Removal Efficiency and Mechanism of Cr(VI) from Aqueous Solution by Maize Straw Biochars Derived at Different Pyrolysis Temperatures
Abstract
:1. Introduction
2. Materials and Methods
2.1. Maize Straw Biochar Preparation
2.2. Physical and Chemical Characterization
2.3. Batch Removal Experiments
2.3.1. Effect of Pyrolysis Temperature on Cr(VI) Removal
2.3.2. Adsorption Isotherm and Capacity
2.3.3. Effect of pH, Temperature, and Co-Existing Anions
2.3.4. Kinetics of Cr(VI) Removal
2.4. Removal Mechanism
3. Results and Discussion
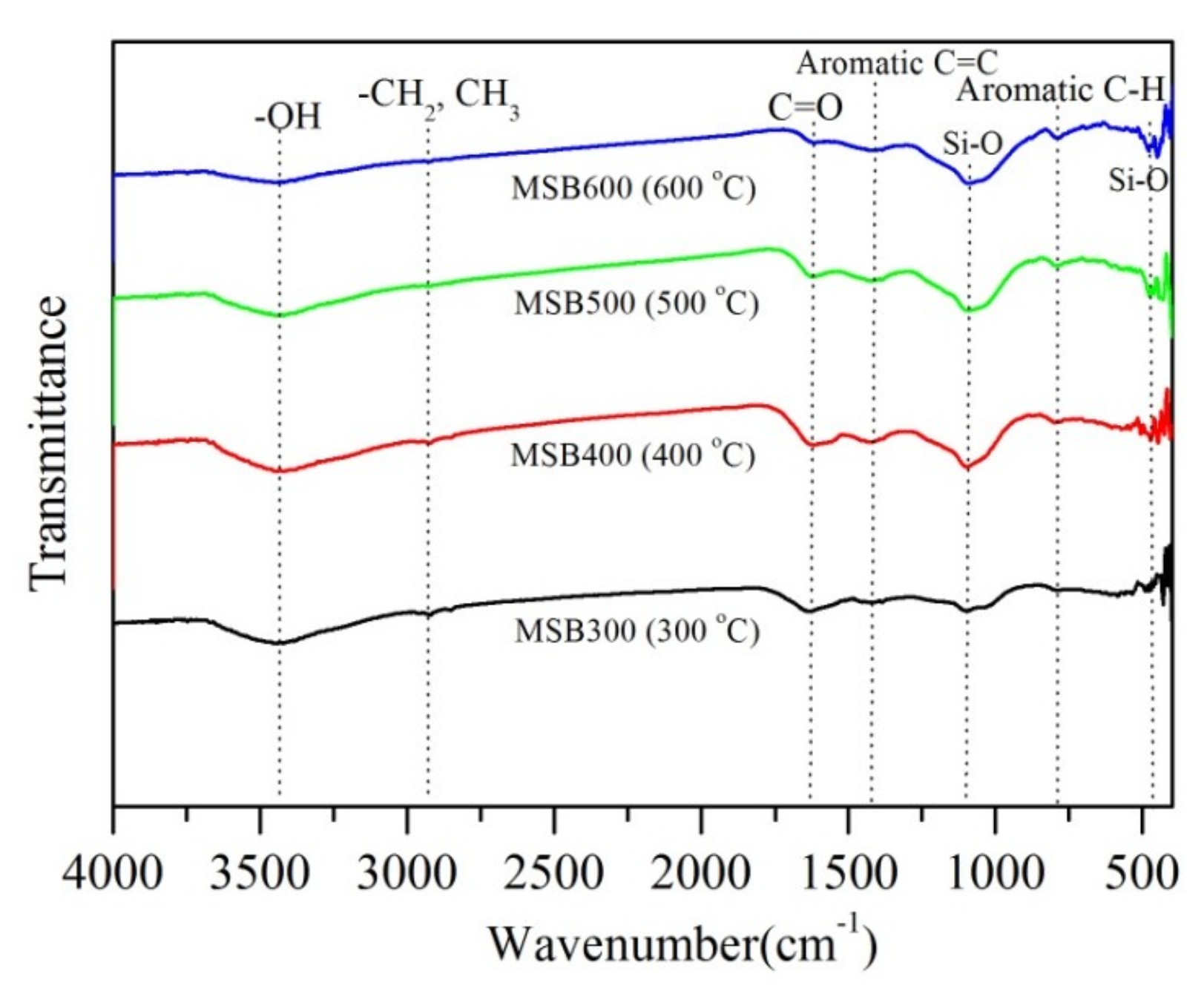
3.1. Effect of Pyrolysis Temperature on Biochar Characterization
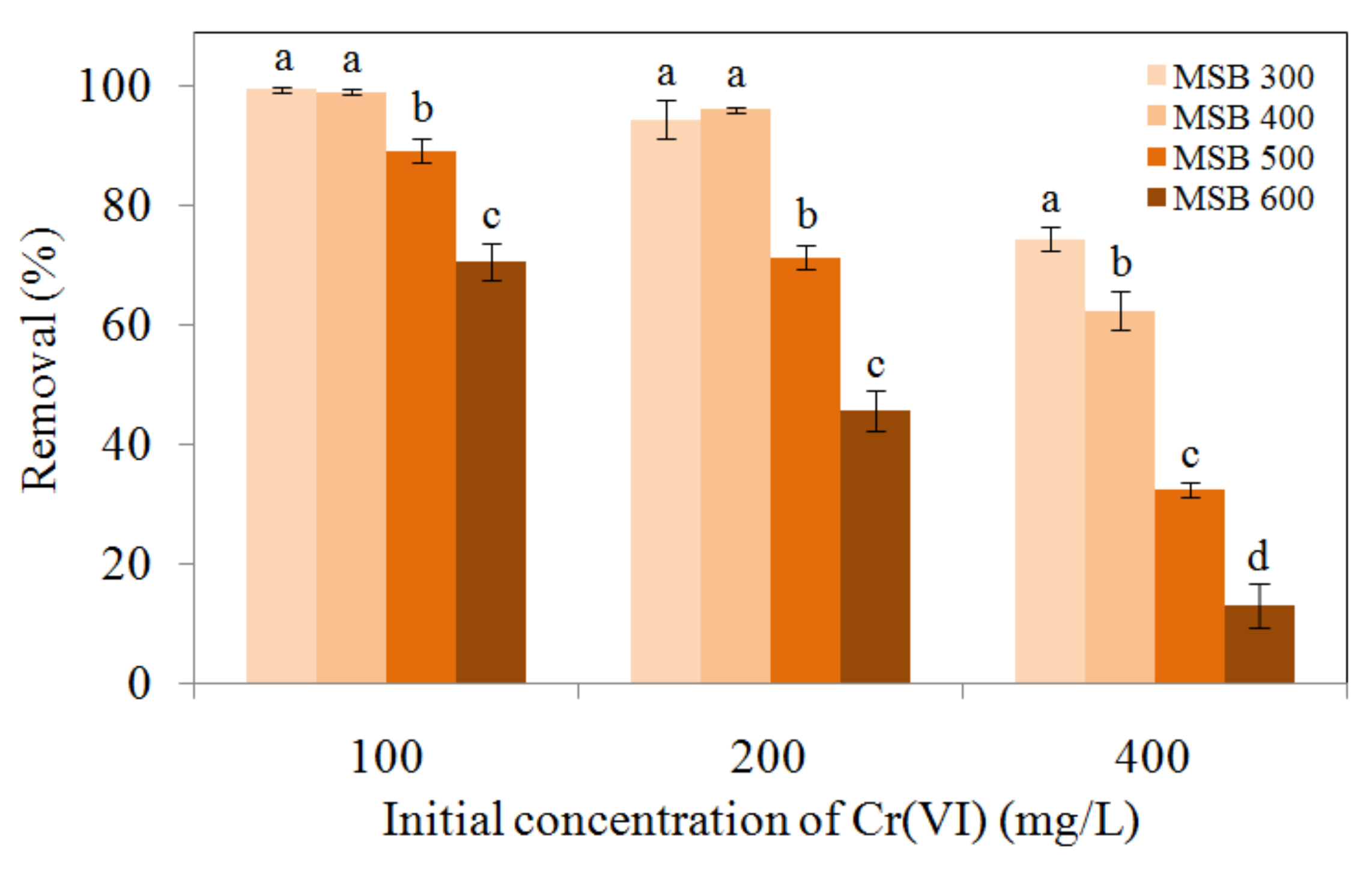
3.2. Comparison of Cr(VI) Removal by the Biochars from Different Pyrolysis Temperatures
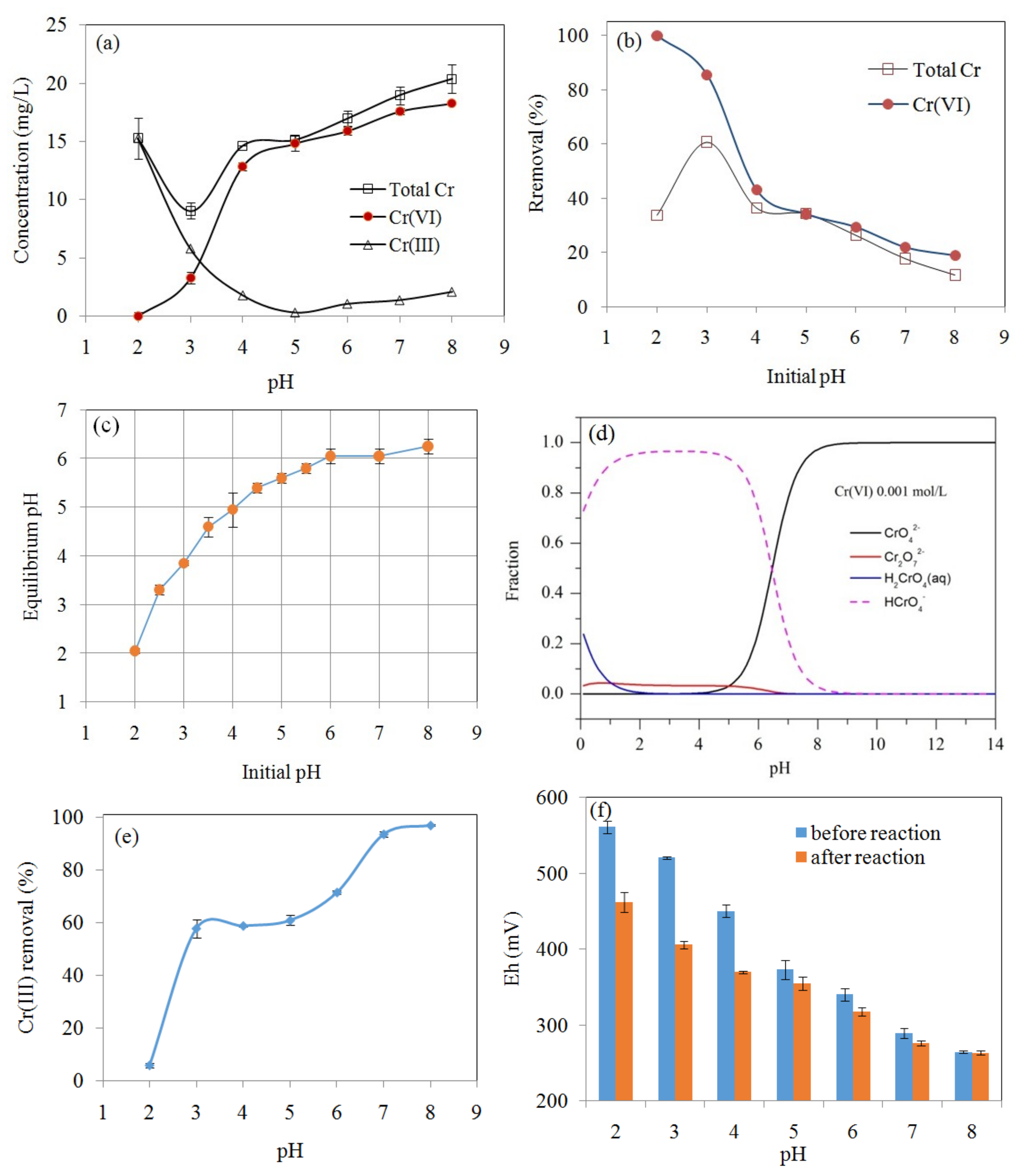
3.3. The Effect of pH on Cr(VI) Removal
3.4. Adsorption Isotherm and Capacity Comparison
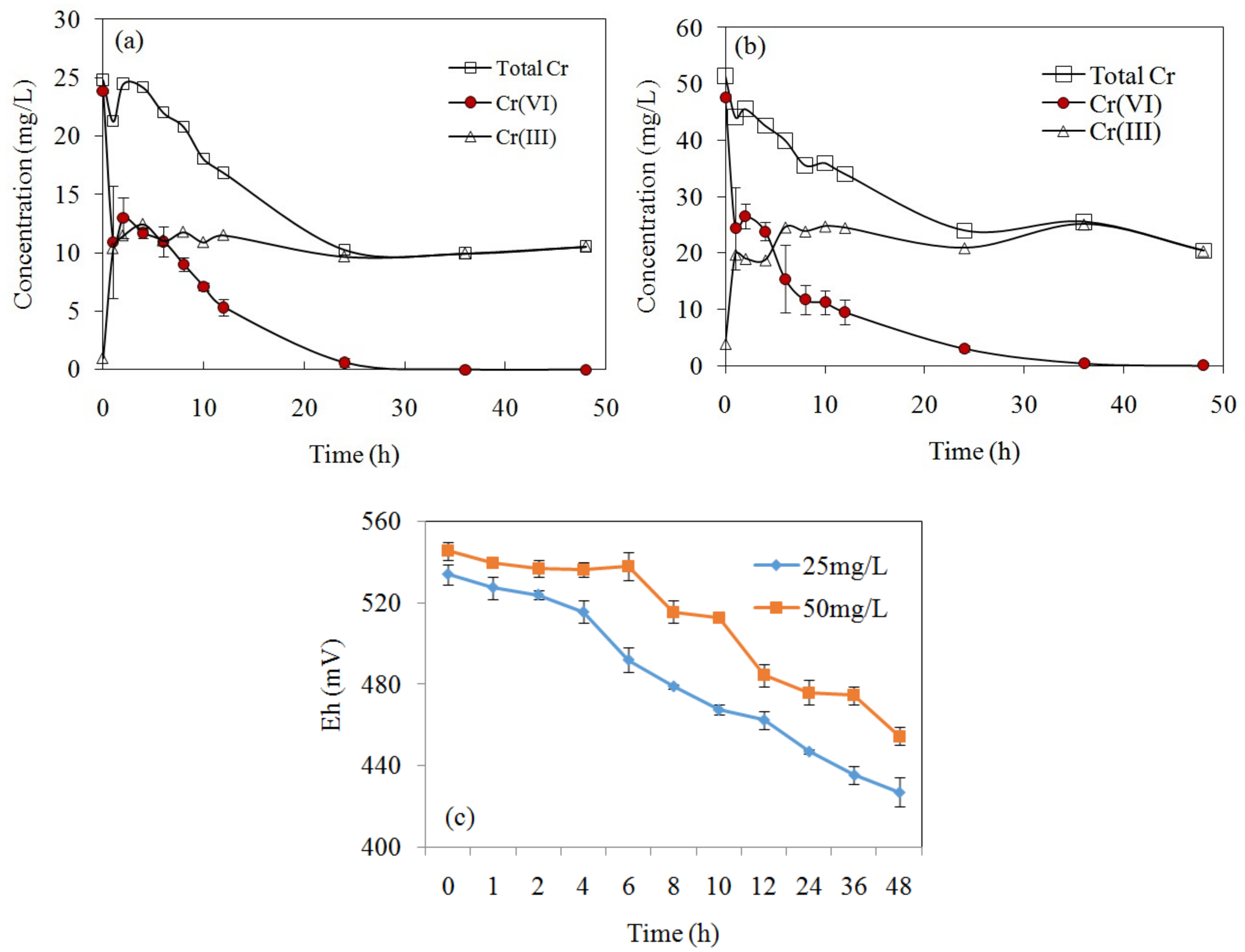
3.5. Cr(VI) Removal Kinetics
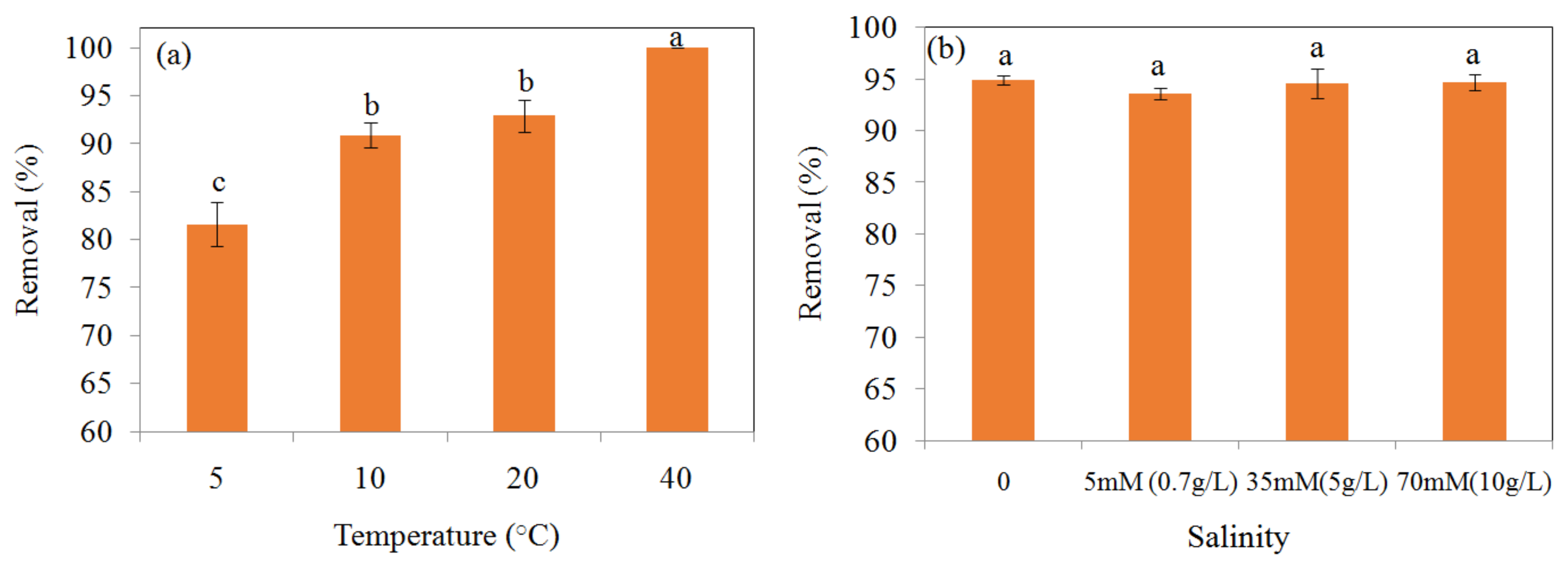
3.6. The Effect of Temperature and Coexisting Anions
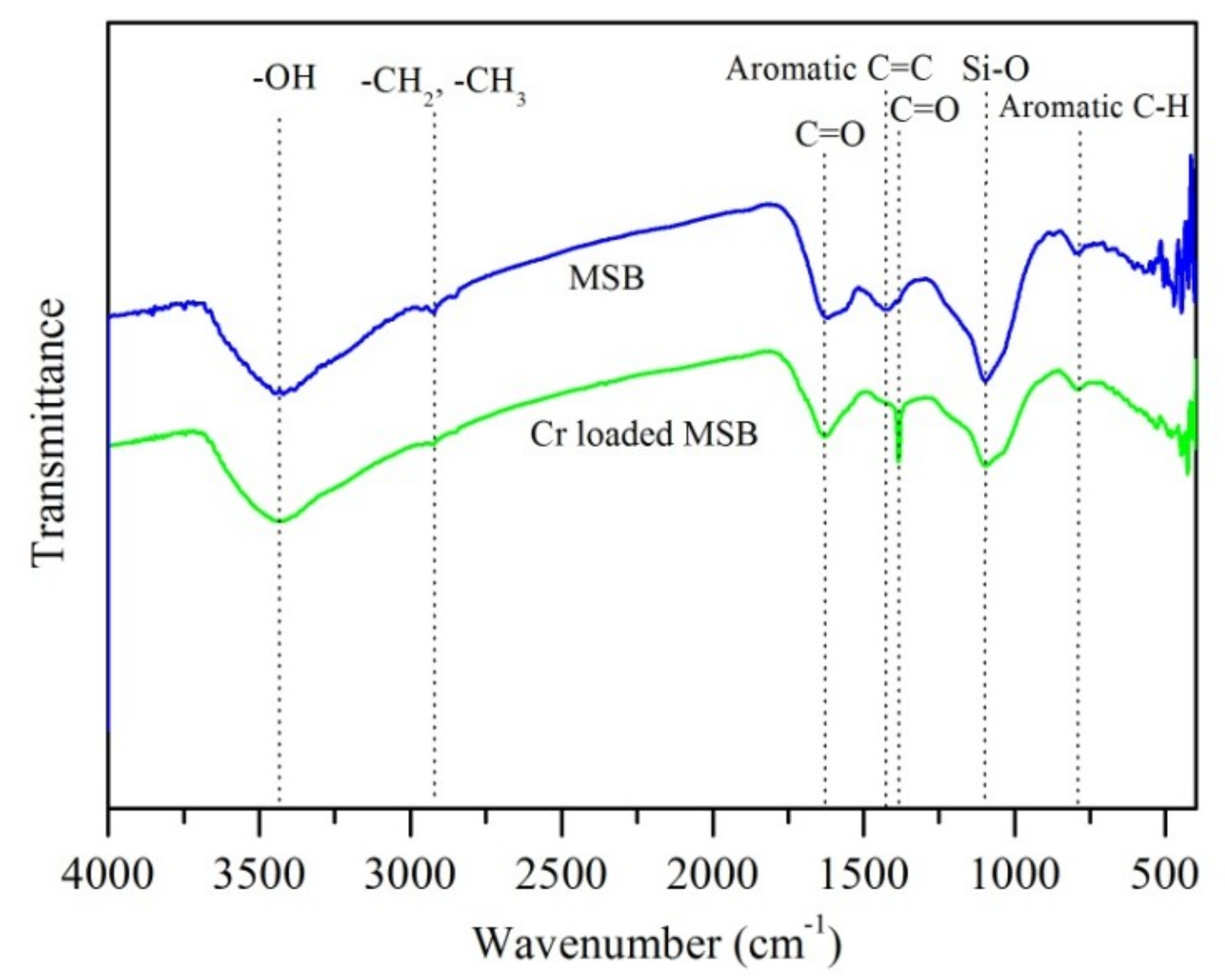
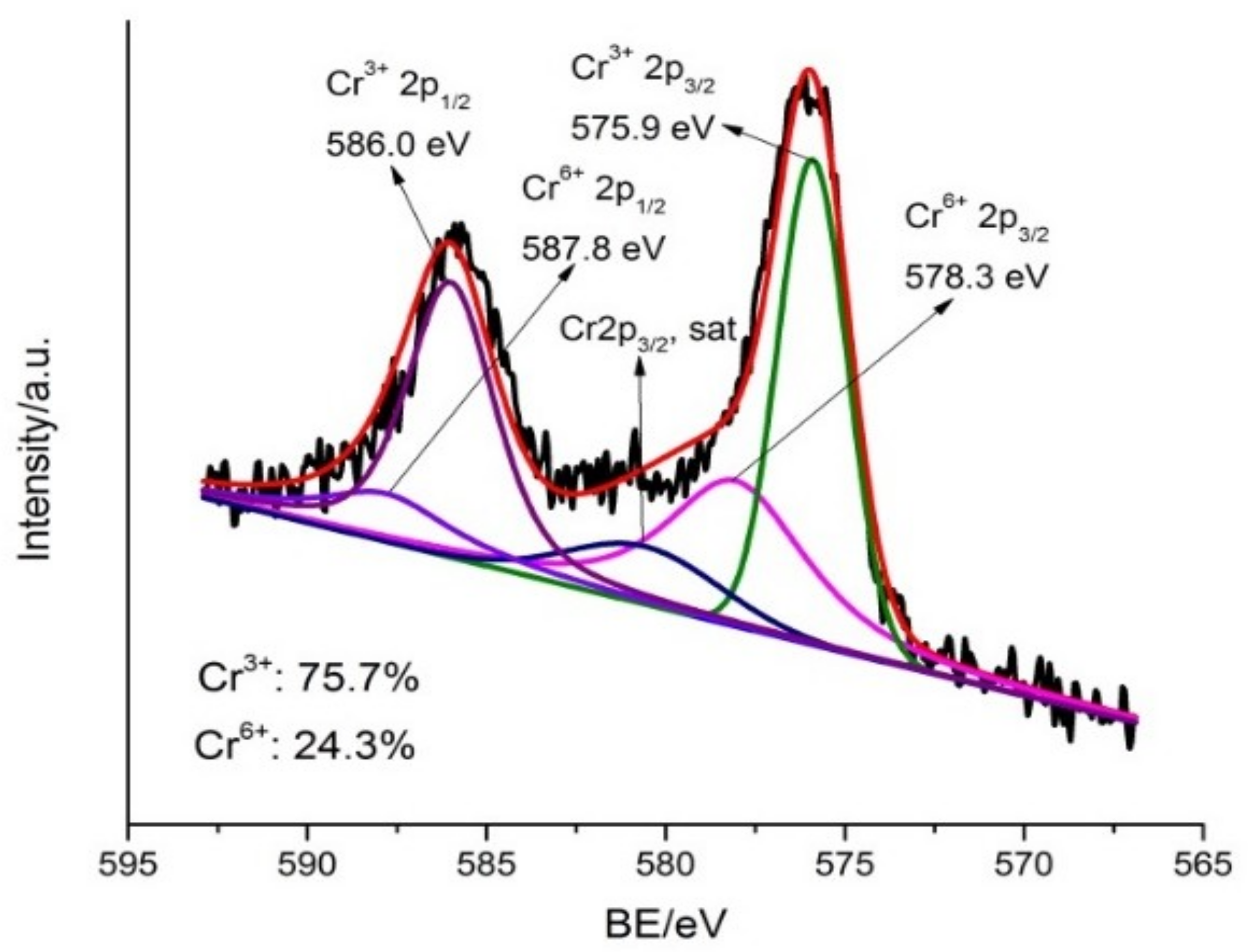
3.7. Cr(VI) Removal Mechanisms
