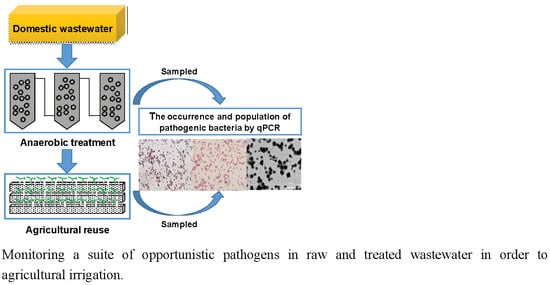
Monitoring Opportunistic Pathogens in Domestic Wastewater from a Pilot-Scale Anaerobic Biofilm Reactor to Reuse in Agricultural Irrigation
Abstract
1. Introduction
2. Materials and Methods
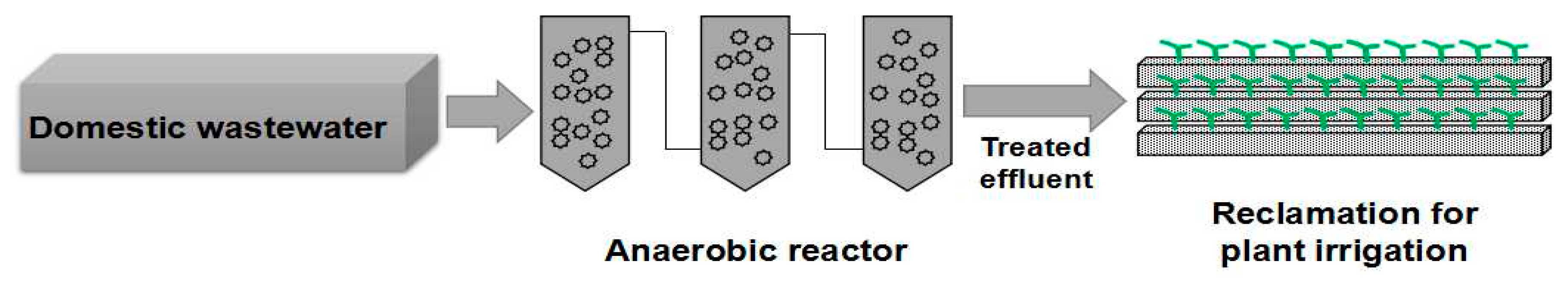
2.1. Reactor Operation and Experimental Design
2.2. Sample Collection, Processing, and DNA Extraction
2.3. Preparation of qPCR Standard Curves
2.4. Quantitative PCR Assay
2.5. Data Analysis
3. Results
3.1. Operational Performance of the Reactor
3.2. Physicochemical Properties of Soils and Lettuces
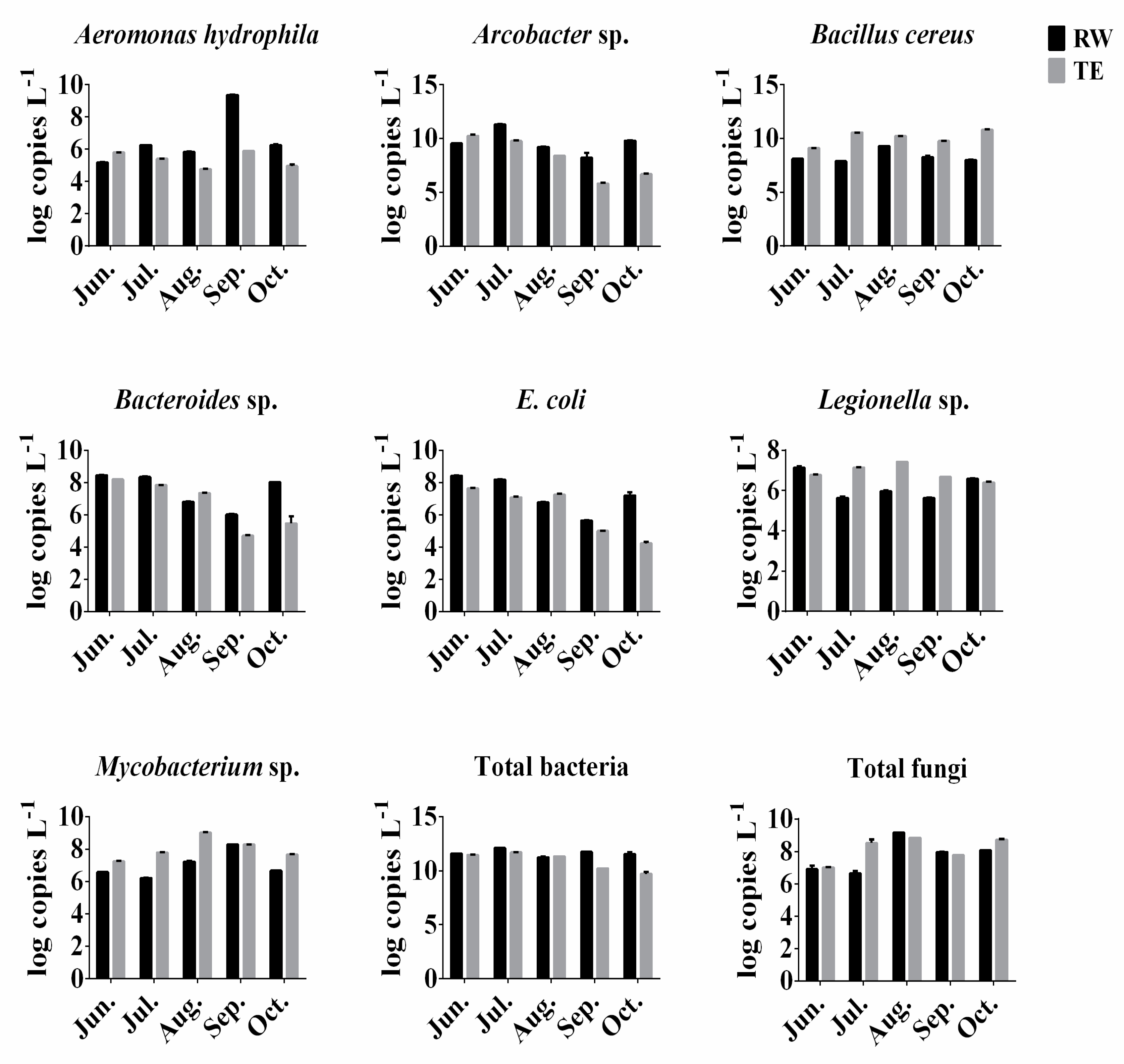
3.3. Abundances of Selected Opportunistic Pathogens in Wastewater
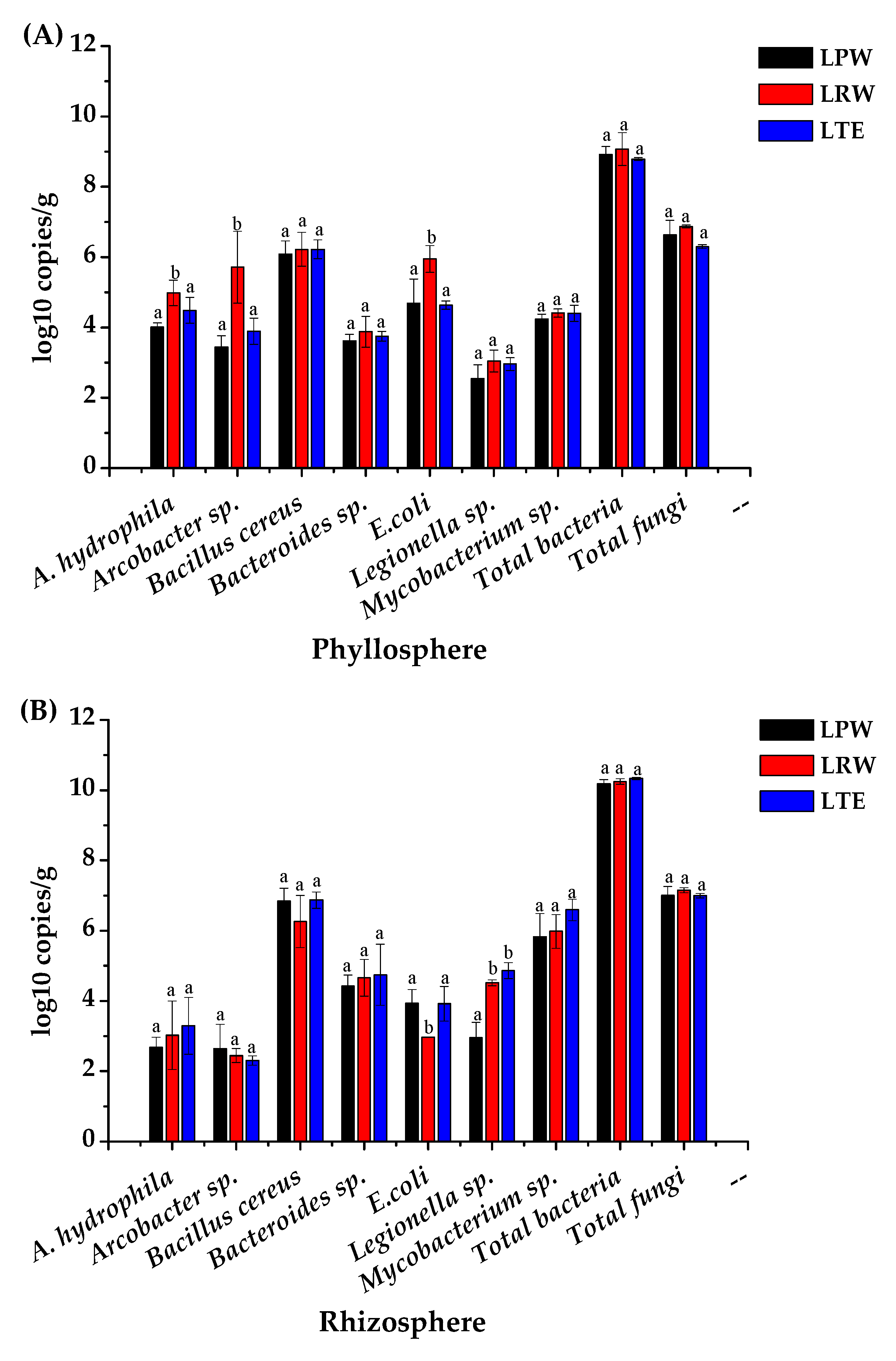
3.4. Abundances of Potential Pathogens in Phyllosphere and Rhizosphere
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lonigro, A.; Montemurro, N.; Rubino, P.; Vergine, P.; Pollice, A. Reuse of treated municipal wastewater for irrigation in Apulia region: The “IN.TE.R.R.A.” project. Environ. Eng. Manag. J. 2015, 14, 1665–1674. [Google Scholar] [CrossRef]
- Carvalho, R.D.S.C.; Bastos, R.G.; Souza, C.F. Influence of the use of wastewater on nutrient absorption and production of lettuce grown in a hydroponic system. Agric. Water Manag. 2018, 203, 311–321. [Google Scholar] [CrossRef]
