Removal of Multi-Class Antibiotic Drugs from Wastewater Using Water-Soluble Protein of Moringa stenopetala Seeds
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instrumentation
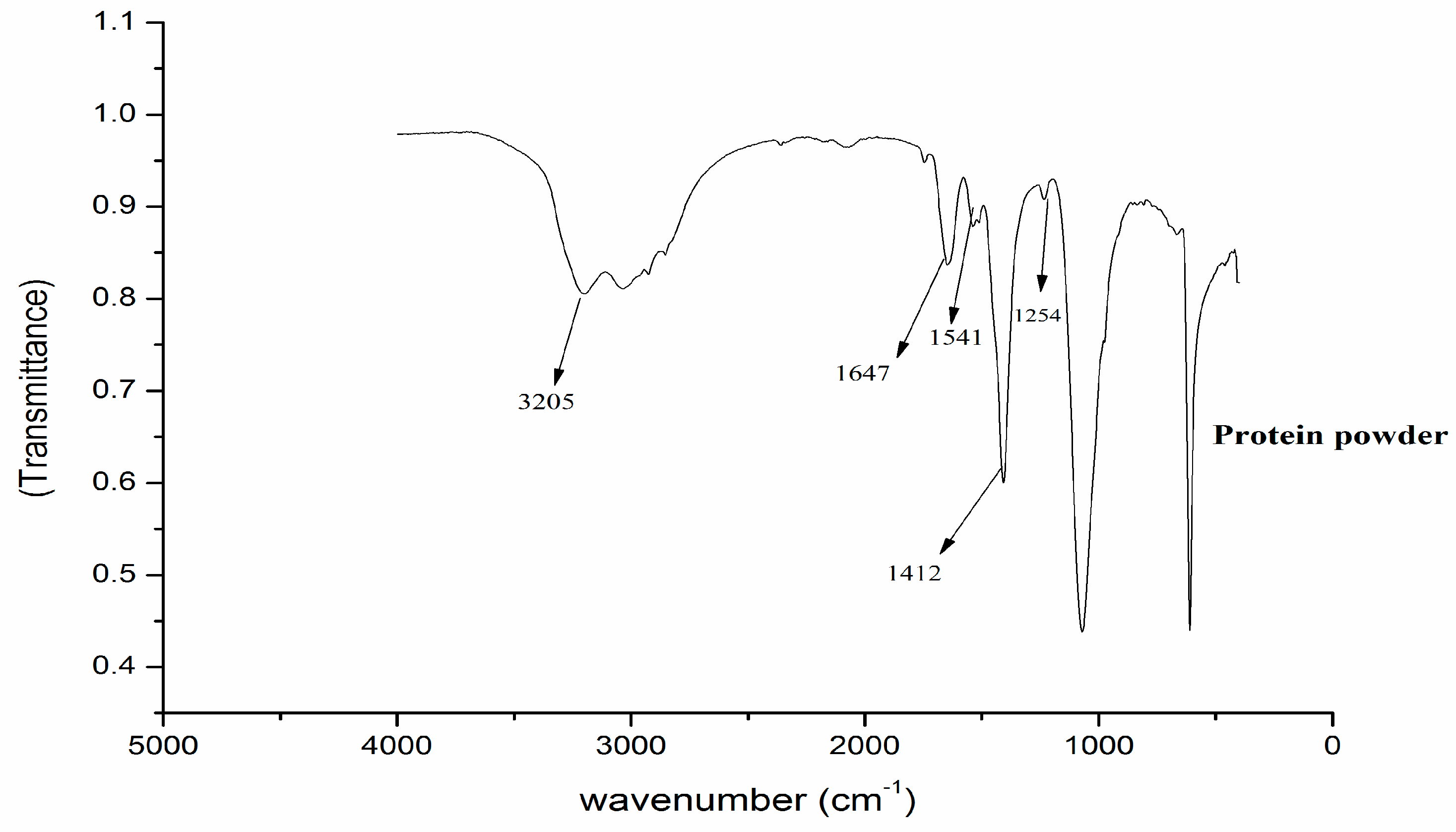
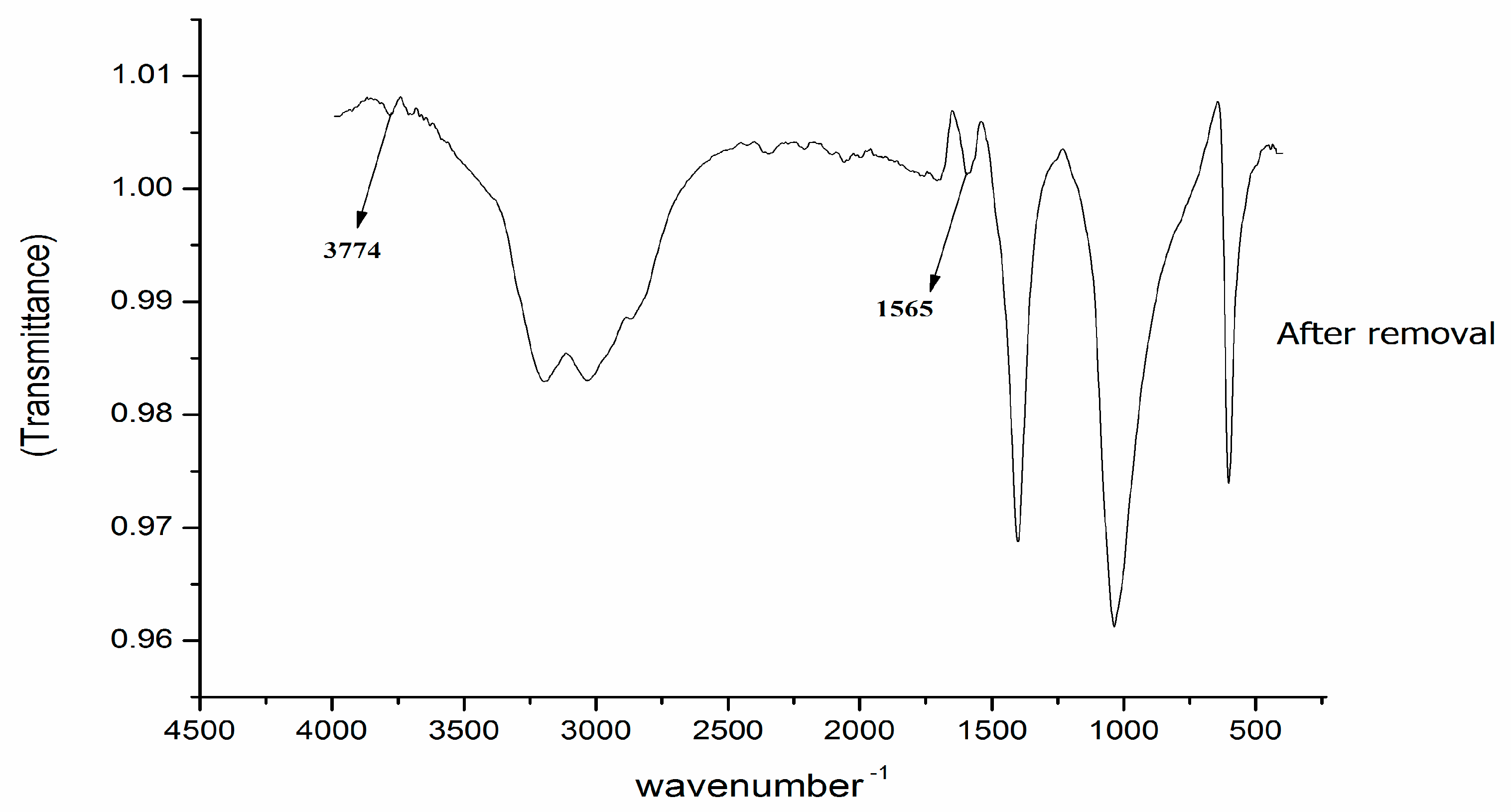
2.3. FTIR Spectroscopy
2.4. Preparation of Standard Solutions
2.5. Extraction of Water-Soluble Proteins
2.6. Preparation of Pure Protein for the Removal of Antibiotics
2.7. Data Analysis
2.8. Sample Collection
3. Results and Discussion
3.1. Characterisation of Moringa Seed Protein Powder
3.2. FTIR Characterisation of Moringa Protein before and after Removal of Antibiotics
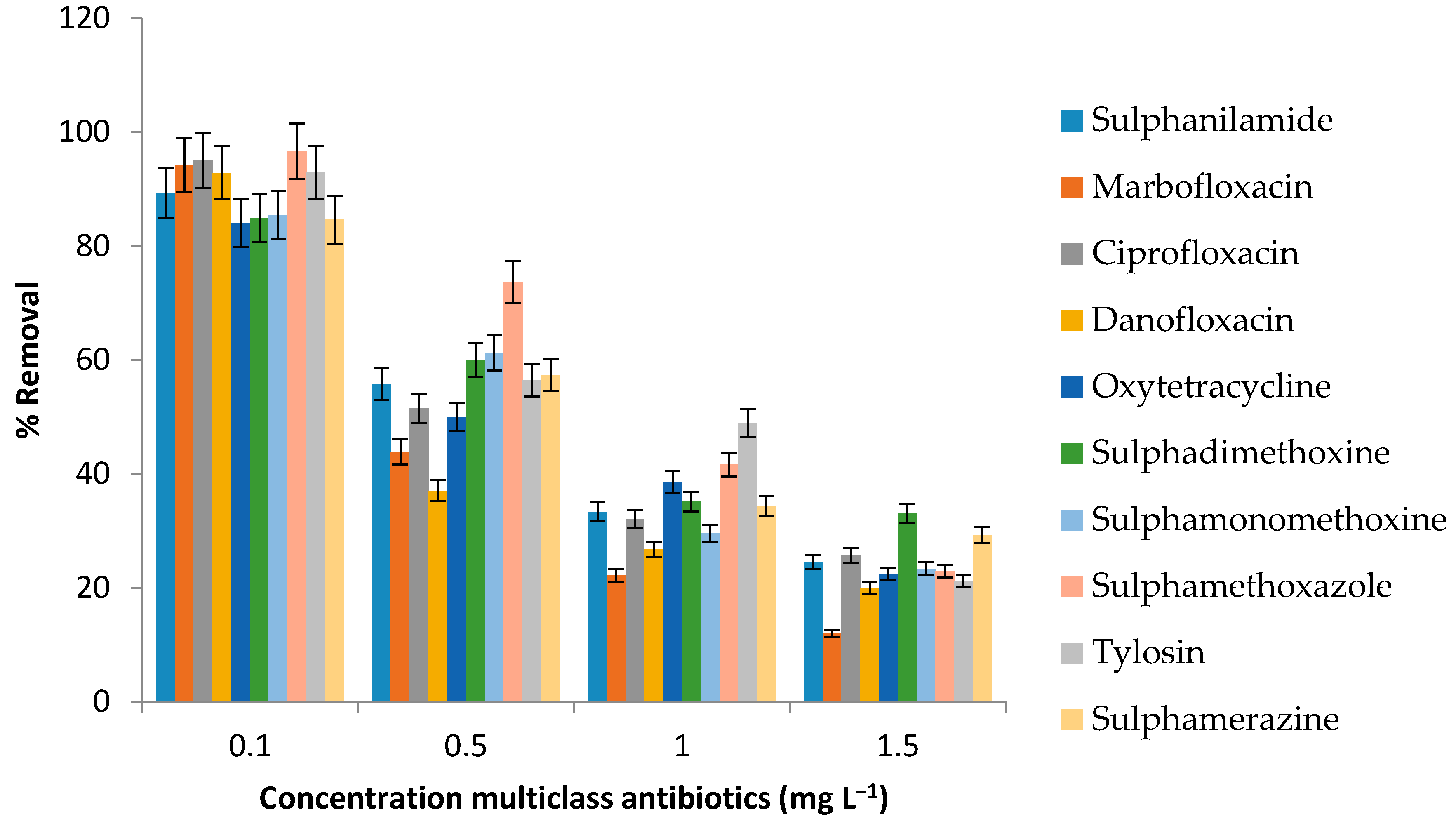
3.3. Removal of Antibiotics Using Moringa Protein
3.4. Effect of Initial Analyte Concentration
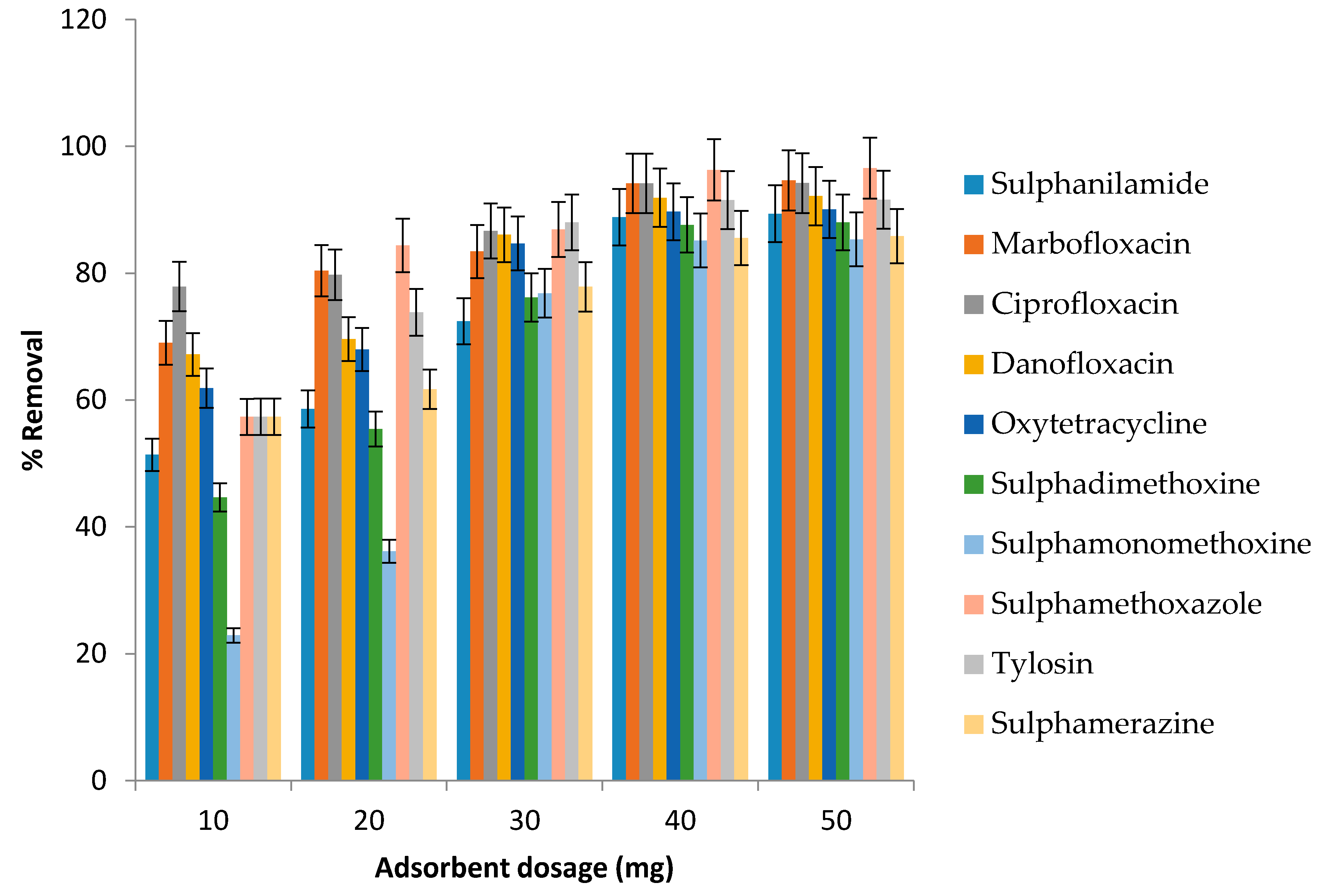
3.5. Effect of Adsorbent Dosage
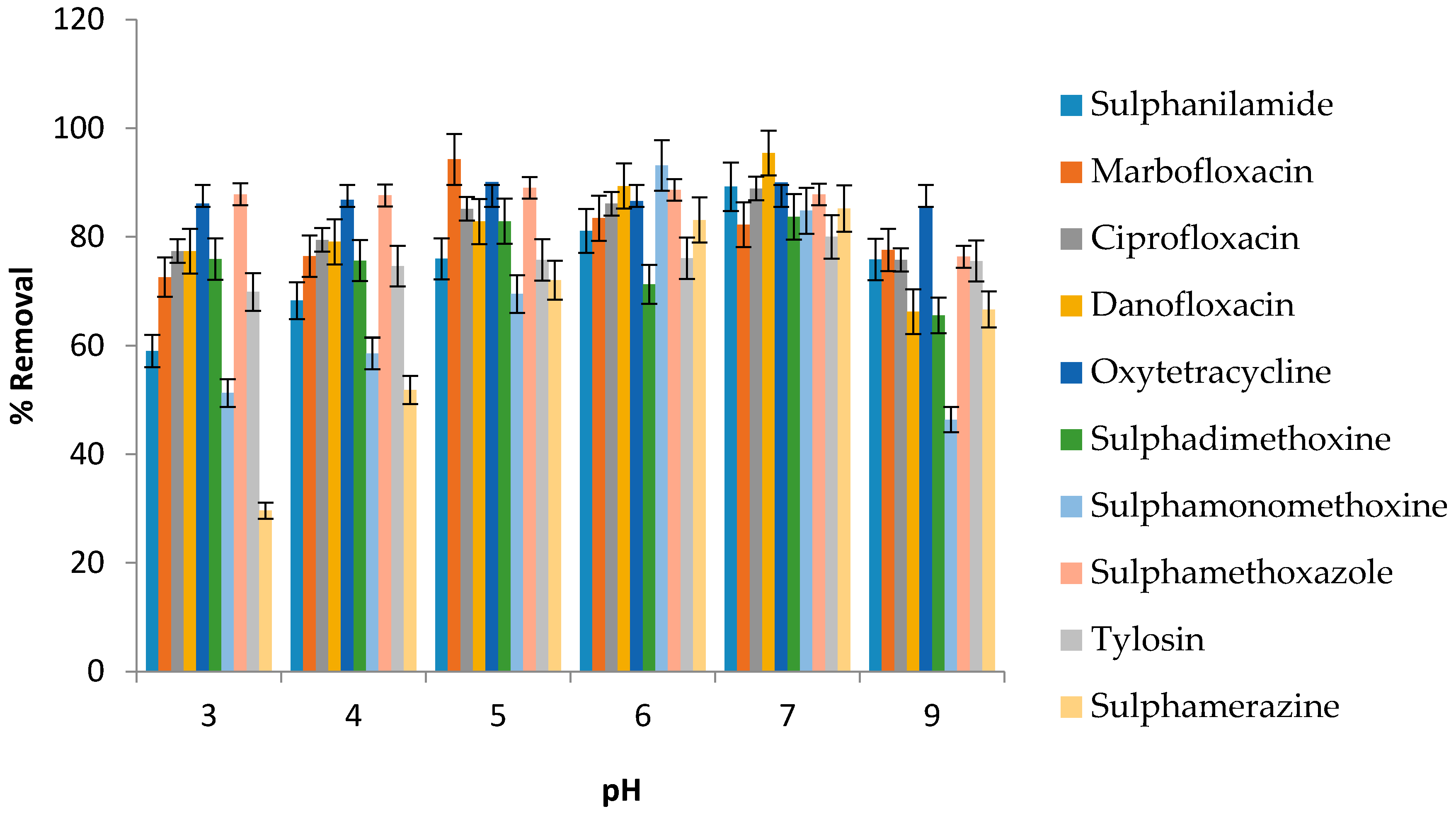
3.6. Effect of pH
3.7. Application of Method on Real Wastewater Sample
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kümmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Thiele-Bruhn, S. Pharmaceutical antibiotic compounds in soils—A review. J. Plant Nutr. Soil Sci. 2003, 166, 145–167. [Google Scholar] [CrossRef]
- Glassmeyer, S.T.; Hinchey, E.K.; Boehme, S.E.; Daughton, C.G.; Ruhoy, I.S.; Conerly, O.; Daniels, R.L.; Lauer, L.; McCarthy, M.; Nettesheim, T.G.; et al. Disposal practices for unwanted residential medications in the United States. Environ. Int. 2009, 35, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Yang, Y.; Zhou, J.; Liu, M.; Nie, M.; Shi, H.; Gu, L. Antibiotics in the surface water of the Yangtze Estuary: Occurrence, distribution and risk assessment. Environ. Pollut. 