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhou, L.; Liu, Y.G.; Liu, S.B.; Yin, Y.C.; Zeng, G.G.; Tan, X.F.; Hu, X.; Hu, X.G.; Jiang, L.H.; Ding, Y.; et al. Investigation of the adsorption-reduction mechanisms of hexavalent chromium by ramie biochars of different pyrolytic temperatures. Bioresour. Technol. 2016, 218, 351–359. [Google Scholar] [CrossRef]
- Zhang, X.; Fu, W.J.; Yin, Y.X.; Chen, Z.H.; Qiu, R.L.; Simonnot, M.O.; Wang, X.F. Adsorption-reduction removal of Cr(VI) by tobacco petiole pyrolytic biochar: Batch experiment, kinetic and mechanism studies. Bioresour. Technol. 2018, 268, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Cibati, A.; Foereid, B.; Bissessur, A.; Hapca, S. Assessment of Miscanthus × giganteus derived biochar as copper and zinc adsorbent: Study of the effect of pyrolysis temperature, pH and hydrogen peroxide modification. J. Clean. Prod. 2017, 162, 1285–1296. [Google Scholar] [CrossRef]
- Li, H.B.; Dong, X.L.; da Silva, E.B.; de Oliveira, L.M.; Chen, Y.S.; Ma, L.Q. Mechanisms of metal sorption by biochars: Biochar characteristics and modifications. Chemosphere 2017, 178, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Dawood, S.; Sen, T.K.; Phan, C. Synthesis and characterization of slow pyrolysis pine cone bio-char in the removal of organic and inorganic pollutants from aqueous solution by adsorption: Kinetic, equilibrium, mechanism and thermodynamic. Bioresour. Technol. 2017, 246, 76–81. [Google Scholar] [CrossRef]