- Becerra-Castro, C.; Lopes, A.R.; Vaz-Moreira, I.; Silva, E.F.; Manaia, C.M.; Nunes, O.C. Wastewater reuse in irrigation: A microbiological perspective on implications in soil fertility and human and environmental health. Environ. Int. 2015, 75, 117–135. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Ma, Z. Wastewater reclamation and reuse in Beijing: Influence factors and policy implications. Desalination 2012, 297, 72–78. [Google Scholar] [CrossRef]
- Friedler, E.; Lahav, O.; Jizhaki, H.; Lahav, T. Study of urban population attitudes towards various wastewater reuse options: Israel as a case study. J. Environ. Manag. 2006, 81, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Larsdotter, K.; Norström, A.; Gumaelius, L.; Jansen, J.L.C.; Dalhammar, G. A small scale hydroponics wastewater treatment system under Swedish conditions. Water Sci. Technol. 2003, 48, 161–167. [Google Scholar]
- Murray, A.; Ray, I. Wastewater for agriculture: A reuse-oriented planning model and its application in peri-urban China. Water Res. 2010, 44, 1667–1679. [Google Scholar] [CrossRef]
- Mara, D.D.; Sleigh, P.A.; Blumentha, U.J.; Carr, R.M. Health risks in wastewater irrigation: Comparing estimates from quantitative microbial risk analyses and epidemiological studies. J. Water Health 2007, 5, 39–50. [Google Scholar] [CrossRef]
- Hamilton, K.A.; Hamilton, M.T.; Johnson, W.; Jjemba, P.; Bukhari, Z.; Lechevallier, M.; Haas, C.N. Health risks from exposure to Legionella in reclaimed water aerosols: Toilet flushing, spray irrigation, and cooling towers. Water Res. 2018, 134, 261–279. [Google Scholar] [CrossRef]
