Potential Use of Biochar from Various Waste Biomass as Biosorbent in Co(II) Removal Processes
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Biochar Samples
2.2. Characterization Methods
2.3. Chemical Reagents
2.4. Sorption/Desorption Experiments
2.5. Isotherm and Kinetics Models
3. Results and Discussion
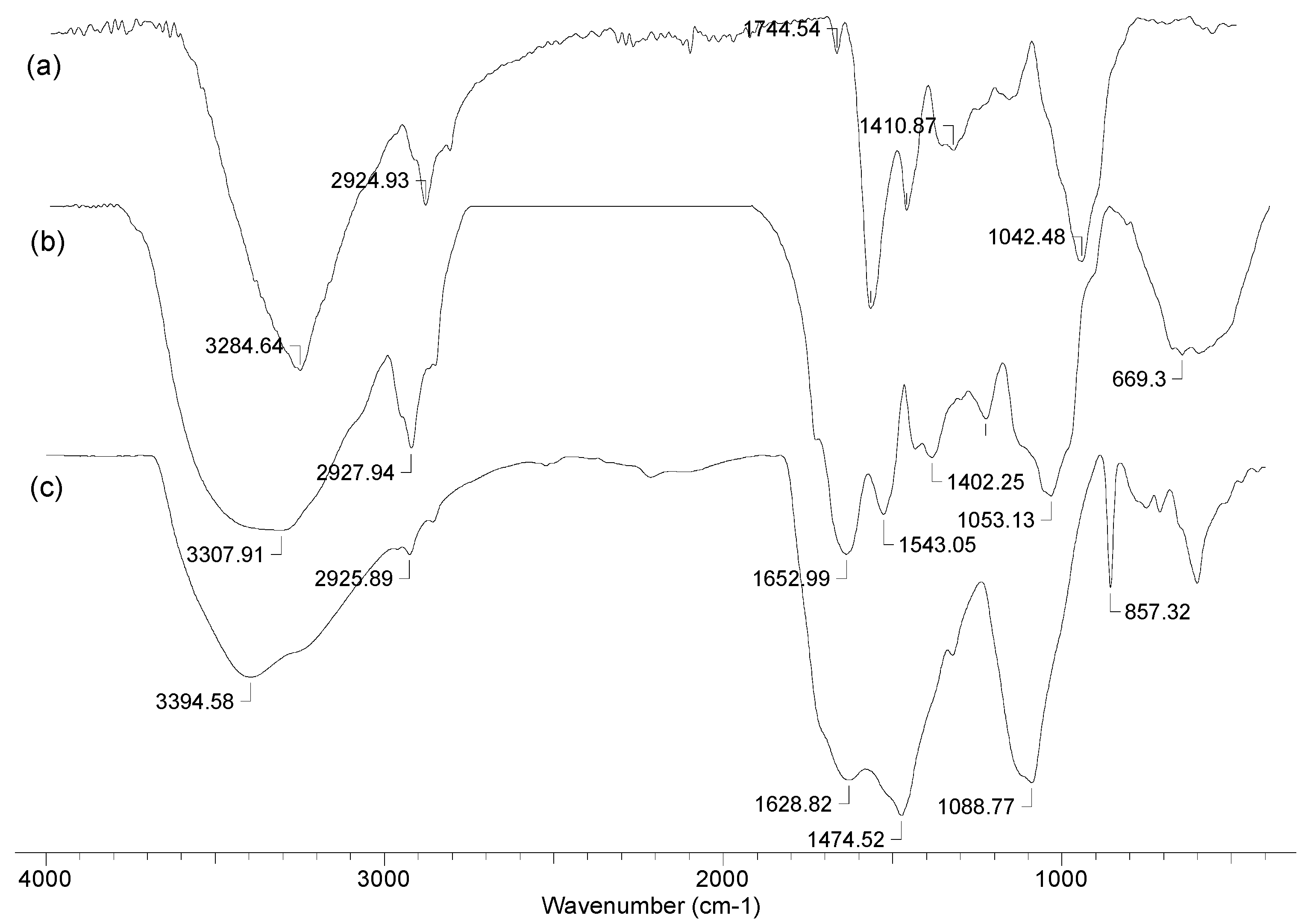
3.1. Characterization of the Biochar Samples
