1. Introduction
Calcium sulfate scales, together with calcium carbonate scales, are the major causes of fouling on process equipment surfaces with severe drawbacks. In particular, scale deposition on heat transfer surfaces increases the resistance to the heat flow [
1] and can accumulate in pipelines, orifices, and other flow passages seriously impeding the process flow. Moreover, calcium sulfate scale is one of the major cause of fouling in reverse osmosis (RO) membranes, resulting in a continuous decline in desalted water production, thus reducing the overall efficiency and increasing operation and maintenance costs [
2] (Di Zio et al., 2005). Therefore, from an economic point of view, the formation of calcium sulfate mineral scales is an obstacle to the recovery of potable water from sea [
3] or brackish waters as well as to the industrial utilization of many natural waters and in addition, from an environmental point of view, to the reuse of water in a closed water cycle approach [
4,
5] Calcium sulfate chemical and mineralogical properties of have been extensively studied: it may crystallize as calcium sulfate dihydrate (CaSO
4·2H
2O), calcium sulfate hemihydrate (CaSO
4·1/2H
2O), and calcium sulfate anhydrite (CaSO
4). The most stable calcium sulfate form in water systems is calcium sulfate dihydrate [
6].
The problem of calcium scaling on membrane is of major concern, not only in reverse osmosis plant for water desalination, but also in the field of superficial water. The use of ultrafiltration for drinking water production has widespread diffused during the past decade [
7]. Membrane fouling may be a major obstacle to the implementation of surface water ultrafiltration, which causes higher costs of energy, operation, and maintenance [
8]. Firstly, the membrane fouling occurs due to the natural total hardness of surface water. The effect is obviously amplified when water is heated or evaporated for process scopes, since the salt concentration, particularly of calcium ions, increases, reaching the saturation. As is well-known from crystallization studies and widely reported in literature [
9], a supersaturation increase in a salt solution at a constant temperature results in the more rapid precipitation (shorter induction times) of a huge quantity of small crystals in solution. In more details, the inorganic ions in water that exceed the equilibrium solubility product firstly reach the nucleation stage and then go through homogeneous or heterogeneous crystal growth processes [
10]. This also occurs when an aqueous solution passes through a membrane [
11]. The process, when intentionally carried out, is known as membrane crystallization [
12]; even if the membrane does not act as a selective barrier in this case, but the separation is based on the phase equilibrium [
13]. Nowadays, membrane crystallization is a mature technology that can replace the conventional crystallizers (e.g., in seawater distillation) because of its advantages due to its adaptability and the lower energy consumption [
14]. Whatever the scope of the process (water separation or salt recovery), the solution to the problem of fouling and scaling in membrane applications is very challenging.
Membrane fouling occurs during an increase in Trans-Membrane Pressure (TMP), to maintain a particular flux or during a decrease in flux when the system is operated at constant pressure [
8]. Fouling can be classified as reversible fouling and irreversible fouling; it is formed due to cake formation (reversible fouling) and pore blocking (irreversible fouling). Efficient pretreatments may relieve the fouling by pre-reacting with the foulants in the feed water, such as coagulation and adsorption [
15]. However, adverse effects from the pretreatment are also claimed. Additionally, operation methods such as running modes, rinsing modes, chemical cleaning, and air scouring can effectively obtain the fouling reduction. Particularly, backflushing appeared to be an efficient way to reduce the backwashable reversible fouling [
8] in ultrafiltration modules [
16]. A particular attention is to be paid to cleaning processes since they may cause membrane disintegration (structural changes and compromised filtration performance) [
17]. Thus far, the problem of membrane fouling is still unsolved in an ultimate way.
A literature analysis has confirmed that the fundamental mechanisms and processes involved in inorganic fouling are not fully understood; it is ascertained that in natural waters (river, groundwater, rainfall, seawater and municipal wastewater), the main basin or acidic inorganic species that are in equilibrium with precipitates or potential scaling components are OH
−, F
−, CO
32−, SO
42−, ortho-phosphate, and silicic acids, while major cations that account for most of the precipitates directly or indirectly are Ca
2+, Mg
2+, Fe
3+, and Al
3+ [
18]. Furthermore, a more recent school of thought affirms that besides feed solution characteristics and operating conditions, membrane fouling due to inorganic salts is also dependent on membrane properties (e.g., porosity, roughness, etc.), as well as module geometry and membrane materials, which influence the crystallization of inorganic salts [
17,
19]. Thus, one of the most straightforward and effective strategies to mitigate fouling is to render antifouling properties through the design of novel membranes [
20]. Nevertheless, there is a limited information on the effect of these parameters on fouling mechanism, mostly circumscribed to the field of reverse osmosis membranes [
21].