2013, 175, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Lee, B. Removal of Antibiotics from Contaminated Waters Using Natural Zeolite. Master’s Thesis, The City University of New York, New York, NY, USA, August 2012. [Google Scholar]
- Karthikeyan, K.G.; Meyer, M.T. Occurrence of antibiotics in wastewater treatment facilities in Wisconsin, USA. Sci. Total Environ. 2006, 361, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Watkinson, A.J.; Murby, E.J.; Kolpin, D.W.; Costanzo, S.D. The occurrence of antibiotics in an urban watershed: From wastewater to drinking water. Sci. Total Environ. 2009, 407, 2711–2723. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhang, G.; Zou, S.; Li, X.; Liu, Y. Determination of selected antibiotics in the Victoria Harbour and the Pearl River, South China using high-performance liquid chromatography-electrospray ionization tandem mass spectrometry. Environ. Pollut. 2007, 145, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Meyer, M.T.; Liu, X.; Zhao, Q.; Chen, H.; Chen, J.A.; Qiu, Z.; Yang, L.; Cao, J.; Shu, W. Determination of antibiotics in sewage from hospitals, nursery and slaughter house, wastewater treatment plant and source water in Chongqing region of Three Gorge Reservoir in China. Environ. Pollut. 2010, 158, 1444–1450. [Google Scholar] [CrossRef] [PubMed]
