Legionella pneumophila as a Health Hazard to Miners: A Pilot Study of Water Quality and QMRA
Abstract
1. Introduction
2. Methods
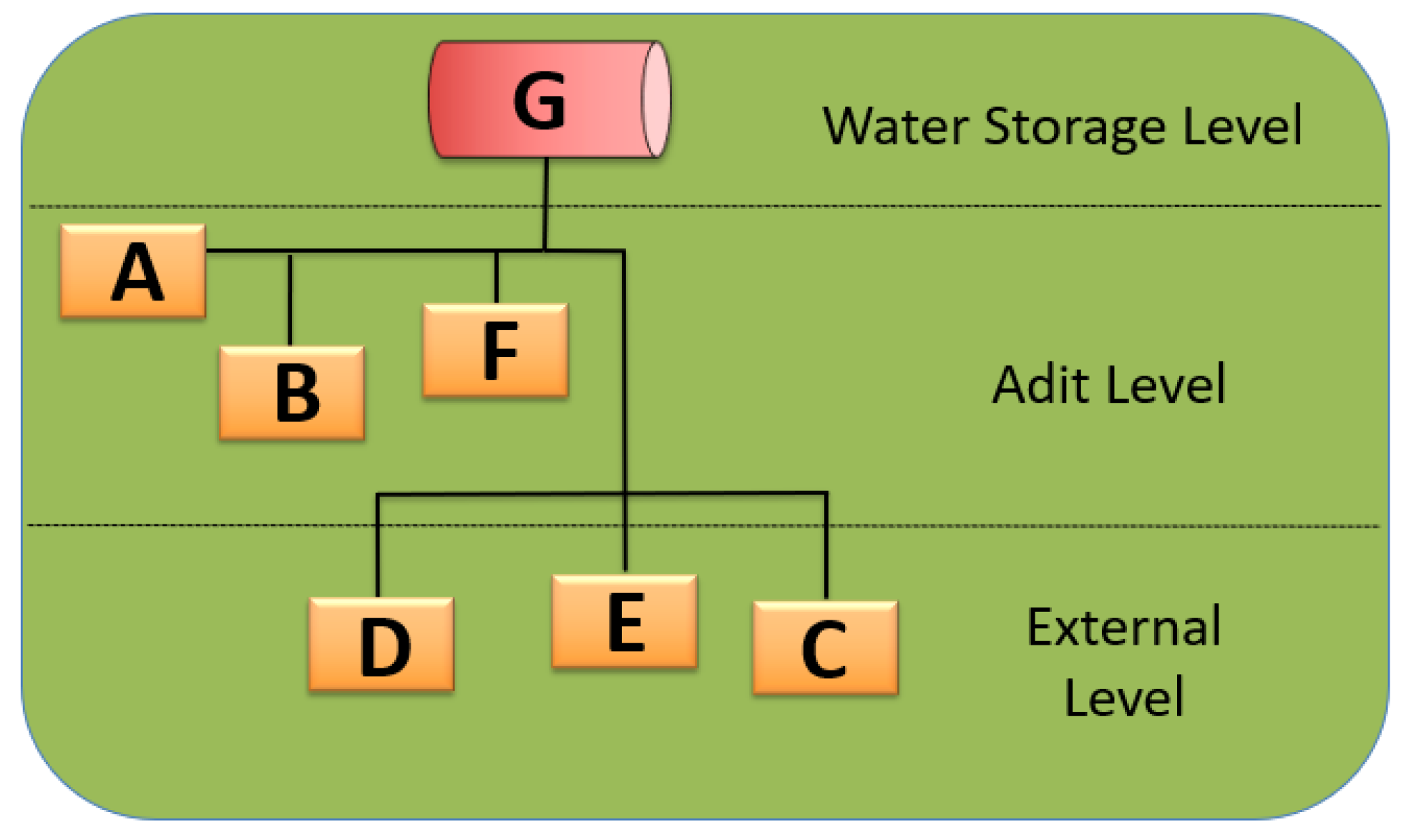
2.1. Site Description Section
2.2. Sample Collection
2.3. Laboratory Analysis
2.4. Data Analysis
2.5. QMRA
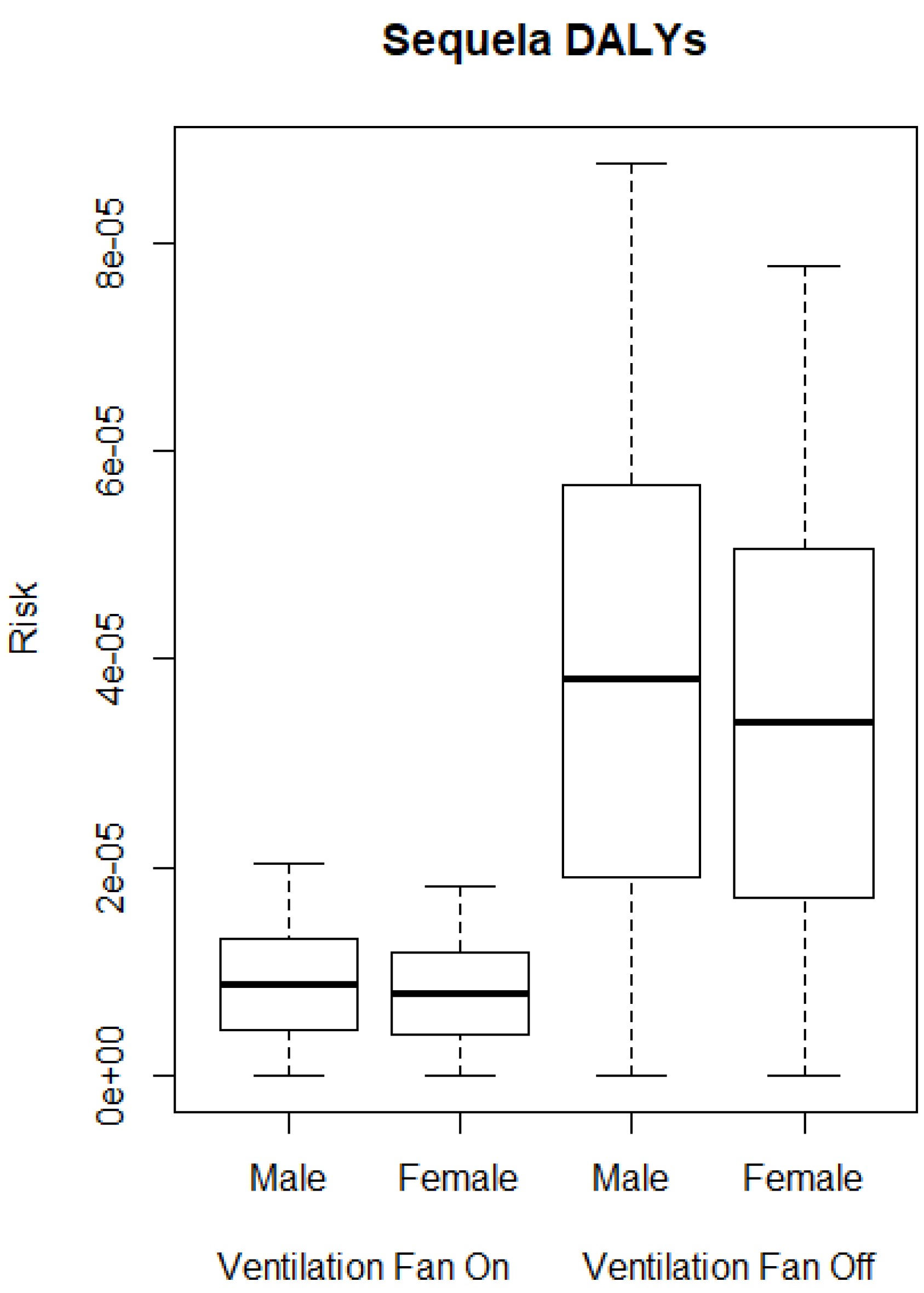
3. Results
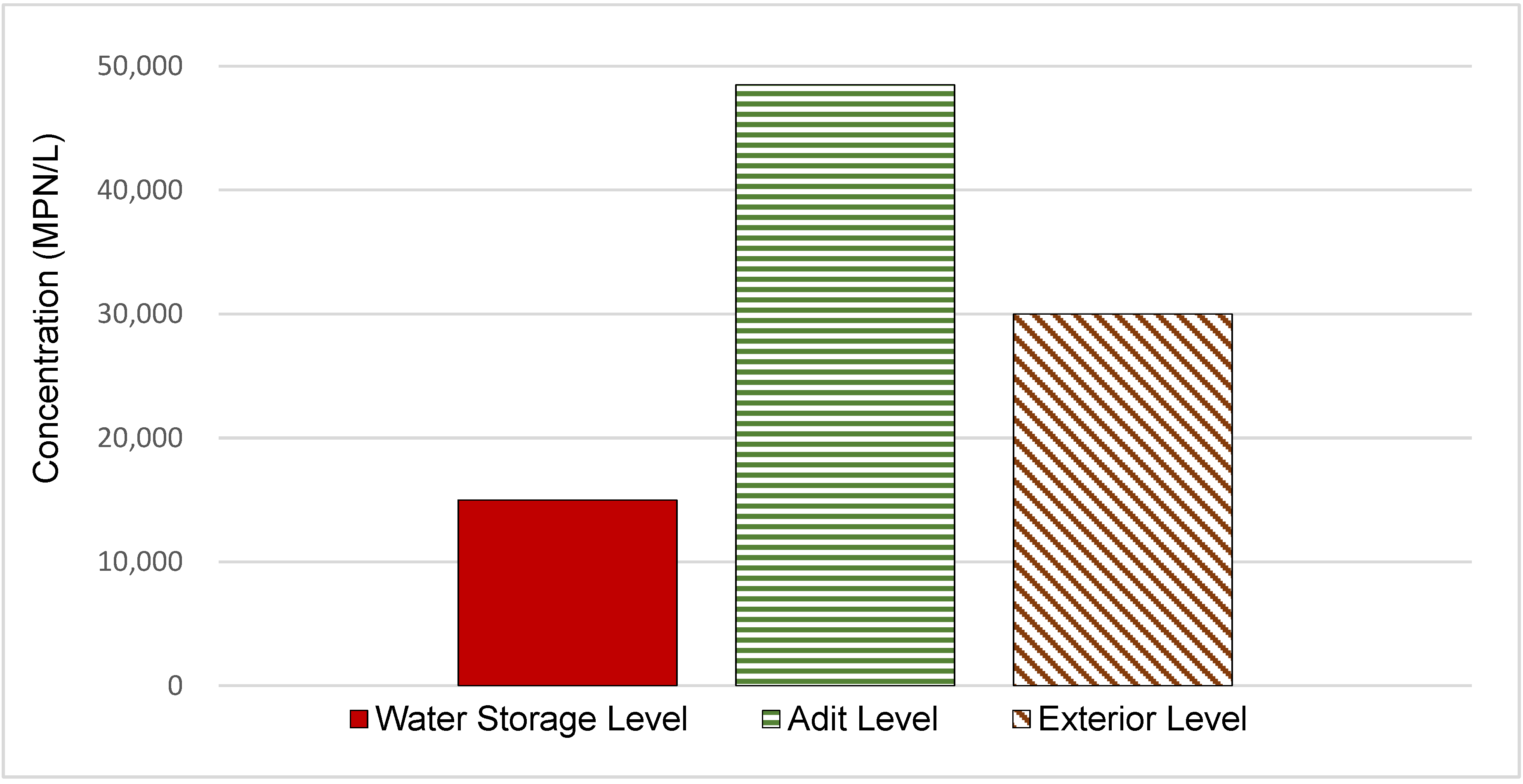
3.1. Characterization of Legionella pneumophila Concentration
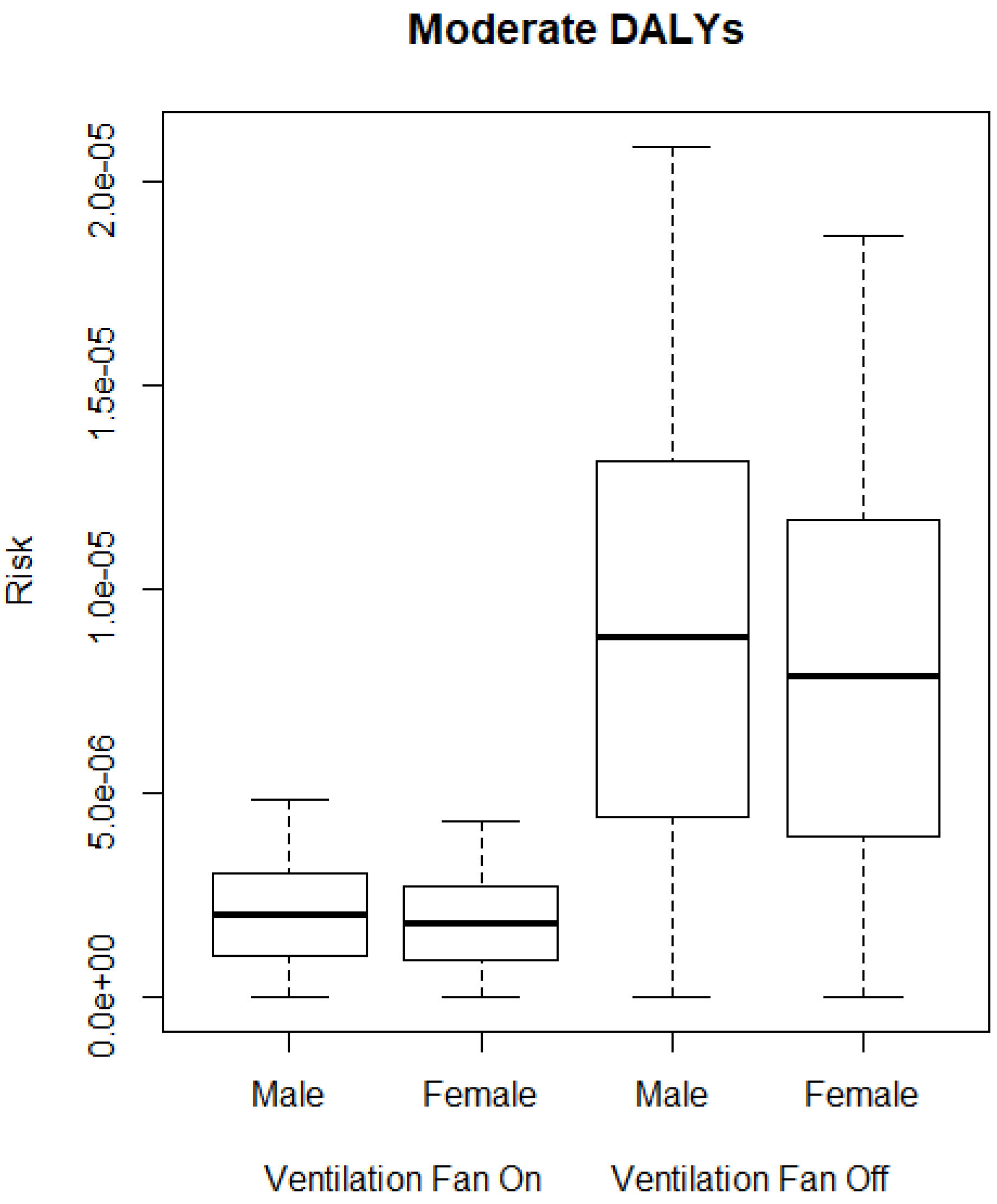
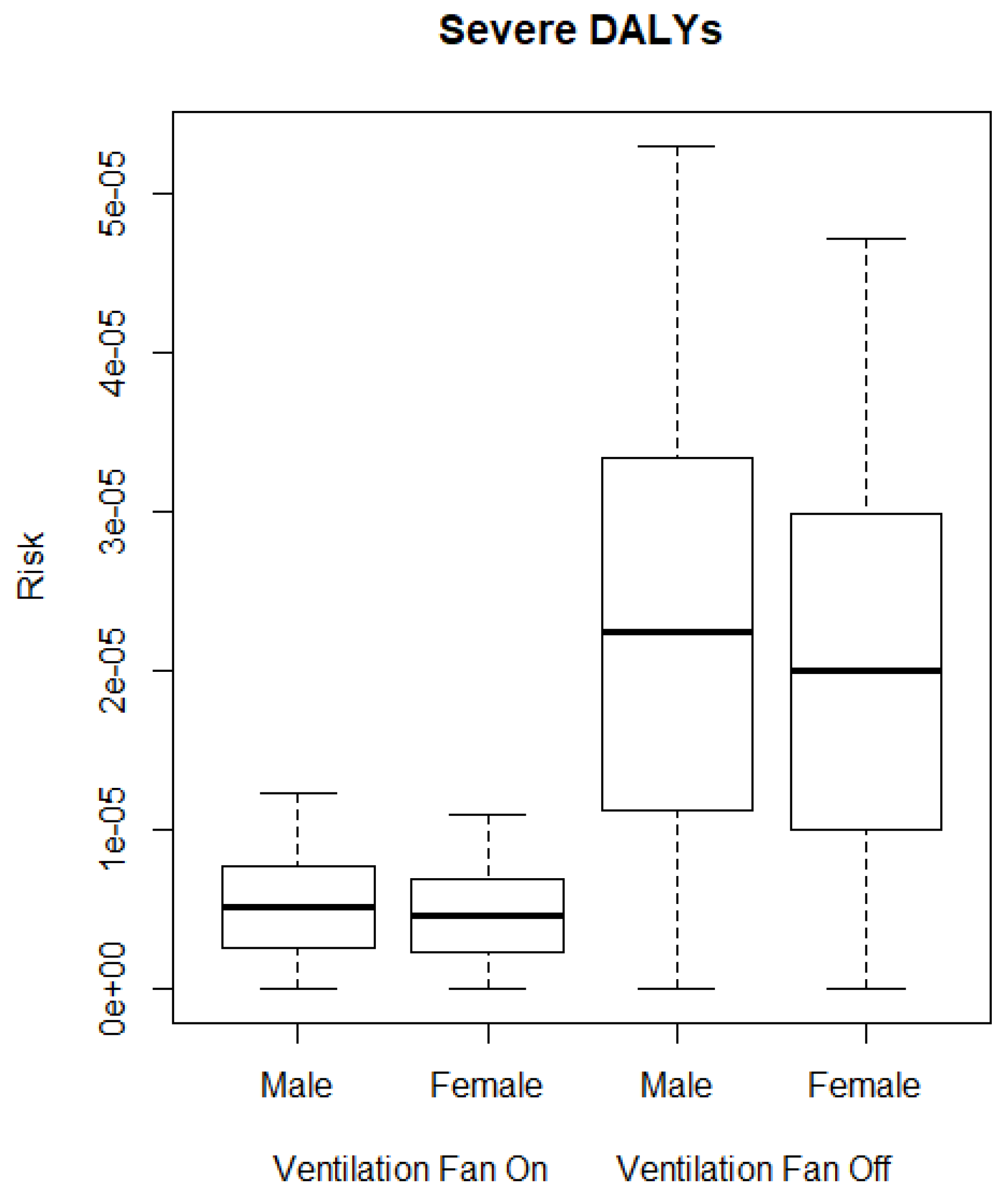
3.2. QMRA Model
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pruckler, J.M.; Forster, T.S.; Donlan, R.M.; Brown, E.; Murga, R.; Fields, B.S. Role of biofilms in the survival of Legionella pneumophila in a model potable-water system. Microbiology 2001, 147, 3121–3126. [Google Scholar]
- Fields, B.S.; Benson, R.F.; Besser, R.E. Legionella and Legionnaires’ Disease: 25 Years of Investigation. Clin. Microbiol. Rev. 2002, 15, 506–526. [Google Scholar] [CrossRef]
