Experimental Determination of Moisture Sorption Isotherm of Fecal Sludge
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fecal Sludge Sampling and Initial Characterization
2.2. Preparation of Saturated Salt Solution
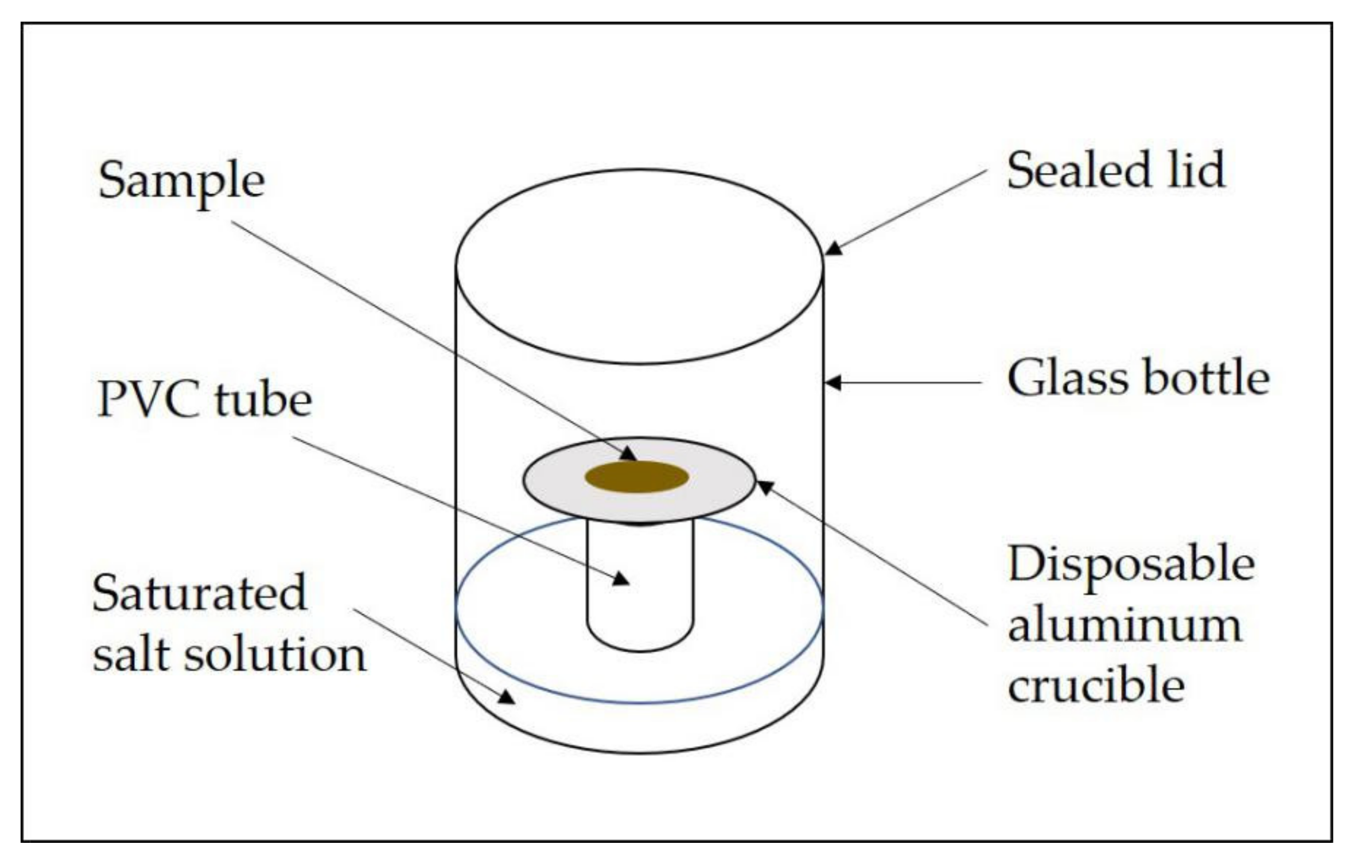
2.3. Determination of Sorption Isotherms
2.4. Mathematical Models of Sorption Isotherms
3. Results and Discussion
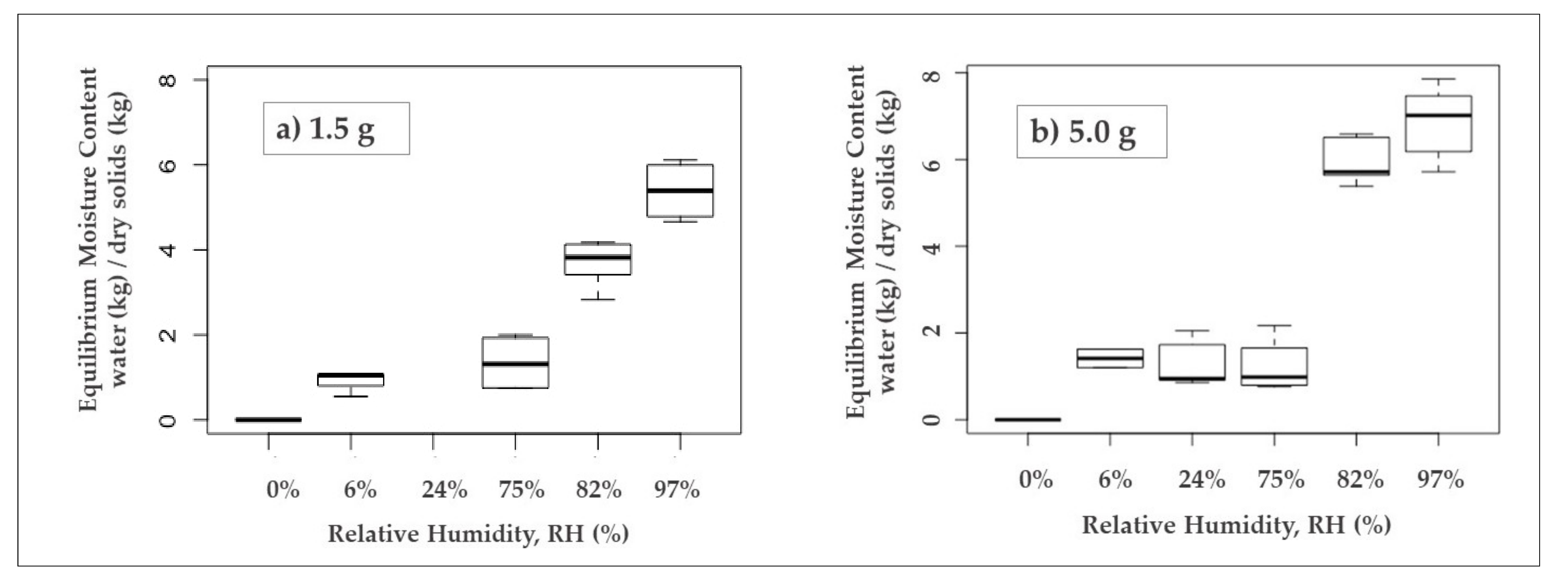
3.1. Moisture Sorption Isotherm from Two Different Sample Masses
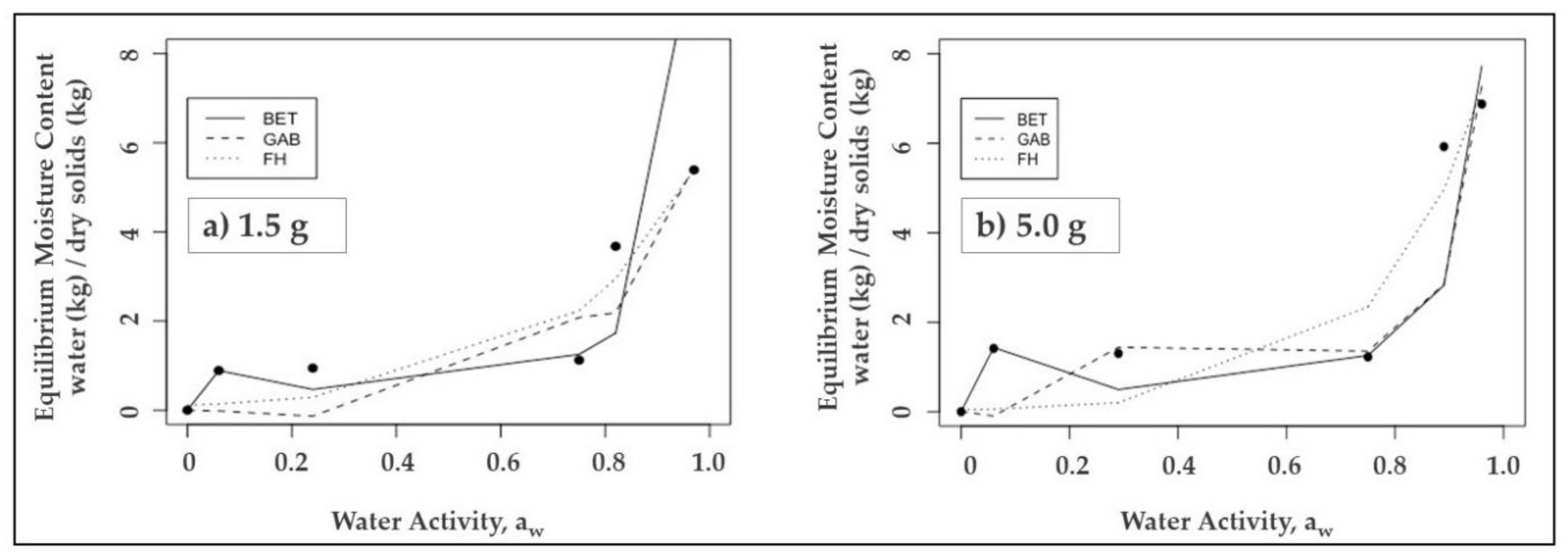
3.2. Modelling of Isotherm
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nakagiri, A.; Niwagaba, C.B.; Nyenje, P.M.; Kulabako, R.N.; Tumuhairwe, J.B.; Kansiime, F. Are pit latrines in urban areas of Sub-Saharan Africa performing? A review of usage, filling, insects and odour nuisances. BMC Public Health 2015, 16, 120. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO)/ the United Nations Children’s Fund (UNICEF). Progress on Drinking Water, Sanitation and Hygiene: 2017 Update and SDG Baselines; WHO and UNICEF: Geneva, Switzerland, 2017. [Google Scholar]
- Strande, L.; Ronteltap, M.; Brdjanovic, D. Fecal Sludge Management—Systems Approach for Implementation and Operation; IWA Publishing: London, UK, 2014. [Google Scholar]
- Mara, D.D.; Feachem, R. Water- and excreta-related diseases: Unitary environmental classification. J. Environ. Eng. ASCE 1999, 125, 334–339. [Google Scholar] [CrossRef]
- Hutton, G.; Varughese, M. The Cost of Meeting the 2030 Sustainable Development Goal Targets on Drinking Water, Sanitation, and Hygiene; Technical Paper; World Bank/Water and Sanitation Programme (WSP): Washington, DC, USA, 2016. [Google Scholar]
