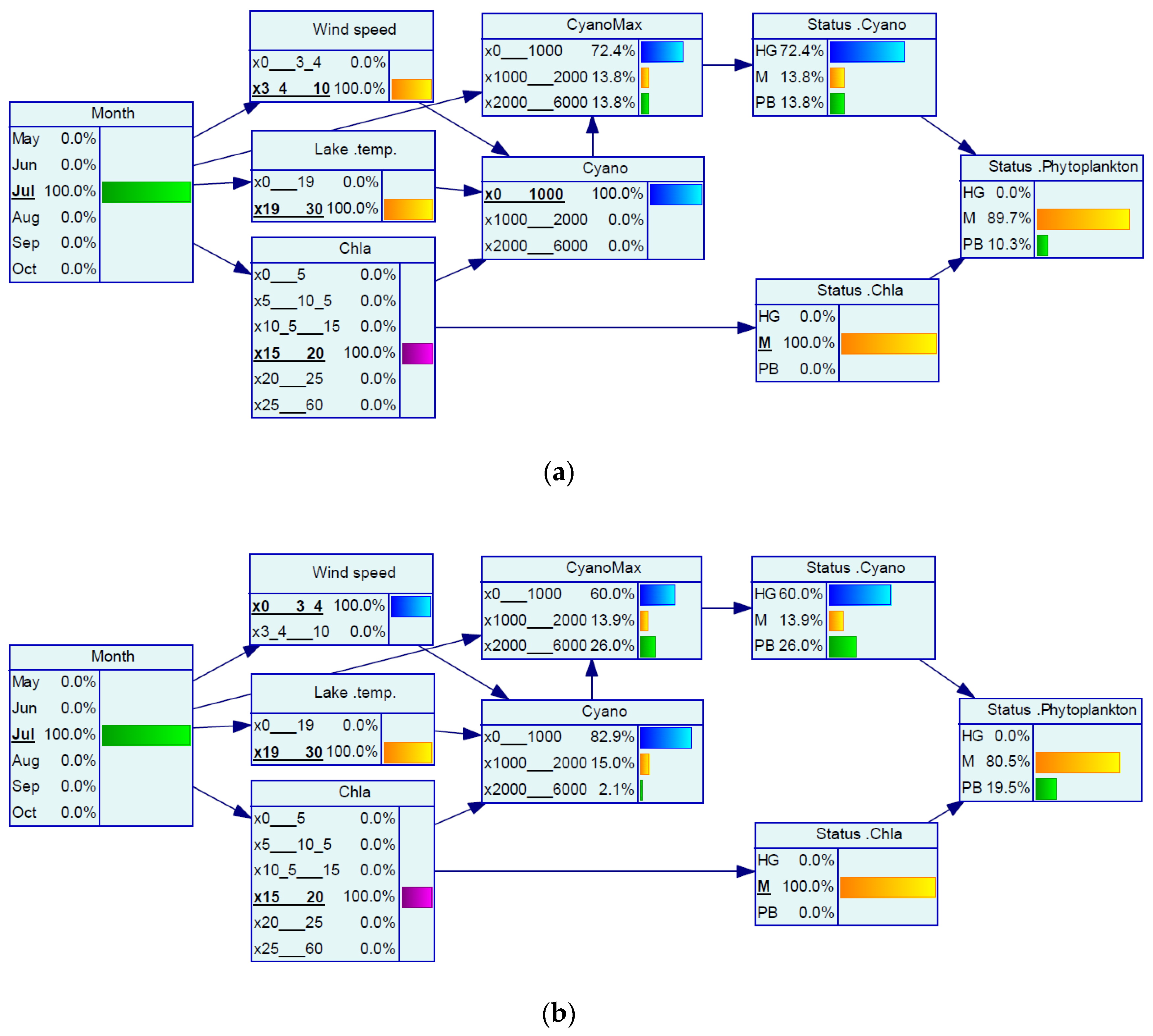
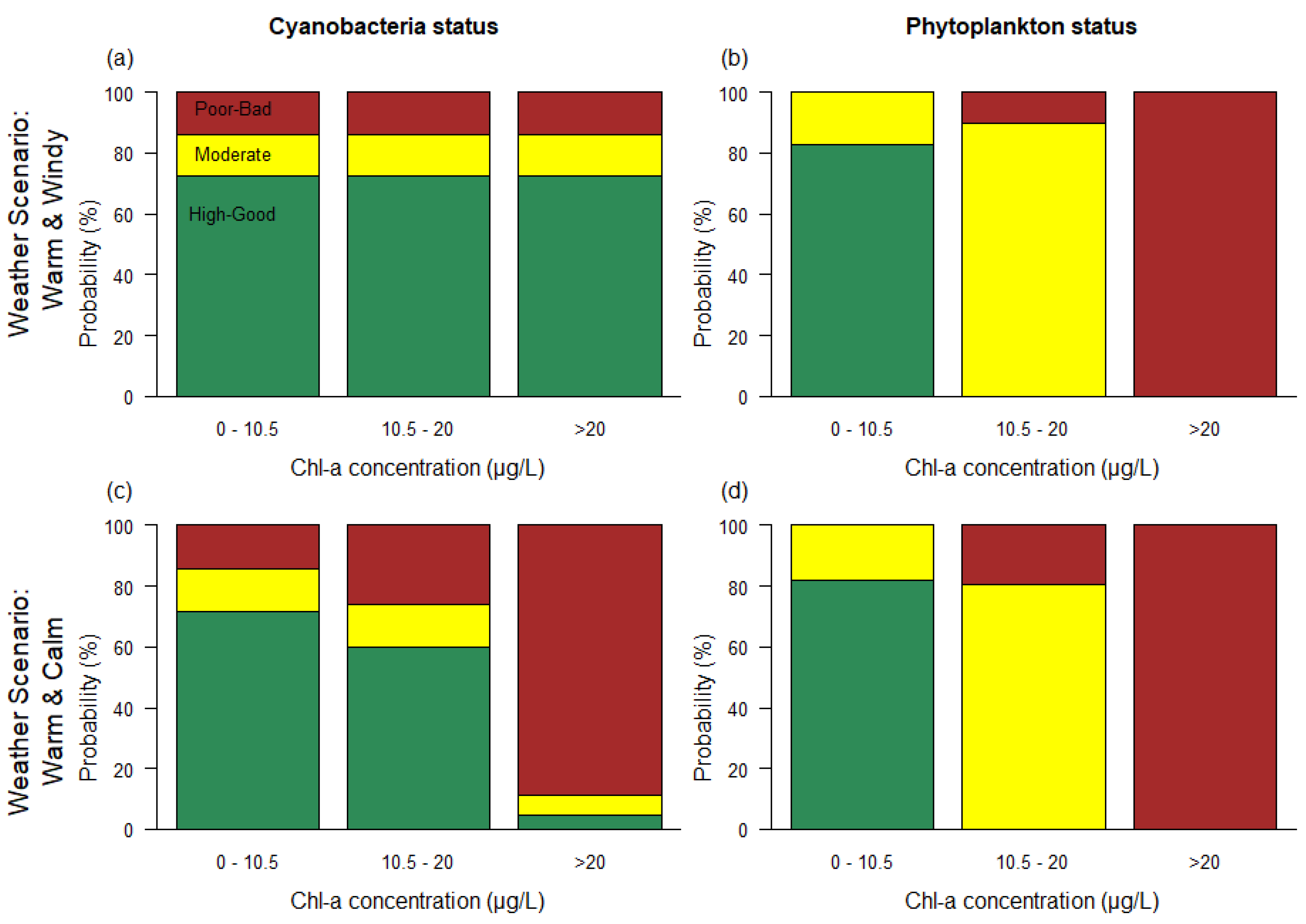

| Variable | Good/Moderate | Moderate/Poor |
|---|---|---|
| Chl-a | 10.5 | 20 |
| CyanoMax | 1000 | 2000 |
| Total P | 20 | 39 |
| Storyline | Climate Scenario | Agricultural Development | Water-Related Development |
|---|---|---|---|
| Consensus World | RCP4.5 | 10% of grassland converted to forest; 30% shift from vegetables and crops to unfertilized grasslands; 50% decrease in fertilization; 50% decrease in erosion | 50% decrease in effluent from scattered dwellings and WWTPs |
| Fragmented World 1 | RCP8.5 | 5% of forest converted to grassland; 30% of grassland converted to arable land; 15% increase in fertilization; 15% increase in erosion | 25% increase in effluent from scattered dwellings and WWTPs |
| Techno World 1 | RCP8.5 | 10% of forest areas converted to grassland; 60% of grassland converted to arable land; 30% increase in fertilization; 30% increase in erosion; | 40% increase in effluent from scattered dwellings and WWTPs |
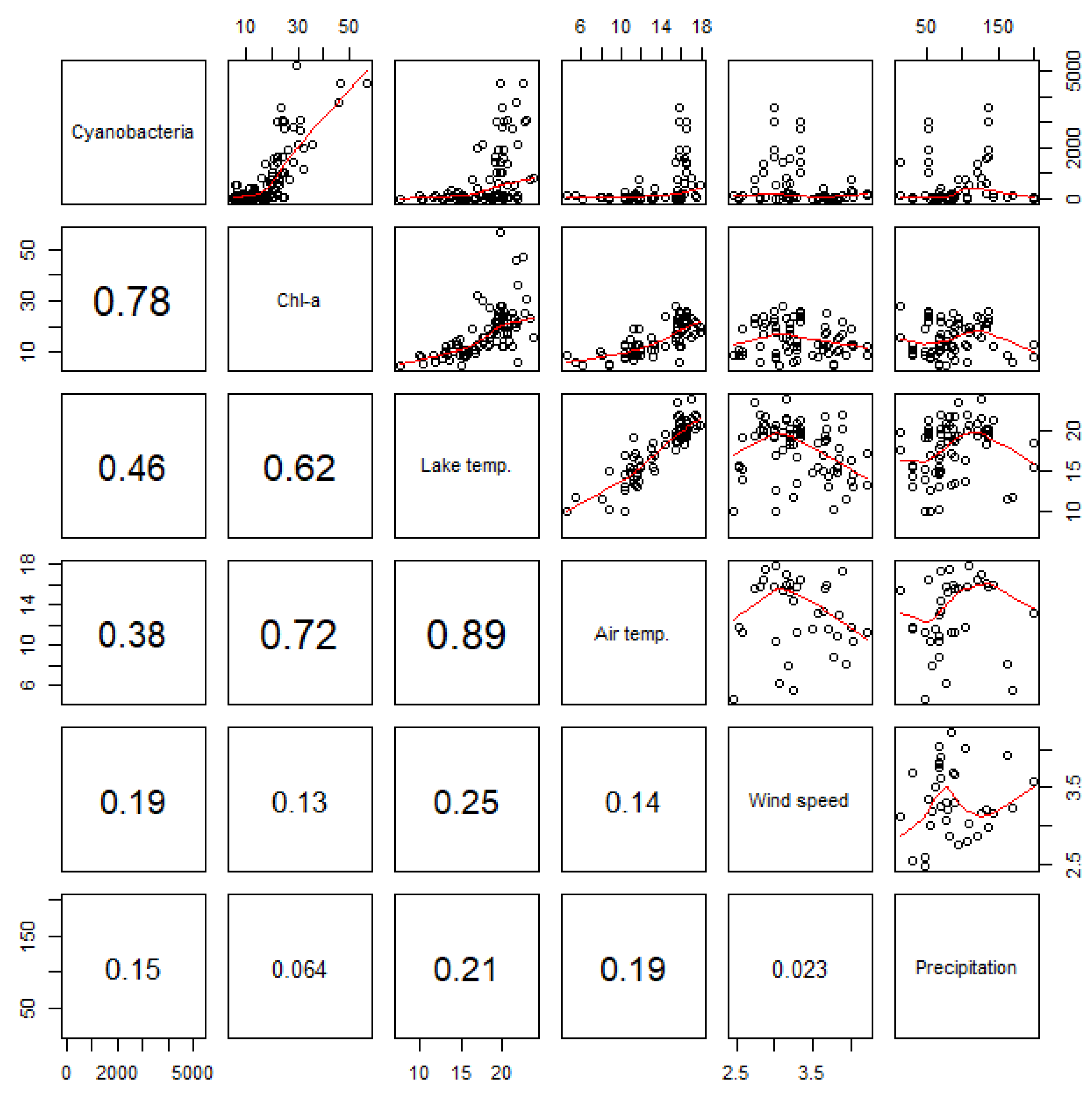