- Berglund, B.; Gengler, S.; Batoko, H.; Wattiau, P.; Errampalli, D.; Leung, K.; Cassidy, M.B.; Kostrzynska, M.; Blears, M.; Lee, H.; et al. Environmental dissemination of antibiotic resistance genes and correlation to anthropogenic contamination with antibiotics. J. Microbiol. Methods 2015, 113, 28564. [Google Scholar] [CrossRef]
- Nghiem, L.D.; Schäfer, A.I.; Elimelech, M. Removal of natural hormones by nanofiltration membranes: Measurements, modeling, and mechanisms. Environ. Sci. Technol. 2004, 38, 1888–1896. [Google Scholar] [CrossRef]
- Milić, N.; Milanović, M.; Letić, N.G.; Sekulić, M.T.; Radonić, J.; Mihajlović, I.; Miloradov, M.V. Occurrence of antibiotics as emerging contaminant substances in aquatic environment. Int. J. Environ. Health Res. 2013, 23, 296–310. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhang, G.; Li, X.; Zou, S.; Li, P.; Hu, Z.; Li, J. Occurrence and elimination of antibiotics at four sewage treatment plants in the Pearl River Delta (PRD), South China. Water Res. 2007, 41, 4526–4534. [Google Scholar] [CrossRef] [PubMed]
- Kemper, N. Veterinary antibiotics in the aquatic and terrestrial environment. Ecol. Indic. 2008, 8, 1–13. [Google Scholar] [CrossRef]
- Terzić, S.; Senta, I.; Ahel, M.; Gros, M.; Petrović, M.; Barcelo, D.; Müller, J.; Knepper, T.; Martí, I.; Ventura, F.; et al. Occurrence and fate of emerging wastewater contaminants in Western Balkan Region. Sci. Total Environ. 2008, 399, 66–77. [Google Scholar] [CrossRef]