- Cirelli, G.L.; Consoli, S.; Licciardello, F.; Aiello, R.; Giuffrida, F.; Leonardi, C. Treated municipal wastewater reuse in vegetable production. Agric. Water Manag. 2012, 104, 163–170. [Google Scholar] [CrossRef]
- Keraita, B.; Konradsen, F.; Drechsel, P.; Abaidoo, R.C. Reducing microbial contamination on wastewater-irrigated lettuce by cessation of irrigation before harvesting. Trop. Med. Int. Health 2007, 12, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Brandes, H.; Gyawali, P.; Sidhu, J.P.S.; Toze, S. Opportunistic pathogens in roof-captured rainwater samples, determined using quantitative PCR. Water Res. 2014, 53, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Szonyi, B.; Gautam, R.; Nightingale, K.; Anciso, J.; Ivanek, R. Risk factors for microbial contamination in fruits and vegetables at the preharvest level: A systematic review. J. Food Protect. 2012, 75, 2055–2081. [Google Scholar] [CrossRef] [PubMed]
- Kisluk, G.; Yaron, S. Presence and persistence of Salmonella enterica serotype Typhimurium in the phyllosphere and rhizosphere of spray-irrigated parsley. Appl. Environ. Microbiol. 2012, 78, 4030–4036. [Google Scholar] [CrossRef] [PubMed]
- De Giglio, O.; Caggiano, G.; Bagordo, F.; Barbuti, G.; Brigida, S.; Lugoli, F.; Grassi, T.; La Rosa, G.; Lucentini, L.; Uricchio, V.F.; et al. Enteric viruses and fecal bacteria indicators to assess groundwater quality and suitability for irrigation. Int. J. Environ. Res. Public Health 2017, 14, 558. [Google Scholar] [CrossRef] [PubMed]