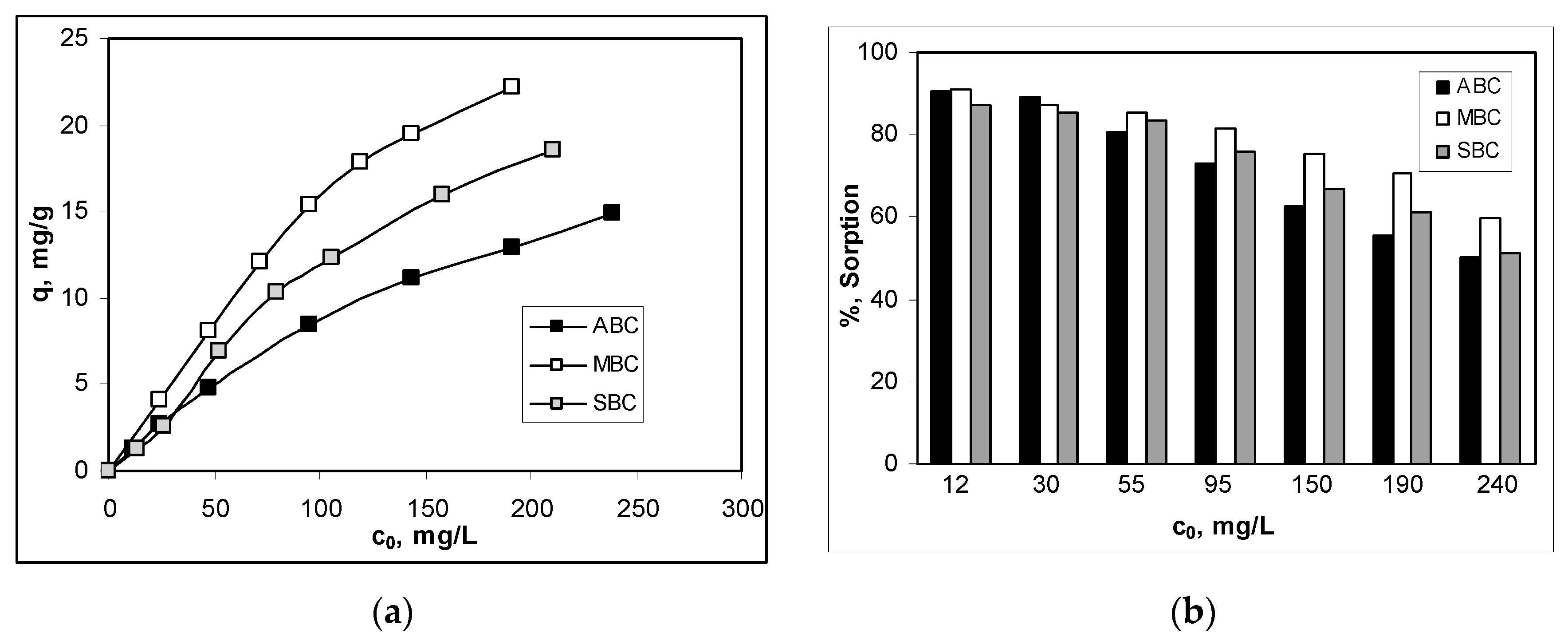
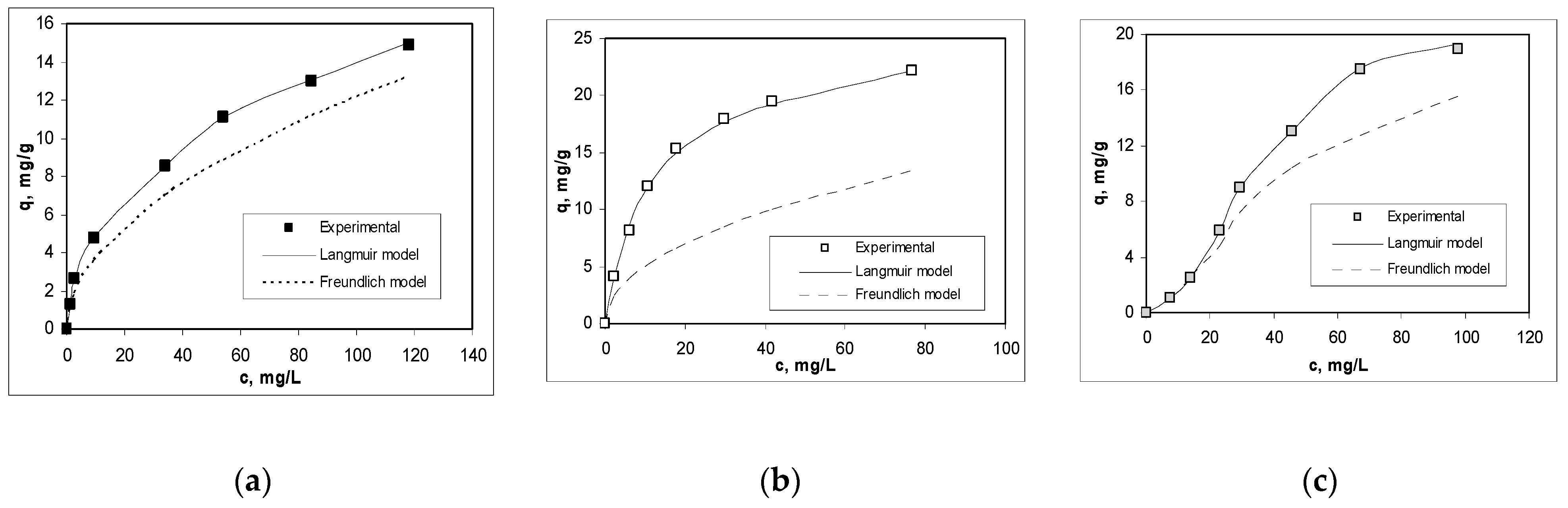
3.2. Effect of Initial Co(II) Concentration and Isotherms Modeling
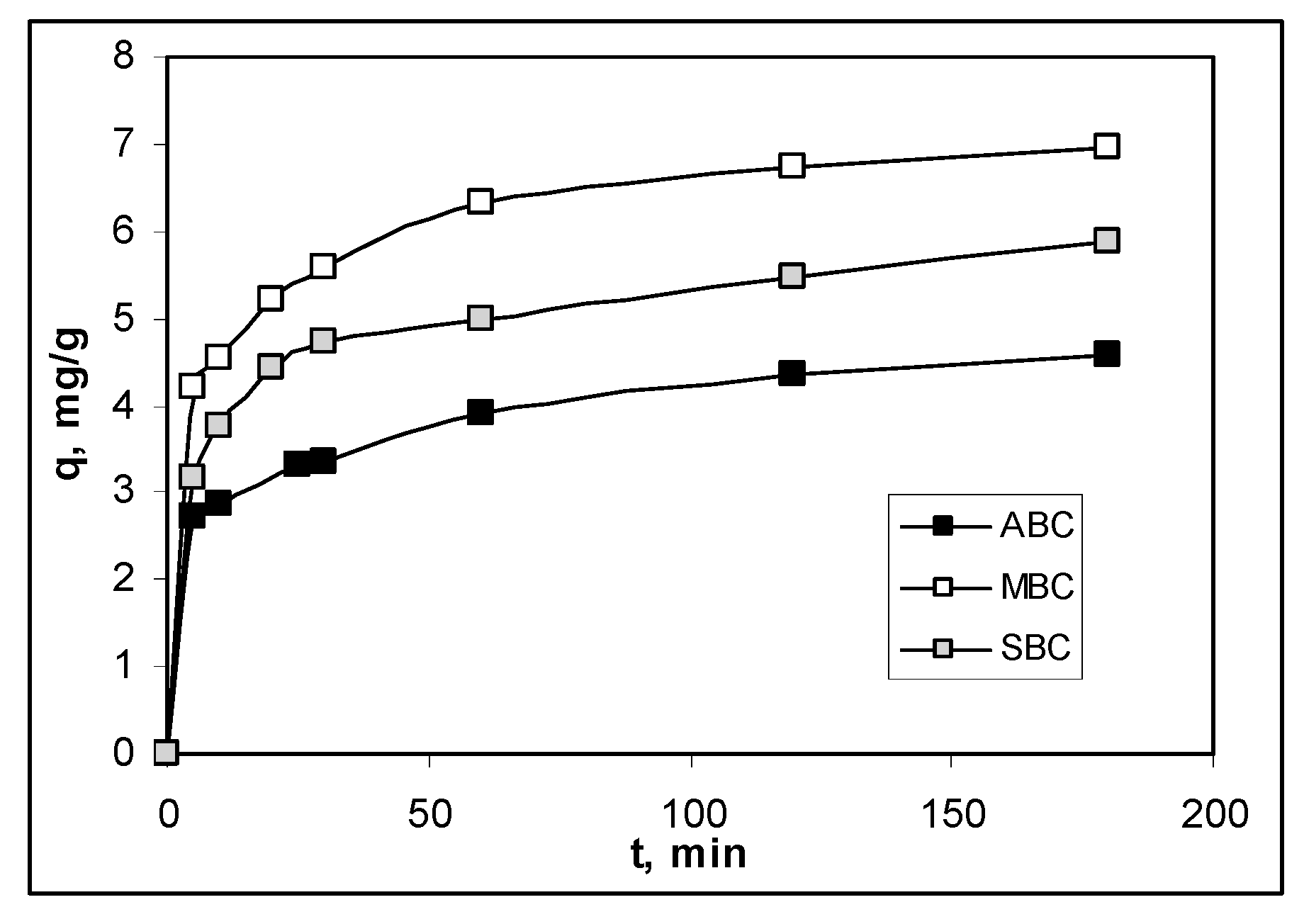
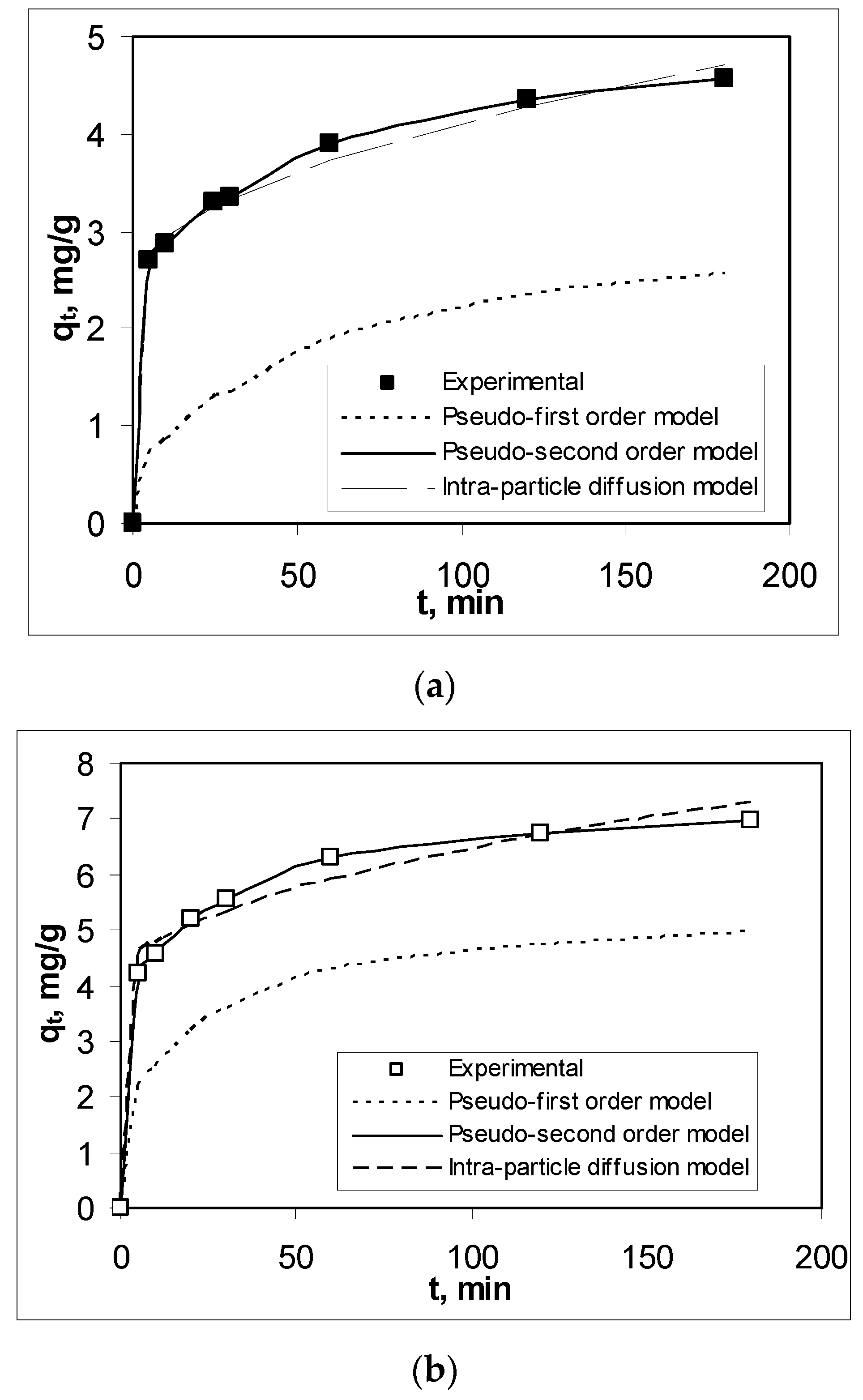
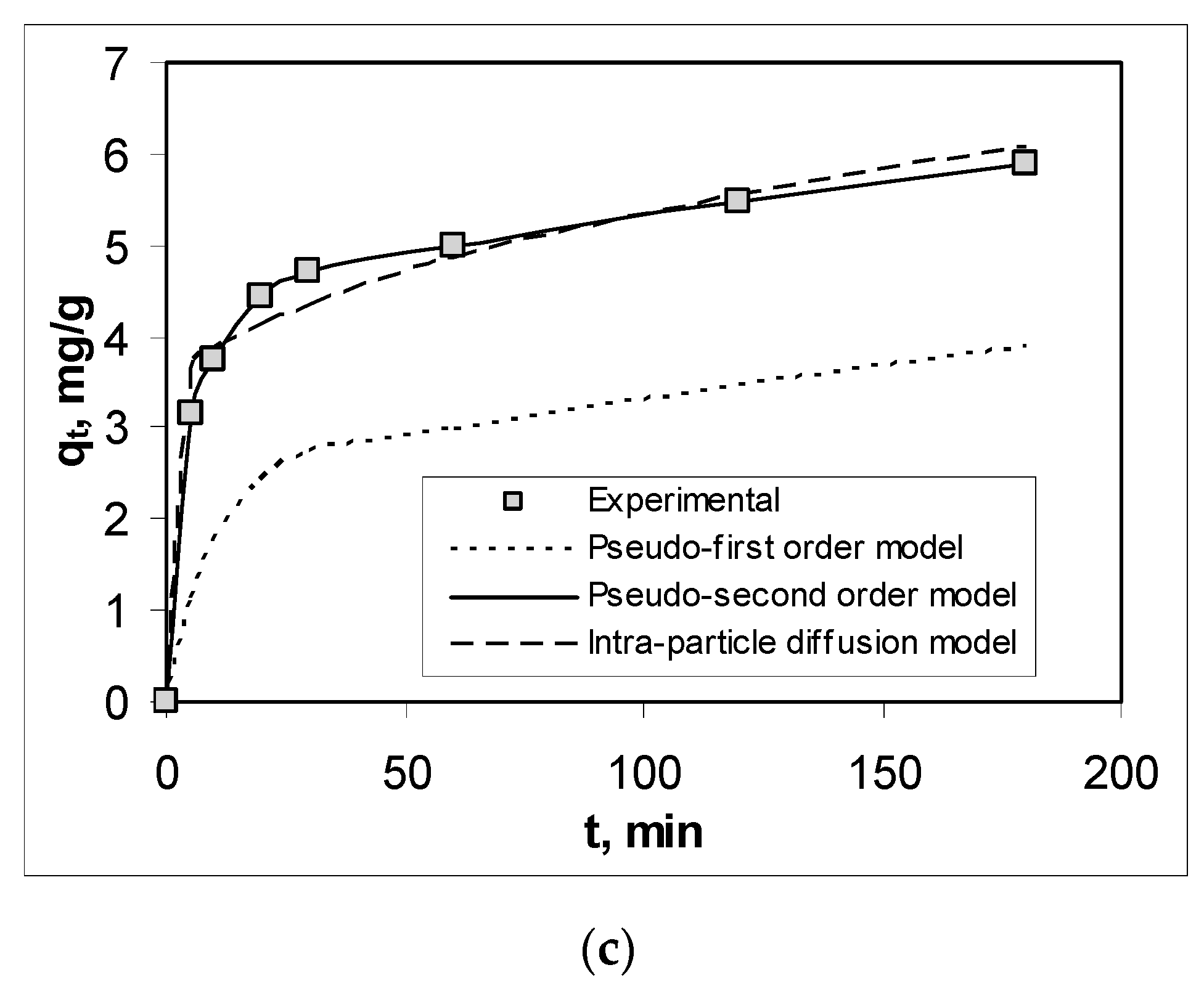
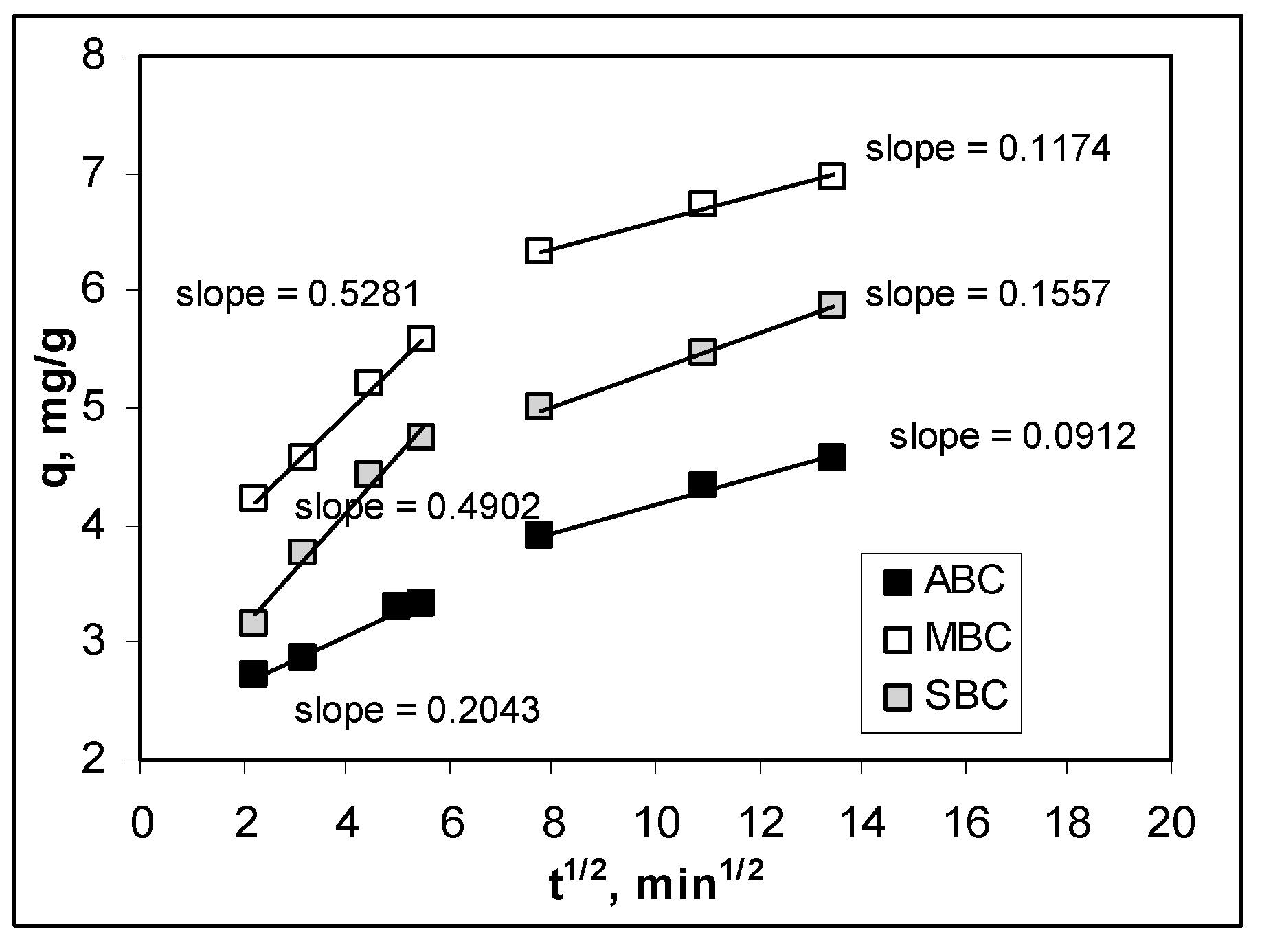
3.3. Effect of Contact Time and Kinetics Modeling
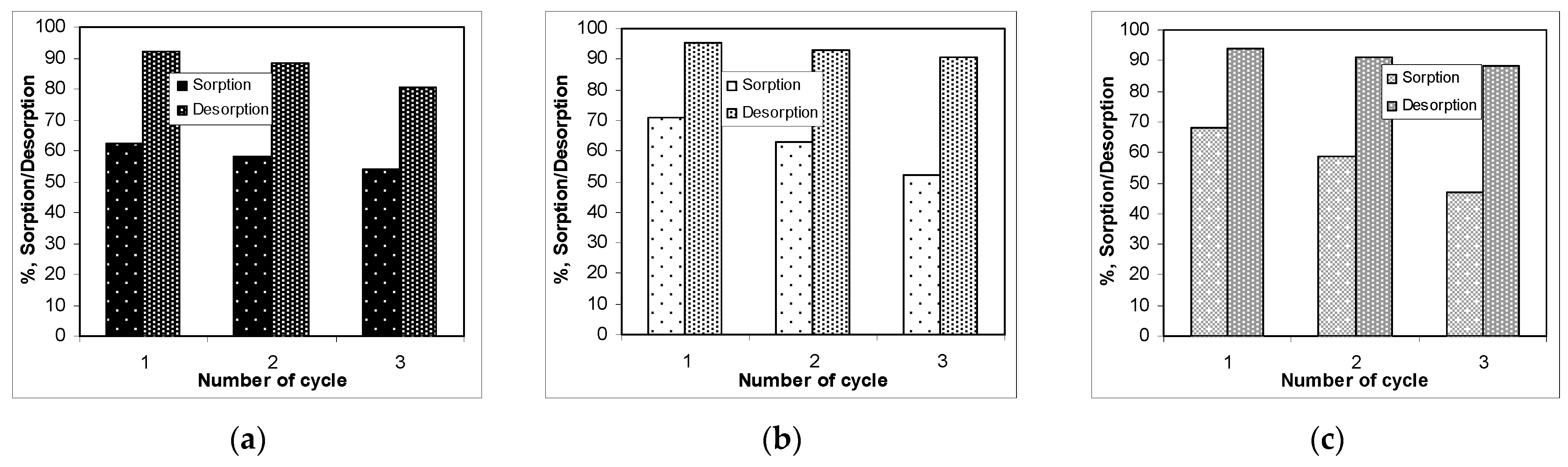
3.4. Recovery of Co(II) Ions and Biochars Regeneration
