Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source, and Bio-Fertilizer. Part 2: Water Recovery
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Preparation
2.2. Experimental Drying Tests
2.3. Water Condensation and Recovery
2.4. Condensed Water Characterization
2.5. Gas Chromatography–Mass Spectrometry
2.5.1. Chemicals and Reagents
2.5.2. Derivatization
2.5.3. GC–MS Instrument Conditions
2.5.4. Chemical Identification
3. Results
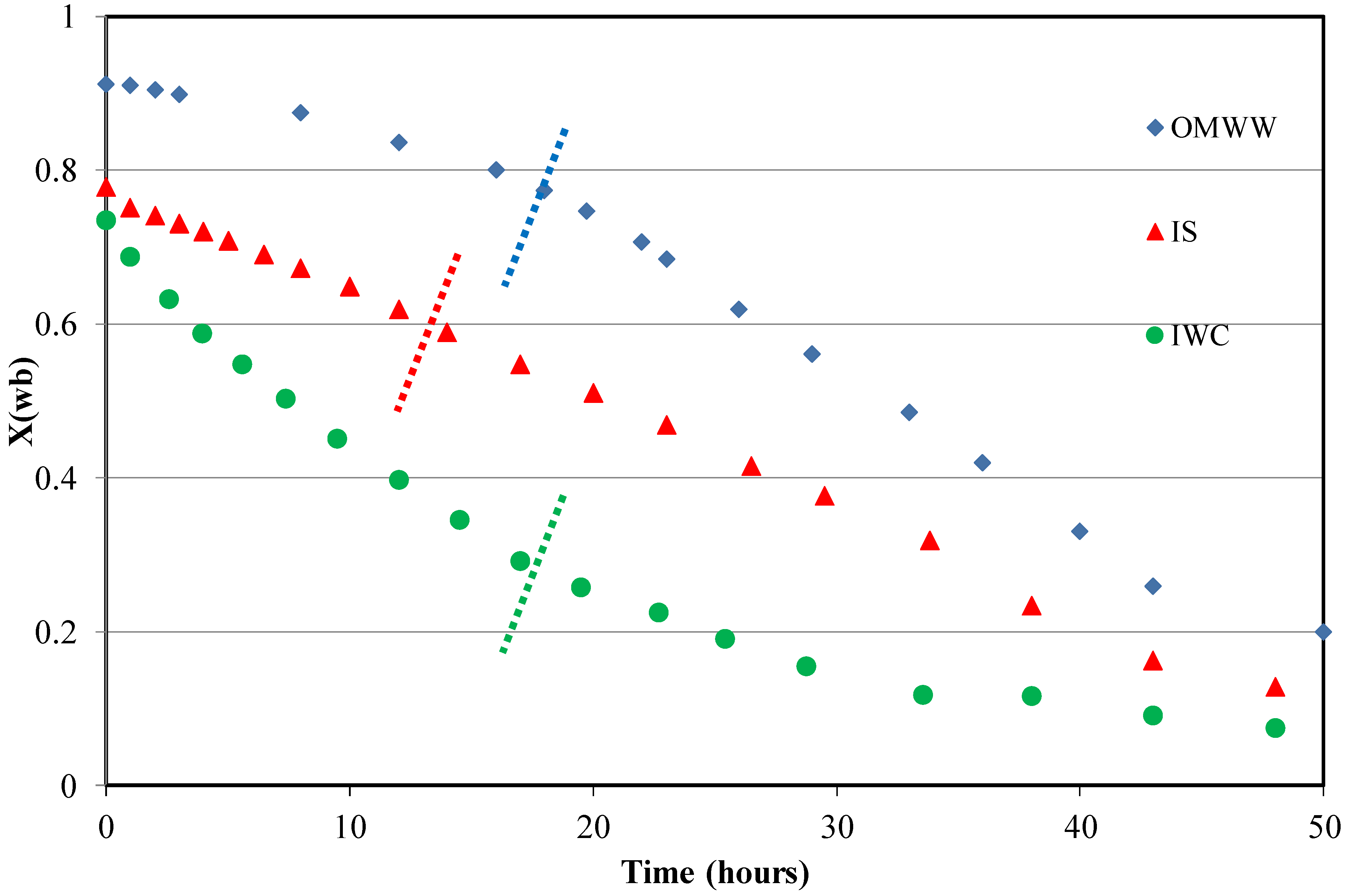
3.1. Kinetic Study
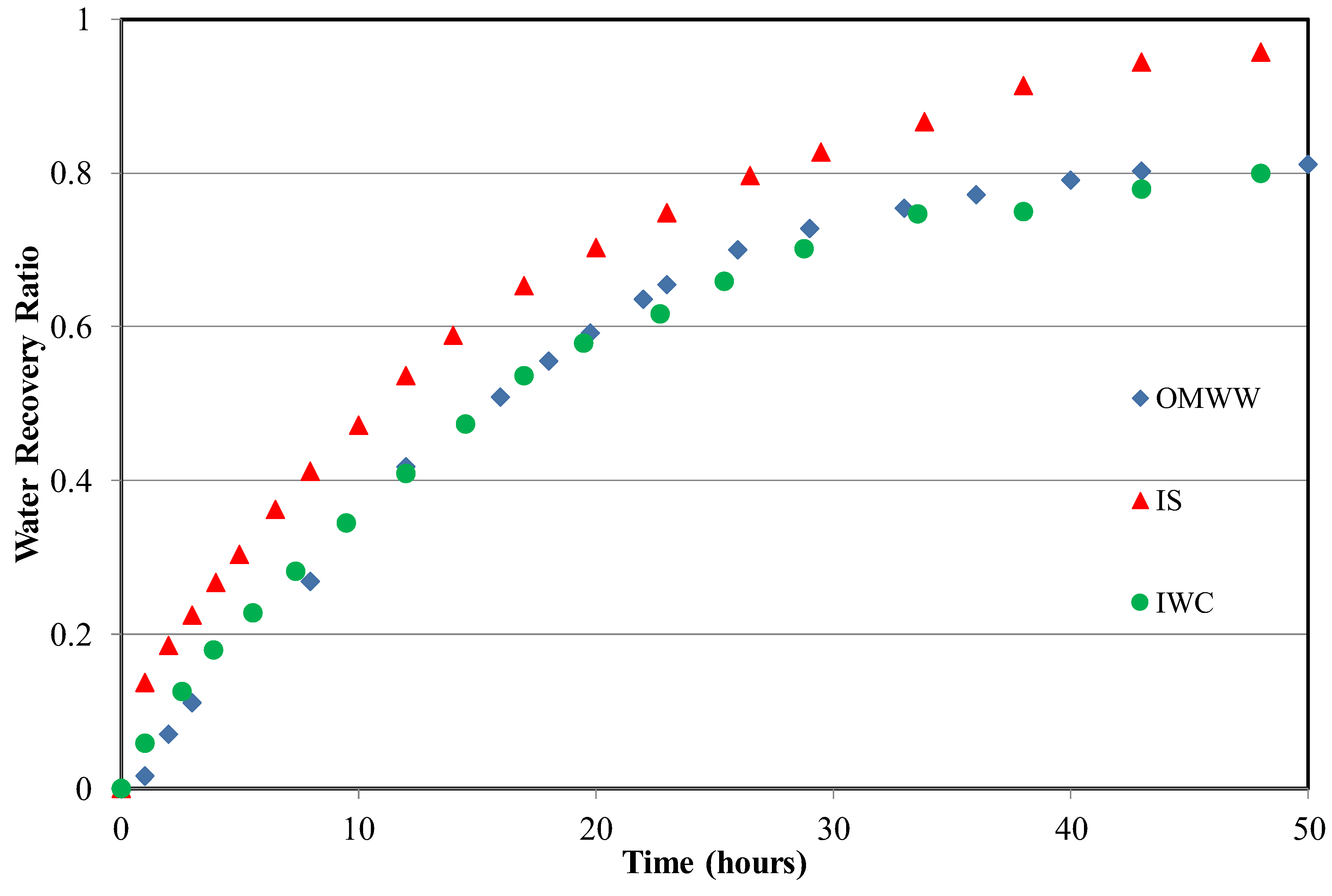
3.2. Water Recovery
3.3. Recovered Water Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Iglesias, A.; Garrote, L.; Flores, F.; Moneo, M. Challenges to Manage the Risk of Water Scarcity and Climate Change in the Mediterranean. Water Resour. Manag. 2007, 21, 775–788. [Google Scholar] [CrossRef]
- Angelakis, A.N.; Marecos Do Monte, M.H.F.; Bontoux, L.; Asano, T. The status of wastewater reuse practice in the Mediterranean basin: Need for guidelines. Water Res. 1999, 33, 2201–2217. [Google Scholar] [CrossRef]
- Mizyed, N.R. Challenges to treated wastewater reuse in arid and semi-arid areas. Environ. Sci. Policy 2013, 25, 186–195. [Google Scholar] [CrossRef]
- Pedrero, F.; Kalavrouziotis, I.; Alarcón, J.J.; Koukoulakis, P.; Asano, T. Use of treated municipal wastewater in irrigated agriculture—Review of some practices in Spain and Greece. Agric. Water Manag. 2010, 97, 1233–1241. [Google Scholar] [CrossRef]
- Khiari, B.; Wakkel, M.; Abdelmoumen, S.; Jeguirim, M. Dynamics and kinetics of cupric ion removal from wastewaters by Tunisian solid crude olive-oilwaste. Materials 2019, 12, 365. [Google Scholar] [CrossRef]
- Wakkel, M.; Khiari, B.; Zagrouba, F. Textile wastewater treatment by agro-industrial waste: Equilibrium modelling, thermodynamics and mass transfer mechanisms of cationic dyes adsorption onto low-cost lignocellulosic adsorbent. J. Taiwan Inst. Chem. Eng. 2019, 96, 439–452. [Google Scholar] [CrossRef]