- Seto, E.Y.; Konnan, J.; Olivieri, A.W.; Danielson, R.E.; Gray, D.M.D. A quantitative microbial risk assessment of wastewater treatment plant blending: Case study in San Francisco Bay. Environ. Sci. Water Res. Technol. 2016, 2, 134–145. [Google Scholar] [CrossRef]
- Ministry of Ecology and Environment of the People’s Republic of China. Discharge Standards of Pollutants for Municipal Wastewater Treatment Plant; Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2002.
- Zhang, B.G.; Bai, Z.H.; Hoefel, D.; Tang, L.; Wang, X.Y.; Li, B.J.; Li, Z.M.; Zhuang, G.Q. The impacts of cypermethrin pesticide application on the non-target microbial community of the pepper plant phyllosphere. Sci. Total Environ. 2009, 407, 1915–1922. [Google Scholar] [CrossRef]
- Whelan, J.A.; Russell, N.B.; Whelan, M.A. A method for the absolute quantification of cDNA using real-time PCR. J. Immunol. Methods 2003, 278, 261–269. [Google Scholar] [CrossRef]
- Ministry of Agriculture and Rural Affairs of the People’s Republic of China. Standards for Irrigation Water Quality; Ministry of Agriculture and Rural Affairs of the People’s Republic of China: Beijing, China, 2005.
- Singh, P.K.; Deshbhratar, P.B.; Ramteke, D.S. Effects of sewage wastewater irrigation on soil properties, crop yield and environment. Agric. Water Manag. 2012, 103, 100–104. [Google Scholar] [CrossRef]
- Ministry of Ecology and Environment of the People’s Republic of China. Soil Environmental Quality-Risk Control Standard for Soil Contamination of Agricultural Land; Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2018.