| Model No. | Explanatory Variables | Number of Obs. | AIC |
|---|---|---|---|
| 1 | Chl-a | 107 | 91.54 |
| 2 | Lake temperature | 90 | 117.9 |
| 3 | Wind speed | 90 | 81.4 |
| 4 | Chl-a + Lake temperature | 90 | 70.7 |
| 5 | Chl-a + Wind speed | 77 | 55.9 |
| 6 | Chl-a + Lake temperature + Wind speed | 73 | 56.4 |
| Node Group | Node Label | State Types | No. of States | Source of Probability Table |
|---|---|---|---|---|
| Scenarios | Scenario no. | Numbers | 25 | (Root node) |
| Climate scenario | Categories | 3 | MARS storylines | |
| Agriculture scenario | Categories | 4 | MARS storylines | |
| Domestic wastewater scenario | Categories | 4 | MARS storylines | |
| Period (time horizon) | Numbers | 3 | MARS storylines | |
| Month | Categories | 6 | (Root node) | |
| Climate | Wind speed | Intervals (unit: m/s) | 2 | Count of data (simulated) |
| Process-based lake model | Lake temperature | Intervals (unit: °C) | 2 | Count of data (simulated) |
| Chl-a | Intervals (unit: µg/L) | 6 | Count of data (simulated) | |