- Khan, S.; Cao, Q.; Zheng, Y.M.; Huang, Y.Z.; Zhu, Y.G. Health risks of heavy metals in contaminated soils and food crops irrigated with wastewater in Beijing, China. Environ. Pollut. 2008, 152, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Wakkel, M.; Khiari, B. Basic red 2 and methyl violet adsorption by date pits: Adsorbent characterization, optimization by RSM and CCD, equilibrium and kinetic studies. Environ. Sci. Pollut. Res. 2018, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Libutti, A.; Gatta, G.; Gagliardi, A.; Vergine, P.; Pollice, A.; Beneduce, L.; Disciglio, G.; Tarantino, E. Agro-industrial wastewater reuse for irrigation of a vegetable crop succession under Mediterranean conditions. Agric. Water Manag. 2018, 196, 1–14. [Google Scholar] [CrossRef]
- Marrakchi, F.; Kriaa, K.; Hadrich, B.; Kechaou, N. Experimental investigation of processing parameters and effects of degumming, neutralization and bleaching on lampante virgin olive oil’s quality. Food Bioprod. Process. 2015, 94, 124–135. [Google Scholar] [CrossRef]
- Souilem, S.; El-Abbassi, A.; Kiai, H.; Hafidi, A.; Sayadi, S.; Galanakis, C.M. Chapter 1—Olive oil production sector: Environmental effects and sustainability challenges. In Olive Mill Waste; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 1–28. [Google Scholar]
- Negro, M.J.; Manzanares, P.; Ruiz, E.; Castro, E.; Ballesteros, M. Chapter 3—The biorefinery concept for the industrial valorization of residues from olive oil industry. In Olive Mill Waste; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 57–78. [Google Scholar]
- Kapellakis, I.E.; Tzanakakis, V.A.; Angelakis, A.N. Land application-based management of olive mill wastewater. Water 2015, 7, 362–376. [Google Scholar] [CrossRef]
- Caputo, A.C.; Scacchia, F.; Pelagagge, P.M. Disposal of by-products in olive oil industry: Waste-to-energy solutions. Appl. Therm. Eng. 2003, 23, 197–214. [Google Scholar] [CrossRef]
- Miranda, T.; Esteban, A.; Rojas, S.; Montero, I.; Ruiz, A. Combustion analysis of different olive residues. Int. J. Mol. Sci. 2008, 9, 512–525. [Google Scholar] [CrossRef] [PubMed]
- Jeguirim, M.; Chouchène, A.; Réguillon, A.F.; Trouvé, G.; Le Buzit, G. A new valorisation strategy of olive mill wastewater: Impregnation on sawdust and combustion. Resour. Conserv. Recycl. 2012, 59, 4–8. [Google Scholar] [CrossRef]
- Kraiem, N.; Jeguirim, M.; Limousy, L.; Lajili, M.; Dorge, S.; Michelin, L.; Said, R. Impregnation of olive mill wastewater on dry biomasses: Impact on chemical properties and combustion performances. Energy 2014, 78, 479–489. [Google Scholar] [CrossRef]