- Ministry of Ecology and Environment of the People’s Republic of China. Environmental Quality Evaluation Standards for Farmland of Edible Agricultural Products; Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2006.
- Public Health Agency of Canada. Pathogen Safety Data Sheets and Risk Assessment. Available online: https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/pathogen-safety-data-sheets-risk-assessment.htmL (accessed on 24 July 2018).
- Kumaraswamy, R.; Amha, Y.M.; Anwar, M.Z.; Henschel, A.; Rodriguez, J.; Ahmad, F. Molecular analysis for screening human bacterial pathogens in municipal wastewater treatment and reuse. Environ. Sci. Technol. 2014, 48, 11610–11619. [Google Scholar] [CrossRef]
- Lonigro, A.; Rubino, P.; Lacasella, V.; Montemurro, N. Faecal pollution on vegetables and soil drip irrigated with treated municipal wastewaters. Agric. Water Manag. 2016, 174, 66–73. [Google Scholar] [CrossRef]
- Gatta, G.; Libutti, A.; Gagliardi, A.; Beneduce, L.; Brusetti, L.; Borruso, L.; Disciglio, G.; Tarantino, E. Treated agro-industrial wastewater irrigation of tomato crop: Effects on qualitative/quantitative characteristics of production and microbiological properties of the soil. Agric. Water Manag. 2015, 149, 33–43. [Google Scholar] [CrossRef]
- Vergine, P.; Saliba, R.; Salerno, C.; Laera, G.; Berardi, G.; Pollice, A. Fate of the fecal indicator Escherichia coli in irrigation with partially treated wastewater. Water Res. 2015, 85, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Popovic, N.T.; Kazazic, S.P.; Strunjak-Perovic, I.; Barisic, J.; Klobucar, R.S.; Kepec, S.; Coz-Rakovac, R. Detection and diversity of aeromonads from treated wastewater and fish inhabiting effluent and downstream waters. Ecotox. Environ. Saf. 2015, 120, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Al-Jassim, N.; Ansari, M.I.; Harb, M.; Hong, P.Y. Removal of bacterial contaminants and antibiotic resistance genes by conventional wastewater treatment processes in Saudi Arabia: Is the treated wastewater safe to reuse for agricultural irrigation? Water Res. 2015, 73, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Houf, K.; On, S.L.W.; Coenye, T.; Mast, J.; Hoof, J.V.; Vandamme, P. Arcobacter cibarius sp. nov., isolated from broiler carcasses. Int. J. Syst. Evol. Microbiol. 2005, 55, 713–717. [Google Scholar] [CrossRef][Green Version]
- Yang, B.; Kong, X.; Cui, B.J.; Jin, D.C.; Deng, Y.; Zhuang, X.L.; Zhuang, G.Q.; Bai, Z.H. Impact of Rural Domestic Wastewater Irrigation on the Physicochemical and Microbiological Properties of Pakchoi and Soil. Water 2015, 5, 1825–1839. [Google Scholar] [CrossRef]
- Webb, A.L.; Taboada, E.N.; Selinger, L.B.; Boras, V.F.; Inglis, G.D. Efficacy of wastewater treatment on Arcobacter butzleri density and strain diversity. Water Res. 2016, 105, 291–296. [Google Scholar] [CrossRef]
- Levican, A.; Collado, L.; Figueras, M.J. The use of two culturing methods in parallel reveals a high prevalence and diversity of Arcobacter spp. in a wastewater treatment plant. BioMed Res. Int. 2016, 1–9. [Google Scholar] [CrossRef]
- Steele, M.; Odumeru, J. Irrigation water as source of foodborne pathogens on fruit and vegetables. J. Food Protect. 2004, 67, 2839–2849. [Google Scholar] [CrossRef]
