Strategy for Flux Enhancement in Biofilm Ceramic Membrane Bioreactor Applying Prepolymerized and Non-Prepolymerized Inorganic Coagulants
Abstract
:1. Introduction
2. Materials and Methods
- (1)
- the selection of the optimum pH at constant coagulant dosage for every tested coagulant during the jar tests;
- (2)
- the jar tests, conducted at previously determined optimum pH for every coagulant, with the variation of the coagulant dose, to determine the relevant optimum dosage ranges;
- (3)
- the total recycle test (TRT), with the pH correction and the application of the tested dosage ranges (depending on the coagulant), which exhibited promising results during the jar tests, to determine the optimum fouling mitigation conditions and the most efficient compound;
- (4)
- the TRT without the pH correction, to assess the coagulant performance regarding fouling mitigation under the conditions, unfavourable for coagulation;
- (5)
- partial least squares analysis using the data from the TRT, with and without pH correction, to study the effect of the selected mixed liquor characteristics on the fouling intensity after the chemical dosing and determine the principal mechanisms of coagulant action;
- (6)
- the determination of the intrinsic coagulant charges and the qualitative analysis of the investigated prepolymerized aluminium coagulants regarding their molecular weight (MW) distribution;
- (7)
- two-level factorial design of the experiment, applying the selected coagulant of the highest performance, to determine the optimum levels of factors for the maximum permeability/filtration time increase, discover the vital factors for the membrane flux enhancement and the tendencies in their interactions.
2.1. Study Object
2.2. Jar Tests
2.3. Total Recycle Test
2.4. Applied Analytical Techniques
2.4.1. Mixed Liquor Analysis
2.4.2. Intrinsic Characteristics of the Coagulants
Potentiometric Titration
Size Exclusion Chromatography
2.5. Statistical Mining of the Relationships in the System
3. Results
3.1. Jar Tests
- (1)
- The ligands, which are representative of ionogenic functional groups and characteristic of the hydrophilic colloids (proteins, polysaccharides and humic substances), such as phosphate, pyrophosphate, oxalate, salicylate (with a carboxyl and an aromatic hydroxyl group), show the tendency to displace the H2O groups in aquo-metal ions of the coagulant, which satisfies the coordinative requirements of Fe(III) and Al(III), which results in the formation of the Me-ligand complexes;
- (2)
- OH− ions have a stronger affinity for the Fe3+ and Al3+ than other ligands, including the representative of functional groups of colloids; however, the latter may compete with OH− for the coordinative sites;
- (3)
- When the ratio ligand/OH− increases, which can be reached by pH decrease in the system, ligands, which originate from the functional groups, may partially or entirely substitute for OH− in the charge neutralization of the metal cations;
- (4)
- Fe3+ has a higher affinity for OH− than Al3+; thus, lower pH is required for the formation of ionized function group-Me complexes to limit the statistical opportunity of the complexation of OH−.
3.2. Total Recycle Test
3.3. TRT with pH Control vs. TRT with Non-Corrected pH
Al(H2O)63+ + H2O ↔ [Al(H2O)5OH]2+ + H3O.
3.4. Investigation of the Characteristics of the Coagulants
3.4.1. Coagulant Charges
3.4.2. Chromatographic Separation of Prepolymerized Aluminium Coagulants
3.5. Statistical Analysis
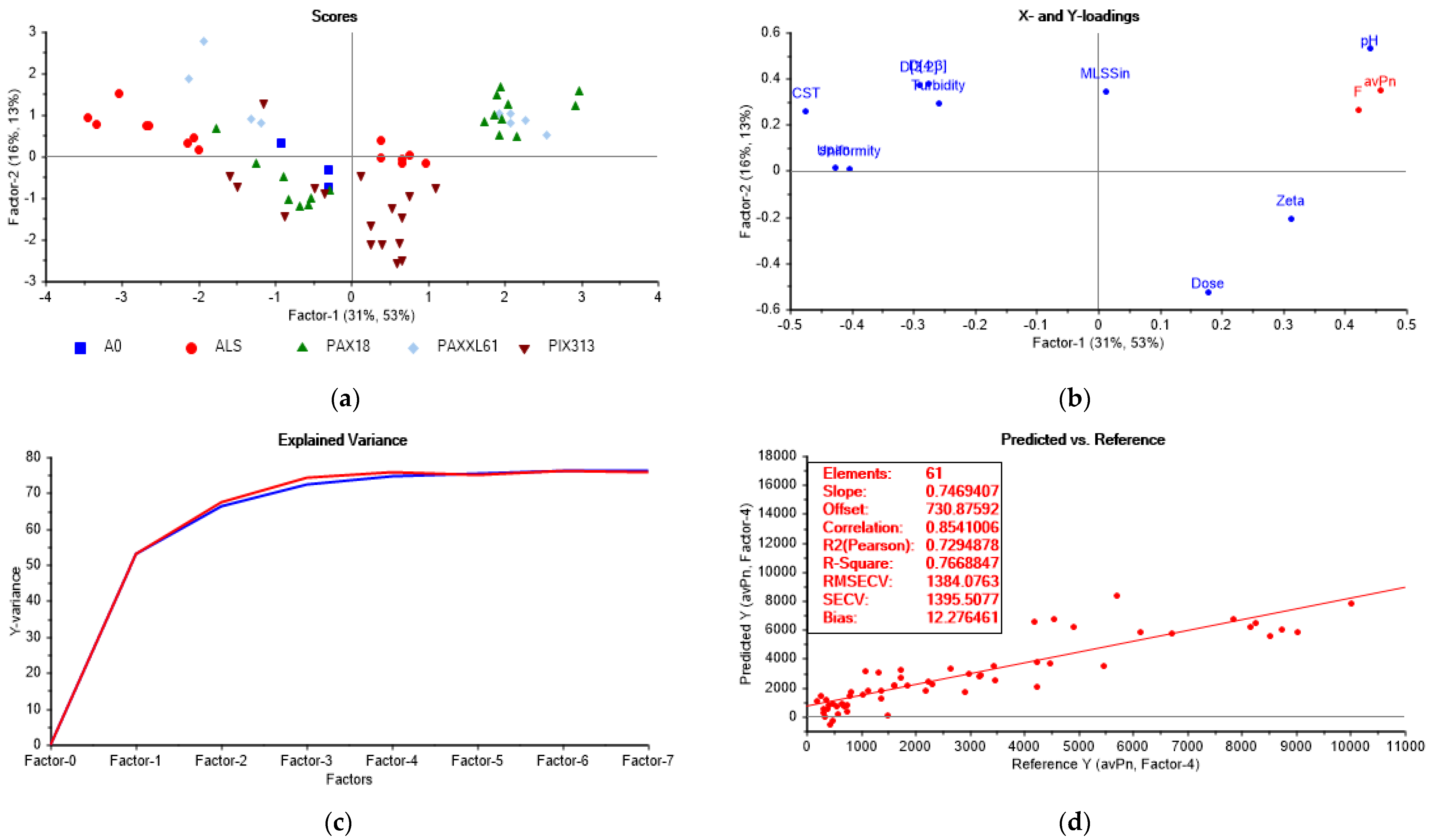
3.5.1. Partial Least Squares Regression Analysis
- enhanced adsorption/charge neutralization (zeta potential −2.9–3.2 mV): PAX18 (100% of all PAX18 samples) > PAXXL61 (55.6%) > PIX313 (41.0%) > ALS (36.0%);
- the increase of relative hydrophobicity of the flocs (CST 17–95 s): PAX18 (100%) > PIX313 (82.0 %) > PAXXL61 (56.0%) > ALS (43.0%);
- the increase in particle size (D[4,3] 32.5–132 µm/D[3,2] 21–83 µm): ALS (100/100%) > PAX18 (78.0/94.0%) > PAXXL61 (78.0/78.0%) > PIX313 (41.0%/53.0%).
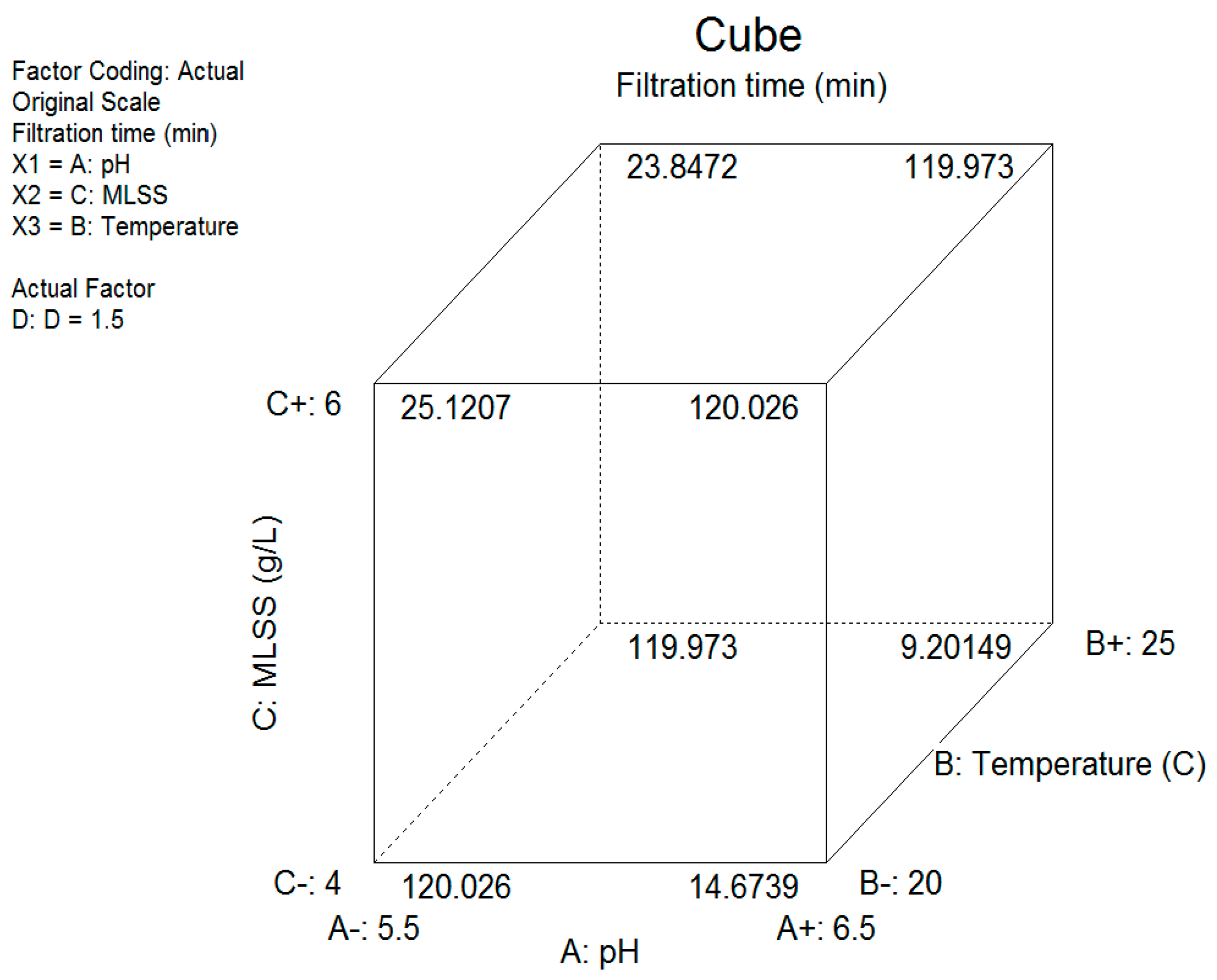
3.5.2. Two-Level Factorial Design of the Experiment
The Average Permeability
105·D − 1.923 × 105·AC − 1.021 × 105·AD,
1.38853 × 106·MLSS + 3.73531 × 106·D − 3.84539 × 105·pH·MLSS − 5.10408 × 105·pH·D,
Filtration Time
+ 8.598 × 105·AC,
2861.8·D + 1.72 × 106·pH·MLSS
- (1)
- A+1 (pH 6.5), B-1 – +1 (T = 20–25 °C), C+1 (MLSS 6 g/L) (values calculated at D = 1.5 µmole Al/mgSS);
- (2)
- A−1 (pH 5.5), B-1 – +1 (T = 20–25 °C), C−1 (MLSS 4 g/L) (values calculated at D = 1.5 µmole Al/mgSS).
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Commission. Regulation of the European Parliament and the Council on Minimum Requirements for Water Reuse; European Commission: Brussels, Belgium, 2018; Volume 169, p. 28. [Google Scholar]
- European Commission. Report on the Review of the European Water Scarcity and Droughts Policy; European Commission: Brussels, Belgium, 2012. [Google Scholar]
- Hankins, N.P.; Singh, R. Emerging Membrane Technology for Sustainable Water Treatment; Marinakis, K., Ed.; Elsevier: Amsterdam, The Netherlands; Oxford, UK; Cambridge, MA, USA, 2016; ISBN 978-0-444-63312-5. [Google Scholar]
- Maletskyi, Z.; Kulesha, O.; Ratnaweera, H.C. Comparison of Al- and Fe-based Membrane Fouling Reducers for BioFilm Membrane BioReactor. In Proceedings of the Euromembrane 2018, Valencia, Spain, 9–13 July 2018. [Google Scholar]
- United States Environmental Protection Agency Water Reuse and Recycling: Community and Environmental Benefits. Available online: https://www3.epa.gov/region9/water/recycling/ (accessed on 22 January 2019).