- Kah, M.; Sigmund, G.; Xiao, F.; Hofmann, T. Sorption of ionizable and ionic organic compounds to biochar, activated carbon and other carbonaceous materials. Water Res. 2017, 124, 673–692. [Google Scholar] [CrossRef]
- Pan, J.; Jiang, J.; Xu, R. Removal of Cr (VI) from aqueous solutions by Na2SO3/FeSO4 combined with peanut straw biochar. Chemosphere 2014, 101, 71–76. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Wang, B.Y.; Xin, J.; Sun, P.; Wu, D. Adsorption behavior and mechanism of Cr(VI) by modified biochar derived from Enteromorpha prolifera. Ecotoxicol. Environ. Saf. 2018, 164, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Yin, Z.; Liu, F.; Zhang, M.; Lv, Y.; Hao, Z.; Pan, G.; Zhang, J. Environmentally persistent free radicals mediated removal of Cr(VI) from highly saline water by corn straw biochars. Bioresour. Technol. 2018, 260, 294–301. [Google Scholar] [CrossRef]
- Jung, K.W.; Kim, K.; Jeong, T.U.; Ahn, K.H. Influence of pyrolysis temperature on characteristics and phosphate adsorption capability of biochar derived from waste-marine macroalgae (Undaria pinnatifida roots). Bioresour. Technol. 2016, 20, 1024–1028. [Google Scholar] [CrossRef]