- Marciano-Cabral, F.; Jamerson, M.; Kaneshiro, E.S. Free–living amoebae, Legionella and Mycobacterium in tap water supplied by a municipal drinking water utility in the USA. J. Water Health 2010, 8, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Whiley, H. Legionella Risk Management and Control in Potable Water Systems: Argument for the Abolishment of Routine Testing. Int. J. Environ. Res. Public Health 2017, 14, 12. [Google Scholar] [CrossRef] [PubMed]
- Jjemba, P.K.; Weinrich, L.A.; Cheng, W.; Giraldo, E.; LeChevallier, M.W. Regrowth of potential opportunistic pathogens and algae in reclaimed-water distribution systems. Appl. Environ. Microbiol. 2010, 76, 4169–4178. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, P.; Olson, N.D.; Paulson, J.N.; Pop, M.; Maddox, C.; Claye, E.; Goldstein, R.E.R.; Sharma, M.; Gibbs, S.G.; Mongodin, E.F.; et al. Conventional wastewater treatment and reuse site practices modify bacterial community structure but do not eliminate some opportunistic pathogens in reclaimed water. Sci. Total Environ. 2018, 639, 1126–1137. [Google Scholar] [CrossRef] [PubMed]
- Beuchat, L.R. Ecological factors influencing survival and growth of human pathogens on raw fruits and vegetables. Microbes Infect. 2002, 4, 413–423. [Google Scholar] [CrossRef]
- Orlofsky, E.; Bernstein, N.; Sacks, M.; Vonshak, A.; Benami, M.; Kundu, A.; Maki, M.; Smith, W.; Wuertz, S.; Shapiro, K.; et al. Comparable levels of microbial contamination in soil and on tomato crops after drip irrigation with treated wastewater or potable water. Agric. Ecosyst. Environ. 2016, 215, 140–150. [Google Scholar] [CrossRef]
- Shannon, K.E.; Lee, D.Y.; Trevors, J.T.; Beaudette, L.A. Application of real-time quantitative PCR for the detection of selected bacterial pathogens during municipal wastewater treatment. Sci. Total Environ. 2007, 382, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Agidi, S.; Vedachalam, S.; Mancl, K.; Lee, J.Y. Effectiveness of onsite wastewater reuse system in reducing bacterial contaminants measured with human-specific IMS/ATP and qPCR. J. Environ. Manag. 2013, 115, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Benami, M.; Gross, A.; Herzberg, M.; Orlofsky, E.; Vonshak, A.; Gillor, O. Assessment of pathogenic bacteria in treated graywater and irrigated soils. Sci. Total Environ. 2013, 458, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Heaton, J.C.; Jones, K. Microbial contamination of fruit and vegetables and the behaviour of enteropathogens in the phyllosphere: A review. J. Appl. Microbiol. 2008, 104, 613–626. [Google Scholar] [CrossRef]
- Wachtel, M.R.; Whitehand, L.C.; Mandrell, R.E. Association of Escherichia coli O157: H7 with preharvest leaf lettuce upon exposure to contaminated irrigation water. J. Food Protect. 2002, 65, 18–25. [Google Scholar] [CrossRef]
- Mottola, A.; Bonerba, E.; Bozzo, G.; Marchetti, P.; Celano, G.V.; Colao, V.; Terio, V.; Tantillo, G.; Figueras, M.J.; Di Pinto, A. Occurrence of emerging food-borne pathogenic Arcobacter spp. isolated from pre-cut (ready-to-eat) vegetables. Int. J. Food Microbiol. 2016, 236, 33–37. [Google Scholar] [CrossRef]