- Watkinson, A.J.; Murby, E.J.; Costanzo, S.D. Removal of antibiotics in conventional and advanced wastewater treatment: Implications for environmental discharge and wastewater recycling. Water Res. 2007, 41, 4164–4176. [Google Scholar] [CrossRef]
- Le-Minh, N.; Khan, S.J.; Drewes, J.E.; Stuetz, R.M. Fate of antibiotics during municipal water recycling treatment processes. Water Res. 2010, 44, 4295–4323. [Google Scholar] [CrossRef]
- Elmolla, E.S.; Chaudhuri, M. Photocatalytic degradation of amoxicillin, ampicillin and cloxacillin antibiotics in aqueous solution using UV/TiO2 and UV/H2O2/TiO2 photocatalysis. Desalination 2010, 252, 46–52. [Google Scholar] [CrossRef]
- González, O.; Sans, C.; Esplugas, S. Sulfamethoxazole abatement by photo-Fenton. Toxicity, inhibition and biodegradability assessment of intermediates. J. Hazard. Mater. 2007, 146, 459–464. [Google Scholar] [CrossRef]
- Avisar, D.; Primor, O.; Gozlan, I.; Mamane, H. Sorption of sulfonamides and tetracyclines to montmorillonite clay. Water. Air. Soil Pollut. 2010, 209, 439–450. [Google Scholar] [CrossRef]
- Wang, Z.; Pan, B.; Xing, B. Norfloxacin sorption and its thermodynamics on surface modified CNTs. Environ. Sci. Technol. 2010, 44, 978–984. [Google Scholar] [CrossRef]
- Kim, Y.; Bae, J.; Park, J.; Suh, J.; Lee, S.; Park, H.; Choi, H. Removal of 12 selected pharmaceuticals by granular mesoporous silica SBA-15 in aqueous phase. Chem. Eng. J. 2014, 256, 475–485. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, X.; Chen, X.; Dai, C.; Zhang, J.; Zhang, Y. Biosorption of clofibric acid and carbamazepine in aqueous solution by agricultural waste rice straw. J. Environ. Sci. 2013, 25, 2384–2395. [Google Scholar] [CrossRef]