- Leoni, E.; De Luca, G.; Legnani, P.; Sacchetti, R.; Stampi, S.; Zanetti, F. Legionella waterline colonization: Detection of Legionella species in domestic, hotel and hospital hot water systems. J. Appl. Microbiol. 2005, 98, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Van Der Kooij, D.; Veenendaal, H.R.; Scheffer, W.J. Biofilm formation and multiplication of Legionella in a model warm water system with pipes of copper, stainless steel and cross-linked polyethylene. Water Res. 2005, 39, 2789–2798. [Google Scholar] [CrossRef] [PubMed]
- Hines, S.A.; Chappie, D.J.; Lordo, R.A.; Miller, B.D.; Janke, R.J.; Lindquist, H.A.; Fox, K.R.; Ernst, H.S.; Taft, S.C. Assessment of relative potential for Legionella species or surrogates inhalation exposure from common water uses. Water Res. 2014, 56, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Mathys, W.; Stanke, J.; Harmuth, M.; Junge-Mathys, E. Occurrence of Legionella in hot water systems of single-family residences in suburbs of two German cities with special reference to solar and district heating. Int. J. Hyg. Environ. Health 2008, 211, 179–185. [Google Scholar] [CrossRef]
- Sungur, E.I.; Cotuk, A. Enumeration of Legionella pneumophila in cooling tower water systems. Environ. Monit. Assess. 2005, 100, 53–58. [Google Scholar]
- Wadowsky, R.M.; Wolford, R.; McNamara, A.M.; Yee, R.B. Effect of temperature, pH, and oxygen level on the multiplication of naturally occurring Legionella pneumophila in potable water. Appl. Environ. Microbiol. 1985, 49, 1197–1205. [Google Scholar]
- Agresta, M.D.; Hanley, E.M.; Urquhart, K.; Schoonmaker, D.; Venezia, R.A. Nosocomial Legionellosis Associated with Aspiration of Nasogastric Feedings Diluted in Tap Water. Infect. Control Hosp. Epidemiol. 1994, 15, 529–533. [Google Scholar]
- Edwards, M.L.; Smith, D.W.; Muller, D. Changes in Iron and Transferrin Levels and Body Temperature in Experimental Airborne Legionellosis. J. Infect. Dis. 1983, 147, 302–307. [Google Scholar]