The addition of additives to a solution, which retards scale formation thus increasing the induction time (i.e., the time that elapses between the establishment of supersaturation in solution and the spontaneous nucleation of the first nuclei) and reducing the amount of salt precipitated, is a reliable and well-assessed technique to hinder or delay the scale formation, mostly in the RO system used for water desalination [
22]. The effect of the additives has been measured mostly with the monitoring of permeate flux decline and salt rejection curves, widely used at the laboratory scale to assess the antiscalant effectiveness on the scaling process for reverse osmosis membranes [
21]. Other techniques—always for RO membranes—are well summarized in the reviews by Shirazi et al. [
19] and Antony and coworkers [
21] include the direct observation of foulant accumulation on the membrane surface (SEM analysy, optical microscope, optical laser sensor), the ultrasonic time domain reflectometry (UTDR) technique and the electrical impedance spectroscopy (EIS).
However, it is important to point out that the behaviors and mechanisms of membrane scaling, as well as the effect of different additives, in various membrane processes may vary significantly, due to the different working principles and feedwater compositions [
23].
The additives commonly employed for gypsum scale control in process equipments are some inorganic and organic substances (e.g., phosphonates, carboxylic acids [
24,
25] capable of altering the surface properties of the crystals, affect nucleation and growth rate, modify crystal shape and agglomeration/dispersion behavior [
26]. As for membrane processes, so far, various commercial anti-scalants (e.g., polyphosphates, organophosphonates and polyelectrolytes) have been employed to mitigate mineral scaling in membrane processes including nanofiltration, reverse osmosis, forward filtration, and membrane desalination [
23]. The proper selection of an antiscalant additive depends on the water chemistry and the system design [
27]. Nowadays, there is the need of finding ecofriendly additives, the so-called green additives, natural organic molecules active as scale inhibitors that are environmentally acceptable compared with conventional inhibitors [
28]. Among them, there is citric acid, widely used across many industries especially in food, beverage, pharmaceutical, nutraceutical and cosmetic products [
29]. This is because of the nice properties of citric acid that is a natural preservative, flavoring, and antioxidant molecule, and most of all it is non-toxic, non-bio accumulating, and biodegradable; moreover, it can be used as an antiscalant [
30], particularly towards the calcium sulfate and carbonates scales.
The aim of the paper is to present the experimental results obtained on the inhibition of fouling due to calcium ions and scaling due to calcium sulfate precipitation on an ultrafiltration membrane surface, also in the presence of citric acid used as an additive. This additive has been typically used as an inhibitor for gypsum scaling on process equipments (industrial water) or RO membrane (seawater), so with waters with characteristics very different from surface waters. Firstly, the effect of water hardness due to calcium ions on membrane permeability has been measured in the range of potable waters. Then the fouling effect of high calcium concentration in solution has been quantified in terms of membrane weight and permeate fluxes as a function of time, with and without the addition of citric acid; moreover, the retarding effect of citric acid has been evaluated through the measurement of the induction times for the nucleation of calcium sulfate dihydrate, used as a model scalant. This coupling between crystallization technique and membrane equipment can be considered a reliable method for evaluating anti-scalant effectiveness of an additive on ultrafiltration membrane surfaces.
2. Materials and Methods
The experimental plant consists in a tangential flow system (MinitanTM-Millipore–see
Figure 1) on a lab scale equipped with a flat sheet ultrafiltration membrane in polyvinylidene fluoride (PVDF), whose cut-off is 650 nm.
Two experimental series were carried out: the first series was aimed at verifying the effect of calcium concentration on membrane fouling in superficial waters acceptable for human consumption. The influence of total water hardness expressed in French degrees (°f) on membrane permeability was studied in the range of potable waters (5 to 50 °f) and compared to that of distilled water. The temperature was constant at the value T = 25 °C, while the transmembrane pressure (TMP) was changed in the range 20 to 500 kPa. In addition, a long-term experiment (200 min) was carried out for three kinds of waters of different hardness (from 5 to 50 °f).
The second experimental series was conducted in a condition of extremely high concentration of calcium and sulfate ions (supersaturated solutions), with and without the use of an additive to retard the salt precipitation, always at room temperature, as shown in
Table 1. All experiments were duplicated and the average values were considered.
Citrid acid (2-hydroxy-1,2,3-propanetricarboxylic acid, C6H8O7) was used as an additive. Supersaturated solutions of calcium sulfate were prepared by mixing two clear equimolar aqueous solutions of reagent-grade CaCl2·2H2O and Na2SO4 with concentrations in the range 55–65 mol/m3.