- World Water Assessment Programme (WWAP). The United Nations World Water Development Report 2017, Wastewater: The Untapped Resource; United Nations Educational, Scientific and Cultural Organization (UNESCO): Paris, France, 2017. [Google Scholar]
- Rose, C.; Parker, A.; Jefferson, B.; Cartmell, E. The Characterization of feces and urine: A review of the literature to inform advanced treatment technology. Crit. Rev. Environ. Sci. Technol. 2015, 45, 1827–1879. [Google Scholar] [CrossRef] [PubMed]
- Gold, M.; Harada, H.; Therrien, J.-D.; Nishida, T.; Cunningham, M.; Semiyaga, S.; Fujii, S.; Dorea, C.; Nguyen, V.-A.; Strande, L. Cross-country analysis of fecal sludge dewatering. Environ. Technol. 2017, 39, 3077–3087. [Google Scholar] [CrossRef] [PubMed]
- Kone, D.; Cofie, O.; Nelson, K. Low-Cost Options for Pathogen Reduction and Nutrient Recovery From Fecal Sludge; Reports, No H042609; International Water Management Institute: Colombo, Sri Lanka, 2010. [Google Scholar]
- Hui, Y.H. Food Drying Science and Technology: Microbiology, Chemistry, Applications; DEStech Publications: Lancaster, England, 2008; p. 792. [Google Scholar]
- Caballero-Cerón, C.; Guerrero-Beltrán, J.A.; Mújica-Paz, H.; Torres, J.A.; Welti-Chanes, J. Moisture Sorption Isotherms of Foods: Experimental Methodology, Mathematical Analysis, and Practical Applications. In Water Stress in Biological, Chemical, Pharmaceutical and Food Systems; Gutiérrez-López, G.F., Alamilla-Beltrán, L., del Pilar Buera, M., Welti-Chanes, J., Parada-Arias, E., Barbosa-Cánovas, G.V., Eds.; Springer New York: New York, NY, USA, 2015; pp. 187–214. [Google Scholar]
- Al-Muhtaseb, A.H.; McMinn, W.A.M.; Magee, T.R.A. Moisture sorption isotherm characteristics of food products: A Review. Food Bioprod. Process. 2002, 80, 118–128. [Google Scholar] [CrossRef]