- Luo, L.; Xu, C.; Chen, Z.; Zhang, S. Properties of biomass-derived biochars: Combined effects of operating conditions and biomass types. Bioresour. Technol. 2015, 192, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Shan, B.Q.; Tang, W.Z.; Zhu, Y.Y. Comparison of cadmium and lead sorption by Phyllostachys pubescens biochar produced under a low-oxygen pyrolysis atmosphere. Bioresour. Technol. 2017, 238, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.L.; Ma, L.Q.; Li, Y.C. Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J. Hazard. Mater. 2011, 190, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.; Rajput, S.; Singh, V.K.; Steele, P.H.; Pittman, C.U., Jr. Modeling and evaluation of chromium remediation from water using low cost bio-char, a green adsorbent. J. Hazard. Mater. 2011, 188, 319–333. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, Y.X.; Wang, H.T.; Lu, W.J.; Zhou, Z.Y.; Zhang, Y.C.; Ren, L.L. Influence of pyrolysis temperature on characteristics and heavy metal adsorptive performance of biochar derived from municipal sewage sludge. Bioresour. Technol. 2014, 164, 47–54. [Google Scholar] [CrossRef]
- Shen, Y.S.; Wang, S.L.; Tzou, Y.M.; Yan, Y.Y.; Kuan, W.H. Removal of hexavalent Cr by coconut coir and derived chars-The effect of surface functionality. Bioresour. Technol. 2012, 104, 165–172. [Google Scholar] [CrossRef]