- Bernal, D.P.; Restrepo, I. Key issues for decentralization in municipal wastewater treatment. In 12th Edition of the World Wide Workshop for Young Environmental Scientists (WWW-YES-2012)—Urban Waters: Resource or Risks? HAL-ENPC: Arcueil, France, 2012. [Google Scholar]
- Kulesha, O.; Maletskyi, Z.; Ratnaweera, H. State-of-the-art of membrane flux enhancement in membrane bioreactor. Cogent Eng. 2018, 5, 1–30. [Google Scholar] [CrossRef]
- Meng, F.; Zhang, S.; Oh, Y.; Zhou, Z.; Shin, H.-S.; Chae, S.-R. Fouling in membrane bioreactors: An updated review. Water Res. 2017, 114, 151–180. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.K.; Nierychlo, M.; Nielsen, A.H.; Larsen, P.; Christensen, M.L.; Nielsen, P.H. Unified understanding of physico-chemical properties of activated sludge and fouling propensity. Water Res. 2017, 120, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Hou, J.; Zhang, Y.; Tian, M.; He, T.; Liu, J.; Chen, V. Polymeric antimicrobial membranes enabled by nanomaterials for water treatment. J. Membr. Sci. 2018, 550, 173–197. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhang, W.; Tang, B.; Ding, J.; Zheng, Y.; Zhang, Z. Membrane fouling mechanism of biofilm-membrane bioreactor (BF-MBR): Pore blocking model and membrane cleaning. Bioresour. Technol. 2018, 250, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Ivanovic, I.; Leiknes, T.O. The biofilm membrane bioreactor (BF-MBR)—A review. Desalin. Water Treat. 2012, 37, 288–295. [Google Scholar] [CrossRef]
- Ødegaard, H. Innovations in wastewater treatment: The moving bed biofilm process. Water Sci. Technol. 2006, 53, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Phattaranawik, J.; Leiknes, T. Study of Hybrid Vertical Anaerobic Sludge-Aerobic Biofilm Membrane Bioreactor for Wastewater Treatment. Water Environ. Res. 2010, 82, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Leiknes, T.; Fredriksen, R.H.; Riviere, E. Comparison of membrane filtration performance between biofilm-MBR and activated sludge-MBR. Desalin. Water Treat. 2012, 48, 285–293. [Google Scholar] [CrossRef]
- Leiknes, T.; Ødegaard, H. The development of a biofilm membrane bioreactor. Desalination 2007, 202, 135–143. [Google Scholar] [CrossRef]
- Dalmau, M.; Atanasova, N.; Gabarrón, S.; Rodriguez-Roda, I.; Comas, J. Comparison of a deterministic and a data driven model to describe MBR fouling. Chem. Eng. J. 2015, 260, 300–308. [Google Scholar] [CrossRef]
- Deng, L.; Guo, W.; Ngo, H.H.; Zhang, H.; Wang, J.; Li, J.; Xia, S.; Wu, Y. Biofouling and control approaches in membrane bioreactors. Bioresour. Technol. 2016, 221, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Germain, E.; Stephenson, T. Biomass characteristics, aeration and oxygen transfer in membrane bioreactors: Their interrelations explained by a review of aerobic biological processes. Rev. Environ. Sci. Biotechnol. 2005, 4, 223–233. [Google Scholar] [CrossRef]
- Sun, F.Y.; Li, P.; Li, J.; Li, H.J.; Ou, Q.M.; Sun, T.T.; Dong, Z.J. Hybrid biofilm-membrane bioreactor (Bf-MBR) for minimization of bulk liquid-phase organic substances and its positive effect on membrane permeability. Bioresour. Technol. 2015, 198, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Nouri, N.; Mehrnia, M.R.; Sarrafzadeh, M.H.; Nabizadeh, R. Performance of membrane bioreactor in presence of flocculants. Desalin. Water Treat. 2013, 52, 2933–2938. [Google Scholar] [CrossRef]
- Ivanovic, I.; Leiknes, T.O. Improved Performance through Particle Surface Modifications by Coagulation with Inorganic Coagulants in a Biofilm Membrane Bioreactor (BF-MBR). Sep. Sci. Technol. 2012, 48, 288–294. [Google Scholar] [CrossRef]
- Deng, L.; Guo, W.; Hao, H.; Farzana, M.; Zuthi, R.; Zhang, J.; Liang, S.; Li, J.; Wang, J.; Zhang, X. Membrane fouling reduction and improvement of sludge characteristics by bioflocculant addition in submerged membrane bioreactor. Sep. Purif. Technol. 2015, 156, 450–458. [Google Scholar] [CrossRef]
- Iversen, V.; Mehrez, R.; Horng, R.Y.; Chen, C.H.; Meng, F.; Drews, A.; Lesjean, B.; Ernst, M.; Jekel, M.; Kraume, M. Fouling mitigation through flocculants and adsorbents addition in membrane bioreactors: Comparing lab and pilot studies. J. Membr. Sci. 2009. [Google Scholar] [CrossRef]
- Wu, J.; Huang, X. Effect of dosing polymeric ferric sulfate on fouling characteristics, mixed liquor properties and performance in a long-term running membrane bioreactor. Sep. Purif. Technol. 2008, 63, 45–52. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, Z.; Mai, S.; Yang, C.; Wang, X.; An, Y.; Zhou, Z. Research and applications of membrane bioreactors in China: Progress and prospect. Sep. Purif. Technol. 2008, 62, 249–263. [Google Scholar] [CrossRef]
- Ji, J.; Li, J.; Qiu, J.; Li, X. Polyacrylamide-starch composite flocculant as a membrane fouling reducer: Key factors of fouling reduction. Sep. Purif. Technol. 2014, 131, 1–7. [Google Scholar] [CrossRef]
- Xiao, Y.; Waheed, H.; Xiao, K.; Hashmi, I.; Zhou, Y. In tandem effects of activated carbon and quorum quenching on fouling control and simultaneous removal of pharmaceutical compounds in membrane bioreactors. Chem. Eng. J. 2018, 341, 610–617. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, B.; Zhao, X.; Gao, Z. Effect of ferric chloride on fouling in membrane bioreactor. Sep. Purif. Technol. 2008, 63, 341–347. [Google Scholar] [CrossRef]
- Guo, W.; Ngo, H.H.; Vigneswaran, S.; Dharmawan, F.; Nguyen, T.T.; Aryal, R. Effect of different flocculants on short-term performance of submerged membrane bioreactor. Sep. Purif. Technol. 2010, 70, 274–279. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Zhang, W.J.; Yang, X.Y.; Xiao, P.; Wang, D.S.; Song, Y. Advanced treatment of effluent from municipal WWTP with different metal salt coagulants: Contaminants treatability and floc properties. Sep. Purif. Technol. 2013, 120, 123–128. [Google Scholar] [CrossRef]
- Holbrook, R.D.; Higgins, M.J.; Murthy, S.N.; Fonseca, A.D.; Fleischer, E.J.; Daigger, G.T.; Grizzard, T.J.; Love, N.G.; Novak, J.T. Effect of alum addition on the performance of submerged membranes for wastewater treatment. Water Environ. Res. 2003, 76, 2699–2702. [Google Scholar] [CrossRef]
- Lee, J.C.; Kim, J.S.; Kang, I.J.; Cho, M.H.; Park, P.K.; Lee, C.H. Potential and limitations of alum or zeolite addition to improve the performance of a submerged membrane bioreactor. Water Sci. Technol. 2001, 43, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Song, K.G.; Kim, Y.; Ahn, K.H. Effect of coagulant addition on membrane fouling and nutrient removal in a submerged membrane bioreactor. Desalination 2008, 221, 467–474. [Google Scholar] [CrossRef]
- Ivanovic, I.; Leiknes, T. Effect of addition of different additives on overall performance of biofilm-MBR (BF-MBR). Desalin. Water Treat. 2011, 34, 129–135. [Google Scholar] [CrossRef]
- Ødegaard, H.; Fettig, J.; Ratnaweera, H.C. Coagulation with Prepolymerized Metal Salts; Hahn, H.H., Klute, R., Eds.; Chemical Water and Wastewater Treatment; Springer: Berlin/Heidelberg, Germany, 1990; ISBN 978-3-642-76093-8. [Google Scholar]
- Gregory, J.; Duan, J. Hydrolyzing metal salts as coagulants. Pure Appl. Chem. 2001, 73, 2017–2026. [Google Scholar] [CrossRef]
- Wu, J.; Chen, F.; Huang, X.; Geng, W.; Wen, X. Using inorganic coagulants to control membrane fouling in a submerged membrane bioreactor. Desalination 2006, 197, 124–136. [Google Scholar] [CrossRef]
- Chen, W.; Liu, J. The possibility and applicability of coagulation-MBR hybrid system in reclamation of dairy wastewater. Desalination 2012, 285, 226–231. [Google Scholar] [CrossRef]
- Gkotsis, P.K.; Batsari, E.L.; Peleka, E.N.; Tolkou, A.K.; Zouboulis, A.I. Fouling control in a lab-scale MBR system: Comparison of several commercially applied coagulants. J. Environ. Manag. 2017, 203, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Bratby, J. Coagulation and Flocculation in Water and Wastewater Treatment, 2nd ed.; IWA Publishing: London, UK; Seattle, WA, USA, 2006; ISBN 9781843391067. [Google Scholar]
- Kulesha, O.; Maletskyi, Z.; Ratnaweera, H. Multivariate Chemometric Analysis of Membrane Fouling Patterns in Biofilm Ceramic Membrane Bioreactor. Water 2018, 10, 982. [Google Scholar] [CrossRef]
- Lee, W.-N.; Chang, I.-S.; Hwang, B.-K.; Park, P.-K.; Lee, C.-H.; Huang, X. Changes in biofilm architecture with addition of membrane fouling reducer in a membrane bioreactor. Process Biochem. 2007, 42, 655–661. [Google Scholar] [CrossRef]
- Yeon, K.-M.; Cheong, W.-S.; Oh, H.-S.; Lee, W.-N.; Hwang, B.-K.; Lee, C.-H.; Beyenal, H.; Lewandowski, Z. Quorum Sensing: A New Biofouling Control Paradigm in a Membrane Bioreactor for Advanced Wastewater Treatment. Environ. Sci. Technol. 2009, 43, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.-S.; Yeon, K.-M.; Yang, C.-S.; Kim, S.-R.; Lee, C.-H.; Park, S.Y.; Han, J.Y.; Lee, J.-K. Control of membrane biofouling in MBR for wastewater treatment by quorum quenching bacteria encapsulated in microporous membrane. Environ. Sci. Technol. 2012, 46, 4877–4884. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. International Standard: ISO 15705:2002. Water Quality—Determination of the Chemical Oxygen Demand Index (ST-COD)—Small-Scale Sealed-Tube Method; International Organization for Standardization: Geneva, Switzerland, 2002; p. 18. [Google Scholar]
- ImageJ: Image Processing and Analysis in Java. Available online: https://imagej.nih.gov/ij/ (accessed on 1 February 2019).
- Rawle, A. Basic Principles of Particle Size Analysis. Available online: http://cat.inist.fr/?aModele=afficheN&cpsidt=14620810 (accessed on 20 November 2018).
- Innopharma Technology. A Guide to D-Values in Pharmaceutical Particle Characterisation. Available online: http://www.innopharmalabs.com/tech/applications-and-processes/particle-size-distribution (accessed on 20 November 2018).
- Horiba Ltd. A Guidebook to Particle Size Analysis. Available online: https://www.horiba.com/fileadmin/uploads/Scientific/Documents/PSA/PSA_Guidebook.pdf (accessed on 20 November 2018).
- Malvern Instruments Limited. A Basic Guide to Particle Characterization. Available online: https://www.cif.iastate.edu/sites/default/files/uploads/Other_Inst/Particle Size/Particle Characterization Guide.pdf (accessed on 20 November 2018).
- Malvern Instruments Ltd. Manual 0101. Available online: Pmbrc.org/index.php/download_file/view/190/ (accessed on 20 November 2018).
- Micrometrix Corporation. Particle Charge Analyser. Operating Manual; Micrometrix Corporation: Suwanee, GA, USA, 2012; p. 19. [Google Scholar]
- American Public Health Association; Water Environment Federation; American Water Works Association. Standard Methods for the Examination of Water and Wastewater, 22nd ed. Rice, E.W., Baird, R.B., Eaton, A.D., Clesceri, L.S., Eds.; American Public Health Association: Washington, DC, USA, 2012; ISBN 9780875530130.