The supersaturation ratio
S was calculated considering the liquid–solid equilibrium between Ca
2+ and SO
42− ions and solid CaSO
4·2H
2O, as described by the following equation:
so that it is:
where a
J represents the activity of the J species (J = Ca
2+, SO
42− and water) expressed as the product of the molality (m
J) and the activity coefficient (γ
J), and K
ps is the solubility product of gypsum. The value of K
ps was calculated as a function of temperature by means of the following relationship:
All the experiments were carried out for four values of additive concentrations (cCA = 0.01, 0.05, 0.10, 0.50 g/L), while the supersaturation ratio changed in the range S = 2.25–2.60. The temperature was kept constant at the value T = 25 °C while the transmembrane pressure was changed in the range 30 to 130 kPa.
For each experimental run on supersaturated solution (
Table 1), the induction time has been measured with a 10% change in solution turbidity. The permeate flux has been measured at the beginning and at the end of run, and the run continued for 15 min after the beginning of gypsum precipitation in solution (induction time). Membrane flat sheet were weighted before each test, soon after the test and after drying in an oven at
T = 50 °C for one hour. As for membrane cleaning, the procedure involves a series of washing with acid and basic solvents and with a specific membrane cleaning reagent Ultrasil. In our case, the fouling was due to calcium salts and therefore an acidic washing with sulfuric acid was sufficient for restoring the initial flux.
Sample of the solids taken form membrane surface were analyzed with XRD (Malvern PANalytical, Malvern, UK) and with an optical microscope (Zeiss, Axioscope FS), equipped for transmitted light work.
3. Results and Discussion