- Haddad, K.; Jeguirim, M.; Jerbi, B.; Chouchene, A.; Dutournié, P.; Thevenin, N.; Ruidavets, L.; Jellali, S.; Limousy, L. Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source and Biofertilizer. ACS Sustain. Chem. Eng. 2017, 5, 8988–8996. [Google Scholar] [CrossRef]
- Jeguirim, M.; Dutournié, P.; Zorpas, A.A.; Limousy, L. Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source and Bio-Fertilizer—Part 1. The Drying Kinetics. Energies 2017, 10, 1423. [Google Scholar]
- Amos, W.A. Report on Biomass Drying Technology; Technical Report; National Renewable Energy Lab.: Golden, CO, USA, 1999. [Google Scholar] [CrossRef]
- Borsato, A.V.; Doni-Filho, L.; Rakocevic, M.; Cocco, L.C.; Paglia, E.C. Chamomile essential oils extracted from flower heads and recovered water during drying process. J. Food Process. Preserv. 2009, 33, 500–512. [Google Scholar] [CrossRef]
- Moore, W.A.; Kroner, R.C.; Ruchhoft, C.C. Dichromate Reflux Method for Determination of Oxygen Consumed. Anal. Chem. 1949, 21, 953–957. [Google Scholar] [CrossRef]
- Montero, I.; Miranda, T.; Arranz, J.I.; Rojas, C.V. Thin Layer Drying Kinetics of By-Products from Olive Oil Processing. Int. J. Mol. Sci. 2011, 12, 7885–7897. [Google Scholar] [CrossRef]
- Khiari, B.; Mihoubi, D.; Mabrouk, S.B.; Sassi, M. Experimental and numerical investigations on water behaviour in a solar tunnel drier. Desalination 2004, 168, 117–124. [Google Scholar] [CrossRef]
- Selimi, A.; Nickelson, R. Effects of liming on soil pH and manganese toxicity in a Goulburn valley pear orchard. Aust. J. Exp. Agric. 1972, 12, 310–314. [Google Scholar] [CrossRef]
- Haddad, K.; Jellali, S.; Jaouadi, S.; Benltifa, M.; Mlayah, A.; Hamzaoui, A.H. Raw and treated marble wastes reuse as low cost materials for phosphorus removal from aqueous solutions: Efficiencies and mechanisms. C. R. Chim. 2015, 18, 75–87. [Google Scholar] [CrossRef]






| Technology | Description | Advantages | Drawbacks |
|---|---|---|---|
| Land spreading | OMWW is used as amendment (30–80 m3 ha−1 year−1) |
|
|
| Composting | Aerobic breakdown of organic content of OMWW |
|
|
| Thermal treatment | OMWW mixing with dry olive husk wastes |
|
|
| Membrane filtration | Separation of the solid fraction using multilayered filters |