| Total P | Intervals (unit: µg/L) | 6 | Count of data (simulated) | |
| Biological monitoring data | Cyanobacteria | Intervals (unit: µg/L) | 3 | Statistical model |
| CyanoMax | Intervals (unit: µg/L) | 3 | Classification system | |
| Ecological status | Status Cyanobacteria | Ordered categories | 3 | Classification system |
| Status Chl-a | Ordered categories | 3 | Classification system | |
| Status Phytoplankton | Ordered categories | 3 | Classification system | |
| Status Total P | Ordered categories | 3 | Classification system | |
| Status of lake | Ordered categories | 3 | Classification system |
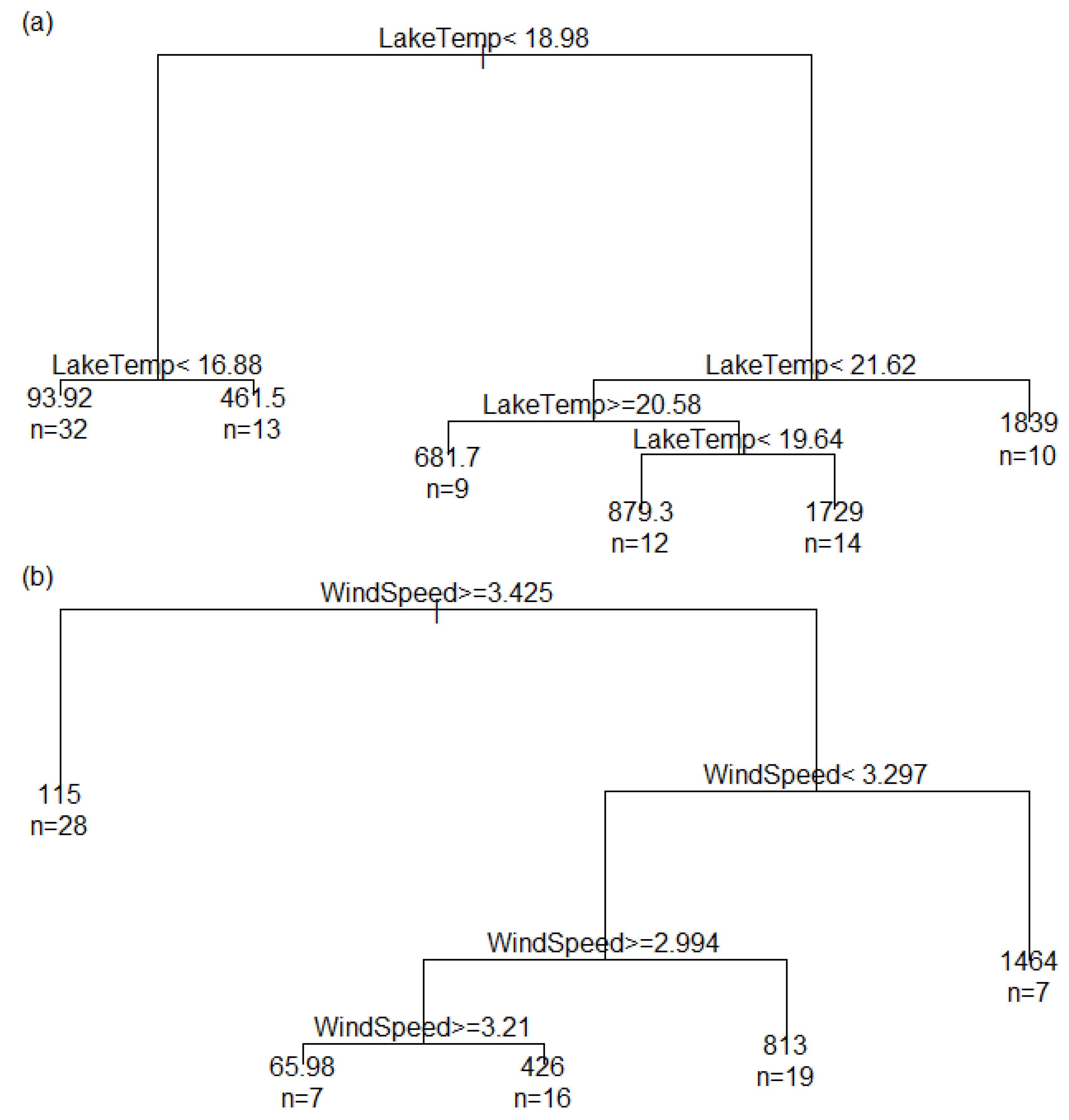
| Variable | Low State Name | Low Interval | High State Name | High Interval |
|---|---|---|---|---|
| Wind speed | Calm | <3.4 m/s | Windy | >3.4 m/s |
| Lake temperature | Cold | <19 °C | Warm | >19 °C |
| Scenario | Climate | Agriculture Scenario | Wastewater Scenario | Wind (m/s) | Lake Temperature (°C) | Total P (µg/L) | Chl-a (µg/L) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Code | Name | 2030 | 2060 | 2030 | 2060 | 2030 | 2060 | 2030 | 2060 | |||