- Kebede, T.G.; Mengistie, A.A.; Dube, S.; Nkambule, T.T.I.; Nindi, M.M. Study on adsorption of some common metal ions present in industrial effluents by Moringa stenopetala seed powder. J. Environ. Chem. Eng. 2018, 6, 1378–1389. [Google Scholar] [CrossRef]
- Kebede, T.G.; Dube, S.; Nindi, M.M. Fabrication and characterization of electrospun nanofibers from moringa stenopetala seed protein. Mater. Res. Express 2018, 5, 125015. [Google Scholar] [CrossRef]
- Schwarz, J.; Thiele-Bruhn, S.; Eckhardt, K.-U.; Schulten, H.-R. Sorption of Sulfonamide Antibiotics to Soil Organic Sorbents: Batch Experiments with Model Compounds and Computational Chemistry. ISRN Soil Sci. 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Bidgood, T.L.; Papich, M.G. Plasma and interstitial fluid pharmacokinetics of enrofloxacin, its metabolite ciprofloxacin, and marbofloxacin after oral administration and a constant rate intravenous infusion in dogs. J. Vet. Pharmacol. Ther. 2005, 28, 329–341. [Google Scholar] [CrossRef]
- Şanli, N.; Şanli, S.; Özkan, G.; Denizli, A. Determination of pKa values of some sulfonamides by LC and LC-PDA methods in acetonitrile-water binary mixtures. J. Braz. Chem. Soc. 2010, 21, 1952–1960. [Google Scholar] [CrossRef]
- Leo, A.; Hansch, C.; Elkins, D. Partition coefficients and their Uses. Chem. Rev. 1971, 71, 525. [Google Scholar] [CrossRef]