- Herwijn, A.J.M. Fundamental Aspects of Sludge Characterization. Ph.D. Thesis, Technische Universiteit Eindhoven, Eindhoven, The Netherlands, 1996. [Google Scholar] [CrossRef]
- Vaxelaire, J. Moisture sorption characteristics of waste activated sludge. J. Chem. Technol. Biotechnol. 2001, 76, 377–382. [Google Scholar] [CrossRef]
- Jannot, Y. Isotherme de Sorption: Modélisation et Charactéristiques; University of Bordeaux, Laboratoire de TREFLE: Bordeaux, France, 2008. [Google Scholar]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater; APHA: Washington, DC, USA, 1998. [Google Scholar]
- CRC. Handbook of chemistry and physics; CRC Press: Boca Raton, FL, USA, 1977. [Google Scholar]
- RCoreTeam. R: A language and environment for statistical computing. R Foundation for Statistical Computing; RCoreTeam: Vienna, Austria, 2017. [Google Scholar]
- Labuza, P.T. Moisture Sorption: Practical Aspects of Isotherm Measurement and Use; Library of Concress Catalog Card Number: 83-73312; Americn Association of Cereal Chemists: Saint Paul, MN, USA, 1984. [Google Scholar]
| Salt | Relative Humidity (%) |
|---|---|
| Sodium hydroxide (NaOH) | 6 |
| Calcium chloride (CaCl2) | 24 |
| Sodium chloride (NaCl) | 75 |
| Potassium chloride (KCl) | 82 |
| Potassium sulphate (K2SO4) | 97 |
| Isotherms | Equation | Parameters |
|---|---|---|
| Brunauer–Emmett–Teller (BET) | M = Moisture content (MC) C = constant M0 = Monolayer MC | |
| Guggenheim–Anderson–de Boer (GAB) | M = Moisture content (MC) C, K’ = constants M0 = Monolayer MC | |
| Flory–Huggins (FH) | M = A exp(Baw) | A, B = constants |