| BL | Extended baseline | Current | Current | Current | 2.16 (0.48) | 2.17 (0.55) | 14.8 (4.8) | 14.9 (4.7) | 16.7 (6.0) | 14.1 (5.6) | 8.3 (4.5) | 6.3 (3.7) |
| 4.5 | RCP4.5 | RCP4.5 | Current | Current | 2.13 (0.54) | 2.11 (0.53) | 17.4 (4.7) | 18.4 (4.5) | 17.6 (5.3) | 15.9 (5.2) | 8.7 (4.0) | 7.2 (3.5) |
| 8.5 | RCP8.5 | RCP8.5 | Current | Current | 2.08 (0.46) | 2.05 (0.51) | 17.6 (4.9) | 19.2 (4.4) | 17.1 (5.3) | 16.0 (5.0) | 8.6 (4.1) | 7.3 (3.5) |
| CW | Consensus World | RCP4.5 | Environmental | Stable | 2.13 (0.54) | 2.11 (0.53) | 17.4 (4.7) | 18.4 (4.5) | 14.1 (4.1) | 12.4 (3.5) | 7.6 (3.7) | 6.2 (3.0) |
| FW | Fragmented World | RCP8.5 | Intermediate | Intermediate | 2.08 (0.46) | 2.05 (0.51) | 17.6 (4.9) | 19.2 (4.4) | 19.1 (6.2) | 18.3 (6.1) | 9.2 (4.3) | 8.0 (3.9) |
| TW | Techno World | Intensive | Increase | 21.4 (7.3) | 21.1 (7.5) | 9.9 (4.7) | 9.0 (4.3) | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moe, S.J.; Couture, R.