| Water Parameters | Raw Wastewater | Treated Effluent | Potable Water | Criteria of Irrigation Water [20] | Discharge Standard [17] |
|---|---|---|---|---|---|
| pH | 6.75–7.41 | 6.36–7.86 | 7.53 | 5.5–8.5 | 6–9 |
| EC (μS·cm−1) | 556–814 | 382–866 | 293 | ≤1000 | - |
| COD (mg·L−1) | 135–218 | 11–54 | 10 | ≤100 a or 60 b | 50–60 |
| TN (mg·L−1) | 11.8–22.5 | 13.2–16.4 | <0.5 | c -- | 15–20 |
| NH3-N (mg·L−1) | 8.4–17.7 | 6.7–17.6 | 0.16 | - | 5–8 |
| TP (mg·L−1) | 2.5–5.2 | 2.0–7.9 | 0.21 | - | 0.5–1 |
| Total bacteria (CFU·mL−1) | 3.6 × 106–4.3 × 106 | 1.67 × 104–3.4 × 105 | <100 | - | - |
| Total Coliforms (CFU·mL−1) | 1.0 × 104–6.0 × 105 | 1.0 × 103–7.17 × 104 | not detectable | - | - |
| Faecal Coliforms (CFU·100 mL−1) | 2.4 × 104 | not detectable (<3) | not detectable | ≤20 a or 10 b | 1–10 |
| Parameters | LPW | LRW | LTE | [22] | [23] |
|---|---|---|---|---|---|
| pH | 7.36 a | 7.18 a | 7.50 a | 6.5–7.5 | 6.5–7.5 |
| EC (μS·cm−1) | 563 a | 1210 b | 1587 b | - | - |
| Organic matter (%) | 1.57 a | 1.67 b | 1.73 b | - | - |
| Total nitrogen (%) | 0.113 a | 0.123 a | 0.130 a | - | - |
| Available P (mg·kg−1) | 53.8 a | 51.7 a | 55.1 a | - | - |
| Available K (mg·kg−1) | 154 a | 172 a | 169 a | - | - |
| Pb (mg·kg−1) | 18.4 a | 23.6 b | 18.9 a | 35–500 | 50–80 |
| Cu (mg·kg−1) | 24.4 a | 25.5 a | 25.4 a | 35–400 | 50–200 |
| Zn (mg·kg−1) | 81.1 a | 98.1 a | 136.5 b | 100–500 | 200–300 |
| Cd (mg·kg−1) | 0.23 a | 0.22 a | 0.24 a | 0.2–1.0 | 0.3–0.6 |
| Cr (mg·kg−1) | 34.45 a | 40.40 a | 43.65 a | 90–300 | 250–300 |
| Hg (mg·kg−1) | 0.111 a | 0.119 a | 0.139 a | 0.15–1.5 | 0.25–0.35 |
| As (mg·kg−1) | 9.74 a | 9.76 a | 10.55 a | 15–40 | 20–30 |
| Total bacteria (CFU·g−1) | 2.1 × 107 a | 2.0 × 107 a | 7.5 × 107 b | - | - |
| Total Coliforms (CFU·g−1) | 1.3 × 106 a | 6.1 × 106 b | 3.1 × 106 c | - | - |
| Parameters | LPW | LRW | LTE |
|---|---|---|---|
| Height (cm) | 25.75 a | 24.55 a | 25.69 a |
| Fresh weight (g) | 50 a | 47 a | 49 a |
| Soluble sugars (%) | 0.79 a | 0.67 a | 0.73 a |
| Soluble proteins (mg·g−1) | 10.99 a | 10.64 a | 10.67 a |
| Nitrate-Stem (mg·kg−1) | 515 a | 521 a | 494 a |
| Nitrate-Leaf (mg·kg−1) | 261 a | 145 b | 170 b |
| Total bacteria (CFU·g−1) | 3.5 × 106 a | 5.3 × 106 a | 4.7 × 106 a |
| Total Coliforms (CFU·g−1) | 1.2 × 104 a | 5.3 × 104 b | 2.0 × 104 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, B.; Liang, S. Monitoring Opportunistic Pathogens in Domestic Wastewater from a Pilot-Scale Anaerobic Biofilm Reactor to Reuse in Agricultural Irrigation. Water 2019, 11, 1283. https://doi.org/10.3390/w11061283
Cui B, Liang S. Monitoring Opportunistic Pathogens in Domestic Wastewater from a Pilot-Scale Anaerobic Biofilm Reactor to Reuse in Agricultural Irrigation. Water. 2019; 11(6):1283. https://doi.org/10.3390/w11061283
Chicago/Turabian StyleCui, Bingjian, and Shengxian Liang. 2019. "Monitoring Opportunistic Pathogens in Domestic Wastewater from a Pilot-Scale Anaerobic Biofilm Reactor to Reuse in Agricultural Irrigation" Water 11, no. 6: 1283. https://doi.org/10.3390/w11061283
APA StyleCui, B., & Liang, S. (2019). Monitoring Opportunistic Pathogens in Domestic Wastewater from a Pilot-Scale Anaerobic Biofilm Reactor to Reuse in Agricultural Irrigation. Water, 11(6), 1283. https://doi.org/10.3390/w11061283