A Pilot Study of an Electromagnetic Field for Control of Reverse Osmosis Membrane Fouling and Scaling During Brackish Groundwater Desalination
Abstract
1. Introduction
2. Materials and Methods
2.1. Groundwater Quality
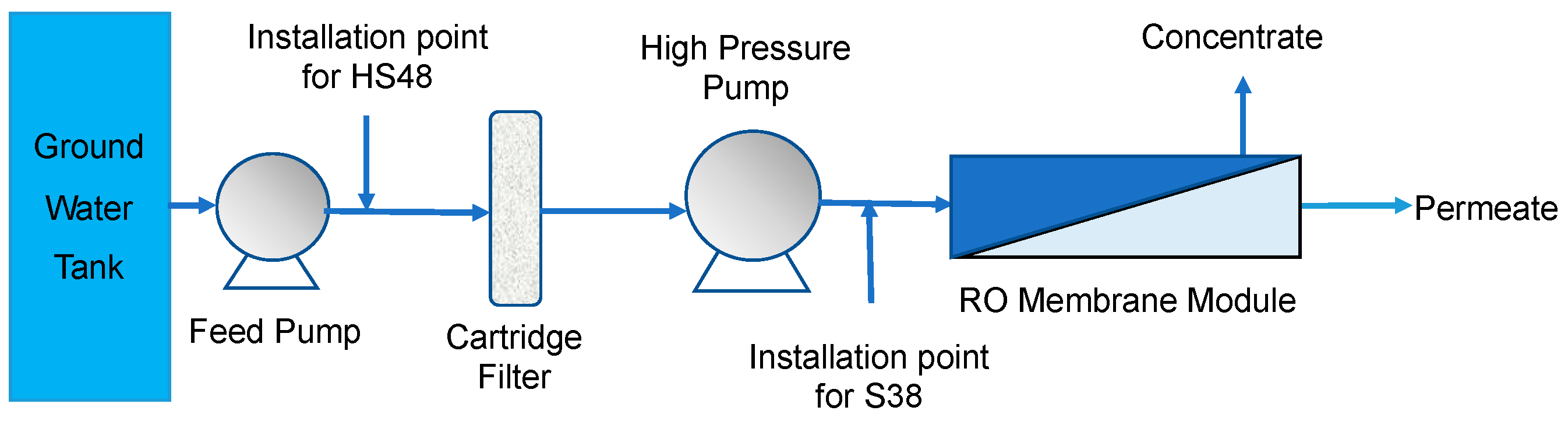
2.2. Pilot-Scale RO System
2.3. HydroFLOW Devices
2.4. Analytical Methods
2.5. Calculations
3. Results and Discussion
3.1. Water Flux of the RO System
3.2. Characterization of RO Membrane Fouling and Scaling
3.3. Solute Transport