- CAMO. The Unscrambler Tutorials. CAMO Process AS 2006; CAMO Software AS: Oslo, Norway, 2006; p. 179. [Google Scholar]
- Olivieri, A.C. Introduction to Multivariate Calibration: A Practical Approach; Springer: Cham, Switzerland, 2018; ISBN 9783319970974. [Google Scholar]
- Van den Broeck, R.; Krzeminski, P.; Van Dierdonck, J.; Gins, G.; Lousada-Ferreira, M.; Van Impe, J.F.M.; van der Graaf, J.H.J.M.; Smets, I.Y.; van Lier, J.B. Activated sludge characteristics affecting sludge filterability in municipal and industrial MBRs: Unraveling correlations using multi-component regression analysis. J. Membr. Sci. 2011, 378, 330–338. [Google Scholar] [CrossRef]
- Wold, S.; Sjostrom, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemom. Intell. Lab. Syst. 2001, 58, 109–130. [Google Scholar] [CrossRef]
- Bratby, J. Coagulation and Flocculation in Water and Wastewater Treatment, 3rd ed.; IWA Publishing: London, UK, 2016; ISBN 9781780407500. [Google Scholar]
- Ratnaweera, H.C. Influence of the Degree of Coagulant Prepolymerization on Wastewater Coagulation Mechanisms. Ph.D. Thesis, The University of Trondheim. The Norwegian Institute of Technology, Trondheim, Norway, 1991. [Google Scholar]
- Stumm, W.; Morgan, J.J. Chemical Aspects of Coagulation. J. Am. Water Works Assoc. 1962, 54, 971–991. [Google Scholar] [CrossRef]
- Stumm, W.; O’Melia, C. Stoichiometry of coagulation. Am. Water Work. Assoc. 1968, 60, 514–539. [Google Scholar] [CrossRef]
- Meyn, T.; Leiknes, T.; Ødegaard, H. Coagulation/flocculation—Ceramic membrane filtration for removal of natural organic matter (NOM) under Norwegian conditions. In Proceedings of the IWA Conference on Membranes for Water and Wastewater Treatment, Harrogate, UK, 15–17 May 2007. [Google Scholar]
- Liang, Z.; Wang, Y.; Zhou, Y.; Liu, H.; Wu, Z. Polishing treatment of molasses wastewater with iron salts: The role of counter-ions. Water Environ. Res. 2009, 81, 2293–2298. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.; Liana, A.E.; Liu, S.; Lim, M.; Chow, C.W.K.; Wang, D.; Drikas, M.; Amal, R. Preparation and characterisation of new-polyaluminum chloride-chitosan composite coagulant. Water Res. 2012, 46, 4614–4620. [Google Scholar] [CrossRef] [PubMed]
- American Water College Zeta Potential—Coagulation and Flocculation Lecture. Available online: https://thewaternetwork.com/article-FfV/zeta-potential-coagulation-and-flocculation-lecture-video-gUbFb9OUygf-evijymdc6g (accessed on 29 November 2018).
- Hall, E.S.; Packham, R.F. Coagulation of Organic Color With Hydrolyzing Coagulants. J. Am. Water Work. Assoc. 1965, 57, 1149–1166. [Google Scholar] [CrossRef]
- Guminska, J.; Klos, M. Effect of Polyaluminium Chlorides Overdosage on Effectiveness of Coagulation and Filtration. Environ. Prot. Eng. 2015, 41, 5–14. [Google Scholar] [CrossRef]
- Bachand, P.A.M.; Bachand, S.M.; Lopus, S.E.; Heyvaert, A.; Werner, I. Treatment with chemical coagulants at different dosing levels changes ecotoxicity of stormwater from the Tahoe basin, California, USA. J. Environ. Sci. Heal. Part A Toxic/Hazard. Subst. Environ. Eng. 2010, 45, 137–154. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wu, Z.; Tang, S. Impact of temperature seasonal change on sludge characteristics and membrane fouling in a submerged membrane bioreactor. Sep. Sci. Technol. 2010, 45, 920–927. [Google Scholar] [CrossRef]
- Sobeck, D.C.; Higgins, M.J. Examination of three theories for mechanisms of cation-induced bioflocculation. Water Res. 2002, 36, 527–538. [Google Scholar] [CrossRef]
- Klute, R. Destabilization and Aggregation in Turbulent Pipe Flow. Chem. Water Wastewater Treat. 1990, 33–54. [Google Scholar] [CrossRef]
- Drews, A. Membrane fouling in membrane bioreactors-Characterisation, contradictions, cause and cures. J. Membr. Sci. 2010, 363, 1–28. [Google Scholar] [CrossRef]
- Miller, D.J.; Kasemset, S.; Paul, D.R.; Freeman, B.D. Comparison of membrane fouling at constant flux and constant transmembrane pressure conditions. J. Membr. Sci. 2014, 454, 505–515. [Google Scholar] [CrossRef]