- Fitzgeorge, R.B.; Baskerville, A.; Broster, M.; Hambleton, P.; Dennis, P.J. Aerosol infection of animals with strains of Legionella pneumophila of different virulence: Comparison with intraperitoneal and intranasal routes of infection. J. Hyg. 1983, 90, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Anderson, J.; Mueller, S.; Gaines, W.; Kendall, A. Literature review—Efficacy of various disinfectants against Legionella in water systems. Water Res. 2002, 36, 4433–4444. [Google Scholar] [CrossRef]
- Prostański, D. Use of Air-and-Water Spraying Systems for Improving Dust Control in Mines. J. Sustain. Min. 2013, 12, 29–34. [Google Scholar] [CrossRef]
- Shevenell, L.; A Connors, K.; Henry, C.D. Controls on pit lake water quality at sixteen open-pit mines in Nevada. Appl. Geochem. 1999, 14, 669–687. [Google Scholar] [CrossRef]
- Bartie, C.; Klugman, K.P. Exposures to Legionella pneumophila and Chlamydia pneumoniae in South African Mine Workers. Int. J. Occup. Environ. Health 1997, 3, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.H.; Hill, E.C.; Howells, C.H.; Ribeiro, C.D. Legionella pneumophila in coal miners. Occup. Environ. Med. 1985, 42, 421–425. [Google Scholar] [CrossRef]