3.4. Preliminary Cost Estimate
4. Conclusions
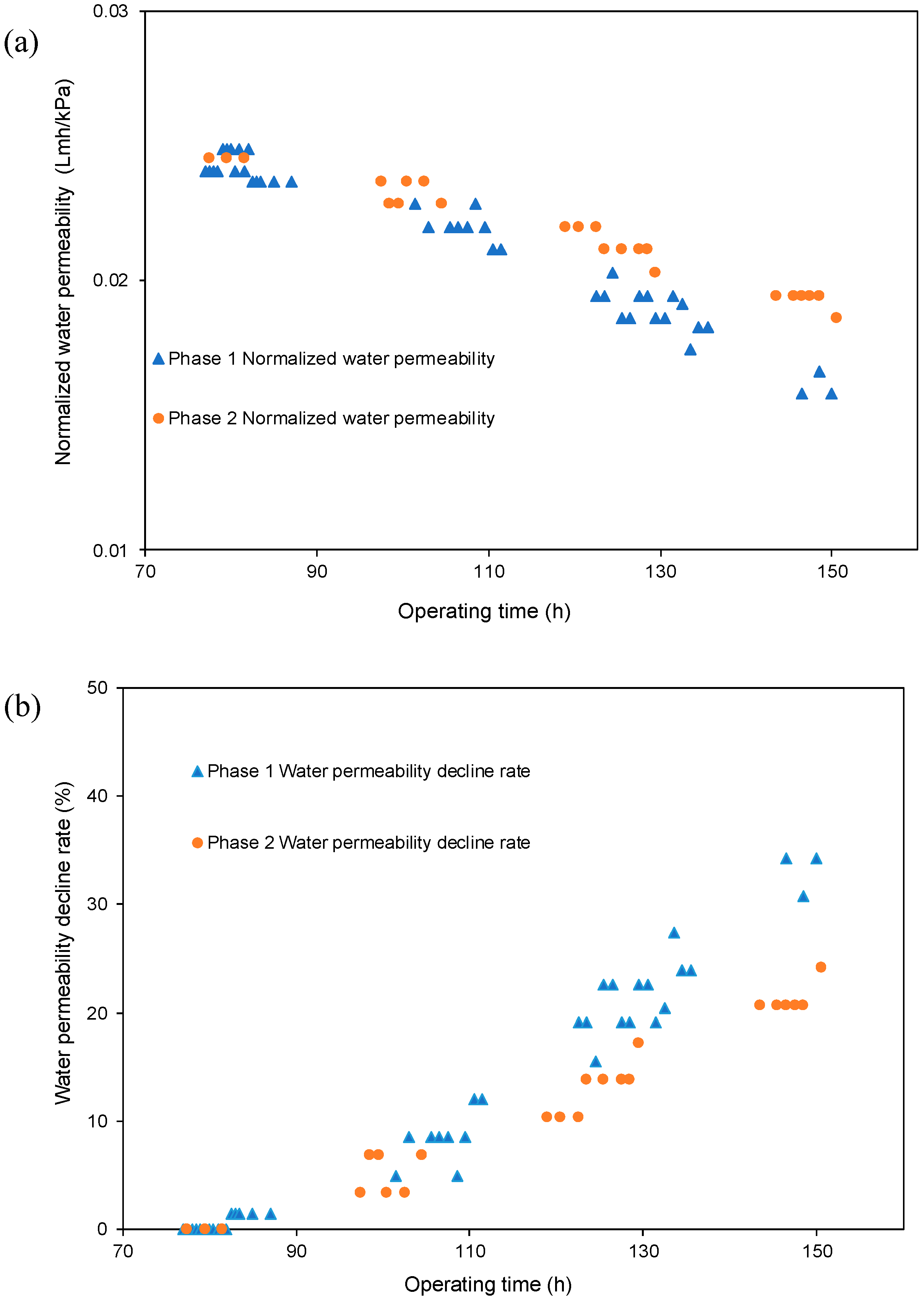
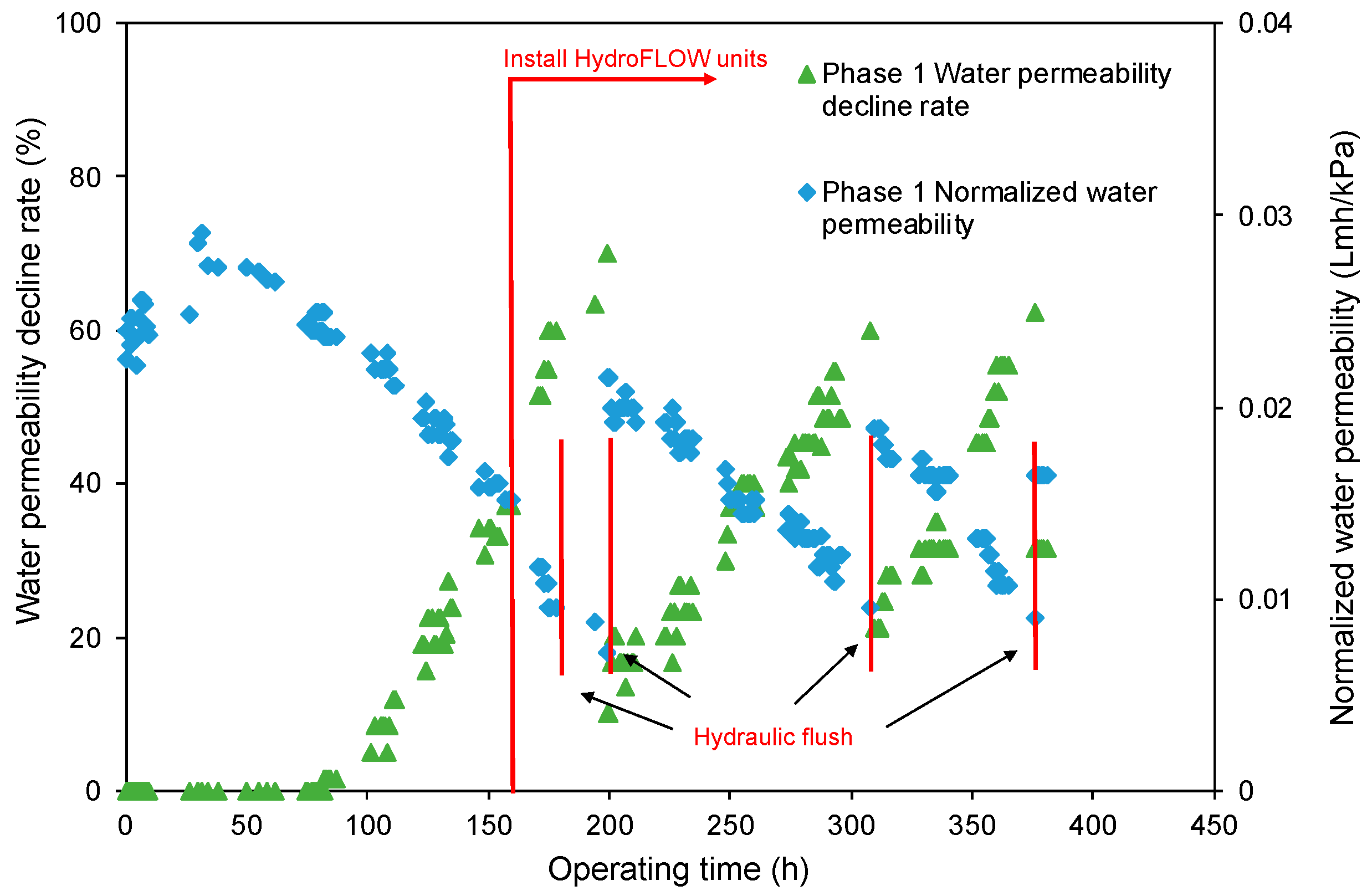
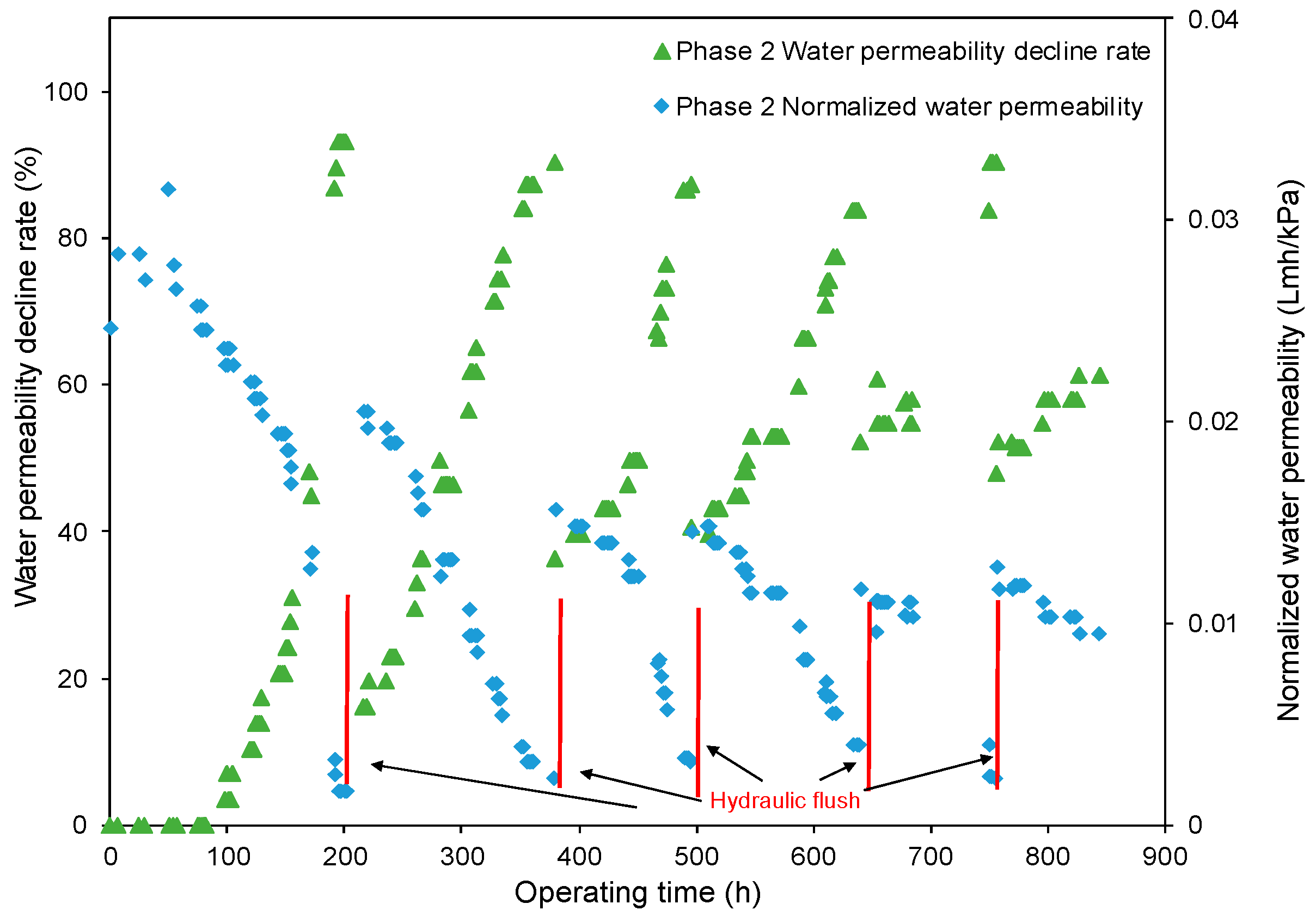
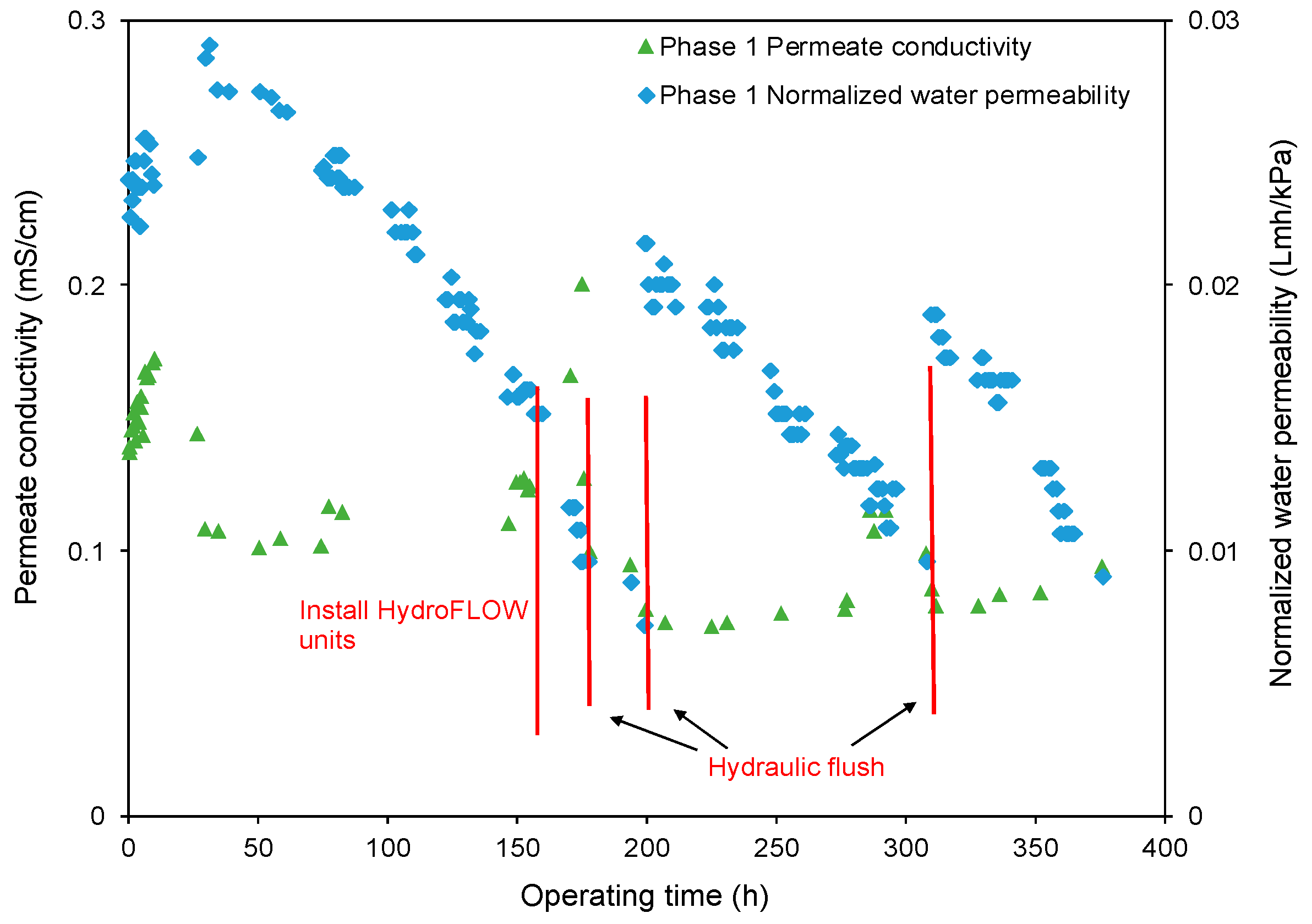
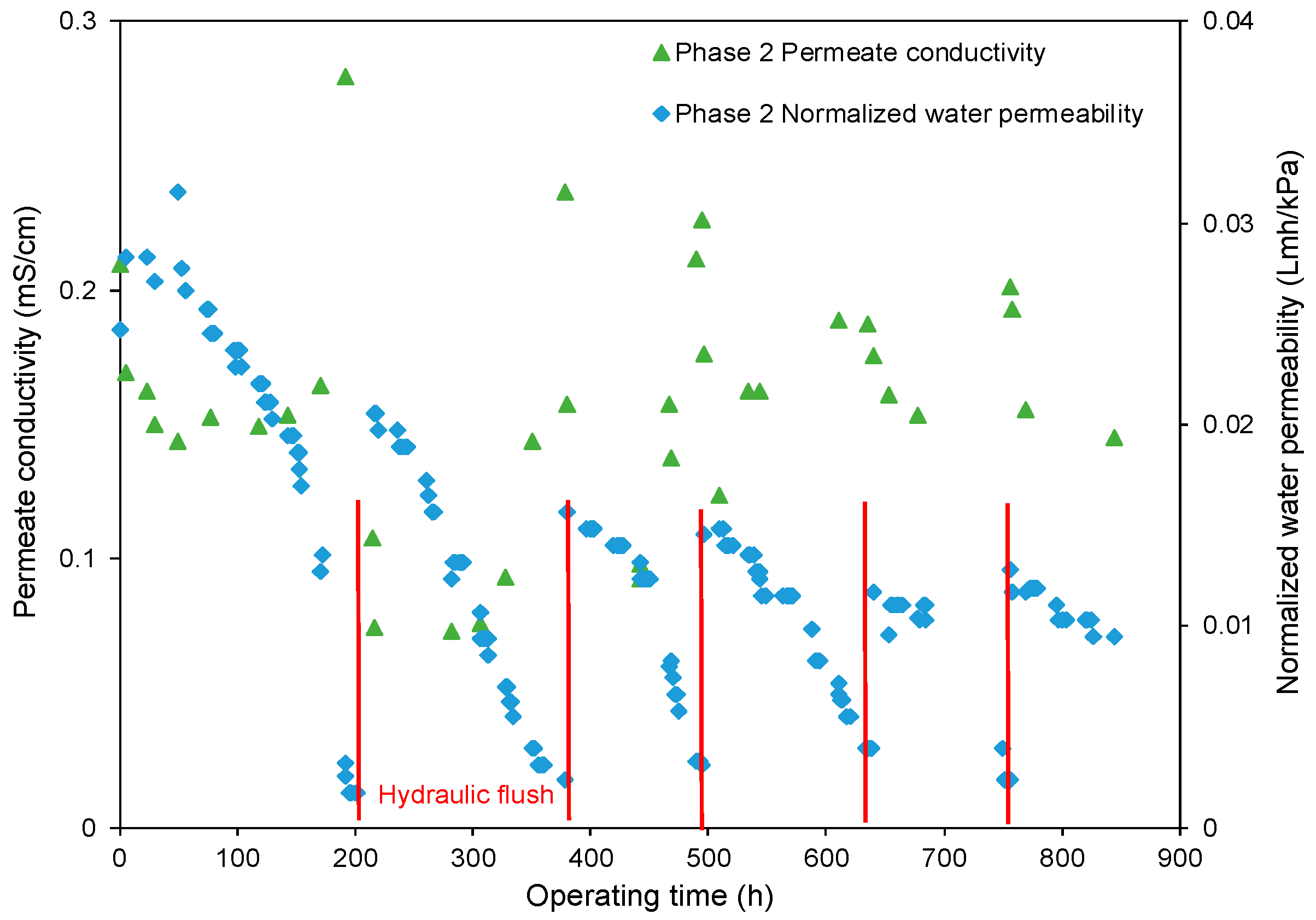
- The decreasing rate of the normalized water permeability (k value) with the HydroFLOW units during Phase 2 (P2) testing improved by 38.3% and 14.3% as compared to the permeate flux in Phase 1 (P1) testing in the 150 h (without HydroFLOW) and 370 h operation, respectively. After 700 h operation and 4 hydraulic flushes, the k value of P2 remained the same as the first 150 h operation, indicating less fouling and that the membrane performance had been improved in P2 by the installation of the EMF devices. The k value also shows less hydraulic flush was required with the EMF devices, which reduces the cleaning downtime. However, the NWP declined continuously despite the installation of HydroFLOW units. This infers the EMF could alleviate the fouling to a certain level but was not able to completely prevent it in the accelerated fouling process.
- In P1 after the installation of HydroFLOW units, the applied EMF loosened and shed the existing fouling layer from the water pipeline and RO membrane surface, resulting in clogging of the feedwater flow channels in the RO elements. Hydraulic flush facilitated the removal of the clogging particles from RO feed flow channels, and partially restored membrane performance.
