Response of Water Chemistry to Long-Term Human Activities in the Nested Catchments System of Subtropical Northeast India
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Long-term Anthropogenic Activity in the Studied Catchments System
2.3. Sampling Design and Collection
2.4. Chemical Analysis
2.5. Statistics
3. Results and Discussion
3.1. Spatial and Seasonal Variation of Physico-chemical Properties of Stream Water
3.1.1. Physical Variables and Major Elements
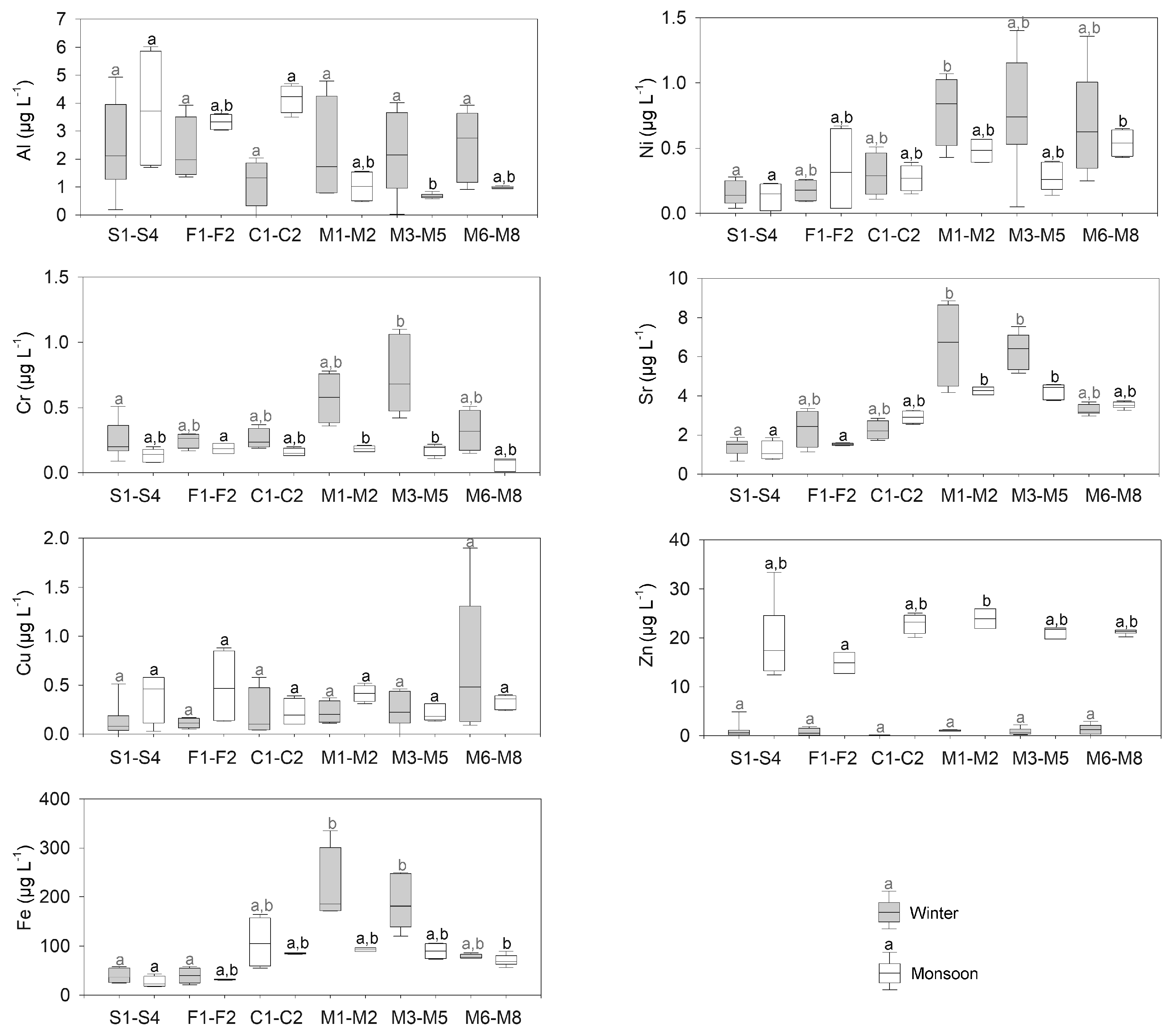
3.1.2. Trace Metals
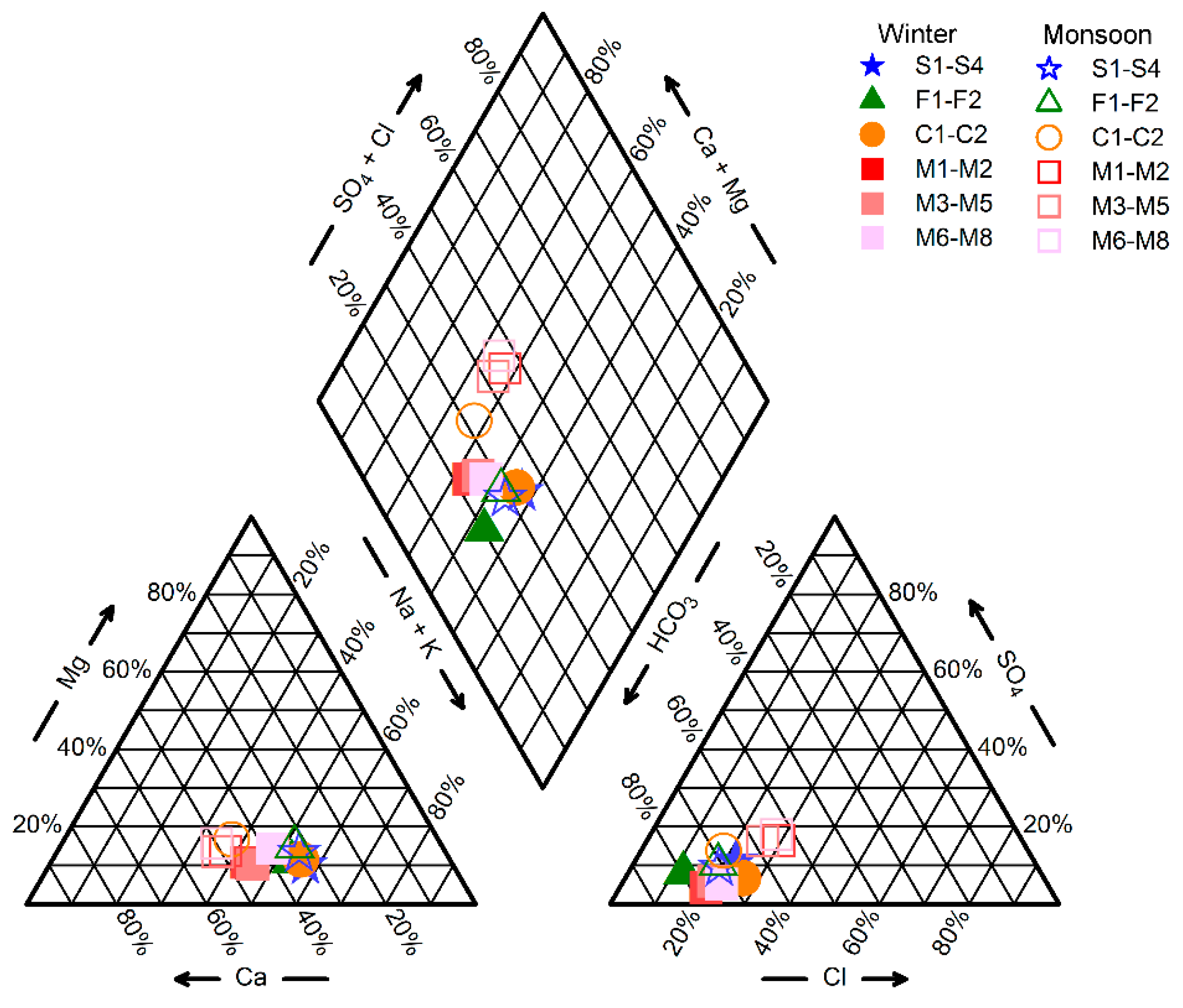
3.1.3. Hydrochemical Facies
3.2. Sources of Solutes and Their Pathways in Light of PCA in the Nongkrem Catchment
3.2.1. Impact of Population Density and Land Use on Water Chemistry
3.2.2. Role of Groundwater in Modification of Water Chemistry
3.2.3. Trace Metal Sources
3.2.4. DOC Concentrations
3.3. Comparison of Selected Chemical Element Concentrations in Headwater Areas of Tropical Zone
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Corlett, R.T. Where are the Subtropics? Biotropica 2013, 45, 273–275. [Google Scholar] [CrossRef]
- Pongratz, J.; Reick, C.; Raddatz, T.; Claussen, M.A. Reconstruction of global agricultural areas and land cover for the last millennium. Glob. Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Hansen, M.C.; Potapov, P.V.; Moore, R.; Hancher, M.; Turubanova, S.A.; Tyukavina, A.; Thau, D.; Stehman, S.V.; Goetz, S.J.; Loveland, T.R.; Kommareddy, A. High-resolution global maps of 21st-century forest cover change. Science 2013, 342, 850–853. [Google Scholar] [CrossRef]
- McKee, L.J.; Eyre, B.D.; Hossain, S.; Pepperell, P.R. Impacts of climate, geology and humans on spatial and temporal variability in nutrient geochemistry in the subtropical Richmond River catchment. Mar. Fresh Res. 2001, 52, 235–248. [Google Scholar] [CrossRef]
- Da Silva, D.M.L.; Ometto, J.P.H.B.; de Lobo, G.A.; de Lima, W.P.; Scaranello, M.A.; Mazzi, E.; da Rocha, H.R. Can land use changes alter carbon, nitrogen and major ion transport in subtropical Brazilian streams? Sci. Agric. 2007, 64, 317–324. [Google Scholar] [CrossRef]
- Wiejaczka, Ł.; Prokop, P.; Kozłowski, R.; Sarkar, S. Reservoir’s Impact on the Water Chemistry of the Teesta River Mountain Course (Darjeeling Himalaya). Ecol. Chem. Eng. S 2018, 25, 73–88. [Google Scholar] [CrossRef]
- Ometo, J.P.H.B.; Martinelli, L.A.; Ballester, M.V.; Gessner, A.; Krusche, A.V.; Victoria, R.L.; Williams, M. Effects of land use on water chemistry and macroinvertebrates in two streams of the Piracicaba river basin, South-East Brazil. Freshw. Biol. 2000, 44, 327–337. [Google Scholar] [CrossRef]
- Subramanian, V. Water quality in South Asia. Asian J. Water Environ. Pollut. 2004, 1, 41–54. [Google Scholar]
- Girija, T.R.; Mahanta, C.; Chandramouli, V. Water quality assessment of an untreated effluent impacted urban stream: The Bharalu tributary of the Brahmaputra River, India. Environ. Monit. Assess. 2007, 130, 221–236. [Google Scholar] [CrossRef]
- Silva, J.S.O.; da Cunha Bustamante, M.M.; Markewitz, D.; Krusche, A.V.; Ferreira, L.G. Effects of land cover on chemical characteristics of streams in the Cerrado region of Brazil. Biogeochemistry 2011, 105, 75–88. [Google Scholar] [CrossRef]
- Allan, J.D. Landscapes and riverscapes: The influence of land use on stream ecosystems. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 257–284. [Google Scholar] [CrossRef]
- Likens, G.E. Some perspectives on long term biogeochemical research from the Hubbarb Brook ecosystem study. Ecology 2004, 85, 2355–2362. [Google Scholar] [CrossRef]