- Heibrt, B.J.; Dorsey, J. mOctanol-Water Partition Coefficient Estimation by Micellar Electrokinetic Capillary Chromatography. Anal. Chem. 1995, 67, 744–749. [Google Scholar]
- Qiang, Z.; Adams, C. Potentiometric determination of acid dissociation constants (pK a) for human and veterinary antibiotics. Water Res. 2004, 38, 2874–2890. [Google Scholar] [CrossRef]
- Yang, S.F.; Lin, C.F.; Lin, A.Y.; Hong, P.K.A. Sorption and biodegradation of sulfonamide antibiotics by activated sludge: Experimental assessment using batch data obtained under aerobic conditions. Water Res. 2011, 45, 3389–3397. [Google Scholar] [CrossRef] [PubMed]
- Kebede, T.G.; Dube, S.; Nindi, M.M. Removal of non-steroidal anti-in fl ammatory drugs (NSAIDs ) and carbamazepine from wastewater using water-soluble protein extracted from Moringa stenopetala seeds. J. Environ. Chem. Eng. 2018, 6, 3095–3103. [Google Scholar] [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S.; Talbot, B.G. Active agents and mechanism of coagulation of turbid waters using Moringa oleifera. Water Res. 1995, 29, 703–710. [Google Scholar] [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S. Quality of water treated by coagulation using Moringa oleifera seeds. Water Res. 1998, 32, 781–791. [Google Scholar] [CrossRef]
- Choi, K.J.; Son, H.J.; Kim, S.H. Ionic treatment for removal of sulfonamide and tetracycline classes of antibiotic. Sci. Total Environ. 2007, 387, 247–256. [Google Scholar] [CrossRef] [PubMed]