- Zhang, W.; Mao, S.; Chen, H.; Huang, L.; Qiu, R. Pb(II) and Cr(VI) sorption by biochars pyrolyzed from the municipal wastewater sludge under different heating conditions. Bioresour. Technol. 2013, 147, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.; Saddique, M.T.; Naeem, A.; Westerhoff, P.; Mustafa, S.; Alum, A. Comparison of different methods for the point of zero charge determination of NiO. Ind. Eng. Chem. Res. 2011, 50, 10017–10023. [Google Scholar] [CrossRef]
- Karunanayake, A.G.; Todd, O.A.; Crowley, M.; Ricchetti, L.; Pittman, C.U., Jr.; Anderson, R.; Mohan, D.; Mlsna, T. Lead and cadmium remediation using magnetized and nonmagnetized biochar from Douglas fir. Chem. Eng. J. 2018, 331, 480–491. [Google Scholar] [CrossRef]
- Chun, Y.; Sheng, G.Y.; Chiou, C.T.; Xing, B.S. Compositions and sorptive properties of crop residue-derived chars. Environ. Sci. Technol. 2004, 38, 4649–4655. [Google Scholar] [CrossRef]
- Zhao, B.; O’Connor, D.; Zhang, J.; Peng, T.; Shen, Z.; Tsang, D.C.W.; Hou, D. Effect of pyrolysis temperature, heating rate, and residence time on rapeseed stem derived biochar. J. Clean. Prod. 2018, 174, 977–987. [Google Scholar] [CrossRef]
- Elangovan, R.; Philip, L.; Chandraraj, K. Biosorption of hexavalent and trivalent chromium by palm flower (Borassus aethiopum). Chem. Eng. J. 2008, 141, 99–111. [Google Scholar] [CrossRef]