|
|
| Biological treatment | Biological degradation of the organic matter in anaerobic digesters |
|
|
| Coagulation-flocculation | Aggregation of the suspended matter through polyers addition |
|
|
| Electrocoagulation | Imposition a current between two electrodes immersed in an electrolyte to produce a coagulant in solution |
|
|
| Adsorption | Elimination of organic and mineral content by using porous media |
|
|
| Parameter | Raw OMWW | OMWW | IS | IWC | HS | HWC | Discharging Norm | Reuse in Agriculture Norm |
|---|---|---|---|---|---|---|---|---|
| pH | 4.8 | 3.5 | 3.9 | 3.8 | 3.8 | 3.7 | 6.5–8.5 | 6.5–8.5 |
| EC (mS/cm) | 9.73 | 0.25 | 0.23 | 0.27 | 0.05 | 0.02 | 5.00 | 7.00 |
| COD (g/L) | 100.00 | 8.38 | 2.10 | 8.40 | 0.10 | 0.04 | 0.13 | 0.09 |
| Na+ (mg/L) | 2465.0 | 2.0 | 3.3 | 1.2 | 3.3 | 2.6 | 700 | - |
| K+ (mg/L) | 5046.5 | 5.0 | 1.9 | 2.0 | 2.1 | 4.5 | 50 | - |
| Mg2+(mg/L) | 1462.5 | 0.4 | 1.1 | 0.4 | 0.8 | 1.0 | 300 | - |
| Ca2+ (mg/L) | 1907.8 | 1.7 | 4.3 | 1.5 | 3.0 | 3.9 | 500 | - |
| Ptotal (mg/L) | 832.1 | 0.7 | 1.6 | 1.4 | 24.8 | 1.9 | 2 | - |
| NO3− (mg/L) | 1056.8 | 1.5 | 1.6 | 0.4 | 3.0 | 5.5 | 600 | - |
| Cl− (mg/L) | 1912.9 | 4.8 | 4.3 | 2.3 | 8.5 | 2.6 | 700 | 2000.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dutournié, P.; Jeguirim, M.; Khiari, B.; Goddard, M.-L.; Jellali, S. Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source, and Bio-Fertilizer. Part 2: Water Recovery. Water 2019, 11, 768. https://doi.org/10.3390/w11040768
Dutournié P, Jeguirim M, Khiari B, Goddard M-L, Jellali S. Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source, and Bio-Fertilizer. Part 2: Water Recovery. Water. 2019; 11(4):768. https://doi.org/10.3390/w11040768
Chicago/Turabian StyleDutournié, Patrick, Mejdi Jeguirim, Besma Khiari, Mary-Lorène Goddard, and Salah Jellali. 2019. "Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source, and Bio-Fertilizer. Part 2: Water Recovery" Water 11, no. 4: 768. https://doi.org/10.3390/w11040768
APA StyleDutournié, P., Jeguirim, M., Khiari, B., Goddard, M.-L., & Jellali, S. (2019). Olive Mill Wastewater: From a Pollutant to Green Fuels, Agricultural Water Source, and Bio-Fertilizer. Part 2: Water Recovery. Water, 11(4), 768. https://doi.org/10.3390/w11040768