- Pradhan, U.K.; Wu, Y.; Shirodkar, P.V.; Zhang, J. Seasonal nutrient chemistry in mountainous river systems of tropical Western Peninsular India. Chem. Ecol. 2015, 31, 199–216. [Google Scholar] [CrossRef]
- Bruijnzeel, L.A. Hydrological functions of tropical forests: Not seeing the soil for the trees? Agric. Ecosyst. Environ. 2004, 104, 185–228. [Google Scholar] [CrossRef]
- Goller, R.; Wilcke, W.; Fleischbein, K.; Valarezo, C.; Zech, W. Dissolved nitrogen, phosphorus, and sulfur forms in the ecosystemfluxes of amontane forest in Ecuador. Biogeochemistry 2006, 77, 57–89. [Google Scholar] [CrossRef]
- Saunders, T.J.; McClain, M.E.; Llerena, C.A. The biogeochemistry of dissolved nitrogen, phosphorus, and organic carbon along terrestrial-aquatic flowpaths of a montane headwater catchment in the Peruvian Amazon. Hydrol. Process. 2006, 20, 2549–2562. [Google Scholar] [CrossRef]
- Biggs, T.W.; Dunne, T.; Domingues, T.F.; Martinelli, L.A. The relative influence of natural watershed properties and human disturbance on stream solute concentrations in the Southwestern Brazilian Amazon basin. Water. Resour. Res. 2002, 38. [Google Scholar] [CrossRef]
- Singh, S.; Mishra, A. Spatiotemporal analysis of the effects of forest covers on stream water quality in Western Ghats of peninsular India. J. Hydrol. 2014, 519, 214–224. [Google Scholar] [CrossRef]
- Recha, J.W.; Lehmann, J.; Walter, M.T.; Pell, A.; Verchot, L.; Johnson, M. Stream water nutrient and organic carbon exports from tropical headwater catchments at a soil degradation gradient. Nutr. Cycl. Agroecosyst. 2013, 95, 145–158. [Google Scholar] [CrossRef]
- Jacobs, S.R.; Breuer, L.; Butterbach-Bahl, K.; Pelster, D.E.; Rufino, M.C. Land use affects total dissolved nitrogen and nitrate concentrations in tropical montane streams in Kenya. Sci. Total Environ. 2017, 603, 519–532. [Google Scholar] [CrossRef] [PubMed]
- Biggs, T.W.; Dunne, T.; Muraoka, T. Transport of water, solutes and nutrients from a pasture hillslope, southwestern Brazilian Amazon. Hydrol. Process. 2006, 20, 2527–2547. [Google Scholar] [CrossRef]
- Germer, S.; Neill, C.; Vetter, T.; Chaves, J.; Krusche, A.V.; Elsenbeer, H. Implications of long-term landuse change for the hydrology and solute budgets of small catchments in Amazonia. J. Hydrol. 2009, 364, 349–363. [Google Scholar] [CrossRef]
- Martinelli, L.A.; Krusche, A.V.; Victoria, R.L.; Camargo, P.B.D.; Bernardes, M.; Ferraz, E.S.; Moraes, J.M.D.; Ballester, M.V. Effects of sewage on the chemical composition of Piracicaba River, Brazil. Water Air Soil Pollut. 1999, 110, 67–79. [Google Scholar] [CrossRef]
- Daniel, M.H.; Montebelo, A.A.; Bernardes, M.C.; Ometto, J.P.; De Camargo, P.B.; Krusche, A.V.; Ballester, M.V.; Victoria, R.L.; Martinelli, L.A. Effects of urban sewage on dissolved oxygen, dissolved inorganic and organic carbon, and electrical conductivity of small streams along a gradient of urbanization in the Piracicaba river basin. Water Air Soil Pollut. 2002, 136, 189–206. [Google Scholar] [CrossRef]
- Bhatt, M.P.; Gardner, K.H. Variation in DOC and trace metal concentration along the heavily urbanized basin in Kathmandu Valley, Nepal. Environ. Geol. 2009, 58, 867–876. [Google Scholar] [CrossRef]
- Ramakrishnan, P.S. Shifting Agriculture and Sustainable Development: An Interdisciplinary Study from North-Eastern India; Parthenon Publications: Carnforth, UK, 1992; pp. 1–424. ISBN 1850703833. [Google Scholar]
- Prokop, P. Land use and land cover changes in the area with the highest rainfall in the world (Meghalaya Plateau, India): Causes and implications. In Environmental Geography of South Asia; Singh, R.B., Prokop, P., Eds.; Springer: Tokyo, Japan, 2016; pp. 143–159. ISBN 978-4-431-55741-8. [Google Scholar]