- Pan, J.; Jiang, J.; Xu, R. Adsorption of Cr (III) from acidic solutions by crop straw derived biochars. J. Environ. Sci. 2013, 25, 1957–1965. [Google Scholar] [CrossRef]
- Agrafioti, E.; Kalderis, D.; Diamadopoulos, E. Arsenic and chromium removal from water using biochars derived from rice husk, organic solid wastes and sewage sludge. J. Environ. Manag. 2014, 133, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Tytlak, A.; Oleszczuk, P.; Dobrowolski, R. Sorption and desorption of Cr (VI) ions from water by biochars in different environmental conditions. Environ. Sci. Pollut. Res. Int. 2015, 22, 5985–5994. [Google Scholar] [CrossRef] [PubMed]
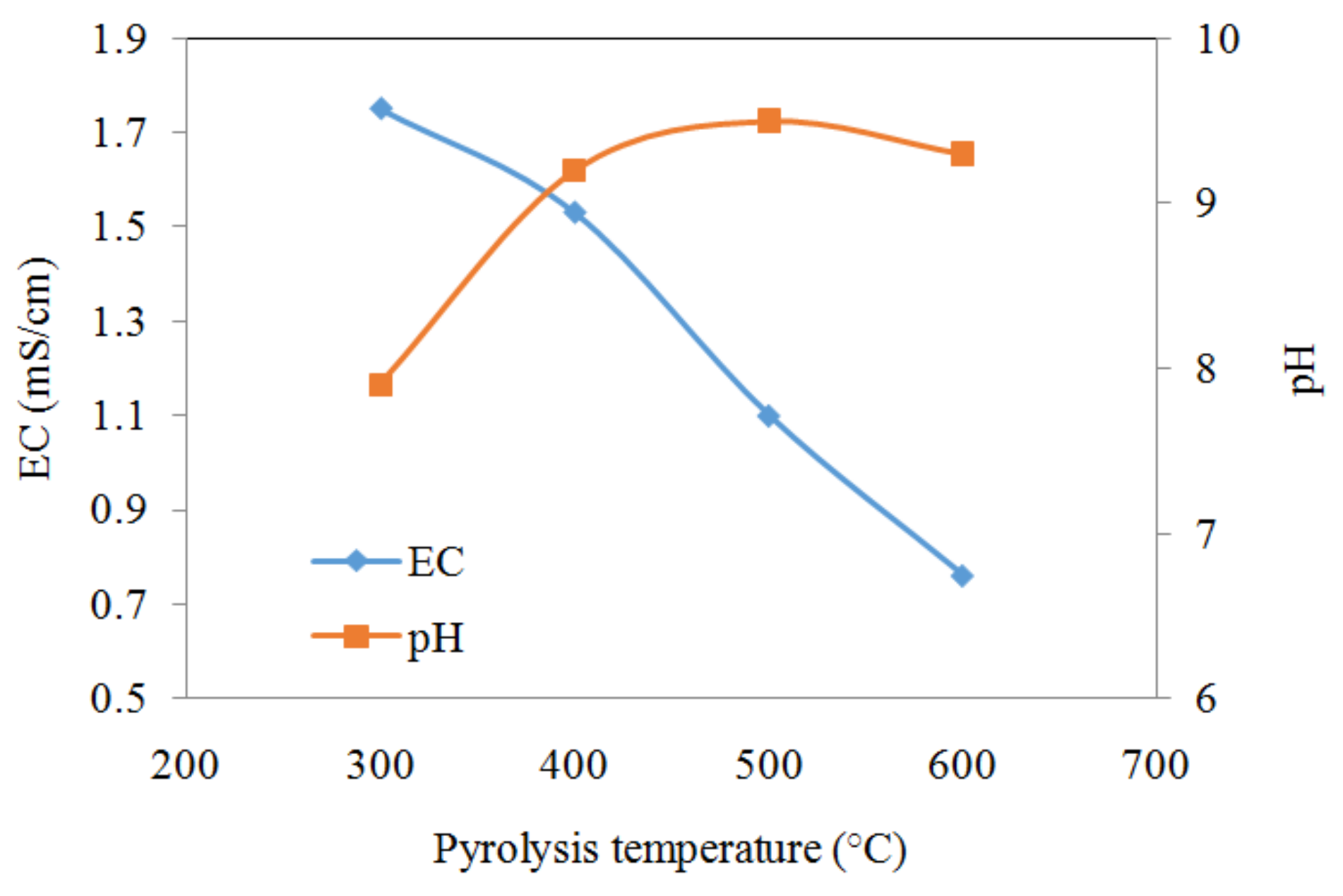


| Biochar | pH | EC (mS/cm) | Redox Potential (mV) | pHpzc | Ash (%) | Elemental Composition | Atomic Ratio | Surface Area (m2/g) | Pore Size (nm) | Pore Volume (cm3/g) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C (%) | H (%) | O (%) | N (%) | S (%) | H/C | (O + N)/C | O/C | |||||||||
| MSB300 | 7.4 | 1.75 | 47 | 7.9 | 50.5 | 24.14 | 2.25 | 20.93 | 2.19 | 0.96 | 1.12 | 0.81 | 0.65 | 10.3 | 13.7 | 0.034 |
| MSB400 | 9.1 | 1.53 | 34 | 9.2 | 57.9 | 25.85 | 1.68 | 14.83 | 1.91 | 1.16 | 0.78 | 0.56 | 0.43 | 17.1 | 11.5 | 0.047 |