Two experimental series were carried out: The first series was aimed at verifying the effect of calcium concentration on membrane fouling in superficial waters acceptable for human consumption. In a second experimental series, mineral scale nucleation and inhibition on membrane surface was evaluated, for calcium sulfate dihydrate (gypsum) used as a model scalant.
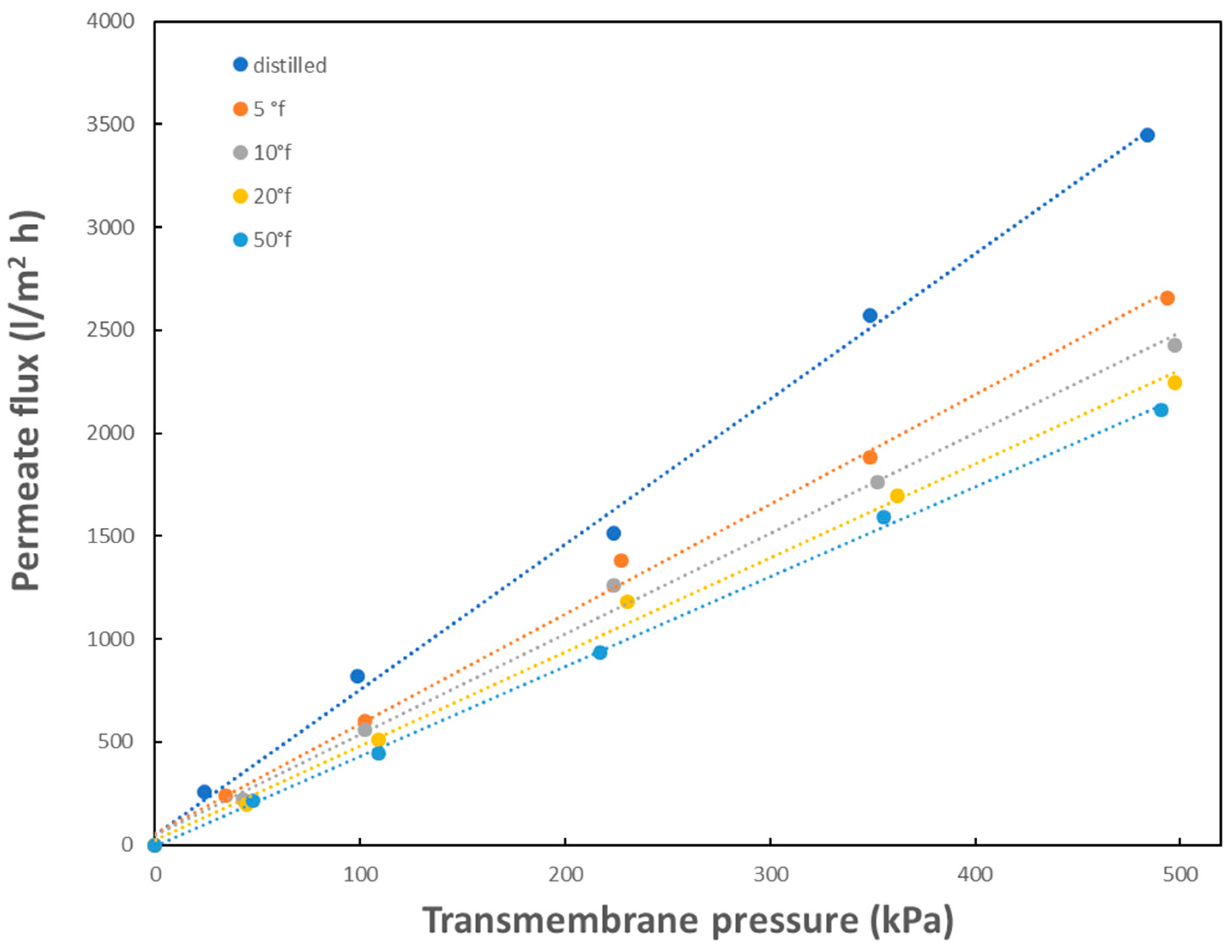
Figure 2 reports some of the results belonging to the first series, showing the effect of total water hardness, expressed in °f, on membrane permeability in the range of potable waters (5 to 50 °f) and compared to the permeability of distilled water, at the constant
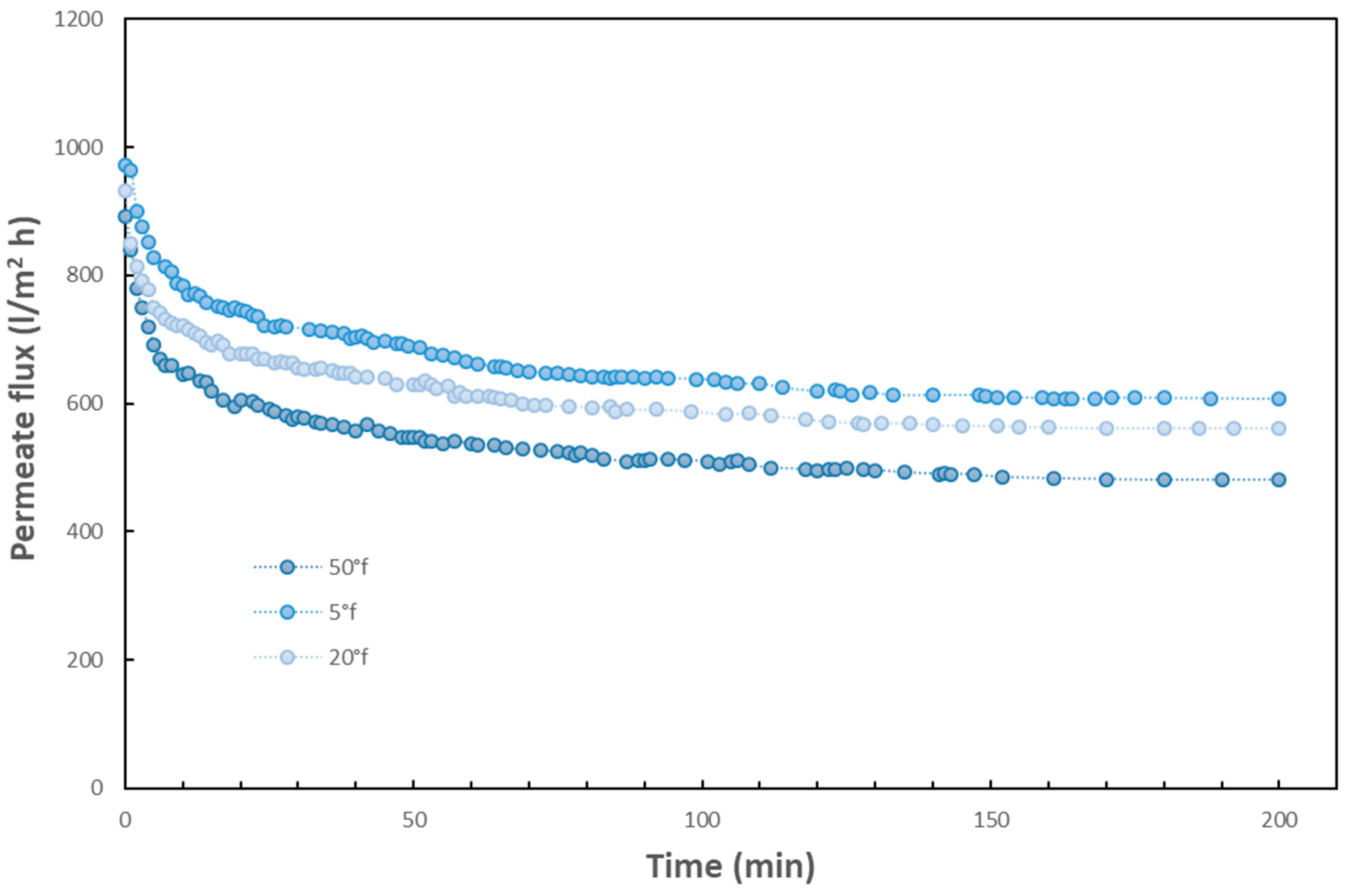
T = 25 °C. The analysis of the figure shows that, as expected, membrane fluxes decrease with increasing water strength, with respect to that of distilled water. In details, the average decrease in flux at 50 °f is reduced of about 20% with respect to the corresponding value at 5 °f and of about 30% with respect to distilled water. The different behavior of water with different harness is more visible in the long-term experiments reported in
Figure 3. As can be seen, relevant decreases of flux with time were observed for the three water samples of different hardness. After about three hours of filtration, there was a flux decay in the range 37%–46% at the constant transmembrane pressure of 100 kPa. Therefore, the membrane fouling due to the sole water hardness is able to cause a decrease in permeate flux with time and a limitation of the separation efficiency. Due to the buildup of a fouling layer mostly made of calcium ions, the rate of water transport through the membrane is decreased. As a result, greater pressure (i.e., energy) is required to produce the same amount of water.