- Council of the European Communities. Directive Concerning Urban Waste Water Treatment (91/271/EEC); Urban Waste Water Directive: Brussels, Belgium, 1991; pp. 40–52. [Google Scholar]
- Ratnaweera, H.; Ødegaard, H.; Fettig, J. Coagulation with prepolymerised aluminium salts and their influence on particle and phosphate removal. Water Sci. Technol. 1992, 6, 1229–1237. [Google Scholar] [CrossRef]
- Zhang, X.; Fan, L.; Roddick, F.A. Feedwater coagulation to mitigate the fouling of a ceramic MF membrane caused by soluble algal organic matter. Sep. Purif. Technol. 2014, 133, 221–226. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. EPA Ambient Water Quality Criteria for Aluminium; Office of Water Regulations and Standards Criteria and Standards Division: Washington, DC, USA, 1988.
- European Commission. EU-Level Instruments on Water Reuse: Final Report to Support the Commission’s Impact Assessment; European Commission: Brussels, Belgium, 2016. [Google Scholar]
- Eignor, D. Draft National 304(a) Aluminum Aquatic Life Criteria. Available online: https://www.epa.gov/sites/production/files/2017-09/documents/aluminum-webinar-9192017.pdf (accessed on 1 December 2018).
- Zhang, Z.; Wang, Y.; Leslie, G.L.; Waite, T.D. Effect of ferric and ferrous iron addition on phosphorus removal and fouling in submerged membrane bioreactors. Water Res. 2015, 69, 210–222. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-M.; Waite, T.D. Role of gelling soluble and colloidal microbial products in membrane fouling. Environ. Sci. Technol. 2009, 43, 9341–9347. [Google Scholar] [CrossRef] [PubMed]
- Dulko, J.M. Stable Concentrated Polyaluminium Chlorosilicate Solutions. US 2016/0244345 A1, issued 2016. US 2016/0244345 A1, 2016. [Google Scholar] [CrossRef]
- Dulko, J.M. Stable Concentrated Polyaluminum Chlorosilicate Solutions. WO 2016/134277 Al, 2016. Available online: https://patents.google.com/patent/WO2016134277A1 (accessed on 28 February 2018).
- Zouboulis, A.I.; Tzoupanos, N.D. Polyaluminium silicate chloride-A systematic study for the preparation and application of an efficient coagulant for water or wastewater treatment. J. Hazard. Mater. 2009, 162, 1379–1389. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.Y.; Yue, Q.Y.; Wang, B.J.; Chu, Y.B. Poly-aluminum-silicate-chloride (PASiC)—A new type of composite inorganic polymer coagulant. Colloids Surfaces A Physicochem. Eng. Asp. 2003, 229, 121–127. [Google Scholar] [CrossRef]
- Zhang, P.; Hahn, H.H.; Hoffmann, E.; Zeng, G. Influence of some additives to aluminium species distribution in aluminium coagulants. Chemosphere 2004, 57, 1489–1494. [Google Scholar] [CrossRef] [PubMed]
- Tzoupanos, N.D.; Zouboulis, A.I.; Tsoleridis, C.A. A systematic study for the characterization of a novel coagulant (polyaluminium silicate chloride). Colloids Surf. A Physicochem. Eng. Asp. 2009, 342, 30–39. [Google Scholar] [CrossRef]
- Ratnaweera, H.; Fettig, J. State of the art of online monitoring and control of the coagulation process. Water (Switzerland) 2015, 7, 6574–6597. [Google Scholar] [CrossRef]
- Adgar, A.; Cox, C.S.; Jones, C.A. Enhancement of coagulation control using the streaming current detector. Bioprocess Biosyst. Eng. 2005, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Brookes, A.; Judd, S.; Reid, E.; Germain, E.; Smith, S.; Alvarez-Vazquez, H.; Le-Clech, P.; Stephenson, T.; Turra, E.; Jefferson, B. Biomass characterisation in membrane bioreactors. In Proceedings of the International Membrane Science and Technology Conference (IMSTEC), Sydney, Australia, 10–14 November 2003. [Google Scholar]
- Striegel, A.M. Size-exclusion chromatography. In Liquid Chromatography: Fundamentals and Instrumentation: Second Edition; Elsevier: Gaithersburg, MD, USA, 2017; pp. 193–224. ISBN 9780128053935. [Google Scholar]
- Montgomery, D.C. Design and Analysis of Experiments, 15th ed.John Wiley & Sons, Inc.: Scottsdale, AZ, USA, 1997. [Google Scholar]
- Stat-Ease Inc. Design-Expert 11.1.0. Documentation. Tutorials. Available online: https://www.statease.com/docs/v11/index.html (accessed on 31 October 2018).