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Volesky, B. Detoxification of metal-bearing efluents biosorption for the next century. Hydrometallurgy 2015, 59, 2003–2216. [Google Scholar]
- Chen, G. Electrochemical technologies in wastewater treatment. Sep. Purif. Tehnol. 2004, 38, 11–41. [Google Scholar] [CrossRef]
- Aji, B.A.; Yavuz, Y.; Koparal, A.S. Electrocoagulation of heavy metals containing model wastewater using monopolar ion electronedes. Sep. Purif. Technol. 2012, 86, 248–254. [Google Scholar] [CrossRef]
- Parab, H.; Joshi, S.; Sudersanan, M.; Shenoy, N.; Lali, A.; Sarma, U. Removal and recovery of cobalt from aqueous solutions by adsorption using low cost lignocellulosic biomass-coir pith. J. Environ. Sci. Health Part A 2010, 45, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Barrio-Parra, F.; Elío, J.; De Miguel, E.; García-González, J.E.; Izquierdo, M.; Álvarez, R. Environmental risk assessment of cobalt and manganese from industrial sources in an estuarine system. Environ. Geochem. Health 2018, 40, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.I.; Arshad, N.; Ahmad, K.; Nadeem, M.; Ashfaq, A.; Wajid, K.; Bashir, H.; Munir, M.; Huma, B.; Memoona, H.; et al. Toxicological potential of cobalt in forage for ruminants grown in polluted soil: A health risk assessment from trace metal pollution for livestock. Environ. Sci. Poll. Res. 2019, 26, 15381–15389. [Google Scholar] [CrossRef] [PubMed]
- The World Health Report 2007—A Safer Future: Global Public Health Security in 21st Century World Health Organization. 2007. Available online: https://www.who.int/whr/2007/en/ (accessed on 17 April 2018).