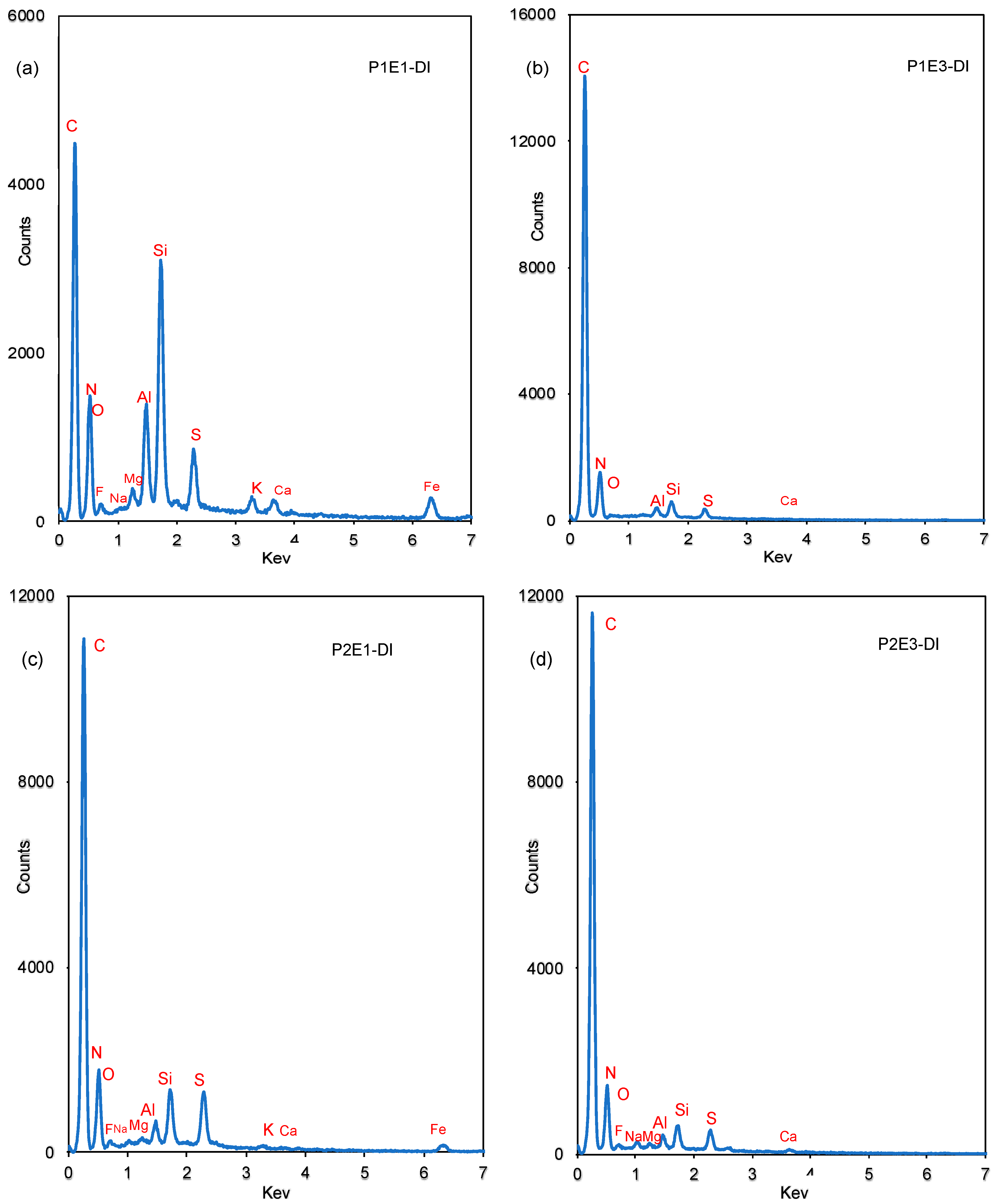
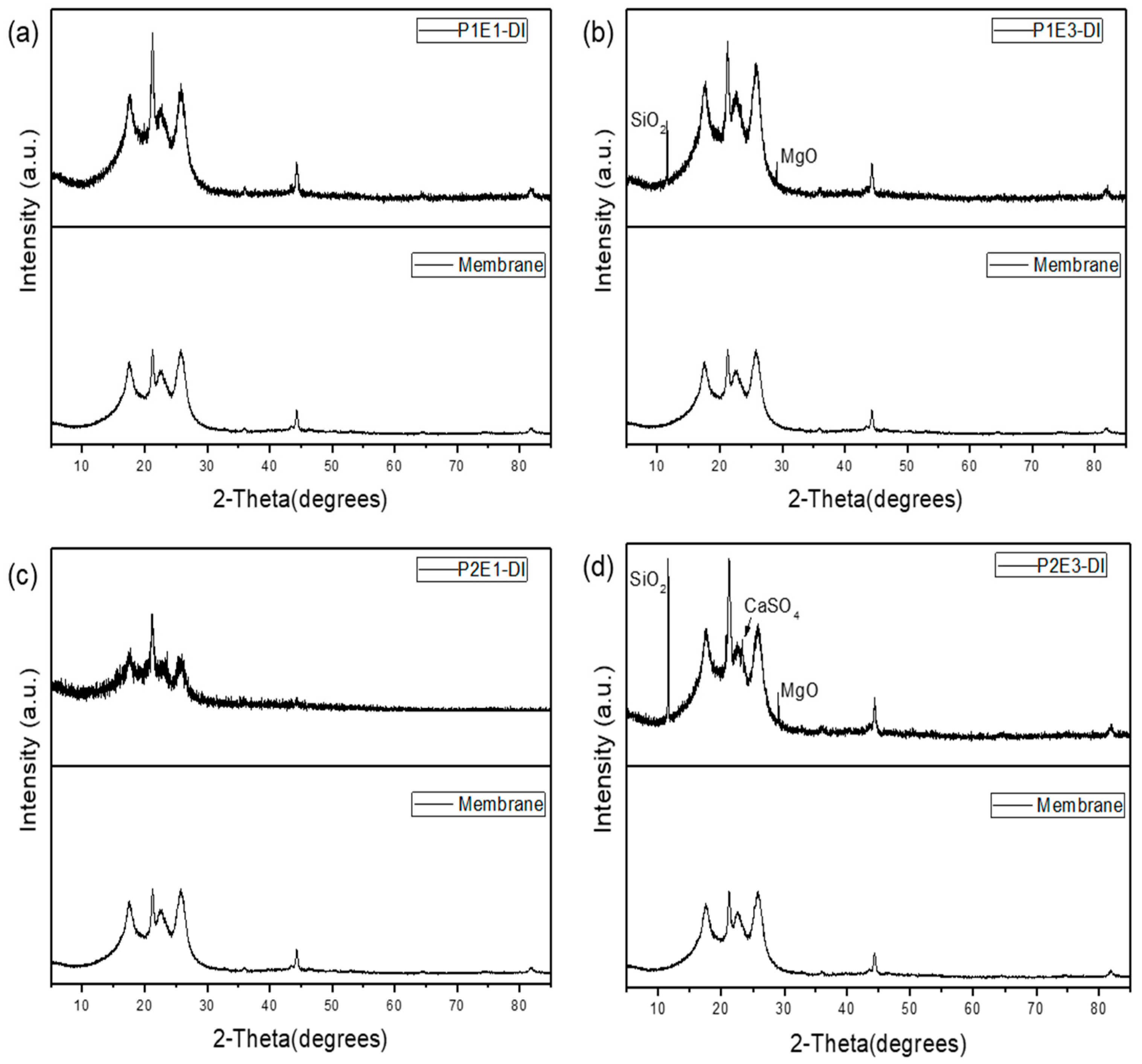
- Membrane autopsy results by SEM, EDX and XRD revealed that in the presence of the EMF, the fouling and scaling layer formed on the RO membrane surface was looser and could be more easily removed by hydraulic flushing. The oscillating electric field induced by the HydroFLOW units improved ion migration which weakened the concentration polarization on the membrane surface and increased nucleation in the bulk solution. The comparison between the lead elements between P1 and P2 showed the HydroFLOW had a positive effect in controlling silica related colloidal fouling. The XRD results show amorphous colloidal clay fouling in the lead elements and there was no crystal formed on the membrane surface in both phases. The SiO2, MgO, and CaSO4 crystals were detected only in tail-end RO elements of both P1 and P2 phases.
- The EMF slightly increased the permeate conductivity during groundwater desalination. The existence of the EMF prevented ions from scaling on the membrane surface and increased the free dissolved ions near the membrane surface, thus resulting in more ions being transported through the membrane. However, further studies are needed to investigate the effect of the EMF on salt transport under different operating conditions.
- For commercial applications, this study suggested that the EMF is more effective in pretreatment rather than cleaning of fouled or scaled membranes. EMF could remove the scales and precipitates in cartridge filters and pipelines, but caused clogging of the membrane feedwater flow channel. Also, HydroFLOW devices with higher intensity than the ones used in this study (model S38 and HS48) would achieve more prominent results.
- The HydroFLOW devices could provide a chemical-free alternative to control membrane fouling and scaling by alleviating the formation of a compact scaling layer on the membrane surface. Further experiments are needed to evaluate the EMF devices in treating various impaired water resources under different operating conditions, e.g., at different water recoveries, with/without addition of acids and scale inhibitors, and with different strengths and durations of hydraulic flushing.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Malaeb, L.; Ayoub, G.M. Reverse osmosis technology for water treatment: State of the art review. Desalination 2011, 267, 1–8. [Google Scholar] [CrossRef]
- Xu, P.; Capito, M.; Cath, T.Y. Selective removal of arsenic and monovalent ions from brackish water reverse osmosis concentrate. J. Hazard. Mater. 2013, 260, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Wenten, I. Reverse osmosis applications: Prospect and challenges. Desalination 2016, 391, 112–125. [Google Scholar] [CrossRef]
- Xu, P.; Bellona, C.; Drewes, J.E. Fouling of nanofiltration and reverse osmosis membranes during municipal wastewater reclamation: Membrane autopsy results from pilot-scale investigations. J. Membr. Sci. 2010, 353, 111–121. [Google Scholar] [CrossRef]
- Melián-Martel, N.; Sadhwani, J.; Malamis, S.; Ochsenkühn-Petropoulou, M. Structural and chemical characterization of long-term reverse osmosis membrane fouling in a full scale desalination plant. Desalination 2012, 305, 44–53. [Google Scholar] [CrossRef]