| Name of Pharmaceuticals | Structure | pKa1 and pKa2 (pKa3) | Log Ko | Reference |
|---|---|---|---|---|
| Sulphanilamide | 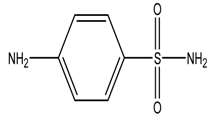 | 1.94/10.67 | −0.62 | [26] |
| Marbofloxacin | 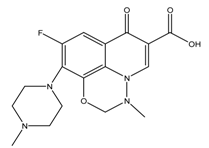 | 5.77/8.22 | 0.07 ± 0.01 | [27] |
| Ciprofloxacin | 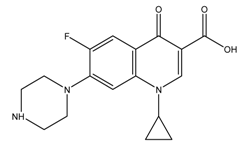 | 1.84 (0.08)/7.63 (0.01) | 0.35 | [28,29] |
| Danofloxacin | 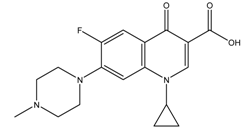 | 2.06/6.90 | 0.44 | [4] |
| Oxytetracycline | 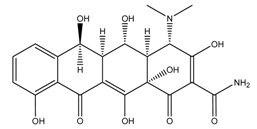 | 3.3/7.55/9.7 | −1.12 | [30,31] |
| Sulphamerazine | 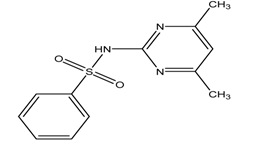 | 2.06/6.90 | 0.44 | [31] |
| Sulphamonomethoxine | 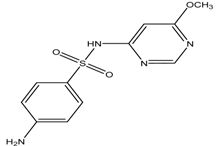 | 2.07(0.05)/6.91 (0.01) | 0.70 | [28,32] |
| Sulphamethoxazole | 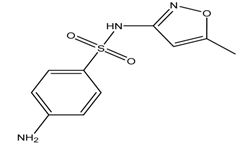 | 1.6/5.7 (7.40 (0.04)) | 0.89 | [4,28] |
| Tylosin tartrate | 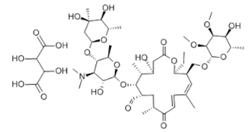 | 3.31/7.5 | - | [31] |
| Sulphadimethoxine | 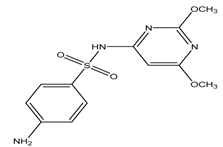 | 2.1/6.3 | 1.63 | [32] |
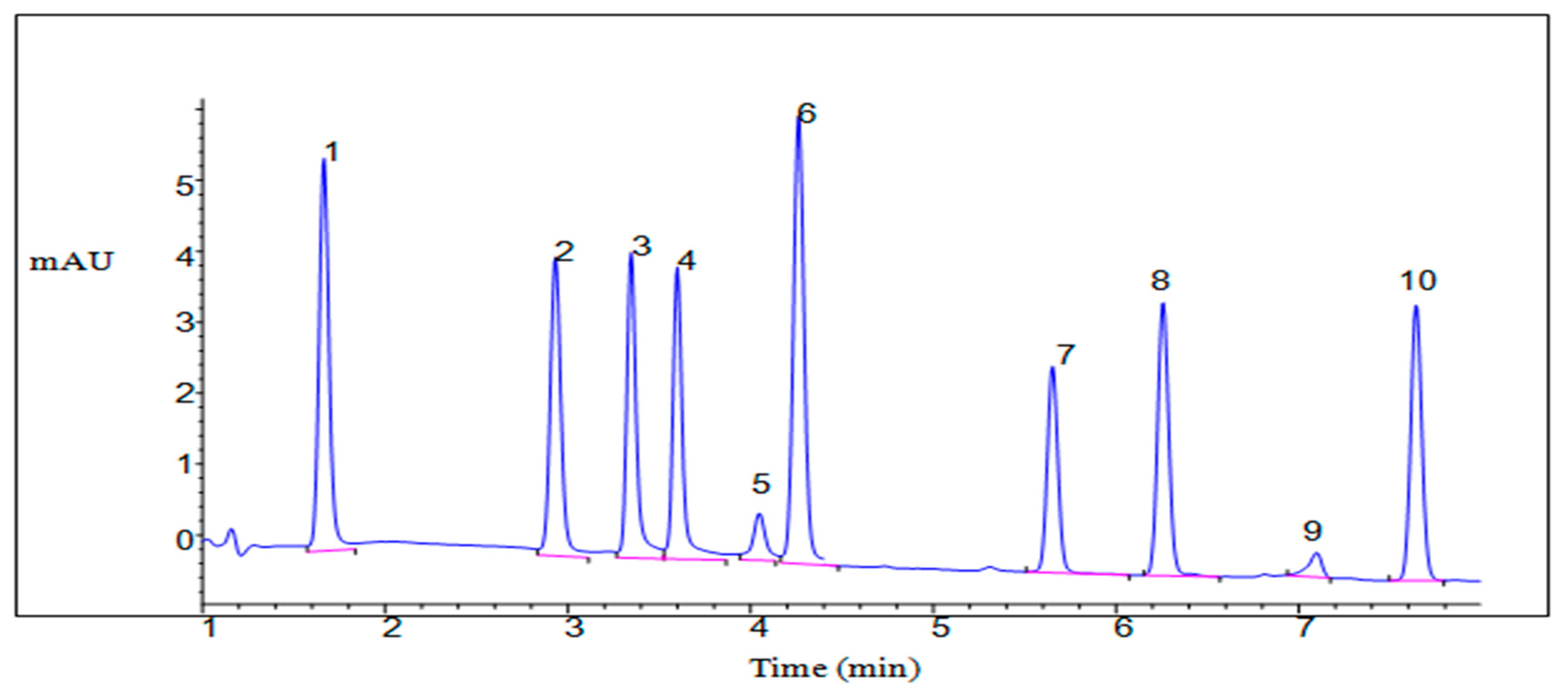
| Analytes | Retention Time | Concentration (mg L−1) before Removal | Percentage Removal in the Ultrahigh Purity Water (%) | Percentage Removal in Effluent (%) | Percentage Removal of Influent (%) |
|---|---|---|---|---|---|
| Sulphanilamide | 1.62 | 0.1 | 88.8 ± 0.05 | 72.4 ± 0.32 | 71.3 ± 0.56 |
| Marbofloxacin | 2.99 | 0.1 | 94.2 ± 0.05 | 88.2 ± 0.45 | 85.2 ± 0.66 |