- Haas, C.N.; Rose, J.B.; Gerba, C.P. Quantitative Microbial Risk Assessment; Wiley: New York, NY, USA, 1999. [Google Scholar]
- Schoen, M.E.; Ashbolt, N.J. An in-premise model for Legionella exposure during showering events. Water Res. 2011, 45, 5826–5836. [Google Scholar] [CrossRef]
- Rose, J.B. Future health assessment and risk-management integration for infectious diseases and biological weapons for deployed US forces. In Strategies to Protect the Health of Deployed U.S. Forces: Assessing Health Risks to Deployed U.S. Forces: Workshop Proceedings; National Academies Press: Washington, DC, USA, 2000; pp. 59–112. [Google Scholar]
- Weir, M.H. Dose-response modeling and use: Challenges and uncertainties in environmental exposure. In Manual of Environmental Microbiology, 4th ed.; American Society of Microbiology: Washington, DC, USA, 2016. [Google Scholar]
- CDC. Sampling Procedure and Potential Sampling Sites. Available online: https://www.cdc.gov/legionella/health-depts/environmental-inv-resources.html (accessed on 9 February 2017).
- IDEXX. Legiolert. Available online: https://idexxcom-live-b02da1e51e754c9cb292133b-9c56c33.aldryn-media.com/filer_public/cb/ee/cbee9faf-58a4-4068-8dce-077e3e21da86/legiolert-procedure-insert.pdf (accessed on 26 March 2017).
- R-Core-Team. R: A Language and Environment for Statistical Computing; The R Development Core Team: Vienna, Austria, 2008. [Google Scholar]
- Kuiper, M.W.; Wullings, B.A.; Akkermans, A.D.L.; Beumer, R.R.; Van Der Kooij, D. Intracellular Proliferation of Legionella pneumophila in Hartmannella vermiformis in Aquatic Biofilms Grown on Plasticized Polyvinyl Chloride. Appl. Environ. Microbiol. 2004, 70, 6826–6833. [Google Scholar] [CrossRef]
- National-Center-for-Environmental-Assessment. Exposure Factors Handbook, 2011 ed.; National Center for Environmental Assessment; Office of Research and Development; U.S. Environmental Protection Agency: Washington, DC, USA, 2011.