- Ogee, A.; Ellis, M.; Stone, B.K.; Scibilia, B.; Pammer, C.; Steele, C. MiniTab 18: What Are T Values and P Values in Statistics? Available online: https://blog.minitab.com/blog/statistics-and-quality-data-analysis/what-are-t-values-and-p-values-in-statistics (accessed on 12 January 2019).
- Mendenhall, W.; Sincich, T. A Second Course in Statistics. Regression Analysis, 7th ed.; Lynch, D., Ed.; Pearson Education, Inc.: Boston, MA, USA, 2012; ISBN 0-321-69169-5. [Google Scholar]
- Winter, B. The F Distribution and the Basic Principle behind ANOVAs. Available online: http://www.bodowinter.com/tutorial/bw_anova_general.pdf (accessed on 1 November 2018).
- The Bayes Information Criterion (BIC). Available online: http://www-math.mit.edu/~rmd/650/bic.pdf (accessed on 1 November 2018).
- Merisaari, H.; Jambor, I.; Jødal, L.; Oikonen, V. Akaike Information Criterium (AIC) in Model Selection. Available online: http://www.turkupetcentre.net/petanalysis/model_aic.html (accessed on 1 November 2018).
- Montgomery, D.C. Design and Analysis of Experiments, 8th ed.; Ratts, L., Melhorn, A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; ISBN 9781118146927. [Google Scholar]
- Hair, J.F.; Black, W.C.; Babin, B.J.; Anderson, R.E. Multivariate Data Analysis. Pearson New International Edition, 7th ed.; Pearson Education Limited: Harlow, UK, 2014; ISBN 9781292021904. [Google Scholar]
- Azami, H.; Sarrafzadeh, M.H.; Mehrnia, M.R. Influence of sludge rheological properties on the membrane fouling in submerged membrane bioreactor. Desalin. Water Treat. 2011, 34, 117–122. [Google Scholar] [CrossRef]
- Meng, F.; Shi, B.; Yang, F.; Zhang, H. Effect of hydraulic retention time on membrane fouling and biomass characteristics in submerged membrane bioreactors. Bioprocess Biosyst. Eng. 2007, 30, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Chang, I.S.; Kim, S.N. Wastewater treatment using membrane filtration—Effect of biosolids concentration on cake resistance. Process Biochem. 2005, 40, 1307–1314. [Google Scholar] [CrossRef]
- Le-Clech, P.; Chen, V.; Fane, T.A.G. Fouling in membrane bioreactors used in wastewater treatment. J. Membr. Sci. 2006, 284, 17–53. [Google Scholar] [CrossRef]
- Ji, L.; Zhou, J. Influence of aeration on microbial polymers and membrane fouling in submerged membrane bioreactors. J. Membr. Sci. 2006, 276, 168–177. [Google Scholar] [CrossRef]
- Hernandez Rojas, M.E.; Van Kaam, R.; Schetrite, S.; Albasi, C. Role and variations of supernatant compounds in submerged membrane bioreactor fouling. Desalination 2005, 179, 95–107. [Google Scholar] [CrossRef]
- Xie, W.M.; Ni, B.J.; Sheng, G.P.; Seviour, T.; Yu, H.Q. Quantification and kinetic characterization of soluble microbial products from municipal wastewater treatment plants. Water Res. 2016, 88, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.; Zhang, S.; Xu, L.; Zhang, M.; Zhu, J. Performance and fouling characteristics in a membrane sequence batch reactor (MSBR) system coupled with aerobic granular sludge. Desalination 2010, 261, 191–196. [Google Scholar] [CrossRef]
- Deng, L.; Guo, W.; Ngo, H.H.; Zhang, J.; Liang, S.; Xia, S.; Zhang, Z.; Li, J. A comparison study on membrane fouling in a sponge-submerged membrane bioreactor and a conventional membrane bioreactor. Bioresour. Technol. 2014, 165, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yu, H.; Zhang, L.; Song, L. Bioresource Technology Stratification structure of polysaccharides and proteins in activated sludge with different aeration in membrane bioreactor. Bioresour. Technol. 2015, 192, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Johir, M.A.H.; Vigneswaran, S.; Sathasivan, A.; Kandasamy, J.; Chang, C.Y. Effect of organic loading rate on organic matter and foulant characteristics in membrane bio-reactor. Bioresour. Technol. 2012, 113, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Zhang, G.; Meng, Q.; Zhang, H.; Xu, L.; Lv, B. Enhanced submerged membrane bioreactor combined with biosurfactant rhamnolipids: Performance for frying oil degradation and membrane fouling reduction. Bioresour. Technol. 2012, 126, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Zhao, W.; Xiao, K.; Huang, X. A systematic insight into fouling propensity of soluble microbial products in membrane bioreactors based on hydrophobic interaction and size exclusion. J. Membr. Sci. 2010, 346, 187–193. [Google Scholar] [CrossRef]