As for the second experimental series,
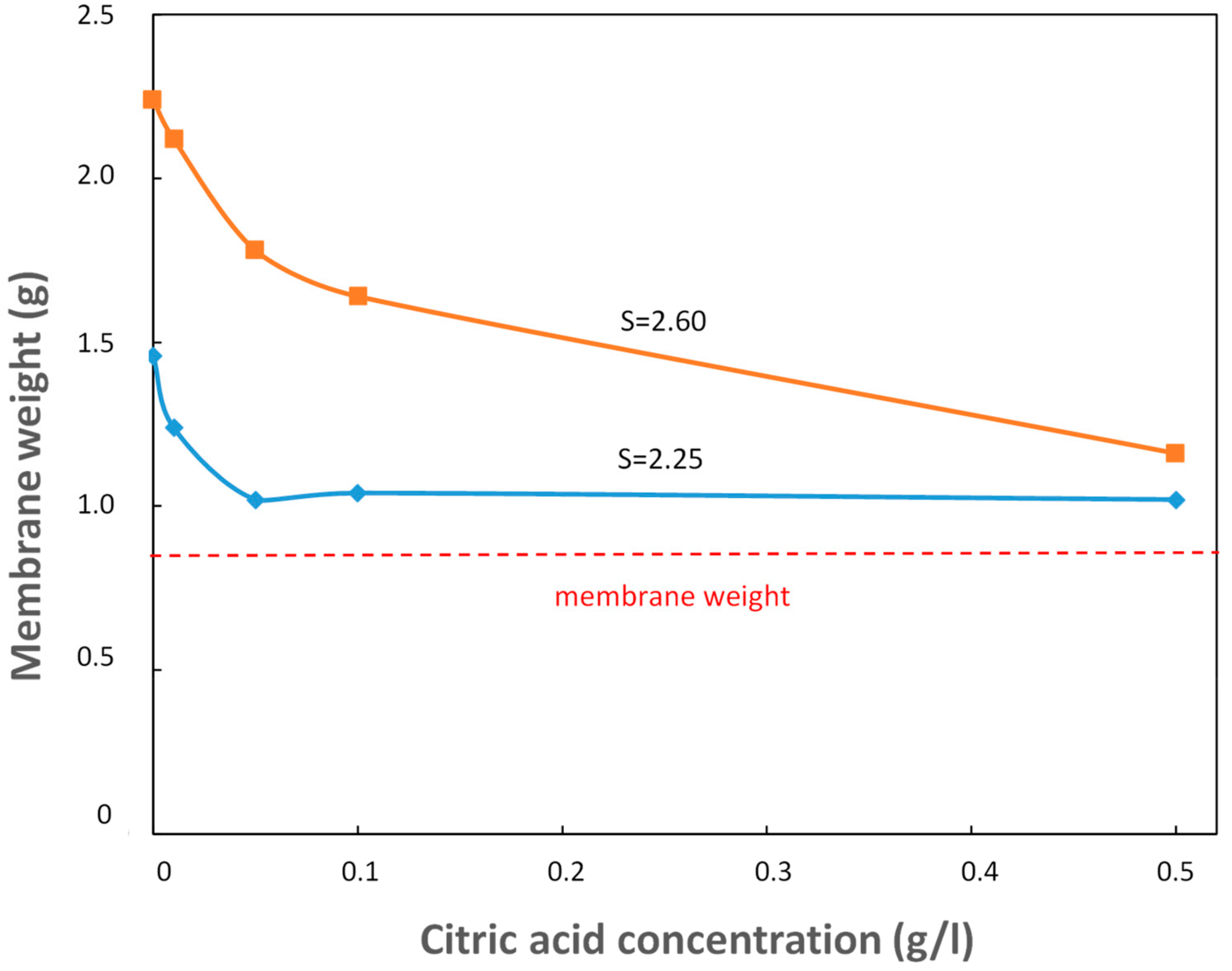
Figure 4 shows the effect of the addiction of citric acid in a calcium sulfate supersaturated solution as a function of the membrane weight for two different supersaturation ratios (
S = 2.25–blue curve and
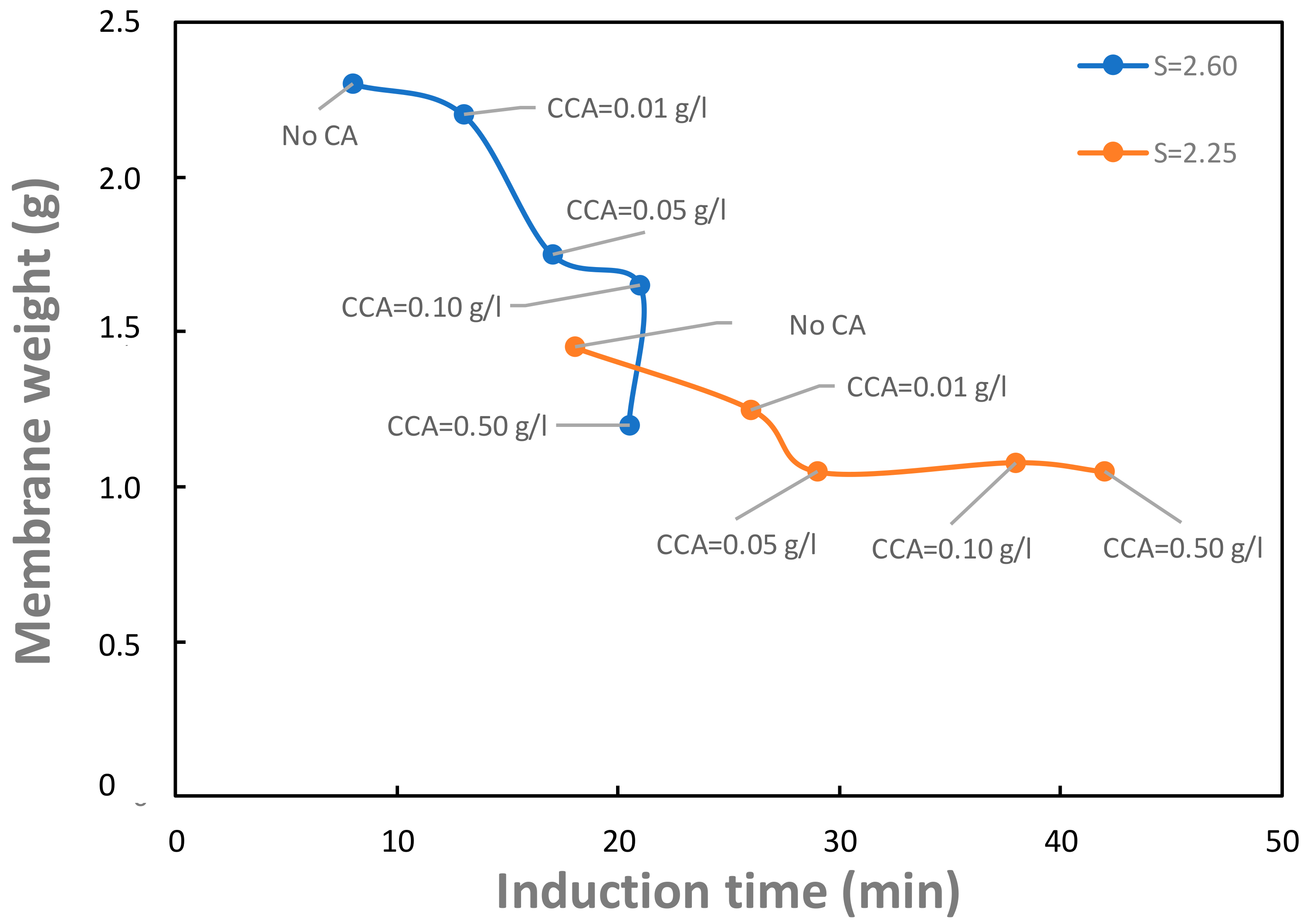
S = 2.60–orange curve). As can be seen, for each supersaturation ratio, the membrane weight decreases while adding citric acid, always remaining above the initial membrane weight (0.86 g), since the effect of the additive is to inhibit the gypsum precipitation, thus diminishing the amount of salt deposited on the membrane surface.