- Vliet, V.N.; Mertz, O.; Heinimann, A.; Langanke, T.; Pascual, U.; Schmook, B.; Adams, C.; Schmidt-Vogt, D.; Messerli, P.; Leisz, S.; et al. Trends, drivers and impacts of changes in swidden cultivation in tropical forest-agriculture frontiers: A global assessment. Glob. Environ. Chang. 2012, 22, 418–429. [Google Scholar] [CrossRef]
- Bhaskar, B.P.; Saxena, R.K.; Vadivelu, S.; Baruah, U.; Butte, P.S.; Dutta, D.P. Pedogenesis in high altitude soils of Meghalaya plateau. Agropedology 2004, 14, 9–23. [Google Scholar]
- Prokop, P.; Kruczkowska, B.; Syiemlieh, H.J.; Bucała-Hrabia, A. Impact of topography and sedentary swidden cultivation on soils in the hilly uplands of North-East India. Land Degrad. Dev. 2018, 29, 2760–2770. [Google Scholar] [CrossRef]
- Prokop, P.; Walanus, A. Variation in the orographic extreme rain events over the Meghalaya Hills in northeast India in the two halves of the twentieth century. Theor. Appl. Climatol. 2015, 121, 389–399. [Google Scholar] [CrossRef]
- Mazumder, S.K. The Precambrian framework of part of the Khasi Hills, Meghalaya. Rec. Geol. Survey India 1986, 117, 1–59. [Google Scholar]
- Ray, J.; Saha, A.; Ganguly, S.; Balaram, V.; Krishna, A.K.; Hazra, S. Geochemistry and petrogenesis of Neoproterozoic Mylliem granitoids, Meghalaya Plateau, Northeastern India. J. Earth Syst. Sci. 2011, 120, 459–473. [Google Scholar] [CrossRef]
- Hazra, S.; Saha, P.; Ray, J.; Polder, A. Simple statistical and mineralogical studies as petrogenetic indicator for Neoproterozoic Mylliem porphyritic granites of East Khasi Hills, Meghalaya, Northeastern India. J. Geol. Soc. India 2010, 75, 760–768. [Google Scholar] [CrossRef]
- Migoń, P.; Prokop, P. Landforms and landscape evolution in the Mylliem granite area, Meghalaya Plateau, Northeast India. Singap. J. Trop. Geogr. 2013, 34, 206–228. [Google Scholar] [CrossRef]
- Soil Survey Staff. Keys to Soil Taxonomy, 12th ed.; USDA-Natural Resources Conservation Service: Washington, DC, USA, 2014; pp. 1–361. [Google Scholar]
- Prokop, P.; Suliga, I. Two thousand years of iron smelting in the Khasi Hills, Meghalaya, North East India. Curr. Sci. India 2013, 104, 761–768. [Google Scholar]
- Prokop, P.; Bhattacharyya, A. Reconnaissance of quaternary sediments from Khasi Hills, Meghalaya. J. Geol. Soc. India 2011, 78, 258–262. [Google Scholar] [CrossRef]
- Rączkowska, Z.; Bucała-Hrabia, A.; Prokop, P. Geomorphological and sedimentological indicators of land degradation (Meghalaya Plateau, NE India). Land Degrad. Dev. 2018, 29, 2746–2759. [Google Scholar] [CrossRef]
- Directorate of Census Operations, Meghalaya. District Census Handbook, East Khasi Hills. Village and Town Wise Primary Census Abstract (PCA); Part XII-B, Series 18; Directorate of Census Operations: Shillong, India, 2011; pp. 1–240. [Google Scholar]
- Christophersen, N.; Hooper, R.P. Multivariate analysis of stream water chemical data: The use of principal component analysis for the end-member mixing problem. Water. Resour. Res. 1992, 28, 99–107. [Google Scholar] [CrossRef]
- Wayland, K.G.; Long, D.T.; Hyndman, D.W.; Pijanowski, B.C.; Woodhams, S.M.; Haack, S.K. Identifying relationships between baseflow geochemistry and land use with synoptic sampling and R-mode factor analysis. J. Environ. Qual. 2003, 31, 180–190. [Google Scholar] [CrossRef]
- Reimann, C.; Filzmoser, P.; Garrett, R.G. Factor analysis applied to regional geochemical data: Problems and possibilities. Appl. Geochem. 2002, 17, 185–206. [Google Scholar] [CrossRef]
- Fitzpatrick, M.L.; Long, D.T.; Pijanowski, B.C. Exploring the effects of urban and agricultural land use on surface water chemistry, across a regional watershed, using multivariate statistics. Appl. Geochem. 2007, 22, 1825–1840. [Google Scholar] [CrossRef]