- Abdel-Aziz, M.H.; Nirdosh, I.; Sedahmed, G.H. Ion-exchange-assisted electrochemical removal of heavy metals from dilute solutions in a stirred-tank electrochemical reactor: A mass-transfer study. Ind. Eng. Chem. Res. 2013, 52, 11655–11662. [Google Scholar] [CrossRef]
- Agwaramgbo, L.; Magee, N.; Nunez, S.K.; Mitt, K. Biosorption and chemical precipitation of lead using biomaterials, molecular sieves, and chlorides, carbonates, and sulfates of Na & Ca. J. Environ. Prot. 2013, 4, 1251–1257. [Google Scholar]
- Park, D.; Yun, Y.S.; Park, J.M. The past, present, and future trends of biosorption. Biotechnol. Bioproc. Eng. 2010, 15, 86–102. [Google Scholar] [CrossRef]
- Wang, J.L.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef] [PubMed]
- Michalak, I.; Chojnacka, K.; Witek-Krowiak, A. State of the Art for the Biosorption Process—A Review. Appl. Biochem. Biotechnol. 2013, 170, 1389–1416. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Nayak, A.; Agarwal, S. Bioadsorbents for remediation of heavy metals: Current status and their future prospects. Environ. Eng. Res. 2015, 20, 1–18. [Google Scholar] [CrossRef]
- Islam, M.A.; Morton, D.W.; Johnson, B.B.; Pramanik, B.K.; Mainali, B.; Angove, M.J. Opportunities and constraints of using the innovative adsorbents for the removal of cobalt (II) from wastewater: A review. Environ. Nanotechnol. Monit. Manag. 2018, 10, 435–456. [Google Scholar] [CrossRef]
- Swelam, A.A.; Awad, M.B.; Salem, A.M.A.; El-Feky, A.S. An economically viable method for the removal of cobalt ions from aqueous solution using raw and modified rice straw. HBRC J. 2018, 14, 255–262. [Google Scholar] [CrossRef]
- Femina Carolin, C.; Senthil Kumar, P.; Saravanan, A.; Joshiba, G.J.; Naushad, M. Efficient techniques for the removal of toxic heavy metals from aquatic environment: A review. J. Environ. Chem. Eng. 2017, 5, 2782–2799. [Google Scholar] [CrossRef]
- Deniz, F.; Karabulut, A. Biosorption of heavy metal ions by chemically modified biomass of coastal seaweed community: Studies on phycoremediation system modeling and design. Ecol. Eng. 2017, 106, 101–108. [Google Scholar] [CrossRef]
- Bulgariu, L.; Bulgariu, D. Functionalized soy waste biomass—A novel environmental-friendly biosorbent for the removal of heavy metals from aqueous solution. J. Clean. Prod. 2018, 197, 875–885. [Google Scholar] [CrossRef]
- Bird, M.; Wurster, C.M.; de Paula Silva, P.H.; Bass, A.M.; de Nys, R. Algal biochar-production and properties. Bioresour. Technol. 2011, 102, 1886–1891. [Google Scholar] [CrossRef]
- De Bhowmick, G.; Sarmah, A.K.; Sen, R. Production and characterization of a value added biochar mix using seaweed, rice husk and pine sawdust: A parametric study. J. Clean. Prod. 2018, 200, 641–656. [Google Scholar] [CrossRef]
- Chang, Y.M.; Tsai, W.T.; Li, M.H. Chemical characterization of char derived from slow pyrolysis of microalgal residue. J. Anal. Appl. Pyrol. 2015, 111, 88–93. [Google Scholar] [CrossRef]
- Huang, Q.; Song, S.; Chen, Z.; Hu, B.; Chen, J.; Wang, X. Biochar-based materials and their applications in removal of organic contaminants from wastewater: State-of-the-art review. Biochar 2019, 1, 45–73. [Google Scholar] [CrossRef]
- Yu, K.L.; Beng, F.L.; Show, P.L.; Ong, H.C.; Ling, T.C.; Chen, W.H.; Ng, E.P.; Chang, J.S. Recent developments on algal biochar production a characterization. Bioresour. Technol. 2017, 246, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Gao, B.; Mosa, A.; Yu, H.; Yin, X.; Bashir, A.; Ghoveisi, H.; Wang, S. Removal of Cu (II), Cd (II) and Pb (II) ions from aqueous solutions by biochars derived from potassium-rich biomass. J. Clean. Prod. 2018, 10, 437–449. [Google Scholar] [CrossRef]
- Nacu, G.; Bulgariu, D.; Harja, M.; Popescu, C.M.; Juravle, D.T.; Bulgariu, L. Removal of Zn (II) ions from aqueous media on thermal activated sawdust Desalin. Water Treat. 2016, 57, 21904–21915. [Google Scholar] [CrossRef]
- Ibrahim, W.M.; Hassan, A.F.; Azab, Y.A. Biosorption of toxic heavy metals from aqueous solution by Ulva lactuca activated carbon. Egypt. J. Basic Appl. Sci. 2016, 3, 241–249. [Google Scholar] [CrossRef]
- Nemeş, L.; Bulgariu, L. Optimization of process parameters for heavy metals biosorption onto mustard waste biomass. Open Chem. 2016, 14, 175–187. [Google Scholar] [CrossRef]
- Rangabhashiyam, S.; Anu, N.; Nandagopal Giri, M.S.; Selvaraju, N. Relevance of isotherm models in biosorption of pollutants by agricultural by-products. J. Environ. Chem. Eng. 2014, 2, 398–414. [Google Scholar] [CrossRef]