- Pramanik, B.K.; Roddick, F.A.; Fan, L. Impact of biological activated carbon pre-treatment on the hydrophilic fraction of effluent organic matter for mitigating fouling in microfiltration. Environ. Technol. 2018, 39, 2243–2250. [Google Scholar] [CrossRef] [PubMed]
- Mu, S.; Wang, S.; Liang, S.; Xiao, K.; Fan, H.; Han, B.; Liu, C.; Wang, X.; Huang, X. Effect of the relative degree of foulant “hydrophobicity” on membrane fouling. J. Membr. Sci. 2019, 570–571, 1–8. [Google Scholar] [CrossRef]
- Ivanovic, I.; Leiknes, T.O.; Ødegaard, H. Fouling control by reduction of submicron particles in a BF-MBR with an integrated flocculation zone in the membrane reactor. Sep. Sci. Technol. 2008, 43, 1871–1883. [Google Scholar] [CrossRef]
- Tian, Y.; Chen, L.; Zhang, S.; Cao, C.; Zhang, S. Correlating membrane fouling with sludge characteristics in membrane bioreactors: An especial interest in EPS and sludge morphology analysis. Bioresour. Technol. 2011, 102, 8820–8827. [Google Scholar] [CrossRef] [PubMed]
- Tarnacki, K.; Lyko, S.; Wintgens, T.; Melin, T.; Natau, F. Impact of extra-cellular polymeric substances on the filterability of activated sludge in membrane bioreactors for landfill leachate treatment. Desalination 2005, 179, 181–190. [Google Scholar] [CrossRef]
- Rosenberger, S.; Evenblij, H.; Te Poele, S.; Wintgens, T.; Laabs, C. The importance of liquid phase analyses to understand fouling in membrane assisted activated sludge processes—Six case studies of different European research groups. J. Membr. Sci. 2005, 263, 113–126. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, Z.; Zhou, Z.; Yu, G.; Gu, G. Sludge rheological and physiological characteristics in a pilot-scale submerged membrane bioreactor. Desalination 2007, 212, 152–164. [Google Scholar] [CrossRef]
- Pan, J.R.; Su, Y.C.; Huang, C.; Lee, H.C. Effect of sludge characteristics on membrane fouling in membrane bioreactors. J. Membr. Sci. 2010, 349, 287–294. [Google Scholar] [CrossRef]
- Jin, B.; Wilén, B.M.; Lant, P. Impacts of morphological, physical and chemical properties of sludge flocs on dewaterability of activated sludge. Chem. Eng. J. 2004, 98, 115–126. [Google Scholar] [CrossRef]
- Niwa, R.; Yin, T.; Oo, M.H.; Noguchi, H.; Watanabe, T.; Razali, L.Y.; Png, H.Y.; Lay, W.C.L.; Ong, K.A.; Alom, M. Performance of a Full-Scale Ceramic MBR System to Treat Domestic Sewage. Water Pract. Technol. 2018, 13, 589–593. [Google Scholar] [CrossRef]
- Gkotsis, P.K.; Mitrakas, M.M.; Tolkou, A.K.; Zouboulis, A.I. Batch and continuous dosing of conventional and composite coagulation agents for fouling control in a pilot-scale MBR. Chem. Eng. J. 2016, 311, 255–264. [Google Scholar] [CrossRef]
- Fan, F.; Zhou, H.; Husain, H. Identification of wastewater sludge characteristics to predict critical flux for membrane bioreactor processes. Water Res. 2006, 40, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Schnoor, J.J., Zehnder, A., Eds.; John Wiley & Soms, Inc.: New York, NY, USA, 1996. [Google Scholar]
- Moran, A.P.; Holst, O.; Brennan, P.J.; von Itzstein, M. (Eds.) Microbial Glycobiology: Structures, Relevance and Applications; Elsevier Inc.: London, UK; Burlington: San Diego, CA, USA, 2009; ISBN 978-0-12-374546-0. [Google Scholar]
- Chen, J.; Lin, H.; Shen, L.; He, Y.; Zhang, M.; Liao, B.Q. Realization of quantifying interfacial interactions between a randomly rough membrane surface and a foulant particle. Bioresour. Technol. 2017, 226, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhou, X.; Shen, L.; Cai, X.; Wang, F.; Chen, J.; Lin, H.; Li, R.; Wu, X.; Liao, B.Q. Quantitative evaluation of the interfacial interactions between a randomly rough sludge floc and membrane surface in a membrane bioreactor based on fractal geometry. Bioresour. Technol. 2017, 234, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.; Zhang, H.; Yang, F.; Zhang, S.; Li, Y.; Zhang, X. Identification of activated sludge properties affecting membrane fouling in submerged membrane bioreactors. Sep. Purif. Technol. 2006, 51, 95–103. [Google Scholar] [CrossRef]