- Connor, N.P.; Sarraino, S.; Frantz, D.E.; Bushaw-Newton, K.; MacAvoy, S.E. Geochemical characteristics of an urban river: Influences of an anthropogenic landscape. Appl. Geochem. 2014, 47, 209–216. [Google Scholar] [CrossRef]
- Olivia, P.; Viers, J.; Dupré, B. Chemical weathering in granitic environments. Chem. Geol. 2003, 202, 225–256. [Google Scholar] [CrossRef]
- Harmon, R.S.; Lyons, W.B.; Long, D.T.; Ogden, F.L.; Mitasova, H.; Gardner, C.B.; Welch, K.A.; Witherow, R.A. Geochemistry of four tropical montane watersheds, Central Panama. Appl. Geochem. 2009, 24, 624–640. [Google Scholar] [CrossRef]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water; Department of the Interior, US Geological Survey: Alexandria, VA, USA, 1985; pp. 1–263. [Google Scholar]
- Davies, P.J.; Wright, I.A.; Jonasson, O.J.; Findlay, S.J. Impact of concrete and PVC pipes on urban water chemistry. Urban Water J. 2010, 7, 233–241. [Google Scholar] [CrossRef]
- Markewitz, D.; Davidson, E.A.; Figueiredo, R.D.O.; Victoria, R.L.; Krusche, A.V. Control of cation concentrations in stream waters by surface soil processes in an Amazonian watershed. Nature 2001, 410, 802–805. [Google Scholar] [CrossRef]
- Bücker, A.; Crespo, P.; Frede, H.G.; Vaché, K.; Cisneros, F.; Breuer, L. Identifying controls on water chemistry of tropical cloud forest catchments: Combining descriptive approaches and multivariate analysis. Aquat. Geochem. 2010, 16, 127–149. [Google Scholar] [CrossRef]
- Boy, J.; Valarezo, C.; Wilcke, W. Water flow paths in soil control element exports in an Andean tropical montane forest. Eur. J. Soil. Sci. 2008, 59, 1209–1227. [Google Scholar] [CrossRef]
- Sung, K.Y.; Yun, S.T.; Park, M.E.; Koh, Y.K.; Choi, B.Y.; Hutcheon, I.; Kim, K.H. Reaction path modeling of hydrogeochemical evolution of groundwater in granitic bedrocks, South Korea. J. Geochem. Explor. 2012, 118, 90–97. [Google Scholar] [CrossRef]
- Meyer, J.; Paul, M.; Taulbee, W. Stream ecosystem function in urbanizing landscapes. J. N. Am. Benthol. Soc. 2005, 24, 602–612. [Google Scholar] [CrossRef]
- Halstead, J.A.; Kliman, S.; Berheide, C.W.; Chaucer, A.; Cock-Esteb, A. Urban stream syndrome in a small, lightly developed watershed: A statistical analysis of water chemistry parameters, land use patterns, and natural sources. Environ. Monit. Assess. 2014, 186, 3391–3414. [Google Scholar] [CrossRef]
- Wright, I.A.; Davies, P.J.; Findlay, S.J.; Jonasson, O.J. A new type of water pollution: Concrete drainage infrastructure and geochemical contamination of urban waters. Mar. Freshwater Res. 2011, 62, 1355–1361. [Google Scholar] [CrossRef]
- Christian, L.N.; Banner, J.L.; Mack, L.E. Sr isotopes as tracers of anthropogenic influences on stream water in the Austin, Texas, area. Chem. Geol. 2011, 282, 84–97. [Google Scholar] [CrossRef]
- Morgan, R.P.; Kline, K.M. Nutrient concentrations in Maryland non-tidal streams. Environ. Monit. Assess. 2011, 178, 221–235. [Google Scholar] [CrossRef]
- Singh, A.K.; Mondal, G.C.; Kumar, S.; Singh, T.B.; Tewary, B.K.; Sinha, A. Major ion chemistry, weathering processes and water quality assessment in upper catchment of Damodar River basin, India. Environ. Geol. 2008, 54, 745–758. [Google Scholar] [CrossRef]
- Zhu, B.; Yang, X.; Rioual, P.; Qin, X.; Liu, Z.; Xiong, H.; Yu, J. Hydrogeochemistry of three watersheds (the Erlqis, Zhungarer and Yili) in northern Xinjiang, NW China. Appl. Geochem. 2011, 26, 1535–1548. [Google Scholar] [CrossRef]
- Salomons, W. Environmental impact of metals derived from mining activities: Processes, predictions, prevention. J. Geochem. Explor. 1995, 52, 5–23. [Google Scholar] [CrossRef]