- Yousef, N.S.; Farouq, R.; Hazza, R. Adsorption kinetics and isotherms for the removal of nickel ions from aqueous solutions by an ion-exchange resin: Application of two and three parameter isotherm models. Desalin. Water Treat. 2016, 57, 21925–21938. [Google Scholar] [CrossRef]
- Zakhama, S.; Dhaouadi, H.; M’Henni, F. Nonlinear modelisation of heavy metal removal from aqueous solution using Ulva lactuca algae. Bioresour. Technol. 2011, 102, 786–796. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Pseudo-second-order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Cha, J.S.; Park, S.H.; Jung, S.C.; Ryu, C.; Jeon, J.K.; Shin, M.C.; Park, Y.K. Production and utilization of biochar: A review. J. Ind. Eng. Chem. 2016, 40, 1–15. [Google Scholar] [CrossRef]
- Chemerys, V.; Baltrėnaitė, E. A review of lignocellulosic biochar modification towards enhanced biochar selectivity and adsorption capacity of potentially toxic elements. Ukranian J. Ecol. 2018, 8, 21–32. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, J.; Liu, H.; Kang, Y.; Yu, J.; Zhang, C. Optimization of the green and low-cost ammoniation-activation method to produce biomass-based activated carbon for Ni (II) removal from aqueous solutions. J. Clean. Prod. 2017, 159, 38–46. [Google Scholar] [CrossRef]
- Yang, T.; Lua, A.C. Characteristics of activated carbons prepared from pistachio-nut shells by physical activation. J. Colloid Interface Sci. 2003, 267, 408–417. [Google Scholar] [CrossRef]
- Escudero, L.B.; Quintas, P.Y.; Wuilloud, R.G.; Dotto, G.L. Recent advances on elemental biosorption. Environ. Chem. Lett. 2019, 17, 409–427. [Google Scholar] [CrossRef]
- Cheung, W.H.; Szeto, Y.S.; McKay, G. Intra-particle diffusion processes during acid dye adsorption onto chitosan. Bioresour. Technol. 2007, 98, 2897–2904. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, S.; Guo, X.; Huang, H. Adsorption of chromium (III) on lignin. Bioresour. Technol. 2008, 99, 7709–7715. [Google Scholar] [CrossRef]
- Bădescu, I.S.; Bulgariu, D.; Ahmad, I.; Bulgariu, L. Valorisation possibilities of exhausted biosorbents loaded with metal ions—A review. J. Environ. Manag. 2018, 224, 288–297. [Google Scholar] [CrossRef]

| Biomass Waste | Algae Waste Biomass | Mustard Waste Biomass | Soy Waste Biomass |
|---|---|---|---|
| Pyrolysis temperature, °C | 600 | 650 | 650 |
| Mass decrease, % | 40.08 | 53.17 | 49.82 |
| Pores volume, m2/g | 0.31 | 0.40 | 0.37 |
| BET surface area, m2/g | 289.70 | 354.07 | 329.93 |
| Biochar | ABC | MBC | SBC | |
|---|---|---|---|---|
| Langmuir model | R2 | 0.9924 | 0.9975 | 0.9983 |
| qmax, mg/g | 11.9047 | 24.2131 | 19.6078 | |
| KL, g/L | 0.1034 | 0.0921 | 0.0983 | |
| Freundlich model | R2 | 0.9872 | 0.9465 | 0.9524 |
| 1/n | 0.5104 | 0.4766 | 0.5231 | |
| KF, g/L | 1.1559 | 1.6932 | 1.4132 | |
| Biochar | ABC | MBC | SBC | |
|---|---|---|---|---|
| qe,exp, mg/g | 4.5705 | 6.9742 | 5.8794 | |
| Pseudo-first order model | R2 | 0.8244 | 0.8976 | 0.8927 |
| qe, mg/g | 0.7037 | 2.7931 | 2.2182 | |
| k1, 1/min | 0.0054 | 0.0092 | 0.0074 | |
| Pseudo-second order model | R2 | 0.9997 | 0.9992 | 0.9997 |
| qe, mg/g | 4.7281 | 7.1684 | 5.9880 | |
| k2, g/mg min | 0.0235 | 0.0211 | 0.0223 | |
| Intra-particle diffusion model | R2 | 0.9695 | 0.9279 | 0.8972 |
| c, mg/L | 2.3902 | 3.9756 | 3.1604 | |
| kdiff, mg/g min1/2 | 0.1729 | 0.2475 | 0.2165 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucaci, A.R.; Bulgariu, D.; Ahmad, I.; Lisă, G.; Mocanu, A.M.; Bulgariu, L. Potential Use of Biochar from Various Waste Biomass as Biosorbent in Co(II) Removal Processes. Water 2019, 11, 1565. https://doi.org/10.3390/w11081565
Lucaci AR, Bulgariu D, Ahmad I, Lisă G, Mocanu AM, Bulgariu L. Potential Use of Biochar from Various Waste Biomass as Biosorbent in Co(II) Removal Processes. Water. 2019; 11(8):1565. https://doi.org/10.3390/w11081565
Chicago/Turabian StyleLucaci, Alina Roxana, Dumitru Bulgariu, Iftikhar Ahmad, Gabriela Lisă, Anca Mihaela Mocanu, and Laura Bulgariu. 2019. "Potential Use of Biochar from Various Waste Biomass as Biosorbent in Co(II) Removal Processes" Water 11, no. 8: 1565. https://doi.org/10.3390/w11081565
APA StyleLucaci, A. R., Bulgariu, D., Ahmad, I., Lisă, G., Mocanu, A. M., & Bulgariu, L. (2019). Potential Use of Biochar from Various Waste Biomass as Biosorbent in Co(II) Removal Processes. Water, 11(8), 1565. https://doi.org/10.3390/w11081565