- Ruiz-García, A.; Melián-Martel, N.; Mena, V. Fouling characterization of RO membranes after 11 years of operation in a brackish water desalination plant. Desalination 2018, 430, 180–185. [Google Scholar] [CrossRef]
- Antony, A.; Low, J.H.; Gray, S.; Childress, A.E.; Le-Clech, P.; Leslie, G. Scale formation and control in high pressure membrane water treatment systems: A review. J. Membr. Sci. 2011, 383, 1–16. [Google Scholar] [CrossRef]
- Kang, G.-d.; Cao, Y.-m. Development of antifouling reverse osmosis membranes for water treatment: A review. Water Res. 2012, 46, 584–600. [Google Scholar] [CrossRef]
- Goh, P.; Lau, W.; Othman, M.; Ismail, A. Membrane fouling in desalination and its mitigation strategies. Desalination 2018, 425, 130–155. [Google Scholar] [CrossRef]
- Jiang, S.; Li, Y.; Ladewig, B.P. A review of reverse osmosis membrane fouling and control strategies. Sci. Total Environ. 2017, 595, 567–583. [Google Scholar] [CrossRef]
- Anis, S.F.; Hashaikeh, R.; Hilal, N. Reverse osmosis pretreatment technologies and future trends: A comprehensive review. Desalination 2019, 452, 159–195. [Google Scholar] [CrossRef]
- Xu, P.; Cath, T.Y.; Robertson, A.P.; Reinhard, M.; Leckie, J.O.; Drewes, J.E. Critical Review of Desalination Concentrate Management, Treatment and Beneficial Use. Environ. Eng. Sci. 2013, 30, 502–514. [Google Scholar] [CrossRef]
- Tang, F.; Hu, H.-Y.; Sun, L.-J.; Sun, Y.-X.; Shi, N.; Crittenden, J.C. Fouling characteristics of reverse osmosis membranes at different positions of a full-scale plant for municipal wastewater reclamation. Water Res. 2016, 90, 329–336. [Google Scholar] [CrossRef]
- Mukhopadhyay, D. Method and Apparatus for High Efficiency Reverse Osmosis Operation. U.S. Patent 6537456B2, 25 March 2003. [Google Scholar]
- Tong, T.; Elimelech, M. The global rise of zero liquid discharge for wastewater management: Drivers, technologies, and future directions. Environ. Sci. Technol. 2016, 50, 6846–6855. [Google Scholar] [CrossRef] [PubMed]
- Oren, Y.; Korngold, E.; Daltrophe, N.; Messalem, R.; Volkman, Y.; Aronov, L.; Weismann, M.; Bouriakov, N.; Glueckstern, P.; Gilron, J. Pilot studies on high recovery BWRO-EDR for near zero liquid discharge approach. Desalination 2010, 261, 321–330. [Google Scholar] [CrossRef]
- Juby, G.; Zacheis, A.; Shih, W.; Ravishanker, P.; Mortazavi, B.; Nusser, M.D. Eastern Municipal Water District and Carollo Engineers. Evaluation and Selection of Available Processes for a Zero-Liquid Discharge System for the Perris, California, Ground Water Basin; DWPR No. 149; U.S. Department of the Interior, Bureau of Reclamation: Denver, CO, USA, 2008.
- Warsinger, D.M.; Tow, E.W.; Nayar, K.G.; Maswadeh, L.A. Energy efficiency of batch and semi-batch (CCRO) reverse osmosis desalination. Water Res. 2016, 106, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Rouina, M.; Kariminia, H.-R.; Mousavi, S.A.; Shahryari, E. Effect of electromagnetic field on membrane fouling in reverse osmosis process. Desalination 2016, 395, 41–45. [Google Scholar] [CrossRef]