- Singh, J.; Ramakrishnan, P.S. Structure and function of a sub-tropical humid forest of Meghalaya I. Vegetation, biomass and its nutrients. Proc. Indian Acad. Sci. Plant Sci. 1982, 91, 241–253. [Google Scholar]
- Prokop, P.; Poręba, G.J. Soil erosion associated with an upland farming system under population pressure in Northeast India. Land Degrad. Dev. 2012, 23, 310–321. [Google Scholar] [CrossRef]
- Collins, R.; Jenkins, A. The impact of agricultural land on stream chemistry in the Middle Hills of the Himalayas, Nepal. J. Hydrol. 1996, 185, 71–86. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Viers, J.; Dupré, B.; Braun, J.J.; Deberdt, S.; Angeletti, B.; Ndam Ngoupayou, J.; Michard, A. Major and trace element abundances, and strontium isotopes in the Nyong basin rivers (Cameroon): Constraints on chemical weathering processes and elements transport mechanisms in humid tropical environments. Chem. Geol. 2000, 169, 211–214. [Google Scholar] [CrossRef]
- Patino, L.C.; Velbel, M.A.; Price, J.R.; Wade, J.A. Trace element mobility during spheroidal weathering of basalts and andesites in Hawaii and Guatemala. Chem. Geol. 2003, 202, 343–364. [Google Scholar] [CrossRef]
- Nakajima, T.; Terakado, Y. Rare earth elements in stream waters from the Rokko granite area, Japan: Effect of weathering degree of watershed rocks. Geochem. J. 2003, 37, 181–198. [Google Scholar] [CrossRef]
- Lorieri, D.; Elsenbeer, H. Aluminium, iron and manganese in near-surface waters of a tropical rainforest ecosystem. Sci. Total Environ. 1997, 205, 13–23. [Google Scholar] [CrossRef]
- Kabata-Pendias, A. Trace Elements in Soils and Plants; CRC Press: Boca Raton, FL, USA, 2010; pp. 1–403. ISBN 0-8493-1575-1. [Google Scholar]
- Van Gaelen, N.; Verheyen, D.; Ronchi, B.; Struyf, E.; Govers, G.; Vanderborght, J.; Diels, J. Identifying the transport pathways of dissolved organic carbon in contrasting catchments. Vadose Zone J. 2014, 13, 1–14. [Google Scholar] [CrossRef]
- Johnson, M.S.; Lehmann, J.; Selva, E.C.; Abdo, M.; Riha, S.; Couto, E.G. Organic carbon fluxes within and streamwater exports from headwater catchments in the Southern Amazon. Hydrol. Process. 2006, 20, 2599–2614. [Google Scholar] [CrossRef]
- Bhaskar, V.V.; Rao, P.S.P. Annual and decadal variation in chemical composition of rain water at all the ten GAW stations in India. J. Atmos. Chem. 2017, 74, 23–53. [Google Scholar] [CrossRef]
- Masese, F.O.; Kitaka, N.; Kipkemboi, J.; Gettel, G.M.; Irvine, K.; McClain, M.E. Litter processing and shredder distribution as indicators of riparian and catchment influences on ecological health of tropical streams. Ecol. Indic. 2014, 46, 23–37. [Google Scholar] [CrossRef]
- Masese, F.O.; Salcedo-Borda, J.S.; Gettel, G.M.; Irvine, K.; McClain, M.E. Influence of catchment land use and seasonality on dissolved organic matter composition and ecosystem metabolism in headwater streams of a Kenyan river. Biogeochemistry 2017, 132, 1–22. [Google Scholar] [CrossRef]
- Silva, D.M.; Camargo, P.B.; Mcdowell, W.H.; Vieira, I.; Salomão, M.S.; Martinelli, L.A. Influence of land use changes on water chemistry in streams in the State of São Paulo, southeast Brazil. An. Acad. Bras. Ciênc. 2012, 84, 919–930. [Google Scholar] [CrossRef]
- Wilcke, W.; Yasin, S.; Valarezo, C.; Zech, W. Change in water quality during the passage through a tropical montane rain forest in Ecuador. Biogeochemistry 2001, 55, 45–72. [Google Scholar] [CrossRef]