Moreover, for a higher supersaturation ratio (orange curve), the increase in weight due to the calcium sulfate deposition of membrane surface, is higher for each citric acid concentration tested. The effect of citric acid seems to be stronger at higher S values. However, the membrane weight goes to an asymptotic value, suggesting the uselessness of increasing the additive concentration above the value of 0.1 g/L.
Figure 5a,b show how the membrane sheets look like after two experimental runs, the best case (
cCA = 0.50 g/L, low supersaturation ratio—run#5) and the worst case (no additive added in solution, high supersaturation ratio—run #6). In the second case, the membrane surface appears covered by a thick layer of gypsum scale, with an increase in weight of about 55%.
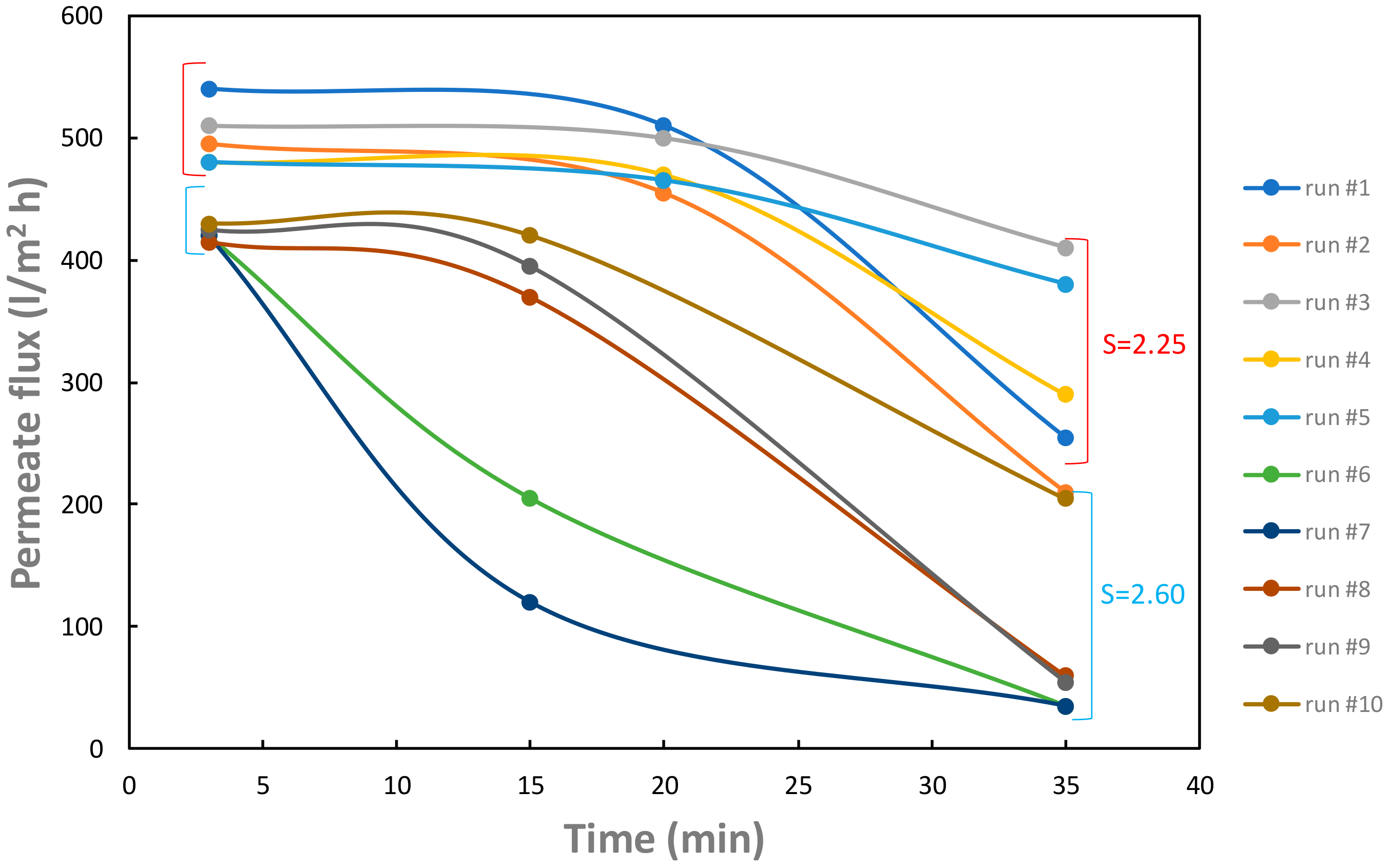
Figure 6 shows the permeate flux through the membrane as a function of time for the ten experimental runs reported in
Table 1.