| Model | Sample Mass | Parameters | Value | Standard Error | t-Value | Pr (>|t|) | RSE |
|---|---|---|---|---|---|---|---|
| BET | 1.5 g | M0 | 0.309 | - | - | - | 2.533 on 18 DoF |
| C | −24.800 | 23.690 | −1.047 | 0.308 | |||
| 5.0 g | M0 | 0.309 | - | - | - | 2.837 on 22 DoF | |
| C | 19.904 | 5.711 | −3.485 | 0.002 ** | |||
| GAB | 1.5 g | M0 | 0.309 | - | - | - | 1.302 on 18 DoF |
| C | −4.997 | 1.658 | −3.013 | 0.00746 ** | |||
| K’ | 0.973 | 0.007 | 142.140 | < 2 × 10−16 *** | |||
| 5.0 g | M0 | 0.309 | - | - | - | 2.361 on 22 DoF | |
| C | −4.519 | 1.497 | −3.017 | 0.00747 ** | |||
| K’ | 0.985 | 0.006 | 157.107 | < 2 × 10−16 *** | |||
| FH | 1.5 g | A | 0.144 | 0.091 | 1.582 | 0.131 | 0.8884 on 18 DoF |
| B | 3.739 | 0.696 | 5.373 | 4.17 × 10−5 *** | |||
| 5.0 g | A | 0.403 | 0.234 | 1.725 | 0.0986 | 1.46 on 22 DoF | |
| B | 2.942 | 0.639 | 4.602 | 000139 *** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bourgault, C.; Lessard, P.; Remington, C.; Dorea, C.C. Experimental Determination of Moisture Sorption Isotherm of Fecal Sludge. Water 2019, 11, 303. https://doi.org/10.3390/w11020303
Bourgault C, Lessard P, Remington C, Dorea CC. Experimental Determination of Moisture Sorption Isotherm of Fecal Sludge. Water. 2019; 11(2):303. https://doi.org/10.3390/w11020303
Chicago/Turabian StyleBourgault, Catherine, Paul Lessard, Claire Remington, and Caetano C. Dorea. 2019. "Experimental Determination of Moisture Sorption Isotherm of Fecal Sludge" Water 11, no. 2: 303. https://doi.org/10.3390/w11020303
APA StyleBourgault, C., Lessard, P., Remington, C., & Dorea, C. C. (2019). Experimental Determination of Moisture Sorption Isotherm of Fecal Sludge. Water, 11(2), 303. https://doi.org/10.3390/w11020303