- Martínez, M.L.; Pérez-Maqueo, O.; Vázquez, G.; Castillo-Campos, G.; García-Franco, J.; Mehltreter, K.; Equihua, M.; Landgrave, R. Effects of land use change on biodiversity and ecosystem services in tropical montane cloud forests of Mexico. For. Ecol. Manag. 2009, 258, 1856–1863. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking Water Quality; WHO: Geneva, Switzerland, 2011; pp. 1–541. ISBN 92-4-154638-7. [Google Scholar]
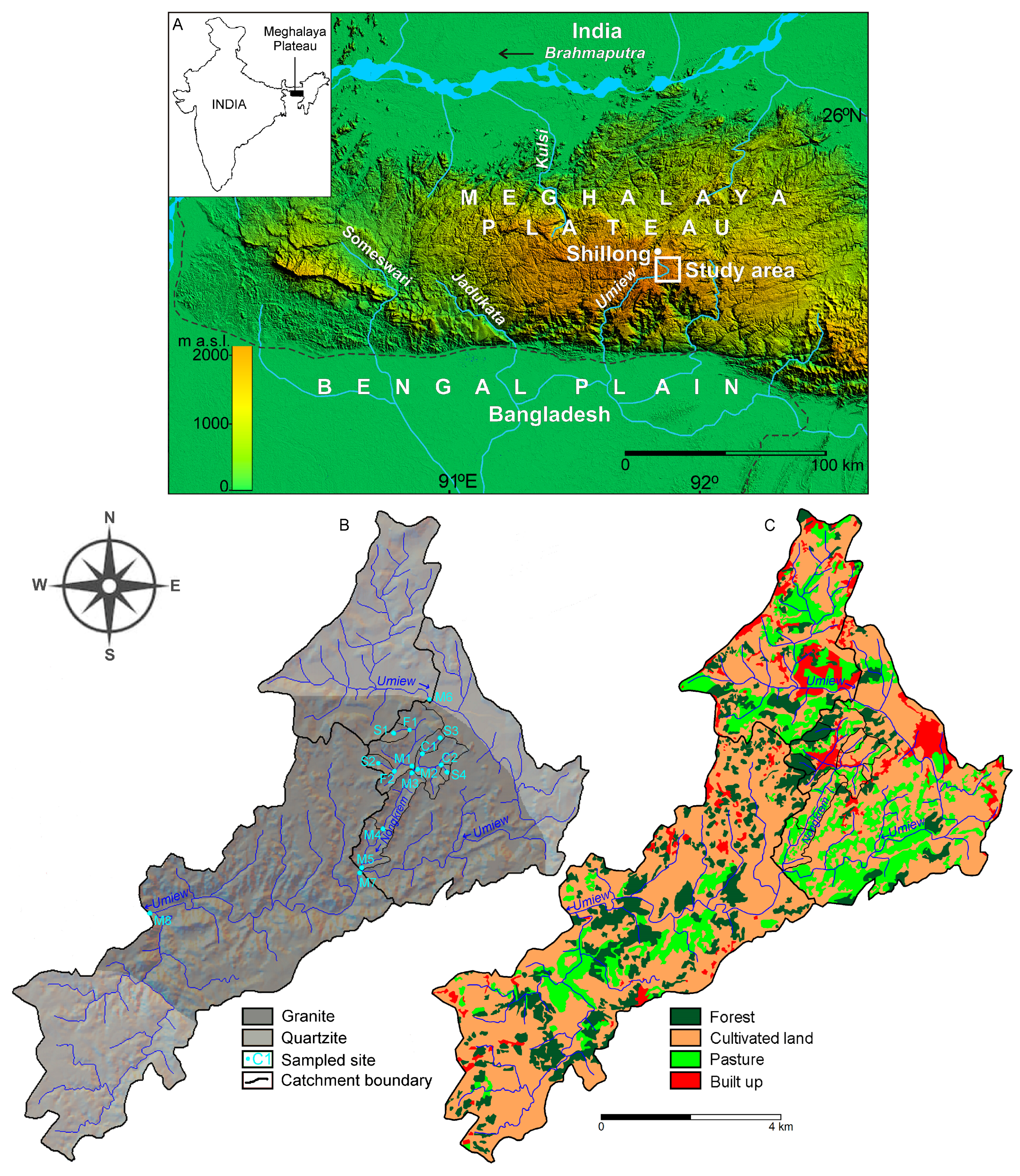
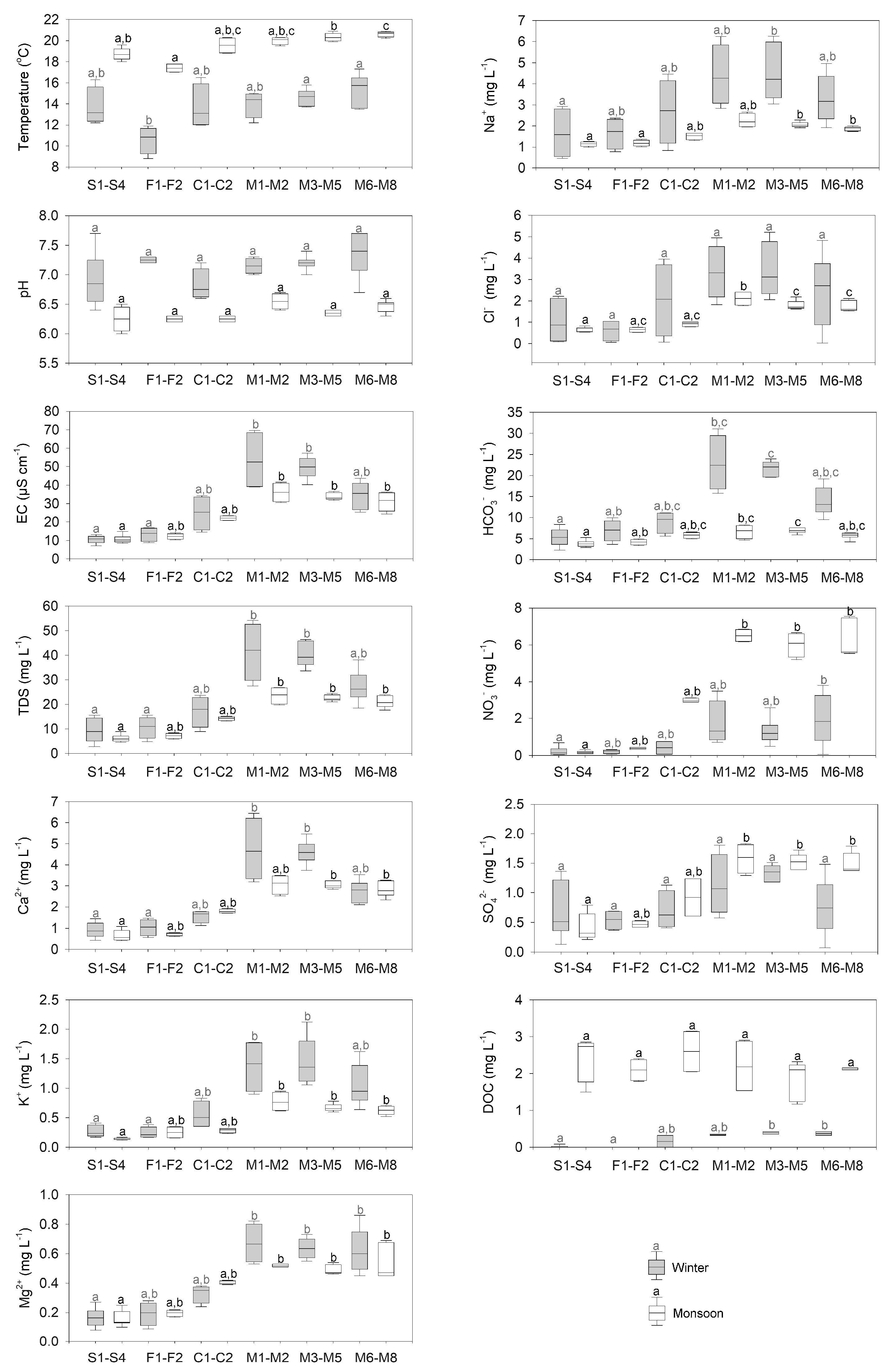
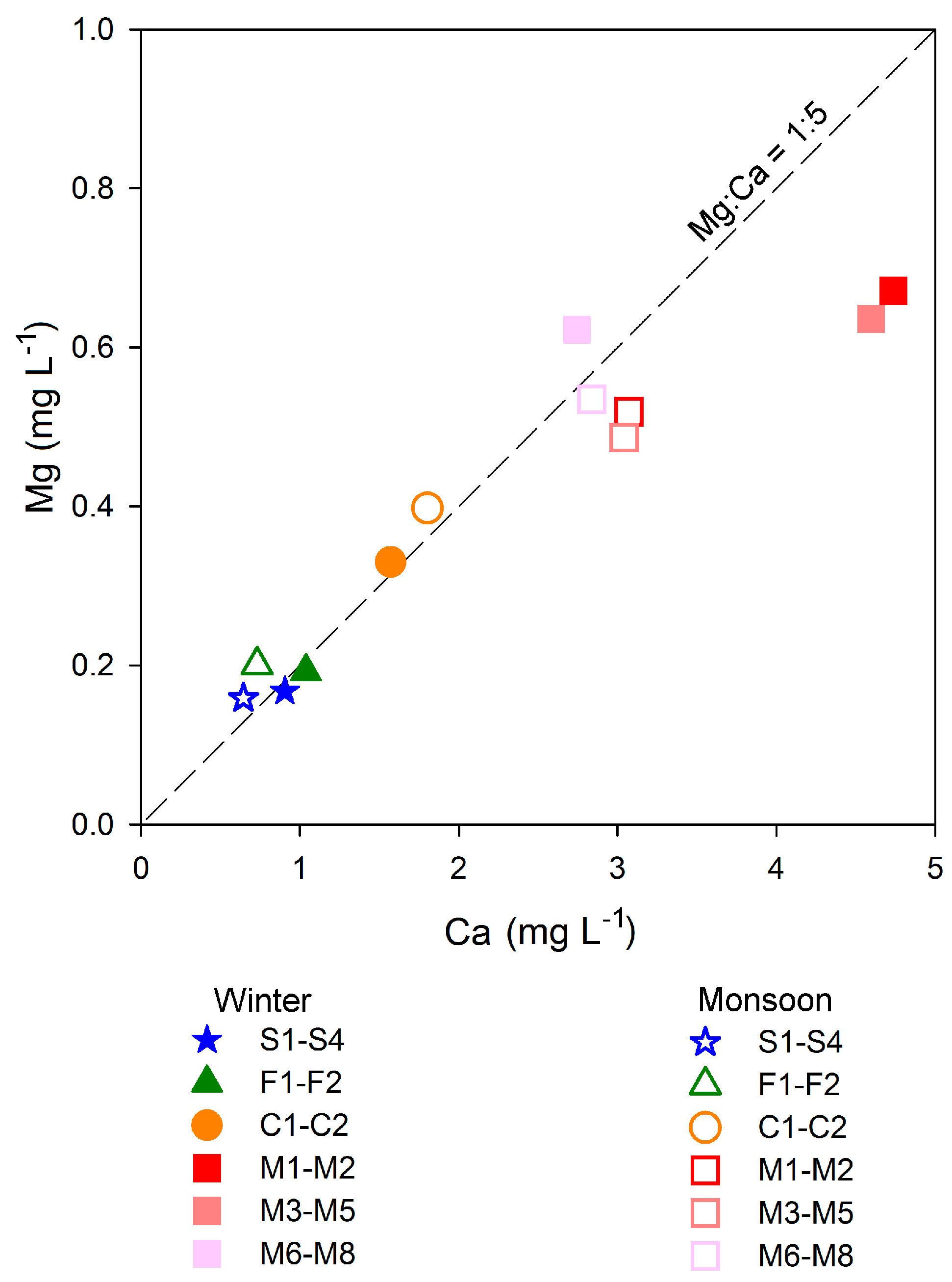
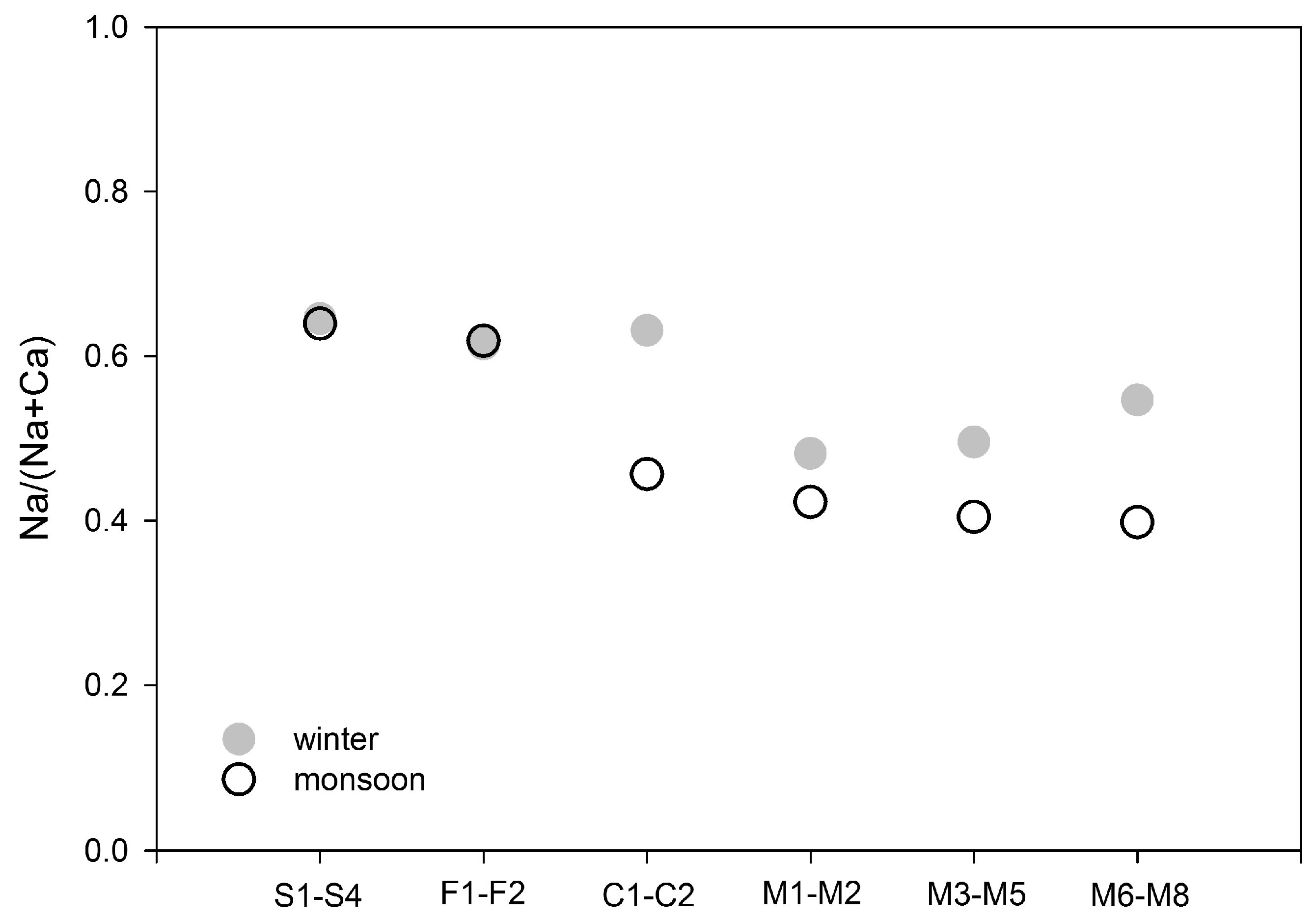
| Site/Sub-Catchment | Average Elevation | Area | Discharge Winter-Monsoon | Stream Order | Population Density | Forest | Grassland | Cultivated Land | Built Up | Geology Granite | Geology Quartzite | Soil Type |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| m a.s.l. | km2 | L s−1 | inhabitants km−2 | % | % | % | % | % | % | |||
| S1–S4 | 1808 | - | 0.2–0.8 | - | - | - | - | - | - | - | - | - |
| F1 | 1800 | 0.20 | 10–20 | 1 | 0 | 90 | 0 | 5 | 0 | 100 | 0 | Ultisol |
| F2 | 1805 | 0.17 | 10–20 | 1 | 0 | 100 | 0 | 0 | 0 | 100 | 0 | Ultisol |
| C1 | 1793 | 0.17 | 10–20 | 1 | 0 | 6 | 6 | 88 | 0 | 100 | 0 | Ultisol |
| C2 | 1778 | 0.14 | 7–15 | 1 | 0 | 0 | 14 | 86 | 0 | 100 | 0 | Ultisol |
| M1 | 1792 | 0.98 | 30–60 | 3 | 651 | 3 | 20 | 70 | 7 | 100 | 0 | Ultisol |
| M2 | 1802 | 1.69 | 40–80 | 3 | 1420 | 34 | 23 | 28 | 15 | 100 | 0 | Ultisol |
| M3 | 1802 | 2.65 | 70–140 | 4 | 1146 | 22 | 22 | 44 | 12 | 100 | 0 | Ultisol |
| M4 | 1795 | 3.34 | 100–200 | 4 | 910 | 12 | 40 | 40 | 8 | 100 | 0 | Ultisol |
| M5 | 1795 | 4.00 | 120–300 | 4 | 760 | 18 | 25 | 47 | 10 | 100 | 0 | Ultisol |
| M6 | 1850 | 10.78 | 490–1200 | 5 | 1031 | 11 | 27 | 47 | 15 | 20 | 80 | Ultisol-Inceptisol |
| M7 | 1830 | 28.96 | 1100–2000 | 5 | 800 | 12 | 25 | 53 | 10 | 52 | 48 | Ultisol-Inceptisol |
| M8 | 1805 | 59.30 | 2950–5300 | 5 | 570 | 16 | 18 | 60 | 6 | 60 | 40 | Ultisol-Inceptisol |
| Variable | Forest | Grassland | Cultivated Land |
|---|---|---|---|
| pH | 4.7 | 4.8 | 4.8 |
| Total C (mg kg−1) | 2608 | 2920 | 2105 |
| Total N (mg kg−1) | 213 | 240 | 195 |
| Total S (mg kg−1) | 100 | 80 | 100 |
| Total P (mg kg−1) | 728 | 580 | 771 |
| Total K (mg kg−1) | 4832 | 7094 | 5864 |
| Exchangeable Al (mg kg−1) | 170 | 182 | 163 |
| Exchangeable K (mg kg−1) | 323 | 374 | 367 |
| Exchangeable Na (mg kg−1) | 112 | 113 | 110 |
| Exchangeable Ca (mg kg−1) | 74 | 130 | 124 |
| Exchangeable Mg (mg kg−1) | 21 | 28 | 27 |
| Cation exchange capacity (cmolc kg−1) | 7.7 | 8.0 | 7.9 |
| Base saturation (%) | 25 | 30 | 29 |
| Site/Sub-Catchment | S1–S4 | F1–F2 | C1–C2 | M1–M2 | M3–M5 | M6–M8 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Season | Winter | Monsoon | Winter | Monsoon | Winter | Monsoon | Winter | Monsoon | Winter | Monsoon | Winter | Monsoon |
| T (°C) | 13.8 | 18.7 | 10.6 | 17.4 | 13.7 | 19.6 | 14.0 | 20.0 | 14.6 | 20.3 | 15.4 | 20.6 |
| pH | 7.1 | 6.3 | 7.3 | 6.3 | 6.8 | 6.3 | 7.2 | 6.6 | 7.2 | 6.4 | 7.4 | 6.5 |
| EC (μS cm−1) | 10.5 | 10.7 | 13.3 | 12.2 | 24.9 | 22.1 | 53.4 | 36.1 | 49.6 | 33.9 | 34.6 | 31.1 |
| TDS (mg L−1) | 10.38 | 7.33 | 11.48 | 8.26 | 17.68 | 14.92 | 40.61 | 23.75 | 39.43 | 22.76 | 27.21 | 21.34 |
| Ca2+ (mg L−1) | 0.90 | 0.64 | 1.04 | 0.73 | 1.57 | 1.80 | 4.74 | 3.07 | 4.60 | 3.04 | 2.75 | 2.84 |
| K+ (mg L−1) | 0.27 | 0.14 | 0.24 | 0.25 | 0.55 | 0.29 | 1.38 | 0.77 | 1.46 | 0.67 | 1.05 | 0.63 |
| Mg2+ (mg L−1) | 0.17 | 0.16 | 0.19 | 0.20 | 0.33 | 0.40 | 0.67 | 0.52 | 0.64 | 0.49 | 0.62 | 0.53 |
| Na+ (mg L−1) | 1.65 | 1.14 | 1.66 | 1.18 | 2.69 | 1.52 | 4.40 | 2.25 | 4.51 | 2.07 | 3.31 | 1.88 |
| Cl− (mg L−1) | 1.05 | 0.62 | 0.62 | 0.65 | 2.05 | 0.92 | 3.35 | 2.11 | 3.43 | 1.80 | 2.47 | 1.74 |
| HCO3− (mg L−1) | 5.32 | 3.85 | 6.92 | 4.17 | 8.97 | 5.80 | 22.91 | 6.68 | 21.69 | 6.93 | 13.86 | 5.73 |