For each curve, the permeate flux as a function of time rapidly decreases mainly due to the precipitation of gypsum on membrane surface. In each group, at the same supersaturation ratio, the addition of citric acid generally allows a final permeate flux (at
t = 35 min) higher with respect to that obtained in the absence of citric acid. The run#10, with
cCA = 0.50 g/L and
S = 2.60 (see
Table 1), notwithstanding a higher initial salt concentration, has a final permeate flux comparable to that obtained in run#2 belonging to the first group, characterized by a lower supersaturation (lower salt concentration) and a lower citric acid concentration (
cCA = 0.01 g/L and
S = 2.25, see
Table 1).
Finally,
Figure 7 shows the membrane weight as a function of induction time for different citric acid concentrations. Specific studies had already shown that the addition of citric acid allows to modify the induction time of gypsum by delaying it [
31]. Data in
Figure 7 show that the increase in induction time caused by citric acid also yields a reduction in the fouling of the membrane surface.
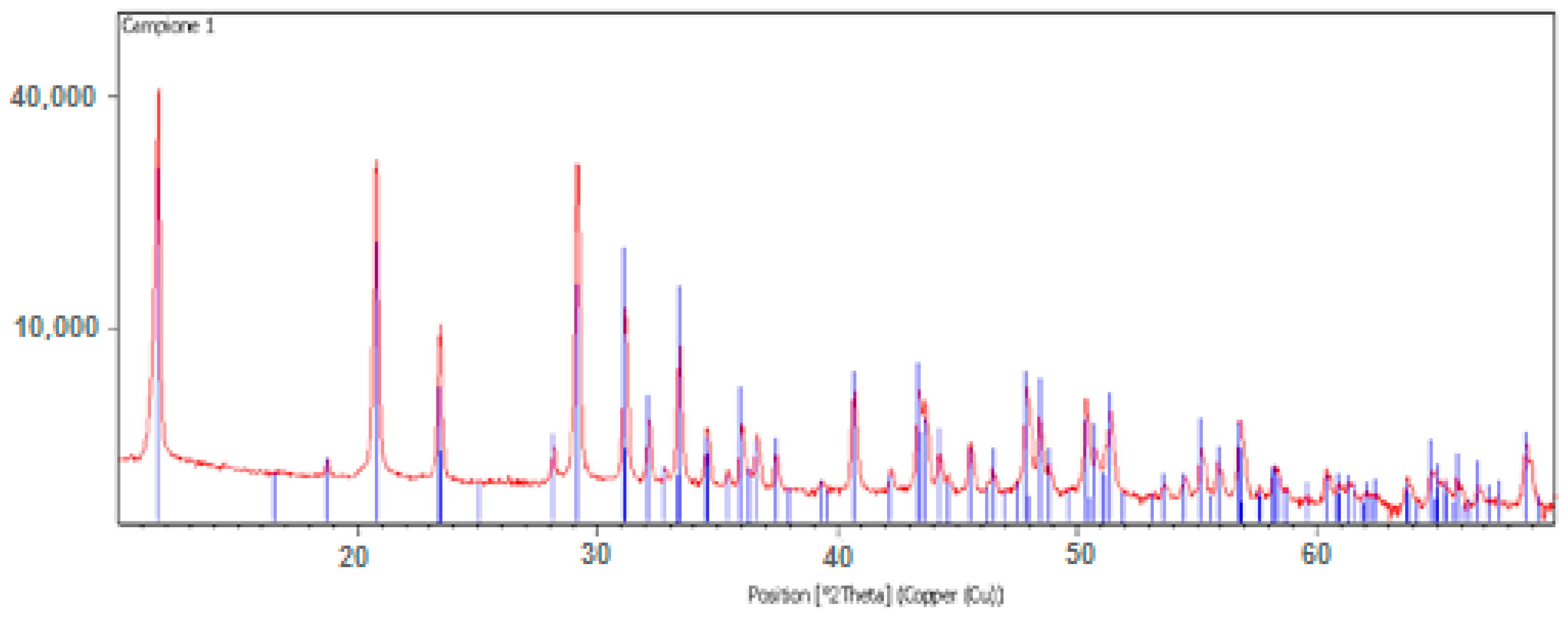
Moreover, an XRD analysis reported in
Figure 8 confirmed that the solid precipitate on membrane surface is mainly made of calcium sulfate dihydrate (gypsum).
Figure 9a,b show the effect of citric acid on the morphology of gypsum crystals. In the samples taken by means of an optical microscope, it can be seen that in the presence of citric acid (
Figure 9b), the morphology of gypsum had changed from the typical long needles (
Figure 9a) [
32] to smaller platelets. This effect of citric acid on gypsum morphology confirms what already found in literature in another field of study, namely the positive outcomes of citric acid that, beside from being a retardant for gypsum nucleation, it is known to increase the compressive strength of the calcium sulphoaluminate cements by changing the shape of crystals with an improved densification of the microstructure [
33].