- Chattopadhyay, S.; Perkins, S.D.; Shaw, M.; Nichols, T.L. Evaluation of Exposure to Brevundimonas diminuta and Pseudomonas aeruginosa during Showering. J. Aerosol Sci. 2017, 114, 77–93. [Google Scholar] [CrossRef]
- Zhou, Y.; Benson, J.M.; Irvin, C.; Irshad, H.; Cheng, Y.-S. Particle Size Distribution and Inhalation Dose of Shower Water under Selected Operating Conditions. Inhal. Toxicol. 2007, 19, 333–342. [Google Scholar] [CrossRef]
- Armstrong, T.W.; Haas, C.N. Legionnaires’ disease: Evaluation of a quantitative microbial risk assessment model. J. Water Health 2008, 6, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Haagsma, J.A.; Graetz, N.; Bolliger, I.; Naghavi, M.; Higashi, H.; Mullany, E.C.; Abera, S.F.; Abraham, J.P.; Adofo, K.; Alsharif, U.; et al. The global burden of injury: Incidence, mortality, disability-adjusted life years and time trends from the Global Burden of Disease study 2013. Inj. Prev. 2016, 22, 3–18. [Google Scholar] [CrossRef] [PubMed]
- CDC. Legionellosis—United States, 2000–2009; 0149-2195; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2011; pp. 1083–1086.
- Aminossadati, S.; Hooman, K. Numerical Simulation of Ventilation Air Flow in Underground Mine Workings; The University of Nevada: Reno, NV, USA, 2008. [Google Scholar]
- De Souza, E. Application of ventilation management programs for improved mine safety. Int. J. Min. Sci. Technol. 2017, 27, 647–650. [Google Scholar] [CrossRef]
- Tickner, J.A.; Ager, B.P. The control of microbiological hazards associated with air-conditioning and ventilation systems. Ann. Occup. Hyg. 1983, 27, 341–358. [Google Scholar]
- Dondero, T.J.; Rendtorff, R.C.; Mallison, G.F.; Weeks, R.M.; Levy, J.S.; Wong, E.W.; Schaffner, W. An Outbreak of Legionnaires’ Disease Associated with a Contaminated Air-Conditioning Cooling Tower. N. Engl. J. Med. 1980, 302, 365–370. [Google Scholar] [CrossRef]
- Ambrose, J.; Hampton, L.M.; Fleming-Dutra, K.E.; Marten, C.; McClusky, C.; Perry, C.; Clemmons, N.A.; McCormic, Z.; Peik, S.; Mancuso, J.; et al. Large outbreak of Legionnaires’ disease and Pontiac fever at a military base. Epidemiol. Infect. 2014, 142, 2336–2346. [Google Scholar] [CrossRef]
- Den Boer, J.W.; Yzerman, E.P.F.; Schellekens, J.; Lettinga, K.D.; Boshuizen, H.C.; Van Steenbergen, J.E.; Bosman, A.; Van den Hof, S.; Van Vliet, H.A.; Peeters, M.F.; et al. A large outbreak of Legionnaires’ disease at a flower show, the Netherlands, 1999. Emerg. Infect. Dis. 2002, 8, 37–43. [Google Scholar] [CrossRef]
- Gilmour, M.W.; Bernard, K.; Tracz, D.M.; Olson, A.B.; Corbett, C.R.; Burdz, T.; Ng, B.; Wiebe, D.; Broukhanski, G.; Boleszczuk, P.; et al. Molecular typing of a Legionella pneumophila outbreak in Ontario, Canada. J. Med. Microbiol. 2007, 56, 336–341. [Google Scholar] [CrossRef][Green Version]
- Principe, L.; Tomao, P.; Visca, P. Legionellosis in the occupational setting. Environ. Res. 2017, 152, 485–495. [Google Scholar] [CrossRef]
- Berjeaud, J.-M.; Chevalier, S.; Schlusselhuber, M.; Portier, E.; Loiseau, C.; Aucher, W.; Lesouhaitier, O.; Verdon, J. Legionella pneumophila: The Paradox of a Highly Sensitive Opportunistic Waterborne Pathogen Able to Persist in the Environment. Front. Microbiol. 2016, 7, 21660. [Google Scholar] [CrossRef]
- Occupational-Safety-and-Health-Administration. Legionellosis (Legionnaire’s Disease and Pontiac Fever). Available online: https://www.osha.gov/SLTC/legionnairesdisease/standards.html (accessed on 26 August 2018).