| Ciprofloxacin | 3.35 | 0.1 | 94.2 ± 0.02 | 88.3 ± 0.56 | 83.5 ± 0.45 |
| Danofloxacin | 3.60 | 0.1 | 95.4 ± 0.12 | 89.5 ± 0.44 | 87.2 ± 0.85 |
| Oxytetracycline | 4.09 | 0.1 | 89.7 ± 0.04 | 92.5 ± 0.84 | 91.5 ± 0.71 |
| Sulphamerazine | 4.32 | 0.1 | 87.6 ± 0.05 | 74.4 ± 0.52 | 76.2 ± 0.32 |
| Sulphamonomethoxine | 5.69 | 0.1 | 85.2 ± 0.01 | 74.2 ± 0.32 | 70.1 ± 0.51 |
| Sulfamethoxazole | 6.28 | 0.1 | 96.3 ± 0.03 | 89.0 ± 0.56 | 83.7 ± 0.61 |
| Tylosin tartrate | 6.99 | 0.1 | 91.5 ± 0.01 | 86.8 ± 0.84 | 81.9 ± 0.55 |
| Sulphadimethoxine | 7.64 | 0.1 | 85.5 ± 0.01 | 74.7 ± 0.56 | 70.4 ± 0.82 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kebede, T.G.; Dube, S.; Nindi, M.M. Removal of Multi-Class Antibiotic Drugs from Wastewater Using Water-Soluble Protein of Moringa stenopetala Seeds. Water 2019, 11, 595. https://doi.org/10.3390/w11030595
Kebede TG, Dube S, Nindi MM. Removal of Multi-Class Antibiotic Drugs from Wastewater Using Water-Soluble Protein of Moringa stenopetala Seeds. Water. 2019; 11(3):595. https://doi.org/10.3390/w11030595
Chicago/Turabian StyleKebede, Temesgen Girma, Simiso Dube, and Mathew Muzi Nindi. 2019. "Removal of Multi-Class Antibiotic Drugs from Wastewater Using Water-Soluble Protein of Moringa stenopetala Seeds" Water 11, no. 3: 595. https://doi.org/10.3390/w11030595
APA StyleKebede, T. G., Dube, S., & Nindi, M. M. (2019). Removal of Multi-Class Antibiotic Drugs from Wastewater Using Water-Soluble Protein of Moringa stenopetala Seeds. Water, 11(3), 595. https://doi.org/10.3390/w11030595