| MSB500 | 9.4 | 1.10 | 33 | 9.5 | 63.4 | 22.08 | 0.99 | 12.94 | 1.41 | 0.97 | 0.54 | 0.55 | 0.44 | 24.5 | 13.9 | 0.065 |
| MSB600 | 10.1 | 0.76 | 53 | 9.3 | 67.9 | 26.44 | 0.68 | 9.54 | 1.46 | 0.99 | 0.31 | 0.37 | 0.27 | 31.1 | 9.0 | 0.062 |
| Absorbents | Langmuir | Freundlich | |||||
|---|---|---|---|---|---|---|---|
| qmax (mg/g) | qmax (mmol/g) | b (L/mg) | R2 | kf (mg1−n·Ln/g) | 1/n | R2 | |
| MSB300 | 91 ± 6 | 1.8 | 0.24 | 0.98 | 29.53 | 0.23 | 0.93 |
| MSB400 | 63 ± 3 | 1.2 | 0.73 | 0.99 | 30.22 | 0.16 | 0.83 |
| MSB500 | 42 ± 1 | 0.8 | 0.26 | 0.98 | 16.09 | 0.16 | 0.95 |
| MSB600 | 27 ± 3 | 0.5 | 0.12 | 0.99 | 0.14 | 0.82 | 0.96 |
| Absorbents | Maximum Adsorption Capacity (mg/g) | Best Fit Isotherm | Reference |
|---|---|---|---|
| sugar beet tailing biochar | 123 | Langmuir | Dong et al., 2011 [13] |
| Enteromorpha prolifera biochar | 88.17 | Langmuir | Chen et al., 2018 [8] |
| ramie residue biochar | 82.23 | Langmuir | Zhou et al., 2016 [1] |
| sewage sludge biochar | 64.1 | Langmuir | Agrafioti et al., 2014 [24] |
| coconut coir biochar | 31.1 | Langmuir | Shen et al., 2012 [16] |
| wood biochar | 25.27 | Langmuir | Zhang et al., 2018 [2] |
| wheat straw biochar | 24.6 | Langmuir | Tytlak et al., 2015 [25] |
| wicker biochar | 23.6 | Langmuir | Tytlak et al., 2015 [25] |