-M.; Haande, S.; Lyche Solheim, A.; Jackson-Blake, L. Predicting Lake Quality for the Next Generation: Impacts of Catchment Management and Climatic Factors in a Probabilistic Model Framework. Water 2019, 11, 1767. https://doi.org/10.3390/w11091767
Moe SJ, Couture R-M, Haande S, Lyche Solheim A, Jackson-Blake L. Predicting Lake Quality for the Next Generation: Impacts of Catchment Management and Climatic Factors in a Probabilistic Model Framework. Water. 2019; 11(9):1767. https://doi.org/10.3390/w11091767
Chicago/Turabian StyleMoe, S. Jannicke, Raoul-Marie Couture, Sigrid Haande, Anne Lyche Solheim, and Leah Jackson-Blake. 2019. "Predicting Lake Quality for the Next Generation: Impacts of Catchment Management and Climatic Factors in a Probabilistic Model Framework" Water 11, no. 9: 1767. https://doi.org/10.3390/w11091767
APA StyleMoe, S. J., Couture, R.-M., Haande, S., Lyche Solheim, A., & Jackson-Blake, L. (2019). Predicting Lake Quality for the Next Generation: Impacts of Catchment Management and Climatic Factors in a Probabilistic Model Framework. Water, 11(9), 1767. https://doi.org/10.3390/w11091767