- Piyadasa, C.; Ridgway, H.F.; Yeager, T.R.; Stewart, M.B.; Pelekani, C.; Gray, S.R.; Orbell, J.D. The application of electromagnetic fields to the control of the scaling and biofouling of reverse osmosis membranes-A review. Desalination 2017, 418, 19–34. [Google Scholar] [CrossRef]
- Brower, J. Magnetic water treatment. Pollut. Eng. 2005, 37, 26–28. [Google Scholar]
- Ambashta, R.D.; Sillanpää, M. Water purification using magnetic assistance: A review. J. Hazard. Mater. 2010, 180, 38–49. [Google Scholar] [CrossRef]
- Alabi, A.; Chiesa, M.; Garlisi, C.; Palmisano, G. Advances in anti-scale magnetic water treatment. Environ. Sci. Water Res. Technol. 2015, 1, 408–425. [Google Scholar] [CrossRef]
- Pelekani, C.; Ostarcevic, E.; Drikas, M.; Patrick, C.; Cook, D. Reverse Osmosis Desalination: Assessment of a Novel Electromagnetic Field for Scale Control. In Proceedings of the International Desalination Association World Congress on Desalination and Water Reuse, Singapore, 11–16 September 2005. [Google Scholar]
- Al-Qahtani, H. Effect of magnetic treatment on Gulf seawater. Desalination 1996, 107, 75–81. [Google Scholar] [CrossRef]
- Zhang, Y.; Chong, T.; Fane, A.; Law, A.; Coster, H.; Winters, H. Implications of enhancing critical flux of particulates by AC fields in RO desalination and reclamation. Desalination 2008, 220, 371–379. [Google Scholar] [CrossRef]
- Gorey, C.; Escobar, I.C.; Gruden, C.L.; Cai, G. Development of microbial sensing membranes. Desalination 2009, 248, 99–105. [Google Scholar] [CrossRef]
- Zeppenfeld, K. Prevention of CaCO3 scale formation by trace amounts of copper (II) in comparison to zinc (II). Desalination 2010, 252, 60–65. [Google Scholar] [CrossRef]
- Sehn, P. Fluoride removal with extra low energy reverse osmosis membranes: Three years of large scale field experience in Finland. Desalination 2008, 223, 73–84. [Google Scholar] [CrossRef]
- Alimi, F.; Tlili, M.; Amor, M.B.; Gabrielli, C.; Maurin, G. Influence of magnetic field on calcium carbonate precipitation. Desalination 2007, 206, 163–168. [Google Scholar] [CrossRef]
- Xiaokai, X.; Chongfang, M.; Yongchang, C. Investigation on the Electromagnetic Anti-Fouling Technology for Scale Prevention. Chem. Eng. Technol. Ind. Chem. Plant Equip. Process Eng. Biotechnol. 2005, 28, 1540–1545. [Google Scholar] [CrossRef]
- Vedavyasan, C. Pontential use of magnetic fields—A perspective. Desalination 2001, 134, 105–108. [Google Scholar] [CrossRef]
- Corbett, B.E. Evaluation of Reverse Osmosis Scaling Prevention Devices at High Recovery; US Department of the Interior: Washington, DC, USA, 2003; pp. 1–35.
- Szkatula, A.; Balanda, M.; Kopeć, M. Magnetic treatment of industrial water. Silica activation. Eur. Phys. J. AP 2002, 18, 41–49. [Google Scholar] [CrossRef]
- Gabrielli, C.; Jaouhari, R.; Maurin, G.; Keddam, M. Magnetic water treatment for scale prevention. Water Res. 2001, 35, 3249–3259. [Google Scholar] [CrossRef]
- Palmer, N.T.; Le, H.; Harrington, P.; Furukawa, D. An EMF Antiscaling System for Desalination of Queensland Nickel’s Wastewater. In Proceedings of the International Desalination Association World Congress, Singapore, 11–16 September 2005. [Google Scholar]
- BGNDRF. Water Quality Analysis at the Brackish Groundwater National Desalination Research Facility. Available online: https://www.usbr.gov/research/bgndrf/water.html (accessed on 5 May 2019).