| MSB300 | 91 | Langmuir | This study |
| Initial Concentration (mg/L) | Pseudo-Second Order | Pseudo-First Order | ||||
|---|---|---|---|---|---|---|
| qe (mg/g) | k2 (g/mg·h) | R2 | qe (mg/g) | k1 (1/h) | R2 | |
| 25 | 6.5 | 0.04 | 0.99 | 5.0 | 0.1 | 0.91 |
| 50 | 12.7 | 0.02 | 0.99 | 8.3 | 0.1 | 0.98 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Zhang, M.; Lv, Q. Removal Efficiency and Mechanism of Cr(VI) from Aqueous Solution by Maize Straw Biochars Derived at Different Pyrolysis Temperatures. Water 2019, 11, 781. https://doi.org/10.3390/w11040781
Wang H, Zhang M, Lv Q. Removal Efficiency and Mechanism of Cr(VI) from Aqueous Solution by Maize Straw Biochars Derived at Different Pyrolysis Temperatures. Water. 2019; 11(4):781. https://doi.org/10.3390/w11040781
Chicago/Turabian StyleWang, Haixia, Mingliang Zhang, and Qi Lv. 2019. "Removal Efficiency and Mechanism of Cr(VI) from Aqueous Solution by Maize Straw Biochars Derived at Different Pyrolysis Temperatures" Water 11, no. 4: 781. https://doi.org/10.3390/w11040781
APA StyleWang, H., Zhang, M., & Lv, Q. (2019). Removal Efficiency and Mechanism of Cr(VI) from Aqueous Solution by Maize Straw Biochars Derived at Different Pyrolysis Temperatures. Water, 11(4), 781. https://doi.org/10.3390/w11040781