- Ji, J.; Qiu, J.; Wai, N.; Wong, F.S.; Li, Y. Influence of organic and inorganic flocculants on physical-chemical properties of biomass and membrane-fouling rate. Water Res. 2010, 44, 1627–1635. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Fan, Z.; Xu, L.; Zhang, G.; Wang, G.; Wu, D.; Long, X.; Meng, Q. A submerged membrane bioreactor with pendulum type oscillation (PTO) for oily wastewater treatment: Membrane permeability and fouling control. Bioresour. Technol. 2015, 183, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Jang, N.; Ren, X.; Choi, K.; Kim, I.S. Comparison of membrane biofouling in nitrification and denitrification for the membrane bioreactor (MBR). Water Sci. Technol. 2006, 53, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Ong, S.L.; Ng, H.Y. Fouling control mechanism by suspended biofilm carriers addition in submerged ceramic membrane bioreactors. J. Membr. Sci. 2013, 427, 250–258. [Google Scholar] [CrossRef]
- Lykkegaard, M.; Niessen, W.; Bøie, N.; Hove, S.; Koustrup, M.; Halkjær, P. Sludge fractionation as a method to study and predict fouling in MBR systems. Sep. Purif. Technol. 2018, 194, 329–337. [Google Scholar] [CrossRef]
- Remy, M.; Potier, V.; Temmink, H.; Rulkens, W. Why low powdered activated carbon addition reduces membrane fouling in MBRs. Water Res. 2010, 44, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Philippe, N.; Racault, Y.; Stricker, A.E.; Sp, M.; Vanrolleghem, P.A. Modelling the long-term evolution of permeability in a full-scale MBR: Statistical approaches. Desalination 2013, 325, 7–15. [Google Scholar] [CrossRef]
- Naessens, W.; Maere, T.; De Temmerman, L.; Nopens, I. Principal component analysis for monitoring membrane bioreactors: Trend detection, outlier detection and optimization. In Proceedings of the 2013 International Workshop—Membrane Bioreactor for Wastewater Reuse: Fundamental, Design and Operation, Tunis, Tunisia, 12–14 November 2013; pp. 2–5. [Google Scholar]
- Kaneko, H.; Funatsu, K. Visualization of Models Predicting Transmembrane Pressure Jump for Membrane Bioreactor. Ind. Eng. Chem. Res. 2012, 51, 9679–9686. [Google Scholar] [CrossRef]
- Choi, B.G.; Cho, J.; Song, K.G.; Maeng, S.K. Correlation between effluent organic matter characteristics and membrane fouling in a membrane bioreactor using advanced organic matter characterization tools. Desalination 2013, 309, 74–83. [Google Scholar] [CrossRef]
- Jacquin, C.; Gambier, N.; Lesage, G.; Heran, M. New insight into fate and fouling behavior of bulk Dissolved Organic Matter (DOM) in a full-scale membrane bioreactor for domestic wastewater treatment. J. Water Process Eng. 2018, 22, 94–102. [Google Scholar] [CrossRef]
- Geladi, P.; Kowalski, B.R. Partial least-squares regression: A tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar] [CrossRef]
- Sivchenko, N. Image Analysis in Coagulation Process Control. Ph.D. Thesis, Norwegian University of Life Sciences (NMBU), Ås, Norway, 2017. [Google Scholar]
- Wang, X.; Kvaal, K.; Ratnaweera, H. Characterization of influent wastewater with periodic variation and snow melting effect in cold climate area. Comput. Chem. Eng. 2017, 106, 202–211. [Google Scholar] [CrossRef]
- Liltved, H.; Ratnaweera, H.; Plo, B.G. Climate change impacts on activated sludge wastewater treatment: A case study from Norway. Water Sci. Technol. WST 2009, 60, 533–541. [Google Scholar] [CrossRef]
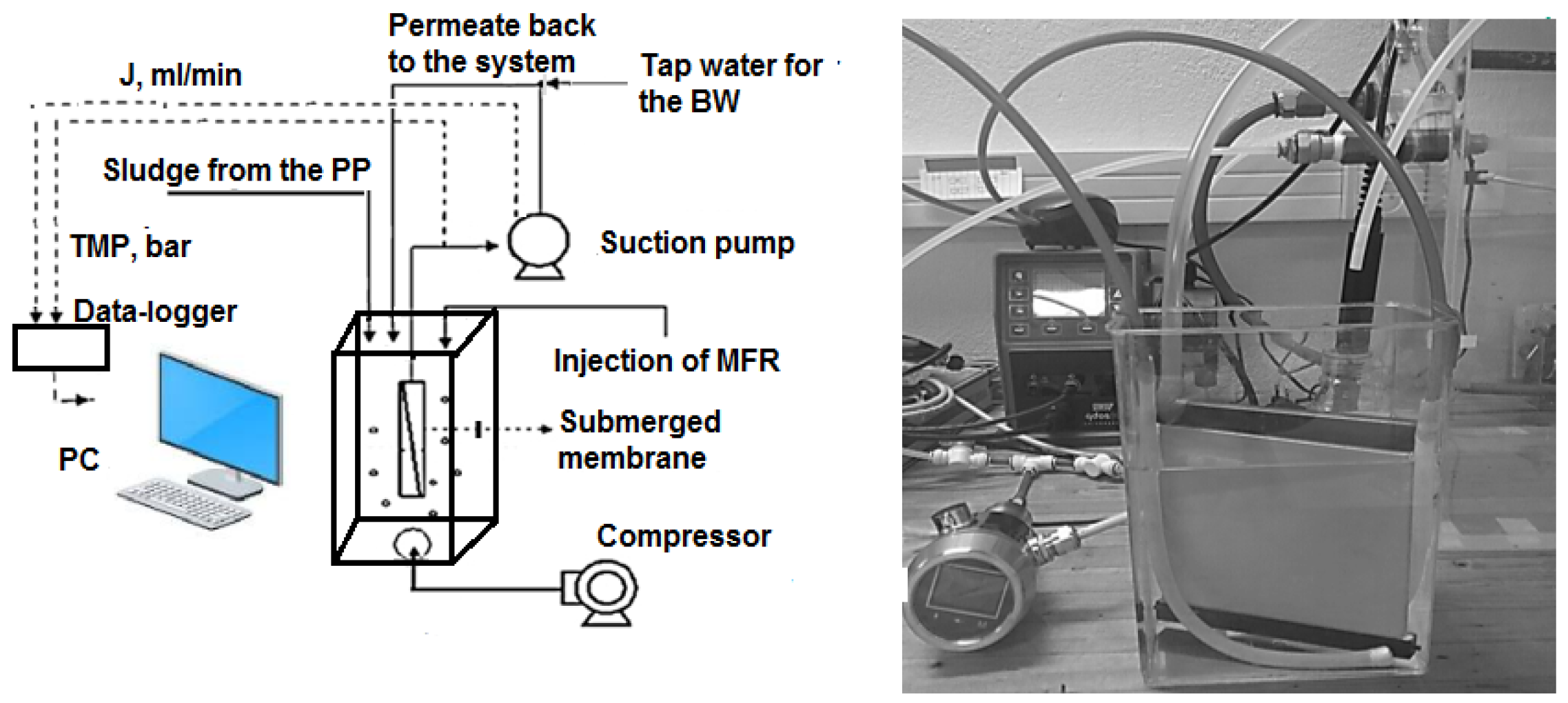
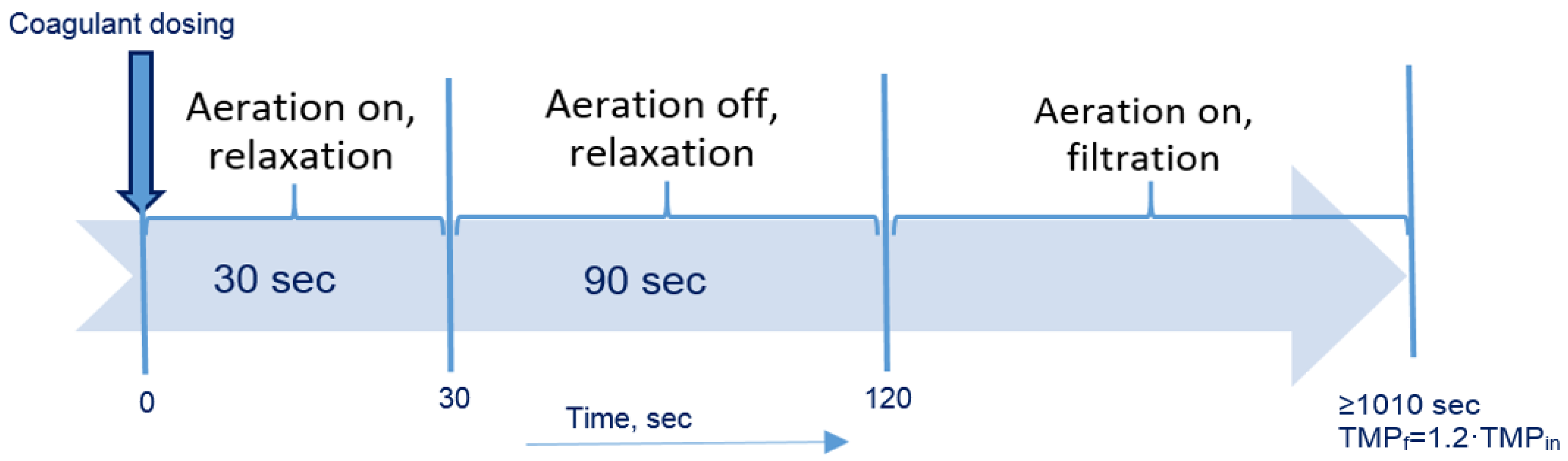

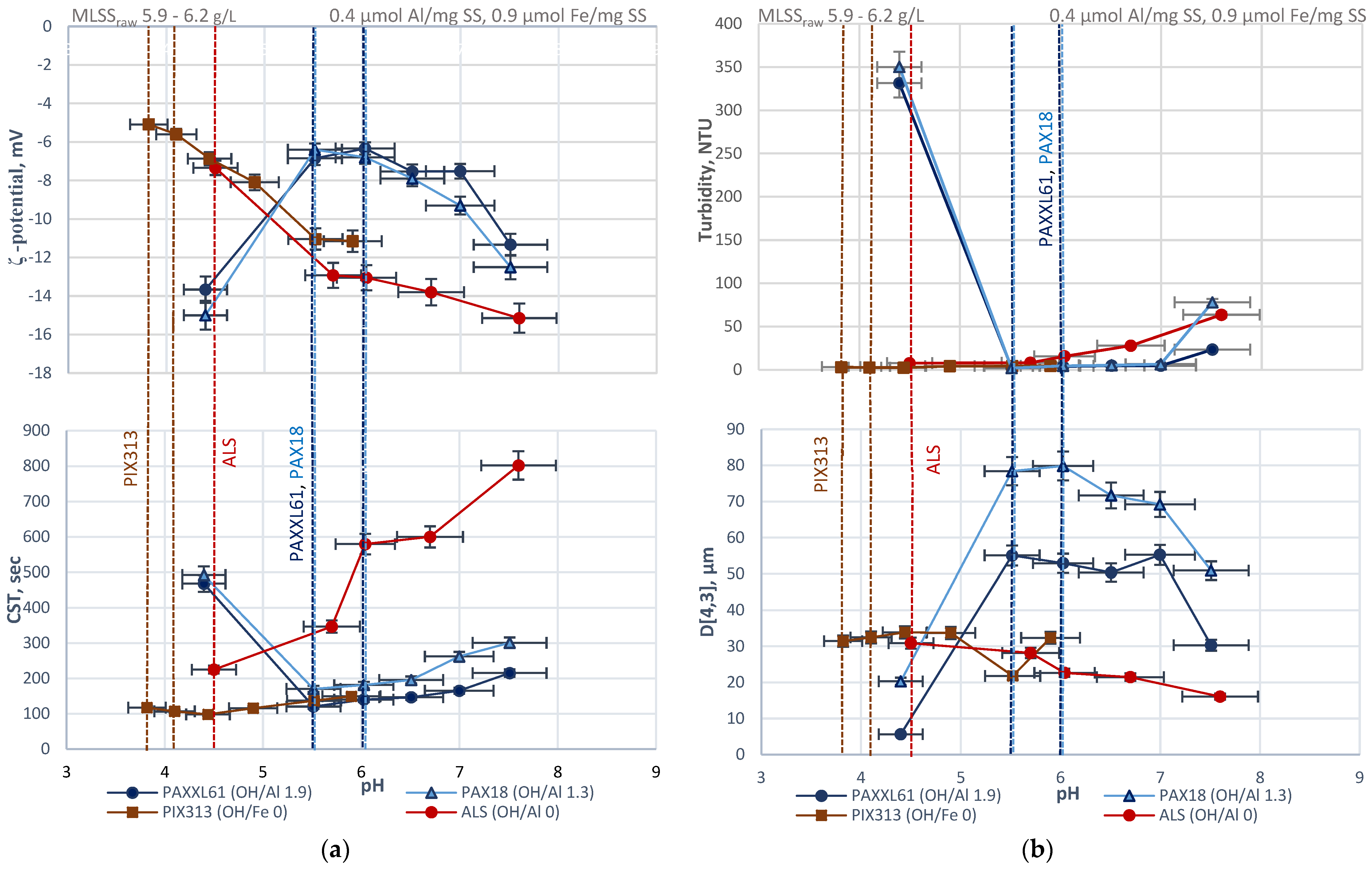
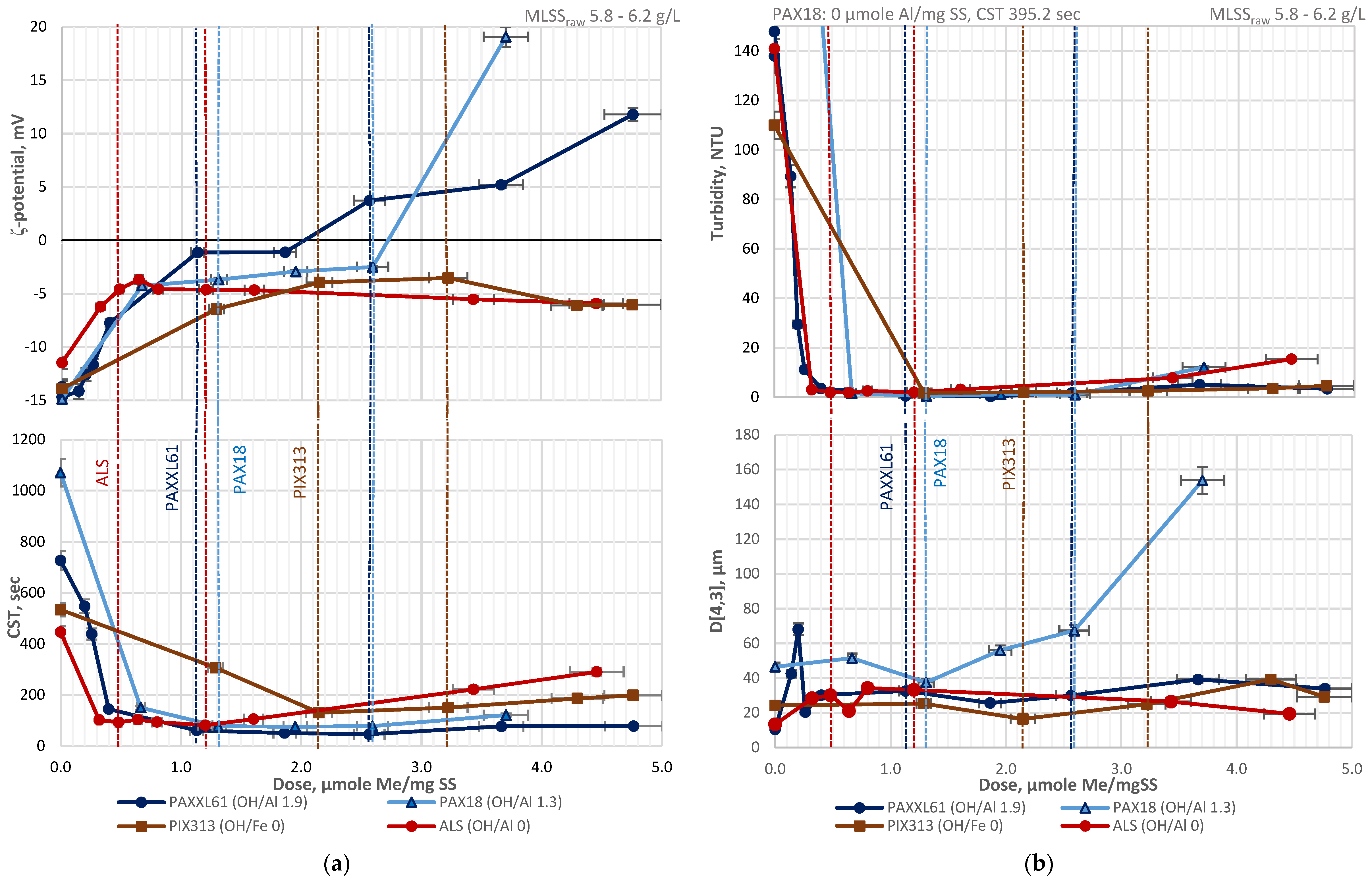
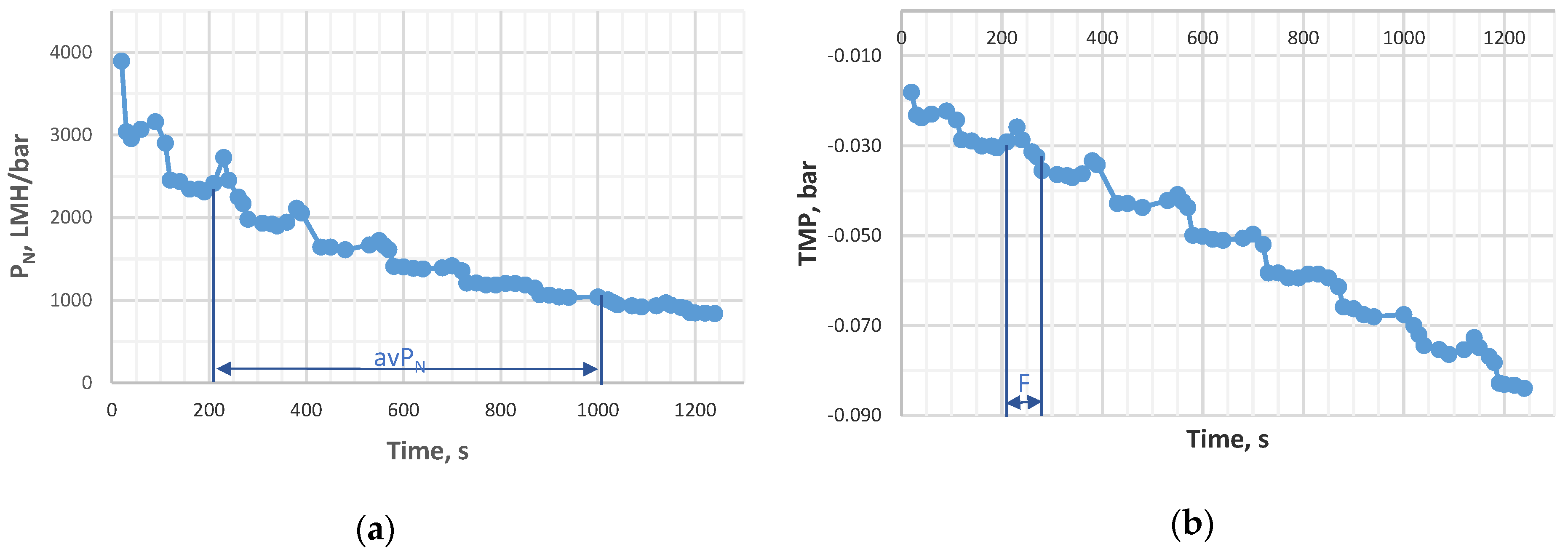
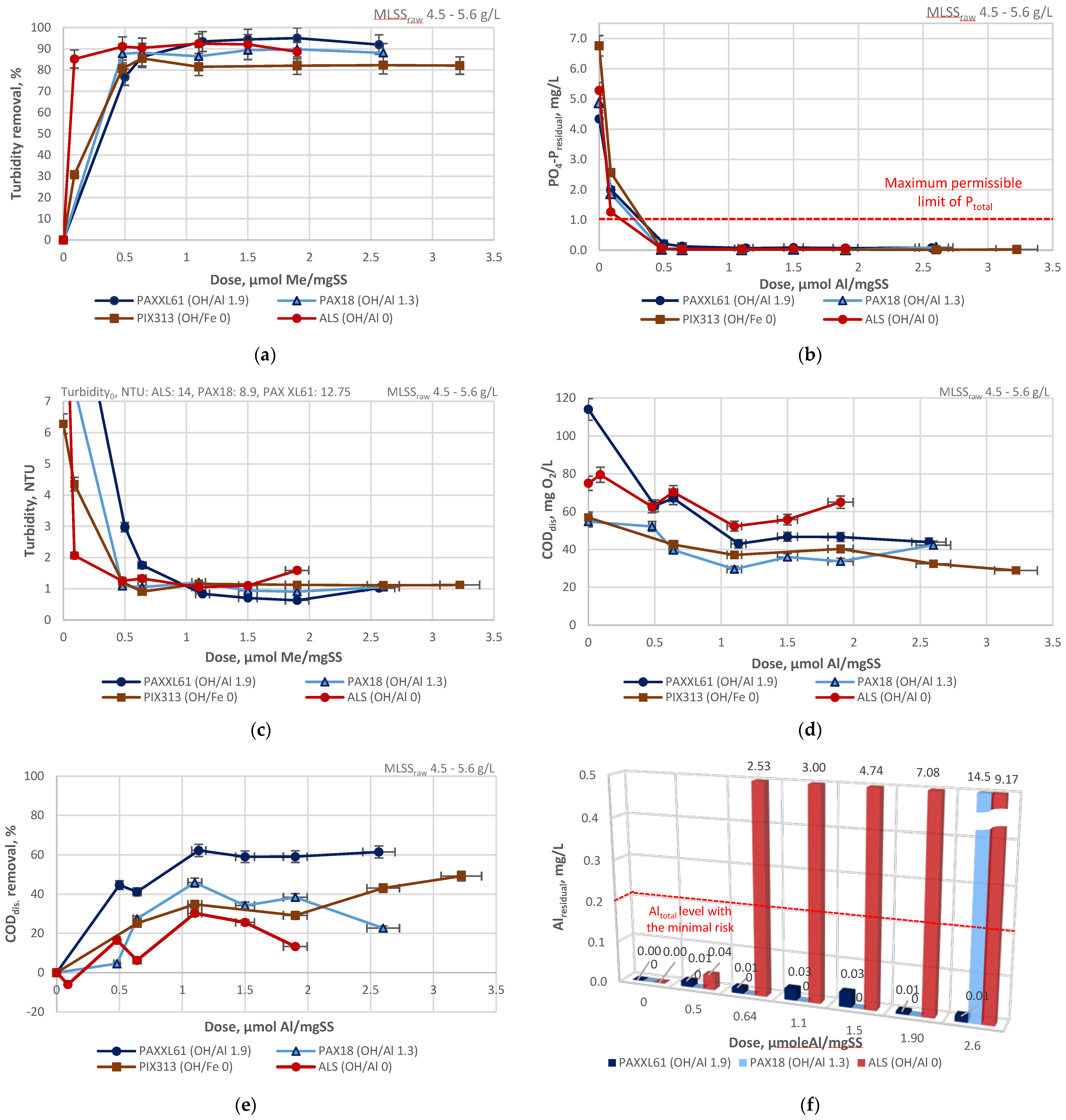
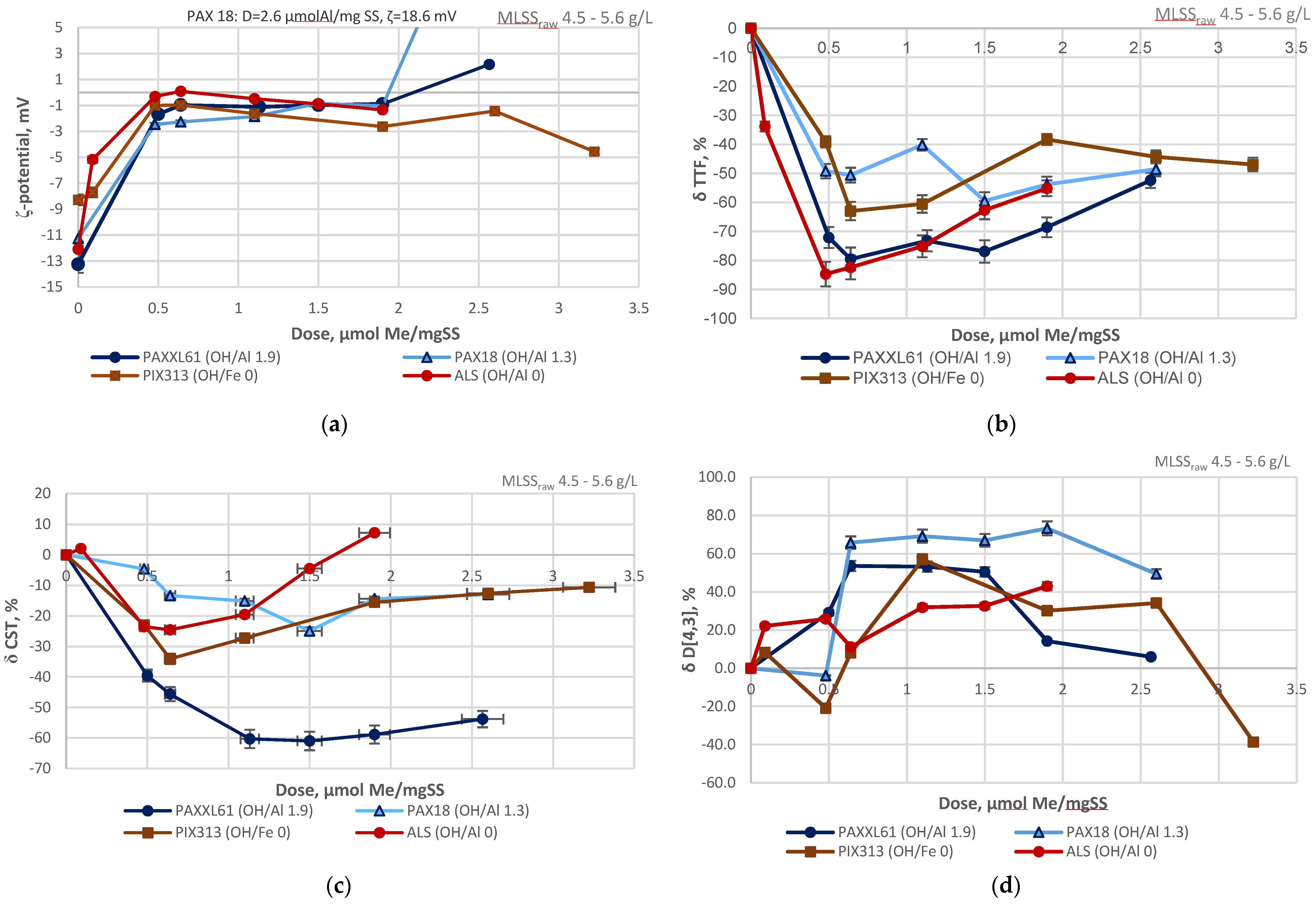
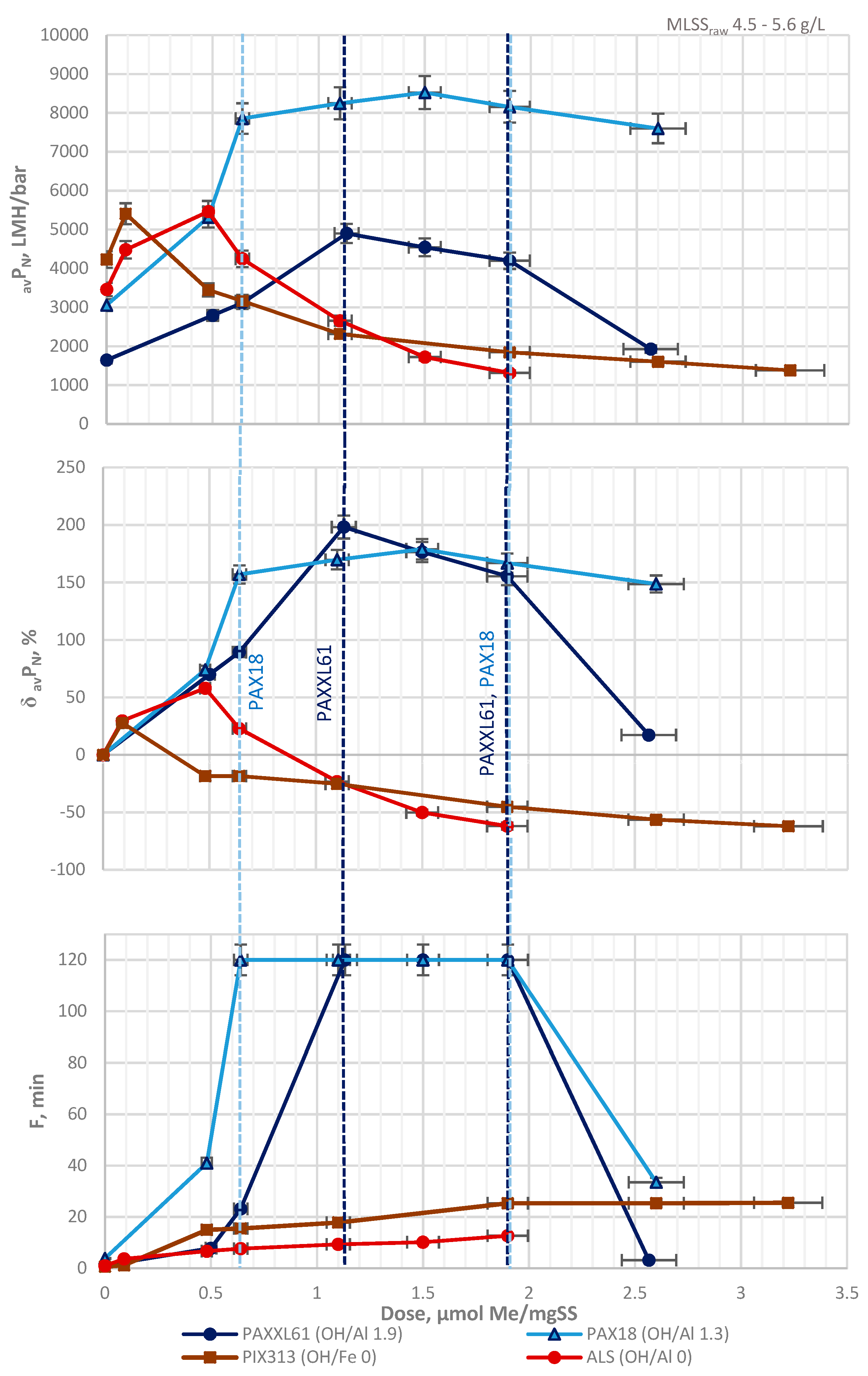
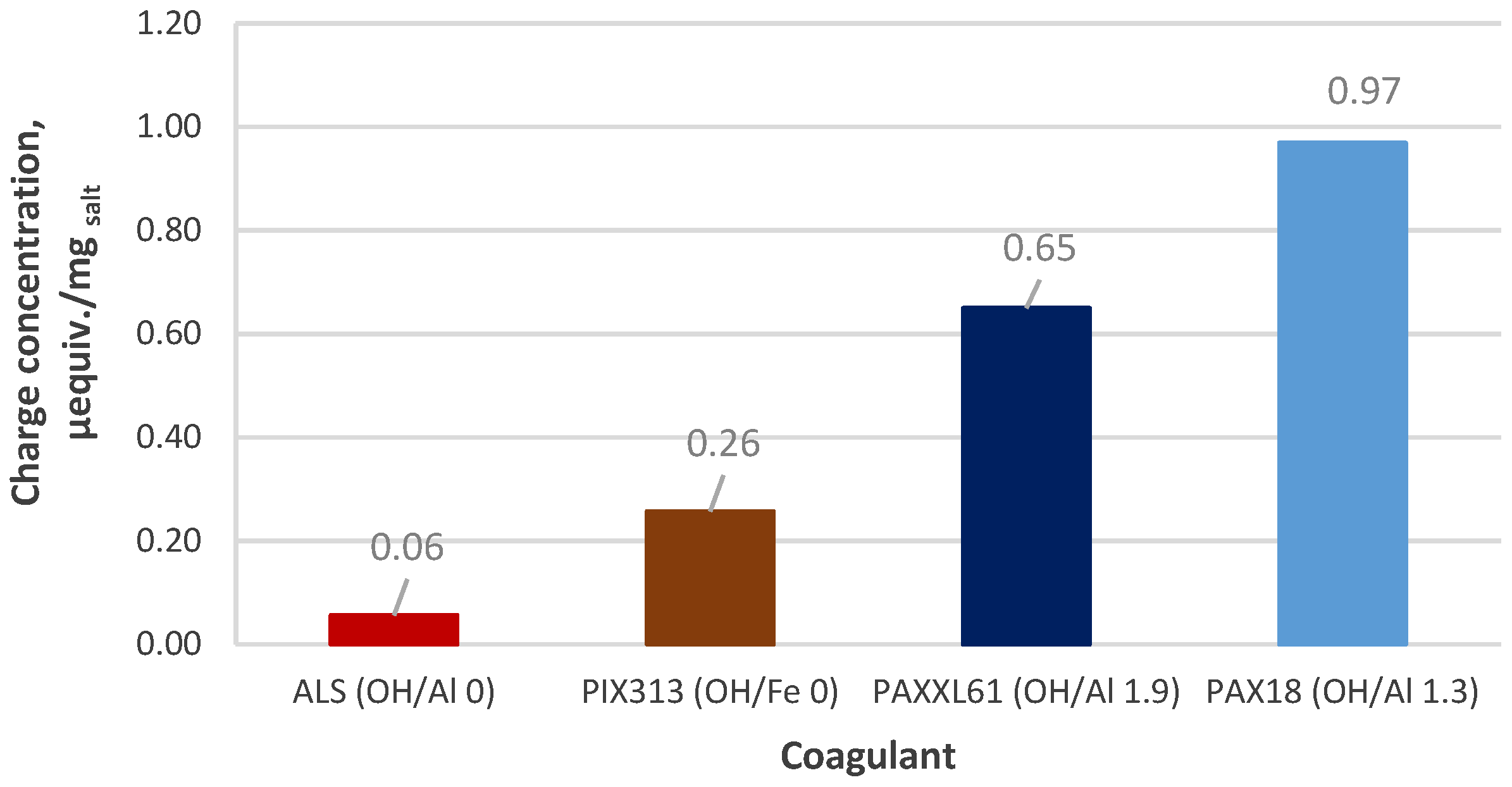
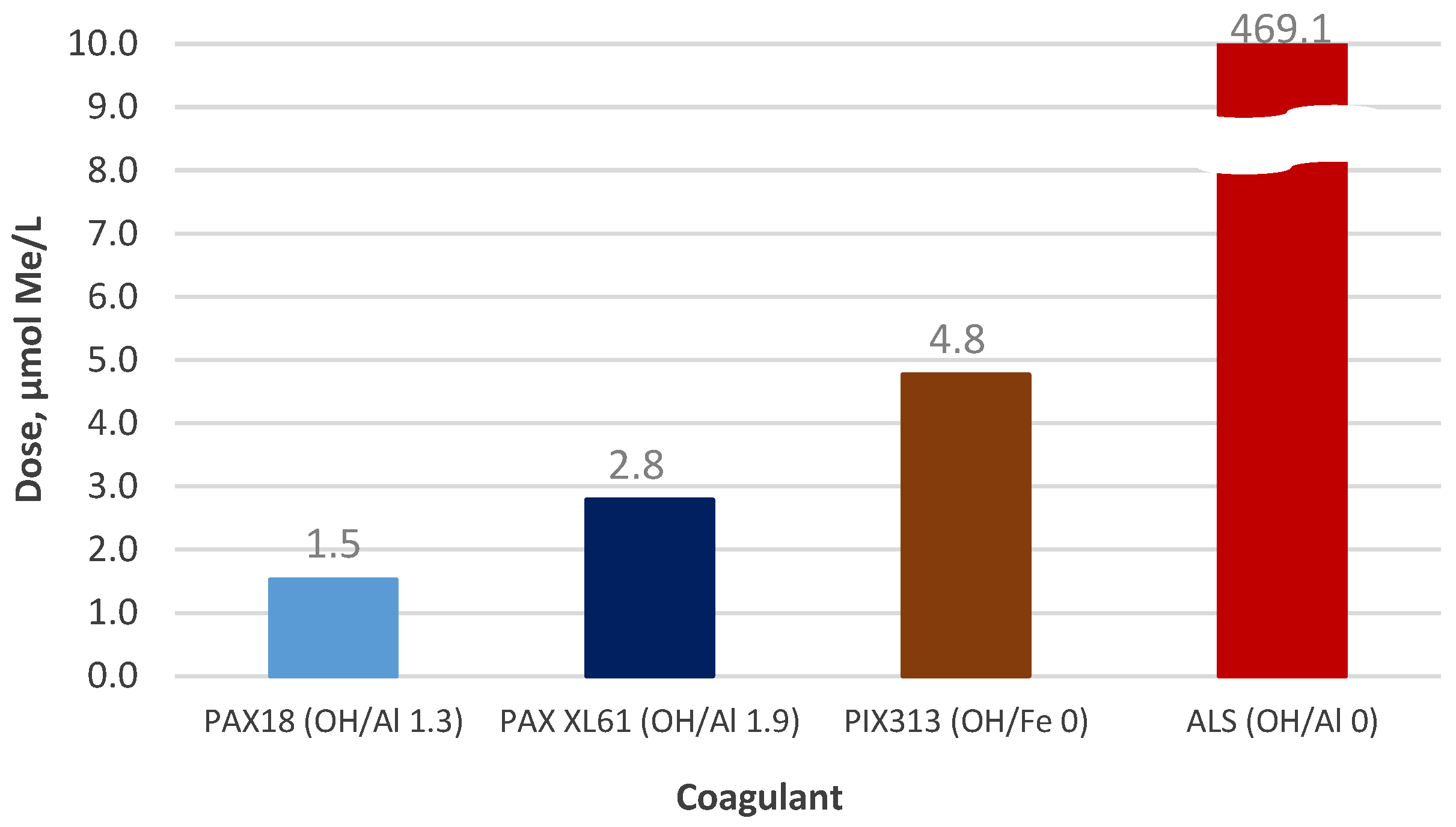