- Wang, Y.; He, W.; Müller, J.-D. Sensitivity analysis and gradient-based optimisation of feed spacer shape in reverse osmosis membrane processes using discrete adjoint approach. Desalination 2019, 449, 26–40. [Google Scholar] [CrossRef]
- Lutz, H. Ultrafiltration for Bioprocessing; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Bard, A.J.; Faulkner, L.R.; Leddy, J.; Zoski, C.G. Electrochemical Methods: Fundamentals and Applications; Wiley: New York, NY, USA, 1980; Volume 2. [Google Scholar]
- Lee, S.; Lee, C.-H. Effect of operating conditions on CaSO4 scale formation mechanism in nanofiltration for water softening. Water Res. 2000, 34, 3854–3866. [Google Scholar] [CrossRef]
- Cho, Y.I.; Lane, J.; Kim, W. Pulsed-power treatment for physical water treatment. Int. Commun. Heat Mass Transf. 2005, 32, 861–871. [Google Scholar] [CrossRef]


| Water Quality Parameter | Unit | Well 1 | Well 2 |
|---|---|---|---|
| Temperature | °C | 37.2 ± 5.1 | 24.1 ± 2.5 |
| pH | pH unit | 7.6 ± 0.2 | 7.3 ± 0.1 |
| Electrical conductivity | µS/cm | 17,123 ± 304 | 6300 ± 353 |
| Total dissolved solids | mg/L | 1179 ± 267 | 5670 ± 346 |
| Langelier Saturation Index | SI | 0.17 ± 0.21 | 0.45 ± 0.18 |
| Total alkalinity (as CaCO3) | mg/L | 151 ± 14 | 222 ± 20 |
| Chloride | mg/L | 36.6 ± 1.3 | 538.5 ± 24.1 |
| Sulfate | mg/L | 641.2 ± 191.1 | 2952.5 ± 234.6 |
| Total hardness (as CaCO3) | mg/L | 209 ± 73 | 2488 ± 42 |
| Calcium | mg/L | 60 ± 21 | 486 ± 15 |
| Magnesium | mg/L | 14.4 ± 4.8 | 309.0 ± 9.2 |
| Potassium | mg/L | 4.7 ± 0.4 | 2.3 ± 0.2 |
| Silicon dioxide | mg/L | 25.4 ± 2.7 | 22.5 ± 1.6 |
| Sodium | mg/L | 305 ± 30 | 691 ± 74 |
| Strontium | mg/L | 1.9 ± 0.4 | 8.2 ± 0.2 |
| Parameter | Feedwater | Concentrate |
|---|---|---|
| Langelier Saturation Index | 1.07 | 1.80 |
| Stiff & Davis Stability Index | 0.69 | 1.16 |
| Ionic Strength (Molar) | 0.14 | 0.29 |
| CaSO4 (%Saturation) | 105 | 238 |
| BaSO4 (%Saturation) | 174 | 352 |
| SrSO4 (%Saturation) | 71 | 150 |
| Operation Period | Operating Time (h) | Total Elapsed Operating Time (h) | k (Lmh/kPa-h) |
|---|---|---|---|
| P1 at water recovery 50% and 150 h operation | 77 | 150 | (1.2 ± 0.32) × 10−4 |
| P1 between 2nd and 3rd flush | 110 | 308 | (0.89 ± 0.14) × 10−4 |
| P1 between 3rd and 4th flush | 66 | 376 | (1.4 ± 0.51) × 10−4 |
| P2 at water recovery 50% and 150 h operation | 77 | 150 | (0.74 ± 0.17) × 10−4 |
| P2 between 1st and 2nd flush | 163 | 379 | (1.2 ± 0.11) × 10−4 |
| P2 between 2nd and 3rd flush | 115 | 495 | (1.2 ± 0.76 × 10−4 |
| P2 between 3rd and 4th flush | 143 | 638 | (0.86 ± 0.32) × 10−4 |
| P2 between 4th and 5th flush | 116 | 753 | (0.77 ± 0.48) × 10−4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, W.; Xu, X.; Lin, L.; Wang, H.; Shaw, R.; Lucero, D.; Xu, P. A Pilot Study of an Electromagnetic Field for Control of Reverse Osmosis Membrane Fouling and Scaling During Brackish Groundwater Desalination. Water 2019, 11, 1015. https://doi.org/10.3390/w11051015
Jiang W, Xu X, Lin L, Wang H, Shaw R, Lucero D, Xu P. A Pilot Study of an Electromagnetic Field for Control of Reverse Osmosis Membrane Fouling and Scaling During Brackish Groundwater Desalination. Water. 2019; 11(5):1015. https://doi.org/10.3390/w11051015
Chicago/Turabian StyleJiang, Wenbin, Xuesong Xu, Lu Lin, Huiyao Wang, Randall Shaw, Daniel Lucero, and Pei Xu. 2019. "A Pilot Study of an Electromagnetic Field for Control of Reverse Osmosis Membrane Fouling and Scaling During Brackish Groundwater Desalination" Water 11, no. 5: 1015. https://doi.org/10.3390/w11051015
APA StyleJiang, W., Xu, X., Lin, L., Wang, H., Shaw, R., Lucero, D., & Xu, P. (2019). A Pilot Study of an Electromagnetic Field for Control of Reverse Osmosis Membrane Fouling and Scaling During Brackish Groundwater Desalination. Water, 11(5), 1015. https://doi.org/10.3390/w11051015