| NO3− (mg L−1) | 0.22 | 0.17 | 0.21 | 0.38 | 0.41 | 3.00 | 1.71 | 6.51 | 1.29 | 6.00 | 1.94 | 6.23 |
| SO42− (mg L−1) | 0.69 | 0.41 | 0.54 | 0.47 | 0.70 | 0.92 | 1.08 | 1.58 | 1.34 | 1.53 | 0.76 | 1.50 |
| DOC (mg L−1) | 0.01 | 2.43 | 0.00 | 2.10 | 0.16 | 2.60 | 0.34 | 2.20 | 0.39 | 1.86 | 0.37 | 2.13 |
| Al (μg L−1) | 2.41 | 3.79 | 2.31 | 3.33 | 1.18 | 4.17 | 2.26 | 1.02 | 2.19 | 0.69 | 2.52 | 0.99 |
| Cr (μg L−1) | 0.25 | 0.13 | 0.25 | 0.18 | 0.26 | 0.16 | 0.58 | 0.19 | 0.74 | 0.18 | 0.33 | 0.07 |
| Cu (μg L−1) | 0.14 | 0.39 | 0.11 | 0.49 | 0.21 | 0.22 | 0.22 | 0.42 | 0.25 | 0.21 | 0.70 | 0.33 |
| Fe (μg L−1) | 39.40 | 26.28 | 39.61 | 31.66 | 107.25 | 84.61 | 219.64 | 92.39 | 187.60 | 89.66 | 78.66 | 70.82 |
| Ni (μg L−1) | 0.16 | 0.13 | 0.18 | 0.34 | 0.30 | 0.27 | 0.80 | 0.48 | 0.78 | 0.28 | 0.69 | 0.54 |
| Sr (μg L−1) | 1.40 | 1.18 | 2.34 | 1.53 | 2.26 | 2.91 | 6.63 | 4.26 | 6.31 | 4.26 | 3.27 | 3.53 |
| Zn (μg L−1) | 1.05 | 19.40 | 0.71 | 14.87 | 0.07 | 22.91 | 1.03 | 23.91 | 0.85 | 21.18 | 1.28 | 21.18 |
| Variable | Factor 1 | Factor 2 | Factor 3 | Factor 4 | Factor 5 |
|---|---|---|---|---|---|
| Population density | 0.912 | ||||
| Forest | −0.932 | ||||
| Grassland | 0.559 | ||||
| Cultivated land | 0.921 | ||||
| Built up | 0.911 | ||||
| T | 0.531 | 0.606 | |||
| pH | 0.637 | ||||
| EC | 0.870 | ||||
| TDS | 0.804 | ||||
| Ca2+ | 0.918 | ||||
| K+ | 0.790 | 0.508 | |||
| Mg2+ | 0.828 | ||||
| Na+ | 0.830 | ||||
| Cl− | 0.809 | ||||
| HCO3− | 0.809 | ||||
| NO3− | 0.923 | ||||
| SO42− | 0.756 | ||||
| DOC | 0.546 | ||||
| Al | 0.938 | ||||
| Cr | 0.700 | ||||
| Cu | 0.911 | ||||
| Fe | |||||
| Ni | 0.653 | 0.706 | |||
| Sr | 0.901 | ||||
| Zn | 0.833 | ||||
| Cumulative variance (%) | 42.0 | 55.2 | 74.5 | 81.0 | 91.6 |
| Variable | Factor 1 | Factor 2 | Factor 3 | Factor 4 |
|---|---|---|---|---|
| Population density | 0.964 | |||
| Forest | −0.885 | |||
| Grassland | 0.829 | |||
| Cultivated land | 0.989 | |||
| Built up | 0.961 | |||
| T | 0.710 | 0.520 | ||
| pH | 0.651 | |||
| EC | 0.881 | |||
| TDS | 0.880 | |||
| Ca2+ | 0.884 | |||
| K+ | 0.885 | |||
| Mg2+ | 0.816 | 0.560 | ||
| Na+ | 0.850 | |||
| Cl− | 0.911 | |||
| HCO3− | 0.610 | 0.629 | ||
| NO3− | 0.920 | |||
| SO42− | 0.840 | |||
| DOC | −0.893 | |||
| Al | −0.923 | |||
| Cr | 0.871 | |||
| Cu | 0.839 | |||
| Fe | 0.655 | 0.696 | ||
| Ni | 0.952 | |||
| Sr | 0.878 | |||
| Zn | 0.625 | |||
| Cumulative variance (%) | 53.2 | 64.8 | 85.7 | 92.7 |
| Location | Altitude (m a.s.l.) | Area (km2) | Soil Type | Land Use | Ca2+ | Cl− | K+ | Mg2+ | Na+ | NO3− | SO42− | DOC | Source |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Forest | |||||||||||||
| Nongkrem catchment, India | 1800 | 0.17–0.20 | Ultisol | evergreen forest | 1.04 ± 0.39 a 0.73 ± 0.08 b | 0.62 ± 0.49 a 0.65 ± 0.12 b | 0.24 ± 0.10 a 0.25 ± 0.10 b | 0.19 ± 0.08 a 0.20 ± 0.02 b | 1.66 ± 0.74 a 1.18 ± 0.15 b | 0.21 ± 0.11 a 0.38 ± 0.07 b | 0.54 ± 0.16 a 0.47 ± 0.06 b | 0.00 ± 0.00 a 2.10 ± 0.30 b | Current study (F1–F2) |
| Kapchorva catchment, Kenya | 1800 | 0.13 | Ultisol, Luvisol | tropical forest | 7.23 | – | 0.36 | 3.01 | 2.99 | 0.40 | – | 1.31 | [19] |
| Mara catchment, Kenya | 1900–2300 | 2.07–31.98 | Andisol | tropical forest | 2.70 ± 0.05 | 3.8 ± 0.40 a 1.0 ± 0.80 b | 4.30 ± 0.20 | 0.90 ± 0.20 | 6.20 ± 0.50 | 0.30 ± 0.10 | 3.20 ± 0.70 a 0.50 ± 0.40 b | 2.70 ± 0.40 a 3.50 ± 0.60 b | [74,75] |
| La Antigua catchment, Mexico | 480–4200 | <0.15 | – | tropical forest | 4.10 ± 0.60 | 4.30 ± 0.02 | 1.20 ± 0.07 | 1.10 ± 0.10 | 3.60 ± 0.40 | 1.40 ± 0.20 | – | – | [78] |