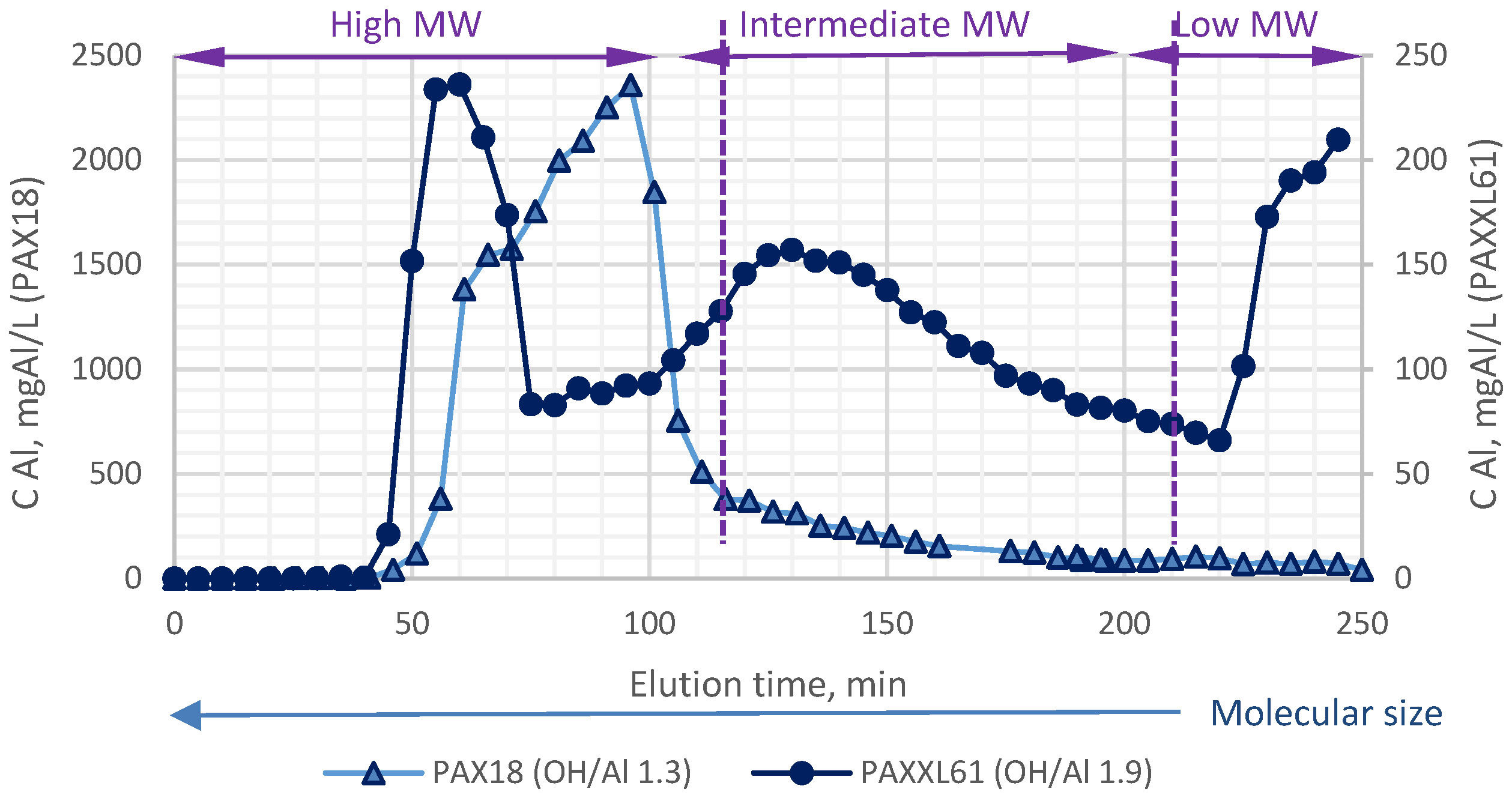
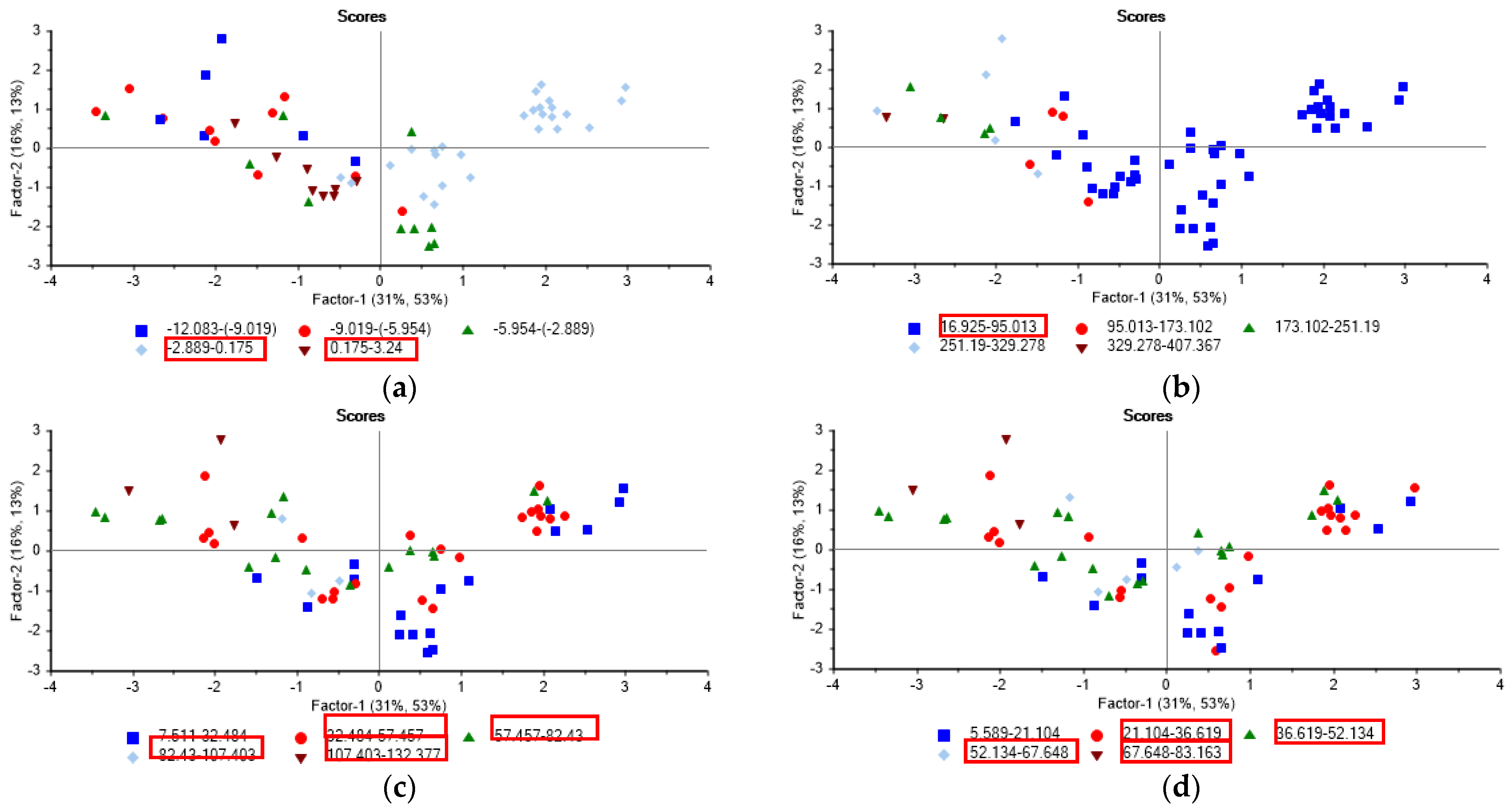
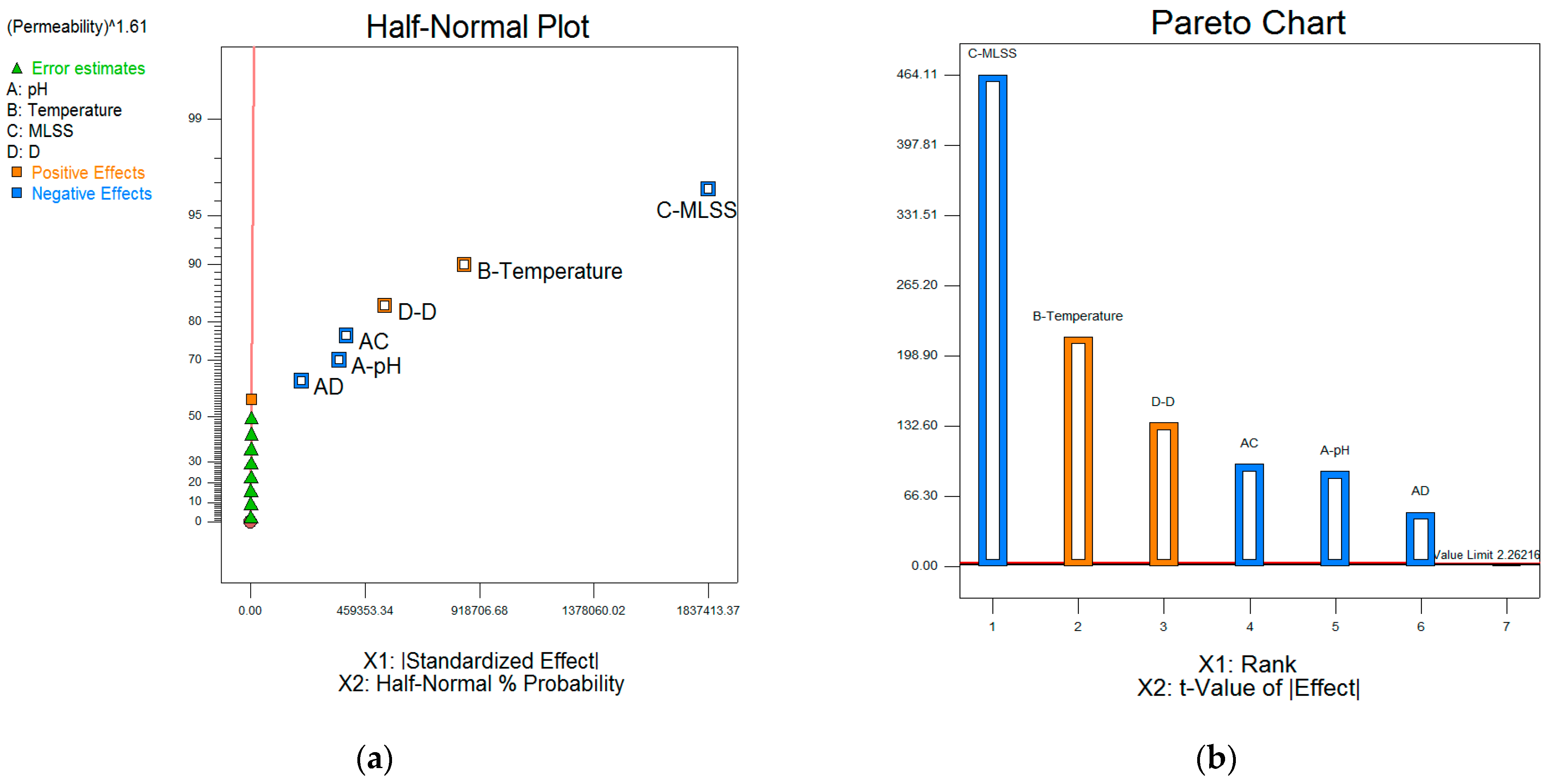
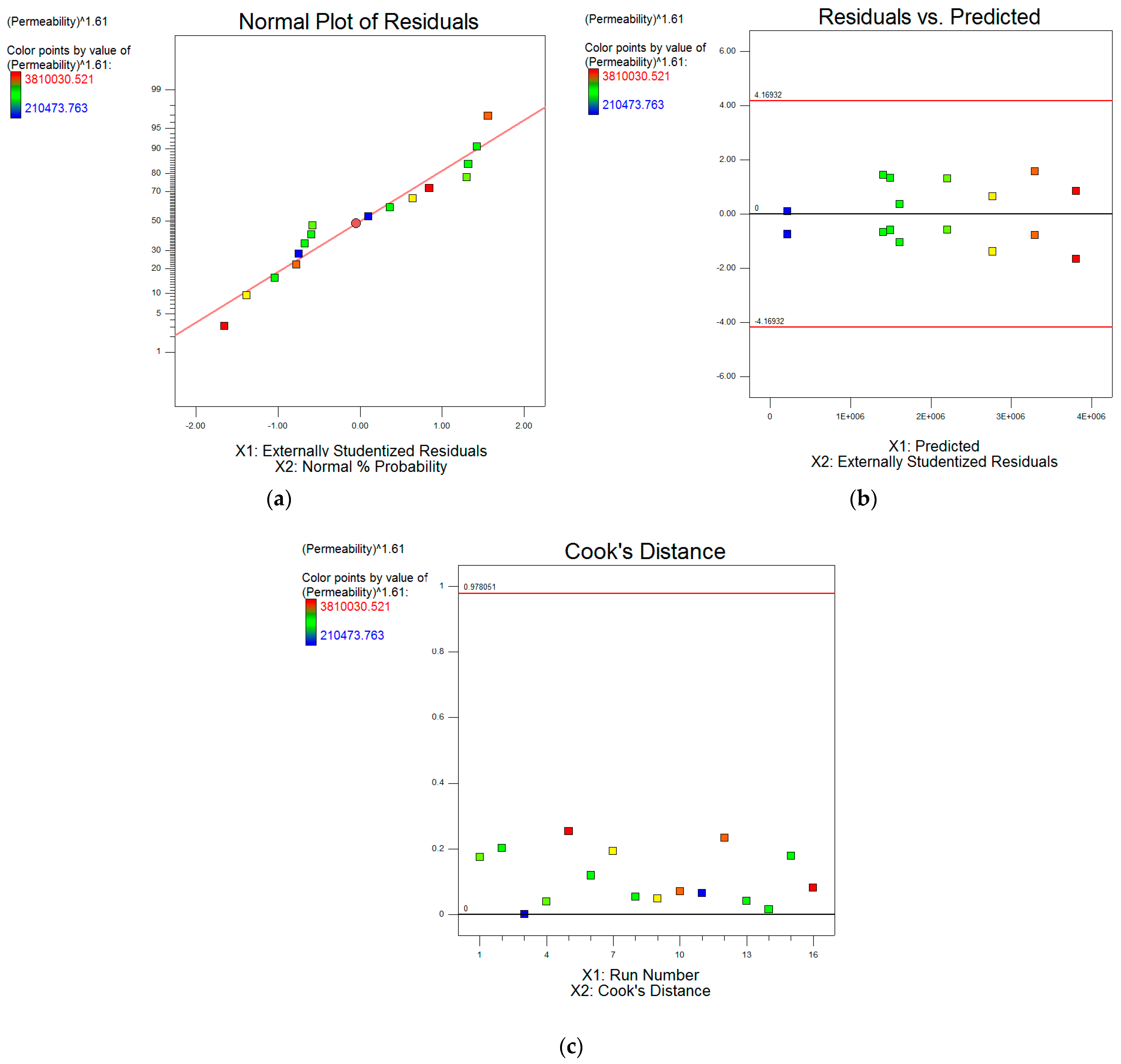
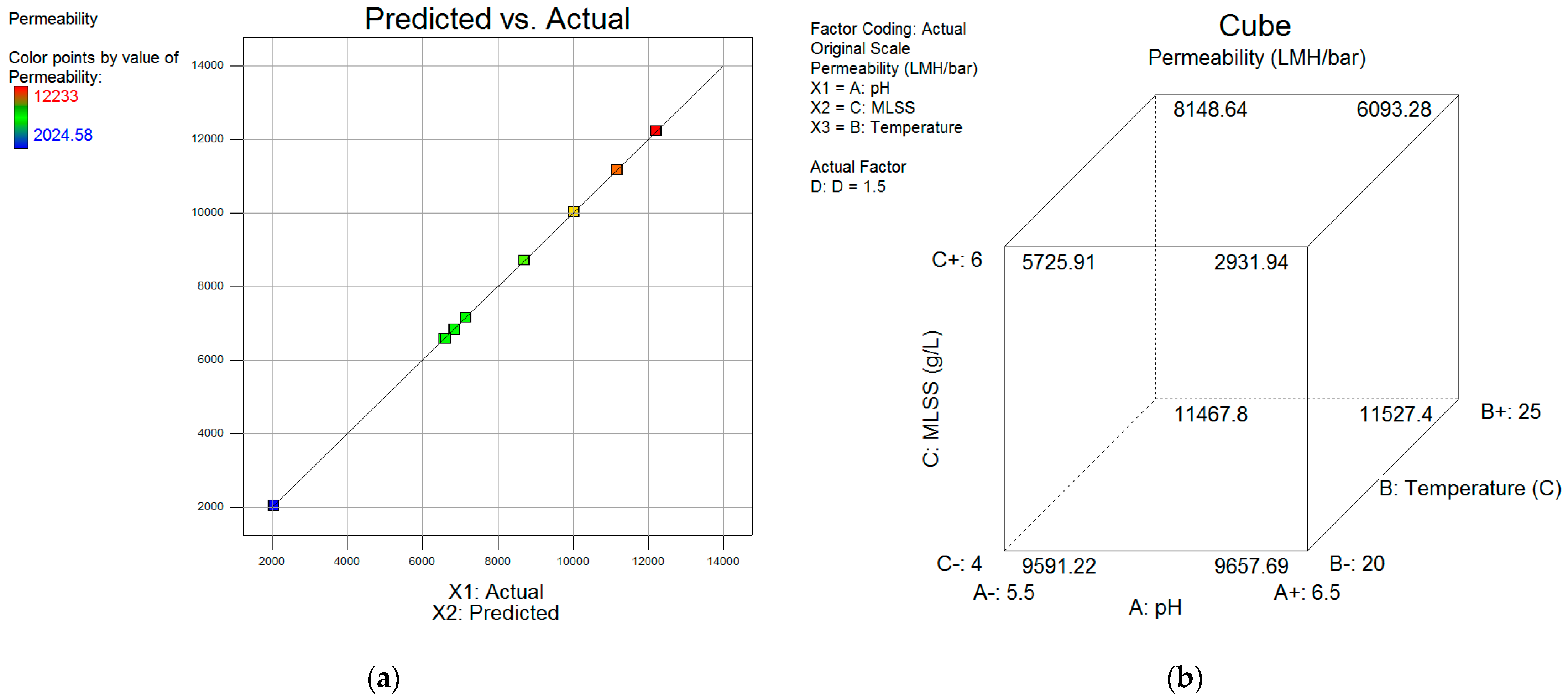
| Designation | Active Compound | Metal Content, % | Basicity (OH/Me) | Density (20 °C), g/cm3 | pH |
|---|---|---|---|---|---|
| PAX18 | [AlClOH]n | 9.0 ± 0.2% | 42.0 ± 2% (1.3) | 1.37 ± 0.03 | 0.6 |
| PAXXL61 | Al(OH)xCl(3-x-2y)(SiO2)y | 5.4 ± 0.3% | 68.0 ± 5 % (1.9) | 1.26 ± 0.03 | 2.7 |
| ALS | Al2(SO4)3 | 4.3 ± 0.1% | - (0) | 1.33 ± 0.01 | 1.8 |
| PIX313 | Fe2(SO4)3 | 11.6 ± 0.4% | - (0) | 1.52 ± 0.06 | <0.5 |
| Predictors (X) | Response (Y) |
|---|---|
| MLSSin1, CST 2, turbidity, pH, D[4,3], D[3,2], span, uniformity, zeta potential, coagulant dosage (Dose) | Average normalized permeability (avPN), Filtration time (F) |
| Mechanism of Action Coagulant | Enhanced Adsorption/Charge Neutralization | The Increase in Relative Hydrophobicity of the Flocs | The Increase in Particle Size | The Reduction of the SMPs Level |
|---|---|---|---|---|
| PAX18 (OH/Al 1.3) | + | + | + | + |
| PAXXL61 (OH/Al 1.9) | + | + | + | + |
| PIX313 (OH/Fe 0) | + | + | ||
| ALS (OH/Al 0) | + |
| Factor | Units | Low Level | High Level |
|---|---|---|---|
| pH | - | 5.5 | 6.5 |
| Temperature | °C | 20 | 25 |
| MLSS | g/L | 4.0 | 6.0 |
| D 1 | µmoleAl/mgSS | 1.1 | 1.9 |
| Factor 1 | Factor 2 | Factor 3 | Factor 4 | Response 1 | |
|---|---|---|---|---|---|
| Run | A:pH | B:Temperature, C | C:MLSS, g/L | D:D, µmolAl/mgSS | Permeability, LMH/bar |
| 1 | 5.5 | 20 | 4.0 | 1.1 | 8725.6 |
| 2 | 6.5 | 25 | 6.0 | 1.9 | 6614.1 |
| 3 | 6.5 | 20 | 6.0 | 1.1 | 2055.5 |
| 4 | 5.5 | 20 | 4.0 | 1.1 | 8698.6 |
| 5 | 5.5 | 25 | 4.0 | 1.9 | 12,204.9 |
| 6 | 5.5 | 25 | 6.0 | 1.1 | 7146.1 |
| 7 | 6.5 | 20 | 4.0 | 1.9 | 10,020.4 |
| 8 | 6.5 | 25 | 6.0 | 1.9 | 6578.7 |
| 9 | 6.5 | 20 | 4.0 | 1.9 | 10,046.9 |
| 10 | 6.5 | 25 | 4.0 | 1.1 | 11,168.8 |
| 11 | 6.5 | 20 | 6.0 | 1.1 | 2024.6 |
| 12 | 6.5 | 25 | 4.0 | 1.1 | 11,196.9 |
| 13 | 5.5 | 20 | 6.0 | 1.9 | 6827.6 |
| 14 | 5.5 | 25 | 6.0 | 1.1 | 7169.4 |
| 15 | 5.5 | 20 | 6.0 | 1.9 | 6859.4 |
| 16 | 5.5 | 25 | 4.0 | 1.9 | 12,233.0 |
| Source | Sum of Squares | Df 1 | Mean Square | F-Value | p-Value Prob > F | |
|---|---|---|---|---|---|---|
| Model | 1.89 × 1013 | 6 | 3.15 × 1012 | 50178.2 | <0.0001 | significant |
| A-pH | 5.06 × 1011 | 1 | 5.06 × 1011 | 8074.3 | <0.0001 | |
| B-Temperature | 2.95 × 1012 | 1 | 2.95 × 1012 | 47019.0 | <0.0001 | |
| C-MLSS | 1.35 × 1013 | 1 | 1.35 × 1013 | 2.154 × 105 | <0.0001 | |
| D-D | 1.16 × 1012 | 1 | 1.16 × 1012 | 18486.6 | <0.0001 | |
| AC | 5.92 × 1011 | 1 | 5.92 × 1011 | 9434.2 | <0.0001 | |
| AD | 1.67 × 1011 | 1 | 1.67 × 1011 | 2659.4 | <0.0001 | |
| Residual | 5.64 × 108 | 9 | 6.27 × 107 | |||
| Lack of Fit | 6.05 × 107 | 1 | 6.05 × 107 | 0.96 | 0.3557 | not significant |
| Pure Error | 5.04 × 108 | 8 | 6.3 × 107 | |||
| Corrected Total | 1.89 × 1013 | 15 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulesha, O.; Maletskyi, Z.; Kvaal, K.; Ratnaweera, H. Strategy for Flux Enhancement in Biofilm Ceramic Membrane Bioreactor Applying Prepolymerized and Non-Prepolymerized Inorganic Coagulants. Water 2019, 11, 446. https://doi.org/10.3390/w11030446
Kulesha O, Maletskyi Z, Kvaal K, Ratnaweera H. Strategy for Flux Enhancement in Biofilm Ceramic Membrane Bioreactor Applying Prepolymerized and Non-Prepolymerized Inorganic Coagulants. Water. 2019; 11(3):446. https://doi.org/10.3390/w11030446
Chicago/Turabian StyleKulesha, Olga, Zakhar Maletskyi, Knut Kvaal, and Harsha Ratnaweera. 2019. "Strategy for Flux Enhancement in Biofilm Ceramic Membrane Bioreactor Applying Prepolymerized and Non-Prepolymerized Inorganic Coagulants" Water 11, no. 3: 446. https://doi.org/10.3390/w11030446
APA StyleKulesha, O., Maletskyi, Z., Kvaal, K., & Ratnaweera, H. (2019). Strategy for Flux Enhancement in Biofilm Ceramic Membrane Bioreactor Applying Prepolymerized and Non-Prepolymerized Inorganic Coagulants. Water, 11(3), 446. https://doi.org/10.3390/w11030446