| Label | Variable | Value | Unit | Distribution | Reference |
|---|---|---|---|---|---|
| CL,S | Concentration of L. pneumophila in non-potable water from the sprinkler system | 8.35 × 10−4 | Most probable number (MPN)/L | N/A | Results of this study |
| PC | Air partitioning coefficient | 10−5 | Unitless | N/A | [26] |
| VS | Water volume released from the sprinkler system | 573 | L/s | N/A | Provided by the mine |
| FA | Fraction of aerosolized organisms | Mean: 0.0337 Std: 0.0098 | Unitless | Normal | [27] |
| Vm,air | Area of the mine | 3.0 × 10−4 | L | N/A | Provided by the mine |
| Vv | Volume of air removed by the ventilation fan | 5663 | L/s | N/A | Provided by the mine |
| CL, JD | Concentration of L. pneumophila in non-potable water from the jackleg drill | 4.65 × 10−4 | MPN/L | N/A | Results of this study |
| VJD | Flow of water released from the jackleg drill | 573 | L/s | N/A | Provided by the mine |
| Imale | Inhalation rate for male workers | Mean: 0.02333 Std: 0.00434 | m3/min | Normal | [25] |
| Ifemale | Inhalation rate for female workers | Mean: 0.02083 Std: 0.0056 | m3/min | Normal | [25] |
| k | Probability of the pathogen surviving to initiate infection in the host | −0.00599 | Unitless | N/A | [28] |
| DWPF | Disability weight for Pontiac Fever (moderate) | Mean: 0.051 Lower: 0.039 Upper: 0.06 | Unitless | Triangular | [29] |
| DWLD | Disability weight for Legionnaires’ disease (severe) | Mean: 0.125 Lower: 0.104 Upper: 0.152 | Unitless | Triangular | [29] |
| DWPC | Disability weight for sequela (post-acute) | Mean: 0.217 Lower: 0.179 Upper: 0.251 | Unitless | Triangular | [29] |
| Sampling Location | Water temperature (°C) | Chlorine levels (mg/L) | Average Concentration of L. pneumophila (MPN/L) | Categories of Sampling Location by Mine Levels |
|---|---|---|---|---|
| Non-potable water tank | 23.6 | ND | 1.50 × 104 | Water Storage Level |
| Jackleg drill connection | 21.6 | ND | 4.65 × 104 | Adit Level |
| Sprinkler system | 21.1 | ND | 8.35 × 104 | Adit Level |
| Jackleg drill hose outside adit level | 30.9 | ND | 1.55 × 104 | Adit Level |
| Sink in hoist house | 19.0 | 0.05 | 2.50 × 104 | Exterior Level |
| Swamp cooler | 16.8 | 0.05 | 4.00 × 104 | Exterior Level |
| Jackleg drill hose in hoist house | 23.3 | ND | 2.50 × 104 | Exterior Level |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madera-García, V.; Mraz, A.L.; López-Gálvez, N.; Weir, M.H.; Werner, J.; Beamer, P.I.; Verhougstraete, M.P. Legionella pneumophila as a Health Hazard to Miners: A Pilot Study of Water Quality and QMRA. Water 2019, 11, 1528. https://doi.org/10.3390/w11081528
Madera-García V, Mraz AL, López-Gálvez N, Weir MH, Werner J, Beamer PI, Verhougstraete MP. Legionella pneumophila as a Health Hazard to Miners: A Pilot Study of Water Quality and QMRA. Water. 2019; 11(8):1528. https://doi.org/10.3390/w11081528
Chicago/Turabian StyleMadera-García, Valerie, Alexis L. Mraz, Nicolás López-Gálvez, Mark H. Weir, James Werner, Paloma I. Beamer, and Marc P. Verhougstraete. 2019. "Legionella pneumophila as a Health Hazard to Miners: A Pilot Study of Water Quality and QMRA" Water 11, no. 8: 1528. https://doi.org/10.3390/w11081528
APA StyleMadera-García, V., Mraz, A. L., López-Gálvez, N., Weir, M. H., Werner, J., Beamer, P. I., & Verhougstraete, M. P. (2019). Legionella pneumophila as a Health Hazard to Miners: A Pilot Study of Water Quality and QMRA. Water, 11(8), 1528. https://doi.org/10.3390/w11081528