| Andes, Equador | 1800–2600 | 1.27 | Inceptisol, Histosol | tropical forest | 0.18 ± 0.03 a | 0.64 ± 0.26 a | 0.22 ± 0.03 a | 0.13 ± 0.02 a | 0.71 ± 0.07 a | 0.64 ± 0.13 a | 0.55 ± 0.14 a | – | [51] |
| Andes, Equador | 1900–2200 | 0.08–0.13 | Inceptisol, Histosol | tropical forest | 0.53–1.03 | 0.28–0.40 | 0.30–0.35 | 0.39–0.45 | 2.77–3.99 | 0.05–0.08 | – | – | [77] |
| Ribeira de Iguape catchment, Brazil | – | – | Inceptisol | ombrophilus forest | 2.45 ± 0.48 | 2.87 ± 0.18 | 0.43 ± 0.16 | 1.41 ± 0.24 | 2.25 ± 0.23 | 1.36 ± 0.19 | 0.87 ± 1.25 | – | [76] |
| Pontal do Paranapanema catchment, Brazil | – | – | Oxisol | semi-evergreen forest | 1.36 ± 0.40 | 0.60 ± 0.21 | 1.33 ± 0.43 | 0.41 ± 0.12 | 0.41 ± 0.11 | 0.37 ± 0.06 | 0.38 ± 0.19 | – | [76] |
| Cultivation | |||||||||||||
| Nongkrem catchment, India | 1790 | 0.14–0.17 | Ultisol | potatoes >100 years, no fertilizers | 1.57 ± 0.30 a 1.80 ± 0.09 b | 2.05 ± 1.73 a 0.92 ± 0.11 b | 0.55 ± 0.23 a 0.29 ± 0.04 b | 0.33 ± 0.06 a 0.40 ± 0.01 b | 2.69 ± 1.53 a 1.52 ± 0.17 b | 0.41 ± 0.39 a 3.00 ± 0.10 b | 0.70 ± 0.32 a 0.92 ± 0.36 b | 0.16 ± 0.18 a 2.60 ± 0.62 b | Current study (C1–C2) |
| Kapchorva catchment, Kenya | 1800 | 0.10 | Ultisol, Luvisol | maize 50 years, nitrogen | 7.16 | – | 1.18 | 2.54 | 5.61 | 4.52 | – | 1.52 | [19] |
| Mara catchment, Kenya | 1900–2300 | 2.02–13.98 | Andisol | maize, beans, potatoes ~40 years, fertilizers | 5.20 ± 0.10 | 5.60 ± 1.40 a 3.90 ± 0.60 b | 9.20 ± 0.80 | 1.30 ± 0.10 | 11.80 ± 1.30 | 6.10 ± 2.60 | 3.80 ± 0.60 a 2.70 ± 0.50 b | 3.60 ± 0.90 a 8.10 ± 0.92 b | [74,75] |
| La Antigua catchment, Mexico | 480–4200 | <0.15 | – | coffe plantation >30 years, potassium chloride, calcium phosphate, nitrogen | 12.00 ± 0.06 | 5.50 ± 0.10 | 2.50 ± 0.10 | 5.40 ± 0.40 | 6.70 ± 0.50 | 3.70 ± 0.60 | – | – | [78] |
| Agua Santa catchment, Brazil | 660–730 | 2.87 | Oxisol | sugar cane, nitrogen, liming | 2.91 ± 0.42 | 4.69 ± 0.92 | 2.87 ± 1.66 | 1.85 ± 0.31 | 1.93 ± 0.38 | 2.43 ± 0.72 | 0.44 ± 0.14 | 2.24 ± 1.62 | [5] |
| Mixed | |||||||||||||
| Nongkrem catchment, India | 1800 | 0.98–1.69 | Ultisol | cultivation 44%, grassland 22%, forest 22%, built-up 12% | 4.74 ± 1.50 a 3.07 ± 0.46 b | 3.35 ± 1.28 a 2.11 ± 0.33 b | 1.38 ± 0.45 a 0.77 ± 0.17 b | 0.67 ± 0.13 a 0.52 ± 0.01 b | 4.40 ± 1.44 a 2.25 ± 0.34 b | 1.71 ± 1.22 a 6.51 ± 0.34 b | 0.99 ± 0.74 a 1.58 ± 0.26 b | 0.34 ± 0.02 a 2.20 ± 0.75 b | Current study (M1–M2) |
| Ribeira de Iguape catchment, Brazil | – | – | Oxisol | pasture 75%, forest 18%, cultivation 4%, settlement 3% | 12.34 ± 3.49 | 4.40 ± 1.49 | 2.58 ± 1.60 | 4.50 ± 0.90 | 6.09 ± 1.56 | 1.30 ± 0.25 | 1.06 ± 0.77 | – | [76] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prokop, P.; Wiejaczka, Ł.; Syiemlieh, H.J.; Kozłowski, R. Response of Water Chemistry to Long-Term Human Activities in the Nested Catchments System of Subtropical Northeast India. Water 2019, 11, 988. https://doi.org/10.3390/w11050988
Prokop P, Wiejaczka Ł, Syiemlieh HJ, Kozłowski R. Response of Water Chemistry to Long-Term Human Activities in the Nested Catchments System of Subtropical Northeast India. Water. 2019; 11(5):988. https://doi.org/10.3390/w11050988
Chicago/Turabian StyleProkop, Paweł, Łukasz Wiejaczka, Hiambok Jones Syiemlieh, and Rafał Kozłowski. 2019. "Response of Water Chemistry to Long-Term Human Activities in the Nested Catchments System of Subtropical Northeast India" Water 11, no. 5: 988. https://doi.org/10.3390/w11050988
APA StyleProkop, P., Wiejaczka, Ł., Syiemlieh, H. J., & Kozłowski, R. (2019). Response of Water Chemistry to Long-Term Human Activities in the Nested Catchments System of Subtropical Northeast India. Water, 11(5), 988. https://doi.org/10.3390/w11050988





